Highlights
-
•
Kawasaki disease (KD) is an acute childhood disease with coronary artery lesions.
-
•
Endothelial cell injury is mediated by T cells and cytokines (TNF-α and IL-1β).
-
•
KD is due to an abnormal immunological response in genetically predisposed individuals.
-
•
Candidate KD susceptibility genes identified by GWAS include BLK, CD40 and FCGR2A.
-
•
Intravenous immunoglobulin and aspirin are the standard treatment of acute KD.
-
•
IVIG-resistant patients are treated with steroids, cyclosporine, anti-IL-1β or anti-TNF.
Abstract
Kawasaki disease (KD) is an acute febrile childhood inflammatory disease, associated with coronary artery abnormalities. The disease is believed to result from an aberrant inflammatory response to an infectious trigger in a genetically predisposed individual. KD is associated with an endothelial cell injury as a consequence of T cell activation and cytotoxic effects of various proinflammatory cytokines. Intravenous immunoglobulin (IVIG) infusion and aspirin are the standard treatment of acute KD. However, 10–20% of patients show resistance to IVIG therapy and present higher risk of coronary vasculitis. The relative roles of second IVIG infusion, corticosteroids, calcineurin inhibitors, interleukin-1 antagonists and anti-tumor necrosis factor agents remain uncertain. In this review, we highlight the predisposing factors, pathogenesis and therapeutic intervention of KD, particularly new therapeutics for IVIG-resistant patients.
Introduction
Kawasaki disease (KD) is an acute febrile childhood vasculitis, associated with the development of coronary artery abnormalities [1]. KD predominantly affects young children aged between 6 months and 4 years, and is the leading cause of acquired heart disease among children in Japan, European countries and the USA. However, KD is also reported in adults [2]. KD involves predominantly small and medium-sized arteries. Patients with complete KD usually have fever of at least 5 days duration and presence of four principal features: changes in peripheral extremities, polymorphous exanthema, bilateral non-exudative conjunctival injection, and changes in the oral cavity and cervical lymphadenopathy (≥1.5 cm diameter). KD is believed to be secondary to an aberrant inflammatory response to an infectious trigger in a genetically predisposed individual. The three distinct diagnostic vasculopathic processes observed in severely affected KD patients are necrotizing arteritis, subacute/chronic vasculitis and luminal myofibroblastic proliferation; and have the potential to cause life-long cardiovascular sequelae or death [3]. In this review, we focus on the predisposing factors, pathogenesis and therapeutic intervention of KD.
Predisposing factors
The etiological basis of the disease is unclear, but it is generally accepted that KD is due to one or more widely distributed infectious agent(s), evoking an abnormal immunological response in genetically susceptible individuals 4, 5. Genetic polymorphisms encoding cytokines, chemokines and enzymes involved in signal transduction might explain differences in the susceptibility to KD itself, as well as the response to intravenous immunoglobulin (IVIG) treatment [6]. Candidate KD susceptibility genes like B-lymphoid tyrosine kinase (BLK), Cluster of differentiation 40 (CD40), Fcgamma-receptor 2A (FCGR2A), inositol-triphosphate 3-kinase C (ITPKC) and caspase 3 (CASP3) have been identified by genome-wide association studies (GWAS) and by linkage analysis (Table 1 ). A recent study also pointed out the key role of calcium mobilization in the immunopathogenesis of KD [7].
Table 1.
Susceptibility genes for KD.
| Gene | Methods | Refs |
|---|---|---|
| ITPKC | Transmission disequilibrium studies | 20, 24 |
| CASP3 | Transmission disequilibrium studies | 21, 25 |
| BLK | GWAS | 19, 22 |
| CD40 | GWAS | 19, 22 |
| FCGR2A | GWAS | [26] |
| HLA | GWAS | [22] |
Abbreviations: GWAS, genome-wide association studies; ITPKC, inositol-triphosphate 3-kinase C; KD, Kawasaki disease.
Infectious triggers
A number of epidemiologic and clinical observations suggest that KD is secondary to an infectious agent. First, the clinical picture of KD overlaps with infectious diseases, such as adenovirus and scarlet fever. Second, seasonal clustering of KD during winter and spring is similar to other viral diseases like influenza viruses and enteroviruses [8]. In fact, temporal clusters of epidemics have been reported in Japan, the USA, Canada and Finland [9]. Presence of IgA plasma cells in the trachea of patients who died of acute KD suggests entry of the KD etiologic agent through the upper respiratory tract, resulting in an IgA immune response with systemic spread to vascular tissue [10]. Light and electron microscopy studies have demonstrated the presence of cytoplasmic inclusion bodies in the inflamed tissues of acute KD [11]. These inclusion bodies are suggestive of an infectious etiology of KD as a result of intracellular pathogens, particularly viruses.
Based on the clues obtained from clinical and epidemiological studies, the investigations into infectious causes of KD were focused mainly on superantigens, bacterial toxins and viral etiologies. Superantigens such as staphylococcal enterotoxins and streptococcal pyrogenic exotoxins do not need classical antigen processing and presentation by antigen-presenting cells (APCs). Superantigens cause extensive T cell proliferation and cytokine secretion following direct binding to major histocompatibility complex class II proteins on the surface of APCs. However, no specific bacteria or superantigens were identified in the KD scenario, but the clinical features of KD are similar to those seen in patients with staphylococcal and streptococcal toxin-mediated disease. The release of proinflammatory cytokines such as interleukin (IL)-1β, IL-6 and tumor necrosis factor (TNF)-α is directly related to the clinical picture of fever, mucosal involvement and desquamation in toxic shock syndrome and KD [12]. The infrequent occurrence of KD among young infants might be caused by passively acquired maternal antibodies to toxic shock syndrome toxin (TSST)-1 superantigen [13]. As compared to a control group, patients with KD are more likely to have positive rates of viral PCR for different viruses including enterovirus, adenovirus, rhinovirus and coronavirus (50.4% vs 16.4%, P < 0.001) [14].
It was previously proposed that a group of normal microflora in the oral cavity, gut or skin of the parents or guardians of infants and young children are etiological agents of KD. Further, environmental factors such as improved public hygiene or western lifestyle in industrialized Asian countries might substitute normal flora with their variants [15]. Also, KD patients in Asian countries and Hawaii may have more probabilities of exposure and colonization of KD agents, and the distribution of some normal flora in parents or guardians of young children in these regions can differ to Western countries, including France, and developing countries. This might explain different racial incidences of KD.
Genetic predisposing factors
A genetic basis of susceptibility to KD is indicated by several observations. Compared with other ethnicities, the incidence rates of KD are much higher among Japanese individuals (and to a somewhat lesser degree among Korean and Taiwanese populations). The siblings and parents of affected patients also show increased incidence of KD 16, 17, 18. Incidence rates in Asians are up to 20-times higher than Caucasians. The number of patients and the incidence rate of KD in Japan are continuing to increase [18]. Further, the genetic basis of susceptibility to KD is complex, and no single HLA class II haplotype is common to most patients. Also, genetic factors might not be associated with racial difference of KD incidence. Table 1 shows candidate KD susceptibility genes that have recently gained interest 19, 20, 21, 22, 23 and genetic study of adaptive immune cell compartments could further help to detect severely affected KD patients. Genetic studies have provided new insights into the pathogenesis and pathophysiology of KD.
Transmission disequilibrium studies. Transmission disequilibrium studies have demonstrated an association of KD with several single nucleotide polymorphisms (SNPs) affecting the function of molecules involved in the calcineurin-nuclear factor of activated T cells (NFAT) pathway, the transforming growth factor (TGF)-β signaling pathway and Fcγ receptor (FcγR) pathway.
ITPKC regulates NOD-like receptor family pyrin domain containing 3 (NLRP3) activation and hence the production of IL-1β via control of Ca2+ mobilization [7]. Therefore, Ca2+ mobilization plays a major part in the immunopathogenesis of KD. The polymorphism in ITPKC might result in increased IL-1β via activation of NLRP3. Data from several studies show that a SNP within the ITPKC gene confers susceptibility to KD as well as an increased risk of coronary artery aneurysms in Japanese, Taiwanese and American populations 20, 24. ITPKC also acts as a negative regulator of T cell activation through the Ca2+/NFAT signaling pathway. The ITPKC polymorphism might result in increased activation of T cells with an increased expression of IL-2. A SNP within the CASP3 gene has also been associated with susceptibility to KD in cohort studies of Asian and American patients 21, 25. It was found that G to A substitution in one commonly associated SNP located in the 5ʹ untranslated region of CASP3 abolished binding of NFAT to the DNA sequence surrounding the SNP [21].
Genome-wide association studies. Some GWAS in KD have been published and a number of biologically plausible loci involved in inflammation, immune responses and cardiovascular status have been identified. The candidate KD susceptibility genes that were identified by GWAS include BLK, CD40 and FCGR2A 19, 22, 26. BLK encodes B lymphoid tyrosine kinase, a Src family tyrosine kinase downstream the B cell receptor. CD40L is expressed on the surface of CD4+ T cells and platelets, and engages with CD40 expressed on the surface of APCs or endothelial cells. FcγRIIA is a low affinity activating receptor for IgG and provides activation signals to innate immune cells. Further, a recent GWAS identified NEBL and tubulin alpha 3C (TUBA3C) as novel susceptibility genes associated with formation of coronary artery aneurysm in KD [27].
Immune deregulation and pathogenesis
KD is characterized by marked immune activation associated with endothelial cell injury, which could be due to abnormal cytokine production (particularly TNF-α) and generation of cytotoxic antibodies against endothelial cells. Experimental studies in mice have indicated a possible role for IL-1β in the pathogenesis of KD 28, 29. It was also suggested that severely affected KD patients with giant aneurysms have improper immune and repair systems against KD agents or injured coronary artery cells [30].
As mentioned earlier, oligoclonal IgA plasma-cell infiltration has been demonstrated in the arterial wall, the upper respiratory tract and the pancreas of patients with acute KD. This suggests an antigen-driven immune response to an etiologic agent with a respiratory portal of entry [10]. The influx of neutrophils characterizes the early stage of KD. The resultant reactive oxygen species and other inflammatory mediators contribute to endothelial inflammation [31]. Infiltrating macrophages, T lymphocytes and cellular components of the arterial wall such as myofibroblasts are also important in the disease pathogenesis. They secrete a number of inflammatory mediators, enzymes and other molecules such as vascular endothelial growth factor (VEGF), which contribute to vascular leakage and edema.
The presence of circulating cytotoxic antiendothelial cell antibodies reactive with antigens on vascular endothelium has been reported [32]. Furthermore, IgG and IgM antibodies derived from acute KD sera were reported to cause lysis of endothelial cells stimulated with IL-1β or TNF-α [33]. These antibodies could also activate immune cells upon binding to activating FcγR such as FcγRIIA, expressed highly on various innate immune cells including macrophages, dendritic cells (DCs) and neutrophils. Therefore stimulation of FcγRIIA results in the activation of these immune cells leading to expression of activation-associated surface molecules, co-stimulatory molecules and secretion of various inflammatory cytokines and chemokines. Of note, a functional polymorphism in FCGR2A encoding an H131R substitution has been identified as a susceptibility locus for KD [26]. In addition, association between genomic hypomethylation of FCGR2A and susceptibility to KD has been reported recently [34]. These studies thus provide a pointer that either functional polymorphism or epigenetic changes lead to enhanced FcγRIIA signaling and contribute to the pathogenesis of KD.
Levels of a variety of inflammatory cytokines such as TNF-α, IL-1β and IL-6 are increased in the serum during acute KD [35]. Several studies have reported direct correlation between elevated levels of IL-1β in acute patients and vascular endothelial cell damage 33, 36. IL-1β is crucially involved in the Lactobacillus casei cell wall extract mediated coronary arteritis and myocarditis seen in the mouse model of KD. These lesions could be efficiently prevented by IL-1 receptor antagonist treatment [28]. The latest results from the same group indicate that CD11c+ DCs and macrophages are absolutely required for the L. casei cell wall extract induced KD vasculitis model and these macrophages seem to be the cellular source of IL-1β in the lesions [29].
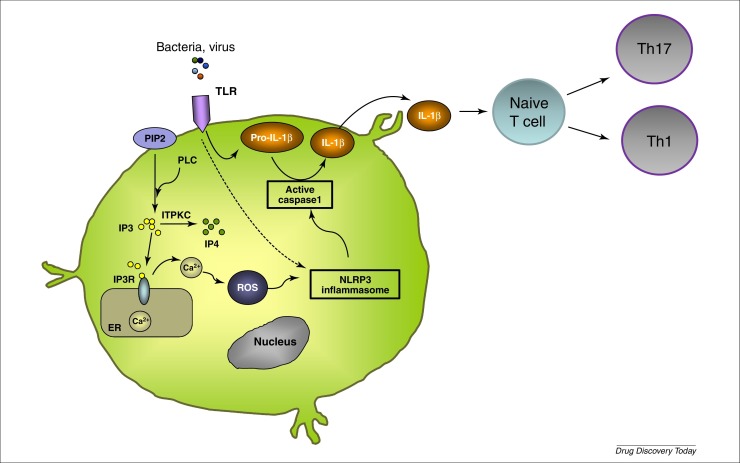
Analysis of serum Th1 and Th2 cytokine levels in KD patients revealed that IL-6, IL-10, TNF-α and interferon (IFN)-γ levels were increased significantly before IVIG treatment [37]. In addition, Th17 cell proportion and expression levels of cytokines associated with Th17 response (IL-17, IL-6 and IL-23) were also significantly upregulated in KD. By contrast, the proportions of regulatory T cells (Tregs), the immunosuppressor cells that are crucial in maintaining immune tolerance and preventing inflammatory responses, were significantly downregulated in acute KD patients [38]. Not only the proportion of Tregs but also expression levels of Treg transcription factor FoxP3 were significantly downregulated in these patients [38]. Thus, the current hypothesis is that an infectious agent activates innate immune cells leading to Ca2+ mobilization and NLRP3 activation leading to IL-1β secretion. IL-1β enhances naive T cell survival and proliferation, and stimulates Th1 and Th17 cells (Fig. 1 ).
Figure 1.
Pathogenesis of Kawasaki disease. It was proposed that an infectious trigger activates TLR and induces accumulation of pro-IL-1β. ITPKC controls Ca2+ homeostasis by phosphorylating IP3 and genome-wide association studies have reported that polymorphisms within the ITPKC gene were associated with susceptibility and severity of Kawasaki disease. IP3 binds to its receptor IP3R expressed on the endoplasmic reticulum membrane and causes the release of Ca2+ into the cytoplasm. Ca2+ mobilization mediates NLRP3 activation. Certain PAMPS are also capable of directly activating the NLRP3 inflammasome. Activation of the inflammasome results in the cleavage of pro-IL-1β into its biologically active form IL-1β, which shapes adaptive immune responses. IL-1β can enhance naive T cell survival and proliferation, via upregulation of the IL-2 receptor. Additionally, a role for IL-1R signaling in Th17 cell differentiation and homeostasis has been reported. Abbreviations: ER, endoplasmic reticulum; IL, interleukin; IP3, inositol 1,4,5-triphosphate; IP4, inositol tetrakisphosphate; ITPKC, inositol-triphosphate 3-kinase C; NLRP3, NOD-like receptor family pyrin domain containing 3; PAMPS, pathogen-associated molecular patterns; PIP2, phosphatidylinositol 4,5-bisphosphate; PLC, phospholipase C; ROS, reactive oxygen species; TLR, Toll-like receptor.
Therapeutic intervention
Standard therapy
IVIG. IVIG consists of a pooled preparation of normal human IgG purified from the plasma of several thousand healthy donors. Initially used for the replacement therapy of primary and secondary immunodeficient patients, high-dose IVIG has been subsequently used to treat a large number of autoimmune and inflammatory disorders. The American Heart Association recommends a single administration of 2 g/kg of IVIG for KD patients within 5–10 days after onset of fever [39]. IVIG treatment not only reduces inflammation (fever, clinical signs, acute phase reactant levels) but also, and more importantly, prevents the development of coronary artery abnormalities 40, 41, 42, 43, 44. It reduces the risk of coronary artery lesions from 20–25% to 2–4%. Because coronary artery lesions begin to occur before the peak of inflammation, it is important to initiate IVIG therapy within a week of fever onset.
IVIG exerts beneficial effects via several mutually nonexclusive mechanisms. These mechanisms include: inhibiting activation of innate cells such as DCs, macrophages, monocytes and neutrophils and of inflammatory mediators secretion; modulation of B cell responses and inhibition of autoantibody production; suppression of pathogenic Th1 and Th17 cells and reciprocal enhancement of Tregs; and inhibition of endothelial cell and complement activation 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58. Although all these mechanisms might not be applicable to KD, several of them have been demonstrated in KD patients following IVIG therapy (Table 2 ). Several studies have shown that IVIG suppresses innate inflammatory cytokines such as IL-1 and IL-6 37, 59, 60, 61. IVIG also reduces nitric oxide production by neutrophils in KD patients [31]. Analysis of serum Th1, Th2 and Th17 cytokine levels revealed that INF-γ, IL-10 and IL-17A decreased rapidly following IVIG treatment 37, 62. By contrast, IVIG reciprocally enhanced the expression of Treg transcription factor FoxP3 [63].
Table 2.
Mechanisms of action of intravenous immunoglobulin reported in Kawasaki disease.
| Mechanisms underlying disease | Mechanisms of IVIG |
|---|---|
| Production of inflammatory cytokines (TNF-α, IL-1, IL-6, IL-17) | Reduction in inflammatory cytokine production 37, 59, 62 |
| Neutrophil activation, migration and production of ROS, nitric oxide | Reduction of nitric oxide production from neutrophils [31] |
| Monocyte and macrophage activation | Reduction of the expression of FcγRI and FcγRIII on monocytes [6] |
| Decreased T regulatory cell/FoxP3 expression | Increase in regulatory T cells [63] |
Abbreviations: IL, interleukin; IVIG, intravenous immunoglobulin; ROS, reactive oxygen species; TNF, tumor necrosis factor.
Genomic studies have reported that the expressions of activating FcγRs: FcγRI and FcγRIII were reduced on monocytes after IVIG therapy [6]. IVIG inhibited TNF-α-induced NF-κB activation in monocytes and macrophages, and blocked FcγRIII on their surface [64]. However, IVIG therapy during the acute stage of KD did not increase the expression of FcγRIIB on peripheral blood CD14+ monocytes and macrophages [65]. Unfortunately, 10–20% of the patients display IVIG resistance and are at higher risk of having coronary vasculitis. The serum levels of IL-1β remained elevated in refractory patients [36]. The level of TNF-α decreased significantly after IVIG treatment in KD patients without coronary artery lesions but increased in IVIG-treated patients with coronary lesions and IVIG-resistant KD patients [37]. Even when treated with high-dose IVIG within the first 10 days of illness, 5% of children with KD develop at least transient coronary artery dilatation and 1% develops giant aneurysms.
To predict resistance to initial IVIG treatment in KD patients, risk-scoring systems have been developed by taking into account age, duration of illness at initial treatment and several blood and biochemical parameters including platelet and neutrophil counts, alanine aminotransferase, sodium and C-reactive protein (CRP). Although these scores demonstrated good specificity, their low sensitivity to predict resistance to IVIG therapy in other cohorts, and particularly in US cohorts, sheds uncertainty on universal utility of those scoring systems [66]. Because a dysregulated immune system and inflammatory response are the hallmarks of KD, a number of inflammatory-mediator-based biomarkers such as damage-associated molecular pattern molecules, granulocyte-colony-stimulating factor, IL-6, IL-1β, TNF-α, polycythemia rubra vera-1 and matrix metalloproteinase-8 have shown a potential for predicting response to IVIG therapy [40]. In resource-limited countries where IVIG therapy is generally unavailable, high-dose methylprednisolone could be used instead of IVIG [67].
Aspirin. Despite the lack of evidence to support therapeutic utility, the use of aspirin in KD continues to be a widely accepted practice. Even the safety of this treatment used at anti-inflammatory doses has been debated. A Cochrane review in 2006 concluded that there was insufficient evidence to support aspirin use for KD [68]. Meta-analysis comparing moderate-dose aspirin (30–50 mg/kg per day) with high-dose aspirin (80–120 mg/kg per day) combined with IVIG found no significant difference in the incidence of coronary abnormalities between the groups [69]. Moderate doses of aspirin are used in the acute phase. A low antiplatelet dose (3–5 mg/kg/day) is used following defervescence. Low-dose aspirin is continued for at least the first 8 weeks of illness, during which time the risk of coronary artery damage is greatest.
Treatments under investigation
Because controlled clinical data are lacking, the relative roles of repeated doses of IVIG, corticosteroids [70], TNF-α antagonists, cytotoxic agents such as methotrexate, cyclophosphamide and cyclosporine A [71], and plasma exchange [72] for patients with refractory KD remain uncertain. Few data from randomized controlled trials exist to guide the treatment of these patients (Table 3 ). In most medical centers, however, a patient who remains febrile or has persistently elevated serum CRP levels, or both, is treated with a second dose of IVIG (2 g/kg).
Table 3.
Current reports on the treatment of patients with refractory Kawasaki disease.
| Treatment | Refs | Study design | Cessation of fever |
|---|---|---|---|
|
Intravenous prednisolone |
[75] | Prospective study | NA/9 |
| [76] | Prospective study | 7/7 | |
| [74] | Retrospective study | 34/44 | |
|
[77] |
Prospective study |
13/13 |
|
|
Infliximab |
[80] | Prospective study | 11/12 |
| [82] | Retrospective study | 17/20 | |
| [81] | Case series | 18/20 | |
|
[83] |
Prospective study |
10/11 |
|
|
Cyclosporine A |
[85] | Retrospective study | 22/28 |
| [84] | Retrospective study | 14/19 | |
|
[79] |
Case series |
6/9 |
|
|
Oral methotrexate |
[87] | Retrospective study | 17/17 |
|
[86] |
Case series |
4/4 |
|
|
Cyclophosphamide |
[90] |
Retrospective study |
2/2 |
| Anakinra | [92] | Case report | 1/1 |
| [91] | Case report | 1/1 | |
| [93] | Case report | 1/1 |
New guidelines have been proposed recently for the management of KD in the UK by taking into consideration of severity of the disease, prior history of IVIG resistance and evolving coronary and/or peripheral aneurysms with ongoing inflammation at the time of presentation [73]. In all these cases, they recommend treating patients with corticosteroids and IVIG. If there is no response to treatment then an anti-TNF-α agent should be considered.
Prednisolone. There are a few prospective studies and one retrospective study about the use of intravenous prednisolone in refractory KD 74, 75, 76, 77. Ogata et al. have demonstrated that steroid pulse therapy was useful to reduce fever duration and medical costs for patients with KD. Steroid pulse therapy and additional IVIG treatment were not significantly different in terms of preventing the development of coronary artery aneurysm [77]. Miura et al. reported that, for KD patients unresponsive to initial IVIG treatment, intravenous methylprednisolone therapy suppressed cytokine levels faster, but were subsequently similar to additional IVIG [76]. It has been shown that addition of prednisolone to the standard regimen of IVIG improves coronary artery outcomes in patients with severe KD in Japan 70, 78. However, another trial in North American patients of mixed ethnicity was unable to demonstrate a benefit of primary steroid treatment with prospective classification of patients in high versus low risk for IVIG resistance [66].
Anti-TNF therapies. Infliximab is a monoclonal antibody against TNF-α and suppresses cytokine-mediated inflammation. A Phase III, randomized, double-blind, placebo-controlled trial explored the use of infliximab as a primary treatment for KD along with IVIG [79]. However, the addition of infliximab to primary treatment in acute KD did not reduce treatment resistance.
Few studies have explored the utility of infliximab in refractory KD 80, 81, 82. In a prospective trial, Burns et al. have shown that infliximab and a second IVIG infusion were safe and well-tolerated in subjects with KD who were resistant to standard IVIG treatment [80]. Another retrospective study has demonstrated that patients with IVIG-resistant KD, whose first retreatment was with infliximab, had faster resolution of fever and fewer days of hospitalization compared with those treated only with IVIG. Coronary artery outcomes and adverse events were similar in both groups [82]. A prospective study has demonstrated that treatment of IVIG-resistant patients with infliximab leads to shorter duration of fever and fewer days of hospitalization than patients retreated with IVIG. However, no differences were observed with coronary outcomes and adverse events [83]. A Phase III trial for the assessment of safety and efficacy of infliximab in IVIG-resistant KD has just been completed (clinicaltrials.gov NCT02298062).
Currently, a placebo-controlled double-blinded randomized study is ongoing to determine if etanercept (0.8 mg/kg subcutaneously) given three times at weekly intervals starting at initial diagnosis is safe and a successful adjunct therapy with IVIG in reducing the incidence of persistent or recurrent fever in KD (clinicaltrials.gov NCT00841789).
Cyclosporine. There is a potential role for calcineurin inhibitors in the management of KD. Two retrospective studies and one case series explored the potential use of cyclosporine in refractory KD 71, 84, 85. Tremoulet et al. demonstrated that cyclosporine was a safe and effective approach that achieved rapid control of inflammation and clinical improvement [71]. Flow-cytometry-based analysis of the T cell phenotype in two subjects displayed a reduction in the circulating activated CD8+ and CD4+ effector memory T cells following cyclosporine treatment. However, suppression of Tregs was not observed [71].
Oral methotrexate. One retrospective study and one case series examined the use of oral methotrexate in refractory KD 86, 87. Lee et al. treated 17 patients who were resistant to one infusion of IVIG with low-dose oral methotrexate (10 mg/m2) once weekly [87]. Administration of methotrexate was continued until CRP levels were normalized. The patients experienced fast resolution of fever and rapid improvement of inflammation markers without adverse effects. However this treatment is now very rarely used.
Atorvastatin. Atorvastatin is a selective competitive inhibitor of 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase. In addition to cholesterol-lowering effects, statins exert improvement in endothelial function and reduction in proinflammatory cytokines and chemokines 88, 89. An open-label, noncomparative Phase II trial is ongoing to evaluate the effect of atorvastatin on the persistent coronary arterial aneurysm in children with KD (clinicaltrials.gov NCT02114099).
Cyclophosphamide. Wallace et al. treated with success two KD patients refractory to two cures of IVIG and intravenous methylprednisolone with intravenous cyclophosphamide at 2 mg/kg/day [90]. Progression of coronary aneurysms was not observed in these patients.
IL-1 receptor antagonists. Only three case reports explored the efficacy of IL-1-targeting drugs in KD 91, 92, 93. Two patients also had a macrophage activation syndrome along with KD. Anakinra, a recombinant IL-1 receptor antagonist, was used at 1, 2 and 9 mg/kg/day with success. KD mice with atherosclerosis after L.-casei-induced coronary arteritis were treated with anakinra [94]. It was found that daily injections of anakinra prevented the acceleration of atherosclerosis.
Currently, two clinical trials are ongoing for exploring the efficacy of anakinra in KD. A Phase IIa multicenter trial is aimed at assessing the efficacy and safety of anakinra in patients with KD who failed to respond to standard treatment (i.e., one infusion of 2 g/kg of IVIG) (clinicaltrials.gov NCT02390596). Another Phase II trial is intended to assess the safety and activity of anakinra in infants and children <2 years old with coronary artery abnormalities (clinicaltrials.gov NCT02179853) [95].
Plasma exchange. Mori et al. reported the effectiveness of plasma exchange in KD patients refractory to IVIG. A comparison of the outcomes in 46 children given plasma exchange treatment with 59 who did not receive it indicated that plasma exchange significantly reduced the frequency of acute-phase coronary lesions by 17.4% versus 40.7% (P = 0.0012) [96]. In another study, 125 patients with refractory KD were treated with plasma exchange. The outcomes were favorable and the effectiveness was excellent, particularly if it was initiated before coronary artery lesions arise [72].
Concluding remarks and future perspectives
About 10–20% of patients with KD are refractory to IVIG therapy. Biomarkers that predict IVIG resistance and responsiveness are needed to reduce the morbidity and treatment costs and to initiate alternative therapy for resistant patients promptly [40]. However, alternative therapies that were explored for resistant patients lack data from randomized, controlled studies, which makes the treatment of refractory KD difficult. Therefore, results from the randomized clinical trials of IL-1β and TNF-α inhibitors are much awaited and are expected to provide more options for the management of resistant patients.
Improved options for the treatment of KD could be derived from the data of pathogenetic mechanisms of the disease. Recent evidence shows that Ca2+ mobilization plays a major part in the pathogenesis of KD by mediating NLRP3 inflammasome activation. NLRP3 converts pro-caspase-1 to active caspase-1, which cleaves pro-IL-1β into its biologically active form IL-1β. MCC950, a potent, selective, small molecule inhibitor of NLRP3, and VX-765, a potent, selective, small molecule inhibitor of caspase-1, have shown promise in various experimental models of autoimmune and inflammatory diseases 97, 98. Combination of IVIG and NLRP3 inflammasome or caspase-1 inhibitors might be examined in refractory patients.
Conflicts of interest
The authors have no conflicts of interest to declare.
Acknowledgments
Supported by Institut National de la Santé et de la Recherche Médicale (INSERM), Université Pierre et Marie Curie, Université Paris Descartes, LFB Biomedicaments, France, and CSL Behring, Switzerland. C.G. is a recipient of fellowship from La Fondation pour la Recherche Médicale (FDM20150633674), France.
References
- 1.Kawasaki T. Acute febrile mucocutaneous syndrome with lymphoid involvement with specific desquamation of the fingers and toes in children. Arerugi. 1967;16:178–222. (in Japanese) [PubMed] [Google Scholar]
- 2.Fraison J.B. Kawasaki disease in adults: observations in France and literature review. Autoimmun. Rev. 2016;15:242–249. doi: 10.1016/j.autrev.2015.11.010. [DOI] [PubMed] [Google Scholar]
- 3.Orenstein J.M. Three linked vasculopathic processes characterize Kawasaki disease: a light and transmission electron microscopic study. PLoS One. 2012;7:e38998. doi: 10.1371/journal.pone.0038998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Onouchi Y. Genetics of Kawasaki disease: what we know and don’t know. Circ. J. 2012;76:1581–1586. doi: 10.1253/circj.cj-12-0568. [DOI] [PubMed] [Google Scholar]
- 5.Rowley A.H. Ultrastructural, immunofluorescence, and RNA evidence support the hypothesis of a new virus associated with Kawasaki disease. J. Infect. Dis. 2011;203:1021–1030. doi: 10.1093/infdis/jiq136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Abe J. Gene expression profiling of the effect of high-dose intravenous Ig in patients with Kawasaki disease. J. Immunol. 2005;174:5837–5845. doi: 10.4049/jimmunol.174.9.5837. [DOI] [PubMed] [Google Scholar]
- 7.Alphonse M.P. Inositol 1,4,5 triphosphate 3-kinase C regulates NLRP3 inflammasome activation in Kawasaki disease. Circulation. 2015;131:A021. [Google Scholar]
- 8.Burns J.C. Seasonality of Kawasaki disease: a global perspective. PLoS One. 2013;8:e74529. doi: 10.1371/journal.pone.0074529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Uehara R., Belay E.D. Epidemiology of Kawasaki disease in Asia, Europe, and the United States. J. Epidemiol. 2012;22:79–85. doi: 10.2188/jea.JE20110131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rowley A.H. IgA plasma cell infiltration of proximal respiratory tract, pancreas, kidney, and coronary artery in acute Kawasaki disease. J. Infect. Dis. 2000;182:1183–1191. doi: 10.1086/315832. [DOI] [PubMed] [Google Scholar]
- 11.Rowley A.H. Cytoplasmic inclusion bodies are detected by synthetic antibody in ciliated bronchial epithelium during acute Kawasaki disease. J. Infect. Dis. 2005;192:1757–1766. doi: 10.1086/497171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Meissner H.C., Leung D.Y. Superantigens, conventional antigens and the etiology of Kawasaki syndrome. Pediatr. Infect. Dis. J. 2000;19:91–94. doi: 10.1097/00006454-200002000-00001. [DOI] [PubMed] [Google Scholar]
- 13.Nomura Y. Maternal antibody against toxic shock syndrome toxin-1 may protect infants younger than 6 months of age from developing Kawasaki syndrome. J. Infect. Dis. 2002;185:1677–1680. doi: 10.1086/340513. [DOI] [PubMed] [Google Scholar]
- 14.Chang L.Y. Viral infections associated with Kawasaki disease. J. Formos. Med. Assoc. 2014;113:148–154. doi: 10.1016/j.jfma.2013.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee K.Y. Kawasaki disease may be a hyperimmune reaction of genetically susceptible children to variants of normal environmental flora. Med. Hypotheses. 2007;69:642–651. doi: 10.1016/j.mehy.2006.12.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huang W.C. Epidemiologic features of Kawasaki disease in Taiwan, 2003–2006. Pediatrics. 2009;123:e401–e405. doi: 10.1542/peds.2008-2187. [DOI] [PubMed] [Google Scholar]
- 17.Kim G.B. Epidemiologic features of Kawasaki disease in South Korea: data from nationwide survey, 2009–2011. Pediatr. Infect. Dis. J. 2014;33:24–27. doi: 10.1097/INF.0000000000000010. [DOI] [PubMed] [Google Scholar]
- 18.Makino N. Descriptive epidemiology of Kawasaki disease in Japan, 2011–2012: from the results of the 22nd nationwide survey. J. Epidemiol. 2015;25:239–245. doi: 10.2188/jea.JE20140089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee Y.C. Two new susceptibility loci for Kawasaki disease identified through genome-wide association analysis. Nat. Genet. 2012;44:522–525. doi: 10.1038/ng.2227. [DOI] [PubMed] [Google Scholar]
- 20.Onouchi Y. ITPKC functional polymorphism associated with Kawasaki disease susceptibility and formation of coronary artery aneurysms. Nat. Genet. 2008;40:35–42. doi: 10.1038/ng.2007.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Onouchi Y. Common variants in CASP3 confer susceptibility to Kawasaki disease. Hum. Mol. Genet. 2010;19:2898–2906. doi: 10.1093/hmg/ddq176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Onouchi Y. A genome-wide association study identifies three new risk loci for Kawasaki disease. Nat. Genet. 2012;44:517–521. doi: 10.1038/ng.2220. [DOI] [PubMed] [Google Scholar]
- 23.Shimizu C. Transforming growth factor-beta signaling pathway in patients with Kawasaki disease. Circ. Cardiovasc. Genet. 2011;4:16–25. doi: 10.1161/CIRCGENETICS.110.940858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kuo H.C. Single-nucleotide polymorphism rs7251246 in ITPKC is associated with susceptibility and coronary artery lesions in Kawasaki disease. PLoS One. 2014;9:e91118. doi: 10.1371/journal.pone.0091118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kuo H.C. CASP3 gene single-nucleotide polymorphism (rs72689236) and Kawasaki disease in Taiwanese children. J. Hum. Genet. 2011;56:161–165. doi: 10.1038/jhg.2010.154. [DOI] [PubMed] [Google Scholar]
- 26.Khor C.C. Genome-wide association study identifies FCGR2A as a susceptibility locus for Kawasaki disease. Nat. Genet. 2011;43:1241–1246. doi: 10.1038/ng.981. [DOI] [PubMed] [Google Scholar]
- 27.Kuo H.C. Genome-wide association study identifies novel susceptibility genes associated with coronary artery aneurysm formation in Kawasaki disease. PLoS One. 2016;11:e0154943. doi: 10.1371/journal.pone.0154943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lee Y. Interleukin-1beta is crucial for the induction of coronary artery inflammation in a mouse model of Kawasaki disease. Circulation. 2012;125:1542–1550. doi: 10.1161/CIRCULATIONAHA.111.072769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lee Y. IL-1 signaling is critically required in stromal cells in Kawasaki disease vasculitis mouse model: role of both IL-1α and IL-1β. Arterioscler. Thromb. Vasc. Biol. 2015;35:2605–2616. doi: 10.1161/ATVBAHA.115.306475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lee K.Y. A common immunopathogenesis mechanism for infectious diseases: the protein-homeostasis-system hypothesis. Infect. Chemother. 2015;47:12–26. doi: 10.3947/ic.2015.47.1.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yoshimura K. Increased nitric oxide production by neutrophils in early stage of Kawasaki disease. Eur. J. Pediatr. 2009;168:1037–1041. doi: 10.1007/s00431-008-0872-1. [DOI] [PubMed] [Google Scholar]
- 32.Grunebaum E. The role of anti-endothelial cell antibodies in Kawasaki disease – in vitro and in vivo studies. Clin. Exp. Immunol. 2002;130:233–240. doi: 10.1046/j.1365-2249.2002.02000.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Leung D.Y. Two monokines, interleukin 1 and tumor necrosis factor, render cultured vascular endothelial cells susceptible to lysis by antibodies circulating during Kawasaki syndrome. J. Exp. Med. 1986;164:1958–1972. doi: 10.1084/jem.164.6.1958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kuo H.C. Identification of an association between genomic hypomethylation of FCGR2A and susceptibility to Kawasaki disease and intravenous immunoglobulin resistance by DNA methylation array. Arthritis Rheumatol. 2015;67:828–836. doi: 10.1002/art.38976. [DOI] [PubMed] [Google Scholar]
- 35.Lin C.Y. Serial changes of serum interleukin-6, interleukin-8, and tumor necrosis factor α among patients with Kawasaki disease. J. Pediatr. 1992;121:924–926. doi: 10.1016/s0022-3476(05)80343-9. [DOI] [PubMed] [Google Scholar]
- 36.Leung D.Y. Endothelial cell activation and high interleukin-1 secretion in the pathogenesis of acute Kawasaki disease. Lancet. 1989;334:1298–1302. doi: 10.1016/s0140-6736(89)91910-7. [DOI] [PubMed] [Google Scholar]
- 37.Wang Y. Evaluation of intravenous immunoglobulin resistance and coronary artery lesions in relation to Th1/Th2 cytokine profiles in patients with Kawasaki disease. Arthritis Rheum. 2013;65:805–814. doi: 10.1002/art.37815. [DOI] [PubMed] [Google Scholar]
- 38.Jia S. The T helper type 17/regulatory T cell imbalance in patients with acute Kawasaki disease. Clin. Exp. Immunol. 2010;162:131–137. doi: 10.1111/j.1365-2249.2010.04236.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Newburger J.W. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation. 2004;110:2747–2771. doi: 10.1161/01.CIR.0000145143.19711.78. [DOI] [PubMed] [Google Scholar]
- 40.Galeotti C. Molecular and immunological biomarkers to predict IVIg response. Trends Mol. Med. 2015;21:145–147. doi: 10.1016/j.molmed.2015.01.005. [DOI] [PubMed] [Google Scholar]
- 41.Greco A. Kawasaki disease: an evolving paradigm. Autoimmun. Rev. 2015;14:703–709. doi: 10.1016/j.autrev.2015.04.002. [DOI] [PubMed] [Google Scholar]
- 42.Newburger J.W. A single intravenous infusion of gamma globulin as compared with four infusions in the treatment of acute Kawasaki syndrome. N. Engl. J. Med. 1991;324:1633–1639. doi: 10.1056/NEJM199106063242305. [DOI] [PubMed] [Google Scholar]
- 43.Galeotti C. Kawasaki disease: aetiopathogenesis and therapeutic utility of intravenous immunoglobulin. Autoimmun. Rev. 2010;9:441–448. doi: 10.1016/j.autrev.2009.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Burns J.C., Franco A. The immunomodulatory effects of intravenous immunoglobulin therapy in Kawasaki disease. Expert Rev. Clin. Immunol. 2015;11:819–825. doi: 10.1586/1744666X.2015.1044980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tha-In T. Modulation of the cellular immune system by intravenous immunoglobulin. Trends Immunol. 2008;29:608–615. doi: 10.1016/j.it.2008.08.004. [DOI] [PubMed] [Google Scholar]
- 46.Trinath J. Intravenous immunoglobulin expands regulatory T cells via induction of cyclooxygenase-2-dependent prostaglandin E2 in human dendritic cells. Blood. 2013;122:1419–1427. doi: 10.1182/blood-2012-11-468264. [DOI] [PubMed] [Google Scholar]
- 47.Othy S. Intravenous gammaglobulin inhibits encephalitogenic potential of pathogenic T cells and interferes with their trafficking to the central nervous system, implicating sphingosine-1 phosphate receptor 1-mammalian target of rapamycin axis. J. Immunol. 2013;190:4535–4541. doi: 10.4049/jimmunol.1201965. [DOI] [PubMed] [Google Scholar]
- 48.Schwab I., Nimmerjahn F. Intravenous immunoglobulin therapy: how does IgG modulate the immune system? Nat. Rev. Immunol. 2013;13:176–189. doi: 10.1038/nri3401. [DOI] [PubMed] [Google Scholar]
- 49.Maddur M.S. Comparison of different IVIg preparations on IL-17 production by human Th17 cells. Autoimmun. Rev. 2011;10:809–810. doi: 10.1016/j.autrev.2011.02.007. [DOI] [PubMed] [Google Scholar]
- 50.Ballow M. The IgG molecule as a biological immune response modifier: mechanisms of action of intravenous immune serum globulin in autoimmune and inflammatory disorders. J. Allergy Clin. Immunol. 2011;127:315–323. doi: 10.1016/j.jaci.2010.10.030. [DOI] [PubMed] [Google Scholar]
- 51.Sharma M. Intravenous immunoglobulin-induced IL-33 is insufficient to mediate basophil expansion in autoimmune patients. Sci. Rep. 2014;4:5672. doi: 10.1038/srep05672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kessel A. Intravenous immunoglobulin therapy affects T regulatory cells by increasing their suppressive function. J. Immunol. 2007;179:5571–5575. doi: 10.4049/jimmunol.179.8.5571. [DOI] [PubMed] [Google Scholar]
- 53.Bayry J. Intravenous immunoglobulin expands regulatory T cells in autoimmune rheumatic disease. J. Rheumatol. 2012;39:450–451. doi: 10.3899/jrheum.111123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Maddur M.S. Intravenous immunoglobulin exerts reciprocal regulation of Th1/Th17 cells and regulatory T cells in Guillain–Barre syndrome patients. Immunologic. Res. 2014;60:320–329. doi: 10.1007/s12026-014-8580-6. [DOI] [PubMed] [Google Scholar]
- 55.Fiebiger B.M. Protection in antibody- and T cell-mediated autoimmune diseases by antiinflammatory IgG Fcs requires type II FcRs. Proc. Natl. Acad. Sci. U. S. A. 2015;112:E2385–E2394. doi: 10.1073/pnas.1505292112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Othy S. Sialylation may be dispensable for reciprocal modulation of helper T cells by intravenous immunoglobulin. Eur. J. Immunol. 2014;44:2059–2063. doi: 10.1002/eji.201444440. [DOI] [PubMed] [Google Scholar]
- 57.Maddur M.S. Intravenous immunoglobulin-mediated expansion of regulatory T cells in autoimmune patients is associated with increased prostaglandin E2 levels in the circulation. Cell. Mol. Immunol. 2015;12:650–652. doi: 10.1038/cmi.2014.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Seite J.F. What is the contents of the magic draft IVIg? Autoimmun. Rev. 2008;7:435–439. doi: 10.1016/j.autrev.2008.04.012. [DOI] [PubMed] [Google Scholar]
- 59.Lin I.C. Augmented TLR2 expression on monocytes in both human Kawasaki disease and a mouse model of coronary arteritis. PLoS One. 2012;7:e38635. doi: 10.1371/journal.pone.0038635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Galeotti C. Heme oxygenase-1 is dispensable for the anti-inflammatory activity of intravenous immunoglobulin. Sci. Rep. 2016;6:19592. doi: 10.1038/srep19592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gupta M. Cytokine modulation with immune gamma-globulin in peripheral blood of normal children and its implications in Kawasaki disease treatment. J. Clin. Immunol. 2001;21:193–199. doi: 10.1023/a:1011039216251. [DOI] [PubMed] [Google Scholar]
- 62.Rasouli M. Downregulation of Th17 cells and the related cytokines with treatment in Kawasaki disease. Immunol. Lett. 2014;162:269–275. doi: 10.1016/j.imlet.2014.09.017. [DOI] [PubMed] [Google Scholar]
- 63.Guo M.M. Th17- and Treg-related cytokine and mRNA expression are associated with acute and resolving Kawasaki disease. Allergy. 2015;70:310–318. doi: 10.1111/all.12558. [DOI] [PubMed] [Google Scholar]
- 64.Ichiyama T. Intravenous immunoglobulin inhibits NF-κB activation and affects Fcγ receptor expression in monocytes/macrophages. Naunyn Schmiedebergs Arch. Pharmacol. 2004;369:428–433. doi: 10.1007/s00210-004-0877-x. [DOI] [PubMed] [Google Scholar]
- 65.Ichiyama T. Intravenous immunoglobulin does not increase FcγRIIB expression on monocytes/macrophages during acute Kawasaki disease. Rheumatology. 2005;44:314–317. doi: 10.1093/rheumatology/keh488. [DOI] [PubMed] [Google Scholar]
- 66.Sleeper L.A. Evaluation of Kawasaki disease risk-scoring systems for intravenous immunoglobulin resistance. J. Pediatr. 2011;158:831–835. doi: 10.1016/j.jpeds.2010.10.031. e833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Singh S. Management of Kawasaki disease in resource-limited settings. Pediatr. Infect. Dis. J. 2015;34:94–96. doi: 10.1097/INF.0000000000000600. [DOI] [PubMed] [Google Scholar]
- 68.Baumer J.H. Salicylate for the treatment of Kawasaki disease in children. Cochrane Database Syst. Rev. 2006;2006:CD004175. doi: 10.1002/14651858.CD004175.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Terai M., Shulman S.T. Prevalence of coronary artery abnormalities in Kawasaki disease is highly dependent on gamma globulin dose but independent of salicylate dose. J. Pediatr. 1997;131:888–893. doi: 10.1016/s0022-3476(97)70038-6. [DOI] [PubMed] [Google Scholar]
- 70.Ogata S. Corticosteroid pulse combination therapy for refractory Kawasaki disease: a randomized trial. Pediatrics. 2012;129:e17–e23. doi: 10.1542/peds.2011-0148. [DOI] [PubMed] [Google Scholar]
- 71.Tremoulet A.H. Calcineurin inhibitor treatment of intravenous immunoglobulin-resistant Kawasaki disease. J. Pediatr. 2012;161 doi: 10.1016/j.jpeds.2012.02.048. 506–512; e501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hokosaki T. Long-term efficacy of plasma exchange treatment for refractory Kawasaki disease. Pediatr. Int. 2012;54:99–103. doi: 10.1111/j.1442-200X.2011.03487.x. [DOI] [PubMed] [Google Scholar]
- 73.Eleftheriou D. Management of Kawasaki disease. Arch. Dis. Child. 2014;99:74–83. doi: 10.1136/archdischild-2012-302841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Furukawa T. Effects of steroid pulse therapy on immunoglobulin-resistant Kawasaki disease. Arch. Dis. Child. 2008;93:142–146. doi: 10.1136/adc.2007.126144. [DOI] [PubMed] [Google Scholar]
- 75.Hashino K. Re-treatment for immune globulin-resistant Kawasaki disease: a comparative study of additional immune globulin and steroid pulse therapy. Pediatr. Int. 2001;43:211–217. doi: 10.1046/j.1442-200x.2001.01373.x. [DOI] [PubMed] [Google Scholar]
- 76.Miura M. Effects of methylprednisolone pulse on cytokine levels in Kawasaki disease patients unresponsive to intravenous immunoglobulin. Eur. J. Pediatr. 2008;167:1119–1123. doi: 10.1007/s00431-007-0642-5. [DOI] [PubMed] [Google Scholar]
- 77.Ogata S. The strategy of immune globulin resistant Kawasaki disease: a comparative study of additional immune globulin and steroid pulse therapy. J. Cardiol. 2009;53:15–19. doi: 10.1016/j.jjcc.2008.08.002. [DOI] [PubMed] [Google Scholar]
- 78.Kobayashi T. Efficacy of immunoglobulin plus prednisolone for prevention of coronary artery abnormalities in severe Kawasaki disease (RAISE study): a randomised, open-label, blinded-endpoints trial. Lancet. 2012;379:1613–1620. doi: 10.1016/S0140-6736(11)61930-2. [DOI] [PubMed] [Google Scholar]
- 79.Tremoulet A.H. Infliximab for intensification of primary therapy for Kawasaki disease: a phase 3 randomised, double-blind, placebo-controlled trial. Lancet. 2014;383:1731–1738. doi: 10.1016/S0140-6736(13)62298-9. [DOI] [PubMed] [Google Scholar]
- 80.Burns J.C. Infliximab treatment of intravenous immunoglobulin-resistant Kawasaki disease. J. Pediatr. 2008;153:833–838. doi: 10.1016/j.jpeds.2008.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Mori M. Efficacy and limitation of infliximab treatment for children with Kawasaki disease intractable to intravenous immunoglobulin therapy: report of an open-label case series. J. Rheumatol. 2012;39:864–867. doi: 10.3899/jrheum.110877. [DOI] [PubMed] [Google Scholar]
- 82.Son M.B. Infliximab for intravenous immunoglobulin resistance in Kawasaki disease: a retrospective study. J. Pediatr. 2011;158 doi: 10.1016/j.jpeds.2010.10.012. 644–649; e641. [DOI] [PubMed] [Google Scholar]
- 83.Youn Y. Infliximab as the first retreatment in patients with Kawasaki disease resistant to initial intravenous immunoglobulin. Pediatr. Infect. Dis. J. 2016;35:457–459. doi: 10.1097/INF.0000000000001039. [DOI] [PubMed] [Google Scholar]
- 84.Hamada H. Inflammatory cytokine profiles during cyclosporin treatment for immunoglobulin-resistant Kawasaki disease. Cytokine. 2012;60:681–685. doi: 10.1016/j.cyto.2012.08.006. [DOI] [PubMed] [Google Scholar]
- 85.Suzuki H. Cyclosporin A treatment for Kawasaki disease refractory to initial and additional intravenous immunoglobulin. Pediatr. Infect. Dis. J. 2011;30:871–876. doi: 10.1097/INF.0b013e318220c3cf. [DOI] [PubMed] [Google Scholar]
- 86.Ahn S.Y., Kim D.S. Treatment of intravenous immunoglobulin-resistant Kawasaki disease with methotrexate. Scand. J. Rheumatol. 2005;34:136–139. doi: 10.1080/03009740510026328. [DOI] [PubMed] [Google Scholar]
- 87.Lee T.J. Low-dose methotrexate therapy for intravenous immunoglobulin-resistant Kawasaki disease. Yonsei Med. J. 2008;49:714–718. doi: 10.3349/ymj.2008.49.5.714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Garg N. Rosuvastatin improves endothelial dysfunction in ankylosing spondylitis. Clin. Rheumatol. 2015;34:1065–1071. doi: 10.1007/s10067-015-2912-3. [DOI] [PubMed] [Google Scholar]
- 89.Yoshimura K. Inhibitory effect of statins on inflammation-related pathways in human abdominal aortic aneurysm tissue. Int. J. Mol. Sci. 2015;16:11213–11228. doi: 10.3390/ijms160511213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Wallace C.A. Initial intravenous gammaglobulin treatment failure in Kawasaki disease. Pediatrics. 2000;105:E78. doi: 10.1542/peds.105.6.e78. [DOI] [PubMed] [Google Scholar]
- 91.Cohen S. A child with severe relapsing Kawasaki disease rescued by IL-1 receptor blockade and extracorporeal membrane oxygenation. Ann. Rheum. Dis. 2012;71:2059–2061. doi: 10.1136/annrheumdis-2012-201658. [DOI] [PubMed] [Google Scholar]
- 92.Shafferman A. High dose anakinra for treatment of severe neonatal Kawasaki disease: a case report. Pediatr. Rheumatol. Online J. 2014;12:26. doi: 10.1186/1546-0096-12-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Miettunen P.M. Successful treatment of severe paediatric rheumatic disease-associated macrophage activation syndrome with interleukin-1 inhibition following conventional immunosuppressive therapy: case series with 12 patients. Rheumatology. 2011;50:417–419. doi: 10.1093/rheumatology/keq218. [DOI] [PubMed] [Google Scholar]
- 94.Chen S. Marked acceleration of atherosclerosis after Lactobacillus casei-induced coronary arteritis in a mouse model of Kawasaki disease. Arterioscler. Thromb. Vasc. Biol. 2012;32:e60–e71. doi: 10.1161/ATVBAHA.112.249417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Tremoulet A.H. Rationale and study design for a phase I/IIa trial of anakinra in children with Kawasaki disease and early coronary artery abnormalities (the ANAKID trial) Contemp. Clin. Trials. 2016;48:70–75. doi: 10.1016/j.cct.2016.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Mori M. Efficacy of plasma exchange therapy for Kawasaki disease intractable to intravenous gamma-globulin. Mod. Rheumatol. 2004;14:43–47. doi: 10.1007/s10165-003-0264-3. [DOI] [PubMed] [Google Scholar]
- 97.Coll R.C. A small-molecule inhibitor of the NLRP3 inflammasome for the treatment of inflammatory diseases. Nat. Med. 2015;21:248–255. doi: 10.1038/nm.3806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wannamaker W. (S)-1-((S)-2-{[1-(4-amino-3-chloro-phenyl)-methanoyl]-amino}-3,3-dimethyl-butanoy l)-pyrrolidine-2-carboxylic acid ((2R,3S)-2-ethoxy-5-oxo-tetrahydro-furan-3-yl)-amide (VX-765), an orally available selective interleukin (IL)-converting enzyme/caspase-1 inhibitor, exhibits potent anti-inflammatory activities by inhibiting the release of IL-1β and IL-18. J. Pharmacol. Exp. Ther. 2007;321:509–516. doi: 10.1124/jpet.106.111344. [DOI] [PubMed] [Google Scholar]