Graphical abstract
Keywords: Epigenetics, microRNA, miRNA, Toxicology, Zebrafish
Highlights
-
•
Zebrafish are an established vertebrate model for toxicity studies.
-
•
Zebrafish have a fully sequenced genome and the capability to create genetic models.
-
•
Zebrafish have over 80 % homology for genes related to human disease.
-
•
Functions of miRNAs in the zebrafish genome are being characterized.
-
•
Zebrafish are ideal for mechanistic studies on how miRNAs regulate toxicity.
Abstract
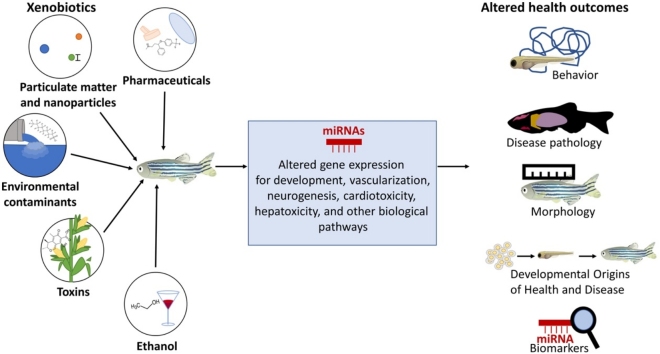
Zebrafish (Danio rerio) are an integrative vertebrate model ideal for toxicity studies. The zebrafish genome is sequenced with detailed characterization of all life stages. With their genetic similarity to humans, zebrafish models are established to study biological processes including development and disease mechanisms for translation to human health. The zebrafish genome, similar to other eukaryotic organisms, contains microRNAs (miRNAs) which function along with other epigenetic mechanisms to regulate gene expression. Studies have now established that exposure to toxins and xenobiotics can change miRNA expression profiles resulting in various physiological and behavioral alterations. In this review, we cover the intersection of miRNA alterations from toxin or xenobiotic exposure with a focus on studies using the zebrafish model system to identify miRNA mechanisms regulating toxicity. Studies to date have addressed exposures to toxins, particulate matter and nanoparticles, various environmental contaminants including pesticides, ethanol, and pharmaceuticals. Current limitations of the completed studies and future directions for this research area are discussed.
1. Introduction
Zebrafish (Danio rerio) are an established model for developmental biology due to their physiology, genetics, and assaying potential. Embryos develop ex vivo, offering a non-invasive approach to study developmental events. Transparency of embryos and larvae in early developmental stages make it easy to visualize internal organs/structures and embryogenesis is complete in only 3 days (Fig. 1). In the first few hours after fertilization, rapid cell division occurs in the zygote, cleavage, blastula, and gastrula periods. Between 24−48 hours post fertilization (hpf) the brain has 5 lobes, the embryo increases rapidly in length, the circulatory system forms, and the heart begins to beat. Between 48–72 hpf, the craniofacial cartilage development allows the mouth to become distinct and rudimentary gills develop. By 72 hpf, most of morphogenesis is completed [1]. From 72−120 hpf, zebrafish are referred to as eleutheroembryos. While in the process of hatching the embryo is not very active, but the eleutheroembryo stage is noted with gradual increased activity (swimming, jaw, eye, and fin movement). At 96−120 hpf it is common for behavioral assays for light/dark, acoustic stimuli, and toxicological responses to be initiated [2]. Behavioral assays, such as the photomotor response can be used to screen thousands of neuroactive drugs [[2], [3], [4]]. In addition, zebrafish have a fully sequenced genome with the capability of creating genetic models using multiple gene editing tools including CRISPR-Cas9 technology, making them ideal for mechanistic and discovery based studies on how genes impact physiology and behavior [5,6]. Due to assaying potential and genetic strengths, zebrafish are a good model for human translation, because zebrafish have 70 % gene homology which increases to 82 % when considering genes related to human disease [7]. Additionally, there is high conservation of metabolizing enzymes [3,8].
Fig. 1.
Zebrafish life cycle. Images are of an embryo at 1 h post fertilization (hpf), an eleutheroembryo at 5 days post fertilization (dpf), and adult female (top) and male (bottom) zebrafish.
As noted above, the zebrafish model organism can be used in high-throughput assays for drug discovery and chemical toxicity screening [3,[9], [10], [11]]. Due to their developmental and biomedical strengths zebrafish are increasingly being used to define mechanisms of toxicity [2,12,13] with recent reviews highlighting the strengths of the zebrafish as a model for toxicology [3,14]. Taking advantage of the developmental and biomedical strengths, the US FDA has used zebrafish toxicity assays for drug approval [3,15]. Many toxicological studies are also evaluating chemical effects on a protein, organ, and organismal level to provide a multi-level approach to understanding how a chemical affects health at different life stages and then determining human translation relevance [16]. Since zebrafish have shorter life periods, there is also the advantage of looking at the effects of a developmental exposure throughout the lifespan in the developmental origins of health and disease (DOHaD) paradigm, and even across generations. These advantages have made the zebrafish the most common fish model to perform multi- and transgenerational studies evaluating epigenetic mechanisms of toxicity [14].
MicroRNAs, also known as miRNAs, are one of three currently known epigenetic mechanisms that help regulate gene expression, alongside methylation and histone modifications, without altering DNA sequence [17,18]. miRNAs are single strand noncoding RNAs that are about 22 nucleotides in length that can regulate gene expression during and after transcription to form proteins [[19], [20], [21]]. miRNAs are highly concerted in a time and tissue specific manner, and are important for gene regulation during development [12,22,23]. miRNAs work through a cascade of molecular mechanisms. miRNAs are transcribed by RNA polymerase II to generate primary miRNA, which are then cleaved by RNAse III enzyme in the nucleus. This cleaved molecule, known as the precursor miRNA is further cleaved by Dicer to form a mature miRNA [24,25]. To inhibit genes, miRNAs arrange into a multi-protein complex to form an RNA-induced silencing complex. This complex then binds to the 3′ UTR ends of the target mRNA of interest to silence the gene [24,26]. miRNAs are important because they help regulate life cycles in eukaryotic cells, molecular and metabolic processes, immunity, and stress response [21]. miRNAs are also important in pathogenesis and genetic disorders. The presence of miRNAs extracellularly was first described in 2008 in a lymphoma patient’s plasma [27]. Research with the zebrafish has also implicated the importance of miRNAs in several biological and disease pathways including heart formation, function, and disease; cancer signaling pathways; and reproduction [18,[28], [29], [30], [31]];[23]; [32]. miRNAs can be altered with disease state or xenobiotic exposure, secreted extracellularly, and detected in biofluids in humans. As such, miRNAs have become a popular biomarker to help predict risk to xenobiotic exposure.
Although many miRNAs are now identified in humans and multiple animal models including the zebrafish (Fig. 2), mechanisms of how xenobiotic exposure affects miRNA regulation is unclear, though several ideas have been proposed. For example, DNA damage can cause p53 and miRNA to work together in the nucleus to repair damage [21]. Nucleophilic sites of miRNA precursors can be bound by electrophilic metabolites, forming adducts, and preventing their ability to be worked on by Dicer in the cytoplasm. miRNAs can be absorbed by long non-coding RNAs (lncRNAs), which can then lead to altered translation and over expression of target genes [21]. In this review, we highlight the intersection of how toxins and xenobiotic exposure affects miRNAs as mechanisms of toxicity. Studies for this review were identified using the search terms: “zebrafish microRNAs, zebrafish miRNA, zebrafish miRNA toxicity, zebrafish miRNA toxicology, microRNA toxicology, or miRNA toxicology” on PubMed and Google Scholar. In this review, miRNAs in zebrafish are referred to as simply “miR-XXX” with human miRNAs denoted as hsa-miR-XXX.
Fig. 2.
Total number of mature miRNAs currently identified in zebrafish, mouse, and human genomes. Numbers were attained from the latest version of miRbase release 22.1 (October 2018).
2. miRNA regulation of toxin response
Mechanisms of miRNA regulation of toxin response is in its infancy with studies beginning to evaluate mycotoxins and cyanobacteria (Table 1). Wu and colleagues through a series of publications detailed the effects of developmental exposures to the mycotoxins citrinin and ochratoxin, which are contaminants of foods including cereals as well as animal feeds. In evaluating cardiotoxicity, the authors focused on two miRNAs that regulate heart development: miR-138 and miR-218a. Citrinin exposure caused decreased expression of miR-138, which was associated with low survival and abnormal heart development. Although miR-218a expression decreased, no cardiotoxicity parameters appeared to be altered with this miRNA [32]. miR-138 expression in zebrafish regulates gene expression in specific areas of the heart chamber, while miR-218a regulates heart loop formation; so alterations in these genes can lead to heart malformations [33]. Developmental exposure to the mycotoxin ochratoxin resulted in increased expression of miR-731 and was associated with decreased expression of renal genes, renal morphological defects, as well as a decrease in expression of vasculature genes [23,34]. miR-731 is an miRNA that has been shown to be regulated in conditions such as hypoxia, and regulates cell survival by inducing apoptosis [35]. As such, it was proposed that miR-731 upregulation could be blocking DNA replication in renal cells, leading to the declines seen in renal genes in the study.
Table 1.
miRNA regulation of toxins found to contaminate food and water sources.
| Toxin | Time of exposure | miRNAs | Health endpoints | References |
|---|---|---|---|---|
| 50 μM citrinin | 24−72 hpf | miR-138 | Decreased expression, leading to lower survival, abnormal heart looping, pericardial edema, cardiac ischemia, miR-138 rescue reduced symptoms and increased survival | [32] |
| miR-218a | Decreased expression but didn’t effect cardiac endpoints assessed | |||
| 0.5 μM ochratoxin | 6−72 hpf | miR-731 | Increased expression, leading to decreased expression of renal genes and morphological defects | [36] |
| 0.5 μM ochratoxin | 6−72 hpf | miR-731 | Increased expression, leading to decreased expression of vasculature genes and cerebral vasculature loss | [34] |
| 12−48 μM Microcystin-RR | 0.75−24 hpf | miR-146a | Up regulation | [37] |
| miR-190b | Down regulation | |||
| miR-126-DP | Down regulation - Rescue partially restores vascularization | |||
| miR-430 | Up regulation | |||
| miR-126 | Up regulation |
Cyanobacteria are microorganisms that can produce toxins and can be found in bodies of water, including drinking water sources [38]. Microcystin-RR (MC-RR) is a commonly found toxin in cyanobacterial blooms, and is associated with hepatotoxicity [39]. Early developmental exposure to MC-RR in zebrafish embryos caused a dose dependent increased rate of malformations such as tail defects and decreased vascularization [37]. These health outcomes were linked to changes in expression of 31 miRNAs (Table 1). miR-126 was decreased with MC-RR exposure and rescue was shown to partially restore vascularization. miR-126′s role in vascularization is supported in zebrafish and mice [40,41]. Additionally, miR-430 was speculated to play a role in developmental defects, which is supported by rescues of miR-430 in dicer mutants [42]. The results from this study suggest MC-RR toxicity alters several miRNAs, involved in development defects and vascularization [37].
3. miRNA regulation of particulate matter and nanoparticle toxicity response
Particulate matter (PM) is a mixture of solid particles and liquid droplets of trace metals and elements that the US EPA classifies by size [43]. PM composition varies spatially and temporally and their presence in the air is linked to respiratory issues and cardiovascular disease [44]. Zebrafish exposed to PM2.5 during development were found to have up regulation of miRNAs involved in DNA repair, hypertension, and immunity [28]. In addition, down regulation of miR-7a/b and miR-19b-3p was observed with the developmental PM2.5 exposure [28]. These miRNAs play a role in cardiomyocyte protection and are important for inhibiting cardiac fibrogenesis [45]. An additional study assessed methyl mercury, a component of PM, influences on miRNA profiles at 48 hpf [29]. Decreases in miR-7147 and miR-26a were observed. Increases in miR-375 and miR-206, which regulate pathways for cardiac muscle contraction, were also found [29]. Past studies focusing on PM pollution found differential expression of miRNAs involved in cardiotoxicity with different exposures in other animal models, further validating risk of adverse cardiac health outcomes and align with the findings reported in this zebrafish study (e.g., [45]). These results also suggest that PM exposure results not only in cardiotoxicity, but has potential for increased risks for cancer, inflammation, and immunity (Table 2).
Table 2.
miRNA regulation of particulate matter and nanoparticle toxicity response.
| Xenobiotic | Time of exposure | miRNAs | Health endpoints | References |
|---|---|---|---|---|
| 100 μg/ml PM2.5 | 6−120 hpf | let-7b | Up regulation - Regulates genes in DNA repair and breast cancer | [28] |
| miR-153 | Up regulation - Associated with hypertension in rats | |||
| miR-122 | Up regulation - Regulates hypoxia based glucose metabolism | |||
| miR-24 | Up regulation - Roles in immunity and inflammation | |||
| let-7i let-7i | Down regulation - Role in immunity | |||
| miR-7a/b | Down regulation - Cardiomyocyte protective | |||
| miR-19b-3p | Down regulation - Inhibits cardiac fibrogenesis | |||
| 3 mg/mL Silica nanoparticle, 0.01 mg/ml MeHg | 48 hpf | miR-7147, miR-26a | Down regulation | [29] |
| miR-26amiR-375, miR-206 | Up regulation(Many miRNAs and genes assessed but the authors focused on these miRNAs that were part of a pathway for cardiac muscle contraction) | |||
| In silico nanoparticles | miR-124 | Key regulator for Wnt signaling and implicated in DNA double strand damage | [20] | |
| miR-144 | Key mediator for oxidative stress | |||
| miR-19a | Important in oncogene regulation and in inflammatory responses | |||
| miR-155a | Inflammatory response mediator with MAPK and TNF signaling as candidate pathways | |||
| miR-223 | Predicted in three networks as a key regulator in tissue damage |
In addition to general PM, miRNA mechanisms of nanoparticle toxicity are also beginning to be identified using the zebrafish model system. Nanoparticles are a microscopic unit of a substance that has a minimum of one dimension less than 100 nm. Nanomaterials are used in medicine, food storage/packaging, and in cosmetics such as toothpastes and sunscreens. Although some nanoparticles are intended for consumption, the health risks for nanomaterials not meant for consumption have become an issue of concern [46]. Currently only one study, which performed an in silico analysis on nanoparticle toxicity, used the zebrafish model [20]. This study evaluated networks related to oxidative stress, DNA damage, and inflammation for nanoparticles of varying chemicals, sizes, shapes, and concentrations. Based on preselected criteria, the analysis found six tissue or biological response-specific miRNA-mRNA networks using bioinformatic algorithms for associating miRNA with mRNA (Table 2). Overall, miR-223 was predicted in all three networks and selected as the key regulator for zebrafish tissue damage induced by nanoparticles. miR-223 was indicated as being highly conserved, playing roles in immune cell development including myeloid cells and inflammation regulation. This study identified that the main mechanism of regulation is between miR-223 and insulin like growth factor-1 receptor (igf1r) [20,47]. This finding is important because this receptor is involved in cell growth and tumor progression [116].
4. miRNA regulation of environmental chemical toxicity response
4.1. miRNA regulation of pesticide toxicity response
Pesticides are chemicals used to control pests, which can range from animals, plants or fungi. Several studies used the zebrafish to investigate the role of miRNAs in insecticide toxicity. Fipronil is an insecticide that blocks ion channels and triazophos is also an insecticide that works through acetylcholinesterase inhibition. Both of these chemicals have been found as co-contaminants in the same environmental areas. Assessing adult zebrafish exposure for four days, mixtures of these chemicals found miR-29b and miR-738 to be down regulated after adjuvant exposure (Wang a et al. 2010). Follow-up studies focusing on fipronil exposure found dose dependent down regulation of miR-155, miR-216b, and miR-499, increasing expression of the target gene cytochrome b561 domain containing 2 (cyb561d2), which is important for electron transfer and cell defense [48,49]. Triazophos exposure resulted in dose dependent down regulation of miR-217, which the authors suggest as a potential biomarker for triazophos exposure [50] (Table 3).
Table 3.
miRNA regulation of pesticide toxicity response.
| Xenobiotic | Time of exposure | miRNAs | Health endpoints | References |
|---|---|---|---|---|
| 0, 4, 4.40, 4.84, 5.32, 5.86 or 6.44 mg/l of 30 % triazophos microemulsifier.0.050, 0.080, 0.128, 0.205, 0.328 or 0.524 mg/l for 1% fipronil ME;1.5, 1.8, 2.16, 2.59, 3.11, or 3.73 mg/l for 31 % triazophos + fipronil ME | 96 h adult exposure | miR-135c, miR-30b, miR-365 | Decrease expression with triazophos exposure | [51] |
| miR-21, miR-31, miR-203b, miR-455 | Increased expression with triazophos exposure | |||
| miR-199, miR-22b, miR-499 | Decreased expression with fipronil exposure | |||
| miR-128, miR-9 | Decreased expression with mixture | |||
| miR-735 | Increased expression with mixture | |||
| miR-181a, let-7i, miR-203b | Increased expression with ME formulation | |||
| miR-155, miR-99 | Decreased expression with ME formulation | |||
| 0.034, 0.068, 0.137, 0.274, 0.548, 1.098 μM fipronil | 96 h adult exposure | miR-155 | Dose dependent down regulation - Associated with increase in cyb561d2 which regulates electron transfer and cell defense | [48] |
| 0.034, 0.068, 0.137, 0.274, 0.548, 1.098 μM fipronil | 96 h adult exposure | miR-216b, miR-499 | Down regulated and interacts with the gene cyb561d2 which was up regulated in a dose dependent manner and is involved in chemical stress | [49] |
| 0.45, 0.9, 1.8, or 3.6 μg/ml triazophos | 96 h adult exposure | miR-217 | Dose dependent down regulation - Suggested as a biomarker for triazophos | [50] |
| 0, 0.3, 3, or 30 μg/l atrazine | 1 hpf to 12, 24, 36, 48, 60 or 72 hpf | miR-126 | Dose dependent up regulation at 36 hpf, suppression at 48 hpf, and up regulation at 60 hpf in 30 μg/l treatment group - Associated with hemorrhage, vascular remodeling and maturation effects in zebrafish embryogenesis | [18] |
| hsa-miR-126-3p | Altered in all 3 concentrations - Important for tumor suppression in several cancers, angiogenesis, and inflammation | |||
| miR-10 | miR-10 family regulates hox genes, specifically hoxb1a and hoxb3a which could lead to morphological malformations | |||
| miR-23a, miR-24 | Regulates angiogenesis | |||
| miR-16a, miR-16b | Key epigenetic factors in identifying prostate, breast and gastric cancer | |||
| miR-18a, miR-18b-5p | Highly expressed in several types of cancer | |||
| miR-143, miR-216b, miR-217 | Important for tumor suppression | |||
| miR-26a | Altered at 30 μg/l - Regulates neurogenesis | |||
| miR-124 | Decreased expression at 30 μg/l - Important for neurogenesis |
Atrazine is an herbicide commonly used in crop fields, and due to concerns over water contamination, the US EPA has set the potable water limit at 3 μg/l (ppb). An embryonic atrazine exposure in zebrafish resulted in a variety of miRNAs with altered expression [52]. hsa-miR-126−3p, which is implicated in cancer and inflammation [53], was found to be altered at all exposure concentrations. miR-10, which regulates hox genes and can lead to morphological malformations [54], was also reported to have altered expression. Two miRNAs that regulate angiogenesis, miR-23a and miR-24, were also changed. Furthermore, miR-124 expression was decreased. This miRNA plays an important role in neurogenesis [55]. The authors also observed several miRNAs differentially regulated that are associated with various types of cancers [52]. The results from these studies show that a single exposure to pesticides can alter several miRNAs that affect gene regulation and responses to chemical stress (Table 3).
4.2. miRNA regulation of other environmental chemicals
A few studies have initiated identification of mechanisms of miRNA regulation in the toxicity of multiple other environmental chemical contaminants including the dioxin 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD), the antimicrobial agent triclosan, the flame retardant hexabromocyclododecane (HBCD), the perfluoroalkyl substance perfluorooctanesulfonic acid (PFOS), the metal copper, the plasticizers bisphenol S (BPS) and bisphenol A (BPA), and the polychlorinated biphenyl (PCB) 1254 (Aroclor 1254) (Table 4).
Table 4.
miRNA regulation of environmental contaminants.
| Xenobiotic | Time of exposure | miRNAs | Health endpoints | References |
|---|---|---|---|---|
| 5 nM TCDD | 30 hpf for 1 h Assessed at 36 and 60 hpf | miR-24 | Up regulation - Proposed regulation of cardiac myocyte apoptosis and cardiac fibrosis | [24] |
| miR-27 | Down regulation - Proposed dysregulation of erythropoiesis regulation and maturation | |||
| miR-144, miR-451 | Up regulation - Proposed cardiac hypertrophy | |||
| miR-23a, miR-204 | Down regulation - Reduced expression of claudin proteins involved in tight junctions | |||
| 62.5, 125, or 250 μg/l triclosan | 4−96 hpf | miR-125b | Dose dependent dysregulation – Involved in fatty acid synthesis and metabolism | [56] |
| miR-205 | Dose dependent up regulation – Involved with DNA repair/cell cycle | |||
| miR-142a | Dose dependent up regulation – Associated with the P53 signaling pathway | |||
| miR-203a | Dose dependent up regulation – Associated with fatty acid synthesis and metabolism | |||
| 62.5, 125, or 250 μg/l triclosan | 6−120 hpf, 6 hpf-90 dpf (for pathology) |
miR-137 | Up regulation in visceral mass, forebrain and olfactory bulb; Down regulation of regulatory genes bcl11aa, mapk6 and runx1; Increasing expression of miR-137 led to bent spines, neuromast decrease, and hypoactivity | [57] |
| 2, 20, or 200 nM Hexa-bromocyclododecane (HBCD) | 1−72 hpf | miR-1 | Down regulation associated with cardiac hypertrophy and downregulation of nkx2.5, which is important for cardiac development | [23] |
| miR-219, miR-145, miR-194b, miR-221, miR-146b | All up regulated, except miR-146b, which was down regulated and is important for cardiovascular related diseases | |||
| 1 μg/mL Perfluorooctane sulfonate (PFOS) |
6 hpf to 24 or 120 hpf | miR-19d, miR-739 miR-181a |
Up regulated - Associated with inhibiting tumor cell proliferation by repressing p27 | [58] |
| miR-19b-c | Up regulated - Targets genes important in brain and nervous system development | |||
| 6.3, 16, or 40 μg/l CuCl2 | 12 mpf zebrafish exposed for 24 h | miR-187, miR-140 | Altered in all concentrations | [59] |
| miR-203a, miR-199*, miR-16a, miR-16c, miR-25 | Up regulated - Associated with mRNA levels of host genes in Ca signaling | |||
| miR-193b, miR-214, miR-183, miR-140* | Down regulated | |||
| let-7 a, e, f, g, h, i | Up regulated - Associated with regulation of development and neurogenesis | |||
| miR-7a | Up regulated - Associated with down regulation of target cytokine signaling 1 | |||
| miR-128 | Up regulated - Associated with neural differentiation | |||
| miR-724, miR-187, miR-126, miR-30c, miR-16c, miR-203a, miR-203b | Up regulated - Associated with neurogenesis | |||
| 5, 50 μg/l bisphenol S | 3−4 mpf adult male zebrafish exposed for 21 d | miR-184, miR-27e, miR-122, miR-204, miR-430a | Uniquely regulated at 5 μg/L | [60] |
| miR-205 | Up regulated - Targets cyp19a1b | |||
| miR-30c, miR-192, miR-430b, miR-454b, miR-499 | Down regulated - Targets cyp19a1b and genes associated with hematopoiesis and development of lymphoid organs and immune system | |||
| miR-223, miR-363 | Down regulated | |||
| miR-193b | Up regulated | |||
| 100 nM bisphenol A | 3 weeks in male zebrafish | miR-2189 | Down regulated - Associated with genes regulating cell cycle | [61] |
| miR-430b | Upregulated - Associated with apoptosis and autophagy | |||
| miR-724, miR-725, miR-193a, miR-202, miR-205, miR-133a | Targets several genes in oxidative phosphorylation, apoptosis, and cell cycle | |||
| 0.125, 0.5, 1.0 mg/l PCB 1254 | 24−120 hpf | miR-21 | Dose dependent up regulation. - Associated with the loss of calcium in zebrafish skeleton | [62] |
TCDD is a contaminant of chlorophenoxy herbicides and a known teratogen, but the mechanism of teratogenicity is not completely understood. To address miRNA dysregulation of TCDD developmental exposure, zebrafish embryos were exposed for 1 h (30–31 hpf) and miRNA profiles determined at 36 and 60 hpf using next generation sequencing (SOLiD) and two different microarray platforms for comparison of the technologies [24]. Surprisingly, the platforms identified dysregulation of different miRNAs, but the two microarray platforms (Agilent and Exiqon) had better overlap in miRNAs and agreement in how those shared targets were differentially expressed. Between all three techniques, only up regulation of miR-27, which is important for craniofacial cartilage development and vascular development, was consistent [63,64] and raises questions on the inconsistency among the platforms.
Triclosan is an antimicrobial agent that the US FDA issued ruling to remove from over the counter anti-microbial soaps [115]. Developmental triclosan exposure in zebrafish causes neurotoxicity such as decreased axonal length, and synaptic density and pathways associated with liver function [65,66]. To understand the role of triclosan exposure on miRNA regulation, zebrafish were exposed during development from 4−96 hpf. Four miRNAs with functions related to fatty acid synthesis and metabolism were up regulated: miR-125b, miR-205, miR-142a, and miR-203a [56]. In another study, triclosan developmental exposure from 6−120 hpf was shown to cause up regulation of miR-137 leading to decreased expression of genes important for neurodevelopment and metabolism [57]. These findings together suggest that triclosan elicits changes in fatty acid metabolism with implications for neurodevelopmental and liver disruption (Table 4).
Hexabromocyclododecane (HBCD) is a flame retardant and is used in polystyrene building insulation [67]. HBCD is an endocrine disrupting chemical and is currently undergoing risk evaluation by the US EPA (2019). Exposures to low concentrations of HBCD during development is associated with increased heart rates and expression alterations in genes that regulate heart rate [68]. To further determine miRNA regulation of HBCD cardiotoxicity, zebrafish were exposed to HBCD during embryogenesis and miRNA expression assessed [23,36]. Altered miRNAs identified in this study were related to cardiac hypertrophy and in regulating cardiac diseases. The authors also associated thicker ventricular walls, collagen deposition, and up regulation of genes associated with the affected miRNA pathways, which agreed with the cardiotoxicity observed [23].
Perfluorooctane sulfonate (PFOS), a perfluoroalkyl substance that was phased out by its major producer in 2002, is still present in environmental sources today due to its persistence. To determine miRNA dysregulation by PFOS exposure, zebrafish embryos were exposed from 6−24 hpf or 6−120 hpf and a microarray platform used to identify altered miRNAs [58]. 39 and 81 different miRNAs were identified at 24 or 120 hpf, respectively. miRNAs affected by PFOS exposure were involved in cell signaling/proliferation, adipose metabolism, and hormone secretion pathways. Targeted gene prediction on the most differentially regulated genes was completed and found that miR-19-b-c targets genes involved in neurodevelopment [58]. The results agree with previous reports that miR-19b acts in a complex to promote neural survival [69] and with the developmental neurotoxicity that is observed in mice with PFOS exposure [70].
The heavy metal copper is a common environmental contaminant that causes developmental toxicity, behavioral hyperactivity in larvae, and a reduction in memory in adult zebrafish [71,72]. Copper is also known as an olfactory toxicant. Focusing on the olfactory system, adult zebrafish were exposed for 24 h to copper and the genome interrogated using the Affymetrix GeneChip miRNA 2.0 array [59]. Results of this study revealed that miRNAs were differentially expressed in a dose dependent manner (Table 4). Based on their findings, the authors predicted that miR-187 has a role in neurogenesis. miR-187 expression has been altered in neural progenitor cells of rats inflicted with stroke, suggesting miR-187 to be important for neural maintenance and disease states [59,73]. miR-140 was changed in all concentrations and plays a role in craniofacial bone and cartilage development [74]. Moreover, 9 of the 11 let-7 isoforms in zebrafish were up regulated, which are important for neurogenesis in the central nervous system. In addition, two miRNAs, miR-128 and miR-138, which are expressed in the olfactory bulb, were also differentially expressed. Overall, several miRNAs with roles in neurogenesis were dysregulated by the copper exposure [59] (Table 4).
Plastic contamination is a growing environmental concern due to the volume of plastic debris present in the ocean, waterways, and animals in the environment [75]. Several papers discuss different types of plastics exposure and altered miRNAs. Lee and colleagues assessed exposure to the plasticizer, bisphenol S, in young adult male zebrafish using the Affymetrix GeneChip miRNA 4.0 Array [60]. 14 miRNAs were differentially expressed by both concentrations utilized in the experiment, and six of these miRNAs target cyp19ab mRNAs [60] (Table 4). Exposure to a similar plasticizer, bisphenol A, in adult male zebrafish caused up regulation of 14 miRNAs, down regulation of one miRNA, and differential expression of 6188 mRNAs in liver [61]. The authors proposed that 15 miRNAs could modulate 50 % of the altered mRNAs. The miRNA/mRNA deregulated pathways that were top ranked were pathways for nonalcoholic fatty liver disease, oxidative phosphorylation, and metabolic pathways [61]. Together, the results of these two studies suggest that male zebrafish exposed to bisphenol compounds results in alterations of miRNA pathways related to reproduction, metabolism, and liver toxicity (Table 4).
Polychlorinated biphenyls (PCBs) are chlorinated hydrocarbons that were used in various industrial processes until 1979 when they were banned in the US [76]. A developmental exposure to PCB 180 in rats was reported to cause sex-specific alterations in cranial size and teeth size [77]. This is due in part to miR-21′s role in regulating osteoclastogenesis and bone metabolism [78]. In agreement with this data, miR-21 was found to be up regulated in a dose-dependent manner in zebrafish exposed to PCB 1254 and to be associated with calcium loss in the zebrafish skeleton [62] (Table 4).
5. miRNA regulation of ethanol toxicity
Ethanol exposure studies are important due to severe developmental effects that are observed in children born to women that consume high amounts of ethanol while pregnant. Several labs have studied the relationship between ethanol exposure and dysregulation of miRNAs and have focused on different physiological outcomes (Table 5). Using a microarray-based approach, Soares et al. [79] identified altered miRNA profiles following exposure to ethanol from 4−24 hpf. Dose dependent differential miRNA expression was observed suggesting different cell responses during development and cell cycle processes (Table 5). miRNAs up regulated in both concentrations were miR-153a, miR-725, miR-30d, let-7k, miR-100, miR-738, and miR-732 [79]. Two other studies evaluating ethanol exposure during gastrulation observed down regulation of miR-9 and miR-153c, which is proposed to occur via methylation, leading to microcephaly, cranial abnormalities, and behavioral hyperactivity [80,81]. Meanwhile, chronic exposure in adult zebrafish for 8 weeks resulted in musculoskeletal damage through down regulation of miR-140−3p [82]. miR-140−3p has been found to target hey1 and notch1, both of which were up regulated in treated zebrafish in this study [82]. The authors proposed that miRNAs that are altered in the Notch pathway induce ethanol-related myopathy, which was supported by the observed 12 % reduction of muscle fiber in the ethanol exposed zebrafish [82].
Table 5.
miRNA regulation of ethanol exposure.
| Xenobiotic | Time of exposure | miRNAs | Health endpoints | References |
|---|---|---|---|---|
| 1 or 1.5 % ethanol | 4−24 hpf | miR-153a, miR-725, miR-30d, miR-let-7k, miR-100, miR-738, miR-732 | Up regulated in both exposures | [79] |
| 430 μM ethanol | 3.5−7.5 hpf | miR-9 | Down regulated - Decreased methylation and decreased expression of fgfr-1 and foxp-2 leading to teratological defects | [80] |
| Up to 300 mM ethanol | 4−24 hpf | miR-153c, miR-204 | Down regulated - Role in altered locomotion | [81] |
| miR-9 | Down regulated – Associated with craniofacial skeletal development | |||
| 0.5% ethanol | 8 weeks in adult zebrafish | miR-140−3p | Down regulated - Targets hey1 and notch1, which are upregulated in treated animals | [82] |
| miR-146 | Up regulated - Targets Notch signaling to suppress myogenesis |
6. miRNA regulation of pharmaceuticals
Pharmaceuticals, such as antidepressants have been ubiquitously found in aquatic systems, downstream of wastewater treatment plants, and in drinking water sources [83,84]. Antidepressant and anxiety medications are prescribed to improve mood and behavior related disorders, but concerns have been raised over developmental effects increasing risk for poor motor development in humans and zebrafish [85,86]. Fluoxetine is a medication synthesized by Eli Lily and is commonly prescribed for depression and anxiety. Zebrafish exposed throughout development to fluoxetine were found to have altered miRNAs in tissues and consistently increased miRNAs in eggs as adults [31] (Table 6). The authors also note their study is the first paper to link toxicant exposure to miRNA expression changes related to egg quality [31]. This finding suggests that a developmental exposure to fluoxetine can change expression of miRNAs in gametes. In another study, adult exposure to fluoxetine at environmentally relevant concentrations was also found to cause similar hepatic miRNA profiles up regulated in metabolic pathways as animals that are fasting [87].
Table 6.
miRNA regulation of pharmaceuticals.
| Pharmaceutical | Time of exposure | miRNAs | Health endpoints | References |
|---|---|---|---|---|
| 54 μg/l Fluoxetine | 3−144 hpf | miR-181a | Up regulated in ovary and fin tissue of exposed females | [31] |
| miR-740 | Down regulated in eggs - Targets multiple stress axis transcripts | |||
| miR-26a, miR-30d, miR-92a, miR-103, miR-30d, miR-92a | Up regulated in eggs from 5 mpf females; Down regulated in eggs from 9 mpf females - miRNA changes transcripts in eggs with reduction of transcripts in eggs from treated females at 5 mpf and increased at 9 mpf | |||
| 540 ng/l fluoxetine | 7 days in adult females | let-7d, miR-140−5p, miR-301a, miR-457b | Up regulated - Similar miRNA expression profile of fluoxetine exposed animals and animals that were fasted | [87] |
| 1 mM valproic acid | 4–48 or 96 hpf | miR-16a, miR-18c, miR-22a, miR-457b | Up regulated at 48 hpf | [12] |
| miR-96, miR-122, miR-124, miR-132, miR-135a, miR-140, miR-182, miR-182*, miR-724 | Down regulated 48 hpf | |||
| miR-10b, miR-10c, miR-16a, miR-16b, miR-18c, miR-22b, miR-210*, miR-217, miR-451, miR-455, miR-457b | Up regulated at 96 hpf | |||
| miR-10b, miR-10c | Up regulated - Regulates hox genes for anterior posterior development | |||
| miR-21, miR-27c, miR-34, miR-122, miR-192, miR-194a, miR-724 | Down regulated at 96 hpf | |||
| miR-29a, miR-29b | Down regulated - Important for neuron survival | |||
| miR-132, miR-212 | Down regulated - Important for neurogenesis | |||
| 1 μg/l venlafaxine and temperature (27° or 32 °C) |
Adults exposed for 21 days | miR-22b-3p, miR-301a | Down regulated - Gene enrichment shows metabolic pathways such as drug metabolism is altered; Observation not seen after recovery period | [88] |
| 0.5, 1, 2 μM tamoxifen 1, 2, 4 μM acetaminophen |
4 dpf for 24 h 3 mpf for 24 h |
miR-122 | Decreased miRNA-122 expression with increasing tamoxifen concentration in adults; Increased miRNA expression with acetaminophen exposure | [89] |
| 20−40 mM acetaminophen | Adult fish (5−24 mpf) exposed for 3 h | miR-122 | Dose dependent increase in circulating expression, which was associated with hepatotoxicity | [97] |
| 5 nM retinoic acid | 6−24 hpf | miR-19a, miR-19c, miR-19d | Decreased expression - Targets cyp26a1, which is associated with posterior curved body axis defects | [90] |
| 6.25 mg/l total composed of: ofloxacin (2.88 μmol/L), ciprofloxacin (3.14 μmol/L) enrofloxacin (2.89 μmol/L), doxycycline (2.03 μmol/L) chlortetracycline (2.02 μmol/L) oxytetracycline (2.26 μmol/L) 12.5 mg/l of combinations: ofloxacin (5.76 μmol/L), ciprofloxacin (6.28 μmol/L), enrofloxacin (5.79 μmol/L), doxycycline (4.06 μmol/L), chlortetracycline (4.04 μmol/L), oxytetracycline (4.52 μmol/L) |
4 hpf - 90 dpf | miR-124, miR-124−3p | Up regulated in 6.25 mg/l treatment; Down regulated in 12.5 mg/l | [30] |
| miR-22a-3p, miR-143, miR-10c-5p, miR-92−3p, miR-148, miR-152, miR-30e, miR-181a-3p, miR-95−5p, miR-184−3p | Dose dependent down regulation in eye and otocyst and decreased expression with ISH -Associated with retinal photoreceptor cysts and larval decreased volume of larval otocyst | |||
| miR-184, miR-96 | Down regulated in atrium - Associated with atrium tissue damage | |||
| miR-92a | Down regulated in the liver - Associated with hepatic parenchyma vacuolar degeneration; Down regulated and reduced expression in the heart - Associated with atrial muscle cysts | |||
| miR-10c | Dose dependent down regulation - Associated with reduced and swollen hepatocytes | |||
| 25 mg/l total, composed of ofloxacin (11.5 μmol/L), ciprofloxacin (12.6 μmol/L), enrofloxacin (11.6 μmol/L), doxycycline (8.1 μmol/L), chlortetracycline (8.1 μmol/L), and oxytetracycline (9.0 μmol/L) | 2−120 hpf | miR-184 | Dose dependent down regulation in otoliths - Associated with delayed otolith development; Rescue improved otic vesicle phenotype | [91] |
| miR-96 | Dose dependent down regulation in otolith - Injection improved otic vesicle phenotype; Rescue restores hair cell numbers following exposure | |||
| 6.25 mg/l total composed of: ofloxacin (2.88 μmol/L), ciprofloxacin (3.14 μmol/L) enrofloxacin (2.89 μmol/L), doxycycline (2.03 μmol/L) chlortetracycline (2.02 μmol/L) oxytetracycline (2.26 μmol/L). 12.5 mg/l of combinations: ofloxacin (5.76 μmol/L), ciprofloxacin (6.28 μmol/L), enrofloxacin (5.79 μmol/L), doxycycline (4.06 μmol/L), chlortetracycline (4.04 μmol/L), oxytetracycline (4.52 μmol/L) |
6 hpf - 3 mpf in F0 fish | miR-125b, miR-430c | Dose dependent increased expression in the F0 adult ovary | [92] |
| miR-124, miR-499 | Highly expressed in the liver, brain, gills, reproductive gills and cloaca of F1 larvae at 7 dpf | |||
| 12.5 or 25.0 mg/l total exposure to ofloxacin, ciprofloxacin, enrofloxacin, doxycycline, chlorotetracycline, and oxytetracycline |
6 hpf- 90 dpf | miR-125b, miR-144 | Increased expression in F0 adult and F1 larvae at 5 dpf - Target genes identified were ppardb, bcl2a, pparaa and pparda | [93] |
A developmental toxicity study was performed with another drug, valproic acid, which is used to treat epilepsy, mood related mental health issues, and migraines [12]. Alterations in differentially expressed miRNAs were then linked to the presence of morphological and behavioral alterations. Exposure to valproic acid resulted in 35 differentially expressed miRNAs, of which seven were confirmed with qPCR (Table 6). Some of the up regulated miRNAs are involved in cancer and cell cycling pathways, while the down regulated miRNAs are involved in MAP kinase signaling, insulin, and axon guidance [12]. This exposure also resulted in altered behavior in the larvae including circular swimming patterns, twitching, and a reduced startle response. Moreover, morphological abnormalities such as pericardial effusion, decreased pigmentation, and spinal curvature were also observed in the exposed larvae [12].
Another drug prescribed for depression and anxiety, venlafaxine, was assessed for altering miRNA profiles in adult zebrafish at two different temperatures [88]. This exposure resulted in down regulation of miR-22b-3p and miR-301a, but no effect of temperature was found. Both miR-22b and miR-301a are involved in metabolic pathways including drug metabolism [88]. In addition, miR-22 is a conserved miRNA important for proper gonadal function [94,95], which also agrees with the pathways identified to be altered in this study.
Nam and colleagues evaluated developmental liver toxicity with exposure to the drugs tamoxifen and acetaminophen [89]. Tamoxifen is a drug used to treat estrogen receptor-positive breast cancers [96], while acetaminophen is a drug used to commonly treats aches and pains. miR-122 was chosen as a potential biomarker for liver injury in this study. Tamoxifen was reported to reduce liver transparency in larvae, which was associated with decreased expression of miR-122, while acetaminophen exposure increased expression of miR-122 [89]. A separate study also evaluated miRNA expression profiles following an adult exposure to acetaminophen [97]. A dose dependent increase in miRNA expression in the liver was reported (Table 6). Liver toxicity due to acetaminophen and tamoxifen exposure is supported in this study, as well other studies in zebrafish and mice with differential expression of genes related to lipid metabolism and hepatotoxicity [[98], [99], [100]]. Several studies in other animal models have evaluated hepatotoxicity and altered expression of miR-122 after exposure to tamoxifen, acetaminophen, or valproic acid. miR-122 has been identified as a highly conserved and integral regulatory miRNA for genes involved in hepatic lipid metabolism, hepatocyte differentiation, and overall liver function, making it an attractive miRNA biomarker for liver damage [[101], [102], [103], [104]] as further demonstrated by these studies in the zebrafish (Table 6).
Retinoic Acid (RA) is important for vitamin A synthesis, neurogenesis, and vertebrate body planning. RA is tightly regulated by cyp26 enzymes [[105], [106], [107]]. In a developmental exposure in zebrafish larvae, RA resulted in down regulation of miR-19a, miR-19c, and miR-19d, which caused posterior curved body defects in larvae through dysregulation of cyp29a1 [90]. Increases in cyp26a1, were also observed in the tailbud with altered expression localized to the dorsal tail tip [90].
β-diketone antibiotics (DKA) are used in human and veterinarian practice to treat infectious diseases. A mixture toxicity study of six DKA drugs: ofloxacin, ciprofloxacin, enrofloxacin, doxycycline, chlortetracycline, and oxytetracycline was completed with exposures spanning embryonic development until adulthood in zebrafish using miRNA sequencing. The expression of miR-96 and miR-184 was found to be altered and at 120 hpf was validated using qPCR, deep sequencing, and in situ hybridization for expression in brain, gills, otoliths, and later line neuromast [30] due to their role in regulating various genes in the nervous system [108]. 20 miRNAs highly present and differentially altered were clustered and found to associate with cell cycle and nervous system pathways. DKA exposure was also associated with cyst formation in the adult retina [30]. A follow up study, focused on changes in miR-96 and miR-184 with a developmental exposure to the same pharmaceuticals found delayed otolith development with both miRNAs over or under expressed. The role of miR-96 in hair cell development was validated with a knockout and a rescue by administering a miR-96 mimic [91]. These findings suggest that DKA exposure causes changes in several miRNAs and that miR-96 and miR-184 are key targets leading to malformations of otolith and hair cell sensory organs. To better understand how chronic developmental exposure effects the health of exposed zebrafish (F0) and the subsequent generation (F1), a generational study evaluating developmental DKA toxicity was performed [92]. In the adult F0 generation, histopathological damage was seen in the ovary tissue, as well as increased expression of miR-125b and miR-430c. miR-125b is proposed as an indicator for ovarian cancers [109] and miR-430c has a known role in germ cell differentiation in zebrafish [110]. These functions align well with the histopathological damage and changes in miRNA expression observed in this study. F1 larvae at 7 dpf had high expression of miR-499 and miR-124 [92]. Expression of miR-124 and miR-499 was increased in several regions of larvae, including the brain. These miRNAs are reported to regulate CNS development and to serve as a biomarker for brain injury [111,112], suggesting DKAs may act as a neurotoxicant. A follow-up study focused on miR-125b and miR-144 with the same DKA exposures found increased expression of these miRNAs resulting in lipid retention in the F0 generation and lipid-metabolism-disorder due to more intense lipid accumulation in the F1 generation [93]. Potential gene targets identified were lipid metabolism genes ppardb and bcl2a, which had decreased expression in the brain, liver, and swim bladder in a dose dependent manner. miR-125′s involvement in lipid metabolism agrees with results in human cells and mice, though there is some debate on miR144′s role in lipid metabolism [113]. These results suggest that miR-125b plays a role in disease and pathology progression as well in metabolism and ovarian health. The collective results from these studies support DKAs as a developmental and reproductive toxicant in zebrafish with sex specific health outcomes at the genetic, tissue, and morphological level. Adverse health outcomes of DKA exposure can also impact the developmental stages of the subsequent generation [30,91,92,93,114].
7. Current limitations and future directions
miRNAs are a critical epigenetic mechanism in the modulation of gene expression in developmental and disease pathways. As a result, miRNAs are a target of interest in toxicity studies. As discussed in this review, zebrafish are a good candidate model organism to better understand toxin and xenobiotic toxicity and are being used to readily identify molecular, physiological, and behavioral alterations. More recently, researchers are beginning to apply the zebrafish to define miRNA regulatory mechanisms of toxicity and are grasping a better understanding of strengths and limitations as publications in this research area continue to grow. Currently, many of the studies using zebrafish have applied -omic based approaches to identify miRNAs altered by xenobiotic exposures, but as noted above some studies applying multiple -omic platforms report inconsistent results bringing to question reproducibility. As such, questions still remain on the accuracy of using miRNAs as toxicity biomarkers, but these questions are being addressed as the literature continues to grow on the mechanisms governing miRNA alterations following xenobiotic exposure. For instance, as noted above some studies are beginning to further apply mechanistic approaches in their studies such as performing rescue validation assays to define miRNA mechanisms of toxicity providing greater confidence.
With the limitations noted above, it is expected that future studies will also help to elucidate heritability of altered miRNAs. So far, fluoxetine exposure has been shown to alter miRNAs in the gametes of exposed females and a developmental exposure to DKAs caused developmental health outcomes seen in the next generation. Moreover, it is expected that studies evaluating chemical mixtures and miRNA dysregulation will expand since most often chemical exposures occur in a mixture state with multiple chemicals. To date, triazophos/fipronil mixtures, DKA mixtures, silica nanoparticles, and MeHg mixtures have been performed with the zebrafish. Overall, as miRNA toxicity studies continue it is expected that more answers will be revealed when considering reproducibility and accuracy in defining how miRNAs regulate toxicity and the zebrafish presents as an excellent complementary vertebrate model for these studies.
Funding
This work was supported by the National Institutes of Health, National Institute of Environmental Health Sciences (R03 ES030545) and by Purdue University as part of AgSEED Crossroads funding to support Indiana’s Agriculture and Rural Development.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Kimmel C.B., Ballard W.W., Kimmel S.R., Ullmann B., Schilling T.F. Stages of embryonic development of the zebrafish. Dev. Dyn. 1995;203:253–310. doi: 10.1002/aja.1002030302. [DOI] [PubMed] [Google Scholar]
- 2.Tegelenbosch R.A.J., Noldus L.P.J.J., Richardson M.K., Ahmad F. Zebrafish embryos and larvae in behavioural assays. Behaviour. 2012;149:1241–1281. doi: 10.1163/1568539X-00003020. [DOI] [Google Scholar]
- 3.Garcia G.R., Noyes P.D., Tanguay R.L. Advancements in zebrafish applications for 21st century toxicology. Pharmacol. Ther. 2016;161:11–21. doi: 10.1016/j.pharmthera.2016.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kokel D., Bryan J., Laggner C., White R., Cheung CYJ Mateus R. Rapid behavior-based identification of neuroactive small molecules in the zebrafish. Nat. Chem. Biol. 2010;6:231–237. doi: 10.1038/nchembio.307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huang P., Xiao A., Zhou M., Zhu Z., Lin S., Zhang B. Heritable gene targeting in zebrafish using customized TALENs. Nat. Biotechnol. 2011;29:699–700. doi: 10.1038/nbt.1939. [DOI] [PubMed] [Google Scholar]
- 6.Peng Y., Clark K.J., Campbell J.M., Panetta M.R., Guo Y., Ekker S.C. Making designer mutants in model organisms. Development. 2014;141:4042–4054. doi: 10.1242/dev.102186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Howe K., Clark M.D., Torroja C.F., Torrance J., Berthelot C., Muffato M. The zebrafish reference genome sequence and its relationship to the human genome. Nature. 2013;496:498–503. doi: 10.1038/nature12111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Goldstone J.V., McArthur A.G., Kubota A., Zanette J., Parente T., Jönsson M.E. Identification and developmental expression of the full complement of Cytochrome P450 genes in Zebrafish. BMC Genomics. 2010;11(643) doi: 10.1186/1471-2164-11-643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cassar S., Adatto I., Freeman J.L., Gamse J.T., Iturria I., Lawrence C. Use of zebrafish in drug Discovery toxicology. Chem. Res. Toxicol. 2020;33:95–118. doi: 10.1021/acs.chemrestox.9b00335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hallare A., Nagel K., Köhler H.-R., Triebskorn R. Comparative embryotoxicity and proteotoxicity of three carrier solvents to zebrafish (danio rerio) embryos. Ecotoxicol. Environ. Saf. 2006;63:378–388. doi: 10.1016/j.ecoenv.2005.07.006. [DOI] [PubMed] [Google Scholar]
- 11.Zon L.I., Peterson R.T. In vivo drug discovery in the zebrafish. Nat. Rev. Drug Discov. 2005;4:35–44. doi: 10.1038/nrd1606. [DOI] [PubMed] [Google Scholar]
- 12.Aluru N., Deak K.L., Jenny M.J., Hahn M.E. Developmental exposure to valproic acid alters the expression of microRNAs involved in neurodevelopment in zebrafish. Neurotoxicol. Teratol. 2013;40:46–58. doi: 10.1016/j.ntt.2013.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bailey J., Oliveri A., Levin E.D. zebrafish model systems for developmental neurobehavioral toxicology: zebrafish developmental neurobehavioral toxicology. Birth Defects. Res. Part C Embryo. Today Rev. 2013;99:14–23. doi: 10.1002/bdrc.21027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Horzmann K.A., Freeman J.L. Making waves: New developments in toxicology with the zebrafish. Toxicol. Sci. 2018;163:5–12. doi: 10.1093/toxsci/kfy044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.He J.-H., Gao J.-M., Huang C.-J., Li C.-Q. Zebrafish models for assessing developmental and reproductive toxicity. Neurotoxicol. Teratol. 2014;42:35–42. doi: 10.1016/j.ntt.2014.01.006. [DOI] [PubMed] [Google Scholar]
- 16.Horzmann K.A., Reidenbach L.S., Thanki D.H., Winchester A.E., Qualizza B.A., Ryan G.A. Embryonic atrazine exposure elicits proteomic, behavioral, and brain abnormalities with developmental time specific gene expression signatures. J. Proteomics. 2018;186:71–82. doi: 10.1016/j.jprot.2018.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jirtle R.L., Skinner M.K. Environmental epigenomics and disease susceptibility. Nat. Rev. Genet. 2007;8:253–262. doi: 10.1038/nrg2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wirbisky S.E., Weber G.J., Schlotman K.E., Sepúlveda M.S., Freeman J.L. Embryonic atrazine exposure alters zebrafish and human miRNAs associated with angiogenesis, cancer, and neurodevelopment. Food Chem. Toxicol. 2016;98:25–33. doi: 10.1016/j.fct.2016.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Freeman J.L., Weber G.J., Sepúlveda M.S. Fishing for microRNAs in toxicology. In: Sahu S.C., editor. microRNAs in Toxicology and Medicine. John Wiley & Sons, Ltd; Chichester, UK: 2013. pp. 49–75. [Google Scholar]
- 20.Hu M., Jovanović B., Palić D. In silico prediction of MicroRNA role in regulation of Zebrafish (Danio rerio) responses to nanoparticle exposure. Toxicol. in Vitro. 2019;60:187–202. doi: 10.1016/j.tiv.2019.05.014. [DOI] [PubMed] [Google Scholar]
- 21.Qiao J., Du Y., Yu J., Guo J. MicroRNAs as potential biomarkers of insecticide exposure: a Review. Chem. Res. Toxicol. Acs. Chemrestox. 2019 doi: 10.1021/acs.chemrestox.9b00236. [DOI] [PubMed] [Google Scholar]
- 22.Lagos-Quintana M., Rauhut R., Yalcin A., Meyer J., Lendeckel W., Tuschl T. Identification of tissue-specific MicroRNAs from mouse. Curr. Biol. 2002;12:735–739. doi: 10.1016/S0960-9822(02)00809-6. [DOI] [PubMed] [Google Scholar]
- 23.Wu M., Wu D., Wang C., Guo Z., Li B., Zuo Z. Hexabromocyclododecane exposure induces cardiac hypertrophy and arrhythmia by inhibiting miR-1 expression via up-regulation of the homeobox gene Nkx2.5. J. Hazard. Mater. 2016;302:304–313. doi: 10.1016/j.jhazmat.2015.10.004. [DOI] [PubMed] [Google Scholar]
- 24.Jenny M.J., Aluru N., Hahn M.E. Effects of short-term exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin on microRNA expression in zebrafish embryos. Toxicol. Appl. Pharmacol. 2012;264:262–273. doi: 10.1016/j.taap.2012.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wienholds E., Plasterk R.H.A. MicroRNA function in animal development. FEBS Lett. 2005;579:5911–5922. doi: 10.1016/j.febslet.2005.07.070. [DOI] [PubMed] [Google Scholar]
- 26.Lim L.P., Lau N.C., Garrett-Engele P., Grimson A., Schelter J.M., Castle J. Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature. 2005;433:769–773. doi: 10.1038/nature03315. [DOI] [PubMed] [Google Scholar]
- 27.Lawrie C.H., Gal S., Dunlop H.M., Pushkaran B., Liggins A.P., Pulford K. Detection of elevated levels of tumour-associated microRNAs in serum of patients with diffuse large B-cell lymphoma. Br. J. Haematol. 2008;141:672–675. doi: 10.1111/j.1365-2141.2008.07077.x. [DOI] [PubMed] [Google Scholar]
- 28.Duan J., Yu Y., Li Y., Jing L., Yang M., Wang J. Comprehensive understanding of PM2.5 on gene and microRNA expression patterns in zebrafish (danio rerio) model. Sci. Total Environ. 2017;586:666–674. doi: 10.1016/j.scitotenv.2017.02.042. [DOI] [PubMed] [Google Scholar]
- 29.Hu H., Shi Y., Zhang Y., Wu J., Asweto C.O., Feng L. Comprehensive gene and microRNA expression profiling on cardiovascular system in zebrafish co-exposured of SiNPs and MeHg. Sci. Total Environ. 2017;607–608:795–805. doi: 10.1016/j.scitotenv.2017.07.036. [DOI] [PubMed] [Google Scholar]
- 30.Li J., Liu J., Zhang Y., Wang X., Li W., Zhang H. Screening on the differentially expressed miRNAs in zebrafish (danio rerio) exposed to trace β-diketone antibiotics and their related functions. Aquat. Toxicol. 2016;178:27–38. doi: 10.1016/j.aquatox.2016.07.009. [DOI] [PubMed] [Google Scholar]
- 31.Martinez R., Vera-Chang M.N., Haddad M., Zon J., Navarro-Martin L., Trudeau V.L. Developmental fluoxetine exposure in zebrafish reduces offspring basal cortisol concentration via life stage-dependent maternal transmission. C.S. Rosenfeld. ed PLOS ONE. 2019;14:e0212577. doi: 10.1371/journal.pone.0212577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wu T.-S., Yang J.-J., Yu F.-Y., Liu B.-H. Cardiotoxicity of mycotoxin citrinin and involvement of MicroRNA-138 in zebrafish embryos. Toxicol. Sci. 2013;136:402–412. doi: 10.1093/toxsci/kft206. [DOI] [PubMed] [Google Scholar]
- 33.Morton S.U., Scherz P.J., Cordes K.R., Ivey K.N., Stainier DYR Srivastava D. microRNA-138 modulates cardiac patterning during embryonic development. Proc. Natl. Acad. Sci. 2008;105:17830–17835. doi: 10.1073/pnas.0804673105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wu T.-S., Lin Y.-T., Huang Y.-T., Yu F.-Y., Liu B.-H. Ochratoxin a triggered intracerebral hemorrhage in embryonic zebrafish: involvement of microRNA-731 and prolactin receptor. Chemosphere. 2020;242:125143. doi: 10.1016/j.chemosphere.2019.125143. [DOI] [PubMed] [Google Scholar]
- 35.Huang C.-X., Chen N., Wu X.-J., Huang C.-H., He Y., Tang R. The zebrafish miR-462/miR-731 cluster is induced under hypoxic stress via hypoxia-inducible factor 1α and functions in cellular adaptations. FASEB J. 2015;29:4901–4913. doi: 10.1096/fj.14-267104. [DOI] [PubMed] [Google Scholar]
- 36.Wu T.-S., Yang J.-J., Wang Y.-W., Yu F.-Y., Liu B.-H. Mycotoxin ochratoxin A disrupts renal development via a miR-731/prolactin receptor axis in zebrafish. Toxicol. Res. 2016;5:519–529. doi: 10.1039/C5TX00360A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhao Y., Xiong Q., Xie P. Analysis of MicroRNA expression in embryonic developmental toxicity induced by MC-RR. C. Schönbach. Ed PLoS ONE. 2011;6:e22676. doi: 10.1371/journal.pone.0022676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.US EPA O . US EPA; 2018. Cyanobacterial Harmful Algal Blooms (CyanoHABs) in Water Bodies. Available: https://www.epa.gov/cyanohabs [Accessed 19 February 2020] [Google Scholar]
- 39.Zhong Q., Sun F., Wang W., Xiao W., Zhao X., Gu K. Water metabolism dysfunction via renin-angiotensin system activation caused by liver damage in mice treated with microcystin-RR. Toxicol. Lett. 2017;273:86–96. doi: 10.1016/j.toxlet.2017.03.019. [DOI] [PubMed] [Google Scholar]
- 40.Fish J.E., Santoro M.M., Morton S.U., Yu S., Wythe J.D., Bruneau B.G. 2009. miR-126 Regulates Angiogenic Signaling and Vascular Integrity. 26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang S., Aurora A.B., Johnson B.A., Qi X., McAnally J., Hill J.A. The endothelial-specific MicroRNA miR-126 governs vascular integrity and angiogenesis. Dev. Cell. 2008;15:261–271. doi: 10.1016/j.devcel.2008.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Giraldez A.J. MicroRNAs regulate brain morphogenesis in zebrafish. Science. 2005;308:833–838. doi: 10.1126/science.1109020. [DOI] [PubMed] [Google Scholar]
- 43.US EPA O . US EPA; 2016. Particulate Matter (PM) Basics. Available: https://www.epa.gov/pm-pollution/particulate-matter-pm-basics [Sccessed 15 January 2020] [Google Scholar]
- 44.Martinelli N., Olivieri O., Girelli D. Air particulate matter and cardiovascular disease: a narrative review. Eur. J. Intern. Med. 2013;24:295–302. doi: 10.1016/j.ejim.2013.04.001. [DOI] [PubMed] [Google Scholar]
- 45.Zou M., Wang F., Gao R., Wu J., Ou Y., Chen X. Autophagy inhibition of hsa-miR-19a-3p/19b-3p by targeting TGF-β R II during TGF-β1-induced fibrogenesis in human cardiac fibroblasts. Sci. Rep. 2016;6:24747. doi: 10.1038/srep24747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fröhlich E., Roblegg E. Models for oral uptake of nanoparticles in consumer products. Toxicology. 2012;291:10–17. doi: 10.1016/j.tox.2011.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gusscott S., Kuchenbauer F., Humphries R.K., Weng A.P. Notch-mediated repression of miR-223 contributes to IGF1R regulation in T-ALL. Leuk. Res. 2012;36:905–911. doi: 10.1016/j.leukres.2012.02.013. [DOI] [PubMed] [Google Scholar]
- 48.Huang H., Zhang K., Zhou Y., Ding X., Yu L., Zhu G. MicroRNA-155 targets cyb561d2 in zebrafish in response to fipronil exposure: MicroRNA-155 Targets cyb561d2 in Zebrafish. Environ. Toxicol. 2016;31:877–886. doi: 10.1002/tox.22099. [DOI] [PubMed] [Google Scholar]
- 49.Zhou Y., Huang H., Zhang K., Ding X., Jia L., Yu L. miRNA-216 and miRNA-499 target cyb561d2 in zebrafish in response to fipronil exposure. Environ. Toxicol. Pharmacol. 2016;45:98–107. doi: 10.1016/j.etap.2016.05.019. [DOI] [PubMed] [Google Scholar]
- 50.Jia L., Zhang D., Huang H., Zhou Y., Zhou S., Guo J. Triazophos-induced toxicity in zebrafish: miRNA-217 inhibits nup43. Toxicol. Res. 2018;7:913–922. doi: 10.1039/C8TX00065D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wang a X., Zhou a S., Ding X., Zhu G., Guo J. Effect of triazophos, fipronil and their mixture on miRNA expression in adult zebrafish. J. Environ. Sci. Health Part B. 2010;45:648–657. doi: 10.1080/03601234.2010.502435. [DOI] [PubMed] [Google Scholar]
- 52.Wirbisky S.E., Sepúlveda M.S., Weber G.J., Jannasch A.S., Horzmann K.A., Freeman J.L. Embryonic atrazine exposure elicits alterations in genes associated with neuroendocrine function in adult male zebrafish. Toxicol. Sci. 2016;153:149–164. doi: 10.1093/toxsci/kfw115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Meister J., Schmidt M.H.H. miR-126 and miR-126*: new Players in Cancer. Sci World J. 2010;10:2090–2100. doi: 10.1100/tsw.2010.198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Woltering J.M., Durston A.J. MiR-10 represses HoxB1a and HoxB3a in zebrafish. D. Raible. Ed PLoS ONE. 2008;3:e1396. doi: 10.1371/journal.pone.0001396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sun E., Shi Y. MicroRNAs: small molecules with big roles in neurodevelopment and diseases. Exp. Neurol. 2015;268:46–53. doi: 10.1016/j.expneurol.2014.08.005. [DOI] [PubMed] [Google Scholar]
- 56.Lin J., Wang C., Liu J., Dahlgren R.A., Ai W., Zeng A. Up-stream mechanisms for up-regulation of miR-125b from triclosan exposure to zebrafish (danio rerio) Aquat. Toxicol. 2017;193:256–267. doi: 10.1016/j.aquatox.2017.10.021. [DOI] [PubMed] [Google Scholar]
- 57.Liu J., Xiang C., Huang W., Mei J., Sun L., Ling Y. Neurotoxicological effects induced by up-regulation of miR-137 following triclosan exposure to zebrafish (Danio rerio) Aquat. Toxicol. 2019;206:176–185. doi: 10.1016/j.aquatox.2018.11.017. [DOI] [PubMed] [Google Scholar]
- 58.Zhang L., Li Y., Zeng H., Wei J., Wan Y., Chen J. MicroRNA expression changes during zebrafish development induced by perfluorooctane sulfonate. J. Appl. Toxicol. 2011;31:210–222. doi: 10.1002/jat.1583. [DOI] [PubMed] [Google Scholar]
- 59.Wang L., Bammler T.K., Beyer R.P., Gallagher E.P. Copper-induced deregulation of microRNA expression in the zebrafish olfactory system. Environ. Sci. Technol. 2013;47:7466–7474. doi: 10.1021/es400615q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lee J., Kho Y., Kim P.-G., Ji K. Exposure to bisphenol S alters the expression of microRNA in male zebrafish. Toxicol. Appl. Pharmacol. 2018;338:191–196. doi: 10.1016/j.taap.2017.11.019. [DOI] [PubMed] [Google Scholar]
- 61.Renaud L., da Silveira W.A., Hazard E.S., Simpson J., Falcinelli S., Chung D. The plasticizer bisphenol a perturbs the hepatic epigenome: a systems level analysis of the miRNome. Genes. 2017;8(269) doi: 10.3390/genes8100269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ju L., Zhou Z., Jiang B., Lou Y., Zhang Z. miR-21 is involved in skeletal deficiencies of zebrafish embryos exposed to polychlorinated biphenyls. Environ. Sci. Pollut. Res. 2017;24:886–891. doi: 10.1007/s11356-016-7874-8. [DOI] [PubMed] [Google Scholar]
- 63.Biyashev D., Veliceasa D., Topczewski J., Topczewska J.M., Mizgirev I., Vinokour E. miR-27b controls venous specification and tip cell fate. Blood. 2012;119:2679–2687. doi: 10.1182/blood-2011-07-370635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kara N., Wei C., Commanday A.C., Patton J.G. miR-27 regulates chondrogenesis by suppressing focal adhesion kinase during pharyngeal arch development. Dev. Biol. 2017;429:321–334. doi: 10.1016/j.ydbio.2017.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Haggard D.E., Noyes P.D., Waters K.M., Tanguay R.L. Phenotypically anchored transcriptome profiling of developmental exposure to the antimicrobial agent, triclosan, reveals hepatotoxicity in embryonic zebrafish. Toxicol. Appl. Pharmacol. 2016;308:32–45. doi: 10.1016/j.taap.2016.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kim J., Oh H., Ryu B., Kim U., Lee J.M., Jung C.-R. Triclosan affects axon formation in the neural development stages of zebrafish embryos (danio rerio) Environ. Pollut. 2018;236:304–312. doi: 10.1016/j.envpol.2017.12.110. [DOI] [PubMed] [Google Scholar]
- 67.US EPA. O . US EPA; 2014. Flame Retardant Alternatives for Hexabromocyclododecane (HBCD) Final Report. [Google Scholar]
- 68.Wu M., Zuo Z., Li B., Huang L., Chen M., Wang C. Effects of low-level hexabromocyclododecane (HBCD) exposure on cardiac development in zebrafish embryos. Ecotoxicology. 2013;22:1200–1207. doi: 10.1007/s10646-013-1107-4. [DOI] [PubMed] [Google Scholar]
- 69.Budde H., Schmitt S., Fitzner D., Opitz L., Salinas-Riester G., Simons M. Control of oligodendroglial cell number by the miR-17-92 cluster. Development. 2010;137:2127–2132. doi: 10.1242/dev.050633. [DOI] [PubMed] [Google Scholar]
- 70.Johansson N., Eriksson P., Viberg H. Neonatal exposure to PFOS and PFOA in mice results in changes in proteins which are important for neuronal growth and synaptogenesis in the developing brain. Toxicol. Sci. 2009;108:412–418. doi: 10.1093/toxsci/kfp029. [DOI] [PubMed] [Google Scholar]
- 71.Acosta D., da S., Danielle N.M., Altenhofen S., Luzardo M.D., Costa P.G., Bianchini A. Copper at low levels impairs memory of adult zebrafish (danio rerio) and affects swimming performance of larvae. Comp Biochem Physiol Part C Toxicol Pharmacol. 2016;185–186:122–130. doi: 10.1016/j.cbpc.2016.03.008. [DOI] [PubMed] [Google Scholar]
- 72.Johnson A., Carew E., Sloman K. The effects of copper on the morphological and functional development of zebrafish embryos. Aquat. Toxicol. 2007;84:431–438. doi: 10.1016/j.aquatox.2007.07.003. [DOI] [PubMed] [Google Scholar]
- 73.Liu X.S., Fan B.Y., Pan W.L., Li C., Levin A.M., Wang X. Identification of miRNomes associated with adult neurogenesis after stroke using argonaute 2-based RNA sequencing. RNA Biol. 2017;14:488–499. doi: 10.1080/15476286.2016.1196320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Papaioannou G., Inloes J.B., Nakamura Y., Paltrinieri E., Kobayashi T. let-7 and miR-140 microRNAs coordinately regulate skeletal development. Proc. Natl. Acad. Sci. 2013;110:E3291–E3300. doi: 10.1073/pnas.1302797110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Conkle J.L., Báez Del Valle C.D., Turner J.W. Are We Underestimating Microplastic Contamination in Aquatic Environments? Environ. Manage. 2018;61:1–8. doi: 10.1007/s00267-017-0947-8. [DOI] [PubMed] [Google Scholar]
- 76.US EPA O . US EPA; 2015. Learn About Polychlorinated Biphenyls (PCBs) Available: https://www.epa.gov/pcbs/learn-about-polychlorinated-biphenyls-pcbs [Accessed 18 February 2020] [Google Scholar]
- 77.Romero A.N., Herlin M., Finnilä M., Korkalainen M., Håkansson H., Viluksela M. Skeletal and dental effects on rats following in utero/lactational exposure to the non-dioxin-like polychlorinated biphenyl PCB 180. H.-J. Lehmler. Ed PLOS ONE. 2017;12 doi: 10.1371/journal.pone.0185241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hu C.-H., Sui B.-D., Du F-Y Shuai Y., Zheng C.-X., Zhao P. miR-21 deficiency inhibits osteoclast function and prevents bone loss in mice. Sci. Rep. 2017;7(43191) doi: 10.1038/srep43191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Soares A.R., Pereira P.M., Ferreira V., Reverendo M., Simões J., Bezerra A.R. Ethanol exposure induces upregulation of specific MicroRNAs in zebrafish embryos. Toxicol. Sci. 2012;127:18–28. doi: 10.1093/toxsci/kfs068. [DOI] [PubMed] [Google Scholar]
- 80.Pappalardo-Carter D.L., Balaraman S., Sathyan P., Carter E.S., W-JA Chen, Miranda R.C. Suppression and epigenetic regulation of MiR-9 contributes to ethanol teratology: evidence from zebrafish and murine fetal neural stem cell models. Alcohol Clin. Exp. Res N/A-N/A. 2013 doi: 10.1111/acer.12139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Tal T.L., Franzosa J.A., Tilton S.C., Philbrick K.A., Iwaniec U.T., Turner R.T. MicroRNAs control neurobehavioral development and function in zebrafish. FASEB J. 2012;26:1452–1461. doi: 10.1096/fj.11-194464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Khayrullin A., Smith L., Mistry D., Dukes A., Pan Y.A., Hamrick M.W. Chronic alcohol exposure induces muscle atrophy (myopathy) in zebrafish and alters the expression of microRNAs targeting the Notch pathway in skeletal muscle. Biochem. Biophys. Res. Commun. 2016;479:590–595. doi: 10.1016/j.bbrc.2016.09.117. [DOI] [PubMed] [Google Scholar]
- 83.Benotti M.J., Trenholm R.A., Vanderford B.J., Holady J.C., Stanford B.D., Snyder S.A. Pharmaceuticals and endocrine disrupting compounds in U.S. Drinking water. Environ. Sci. Technol. 2009;43:597–603. doi: 10.1021/es801845a. [DOI] [PubMed] [Google Scholar]
- 84.Cunningham V.L., Buzby M., Hutchinson T., Mastrocco F., Parke N., Roden N. Effects of human pharmaceuticals on aquatic life: next steps. Environ. Sci. Technol. 2006;40:3456–3462. doi: 10.1021/es063017b. [DOI] [PubMed] [Google Scholar]
- 85.Grove K., Lewis A.J., Galbally M. Prenatal antidepressant exposure and child motor development: a meta-analysis. Pediatrics. 2018;142 doi: 10.1542/peds.2018-0356. [DOI] [PubMed] [Google Scholar]
- 86.Wu M., Liu S., Hu L., Qu H., Pan C., Lei P. Global transcriptomic analysis of zebrafish in response to embryonic exposure to three antidepressants, amitriptyline, fluoxetine and mianserin. Aquat. Toxicol. 2017;192:274–283. doi: 10.1016/j.aquatox.2017.09.027. [DOI] [PubMed] [Google Scholar]
- 87.Craig P.M., Trudeau V.L., Moon T.W. Profiling hepatic microRNAs in zebrafish: fluoxetine exposure mimics a fasting response that targets AMP-activated protein kinase (AMPK). E. Buratti. Ed PLoS ONE. 2014;9:e95351. doi: 10.1371/journal.pone.0095351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ikert H., Craig P.M. Chronic exposure to venlafaxine and increased water temperature reversibly alters microRNA in zebrafish gonads (Danio rerio) Comp. Biochem. Physiol. Part D Genomics Proteomics. 2020;33:100634. doi: 10.1016/j.cbd.2019.100634. [DOI] [PubMed] [Google Scholar]
- 89.Nam H.-S., Hwang K.-S., Jeong Y.-M., Ryu J.-I., Choi T.-Y., Bae M.-A. Expression of miRNA-122 induced by liver toxicants in zebrafish. Biomed Res. Int. 2016;2016:1–7. doi: 10.1155/2016/1473578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Franzosa J.A., Bugel S.M., Tal T.L., La Du J.K., Tilton S.C., Waters K.M. Retinoic acid-dependent regulation of miR-19 expression elicits vertebrate axis defects. FASEB J. 2013;27:4866–4876. doi: 10.1096/fj.12-225524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Li J., Ling Y., Huang W., Sun L., Li Y., Wang C. Regulatory mechanisms of miR-96 and miR-184 abnormal expressions on otic vesicle development of zebrafish following exposure to β-diketone antibiotics. Chemosphere. 2019;214:228–238. doi: 10.1016/j.chemosphere.2018.09.118. [DOI] [PubMed] [Google Scholar]
- 92.Zheng Y., Lin J., Li J., Zhang H., Ai W., Wang X. Effects of β-diketone antibiotics on F1-zebrafish (Danio rerio) based on high throughput miRNA sequencing under exposure to parents. Chemosphere. 2016;164:41–51. doi: 10.1016/j.chemosphere.2016.07.057. [DOI] [PubMed] [Google Scholar]
- 93.Wang X., Zheng Y., Ma Y., Du L., Chu F., Gu H. Lipid metabolism disorder induced by up-regulation of miR-125b and miR-144 following β-diketone antibiotic exposure to F0-zebrafish (danio rerio) Ecotoxicol. Environ. Saf. 2018;164:243–252. doi: 10.1016/j.ecoenv.2018.08.027. [DOI] [PubMed] [Google Scholar]
- 94.Guo Y., Sun J., Lai D. Role of microRNAs in premature ovarian insufficiency. Reprod. Biol. Endocrinol. 2017;15(38) doi: 10.1186/s12958-017-0256-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Nagaraja A.K., Creighton C.J., Yu Z., Zhu H., Gunaratne P.H., Reid J.G. A link between mir-100 and FRAP1/mTOR in clear cell ovarian cancer. Mol. Endocrinol. 2010;24:447–463. doi: 10.1210/me.2009-0295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Porta M., Last J.M.L.M. Tamoxifen. In: Last J.M., editor. A Dictionary of Public Health. Oxford University Press; 2018. [Google Scholar]
- 97.Vliegenthart A.D.B., Starkey Lewis P., Tucker C.S., Del Pozo J., Rider S., Antoine D.J. Retro-orbital blood acquisition facilitates circulating microRNA measurement in zebrafish with paracetamol hepatotoxicity. Zebrafish. 2014;11:219–226. doi: 10.1089/zeb.2013.0912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Lee M.-H., Kim J.-W., Kim J.-H., Kang K.-S., Kong G., Lee M.-O. Gene expression profiling of murine hepatic steatosis induced by tamoxifen. Toxicol. Lett. 2010;199:416–424. doi: 10.1016/j.toxlet.2010.10.008. [DOI] [PubMed] [Google Scholar]
- 99.North T.E., Babu I.R., Vedder L.M., Lord A.M., Wishnok J.S., Tannenbaum S.R. PGE2-regulated wnt signaling and N-acetylcysteine are synergistically hepatoprotective in zebrafish acetaminophen injury. Proc. Natl. Acad. Sci. 2010;107:17315–17320. doi: 10.1073/pnas.1008209107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Reilly T.P., Bourdi M., Brady J.N., Pise-Masison C.A., Radonovich M.F., George J.W. Expression profiling of acetaminophen liver toxicity in mice using microarray technology. Biochem. Biophys. Res. Commun. 2001;282:321–328. doi: 10.1006/bbrc.2001.4576. [DOI] [PubMed] [Google Scholar]
- 101.Landgraf P., Rusu M., Sheridan R., Sewer A., Iovino N., Aravin A. A mammalian microRNA expression atlas based on small RNA library sequencing. Cell. 2007;129:1401–1414. doi: 10.1016/j.cell.2007.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Thakral S., Ghoshal K. miR-122 is a unique molecule with Great potential in diagnosis, prognosis of liver disease, and therapy both as miRNA mimic and antimir. Curr. Gene Ther. 2015;15:142–150. doi: 10.2174/1566523214666141224095610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ward J., Kanchagar C., Veksler-Lublinsky I., Lee R.C., McGill M.R., Jaeschke H. Circulating microRNA profiles in human patients with acetaminophen hepatotoxicity or ischemic hepatitis. Proc. Natl. Acad. Sci. 2014;111:12169–12174. doi: 10.1073/pnas.1412608111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Wienholds E. MicroRNA expression in zebrafish embryonic development. Science. 2005;309:310–311. doi: 10.1126/science.1114519. [DOI] [PubMed] [Google Scholar]
- 105.Pennimpede T., Cameron D.A., MacLean G.A., Li H., Abu‐Abed S., Petkovich M. The role of CYP26 enzymes in defining appropriate retinoic acid exposure during embryogenesis. Birt. Defects Res. A Clin. Mol. Teratol. 2010;88:883–894. doi: 10.1002/bdra.20709. [DOI] [PubMed] [Google Scholar]
- 106.Sakai Y. The retinoic acid-inactivating enzyme CYP26 is essential for establishing an uneven distribution of retinoic acid along the anterio-posterior axis within the mouse embryo. Genes Dev. 2001;15:213–225. doi: 10.1101/gad.851501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Thatcher J.E., Isoherranen N. The role of CYP26 enzymes in retinoic acid clearance. Expert Opin. Drug Metab. Toxicol. 2009;5:875–886. doi: 10.1517/17425250903032681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Liang X., Meng W., Hu L., Li L., Xing H., Xie G. MicroRNA-184 modulates human central nervous system lymphoma cells growth and invasion by targeting iASPP: MicroRNA-184 modulates human central nervous system. J. Cell. Biochem. 2017;118:2645–2653. doi: 10.1002/jcb.25856. [DOI] [PubMed] [Google Scholar]
- 109.Zuberi M., Khan I., Mir R., Gandhi G., Ray P.C., Saxena A. Utility of serum miR-125b as a diagnostic and prognostic indicator and its Alliance with a panel of tumor suppressor genes in epithelial ovarian cancer. R.B. Ray. ed PLOS ONE. 2016;11:e0153902. doi: 10.1371/journal.pone.0153902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Mishima Y., Giraldez A.J., Takeda Y., Fujiwara T., Sakamoto H., Schier A.F. Differential regulation of germline mRNAs in Soma and germ cells by zebrafish miR-430. Curr. Biol. 2006;16:2135–2142. doi: 10.1016/j.cub.2006.08.086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ristori E., Nicoli S. Essentials of Noncoding RNA in Neuroscience. Elsevier; 2017. Comparative functions of miRNAs in embryonic neurogenesis and neuronal network formation; pp. 265–282. [Google Scholar]
- 112.Yang T., Song J., Bu X., Wang C., Wu J., Cai J. Elevated serum miR-93, miR-191, and miR-499 are noninvasive biomarkers for the presence and progression of traumatic brain injury. J. Neurochem. 2016;137:122–129. doi: 10.1111/jnc.13534. [DOI] [PubMed] [Google Scholar]
- 113.Li D., Wang X., Lan X., Li Y., Liu L., Yi J. Down-regulation of miR-144 elicits proinflammatory cytokine production by targeting toll-like receptor 2 in nonalcoholic steatohepatitis of high-fat-diet-induced metabolic syndrome E3 rats. Mol. Cell. Endocrinol. 2015;402:1–12. doi: 10.1016/j.mce.2014.12.007. [DOI] [PubMed] [Google Scholar]
- 114.Yin X., Wang H., Zhang Y., Dahlgren R.A., Zhang H., Shi M. Toxicological assessment of trace β-diketone antibiotic mixtures on zebrafish (danio rerio) by proteomic analysis. X. Xu. Ed PLoS ONE. 2014;9:e102731. doi: 10.1371/journal.pone.0102731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.FDA issues final rule on safety and effectiveness of antibacterial soaps | FDA. Available: https://www.fda.gov/news-events/press-announcements/fda-issues-final-rule-safety-and-effectiveness-antibacterial-soaps [Accessed 18 February 2020].
- 116.IGF1R Gene - GeneCards | IGF1R Protein | IGF1R Antibody. Available: https://www.genecards.org/cgi-bin/carddisp.pl?gene=IGF1R [Accessed 18 February 2020].