Abstract
Background:
Continuous glucose monitors (CGMs) enable people with diabetes to proactively manage their blood glucose and reduce the risk of hypoglycemia. Commercially available CGMs utilize percutaneous electrodes that, after days to weeks of implantation, are subjected to the foreign body response that severely reduces sensor accuracy. The previous work demonstrated the use of hydrogels containing a glucose-responsive viologen that quenches a nearby fluorophore. Here, we investigate the immobilization of this sensing motif onto a nanoparticle surface and optimize local surface concentrations for optical glucose sensing.
Methods:
A viologen quencher-fluorescent dye system was incorporated into poly(2-hydroethyl methacrylate) hydrogels in varying quantities to assess the effect of quencher-fluorophore concentration on glucose responsiveness. The sensing motif was then immobilized onto silica nanoparticles by carbodiimide chemistry. Nanosensors with a range of dye and quencher concentrations were challenged for glucose responsiveness to determine the optimal sensor formulation.
Results:
When incorporated into a hydrogel, high concentrations of viologen quencher and fluorophore were required to permit electron transfer between the two components and yield a detectable glucose response. Immobilization of this glucose-responsive system onto a silica nanoparticle facilitated this electron transfer to yield detectable responses at even low concentrations. Increasing quencher concentration on the nanoparticle, relative to the fluorophore, resulted in the greatest apparent glucose response.
Conclusion:
The nanoparticle sensors demonstrated excellent glucose response in the physiological range and are a promising tool for real-time glucose tracking.
Keywords: CGM, glucose sensors, fluorescence, subcutaneous, implantable
Introduction
Diabetes mellitus affects over 30 million people in the United States, costing the American healthcare system over $245 billion annually in direct and indirect costs.1 In order to prevent morbidities associated with hyper- and hypoglycemia, individuals with diabetes must constantly monitor and control their blood glucose levels. Commercially available continuous glucose monitors (CGMs) have simplified this process by enabling the tracking of blood glucose throughout the day in real time, significantly improving the quality of life of these patients. The majority of CGMs on the market today are implantable electrochemical sensors that continuously detect blood glucose through percutaneous electrodes.2-5 However, the insertion of these electrodes causes undesired tissue trauma and carry a high risk for infection, resulting in substantial immune responses and eventual fibrotic encapsulation. In turn, the sensitivity of these sensors becomes severely compromised after implantation and requires recalibration or replacement every seven to ten days.5-7
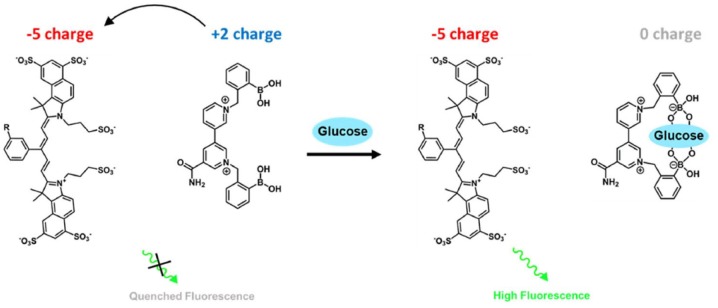
Light-based glucose-responsive systems have been proposed as an alternative noninvasive method for continuous monitoring, as they can be interrogated through the skin without requiring transcutaneous connections. Recently, a two-component sensing system was developed utilizing a boronic acid-based viologen quencher and an anionic fluorescent dye.8-11 In this system, the fluorophore is quenched by a physically separate boronic acid-substituted viologen quencher through electrostatic association, where the viologen acts as an electron receptor (Scheme 1). Binding of the boronic acid moiety with glucose neutralizes the dicationic viologen and diminishes the quenching efficiency, leading to increased fluorescence intensity in a glucose concentration-dependent manner. For in vivo sensing applications, these components must be immobilized onto a scaffold material, such as a hydrogel, and presented in a way that permits these electronic associations to occur.
Scheme 1.
A dual component system for fluorescence-based glucose sensing. A boronic-acid modified viologen molecule associates with an anionic fluorophore to quench fluorescence. Upon binding of glucose with the boronic acid moieties, the viologen is neutralized to increase the effective fluorescence of a near-infrared dye.
Here, we present the synthesis and optimization of nanoparticle sensors in which the two-component sensing motifs are immobilized onto a nanoparticle surface. These nanosensors can be embedded into a microporous poly(2-hydroxyethyl methacrylate) scaffold with high surface area for analyte diffusion (Scheme 2). Blood vessels grow into and throughout these hydrogels, reducing the diffusion distances between blood vessels and the nanosensors for more rapid response times.12 Further, these biocompatible scaffolds have been shown to resist fibrotic encapsulation and can be optimized separately from the nanosensor components. Combining these two materials together—a biocompatible scaffold and nanoparticle optical sensors—yields an effective biosensor for long-term, noninvasive glucose monitoring.
Scheme 2.
Illustration of subcutaneous optical detection of blood analytes via an implantable nanosensor-hydrogel composite. Microporous hydrogel scaffolds resist fibrotic encapsulation and facilitate tissue integration, yielding shorter diffusion distances between blood analytes and optical nanosensors.
Methods
Materials
Tetraethylene glycol dimethacrylate (TEGDMA) and 2-hydroxyethyl methacrylate (HEMA) were purchased from Sigma Aldrich (St. Louis, MO, United States). 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC) and N-hydroxysulfosuccinimide (NHSS) were purchased from Thermo-Fisher Scientific (Waltham, MA, United States). A total of 100 nm amine-terminated silica nanoparticles were purchased from Nanocomposix, Inc. (San Diego, CA, United States). A total of 200 μm poly(methyl methacrylate) (PMMA) microbeads were purchased from Polysciences, Inc. (Warrington, PA, United States). VA044 thermal initiator was purchased from Wako Chemicals USA, Inc. (Richmond, VA, United States). Viologen boronic acid (VBA) and IRDye680 were provided by Profusa, Inc. (South San Francisco, CA, United States).
Preparation of Microporous Hydrogels
Hydrogel scaffolds were fabricated utilizing previously described techniques.12-14 Briefly, a 3 mm teflon spacer was sandwiched between two glass slides and used as a mold for hydrogel polymerization. A prepolymer solution consisting of 49% HEMA, 1% TEGDMA crosslinker, 35% dimethyl sulfoxide (DMSO), VA044 thermal initiator (5 mg/mL), and water/nanoparticle solution/free dye solution was prepared. The prepolymer solution was injected into the prepared mold and incubated at 37°C for at least four hours. The crosslinked hydrogel was gently removed from the mold and allowed to swell in phosphate buffered saline.
In order to form hydrogel scaffolds with interconnected micropores, PMMA microparticles were packed into the assembly described above and sintered to form a bead cake.15 Prepolymer solution was then injected and crosslinked as described above to form a hydrogel with interconnected PMMA beads. The PMMA beads were leached out by incubation in dichloromethane, leaving behind a microporous hydrogel scaffold.
Synthesis of Glucose Nanosensors
An acid-terminated near-infrared (IR) wavelength fluorescent dye, IRDye680, and a boronic acid-modified viologen quencher, VBA, were custom synthesized. Both compounds were attached to amine-terminated silica nanoparticles using EDC/NHSS crosslinking chemistry. Viologen boronic acid and IRDye680 were dissolved in DMSO in a range of concentrations and activated with a tenfold excess of EDC and 20-fold excess of NHSS for 30 minutes. This solution was added to 10 mg/mL of aminated silica nanoparticles in 100 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer (pH 8.0) and allowed to react overnight on a shaker. Conjugated nanoparticles were centrifuged, washed thrice in HEPES buffer, and stored until further use. Absorbance spectra were obtained in order to account for any shifts in peak excitation and emission wavelengths of IRDye680 after conjugation to a nanoparticle.
Quantification of IRDye680 and VBA on Glucose Nanosensors
IRDye680 content on nanoparticles was quantified by absorbance at 680 nm and subtracting baseline absorbance from blank nanoparticles. A degradation assay was developed to determine VBA content on the nanosensors. A total of 200 µL of glucose nanosensors and standard solutions of free VBA were treated with 100 µL of 50 mM sodium hydroxide and incubated for five minutes. The absorbance of the glucose nanosensor sample was measured at 405 nm and compared to the standard curve to calculate VBA content.
Characterization of Glucose Sensitivity
To test the optical response of the nanosensors, samples were placed in a 96-well plate and challenged with gradual additions of glucose. Fluorescence was measured (excitation = 680 nm, emission = 710 nm) using a Molecular Devices SpectraMax M5 spectrophotometer (San Jose, CA, United States). Baseline fluorescence was first measured for ten minutes prior to glucose challenge. The concentration of glucose was then increased to 50 mg/dL by the addition of concentrated glucose (20 mg/mL) and the fluorescence was measured continuously until the signal stabilized. This process was repeated several times to reach 100, 200, and 600 mg/dL. Glucose sensitivity was reported as a ratio of intensity at 200 mg/dL over intensity at 50 mg/dL.
Results
Microbead templating was used to fabricate microporous hydrogels that support blood vessel growth and facilitate tissue integration. Poly(methyl methacrylate) microbeads were partially fused together by sintering at 140°C. The resulting “beadcake” was filled with a nanosensor/HEMA precursor solution and crosslinked at 40°C using a thermal initiator. The final microporous hydrogel was formed by dissolving the sintered microparticles in dichloromethane to leave behind interconnected micropores that are surrounded by the matrix material (Figure 1).
Figure 1.
Scattering electron microscopy image of microporous hydrogels fabricated by microbead templating. Sintering at 140°C for 18 hours fuses microbeads to serve as a template for fabricating microporous hydrogel. Crosslinking of 2-hydroxyethyl methacrylate around sintered microbeads yields hydrogels with large interconnected pores throughout the scaffold (scale bars = 100 μm).
A variety of nanosensors can be incorporated into these systems to detect blood analytes by transcutaneous fluorescence readings. For glucose sensing, a two-component sensing system consisting of a near-IR dye (IRDye680) and a boronic acid-based viologen (VBA) quencher was used (Scheme 1). 2-Hydroxyethyl methacrylate based hydrogels were fabricated, containing IRDye680 and VBA in a fixed 1:10 ratio. The total amount of dye and quencher was varied in order to demonstrate the effect of the molecular proximity on sensitivity (Figure 2). When challenged with glucose, hydrogel sensors with higher concentrations of IRDye680 and VBA demonstrated higher sensitivity to glucose, while those with less than 5 mM VBA and 0.5 mM IRDye680 had limited response that were more similar to drift than stepwise increases in fluorescence.
Figure 2.
Two-component glucose sensing chemistry is concentration dependent. (a) 2-Hydroxyethyl methacrylate hydrogels containing IRDye680 and viologen boronic acid at a fixed 10:1 ratio in a range of concentrations were challenged with the addition of glucose (i, 1:0.1; ii, 5:0.5; iii, 10:1; and iv, 15:1.5). (b) Intensity ratios of glucose sensitive hydrogels reveal that higher total concentrations of IRDye680 and viologen boronic acid improve glucose sensitivity.
The close positioning of dye and quencher resulting from high total concentrations can be reproduced on a nanoparticle, where each component is concentrated onto the nanoparticle surface. A colorimetric assay was developed to determine the concentration of VBA, which is normally colorless, after conjugation onto nanoparticles. Upon treatment with a strong base, the viologen degrades and obtains an absorbance peak at 400 nm (Figure 3(a)). A range of concentrations of VBA were treated with increasing concentrations of sodium hydroxide to determine the degradation kinetics of VBA. A total of 100 mM NaOH was required for complete degradation of VBA in the detectable range (Figure 3(b)). Standard curves generated with each concentration of NaOH were compared with 10 mM NaOH to 100 mM NaOH treatment resulting in similar standard curves (Figure 3(c)).
Figure 3.

Detection of viologen boronic acid quencher on glucose nanosensors. (a) Treatment of viologen boronic acid with NaOH results in the appearance of an absorbance peak at 400 nm. (b) Absorbance of viologen boronic acid at all concentrations plateau after treatment with 10 mM NaOH. (c) Standard curves of viologen boronic acid are indistinguishable after treatment with at least 10 mM NaOH.
Viologen boronic acid and IRDye680 were then conjugated onto an amine-coated silica nanoparticle. Both VBA and IRDye680 were activated by EDC/NHS chemistry and attached to the nanoparticle under basic conditions (Figure 4(a)). The ratio of VBA and IRDye680 was varied from 1:15 to 1:50. Control without EDC/NHS activation yielded nanoparticles with no conjugation of quencher and dye, in contrast to particles treated with EDC/NHS activated VBA and IRDye680 that appeared bright blue (Figure 4(b)). All nanoparticles containing VBA and IRDye680 exhibited sensitivity to glucose, with 1:50 ratio of IRDye680:VBA having the greatest glucose modulation (Figure 4(c) and (d)). When the nanosensors were treated with control solution, sensors with a 1:15 ratio of IRDye680:VBA had some drift in fluorescence, while 1:50 sensors had little to no response.
Figure 4.
Optimization of glucose nanosensor fabrication. (a) Synthesis scheme of glucose nanosensors with IRDye680 and viologen boronic acid quencher. (b) Comparison of silica nanoparticles reacted with IRDye680/viologen boronic acid (left) and 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride/N-hydroxysulfosuccinimide activated IRDye680/viologen boronic acid (right). (c) Glucose modulation of glucose nanosensors containing a range of IRDye680:viologen boronic acid ratios show that higher ratios provide better modulation. (d) Table of intensity ratios for particles treated with glucose and particles treated with control (saline) indicate that higher IRDye680:viologen boronic acid ratios have better modulation within physiological ranges (I200/I50) and limited drift in fluorescence upon addition of saline.
Discussion
In this study, we describe the synthesis of a novel nanoparticle sensors for continuous measurement of blood glucose. These sensors utilize a near-IR dye in combination with a glucose-sensitive quenching mechanism to modulate fluorescence. These dyes can be excited and detected through skin with minimal loss in signal due to the exceptional penetration depth of near-IR wavelengths.
The ideal optical sensor would rapidly integrate with the host tissue, avoid fibrotic encapsulation, and maintain sensitivity and specificity for extended periods of time. Our nanoparticle sensors were embedded into a microporous hydrogel scaffold that supports ingrowth of blood vessels to promote intimate integration with the host tissue. This design also reduces the diffusion distance between the vasculature and the nanosensors for rapid detection. Further, scaffolds with this added microarchitecture have been shown to resist the foreign body response and fibrotic encapsulation that would normally compromise sensor activity.16 Since the sensing chemistries were not immobilized to the scaffold material directly, but instead onto an entrapped nanoparticle, the glucose sensitivity could be independently optimized without affecting the bulk hydrogel properties. Additionally, this separation of function also enables the incorporation of multiple different types of nanosensors within the same scaffold for simultaneous monitoring of multiple analytes.
The ability to localize the sensing chemistries onto a nanoparticle surface was crucial for the development of the glucose sensor described in this work. This novel optical sensing chemistry utilized a near-IR wavelength dye that is modulated by a nearby quencher molecule. This quencher molecule is responsive to glucose due to the inclusion of two boronic acid groups, which upon binding to diols gains additional negative charges that significantly hinder electron transfer between the quencher and the fluorophore.8-11 It has been previously demonstrated that positioning of the boronic acid moiety plays a large role in glucose modulation.17 The binding of glucose to the boronic acid groups yields an anionic boronate that can electrostatically interact with the quaternary nitrogen on the viologen. When the boronate is in the ortho position, it is more proximal to the quaternary nitrogen leading to more effective neutralization of the positive charge. In the two-component sensing scheme, the overall charge of the viologen must neutralize the anionic charge on the fluorophore. Therefore, it is reasonable that the sensitivity of a hydrogel scaffold consisting of these components would heavily depend on proximity of the dye and quencher.
We hypothesized that the distance between the fluorophore and the quencher could be tuned by adjusting their total concentrations within the hydrogel scaffold, allowing for optimization of sensitivity. This was confirmed through the generation of a library of sensors with both IRDye680 and VBA covalently incorporated into the hydrogel network. Our results indicated that with a fixed ratio of dye to quencher, increasing their absolute concentrations, and by extension, their proximity to each other, improved the sensitivity of these hydrogel scaffolds to glucose. Gamsey et al previously fabricated glucose sensors with high concentrations of these sensing components immobilized to the hydrogel matrix for optimal sensitivity.11 Conjugation of these moieties to a nanoparticle surface allowed us to significantly reduce the distance between dye and quencher, thus increasing the sensitivity without having to increase the total concentrations in the hydrogel scaffold. We further optimized these glucose nanosensors by optimizing the ratio of dye to quencher. With minimal amounts of quencher, there was increased fluorescent signal due to limited fluorescence inhibition. However, there is also reduced glucose modulation due to a reduction of glucose responsive groups. By increasing the amount of quencher, there was greater glucose modulation that was accompanied by overall lower fluorescence values. This trend continued up to a 1:50 ratio of dye to quencher. For all ratios, there were no additional increases in fluorescence after 600 mg/dL of glucose, which exceeds the physiologically relevant range.
While boronic acid-based quenching is a promising technology for noninvasive glucose sensing, boronic acids are also known to bind to a variety of common sugars, such as fructose and galactose, with high affinity. This lack of specificity can impair accurate glucose measurements and reduce the reliability of these systems. The viologen quencher used in this study contains boronic acids in the ortho position, shortening the molecular distance between the boronic acids to enable cooperative binding and impart specificity for glucose. This modification was demonstrated to improve saccharide selectivity of the quencher molecules over fructose (1.7-fold selectivity) and galactose (11-fold selectivity).18 While the selectivity toward maltose and xylose was not examined for these nanosensors, Lorenz et al recently demonstrated that the sensitivity of a similar bis-boronic acid quencher molecule was not significantly affected by these sugars.19 Since both systems utilize the same glucose binding mechanism with bis-boronic acids in the ortho position, we anticipate limited interference by maltose and xylose on these nanosensors as well. However, further investigation is required to confirm the effect of these common interferents on nanosensor performance.
This study shows that our optical nanosensors are a promising technology for the treatment of diabetes. Despite successes in the optimization of sensitivity, the long-term stability of these materials has yet to be assessed. For clinically relevant applications, it is necessary to have continuous sensors that can accurately measure and report blood glucose levels over a long period of time without loss in sensitivity or signal. With these hydrogel-nanoparticle composite sensors, additional sensors can easily be implanted to replace inactive sensors, as these hydrogel sensors are biocompatible and can be fabricated with a very small form factor. While not addressed in the present study, several groups have developed instrumentation that can noninvasively excite and detect fluorescent signals from these optical sensing chemistries and transmit these data to a remote device.12 The combination of this instrumentation with our novel nanosensor technology can empower individuals with diabetes to take control of their disease state via on-demand blood glucose readings and in-depth data on patterns or trends in blood glucose fluctuations through continuous sensing.
Conclusion
Nanoparticle sensors capable of detecting glucose were synthesized and incorporated into tissue-integrating hydrogel scaffolds to form a composite implantable sensor. These materials utilize dyes in the near-IR spectrum and can be noninvasively excited and detected through tissue. The sensing ensemble utilizes a distance-dependent quenching mechanism based on electrostatic association, which was immobilized on the nanoparticle surface. Glucose sensitivity was optimized in vitro by varying relative concentrations of the near-IR dye and the viologen quencher components, respectively. Future work will involve calibrating this sensor with an in vivo model, as well as incorporating additional nanoparticles sensors for multi-analyte sensing.
Footnotes
Declaration of Conflicting Interests: The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Natalie Wisniewski and Soya Gamsey are full-time employees of Profusa, Inc.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Institutes of Health (5R01EB016414-03).
ORCID iDs: Long V. Le  https://orcid.org/0000-0003-1412-0612
https://orcid.org/0000-0003-1412-0612
Tejal A. Desai  https://orcid.org/0000-0003-3409-9208
https://orcid.org/0000-0003-3409-9208
References
- 1. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2017. Atlanta, GA: CDC. [Google Scholar]
- 2. Bindra DS, Zhang Y, Wilson GS, et al. Design and in vitro studies of a needle-type glucose sensor for subcutaneous monitoring. Anal Chem. 1991;63(17):1692-1696. [DOI] [PubMed] [Google Scholar]
- 3. Wang H-C, Lee A-R. Recent developments in blood glucose sensors. J Food Drug Anal. 2015;23(2):191-200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Klonoff DC. Continuous glucose monitoring: roadmap for 21st century diabetes therapy. Diabetes Care. 2005;28(5):1231-1239. [DOI] [PubMed] [Google Scholar]
- 5. Koschwanez HE, Reichert WM. In vitro, in vivo and post explantation testing of glucose-detecting biosensors: current methods and recommendations. Biomaterials. 2007;28(25):3687-3703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Pickup JC, Claremont DJ, Shaw GW. Responses and calibration of amperometric glucose sensors implanted in the subcutaneous tissue of man. Acta Diabetol. 1993;30(3):143-148. [DOI] [PubMed] [Google Scholar]
- 7. Yu B, Ju Y, West L, Moussy Y, Moussy F. An investigation of long-term performance of minimally invasive glucose biosensors. Diabetes Technol Ther. 2007;9(3):265-275. [DOI] [PubMed] [Google Scholar]
- 8. Suri JT, Cordes DB, Cappuccio FE, Wessling RA, Singaram B. Monosaccharide detection with 4,7-phenanthrolinium salts: charge-induced fluorescence sensing. Langmuir. 2003;19(12):5145-5152. [Google Scholar]
- 9. Cordes DB, Gamsey S, Sharrett Z, et al. The interaction of boronic acid-substituted viologens with pyranine: the effects of quencher charge on fluorescence quenching and glucose response. Langmuir. 2005;21(14):6540-6547. [DOI] [PubMed] [Google Scholar]
- 10. Cordes DB, Miller A, Gamsey S, et al. Optical glucose detection across the visible spectrum using anionic fluorescent dyes and a viologen quencher in a two-component saccharide sensing system. Org Biomol Chem. 2005;3(9):1708-1713. [DOI] [PubMed] [Google Scholar]
- 11. Gamsey S, Suri JT, Wessling RA, Singaram B. Continuous glucose detection using boronic acid-substituted viologens in fluorescent hydrogels: linker effects and extension to fiber optics. Langmuir. 2006;22(21):9067-9074. [DOI] [PubMed] [Google Scholar]
- 12. Wisniewski NA, Nichols SP, Gamsey SJ, Pullins S, Au-Yeung KY, Klitzman B, et al. Tissue-Integrating Oxygen Sensors: Continuous Tracking of Tissue Hypoxia. In: Halpern H, LaManna J, Harrison D, Epel B, eds. Adv Exp Med Biol. New York, NY: Springer; 2017:377–383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Andrus L, Unruh R, Wisniewski N, McShane M. Characterization of lactate sensors based on lactate oxidase and palladium benzoporphyrin immobilized in hydrogels. Biosensors. 2015;5(3):398-416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Roberts JR, Park J, Helton K, Wisniewski N, McShane MJ. Biofouling of polymer hydrogel materials and its effect on diffusion and enzyme-based luminescent glucose sensor functional characteristics. J Diabetes Sci Technol. 2012;6(6):1267-1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Long TJ, Takeno M, Sprenger CC, Plymate SR, Ratner BD. Capillary force seeding of sphere-templated hydrogels for tissue-engineered prostate cancer xenografts. Tissue Eng Part C Methods. 2013;19(9):738-744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Wisniewski N, Klitzman B, Miller B, Reichert WM. Decreased analyte transport through implanted membranes: differentiation of biofouling from tissue effects. J Biomed Mater Res. 2001;57(4):513-521. [DOI] [PubMed] [Google Scholar]
- 17. Gamsey S, Baxter NA, Sharrett Z, et al. The effect of boronic acid-positioning in an optical glucose-sensing ensemble. Tetrahedron. 2006;62(26):6321-6331. [Google Scholar]
- 18. Gamsey S, Miller A, Olmstead MM, et al. Boronic acid-based bipyridinium salts as tunable receptors for monosaccharides and α-hydroxycarboxylates. J Am Chem Soc. 2007;129(5):1278-1286. [DOI] [PubMed] [Google Scholar]
- 19. Lorenz C, Sandoval W, Mortellaro M. Interference assessment of various endogenous and exogenous substances on the performance of the eversense long-term implantable continuous glucose monitoring system. Diabetes Technol Ther. 2018;20(5):344-352. [DOI] [PMC free article] [PubMed] [Google Scholar]