Abstract
Sestrin2 (Sesn2), a metabolic regulator, accumulates in response to a diverse array of cellular stresses. Sesn2 regulates cellular metabolism by inhibiting the mammalian target of rapamycin complex 1 through the AMP-activated protein kinase (AMPK) signaling pathway. Recently, researchers reported that Sesn2 regulates the differentiation and function of innate immune cells and T cells; however, the role of Sesn2 in B cells is largely unknown. In this study, we investigated the role of Sesn2 in Ig class switching and Ig production in mouse B cells. We observed that mouse B cells express Sesn2 mRNA. Interestingly, the expression of germline ε transcripts (GLTε) was selectively decreased in lipopolysaccharide-stimulated Sesn2−/− splenocytes. Overexpression of Sesn2 increased GLTε promoter activity in B cells. In addition, AICAR (an activator of AMPK) selectively increased IL-4-induced GLTε expression and surface IgE (sIgE) expression in splenocytes. Furthermore, AICAR selectively enhanced IL-4-induced GLTε expression, sIgE expression, and IgE production by anti-CD40-stimulated B cells. We observed that ovalbumin (OVA)-specific IgE concentration was reduced in OVA-challenged Sesn2−/− mice. Taken together, these results indicate that Sesn2-AMPK signaling selectively enhances IL-4-induced IgE class switching and IgE production by B cells, suggesting that this could be a therapeutic strategy targeting Sesn2 in IgE-mediated allergic diseases.
Keywords: B cells, Sesn2, AMPK, Germline ε transcripts, IgE
INTRODUCTION
Sestrins (Sesns) is a highly conserved stress-inducing protein in several species including mammals, Drosophila melanogaster, and Caenorhabditis elegans (1). Sesn family was known to perform protective functions through regulation of various mechanisms such as endoplasmic reticulum stress, autophagy, metabolic homeostasis, inflammation, and oxidative stress in most physiological and pathological conditions (2). Three Sesn genes, Sesn1 (PA26), Sesn2 (Hi95), and Sesn3, are identified in vertebrates (3). Sesn1 and Sesn2 are mainly responsive to p53, while Sesn3 is activated by forkhead transcription factors family (4). Sesn1 is involved in autophagy-related genes and can suppress mTOR complex 1 (mTORC1) or reactive oxygen species in cells. Sesn2 activates AMP-activated protein kinase (AMPK) and inhibits mTORC1 signaling, and has antioxidant properties. Sesn3 activates the AMPK/tuberous sclerosis complex 1/2 axis to inhibit mTORC1 activity and maintain Akt activity. Since the discovery of Sesn in 2002, Sesn2 has been the most active research among Sesn family members, whereas investigations on the function or structure of Sesn1 and Sesn3 have been limited (3).
Sesn2 exhibits pleiotropic biological functions such as survival, inflammation, and senescence of immune cells (1,5). Therefore, Sesn2 plays a protective role in various diseases, including cardiovascular and metabolic disorders, neurodegenerative diseases, and cancer (6). Sesn2 regulates metabolic homeostasis via upstream regulation of mTORC1 and AMPK signaling pathways, which are critical for energy and nutrient sensing in cells (1,7). Sesn2 inhibits mTORC1 activation in cells mainly through the activation of AMPK and phosphorylation of tuberous sclerosis 2. Genetic silencing and knockdown of Sesn2 in vitro and in vivo cause sustained activation of mTOR signaling in multiple cell types, including liver, indicating the essential role of Sesn2 in mTOR inhibition (6).
Recently, many studies were conducted on the function and role of Sesn2 in immunity, and most of these studies focused on macrophages and T cells. Sesn2 and Sesn3 suppress NK cell-mediated cytotoxic activity on ovarian cancer cells through AMPK and mTORC1 signaling (8). Upregulation of Sesn2 expression is mediated by NOS2-generated NO or AP-1, Nrf2, and the ubiquitin-proteasome system in macrophages (9,10,11), and Sesn2 upregulation induces mitophagy activation, which contributes to inhibition of the prolonged NLRP3 inflammasome activation (10). In addition, increased expression of Sesn2 could promote the survival of macrophages to apoptosis and reduce the expression of proinflammatory cytokines, which may contribute to the improvement of inflammatory diseases (12,13). Inhibition of Sesn1, Sesn2, and Sesn3 in senescent T cells results in broad functional reversal of senescence, apparent as the enhancement of cell viability (5,14). Mechanically, they demonstrate that the MAP kinases, including ERK, JNK, and p38, mediate the prosenescent function of the Sesns in CD4+ T cells through the formation of a new immunosuppressive complex (Sesn-MAPK activation complex), rather than the mTOR pathway (14). Thus, Sesn2 performs a variety of functions in immune cells. However, the study of the role of Sesn2 in B cells has not been investigated to date. As mentioned above, Sesn2 inhibits mTORC1 activation. Interestingly, mTORC1 negatively regulates IL-4-induced STAT6 signaling in Th2 cell differentiation (15). In B cells, the IL-4-induced STAT6 signaling is essential for IgE class switch recombination (CSR) (16). Therefore, in the present study, we focused on the role of Sesn2 in B cell Ig CSR.
Ig CSR occurs in B cells by deletion of the internal germline gene in the Ig heavy (H) chain and causes switching from IgM to IgG or IgE or IgA producing B cells (17). The initiation of Ig CSR requires the activation of B cells through stimuli, such as CD40 ligand, LPS, and cytokines. In this process, cytokine-induced germline transcripts (GLT) are a prerequisite for Ig CSR, and activation-induced cytidine deaminase (AID) is an essential enzyme for the initiation of double strand breaks in the switch region of the IgH gene (17). The representative cytokines regulating GLTs transcription are IL-4, TGF-β1, and IFN-γ. GLTε/GLTγ1, GLTα/GLTγ2b, and GLTγ2a (GLTγ2c)/GLTγ3 are induced by IL-4, TGF-β1, and IFN-γ, respectively, in mouse B cells (18). After Ig CSR, the GLTµ promoter becomes associated with each constant region gene and continues to be active, generating transcripts termed “post-switch transcripts” (PST) (19). Thus, one may monitor GLT and PST expression as indicative of active Ig CSR.
In this study, we evaluate the roles of Sesn2 and AMPK in GLT expression, surface Ig (sIg) expression, and Ig production in anti-CD40/IL-4-stimulated mouse B cells. Our results suggest that Sesn2-AMPK signaling selectively increases IgE production by promoting IgE class switching in B cells.
MATERIALS AND METHODS
Animals
C57BL/6 mice were purchased from Damool Science (Daejeon, Korea), and Sesn2−/− (Sesn2 knockout) mice in the CL57BL/6 background were provided by Ehwa Woman's University (Seoul, Korea). Mice were maintained in an 8:16 h light:dark cycle in an animal environmental control chamber. Eight- to 12-wk-old mice were used, and animal care was conducted in accordance with the institutional guidelines of the Institutional Animal Care and Use Committee of Konyang University (approval number: P-16-16-E-01).
Cell culture and reagents
Mouse resting B cells were purified as previously described (20). After staining the cells with anti-B220-PE (BD Biosciences, San Jose, CA, USA) and anti-CD43-FITC (eBioscience, San Hose, CA, USA), the purity of resting B cells (CD43-B220+, ≥97%) was assessed by CytoFLEX (Beckman Coulter, Pasadena, CA, USA). The mouse B cell line, CH12F3-2A (surface µ+), was provided by Dr. T. Honjo (Kyoto University, Kyoto, Japan). The mouse B cell line, L10A6.2 (surface µ+, mature B cell line), was provided by Dr. J. Stavnezer (University of Massachusetts Medical School, Worcester, MA, USA). Cells were cultured in a RPMI-1640 medium (WelGENE, Gyeongsan, Korea) supplemented with 50 µM 2-merchaptoethanol, 10% FBS (WelGENE), and penicillin (50 U/ml)/streptomycin (50 µg/ml), and the cells were incubated at 37°C in a humidified 5% CO2 incubator (Forma Scientific, Marietta, OH, USA). Cells were treated with LPS (Sigma Aldrich, Saint Louis, MO, USA) or anti-CD40 Ab (eBioscience), rmIL-4 (R&D Systems, Minneapolis, MN, USA), AICAR (LC Laboratories, Woburn, MA, USA), rapamycin (LC Laboratories), and everolimus (Invivogen, San Diego, CA, USA).
RNA isolation and RT-PCR
RNA isolation and RT-PCR were performed as previously described (21). The PCR primers (Supplementary Table 1) were synthesized by Bioneer (Daejeon, Korea). PCR for β-actin was performed in parallel to normalize cDNA concentrations within each set of samples. PCR products were resolved by electrophoresis on 2% agarose gel. Semi-quantitative RT-PCR analysis was performed using cDNA dilutions.
Flow cytometric analysis
Surface Ig staining was performed with anti-mouse IgE-FITC, anti-mouse IgE-PE (eBioscience), anti-mouse B220-PE, anti-mouse B220-FITC, and anti-mouse CD69 (BD Biosciences) in the dark for 30 min at 4°C. The stained cells were analyzed by flow cytometry (CytoFLEX, Beckman Coulter).
Cell viability assay
Cell viability was determined using the EZ-Cytox cell viability assay (DaeilLab Service Co., Ltd., Seoul, Korea) according to the manufacturer's instructions (21).
Transfection and luciferase reporter assays
Luciferase reporter plasmids pXP2-GLTε[−162/+53], pGL3-GLTγ1[−148/+202], and pGL3-GLTα[−3,028/+72] which, respectively, contain mouse GLTε, GLTγ1, and GLTα promoter. The expression plasmid for Sesn2 (pLU-Sesn2) and pLU-GFP were provided by Dr. Budanov (Trinity College, Dublin, Ireland). The expression plasmid for Nrf2 (pcDNA3-Myc3-Nrf2) was obtained from AddGene (Cambridge, MA, USA). Transfection was performed by electroporation with the Gene Pulser II electroporation system (Bio-Rad Laboratories, Hercules, CA, USA), as described previously (22). Reporter plasmids were co-transfected with pCMVβgal (Stratagene, La Jolla, CA, USA), and luciferase and β-gal assays were performed as described previously (22).
Western blot analysis
Cell lysates were prepared in lysis buffer(RIPA buffer [iNtRON Biotechnology, Seongnam, Korea] with protease inhibitor [Sigma], and phosphatase inhibitor [Sigma] mixture). Proteins were subjected for SDS/PAGE and transferred onto Nitrocellulose Membrane (Dogen, Seoul, Korea). Membranes were probed with the antibodies below and detected with Amersham ECL Prime Western Blotting Detection Reagent (Amersham Biosciences, Roosendaal, The Netherlands) or Ezwest Lumi one (ATTO, Tokyo, Japan). The following antibodies were used: rabbit anti-STAT6, mouse anti-β-actin (Santa Cruz Biotechnology, Santa Cruz, CA, USA), rabbit anti-phospho-STAT6 (Millipore, Bedford, MA, USA), rabbit anti-phospho-AMPK, and rabbit anti-AMPK (Cell Signaling Technology, Beverly, MA, USA).
Isotype-specific ELISA
Abs produced in B cell cultures were detected using isotype-specific ELISA (21).
Immunization and detection of ovalbumin (OVA)-specific Ig levels by ELISA
Mice were immunized with OVA (Sigma Aldrich) and alum (Thermo Fisher Scientific, Rockford, IL, USA) by intraperitoneal injection on day 0 and day 7, and serums were collected on day 14 after immunization. A 0.1 M sodium bicarbonate buffer (pH 9.3) containing 10 μg/ml of OVA was added to 96-well U-bottomed polyvinyl microplates (Falcon, Becton Dickinson & Co., Oxnard, CA, USA) and incubated overnight at 4°C. After washing with PBS containing 0.05% Tween-20 (PBST), the plates were blocked for 1 h with 2% BSA solution. After washing with PBST again, 50 µl of serum was added to each well and incubated for 2 h at 37°C. After washing with PBST, HRP-conjugated anti-isotype specific Abs (Southern Biotechnology, Birmingham, AL, USA) were added to each well and incubated for 1 h. Plates were then washed, and TMB substrate (BD Biosciences) was added. After incubation for 5–10 min, 0.05 M sulfuric acid was added to each well, and the colorimetric reaction was measured at 450 nm with an absorbance microplate reader (BioTek Instruments, Inc., Winooski, VT, USA).
Statistical analysis
Statistical differences between experimental groups were determined by analysis of variances. All p values were calculated using unpaired 2-tailed Student's t-tests to consider statistical significance.
RESULTS AND DISCUSSION
Effects of Sesn2 and AMPK on IL-4-induced IgE class switching in B cells
To determine whether B cells express Sesn2, we performed RT-PCR with mRNAs from mouse B cell lines (L10A6.2 and A20.3), purified mouse splenic B cells, and a mouse macrophage cell line, RAW 264.7–used as a positive control for Sesn2 expression (Supplementary Fig. 1) (9). Sesn2 was expressed in the B cell lines as well as the RAW 264.7 cell line. Mouse splenic B cells also expressed Sesn2 mRNA, and this was further enhanced by LPS stimulation.
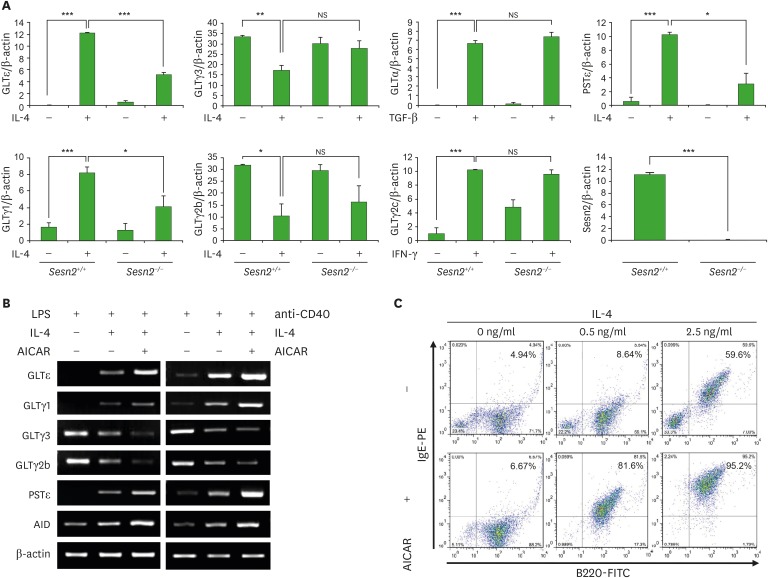
Next, to evaluate the effect of Sesn2 on Ig CSR by B cells, we performed RT-PCR for GLTs and PSTs with cytokines-stimulated splenocytes from Sesn2 knockout (Sesn2−/−) and wild-type (Sesn2+/+) mice. As shown in Fig. 1A (Supplementary Fig. 2), IL-4-induced GLTε expression was reduced in Sesn2−/−. IL-4-induced GLTγ1 was also reduced in Sesn2−/−, suggesting that Sesn2 affects sequential switching to IgE through IgG1 by IL-4 because IL-4 induces both GLTγ1 and GLTε transcription and successive IgM→IgG1→IgE CSR (23,24,25,26). Conversely, Sesn2 knockout did not decrease other stimuli-induced GLTs (GLTγ3, GLTγ2b, GLTα, and GLTγ2c). Importantly, IL-4-induced PSTε expression was reduced in Sesn2−/− (Fig. 1A). In addition, we performed a luciferase reporter assay to investigate the effect of Sesn2 on GLTε promoter activity (Supplementary Fig. 3). Sesn2 overexpression enhanced IL-4-induced GLTε promoter activity as well as GLTγ1 activity but not TGF-β1-induced GLTα activity. These results indicate that Sesn2 selectively mediates the IgE CSR by enhancing IL-4-induced GLTε transcription.
Figure 1. Effects of Sesn2 and AMPK on Ig class switching. (A) Splenocytes (1 × 106 cells/ml) from Sesn2+/+ or Sesn2−/− mice were stimulated with TGF-β1 (0.2 ng/ml), IL-4 (0.5 ng/ml), and IFN-γ (10 ng/ml) in the presence of LPS (12.5 µg/ml). After 2.5 days of culture, RNAs were isolated and the levels of GLTs and PSTε were measured by RT-PCR. Graphs indicate relative cDNA levels normalized to β-actin cDNA expression using ImageJ (National Institutes of Health, Bethesda, MD, USA) analysis. (B, C) Splenocytes were stimulated with IL-4 (0.5 or 2.5 ng/ml), and AICAR (200 µM) in the presence of LPS (12.5 µg/ml) or anti-CD40 (2 µg/ml). After 2.5 days of culture, RNAs were isolated and the levels of GLTs and AID mRNA were measured by RT-PCR (B). After 4 days of culture, cells were stained with anti-IgE-PE and anti-B220-FITC, and surface Ig expression was analyzed by flow cytometry (C). Data presented are the means±SEM from three independent experiments.
NS, not significant.
*p<0.05, **p<0.01, ***p<0.001.
mTORC1 negatively constrains STAT6 signaling in Th2 cell differentiation (15). In many cell types, Sesn2 activates AMPK and activated AMPK signaling inhibits mTORC1 activation (1,6,7). To determine the roles of AMPK and mTORC1 in IL-4-induced GLTε expression, we tested the effects of an AMPK activator (AICAR) and the mTORC1 inhibitors (rapamycin and everolimus) on the GLTs and PSTε expression using in vitro cell culture. As shown in Fig. 1B, AICAR selectively enhanced IL-4-induced GLTε/GLTγ1 and PSTε expression. AICAR also increased IL-4-induced AID expression. In addition, AICAR promoted IL-4-induced sIgE expression (Fig. 1C). However, rapamycin and everolimus did not enhance IL-4-induced-GLTε expression without compromising cell viability (Supplementary Fig. 4), suggesting that mTORC1 does not negatively regulate IgE class switching in B cells.
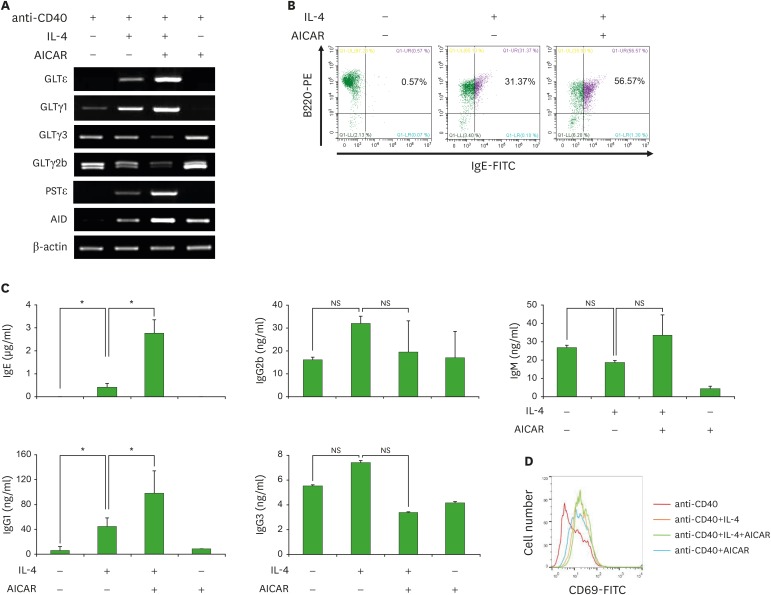
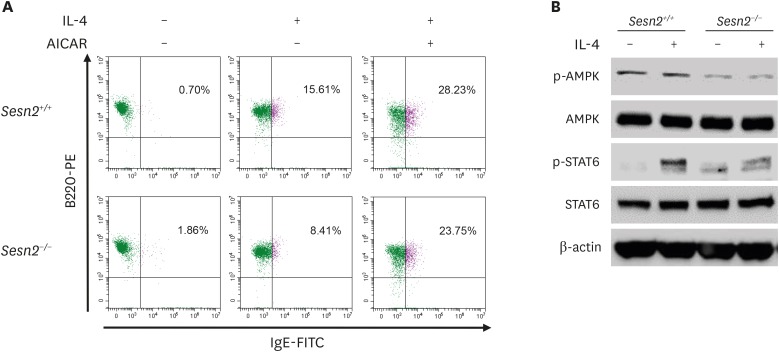
To further explore the role of AICAR in B cell IgE class switching and IgE production, we assessed GLTε transcription, sIgE expression, and IgE production in IL-4-stimulated mouse splenic B cells in the presence or absence of AICAR (Fig. 2). As shown in Fig. 2A, AICAR selectively increased IL-4-induced GLTε/GLTγ1 and PSTε expression. In addition, AICAR further increased the proportion of IL-4-induced sIgE+ B cells and the production of IL-4-induced IgE by B cells (Fig. 2B and C). We examined the expression of surface CD69 (a marker of lymphocyte activation), to determine the effect of AICAR on B cell activation (Fig. 2D). IL-4 and AICAR enhanced CD69 expression in the presence of anti-CD40, but AICAR did not further increase anti-CD40/IL-4-induced CD69 expression. To verify the role of AMPK in Sesn2-mediated IgE CSR, we tested the effect of AICAR and the activation of AMPK in Sesn2−/− B cells (Fig. 3). IL-4-induced sIgE expression was reduced in Sesn2−/− B cells (15.61%→8.41%), and AICAR rescued the reduction (8.41%→23.75%) (Fig. 3A). AMPK activation (phosphorylated AMPK; p-AMPK) was reduced in Sesn2−/− B cells (Fig. 3B).
Figure 2. Effect of AICAR on GLTε transcription, surface IgE expression, and IgE production by anti-CD40-stimulated B cells. Purified mouse B cells (1×106 cells/ml) were stimulated with IL-4 (1 or 2.5 or 5 ng/ml) and AICAR (200 µM) in the presence of anti-CD40 (2 µg/ml). (A) After 2.5 days of culture, RNAs were isolated and the levels of GLTs and AID mRNA were measured by RT-PCR. (B) After 4 days of culture, cells were stained with anti-IgE-FITC and anti-B220-PE, and surface Ig expression was analyzed by flow cytometry. (C) After 7 days of culture, supernatants were harvested, and the levels of Ig production were measured by isotype-specific ELISA. Data presented are the means±SEM from three independent experiments. (D) After 4 days of culture, cells were stained with anti-CD69-FITC and B cell activation was analyzed by flow cytometry.
NS, not significant.
*p<0.05.
Figure 3. Effects of Sesn2 and AICAR on the expression of surface IgE and the activation of STAT6 and AMPK. (A) Purified B cells (1×106 cells/ml) from Sesn2+/+ or Sesn2−/− mice were stimulated with IL-4 (2.5 ng/ml) and AICAR (200 µM) in the presence of anti-CD40 (2 µg/ml). After 4 days of culture, cells were stained with anti-IgE-FITC and anti-B220-PE, and surface Ig expression was analyzed by flow cytometry. (B) Purified B cells (5×106 cells/ml) from Sesn2+/+ or Sesn2−/− mice were stimulated with IL-4 (100 ng/ml) for 30 min in the presence of anti-CD40 (2 µg/ml). Cells were harvested to measure expression of the indicated proteins.
Collectively, these results indicate that the Sens2-AMPK signaling pathway may positively regulate IL-4-induced IgE class switching and IgE production by B cells. However, AMPK-independent AICAR effects were also reported (27). Therefore, the AMPK-mediated IgE response needs to be further proved by reagents that demonstrate absolute specificity for AMPK, since AICAR could potentially induce other signal pathways.
The transcription factors, STAT6 (which is essential for the induction of IL-4-specific gene expression) and NFIL3, positively regulate mouse GLTε promoter activity (17), while BCL6 negatively regulates mouse GLTε promoters (28). In addition, CREB is activated by AMPK and activates NFIL3 in various cells (29,30). SOCS1 suppresses STAT6 in leukocytes (31). To elucidate the mechanism by which AICAR regulates IL-4-induced GLTε transcription, we investigated the effect of AICAR on mRNA expression of the transcription factors, STAT6, NFIL3, BCL6, CREB, and SOCS1. AICAR did not affect the mRNA expression of the transcription factors in anti-CD40/IL-4-activated B cells (Supplementary Fig. 5A). Sesn2 facilitates the nuclear translocation of the stress responsive transcription factor Nrf2 in liver cells (6), and AMPK phosphorylates Nrf2 in HepG2 cells and HEK293 cells (32). AICAR also exhibited no effect on Nrf2 mRNA expression in anti-CD40/IL-4-stimulated B cells (Supplementary Fig. 5A). Nrf2 binds to antioxidant response elements (ARE, RTGAYnnnGC) (33,34) that mediate the transcriptional induction of genes (6). Since the GLTε promoter exhibits putative AREs (Supplementary Fig. 5B), we examined the effect of Nrf2 overexpression on GLTε promoter activity. As shown in Supplementary Fig. 5C, overexpression of Nrf2 decreased rather than increased overall GLTε promoter activity with or without IL-4 in B cells. Next, we investigated the effect of Sesn2 on activation of STAT6 in IL-4-stimulated B cells. As shown in Fig. 3B, IL-4-induced phosphorylated STAT6 was reduced in Sesn2−/− B cells. These results suggest that STAT6 is a crucial transcriptional regulator of Sesn2-AMPK signaling in IL-4-induced GLTε transcription. Researchers reported that the activation of p38 and JNK is required for IgE CSR (35,36), in addition to the finding that AMPK activates p38 and JNK (37,38). These facts raise the possibility that p38 or JNK can act downstream of Sesn2-AMPK signaling. Further studies are needed to elucidate the downstream regulators of the Sesn2-AMPK pathway in IL-4-induced GLTε transcription and IgE class switching.
OVA-specific IgE production in OVA-challenged Sesn2−/− mice
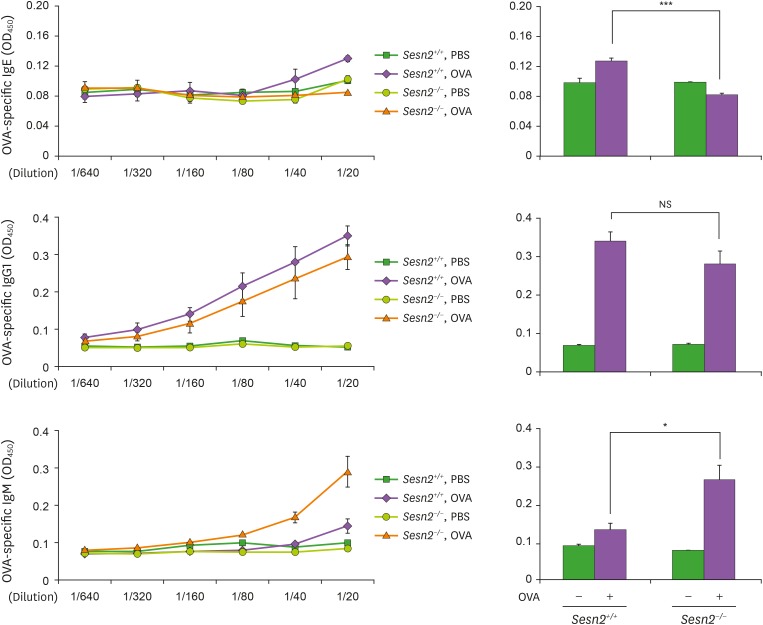
Our current findings indicate that Sesn2 is a positive regulator of IgE class switching and IgE production in cell culture, at least in vitro. To determine the role of Sesn2 in IgE production in vivo, we monitored IgE concentration in serum from OVA allergen-challenged Sesn2−/− mice (Fig. 4). Immunization with OVA induced OVA-specific IgE, IgG1, and IgM production in Sesn2 wild-type (Sesn2+/+) mice. In Sesn2−/− mice, OVA-specific IgE concentration was reduced, while IgM concentration was increased. Therefore, we can conclude that Sesn2 is involved in allergen-induced IgE production.
Figure 4. OVA-specific IgE production in serum is reduced in Sesn2−/− mice. Sesn2+/+ and Sesn2−/− mice were immunized with OVA following the protocol described in the Materials and Methods section. Control animals received PBS only. Serum was collected at 7 days after the last immunization, and OVA-specific Ig levels in the indicated serum dilutions were determined by ELISA. Data presented are the means±SEM from three mice from each group (right panel). Sesn2+/+ mice (n=3), Sesn2−/− mice (n=3).
NS, not significant.
*p<0.05, ***p<0.001.
In conclusion, our findings suggest that Sesn2 and AMPK are positive regulators of IL-4-induced GLTε transcription, resulting in IgE class switching and IgE production in B cells. This study reveals not only an important regulatory role of the Sesn2-AMPK signaling pathway in the B cell IgE response but also the scope of the challenge in the development of therapeutic strategies that target Sesn2 and AMPK in IgE-mediated allergic diseases.
ACKNOWLEDGEMENTS
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science, and Technology (MEST) (NRF-2016R1D1A1B04935588) and the Priority Research Centers Program through the NRF funded by the MEST (NRF-2017R1A6A1A03015713). This work was also supported in part by Konyang University Myunggok Research Fund of 2016.
Footnotes
Conflict of Interest: The author declares no potential conflicts of interest.
- Conceptualization: Park SR.
- Data curation: Shim YS, Park SR.
- Formal analysis: Shim YS, Park SR.
- Funding acquisition: Park SR.
- Investigation: Shim YS, Lee S, Park HW, Park SR.
- Resources: Park SR, Park HW.
- Supervision: Park SR.
- Writing - original draft: Shim YS, Park SR.
SUPPLEMENTARY MATERIALS
RT-PCR primers
Mouse B cells express Sesn2. Total RNA were isolated from the indicated cells, and the levels of Sesn2 mRNA were measured by RT-PCR.
GLTs and PSTε expression in LPS-stimulated Sesn2−/− splenocytes. Splenocytes (1 × 106 cells/ml) from Sesn2+/+ or Sesn2−/− mice were stimulated with TGF-β1 (0.2 ng/ml), IL-4 (0.5 ng/ml), and IFN-γ (10 ng/ml) in the presence of LPS (12.5 μg/ml). After 2.5 days of culture, RNAs were isolated and the levels of GLTs and PSTε were measured by semi-quantitative RT-PCR.
Effect of Sesn2 overexpression on GLTε promoter activity. Reporter plasmids (GLTε, GLTγ1, and GLTα promoter reporters) (15 μg) and pLU-Sesn2 (5 μg) were transfected into B cell lines (L10A6.2 and CH12F3-2A). The transfected cells were stimulated with IL-4 (10 ng/ml) or TGF-β1 (0.2 ng/ml) in the presence of LPS (12.5 μg/ml) or anti-CD40 (2 μg/ml), and luciferase activity was assayed 16 h later. Each value of luciferase activity was normalized by β-galactosidase activity. Data represent mean±SEM luciferase activities from three independent transfections.
Effects of rapamycin and everolimus on GLTε expression and cell viability. (A) Purified mouse B cells (1×106 cells/ml) were stimulated with IL-4 (1 ng/ml), AICAR (200 μM), rapamycin (2.5 nM), and everolimus (1.25 nM) in the presence of anti-CD40 (2 μg/ml). After 2.5 days of culture, RNAs were isolated and the levels of GLTε were measured by RT-PCR. (B) Cell viability was measured by Ez-Cytox assay at 2 and 3 days of culture.
Effect of AICAR on the expression of GLTε-related transcription factors, and effect of Nrf2 overexpression on GLTε promoter activity. (A) Purified mouse B cells (1×106 cells/ml) were stimulated with IL-4 (0.5 ng/ml), AICAR (200 μM) in the presence of anti-CD40 (2 μg/ml). After 2.5 days of culture, RNAs were isolated and the levels of transcription factor mRNAs were measured by RT-PCR. (B) Sequences (−150 to +100) of GLTε promoter. pARE, putative antioxidant response element; IL-4 RE, IL-4 responsive element; STAT6, STAT6 binding site. (C) Reporter plasmid (GLTε) (15 μg) and pcDNA3-Myc3-Nrf2 (15 μg) were transfected into L10A6.2 and the cells were stimulated with IL-4 (10 ng/ml) in the presence of LPS (12.5 μg/ml) or anti-CD40 (0.2 μg/ml). After 16 h, the promoter activities were measured by luciferase assay. Each value of luciferase activity was normalized by β-galactosidase activity. Data represent mean±SEM luciferase activities from three independent transfections.
References
- 1.Sanli T, Linher-Melville K, Tsakiridis T, Singh G. Sestrin2 modulates AMPK subunit expression and its response to ionizing radiation in breast cancer cells. PLoS One. 2012;7:e32035. doi: 10.1371/journal.pone.0032035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen SD, Yang JL, Lin TK, Yang DI. Emerging roles of sestrins in neurodegenerative diseases: counteracting oxidative stress and beyond. J Clin Med. 2019;8:e1001. doi: 10.3390/jcm8071001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang LX, Zhu XM, Yao YM. Sestrin2: its potential role and regulatory mechanism in host immune response in diseases. Front Immunol. 2019;10:2797. doi: 10.3389/fimmu.2019.02797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sánchez-Álvarez M, Strippoli R, Donadelli M, Bazhin AV, Cordani M. Sestrins as a therapeutic bridge between ROS and autophagy in cancer. Cancers (Basel) 2019;11:e1415. doi: 10.3390/cancers11101415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang M, Xu Y, Liu J, Ye J, Yuan W, Jiang H, Wang Z, Jiang H, Wan J. Recent insights into the biological functions of sestrins in health and disease. Cell Physiol Biochem. 2017;43:1731–1741. doi: 10.1159/000484060. [DOI] [PubMed] [Google Scholar]
- 6.Pasha M, Eid AH, Eid AA, Gorin Y, Munusamy S. Sestrin2 as a novel biomarker and therapeutic target for various diseases. Oxid Med Cell Longev. 2017;2017:3296294. doi: 10.1155/2017/3296294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bodmer D, Levano-Huaman S. Sesn2/AMPK/mTOR signaling mediates balance between survival and apoptosis in sensory hair cells under stress. Cell Death Dis. 2017;8:e3068. doi: 10.1038/cddis.2017.457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang X, Liu W, Zhuang D, Hong S, Chen J. Sestrin2 and sestrin3 suppress NK-92 cell-mediated cytotoxic activity on ovarian cancer cells through AMPK and mTORC1 signaling. Oncotarget. 2017;8:90132–90143. doi: 10.18632/oncotarget.21487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Essler S, Dehne N, Brüne B. Role of sestrin2 in peroxide signaling in macrophages. FEBS Lett. 2009;583:3531–3535. doi: 10.1016/j.febslet.2009.10.017. [DOI] [PubMed] [Google Scholar]
- 10.Kim MJ, Bae SH, Ryu JC, Kwon Y, Oh JH, Kwon J, Moon JS, Kim K, Miyawaki A, Lee MG, et al. SESN2/sestrin2 suppresses sepsis by inducing mitophagy and inhibiting NLRP3 activation in macrophages. Autophagy. 2016;12:1272–1291. doi: 10.1080/15548627.2016.1183081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kim MG, Yang JH, Kim KM, Jang CH, Jung JY, Cho IJ, Shin SM, Ki SH. Regulation of Toll-like receptor-mediated Sestrin2 induction by AP-1, Nrf2, and the ubiquitin-proteasome system in macrophages. Toxicol Sci. 2015;144:425–435. doi: 10.1093/toxsci/kfv012. [DOI] [PubMed] [Google Scholar]
- 12.Hu HJ, Shi ZY, Lin XL, Chen SM, Wang QY, Tang SY. Upregulation of Sestrin2 expression protects against macrophage apoptosis induced by oxidized low-density lipoprotein. DNA Cell Biol. 2015;34:296–302. doi: 10.1089/dna.2014.2627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yang JH, Kim KM, Kim MG, Seo KH, Han JY, Ka SO, Park BH, Shin SM, Ku SK, Cho IJ, et al. Role of sestrin2 in the regulation of proinflammatory signaling in macrophages. Free Radic Biol Med. 2015;78:156–167. doi: 10.1016/j.freeradbiomed.2014.11.002. [DOI] [PubMed] [Google Scholar]
- 14.Lanna A, Gomes DC, Muller-Durovic B, McDonnell T, Escors D, Gilroy DW, Lee JH, Karin M, Akbar AN. A sestrin-dependent Erk-Jnk-p38 MAPK activation complex inhibits immunity during aging. Nat Immunol. 2017;18:354–363. doi: 10.1038/ni.3665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chi H. Regulation and function of mTOR signalling in T cell fate decisions. Nat Rev Immunol. 2012;12:325–338. doi: 10.1038/nri3198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Geha RS, Jabara HH, Brodeur SR. The regulation of immunoglobulin E class-switch recombination. Nat Rev Immunol. 2003;3:721–732. doi: 10.1038/nri1181. [DOI] [PubMed] [Google Scholar]
- 17.Tong P, Wesemann DR. Molecular mechanisms of IgE class switch recombination. Curr Top Microbiol Immunol. 2015;388:21–37. doi: 10.1007/978-3-319-13725-4_2. [DOI] [PubMed] [Google Scholar]
- 18.Vercelli D, Geha RS. Regulation of isotype switching. Curr Opin Immunol. 1992;4:794–797. doi: 10.1016/0952-7915(92)90064-l. [DOI] [PubMed] [Google Scholar]
- 19.Li SC, Rothman PB, Zhang J, Chan C, Hirsh D, Alt FW. Expression of I mu-C gamma hybrid germline transcripts subsequent to immunoglobulin heavy chain class switching. Int Immunol. 1994;6:491–497. doi: 10.1093/intimm/6.4.491. [DOI] [PubMed] [Google Scholar]
- 20.Lee SH, Park SR. Toll-like receptor 1/2 agonist Pam3CSK4 suppresses lipopolysaccharide-driven IgG1 production while enhancing IgG2a production by B cells. Immune Netw. 2018;18:e10. doi: 10.4110/in.2018.18.e10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Seo BS, Park HY, Yoon HK, Yoo YC, Lee J, Park SR. Dectin-1 agonist selectively induces IgG1 class switching by LPS-activated mouse B cells. Immunol Lett. 2016;178:114–121. doi: 10.1016/j.imlet.2016.08.010. [DOI] [PubMed] [Google Scholar]
- 22.Park SR, Lee JH, Kim PH. Smad3 and Smad4 mediate transforming growth factor-β1-induced IgA expression in murine B lymphocytes. Eur J Immunol. 2001;31:1706–1715. doi: 10.1002/1521-4141(200106)31:6<1706::aid-immu1706>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- 23.Mao CS, Stavnezer J. Differential regulation of mouse germline Ig γ1 and ε promoters by IL-4 and CD40. J Immunol. 2001;167:1522–1534. doi: 10.4049/jimmunol.167.3.1522. [DOI] [PubMed] [Google Scholar]
- 24.Stavnezer J. Molecular processes that regulate class switching. Curr Top Microbiol Immunol. 2000;245:127–168. doi: 10.1007/978-3-642-59641-4_6. [DOI] [PubMed] [Google Scholar]
- 25.Mandler R, Finkelman FD, Levine AD, Snapper CM. IL-4 induction of IgE class switching by lipopolysaccharide-activated murine B cells occurs predominantly through sequential switching. J Immunol. 1993;150:407–418. [PubMed] [Google Scholar]
- 26.Wu LC, Zarrin AA. The production and regulation of IgE by the immune system. Nat Rev Immunol. 2014;14:247–259. doi: 10.1038/nri3632. [DOI] [PubMed] [Google Scholar]
- 27.Santidrián AF, González-Gironès DM, Iglesias-Serret D, Coll-Mulet L, Cosialls AM, de Frias M, Campàs C, González-Barca E, Alonso E, Labi V, et al. AICAR induces apoptosis independently of AMPK and p53 through up-regulation of the BH3-only proteins BIM and NOXA in chronic lymphocytic leukemia cells. Blood. 2010;116:3023–3032. doi: 10.1182/blood-2010-05-283960. [DOI] [PubMed] [Google Scholar]
- 28.Harris MB, Chang CC, Berton MT, Danial NN, Zhang J, Kuehner D, Ye BH, Kvatyuk M, Pandolfi PP, Cattoretti G, et al. Transcriptional repression of Stat6-dependent interleukin-4-induced genes by BCL-6: specific regulation of iepsilon transcription and immunoglobulin E switching. Mol Cell Biol. 1999;19:7264–7275. doi: 10.1128/mcb.19.10.7264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.McGee SL, Hargreaves M. AMPK-mediated regulation of transcription in skeletal muscle. Clin Sci (Lond) 2010;118:507–518. doi: 10.1042/CS20090533. [DOI] [PubMed] [Google Scholar]
- 30.MacGillavry HD, Stam FJ, Sassen MM, Kegel L, Hendriks WT, Verhaagen J, Smit AB, van Kesteren RE. NFIL3 and cAMP response element-binding protein form a transcriptional feedforward loop that controls neuronal regeneration-associated gene expression. J Neurosci. 2009;29:15542–15550. doi: 10.1523/JNEUROSCI.3938-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tamiya T, Kashiwagi I, Takahashi R, Yasukawa H, Yoshimura A. Suppressors of cytokine signaling (SOCS) proteins and JAK/STAT pathways: regulation of T-cell inflammation by SOCS1 and SOCS3. Arterioscler Thromb Vasc Biol. 2011;31:980–985. doi: 10.1161/ATVBAHA.110.207464. [DOI] [PubMed] [Google Scholar]
- 32.Joo MS, Kim WD, Lee KY, Kim JH, Koo JH, Kim SG. AMPK facilitates nuclear accumulation of Nrf2 by phosphorylating at serine 550. Mol Cell Biol. 2016;36:1931–1942. doi: 10.1128/MCB.00118-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jyrkkänen HK, Kuosmanen S, Heinäniemi M, Laitinen H, Kansanen E, Mella-Aho E, Leinonen H, Ylä-Herttuala S, Levonen AL. Novel insights into the regulation of antioxidant-response-element-mediated gene expression by electrophiles: induction of the transcriptional repressor BACH1 by Nrf2. Biochem J. 2011;440:167–174. doi: 10.1042/BJ20110526. [DOI] [PubMed] [Google Scholar]
- 34.Erickson AM, Nevarea Z, Gipp JJ, Mulcahy RT. Identification of a variant antioxidant response element in the promoter of the human glutamate-cysteine ligase modifier subunit gene. Revision of the ARE consensus sequence. J Biol Chem. 2002;277:30730–30737. doi: 10.1074/jbc.M205225200. [DOI] [PubMed] [Google Scholar]
- 35.Zhang K, Zhang L, Zhu D, Bae D, Nel A, Saxon A. CD40-mediated p38 mitogen-activated protein kinase activation is required for immunoglobulin class switch recombination to IgE. J Allergy Clin Immunol. 2002;110:421–428. doi: 10.1067/mai.2002.126382. [DOI] [PubMed] [Google Scholar]
- 36.Jabara HH, Geha RS. Jun N-terminal kinase is essential for CD40-mediated IgE class switching in B cells. J Allergy Clin Immunol. 2005;115:856–863. doi: 10.1016/j.jaci.2005.01.020. [DOI] [PubMed] [Google Scholar]
- 37.Chaube B, Malvi P, Singh SV, Mohammad N, Viollet B, Bhat MK. AMPK maintains energy homeostasis and survival in cancer cells via regulating p38/PGC-1α-mediated mitochondrial biogenesis. Cell Death Dis. 2015;1:15063. doi: 10.1038/cddiscovery.2015.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Meisse D, Van de Casteele M, Beauloye C, Hainault I, Kefas BA, Rider MH, Foufelle F, Hue L. Sustained activation of AMP-activated protein kinase induces c-Jun N-terminal kinase activation and apoptosis in liver cells. FEBS Lett. 2002;526:38–42. doi: 10.1016/s0014-5793(02)03110-1. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
RT-PCR primers
Mouse B cells express Sesn2. Total RNA were isolated from the indicated cells, and the levels of Sesn2 mRNA were measured by RT-PCR.
GLTs and PSTε expression in LPS-stimulated Sesn2−/− splenocytes. Splenocytes (1 × 106 cells/ml) from Sesn2+/+ or Sesn2−/− mice were stimulated with TGF-β1 (0.2 ng/ml), IL-4 (0.5 ng/ml), and IFN-γ (10 ng/ml) in the presence of LPS (12.5 μg/ml). After 2.5 days of culture, RNAs were isolated and the levels of GLTs and PSTε were measured by semi-quantitative RT-PCR.
Effect of Sesn2 overexpression on GLTε promoter activity. Reporter plasmids (GLTε, GLTγ1, and GLTα promoter reporters) (15 μg) and pLU-Sesn2 (5 μg) were transfected into B cell lines (L10A6.2 and CH12F3-2A). The transfected cells were stimulated with IL-4 (10 ng/ml) or TGF-β1 (0.2 ng/ml) in the presence of LPS (12.5 μg/ml) or anti-CD40 (2 μg/ml), and luciferase activity was assayed 16 h later. Each value of luciferase activity was normalized by β-galactosidase activity. Data represent mean±SEM luciferase activities from three independent transfections.
Effects of rapamycin and everolimus on GLTε expression and cell viability. (A) Purified mouse B cells (1×106 cells/ml) were stimulated with IL-4 (1 ng/ml), AICAR (200 μM), rapamycin (2.5 nM), and everolimus (1.25 nM) in the presence of anti-CD40 (2 μg/ml). After 2.5 days of culture, RNAs were isolated and the levels of GLTε were measured by RT-PCR. (B) Cell viability was measured by Ez-Cytox assay at 2 and 3 days of culture.
Effect of AICAR on the expression of GLTε-related transcription factors, and effect of Nrf2 overexpression on GLTε promoter activity. (A) Purified mouse B cells (1×106 cells/ml) were stimulated with IL-4 (0.5 ng/ml), AICAR (200 μM) in the presence of anti-CD40 (2 μg/ml). After 2.5 days of culture, RNAs were isolated and the levels of transcription factor mRNAs were measured by RT-PCR. (B) Sequences (−150 to +100) of GLTε promoter. pARE, putative antioxidant response element; IL-4 RE, IL-4 responsive element; STAT6, STAT6 binding site. (C) Reporter plasmid (GLTε) (15 μg) and pcDNA3-Myc3-Nrf2 (15 μg) were transfected into L10A6.2 and the cells were stimulated with IL-4 (10 ng/ml) in the presence of LPS (12.5 μg/ml) or anti-CD40 (0.2 μg/ml). After 16 h, the promoter activities were measured by luciferase assay. Each value of luciferase activity was normalized by β-galactosidase activity. Data represent mean±SEM luciferase activities from three independent transfections.