Summary
Objective Following the severe acute respiratory syndrome (SARS) outbreak, many survivors were observed to suffer from psychosomatic symptoms reminiscent of various endocrine disorders. Hence, we sought to determine the existence of any chronic endocrine sequelae in SARS survivors.
Design, patients, measurements Sixty‐one survivors of SARS prospectively recruited were analysed for hormonal derangements 3 months following recovery. Patients with pre‐existing endocrine disorders were excluded. Any endocrine abnormalities diagnosed were investigated and treated where indicated up to a year. Serial evaluation facilitated characterization of trends and prognostication of any endocrinological aberrations.
Results Twenty‐four (39·3%) patients had evidence of hypocortisolism. The hypothalamic–pituitary–adrenal (HPA) axis dysfunction of the majority resolved within a year. Two (3·3%) of the hypocortisolic cohort had transient subclinical thyrotoxicosis. Four (6·7%) were biochemically hypothyroid, being comprised of three with central hypothyroidism and one with primary hypothyroidism. Two of the three with central hypothyroidism had concomitant central hypocortisolism. Eight had subnormal DHEAS levels.
Conclusions These preliminary findings highlight a possible aetiologic role of SARS‐associated coronavirus in causing a reversible hypophysitis or direct hypothalamic effect, with the HPA axis affected more frequently than the HPT axis.
Introduction
Ever since Singapore was stricken in March 2003 with severe acute respiratory syndrome (SARS), it became apparent that many survivors experienced chronic pulmonary morbidities and a multitude of psychosomatic manifestations.
We hypothesize that an endocrinological basis could account for some of these extrapulmonary symptoms due to their resemblance to various hormonal and metabolic disorders that frequently feature nonspecific symptomatology. 1 Studies have linked aberrations in the hypothalamic–pituitary–adrenal (HPA) axis to psychiatric conditions. 2 Moreover, correlations of hypothalamo–pituitary–thyroid (HPT) axis dysfunction with certain psychosomatic syndromes have been described. 3
The primary focus of this prospective study is to determine the existence of any chronic HPA axis sequelae in SARS survivors, while characterization of their prognostic outcomes and the presence of any HPT axis dysfunction constitutes its secondary objective. Given that the endocrinopathic properties of the SARS‐associated coronavirus (SARS‐CoV) is currently an inadequately explored domain, any hormonal aberrations unravelled through this preliminary investigation will contribute new scientific insights to the medical database with the potential of translating into clinically relevant therapeutic strategies for SARS‐associated endocrinopathies.
Patients and methods
Subjects recruitment
This research was conducted in Tan Tock Seng Hospital, the only nationally designated infirmary for isolation and treatment of all SARS patients, with approval from its Institutional Review Board. A total of 238 were infected with SARS, of whom 33 died. All survivors of probable SARS in Singapore aged 21 years and above were eligible. Those with pre‐existing endocrine disorders before SARS were excluded. Out of 205 survivors, two with underlying primary hypothyroidism were excluded, and 61 who fulfilled the inclusion criteria and provided written informed consent were prospectively enrolled about 3 months postdischarge, with the last batch of patients entered into the study by 30 September 2003 and followed up till 1 October 2004. Most of the remaining survivors not recruited did not consent for a variety of personal reasons, many of whom cited inconvenience, inability to be followed up over an extended period of 1 year or their reluctance to be subjected to laboratory investigations. A number of survivors refused to participate on grounds of being psychologically unprepared, or being in a state of grief over their deceased relatives who expired from SARS. Some remained noncontactable and could not be recruited and several were underaged individuals excluded for the purpose of this study. Using admission to the intensive care unit (ICU) as an arbitrary criterion for severe SARS, 46 were severe SARS cases admitted to the ICU of whom 20 survived; among them, eight severe SARS subjects were enrolled while 12 declined participation. Thus, among the survivors with severe SARS, 13·1% (8/61) were recruited while 8·3% (12/144) were not recruited. As such, more subjects with severe SARS were represented in the study cohort than those who were not entered into the study.
Sample collection and protocol
Blood samples were collected at 0800 h by venepuncture into EDTA and plain tubes for: 1) full blood count, 2) electrolytes, 3) cortisol, 4) ACTH, 5) free T4, 6) free T3, 7) TSH and 8) dehydroepiandrosterone sulphate (DHEAS). Twenty‐four‐hour urinary free cortisol (UFC) was done to assess integrated cortisol secretion. Subjects with serum cortisol below 275 nmol/l as cut‐off underwent dynamic HPA axis evaluation using low dose (1 µg), short Synacthen (ACTH‐1‐24) test (SST). Subjects with 0800 h serum cortisol below 138 nmol/l and/or poststimulation serum cortisol under 550 nmol/l at 30 min were deemed hypocortisolic. SST assessment for HPA axis recovery was repeated at about three to six monthly intervals. Although the study was terminated at 1 year, reviews were extended for those with persistent hypocortisolism as medically appropriate. Symptomatic patients with orthostatic hypotension were prescribed physiological doses of hydrocortisone replacement until their SST normalized.
Biochemical and immunoassay determinations
Blood was centrifuged, plasma separated, frozen and stored at −30 °C until assayed. Hormone measurement techniques including immunochemiluminometric assay (ICMA), radioimmunoassay (RIA) and immunoradiometric assay (IRMA) were employed according to standardized manufacturers’ protocols with detection limits, intra‐ and interassays coefficients of variations published in package inserts: FT4 (chemiluminescence immunoassay–ACS‐180‐Plus analyser, CIBA–Corning Diagnostic Corp., Medfield, MA), TSH (3rd generation ICMA‐Access TSH Assay, Sanofi Diagnostics Pasteur, Inc., Chaska, MN), FT3 (RIA‐Becton Dickinson and Co. kit, SmithKline Beecham Laboratories, Van Nuys, CA), DHEAS (RIA‐ Coat‐A‐Count DHEA‐SO4 kit, Diagnostic Product Corp., LA, CA), cortisol (solid phase RIA‐Diagnostic Product Corp., LA, CA), ACTH (immunoradiometric assay, Nichols Institute Diagnostics, San Juan Capistrano, CA). Twenty‐four‐hour UFC was measured by enhanced chemiluminescence immunoassay using Vitros ECi immunodiagnostic analyser.
Statistical analysis
Descriptive statistical analysis was performed on raw data where applicable. For stimulated cortisol end‐points at sequential assessments, paired numerical data were compared with the use of the nonparametric Wilcoxon signed ranks test. A two‐tailed P value of 0·05 or less was used as a criterion to indicate statistical significance. Analysis was done using spss statistical software package on a personal computer (version 12·0).
Ethical approval
The institutional review board (Research and Ethics Committee, Tan Tock Seng Hospital) approved the study on the basis that it complied with the declaration of Helsinki, 4 and that the protocol followed existing good clinical practice guidelines.
Results
Table 1 shows the demographic and clinical profile of the study population. Twenty‐four (39·3%) patients had hypocortisolism, of which 20 (83·3%) had unequivocal central hypocortisolism as evidenced by concomitant low or inappropriately normal ACTH levels (Table 2). Four (16·7%) had hypocortisolism associated with plasma ACTH above the upper reference limit of 11 pmol/l (50 pg/ml). Although this may superficially suggest primary hypocortisolism, the levels of ACTH were not elevated sufficiently to be diagnostic of primary hypocortisolism. In the absence of renin–aldosterone data and overt addisonian crisis, primary adrenal insufficiency was most unlikely, whereas secondary hypocortisolism appear to be the main form of HPA axis dysfunction encountered here. Indeed, the raised ACTH may in fact be a reflection of a recovering HPA axis following steroid‐induced suppression. Among these four patients, one who was given systemic steroids during SARS was probably a case of central hypocortisolism rather than primary hypocortisolism, being recruited at a time when the hypothalamo‐pituitary unit was recovering from transient HPA axis suppression, while the other three with elevated ACTH who had no steroids exposure during SARS or prior to SARS could arguably be cases of SARS‐induced central hypocortisolism showing evidence of early hypothalamo‐pituitary recovery rather than primary adrenal insufficiency.
Table 1.
Clinical characteristics of the study population (n = 61)
| Variable | Value |
|---|---|
| Age | 36·5 (25·5–47·5) |
| Gender (male:female) | 14:47 |
| Race | Chinese (39), Malay (13), Indian (7), others (2) |
| Use of corticosteroids during SARS | 10 |
| Number who required ICU care | 8 |
| Mechanical ventilation | 7 |
| Need for tracheostomy | 3 |
Table 2.
SST1, SST2 and SST3 represent the sequential order (i.e. 1st, 2nd, and 3rd) of the low dose short Synacthen test being performed during the follow‐up of hypocortisolic patients. Wilcoxon signed ranked test on 30 min F between SST1 and SST2 revealed a statistically significant improvement in stimulated serum cortisols over time (P < 0·001). (Abbrev: F is serum cortisol, UFC is urinary free cortisol, NR is normal range, while n is the number of patients)
| Patient | p.ACTH (pmol/l) (NR: 2–11) | 24 h UFC (nmol/day) (NR: 59–413) | SST1: F (nmol/l) n = 21 | SST2: F (nmol/l) n = 21 | SST3: F (nmol/l) n = 8 | |||
|---|---|---|---|---|---|---|---|---|
| 0 min | 30 min | 0 min | 30 min | 0 min | 30 min | |||
| 1 | 4·3 | 42 | 108 | 174 | 170 | 582 | – | – |
| 2 | 11·6 | 160 | 259 | 307 | 253 | 504 | Default | – |
| 3 | 16·9 | 106 | 368 | 391 | 322 | 588 | – | – |
| 4 | 3·0 | 118 | 58 | 207 | 269 | 649 | – | – |
| 5 | 4·6 | Not done | 214 | 279 | 188 | 485 | Default | – |
| 6 | 2·9 | 200 | 159 | 244 | 216 | 720 | – | – |
| 7 | 2·3 | Not done | 254 | 272 | 213 | 586 | – | – |
| 8 | 18·7 | 56 | 90 | 443 | 106 | 469 | Default | – |
| 9 | 3·7 | 150 | 225 | 262 | 251 | 708 | – | – |
| 10 | 3·2 | Not done | 146 | 273 | 190 | 528 | Default | – |
| 11 | 3·9 | 128 | 104 | 133 | 220 | 579 | – | – |
| 12 | 6·1 | 173 | 279 | 299 | 519 | 760 | – | – |
| 13 | 6·1 | Not done | 181 | 189 | 268 | 652 | – | – |
| 14 | 2·3 | 73 | 201 | 384 | 274 | 363 | 223 | 537 |
| 15 | 7·4 | 24 | 90 | 205 | 171 | 483 | 526 | 575 |
| 16 | 6·4 | 84 | 191 | 225 | 174 | 310 | 131 | 662 |
| 17 | 2·0 | 102 | 93 | 117 | 167 | 234 | 196 | 676 |
| 18 | 3·9 | 139 | 201 | 222 | 262 | 299 | 108 | 554 |
| 19 | 7·3 | 76 | 280 | 322 | 441 | 455 | 565 | 750 |
| 20 | 4·6 | 13 | 165 | 240 | 226 | 309 | 182 | 678 |
| 21 | 16·9 | 133 | 130 | 151 | 294 | 305 | 185 | 255 |
Among the 24 patients with hypocortisolism, three were deemed hypocortisolic based on 0800 h serum cortisol below 138 nmol/l, while 21 patients with borderline values underwent low‐dose SST for evaluation of HPA axis integrity. Because the treatment protocol for critically ill SARS patients managed in the ICU included the use of high‐dose systemic glucocorticoids, HPA axis suppression as a cause of central hypocortisolism from exogenous steroid exposure remains a possibility. Of the 24 patients with hypocortisolism, six received systemic glucocorticoids during SARS while one was an asthmatic on inhaled corticosteroids. Among the six patients treated with systemic glucocorticoids, three were on prednisolone up to a dose of 60 mg given for 2–5 days; the other three patients had intravenous methylprednisolone 200 mg for 3 days, followed by hydrocortisone 100 mg 8 hourly for about a week before being switched to oral prednisolone tapered over 10 days. Notably, the majority (18 patients) of those with hypocortisolism were glucocorticoid naïve. Four of six patients treated with high‐dose parenteral glucocorticoids who escaped hypocortisolism post‐SARS demonstrated the lack of prolonged suppression on the HPA axis with the corticosteroid regimen and doses utilized.
To support the notion that none in the study had pre‐existing subclinically impaired HPA axis, retrospective data of the standard 250 µg SST performed prior to the administration of glucocorticoids revealed 30 min stimulated serum cortisol exceeding 1000 nmol/l for most critically ill steroid‐treated cases in the ICU (Table 3), indicating intact HPA axis at the time of acquiring SARS. As a corroborative measure, to determine if hypocortisolism occurred during acute SARS in those steroid‐naïve patients without SST data, retrospective data of blood pressures recorded at the height of the febrile period were compared against that during apyrexial phase at discharge. This assumes that any patient with acute hypocortisolism developing in the midst of SARS would manifest profound hypotension. However, the average decrease in mean arterial pressure (MAP) was modest and comparable between the group who had hypocortisolism post‐SARS (−9·8 mmHg, n = 15) and those who remained eucortisolic (−10·4 mmHg, n = 17).
Table 3.
Baseline and stimulated plasma cortisols of 15 critically ill SARS patients (eight of whom survived and participated in our study) prior to parenteral glucocorticoid therapy
| Patient | Plasma Cortisol (nmol/l) 0 min | Plasma cortisol (nmol/l) 30 min | Plasma cortisol (nmol/l) 60 min |
|---|---|---|---|
| A | 1220 | 1640 | – |
| B | 619 | 1190 | 1410 |
| C | > 1700 | > 1700 | – |
| D | 683 | 1440 | 1700 |
| E | 877 | 1310 | – |
| F | 182 | 643 | – |
| G | 1060 | 1340 | – |
| H | 354 | 828 | – |
| I | 664 | 1100 | – |
| J | 633 | 660 | 691 |
| K | 880 | 1500 | 1600 |
| L | 1330 | 1420 | – |
| M | 702 | 1070 | 1250 |
| N | 1650 | > 1700 | – |
| O | 558 | 858 | – |
Using a poststimulated serum cortisol cut‐off of greater than 550 nmol/l for complete recovery of the HPA axis, 15 (62·5%) resolved within a year paralleled by resolution of orthostatic hypotension and a subjective sense of improved well‐being (i.e. reduced fatigue and postural dizziness compared with their perceived symptoms at the time of entering the study), while six (25%) had residual hypocortisolism of whom only one still required hydrocortisone replacement. Although we did not formally evaluate the spectrum of symptoms between those with normal vs. impaired HPA axis using standardized psychometric scales or instruments of health outcomes/quality of life (QoL) measurements (e.g. SF‐36), only 37·8% (14/37) of those with normal HPA axis compared with 87·5% (21/24) of those with impaired HPA axis volunteered having experienced fatigue and/or orthostatic dizziness at the point of recruitment.
Serial SST revealed a statistically significant improvement in stimulated cortisols among all hypocortisolic patients over time (P < 0·001) (Table 2). Four patients with suboptimal stimulated cortisols defaulted follow‐up. For those whose HPA axis resolved completely within a year, the average duration for recovery of the hypofunctional HPA axis was 5·9 ± 3·1 months. Among those patients with delayed recovery of the HPA axis beyond 12 months, there was no obvious linkage to age, sex, race or severity of illness. However, there was a clear trend of improvement over the course of follow‐up in stimulated adrenal cortisol output in all cases with hypocortisolism (Fig. 1).
Figure 1.
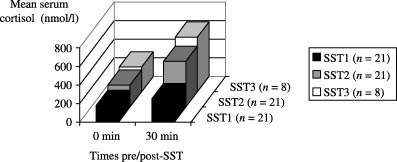
Results of short Synacthen test (SST) showing mean serum cortisol responses over a year of follow‐up of 21 survivors of SARS with hypocortisolism. SST1, SST2 and SST3 represent respectively, SST performed at the first, second and third clinic visit.
Two of the hypocortisolic cohort had subclinical thyrotoxicosis. Four were biochemically hypothyroid, comprising three with central hypothyroidism and one with primary hypothyroidism. Thyroid autoantibodies were positive only in the male patient with primary hypothyroidism (antithyroglobulin antibody positive and antithyroperoxidase antibody > 500 mIU/ml), though he had neither previous symptoms nor a history of thyroid disorder. Two of the three with central hypothyroidism had concomitant central hypocortisolism. Except for the patient with primary hypothyroidism who never recovered and still required L‐thyroxine 100 µg daily replacement at the close of the study, the other cases of thyroid dysfunction were transient and resolved between 3 and 9 months. We found low levels of DHEAS in eight (13·1%) patients after adjusting for age, sex and menopausal status. Within this group, there was only a male while the rest were women.
Discussion
SARS is a novel infection with significant morbidity and mortality posing a serious threat to mankind in this new millennium. 5 Observations of survivors of SARS during clinical follow‐up revealed a cluster of chronic extrapulmonary symptoms such as lethargy, malaise, lassitude, fatigue, weakness, orthostatic dizziness, anorexia, apathy, anxiety and depression. Although mostly vague, nonspecific and ill‐defined, these symptoms were reminiscent of the complaints seen in a host of endocrine disorders, including hypothyroidism, hypocortisolism, thyrotoxicosis, hypercalcaemia, hypopituitarism and Cushing's syndrome. Theoretically, this coronavirus can involve any organ during the viraemic phase. Because certain forms of hypophysitis, thyroiditis and adrenalitis have viral aetiologies, 6 , 7 , 8 it is instructive to determine the endocrinopathic impact of SARS‐CoV. Recently, researchers have examined the pulmonary and psychological aftermath of SARS survivors. 9 , 10 However, most studies were cross‐sectional with limited longitudinal data, and to our knowledge, endocrine derangements have not been reported thus far.
Our investigation set on record an unprecedented effort to chronicle the natural history of endocrine dysfunction, in particular that of the HPA axis, in SARS survivors for up to a year. Serial longitudinal evaluation facilitated characterization of trends and prognostication. A sizeable proportion surprisingly exhibited cortisol deficiencies rather than up‐regulated HPA axis or thyroid status typical in depression and panic disorder. 11 , 12 , 13 In this respect, the cortisol disturbances observed are analogous to that reported in chronic fatigue syndrome, post‐traumatic stress disorder and primary fibromyalgia syndrome, whereby the HPA axis appears to be under‐active. 14 , 15
Central hypocortisolism consequent to a pathological effect of SARS rather than glucocorticoid‐induced HPA axis suppression was suggested by absence of steroid use in nearly two‐thirds of this group. Moreover, the transient and minimally suppressive effect on the HPA axis of the glucocorticoid regimen used is backed by those with preservation of a normal response to the low‐dose SST despite having received high‐dose glucocorticoids. Taken together, this implies that the HPA axis impairment is probably a delayed pathological complication post‐SARS rather than merely be ascribed to suppression by exogenous steroid use. Other than mild reduction in MAP during pyrexia from SARS, the absence of overt hypotension during the acute febrile phase of SARS in survivors who developed hypocortisolism subsequently also suggests that SARS‐associated hypocortisolism is a late‐onset phenomenon. This is supported by evidence of those with intact HPA axis by standard dose SST during acute SARS who subsequently revealed impaired HPA axis by low dose SST at 3 months postdischarge.
Although speculative, the overall data are interpreted to be consistent with the hypothesis that hypocortisolism in SARS survivors develops gradually as a late complication several weeks after the onset of SARS infection. Because the majority of these patients were young (mean age 36·5 years), previously healthy and had no prior corticosteroid exposure, there is a strong likelihood that the SARS‐CoV was the aetiologic agent responsible for hypocortisolism.
To address the integrity of the HPA axis of SARS survivors postdischarge, the low‐dose 1 µg SST was employed as it was deemed potentially more appropriate and sensitive than the 250 µg SST in detection of any recent onset central hypocortisolism associated with minimal adrenal atrophy, in whom false negatives could result with the supraphysiological 250 µg SST. 16 , 17 , 18 , 19
The negative thyroid autoantibodies in all but one patient implicate SARS‐CoV rather than autoimmunity as a pathogenetic mechanism causing transient subclinical thyrotoxicosis or hypothyroidism. Nonthyroidal illness (i.e. ‘euthyroid sick syndrome’) was an unlikely confounder as these patients were completely apyrexial at the time of enrolment into the study. Although no radionuclide scans or thyroid biopsies were performed, we postulate that SARS‐CoV induced thyroiditis or hypophysitis, either alone or in combination, was the most probable reason for the thyroid dysfunction.
We had assayed DHEAS because there is evidence in the medical literature suggesting a correlation between a low DHEAS level and psychosomatic symptoms. 20 , 21 However, we found a much smaller number with DHEAS deficiency compared to those with hypocortisolism. This finding may be explained by the fact that adrenal androgen production is not solely regulated by ACTH, but also by extra‐adrenal peptides including β‐endorphin and β‐lipotropin, and by an intra‐adrenal neuroimmune system. 22 , 23 It is possible that DHEA replacement could further ameliorate symptoms in those with DHEAS deficiency. Because of the dramatic improvement of energy level, mood and resolution of dizziness after achieving satisfactory cortisol levels, DHEA replacement was not instituted in those affected, predominantly female patients in case adverse effects outweigh its benefits. We elected not to investigate the rest of the pituitary hormones and only to focus our attention on the HPA axis and thyroid dysfunction which were better defined in postviral syndromes.
Although there was a suggestion in the course of clinical follow‐up that patients with impaired HPA axis were relatively more symptomatic initially compared to a year later, we did not formally correlate endocrine dysfunction with psychosomatic symptoms to permit a rigorous conclusion as to whether subjects with impaired HPA axis had more symptoms than those with an intact HPA axis, or that symptoms significantly improved with recovery or treatment.
Intriguingly, such a link, if it exists, may be mediated by angiotensin‐converting enzyme (ACE), possibly interacting with neurotransmitter pathways yet to be understood. In this regard, alterations in the insertion/deletion (I/D) polymorphism in the ACE gene are associated with increased risk of chronic fatigue syndrome and idiopathic chronic fatigue. 24 Coincidentally, SARS‐CoV utilizes the angiotensin‐converting enzyme‐2 (ACE2) 25 and a C‐type lectin surface glycoprotein called L‐SIGN or CD209L 26 as critical portals of cellular entry. Perhaps ACE‐2 and/or CD209L could be expressed in the hypothalamus and pituitary where they may play a role in post‐viral fatigue pathogenesis, given our data of post‐SARS hypocortisolism, even though neither ACE2 nor CD209L has been shown to date to be expressed on any endocrine organ.
Finally, the overall prognosis of hypocortisolism in post‐SARS survivors appears favourable. Our results provide compelling evidence that the HPA and HPT axes are candidate targets of SARS‐CoV. Pending future SARS epidemics, adequately powered clinical studies to clarify this could prove daunting. In vitro studies and animal models of SARS might better define the endocrine lesions.
Conclusion
Overall, the data seem most consistent with SARS causing a reversible hypophysitis or direct hypothalamic effect, with the HPA axis more frequently perturbed than the HPT axis in SARS survivors. Although speculative, these findings highlight a possible aetiologic role of SARS‐CoV as an endocrinopathic factor deserving of future elucidation by in vitro, animal and clinical studies.
Acknowledgements
We appreciate nurse, SRN. Lee Mai for assisting in patient recruitment and biostatistician, Dr Zhu Wei, for providing helpful statistical analysis, and Ms. Veronica May – Gwen Leow for her excellent secretarial assistance. This study was supported by a grant from the Agency for Science, Technology and Research (A*STAR) Biomedical Research Council.
References
- 1. Frohman, L.A. & Felig, P. (2001) The clinical manifestations of endocrine disease In: Felig P., Frohman L.A. eds. Endocrinology and Metabolism, 4th edn McGraw‐Hill, New York, 19–28. [Google Scholar]
- 2. Kathol, R.G. , Noyes, R. & Lopez, A. (1988) Similarities in hypothalamic–pituitary–adrenal axis activity between patients with panic disorder and those experiencing external stress. Psychiatric Clinics of North America, 11, 335–348. [PubMed] [Google Scholar]
- 3. Kjellman, B.F. , Thorell, L.H. , Orhagen, T. , D’Elia, G. & Kagedal, B. (1993) The hypothalamic–pituitary–thyroid axis in depressive patients and healthy subjects in relation to the hypothalamic–pituitary–adrenal axis. Psychiatry Research, 47, 7–21. [DOI] [PubMed] [Google Scholar]
- 4. World Medical Association General Assembly. (2004) World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. Journal of International Bioethique, 15, 124–129. [PubMed] [Google Scholar]
- 5. Berger, A. , Drosten, C.H. , Doerr, H.W. , Sturmer, M. & Preiser, W. (2004) Severe acute respiratory syndrome (SARS) – paradigm of an emerging viral infection. Journal of Clinical Virology, 29, 13–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Sellmeyer, D.E. & Grunfeld, C. (1996) Endocrine and metabolic disturbances in human immunodeficiency virus infection and the acquired immune deficiency syndrome. Endocrine Reviews, 17, 518–532. [DOI] [PubMed] [Google Scholar]
- 7. Hautala, T. , Sironen, T. , Vapalahti, O. , Paakko, E. , Sarkioja, T. , Salmela, P.I. , Vaheri, A. , Plyusnin, A. & Kauma, H. (2002) Hypophyseal hemorrhage and panhypopituitarism during Puumala Virus Infection: Magnetic Resonance Imaging and detection of viral antigen in the hypophysis. Clinical Infectious Diseases, 35, 96–101. [DOI] [PubMed] [Google Scholar]
- 8. Parmar, R.C. , Bavdekar, S.B. , Sahu, D.R. , Warke, S. & Kamat, J.R. (2001) Thyroiditis as a presenting feature of mumps. Pediatric Infectious Disease Journal, 20, 637–638. [DOI] [PubMed] [Google Scholar]
- 9. Tso, E.Y. , Tsang, O.T. , Choi, K.W. , Wong, T.Y. , So, M.K. , Leung, W.S. , Lai, J.Y. , Ng, T.K. & Lai, T.S. , Princess Margaret Hospital SARS Study Group. (2004) Persistence of physical symptoms in and abnormal laboratory findings for survivors of severe acute respiratory syndrome. Clinical Infectious Diseases, 38, 1338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Chan, K.S. , Zheng, J.P. , Mok, Y.W. , Li, Y.M. , Liu, Y.N. , Chu, C.M. & Ip, M.S. (2003) SARS: prognosis, outcome and sequelae. Respirology, 8, S36–S40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Tafet, G.E. & Bernardini, R. (2003) Psychoneuroendocrinological links between chronic stress and depression. Progress in Neuropsychopharmacology and Biological Psychiatry, 27, 893–903. [DOI] [PubMed] [Google Scholar]
- 12. Ehlert, U. , Gaab, J. & Heinrichs, M. (2001) Psychoneuroendocrinological contributions to the etiology of depression, posttraumatic stress disorder, and stress‐related bodily disorders: the role of the hypothalamic‐pituitary‐adrenal axis. Biological Psychology, 57, 141–152. [DOI] [PubMed] [Google Scholar]
- 13. Abelson, J.L. & Curtis, G.C. (1996) Hypothalamic–pituitary–adrenal axis activity in panic disorder. 24‐hour secretion of corticotropin and cortisol. Archives of General Psychiatry, 53, 323–331. [DOI] [PubMed] [Google Scholar]
- 14. Demitrack, M.A. & Crofford, L.J. (1988) Evidence for and pathophysiologic implications of hypothalamic–pituitary–adrenal axis dysregulation in fibromyalgia and chronic fatigue syndrome. Annals of the New York Academy of Sciences, 840, 684–697. [DOI] [PubMed] [Google Scholar]
- 15. Yehuda, R. (2003) Hypothalamic–pituitary–adrenal alterations in PTSD: are they relevant to understanding cortisol alterations in cancer? Brain, Behavior, and Immunity, 17 (Suppl. 1), S73–S83. [DOI] [PubMed] [Google Scholar]
- 16. Oelkers, W. (1996) Dose‐response aspects in the clinical assessment of the hypothalamic–pituitary–adrenal axis, and the low‐dose adrenocorticotropin test. European Journal of Endocrinology, 135, 27–33. [DOI] [PubMed] [Google Scholar]
- 17. Rasmuson, S. , Olsson, T. & Hagg, E. (1996) A low dose ACTH test to assess the function of the hypothalamic–pituitary–adrenal axis. Clinical Endocrinology, 44, 151–156. [DOI] [PubMed] [Google Scholar]
- 18. Dickstein, G. , Spigel, D. , Arad, E. & Shechner, C. (1997) One microgram is the lowest ACTH dose to cause a maximal cortisol response. There is no diurnal variation of cortisol response to submaximal ACTH stimulation. European Journal of Endocrinology, 137, 172–175. [DOI] [PubMed] [Google Scholar]
- 19. Abdu, T.A.M. , Elhadd, T.A. , Neary, R. & Clayton, R.N. (1999) Comparison of the low dose short Synacthen test (1 µg), the conventional dose short Synacthen test (250 µg), and the insulin tolerance test for the assessment of the hypothalamic–pituitary–adrenal axis in patients with pituitary disease. Journal of Clinical Endocrinology and Metabolism, 84, 838–843. [DOI] [PubMed] [Google Scholar]
- 20. Kuratsune, H. , Yamuguti, K. , Sawada, M. , Kodate, S. , Machii, T. , Kanakura, Y. & Kitani, T. (1998) Dehydroepiandrosterone sulfate deficiency in chronic fatigue syndrome. International Journal of Molecular Medicine, 1, 143–146. [DOI] [PubMed] [Google Scholar]
- 21. Heuser, I. , Deuschle, M. , Luppa, P. , Schweiger, U. , Standhardt, H. & Weber, B. (1998) Increased diurnal plasma concentrations of dehydroepiandrosterone in depressed patients. Journal of Clinical Endocrinology and Metabolism, 83, 3130–3133. [DOI] [PubMed] [Google Scholar]
- 22. Genazzani, A.R. , Facchinetti, F. , Petraglia, F. , Pintor, C. , Bagnoli, F. , Puggioni, R. & Corda, R. (1983) Correlations between plasma levels of opioid peptides and adrenal androgens in prepuberty and puberty. Journal of Steroid Biochemistry, 19, 891–895. [DOI] [PubMed] [Google Scholar]
- 23. Ehrhart‐Bornstein, M. , Hinson, J.P. , Bornstein, S.R. , Scherbaum, W.A. & Vinson, G.P. (1998) Intraadrenal interactions in the regulation of adrenocortical steroidogenesis. Endocrine Reviews, 19, 101–143. [DOI] [PubMed] [Google Scholar]
- 24. Vladutiu, G.D. & Natelson, B.H. (2004) Association of medically unexplained fatigue with ACE insertion/deletion polymorphism in Gulf War veterans. Muscle and Nerve, 30, 38–43. [DOI] [PubMed] [Google Scholar]
- 25. Li, W. , Moore, M.J. , Vasilieva, N. , Sui, J. , Wong, S.K. , Berne, M.A. , Somasundaran, M. , Sullivan, J.L. , Luzuriaga, K. , Greenough, T.C. , Choe, H. & Farzan, M. (2003) Angiotensin‐converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature, 426, 450–454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Jeffers, S.A. , Tusell, S.M. , Gillim‐Ross, S.L. , Hemmila, E.M. , Achenbach, J.E. , Babcock, G.J. , Thomas, W.D. Jr , Thackray, L.B. , Young, M.D. , Mason, R.J. , Ambrosino, D.M. , Wentworth, D.E. , Demartini, J.C. & Holmes, K.V. (2004) CD209L (L‐SIGN) is a receptor for severe acute respiratory syndrome coronavirus. Proceedings of the National Academy of Sciences USA, 101, 15748–15753. [DOI] [PMC free article] [PubMed] [Google Scholar]


