Highlights
-
•
PAK1 is the major ‘pathogenic’ kinase.
-
•
Several potent PAK1 blockers developed are introduced for treatment of a wide variety of PAK1-dependent diseases including cancers and pandemic COVID-19 infection.
Abstract
To celebrate the 25th anniversary of the cloning of the first mammalian p21-activated kinases (PAKs) (RAC/CDC42-activated kinases) by Ed Manser, the first international PAK symposium was held in NYC in October 2019. Among six distinct PAKs in mammals, PAK1 is the major ‘pathogenic kinase’, the abnormal activation of which is responsible for a wide variety of diseases and disorders including cancers, ageing processes and infectious and inflammatory diseases such as pandemic coronaviral infection. Recently, for a clinical application, a few potent (highly cell-permeable and water-soluble) PAK1 blockers have been developed from natural or synthetic PAK1 blockers (triptolide, vitamin D3 and ketorolac) via a series of ‘chemical evolutions’ that boost pharmacological activities >500 times.
Amoeba PAK (myosin I heavy chain kinase)
More than four decades ago, an NIH team discovered a peculiar kinase in a soil amoeba that phosphorylated the heavy chain of an unusual myosin: a single-headed myosin (myosin I), and this phosphorylation led to a robust activation of myosin I ATPase by actin fiber (F-actin) [1]. Interestingly, myosin I is essential for amoeboid movement including phagocytosis and cell migration but not for cell division (cytokinesis), which depends on another myosin: double-headed myosin (myosin II). Curiously, the amoeba myosin I heavy-chain kinase phosphorylates the regulatory light chain of smooth muscle myosin II from mammals, leading to a robust activation of this muscle myosin II ATPase by F-actin. Therefore, if such a kinase should exist in mammals as well, the mammalian counterpart was anticipated to raise the blood pressure by triggering smooth muscle contraction along blood vessel walls.
Mammalian PAK
Eventually, 17 years later, according to an early 1994 issue of Nature [2], a similar kinase was discovered in the mammalian brain by a British scientist (Dr Ed Manser) in Singapore – and named p21-activated kinase (PAK)1 (RAC/CDC42-activated kinase 1). Because the G proteins called RAC and CDC42 were known to be activated by the oncogenic G protein RAS, which is responsible for >30% of malignant transformation of human cells (such as pancreatic, colon and lung cancers), we wondered whether PAK1 might be responsible for RAS-induced malignant transformation. Soon, Ed Manser’s team discovered another protein called PIX, which is essential for the activation of PAK1, and found that the PIX–PAK1 interaction is essential for a phenomenon called ‘membrane ruffling’ of cancer cells 3, 4. The SH3 domain of PIX binds to the Pro-rich domain of PAK1 called PAK18, consisting of 18 amino acids [4]. Because the actomyosin-based membrane ruffling is closely linked to RAS-induced malignant transformation, a team at Ludwig Institute for Cancer Research (Melbourne Branch) synthesized a highly cell-permeable peptide called WR–PAK18, in which the cell-permeable (Trp- and Arg-rich) peptide vector WR is linked to PAK18, and they confirmed that WR–PAK18 inhibits the RAS-induced malignant transformation with an IC50 below 5 μM by blocking the PIX–PAK1 interaction in cells [5]. This peptide was the very first PAK1 blocker that suppressed the growth of cancer cells selectively, without affecting normal cell growth.
PAK1-dependent diseases and disorders
Since then, by means of WR/TAT-PAK18 and many other PAK1 blockers, it has been revealed that abnormal activation of PAK1 is responsible not only for cancers but also for so many other diseases and disorders such as hypertension, diabetes (type 2), infectious and inflammatory diseases including coronavirus pandemic, neuronal diseases such Alzheimer’s disease (PD), Parkinson’s disease (PD), epilepsy, schizophrenia, depression, autism, immune-suppression, osteoporosis, hair loss and hyper-pigmentation 6, 7. Furthermore, it was found that a PAK1-deficient mutant of Caenorhabditis elegans lives 60% longer than the wild-type [6], proving that even a normal level of PAK1 is sufficient to shorten the healthy lifespan. Thus, in theory, PAK1 blockers could promote longevity as well. In fact, several natural PAK1 blockers such as propolis and melatonin significantly extend the healthy lifespan of small animals such as C. elegans and mice 6, 7. Undoubtedly, therefore, the potential market value of PAK1 blockers could be huge in the pharmaceutical and cosmetics industries. Thus, celebrating the 25th anniversary of the mammalian pathogenic kinase (PAK1) cloning, the first international PAK symposium (12 October 2019) entitled ‘Pathogenic Roles of PAK1: Anti-PAK1 Therapy Promoting the Longevity’ was held in New York City (https://www.somatopublications.com/pathogenic-roles-of-pak1-including-oncogenesis-and-ageing.pdf).
Among six distinct members of the PAK family in mammals, only PAK1 and PAK4 turned out to be pathogenic (in particular oncogenic). However, because CDC42-activated kinase 4 (PAK4) is essential for embryogenesis [6], developing PAK1 blockers has been the focus for clinical application. Although a bunch of reviews on PAK1 have been published during the past several years, most of them concern only PAK1 inhibitors that are useful only as laboratory reagents, and they are basically useless for clinical application, mainly owing to either poor water solubility or poor cell permeability.
Natural PAK1 blockers
Instead, we have focused on identifying or developing a series of clinically useful PAK1 blockers (natural or synthetic) and their chemical potentiation for the past two decades [7]. One of the natural PAK1 blockers available on the market turned out to be a bee product (alcohol-extract of beehives) called propolis. Around 1988, caffeic acid phenethyl ester (CAPE)-based propolis was found, by a team at Columbia University, to kill cancer cells selectively without any effect on normal cell growth [8]. However, the molecular mechanism underlying propolis therapy of cancers remained unknown until 2005 when CAPE or caffeic acid (CA) in propolis was revealed to downregulate RAC, just upstream of PAK1 [9]. Interestingly, although the major anticancer ingredients in propolis vastly differ from one product to another, depending on the major plant sources from which bees prepare hives, all propolis products contain PAK1 blockers such as CAPE, artepillin C (ARC) and nymphaeols, so far without any exception [10]. Moreover, unlike conventional anticancer drugs (chemotherapeutics), which cause several serious side effects such as immune suppression and hair loss, propolis caused no side effects and even promotes the immune system, hair growth and longevity 11, 12.
Highly cell-permeable synthetic PAK1 blockers
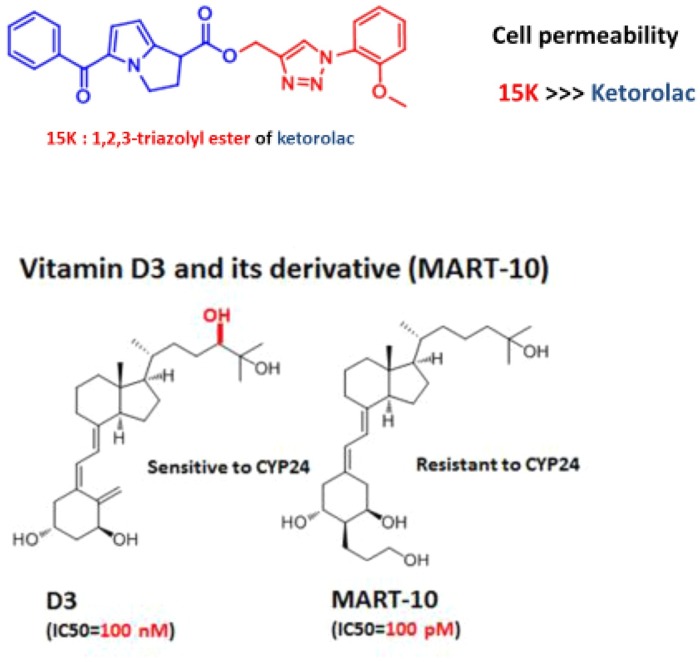
Either water insolubility or poor cell permeability (therefore low bioavailability) of propolis has been the major problem limiting worldwide clinical application. For example, ARC and CA bear the COOH moiety which blocks their free penetration through negatively charged plasma membranes of target cells, whereas CAPE is poorly water-soluble. Thus, a few years ago, our Melbourne team decided to potentiate these PAK1 blockers of propolis origin by a unique esterization with a water-soluble 1,2,3-triazolyl alcohol via click chemistry (CC), which was developed by Barry Sharpless (2001 Nobel Laureate) and his team [13]. Mainly owing to a robust increase in their cell permeability, 1,2,3-triazolyl ester of ARC (15A) and of CA (15C) are 100- and 400-times more potent in anticancer and anti-PAK1 activities than ARC and CA, respectively, with IC50s ∼200 nM [14]. Eventually from an old pain killer (ketorolac), we developed a far more effective PAK1 blocker called 15 K via CC [15] (Fig. 1 a).
Figure 1.
(a) 15 K: highly cell-permeable ester of ketorolac. (b) Vitamin D3 and its [cytochrome P450 (CYP)24-resistant] derivative MART-10. Hydroxylation of D3 at position 24 (highlighted in red) by human CYP24 inactivates D3.
Ketorolac is a synthetic racemic compound and its S-form has been long known to inhibit cyclooxygenase (COX)-2 directly, COX-2 is responsible for synthesis of prostaglandin (a pain source). Interestingly, a few years ago its R-form was found by a team at University of New Mexico to downregulate RAC, thereby blocking PAK1 with an IC50 ∼13 μM [16]. However, ketorolac bears a COOH moiety that hampers its free penetration through the negatively charged plasma membranes of target cells; and tends to cause an ulcer as well if used regularly. Thus, we decided to boost its cell permeability by esterization with 1,2,3-triazolyl alcohol via CC as well [15]. This esterization led to a great leap (>500 times) in the anti-PAK1 and anticancer properties of ketorolac [15]. The IC50 of 15 K is in the range 5–24 nM against different cancer cell lines, and it increases the heat resistance of C. elegans by nine times even at 10 nM ∼35 °C (nonpermissive temperature), inducing a heat shock gene called HSP16.2 via the longevity transcription factor FOXO, and concomitantly extends the healthy lifespan of this worm by 15–30% ∼20 °C (permissive temperature) [17]. Facing the current climate crisis namely ‘global warming or extreme heat’ (e.g., frequent summer bushfires in California and the southern half of Australia), 15 K and other PAK1 blockers could provide us with a practical alternative (chemical evolution) of the genetic evolution (knockout of PAK1 gene) for our survival of the extreme heat, which might eventually lead to extinction of the whole animal kingdom from the surface of this planet (none of the plant kingdom carries the pathogenic PAK1 gene). If we understand this correctly, once the animal kingdom (in particular the over-populated Homo sapiens) perishes, the earth would be cooled down, back to another ice age.
Notably, 90% of human pancreatic cancers are resistant to chemotherapies such as gemcitabine (GEM), and their chemoresistance is mainly the result of chemo-induced abnormal activation of PAK1 [18]. Remarkably, 15 K suppresses growth and metastasis of GEM-resistant human pancreatic cancer xenografts in mice with an IC50 below 0.1 mg/kg/day, causing no side effect in mice even with doses of 5 mg/kg/day [19].
Vitamin D3 and its potent derivative MART-10
The best-known physiological role of vitamin D3 is calcemic action promoting calcium absorption into bone tissues. However, back in 1987, one of its new roles was discovered by an Australian team: suppressing the growth of colon cancer xenografts in mice even with 5 μg/kg/day on low-calcium diet taming its calcemic effect [20]; but not melanomas which lack D3 receptor (VDR), strongly suggesting that the anticancer activity of D3 is also through VDR, a nuclear transcription factor. However, a D3 metabolite, which is hydroxylated at position 24 (Fig. 1b) by an enzyme called CYP24, was ineffective, mainly owing to its failure in activating VDR.
Two decades later, a team at Teikyo University in Tokyo developed a series of vitamin D3 derivatives. Among them is MART-10 (Fig. 1b) which is 100-times more-resistant to CYP24 [21]. To their great surprise, the anticancer activity of MART-10 (IC50 ∼100 pM) was found to be 1000-times more potent than D3 (IC50 ∼100 nM), although it is poorly calcemic [21]. Excitingly, D3 was recently found by a German team to downregulate RAC, thereby blocking the PAK1/LIM kinase/cofilin pathway and depolymerizing actin filaments [22]. In other words, there are at least two distinct D3-induced anticancer signaling pathways: one activating VDR and the other inactivating RAC/PAK1, both eventually downregulating an oncogenic transcription factor called β-catenin (Fig. 2 a). Furthermore, it was revealed that MART-10 also depolymerizes actin filaments in cancer cells, strongly suggesting that MART-10 is also a potent PAK1 blocker [23]. D3 was known to extend the healthy lifespan of C. elegans [24], and MART-10 suppresses growth and metastasis of GEM-resistant human pancreatic cancer xenografts in mice with an IC50 below 0.1 mg/kg/day [25]. Thus, 15 K and MART-10 (made in Japan) are among the most potent synthetic PAK1 blockers, and both of us are very keen to initiate their clinical trials for GEM-resistant pancreatic cancers as soon as possible with a generous financial support and/or backup from one of the keen pharmaceutical giants.
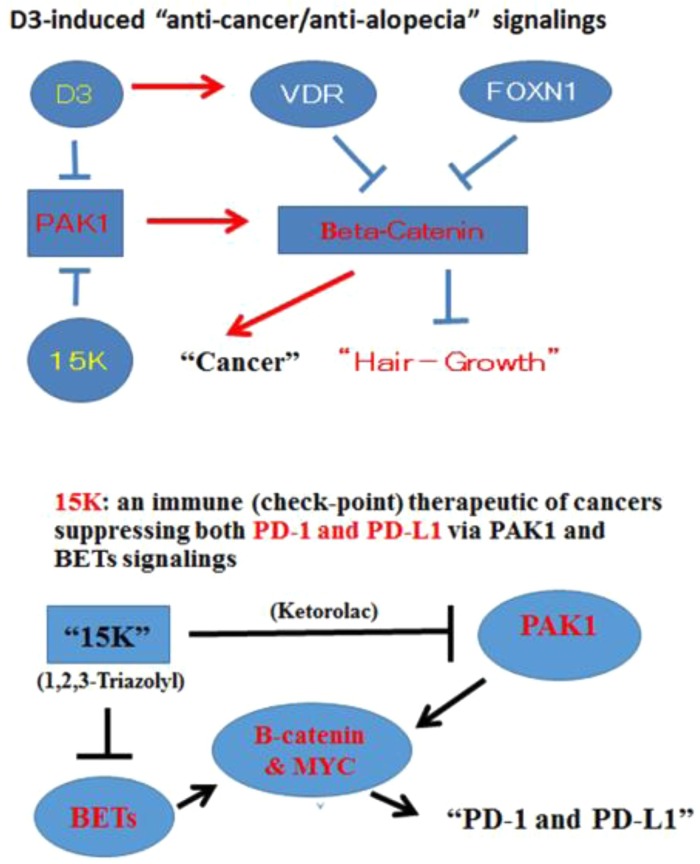
Figure 2.
(a) D3-induced anticancer/anti-alopecia signaling pathways. (b) 15 K: a potent immune (checkpoint) cancer therapeutic suppressing PD-1 and PD-L1 via p21-activated kinase (PAK)1 and bromodomain and ex-terminal domain proteins (BETs) signaling. 15 K blocks PAK1 via ketorolac and BETs through the 1,2,3-triazolyl ring, suppresses their common downstream targets (β-catenin and MYC) and eventually PD-1 and PD-L1 expression.
A third potent synthetic PAK1 blocker called minnelide, a water-soluble phosphatase-sensitive prodrug (phosphorylated form) of triptolide, was synthesized by a team at University of Minnesota led by Gunda Georg, a German organic chemist. This also inhibits RAC with an IC50 ∼30 nM [26] and is currently in clinical trials (Phase II) for cancers by Minneamrita Therapeutics in the USA. Amazingly, the phosphorylation of triptolide alone boosts its water solubility >3000 times, thereby its bioavailability as well [27].
Pathogenic roles of PAK1 in immune system and hair growth
Back in 1962, upon a spontaneous genetic mutation, a hair-less and thymus-deficient mouse strain: the nude (nu/nu) mouse, was isolated by Norman Grist in Scotland [28]. The lifespan of nu/nu mice is only 6 months, one-fifth compared with the wild-type, mainly because of no immunological defense against a wide variety of pathogenic bacteria and viruses. However, under sterile conditions and controlled temperatures, they could live as long as the wild type. Because nu/nu mice do not reject either allografts or xenografts, they have often been used to test the anticancer effects of new drugs against human cancer xenografts, before their clinical trials. Around 2000, deficiency of a gene called FOXN1 was found to be solely responsible for their phenotypes (i.e., hair-less with thymic deficiency) 28, 29. However, the signaling pathway(s) underlying FOXN1-dependent hair-growth and thymus development remained unknown until recently.
Finally in 2018, a Chinese group solved this mystery. Downregulation of FOXN1 in human lung cancer cells (A549) activates the β-catenin gene, just downstream of PAK1, promoting the growth of cancer cells [30], clearly indicating that FOXN1 is a tumor suppressor gene that blocks the oncogenic PAK1/β-catenin pathway. This finding is not entirely a surprise, because in 2002 a few potent D3 analogs developed by Milan Uskokovic’s team at Roche promoted hair-growth in the nu/nu mice even with the daily dose below 50 ng/kg [31]; and a few other natural PAK1 blockers such propolis, melatonin and cucurbitacins also promoted hair growth in cell culture or in vivo 7, 32, 33. It is also worth noting that VDR-null mice are also hair-less, and D3 could restore hair-growth in VDR-null nice [34], probably by inactivating RAC/PAK1/β-catenin signaling [35] (Fig. 2a). Interestingly, downregulation of PAK1/β-catenin signaling in mice also boosts their immune system [36].
Although a PAK1-KO mutant of C. elegans lives 60% longer than the wild type [6], so far nobody has been motivated to examine the heathy lifespan of PAK1-KO mice as yet, partly because even the wild-type lives as long as 3 years (>75-times longer than C. elegans). Nevertheless, it is expected that PAK1-deficient mice live significantly longer than the wild-type, mainly because mice treated with either melatonin or rapamycin, in which PAK1 or TOR (an oncogenic kinase downstream of PAK1) is suppressed, are far more resistant to pathogens and stresses such as coldness and UV light in the wildness, unlike the ‘nude’ (FOXN1-deficient) mice, and indeed live significantly longer than the control wild mice 6, 7. In other words, phenotypes of PAK1-deficient mice must be precisely opposite to those of the fragile short-lived nu/nu mice.
The ED therapeutic Viagra® is also a PAK1 blocker
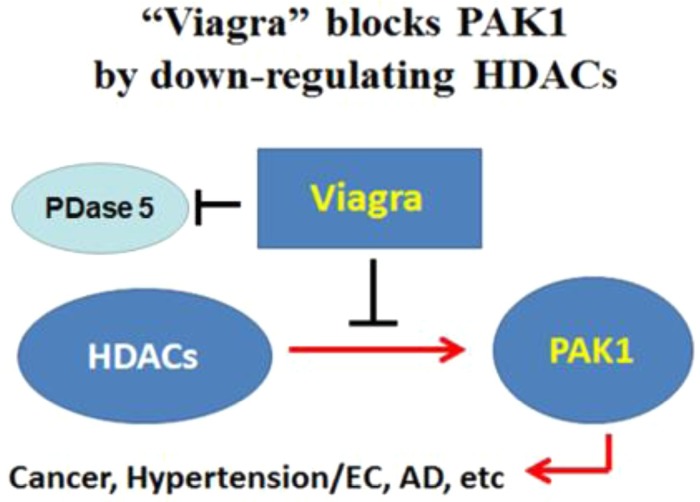
More than two decades ago, Pfizer marketed a new antihypertension drug called sildenafil mainly for the therapy of erectile disfunction (ED) under the brand name Viagra®. It is a cyclic GMP analog that inhibits selectively the cyclic GMP phosphodiesterase (PDase 5) with an IC50 below 5 nM [37]. It is well known that 1 in 6 males suffer from ED worldwide. However, it was recently revealed that Viagra® suppresses the PAK1-dependent expression of PD-L1 in human lung cancer cell lines such as A549 by downregulating histone deacetylases (HDACs) with the IC50 ∼100 nM [38], which eventually blocks PAK1 [6] (Fig. 3 ). Thus, a combination of Viagra® and anti-PD-L1 monoclonal antibodies (checkpoint therapeutics) is currently in clinical trials for cancers. Furthermore, back in 2009, a group at Columbia University reported that Viagra® (3 mg/kg i.p.) effectively restores memory loss in an Alzheimer’s disease (AD) model of mice [39]. Because AD is also among the PAK1-dependent diseases, these findings strongly suggest that Viagra® is a potent PAK1 blocker already on the market. Interestingly, unlike a more potent HDAC inhibitor FK228 (brand name Istodax®), which unfortunately fails to pass through the blood–brain barrier (BBB), Viagra® is able to cross the BBB effectively, and therefore could be potentially useful for therapy of a wide variety of PAK1-dependent neuronal diseases. It is also predicted that Viagra® could contribute to the extension (improvement) of our healthy lifespan, in particular of males, if it is properly used.
Figure 3.
Viagra® blocks p21-activated kinase (PAK)1 by downregulating histone deacetylases (HDACs).
Suppression of PD-1 and PD-L1 by 15 K via the PAK1 and BET signaling pathways
According to a 2018 Nobel Laureate, Tasuku Honjo at Kyoto University, the interaction of a ligand called PD-L1 in cancer cells with its receptor called PD-1 in T cells destroys the anticancer T cells responsible for so-called immuno-surveillance of cancers. Thus, monoclonal antibodies against PD-L1, PD-1 or both could be useful for cancer therapy – so-called checkpoint immunotherapy. Interestingly, several PAK1 blockers such as triptolide, curcumin, resveratrol and melatonin have been shown to suppress the expression of PD-L1, strongly suggesting that PD-L1 expression might depend on PAK1 [40]. In fact, recently this notion or hypothesis was proven genetically in mice and pancreatic cancer cells by a team at Melbourne University [41]. Thus, it is possible that the potent PAK1 blocker 15 K also suppresses PD-L1 expression.
In this context, it is worth noting that the new role of bromodomain and ex-terminal domain proteins (BETs) was recently identified. BETs are responsible for expression of PD-1 and PD-L1 via the β-catenin/MYC pathway [42]. Furthermore, a few years ago, a series of 1,2,3-triazol compounds were found to inhibit BETs [43]. Because 15 K is also among 1,2,3-triazol compounds, we are planning to test whether 15 K suppresses PD-1 and PD-L1 by blocking not only PAK1 but also BETs, both of which are essential for the activation of the oncogenic β-catenin/MYC pathway (Fig. 2b). If this working hypothesis is proven, it is most likely that 15 K could be a more effective (safer and less expensive as well) therapeutic than the Nobel-prize-winning monoclonal-based (checkpoint) therapeutics that have never worked for the treatment of brain tumors and pancreatic cancers.
PAK1 blockers for alternative therapeutics of coronavirus infection
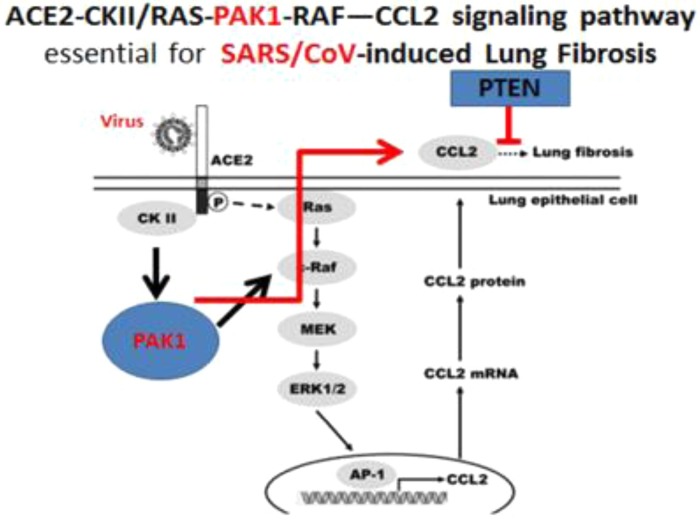
Since the end of 2019, deadly coronavirus infection started to spread rapidly worldwide from Wuhan in China, at the time of writing >3,000,000 people were infected, and the death toll had reached >20,000 people, https://corona.help/. Traditionally, a specific vaccine would be the way forward to combat viral diseases. However, it would take at least 12 months (and realistically 18 months) to prepare a vaccine (according to WHO). Until that time, it was feared the majority of coronavirus victims would perish. However, very recently, a team at University Colorado found that a tumor-suppressing phosphatase called phosphatase and tensin homolog (PTEN), which inactivates PAK1, could suppress the coronavirus-caused CCL2-dependent fibrosis of lungs [44]. Furthermore, a decade ago, a group at Taipei found that expression of CCL2 in lung cells depends on the oncogenic signaling pathway including trans-membrane viral receptor called angiotensin-converting enzyme 2 (ACE-2), kinases CK2, PAK1, RAF and the transcription factor AP-1 [45] (Fig. 4 ).
Figure 4.
Phosphatase and tensin homolog (PTEN), a p21-activated kinase (PAK)1 blocker, interferes with coronavirus-induced PAK1-dependent signaling pathway leading to lung fibrosis. Adapted, with permission, from 44, 45.
Interestingly, a few years after the 2003 SARS outbreak, a CDCP team at Atlanta reported that an old antimalaria drug called chloroquine suppresses the SARS/coronaviral infection in cell culture with an IC50 ∼1 μM [46]. Malaria infection requires PAK1 in host cells [6], and chloroquine upregulates p21 (a CDK inhibitor) whose expression is suppressed by PAK1 6, 47. Its anticoronaviral effect was recently confirmed in vitro by a team at the Chinese Academy of Sciences as well (https://www.nature.com/articles/s41422-020-0282-0/). Thus, these encouraging findings altogether strongly suggest, albeit not yet proven clinically, that PAK1 blockers such as propolis, ivermectin, melatonin, vitamin D3, ketolorac, HQ and AZ (Azithromycin) readily available on the market [7] could be potentially useful for treating or easing the pandemic coronavirus infection, until its vaccine becomes available.
Concluding remarks
In the past (over a decade ago), development of anti-PAK1 drugs conducted mainly by pharmaceutical giants focused on PAK1 specificity. However, in a common sense it would be very difficult to imagine that a given compound inhibits only PAK1, and not any other kinases or enzymes. Instead, here we have emphasized a chemical evolution of pre-existing natural or synthetic compounds known to inhibit PAK1 directly or upstream enzymes; but without any side effect in vivo (in small animals such as C. elegans and mice). This approach could boost either their cell permeability, water solubility or metabolism resistance. In a sense this ‘wise’ (reasonably flexible) approach simply follows the million-plus years of history regarding our own genetic and cultural evolution from primates that Charles Darwin revealed >150 years ago. Lastly it should be highlighted that the major purpose of PAK1 blockers for clinical application is to reduce the abnormally activated PAK1 to the ‘normal’ level (instead of ‘null’ level) in patients suffering from PAK1-dependent diseases. Thus, it is very unlikely that such a cautious measure or approach would cause any serious side effect.
Conflicts of interest
Neither author has any conflicts of interest to declare.
Acknowledgments
H.M. wrote a large part of this Feature and A.K. was in charge of the MART-10 section.
References
- 1.Maruta H., Korn E.D. Acanthamoeba cofactor protein is a heavy chain kinase required for actin activation of the Mg2+-ATPase activity of Acanthamoeba myosin I. J. Biol. Chem. 1977;252:8329–8332. [PubMed] [Google Scholar]
- 2.Manser E. A brain serine/threonine protein kinase activated by Cdc42 and Rac1 (PAK) Nature. 1994;367:40–46. doi: 10.1038/367040a0. [DOI] [PubMed] [Google Scholar]
- 3.Manser E. PAK kinases are directly coupled to the PIX family of nucleotide exchange factors. Mol. Cell. 1998;1:183–192. doi: 10.1016/s1097-2765(00)80019-2. [DOI] [PubMed] [Google Scholar]
- 4.Obermeier A. PAK promotes morphological changes by acting upstream of Rac. EMBO J. 1998;17:4328–4339. doi: 10.1093/emboj/17.15.4328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.He H. Signal therapy for RAS-induced cancers in combination of AG 879 and PP1, specific inhibitors for ErbB2 and Src family kinases, that block PAK activation. Cancer J. 2001;7:191–202. [PubMed] [Google Scholar]
- 6.Maruta H. Herbal therapeutics that block the oncogenic kinase PAK1: a practical approach towards PAK1-dependent diseases and longevity. Phytother. Res. 2014;28:656–672. doi: 10.1002/ptr.5054. [DOI] [PubMed] [Google Scholar]
- 7.Maruta H., Ahn M.R. From bench (laboratory) to bed (hospital/home): how to explore effective natural and synthetic PAK1-blockers/longevity-promoters for cancer therapy. Eur. J. Med. Chem. 2017;142:229–243. doi: 10.1016/j.ejmech.2017.07.043. [DOI] [PubMed] [Google Scholar]
- 8.Grunberger D. Preferential cytotoxicity on tumor cells by caffeic acid phenethyl ester isolated from propolis. Experientia. 1988;44:230–232. doi: 10.1007/BF01941717. [DOI] [PubMed] [Google Scholar]
- 9.Xu J.W. Downregulation of Rac1 activation by caffeic acid in aortic smooth muscle cells. Life Sci. 2005;76:2861–2872. doi: 10.1016/j.lfs.2004.11.015. [DOI] [PubMed] [Google Scholar]
- 10.Nguyen B.C. Frondoside A from sea cucumber and nymphaeols from Okinawa propolis: natural anticancer agents that selectively inhibit PAK1 in vitro. Drug Discov. Ther. 2017;11:110–114. doi: 10.5582/ddt.2017.01011. [DOI] [PubMed] [Google Scholar]
- 11.Taira N. Effect of Okinawa propolis on PAK1 activity, C. elegans longevity, melanogenesis, and growth of cancer cells. J. Agric. Food Chem. 2016;64:5484–5489. doi: 10.1021/acs.jafc.6b01785. [DOI] [PubMed] [Google Scholar]
- 12.Nguyen B.C. Artepillin C and other herbal PAK1-blockers: effects on hair cell proliferation and related PAK1-dependent biological function in cell culture. Phytother. Res. 2016;30:120–127. doi: 10.1002/ptr.5510. [DOI] [PubMed] [Google Scholar]
- 13.Kolb H.C. Click chemistry: diverse chemical function from a few good reactions. Angew. Chem. Int. Ed. Engl. 2001;40:2004–2021. doi: 10.1002/1521-3773(20010601)40:11<2004::AID-ANIE2004>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 14.Takahashi H. 1,2,3-Triazolyl esterization of PAK1-blocking propolis ingredients, artepillin C (ARC) and caffeic acid (CA), for boosting their anti-cancer/anti-PAK1 activities along with cell-permeability. Drug Discov. Ther. 2017;11:104–109. doi: 10.5582/ddt.2017.01009. [DOI] [PubMed] [Google Scholar]
- 15.Nguyen B.C. 1,2,3-Triazolyl ester of ketorolac: a “click chemistry”-based highly potent PAK1-blocking cancer-killer. Eur. J. Med. Chem. 2016;126:270–276. doi: 10.1016/j.ejmech.2016.11.038. [DOI] [PubMed] [Google Scholar]
- 16.Guo Y. R-ketorolac targets Cdc42 and Rac1 and alters ovarian cancer cell behaviors critical for invasion and metastasis. Mol. Cancer Ther. 2015;14:2215–2227. doi: 10.1158/1535-7163.MCT-15-0419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nguyen B.C. 1,2,3-Triazolyl ester of ketorolac (15K): boosting both heat-endurance and lifespan of C. elegans by down-regulating PAK1 at nM levels. Drug Discov. Ther. 2018;12:92–96. doi: 10.5582/ddt.2018.01018. [DOI] [PubMed] [Google Scholar]
- 18.Jagadeeshan S. p21-activated kinase (PAK1) influences therapeutic outcome in pancreatic cancer. Annal. Oncol. 2016;27:1546–1556. doi: 10.1093/annonc/mdw184. [DOI] [PubMed] [Google Scholar]
- 19.Hennig R. 1,2,3-Triazolyl ester of ketorolac (15K), a potent PAK1-blocker, inhibits both growth and metastasis of human pancreatic cancer orthotopic xenografts in mice. Drug Discov. Ther. 2019;13:248–255. doi: 10.5582/ddt.2019.01068. [DOI] [PubMed] [Google Scholar]
- 20.Eisman J.A. Suppression of in vivo growth of human cancer solid tumor xenografts by 1,25-dihydroxy vitamin D3. Cancer Res. 1987;47:21–25. [PubMed] [Google Scholar]
- 21.Flanagan J.N. Evaluation of 19-nor-2alpha-(3-hydroxypropyl)-1alpha,25-dihydroxy vitamin D3 (MART-10) as a therapeutic agent for androgen-dependent prostate cancer. Anticancer Res. 2009;29:3547–3553. [PubMed] [Google Scholar]
- 22.Zeng N. 1α,25 (OH) 2D3 induces actin depolymerization in endometrial carcinoma cells by targeting RAC1 and PAK1. Cell Physiol. Biochem. 2016;40:1455–1464. doi: 10.1159/000453197. [DOI] [PubMed] [Google Scholar]
- 23.Chiang K.C. The vitamin D analog, MART-10, attenuates triple negative breast cancer cells metastatic potential. Int. J. Mol. Sci. 2016;17:E606. doi: 10.3390/ijms17040606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Messing J.A. Effect of vitamin D3 on lifespan in Caenorhabditis elegans. Curr. Aging Sci. 2013;6:220–224. doi: 10.2174/18746098113066660038. [DOI] [PubMed] [Google Scholar]
- 25.Chiang K.C. Evaluation of the potential therapeutic role of a new generation of vitamin D analog, MART-10, in human pancreatic cancer cells in vitro and in vivo. Cell Cycle. 2013;12:1316–1325. doi: 10.4161/cc.24445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang Z. Triptolide downregulates Rac1 and the JAK/ STAT3 pathway and inhibits colitis-related colon cancer progression. Exp. Mol. Med. 2009;41:717–727. doi: 10.3858/emm.2009.41.10.078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Patil S. Phosphonooxymethyl prodrug of triptolide (minnelide): synthesis, physicochemical characterization, and efficacy in human colon adenocarcinoma and ovarian cancer xenografts. J. Med. Chem. 2015;58:9334–9344. doi: 10.1021/acs.jmedchem.5b01329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mecklenburg L. The nude mouse skin phenotype: the role of Foxn1 in hair follicle development and cycling. Exp. Mol. Pathol. 2001;71:171–178. doi: 10.1006/exmp.2001.2386. [DOI] [PubMed] [Google Scholar]
- 29.Cunliffe V.T. Complete rescue of the nude mutant phenotype by a wild-type Foxn1 transgene. Mamm. Genome. 2002;13:245–252. doi: 10.1007/s00335-001-3079-6. [DOI] [PubMed] [Google Scholar]
- 30.Ji X. Forkhead box N1 inhibits the progression of non-small cell lung cancer and serves as a tumor suppressor. Oncol. Lett. 2018;15:7221–7230. doi: 10.3892/ol.2018.8210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vegesna V. Vitamin D3 analogs stimulate hair growth in nude mice. Endocrinology. 2002;143:4389–4396. doi: 10.1210/en.2002-220118. [DOI] [PubMed] [Google Scholar]
- 32.Rose J. Induction of winter fur growth in mink (Mustela vison) with melatonin. J. Anim. Sci. 1984;58:57–61. doi: 10.2527/jas1984.58157x. [DOI] [PubMed] [Google Scholar]
- 33.Nguyen B.C. Artepillin C and other herbal PAK1-blockers: effects on hair cell proliferation and related PAK1-dependent biological function in cell culture. Phytother. Res. 2016;30:120–127. doi: 10.1002/ptr.5510. [DOI] [PubMed] [Google Scholar]
- 34.Chen C.H. Targeting expression of the human vitamin D receptor to the keratinocytes of vitamin D receptor null mice prevents alopecia. Endocrinology. 2001;142:5386–5389. doi: 10.1210/endo.142.12.8650. [DOI] [PubMed] [Google Scholar]
- 35.Zheng W. Vitamin D-induced vitamin D receptor expression induces tamoxifen sensitivity in MCF-7 stem cells via suppression of Wnt/β-catenin signaling. Biosci. Rep. 2018;38 doi: 10.1042/BSR20180595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Huynh N. Depletion of p21-activated kinase 1 (PAK1) up-regulates the immune system of APCΔ14/+ mice and inhibits intestinal tumorigenesis. BMC Cancer. 2017;17:31. doi: 10.1186/s12885-017-3432-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Padma-Nathan H., Giuliano F. Oral drug therapy for erectile dysfunction. Urol. Clin. North Am. 2001;28:321–334. doi: 10.1016/s0094-0143(05)70141-5. [DOI] [PubMed] [Google Scholar]
- 38.Booth L. [Pemetrexed + sildenafil], via autophagy-dependent HDAC downregulation, enhances the immunotherapy response of NSCLC cells. Cancer Biol. Ther. 2017;18:705–714. doi: 10.1080/15384047.2017.1362511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Puzzo D. Phosphodiesterase 5 inhibition improves synaptic function, memory, and amyloid-beta load in an Alzheimer’s disease mouse model. J. Neurosci. 2009;29:8075–8086. doi: 10.1523/JNEUROSCI.0864-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Maruta H. Breakthrough: PAK1-dependent expression of PD-L1 (programmed death ligand) Integr. Mol. Med. 2019;6:1–2. [Google Scholar]
- 41.Wang K. Inhibition of PAK1 suppresses pancreatic cancer by stimulation of anti-tumour immunity through down-regulation of PD-L1. Cancer Lett. 2020;472:8–18. doi: 10.1016/j.canlet.2019.12.020. [DOI] [PubMed] [Google Scholar]
- 42.Andrieu G.P. BET protein targeting suppresses the PD-1/PD-L1 pathway in triple-negative breast cancer and elicits anti-tumor immune response. Cancer Lett. 2019;465:45–58. doi: 10.1016/j.canlet.2019.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sharp P.P. Design, synthesis, and biological activity of 1,2,3-triazolobenzodiazepine BET bromodomain inhibitors. ACS Med. Chem. Lett. 2017;8:1298–1303. doi: 10.1021/acsmedchemlett.7b00389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lu S. PTEN (phosphatase and tensin homolog) protects against Ang II (angiotensin II)-induced pathological vascular fibrosis and remodeling. Brief report. Arterioscler. Thromb. Vasc. Biol. 2020;40:394–403. doi: 10.1161/ATVBAHA.119.313757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chen I.Y. Upregulation of the chemokine (C-C motif) ligand 2 via a severe acute respiratory syndrome coronavirus spike-ACE2 signaling pathway. J. Virol. 2010;84:7703–7712. doi: 10.1128/JVI.02560-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Vincent M.J. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol. J. 2005;2:69. doi: 10.1186/1743-422X-2-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Oh S. Anti-inflammatory activity of chloroquine and amodiaquine through p21-mediated suppression of T cell proliferation and Th1 cell differentiation. Biochem. Biophys. Res. Commun. 2016;474:345–350. doi: 10.1016/j.bbrc.2016.04.105. [DOI] [PMC free article] [PubMed] [Google Scholar]