Abstract
Severe COVID-19 associated pneumonia patients may exhibit features of systemic hyper-inflammation designated under the umbrella term of macrophage activation syndrome (MAS) or cytokine storm, also known as secondary haemophagocytic lymphohistocytosis (sHLH). This is distinct from HLH associated with immunodeficiency states termed primary HLH -with radically different therapy strategies in both situations. COVID-19 infection with MAS typically occurs in subjects with adult respiratory distress syndrome (ARDS) and historically, non-survival in ARDS was linked to sustained IL-6 and IL-1 elevation. We provide a model for the classification of MAS to stratify the MAS-like presentation in COVID-19 pneumonia and explore the complexities of discerning ARDS from MAS. We discuss the potential impact of timing of anti-cytokine therapy on viral clearance and the impact of such therapy on intra-pulmonary macrophage activation and emergent pulmonary vascular disease.
1. Introduction
We live in extraordinary times with intensive care units around the globe being overwhelmed with severe COVID-19 viral pneumonia that leads to severe adult respiratory distress syndrome (ARDS). Given the lack of a vaccine or proven effective anti-viral therapy and non-existent herd immunity, anti-cytokine therapy, most notably anti-IL-6 and others including IL-1 antagonism have been proposed for mitigating against the hyper-inflammation that may develop in conjunction with this virally induced ARDS [1,2]. The backdrop for cytokine antagonism of hyper-inflammation in non-immunodeficient patients emerged in children with systemic onset juvenile inflammatory arthritis (sJIA) also known as Stills disease where a severe hyer-cytokinemic inflammatory state variously termed as a cytokine storm, macrophage activation syndrome (MAS) or secondary haemophagocytic lymphohistocytosis (sHLH) may occur [[3], [4], [5]]. Impressive responses to antagonism of either IL-6 or IL-1 have been reported in sJIA [6,7].
The suspicion that overzealous immune responses associated with MAS/sHLH may be driving COVID-19 related ARDS has created a tremendous interest for anti-cytokine therapy for dampening of such exaggerated immune responses as a beneficial therapeutic strategy [2]. The focus of this article is to explore COVID-19 pulmonary immunopathology and describe the potential benefits or disadvantages of IL-6 antagonism is patients with severe inflammatory responses that has implications for other anti-cytokine strategies including IL-1, IL-18 or IFNγ antagonism. Other DMARDs may also have potential use for treating COVID-19 patients, and excellent reviews can be found elsewhere [8].
2. Macrophage activation syndrome in COVID-19 pneumonia?
Although pneumonitis may occur, the classic MAS/sHLH picture in sJIA and other settings most often occurs outside the lungs and manifests as fevers, adenopathy, hepatosplenomegaly, anaemia, other cytopenias, liver function derangement and the activation of intravascular coagulation cascades secondary to inflammation, and is accompanied by marked hypercytokinaemia. (Fig. 1A) Clinical and laboratory parameters in the MAS/sHLH phenotype are similar to primary HLH but the latter is invariably autosomal recessive, presenting in childhood, and is typically due to mutations that impair NK and CD8+ cytotoxic T-cell function [[9], [10], [11]], although there are emergent overlaps. As the focus of this perspective is restricted to IL-6 in potential MAS in COVID-19 related pneumonia, the reader is referred to several recent articles on primary and secondary HLH and sJIA with MAS [[12], [13], [14], [15]].
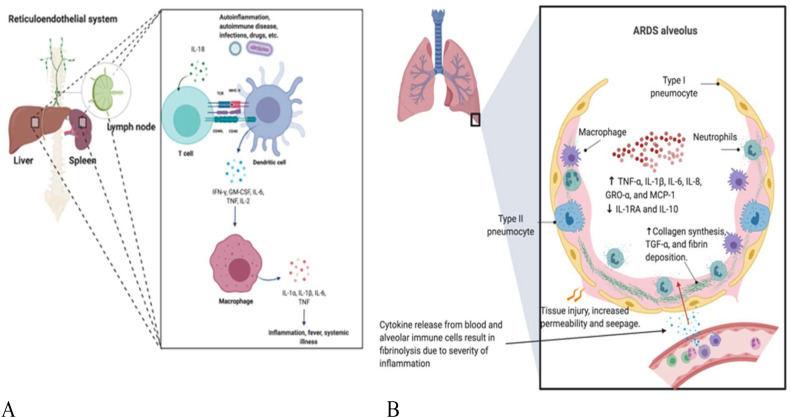
Fig. 1.
Classical MAS Organ Pattern versus COVID-19 Related Picture.
Panel A). The MAS/sHLH picture is associated with viral and other infections that does not have a special tropism for pulmonary tissues. The hyper-activation of T-cells is associated with lymphoid organ hyperplasia with hepatosplenomegaly and adenopathy. The resulting excessive reticuloendothelial system activity manifests as bone marrow haemophagocytosis and deranged liver function tests, deranged lipid profiles as a result of liver Kupffer cell dysfunction. Systemically circulating activated macrophages contribute to disseminated intravascular coagulation (DIC). A similar picture can develop in primary HLH that represents a rare group of immunodeficiency, linked to impaired cytolytic activity due to perforin pathway loss of function, with expansion of immune cells and the lymphoid organs that house them. In primary HLH, cytokine antagonism used with anti-microbial therapy may temporarily help prior to definitive bone marrow transplantation. Pulmonary involvement may be a feature of both pHLH and sHLH.
Panel B). There is evidence for MAS/sHLH emerging in the COVID-19 setting that is supported by the abnormal laboratory parameters without reporting of the classical organomegaly pattern described in Fig. 1A. Hyperactivation and over-zealous immune activity appears to be more confined to the lung parenchyma and immediately adjacent bronchial alveolar lymphoid tissue and is associated with ARDS development. Pulmonary haemophagocytosis has has been occasionally reported in human coronavirus infection but not yet been reported in COVID-19 associated pneumonia [56].
.It is proposed that severe widespread alveolar and interstitial inflammation extends to the closely juxtaposed pulmonary vasculature. The normal circulatory fibrinogen levels and regional fibrinolysis with elevated D-dimer formation seen in early COVID-19 pneumonia is not a feature of typical acute onset MAS/HLH. This MAS-like intra-pulmonary inflammation might influence a propensity towards severe local vascular dysfunction including micro-thrombosis and haemorrhage resulting in a lung centric pulmonary intravascular coagulopathy (PIC) presentation rather than a DIC presentation
Laboratory parameters including highly elevated CRP and hyperferritinaemia, the latter of which may play a complex role in disease [[16], [17], [18]], are key to the diagnosis of MAS/HLH and are elevated in many severe COVID-19 pneumonia cases. Other features including coagulopathy and abnormal liver function may be evident suggesting that a subgroup of COVID-19 pneumonia cases also have MAS/sHLH [19,20], but the immunopathology is very lung-centric with emergent ARDS and the absence of organomegaly (Fig. 1B). A cytokine profile, resembling MAS/sHLH has also been noted in COVID-19 patients, notably increased IL-1β, IL-2, IL-6, IL-17, IL-8, TNF and CCL2 [21]. The apparent recognition of MAS/sHLH complicating COVID-19 pneumonia forms the backdrop for cytokine suppression strategies [6,[22], [23], [24]], although the evidence for these cytokines being effective for active MAS complicating sJIA itself is not compelling [25].
Not only do COVID-19 pneumonia patients have serological markers associated with MAS development including hyperferritinaemia, deranged liver function tests with coagulopathy but also preliminary trials demonstrate evidence for efficacy for anti-IL-6R blockade with tocilizumab [26,27]. Furthermore, a MAS/sHLH has been described in the related SARS coronavirus sufferers in prior studies [26,27]. In common with the disseminated intravascular coagulation (DIC) associated with MAS/HLH, there is evidence of D-dimer level elevation in COVID-19 pneumonia which might represent an extension of this novel virally induced hyper-inflammatory pulmonary immunopathology to the adjacent microcirculation with extensive secondary fibrinolytic activation (Fig. 1B). This emerging COVID-19 immunopatholgoy could thus be associated with extensive pulmonary microthrombosis rather than the DIC that typically occurs with advanced MAS. The MAS that supervenes COVID-19 pneumonia is mostly anatomically compartmentalized to the lungs and thorax making clinical recognition difficult from ARDS and indeed for fully disentangling the precise pathological picture(19), (Fig. 1), which in some cases could be possibly mixed.
The limited COVID-19 post mortem data shows prominent alveolar oedema, hyalonosis (intra-alveolar proteinosis) and fibrin deposition with pneumocytes viral cytopathic change and immune cell infiltration including lymphocytes is typical or ARDS [26] as is evolving severe multi-organ damage including renal, cardiac and liver dysfunction with hypoproteinaemia [28]. In patients with ARDS (not generally due to viral pneumonia, but other causes) the elevation in baseline plasma levels of IL-6 predicted a poor survival [29] as did even higher bronchoalveolar (BAL) fluid levels indicating a pulmonary, rather than systemic origin for these cytokines in ARDS pathology [30]. Therefore, the overlapping cytokine profiles between severe ARDS and MAS/sHLH may limit the utility of cytokine profiling in the differentiation between both conditions and many of the laboratory changes reported in COVID-19 could predominantly reflect ARDS. (Fig. 2 ).
Fig. 2.
Hyper-cytokinaemic Overlaps Between ARDS and MAS.
Elevated cytokine levels or hypercytokinaemia are associated with the current COVID-19 infection [20] and independently found in the MAS spectrum and many other settings including sepsis [17]. Cytokines including IL-6 may be substantially elevated in isolated ARDS. The magnitude of cytokine level changes may not be reliable in the differentiation from other cause of elevated cytokine levels including severe infection or tissue destruction and dependable cut-off values do not exist. Superimposed second viral or bacterial infections in either setting may further drive systemic cytokine responses. Therefore, phase 3 studies based on elevated IL-6 levels may encompass a group of ARDS cases without the bone fide MAS phenotype [57].
3. A new proposed integrated innate and adaptive immune mechanisms in MAS/HLH
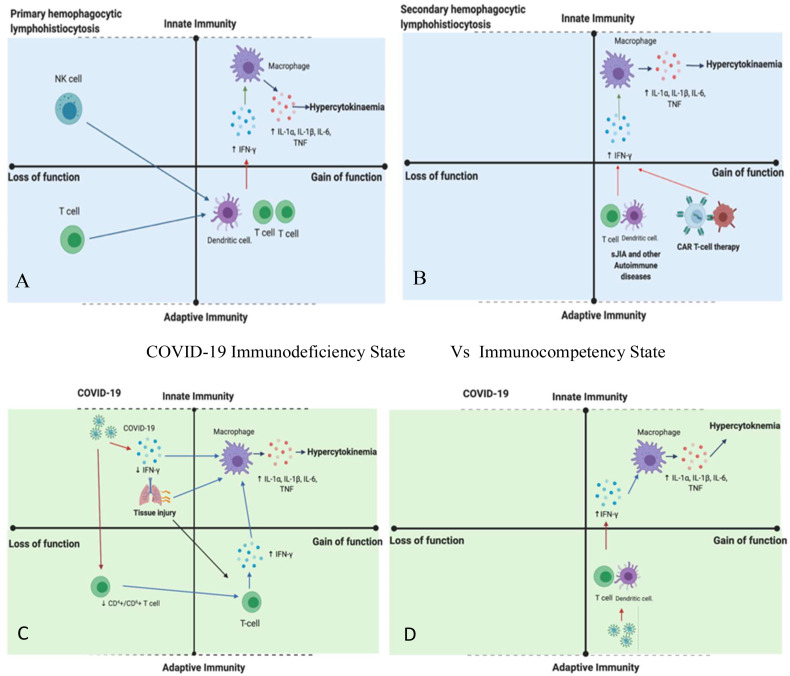
The link between a COVID-19 induction of a temporary immunodeficiency states (with features resembling primary HLH) rather than MAS/sHLH (that may occur in completely immunocompetent states) is not something yet that is fully appreciated. At first glance, the notion of hyper-inflammation in the face of immunodeficiency seems counter-intuitive, but that is exactly what happens in primary HLH. Inflammation against self occurs along an immunological disease continuum with pure innate and pure adaptive immunity at opposite boundaries [31]. Within these clear boundaries it is possible to stratify immune responses as being loss of function, normal or gain of function which then permits a better conceptual understanding of the integrated workings of the system [32] (Fig. 3 ).
Fig. 3.
Relative Innate and Adaptive Immune Mechanisms in HLH Spectrum Pathology.
Immunodeficiency State (Primary HLH) Vs Immunocompetency State (MAS/sHLH.
COVID-19 Immunodeficiency State Vs Immunocompetency State.
The HLH responses can occur in the setting of both immunodeficiency states and in immunocompetence settings and following adaptive immune system engineering (Panel A and B in blue). In humans with defects in NK and CD8+ T-cells, the inability to kill virally infected cells results in primary HLH with hyper-cytokinaemia that can't be cured with anti-cytokine strategies as genetic defect typically in perforin pathway machinery makes pathogen elimination impossible [58] (Panel A). In CAR-T cell therapy hyper proliferating engineered T-cells may drive MAS/sHLH that only lasts for the duration of the presence of detectable tumour antigen (Panel B). Similar mechanisms may occur in sJIA or autoimmune diseases, but this remains to be fully defined. This model is based on the immunological disease continuum modified for gain or loss of function in innate or adaptive immunity (Reference 43).
When COVID-19 hyper-inflammatory or cytokine storm reactions are viewed through the lens of primary and secondary HLH, it is noteworthy that virally induced immunosuppression may play a key role (Panel C). Coronavirus family members including COVID-19 pneumonia is associated with robust interferon suppression, blood lymphopenia including NK cell abnormalities. Such scenarios likely drive macrophage infiltration and the “second wave” of non-type-1 interferon pathway cytokines including IL-6, IL-1, IL-18, INFγ, GM-CSF and others that lead to a blood hyper-cytokinaemic picture (Panel B). It remains to be seen how many cases of COVID-19 related fatalities have mutations in genes associated with the primary HLH disorders and thus closely resemble primary HLH (Panel A). Based on persistent viral shedding from the respiratory tract during CORVID-19 related disease, then an exaggerated immune response including IFN driven and T-cell driven responses that clears the virus but induces pulmonary immune system collateral damage and ARDS offers an explanation for the more typical MAS/sHLH phenotype. Viral clearance in the face of marked hyper-inflammation would be expected in the face of brisk anti-viral antigen responses.
Primary HLH broadly defines hyper inflammatory immunodeficiency states that often equate with failure of the perforin and NK and CD8+ cytotoxic T-cells killing machinery that forms pores leading to cell lysis to initiate apoptosis of virally infected cells [9,33,34]. This leads to widespread T-cell mediated IFNγ driven secondary cytokine driven macrophage activation (Fig. 3A). Vigorous immunosuppression and sometimes anti-viral chemotherapy in these settings only represents a bridging strategy towards definitive allogeneic bone marrow transplantation strategies with immunosuppression being futile. Indeed evidence for such a primary HLH with immunodeficiency picture in adults succumbing to HIN1 influenza viral pneumonia has been reported [19], making it possible that patients succumbing to COVID-19 may also occasionally harbour perforin pathway mutations.
The COVID-19 associated pneumonia is associated with lung damage and ARDS and robust interferon suppression with lymphopenia as part of the virally induced immunosuppression. Also, preliminary data suggest that disease severity in COVID-19 may be associated with low IFN-γ production by CD4 + T-cells [35]. The related SARS-CoV virus open reading frame (ORF) and N proteins, act as antagonists to the interferon pathway by regulating IFN-β synthesis and signalling [36] which was mirrored in another experimental model [37]. In MERS-CoV–infected rhesus macaques, treatment with interferon-α2b was able to improve outcome [38]. Both IFN-β and IFN-γ inhibit the replication of SARS-CoV [39]. A characteristic feature of primary HLH but not sHLH/MAS is defective NK function which is also reported in COVID-19 infection, but by different mechanisms [40].
Analogous to primary HLH, the loss of “front line” anti-viral defence mechanism may activate a “second wave” of more tissue aggressive immunity including exaggerated IL-6 production with a secondary cytokine storm supervening with increased tissue damage (Fig. 3C) and (Fig. 4 ). Other “second wave” of non-type-1 interferon pathway myeloid and stromal derived cytokines including IL-1, TNF, IL-18, GM-CSF would be expected to be part of blood hyper-cytokinaemic and MAS picture (Panel 4C). Accordingly, there are similarities between COVID-19 and primary HLH which would point towards the importance of viral load reduction in COVID-19 (Fig. 3C).
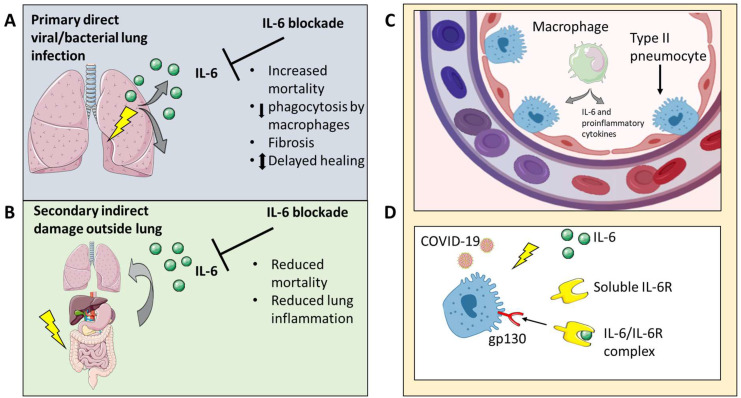
Fig. 4.
IL-6 in Viral Pneumoia and Potential Role in Type II Pneumocyte COVID-19 infection.
Panel A). Some experimental models of viral pulmonary infection triggering ARDS show that IL-6 may have a protective role with its blockade increasing mortality including reduction and phagocytosis and increased fibrosis depending on model. Panel B). Bacterial, rather than viral infections, originating outside the lung that also trigger ARDS but in this setting IL-6 blockade may be beneficial. The latter is more akin to classical MAS/sHLH where pulmonary pathology, when present, may be due to exogenous factors. However, the fact that IL-6 is detrimental in murine virally induced ARDS cannot be extrapolated to the novel COVID-19 virus infection that shows a tropism for type 2 pneumocytes (Panel C). The COVID-19 suppression of first line interferon responses and abrogation of T-cell responses suggests a role for type 2 pneunocyte gp130 receptor expression and IL-6 in pulmonary pathology [59]. Also in experimental models, IL-6 may either suppress or facilitate viral replication depending on the virus [49], so data on COVID-19 related disease is eagerly awaited.
It is also postulated that the typical MAS/sHLH pathology that arises in immunocompetent cases (Fig. 3B) may also arise in the COVID-19 infection (Fig. 3D). However, how this occurs in the face of active infection and how these two patterns of MAS could be differentiated and whether this clinically matters, especially in advanced ARDS is presently unclear. Stated differently, such a hypersensitive T-cell mediated reaction against virally infected cells would be expected to clear the actual infection, but contributing to ongoing damage and ARDS (Fig. 4D). Viraemia has been reported in up to 40% of cases [41] with one study showing a strong correlation between serum viral RNA load and ARDS severity [42]. A simple serological score including serial measurements of CRP, ferritin and blood viral load could therefore be used to evaluate therapy strategies for these different types of MAS (Table 2), but it is essential to appreciate that the hypoxaemic environment of ARDS complicates the perceived MAS picture.
Table 2.
Therapy considerations based on COVID-19 MAS-like picture.
| Primary HLH features | Secondary HLH pattern | |
|---|---|---|
| Immune state | Viral induced immunodeficiency | Normal Immune response/ Hypersensitivity |
| Viral Load | Persistent Viral Shedding more likely | Initial Viraemia |
| VViral shedding expected to disappear with vigorous T-cells responses to COVID-19 infected cells. | ||
| Progression rate | Unclear | Unclear |
| Rapid deterioration 2nd week? | ||
| Resolution rate | Slower resolution | May quickly improve on viral load elimination? |
| CRP | Elevated ++ | Elevated+++ |
| Ferritin | Elevated++ | Elevated ++ (possible rapid rise) |
| Anti-viral therapy | Consider Anti-viral therapy | ? |
| Corticosteroids | Caution (increase viraemia) | consider if viraemia cleared/clearing |
| Anti-IL-6R and biologics | Less likely to benefit (may cause harm) | More likely benefit |
| Frequency | Very common | Less common |
4. Interleukin-6 and immune function in COVID-19 related pneumonia
Clues as to how the increased levels of IL-6 and other cytokines that arise in ARDS impact on immunity come from experimentally induced viral lung infection where IL-6 may have contextual protective or exacerbating roles including severity of infection, survival and tissue remodeling, but there are very limited data on coronavirus family members in general (Table 1 ). Interleukin-6 also plays an important role in lung repair responses following viral or chemical insults indicating that timing of administration of anti-IL6R could impact on proper tissue remodeling (Table 1). In human epithelial cells, SARS-CoV was able to induce greater IL-6 when compared to Influenza-A virus and human parainfluenza virus type 2, but interestingly induced less SOC3 than other viruses, suggesting a potential basis for exaggerated IL-6 responses with this family of viruses [43].
Table 1.
Impact of Interleukin-6 on viral pneumonitis and lung Injury.
| Experimental model | Key findings | Reference |
|---|---|---|
| Infection of human epithelial cells with SARS-CoV |
|
[43] |
| Infection of monocytes/macrophages with SARS-spike protein |
|
[60,61] |
| Murine SARS-CoV model |
|
[62] |
| Influenza infection with IL-6 KO mice | IL-6 KO mice show
|
[63,64] |
| HSV-1 respiratory infection with IL-6KO mice |
|
[65] |
| Experimental ARDS induced by the intratracheal instillation of bacterial endotoxin |
|
[66,67] |
| Infectious models of tuberculosis, pneumococcal pneumonia and pulmonary aspergillosis |
|
[[68], [69], [70]] |
| Bleomycin lung injury model with IL-6 neutralization | IL-6 neutralization resulted in:
|
[71] |
| Rat model of bacterial sepsis remote from the lung with tocilizumab | Tocilizumab (anti-IL-6) resulted in:
|
[72] |
A picture emerges of COVID-19 ARDS and “second wave” pro-inflammatory cytokines including IL-6 and others leading to the MAS like pathology (Fig. 4C and D). The biology of IL-6 is complex with cytokine engagement of membrane anchored IL-6R and gp130 co-receptor being known to have tissue homeostatic and repair responses [44]. However, many non-immune cells including stromal and epithelial cells can induce marked inflammatory responses when soluble IL-6R-IL-6 anchors to membrane gp130 in what is termed trans-signalling. This engagement potently activates inflammatory responses [45] (Fig. 4B). Reports indicate that murine pulmonary stromal cells including myofibroblasts signal via both IL-6R and trans signalling but type 2 pneumocytes lack the soluble membrane IL-6 receptor indicating that these signals exclusively via IL-6R trans signalling [46] (Fig. 4D). Given that trans signalling typically drives inflammatory reactions this may impact on COVID-19 immunity [[47], [48], [49]].
5. Key messages and considerations for IL-6 for COVID-19 pneumonia, ARDS and MAS
We highlight how COVID-19 pneumonia may represent a novel viral MAS-like immunopathology, where hyper-inflammation may be key to virus control in the face of disabled type-1 interferon responses. Furthermore, the recognition of MAS/sHLH is problematic in COVID-19 pneumonia cases with the severe inflammation emanating from the pulmonary compartment mimicking MAS, but the lack of other classical systemic clinical features making MAS presentation atypical and diagnosis more difficult. Consequently, many cases receiving anti-IL-6R or other cytokine inhibitors therapy may have severe infection related ARDS without superimposed MAS. Although inflammation is more lung centered, than multi-organ, the argument for IL-6 involvement in COVID-19 related MAS comes from biochemical parameters changes including ferritin and the preliminary open reports of anti-IL6R efficacy [28,50]. We appear to be dealing with a pulmonary pathology distinct from MAS with DIC with both the macrophage activation and associated coagulopathy being more cantered on the lung and not systemic (Figure 1B).
Considerations around timing of anti-IL-6 therapy and its use outside a clear-cut systemic MAS pictures are key. Early use of anti-retroviral therapy strategies to reduce viral load appear crucial to preventing the relative immunosuppression that might be contributing to the MAS like picture development. The subtle overlap in features of severe COVID-19 pneumonia with primary HLH and the sub-analysis of the failed anti-retroviral trial in COVID-19 pneumonia suggested a potential benefit of early anti-viral therapy initiation [1,51].
It is presently unclear if elevated IL-6 levels are detrimental or beneficial in COVID-19 pneumonia. In experimental model systems, IL-6 can either suppress or facilitate viral replication [49], so studies on COVID-9 are urgently needed. Timing of anti-IL-6R, if too early might adversely affect viral clearance which needs to be assessed in trials. If it emerges that blocking IL-6R early in the course of COVID pneumonia MAS-like disease has a detrimental impact on type-2 pneumocyte anti-viral immunity, then local augmentation of IL-6 could be considered. Such are the complexities that only trial results will clarify.
In this perspective we focused on IL-6 and its relationship to the COVID-19 MAS-like pathology but several other relevant cytokines including Il-18, IFNγ, and the JAK1 pathway critically control macrophage function including IL-6 production during MAS states [52]. Antagonism of either IFNγ or IL-18 that may be upstream of IL-6 and IL-1 has been associated with efficacy in human HLH and SJIA [53,54]. Both the short term and long-term outcome of trials of IL-6 blockers in COVID-19 pneumonia are eagerly awaited to clarify nature of the MAS-like state.
If a MAS-like state exists and excessive IL-6 levels are detrimental-why shouldn't corticosteroids be first line therapy as these will vigerously suppress IL-6 and a raft of other cytokines? Although the recent open label study from Wu and colleagues showed a benefit for corticosteroids, the consensus is that these should not be used based on clinical experience in SARS-CoV, MER-CoV and other infections including influenza and respiratory syncytial virus infection, where collectively there is evidence for delayed viral clearance [20,55]. The MAS-like state in COVID-19 exhibits features of both primary and secondary HLH with death being linked to respiratory viral persistence in the aforementioned Wu et al. study, indicating that, analogous to primary HLH, ongoing infection may be a driver. The role of IL-6 and other cytokines in what could be a distinct MAS-like lung inflammation with associated inflammation driven pulmonary vascular disease awaits clarification.
Financial disclosure
Conflict of Interest: Dennis McGonagle has received speaker fees and honoraria from Roche, Sobi and Novartis and research grants from Novartis.
Acknowledgements
Dr. Rachel Tattersall and Dr. Jessica Manson for gathering together the UK HLH Across Specialty Collaboration Group and the group members themselves for discussions on Policy Development towards MAS in COVID-19 Pneumonia.
Figures were created using Biorender.com
References
- 1.Cao B., Wang Y., Wen D., Liu W., Wang J., Fan G. A trial of Lopinavir–Ritonavir in adults hospitalized with severe Covid-19. New Engl J Med. 2020 doi: 10.1056/NEJMoa2001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020 doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schulert G.S., Yasin S., Carey B., Chalk C., Do T., Schapiro A.H. Systemic Juvenile Idiopathic arthritis-associated lung disease: characterization and risk factors. Arthritis Rheumatol (Hoboken, NJ) 2019;71:1943–1954. doi: 10.1002/art.41073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Martini A.J.A.R. Vol. 12. 2012. Systemic juvenile idiopathic arthritis; pp. 56–59. [Google Scholar]
- 5.Behrens E.M.J.A.R. Vol. 7. 2008. Macrophage activation syndrome in rheumatic disease: what is the role of the antigen presenting cell? pp. 305–308. [DOI] [PubMed] [Google Scholar]
- 6.De Benedetti F., Brunner H.I., Ruperto N., Kenwright A., Wright S., Calvo I. Randomized trial of tocilizumab in systemic juvenile idiopathic arthritis. N Engl J Med. 2012;367:2385–2395. doi: 10.1056/NEJMoa1112802. [DOI] [PubMed] [Google Scholar]
- 7.Feist E., Quartier P., Fautrel B., Schneider R., Sfriso P., Efthimiou P. Efficacy and safety of canakinumab in patients with Still's disease: exposure-response analysis of pooled systemic juvenile idiopathic arthritis data by age groups. Clin Exp Rheumatol. 2018;36:668–675. [PubMed] [Google Scholar]
- 8.Favalli E.G., Ingegnoli F., De Lucia O., Cincinelli G., Cimaz R., Caporali R.J.A.R. COVID-19 infection and rheumatoid arthritis: Faraway, so close! Autoimmun Rev. 2020;102523 doi: 10.1016/j.autrev.2020.102523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Crayne C.B., Albeituni S., Nichols K.E., Cron R.Q. The immunology of macrophage activation syndrome. Front Immunol. 2019;10 doi: 10.3389/fimmu.2019.00119. 119–119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Martinez F.O., Sica A., Mantovani A., Locati M. Macrophage activation and polarization. Front Biosci. 2008;13:453–461. doi: 10.2741/2692. [DOI] [PubMed] [Google Scholar]
- 11.Carter S.J., Tattersall R.S., Ramanan A.V. Macrophage activation syndrome in adults: recent advances in pathophysiology, diagnosis and treatment. Rheumatology (Oxford) 2019;58:5–17. doi: 10.1093/rheumatology/key006. [DOI] [PubMed] [Google Scholar]
- 12.Ravelli A., De Benedetti F., Viola S., Martini A. Macrophage activation syndrome in systemic juvenile rheumatoid arthritis successfully treated with cyclosporine. J Pediatr. 1996;128:275–278. doi: 10.1016/s0022-3476(96)70408-0. [DOI] [PubMed] [Google Scholar]
- 13.Davi S., Consolaro A., Guseinova D., Pistorio A., Ruperto N., Martini A. An international consensus survey of diagnostic criteria for macrophage activation syndrome in systemic juvenile idiopathic arthritis. J Rheumatol. 2011;38:764–768. doi: 10.3899/jrheum.100996. [DOI] [PubMed] [Google Scholar]
- 14.de Benedetti F., Massa M., Robbioni P., Ravelli A., Burgio G.R., Martini A. Correlation of serum interleukin-6 levels with joint involvement and thrombocytosis in systemic juvenile rheumatoid arthritis. Arthritis Rheum. 1991;34:1158–1163. doi: 10.1002/art.1780340912. [DOI] [PubMed] [Google Scholar]
- 15.Poddighe D., Cavagna L., Brazzelli V., Bruni P., Marseglia G.L.J.A.R. Vol. 13. 2014. A hyper-ferritinemia syndrome evolving in recurrent macrophage activation syndrome, as an onset of amyopathic juvenile dermatomyositis: a challenging clinical case in light of the current diagnostic criteria; pp. 1142–1148. [DOI] [PubMed] [Google Scholar]
- 16.Colafrancesco S., Priori R., Alessandri C., Astorri E., Perricone C., Blank M. Vol. 60. 2014. sCD163 in AOSD: a biomarker for macrophage activation related to hyperferritinemia; pp. 177–183. [DOI] [PubMed] [Google Scholar]
- 17.Rosário C., Zandman-Goddard G., Meyron-Holtz E.G., D'Cruz D.P., Shoenfeld Y.J.B.M. Vol. 11. 2013. The hyperferritinemic syndrome: macrophage activation syndrome, Still's disease, septic shock and catastrophic antiphospholipid syndrome; p. 185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sharif K., Vieira Borba V., Zandman-Goddard G., Shoenfeld Y.J.C., E. Immunology . Vol. 191. 2018. Eppur Si Muove: ferritin is essential in modulating inflammation; pp. 149–150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bracaglia C., Prencipe G., De Benedetti F. Macrophage activation syndrome: different mechanisms leading to a one clinical syndrome. Pediatr Rheumatol Online J. 2017;15 doi: 10.1186/s12969-016-0130-4. 5–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wu C., Chen X., Cai Y., Xia J.A., Zhou X., Xu S. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med. 2020 doi: 10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wan S., Yi Q., Fan S., Lv J., Zhang X., Guo L. Characteristics of lymphocyte subsets and cytokines in peripheral blood of 123 hospitalized patients with 2019 novel coronavirus pneumonia (NCP) medRxiv. 2020 2020.02.10.20021832. [Google Scholar]
- 22.Vastert S.J., Jamilloux Y., Quartier P., Ohlman S., Osterling Koskinen L., Kullenberg T. Anakinra in children and adults with Still's disease. Rheumatology (Oxford) 2019;58 doi: 10.1093/rheumatology/kez350. vi9-vi22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Quartier P., Allantaz F., Cimaz R., Pillet P., Messiaen C., Bardin C. A multicentre, randomised, double-blind, placebo-controlled trial with the interleukin-1 receptor antagonist anakinra in patients with systemic-onset juvenile idiopathic arthritis. ANAJIS trialAnn Rheum Dis. 2011;70:747–754. doi: 10.1136/ard.2010.134254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yokota S., Imagawa T., Mori M., Miyamae T., Aihara Y., Takei S. Efficacy and safety of tocilizumab in patients with systemic-onset juvenile idiopathic arthritis: a randomised, double-blind, placebo-controlled, withdrawal phase III trial. Lancet (London, England) 2008;371:998–1006. doi: 10.1016/S0140-6736(08)60454-7. [DOI] [PubMed] [Google Scholar]
- 25.Yokota S., Itoh Y., Morio T., Sumitomo N., Daimaru K., Minota S. Macrophage activation syndrome in patients with systemic juvenile idiopathic arthritis under treatment with Tocilizumab. J Rheumatol. 2015;42:712–722. doi: 10.3899/jrheum.140288. [DOI] [PubMed] [Google Scholar]
- 26.Yao X.H., Li T.Y., He Z.C., Ping Y.F., Liu H.W., Yu S.C. A pathological report of three COVID-19 cases by minimally invasive autopsies. Zhonghua Bing Li Xue Za Zhi. 2020;49 doi: 10.3760/cma.j.cn112151-20200312-00193. E009. [DOI] [PubMed] [Google Scholar]
- 27.Tisoncik J.R., Korth M.J., Simmons C.P., Farrar J., Martin T.R., Katze M.G. Into the eye of the cytokine storm. Microbiol Mol Biol Rev. 2012;76:16–32. doi: 10.1128/MMBR.05015-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020 doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Meduri G.U., Headley S., Kohler G., Stentz F., Tolley E., Umberger R. Vol. 107. 1995. Persistent elevation of inflammatory cytokines predicts a poor outcome in ARDS: plasma IL-1β and IL-6 levels are consistent and efficient predictors of outcome over time; pp. 1062–1073. [DOI] [PubMed] [Google Scholar]
- 30.Meduri G.U., Kohler G., Headley S., Tolley E., Stentz F., Postlethwaite A.J.C. Vol. 108. 1995. Inflammatory cytokines in the BAL of patients with ARDS: persistent elevation over time predicts poor outcome; pp. 1303–1314. [DOI] [PubMed] [Google Scholar]
- 31.McGonagle D., McDermott M.F. A proposed classification of the immunological diseases. PLoS Med. 2006;3 doi: 10.1371/journal.pmed.0030297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Grateau G., Hentgen V., Stojanovic K.S., Jeru I., Amselem S., Steichen O. How should we approach classification of autoinflammatory diseases? Nat Rev Rheumatol. 2013;9:624–629. doi: 10.1038/nrrheum.2013.101. [DOI] [PubMed] [Google Scholar]
- 33.Grom A.A. Primary hemophagocytic lymphohistiocytosis and macrophage activation syndrome: the importance of timely clinical differentiation. J Pediatr. 2017;189:19–21. doi: 10.1016/j.jpeds.2017.07.025. e1. [DOI] [PubMed] [Google Scholar]
- 34.Brisse E., Matthys P., Wouters C.H. Understanding the spectrum of haemophagocytic lymphohistiocytosis: update on diagnostic challenges and therapeutic options. Br J Haematol. 2016;174:175–187. doi: 10.1111/bjh.14144. [DOI] [PubMed] [Google Scholar]
- 35.Chen G., Wu D., Guo W., Cao Y., Huang D., Wang H. 2020. Clinical and immunologic features in severe and moderate forms of Coronavirus Disease 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kopecky-Bromberg S.A., Martínez-Sobrido L., Frieman M., Baric R.A., Palese P. Severe acute respiratory syndrome coronavirus open reading frame (ORF) 3b, ORF 6, and nucleocapsid proteins function as interferon antagonists. J Virol. 2007;81:548–557. doi: 10.1128/JVI.01782-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cameron M.J., Kelvin A.A., Leon A.J., Cameron C.M., Ran L., Xu L. Lack of innate interferon responses during SARS coronavirus infection in a vaccination and reinfection ferret model. PLoS One. 2012;7 doi: 10.1371/journal.pone.0045842. e45842-e45842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Falzarano D., de Wit E., Rasmussen A.L., Feldmann F., Okumura A., Scott D.P. Treatment with interferon-alpha2b and ribavirin improves outcome in MERS-CoV-infected rhesus macaques. Nat Med. 2013;19:1313–1317. doi: 10.1038/nm.3362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sainz B., Jr., Mossel E.C., Peters C., Garry R.F.J.V. Vol. 329. 2004. Interferon-beta and interferon-gamma synergistically inhibit the replication of severe acute respiratory syndrome-associated coronavirus (SARS-CoV) pp. 11–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zheng M., Gao Y., Wang G., Song G., Liu S., Sun D. 2020. Functional exhaustion of antiviral lymphocytes in COVID-19 patients; pp. 1–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang W., Du R.-H., Li B., Zheng X.-S., Yang X.-L., Hu B. Vol. 9. 2020. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes; pp. 386–389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen W., Lan Y., Yuan X., Deng X., Li Y., Cai X. Vol. 9. 2020. Detectable 2019-nCoV viral RNA in blood is a strong indicator for the further clinical severity; pp. 469–473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Okabayashi T., Kariwa H., Yokota S., Iki S., Indoh T., Yokosawa N. Cytokine regulation in SARS coronavirus infection compared to other respiratory virus infections. J Med Virol. 2006;78:417–424. doi: 10.1002/jmv.20556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Mihara M., Hashizume M., Yoshida H., Suzuki M., Shiina M.J.C.S. Vol. 122. 2012. IL-6/IL-6 receptor system and its role in physiological and pathological conditions; pp. 143–159. [DOI] [PubMed] [Google Scholar]
- 45.Jones S.A., Richards P.J., Scheller J., Rose-John S.J.J.O.I., C. Research . Vol. 25. 2005. IL-6 transsignaling: the in vivo consequences; pp. 241–253. [DOI] [PubMed] [Google Scholar]
- 46.Le T.-T.T., Karmouty-Quintana H., Melicoff E., Le T.-T.T., Weng T., Chen N.-Y. Blockade of IL-6 <em>trans</em> Signaling attenuates pulmonary fibrosis. J Immunol. 2014;193:3755–3768. doi: 10.4049/jimmunol.1302470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Scheller J., Ohnesorge N., Rose-John S. Interleukin-6 trans-signalling in chronic inflammation and cancer. Scand J Immunol. 2006;63:321–329. doi: 10.1111/j.1365-3083.2006.01750.x. [DOI] [PubMed] [Google Scholar]
- 48.Garbers C., Rose-John S. Dissecting Interleukin-6 classic- and trans-signaling in inflammation and cancer. Methods Mol Biol (Clifton, NJ) 2018;1725:127–140. doi: 10.1007/978-1-4939-7568-6_11. [DOI] [PubMed] [Google Scholar]
- 49.Velazquez-Salinas L., Verdugo-Rodriguez A., Rodriguez L.L., Borca M.V. Vol. 10. 2019. The role of interleukin 6 during viral infections; p. 1057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ruan Q., Yang K., Wang W., Jiang L., Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020 doi: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Baden L.R., Rubin E.J. Covid-19 — the search for effective therapy. New Engl J Med. 2020 doi: 10.1056/NEJMe2005477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bracaglia C., Caiello I., De Graaf K., D'Ario G., Guilhot F., Ferlin W. Interferon-gamma (IFNy) in macrophage activation syndrome (MAS) associated with systemic juvenile idiopathic arthritis (sJIA). High levels in patients and a role in a murine mas model. Pediatr Rheumatol Online J. 2014;12 O3-O3. [Google Scholar]
- 53.Vallurupalli M., Berliner N. Emapalumab for the treatment of relapsed/refractory hemophagocytic lymphohistiocytosis. Blood. 2019;134:1783–1786. doi: 10.1182/blood.2019002289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Dinarello C.A. Targeting interleukin 18 with interleukin 18 binding protein. Ann Rheum Dis. 2000;59(Suppl. 1):i17–i20. doi: 10.1136/ard.59.suppl_1.i17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Russell C.D., Millar J.E., Baillie J.K. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet. 2020;395:473–475. doi: 10.1016/S0140-6736(20)30317-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020 doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Registry C.C.T. 2020. A multicenter, randomized controlled trial for the efficacy and safety of tocilizumab in the treatment of new coronavirus pneumonia (COVID-19) [Google Scholar]
- 58.Gupta S., Weitzman S. Primary and secondary hemophagocytic lymphohistiocytosis: clinical features. Pathogen Ther. 2010;6:137–154. doi: 10.1586/eci.09.58. [DOI] [PubMed] [Google Scholar]
- 59.Le T.-T.T., Karmouty-Quintana H., Melicoff E., Le T.-T.T., Weng T., Chen N.-Y. Vol. 193. 2014. Blockade of IL-6 Trans signaling attenuates pulmonary fibrosis; pp. 3755–3768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Dosch S.F., Mahajan S.D., Collins A.R.J.V. Vol. 142. 2009. SARS coronavirus spike protein-induced innate immune response occurs via activation of the NF-κB pathway in human monocyte macrophages in vitro; pp. 19–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wang W., Ye L., Ye L., Li B., Gao B., Zeng Y. Vol. 128. 2007. Up-regulation of IL-6 and TNF-α induced by SARS-coronavirus spike protein in murine macrophages via NF-κB pathway; pp. 1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Channappanavar R., Fehr A.R., Vijay R., Mack M., Zhao J., Meyerholz D.K. Vol. 19. 2016. Dysregulated type I interferon and inflammatory monocyte-macrophage responses cause lethal pneumonia in SARS-CoV-infected mice; pp. 181–193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Dienz O., Rud J.G., Eaton S.M., Lanthier P.A., Burg E., Drew A. Vol. 5. 2012. Essential role of IL-6 in protection against H1N1 influenza virus by promoting neutrophil survival in the lung; pp. 258–266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Yang M.-L., Wang C.-T., Yang S.-J., Leu C.-H., Chen S.-H., Wu C.-L. Vol. 7. 2017. IL-6 ameliorates acute lung injury in influenza virus infection; pp. 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Murphy E.A., Davis J.M., Brown A.S., Carmichael M.D., Ghaffar A., Mayer E.P.J.J. Effect of IL-6 deficiency on susceptibility to HSV-1 respiratory infection and intrinsic macrophage antiviral resistance. 2008;28:589–596. doi: 10.1089/jir.2007.0103. [DOI] [PubMed] [Google Scholar]
- 66.Bhargava R., Janssen W., Altmann C., Andrés-Hernando A., Okamura K., Vandivier R.W. Vol. 8. 2013. Intratracheal IL-6 protects against lung inflammation in direct, but not indirect, causes of acute lung injury in mice. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Voiriot G., Razazi K., Amsellem V., Van Nhieu J.T., Abid S., Adnot S. Interleukin-6 displays lung anti-inflammatory properties and exerts protective hemodynamic effects in a double-hit murine acute lung injury. 2017;18:64. doi: 10.1186/s12931-017-0553-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ladel C.H., Blum C., Dreher A., Reifenberg K., Kopf M., S.J.I. Kaufmann and immunity . Vol. 65. 1997. Lethal tuberculosis in interleukin-6-deficient mutant mice; pp. 4843–4849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.van der Poll T., Keogh C.V., Guirao X., Buurman W.A., Kopf M., Lowry S.F.J.J. Vol. 176. 1997. Interleukin-6 gene-deficient mice show impaired defense against pneumococcal pneumonia; pp. 439–444. [DOI] [PubMed] [Google Scholar]
- 70.Cenci E., Mencacci A., Casagrande A., Mosci P., Bistoni F., Romani L.J.T.J. Vol. 184. 2001. Impaired antifungal effector activity but not inflammatory cell recruitment in interleukin-6-deficient mice with invasive pulmonary aspergillosis; pp. 610–617. [DOI] [PubMed] [Google Scholar]
- 71.Kobayashi T., Tanaka K., Fujita T., Umezawa H., Amano H., Yoshioka K. Vol. 16. 2015. Bidirectional role of IL-6 signal in pathogenesis of lung fibrosis; p. 99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ibrahim Y.F., Moussa R.A., Bayoumi A.M., Ahmed A.-S.F.J.I. Vol. 28. 2020. Tocilizumab attenuates acute lung and kidney injuries and improves survival in a rat model of sepsis via down-regulation of NF-κB/JNK: a possible role of P-glycoprotein; pp. 215–230. [DOI] [PubMed] [Google Scholar]