Summary
Ten calves which had contracted acute diarrhoea caused by rotavirus, coronavirus and Cryptosporidium were used to test the hypothesis that feeding lactose‐hydrolysed cow's milk instead of unprocessed cow's milk improves sugar absorption in diarrhoeic calves. The animals were rehydrated with an orally administered solution containing electrolytes and glucose. Thereafter the calves received one test meal of whole fresh cow's milk whose lactose had been hydrolysed by added lactase and one test meal of unprocessed cow's milk at an interval of 24 h in a cross‐over design trial. In comparison with unprocessed milk, the intake of milk containing hydrolysed lactose resulted in a slight decrease of mean breath hydrogen concentration (P = 0.18), but also a slight decrease of mean blood galactose concentration (P = 0.14). There was no treatment effect on mean plasma glucose concentration. Peak plasma glucose and blood galactose concentration tended to be delayed after the intake of lactose‐hydrolysed milk, which implies that gastric emptying was probably delayed. The results show that feeding milk which contains hydrolysed lactose does not significantly improve lactose utilization in calves that are suffering from benign infectious diarrhoea.
Introduction
Dehydration and acidosis, which develop during acute diarrhoea in newborn calves, are best corrected by oral or parenteral use of solutions containing electrolytes and glucose, but rehydration solutions contain insufficient amounts of energy and nutrients to meet the animal's requirements. The question of whether calves suffering from diarrhoea should receive milk is controversial. The activity of the small intestinal β‐galactosidase (lactase) is reduced in calves with diarrhoea ( Tzipori et al., 1983; Torres‐Medina et al., 1985). Lactase deficiency would theoretically result in malabsorptive diarrhoea with loss of fluid and electrolytes when milk feeding is resumed. On the other hand, the use of oral rehydration solutions as the sole feed for several days results in reduced regeneration of the intestinal mucosa and in starvation ( Heath et al., 1989). The feeding of milk which has been incubated with lactase should theoretically alleviate the problems related to insufficient endogenous lactase synthesis. Humans who suffer from lactose maldigestion respond favourably to milk which has been treated with lactase ( Suarez et al., 1995), and the successful use of lactose‐hydrolysed milk in a calf which presumably suffered from congenital lactase deficiency has been described ( Olchowy et al., 1993). Although this procedure could theoretically be useful in diarrhoeic calves with reduced intestinal lactase activity, there is no published report on the use of lactase in calves with diarrhoea. In the present study the hypothesis was tested that calves suffering from infectious enteritis suffer from lactose malabsorption due to lactase deficiency, and that under these circumstances the use of lactose‐hydrolysed milk improves sugar absorption.
Materials and Methods
Animals and experimental design
Ten Simmental × Red Holstein and Swiss Brown calves of both sexes, which were born at the research station and had received fresh colostrum, and later on fresh cow's milk, contracted acute diarrhoea when they were 12 ± 2.3 days old. When watery diarrhoea had lasted over 12 h, they were chosen to participate in the study. Faecal samples were collected and were analysed for enteropathogenic bacteria, for Cryptosporidium, rotavirus and coronavirus. The calves, who were able to stand and to suckle, received an oral rehydration solution containing glucose, glycine and electrolytes (Rehydran®; Veterinaria AG, Zürich, Switzerland; content in mmol/l: glucose, 131; glycine, 120; gluconate, 2; sodium, 85; potassium, 27; calcium, 2; magnesium, 2; chloride, 85; dihydrogen phosphate, 27; sulphate, 1) during the following 24–36 h. No medication was administered in addition to the rehydration solution. After the rehydration period the animals were weighed and entered the clinical trial which was designed as a simple cross‐over study with 10 repetitions per treatment.
Experimental protocol
Fresh bulk milk from the 60 cows of the research herd was used in the trial. The milk, which was fed without pre‐treatment was obtained 1 h before feeding and was heated to 40°C. For the production of hydrolysed milk, tank milk was obtained about 13 h before feeding. Four millilitres of lactase (Lactozym®; Novo Nordisk Ferment AG, Dittingen, Switzerland) were added per litre, the milk was left standing at 5°C for 12 h and was heated to 40°C before it was fed. Samples of lactose‐hydrolysed milk were collected immediately before feeding, the casein was precipitated with rennet, and the whey samples were stored at −20°C until the galactose was analysed.
About 12 h after the calves had received the last meal of the rehydration solution, a catheter (Cavafix®, Braun, Emmenbrücke, Switzerland] was placed into their jugular vein, and 1 h later they received their first test meal. Five randomly selected calves received a first test meal of lactose‐hydrolysed milk and 24 h later a second test meal of unprocessed whole cow's milk, whereas the other five calves received unprocessed milk as the first test meal and lactose‐hydrolysed milk 24 h later. Each calf was offered 0.5 l of milk per 10 kg body weight (BW) during the first absorption test and was allowed to drink for 5 min. The amount of ingested milk was recorded, and on the second test day, each calf was offered the amount of milk which it had consumed the previous day. Between the two test meals, the calves received only 2 l of the rehydration solution (Rehydran®) but no milk. Blood and breath samples were collected 15 and 5 min before the test meals were fed and 0.5, 1, 1.5, 2, 3, 4, 5 and 6 h after the test meals. Blood was drawn from the catheter with a syringe. Blood for glucose analysis was put into tubes containing EDTA and sodium fluoride, and blood for galactose determination was deproteinized with perchloric acid immediately after collection. The blood was centrifuged at 2000 g for 15 min within 15 min after sample collection, and aliquots were stored at −20°C until they were analysed. Total exhaled breath gas samples were collected with a breathing bag as described by Holland et al. (1986). Briefly, the face mask which was connected by a non‐rebreathing valve to a 5 l breathing bag was placed over the nostrils and the mouth of the calves. When the breathing bag was filled with breath gas it was manually compressed and allowed to refill. Breath gas samples were analysed for H2 within 15 min after collection.
Analytical procedures
Plasma glucose was determined using the hexokinase method (Gluco‐quant®; Boehringer, Mannheim, Germany) on an automated analyser (Cobas Mira®; Hoffmann‐La Roche, Basel, Switzerland).
Galactose in deproteinized blood and in whey was analysed using an enzymatic method (Test 176303®; Boehringer) on a Cobas Mira® analyser.
Breath H2 concentration was measured electrochemically by use of an exhaled H2 monitor (monitor 81 HP®; Gas Measurement Instruments Ltd, Renfrew, Scotland). The monitor, which has a sensitivity of 2 mg H2 per litre, was calibrated before each run with a calibrated gas containing 94 mg H2 per litre and with fresh air containing no detectable amounts of H2.
Data analysis
Means and standard errors of means are used to describe the data. For the statistical evaluation, areas under the curves (AUC) of blood galactose, plasma glucose and breath H2 were calculated by use of the trapezoid method. AUC of breath H2 was calculated for the whole 6 h period and for the period between 2 and 6 h after milk intake, as the estimated orocaecal transit time for sugars in calves is about 2 h ( Holland et al., 1986). The data which were obtained after the intake of lactose‐hydrolysed milk and of unprocessed milk were compared using the two‐tailed paired t‐test.
Results
The 10 calves which had contracted acute diarrhoea showed no clinical signs of severe dehydration. Although they voided watery faeces, they were able to stand and to suckle the rehydration solution.
Rotavirus, coronavirus and Cryptosporidium were detected in six, seven and two faecal samples; one pathogen was found in five samples and two pathogens were found in the other five samples. No enteropathogenic bacteria were isolated. The lactose‐hydrolysed milk contained 23.5 ± 0.25 g of galactose per litre, which corresponds to 47 ± 0.5 g of cleaved lactose per litre. The added lactase had thus cleaved more than 90 % of the lactose since fresh whole milk contains about 50 g of lactose per litre and only traces of glucose and galactose ( Bird et al., 1995). The calves, which weighed 48 ± 3.2 kg, drank 0.40 ± 0.02 l of milk per 10 kg BW. Thus the calves ingested about 2 ± 0.08 g of sugar as lactose or glucose plus galactose per kg BW at each test meal. Each calf drank identical amounts of milk on both test days within 5 min.
Pre‐prandial concentration of plasma glucose, blood galactose and breath H2 were almost identical in both treatments ( 1, 2, 3). Glucose and galactose tended to reach the peak value later after the intake of lactose‐hydrolysed milk than after the intake of unprocessed milk (249 ± 33 versus 200 ± 27 min, P = 0.15, and 237 ± 31 versus 165 ± 22 min, P = 0.06). There was no treatment effect on plasma glucose concentration (P = 0.88). Blood galactose concentration tended to be lower after the intake of lactose‐hydrolysed milk than after the intake of unprocessed milk (P = 0.14). Breath H2 concentration in the whole 6 h period and between 2 and 6 h after milk intake did not differ between the two treatments (P = 0.18 and P = 0.19). Pre‐prandial concentration of blood galactose and of breath hydrogen as well as post‐prandial concentration of plasma glucose, blood galactose and breath hydrogen concentration did not differ between day 1 and day 2 of the trial. On the other hand, fasting plasma glucose concentration was higher (4.5 ± 0.6 versus 3.9 ± 0.9 mmol/l, P < 0.05), and the post‐prandial increase above fasting level tended to be lower (P = 0.06) on the second compared with the first day of the trial. This carry‐over effect was not dependent on the type of test meal, unprocessed and lactose‐hydrolysed milk, which had been consumed on the first test day.
Figure 1.
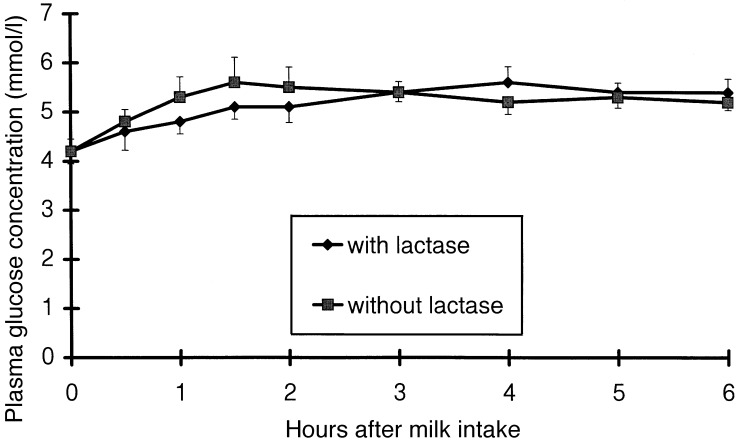
Plasma glucose concentration after the intake of lactose‐hydrolysed cow's milk (with lactase) and of unprocessed cow's milk (without lactase). Data (10 observations at each point of measurement) are shown as mean and SEM.
Figure 2.
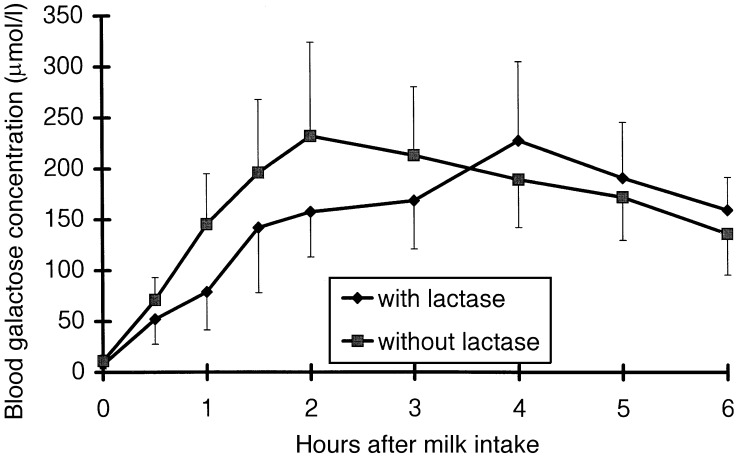
Blood galactose concentration after the intake of lactose‐hydrolysed cow's milk (with lactase) and of unprocessed cow's milk (without lactase). Data (10 observations at each point of measurement) are shown as mean and SEM.
Figure 3.
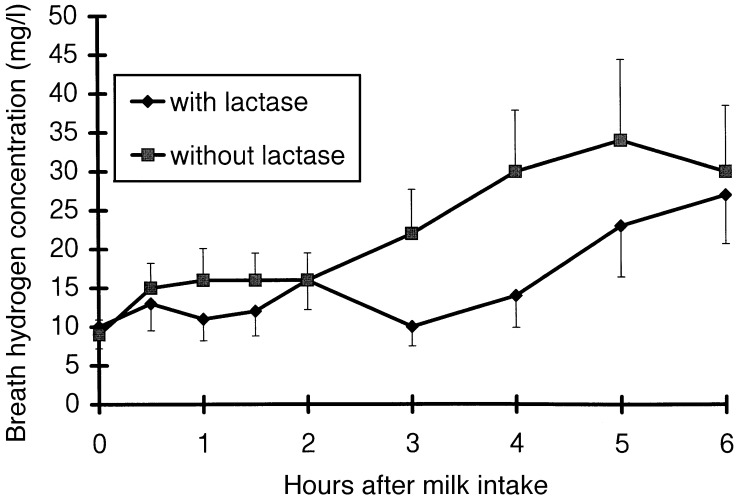
Total breath hydrogen concentration after the intake of lactose‐hydrolysed cow's milk (with lactase) and of unprocessed cow's milk (without lactase). Data (10 observations at each point of measurement) are shown as mean and SEM.
Discussion
Rotavirus, coronavirus and Cryptosporidium are among the most commonly found pathogens in new‐born calves with undifferentiated acute diarrhoea ( Radostits et al., 1994). The calves of the present study can therefore be regarded as representative of naturally infected neonatal calves suffering from malabsorption diarrhoea. Since the calves of the research herd were closely supervised, signs of diarrhoea were noticed without delay, and the oral rehydration was started soon after the onset of the disease. The rapid initiation of oral rehydration probably prevented the development of severe signs of dehydration. Nevertheless, the observed increase in breath hydrogen concentration after the intake of milk, a phenomenon which is not seen in healthy calves receiving moderate amounts of lactose ( Nappert et al., 1993), shows that carbohydrate malabsorption occurred in the experimental animals although they had not been critically ill. This finding confirms the observation of Nappert et al. (1993) that the extent of carbohydrate malabsorption is not dependent on the clinical condition of the calves suffering from diarrhoea.
The amount of sugar which the calves received in their test meals is comparable with the amount which diarrhoeic calves have received in other digestibility studies ( Holland et al., 1989; Gutzwiller and Blum, 1996). In comparison with the blood values in diarrhoeic calves which had received 2 g/kg BW of lactose as an aqueous solution ( Gutzwiller and Blum, 1996), blood galactose concentration reached the peak value much later, and plasma glucose concentration rose to a lesser degree in the calves of this study which had received 2 g/kg BW of sugar in milk. A delay in gastric emptying after the intake of milk instead of an aqueous lactose solution, as has been shown in humans ( Solomons et al., 1979), is probably the cause of this phenomenon. Results obtained from oral tolerance tests using lactose solutions may therefore differ from the more natural condition encountered during milk feeding. The tendency of the galactose concentration to be lower and to reach a peak later after the intake of milk which contains hydrolysed lactose is an indication that the monosaccharides of the processed milk leave the abomasum more slowly than does lactose of unprocessed milk. A lower rate of galactose absorption would result in a more complete hepatic transformation of galactose to glucose and consequently in a lower galactose concentration in the peripheral blood. The presence of glucose, but not of lactose, in the duodenum has been shown to inhibit abomasal emptying in the milk‐fed calf ( Bell and Mostaghini, 1975), which might explain the postulated effect of lactose‐hydrolysed milk on abomasal emptying. Since a delayed gastric emptying has been shown to improve lactose tolerance in humans ( Solomons et al., 1979; Vesa et al., 1997), lactose‐hydrolysed milk should theoretically improve sugar utilization both by delaying gastric emptying and by alleviating the negative effects of lactase deficiency.
These assumptions are not supported by the results of the present trial since the measured breath H2 concentrations, which indicate that carbohydrates were fermented in the large intestine, did not differ between the two treatments. The increased pre‐prandial plasma glucose concentration on day 2 of the trial is a sign of the improved energy status of the calves, since milk contains about five times more energy per litre than does the rehydration solution which was administered before the first test day. The fact that plasma glucose concentration after milk intake tended to increase less when pre‐prandial glucose concentration was increased demonstrates that, due to hormonal control mechanisms, plasma glucose concentration is not a sensitive parameter for intestinal glucose absorption, as has been shown previously ( Gutzwiller and Blum, 1996). Breath hydrogen determination, which is the most widely used method to measure lactose malabsorption in humans ( Brummer et al., 1993), is probably better suited to test lactose maldigestion in calves. The results of the absorption tests suggest that a deficiency of endogenous lactase may not play an important role in the pathogenesis of malabsorption diarrhoea in most calves suffering from undifferentiated neonatal diarrhoea. In healthy young milk‐fed calves, intestinal lactase is not the limiting step in the process of lactose digestion and the subsequent absorption of glucose and galactose ( Coombe and Smith, 1973).
Although lactase activity is reduced in calves which are infected with rota‐ and coronavirus and with Cryptosporidium ( Tzipori et al., 1983; Torres‐Medina et al., 1985), monosaccharide malabsorption also occurs in viral and cryptosporidial enteritis ( Nappert et al., 1993; Gutzwiller and Blum, 1996). The results of the present study suggest that the impaired capacity of the small intestine to absorb monosaccharides rather than the reduced intestinal lactase activity is the limiting step in carbohydrate utilization in calves with diarrhoea. The study shows that unprocessed cow's milk seems to be just as suitable as lactose‐hydrolysed cow's milk for the majority of calves which suffer from diarrhoea caused by viruses or Cryptosporidium. The 10 calves which were used in this study recovered rapidly from the acute disease. Although the severity of the acute diarrhoeal disease does not seem to be correlated with the degree of malabsorption in calves ( Nappert et al., 1993), the results of the present investigation do not rule out the possibility that individual calves suffering from chronic severe diarrhoea might respond favourably to lactose‐hydrolysed milk.
References
- Bell, F. & Mostaghini K., 1975:Duodenal control of gastric emptying in the milk‐fed calf. J. Physiol. 245,387 407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bird, P. C. , Atwood, And , Hartmann P., 1995:The response of blood galactose to oral doses of lactose, galactose plus glucose and milk to piglets. Br. J. Nutr. 73,753 761. [DOI] [PubMed] [Google Scholar]
- Brummer, R. , Karibe M., Stockbrügger R., 1993:Lactose malabsorption. Optimalization of investigational methods. Scand. J. Gastroenterol. 28(Suppl. 200),65 69. [PubMed] [Google Scholar]
- Coombe, N. & Smith R., 1973:Absorption of glucose and galactose and digestion and absorption of lactose by the preruminant calf. Br. J. Nutr. 30,331 344. [DOI] [PubMed] [Google Scholar]
- Gutzwiller, A. & Blum J., 1996:Effects of oral lactose and xylose loads on blood glucose, galactose, xylose, and insulin values in healthy calves and calves with diarrhea. Am. J. Vet. Res. 57,560 563. [PubMed] [Google Scholar]
- Heath, S. , Naylor J., Guedo B., Petrie L., Rousseaux C., Radostits O., 1989:The effects of feeding milk to diarrheic calves supplemented with oral electrolytes. Can. J. Vet. Res. 53,477 485. [PMC free article] [PubMed] [Google Scholar]
- Holland, R. , Herdt T., Refsal K., 1986:Breath hydrogen concentration and small intestinal malabsorption in calves. Am. J. Vet. Res. 47,2020 2024. [PubMed] [Google Scholar]
- Holland, R. , Herdt T., Refsal K., 1989:Pulmonary excretion of H2 in calves with Cryptosporidium‐induced malabsorption. Dig. Dis. Sci. 34,1399 1404. [DOI] [PubMed] [Google Scholar]
- Nappert, G. , Hamilton D., Petrie L., Naylor J., 1993:Determination of lactose and xylose malabsorption in preruminant diarrheic calves. Can. J. Vet. Res. 57,152 158. [PMC free article] [PubMed] [Google Scholar]
- Olchowy, T. , Linnabary R., Andrews F., Longshore R., 1993:Lactose intolerance in a calf. J. Vet. Int. Med. 7,12 15. [DOI] [PubMed] [Google Scholar]
- Radostits, O. , Blood D., Gay C., 1994:Veterinary Medicine,8th edn. Baillière Tindall,London. [Google Scholar]
- Solomons, N. , Garcia‐Ibanes R., Viteri F., 1979:Reduced rate of breath hydrogen excretion with lactose tolerance tests in young children using whole milk. Am. J. Clin. Nutr. 32,783 786. [DOI] [PubMed] [Google Scholar]
- Suarez, F. , Savaiano D., Levitt M., 1995:The treatment of lactose intolerance. Aliment. Pharmacol. Ther. 9,589 597. [DOI] [PubMed] [Google Scholar]
- Torres‐Medina, A. , Schlafer D., Mebus C., 1985:Rotaviral and coronaviral diarrhea. Vet. Clin. North Am. Food Anim. Pract. 1,471 493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzipori, S. , Smith M., Halpin C., Angus K., Sherwood D., Campbell I., 1983:Experimental cryptosporidiosis in calves: clinical manifestations and pathological findings. Vet. Rec. 112,116 120. [DOI] [PubMed] [Google Scholar]
- Vesa, T. , Marteau P., Briet F., Boutron‐Ruault M., Rambaud J., 1997:Raising milk energy content retards gastric emptying of lactose in lactose‐intolerant humans with little effect on lactose digestion. J. Nutr. 127,2316 2320. [DOI] [PubMed] [Google Scholar]


