Abstract
Community-acquired pneumonia may present with similar clinical symptoms, regardless of viral or bacterial cause. Diagnostic assays are needed to rapidly discriminate between causes, because this will guide decisions on appropriate treatment. Therefore, a quantitative real-time PCR (qPCR) assay with duplex reactions targeting eight bacteria and six viruses was developed. Technical performance was examined with linear plasmids. Upper and lower respiratory tract specimens were used to compare the qPCR assay with standard microbiological methods. The limit of detection was 5 to 20 DNA template copies with approximately 1000-fold differences in concentrations of the two competing templates. SDs for positive controls were <5%. The use of the qPCR assay resulted in 113 positive identifications in 94 respiratory specimens compared with 38 by using standard diagnostics. Diagnostic accuracy of the qPCR assay varied between 60% positive agreement with standard tests for Streptococcus pneumoniae and 100% for Mycoplasma pneumoniae, Moraxella catarrhalis, and Staphylococcus aureus. Negative percentage of agreement was >95% for M. pneumoniae, Streptococcus pyogenes, respiratory syncytial virus, and influenza A virus; whereas it was only 56% for Haemophilus influenzae. Multiple microbial agents were identified in 19 of 44 sputum and 19 of 50 nasopharynx specimens. We conclude that in parallel qPCR detection of the targeted respiratory bacteria and viruses is feasible. The results indicate good technical performance of the assay in clinical specimens.
Community-acquired pneumonia (CAP) represents a major challenge for institutions of rational medical treatment because of its diverse cause. Viruses and bacteria may present with similar symptoms, making them difficult to distinguish clinically.1 When physicians lack information about the causative agent for their decision on appropriate treatment, it leads to increased use of ecologically unfavorable broad-spectrum antibiotics.2 To provide appropriate and targeted treatment, physicians need diagnostic methods that are rapid, sensitive, and specific, the combination of which current standard diagnostic methods lack. Although present viral diagnostics mostly rely on nucleic acid detection, the properties of traditional cultivation techniques for bacteria limit the possibilities to develop a rapid method for detection of all tentative causes. Thus, a number of recent studies have aimed to develop molecular methods to detect bacterial and viral pathogens. To overcome the limitations of cultivation, quantitative real-time PCR (qPCR) assays were implemented because they allow rapid detection and are generally applicable. The method was implemented for common bacterial pathogens such as Streptococcus pneumoniae, Staphylococcus aureus, Streptococcus pyogenes, Haemophilus influenzae, Moraxella catarrhalis, Legionella pneumophila, Mycoplasma pneumoniae, and Chlamydophila pneumoniae.3, 4, 5, 6, 7, 8 A vast number of studies have also explored diagnostic qPCR for viral causes of CAP, in particular influenza A and B, respiratory syncytial virus (RSV), and parainfluenza virus (PIV).9, 10, 11, 12 Nevertheless, few studies have aimed to develop combined molecular detection assays for viruses and bacteria with relevance to CAP. A recent evaluation of two available systems found that, although one clinically relevant pathogen could readily be detected, there were performance limitations when two pathogens were present in a specimen.13
Because of the rapid turnaround time and a low limit of detection, implementing nucleic acid-based detection methods for CAP pathogens may shorten the time to diagnosis and increase sensitivity. Standard cultivation methods provide limited sensitivity for important causes, including H. influenzae, probably because of the fastidious nature of the bacterium, and even more so for S. pneumoniae. Because of autolytic behavior, the latter seems to survive poorly during both transport and after cultivation.14
To fully take advantage of the molecular methods' sensitivity and specificity, target genes must be chosen with care. Capitalizing on previous work that validated suitable targets for several respiratory pathogens, we have developed a qPCR assay for causative diagnosis of CAP.3, 12, 15, 16, 17, 18, 19, 20, 21, 22, 23 We have emphasized high technical performance, including the ability to detect co-infections when multiple pathogens are present at different concentrations. Here, we present a multiplex, qPCR method for simultaneous detection of eight bacterial and six viral pathogens with relevance for diagnosis of CAP.
Materials and Methods
Experimental Practices for Designing, Performing, and Interpreting qPCR Experiments
This work was conducted at the maximum possible extent according to the guidelines on minimum information for publication of qPCR experiments.24
DNA and RNA Extraction
Sputum specimens were homogenized with equal volume N-acetyl cysteine solution as described previously,25 before extraction of nucleic acids. Automated DNA/RNA extraction by the Nordiag Arrow instrument and the Viral NA kit (DiaSorin, Saluggia VC, Italy) was used for all clinical specimens and viral strains with a sample volume of 550 μL and an elution volume of 100 μL. The eluate of clinical samples was then diluted with 100 μL of nuclease-free water (Ambion, Life Technologies, Carlsbad, CA) before being stored at −80°C until analysis. DNA of bacterial strains was extracted by a previously described method,26 based on binding of DNA to uniform glass beads, before being stored at −20°C until analysis. Plasmid DNA was extracted by the QIAprep Spin Miniprep Kit (Qiagen Inc., Valencia, CA), according to the manufacturer's instructions.
Positive Controls and Standards
Linear plasmids with inserts of the target sequences were used as positive controls for all assays. Plasmids were ordered from Eurofins MWG Operon (Ebersberg, Germany), except the plasmid used for C. pneumoniae that was an in-house construct. Plasmids were transformed into One Shot TOP10 Electrocom Escherichia coli (Invitrogen, Life Technologies, Carlsbad, CA); cells were plated on kanamycin-containing Luria Broth-agar plates. Single colonies were used for DNA extraction as described in DNA and RNA Extraction, and the concentration was measured in duplicates with the NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies, Wilmington, DE). A positive control solution of 2 × 104 plasmid copies per mL that corresponded to 100 plasmid copies per 25 μL of PCR reaction was prepared.
Primers Probes and qPCR Conditions
Primer and probe sequences for L. pneumophila, M. pneumoniae, M. catarrhalis, C. pneumoniae, S. aureus, PIV type 1 (PIV1), PIV2, RSV, and influenza A1 and B were obtained from previous publications,3, 12, 15, 20, 22 and PCR conditions were optimized for duplex reactions (Table 1 ). A new influenza A H3 strain with point mutations in the target sequence of primers and probes of the influenza A1 assay appeared during the course of our work, and our qPCR assay was therefore updated according to the recommendations of the Public Health Agency of Sweden (M. Brytting, unpublished data) to include an assay-denoted influenza A2. Primers and probes for H. influenzae and S. pneumoniae were already established as uniplex qPCR assays for analysis of spinal fluid at our laboratory but not published before. Primers and probes for S. pyogenes were constructed with the Primer Express version 3.0 (Applied Biosystems, Life Technologies, Carlsbad, CA). All probes used the hydrolysis technology with black hole quenchers and fluorophores 6-carboxyfluorescein or VIC (Applied Biosystems). The TaqMan Fast Advanced Master Mix for DNA detection and the TaqMan Fast Virus 1-step Master Mix for RNA detection were used for bacteria and viruses, respectively (Applied Biosystems). Total reaction volume was 25 μL, consisting of 12.5/6.25 μL of 2× DNA/4× RNA Master Mix, primers, probes, nuclease-free water, and 5 μL of template or clinical specimen. All runs included triplicates of no-template controls and duplicates of positive controls. Amplification, detection, and analysis were performed by a 7900HT Fast Real-Time PCR System (Applied Biosystems). The thermal profile was 5 minutes at 50°C, 20 seconds at 95°C, and subsequently 45 cycles of 15 seconds at 95°C and 60 seconds at 60°C.
Table 1.
Primers and Probes
| Primer/probe target (gene or protein name) | Sequence | Reporter/quencher | Concentration, nmol/L | Reference | Duplex name |
|---|---|---|---|---|---|
| Legionella pneumophila (mip) | Forward: 5′-AAAGGCATGCAAGACGCTATG-3′ | 100 | 3 | A | |
| Reverse: 5′-TGTTAAGAACGTCTTTCATTTGCTG-3′ | 100 | ||||
| Probe: 5′-TGGCGCTCAATTGGCTTTAACCGA-3′ | VIC-MGBNFQ | 250 | |||
| Mycoplasma pneumonia (P1) | Forward: 5′-GGAATCCCAATGCACAAGAACA-3′ | 600 | 12 | ||
| Reverse: 5′-GCTTTGGTCAACACATCAACCTT-3′ | 300 | ||||
| Probe: 5′-GCCTTGAAGGCTGGGTTTGCGCTA-3′ | 6FAM-MGBNFQ | 250 | |||
| Streptococcus pneumoniae (lytA) | Forward: 5′-AACTCTTACGCAATCTAGCAGATGAA-3′ | 300 | This study | B | |
| Reverse: 5′-CGTGCAATACTCGTGCGTTTTA-3′ | 300 | ||||
| Probe: 5′-CCGAAAACGCTTGATACA-3′ | 6FAM-MGBNFQ | 250 | |||
| Moraxella catarrhalis (uspA1) | Forward: 5′-GTCAAACAGCTGGAGGTATTGC-3′ | 300 | 15 | ||
| Reverse: 5′-GACATGATGCTCACCTGCTCTA-3′ | 600 | ||||
| Probe: 5′-ATCGCAATTGCAACTTT-3′ | VIC-MGBNFQ | 200 | |||
| Chlamydophila pneumonia (momp) | Forward: 5′-CAAGGGCTATAAAGGCGTTGCT-3′ | 200 | 12 | C | |
| Reverse: 5′-ATGGTCGCAGACTTTGTTCCA-3′ | 200 | ||||
| Probe: 5′-TCCCCTTGCCAACAGACGCTGG-3′ | VIC-MGBNFQ | 250 | |||
| Staphylococcus aureus (nuc) | Forward: 5′-AAATTACATAAAGAACCTGCGACA-3′ | 300 | 20 | ||
| Reverse: 5′-GAATGTCATTGGTTGACCTTTGTA-3′ | 600 | ||||
| Probe: 5′-AATTTAACCGTATCACCATCAATCGCTTT-3′ | 6FAM-MGBNFQ | 250 | |||
| Streptococcus pyogenes grp A (SPy_1285) | Forward: 5′-CTCGACAAGTCCTCAATCAAACC-3′ | 200 | This study | D | |
| Reverse: 5′-ATGAGTTGCGGAAATTTGAGGTA-3′ | 200 | ||||
| Probe: 5′-CATAGAGAATTTATAACCGCACTC-3′ | 6FAM-MGBNFQ | 250 | |||
| Haemophilus influenza (P6) | Forward: 5′-CTTGGTCTCCATACTTAACTAAATAAAAAACTC-3′ | 400 | This study | ||
| Reverse: 5′-GAACTACAAGCCGCTAATGCAG-3′ | 400 | ||||
| Probe: 5′-CATTATTAGTTGCAGGTTCT-3′ | VIC-MGBNFQ | 250 | |||
| Parainfluenza virus 1 (HN) | Forward: 5′-GTTGTCAATGTCTTAATTCGTATCAATAATT-3′ | 300 | 22 | E | |
| Reverse: 5′-GTAGCCTMCCTTCGGCACCTAA-3′ | 300 | ||||
| Probe: 5′-TAGGCCAAAGATTGTTGTCGAGACTATTCCAAGCATTTCCAATCTTCAGGACTATGA-3′ | 6FAM-MGBNFQ | 150 | |||
| Parainfluenza virus 2 (HN) | Forward: 5′-GCATTTCCAATCTTCAGGACTATGA-3′ | 300 | 22 | ||
| Reverse: 5′-ACCTCCTGGTATAGCAGTGACTGAAC-3′ | 300 | ||||
| Probe: 5′-CCATTTACCTAAGTGATGGAATCAATCGCAAA-3′ | VIC-MGBNFQ | 250 | |||
| Parainfluenza virus 3 (HN) | Forward: 5′-AAATGATCTGATTTATGCTTATACCTC-3′ | 300 | LP Nielsen, unpublished data | F | |
| Reverse: 5′-TCAGGTACCAAGTCTGAGTTTACA-3′ | 300 | Copenhagen | |||
| Probe: 5′-CGAGGTTGYCAGGATATAGGAAAATCA-3′ | 6FAM-MGBNFQ | 250 | |||
| Respiratory syncytial virus (M) | Forward: 5′-GCAAATATGGAAACATACGTGAACA-3′ | 200 | 12 | ||
| Reverse: 5′-GCACCCATATTGTWAGTGATGCA-3′ | 200 | ||||
| Probe: 5′-CTTCACGAAGGCTCCACATACACAGCWG-3′ | VIC-MGBNFQ | 150 | |||
| Influenza A 1 (M1)∗ | Forward: 5′-AAGACCAATYCTGTCACCTCTGA-3′ | 600 | 12 | G | |
| Reverse: 5′-CAAAGCGTCTACGCTGCAGTCC-3′ | 300 | ||||
| Probe: 5′-TTTGTGTTCACGCTCACCGT-3′ | VIC-MGBNFQ | 250 | |||
| Influenza A 2 (Segment 7/M1)∗ | Forward 1: 5′-AGGACCYCTGTCACCTCTGA-3′ | 300 | M Brytting, unpublished data | ||
| Forward 2: 5′-CAAGACCAATCTTGTCACTCTGA-3′ | 300 | Public Health Agency of Sweden | |||
| Reverse 1: 5′-TCTCGCTCACTGGGCA-3′ | 300 | ||||
| Reverse 2: 5′-TCCTCGCTCACTTGGCA-3′ | 300 | ||||
| Probe 1: 5′-TTGTGTTCACGCTCACC-3′ | 6FAM-MGBNFQ | 112 | |||
| Probe 2: 5′-TTTGTTTTCACGCTCACCG-3′ | 6FAM-MGBNFQ | 112 | |||
| Probe 3: 5′-TTTGTATTCACGCTCACCG-3′ | 6FAM-MGBNFQ | 112 | |||
| Influenza B (HA) | Forward: 5′-AAATACGGTGGATTAAAYAAAAGCAA-3′ | 600 | 12 | ||
| Reverse: 5′-CCAGCAATAGCTCCGAAGAAA-3′ | 600 | ||||
| Probe: 5′-CACCCATATTGGGCAATTTCCTATGGC-3′ | VIC-MGBNFQ | 250 |
6FAM, 6-carboxyfluorescein.
Bold indicates the use of a primer mix to target two different bases.
For the detection of influenza A, two different primer pairs were used during the study because a new influenza A strain emerged, and we adjusted our assay accordingly.
Tests of Analytical Specificity, Sensitivity, Efficiency, and Reproducibility
Duplex qPCR assays were combined as described in Table 1. Analytical specificity of primers and probes was tested bioinformatically and experimentally with primer BLAST27 and a test panel of 34 reference strains (Supplemental Table S1), respectively. Limit of detection was assessed as recommended,28 by serial dilution of linear plasmid by using six replicates with concentrations of 20, 10, 5, 2, 1, and 0.1 copies per reaction, respectively. The ability of the duplex assay to simultaneously detect both agents targeted was tested by keeping one of the agents at high concentration (104 copies per reaction) while testing for the other agent. For PIV1 and PIV2 equal concentrations were used. The limit of detection was defined as the concentration at which all six replicates were detected, for theoretical reasons five copies per reactions was set as detection limit. An experiment was approved only if zero to one of the six replicates in the 0.1 dilution were positive. Efficiency was tested by dilution series of plasmid standards, ranging from approximately 1 to 105 copies per reaction, with equal concentrations of both targets of the duplex reaction. The analyses were performed in duplicates or triplicates, and detection of at least five dilution steps was required for an approved result. Standard curves were generated by plotting the quantification cycle (Cq) value against 10-logarithm of the concentration. Linearity (R 2 value) and slope of the standard curve was calculated with the data analysis program SDS version 2.3 (Applied Biosystems). Reproducibility of assays were assessed by comparing positive control results from 26 runs in two separate analysis batches performed >6 months apart by two persons with the use of separate plasmid preparations and dilution series. Relative SD of the Cq values was calculated as a measure of stability. In addition, reproducibility was assessed with clinical specimens spiked with low (n = 5) or high (n = 5) concentration of target DNA or RNA and by using separate qPCR machines in five consecutive runs on three different days. The relative SDs of the Cq values were calculated as a measure of intrarun variability.
Tests of Inhibition
The presence and frequency of PCR inhibition was investigated by analysis of 22 clinical nasopharyngeal aspirates (NpAs) and 38 sputum specimens, as previously described.29 Specimens were spiked with 100 copies per reaction with each of the plasmid standards, and the results were normalized to the mean of the 100 copies per reaction positive controls from the same run. Absence of signal or a normalized Cq value >2.5 SD above the mean of the positive controls was considered as inhibition. To verify that the method could detect viruses in sputum, sputum specimens each with a volume of 350 μL were spiked with100 μL from a NpA specimen positive for either RSV or influenza A before nucleic acid extraction and qPCR assay analysis were performed as described in the sections DNA and RNA Extraction and Primers Probes and qPCR Conditions, respectively.
Inclusion and the Handling of Clinical Specimens
Lower respiratory tract specimens (expectorated or induced sputum) and upper respiratory tract specimens (NpA) sent for respiratory infection diagnostics to the Clinical Microbiology Laboratory (Norrlands universitetssjukhus, Umeå, Sweden) were consecutively included in the evaluation of the qPCR assay. Sputum specimens with an original volume of >0.5 mL, >25 leucocytes, and <10 epithelial cells per 100× power field as determined by Gram staining were included. Sputum specimens were split into one portion for standard tests and another for storage at −80°C until used for evaluation by the qPCR assay. Before culture, specimens were treated with a 1:1 volume of N-acetyl cysteine and diluted 1:100 and 1:10,000 in phosphate-buffered saline.
Laboratory Workflow for Comparing the qPCR Assay and Standard Microbiological Tests
The qPCR assay was run in small batches in parallel with standard microbiological tests, but its results were not included in the laboratory reports sent to the clinicians. The qPCR assay results were compared with all available standard test results that corresponded to the day of sampling and to the specimen type, NpA, nasopharyngeal swab, or sputum. The standard diagnostics at our laboratory uses NpA for detection of viruses, M. pneumoniae, and C. pneumoniae, whereas nasopharyngeal swab is used for other bacteria present in the upper respiratory tract. Standard diagnostic work was performed by well-trained clinical laboratory scientists in cooperation with senior consultants in clinical microbiology. Standard tests were uniplex qPCR for L. pneumophila,30 PIV1 to PIV3 (in-house method; Y. Lidgren, unpublished data), and influenza A and B (as recommended by the Public Health Agency of Sweden; M. Brytting, unpublished data). A duplex qPCR was used as the standard test for M. pneumoniae and C. pneumoniae.4 For detection of S. pneumoniae, S. aureus, S. pyogenes, H. influenzae, and M. catarrhalis, the standard test was semiquantitative bacterial culture of sputum and nasopharyngeal swab according to Swedish reference methodology.31 Bacteria were incubated on blood agar and chocolate agar at 37°C in 5% carbon dioxide atmosphere for 24 to 48 hours and identified according to standard phenotypical methods.31
Statistical Analysis
Raw data of the qPCRs were processed with SDS version 2.3 (Applied Biosystems). Further analyses and statistical tests were performed with GraphPad Prism version 6 (GraphPad Software, Inc., La Jolla, CA) and Microsoft Excel 2010 (Microsoft Corp., Redmond, WA). Pearson's χ2 test, Fishers' exact test, U-test, and analysis of variance were used for comparison of proportions and comparison of means, respectively. P < 0.05 was regarded as significant. Positive and negative percentage of agreement and overall accuracy were calculated according to recommendations by the US Food and Drug Administration (http://www.fda.gov/downloads/MedicalDevices/DeviceRegulationandGuidance/GuidanceDocuments/ucm071287.pdf, last accessed December 15, 2014).
Results
Technical Performance of the qPCR Assay
Analysis of the sensitivity for a given target in the duplex reaction revealed that the panel indicated good amplification linearity, efficiency, and sensitivity for each of the tested targets (Table 2 ). The limit of detection for each of the targets varied between 5 and 20 copies when there were approximately 1000-fold differences in template concentrations. Reproducibility tests found that the calculated relative SDs for all positive controls (26 runs) were <5% (1.1% to 4.3%). The intrarun variability was <2% in all assays, 0.1% to 1.8% in high-positive samples and 0.2% to 1.2% in low-positive samples.
Table 2.
Summary of the Technical Performance
| Primer/probe | LOD, copies per reaction | Cq at LOD | Cq of second target | R2 | Amplification efficiency, % |
|---|---|---|---|---|---|
| L. pneumophila | 5 | 36.6 | 29.4 | 0.994 | 94.9 |
| M. pneumoniae | 5 | 39.6 | 26.2 | 0.996 | 100.5 |
| S. pneumoniae | 5 | 40.6 | 26.3 | 0.994 | 96.8 |
| M. catarrhalis | 5 | 41.1 | 25.2 | 0.998 | 93.8 |
| C. pneumoniae | 5 | 35.8 | 28.5 | 0.997 | 101.8 |
| S. aureus | 5 | 35.5 | 28.4 | 0.999 | 101.8 |
| S. pyogenes (GAS) | 5 | 40.8 | 27.4 | 0.996 | 108.7 |
| H. influenzae | 5 | 39.3 | 26.3 | 0.998 | 102.7 |
| PIV1 | 20 | 38.4 | —∗ | 0.996 | 92.0 |
| PIV2 | 20 | 38.4 | —∗ | 0.996 | 97.2 |
| PIV3 | 10 | 39.7 | 26.8 | 1.000 | 96.8 |
| RSV | 5 | 38.0 | 29.4 | 0.998 | 99.3 |
| Influenza A 1 | 10 | 38.5 | 31.0 | 0.998 | 91.6 |
| Influenza A 2 | 10 | 37.1 | 26.3 | 0.998 | 98.0 |
| Influenza B | 10 | 36.9 | 28.9 | 0.998 | 94.2 |
Cq, quantitation value; LOD, limit of detection; PIV, parainfluenza virus; RSV, respiratory syncytial virus.
LOD for PIV1 and PIV2 was determined in equal concentrations.
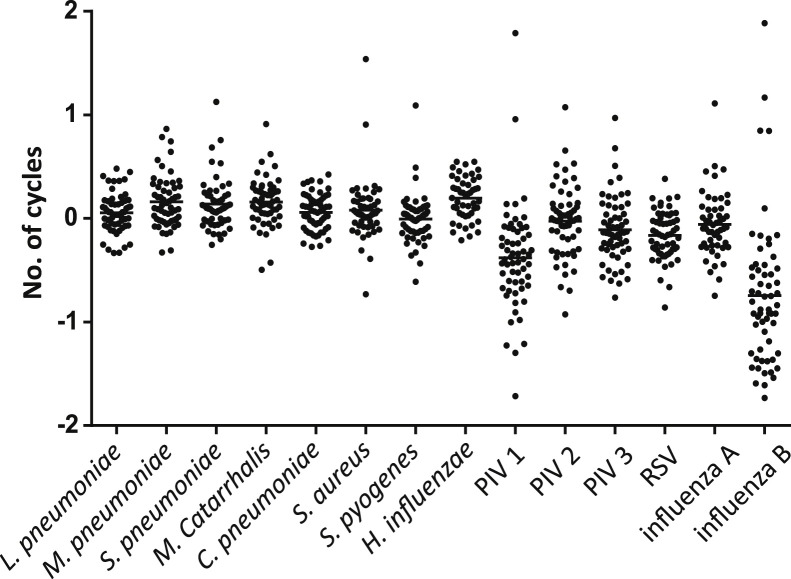
PCR amplification inhibition by using clinical specimens spiked with control plasmids was low for all agents, with variations in Cq values of <2 cycles compared with the positive control (Figure 1 ). Substantial inhibition was present for 2 of 38 sputum specimens tested with 9 of 14 and 7 of 14 detection systems inhibited, respectively. In two additional sputum specimens and one NpA specimen, results for 1 of the 14 detection systems were >2.5 SD above the mean of the positive controls, indicating a presence of limited inhibition. In the evaluation of the assay performance for detection of virus in sputum, we found that sputum specimens spiked with influenza A- or RSV-containing NpA specimens were positive at 25.9 ± 0.6 (n = 5) and 27.7 ± 1.1 (n = 5) cycles, respectively.
Figure 1.
Number of quantitative real-time PCR cycles between clinical specimens and positive control spikes with the same amount of target nucleic acid to reveal possible amplification inhibition. PIV, parainfluenza virus; RSV, respiratory syncytial virus.
Findings by Standard Microbiological Tests
Among sputum specimens consecutively collected between October 10, 2011, and November 24, 2011, 44 were eligible for inclusion. Patient median age was 63.5 years (range, 17 to 83 years; n = 44), with male/female distribution of 12/32. Standard bacterial cultures identified a common respiratory pathogen in 12 specimens: S. pneumoniae (n = 3), M. catarrhalis (n = 1), S. aureus (n = 3), and H. influenzae (n = 5). In addition, Klebsiella spp. were identified in three specimens and Exophiala dermatitidis in one specimen. Fifty NpA specimens were consecutively collected between January 2, 2013, and January 18, 2013. Patient median age was 60.5 years (range, 0 to 93 years; n = 50), with male/female distribution of 29/21. Nasopharyngeal swab bacterial culture results were available for 14 specimens, with findings of S. pneumoniae (n = 2), M. catarrhalis (n = 1), and H. influenzae (n = 1). Standard qPCR results were available for influenza A and B in 48 of 50 specimens, for PIV1 to PIV3 in 10 of 50, for C. pneumoniae in 10 of 50, for M. pneumoniae in 10 of 50, and for RSV in 12 of 50. The positive identifications were M. pneumoniae (n = 2), RSV (n = 3), and influenza A (n = 17). No standard diagnostic tests for L. pneumophila were performed.
Causative Findings by Use of the qPCR Assay
The results of the qPCR assay are shown in Table 3 . H. influenzae was the most common finding detected in 26 of 44 sputum specimens and 4 of 50 NpA specimens. Notably, Cq mean values were high, indicating relatively low numbers of bacteria in many specimens. Similarly, S. aureus was also a common finding and likewise displayed high Cq mean values. A single agent was identified in 16 of 44 sputum specimens and in 16 of 50 NpA specimens. Two agents were detected in 14 of 44 sputum specimens and in 16 of 50 NpA specimens, and three agents were detected in 4 of 44 sputum specimens and 1 of 50 NpA specimens. Four agents were identified in a single sputum specimen. The qPCR assay was negative for 9 of 44 sputum specimens and 17 of 50 NpA specimens.
Table 3.
Result of the qPCR Assay Applied to Upper and Lower Respiratory Tract Specimens
| Organism and specimen type∗ | Result of the qPCR assay |
||
|---|---|---|---|
| Positive, n | Negative, n | Cq means ± SEM | |
| M. pneumoniae | |||
| NpA | 0 | 50 | |
| Sputum† | 4 | 40 | 27.3 ± 1.6 |
| S. pneumoniae | |||
| NpA | 5 | 45 | 32.2 ± 2.3 |
| Sputum | 7 | 36 | 28.8 ± 1.8 |
| M. catarrhalis | |||
| NpA | 8 | 42 | 32.1 ± 1.5 |
| Sputum | 6 | 38 | 28.4 ± 3.5 |
| S. aureus | |||
| NpA | 11 | 39 | 35.4 ± 0.8 |
| Sputum | 15 | 29 | 33.5 ± 1.3 |
| S. pyogenes | |||
| NpA | 5 | 45 | 32.5 ± 1.4 |
| Sputum | 2 | 40 | 36.9 ± 2.2 |
| H. influenzae | |||
| NpA | 4 | 46 | 38.3 ± 2.1 |
| Sputum | 26 | 18 | 34.3 ± 1.0 |
| RSV | |||
| NpA | 5 | 45 | 24.7 ± 1.0 |
| Sputum | 0 | 44 | |
| Influenza A 1 | |||
| NpA | 13 | 37 | 31.0 ± 1.1 |
| Sputum | 0 | 43 | |
| Influenza A 2 | |||
| NpA | 15 | 35 | 29.5 ± 1.3 |
| Sputum | 0 | ||
Cq, quantitation value; NpA, nasopharyngeal aspirate; qPCR, quantitative real-time PCR; RSV, respiratory syncytial virus.
No positive results were obtained for L. pneumophila, C. pneumoniae, PIV1 to PIV3, and influenza B.
For some organisms, the number of results for sputum is <44, because the specimen volume was consumed so not all duplex reactions could be completed.
Comparison of Methods
Measures of agreement between results of standard microbiological tests and the qPCR assay are shown in Table 4 . Positive percentage of agreement varied between 60.0% for S. pneumoniae and 100% for M. pneumoniae, M. catarrhalis, S. aureus, and RSV. Negative percentage of agreement exceeded 95% for M. pneumoniae, S. pyogenes, RSV, and influenza A and was lowest, 55.8%, for H. influenzae.
Table 4.
Findings of Agreement and Overall Accuracy between the qPCR Assay and Standard Microbiological Tests
| Assay∗ | PPA, % | (95% CI) | NPA, % | (95% CI) | Accuracy, % |
|---|---|---|---|---|---|
| M. pneumoniae | 100.0 | (15.8–100) | 100.0 | (63.1–100) | 100.0 |
| S. pneumoniae | 60.0 | (14.7–94.7) | 84.9 | (72.4–93.3) | 82.8 |
| M. catarrhalis | 100.0 | (15.8–100) | 89.3 | (78.1–96.0) | 89.7 |
| S. aureus | 100.0 | (29.2–100) | 69.1 | (55.2–80.0) | 70.7 |
| S. pyogenes | 96.5 | (87.9–99.6) | 96.6 | ||
| H. influenza | 83.3 | (35.9–99.6) | 55.8 | (41.3–69.5) | 58.6 |
| RSV | 100.0 | (29.2–100) | 100.0 | (66.4–100) | 100.0 |
| Influenza A 1 | 76.5 | (50.1–93.2) | 100.0 | (89.4–100) | 92.0 |
| Influenza A 2 | 88.2 | (63.6–98.5) | 100.0 | (88.8–100) | 96.0 |
NPA, negative percentage of agreement; PPA, positive percentage of agreement; qPCR, quantitative real-time PCR; RSV, respiratory syncytial virus.
L. pneumophila, C. pneumoniae, parainfluenza virus type 1 to type 3, and influenza B are not shown, because of lack of positive findings.
Overall, the qPCR assay detected higher numbers of microbial agents compared with standard diagnostic methods, with a total of 113 and 38 agents, respectively. The difference in diagnostic yield in large part resulted from the panel design of the qPCR assay constantly targeting eight bacterial and six viral agents compared with standard diagnostic tests variably being applied to respiratory specimens from the corresponding patients. Of the 50 NpA specimens tested with the qPCR assay, 33 were analyzed by standard tests for specific viruses or M. pneumoniae and C. pneumoniae, but no corresponding respiratory specimens were tested for the presence of other bacteria. Nineteen additional observations of bacteria resulted from using the qPCR assay. Of 44 sputum specimens, 38 were tested by standard culture test, but no corresponding tests were performed for identifying viruses or difficult-to-culture bacteria. Two additional M. pneumoniae but no additional observations of viruses resulted from using the qPCR assay. Generally, the increase in detection rate by the qPCR assay was most pronounced for H. influenzae, S. aureus, M. pneumoniae, and RSV (Table 5 and Supplemental Table S2).
Table 5.
Comparison of Diagnostic Outcomes of the qPCR Assay and Standard Diagnostics Applied to Patients Sampled by NpA (n = 50) and Collection of Sputa (n = 44)
| Assay | Positive by the qPCR assay, n |
Positive by standard tests, n |
||
|---|---|---|---|---|
| Total | Unique findings∗ | Total | Unique findings∗ | |
| M. pneumoniae | 4 | 2 | 2 | 0 |
| S. pneumoniae | 12 | 9 | 3 | 2 |
| M. catarrhalis | 12 | 10 | 2 | 0 |
| S. aureus | 26 | 22 | 4 | 0 |
| S. pyogenes | 7 | 6 | 1 | 0 |
| H. influenzae | 31 | 26 | 6 | 1 |
| RSV | 5 | 2 | 3 | 0 |
| Influenza A 1 | 13 | 0 | 17 | 4 |
| Influenza A 2 | 15 | 0 | 17 | 2 |
qPCR, quantitative real-time PCR; RSV, respiratory syncytial virus.
Unique to the qPCR assay or standard tests, respectively. Note that by standard testing, some organisms were sometimes not clinically suspected and consequently were not tested.
Unique observations by the qPCR assay versus the standard tests and vice versa are highlighted in Table 5. Notably, two of five specimens were positive for RSV and two of four specimens were positive for M. pneumoniae by the qPCR assay only. Analysis of mean Cq values for positive identification by the qPCR assay found that specimens culture-positive for H. influenzae or S. aureus were significantly lower than values of culture-negative specimens, (P = 0.0005 and P = 0.0088, respectively), suggesting that high Cq values represent numbers of bacteria below the detection limit of culture (data not shown). Two of five culture-positive sputum specimens were false negative by the qPCR assay for S. pneumoniae. One of them was also false negative for H. influenzae and positive in the inhibition control assay, indicating the presence of a potent inhibiting substance. In addition, two NpA specimens were false negative for influenza A by the qPCR assay, both with Cq values of >36 cycles in the standard qPCR test, indicative of low viral RNA abundance.
Discussion
With the aim to develop a qPCR assay to provide clinically useful information for a physician to evaluate CAP patients, we developed a panel that targeted eight bacteria and six viruses. The performance of the assay was evaluated with spiked samples and clinical respiratory specimens. This resulted in a qPCR assay that showed high technical performance and, in addition, showed superior overall performance compared with the standard diagnostic methods used at our laboratory. Microorganisms commonly regarded as clinically relevant were identified by the new assay but not by standard tests. Importantly, the main difference was not only because of a higher sensitivity of the new qPCR assay compared with standard tests. The improved performance resulted largely from the change of the diagnostic approach; standard diagnostic testing involved clinical decisions on including or excluding specific test, whereas the new assay approach relied on analysis of a panel of causes relevant to the evaluation of all patients with suspected CAP. A distinct advantage of the new assay was that common causes of CAP were not disregarded because of nontypical clinical presentations.
There is a challenge when designing a diagnostic PCR assay to find an appropriate balance between technical performances on the one hand and the usability and cost on the other hand. We have designed this assay such that the high demands of diagnostic performance could be combined with ease of use at an acceptable cost. Clinical specimens are analyzed in duplicate with our protocol as means of providing technical control of pipetting or other potential handling errors during pre-PCR processing. Downscaling to one reaction per sample would be a simple task if this is preferred at other laboratories. We strongly endorse, however, to not compromise the inclusion of external positive controls and the negative no-template controls to ensure proper quality control.
The qPCR assay uniformly detected <20 gene target copies per reaction, and most importantly this was true for both agents targeted in each PCR reaction, even when there was a 1000-fold difference in target DNA copy numbers. It is well known that a potential limitation of all diagnostic PCR assays is poor amplification efficiency that may result from inhibitory substances in the biological material analyzed, interfering with the DNA preparation step or the PCR amplification step. As should be expected in well-designed assays, we noted stable and uniform amplification efficiency of >90% for all microbial DNA targets in the dilution experiments performed with linear plasmids, but, most importantly, the inhibition rate was low in the evaluation of clinical specimens. Extensive inhibition was identified in only 2 of 38 sputum specimens, and minor inhibition was observed as an increased Cq value in one of the duplicate reactions for 2 of 38 sputum specimens and 1 of 22 NpA specimens. In support of such reasoning, we found that even for the two sputum specimens with substantial inhibition of the amplification of some target; all other targets were still amplified.
There is currently a knowledge gap about the role of co-infections in patients with symptoms of CAP, and we hope that the availability of assays like the one developed here will help fill the gap. Some reports have indicated clinical implications of co-infection in children and in adults,32, 33, 34 whereas others suggest little effects of co-infection on disease severity and outcome.35, 36 More studies are clearly needed on this topic to ensure that future patients with a suspicion of CAP or other respiratory infections receive the best possible treatment.
Evaluating the qPCR assay on consecutively collected sputum or NpA specimens found an added value of combining the detection of bacteria and viruses in the same assay. Compared with the more traditional strategy of analyzing respiratory specimens for either bacterial or viral causes, the combined approach showed superior detection of agents that are regarded as clinically relevant. Thus, the added value was a combination of the high sensitivity of qPCR assays in general, and, in particular, the fact that infectious agents were detected which were not initially suspected. It was evident that bacteria were detected more often by the qPCR assay than by standard tests as exemplified by the detection of S. pneumoniae in 12 specimens by the former assay but only in five specimens by the standard culture method. The superior performance of the qPCR assay of detecting S. pneumoniae applied to both nasopharyngeal and sputum specimens, but especially so for sputum, and, notably, the Cq mean values of the latter were often low, indicating that several culture-negative specimens contained abundant S. pneumoniae-specific nucleic acids. These findings indicate that PCR-based methods may be preferable to culture-based methods for detection of S. pneumoniae in sputum, and this is noteworthy because it is the most important CAP pathogen. Likewise, the qPCR assay was superior for detection of RSV and M. pneumoniae, and in this case it was obvious that the change in the diagnostic approach provided the advantage. The additional detections of RSV and M. pneumoniae were in NpA and sputum specimens, respectively, and occurred at low Cq values, indicating abundant nucleic acids. These two agents were not targeted by the standard tests performed, although they, in retrospect, likely were clinically relevant.
We selected eight bacteria and six viral agents to be included in the new assay on the basis of a judgment that these would be the most important agents to test for in all patients with CAP admitted to hospital. This selection may be questioned, and we acknowledge that there are several other targets that could be included in a panel relevant to the evaluation of CAP patients. We believe, however, that the agents selected represent a reasonable compromise of agents that are interesting for inclusion and exclusion purposes at the evaluation of suspected CAP. Naturally, additional microbial agents should be targeted in, for example, situations with severe disease at the intensive care unit, when there is an epidemiology to suggest a spread of highly pathogenic corona viruses, for patient populations with chronic lung disease, or when long-term hospitalized patients are suspected with respiratory infection.
The strengths of this study include the high technical performance of the assay, and that the evaluation and comparison between the new assay and the standard diagnostic approach generally found good agreement with enhanced capabilities of the new assay to detect microbial agents commonly regarded as clinically relevant, for example, S. pneumoniae, M. pneumoniae, and RSV. Limitations of the study include the fact that the study design precluded an effective evaluation of the clinical relevance of the microbial findings. The relevance of, for example, an increased detection of S. aureus and H. influenzae by the qPCR assay is uncertain. Findings of S. aureus by the standard culture method in respiratory specimens are by clinical tradition and experience regarded important only in a few scenarios, for example, in chronic lung disease or with concomitant severe influenza A pneumonitis. Consequently, an increased detection by the new assay may or may not be relevant. Likewise, some clinicians may argue that the clinical value of increased detection of H. influenzae is uncertain.37
Conclusion
The results of this study suggest that the application of a qPCR assay to in parallel detect both bacterial and viral agents in respiratory specimens from CAP patients is feasible. The qPCR approach confers the obvious advantage of being much more rapid than standard microbiological methods and was found to be technically robust with good sensitivity for clinical specimens. In addition, an obvious and important clinical advantage is the panel-based strategy. This minimizes the risk that the causative search is biased by overlapping clinical presentations of distinct causes. Importantly, the assay can easily be modified, and additional target panels can be included to detect other causes, for example, those relevant for hospital-acquired pneumonia.
Acknowledgments
We thank Mats Sellin, Helen Edebro, Rafael Björk, Nina Ottosson, and Anna Kauppi for technical assistance and theoretical advice during the study. The work was performed in part at the Umeå Centre for Microbial Research (UCMR).
Footnotes
Supported by a grant connected to a Clinical Fellowship (A.E.) at the Laboratory for Molecular Infection Medicine Sweden and by Västerbotten County Council, Sweden (A.J.), grants ALFVLL-151871 and ALFVLL-402201.
Disclosures: None declared.
Supplemental material for this article can be found at http://dx.doi.org/10.1016/j.jmoldx.2015.01.005.
Supplemental Data
References
- 1.Huijskens E.G., Koopmans M., Palmen F.M., van Erkel A.J., Mulder P.G., Rossen J.W. The value of signs and symptoms in differentiating between bacterial, viral and mixed aetiology in patients with community-acquired pneumonia. J Med Microbiol. 2014;63(Pt 3):441–452. doi: 10.1099/jmm.0.067108-0. [DOI] [PubMed] [Google Scholar]
- 2.Caliendo A.M., Gilbert D.N., Ginocchio C.C., Hanson K.E., May L., Quinn T.C., Tenover F.C., Alland D., Blaschke A.J., Bonomo R.A., Carroll K.C., Ferraro M.J., Hirschhorn L.R., Joseph W.P., Karchmer T., MacIntyre A.T., Reller L.B., Jackson A.F., Infectious Diseases Society of America (IDSA) Better tests, better care: improved diagnostics for infectious diseases. Clin Infect Dis. 2013;57(Suppl 3):S139–S170. doi: 10.1093/cid/cit578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Welti M., Jaton K., Altwegg M., Sahli R., Wenger A., Bille J. Development of a multiplex real-time quantitative PCR assay to detect Chlamydia pneumoniae, Legionella pneumophila and Mycoplasma pneumoniae in respiratory tract secretions. Diagn Microbiol Infect Dis. 2003;45:85–95. doi: 10.1016/s0732-8893(02)00484-4. [DOI] [PubMed] [Google Scholar]
- 4.Gullsby K., Storm M., Bondeson K. Simultaneous detection of Chlamydophila pneumoniae and Mycoplasma pneumoniae by use of molecular beacons in a duplex real-time PCR. J Clin Microbiol. 2008;46:727–731. doi: 10.1128/JCM.01540-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Abdeldaim G.M., Strålin K., Korsgaard J., Blomberg J., Welinder-Olsson C., Herrmann B. Multiplex quantitative PCR for detection of lower respiratory tract infection and meningitis caused by Streptococcus pneumoniae, Haemophilus influenzae and Neisseria meningitidis. BMC Microbiol. 2010;10:310. doi: 10.1186/1471-2180-10-310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Greiner O., Day P.J., Altwegg M., Nadal D. Quantitative detection of Moraxella catarrhalis in nasopharyngeal secretions by real-time PCR. J Clin Microbiol. 2003;41:1386–1390. doi: 10.1128/JCM.41.4.1386-1390.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Peters R.P., van Agtmael M.A., Gierveld S., Danner S.A., Groeneveld A.B., Vandenbroucke-Grauls C.M., Savelkoul P.H. Quantitative detection of Staphylococcus aureus and Enterococcus faecalis DNA in blood to diagnose bacteremia in patients in the intensive care unit. J Clin Microbiol. 2007;45:3641–3646. doi: 10.1128/JCM.01056-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kais M., Spindler C., Kalin M., Ortqvist A., Giske C.G. Quantitative detection of Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis in lower respiratory tract samples by real-time PCR. Diagn Microbiol Infect Dis. 2006;55:169–178. doi: 10.1016/j.diagmicrobio.2006.01.007. [DOI] [PubMed] [Google Scholar]
- 9.Chen Y., Cui D., Zheng S., Yang S., Tong J., Yang D., Fan J., Zhang J., Lou B., Li X., Zhuge X., Ye B., Chen B., Mao W., Tan Y., Xu G., Chen Z., Chen N., Li L. Simultaneous detection of influenza A, influenza B, and respiratory syncytial viruses and subtyping of influenza A H3N2 virus and H1N1 (2009) virus by multiplex real-time PCR. J Clin Microbiol. 2011;49:1653–1656. doi: 10.1128/JCM.02184-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Coiras M.T., Aguilar J.C., García M.L., Casas I., Pérez-Breña P. Simultaneous detection of fourteen respiratory viruses in clinical specimens by two multiplex reverse transcription nested-PCR assays. J Med Virol. 2004;72:484–495. doi: 10.1002/jmv.20008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lassaunière R., Kresfelder T., Venter M. A novel multiplex real-time RT-PCR assay with FRET hybridization probes for the detection and quantitation of 13 respiratory viruses. J Virol Methods. 2010;165:254–260. doi: 10.1016/j.jviromet.2010.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brittain-Long R., Nord S., Olofsson S., Westin J., Anderson L.M., Lindh M. Multiplex real-time PCR for detection of respiratory tract infections. J Clin Virol. 2008;41:53–56. doi: 10.1016/j.jcv.2007.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Driscoll A.J., Karron R.A., Bhat N., Thumar B., Kodani M., Fields B.S., Whitney C.G., Levine O.S., O'Brien K.L., Murdoch D.R. Evaluation of fast-track diagnostics and TaqMan array card real-time PCR assays for the detection of respiratory pathogens. J Microbiol Methods. 2014;107:222–226. doi: 10.1016/j.mimet.2014.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jedrzejas M.J. Pneumococcal virulence factors: structure and function. Microbiol Mol Biol Rev. 2001;65:187–207. doi: 10.1128/MMBR.65.2.187-207.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Heiniger N., Spaniol V., Troller R., Vischer M., Aebi C. A reservoir of Moraxella catarrhalis in human pharyngeal lymphoid tissue. J Infect Dis. 2007;196:1080–1087. doi: 10.1086/521194. [DOI] [PubMed] [Google Scholar]
- 16.Liu D., Hollingshead S., Swiatlo E., Lawrence M.L., Austin F.W. Rapid identification of Streptococcus pyogenes with PCR primers from a putative transcriptional regulator gene. Res Microbiol. 2005;156:564–567. doi: 10.1016/j.resmic.2005.01.010. [DOI] [PubMed] [Google Scholar]
- 17.Strålin K., Bäckman A., Holmberg H., Fredlund H., Olcén P. Design of a multiplex PCR for Streptococcus pneumoniae, Haemophilus influenzae, Mycoplasma pneumoniae and Chlamydophila pneumoniae to be used on sputum samples. APMIS. 2005;113:99–111. doi: 10.1111/j.1600-0463.2005.apm1130203.x. [DOI] [PubMed] [Google Scholar]
- 18.Abdeldaim G.M., Strålin K., Kirsebom L.A., Olcén P., Blomberg J., Herrmann B. Detection of Haemophilus influenzae in respiratory secretions from pneumonia patients by quantitative real-time polymerase chain reaction. Diagn Microbiol Infect Dis. 2009;64:366–373. doi: 10.1016/j.diagmicrobio.2009.03.030. [DOI] [PubMed] [Google Scholar]
- 19.Martinon A., Cronin U.P., Wilkinson M.G. Development of defined microbial population standards using fluorescence activated cell sorting for the absolute quantification of S. aureus using real-time PCR. Mol Biotechnol. 2012;50:62–71. doi: 10.1007/s12033-011-9417-3. [DOI] [PubMed] [Google Scholar]
- 20.Fosheim G.E., Nicholson A.C., Albrecht V.S., Limbago B.M. Multiplex real-time PCR assay for detection of methicillin-resistant Staphylococcus aureus and associated toxin genes. J Clin Microbiol. 2011;49:3071–3073. doi: 10.1128/JCM.00795-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Templeton K.E., Scheltinga S.A., Beersma M.F., Kroes A.C., Claas E.C. Rapid and sensitive method using multiplex real-time PCR for diagnosis of infections by influenza a and influenza B viruses, respiratory syncytial virus, and parainfluenza viruses 1, 2, 3, and 4. J Clin Microbiol. 2004;42:1564–1569. doi: 10.1128/JCM.42.4.1564-1569.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Watzinger F., Suda M., Preuner S., Baumgartinger R., Ebner K., Baskova L., Niesters H.G., Lawitschka A., Lion T. Real-time quantitative PCR assays for detection and monitoring of pathogenic human viruses in immunosuppressed. J Clin Microbiol. 2004;42:5189–5198. doi: 10.1128/JCM.42.11.5189-5198.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ward C.L., Dempsey M.H., Ring C.J., Kempson R.E., Zhang L., Gor D., Snowden B.W., Tisdale M. Design and performance testing of quantitative real time PCR assays for influenza A and B viral load measurement. J Clin Virol. 2004;29:179–188. doi: 10.1016/S1386-6532(03)00122-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bustin S.A., Benes V., Garson J.A., Hellemans J., Huggett J., Kubista M., Mueller R., Nolan T., Pfaffl M.W., Shipley G.L., Vandesompele J., Wittwer C.T. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem. 2009;55:611–622. doi: 10.1373/clinchem.2008.112797. [DOI] [PubMed] [Google Scholar]
- 25.Tebbutt G.M., Coleman D.J. Evaluation of some methods for the laboratory examination of sputum. J Clin Pathol. 1978;31:724–729. doi: 10.1136/jcp.31.8.724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ibrahim A., Norlander L., Macellaro A., Sjöstedt A. Specific detection of Coxiella burnetii through partial amplification of 23S rDNA. Eur J Epidemiol. 1997;13:329–334. doi: 10.1023/a:1007385104687. [DOI] [PubMed] [Google Scholar]
- 27.Ye J., Coulouris G., Zaretskaya I., Cutcutache I., Rozen S., Madden T.L. Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics. 2012;13:134. doi: 10.1186/1471-2105-13-134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Codex Committee on Methods of Analysis and Sampling. 27th Session of the European Community Comments on items 4, 7 and 9, 2006 May 15-19. Budapest, Hungary. pp 1–6
- 29.Gallup J.M., Sow F.B., Van Geelen A., Ackermann M.R. SPUD qPCR assay confirms PREXCEL-Q softwares ability to avoid qPCR inhibition. Curr Issues Mol Biol. 2010;12:129–134. [PMC free article] [PubMed] [Google Scholar]
- 30.Nazarian E.J., Bopp D.J., Saylors A., Limberger R.J., Musser K.A. Design and implementation of a protocol for the detection of Legionella in clinical and environmental samples. Diagn Microbiol Infect Dis. 2008;62:125–132. doi: 10.1016/j.diagmicrobio.2008.05.004. [DOI] [PubMed] [Google Scholar]
- 31.Hallander H., Thore M., Olsson-Liljequist B., Linde A., Wollin R., editors. Referensmetodik: Nedre luftvägsinfektioner. ed 2. Swedish Institute for Infectious Disease Control; Solna: Sweden: 2005. [Google Scholar]
- 32.Johansson N., Kalin M., Hedlund J. Clinical impact of combined viral and bacterial infection in patients with community-acquired pneumonia. Scand J Infect Dis. 2011;43:609–615. doi: 10.3109/00365548.2011.570785. [DOI] [PubMed] [Google Scholar]
- 33.Musher D.M., Roig I.L., Cazares G., Stager C.E., Logan N., Safar H. Can an etiologic agent be identified in adults who are hospitalized for community-acquired pneumonia: results of a one-year study. J Infect. 2013;67:11–18. doi: 10.1016/j.jinf.2013.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Harada Y., Kinoshita F., Yoshida L.M., Minh le N., Suzuki M., Morimoto K., Toku Y., Tomimasu K., Moriuchi H., Ariyoshi K. Does respiratory virus coinfection increases the clinical severity of acute respiratory infection among children infected with respiratory syncytial virus? Pediatr Infect Dis J. 2013;32:441–445. doi: 10.1097/INF.0b013e31828ba08c. [DOI] [PubMed] [Google Scholar]
- 35.Goka E.A., Vallely P.J., Mutton K.J., Klapper P.E. Single and multiple respiratory virus infections and severity of respiratory disease: a systematic review. Paediatr Respir Rev. 2014;15:363–370. doi: 10.1016/j.prrv.2013.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Luchsinger V., Ruiz M., Zunino E., Martínez M.A., Machado C., Piedra P.A., Fasce R., Ulloa M.T., Fink M.C., Lara P., Gebauer M., Chávez F., Avendaño L.F. Community-acquired pneumonia in Chile: the clinical relevance in the detection of viruses and atypical bacteria. Thorax. 2013;68:1000–1006. doi: 10.1136/thoraxjnl-2013-203551. [DOI] [PubMed] [Google Scholar]
- 37.Hare K.M., Marsh R.L., Binks M.J., Grimwood K., Pizzutto S.J., Leach A.J., Chang A.B., Smith-Vaughan H.C. Quantitative PCR confirms culture as the gold standard for detection of lower airway infection by nontypeable Haemophilus influenzae in Australian Indigenous children with bronchiectasis. J Microbiol Methods. 2013;92:270–272. doi: 10.1016/j.mimet.2012.12.013. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.