Abstract
A wide array of molecular pathways has been investigated during the past decade in order to understand the mechanisms by which the practice of physical exercise promotes neuroprotection and reduces the risk of developing communicable and non-communicable chronic diseases. While a single session of physical exercise may represent a challenge for cell homeostasis, repeated physical exercise sessions will improve immunosurveillance and immunocompetence. Additionally, immune cells from the central nervous system will acquire an anti-inflammatory phenotype, protecting central functions from age-induced cognitive decline. This review highlights the exercise-induced anti-inflammatory effect on the prevention or treatment of common chronic clinical and experimental settings. It also suggests the use of pterins in biological fluids as sensitive biomarkers to follow the anti-inflammatory effect of physical exercise.
Keywords: Inflammation, Physical exercise, Neuroprotection, Communicable chronic diseases, Non-communicable chronic diseases, Biomarker
Highlights
-
•
Physical exercise provokes an adaptive response which depends on the type, duration and intensity of the stimuli.
-
•
The beneficial consequences of physical exercise on the health outcomes involve the modulation of the immune system.
-
•
Regular practice of moderate-intensity physical exercise directs the immune response to an anti-inflammatory status.
-
•
The measurement of BH4-related metabolites may represent a feasible way to assess the immune system status.
1. Introduction
The practice of physical activity and/or physical exercise is an efficient free-drug strategy for the prevention and treatment of several chronic diseases. A great body of scientific evidence has contributed to establish a positive correlation between a physically active lifestyle and health benefits [1]. According to the Physical Activity Guidelines for Americans, individuals affected by chronic diseases should practice a minimum of 150 min per week of moderate-intensity exercise, or at least 75 min per week of vigorous-intensity aerobic physical activity to improve health [2]. Additional health benefits can also be promoted by performing muscle-strengthening activities involving all main muscle groups at least twice a week [2].
Due to enhanced musculoskeletal function, cardiorespiratory and metabolic health, sleep, pain management, cognition, learning, memory and more, the practice of physical exercise represents a non-pharmacological primary clinical tool for the prevention of many diseases whose physiopathology is linked to acute or sustained immune system activation [[3], [4], [5]]. In general repeated moderate intensity exercise enhances the immune function response, reinforces the antioxidative capacity, reduces oxidative stress, and increases the efficiency of energy generation, therefore reducing the incidence of inflammatory diseases [6,7]. Acute bouts of physical exercise also regulate the immune response, i.e. by transiently redistributing immune cells to peripheral tissues, resulting in a heightened state of immunocompetence [8].
Inflammation is a biological response of the immune system that prevents, limits, and repairs damage by invading pathogens or endogenous biomolecules. Although acute inflammation is a transient inflammatory response and is beneficial to the organism, a persitent inflammatory response is associated with tissue dysfunction and pathology [9]. Examples of chronic non-communicable diseases with inflammation as a physiopathological key component are obesity, type 2 diabetes mellitus (T2DM), atherosclerosis, cardiovascular diseases, cancer, asthma, dementia and neurodegenerative diseases [6,[10], [11], [12], [13], [14], [15], [16]]. COVID-19 (coronavirus disease 2019) is a communicable disease responsible for the ongoing global pandemic [17]. Recent evidence has demonstrated that COVID-19 impairs the immune system response by inducing leukopenia with increased levels of pro-inflammatory mediators. Furthermore, this immune dysregulation might have a fatal outcome mainly in individuals with pre-existing medical conditions and in elderly patients in whom a chronic inflammation status is expected [18,19]. In this scenario, it has been shown that physical exercise improves overall health and reduces all-cause mortality risk (for a revision see ref. [20]). Indeed, the practice of physical exercise helps to manage T2DM [21], lower blood pressure, reduce the risk of heart attacks, hypertension and stroke [12], reduce risk of developing breast and colon cancer [22], protect from accelerated cognitive decline [23,24], and enhance immune responses to pathogens and vaccinations [8,25].
The activation of the immune system results in the release of cytokines that can be classified into pro- (including IL-1, IL-8, TNF-α, IFN-γ, VEGF, among others) or anti-inflammatory (i.e. IL-2, IL-4, IL-10, IL-13) factors [26]. Chronic physical exercise can guide the immune system response by favoring an anti-inflammatory status, which appears to be the key factor in improving health, mainly in chronic diseases [6]. In this review, we highlight the main findings in the literature on the (i) beneficial effect of physical exercise on the antioxidant system, on neurotrophic factors and on the microglia phenotype to promote neuroprotection; (ii) anti-inflammatory effect of the regular practice of moderate-intensity physical exercise on communicable and chronic non-communicable diseases, and finally we suggest (iii) the use of immune system activation biomarkers to follow the anti-inflammatory effects of physical exercise.
2. Material and methods
2.1. Search strategy and selection criteria
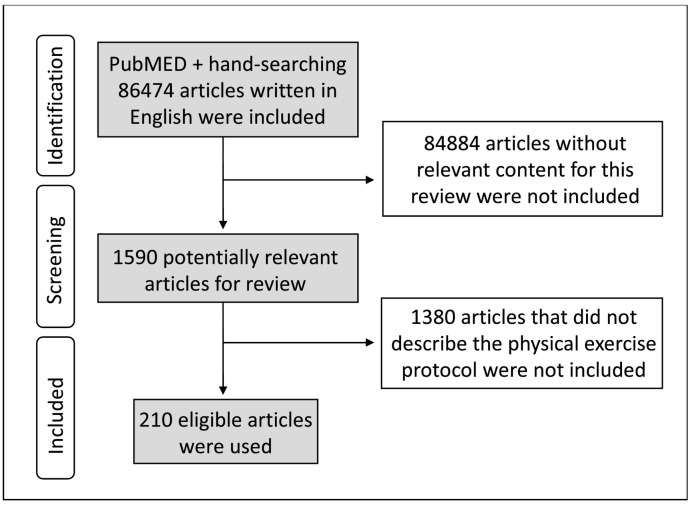
The United States' electronic database National Library of Medicine (PubMED) platform was used as a searching tool with the following indexers: physical exercise or physical activity combined with inflammation, immune system, biomarkers, neopterin, central nervous system, tetrahydrobiopterin or pain. Only articles written in English, published between 1973 and 2020, with the terms physical exercise or physical activity plus another key word (or synonym) in the title, abstract or keywords list were included. Articles without relevant content for this review, or that did not describe the physical exercise protocol were not included. Additional references were sought through hand-searching the bibliographies of relevant studies (Fig. 1 ). The searches were carried out between June 2019 and February 2020.
Fig. 1.
- Flowchart of literature search.
3. Definition of physical exercise
The Physical Activity Guidelines for Americans stresses that the effects of physical exercises that promote biochemical and physiological adaptations favoring health benefits are dependent on overload, progression, and specificity [2]. Physical activity is defined as any bodily movement produced by skeletal muscles, which results in energy expenditure above resting levels [27]. Examples of physical activity includes occupational activities (e.g., work, domestic cleaning), transport (e.g., walking or cycling to work) and leisure-time activities (e.g., playing, dancing) [27]. When physical activity is planned, structured, and repetitive and has as a final or intermediate objective the improvement or maintenance of physical fitness, it is defined as physical exercise [27] (the definition of the main terms related to exercise are presented in Table 1 ). Exercise bouts disrupt bodily homeostasis by provoking perturbations in different cells, tissues, and organs and thus increasing body's metabolic activity. The contracting skeletal muscles engaged in exercise consequently increase muscle energy and oxygen demands [27,28].
Table 1.
– Definition of physical exercise-related terms.
| Terms | Definitions |
|---|---|
| Physical activity | Any bodily movement generated by skeletal muscles which results in energy expenditure above resting levels [1,27] |
| Physical exercise | Planned, structured, and repetitive bodily movement performed in order to improve and/or maintain one or more components of physical fitness [1,27] |
| Physical fitness | A set of attributes that are either health- or skill-related. The degree to which these attributes can be measured by specific tests as stated in [27] |
| Physical function | Capacity of an individual to carry out physical activities. It is a direct reflection of motor function and control. Physical fitness and habitual physical activity are independent predictors of functional independence, disability, morbidity, and mortality [1] |
| Physical inactivity | State in which bodily movements are minimal and energy expenditure approximates the resting metabolic rate [29] |
| Sedentary behavior | Activity that involves little or no movement or physical activity, characterized by energy expenditure ≤1.5 METa, e.g., in a sitting, reclining, or lying posture [1] |
MET: metabolic equivalent of task (index of energy expenditure; 1 MET = 3.5 mL oxygen/kg/min uptake).
The practice of physical exercise can elicit acute or chronic biochemical and physiological responses based on the exercise frequency, volume and intensity (Fig. 1). Acute physiological responses are associated, for example, with the immediate effects of a single bout of exercise, which induce alterations in the homeostasis of the whole body, e.g., increase in the heart rate, transient tissue redistribution of immune cells [[30], [31], [32]]. Physical activity and physical exercise can be both acute and chronic. When a single bout of exercise (acute physical exercise) is repeated over time it becomes a chronic exercise, which can also be defined as exercise training [30,33]. The intensity of exercise determines the overall physiological responses to exercise training (see the classification of physical exercise intensities in Table 2 ), and it is typically expressed as a percentage of the maximum oxygen uptake (VO2max) of the individual. [1,28]. VO2max represents the maximum aerobic capacity and high energy use during exercise, and it is defined as the combined capacities of the pulmonary and cardiovascular systems to deliver oxygen to skeletal muscles, and the ability of those muscles to utilize it [28]. At rest, the average of the whole-body oxygen consumption in an adult individual is about 3.5 mL/kg/min. The resting body oxygen consumption is about 20%–25% of this value. Endurance athletes can exceed 25 times resting values, while in untrained subjects usually does not exceed 15 times [34].
Table 2.
– Intensities of physical exercise.
| Physical exercise intensity | VO2max* | MET& |
|---|---|---|
| Very light | <37% | <2 |
| Light | 37–45% | 2–2.9 |
| Moderate | 46–63% | 3–5.9 |
| Vigorous | 64–90% | 6–8.7 |
| Near-maximal to maximal | ≥ 91% | ≥ 8.8 |
MET: Metabolic equivalent of task (index of energy expenditure; 1 MET = 3.5 mL/kg/min oxygen uptake). *VO2max: Combined capacities of the pulmonary and cardiovascular systems to deliver oxygen to contracting skeletal muscles, and the ability of those &muscles to utilize it.
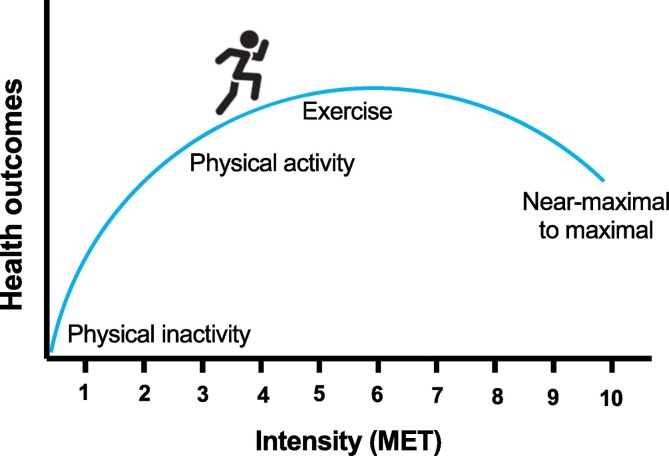
Many literature reports show that the regular practice of physical exercise can be considered as a tool to prevent and/or to treat numerous clinical conditions, e.g., high blood pressure, cardiovascular risk, insulin resistance and body mass percentage [12,21,22,35]. However, if exceeded the minimum recommended amounts of physical activity, more significant improvements could be obtained in terms of quality and physical fitness [1]. On the other hand, physical exercise below a minimum intensity, will not cause changes in body homeostasis, and therefore will not stimulate biochemical and physiological adaptations favoring a healthier lifestyle [1]. Indeed, a sedentary behavior increases, for example, the risk of dementia and the development of cognitive impairment [36]. On the other hand, vigorous intensity physical exercise is usually associated to adverse cardiovascular events such as sudden cardiac death or acute myocardial infarction [1,2]. Therefore, there is a dose–response relationship between physical exercise and health outcomes (Fig. 2 ). It implies that the practice of physical exercise, a free-drug treatment, takes into account the health or fitness outcome that the individual craves.
Fig. 2.
The practice of physical activity, physical exercise and exercise training induce beneficial health outcomes.
4. Immune system modulation by the practice of physical exercise
The practice of physical exercise inherently consumes energy, generates reactive oxygen species (ROS), and activates the immune system, having both, positive and eventually harmful effects depending on the type and degree of the activated immune system responses [37]. It has been widely described that the regular practice of physical exercise of moderate intensity promotes an anti-inflammatory status, and therefore, in long term, prevents the development of chronic diseases. However, high-intensity physical exercise or competitions, have been shown able to promote a pro-inflammatory scenario [[38], [39], [40]]. Although the hypothesis of the immunological “open window” after intense physical exercise is widely disseminated in the literature, some aspects regarding the immunological dynamics after acute bout of physical exercise remains controversial (for review see [8]).
The “open-window” hypothesis suggests that an impairment of the immune system after vigorous exercise increases the risk of contracting an upper respiratory system infection [3,8,32]. The immune system activation is a response to a stressor, aiming to restore cellular homeostasis. The inflammatory process plays a crucial role in the homeostasis, mainly through active defense against various harmful stimuli such as neurotropic viral infections and/or traumatic damage, promoting the reestablishment of cellular and tissue function [41,42]. The innate (natural) and acquired (adaptive) immune system are responsible for providing the primary defense against pathogens [43].
Pathogenic stimuli are conserved microbial structures, such as bacterial lipopolysaccharide (LPS), lipoproteins, flagellin, or bacterial and viral nucleic acids [42]. These structures are classified as molecular patterns associated with pathogens (PAMPs) or microorganisms (MAMPs) [44,45]. When they invade tissues and/or the circulatory system, PAMPs/MAMPs can initiate an inflammatory response mediated by cells of innate immunity, with consequent release of damage-associated molecular patterns (DAMPs). DAMPs are endogenous stimuli that can be damaged cells (debris) or molecules released during cell death such as ATP, which induce the production of pro-inflammatory cytokines in response to damage or stress [46].
Natural killer cells and phagocytes, including, neutrophils, monocytes and macrophages, represent the first line of innate immune system against viral infection and are highly responsive to acute aerobic exercise [47,48]. However, an increased susceptibility to upper respiratory tract infections (URTI), e.g., cold or influenza, can occur when the innate immune system is compromised [3,8]. Regular moderate-intensity exercise may reduce URTI symptoms; while vigorous acute physical exercise may increase the susceptibility to contracting an infection [43]. However, the modulation of the innate immune system to physical exercise can change according to the type, intensity and volume of exercise, or even with timing of measurement in relation to the training session. For example, after acute vigorous exercise occurs a dramatic influx of natural killer cells and CD8+ T lymphocytes which exhibit elevated cytotoxicity and tissue migrating potential [32].
The acquired immune response is developed throughout life, which can be through exposure to diseases or immunization by vaccines. Lymphocytes are the main cells involved in acquired immunity, and are responsible for secreting antibodies and cytokines, including IFN-γ and TNF- α [43,49]. During vigorous aerobic exercise is observed that peripheral blood lymphocyte is increased, suggesting an exercise-induced immune system activation [8]. However, 1-2 h post-exercise a decreased in the peripheral blood lymphocyte is observed, representing to the athlete a period of risk to contract infections and reinforcing the hypothesis that exercise promote a short-term window of immune suppression [32]. On the other hand, the lymphopenia described as a resulting of performing vigorous-intensity exercise might represent a heightened state of immunosurveillance and competence driven by a preferential mobilization of cells to more susceptible areas to infection after physical exercise (e.g., lungs and gut) [8].
The regular practice of moderate-intensity of exercise has been extensively recommended to counteract sustained low-grade chronic inflammation in the periphery and also in the brain [6,26]. Many experimental studies using mouse models have shown that physical exercise decreased hippocampal proliferation of microglia (macrophage-like immune cells of the CNS) [26,50,51], hippocampal expression of immune-related genes [52], nuclear NF-κB activation [52], and the expression of pro-inflammatory cytokines, such as TNF-α [53], IFN-γ [26] and IL-1β [54]. The anti-inflammation induced by chronic physical exercise has been demonstrated to involve a differential cytokine response represented by increased circulating IL-6 levels followed by a rise in IL-1ra and IL-10 levels and a suppression of TNF production [26,55]. The sustained modulation on the circulating levels of cytokines may influence the brain function, from behavior to several other vital functions [56]. For example, chronic exercise promotes the production IL-4 to enhance hippocampal-regulated learning and cognition by stimulating the synthesis of the neurotrophin brain-derived neurotrophic factor (BDNF) [56], to induce microglia to release BDNF, insulin-like growth factor (IGF-1) and transforming growth factor β (TGF-β), for enhancing neuronal functioning [56]. An anti-inflammatory status has also shown to increase the activity of specific transcription factors, i.e., REST (RE1-silencing transcription factor; otherwise called neuron-restrictive silence factor). REST, currently described as a facilitator of cognition, was demonstrated to be inactivated in individuals affected by cognitive disorders, including Alzheimer's disease and increased in the hippocampus of aged-mice submitted to a protocol of physical activity [57].
4.1. Blood–brain barrier and inflammation
The blood–brain barrier (BBB) regulates the passage of substances essential for brain health, such as nutrients and vitamins, besides defending the brain from invading agents, being therefore, essential for homeostasis and brain function [58,59]. The BBB is a complex structure consisting of endothelial cells, pericytes, vascular smooth muscle cells, astrocytes, microglia and neurons [59,60]. Interactions between these components have led to the concept of the neurovascular unit, where each cell type contributes to the BBB function [60,61]. The main structures responsible for the integrity of the BBB are the tight junctions [59]. The maintenance of adherence, gap and tight junctions between the different cell types within the neurovascular unit of the BBB is essential for CNS homeostasis [59]. The structural integrity of the barrier maintains the functionality of the brain, and its rupture or increased permeability might be associated with dysfunction and the development of many autoimmune neurological disorders [59,60].
BBB dysfunction indicate a decrease in barrier tightness and a gain of permeability [60]. The BBB disruption exposes the brain to peripheral cytokines and immune cells, which subsequently activates microglial cells and changes the extracellular environment promoting neurotoxicity (see the brain resident immune cells in Table 3 ) [62]. Impaired permeability also favor the entry of plasma proteins, toxins and leukocytes, which may compromise different brain regions, resulting in motor and cognitive impairment [59,62]. This process further increases the activation of the immune system creating a vicious cycle. In this context, the anti-inflammatory effect of physical exercise could either reduce peripheral inflammation, decreasing the entry of inflammatory cells in the brain or maintaining the density of tight junctions in the BBB (for a revision see ref. [61]).
Table 3.
– Function of brain resident immune cells.
| Brain resident immune cells | |
|---|---|
| Cell type | Physiological cell function |
| Microglia | Glial cell that inspect the local microenvironment and respond to injuries by releasing pro-inflammatory molecules; phagocytosis clearance of apoptotic cells [63] |
| Astrocytes | Help form the physical structure of the brain and play a number of active roles, including the secretion or absorption of neural transmitters and maintenance of the BBB [63] |
| Pericytes | Regulate the BBB permeability; the clearance and phagocytosis cellular debris; and has a neurogenic potential being able to differentiate into neurons [63] |
| Perivascular macrophages | Phagocytosis of cellular and pathogenic debris; maintains brain homeostasis; maintains tight junctions; initiation of the CNS acute phase response via the production of prostaglandins [63] |
BBB: blood brain barrier; CNS: central nervous system.
4.2. The anti-inflammatory facet of IL-6 driven by exercise
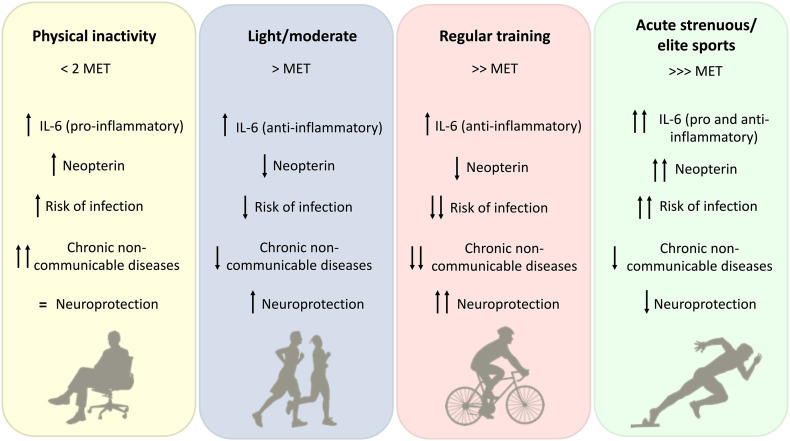
IL-6 is in general classified as a pro-inflammatory cytokine, since secreted by T-cells and macrophages, it promotes the immune system activation, leading to inflammation [64]. However, it has been shown that IL-6 and IL-6-induced acute phase proteins have anti-inflammatory and immunosuppressive effects when they come from skeletal muscle (myokine function), decreasing the pro-inflammatory response of the immune system [65]. This differential effect of IL-6 has been shown to be induced by physical exercise (Fig. 3 ). Muscle IL-6 production is controlled by Ca+2 and glycogen/p38 mitogen activated protein kinase (MAPK) stimuli, known to be associated to muscle contraction [66]. Usually, plasma IL-6 peak occurs as soon as the physical exercise ends, returning to basal levels in a few hours after recovery [67,68]. Modest increases of IL-6 are expected after exercise of low to moderate intensity or in intermittent physical exercise protocols of a shorter duration [68]. Under long-term physical exercise (over 2.5 h; e.g. marathon runners) muscle and blood IL-6 levels increase up to 100-fold, which is accompanied by raised levels of anti-inflammatory and cytokine inhibitors such as IL-1rα and IL-10 levels [26,55,68]. The release of these mediators by blood mononuclear cells may possibly represent a mechanisms to control exacerbated inflammation, especially in elite athlete [38,69,70].
Fig. 3.
Effects of physical inactivity and different intensities of physical exercise on the inflammatory response (IL-6 and neopterin) and health outcome (risk of infection, chronic non-communicable diseases and neuroprotection). MET: metabolic equivalent of task.
Reduction of the pro-inflammatory mediators IL-18, C-reactive protein, TNF-α and IL-1β levels [[71], [72], [73]], and a marked increase in IL-10 have been identified in blood from exercised individuals [74]. The increased anti-inflammatory IL-10 levels can counteract the effect of pro-inflammatory cytokines and to banish immune cells tissue infiltration by reducing the levels of adhesion molecules, such as ICAM-1, demonstrated through in vitro experiments using bone marrow cells [26,75].
The exercise-induced production of circulating IL-6 increases during and after physical exercise, while in the brain the levels rise more slowly [67,68,76,77]. By measuring the levels of IL-6 in arterial and jugular venous during and after prolonged exhaustive exercise, it was demonstrated that IL-6 is released also from the brain [76,77]. Increased brain IL-6 levels driven by physical exercise has been shown to promote many effects on glutamate release by astrocytes, therefore, controlling neuronal excitability, signal propagation within glial networks and synaptic transmission, and the release of MKP-1 (systemic macrophage-released MAPK phosphatise-1) that negatively regulates pro-inflammatory macrophages activation, demonstrated in male mice after chronic treadmill exercise-training [78].
5. Anti-inflammatory effects of physical exercise
The anti-inflammatory cytokines, including IL-10 and IL-6, belong to a group of regulatory mediators that govern the pro-inflammatory cytokine response mediated by IL-1β, IFN-γ, TNF-α, and others. In this scenario, the regular practice of physical exercise of moderate intensity can be considered as an immune system controller, able to improve defenses against infections and decrease the chances of chronic diseases (Fig. 3) [3,20].
The anti-inflammatory effects of physical exercise appear to be controlled by multiple mechanisms. Some of them are represented by (i) increased production of adrenaline, cortisol, growth hormone, prolactin, among others that have immunomodulatory effects by influencing leukocyte trafficking and functions [13,70]; (ii) visceral fat loss [79]; (iii) improved levels of anti-inflammatory myokines by the working skeletal muscle (see Table 4 ) [80], and (iv) diminishing the expression of TLRs in immune cells.
-
(i)
Physical exercise-induced release of stress hormones. Adrenaline and noradrenaline are the fast-acting fight or flight hormones produced by the adrenal glands on direct signaling from the brain under a stressful stimulus. Cortisol is a steroid hormone also produced by the adrenal glands, not released instantly as adrenalin, but rather taking minutes since it involves a stepwise activation of the amygdala. Both, adrenaline and cortisol, rapidly increase during exercise and once released, adrenaline activates the sequential releases of corticotropin-releasing hormone by the hypothalamus, adrenocorticotropic hormone by the pituitary gland, and finally of cortisol by the adrenal glands (for revision see ref. [26,70]). The responses induced by the release of adrenaline and cortisol typically are upstream of the immune system response.
Catecholamine signaling occurs though of the β-adrenoceptors present on T-cells, natural killer cells, macrophages and neutrophils, which increase intracellular cyclic adenosine monophosphate levels by G-coupled protein activation [70]. Ex vivo experiments using blood of healthy volunteers has been shown that catecholamines are able to reduce the production of cytokines induced by LPS, e.g. TNF-α and IL-1β, presenting an anti-inflammatory role [81]. In addition, the increased plasma cortisol levels appears to be mediated by exercise-induced IL-6 [55] and dependent to the intensity and duration of physical exercise [70]. After long-term physical exercise the cortisol show an immunosuppressive action, since cortisol has a role in maintaining the neutrophilia and lymphopenia [26].
-
(ii)
Physical exercise-induced anti-inflammatory effects on adiposity. Increased visceral adipose tissue (white adipose tissue; WAT) deposition is a risk factor for the developing of many chronic inflammatory conditions, including the pandemics of obesity, and T2DM, and the public health problems, chronic pain and cardiovascular diseases [[14], [15], [16]]. WAT is known to produce many adipokines, including TNF-α, IL-6, IL-18, leptin and retinol-binding protein 4. Excessive WAT provokes the reduction of plasma anti-inflammatory mediators, leading to the development of a chronic low-grade systemic inflammatory state [13,82]. Immune infiltration (macrophages and T-cells) into the WAT are known to facilitate and perpetuate the inflammatory state of this endocrine tissue in obesity [13]. Abdominal adiposity is known to positively correlate with a sedentary behavior [79]. In this scenario, it is widely known that the regular practice of physical exercise decreases the activation of the immune system in the WAT and prevents the risk of diabetes. The volume of physical exercise is proportional to the induced anti-inflammatory effect, for example, the greater the volume of the training (> 45 min), the bigger is the visceral adipose tissue mass loss [79].
-
(iii)
Increased production and release of anti-inflammatory myokines from contracting skeletal muscle. Contracting skeletal muscle produces myokines, such as IL-6, IL-15 and irisin, which exert beneficial effects on the adipocyte metabolism [79]. It has been demonstrated that infusion of IL-6 in healthy individuals delays gastric emptying with positive effects on postprandial glycemia [83], increases glucose uptake [84] and lipid mobilization and oxidation [85], reducing therefore the risk for metabolic disorders. Indeed, it has been demonstrated that the blockage of IL-6 signaling compromised the exercise-induced reduction of WAT in obese individuals [79].
IL-15 is also an anabolic factor present in the muscle and related with reduced WAT mass [80,86]. The high expression of IL-15 prevents rodents from the visceral fat deposit by increasing lean body mass, and it has been shown to be increased in trained human muscle [[86], [87], [88]]. Therefore, either IL-6 and IL-15 have a significant role in lipid metabolism by preventing obesity development.
Irisin, another myokine released into the circulation by exercise training, has demonstrated to induce thermogenic actions in adipose tissue in rodents and humans [89,90]. Irisin is a cleaved product from fibronectin type III domain-containing protein 5 (FNDC5), a transmembrane precursor protein expressed in muscle and released into the bloodstream through the control of the peroxisome proliferator-activated receptor-γ coactivator 1α (PGC-1α) [89]. Irisin has been recently shown to be linked to the exercise-induced beneficial effects in the cardiovascular, digestive, immune and adipose systems [89,91]. Moderate increased circulating irisin levels induced browning of WAT with increased expression of uncoupling protein 1 (UCP1), reducing obesity and insulin resistance in mice [89]. It has also been demonstrated that irisin has additional benefits by counterbalancing some of the comorbidities induced by obesity. Physical exercise-liked increased irisin in the blood and brain protects from cognitive impairment and improves memory in mice [24]. Irisin also has been correlated to bone mineral density in soccer players [92] and prevented bone loss in healthy mice [93].
Table 4.
– Example of myokines regulated by exercise in humans.
| Myokine | Biological function stimulated | Biological sample |
|---|---|---|
| IL-6 | Anti-inflammation | Plasma [94], skeletal muscle [95] |
| IL-7 | T-and B-cell development | Plasma [96], skeletal muscle [97] |
| IL-8 | Endothelial cell proliferation angiogenic factor | Plasma [96], skeletal muscle [98] |
| IL-10 | Anti-inflammation | Plasma [38], skeletal muscle [99], adipose tissue [100] |
| IL-15 | Maturation and redistribution of killer cells | Plasma [101], skeletal muscle [102] |
| BDNF | Neuronal development; synaptic plasticity; cell survival; differentiation | Serum [103] |
| Irisin | Thermogenesis and training adaptation to exercise; neuronal mitochondrial activity; neuroprotection; bone resorption | Plasma [24], CSF [24] |
| LIF | Platelet formation, haematopoietic cell proliferation, bone formation, neural survival and formation; muscle satellite cell proliferation | Skeletal muscle [104] |
| IGF-1 | Osteogenic factor | Plasma [105], skeletal muscle [106] |
| VEGF | Angiogenic factor | Plasma [107], CSF [108], skeletal muscle [109] |
BDNF: brain-derived neurotrophic factor; CSF: cerebrospinal fluid, IGF-1: insulin-like growth factor; IL: interleukin; LIF: leukemia inhibitor factor; VEGF: vascular endothelial growth factor.
More examples of myokines released by the practice of physical exercise are presented in Table 4.
-
(iv)
Reduced expression of TLRs in immune cells. Systemic inflammation is controlled by the activation of TLRs signaling. TLRs are highly conserved transmembrane proteins that play an important role in the detection and recognition of microbial pathogens, and they can also be activated by endogenous danger signals of tissue damage, such as heat shock proteins [26]. It has been demonstrated that physical inactivity correlates with augmented TLRs activation, and consequently, in the development of chronic non-communicable diseases. On the other hand, chronic exercise has been demonstrated to decrease cell-surface expression of TLRs on immune cells [72].
6. Physical exercise as a non-pharmacological intervention for neuroprotection
The regular practice of physical exercise is known to be beneficial for the CNS health and function, counteracting age-related cognitive decline and dementia risk in humans and rodents [36,110]. A great body of evidence has shown that exercise improves neurological impairments induced by brain injury and promotes functional recovery by preventing neuronal loss [111]. Moderate physical exercise training has been shown to promote neuroplasticity [112], neurogenesis [113] and neuroprotection [114], in addition to hippocampus-dependent learning and memory [115,116].
Most of the pre-clinical studies reporting physical exercise-induced neuroprotection have been based on the voluntary paradigm by using the running wheel. The beneficial effects of voluntary exercise in the brain are postulated to be related to increased expressions of neurotrophic factors, including BDNF [115,117], glial cell-derived neurotrophic factor (GDNF) [112], fibroblast growth factor 2 [118], IGF-1 [119] and REST [57,120]. Additionally, mediators circulating in the blood released by the skeletal muscle, such as irisin, also regulate the positive effects of exercise on the CNS, stimulating BDNF expression in the mice hippocampus [24,121]. In line, it has been shown that experimental peripheral inflammation compromises neurogenesis, reduces the content of neurotrophic factors [122] and impairs cognitive function in mice [123].
Neuroinflammation is a common hallmark in many disorders of the CNS, e.g., depression, Alzheimer's disease and Parkinson's disease [[124], [125], [126]], and it is responsible for the pathological reactive activation of glial cells (microglia and astrocytes) [125]. Microglial cells may support neuronal function due to the capacity to secrete anti-inflammatory mediators, facilitate phagocytosis and cell repair when required, and to scavenge ROS. Microglia is the primary immune cell in the CNS and under stress adopts a pro-inflammatory phenotype releasing, e.g., TNF-α and IL-1β [127]. In line, treadmill exercise protocol with low to moderate intensity decrease the hypothalamic microglial activation in an animal model of obesity induced by high-fat diet [128]. In addition, similar training protocol improved motor behavior and microglial activation by reducing the NOS II (nitric oxide synthase II) expression, involved in microglial oxidative burst [129]. Microglia activation has been related with reduction of dopaminergic neurotransmission in the substantia nigra pars compacta (SNpc), contributing to the physiopathology of Parkinson's disease [130]. In this context, the beneficial effects of physical exercise protocols, voluntary or forced, on the brain of animal models of Parkinson's disease have been widely demonstrated in the literature. Six weeks of moderate-intensity exercise on the treadmill improved dopamine metabolism in the striatum of C57BL/6 mice treated with the neurotoxin 1-methyl-4phenyl-1,2,3,6-tetrahydropyridine (MPTP) [131]. Additionally, eight weeks of strength or running treadmill protocols were able to promote neuroprotection in striatum and hippocampus of adult male mice treated with 6-hydroxydopamine (6-OHDA) [132].
Other CNS alterations has shown altered monoaminergic metabolism, like the down-regulation of the biosynthesis of serotonin, which is implicated in the physiopathology of depression [133]. In addition, increased blood levels of pro-inflammatory mediators, e.g., IL-8 and TNF-α, with low levels of BDNF have been documented in individuals affected by depression [[134], [135], [136]]. Exercise is considered a first-line treatment in mild to moderate depression [135,137,138]. It has been shown that aerobic exercise on the treadmill significantly upregulated the expression and content of central components of the BDNF-serotonin system in different regions of the brain, e.g. cortex and hippocampus, in adult rats physically trained lifelong (from 2 to 18 months of age) [139]. In addition, BDNF released from human brain has been shown to increase with an acute bout of exercise [140,141]. Besides that, six weeks of voluntary exercise on the running wheel decrease anxiety-like behaviors and exerted antidepressant-like effects, which were accompanied by stimulated hippocampal neuroplasticity, mitochondrial activity and gene expression associated energy metabolism and BDNF regulation in C57BL/6 mice [112].
In this context, irisin stimulates the expression of neurotrophins (e.g., BDNF) and neuroprotective proteins (e.g., cFOS and ARC) in the mouse CNS [24]. Indeed, recently it was demonstrated that FNDC5/irisin is an essential pathway involved in synaptic plasticity and memory induced by exercise in an Alzheimer's disease experimental model using C57BL/6 mice [24]. Another important brain factor stimulated by exercise is REST ([120] and revised in [23]). REST is a master transcription factor that down-regulates genes linked to cell-death in differentiated neurons and enhances the expression of other genes that induce resistance to the ageing process and the preservation of cognitive function [23]. Our group demonstrated that experimental chronic hyperglycemia in Wistar rats provoked a marked REST reduction specifically in the hippocampus [120,142]. However, our group also demonstrated that eight weeks of voluntary physical activity increased REST expression, which was also accompanied by an increase in BDNF expression in the hippocampus of aged C57BL/6 male mice [57]. Thus, the relationship between enhanced cognition and REST expression appears to be related to the repression of cell death-associated genes, suggesting that REST might be neuroprotective [23].
7. Physical exercise and neopterin
Neopterin has been used as a sensitive clinical biomarker for immune system activation for over decades. Increased neopterin levels in biological fluids have been proposed by our group and others to be a biomarker of exhaustive or high intensity physical exercise-induced inflammation [39,40,135,[143], [144], [145], [146]]. Many reports have demonstrated that a sedentary lifestyle or reduced physical activity have increased levels of several markers of inflammation, such as C-reactive protein, IL-18, IL-6, TNF-α, that correlated with increased neopterin levels in the blood [64,145,147]. These inflammatory mediators have also shown to be increased in individuals with cognitive impairment [145,147]; indeed, our group showed for the first time in a pre-clinical paradigm that neopterin is an endogenous compound that facilitates learning and memory in a non-inflammatory scenario [148]. In addition, we showed that neopterin increases the antioxidant capacity of the cell, enhances mitochondrial bioenergetics, and reduces inflammation and oxidative stress in the brain of naïve mice [149,150]. In agreement, the regular practice of moderate-intensity physical exercise has demonstrated to correlate with lower levels of neopterin in biological fluids [143]. Therefore, exercise can tailor the immune responses and be quantified by assessing neopterin levels.
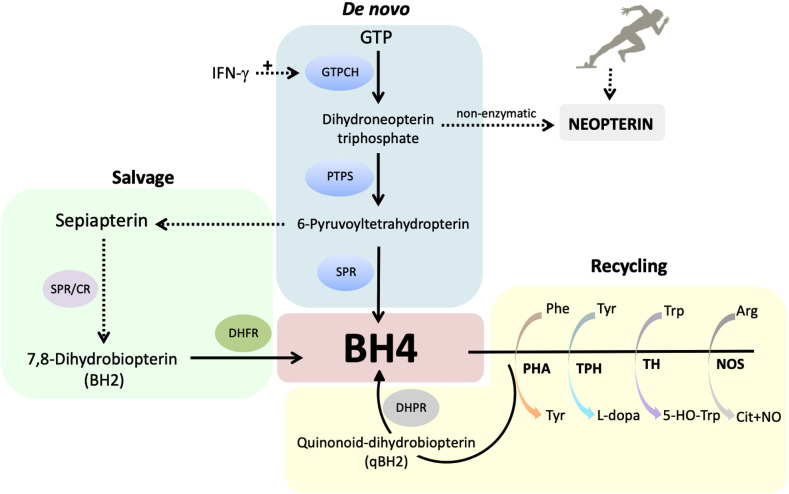
Neopterin is a pteridine and a byproduct of the de novo tetrahydrobiopterin (BH4) biosynthesis. Guanosine triphosphate cyclohydrolase I (GTPCH; gene: Gch1) is the rate-limiting enzyme of the de novo pathway and it is transcriptionally controlled by inflammation. Gch1 expression can increase up to 100-fold during inflammation, however, the two subsequent enzymatic steps responsible for the synthesis of BH4 remain unchanged or slightly increased, determining a pseudo-metabolic blockage with accumulation of a metabolic intermediate that finally will generate neopterin. The production of this soluble compound is mainly favored by IFN-γ stimulation in a variety of cells (see Fig. 4 ) (metabolic pathways have been reviewed by Ghisoni and collaborators [149]).
Fig. 4.
Intracellular levels of tetrahydrobiopterin (BH4) are controlled by three metabolic pathways: de novo synthesis, salvage and recycling pathways. CR: carbonil reductase; DHFR: dihydrofolate reductase; DHPR: dihydropteridin reductase; GTPCH: GTP cyclohydrolase I; IFN-γ: interferon-γ; NO: nitric oxide; NOS: nitric oxide synthase; PHA: phenylalanine hydroxylase; PTPS: 6-pyruvoyl tetrahydropterin synthase; SPR: sepiapterin reductase; TH: tyrosine hydroxylase; TPH: tryptophan hydroxylase.
Our group was the first to demonstrate that human primary neurons, astrocytes and microglia, release neopterin under inflammatory stimuli, being therefore the measuring of neopterin also an index of neuroinflammation [151]. We also showed that a pro-inflammatory state stimulates the production and release of neopterin by brain cells in the mouse hippocampus and striatum [149]. A well-known and potent stimulator for the formation of neopterin is the pro-inflammatory cytokine IFN-γ, that is secreted by Th1 cells during activation of the immune system [149,151]. Neopterin levels also increase in plasma from patients with neurodegenerative diseases and with progressive dementia [152] and patients with acute bacterial infections [153].
When neopterin production is induced by physical exercise, its levels depend on the physical exercise workload, training volume and specificity [40,135,146] (see Table 5 ). Our group showed that neopterin is increased in the blood of athletes who completed a multi-sport competition, including off-road running, mountain biking and kayaking (total 90 km). The increased levels of neopterin occurred in parallel with muscle damage [40]. Urinary neopterin levels were also shown to be increased in rugby and marathon athletes and the levels correlated with other inflammatory and oxidative biomarkers [39,154]. Neopterin has also been used as a biomarker in response to mixed martial arts [155] and body-building training [156] and has been proposed as a biomarker of fatigue and overtraining [157]. However, recently, experimental data from our group indicated that moderate intensity-treadmill running training induces an anti-inflammatory environment with reduced levels of neopterin in an animal model of inflammation induced by LPS. Mechanistically, neopterin may activate the transcription factor Nrf2, a key regulator of the cellular antioxidant response, which will further enhance the cellular antioxidant defenses, mitochondrial activity and the anti-inflammatory environment [[148], [149], [150], [151]]. Supporting this state, our group demonstrated that six weeks of moderate-intensity physical exercise on a treadmill induced the expression of Nrf2 in the brain mouse [114].
Table 5.
– Physical exercise modality and neopterin levels.
| Physical exercise type | Intervention/training protocol | Biological sample | Method of analysis | Levels | Effect on neopterin levels |
|---|---|---|---|---|---|
| Rodents | |||||
| Moderate running in a treadmill [143] | 5 session/week for 3 weeks (30, 40 and 50 min/day), no inclination | Urine | HPLC | ~12 μmol/mol creatinine | Reduction of exacerbated levels under inflammation |
| Humans | |||||
| Barcelona Olympic Games [157] | 17-day period of training at altitude (2030 m) followed by a 10-day period of heat acclimatization (mean daily maximum ambient temperature 27 °C). The following week, the athletes performed the Olympic competition. The average training time was approximated to 3 h/day | Urine | HPLC | ~219 μmol/mol creatinine | Increased from pre-training on day 19 of training |
| Ultra-endurance cycling athletes Race Across America [158] | 4763 km from San Diego to Atlantic City | Urine | HPLC | ~290 μmol/mol creatinine | Increased on day two to four of the race |
| Rugby players [39] | Rugby game | Urine | HPLC | ~4000 nmol/SG1.020 | Increased post-game |
| Semi-professional rugby players [159] | Rugby game | Urine | HPLC | ~10,000 nmol/SG1.020 | Increased post-game |
| Ultra-endurance athletes MultiSport race [40] | 28.5 km of trail running, 42.5 km mountain biking and 17.5 km kayaking | Plasma | HPLC | ~18 nmol/L | Increased after race |
| Body builders [156] | Each subject would train 1 to 2 body parts per day, 3–6 exercises per body part, 3–6 sets per exercise and 8–12 repetitions/set | Urine | HPLC | ~200 μmol/mol creatinine or ~3000 nmol/SG1.020 | No changes over the week |
| Ultra-marathon runners [154] | 330 km, altitude difference of 24,000 m (3 times the Everest), 25 mountain passes above 2000 m sea level | Urine | HPLC | ~300 μmol/mol creatinine | Increased after race |
| Exhaustive aerobic physical exercise [144] | Cycle ergometer physical exercise started at a workload of 50/75 W (women/men) for 5 min (warm up) with an increase in workload of 25 W/min until exhaustion. After a 20 min resting period, athletes with a maximal aerobic capacity performed a 20-min maximal time-trial | Serum | ELISA | ~11 nmol/L | Increased post physical exercise |
| Mixed martial arts [155] | 3 rounds of 5 min in a simulated contest with 1 min between rounds. Each subject competed against several opponents throughout 15 min session. Each participant was subsequently involved in the other subject's “rounds” resulting in a combined time totaling 90 min of physical exercise | Urine | HPLC | ~8000 nmol/SG1.020 | Increased post-session |
SG1.020 = Specific gravity (SG) corrected concentration; HPLC = high performance liquid chromatography.
Table 5 shows studies from literature relating the type of physical activity with neopterin levels in humans and animals.
7.1. Physical exercise and ROS production
It is well stablished in the literature that physical exercise induces alterations in oxidative metabolism in different tissues [38,40,160,161]. Cellular stress response represents an adaptive protective mechanism to maintain or reestablish cellular homeostasis to survive under unfavorable environmental conditions [162]. The balance between ROS generation and antioxidant system is maintained by many interplaying mechanisms, and a disbalance may determine the degree of oxidative stress [163,164]. ROS and moderate-intensity physical exercise can induce appropriate levels of eustress, an important stimulus for adaptations and health benefits induced by physical exercise [161,165]. On the other hand, the disruption of redox homeostasis will induce oxidative stress provoking deleterious effects damaging biomolecules, including lipids, proteins and DNA, leading to severe cell damage and contributing to the physiopathology of a variety of chronic pathological conditions [164,166]. In fact, decreased performance, fatigue and muscle damage have been associated with enhanced production of ROS [10,40].
Under inflammatory conditions phagocytic cells like neutrophils and macrophages generate large amounts of ROS, reactive nitrogen species (RNS) and chlorine species [167]. Inflammation-driven oxidative stress activates NF-κB, a master transcription factor that controls the priming of inflammation and enhances oxidative metabolism [168]. This mechanism has been demonstrated in many scenarios, including in immune cells-infiltrated muscle [167,169]. Additionally, ROS production has been shown to be an activator of the NLRP3 inflammasome, an intracellular receptor for DAMPs that promotes the activation of pro-inflammatory cytokines [151]. ROS activating inflammasomes may be formed in the mitochondria and also in the cytosol by xanthine oxidase [170]. The generated of reactive species is crucial to the remodeling that occurs in skeletal muscle in response to physical exercise training. However, the changes caused by the physical exercise in the oxidative metabolism depend on the type, volume and workload of the training and are specific to each tissue [12,171]. For example, it has been shown that the inflammatory peak occurs in the muscle during the first hours after an acute bout of resistance exercise [[172], [173], [174]]. However, moderate and chronic physical exercise reduces serum prostaglandin (biomarker of inflammation) levels in humans [175].
NADPH oxidase is also a potential ROS generator in the skeletal muscle during exercise [176]. In the presence of ADP and Fe3+, NADPH oxidase catalyzes electron transfer from NADPH to molecular oxygen to form radical superoxide [177]. Superoxide production is increased by NADPH oxidase during the respiratory burst, which if promptly converted into hydrogen peroxide by the dismutase superoxide, and finally into hydroxyl radicals and hypochlorous acid by the Fenton reaction or the action of myeloperoxidase [177]. During and after submaximal physical exercise the myeloperoxidase enzyme is significantly increased [178]. In addition, ROS generation, such as hypochlorous acid, is able to inhibit ATPase activity via a modification of sulfhydryl groups on the protein in the muscle cells [179]. ROS production during high-intensity or acute physical exercise is also followed by a decrease in antioxidant system [180]. On the other hand, studies have demonstrated that endurance training favor the cellular antioxidant defenses and appropriate oxidative capacity in many tissues, such as skeletal muscle [7,38,143], the adipose tissue [181] and the brain [112,182].
8. Physical exercise and inflammation-induced pain: Relevance of BH4
Pain is a distressing experience associated with actual or potential tissue damage with sensory, emotional, cognitive, and social components [183,184]. Inflammatory response has an important role in pain and nociception by sensitizing nociceptor neurons, mainly through the production of inflammatory cytokines [185]. The physiopathology of many painful diseases, such as, rheumatoid arthritis and inflammatory bowel diseases have been linked to the exacerbated production of pro-inflammatory cytokines [186,187]. Inflammation-induced pain is the pain induced by tissue damage and immune system activation. Non-steroidal anti-inflammatory drugs (NSAIDS) have long been used to treat acute pain, and after the disease is solved the hypersensitivity typically returns to basal levels [188]. However, when inflammation is sustained, classical painkillers are not effective to resolve pain and the condition becomes chronic. Chronic pain is classified by the International Association for the Study of Pain as a pain that lasts or recurs for more than three months [189]. Chronic pain is characterized by its long-term nature and abnormal sensitivity to thermal and mechanical stimuli. Such hypersensitivity can be expressed as hyperalgesia, an excessive reaction to normally painful input; allodynia, a painful response to normally innocuous stimuli; or spontaneous pain with no identifiable cause [189]. Chronic pain has not a safe and effective pharmacological treatment; therefore, the management of chronic pain is a major unmet clinical.
Several studies have shown that the exacerbated production of pro-inflammatory cytokines in the periphery and the CNS is associated with the appearance and progress of pain [190]. Tissue infiltration of macrophages and monocytes has been extensively demonstrated to be involved in chronic painful disease conditions [[191], [192], [193]]. Tissue injury is sensed by both tissue-resident macrophages, that produce inflammatory substances and tissue repair, and by nociceptors which allow pain feeling in the injured area [190]. Cytokines IL-1β, IL-6, TNF-α, IL-17A and IL-15 are known stressors of sensory neurons [186]. Furthermore, the silencing of the neuro-immune interphase has been proposed as a new avenue to treat chronic pain [194].
The relationship between the development of inflammatory and neuropathic pain with pathological BH4 production has been recently demonstrated by our group in multiple animal models of chronic pain [187,195,196]. BH4 is an essential cofactor for the enzyme activity of aromatic amino-acid hydroxylases, all nitric oxide synthase isoforms, and alkylglycerol monooxygenase [197]. BH4 intracellular levels play, therefore, a key role in various biological systems, e.g., monoamine neurotransmitter synthesis [197], cardiovascular and endothelial functions [198], the immune system activation [195] and pain thresholds [195,196,199]. The levels of BH4 are tuned and finely regulated by three metabolic pathways: de novo synthesis, recycling and the salvage pathway (see BH4 pathways in Fig. 4) [200]. The de novo pathways produce BH4 from GTP by three enzymatic steps catalyzed by the enzymes GTPCH, PTPS and sepiapterin reductase (SPR) [200]. The salvage pathway rescues metabolic intermediates of the de novo pathway to produce sepiapterin and 7,8-dihydrobiopterin (BH2) to form BH4 or it directly reduces the de novo intermediate to BH4, maintaining BH4 appropriate cell concentrations, while saving ATP (in equilibrium with GTP) in energy-demanding situations like inflammation [197]. Finally, once BH4 is oxidized to BH2, during its role as an enzyme cofactor, a NADH-dependent reaction will efficiently recycles it back to BH4 [200].
The initial identification of single nucleotide polymorphisms in the Gch1 locus and its association with reduced clinical pain scores in individuals affected by chronic painful conditions, allowed to validate the involvement of the BH4 metabolism in human chronic pain [199]. Our group demonstrated that the BH4 pathway is upregulated in damaged sensory neurons and that the reduction of exacerbated and pathological levels of BH4 confers analgesia in multiple animal models for chronic pain [187,195,196].
It is known that having an active lifestyle helps to manage and prevent chronic pain. The regular practice of physical activity has been indicated to non-pharmacologically treat many painful disorders, since it had similar clinical outcomes as to the use of NSAIDS and non-narcotic analgesics [201,202]. Low-intensity aerobic physical exercise during four weeks showed to reduce neuropathic pain by decreasing mechanical hypersensitivity and pro-inflammatory cytokines in the sciatic nerve and spinal cord [203]. Physically active animals (two weeks of exercise on a treadmill) with neuropathic pain show increased M2 (secrete anti-inflammatory cytokines) and less M1(secrete pro-inflammatory cytokines) polarization of macrophages at the site of the injury, while sedentary mice showed increased M1 over M2 macrophages in the sciatic nerve [204]. Moreover, the blockade of IL-10 systemically and locally in the muscle reduces the physical exercise-induced analgesic effect [205]. Regular practice of six weeks of voluntary running wheel has also shown to increase the anti-inflammatory cytokines in serum after chronic constriction injury [206]. In addition, low-intensity aerobic exercise on the treadmill has shown to reduce IL-1β and TNF-α levels in sciatic nerve and spinal cord in an animal model of sciatic nerve crush injury [206].
Serotonergic neurotransmission has been suggested to be involved in the exercise-induced analgesia [54]. Many reports have shown that noxious stimuli-induced response is inhibited by medullary serotonergic nuclei activation [207,208]. Physical exercise improves serotonergic metabolism in many brain regions, including, brainstem, cerebral cortex, hippocampus and others [54,209,210]. Low-intensity exercise on the treadmill demonstrated increase serotonin levels and serotonin receptor mRNA expression, decrease pro-inflammatory cytokines levels in the brainstem and produced analgesia after nerve injury [54]. In addition, were showed that the reduction of serotonin in the brainstem induced by tryptophan hydroxylase inhibitor, prevented the analgesic effect induced by physical exercise training after nerve injury [54]. In this way, since BH4 is a mandatory cofactor for neurotransmitter synthesis, it is possible to suggest that exercise-induced analgesia is dependent on BH4 metabolism.
Altogether, physical exercise normalized exacerbated neuroimmune signaling in the CNS and reduced the sensation of pain, supporting the fact that exercise is also a neuroprotective non-pharmacological tool with the ability to manage central pain. Moreover, a better understanding of BH4 metabolism may contribute to the development of new approaches to treating chronic pain.
9. Summary and future directions
The positive effects of physical exercise in reducing the risk of all-cause mortality and improving the lifespan have been extensively documented. Chronic physical exercise offers protection against chronic non-communicable diseases, dementia and cognitive decline, among other diseases. Regular exercise training also protects against bacteria and viruses infections and enhance immune responses to pathogens and vaccines. This makes the physical exercise a valuable tool to prevent infectious diseases, such as COVID-19, or non-infectious ones, such as obesity. The impact of physical exercise on tissue homeostasis provokes an adaptive response which depends on the type, duration and intensity of the stimuli. The beneficial consequences of exercise on the health outcomes involve the modulation of the immune system. A pro-inflammatory phenotype is favored by physical inactivity. Conversely, the regular practice of moderate intensity physical exercise directs the immune response to an anti-inflammatory status, that is believed to be the main desired molecular mechanism to improve the health outcomes. Thus, the preventive and/or therapeutic effect of physical exercise depends of exercise workload and dose-response relationships, which could be quantified by monitoring markers of the immune response in biological fluids. We also suggest the measurement of BH4-related metabolites in biological fluids may represent a feasible way to assess the tissue immune system status and the impact of physical exercise intervention in clinical setups on chronic inflammation.
Funding
This work was supported by the CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico, Brazil) and CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brazil). Latini A is a CNPq fellow.
Declarations of competing interest
The authors declare no conflict of interest.
References
- 1.Garber C.E., Blissmer B., Deschenes M.R., Franklin B.A., Lamonte M.J., Lee I.M., Nieman D.C., Swain D.P. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011:1334–1359. doi: 10.1249/MSS.0b013e318213fefb. [DOI] [PubMed] [Google Scholar]
- 2.U.S. Department of Health and Human Services . 2008. Physical Activity Guidelines for Americans, Washington, D.C. [Google Scholar]
- 3.Nieman D.C., Wentz L.M. The compelling link between physical activity and the body’s defense system. J. Sport Health Sci. 2019:201–217. doi: 10.1016/j.jshs.2018.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Llamas-Velasco S., Villarejo-Galende A., Contador I., Lora Pablos D., Hernández-Gallego J., Bermejo-Pareja F. Physical activity and long-term mortality risk in older adults: a prospective population based study (NEDICES) Prev. Med. Rep. 2016;4:546–550. doi: 10.1016/j.pmedr.2016.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shinkai S., Konishi M., Shephard R.J. Aging and immune response to exercise. Can. J. Physiol. Pharmacol. 1998;76:562–572. doi: 10.1139/y98-043. [DOI] [PubMed] [Google Scholar]
- 6.Petersen A.M.W., Pedersen B.K. The anti-inflammatory effect of exercise. J. Appl. Physiol. 2005:1154–1162. doi: 10.1152/japplphysiol.00164.2004. [DOI] [PubMed] [Google Scholar]
- 7.Radák Z., Kaneko T., Tahara S., Nakamoto H., Ohno H., Sasvári M., Nyakas C., Goto S. The effect of exercise training on oxidative damage of lipids, proteins, and DNA in rat skeletal muscle: evidence for beneficial outcomes. Free Radic. Biol. Med. 1999:69–74. doi: 10.1016/S0891-5849(99)00038-6. [DOI] [PubMed] [Google Scholar]
- 8.Campbell J.P., Turner J.E. Debunking the myth of exercise-induced immune suppression: redefining the impact of exercise on immunological health across the lifespan. Front. Immunol. 2018;9:1–21. doi: 10.3389/fimmu.2018.00648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Okin D., Medzhitov R. Evolution of inflammatory diseases. Curr. Biol. 2012:R733–R740. doi: 10.1016/j.cub.2012.07.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gleeson M. Immune function in sport and exercise. J. Appl. Physiol. 2007:693–699. doi: 10.1152/japplphysiol.00008.2007. [DOI] [PubMed] [Google Scholar]
- 11.McFarlin B.K., Flynn M.G., Campbell W.W., Craig B.A., Robinson J.P., Stewart L.K., Timmerman K.L., Coen P.M. Physical activity status, but not age, influences inflammatory biomarkers and toll-like receptor 4. Journals Gerontol. - Ser. A Biol. Sci. Med. Sci. 2006:388–393. doi: 10.1093/gerona/61.4.388. [DOI] [PubMed] [Google Scholar]
- 12.Rosa F., Jr., Pedro Fuentes J., Pertile Remor A., Ghisoni K., Silveira P. César Lock, Costa V., Prim R. Luiz, da Luz Scheffer D., Silva Aguiar A., Jr., Guglielmo L.G. Antonacci, Latini A. A tennis-based health program for middle-aged men who are at risk for heart disease. Integr. Obes. Diabetes. 2017;3:1–6. doi: 10.15761/IOD.1000172. [DOI] [Google Scholar]
- 13.Gleeson M., Bishop N.C., Stensel D.J., Lindley M.R., Mastana S.S., Nimmo M.A. The anti-inflammatory effects of exercise: mechanisms and implications for the prevention and treatment of disease. Nat. Rev. Immunol. 2011:607–615. doi: 10.1038/nri3041. [DOI] [PubMed] [Google Scholar]
- 14.Handschin C., Spiegelman B.M. The role of exercise and PGC1alpha in inflammation and chronic disease. Nature. 2008;454:463–469. doi: 10.1038/nature07206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gielen S., Adams V., Linke A., Erbs S., Möbius-Winkler S., Schubert A., Schuler G., Hambrecht R. Exercise training in chronic heart failure: correlation between reduced local inflammation and improved oxidative capacity in the skeletal muscle. Eur. J. Prev. Cardiol. 2005;12(4):393–400. doi: 10.1097/01.hjr.0000174824.94892.43. [DOI] [PubMed] [Google Scholar]
- 16.WHO . WHO; 2018. WHO | Overweight and obesity. [Google Scholar]
- 17.WHO Coronavirus disease (COVID-19) outbreak. Emergencies - Dis. 2020;12:393–400. [Google Scholar]
- 18.Chen N., Zhou M., Dong X., Qu J., Gong F., Han Y., Qiu Y., Wang J., Liu Y., Wei Y., Xia J., Yu T., Zhang X., Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kelly P., Kahlmeier S., Götschi T., Orsini N., Richards J., Roberts N., Scarborough P., Foster C. Systematic review and meta-analysis of reduction in all-cause mortality from walking and cycling and shape of dose response relationship. Int. J. Behav. Nutr. Phys. Act. 2014;11:1–15. doi: 10.1186/s12966-014-0132-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Colberg S.R., Sigal R.J., Fernhall B., Regensteiner J.G., Blissmer B.J., Rubin R.R., Chasan-Taber L., Albright A.L., Braun B. Exercise and type 2 diabetes: the American College of Sports Medicine and the American Diabetes Association: joint position statement. Diabetes Care. 2010:2692–2696. doi: 10.2337/dc10-9990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Oruç Z., Kaplan M.A. Effect of exercise on colorectal cancer prevention and treatment. World J. Gastrointest. Oncol. 2019;11:348–366. doi: 10.4251/wjgo.v11.i5.348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Barros L., Eichwald T., Solano A.F., da Luz Scheffer D., da Silva R.A., Gaspar J.M., Latini A. Epigenetic modifications induced by exercise: drug-free intervention to improve cognitive deficits associated with obesity. Physiol. Behav. 2019:309–323. doi: 10.1016/j.physbeh.2019.03.009. [DOI] [PubMed] [Google Scholar]
- 24.Lourenco M.V., Frozza R.L., de Freitas G.B., Zhang H., Kincheski G.C., Ribeiro F.C., Gonçalves R.A., Clarke J.R., Beckman D., Staniszewski A., Berman H., Guerra L.A., Forny-Germano L., Meier S., Wilcock D.M., de Souza J.M., Alves-Leon S., Prado V.F., Prado M.A.M., Abisambra J.F., Tovar-Moll F., Mattos P., Arancio O., Ferreira S.T., De Felice F.G. Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer’s models. Nat. Med. 2019;25:165–175. doi: 10.1038/s41591-018-0275-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Long J.E., Ring C., Drayson M., Bosch J., Campbell J.P., Bhabra J., Browne D., Dawson J., Harding S., Lau J., Burns V.E. Vaccination response following aerobic exercise: can a brisk walk enhance antibody response to pneumococcal and influenza vaccinations? Brain Behav. Immun. 2012;26:680–687. doi: 10.1016/j.bbi.2012.02.004. [DOI] [PubMed] [Google Scholar]
- 26.Svensson M., Lexell J., Deierborg T. Effects of physical exercise on neuroinflammation, neuroplasticity, neurodegeneration, and behavior. Neurorehabil. Neural Repair. 2015:577–589. doi: 10.1177/1545968314562108. [DOI] [PubMed] [Google Scholar]
- 27.Caspersen C.J., Powell K.E., Christenson G.M. Physical activity, exercise and physical fitness definitions for health-related research. Public Health Rep. 1985:126–131. [PMC free article] [PubMed] [Google Scholar]
- 28.Hawley J.A., Hargreaves M., Joyner M.J., Zierath J.R. Integrative biology of exercise. Cell. 2014:738–749. doi: 10.1016/j.cell.2014.10.029. [DOI] [PubMed] [Google Scholar]
- 29.International Agency for Research on Cancer (IARC) - World Health Organization . 2002. IARC Handbooks of Cancer Prevention: Weight Control and Physical Activity. [Google Scholar]
- 30.Budde H., Schwarz R., Velasques B., Ribeiro P., Holzweg M., Machado S., Brazaitis M., Staack F., Wegner M. The need for differentiating between exercise, physical activity, and training. Autoimmun. Rev. 2016:110–111. doi: 10.1016/j.autrev.2015.09.004. [DOI] [PubMed] [Google Scholar]
- 31.Peake J.M., Neubauer O., Walsh N.P., Simpson R.J. Recovery of the immune system after exercise. J. Appl. Physiol. 2017;122:1077–1087. doi: 10.1152/japplphysiol.00622.2016. [DOI] [PubMed] [Google Scholar]
- 32.Kakanis M.W., Peake J., Brenu E.W., Simmonds M., Gray B., Hooper S.L., Marshall-Gradisnik S.M. The open window of susceptibility to infection after acute exercise in healthy young male elite athletes. Exerc. Immunol. Rev. 2010;16:119–137. doi: 10.1016/j.jsams.2010.10.642. [DOI] [PubMed] [Google Scholar]
- 33.Walsh N.P., Gleeson M., Shephard R.J., Gleeson M., Woods J.A., Bishop N.C., Fleshner M., Green C., Pedersen B.K., Hoffman-Goetz L., Rogers C.J., Northoff H., Abbasi A., Simon P. Position statement part one: immune function and exercise. Exerc. Immunol. Rev. 2011;17:6–63. [PubMed] [Google Scholar]
- 34.Saltin B., Astrand P.O. Maximal oxygen uptake in athletes. J. Appl. Physiol. 2017:353–358. doi: 10.1152/jappl.1967.23.3.353. [DOI] [PubMed] [Google Scholar]
- 35.Pedersen B.K., Saltin B. Evidence for prescribing exercise as therapy in chronic disease. Scand. J. Med. Sci. Sports. 2006:3–63. doi: 10.1111/j.1600-0838.2006.00520.x. [DOI] [PubMed] [Google Scholar]
- 36.Voss M.W., Soto C., Yoo S., Sodoma M., Vivar C., van Praag H. Exercise and hippocampal memory systems. Trends Cogn. Sci. 2019:318–333. doi: 10.1016/j.tics.2019.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Finsterer J. Biomarkers of peripheral muscle fatigue during exercise. BMC Musculoskelet. Disord. 2012:1–13. doi: 10.1186/1471-2474-13-218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pinho R. a, Silva L. a, Pinho C. a, Scheffer D.L., Souza C.T., Benetti M., Carvalho T., Dal-Pizzol F. Oxidative stress and inflammatory parameters after an Ironman race. Clin. J. Sport Med. 2010:306–311. doi: 10.1097/JSM.0b013e3181e413df. [DOI] [PubMed] [Google Scholar]
- 39.Lindsay A., Lewis J., Scarrott C., Draper N., Gieseg S.P. Changes in acute biochemical markers of inflammatory and structural stress in rugby union. J. Sports Sci. 2015:882–891. doi: 10.1080/02640414.2014.971047. [DOI] [PubMed] [Google Scholar]
- 40.Dantas de Lucas R., Caputo F., Mendes de Souza K., Sigwalt A.R., Ghisoni K., Lock Silveira P.C., Remor A.P., de Luz Scheffer, Guglielmo L.G. Antonacci, Latini A. Increased platelet oxidative metabolism, blood oxidative stress and neopterin levels after ultra-endurance exercise. J. Sports Sci. 2014;32:22–30. doi: 10.1080/02640414.2013.797098. [DOI] [PubMed] [Google Scholar]
- 41.Butovsky O., Ziv Y., Schwartz A., Landa G., Talpalar A.E., Pluchino S., Martino G., Schwartz M. Microglia activated by IL-4 or IFN-γ differentially induce neurogenesis and oligodendrogenesis from adult stem/progenitor cells. Mol. Cell. Neurosci. 2006;31:149–160. doi: 10.1016/j.mcn.2005.10.006. [DOI] [PubMed] [Google Scholar]
- 42.Medzhitov R. Inflammation 2010: new adventures of an old flame. Cell. 2010:771–776. doi: 10.1016/j.cell.2010.03.006. [DOI] [PubMed] [Google Scholar]
- 43.Malaguarnera L., Cristaldi E., Vinci M., Malaguarnera M. The role of exercise on the innate immunity of the elderly. Eur. Rev. Aging Phys. Act. 2008;5:43–49. doi: 10.1007/s11556-007-0028-8. [DOI] [Google Scholar]
- 44.Souza D.G., Vieira A.T., Soares A.C., Pinho V., Nicoli J.R., Vieira L.Q., Teixeira M.M. The essential role of the intestinal microbiota in facilitating acute inflammatory responses. J. Immunol. 2004;173:4137–4146. doi: 10.4049/jimmunol.173.6.4137. [DOI] [PubMed] [Google Scholar]
- 45.Ozinsky A., Underhill D.M., Fontenot J.D., Hajjar A.M., Smith K.D., Wilson C.B., Schroeder L., Aderem A. The repertoire for pattern recognition of pathogens by the innate immune system is defined by cooperation between toll-like receptors. Proc. Natl. Acad. Sci. U. S. A. 2000;97:13766–13771. doi: 10.1073/pnas.250476497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Perregaux D., Gabel C.A. Interleukin-1β maturation and release in response to ATP and nigericin. Evidence that potassium depletion mediated by these agents is a necessary and common feature of their activity. J. Biol. Chem. 1994;269:15195–15203. [PubMed] [Google Scholar]
- 47.Nieman D.C., Nehlsen-Cannarella S.L., Markoff P.A., Balk-Lamberton A.J., Yang H., Chritton D.B.W., Lee J.W., Arabatzis K. The effects of moderate exercise training on natural killer cells and acute upper respiratory tract infections. Int. J. Sports Med. 1990;11:467–473. doi: 10.1055/s-2007-1024839. [DOI] [PubMed] [Google Scholar]
- 48.Evans E.S., Hackney A.C., McMurray R.G., Randell S.H., Muss H.B., Deal A.M., Battaglini C.L. Impact of acute intermittent exercise on natural killer cells in breast cancer survivors. Integr. Cancer Ther. 2015;14:436–445. doi: 10.1177/1534735415580681. [DOI] [PubMed] [Google Scholar]
- 49.Grindvik H. Exercise and immunity. Curr. Issues Sport. Exerc. Med. 2013:121–140. doi: 10.5772/54681. [DOI] [Google Scholar]
- 50.Kohman R.A., Bhattacharya T.K., Wojcik E., Rhodes J.S. Exercise reduces activation of microglia isolated from hippocampus and brain of aged mice. J. Neuroinflammation. 2013:1–9. doi: 10.1186/1742-2094-10-114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Vukovic J., Colditz M.J., Blackmore D.G., Ruitenberg M.J., Bartlett P.F. Microglia modulate hippocampal neural precursor activity in response to exercise and aging. J. Neurosci. 2012:6435–6443. doi: 10.1523/JNEUROSCI.5925-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Parachikova A., Nichol K.E., Cotman C.W. Short-term exercise in aged Tg2576 mice alters neuroinflammation and improves cognition. Neurobiol. Dis. 2008:121–129. doi: 10.1016/j.nbd.2007.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Gomes da Silva S., Simões P.S.R., Mortara R.A., Scorza F.A., Cavalheiro E.A., da Graça Naffah-Mazzacoratti M., Arida R.M. Exercise-induced hippocampal anti-inflammatory response in aged rats. J. Neuroinflammation. 2013:1–6. doi: 10.1186/1742-2094-10-61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bobinski F., Ferreira T.A.A., Córdova M.M., Dombrowski P.A., Da Cunha C., Santo C.C.D.E., Poli A., Pires R.G.W., Martins-Silva C., Sluka K.A., Santos A.R.S. Role of brainstem serotonin in analgesia produced by low-intensity exercise on neuropathic pain after sciatic nerve injury in mice. Pain. 2015;156:2595–2606. doi: 10.1097/j.pain.0000000000000372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Steensberg A., Fischer C.P., Keller C., Møller K., Pedersen B.K. IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am. J. Physiol. Metab. 2003;285:E433–E437. doi: 10.1152/ajpendo.00074.2003. [DOI] [PubMed] [Google Scholar]
- 56.Di Benedetto S., Müller L., Wenger E., Düzel S., Pawelec G. Contribution of neuroinflammation and immunity to brain aging and the mitigating effects of physical and cognitive interventions. Neurosci. Biobehav. Rev. 2017:114–128. doi: 10.1016/j.neubiorev.2017.01.044. [DOI] [PubMed] [Google Scholar]
- 57.Dallagnol K.M.C., Remor A.P., da Silva R.A., Prediger R.D., Latini A., Aguiar A.S. Running for REST: physical activity attenuates neuroinflammation in the hippocampus of aged mice. Brain Behav. Immun. 2017:31–35. doi: 10.1016/j.bbi.2016.07.159. [DOI] [PubMed] [Google Scholar]
- 58.Van Dyken P., Lacoste B. Impact of metabolic syndrome on neuroinflammation and the blood–brain barrier. Front. Neurosci. 2018:1–19. doi: 10.3389/fnins.2018.00930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zlokovic B.V. The blood-brain barrier in health and chronic neurodegenerative disorders. Neuron. 2008:178–201. doi: 10.1016/j.neuron.2008.01.003. [DOI] [PubMed] [Google Scholar]
- 60.Daneman R., Prat A. The blood brain barrier (BBB) Cold Spring Harb. Perspect. Biol. 2014:1–23. doi: 10.1007/978-3-662-43787-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Małkiewicz M.A., Szarmach A., Sabisz A., Cubała W.J., Szurowska E., Winklewski P.J. Blood-brain barrier permeability and physical exercise. J. Neuroinflammation. 2019:1–15. doi: 10.1186/s12974-019-1403-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Klein R.S., Garber C., Howard N. Infectious immunity in the central nervous system and brain function. Nat. Immunol. 2017:132–141. doi: 10.1038/ni.3656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Leiter O., Kempermann G., Walker T.L. A common language: how neuroimmunological cross talk regulates adult hippocampal neurogenesis. Stem Cells Int. 2016:1–13. doi: 10.1155/2016/1681590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Fischer C.P., Berntsen A., Perstrup L.B., Eskildsen P., Pedersen B.K. Plasma levels of interleukin-6 and C-reactive protein are associated with physical inactivity independent of obesity. Scand. J. Med. Sci. Sports. 2007:580–587. doi: 10.1111/j.1600-0838.2006.00602.x. [DOI] [PubMed] [Google Scholar]
- 65.Mathur N., Pedersen B.K. Exercise as a mean to control low-grade systemic inflammation. Mediat. Inflamm. 2008:1–6. doi: 10.1155/2008/109502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Febbraio M.A., Pedersen B.K. Muscle-derived interleukin-6: mechanisms for activation and possible biological roles. FASEB J. 2002:1335–1347. doi: 10.1096/fj.01-0876rev. [DOI] [PubMed] [Google Scholar]
- 67.Keller C., Steensberg A., Hansen A.K., Fischer C.P., Plomgaard P., Pedersen B.K. Effect of exercise, training, and glycogen availability on IL-6 receptor expression in human skeletal muscle. J. Appl. Physiol. 2005:2075–2079. doi: 10.1152/japplphysiol.00590.2005. [DOI] [PubMed] [Google Scholar]
- 68.Fischer C.P. Interleukin-6 in acute exercise and training: what is the biological relevance? Exerc. Immunol. Rev. 2006:6–33. [PubMed] [Google Scholar]
- 69.Pedersen B.K., Febbraio M.A. Muscle as an endocrine organ: focus on muscle-derived interleukin-6. Physiol. Rev. 2008:1379–1406. doi: 10.1152/physrev.90100.2007. [DOI] [PubMed] [Google Scholar]
- 70.Pedersen B.K., Hoffman-Goetz L. Exercise and the immune system: regulation, integration, and adaptation. Physiol. Rev. 2000:1055–1081. doi: 10.1152/physrev.2000.80.3.1055. [DOI] [PubMed] [Google Scholar]
- 71.Eyre H., Baune B.T. Neuroimmunological effects of physical exercise in depression. Brain Behav. Immun. 2012:251–266. doi: 10.1016/j.bbi.2011.09.015. [DOI] [PubMed] [Google Scholar]
- 72.Stewart L.K., Flynn M.G., Campbell W.W., Craig B.A., Robinson J.P., McFarlin B.K., Timmerman K.L., Coen P.M., Felker J., Talbert E. Influence of exercise training and age on CD14+ cell-surface expression of toll-like receptor 2 and 4. Brain Behav. Immun. 2005;19:389–397. doi: 10.1016/j.bbi.2005.04.003. [DOI] [PubMed] [Google Scholar]
- 73.Donges C.E., Duffield R., Drinkwater E.J. Effects of resistance or aerobic exercise training on interleukin-6, C-reactive protein, and body composition. Med. Sci. Sports Exerc. 2010;42:304–313. doi: 10.1249/MSS.0b013e3181b117ca. [DOI] [PubMed] [Google Scholar]
- 74.Kadoglou N.P.E., Iliadis F., Angelopoulou N., Perrea D., Ampatzidis G., Liapis C.D., Alevizos M. The anti-inflammatory effects of exercise training in patients with type 2 diabetes mellitus. Eur. J. Cardiovasc. Prev. Rehabil. 2007;14:837–843. doi: 10.1097/HJR.0b013e3282efaf50. [DOI] [PubMed] [Google Scholar]
- 75.Chatelain R., Wollenberg A., Martin C., Panhans-Gross A., Bieber T., Degitz K., Heckmann M. IL-10 inhibits ICAM-1 expression on human Langerhans cells but not on keratinocytes, dermal endothelial cells or fibroblasts. Arch. Dermatol. Res. 1998;290:477–482. doi: 10.1007/s004030050339. [DOI] [PubMed] [Google Scholar]
- 76.Dalsgaard M.K., Ott P., Dela F., Juul A., Pedersen B.K., Warberg J., Fahrenkrug J., Secher N.H. The CSF and arterial to internal jugular venous hormonal differences during exercise in humans. Exp. Physiol. 2004:271–277. doi: 10.1113/expphysiol.2003.026922. [DOI] [PubMed] [Google Scholar]
- 77.Nybo L., Nielsen B., Pedersen B.K., Møller K., Secher N.H. Interleukin-6 release from the human brain during prolonged exercise. J. Physiol. 2002:991–995. doi: 10.1113/jphysiol.2002.022285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Chen M.F., Chen H.I., Jen C.J. Exercise training upregulates macrophage MKP-1 and affects immune responses in mice. Med. Sci. Sports Exerc. 2010;42:2173–2179. doi: 10.1249/MSS.0b013e3181e2158d. [DOI] [PubMed] [Google Scholar]
- 79.Wedell-Neergaard A.S., Lang Lehrskov L., Christensen R.H., Legaard G.E., Dorph E., Larsen M.K., Launbo N., Fagerlind S.R., Seide S.K., Nymand S., Ball M., Vinum N., Dahl C.N., Henneberg M., Ried-Larsen M., Nybing J.D., Christensen R., Rosenmeier J.B., Karstoft K., Pedersen B.K., Ellingsgaard H., Krogh-Madsen R. Exercise-induced changes in visceral adipose tissue mass are regulated by IL-6 signaling: a randomized controlled trial. Cell Metab. 2019:844–855. doi: 10.1016/j.cmet.2018.12.007. [DOI] [PubMed] [Google Scholar]
- 80.Pedersen B.K. Anti-inflammatory effects of exercise: role in diabetes and cardiovascular disease. Eur. J. Clin. Investig. 2017:600–611. doi: 10.1111/eci.12781. [DOI] [PubMed] [Google Scholar]
- 81.Bergmann M., Gornikiewicz A., Sautner T., Waldmann E., Weber T., Mittlböck M., Roth E., Függer R. Attenuation of catecholamine-induced immunosuppression in whole blood from patients with sepsis. Shock. 1999;12:421–427. doi: 10.1097/00024382-199912000-00002. [DOI] [PubMed] [Google Scholar]
- 82.Yudkin J.S. Inflammation, obesity, and the metabolic syndrome. Horm. Metab. Res. 2007:707–709. doi: 10.1055/s-2007-985898. [DOI] [PubMed] [Google Scholar]
- 83.Lang Lehrskov L., Lyngbaek M.P., Soederlund L., Legaard G.E., Ehses J.A., Heywood S.E., Wewer Albrechtsen N.J., Holst J.J., Karstoft K., Pedersen B.K., Ellingsgaard H. Interleukin-6 delays gastric emptying in humans with direct effects on glycemic control. Cell Metab. 2018;27:1201–1211.e3. doi: 10.1016/j.cmet.2018.04.008. [DOI] [PubMed] [Google Scholar]
- 84.Carey A.L., Steinberg G.R., Macaulay S.L., Thomas W.G., Holmes A.G., Ramm G., Prelovsek O., Hohnen-Behrens C., Watt M.J., James D.E., Kemp B.E., Pedersen B.K., Febbraio M.A. Interleukin-6 increases insulin-stimulated glucose disposal in humans and glucose uptake and fatty acid oxidation in vitro via AMP-activated protein kinase. Diabetes. 2006:2688–2697. doi: 10.2337/db05-1404. [DOI] [PubMed] [Google Scholar]
- 85.Van Hall G., Steensberg A., Sacchetti M., Fischer C., Keller C., Schjerling P., Hiscock N., Møller K., Saltin B., Febbraio M.A., Pedersen B.K. Interleukin-6 stimulates lipolysis and fat oxidation in humans. J. Clin. Endocrinol. Metab. 2003:3005–3010. doi: 10.1210/jc.2002-021687. [DOI] [PubMed] [Google Scholar]
- 86.Nielsen A.R., Hojman P., Erikstrup C., Fischer C.P., Plomgaard P., Mounier R., Mortensen O.H., Broholm C., Taudorf S., Krogh-Madsen R., Lindegaard B., Petersen A.M.W., Gehl J., Pedersen B.K. Association between interleukin-15 and obesity: interleukin-15 as a potential regulator of fat mass. J. Clin. Endocrinol. Metab. 2008:4486–4493. doi: 10.1210/jc.2007-2561. [DOI] [PubMed] [Google Scholar]
- 87.Rinnov A., Yfanti C., Nielsen S., Åkerström T.C.A., Peijs L., Zankari A., Fischer C.P., Pedersen B.K. Endurance training enhances skeletal muscle interleukin-15 in human male subjects. Endocrine. 2014:271–278. doi: 10.1007/s12020-013-9969-z. [DOI] [PubMed] [Google Scholar]
- 88.Grabstein K.H., Eisenman J., Shanebeck K., Rauch C., Srinivasan S., Fung V., Beers C., Richardson J., Schoenborn M.A., Ahdieh M., Johnson L., Alderson M.R., Watson J.D., Anderson D.M., Giri J.G. Cloning of a T cell growth factor that interacts with the β chain of the interleukin-2 receptor. Science (80-. ) 1994:965–968. doi: 10.1126/science.8178155. [DOI] [PubMed] [Google Scholar]
- 89.Boström P., Wu J., Jedrychowski M.P., Korde A., Ye L., Lo J.C., Rasbach K.A., Boström E.A., Choi J.H., Long J.Z., Kajimura S., Zingaretti M.C., Vind B.F., Tu H., Cinti S., Højlund K., Gygi S.P., Spiegelman B.M. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012:463–468. doi: 10.1038/nature10777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Jedrychowski M.P., Wrann C.D., Paulo J.A., Gerber K.K., Szpyt J., Robinson M.M., Nair K.S., Gygi S.P., Spiegelman B.M. Detection and quantitation of circulating human irisin by tandem mass spectrometry. Cell Metab. 2015:734–740. doi: 10.1016/j.cmet.2015.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.de Oliveira Bristot V.J., de Bem Alves A.C., Cardoso L.R., da Luz Scheffer D., Aguiar A.S. The role of PGC-1α/UCP2 signaling in the beneficial effects of physical exercise on the brain. Front. Neurosci. 2019:1–9. doi: 10.3389/fnins.2019.00292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Colaianni G., Notarnicola A., Sanesi L., Brunetti G., Lippo L., Celi M., Moretti L., Pesce V., Vicenti G., Moretti B., Colucci S., Grano M. Irisin levels correlate with bone mineral density in soccer players. J. Biol. Regul. Homeost. Agents. 2017;31:21–28. [PubMed] [Google Scholar]
- 93.Colaianni G., Cuscito C., Mongelli T., Pignataro P., Buccoliero C., Liu P., Lu P., Sartini L., Comite M.D., Mori G., Benedetto A.D., Brunetti G., Yuen T., Sun L., Reseland J.E., Colucci S., New M.I., Zaidi M., Cinti S., Grano M. The myokine irisin increases cortical bone mass. Proc. Natl. Acad. Sci. U. S. A. 2015;112:12157–12162. doi: 10.1073/pnas.1516622112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Morettini M., Palumbo M.C., Sacchetti M., Castiglione F., Mazzà C. A system model of the effects of exercise on plasma interleukin-6 dynamics in healthy individuals: role of skeletal muscle and adipose tissue. PLoS One. 2017;12:1–12. doi: 10.1371/journal.pone.0181224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ostrowski K., Rohde T., Zacho M., Asp S., Pedersen B.K. Evidence that interleukin-6 is produced in human skeletal muscle during prolonged running. J. Physiol. 1998:949–953. doi: 10.1111/j.1469-7793.1998.949bp.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Andersson H., Bøhn S.K., Raastad T., Paulsen G., Blomhoff R., Kadi F. Differences in the inflammatory plasma cytokine response following two elite female soccer games separated by a 72-h recovery. Scand. J. Med. Sci. Sports. 2010:740–747. doi: 10.1111/j.1600-0838.2009.00989.x. [DOI] [PubMed] [Google Scholar]
- 97.Haugen F., Norheim F., Lian H., Wensaas A.J., Dueland S., Berg O., Funderud A., Skålhegg B.S., Raastad T., Drevon C.A. IL-7 is expressed and secreted by human skeletal muscle cells. Am. J. Physiol. Physiol. 2010:C807–C816. doi: 10.1152/ajpcell.00094.2009. [DOI] [PubMed] [Google Scholar]
- 98.Chan M.H.S., Carey A.L., Watt M.J., Febbraio M.A. Cytokine gene expression in human skeletal muscle during concentric contraction: evidence that IL-8, like IL-6, is influenced by glycogen availability. Am. J. Physiol. Integr. Comp. Physiol. 2004:R322–R327. doi: 10.1152/ajpregu.00030.2004. [DOI] [PubMed] [Google Scholar]
- 99.Nieman D.C., Davis J.M., Henson D.A., Walberg-Rankin J., Shute M., Dumke C.L., Utter A.C., Vinci D.M., Carson J.A., Brown A., Lee W.J., McAnulty S.R., McAnulty L.S. Carbohydrate ingestion influences skeletal muscle cytokine mRNA and plasma cytokine levels after a 3-h run. J. Appl. Physiol. 2003:1917–1925. doi: 10.1152/japplphysiol.01130.2002. [DOI] [PubMed] [Google Scholar]
- 100.Rocha-Rodrigues S., Rodríguez A., Gonçalves I.O., Moreira A., Maciel E., Santos S., Domingues M.R., Frühbeck G., Ascensão A., Magalhães J. Impact of physical exercise on visceral adipose tissue fatty acid profile and inflammation in response to a high-fat diet regimen. Int. J. Biochem. Cell Biol. 2017;87:114–124. doi: 10.1016/j.biocel.2017.04.008. [DOI] [PubMed] [Google Scholar]
- 101.Tamura Y., Watanabe K., Kantani T., Hayashi J., Ishida N., Kaneki M. Upregulation of circulating IL-15 by treadmill running in healthy individuals: is IL-15 an endocrine mediator of the beneficial effects of endurance exercise? Endocr. J. 2011:211–215. doi: 10.1507/endocrj.k10e-400. [DOI] [PubMed] [Google Scholar]
- 102.Nielsen A.R., Mounier R., Plomgaard P., Mortensen O.H., Penkowa M., Speerschneider T., Pilegaard H., Pedersen B.K. Expression of interleukin-15 in human skeletal muscle - effect of exercise and muscle fibre type composition. J. Physiol. 2007:305–312. doi: 10.1113/jphysiol.2007.139618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ferris L.T., Williams J.S., Shen C.L. The effect of acute exercise on serum brain-derived neurotrophic factor levels and cognitive function. Med. Sci. Sports Exerc. 2007:728–734. doi: 10.1249/mss.0b013e31802f04c7. [DOI] [PubMed] [Google Scholar]
- 104.Broholm C., Mortensen O.H., Nielsen S., Akerstrom T., Zankari A., Dahl B., Pedersen B.K. Exercise induces expression of leukaemia inhibitory factor in human skeletal muscle. J. Physiol. 2008:2195–2201. doi: 10.1113/jphysiol.2007.149781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Cappon J., Brasel J.A., Mohan S., Cooper D.M. Effect of brief exercise on circulating insulin-like growth factor I. J. Appl. Physiol. 2017:2490–2496. doi: 10.1152/jappl.1994.76.6.2490. [DOI] [PubMed] [Google Scholar]
- 106.Hameed M., Orrell R.W., Cubbold M., Goldspink G., Harridge S.D.R. Expression of IGF-I splice variants in young and old human skeletal muscle after high resistance exercise. J. Physiol. 2003:247–254. doi: 10.1113/jphysiol.2002.032136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Wahl P., Zinner C., Achtzehn S., Behringer M., Bloch W., Mester J. Effects of acid-base balance and high or low intensity exercise on VEGF and bFGF. Eur. J. Appl. Physiol. 2011:1405–1413. doi: 10.1007/s00421-010-1767-1. [DOI] [PubMed] [Google Scholar]
- 108.Yang J., Shanahan K.J., Shriver L.P., Luciano M.G. Exercise-induced changes of cerebrospinal fluid vascular endothelial growth factor in adult chronic hydrocephalus patients. J. Clin. Neurosci. 2016:52–56. doi: 10.1016/j.jocn.2015.08.019. [DOI] [PubMed] [Google Scholar]
- 109.Gavin T.P., Robinson C.B., Yeager R.C., England J.A., Nifong L.W., Hickner R.C. Angiogenic growth factor response to acute systemic exercise in human skeletal muscle. J. Appl. Physiol. 2004:19–24. doi: 10.1152/japplphysiol.00748.2003. [DOI] [PubMed] [Google Scholar]
- 110.Larson E.B., Wang L., Bowen J.D., McCormick W.C., Teri L., Crane P., Kukull W. Exercise is associated with reduced risk for incident dementia among persons 65 years of age and older. Ann. Intern. Med. 2006:73–81. doi: 10.7326/0003-4819-144-2-200601170-00004. [DOI] [PubMed] [Google Scholar]
- 111.Carro E., Trejo J.L., Busiguina S., Torres-Aleman I. Circulating insulin-like growth factor I mediates the protective effects of physical exercise against brain insults of different etiology and anatomy. J. Neurosci. 2001:5678–5684. doi: 10.1523/jneurosci.21-15-05678.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Aguiar A.S., Stragier E., da Luz Scheffer D., Remor A.P., Oliveira P.A., Prediger R.D., Latini A., Raisman-Vozari R., Mongeau R., Lanfumey L. Effects of exercise on mitochondrial function, neuroplasticity and anxio-depressive behavior of mice. Neuroscience. 2014;271:56–63. doi: 10.1016/j.neuroscience.2014.04.027. [DOI] [PubMed] [Google Scholar]
- 113.van Praag H., Kempermann G., Gage F.H. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat. Neurosci. 1999;2:266–270. doi: 10.1038/6368. [DOI] [PubMed] [Google Scholar]
- 114.Aguiar A.S., Duzzioni M., Remor A.P., Tristão F.S.M., Matheus F.C., Raisman-Vozari R., Latini A., Prediger R.D. Moderate-intensity physical exercise protects against experimental 6-hydroxydopamine-induced hemiparkinsonism through Nrf2-antioxidant response element pathway. Neurochem. Res. 2016;41:64–72. doi: 10.1007/s11064-015-1709-8. [DOI] [PubMed] [Google Scholar]
- 115.Gomez-Pinilla F., Zhuang Y., Feng J., Ying Z., Fan G. Exercise impacts brain-derived neurotrophic factor plasticity by engaging mechanisms of epigenetic regulation. Eur. J. Neurosci. 2011;33:383–390. doi: 10.1111/j.1460-9568.2010.07508.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.van Praag H. Exercise enhances learning and hippocampal neurogenesis in aged mice. J. Neurosci. 2005:8680–8685. doi: 10.1523/jneurosci.1731-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Neeper S.A., Gómez-Pinilla F., Choi J., Cotman C.W. Physical activity increases mRNA for brain-derived neurotrophic factor and nerve growth factor in rat brain. Brain Res. 1996:49–56. doi: 10.1016/0006-8993(96)00273-9. [DOI] [PubMed] [Google Scholar]
- 118.Gómez-Pinilla F., Dao L., So V. Physical exercise induces FGF-2 and its mRNA in the hippocampus. Brain Res. 1997:1–8. doi: 10.1016/S0006-8993(97)00375-2. [DOI] [PubMed] [Google Scholar]
- 119.Kohman R.A., DeYoung E.K., Bhattacharya T.K., Peterson L.N., Rhodes J.S. Wheel running attenuates microglia proliferation and increases expression of a proneurogenic phenotype in the hippocampus of aged mice. Brain Behav. Immun. 2012:803–810. doi: 10.1016/j.bbi.2011.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Lu T., Aron L., Zullo J., Pan Y., Kim H., Chen Y., Yang T.-H., Kim H.-M., Drake D., Liu X.S., Bennett D.A., Colaiácovo M.P., Yankner B.A. REST and stress resistance in ageing and Alzheimer’s disease. Nature. 2014;507:448–454. doi: 10.1038/nature13163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Pedersen B.K. Physical activity and muscle–brain crosstalk. Nat. Rev. Endocrinol. 2019:383–392. doi: 10.1038/s41574-019-0174-x. [DOI] [PubMed] [Google Scholar]
- 122.Ekdahl C.T., Claasen J.-H., Bonde S., Kokaia Z., Lindvall O. Inflammation is detrimental for neurogenesis in adult brain. Proc. Natl. Acad. Sci. 2003:13632–13637. doi: 10.1073/pnas.2234031100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Arai K., Matsuki N., Ikegaya Y., Nishiyama N. Deterioration of spatial learning performances in lipopolysaccharide-treated mice. Jpn. J. Pharmacol. 2001:195–201. doi: 10.1254/jjp.87.195. [DOI] [PubMed] [Google Scholar]
- 124.Whitton P.S. Inflammation as a causative factor in the aetiology of Parkinson’s disease. Br. J. Pharmacol. 2007:963–976. doi: 10.1038/sj.bjp.0707167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Zipp F., Aktas O. The brain as a target of inflammation: common pathways link inflammatory and neurodegenerative diseases. Trends Neurosci. 2006:518–527. doi: 10.1016/j.tins.2006.07.006. [DOI] [PubMed] [Google Scholar]
- 126.Dinan T.G., Dinan T. Inflammatory markers in depression. Curr. Opin. Psychiatry. 2009:32–36. doi: 10.1097/YCO.0b013e328315a561. [DOI] [PubMed] [Google Scholar]
- 127.Perry V.H., Teeling J. Microglia and macrophages of the central nervous system: the contribution of microglia priming and systemic inflammation to chronic neurodegeneration. Semin. Immunopathol. 2013:601–612. doi: 10.1007/s00281-013-0382-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Yi C.X., Al-Massadi O., Donelan E., Lehti M., Weber J., Ress C., Trivedi C., Müller T.D., Woods S.C., Hofmann S.M. Exercise protects against high-fat diet-induced hypothalamic inflammation. Physiol. Behav. 2012:485–490. doi: 10.1016/j.physbeh.2012.03.021. [DOI] [PubMed] [Google Scholar]
- 129.Sung Y.H., Kim S.C., Hong H.P., Park C.Y., Shin M.S., Kim C.J., Seo J.H., Kim D.Y., Kim D.J., Cho H.J. Treadmill exercise ameliorates dopaminergic neuronal loss through suppressing microglial activation in Parkinson’s disease mice. Life Sci. 2012:1309–1316. doi: 10.1016/j.lfs.2012.10.003. [DOI] [PubMed] [Google Scholar]
- 130.Wu S.Y., Wang T.F., Yu L., Jen C.J., Chuang J.I., Sen Wu F., Wu C.W., Kuo Y.M. Running exercise protects the substantia nigra dopaminergic neurons against inflammation-induced degeneration via the activation of BDNF signaling pathway. Brain Behav. Immun. 2011:135–146. doi: 10.1016/j.bbi.2010.09.006. [DOI] [PubMed] [Google Scholar]
- 131.Aguiar A.S., Lopes S.C., Tristão F.S.M., Rial D., de Oliveira G., da Cunha C., Raisman-Vozari R., Prediger R.D. Exercise improves cognitive impairment and dopamine metabolism in MPTP-treated mice. Neurotox. Res. 2016;29:118–125. doi: 10.1007/s12640-015-9566-4. [DOI] [PubMed] [Google Scholar]
- 132.Tuon T., Souza P.S., Santos M.F., Pereira F.T., Pedroso G.S., Luciano T.F., De Souza C.T., Dutra R.C., Silveira P.C.L., Pinho R.A. Physical training regulates mitochondrial parameters and neuroinflammatory mechanisms in an experimental model of Parkinson’s disease. Oxidative Med. Cell. Longev. 2015;261809:1–10. doi: 10.1155/2015/261809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Owens M.J., Nemeroff C.B. Role of serotonin in the pathophysiology of depression: focus on the serotonin transporter. Clin. Chem. 1994:288–295. [PubMed] [Google Scholar]
- 134.Karege F., Perret G., Bondolfi G., Schwald M., Bertschy G., Aubry J.M. Decreased serum brain-derived neurotrophic factor levels in major depressed patients. Psychiatry Res. 2002:143–148. doi: 10.1016/S0165-1781(02)00005-7. [DOI] [PubMed] [Google Scholar]
- 135.de Paula Martins R., Lim C.K., Ghisoni K., Staats A., Dallagnol K., Solano A., Guillemin G.J., Silva Aguiar A., Jr., Latini A. Treating depression with exercise: the inflammasome inhibition perspective. J. Syst. Integr. Neurosci. 2016:158–161. doi: 10.15761/JSIN.1000147. [DOI] [Google Scholar]
- 136.Merendino R.A., Di Rosa A.E., Di Pasquale G., Minciullo P.L., Mangraviti C., Costantino A., Ruello A., Gangemi S. Interleukin-18 and CD30 serum levels in patients with moderate-severe depression. Mediat. Inflamm. 2002:265–267. doi: 10.1080/096293502900000131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Hallgren M., Helgadóttir B., Herring M.P., Zeebari Z., Lindefors N., Kaldo V., Öjehagen A., Forsell Y. Exercise and internet-based cognitive-behavioural therapy for depression: multicentre randomized controlled trial with 12-month follow-up. Br. J. Psychiatry. 2016:414–420. doi: 10.1192/bjp.bp.115.177576. [DOI] [PubMed] [Google Scholar]
- 138.Sigwalt A.R., Budde H., Helmich I., Glaser V., Ghisoni K., Lanza S., Cadore E.L., Lhullier F.L., de Bem A.F., Hohl A., de Matos F.J., de Oliveira P.A., Prediger R.D., Guglielmo L.G., Latini A. Molecular aspects involved in swimming exercise training reducing anhedonia in a rat model of depression. Neuroscience. 2011;192:661–674. doi: 10.1016/j.neuroscience.2011.05.075. [DOI] [PubMed] [Google Scholar]
- 139.Pietrelli A., Matković L., Vacotto M., Lopez-Costa J.J., Basso N., Brusco A. Aerobic exercise upregulates the BDNF-serotonin systems and improves the cognitive function in rats. Neurobiol. Learn. Mem. 2018;155:528–542. doi: 10.1016/j.nlm.2018.05.007. [DOI] [PubMed] [Google Scholar]
- 140.Rasmussen P., Brassard P., Adser H., Pedersen M.V., Leick L., Hart E., Secher N.H., Pedersen B.K., Pilegaard H. Evidence for a release of brain-derived neurotrophic factor from the brain during exercise. Exp. Physiol. 2009:1062–1069. doi: 10.1113/expphysiol.2009.048512. [DOI] [PubMed] [Google Scholar]
- 141.Seifert T., Brassard P., Wissenberg M., Rasmussen P., Nordby P., Stallknecht B., Adser H., Jakobsen A.H., Pilegaard H., Nielsen H.B., Secher N.H. Endurance training enhances BDNF release from the human brain. Am. J. Physiol. Integr. Comp. Physiol. 2010:R372–R377. doi: 10.1152/ajpregu.00525.2009. [DOI] [PubMed] [Google Scholar]
- 142.Remor A.P., da Silva R.A., de Matos F.J., Glaser V., de Paula Martins R., Ghisoni K., da Luz Scheffer D., Andia D.C., Portinho D., de Souza A.P., de Oliveira P.A., Prediger R.D., Torres A.I., Linhares R.M.M., Walz R., Ronsoni M.F., Hohl A., Rafacho A., Aguiar A.S., de Paul A.L., Latini A. Chronic metabolic derangement-induced cognitive deficits and neurotoxicity are associated with REST inactivation. Mol. Neurobiol. 2018:1539–1557. doi: 10.1007/s12035-018-1175-9. [DOI] [PubMed] [Google Scholar]
- 143.D da Scheffer L., Ghisoni K., Aguiar A.S., Latini A. Moderate running exercise prevents excessive immune system activation. Physiol. Behav. 2019:248–255. doi: 10.1016/j.physbeh.2019.02.023. [DOI] [PubMed] [Google Scholar]
- 144.Strasser B., Geiger D., Schauer M., Gatterer H., Burtscher M., Fuchs D. Effects of exhaustive aerobic exercise on tryptophan-kynurenine metabolism in trained athletes. PLoS One. 2016:1–10. doi: 10.1371/journal.pone.0153617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Valdiglesias V., Sánchez-Flores M., Maseda A., Lorenzo-López L., Marcos-Pérez D., López-Cortón A., Strasser B., Fuchs D., Laffon B., Millán-Calenti J.C., Pásaro E. Immune biomarkers in older adults: role of physical activity. J. Toxicol. Environ. Heal. - Part A Curr. Issues. 2017:605–620. doi: 10.1080/15287394.2017.1286898. [DOI] [PubMed] [Google Scholar]
- 146.Lindsay A., Baxter-Parker G., Gieseg S.P. Pterins as diagnostic markers of mechanical and impact-induced trauma: a systematic review. J. Clin. Med. 2019:53–62. doi: 10.3390/jcm8091383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Lin F., Friedman E., Quinn J., Chen D.G.D., Mapstone M. Effect of leisure activities on inflammation and cognitive function in an aging sample. Arch. Gerontol. Geriatr. 2012:e398–e404. doi: 10.1016/j.archger.2012.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Ghisoni K., Aguiar A.S., de Oliveira P.A., Matheus F.C., Gabach L., Perez M., Carlini V.P., Barbeito L., Mongeau R., Lanfumey L., Prediger R.D., Latini A. Neopterin acts as an endogenous cognitive enhancer. Brain Behav. Immun. 2016:156–164. doi: 10.1016/j.bbi.2016.02.019. [DOI] [PubMed] [Google Scholar]
- 149.Ghisoni K., de P. Martins R., Barbeito L., Latini A. Neopterin as a potential cytoprotective brain molecule. J. Psychiatr. Res. 2015;71:134–139. doi: 10.1016/j.jpsychires.2015.10.003. [DOI] [PubMed] [Google Scholar]
- 150.Ghisoni K., Latini A., Kuehne L.K., Reiber H., Bechter K., Hagberg L., Fuchs D. Cerebrospinal fluid neopterin is brain-derived and not associated with blood-CSF barrier dysfunction in non-inflammatory affective and schizophrenic spectrum disorders. J. Psychiatr. Res. 2015;63:141–142. doi: 10.1016/j.jpsychires.2015.02.002. [DOI] [PubMed] [Google Scholar]
- 151.de P. Martins R., Ghisoni K., Lim C.K.C.K., Jr A.S.A., Guillemin G.J.G.J., Latini A., de Paula Martins R., Ghisoni K., Lim C.K.C.K., Aguiar A.S., Guillemin G.J.G.J., Latini A. Neopterin preconditioning prevents inflammasome activation in mammalian astrocytes. Free Radic. Biol. Med. 2018:371–382. doi: 10.1016/j.freeradbiomed.2017.11.022. [DOI] [PubMed] [Google Scholar]
- 152.Wissmann P., Geisler S., Leblhuber F., Fuchs D. Immune activation in patients with Alzheimer’s disease is associated with high serum phenylalanine concentrations. J. Neurol. Sci. 2013:29–33. doi: 10.1016/j.jns.2013.03.007. [DOI] [PubMed] [Google Scholar]
- 153.Pourakbari B., Mamishi S., Zafari J., Khairkhah H., Ashtiani M.H., Abedini M., Afsharpaiman S., Rad S.S. Evaluation of procalcitonin and neopterin level in serum of patients with acute bacterial infection. Braz. J. Infect. Dis. 2010:252–255. [PubMed] [Google Scholar]
- 154.Mrakic-Sposta S., Gussoni M., Moretti S., Pratali L., Giardini G., Tacchini P., Dellanoce C., Tonacci A., Mastorci F., Borghini A., Montorsi M., Vezzoli A. Effects of mountain ultra-marathon running on ROS production and oxidative damage by micro-invasive analytic techniques. PLoS One. 2015:1–19. doi: 10.1371/journal.pone.0141780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Lindsay A., Petersen C., Gieseg S.P., Lewis J.G., Carr S., Cross S. The physiological response to cold-water immersion following a mixed martial arts training session. Appl. Physiol. Nutr. Metab. 2017:529–536. doi: 10.1139/apnm-2016-0582. [DOI] [PubMed] [Google Scholar]
- 156.Lindsay A., Janmale T., Draper N., Gieseg S.P. Measurement of changes in urinary neopterin and total neopterin in body builders using SCX HPLC. Pteridines. 2014:53–62. doi: 10.1515/pteridines-2014-0003. [DOI] [Google Scholar]
- 157.Jakeman P.M., Weller A., Warrington G. Cellular immune activity in response to increased training of elite oarsmen prior to olympic competition. J. Sports Sci. 1995:207–211. doi: 10.1080/02640419508732229. [DOI] [PubMed] [Google Scholar]
- 158.Moser B., Schroecksnadel K., Hörtnagl H., Rieder J., Fuchs D., Gottardis M. Influence of extreme long endurance sports activity on neopterin excretion. Pteridines. 2008:114–119. doi: 10.1515/pteridines.2008.19.1.114. [DOI] [Google Scholar]
- 159.Lindsay A., Lewis J.G., Scarrott C., Gill N., Gieseg S.P., Draper N. Assessing the effectiveness of selected biomarkers in the acute and cumulative physiological stress response in professional rugby union through non-invasive assessment. Int. J. Sports Med. 2015:446–454. doi: 10.1055/s-0034-1398528. [DOI] [PubMed] [Google Scholar]
- 160.Scheffer D.L., Silva L.A., Tromm C.B., da Rosa G.L., Silveira P.C., de Souza C.T., Latini A., Pinho R.A. Impact of different resistance training protocols on muscular oxidative stress parameters. Appl. Physiol. Nutr. Metab. 2012;37:1239–1246. doi: 10.1139/h2012-115. [DOI] [PubMed] [Google Scholar]
- 161.Jackson M.J. Control of reactive oxygen species production in contracting skeletal muscle. Antioxid. Redox Signal. 2011;15:2477–2486. doi: 10.1089/ars.2011.3976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Halliwell B., Cross C.E. Oxygen-derived species: their relation to human disease and environmental stress. Environ. Health Perspect. 1994;102:5–12. doi: 10.2307/3432205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.SIES H. Oxidative stress: introductory remarks. Oxidative Stress. 1985:1–8. doi: 10.1016/b978-0-12-642760-8.50005-3. [DOI] [Google Scholar]
- 164.Sies H. Oxidative stress: a concept in redox biology and medicine. Redox Biol. 2015;4:180–183. doi: 10.1016/j.redox.2015.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Sanchis-Gomar F., Garcia-Gimenez J.L., Perez-Quilis C., Gomez-Cabrera M.C., Pallardo F.V., Lippi G. Physical exercise as an epigenetic modulator: eustress, the “positive stress” as an effector of gene expression. J. Strength Cond. Res. 2012;26:3469–3472. doi: 10.1519/JSC.0b013e31825bb594. [DOI] [PubMed] [Google Scholar]
- 166.Watson J.D. Type 2 diabetes as a redox disease. Lancet. 2014;383:841–843. doi: 10.1016/S0140-6736(13)62365-X. [DOI] [PubMed] [Google Scholar]
- 167.Fialkow L., Wang Y., Downey G.P. Reactive oxygen and nitrogen species as signaling molecules regulating neutrophil function. Free Radic. Biol. Med. 2007;42:153–164. doi: 10.1016/j.freeradbiomed.2006.09.030. [DOI] [PubMed] [Google Scholar]
- 168.Ji L.L. Modulation of skeletal muscle antioxidant defense by exercise: role of redox signaling. Free Radic. Biol. Med. 2008:142–152. doi: 10.1016/j.freeradbiomed.2007.02.031. [DOI] [PubMed] [Google Scholar]
- 169.Dröge W. Redox regulation in anabolic and catabolic processes. Curr. Opin. Clin. Nutr. Metab. Care. 2006:190–195. doi: 10.1097/01.mco.0000222098.98514.40. [DOI] [PubMed] [Google Scholar]
- 170.Gomez-Cabrera M.C., Borrás C., Pallardo F.V., Sastre J., Ji L.L., Viña J. Decreasing xanthine oxidase-mediated oxidative stress prevents useful cellular adaptations to exercise in rats. J. Physiol. 2005;567:113–120. doi: 10.1113/jphysiol.2004.080564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Pinho R.A., Andrades M.E., Oliveira M.R., Pirola A.C., Zago M.S., Silveira P.C., Dal-Pizzol F., Moreira J.C. Imbalance in SOD/CAT activities in rat skeletal muscles submitted to treadmill training exercise. Cell Biol. Int. 2006;30:848–853. doi: 10.1016/j.cellbi.2006.03.011. [DOI] [PubMed] [Google Scholar]
- 172.Paulsen G., Egner I.M., Drange M., Langberg H., Benestad H.B., Fjeld J.G., Hallén J., Raastad T. A COX-2 inhibitor reduces muscle soreness, but does not influence recovery and adaptation after eccentric exercise. Scand. J. Med. Sci. Sports. 2010;20:1–13. doi: 10.1111/j.1600-0838.2009.00947.x. [DOI] [PubMed] [Google Scholar]
- 173.Markworth J.F., Vella L., Lingard B.S., Tull D.L., Rupasinghe T.W., Sinclair A.J., Maddipati K.R., Cameron-Smith D. Human inflammatory and resolving lipid mediator responses to resistance exercise and ibuprofen treatment. Am. J. Phys. Regul. Integr. Comp. Phys. 2013;305:R1281–R1296. doi: 10.1152/ajpregu.00128.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Vella L., Markworth J.F., Farnfield M.M., Maddipati K.R., Russell A.P., Cameron-Smith D. Intramuscular inflammatory and resolving lipid profile responses to an acute bout of resistance exercise in men. Phys. Rep. 2019;7:1–13. doi: 10.14814/phy2.14108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Martínez M.E., Heddens D., Earnest D.L., Bogert C.L., Roe D., Einspahr J., Marshall J.R., Alberts D.S. Physical activity, body mass index, and prostaglandin E2 levels in rectal mucosa. J. Natl. Cancer Inst. 1999;91:950–953. doi: 10.1093/jnci/91.11.950. [DOI] [PubMed] [Google Scholar]
- 176.Bejma J., Ji L.L. Aging and acute exercise enhance free radical generation in rat skeletal muscle. J. Appl. Physiol. 1999;87:465–470. doi: 10.1152/jappl.1999.87.1.465. [DOI] [PubMed] [Google Scholar]
- 177.Tidball J.G. Inflammatory processes in muscle injury and repair. Am. J. Phys. Regul. Integr. Comp. Phys. 2005;288:R345–R353. doi: 10.1152/ajpregu.00454.2004. [DOI] [PubMed] [Google Scholar]
- 178.Bury T.B., Pirnay F. Effect of prolonged exercise on neutrophil myeloperoxidase secretion. Int. J. Sports Med. 1995;16:410–412. doi: 10.1055/s-2007-973029. [DOI] [PubMed] [Google Scholar]
- 179.Favero T.G., Colter D., Hooper P.F., Abramson J.J. Hypochlorous acid inhibits Ca2+ -ATPase from skeletal muscle sarcoplasmic reticulum. J. Appl. Physiol. 1998;84:425–430. doi: 10.1152/jappl.1998.84.2.425. [DOI] [PubMed] [Google Scholar]
- 180.Margonis K., Fatouros I.G., Jamurtas A.Z., Nikolaidis M.G., Douroudos I., Chatzinikolaou A., Mitrakou A., Mastorakos G., Papassotiriou I., Taxildaris K., Kouretas D. Oxidative stress biomarkers responses to physical overtraining: implications for diagnosis. Free Radic. Biol. Med. 2007:901–910. doi: 10.1016/j.freeradbiomed.2007.05.022. [DOI] [PubMed] [Google Scholar]
- 181.Kawanishi N., Yano H., Yokogawa Y., Suzuki K. Exercise training inhibits inflammation in adipose tissue via both suppression of macrophage infiltration and acceleration of phenotypic switching from M1 to M2 macrophages in high-fat-diet-induced obese mice. Exerc. Immunol. Rev. 2010:105–118. [PubMed] [Google Scholar]
- 182.Speck A.E., Tromm C.B., Pozzi B.G., Paganini C.S., Tuon T., Silveira P.C.L., Aguiar A.S., Pinho R.A. The dose-dependent antioxidant effects of physical exercise in the hippocampus of mice. Neurochem. Res. 2014:1496–1501. doi: 10.1007/s11064-014-1339-6. [DOI] [PubMed] [Google Scholar]
- 183.Williams A.C.D.C., Craig K.D. Updating the definition of pain. Pain. 2016:2420–2423. doi: 10.1097/j.pain.0000000000000613. [DOI] [PubMed] [Google Scholar]
- 184.Nicholas M., Vlaeyen J.W.S., Rief W., Barke A., Aziz Q., Benoliel R., Cohen M., Evers S., Giamberardino M.A., Goebel A., Korwisi B., Perrot S., Svensson P., Wang S.J., Treede R.D. The IASP classification of chronic pain for ICD-11: chronic primary pain. Pain. 2019:28–37. doi: 10.1097/j.pain.0000000000001390. [DOI] [PubMed] [Google Scholar]
- 185.Woolf C.J., Allchorne A., Safieh-Garabedian B., Poole S. Cytokines, nerve growth factor and inflammatory hyperalgesia: the contribution of tumour necrosis factor α. Br. J. Pharmacol. 1997:417–424. doi: 10.1038/sj.bjp.0701148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Pinho-Ribeiro F.A., Verri W.A., Chiu I.M. Nociceptor sensory neuron–immune interactions in pain and inflammation. Trends Immunol. 2017:5–19. doi: 10.1016/j.it.2016.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Fujita M., da Luz Scheffer D., Lenfers Turnes B., Cronin S.J.F., Latrémolière A., Costigan M., Woolf C.J., Latini A., Andrews N.A. Sepiapterin reductase inhibition selectively reduces inflammatory joint pain and increases urinary sepiapterin. Arthritis Rheum. 2019:1–10. doi: 10.1002/art.41060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Woolf C.J., Salter M.W. Neuronal plasticity: increasing the gain in pain. Science (80-. ) 2000;288:1765–1768. doi: 10.1126/science.288.5472.1765. [DOI] [PubMed] [Google Scholar]
- 189.IASP . 2017. IASP Terminology, IASP. [Google Scholar]
- 190.Latremoliere A., Woolf C.J. Central sensitization: a generator of pain hypersensitivity by central neural plasticity. J. Pain. 2009;10:895–926. doi: 10.1016/j.jpain.2009.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Kobayashi Y., Kiguchi N., Fukazawa Y., Saika F., Maeda T., Kishioka S. Macrophage-T cell interactions mediate neuropathic pain through the glucocorticoid-induced tumor necrosis factor ligand system. J. Biol. Chem. 2015:12603–12613. doi: 10.1074/jbc.M115.636506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Schuh C.D., Pierre S., Weigert A., Weichand B., Altenrath K., Schreiber Y., Ferreiros N., Zhang D.D., Suo J., Treutlein E.M., Henke M., Kunkel H., Grez M., Nüsing R., Brüne B., Geisslinger G., Scholich K. Prostacyclin mediates neuropathic pain through interleukin 1β-expressing resident macrophages. Pain. 2014:545–555. doi: 10.1016/j.pain.2013.12.006. [DOI] [PubMed] [Google Scholar]
- 193.Trevisan G., Benemei S., Materazzi S., De Logu F., De Siena G., Fusi C., Fortes Rossato M., Coppi E., Marone I.M., Ferreira J., Geppetti P., Nassini R. TRPA1 mediates trigeminal neuropathic pain in mice downstream of monocytes/macrophages and oxidative stress. Brain. 2016:1361–1377. doi: 10.1093/brain/aww038. [DOI] [PubMed] [Google Scholar]
- 194.Talbot S., Abdulnour R.E.E., Burkett P.R., Lee S., Cronin S.J.F., Pascal M.A., Laedermann C., Foster S.L., Tran J.V., Lai N., Chiu I.M., Ghasemlou N., DiBiase M., Roberson D., Von Hehn C., Agac B., Haworth O., Seki H., Penninger J.M., Kuchroo V.K., Bean B.P., Levy B.D., Woolf C.J. Silencing nociceptor neurons reduces allergic airway inflammation. Neuron. 2015;87:341–354. doi: 10.1016/j.neuron.2015.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Latremoliere A., Latini A., Andrews N., Cronin S.J., Fujita M., Gorska K., Hovius R., Romero C., Chuaiphichai S., Painter M., Miracca G., Babaniyi O., Remor A.P., Duong K., Riva P., Barrett L.B., Ferreirós N., Naylor A., Penninger J.M., Tegeder I., Zhong J., Blagg J., Channon K.M., Johnsson K., Costigan M., Woolf C.J. Reduction of neuropathic and inflammatory pain through inhibition of the tetrahydrobiopterin pathway. Neuron. 2015;86:1393–1406. doi: 10.1016/j.neuron.2015.05.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Cronin S.J.F., Seehus C., Weidinger A., Talbot S., Reissig S., Seifert M., Pierson Y., McNeill E., Longhi M.S., Turnes B.L., Kreslavsky T., Kogler M., Hoffmann D., Ticevic M., da Luz Scheffer D., Tortola L., Cikes D., Jais A., Rangachari M., Rao S., Paolino M., Novatchkova M., Aichinger M., Barrett L., Latremoliere A., Wirnsberger G., Lametschwandtner G., Busslinger M., Zicha S., Latini A., Robson S.C., Waisman A., Andrews N., Costigan M., Channon K.M., Weiss G., Kozlov A.V., Tebbe M., Johnsson K., Woolf C.J., Penninger J.M. The metabolite BH4 controls T cell proliferation in autoimmunity and cancer. Nature. 2018;563:564–568. doi: 10.1038/s41586-018-0701-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Werner E.R., Blau N., Thöny B. Tetrahydrobiopterin: biochemistry and pathophysiology. Biochem. J. 2011;438:397–414. doi: 10.1042/BJ20110293. [DOI] [PubMed] [Google Scholar]
- 198.Channon K.M. Tetrahydrobiopterin: regulator of endothelial nitric oxide synthase in vascular disease. Trends Cardiovasc. Med. 2004:323–327. doi: 10.1016/j.tcm.2004.10.003. [DOI] [PubMed] [Google Scholar]
- 199.Tegeder I., Costigan M., Griffin R.S., Abele A., Belfer I., Schmidt H., Ehnert C., Nejim J., Marian C., Scholz J., Wu T., Allchorne A., Diatchenko L., Binshtok A.M., Goldman D., Adolph J., Sama S., Atlas S.J., Carlezon W.A., Parsegian A., Lotsch J., Fillingim R.B., Maixner W., Geisslinger G., Max M.B., Woolf C.J. GTP cyclohydrolase and tetrahydrobiopterin regulate pain sensitivity and persistence. Nat. Med. 2006;12:1269–1277. doi: 10.1038/nm1490. [DOI] [PubMed] [Google Scholar]
- 200.Thöny B., Auerbach G., Blau N., Thöny B., Auerbach G., Blau N. Tetrahydrobiopterin biosynthesis, regeneration and functions. Biochem. J. 2000;347(Pt 1):1–16. doi: 10.1042/0264-6021:3470001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Giannotti E., Koutsikos K., Pigatto M., Rampudda M.E., Doria A., Masiero S. Medium-/long-term effects of a specific exercise protocol combined with patient education on spine mobility, chronic fatigue, pain, aerobic fitness and level of disability in fibromyalgia. Biomed. Res. Int. 2014:1–9. doi: 10.1155/2014/474029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Kelley G.A., Kelley K.S. Effects of exercise on depressive symptoms in adults with arthritis and other rheumatic disease: a systematic review of meta-analyses. BMC Musculoskelet. Disord. 2014:1–9. doi: 10.1186/1471-2474-15-121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Bobinski F., Martins D.F., Bratti T., Mazzardo-Martins L., Winkelmann-Duarte E.C., Guglielmo L.G.A., Santos A.R.S. Neuroprotective and neuroregenerative effects of low-intensity aerobic exercise on sciatic nerve crush injury in mice. Neuroscience. 2011;194:337–348. doi: 10.1016/j.neuroscience.2011.07.075. [DOI] [PubMed] [Google Scholar]
- 204.Bobinski F., Teixeira J.M., Sluka K.A., Santos A.R.S. Interleukin-4 mediates the analgesia produced by low-intensity exercise in mice with neuropathic pain. Pain. 2018:437–450. doi: 10.1097/j.pain.0000000000001109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Leung A., Gregory N.S., Allen L.A.H., Sluka K.A. Regular physical activity prevents chronic pain by altering resident muscle macrophage phenotype and increasing interleukin-10 in mice. Pain. 2016:70–79. doi: 10.1097/j.pain.0000000000000312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Grace P.M., Fabisiak T.J., Green-Fulgham S.M., Anderson N.D., Strand K.A., Kwilasz A.J., Galer E.L., Walker F.R., Greenwood B.N., Maier S.F., Fleshner M., Watkins L.R. Prior voluntary wheel running attenuates neuropathic pain. Pain. 2016:2012–2023. doi: 10.1097/j.pain.0000000000000607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Rahman W., Suzuki R., Webber M., Hunt S.P., Dickenson A.H. Depletion of endogenous spinal 5-HT attenuates the behavioural hypersensitivity to mechanical and cooling stimuli induced by spinal nerve ligation. Pain. 2006;123:264–274. doi: 10.1016/j.pain.2006.02.033. [DOI] [PubMed] [Google Scholar]
- 208.Veasey S.C., Fornal C.A., Metzler C.W., Jacobs B.L. Single-unit responses of serotonergic dorsal raphe neurons to specific motor challenges in freely moving cats. Neuroscience. 1997;79:161–169. doi: 10.1016/S0306-4522(96)00673-2. [DOI] [PubMed] [Google Scholar]
- 209.Gomez-Merino D., Béquet F., Berthelot M., Chennaoui M., Guezennec C.Y. Site-dependent effects of an acute intensive exercise on extracellular 5-HT and 5-HIAA levels in rat brain. Neurosci. Lett. 2001;301:143–146. doi: 10.1016/S0304-3940(01)01626-3. [DOI] [PubMed] [Google Scholar]
- 210.Brown B.S., Payne T., Kim C., Moore G., Krebs P., Martin W. Chronic response of rat brain norepinephrine and serotonin levels to endurance training. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1979;46:19–23. doi: 10.1152/jappl.1979.46.1.19. [DOI] [PubMed] [Google Scholar]