An increased risk of B cell lymphomas in patients with myeloproliferative neoplasm (MPN) treated with Janus kinase (JAK) inhibitors has recently been explored.1–3 In light of contradictory results, it is highly important to assess whether JAK inhibitors might be associated with a higher risk of lymphoma, notably in patients with pre-existing B cell clones that can escape immunosurveillance after exposure to JAK inhibitors.
This situation is very similar to what is observed in patients with autoimmune diseases, especially rheumatoid arthritis (RA). B cell clones are present in patients with RA, and dominant BCR clones in the peripheral blood predict the onset of the disease in at-risk individuals.4 In addition, RA patients have a 2–3-fold increased risk of lymphoma, and the impact of immunosuppressive drugs remains unclear. Tofacitinib, a JAK 1/3 inhibitor, has been approved for use in RA and, its use is increasing. To date, lymphomas have not been found to occur significantly more frequently in RA patients treated with tofacitinib. However, the frequency of their association with Epstein–Barr virus (EBV) has been found to be slightly increased compared to what is expected in RA patients.5 Thus the hypothesis is that JAK inhibitors might alter the control of B cell clones present in some MPN or RA patients and might promote the occurrence of lymphomas in these at-risk patients. In this context, we aimed to better understand the cellular targets of tofacitinib, focusing on natural killer (NK) cells, which are cellular actors of antitumor immunosurveillance, to anticipate the safety profile of the drug. We assessed the impact of tofacitinib on the phenotype and function of NK cells. NK cells isolated from healthy blood donors were cultured in interleukin (IL)-2 in the presence of tofacitinib at 3 concentrations (10, 50, and 100 nM) or control for 6 days. The 50 and 100 nM doses of tofacitinib in vitro were equivalent to the 5 and 10 mg doses, respectively, used BID (twice daily) for human RA, and 10 nM tofacitinib represented a subtherapeutic dose for RA treatment.6 At the end of culture, NK cells were phenotyped. First, we observed that tofacitinib was not cytotoxic to NK cells regardless of the concentration. However, tofacitinib significantly decreased IL-2-induced activation, as assessed by CD69 expression. In addition, exposure to tofacitinib (50 and 100 nM) led to decreased expression of natural cytotoxic receptors, including NKp44 and NKp30 but not NKp46. NKG2D expression was also decreased following exposure to tofacitinib at the highest concentration. Finally, 100 nM tofacitinib blocked the maturation of NK cells, as assessed by decreased CD57 and Tim-3 expression.
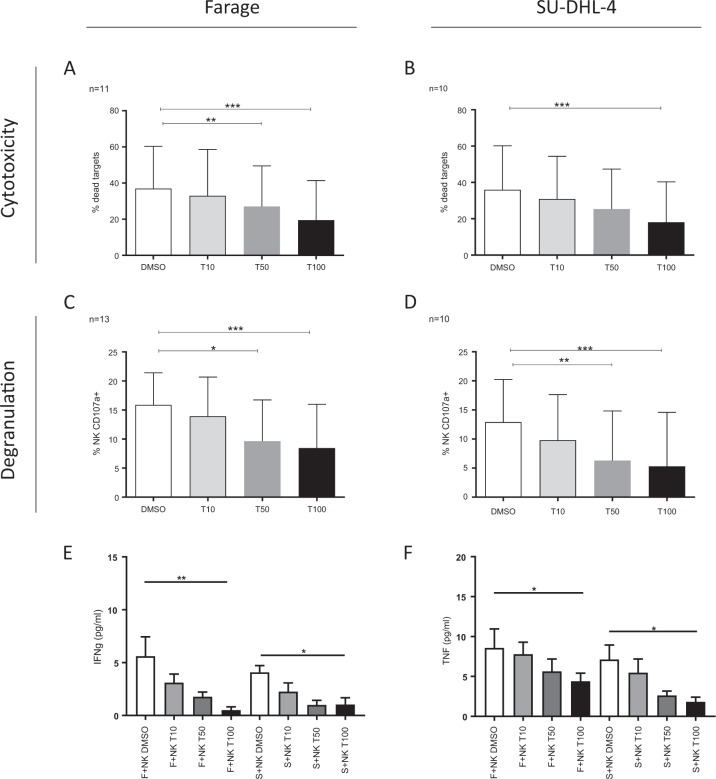
Then, to test the functional consequences of these alterations, we cocultured NK cells exposed to tofacitinib or control (dimethyl sulfoxide) with B cell lymphoma cells. We tested one lymphoma cell line positive for EBV (Farage) and one lymphoma cell line negative for EBV (SU-DHL-4). We observed decreased capacity for lysis of B cell lymphoma targets by NK cells exposed to tofacitinib (Fig. 1a, b). In addition, we observed decreased capacity for degranulation (Fig. 1c, d) after tofacitinib exposure. NK cells can mediate tumor rejection via cytotoxicity and the production of cytokines such as interferon-γ and tumor necrosis factor.7 These cytokines were decreased in the supernatant of cocultures exposed to tofacitinib (Fig. 1e, f).
Fig. 1.
Antilymphoma activity of NK cells exposed to tofacitinib. After a 6-day culture with tofacitinib or control, NK cells were cocultured with B-NHL (Farage (EBV-positive) and SU-DHL-4 (EBV-negative)) cells. The percentage of lysis of Farage (a) and SU-DHL-4 (b) cells is shown (upper panel). The percentage of NK-mediated lysis was calculated as the percentage of dead target cells under specific conditions − the percentage of dead target cells cultured alone. The percentage of degranulating NK cells (CD107a-positive) is shown in the lower panel (c Farage cells, d SUDHL4 cells). IFNγ (e) and TNF (f) secretion was assessed by ELISA. The results are presented as mean and SD. Mann–Whitney t test. *p < 0.05, **p < 0.01, ***p < 0.001
In summary, these experiments demonstrated that exposure to tofacitinib might impair NK cell activation and lymphoma cell killing efficacy by decreasing their capacity for degranulation and cytokine secretion. The inhibition was dose dependent and was observed at concentrations equivalent to that used in RA (5 or 10 mg BID (steady-state plasma concentration)).6
Our results are consistent with those described for ruxolitinib (a JAK 1/2 inhibitor used in MPN) since it has been demonstrated that NK cells exposed to ruxolitinib are less activated and display reduced effector functions, including degranulation and cytokine secretion. In light of these observations, further studies are needed, and caution should be taken in at-risk patients. Lymphoma occurrence is a rare event that is difficult to detect in short-term follow-up.5 Thus long-term evaluation of RA and MPN patients treated with JAK inhibitors based on randomized clinical trials and population-based observational research remains mandatory.
Acknowledgements
Grants were obtained from Fondation pour la Recherche Médicale (DEQ20150934719: Sjögren’s syndrome and autoimmunity-associated lymphomas (SAIL). Grants were also obtained from Pfizer (“Assessment of the risk of lymphoproliferative disorders associated with specific inhibition of the JAK pathway_modified,” #IIR WI199662 and “JAKinNaK: Assessment of the impact of different JAK inhibitors on NK cell functions”, #WI236322).
Author contributions
G.N., J.P., B.L., and F.T. performed the experiments; G.N. and X.M. analyzed the results, designed the research, and wrote the paper.
Competing interests
X.M. received an honorarium from Pfizer.
References
- 1.Pemmaraju N, et al. Characteristics of patients with myeloproliferative neoplasms with lymphoma, with or without JAK inhibitor therapy. Blood. 2019;133:2348–2351. doi: 10.1182/blood-2019-01-897637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Porpaczy E, et al. Aggressive B-cell lymphomas in patients with myelofibrosis receiving JAK1/2 inhibitor therapy. Blood. 2018;132:694–706. doi: 10.1182/blood-2017-10-810739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rumi E, Zibellini S. JAK inhibitors and risk of B-cell lymphomas. Blood. 2019;133:2251–2253. doi: 10.1182/blood-2019-03-900951. [DOI] [PubMed] [Google Scholar]
- 4.Tak PP, et al. Dominant B cell receptor clones in peripheral blood predict onset of arthritis in individuals at risk for rheumatoid arthritis. Ann. Rheum. Dis. 2017;76:1924–1930. doi: 10.1136/annrheumdis-2017-211351. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 5.Mariette X, Chen C, Biswas P, Kwok K, Boy MG. Lymphoma in the tofacitinib rheumatoid arthritis clinical development program. Arthritis Care Res. 2018;70:685–694. doi: 10.1002/acr.23421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dowty ME, et al. Preclinical to clinical translation of tofacitinib, a Janus kinase inhibitor, in rheumatoid arthritis. J. Pharm. Exp. Ther. 2014;348:165–173. doi: 10.1124/jpet.113.209304. [DOI] [PubMed] [Google Scholar]
- 7.Marcus A, et al. Recognition of tumors by the innate immune system and natural killer cells. Adv. Immunol. 2014;122:91–128. doi: 10.1016/B978-0-12-800267-4.00003-1. [DOI] [PMC free article] [PubMed] [Google Scholar]