Abstract
Aims: To investigate the performance of an iodine‐releasing filter medium for use as a protective device against airborne pathogens.
Methods and Results: The filter’s physical and viable removal efficiencies (VRE) were investigated with challenges of MS2 bacteriophage aerosols, and the infectivity of MS2 collected on the filter was analysed. To test a proposed inactivation mechanism, media containing thiosulfate or bovine serum albumin (BSA) were put in impingers to quench and consume I2 released from the filter. In direct plating experiments, treated filters presented significantly higher VREs than did untreated filters; however, collection in excess BSA decreased VRE by half and in thiosulfate the apparent VRE decreased drastically. No significant difference in infectivity of retained viruses on treated and untreated filters was observed at the same environmental condition.
Conclusions: Evidence presented herein for competition by dissolved I2 in infectivity assays supports a mechanism of induced displacement and capture of I2. It also requires that dissociation of iodine from the filter and capture of iodine by MS2 aerosols as they pass through the filter be factored in the design of the assessment methodology. The filter’s strong retention capability minimizes reaerosolization but also makes it difficult to discriminate the antimicrobial effect at the surface.
Significance and Impact of the Study: This study shows the direct plating assay method to be sensitive to interference by iodine‐releasing materials. This requires reevaluation of earlier reports of VRE measurements.
Keywords: antimicrobial, biocidal filter, inactivation, iodine, MS2, removal efficiency
Introduction
Increasing concerns about bioterrorism and the spread of such airborne pathogens as severe acute respiratory syndrome (SARS) and avian flu viruses have attracted the public attention to bioaerosols and spurred the development of respiratory protection methods. For example, SARS is caused by a corona virus for which there is no vaccine. First reported in Asia in February 2003, it spread to more than two dozen countries in North America, South America, Europe and Asia over the following few months. Transmission of SARS virus is suspected to occur through droplets generated from sneezing or coughing of an infected person, which subsequently deposit on the mucous membranes of the mouth, nose or eyes of nearby persons (CDC 2005). Various diseases, such as tuberculosis, mumps, measles, pneumonia, influenza in addition to many not known to humans, are transmitted by the airborne route (Biswas and Wu 2005; Fiegel et al. 2006). Through sneezing and coughing, thousands of droplets containing viable micro‐organisms can be released (Jennison 1942). The smaller droplets quickly evaporate to droplet nuclei, which can suspend in air for a long time and travel considerable distances either in free form or by attaching to other aerosols. Because of droplet encasement, virus infectivity can be shielded from external forces (e.g. drying, sunlight and temperature) (Tyrrell 1967).
With the advantages of simplicity, versatility and economical collection of aerosol particles, filtration is the most commonly used method for aerosol removal in heating, ventilation and air conditioning (HVAC) systems, as well as in respiratory protection devices (Hinds 1999). However, conventional high efficiency filters require high energy consumption (for HVAC system) or breathing exertion (respirators) because of its high pressure drop. Moreover, the surviving micro‐organisms collected on the filter can multiply and may be released into the air passing through the filter medium, thus resulting in microbial contamination of the breathing air (Richardson et al. 2006). Therefore, there have been efforts to incorporate antimicrobial agents into air filters, intended to destroy or inhibit the survival and growth of micro‐organisms (Foarde and James 2001; Cecchini et al. 2004).
Iodine, a halogen species, has been used for water treatment as an antimicrobial agent and has some advantages over chlorine. Iodine is slightly soluble and stable in water over the pH range of 6–8, and it is less reactive with organic nitrogenous contaminants than chlorine (Bruchertseifer et al. 2003). Among the various iodine species, iodine molecules (I2) and hypoiodous acid (HOI), which is produced by hydrolysis of I2, have disinfection capability (Chang 1958). Iodine can be attached to a quaternary ammonium strong‐base anion resin in the form of triiodide (I3 −) and pentaiodide (I5 −) ions. This iodinated resin filter, being an electret medium, is expected to possess high removal efficiency for bioaerosols and lower pressure drop than conventional mechanical filter media.
Negatively charged micro‐organisms can polarize iodine complexes on the filter during near contact encounters and transfer I2 (Ratnesar‐Shumate et al. 2008). The mechanism by which iodine inactivates viruses involves structural damage to the capsid protein (Maillard 2001). Oxidation of sulfhydryl (‐SH) groups or substitution onto tyrosine and histidine residues results in the disruption of normal functions of these amino acids (Carroll 1955). On the other hand, iodine does not inactivate either infectious RNA or DNA (Hsu 1964).
Studies on the disinfection capacity of iodinated resin filters for the treatment of bacteria and viruses in water were conducted three decades ago and reported disinfection capacities over 99·99% (Taylor et al. 1970; Gilmour and Wicksell 1972; Hatch et al. 1980; Fina et al. 1982; Marchin et al. 1997). However, only limited studies have been conducted recently on the disinfection capacity of iodinated resin filters for air treatment (Messier 2004; Heimbuch and Wander 2006; Heimbuch et al. 2007; Eninger et al. 2008; Lee et al. 2008; Ratnesar‐Shumate et al. 2008). The previous study in this series (Ratnesar‐Shumate et al. 2008) – investigating the performance of iodine‐treated filters for inert fluorescent particles and vegetative bacterial cells (Micrococcus luteus, Escherichia coli) – proposed a near‐contact transfer mechanism between the iodine‐treated filter and micro‐organisms penetrating the filter as an inactivation mechanism, but without solid proof. In the current study, a second possible source of inactivation was considered – I2 released from the filter. I2 released from the filter could cause inactivation in the sampling device, whereas I2 captured by micro‐organisms passing through the filter could inactivate them in their airborne state and/or continue the inactivation process after collection in the sampling device, either bound to the particle or by dissolving in the aqueous medium. Identification of inactivation by dissolved I2 could confound the results in earlier reports (Messier 2004; Heimbuch and Wander 2006; Heimbuch et al. 2007; Eninger et al. 2008; Lee et al. 2008; Ratnesar‐Shumate et al. 2008) that used plating methods to measure viable removal efficiency, which would require an independent experimental method to quantify the relative importance of two competing inactivation mechanisms.
The objective of this study was to evaluate an iodine‐treated filter medium for removal and inactivation of viral aerosols as a respiratory protective device against airborne biological agents under various environmental conditions. Physical removal efficiency (PRE), viable removal efficiency (VRE), pressure drop, I2 concentration in the impinger medium and the infectivity of collected viruses on the iodine‐treated filter were investigated and compared with those of an untreated filter. Furthermore, the inactivation mechanism proposed earlier for the iodine‐treated filter was examined by measuring VRE downstream of the filter using various collection media that were inert, moderately reactive and aggressively reactive to I2.
Materials and methods
Test filters
Samples of the iodine‐treated (polyester–cotton, 125 g m−2 triiodide resin; Safe Life Corp., San Diego, CA, USA) and untreated (polyester–cotton; Safe Life Corp.) filter media, both as flat sheets 1‐mm thick, were tested as discs 47 mm in diameter. Triiodide ion, prepared by mixing stoichiometric amounts of I2 and KI in a minimum amount of water, was contacted with a quaternary ammonium anion exchange resin to substitute the anion with triiodide. Details of the preparation are available in the patent by Messier (2004). The I2 concentration in effluent air passing through the iodine‐treated filter can be measured by Occupational Safety and Health Administration (OSHA) analytical method ID‐212 for I2 in workplace atmospheres. I2 collected in the impinger medium (1·5 mmol l−1 Na2CO3 and 1·5 mmol l−1 NaHCO3) can be analysed as iodide by ion chromatography (OSHA 1994). The I2 concentration was measured to be 0·004 mg I2 per m3.
Test micro‐organism
MS2 bacteriophage (ATCC® 15597‐B1™) was selected as a representative virus aerosol. In the selection of a model virus, its resistance to antimicrobial agents should be considered, because resistance varies from one virus to another (Berg et al. 1964; Sobsey et al. 1990). MS2 is a nonenveloped, icosahedron‐shaped, single‐stranded RNA with a single‐capsid size of 27·5 nm, and it infects only male E. coli (Prescott et al. 2002). MS2 has been used as a surrogate for small RNA enteroviruses pathogenic to humans, because they both have no lipid component surrounding the protein coat and are considered to have similar resistance (Aranha‐Creado and Brandwein 1999; Brion and Silverstein 1999). Freeze‐dried MS2 was suspended with filtered deionized (DI) water to a concentration of 108–109 PFU ml−1 as the virus stock suspension and stored at 4°C.
Experimental system and conditions
The experimental set‐up for testing the removal efficiency of filters is shown in Fig. 1. Seven litres per minute (Lpm) of dry, filtered compressed air was passed though a six‐jet Collison nebulizer (Model CN25; BGI Inc., Waltham, MA, USA) to aerosolize the viral suspension. The virus concentration in the Collison nebulizer was 105–106 PFU ml−1 and was prepared by diluting 0·10 or 0·20 ml of virus stock suspension in 50 ml sterile DI water. The aerosolized particles were dried with filtered compressed air in a 2·3‐l glass dilution dryer. A flow rate of 8 l min−1, which corresponds to a face velocity of 14·2 cm s−1, was used for each stream (i.e. control and experimental) and controlled by a calibrated rotameter. Based on the velocity, flight time through the 1‐mm filter is estimated to be 0·007 s. This face velocity, used by Safe Life Corp., corresponds to a certification testing of 100 cm2 media (Di Ionno and Messier 2004) against the 85‐Lpm flow rate specified by the National Institute for Occupational Safety and Health (NIOSH 2005). Pressure drop across each filter was monitored with a Magnehelic gauge measuring 0–2491 Pa and recorded every 30 min. The viral aerosols entering and penetrating the test and control filters were collected in an AGI‐30 impinger (Ace Glass Inc., Vineland, NJ, USA) containing 20 ml of sterile phosphate‐buffered saline (PBS). The collection medium in each impinger was replaced by fresh solution every 30 mins and assayed to determine the virus concentration by using suitable dilution to an adequate count (i.e. 30–300 PFU). Five 2‐h trials were conducted, and thus total evaluation time was 10 h.
Figure 1.
Experimental set‐up for the (a) viable removal efficiency (b) physical removal efficiency of the test filters.
As I2 and HOI are disinfective forms, an increased VRE of the iodine‐treated biocidal filter at high temperature and increased relative humidity (RH) was hypothesized possibly because of iodine’s sublimation and increased dissolution through the hydrolysis of I2 to HOI respectively. Therefore, various environmental conditions were considered: room temperature (23 ± 2°C) and low relative humidity (35 ± 5%, RT/LRH), high temperature (30 ± 2°C) and LRH (HT/LRH), RT and medium RH (50 ± 5%, RT/MRH). Because the maximum inactivation of MS2 aerosolized from 0·1 mol l−1 NaCl was reported to occur at 75% (Trouwbor and de Jong 1973), RHs below this level were considered. Temperature and RH were adjusted by wrapping the dilution dryer with a heating jacket and adding dry or humid dilution air to the system respectively.
Removal efficiency
Removal efficiency of viral aerosols by the test filters can be expressed as both PRE and VRE. The particle size distribution (PSD) of the aerosols entering and penetrating the test filters was measured by using a Scanning Mobility Particle Sizer (SMPS, Model 3936; TSI Inc., Shoreview, MN, USA), and the PRE was determined as
| (1) |
where N E is the number of particles entering the filter and N p is the number of particles penetrating the filter.
The VRE depends on the infectivity of viruses collected in the impingers. The VRE was determined by counting plaques on each Petri dish from both control and experimental impingers, and calculated according to eqn (1). In calculating the viral concentration, a dilution factor was used, which depends on the number of transfers of the impinger solution. Thus, the viral concentration in the impinger, C v (PFU ml−1), was determined as
| (2) |
where PFU is the number of plaque‐forming units, V is the volume of diluted solution and n is the dilution factor. The final mean viral concentration was determined by averaging all values in each dilution.
Inactivation mechanism of the iodine‐treated biocidal filter
Two possible inactivation mechanisms of the iodine‐treated filter were considered: (i) inactivation of viruses downstream of the filter by reaction with I2 released from the filter and (ii) direct transfer of I2 during near contact as viral aerosols pass through the iodine‐treated filter.
Sublimation and dissolution of iodine molecules released from the filter
To investigate the effects of iodine released from the iodine‐treated filter, filtered clean air passing the test filter at various environmental conditions was drawn into impingers containing a viral suspension of known concentration. The virus in the experimental impinger might lose its infectivity because of both the operation of the impinger (e.g. swirling effect and reaerosolization) and the action of I2. Meanwhile, the infectivity of viruses in the control impinger will be affected only by the operation of the impinger. Therefore, by comparing the results of the control and the experimental impingers, the loss of virus infectivity by the operation of the impinger was excluded.
How I2 disinfects virus in the impinger was studied by using sodium thiosulfate solution to quench the reactivity of I2 available in the impinger. The same experimental procedure described previously for sublimation and dissolution of I2 was followed except that the impinger medium was replaced by a 0·1‐mol l−1 solution of sodium thiosulfate. Thiosulfate anion (S2O3 2−) reacts stoichiometrically with I2 and reduces it to iodide, which is not virucidal (Berg et al. 1964).
Transfer of I2 to viral aerosols
To investigate the inactivation mechanism of direct transfer of I2 to viral aerosols, the effect of sublimation and dissolution of I2 released from the iodine‐treated filter should be excluded. The use of thiosulfate solution has a limitation in this exclusion because it can react with both I2 existing free in the impinger solution and I2 residing on the MS2. Therefore, a halogen‐demanding substance, bovine serum albumin (BSA), was used, which consumes free I2 in the impinger solution but competes less aggressively than thiosulfate for I2 on the MS2. The capacity of BSA to consume all of the I2 released from the filter was predetermined by using the same experimental configuration for sublimation and dissolution of I2 except that the impinger contained 0·3%, 3% and 6% BSA and a virus suspension of known concentration. The filtration experiment was then performed using the selected concentration. Viral aerosols were delivered as challenges to the iodine‐treated filter and collected in both control and experimental impingers for 1, 5, 10 and 15 mins. The MS2 in the experimental impinger was compared to the penetrating MS2. For comparison, the same experiment was performed with thiosulfate solution as the collection medium of the impinger for 15 mins.
Infectivity of viruses on the filter
After 10 h of removal efficiency experiments, the test filters were retrieved from the filter holder in the experimental system and subjected to a vortex mixer (Model M16715; Barnstead, Dubuque, IA, USA) to investigate the infectivity of viruses collected on the filter. The filter was immersed in 40 ml sterile DI water in a 250‐ml beaker and vortexed for a designated time (i.e. 0, 1, 3 and 5 min) to investigate the optimal extraction time. The vortexing solution was assayed to determine the infectivity of viruses, and the number of viruses (N v) was determined as:
| (3) |
where V 1 is the volume of extraction fluid and V 2 is the volume of original or diluted suspension assayed with host cells. The total infectivity of extracted viruses was calculated by averaging the results at all vortexing times, because the number of extracted viruses at each designated vortexing time was found to be similar. The extracted fraction – the ratio of the infectivity count in the extraction solution to the total viruses collected on the filter – was used to compare the result of the iodine‐treated filter with the untreated filter.
Effects of free iodine molecules
In an aqueous suspension for the vortexing experiment, the resin surfaces are expected to release I2 that can inactivate viruses. This reaction raises a question whether viruses lose their infectivity in the extract solution because of the free I2 residual or on the filter. To investigate this question, the solution after vortexing a clean iodine‐treated filter for a designated length of time (0, 1, 3 and 5 min) was inoculated with a virus suspension of known concentration. Because it took 15 min to finish the vortexing experiment including dilution and assay, the infectivity of virus in the mixed suspension was analysed after 15‐min exposure to the free I2 in the suspension. The I2 concentration in the vortexing solution was analysed by the N, N‐diethyl‐p‐phenylenediamine (DPD) colorimetric method adapted from Standard Methods for the Examination of Water and Wastewater 4500‐CI G (APHA 1995). Ten millilitres of solution vortexed with the iodine‐treated filter was analysed at 530 nm by using a DR/4000 V Spectrophotometer (Hach, Loveland, CO, USA). I2 in the solution reacts with DPD to form a pink colour, the intensity of which is proportional to the total I2 concentration (Hach 2003). The effect of vortexing alone on the infectivity of viruses was also investigated by following the same vortexing procedure with a virus suspension of known concentration.
Results
Physical removal efficiency and pressure drop
The PRE of the test filters was determined by comparing the PSDs of the aerosols entering and penetrating the test filters as shown in Fig. 2. The PSD of the aerosols entering the test filters showed its mode at approx. 25 nm. As a baseline, sterile DI water without virus was aerosolized from the nebulizer, and the PSD of that was measured, defining the background noise. Therefore, the PSD of the aerosols above the noise level in the window from 9·82 to 162·5 nm was considered for the calculation. The PREs (mean ± SD) of the iodine‐treated and untreated filters for this size range were 41 ± 3% and 39 ± 2% respectively. Statistical evaluation of the two values by a one‐tailed student’s t‐test indicated that the difference was insignificant (P‐value > 0·05).
Figure 2.
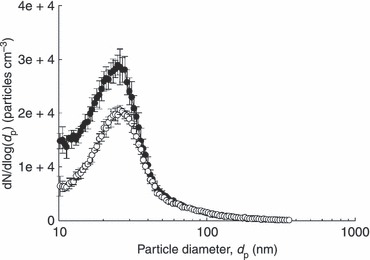
The number‐based particle size distribution of aerosols entering and penetrating the filter at room temperature and medium relative humidity. ( ) Entering the filter; (
) Entering the filter; ( ) penetrating the filter.
) penetrating the filter.
The initial pressure drop of the test filters was around 50–100 Pa, and the variation in pressure drop during the entire experiment was negligible. This value is much less than the inhalation and exhalation resistances of the respirator certified by NIOSH, which cannot exceed 343 and 196 Pa respectively (CFR 2002). No significant difference in the pressure drop between the iodine‐treated and untreated filters was observed.
Viable removal efficiency
The VRE of the test filters was calculated by analysing the infectivity of viruses collected on both control and experimental impingers for challenging and penetrating viruses from the filter, respectively. The result is presented as an average of five 2‐h experimental runs for each filter indicated as No.1 and No.2 in Table 1. As shown, the iodine‐treated filter presented a significantly higher VRE from the standard assay than that of the untreated filter (P‐value < 0·05) at various environmental conditions. At HT/LRH, a significantly higher value of the iodine‐treated filter than the other conditions was observed, according to one‐way anova (P‐value < 0·05), because of the increased release of I2 from the filter. Meanwhile, the difference between RT/LRH and RT/MRH was not significant (P‐value > 0·05), indicating that the release of HOI into air because of the hydrolysis of I2 at increased RH is negligible.
Table 1.
Removal efficiency of the iodine‐treated and untreated filters at various environmental conditions in impingers containing phosphate buffered saline
| Environmental conditions | Filter media | Virus concentration (PFU per ml)* | Removal efficiency (%)* | ||
|---|---|---|---|---|---|
| Challenge | Penetration | ||||
| Room temperature (23 ± 2°C) Low relative humidity (RH) (35 ± 5%) | Iodine‐treated | No.1 | 1·0 × 105 ± 4·3 × 104 | 5·3 × 102 ± 2·5 × 102 | 99·4 ± 0·5 |
| No.2 | 1·4 × 105 ± 5·8 × 104 | 4·1 × 102 ± 3·4 × 102 | 99·7 ± 0·4 | ||
| Untreated | No.1 | 6·3 × 104 ± 5·6 × 104 | 5·0 × 103 ± 4·4 × 103 | 92·4 ± 1·8 | |
| No.2 | 3·7 × 104 ± 1·2 × 104 | 3·3 × 103 ± 1·1 × 103 | 90·7 ± 2·2 | ||
| High temperature (30 ± 2°C) Low RH | Iodine‐treated | No.1 | 1·4 × 105 ± 7·0 × 104 | ND† | >99·9995 |
| No.2 | 3·0 × 104 ± 2·5 × 104 | 3·2 × 100 ± 2·4 × 100 | 99·98 ± 0·05 | ||
| Untreated | No.1 | 3·3 × 105 ± 1·5 × 105 | 1·6 × 104 ± 6·9 × 103 | 94·0 ± 3·8 | |
| No.2 | 9·6 × 104 ± 3·0 × 104 | 7·2 × 103 ± 2·7 × 103 | 91·4 ± 4·8 | ||
| Room temperature Medium RH (50 ± 5%) | Iodine‐treated | No.1 | 2·4 × 104 ± 1·8 × 104 | 6·7 × 101 ± 6·9 × 101 | 99·8 ± 0·3 |
| No.2 | 7·6 × 103 ± 3·2 × 103 | 4·2 × 100 ± 8·8 × 100 | 99·8 ± 0·8 | ||
| Untreated | No.1 | 2·3 × 105 ± 2·4 × 105 | 1·4 × 104 ± 1·3 × 104 | 93·4 ± 2·1 | |
| No.2 | 1·0 × 105 ± 3·8 × 104 | 8·9 × 103 ± 3·5 × 103 | 91·3 ± 2·0 | ||
*The average (±SD) of five 2‐h trials.
†ND, not detected.
Inactivation mechanism of the iodine‐treated biocidal filter
The effect of sublimation and dissolution of I2 was investigated by using the impingers containing a virus suspension of known concentration in either the PBS or the sodium thiosulfate solution. As shown in Table 2, no surviving virus was detected in the experimental impinger until >104 PFU in the PBS was added to the impingers. As the virus concentration in the impinger increased, the number of surviving viruses also increased. Meanwhile, the survival fraction of viruses in the thiosulfate solution was much higher than that in the PBS. Most viruses suspended in the thiosulfate solution survived in the experimental impinger because of the quenching by reaction with thiosulfate of the I2 released from the iodine‐treated filter and/or transferred I2 to viral aerosols. Hatch et al. (1980) proposed spontaneous dissociation of I2 from the polyiodide–resin complex as one of three possible inactivation mechanisms of their iodinated resin filter in water treatment. In another study (Marchin et al. 1983), acquisition of I2 by a cyst during passage through an iodinated resin column was hypothesized. The authors observed that cysts regained viability because of the reduction of I2 by thiosulfate solution for up to 3 min. A more recent study (Brion and Silverstein 1999) reported reversal of MS2 inactivation after a few minutes (<5 min) of iodine treatment by adding 0·3% BSA. It must be noted that these studies were performed in water, so their applicability to inactivation mechanisms of iodine in air treatment is uncertain.
Table 2.
The survived MS2 among various MS2 concentrations in the impingers with phosphate buffered saline, thiosulfate solution and bovine serum albumin at various environmental conditions due to iodine released from the filter
| Environmental conditions | Collection medium in the impinger | Virus count (PFU) in the impinger (Average ± SD) | Survival fraction* | |
|---|---|---|---|---|
| Control | Experimental | |||
| Room temperature (23 ± 2°C) Low relative humidity (RH) (35 ± 5%) | PBS† | 5·6 × 103 | 0 | 0 |
| 1·1 × 104 | 1·0 × 102 | 9·1 × 10−3 | ||
| 2·3 × 105 | 8·0 × 102 | 3·4 × 10−3 | ||
| Sodium thiosulfate | 1·9 × 103 ± 4·9 × 102 | 1·7 × 103 ± 4·9 × 102 | 9·0 × 10−1 ± 0·0 | |
| High temperature (30 ± 2°C) Low RH | PBS | 6·3 × 103 ± 7·1 × 101 | 0 | 0 |
| 5·3 × 104 ± 8·5 × 103 | 8·6 × 101 ± 3·4 × 101 | 1·6 × 10−3 ± 3·5 × 10−4 | ||
| 2·1 × 105 ± 3·5 × 104 | 5·0 × 102 ± 2·1 × 102 | 2·4 × 10−3 ± 4·9 × 10−4 | ||
| Sodium thiosulfate | 1·4 × 103 ± 1·4 × 102 | 1·2 × 103 ± 0·0 × 100 | 8·5 × 10−1 ± 7·1 × 10−2 | |
| Room temperature Medium RH (50 ± 5%) | PBS | 6·2 × 103 ± 2·5 × 103 | 0 | 0 |
| 6·5 × 104 ± 7·1 × 103 | 4·9 × 101 ± 1·9 × 101 | 7·3 × 10−4 ± 2·1 × 10−4 | ||
| 2·9 × 105 ± 1·4 × 104 | 5·6 × 102 ± 4·2 × 101 | 2·0 × 10−3 ± 7·1 × 10−5 | ||
| Sodium thiosulfate | 3·2 × 103 ± 2·5 × 103 | 2·2 × 103 ± 1·5 × 103 | 7·5 × 10−1 ± 7·1 × 10−2 | |
| 0·3% Bovine serum albumin | 1·6 × 103 ± 6·7 × 102 | 9·1 × 102 ± 1·3 × 102 | 5·9 × 10−1 ± 1·7 × 10−1 | |
| 3% Bovine serum albumin | 1·6 × 103 ± 7·4 × 102 | 1·5 × 103 ± 6·1 × 102 | 9·5 × 10−1 ± 6·1 × 10−2 | |
| 6% Bovine serum albumin | 1·9 × 103 ± 7·6 × 102 | 1·7 × 103 ± 6·4 × 102 | 9·5 × 10−1 ± 4·3 × 10−2 | |
*PFU in the experimental impinger divided by PFU in the control impinger.
†PBS, phosphate‐buffered saline.
In the experiments measuring I2 demand of BSA, various concentrations of BSA were evaluated. As shown in Table 2, the survival fractions of MS2 in the experimental impinger having 3% and 6% BSA were similar to those in the control impinger (approx. 0·95). The result indicates that both 3% and 6% BSA solutions contain sufficient protein to exhaust I2 released from the filter and thus isolate MS2 in the experimental impinger from inactivation by I2 in solution. The history of iodination of albumins suggests significant dependence on conditions. Muus et al. (1941) reported rapid uptake of 15 wt% of iodine by horse serum albumin (HSA) from approx. 0·2 N I2/KI in aqueous ethanol, and Shahkrokh (1943) added 8 wt% of iodine to HSA with a similar concentration of I2/KI in water. Hughes and Straessle (1950) incorporated 30 molar equivalents of iodine into human serum albumin in 0·1 N aqueous I2, converting 70% of l‐tyrosine residues into diiodotyrosine. Small‐scale preparations adding chloramine‐T to similar concentrations of K131I in water achieved fast and efficient incorporation of the 131I into human growth hormone (Greenwood et al. 1963), BSA (Opresko et al. 1980) and BSA microspheres (Smith et al. 1984). Lee and Ellis (1991) proposed the reaction with iodine solutions as a method to visualize serum albumins on polyacrylamide gels. However, Shahkrokh (1943) also showed that the extent of reaction of HSA with I2 falls off rapidly with decreasing concentration, and Portenier et al. (2001) reported that an equimolar amount of BSA did not suppress the bactericidal activity of a 0·2% solution of I2/KI.
In the experiments in which aerosolized MS2 penetrated the iodinated filter, collection in a medium containing thiosulfate effectively neutralized all of the iodine released, whether displaced and captured or dissociated, as no decrease in viable penetration was observed, as shown in Fig. 3. In contrast, a similar experiment in which the penetrating particles and free iodine were collected in 3% BSA medium showed that half the penetrating MS2 virions were inactivated initially, and a moderate increase in survival was seen after 10 min. The initial observation is consistent with the mechanism proposed by Ratnesar‐Shumate et al. (2008), because the data in Table 2 show that the capture medium is able to consume all of the free iodine coming off the filter. Thus, at least half of the MS2 viral particles penetrating the filter in this experiment appear to have acquired and bound a lethal dose of I2 as they traversed the iodine‐treated filter. The distinguishable increase of surviving MS2 at 10 mins of collection time parallels a delayed reactivation of MS2 observed in aqueous iodine solutions (Brion and Silverstein 1999), and it is tempting to conclude that the deactivation processes in water and in this system are similar after iodine has been transferred to the virion. However, some combination of direct transfer of I2 from the filter plus dissociation of I2 from weaker binding sites on penetrating particles reproduces their general conditions and appears to cause almost half the observed inactivation of viral aerosols penetrating the iodine‐treated filter and collected in PBS medium. After submission of this manuscript, Triosyn Corp. (P.J. Messier, 2009, unpublished data) disclosed data showing a threshold for inactivation of MS2 and of Staphylococcus aureus at 0·5∼0·6 ppm I2 in PBS medium, which is consistent with results presented herein and defines a boundary condition to anticipate significant interference by dissolved iodine. We then verified that the data reported by Heimbuch and Wander (2006) and by Heimbuch et al. (2007) were measured under conditions that inactivation by free I2 did not contribute significantly.
Figure 3.
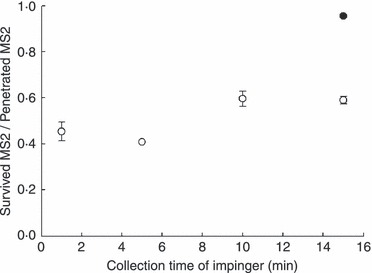
The survived MS2 aerosols among penetrated MS2 aerosols from the iodine‐treated filter with thiosulfate solution and 3% bovine serum albumin (BSA) as the collection medium of the impinger at room temperature and medium relative humidity. ( ○ ) Three per cent BSA; ( • ) thiosulfate solution.
Eninger et al. (2008) collected MS2 aerosols penetrating an iodinated medium into gelatin‐coated plates, which they washed out into water and plated in a plaque assay. They observed no kill of MS2 and concluded that the treatment was ineffective. However, their observation of no inactivation by iodine during the steps of their workup that were executed in water shows clearly that the overwhelming excess of protein in their collection surface consumed all of the iodine displaced, released or captured from the iodinated medium. Whereas their experiment thus does not support the conclusion that the treatment is inactive, in the absence of measurements of I2 concentrations in the impingers, we can make no quantitative statement about the relative importance in our dataset of these potentially competing processes for inactivation. However, we note that, even though sufficient I2 is released to confound the environment in the impingers, the airborne concentration of I2 released from the filter was much less than the 8‐h time weighted average‐threshold limit value of 1 mg m−3, the level below which a worker is expected to have no adverse health effect resulting from chronic exposure (OSHA 2000; Di Ionno and Messier 2004). Hence, whatever activity is present is realistically available for use in respiratory protection.
Effects of free iodine molecules and extracted fraction
To account for the effect of free iodine in the extract solution, the infectivity of viruses mixed with the vortexing solution from a clean iodine‐treated filter after each designated vortexing time was analysed and expressed as survival fraction (C S/C I, C S, surviving MS2; C I, initial MS2 in the suspension). The average value of the survival fraction at all vortexing times, 0·17 (i.e. 83% attenuation), was used to correct the value for the infectivity of viruses collected on the filter. As presented in Table 3, the I2 concentration in the vortexing solution measured by the DPD colorimetric method was around 1·0 mg l−1 I2. Some I2 was released from the iodine‐treated filter before subjecting to the vortexing, designated as ‘0’ vortexing time. No further increase of I2 extraction from the filter by increasing vortexing time was observed.
Table 3.
Iodine concentration (mg I2 per litre)* in the vortexing solution at each vortexing time
| Filter media | Vortexing time (min) | |||
|---|---|---|---|---|
| 0 | 1 | 5 | 10 | |
| Iodine‐treated filter | 0·62 ± 0·11 | 0·98 ± 0·04 | 0·91 ± 0·13 | 0·98 ± 0·08 |
*The average measurement in triplicate.
The infectivity of viruses collected on the filter is presented as the extracted fraction, C E/C C (C E, MS2 extracted from the filter; C C, MS2 collected on the filter). C C for the iodine‐treated filter was determined from the VRE of the untreated filter because both iodine‐treated and untreated filters had a similar PRE. The effect of vortexing on the viruses was negligible, because the infectivity of viruses vortexed at various times did not have observable variation. Table 4 presents both observed and corrected values of the extracted fraction. The corrected values were determined by dividing the observed values by the survival fraction (0·17) to consider the effects of free I2. As shown, no significant difference in the corrected extracted fraction between iodine‐treated and untreated filters at the same environmental condition was exhibited (P‐value > 0·05). Both iodine‐treated and untreated filters tested at MRH showed the lowest value among the survival fractions presumably because of the sensitivity of MS2 to the MRH (Dubovi and Akers 1970). The lower values of free I2 from the iodine‐treated filter tested at HT/LRH and RT/MRH than that at RT/LRH indicate measurable loss of I2 from the iodine‐treated filter. Although the filter constantly experienced loss of I2, it was observed that the efficacy of the iodine‐treated biocidal filter did not deteriorate during 10 h of experiment.
Table 4.
Extracted fraction of MS2 on the iodine‐treated and untreated filters at various environmental conditions
| Environmental conditions | Filter media | Average ± SD | Iodine in vortexed solution (mg l−1) | |
|---|---|---|---|---|
| Observed | Corrected* | |||
| Room temperature (23 ± 2°C) Low relative humidity (RH) (35 ± 5%) | Iodine‐treated | 3·4 × 10−3 ± 1·4 × 10−3 | 2·0 × 10−2 ± 8·4 × 10−3 | 0·93 ± 0·01 |
| Untreated | 3·6 × 10−2 ± 3·4 × 10−2 | – | ||
| High temperature (30 ± 2°C) Low RH | Iodine‐treated | 3·3 × 10−3 ± 2·0 × 10−3 | 2·0 × 10−2 ± 1·2 × 10−2 | 0·575 ± 0·007 |
| Untreated | 3·3 × 10−2 ± 2·7 × 10−2 | – | ||
| Room temperature Medium RH (50 ± 5%) | Iodine‐treated | 1·2 × 10−3 ± 5·0 × 10−4 | 6·9 × 10−3 ± 2·9 × 10−3 | 0·76 ± 0·06 |
| Untreated | 5·5 × 10−3 ± 9·2 × 10−4 | – | ||
*The value was obtained by dividing the observed values by the survival fraction (0·17).
After vortexing, one tested filter and one unused iodine‐treated filter were examined under a scanning electron microscope (SEM) (JSM‐6330F; JEOL Ltd., Tokyo, Japan). As shown in Fig. 4, abundant particles were observed in the tested filter compared to the unused filter.
Figure 4.

Scanning electron microscope images of the (a) unused iodine‐treated filter and (b) tested iodine‐treated filter at 2700×.
Discussion
Intrinsic differences in test methods complicate comparison of PRE and VRE values measured for test filters. The PRE was measured for ultrafine particles (i.e. 9·82–162·5 nm), whereas the VRE was measured over the entire particle size range generated from the nebulizer. Even if the PRE for the entire particle size range is calculated by particle counting, its value will still be different from the VRE because of aggregation of virus aerosols and fewer counts of viable virus available for disaggregation in smaller particles than in bigger particles. A viral aggregate is measured by the particle counter as one particle, but it can be assayed as several viruses after collection in the impinger because of dispersion in the collection medium. The number of viable viruses in a big particle is larger than that in an ultrafine particle; thus, the contribution of larger particles collected in the impingers to the infectivity results will be much greater than that of ultrafine particles. This effect was observed in a prior study (Hogan et al. 2005), which reported that the possibility of containing viable viruses increases with the size of particles from MS2 suspension.
In the experiment for sublimation and dissolution of I2, increase of survived viruses as increase of virus concentration in the impinger is presumably due primarily to exhausting the supply of I2 but might also include some shielding effect of aggregated/encased viruses if the aggregate persists in the impinger. Berg et al. (1964) reported that deactivation of viruses by iodine follows first‐order reaction kinetics, and thus reaction rates of iodine with viruses depend on the number and availability of vital sites on the virion. They mentioned a lagged deactivation curve of iodine because of virus clumping and the necessity of time for virus clumping to be separated. A study of the survival of viral particles in aqueous suspension irradiated with ultraviolet light demonstrated that virus survival was strongly dependent on the degree of aggregation among the viral particles (Galasso and Sharp 1965).
The SEM images of the tested filter in Fig. 4 show that many particles still remained on the filter after extraction. One can argue that it is because of inefficient extraction of the vortexing process. However, the extracted fraction from glass fibre HEPA filters (162 ± 61) following the same vortexing procedure was much higher, demonstrating that vortexing extraction was efficient for regular filter media (Li et al. 2008). High retention capability of the electret test filter can be a reason for the low extracted fraction caused by the electrostatic attraction between viral particles and filter media. In the same context, insignificant reaerosolization of the viruses from the test filters is expected. It should be noted that both iodine‐treated and untreated filters presumably have similar retention of viruses. In the test filters, the negatively charged surface of viruses is influenced by an attractive force with the positive resin surface and repulsive force caused by negatively charged functional groups on the filter medium. This property of the test filter implies that a filter medium that is depleted of I2 over time can still serve as an effective medium for trapping viruses, because it has the same attractive and repulsive forces as the untreated filter – the resin surface and by‐product iodide ions remain after consumption of the iodine molecules from the triiodide ions.
The effect of iodine on the infectivity of MS2 collected on the iodine‐treated filter is less certain than previously thought, because similar viable recoveries were observed for the iodine‐treated and untreated filters; however, a strong virucidal effect of I2 was observed in both the VRE of the iodine‐treated filter and free I2 residual experiments. This phenomenon can be explained by two possible reasons: (i) shielding effect of aggregated particles collected on the filter and (ii) high retention capability of the test filters.
Shielding effect
MS2 in suspension is vulnerable to iodine, because the virus is better dispersed in an aqueous medium, whereas in the air it can be aggregated or encased in other constituents of particles that protect it from iodine inactivation. This assertion is supported by the SEM images shown in Fig. 4. Most particles observed in the tested filter are orders of magnitude larger than a single naked MS2, which can be either the MS2 aggregates or substances with MS2 generated from the nebulizer suspension (virus stock suspension in the nebulizer contains milk proteins and organic molecules for virus preservation). Therefore, infectivity of MS2 can be shielded by the outer layer of the aggregates or by encasement in substances present in the nebulization medium. MS2 aggregation generated from the nebulizer, which is caused by hydrophobic interactions between neighbouring protein capsids, has been observed by previous studies (Hogan et al. 2004; Balazy et al. 2006).
High retention capability of the filter
The extracted fractions of both iodine‐treated and untreated filters are significantly lower than the other regular filter media because of the expected high retention of particles on filter media resulting from electrostatic interaction between filter media and charged surface of viral particles, as discussed earlier. It should be stated that this interaction will persist because of the inherent electret property of the resin‐treated surface. Extracted values close to the detection limit can make the effect of iodine on the virus infectivity indistinguishable.
The control experiments carried out in this study with thiosulfate and BSA require that reported data generated in experiments collecting aerosols in aqueous media or on protein gels to measure the biocidal capacity of the iodine‐treated filter be reexamined to consider the possibility of competition by dissolved I2. Significant support for the previously proposed mechanism of charge‐induced capture of iodine from bound triiodide is found in the observation of significant inactivation persisting in a BSA medium that was able to protect suspended virions from inactivation by impinging I2 vapour. However, toxicity of iodine dissolved in the collection medium is likely to be a competing mechanism in warm environments, and the relative importance of each must be determined – or at least factored into the design and analysis processes – at different conditions. Data from a different experimental approach might not encounter this uncertainty, and the assay is only a surrogate for the goal of the technology, enhancing respiratory protection against bioaerosol transmission of pathogens. Both the medium in the impinger and the protein gel have elements in common with respiratory mucosa, and for a person wearing individual protective gear, the time of transit from filter to mucosal surface is similar. However, competition by water and by proteins at the site of impaction might or might not behave the same as in the in vitro systems tested to date. Thus, the ultimate measure of enhancement of protection by surface‐bound iodine – or any other reactive surface on filter fibres – will require data from animal exposure studies.
Acknowledgements
This research was supported by the United States Air Force through grant FA8651‐05‐C‐0136. We are grateful to Safe Life Corp. for supplying the filters for testing. We also thank Yong‐Ho Choi and Ki‐Joon Jeon for their assistance in taking the SEM images and the Particle Engineering Research Center in the University of Florida for providing the SEM.
References
- APHA (1995) Standard Methods for the Examination of Water and Wastewater, Method 4500 CI G. 19th edn Washington, DC: American Public Health Association. [Google Scholar]
- Aranha‐Creado, H. and Brandwein, H. (1999) Application of bacteriophages as surrogates for mammalian viruses: a case for use in filter validation based on precedents and current practices in medical and environmental virology. PDA J Pharm Sci Technol 53, 75–82. [PubMed] [Google Scholar]
- Balazy, A. , Toivola, M. , Adhikari, A. , Sivasubramani, S.K. , Reponen, T. and Grinshpun, S.A. (2006) Do N95 respirators provide 95% protection level against airborne viruses, and how adequate are surgical masks? Am J Infect Control 34, 51–57. [DOI] [PubMed] [Google Scholar]
- Berg, G. , Chang, S.L. and Harris, E.K. (1964) Devitalization of microorganisms by iodine. Virol 22, 469–481. [DOI] [PubMed] [Google Scholar]
- Biswas, P. and Wu, C.Y. (2005) Nanoparticles and the environment. J Air Waste Manag Assoc 55, 708–746. [DOI] [PubMed] [Google Scholar]
- Brion, G.M. and Silverstein, J. (1999) Iodine disinfection of a model bacteriophage, MS2, demonstrating apparent rebound. Water Res 33, 169–179. [Google Scholar]
- Bruchertseifer, H. , Cripps, R. , Guentay, S. and Jaeckel, B. (2003) Analysis of iodine species in aqueous solutions. Anal Bioanal Chem 375, 1107–1110. [DOI] [PubMed] [Google Scholar]
- Carroll, B. (1955) The relative germicidal activity of triiodide and diatomic iodine. J Bacteriol 69, 413–417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CDC (2005) Fact sheet: Basic Information about SARS. Atlanta, GA: Center for Disease Control and Prevention; Available at: http://www.cdc.gov/ncidod/SARS/pdf/factsheet.pdf. [Google Scholar]
- Cecchini, C. , Verdenelli, M.C. , Orpianesi, C. , Dadea, G.M. and Cresci, A. (2004) Effects of antimicrobial treatment on fiberglass‐acrylic filters. J Appl Microbiol 97, 371–377. [DOI] [PubMed] [Google Scholar]
- Chang, S.L. (1958) The use of iodine as a water disinfectant. J Am Pharm Assoc 48, 417–423. [PubMed] [Google Scholar]
- Code of Federal Regulations (CFR) (2002) Respiratory Protective Devices, 42 CFR Part 84. Washington, DC: U.S. Government Printing Office. [Google Scholar]
- Di Ionno, L. and Messier, P.J. (2004) Iodinated thermal‐fused polymer as a biocidal additive to commercial and chemical‐resistant coatings. Surf Coatings Int B: Coating Trans 87, 235–308. [Google Scholar]
- Dubovi, E.J. and Akers, T.G. (1970) Airborne stability of tailless bacterial viruses S‐13 and MS‐2. Appl Microbiol 19, 624–628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eninger, R.M. , Adhikari, A. , Reponen, T. and Grinshpun, S.A. (2008) Differentiating between physical and viable penetrations when challenging respirator filters with bioaerosols. Clean–Soil Air Water 36, 615–621. [Google Scholar]
- Fiegel, J. , Clarke, R. and Edwards, D.A. (2006) Airborne infectious disease and the suppression of pulmonary bioaerosols. Drug Discov Today 11, 51–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fina, L.R. , Hassouna, N. , Horacek, G.L. , Lambert, J.P. and Lambert, J.L. (1982) Viricidal capability of resin‐triiodide demand‐type disinfectant. Appl Environ Microbiol 44, 1370–1373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foarde, K.K. and James, T.H. (2001) Determine the efficacy of antimicrobial treatments of fibrous air filters. ASHRAE Trans 107, 156–170. [Google Scholar]
- Galasso, G.J. and Sharp, D.G. (1965) Effect of particle aggregation on survival of irradiated vaccinia virus. J Bacteriol 90, 1138–1142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilmour, M.N. and Wicksell, E.K. (1972) Bactericidal and sporicidal activity of a quaternary ammonium resin‐triiodide complex. Antimicrob Agents Chemother 22, 417–418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenwood, F.C. , Hunter, W.M. and Glover, J.S. (1963) The preparation of 131I‐labelled human growth hormone of high specific radioactivity. Biochem J 89, 114–123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hach (2003) Iodine Method 8031. Loveland, CO: Hach company. [Google Scholar]
- Hatch, G.L. , Lambert, J.L. and Fina, L.R. (1980) Some properties of the quaternary ammonium anion‐exchange resin‐triiodide disinfectant for water. Ind Eng Chem Prod Res Dev 19, 259–263. [Google Scholar]
- Heimbuch, B.K. and Wander, J.D. (2006) Bioaerosol Challenges to Antimicrobial Surface Treatments: Enhanced Efficacy Against MS2 Coli Phage of Air Filter Media Coated with Polystyrene‐4‐Methyltrimethylammonium Triiodide. Report No. AFRL‐ML‐TY‐TP‐2006‐4527, Tyndall AFB, FL: Air Force Research Laboratory. [Google Scholar]
- Heimbuch, B.K. , Hodge, J.E. and Wander, J.D. (2007) Viral Penetration of High Efficiency Particulate Air (HEPA) Filters. Report No. AFRL‐ML‐TY‐TP‐2007‐4512, Tyndall AFB, FL: Air Force Research Laboratory. [Google Scholar]
- Hinds, W.C. (1999) Filtration. In Aerosol Technology. 2nd edn pp. 182–205. New York, NY: John Wiley & Sons, Inc. [Google Scholar]
- Hogan, C.J. , Lee, M.H. and Biswas, P. (2004) Capture of viral particles in soft X‐ray‐enhanced corona systems: charge distribution and transport characteristics. Aerosol Sci Technol 38, 475–486. [Google Scholar]
- Hogan, C.J. , Kettleson, E.M. , Lee, M.H. , Ramaswami, B. , Angenent, L.T. and Biswas, P. (2005) Sampling methodologies and dosage assessment techniques for submicrometre and ultrafine virus aerosol particles. J Appl Microbiol 99, 1422–1434. [DOI] [PubMed] [Google Scholar]
- Hsu, Y.C. (1964) Resistance of infectious RNA and transforming DNA to iodine which inactivates F2 phage and cells. Nature 203, 152–153. [Google Scholar]
- Hughes, W.L.J. and Straessle, R. (1950) Preparation and properties of serum and plasma proteins. XXIV. Iodination of human serum albumin. J Am Chem Soc 72, 452–457. [Google Scholar]
- Jennison, M.W. (1942) Atomizing of mouth and nose secretions into the air as revealed by high speed photography. Aerobiology 17, 106–128. [Google Scholar]
- Lee, K.‐K. and Ellis, A.E. (1991) A novel method for specific visualization of serum albumin in polyacrylamide gels by iodine staining. Electrophoresis 12, 382–383. [DOI] [PubMed] [Google Scholar]
- Lee, J.H. , Wu, C.Y. , Wysocki, K.M. , Farrah, S. and Wander, J. (2008) Efficacy of iodine‐treated biocidal filter media against bacterial spore aerosols. J Appl Microbiol 105, 1318–1326. [DOI] [PubMed] [Google Scholar]
- Li, H.W. , Wu, C.Y. , Tepper, F. , Lee, J.H. and Lee, C.N. (2008) Removal and retention of viral aerosols by a novel alumina nanofiber filter. J Aerosol Sci 40, 65–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maillard, J.Y. (2001) Virus susceptibility to biocides: an understanding. Rev Med Microbiol 12, 63–74. [Google Scholar]
- Marchin, G.L. , Fina, L.R. , Lambert, J.L. and Fina, G.T. (1983) Effect of Resin disinfectants I3 and I5 on Giardia muris and Giardia lamblia . Appl Environ Microbiol 46, 965–969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchin, G.L. , Silverstein, J. and Brion, G.M. (1997) Effect of microgravity on Escherichia coli and MS‐2 bacteriophage disinfection by iodinated resins. Acta Astronaut 40, 65–68. [DOI] [PubMed] [Google Scholar]
- Messier, P.J. (2004) Method and System for Treating Air with an Iodinated Resin. US 6,696,055 B2. Williston, VT, USA: Triosyn Holdings Inc. [Google Scholar]
- Muus, J. , Coons, A.H. and Salter, W.T. (1941) Thyroidal activity of iodinated serum albumin. IV. The effect of progressive iodination. J Biol Chem 139, 135–144. [Google Scholar]
- NIOSH (2005) Determination of Particulate Filter Penetration to Test Against Liquid Particulates for Negative Pressure, Air‐Purifying Respirators Standard Testing Procedure (STP). Procedure No. RCT‐APR‐STP‐0051, 0052, 0053, 0054, 0055, 0056, Revision 1.1. Pittsburgh, PA: National Institute for Occupational Safety and Health, National Personal Protective Technology Laboratory. [Google Scholar]
- Opresko, L. , Wiley, H.S. and Wallace, R.A. (1980a) Proteins iodinated by the chloramine‐T method appear to be degraded at an abnormally rapid rate after endocytosis. Proc Natl Acad Sci U S A 77, 1556–1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OSHA (1994) Iodine in Workplace Atmosphere (Impregnated Activated Beaded Carbon). Washington, DC: Occupational Safety & Health Administration. [Google Scholar]
- OSHA (2000) Safety and Health Topics: Iodine. Washington, DC: Occupational Safety & Health Administration. [Google Scholar]
- Portenier, I. , Haapasalo, H. , Rye, A. , Waltimo, T. , Ørstavik, D. and Haapasalo, M. (2001) Inactivation of root canal medicaments by dentine, hydroxyapatite and bovine serum albumin. Int Endod J 34, 184–188. [DOI] [PubMed] [Google Scholar]
- Prescott, L.M. , Harley, J.P. and Klein, D.A. (2002) Microbiology. New York, NY: McGraw‐Hill Companies Inc. [Google Scholar]
- Ratnesar‐Shumate, S. , Wu, C.Y. , Wander, J. , Lundgren, D. , Farrah, S. , Lee, J.H. , Wanakule, P. , Blackburn, M. et al. (2008) Evaluation of physical capture efficiency and disinfection capability of an iodinated biocidal filter medium. Aerosol Air Qual Res 8, 1–18. [Google Scholar]
- Richardson, A.W. , Eshbaugh, J.P. , Hofacre, K.C. and Gardner, P.D. (2006) Respirator Filter Efficiency Against Particulate and Biological Aerosols Under Moderate to High Flow Rates. Report No. ECBC‐CR‐085, Aberdeen Proving Ground, MD: U.S. Army Edgewood Chemical Biological Center. [Google Scholar]
- Shahkrokh, B.K. (1943) Preparation and antigenic properties of a crystalline labeled antigen. J Biol Chem 151, 659–664. [Google Scholar]
- Smith, M.F. , Bryant, S. , Welch, S. and Digenis, G.A. (1984) Labeling suspended aerosol particles with short‐lived radionuclides for determination of particle deposition. J Pharm Sci 73, 1091–1094. [DOI] [PubMed] [Google Scholar]
- Sobsey, M.D. , Oldham, C.E. and McCall, D.E. (1990) Comparative inactivation of hepatitis a virus and other enteroviruses in water by iodine. Water Sci Technol 24, 331–337. [Google Scholar]
- Taylor, S.L. , Fina, L.R. and Lambert, J.L. (1970) New water disinfectant – an insoluble quaternary ammonium resin‐triiodide combination that releases bactericide on demand. Appl Microbiol 20, 720–722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trouwbor, T. and De Jong, J.C. (1973) Interaction of some factors in mechanism of inactivation of bacteriophage‐MS2 in aerosols. Appl Microbiol 26, 252–257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyrrell, D.A.J. (1967) The spread of viruses of the respiratory tract by the airborne route. Symp Soc Gen Microbiol 17, 286–306. [Google Scholar]


