Abstract
The average respiration rate for an adult is 12–20 breaths per minute, which constantly exposes the lungs to allergens and harmful particles. As a result, respiratory diseases, which includes asthma, chronic obstructive pulmonary disease (COPD) and acute lower respiratory tract infections (LTRI), are a major cause of death worldwide. Although asthma, COPD and LTRI are distinctly different diseases with separate mechanisms of disease progression, they do share a common feature – airway inflammation with intense recruitment and activation of granulocytes and mast cells. Neutrophils, eosinophils, basophils, and mast cells are crucial players in host defense against pathogens and maintenance of lung homeostasis. Upon contact with harmful particles, part of the pulmonary defense mechanism is to recruit these cells into the airways. Despite their protective nature, overactivation or accumulation of granulocytes and mast cells in the lungs results in unwanted chronic airway inflammation and damage. As such, understanding the bright and the dark side of these leukocytes in lung physiology paves the way for the development of therapies targeting this important mechanism of disease. Here we discuss the role of granulocytes in respiratory diseases and summarize therapeutic strategies focused on granulocyte recruitment and activation in the lungs.
Keywords: Granulocytes, Asthma, Chronic obstructive pulmonary disease, Acute lower respiratory tract infections, Targeted therapy in the lungs, Inflammation
1. Introduction
The lungs are constantly exposed to chemicals, particles, allergens and microorganisms from the external environment, resulting in their susceptibility to infections and injury. Respiratory diseases are known to be one of the leading causes of death worldwide, accounting for more than 10 % of all disability-adjusted life-years (DALYs) – an estimate of the amount of active life lost due to a disease [1]. Common respiratory disorders can be grouped into allergic inflammation (e.g. asthma), destructive pathologies, such as chronic obstructive pulmonary disease (COPD) and acute lower respiratory infections. Among the chronic respiratory pathological conditions, COPD, asthma and pulmonary fibrosis still lack efficient treatment. Moreover, despite the availability of vaccines and antibiotics, acute lower respiratory tract infections are still the sixth leading cause of death among all diseases, and the first cause of death in children under the age of five [2]. Therefore, understanding the pathophysiology of these respiratory tract diseases is crucial for the development of new therapeutic strategies to decrease or prevent disease burden.
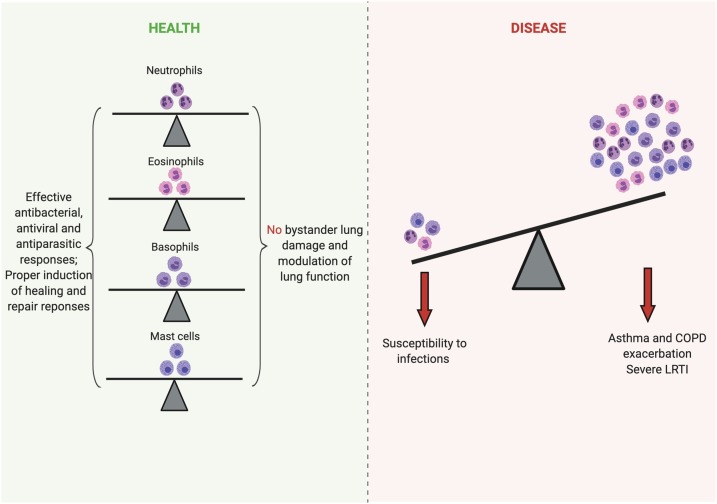
Inflammation is a common feature of several respiratory diseases. Of note, the influx of granulocytes – neutrophils, eosinophils, basophils – and mast cells activation are important to control proliferation of pathogens and induce tissue repair programs favoring the re-establishment of pulmonary homeostasis. On the other hand, the overactivation or the excessive recruitment of granulocytes in the lungs could lead to severe injury that may exacerbate the disease or worsen its prognosis (Fig. 1 ). In this regard, several pharmacological approaches targeting the recruitment and/or the function of granulocytes have been suggested and, in many cases, implemented in the management of respiratory diseases.
Fig. 1.
Granulocytes in health and disease. Neutrophils, eosinophils, basophils and mast cells are crucial for the maintenance of lung health by preventing potential infections and inducing repair responses when needed. However, overactivation or recruitment of these cells can induce increased lung injury and bronchoconstriction, which aggravates and exacerbates respiratory diseases such as asthma, COPD, and lower respiratory tract infections. (Created with Biorender.com ®).
Basophils, eosinophils and mast cells are classically considered as harmful components in allergies and asthma [[3], [4], [5]]. However, these cells also play important physiological roles in the coordination of defense responses against parasitic infections, tissue repair, tumor control, angiogenesis, among others [[6], [7], [8], [9]]. On the other hand, neutrophils are crucial to control bacterial and fungal infections but may also lead to tissue damage once dysregulated [10,11]. Therefore, a challenging question remains: are granulocytes friends or foes of the inflammatory process?
Here, we summarize the role of granulocytes and mast cells in asthma, COPD and acute respiratory tract infections and discuss the available and novel therapies targeting basophils, neutrophils, mast cells and eosinophils to control these lung diseases.
2. Types of granulocytes and their functions
In general, granulocytes are defined as immune cells that have specialized granules in the cytoplasm and traditionally include neutrophils, eosinophils, and basophils [12]. Mast cells are also granule-containing immune cells, yet being tissue resident cells, they are not typically classified as granulocytes [13].
The granular content of each type of granulocyte is related to different cell functions and is used for the classification of these cells by light microscopy using cytochemical staining methods. Among others, various enzymes, inflammatory mediators and antimicrobial peptides are packed in the cytoplasmic granules of granulocytes and are released upon cell activation.
Granulocytes and mast cells are produced in the bone marrow through the process of haematopoiesis [12,13]. The bone marrow provides suitable niches for the production of the pluripotent and self-renewed hematopoietic stem cells (HSCs) that give rise to the lymphoid or myeloid multipotent progenitors (MPPs) [14,15]. MPPs eventually generate the lymphoid or myeloid lineages that include, among other cell types, lymphocytes and granulocytes, respectively [14]. Of interest, although the lineage origin of mast cells is still debated, mature mast cells arise from mast cell progenitors that circulate as agranular cells and enter the tissues where they complete their development into specific subsets of long-lived differentiated mast cells [15,16].
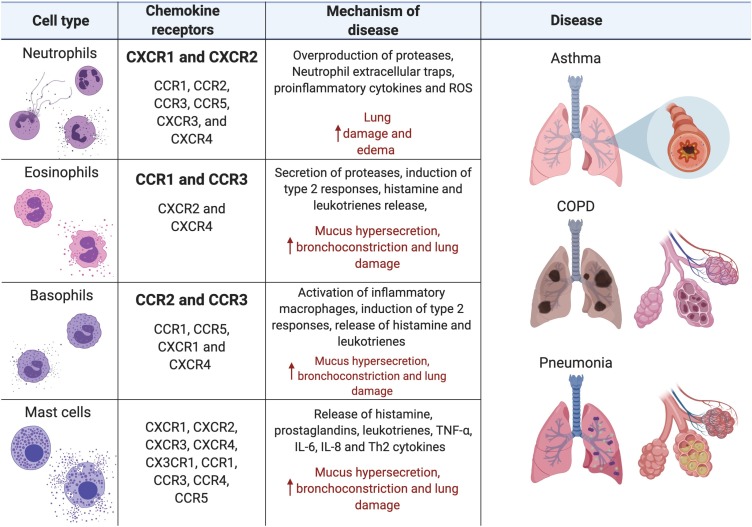
The proliferation and maturation of granulocytes in the bone marrow requires approximately 7–12 days before their release into the bloodstream (circulating leukocytes) and their homing to different tissues (resident leukocytes), including the lungs [17,18]. Resident eosinophils, neutrophils, basophils and mast cells, present in the lung mucosa in physiological conditions, are important for the mucosal immune surveillance and the maintenance of organ homeostasis [6,7,19]. A given insult in the lungs, such as infection, injury and allergens, induces the recruitment of mature granulocytes from the circulation through different chemokine gradients depending on the nature of the stimulus [20]. Upon activation, resident mast cells and granulocytes can also secrete cytokines and chemokines, thus enhancing the accumulation of leukocytes into the airways [21]. Different stimuli are detected by innate immune receptors in pulmonary cells (e.g. pattern recognition receptors – PRRs) and trigger inflammatory signaling cascades that eventually result in the production of distinct repertoires of chemokines. Receptors on the surface of eosinophils, neutrophils and basophils can recognize specific chemokines mediating the recruitment of specific subsets of granulocytes in response to a given stimulus (Fig. 2 ) [20]. Activated granulocytes and/or mast cells in the lungs mediate defense responses against invaders but can also contribute to immunopathology.
Fig. 2.
Granulocyte Mechanisms of Disease. Neutrophil activation and degranulation lead to the release of proteases, antimicrobial peptides, peroxidases, cytokines, and ROS that increase lung edema and damage lung epithelial and endothelial cells. Eosinophil, basophil and mast cells activation magnify type 2 responses while the secretion of proteases, histamine, leukotrienes and prostaglandins increase mucus secretion, bronchoconstriction and damage to the epithelial cells. (Created with Biorender.com ®).
2.1. Neutrophils
Neutrophils are the most abundant granulocytes in the blood and rapidly respond to viral, bacterial and fungal infections [20]. Morphologically, human neutrophils have a multilobe nucleus and numerous, rather heterogeneous, cytoplasmic granules that are small and stain light-pink or purple-blue following treatment with chemically neutral dyes. The granular content of mature neutrophils include a number of antimicrobial peptides, myeloperoxidase (MPO), the serine proteases proteinase 3 (PR3), cathepsin G, neutrophil elastase (NE), the enzymatic inactive protease cationic antimicrobial protein of 37 kd (CAP37, aka azurocidin) and the neutrophil serine protease 4 (NSP4), cysteine proteases, lactoferrin, lipocalin, metalloproteinases and gelatinase [22]. In health, circulating neutrophils infiltrate the lungs, in a diurnal pattern coordinating physiological surveillance responses [10] and regulating the numbers of alveolar macrophages [19].
The recognition of a given inflammatory stimulus by resident alveolar macrophages and pulmonary parenchyma cells lead to the production of chemokines (e.g. CXCL1, CXCL2, CXCL8), cytokines (e.g. IL-6, TNF-α) and other pro-inflammatory mediators (e.g. leukotriene (LT)B4) that promote a significant and rapid recruitment of neutrophils into the lungs [23]. Neutrophil inflammatory chemokine receptors are classically CXCR1 and CXCR2 [24], however, infiltrating neutrophils can increase expression of other receptors such as CCR1, CCR2, CCR3, CCR5, CXCR3, and CXCR4 [25]. Gradients of proinflammatory mediators increase neutrophil stiffness and expression of adhesion molecules, leading to their consequent retention in pulmonary capillary beds and subsequent transmigration to the sites of infection/injury in the lungs [26]. Activated neutrophils degranulate and release antimicrobial peptides and other preformed mediators that mediate pathogen clearance at the site of infection. Additionally, neutrophils can kill potential pathogens by the extrusion of neutrophil extracellular traps (NETs) and by phagocytosis [20]. Therefore, neutrophils are considered the first line of defence against infections in the lungs [23]. Besides their classical described functions, neutrophils were recently shown to mediate tissue repair [27] and anti-tumour responses [28].
In contrast, the permanence and overactivation of neutrophils in the lungs can cause injury, pulmonary dysfunction and even death [29,30]. Several airway diseases including COPD, bronchiolitis, pneumonia, asthma and acute lung injury are characterized by neutrophil infiltration of the airway wall [31]. Both clinical and experimental data have shown an association between neutrophil numbers and the severity and progression of these airway diseases [[32], [33], [34]]. This dual “face” of neutrophils is further evidenced by the different phenotypes of these cells (N1 versus N2) that can either promote or inhibit lung cancer [35]. In view of the opposing roles of neutrophils in airway diseases, a finely tuned response is ideal to promote an efficient clearance of potential invaders while preventing immune mediated lung injury.
2.2. Eosinophils
Eosinophils are not as abundant as neutrophils in the blood and are characterized by basic granules that are stained in pink or orange with the classical cytology dyes such as eosin. The granular content of eosinophils includes cationic proteins such as eosinophil cationic protein (ECP, aka RNase3), eosinophil-derived neurotoxin (EDN, also known as RNase2), major basic protein (MBP), eosinophil peroxidase (EPO), hydrolytic enzymes and a diverse repertoire of preformed cytokines, chemokines, and growth factors [36]. Eosinophils are recruited into the lungs mostly in response to IL-5, eotaxin or histamine production [37,38]. Moreover, IL-5 is crucial for the production, maturation and survival of eosinophils [39]. Among the inflammatory chemokine receptors, CCR1 and CCR3 are highly expressed in eosinophils [24]. Historically, eosinophils are thought to promote defences against parasitic infections, through the release of their cytoplasmic granular content. However, some studies have shown that eosinophils can also enhance antiviral responses [40,41] and have immunomodulatory functions [42].
The “dark” side of eosinophils is evidenced by their role in asthma. Eosinophil recruitment and activation in the lungs are associated with asthma severity and, therefore, are targets for the development of therapeutic strategies [43]. Because of the contrasting roles of eosinophils in homeostasis and disease, it has been suggested that different phenotypes are associated to distinct contexts. Indeed, the lungs contain a morphological and functional distinct population of resident eosinophils that are important regulators of the T helper (Th) 2 responses during asthma, in contrast to the inflammatory recruited eosinophils [6]. Thus, new eosinophil-directed therapies are expected to consider targeting selected eosinophil phenotypes that are associated with disease development, rather than with their protective functions.
2.3. Basophils
Basophils are easily recognized by their numerous metachromatic-stained granules. Basic pigments such as methylene and toluidine blue stain basophil granules dark purple, in contrast to the blue colour seen with mast cells granules [44]. Basophils constitute a relatively rare population in the bloodstream, thus making them difficult to isolate and study [45]. As such, the immunological roles of basophils have been neglected in comparison to other leukocytes. Basophil recruitment is mainly associated with the activation of CCR2 and CCR3 by inflammatory CCL chemokines (CCL2, CCL5, CCL7, CCL8, CCL11 CCL12, CCL13) [24]. Based on their granular content, basophils share some functions with mast cells. Histamine, chondroitin sulphate, proteolytic enzymes, cysteinyl leukotrienes (cysLTs), PAF and cytokines are released upon basophil activation, and similarly to mast cells, they are related to the pathophysiology of allergies and asthma [46,47]. Moreover, basophils can act as antigen presenting cells and may induce Th2 responses, thus contributing to both the host defence against helminth parasites and chronic allergic inflammation [48].
Interestingly, basophils were also recently shown to regulate alveolar macrophage function and development through the production of granulocyte-macrophage colony-stimulating factor (GM-CSF) [7]. While the physiological actions of basophils are poorly described, their contribution to airway diseases, such as asthma, has been relatively more widely explored. Together with mast cells, activated basophils contribute to type 2 inflammation by secretion of cytokines such as IL-5, IL4, IL-13 and thymic stromal lymphopoietin (TSLP), among others [44]. However, basophils can be distinguished from mast cells by not only their morphological characteristics, but also by their tissue distribution, lifespan and ontogeny. In contrast to mast cells, basophils complete their maturation in the bone marrow and are short-lived cells in the circulation (2–3 days), being recruited to the lungs during inflammation [44]. Yet, new approaches of cell isolation and signalling mapping are required in order to shed light on the function of basophils in both health and disease.
2.4. Mast cells
Mast cells are highly granulated, mononucleated cells developed from the CD34+ progenitors. Once expanded in the bone marrow, these progenitors circulate in the bloodstream to become tissue resident cells. In the tissues mast cell progenitors mature under the influence of cytokines, especially by stem cell factor (SCF), the high affinity ligand of the cKit [49]. Mast cells are mostly known to be activated by the immunoglobulin (Ig) E-dependent pathway. However, they can also be stimulated by several IgE-independent triggers that activate different surface receptors, such as toll-like receptors (TLR), G protein-coupled receptors (GPCRs), complement receptors, lectin receptors and the recently identified human Mas-related G-protein coupled receptor member X2 (MGRX2) [50]. Mast cell chemokine receptors such as CXCR1–4, CX3CR1, CCR1, CCR3–5 are important for mast cell activation [51]. Upon activation, mast cells degranulate and release highly bioactive mediators that are grouped into three main categories: the granule-associated pre-formed mediators, such as tryptase, chymase, histamine, heparin and TNF-α; the de-novo synthesized lipid mediators such as LTC4, D4, E4 and B4, platelet activating factor (PAF) and prostaglandin (PG)D2; and last but not least, an array of cytokines and chemokines, including IL-6, IL-8, IL-4, IL-5, IL-10 and eotaxin, among others [52]. While preformed and the lipid mediators are released within minutes, cytokines and chemokines are released several hours after mast cell activation, during the late phase of the response. Notably, depending on a specific stimulus, mast cells can release all kinds of mediator, or they may differentially release certain kinds of mediators [53].
Human mast cells are typically distinguished by their protease content, being referred to as tryptase-containing (MCT), chymase-containing (MCC) and tryptase/chymase-containing (MCTC) mast cells [52]. In addition, there is strong evidence of further mast cell heterogeneity with respect to their receptors, mediators and their consequent functional tissue specificity. For instance, lung mast cells express high levels of TLRs, while skin mast cells exhibit low levels of this class of receptors [54]. Moreover, anti-IgE-mediated activation was recently shown to cause higher release of LTC4 and PGD2 from isolated lung mast cells, compared to their skin, heart and synovial cavity counterparts [55]. On the other hand, substance P, a MRGPRX2 agonist, failed to induce lipid mediator production from lung mast cells and caused no histamine and tryptase release from both lung and heart mast cells, whereas it induced a significant concentration-dependent release of these mediators from skin mast cells [55]. In the lungs, the main population of mast cells is of the MCT type, which is located mainly in the bronchial and bronchiolar lamina propria [54,56]. Mast cells have also been shown to be activated by several respiratory pathogens, such as Mycobacterium tuberculosis, Mycoplasma pneumonia, influenza virus and the respiratory syncytial virus (RSV) [57,58] and were recently shown to act as antigen presenting cells [59]. Thus, in the lungs, mast cells play a dual role: hey contribute to allergic asthma when activated mainly by IgE-mechanisms, and they also seem to be sentinel cells against different lung pathogens.
3. Granulocytes in airway diseases
3.1. Asthma
Asthma is a chronic inflammatory disease of the airways, affecting over 235 million people worldwide [60,61] and classically characterized by inflammation, mucus hypersecretion, airway hyper-responsiveness (AHR) and airway remodeling [62]. This heterogeneous chronic disorder is orchestrated by various inflammatory cells and cellular components to mount diverse clinical phenotypes and complex underlying endotypes [63]. Granulocytes play a major role in the development of asthma, and distinct granulocyte populations seem to be associated with particular phenotypes and endotypes. Thus, the inflammatory and immune processes in the allergic phenotype are linked to the recruitment of eosinophils into the airways, a cardinal feature of the Th2 response in the lung [64]. On the other hand, there is a sub-population of asthmatic patients that are considered as non-Th2 immunologic responsive, where neutrophils are the main inflammatory cells involved in the pathogenesis. Additional phenotypes/endotypes of asthma include Th1 skewed responses, Th17-high inflammation, obesity– and smoking-associated asthma, exercise-induced bronchoconstriction, and the very late-onset asthma that is associated with the decreased function of the ageing lung [44,63].
Allergic asthma is commonly initiated by an inappropriate immune response towards inhaled allergens [65], such as house dust mite, spores, pollens, animal dander, etc. [66]. The exposure of allergens in the airways can activate receptors on the airway epithelium, such as TLRs and PRRs, to initiate allergic responses [67]. This results in the capture and processing of these antigens by dendritic cells in the basement membrane of the airway epithelium, where they mature and migrate to lymph nodes to present the processed antigen to naïve CD4+ T cells [68,69]. The presence of IL-4 drives the differentiation of naïve CD4+ T cells into Th2 cells. Transmigration of eosinophils into the airway involves a cascade of signaling pathways, where the priming and activation of Th2 cells releases cytokines, including IL-5. Besides mediating eosinophil maturation, migration and survival [39], IL-5, in combination with chemokines like eotaxin, adhesion molecules, such as ICAM-1, VCAM-1, E-selectin and the integrin VLA-4, induces eosinophilia in the airways [70,71]. In fact, the pivotal role of IL-5 in asthma has been evidenced in earlier studies in IL-5-deficient mice showing reduced eosinophil trafficking into the airways and decreased AHR [72]. When activated, eosinophils secrete cytotoxic granule proteins like MBP and EPO, which result in the damage of airway epithelial cells and induce histamine release from mast cells and basophils [73]. Besides eliciting damage to the airways directly through the production of granular proteins, studies have shown that eosinophils are the source of LTC4, which, following conversion to LTD4 and LTE4, is involved in the AHR, mucus hypersecretion and bronchoconstriction in asthma [74].
Mast cells and basophils are also involved in the pathogenesis of asthma [48]. The cytokines IL-4 and IL-13, generated mostly from Th2 cells, induce B cells to undergo isotype class switching from IgM to IgE [75]. The interaction between Th2 and B cells (through CD40 – CD40 L signaling) activates B cells to produce IgE antibodies into the bloodstream, which bind to the high affinity IgE receptors (FcεRI) on the surface of mast cells and basophils [76]. When the lungs are re-exposed to the allergen, the resulting IgE-allergen complexes lead to FcεRI cross-linking, thus triggering mast cell activation and degranulation [76]. The release of pre-formed histamine and TNF-α and the newly-formed arachidonic acid metabolites are largely responsible for the early symptoms of the asthmatic reaction including bronchoconstriction [5,77]. Mast cells also have an active role in the late phase reaction that is mediated via the release of cytokines and chemokines [78]. Another type of histamine-producing cell would be the basophil, although it is not as prominent as mast cells in the IgE-driven responses that is characteristic of allergic asthma [79]. Basophils produce cytokines IL-4 and IL-13 that contribute to the shift towards Th2 inflammation in asthma [80]. While there is evidence showing that the lifespan of basophil can be extended by the binding of IgE to the FcεRI [81], the full extent of basophil contribution to innate immunity and asthma remains to be elucidated.
Neutrophils are also known to play a role in the pathogenesis of asthma. Minimal infiltration of neutrophils is observed in the airways of patients with mild-to-moderate asthma, but it is noticeable in the airways of patients with severe asthma and acute asthma exacerbations [82,83]. Neutrophils can attract eosinophils through IL-8 (or via CXCL1/2 in mice), and induce eosinophil degranulation by secreting neutrophilic lactoferrin and elastase [84]. In addition, augmented levels of IL-17 were shown to correlate with increased neutrophil recruitment and disease severity in asthma patients [85]. Matrix metalloproteinase (MMP)-9 is primarily produced by neutrophils and also promotes eosinophil migration and airway remodeling [86]. Dendritic cell presentation requires MMP-9 for antigen uptake in the airway; therefore, MMP-9 knockout mice display a reduction in allergic airway inflammation [87].
3.2. Granulocyte-targeted therapies for asthma
Corticosteroids (CS) are the gold standard in asthma therapy, and although they are effective at abating airway eosinophilia, it has been consistently shown that they are ineffective against neutrophilic inflammation [88,89]. Furthermore, neutrophilic asthma is generally seen in patients treated with CS, as these drugs can decrease apoptosis of neutrophils and potentially contribute to neutrophil activation [64]. Of note, different studies have evaluated the effect of macrolides, anti-IL17 therapy and CXCR1/2 antagonism in reducing neutrophil recruitment in asthma [[90], [91], [92]]. However, no significant improvement was shown, and further research is needed to evaluate the effect of these treatment strategies in different asthma subpopulations.
Current therapies of asthma can be grouped into those targeting allergic asthma (high serum IgE and atopy), eosinophilic asthma (exacerbations, sputum eosinophils and steroid-dependent asthma), neutrophilic asthma (sputum neutrophils, steroid-resistant and/or non-Th2 phenotype) and chronic airflow obstruction (low lung function and high serum periostin) [93]. Inhaled selective β2-agonists are the most common medication that provides rapid relief of asthma symptoms [94]. They are effective bronchodilators, but they are unable to suppress the ongoing airway inflammation. Short-acting β2-agonists (SABA) provide short-term relief (onset of action in 5 min, duration 4−6 h), while long-acting β2-agonists (LABA) deliver a longer (more than 12 h) bronchodilation [62]. The reduction of AHR by LABA without abating the airway inflammation, leads to false perception of controlling the disease, and result in uncontrolled progression of the inflammatory process [95].
For controlling airway inflammation and preventing damage/remodeling of the airways, inhaled corticosteroids (ICS) are the first line of treatment and, in combination with LABA, they are regarded as the gold standard in the management of asthma. ICS reduce inflammation-associated leukocyte infiltration into the airways and suppress airway inflammation, thus leading to asthma relief and more effective management of the disease [62,96]. Although ICS suppress airway inflammation, they do not cure the disease. Another major drawback is the development of resistance against ICS in a subset of patients, which necessitates the use of higher doses, and, eventually, oral CS administration is needed to systemically suppress the uncontrolled inflammation [62,97,98]. Prolonged oral CS treatment is not ideal due to their numerous side effects, including water retention, lipid and cortisol metabolism dysfunction, cataracts, glaucoma, osteoporosis and increased risk of opportunistic infections [62,96]. It is of note that approximately 10 % of the asthmatic patients respond poorly or do not respond to CS at all, accounting for about 50 % of the total healthcare cost in managing asthma [99].
An alternative approach to reduce bronchoconstriction and eosinophilia is the use of leukotriene receptor antagonists, such as montelukast and pranlukast. They bind to CysLT1 receptors expressed on the airway smooth muscle and block the action of LTC4, LTD4 and LTE4, resulting in bronchodilation and reduction of circulating eosinophils in the blood [100]. While CysLT1 receptor antagonists display clinical improvements in asthmatic symptoms and lung function, they are less effective than ICS. However, they are still in use, as they are orally effective, have less unwanted side effects than CS, and provide an alternative treatment for patients who are resistant to CS [101]. Furthermore, it was recently shown that the activation of the epithelial P2Y receptors can induce bronchoconstriction, whereas montelukast acts as an antagonist for the P2Y6 receptor, suggesting an additional potential action of montelukast in asthma [102].
Rather than broadly suppressing systemic inflammation, recent asthma therapies adopt newer targeted approaches and use therapeutic monoclonal antibodies (mAbs) to manage the disease [103]. The first-in-class biologic intended for the treatment of persistent allergic asthma in patients with high serum IgE and mast cell levels is the IgE-neutralizing humanized mAb omalizumab, which was approved by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) in 2003 and 2005, respectively [99]. In addition to its binding to the portion of IgE that interacts with FcεRI on the surface of mast cells and basophils, omalizumab can down-regulate the expression of FcεRI, thus moderating IgE-mediated responses in asthma [62].
For targeted therapies against eosinophilic asthma, mepolizumab and reslizumab are mAbs that target IL-5 to reduce eosinophilia. Both antibodies neutralize circulating IL-5 and inhibit its binding to the IL-5 receptor (IL-5R), thus resulting in decreased blood eosinophils to be trafficked to the lungs. They were initially shown to be ineffective in clinical trials of unselected asthma patients but they proved effective against placebo when patients with severe eosinophilic asthma were selected [104,105]. Both drugs were approved by the FDA and EMA in 2015 and 2016, respectively. In addition, instead of targeting IL-5 itself, benralizumab is a humanized mAb that binds to the IL-5Rα subunit on human eosinophils and basophils and blocks ligand-independent IL-5R signaling. It also acts via antibody-dependent cell-mediated cytotoxicity (ADCC) and consequently depletes IL-5Rα-expressing cells [106]. Several Phase IIb and III clinical trials had shown the clinical efficacy of benralizumab in reducing exacerbations and controlling severe asthma [107,108], prior to its approval by the FDA in 2017 and by the EMA in 2018 for the treatment of patients with severe eosinophilic asthma.
Besides IL-5, the cytokines IL-4 and IL-13 are also important drivers of Th2-mediated inflammation and B-cell differentiation, as well as supporting the recruitment of eosinophils and inducing airway bronchoconstriction [75]. It was hypothesized that targeting IL-4 and/or IL-13 would benefit the downregulation of type 2 inflammation, eosinophils trafficking, and AHR [109,110].While both IL-4 and IL-13 can activate the heterodimeric IL-4R complexes, IL-4 preferentially binds to the IL-4Rα subunit and regulates Th2 function and IgE class switching, whereas IL-13 induces chemokines for the recruitment of eosinophils into the lungs [76,93]. The anti-IL-4 mAb pascolizumab showed some promise in preclinical studies [111], but it was not further developed due to lack of clinical benefit in a pilot study in patients with symptomatic steroid-naive asthma (ClinicalTrials.gov Identifier: NCT00024544). In addition, pitrakinra (Aerovant) is a recombinant human IL-4 variant that binds to the IL-4Rα subunit and acts as a dual IL-4/IL-13 antagonist, it was initially reported to lack efficacy in a clinical trial that recruited not fully controlled asthmatic patients with ICS and LABA (ClinicalTrials.gov Identifier: NCT00801853). Yet, further analysis revealed that pitrakinra was able to reduce asthma exacerbations in selected trial participants with eosinophilic asthma (ClinicalTrials.gov Identifier: NCT00801853). Similarly, the fully human anti-IL-4Rα mAb AMG 317 also failed to demonstrate clinical efficacy across the entire group of patients with moderate to severe asthma in a Phase II clinical trial [112]. On the other hand, dupilumab, a human mAb targeting the IL-4Rα subunit, blocking both IL-4 and IL-13 pathways, and approved since 2017 for the treatment of moderate-to-severe atopic dermatitis, has been shown to reduce exacerbations and to improve lung function in patients with uncontrolled asthma [113]. Being effective in asthmatic patients that had withdrawn ICS and LABA [114], dupilumab was approved by FDA and EMA in 2018 and 2019, respectively as add-on maintenance therapy for patients with severe uncontrolled asthma characterized by raised blood eosinophils.
Considering the activation of the heterodimeric type II IL-4R complexes by IL-13, this cytokine is also a candidate target for the development of treatment options for chronic airway disease [93]. However, lebrikizumab, a human monoclonal antibody against IL-13, did not significantly improve lung function in patients with severe uncontrolled asthma [115]. Interestingly, patients with higher serum levels of the matricellular protein periostin that may be implicated in asthma pathophysiology, responded better to lebrikizumab than the patients with low periostin levels [115]. The drug was eventually discontinued when another phase III trial displayed lack of efficacy in reducing asthma exacerbations in patients. Lebrikizumab was repositioned by being granted Fast Track designation by the FDA in 2019 for the treatment of atopic dermatitis. Tralokinumab, another anti-IL-13 mAb, also showed inconsistent effects on asthma exacerbation rate in patients with severe, uncontrolled asthma [116] and, similarly to lebrikizumab, was recently been reported to benefit patients with moderate-to-severe atopic dermatitis in a phase III clinical trial [117].
The sustained granulocytic inflammation in asthma suggests that part of asthma pathobiology may be related to an impairment of resolution of inflammation [118]. Resolution of inflammation is an active process coordinated by specialized pro-resolving mediators (SPMs) that leads to termination of inflammation [119]. SPMs mainly act through binding to receptors in cell surface. Of note, eosinophils, neutrophils, mast cells and lung epithelial cells express SPM receptors being able to produce and respond to these molecules [120]. A proper resolution of inflammation assures the reduction of secretion of pro-inflammatory mediators and recruitment of granulocytes while increases clearance of apoptotic cells and induces tissue repair responses [119]. Severity of asthma has been correlated with reduction in the levels of SPMs in bronchoalveolar lavage fluid of patients [121] and treatment strategies that induce resolution have been protective in several preclinical studies by decreasing eosinophil counts in the lungs and preventing degranulation of mast cells [[121], [122], [123]].
Evidently, the outcomes of both preclinical and clinical investigations point to the urgent need to appropriately identify the various phenotypes and endotypes of asthma that may guide the development of more beneficial targeted therapeutic interventions.
Chronic obstructive pulmonary disease (COPD)
COPD is a chronic inflammatory lung disease with persistent airflow obstruction that is presented clinically as emphysema, obstructive bronchitis, exacerbation and lung function decline [124]. COPD continues to be a leading cause of morbidity and mortality worldwide, with an estimated prevalence of 328 million people already being the third leading cause of death worldwide [125]. Neutrophils are the major contributor to the pathogenesis of COPD in which heightened airway neutrophilia is observed in patients, correlating very well with disease severity [32]. This could in part be due to the proteases released by neutrophils that can cause host damage and lead to emphysema [126]. Smoking can release both GM-CSF and granulocyte colony-stimulating factor (G-CSF) from epithelial cells and macrophages, which can stimulate granulocyte production, release and survival [127]. Neutrophils in the peripheral blood bind to endothelial cells via E-selectin and are drawn to the airway by neutrophil chemoattractants, such as CXC ligand-1 (CXCL1), CXCL5, CXCL8 and LTB4 [124]. Secreted granule proteins and serine proteases from neutrophils contribute towards alveolar destruction, inflammation and oxidative stress. Cathepsin G, NE and PR3 have also been reported to contribute towards mucus hypersecretion [128,129]. NETs secreted by activated neutrophils, contain histones, NE and MPO, and are found to be excessive in COPD patients, contributing to disease progression [130].
3.3. Granulocyte-targeted therapies for COPD
Targeting neutrophilic inflammation in COPD is a potential treatment for the disease. Tyrphostin AG825, an inhibitor of the ErbB family of receptor tyrosine kinases (RTKs), was shown to prevent GM-CSF-mediated survival of isolated neutrophils from the blood of COPD patients. This was further validated in mice nebulized with lipopolysaccharide (LPS) and immediately given Tyrphostin AG825 via i.p., in which Tyrphostin AG825 increased percentage of neutrophil apoptosis [131]. Matrine, a bioactive component of Sophora flavescens Ait (Kushen), was administered daily via gavage in mice exposed to 4 days of cigarette smoke. Matrine was observed to reduce lung neutrophilia and NE activity, mainly through the apoptosis of neutrophils [132]. AZD8999, a novel muscarinic acetylcholine receptor antagonist and β2-adrenoceptor agonist (MABA), was shown to inhibit IL-8, IL-1β and MMP-9 release from human peripheral blood neutrophils stimulated with LPS [133]. AZD5069, a chemokine (C-X-C motif) receptor 2 (CXCR2) antagonist, was shown to reduce NET formation and levels of IL-8/CXCL-8 in sputum and blood neutrophils isolated from COPD patients [134]. Galectin (Gal)-9, a beta-galactoside lectin protein, was able to reduce the number of neutrophils and the levels of MMP-2 and MMP-9 in the bronchoalveolar lavage fluid (BALF) in a porcine pancreatic elastase (PPE)-induced emphysema model. Gal-9 was also able to inhibit the chemotactic activity of neutrophils in vitro [135].
Another candidate for neutrophil-targeted therapy may be IL-26, which is a neutrophil mobilizing cytokine that is increased in BALF of COPD patients and long term smokers [136]. In addition, an alternative treatment strategy may be the use of AZD7986, a competitive and reversible inhibitor of dipeptidyl peptidase 1 (DPP1), which was shown to inhibit whole blood NE activity in healthy volunteers after once-daily dosing for 21–28 days with no serious side effects [137]. Nanomedicine is also opening new avenues for cell-targeted drug treatments [138]. PEGylated immune-conjugated poly(lactic-co-glycolic acid) (PLGA)-nanoparticle (PINP) have been used to deliver drugs specifically to neutrophils [139].
As mentioned above, neutrophils play a major role in microbial killing [20,23], and inhibiting their function may leave the patient susceptible to future infections. Most of the novel therapies highlighted above for neutrophilic inflammation in COPD involve either the apoptosis of neutrophils or inhibition of chemotactic factors modulating neutrophil migration.
Although COPD is mainly observed to have neutrophilic inflammation, eosinophilic inflammation is enhanced during periods of severe exacerbations. Therefore, the eosinophil is a potential target for managing COPD exacerbation [140]. Increased numbers of eosinophils have been reported in the airway of COPD patients, with 40 % of them having eosinophilic airway inflammation, predicting a more favorable response towards bronchodilators and CS therapy [141]. Heightened level of blood eosinophil is also associated to the risk of COPD exacerbation, increased mortality or decreased lung function [141]. The increased eosinophils in COPD could be recruited by IL-5 and type 2 innate lymphoid cells (ILC2), in which ILC2 is recruited by epithelial mediators (e.g. IL-33) [127].
Generally, established anti-eosinophilic therapy using mAbs against IL-5 or IL-5Rα are safe, albeit an increased susceptibility to helminthic infection [142]. Benralizumab, was tested in patients with eosinophilic COPD (ClinicalTrials.gov Identifier: NCT01227278) administered every 4 weeks (Q4W) (weeks 1, 4 and 8), and then Q8W (weeks 16, 24, 32, 40 and 48). It was observed that genes related to basophil or eosinophil, such as the serine protease PRSS33 were downregulated by benralizumab [143]. SB010, a GATA3-specific DNAzyme, was tested in COPD patients with sputum eosinophilia (DRKS00006087), in which patients inhaled SB010 for 28 days. SB010 was observed to decrease sputum eosinophil and IL-5 whilst increasing blood IFN-γ [144]. In general, anti-neutrophilic and anti-eosinophilic therapies are highly relevant to COPD and will play a major role in alleviating disease burden.
Mast cells increase numbers can be observed in COPD patients with centrilobular emphysema, and most likely contribute to airway hyperresponsiveness [127]. Moreover, disease severity has been found to be correlated with increased numbers of MCT in the sputum and serum of COPD patients. However, a recent publication has shown that MCTC numbers are increasing in COPD patients and linked this class of mast cells to improved lungs functions (reviewed in [145]).
Basophils currently play an obscure role in COPD, however a recent study showed that basophil-derived IL-4 plays a crucial role in the initiation of emphysema in a murine COPD model, driving the production of MMP-12 by interstitial macrophages [146]. This highlights that basophils may play a bigger role in COPD and this is worth further investigation.
Acute Lower respiratory tract Infections
For several years, acute lower respiratory tract infections (LRTI) have been one the main causes of death worldwide [2]. According to the Global Burden of Diseases, Injuries, and Risk Factors Study (GBD), pneumonia and bronchiolitis are considered as LTRI, which caused nearly 2.38 million deaths in 2016 [1]. The broad spectrum of pathogens that can cause LTRI –bacteria, viruses and/or fungi- in addition to the crescent antibiotic resistance rates, create challenges for the diagnosis, prevention and treatment of these diseases [147]. Moreover, the pathogenesis of respiratory infections involves a complex interplay between pathogen virulence factors and the host defense responses. Treatment guidelines for LRTI vary among countries, but in general, the recommendation is the use of different pharmacological classes of antimicrobial agents depending on the etiology and severity [147]. Of note, in severe cases of LRTI, antibiotic therapy does not reduce mortality as the exuberant inflammatory responses triggered in the lungs, rather than pathogen proliferation, lead to severe lung injury and respiratory failure [148]. Therefore, the immune responses triggered by infection must be efficient, yet regulated, to assure proper pathogen clearance and minimum tissue damage.
Inflammation is a common feature of LTRI. Potential pathogens that evade the physical barriers of the respiratory tract and reach the lungs, will be detected by immune and non-immune resident cells via PRRs that bind to microbial associated molecular patterns (MAMPs) [149]. Different families of PRRs, such as TLRs, nucleotide-binding and oligomerization-domain proteins, and caspase-recruitment domain helicases recognize several MAMPs of any one microbe. Activation of signaling pathways by PRRs converge to the transcription of several pro-inflammatory genes as adhesion molecules, chemokines and other cytokines. The production of pro-phlogistic mediators culminates with the recruitment of leukocytes into the airways, specially neutrophils [150]. The recruitment of these cells into the airways occurs rapidly, and is mediated by the chemokine gradient produced by activated lung epithelial cells, macrophages and dendritic cells [33]. At the site of infection, neutrophils mediate important host defense functions such as phagocytosis of microorganisms, production of reactive oxygen species (ROS), antimicrobial peptides and proteases, and extrusion of NETs. The crucial role of neutrophils is evidenced by the higher susceptibility of patients to lung infections with deficits in neutrophil quantity (neutropenia) or defects in function (chronic granulomatous disease) [151,152]. Furthermore, neutrophils were shown to be important for the development of adaptive immune responses following viral lung infections [153].
Besides the crucial role of neutrophils conferring protection against pulmonary infections, the exacerbated activation or prolonged recruitment of these cells into the airways may also be detrimental as it can damage the lungs, resulting in lung injury [154]. There are several microorganisms that had evolved strategies to neutralize the neutrophil responses in the lungs by evading phagocytosis, or blocking/cleaving proteases, antimicrobial peptides and ROS [11]. The vast accumulation of activated neutrophils is often associated with a worse prognosis during severe LTRI [33].
Highly virulent influenza A virus (IAV) infections lead to massive recruitment and activation of neutrophils in the airways [155,156]. In this context, instead of being protective, neutrophils are the main inducers of lung injury through the release of tissue-destructive proteases, cytokines and ROS. In other viral lung infections, such as the one caused by RSV, neutrophils are related to severe bronchiolitis, tissue injury and correlated with disease severity [157,158]. On the other hand, the depletion of neutrophils before or during the early-stages of an ongoing viral infection, leads to increased proliferation of the virus which contributes to higher morbidity and mortality [159]. The complex, often contrasting functions of neutrophils during respiratory viral infections are also highlighted by their putative dual contribution in the pulmonary inflammatory responses in rats infected with the rat coronavirus (rCoV) [160], as well as by the identification of the high ratio of neutrophils to lymphocytes as an independent risk factor for severe pneumonia during the coronavirus disease 2019 (COVID-19) pandemic [161,162]. Indeed, the neutrophilic infiltration and increased secretion of pro-inflammatory cytokines has been shown to contribute with the severity of COVID-19 demanding efforts to reduce inflammation in the advanced stage of disease [163].
Therefore, a timely regulated neutrophilic response must be achieved to confer proper clearance of virus without being harmful to the host. In order to assess whether modulating neutrophilic inflammation during viral lung infections would improve disease outcome, several preclinical studies have been done [164]. Treatment strategies that decrease, rather than block, the recruitment of neutrophils shows interesting potential in controlling lung viral infections [33]. Similarly, bacterial and fungal pulmonary infections are often associated with a pronounced recruitment of neutrophils. The accumulation and over activation of these cells impair the gaseous exchange in the lungs and increase cell wall disruption [165]. Interestingly, the unregulated inflammatory responses in the lungs can result in bacteria overgrowth and dissemination [166,167]. The production of proteases and cytokines by neutrophils contributes to the lung epithelial barrier disruption that results in bacteremia [168].
3.4. Granulocyte-targeted therapies for LRTI
Drugs that are used to impact neutrophil function or numbers in inflammatory diseases, such as the 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitors (statins), CSs and macrolides, are considered potential adjunctive therapy strategy targeting the inflammatory exacerbations in severe pulmonary infections [[169], [170], [171]]. However, associative and clinical studies evaluating the impact of these drugs are not consistent, as larger number of patients is required for clearer results. Based on their pleiotropic anti-inflammatory and immunomodulatory effects, HMG-CoA reductase inhibitors were tested as potential therapies for severe pneumonia [172,173]. In experimental models of pulmonary infection, statins were shown to diminish lung injury, neutrophilic inflammation and improve survival, with greater response when combined with antibiotics [174]. However, numerous retrospective clinical studies have obtained conflicting results while evaluating the impact of statins on respiratory tract infections. While most of these studies observed some beneficial effects of statins on pneumonia severity [[175], [176], [177], [178]], others found no significant disease improvement in the treated group against placebo [179]. Of note, while the reduced recruitment of neutrophils into the lungs after treatment with statins might decrease post-infection lung injury [180], changes in neutrophil function, rather than numbers, were shown to be crucial for the beneficial role of statins during pneumonia. A recent clinical trial showed that treatment of pneumonia patients with HMG-CoA reductase inhibitors was protective by improving neutrophil function and chemotaxis [181]. Unfortunately, it is unclear whether the protective effects observed would be seen in other populations (such as young adults, or patients taking statins chronically). In addition, the treatment schedules, route of administrations and the potential interactions of drugs used to treat pneumonia (such as clarithromycin) are of concern [182].
Similar controversies have been shown when using CSs to treat severe LRTI patients. Current clinical data suggest a potential beneficial effect in the management of severe pneumonia characterized by a “hyper inflammatory” state [[183], [184], [185], [186], [187]]. The mechanisms underlying this protective role of CSs as adjunctive therapy for pneumonia and bronchiolitis include the decreased production of pro-inflammatory cytokines and the consequent decreased recruitment of neutrophils [188]. Nevertheless, there is no consensus in the dosage and type of CSs therapy among different clinical trials, and the induction of immunosuppression by steroids might worsen, instead of treating LRTI.
Recently, new therapeutic strategies directly targeting the recruitment of neutrophils have been carried out. Drugs developed to block the receptor CXCR2 which serves as a binding site for neutrophil-related chemokines, had been tested to treat pulmonary infections caused by influenza and S. pneumoniae [[189], [190], [191]], those included danirixin (GSK1325756) and DF2162. Preclinical studies have shown that inhibition of CXCR2 during influenza infection reduced neutrophil numbers in the airways, and prevented lung injury and mortality, with additive effects when combined with antiviral drugs [191,192]. Phase II clinical studies were recently completed, and safety was not compromised since the viral clearance was not impaired. However, the assessment of treatment efficacy in reducing the severity of disease outcomes was limited due to the small number of patients evaluated (ClinicalTrials.gov Identifier: NCT02927431) [193].
Ideally, therapeutic strategies for the exacerbations of the neutrophilic responses during LRTI should target the harmful potentials of neutrophils while maintaining or enhancing their capacity to kill invading pathogens. Targeting specific neutrophil functions, rather than completely blocking their recruitment, might yield some benefit. In this regard, inhibiting NET release [194] or neutrophil serine proteases [195] have been shown to attenuate the deleterious role of inflammation during pneumonia. In addition, induction of resolution of inflammation has been pointed as a remarkably interesting strategy to reduce disease severity during pneumonia [119]. One pivotal feature of immunoresolvents is enhancing antimicrobial responses while modulating neutrophilic inflammation, therefore, the harmful immunosuppression caused by anti-inflammatory drugs is prevented [196]. Annexin A1 and SPMs such as lipoxins and resolvins have been shown to be protective during viral and bacterial pneumonia reducing neutrophil recruitment and activation while preventing lung damage [[197], [198], [199], [200]]. Of interest, part of the immunomodulatory effects of statins might be related to SPM induction [201]. In order to effectively translate these findings to new therapeutics development, proof of concept studies in humans are necessary.
In addition to neutrophils, other granulocytes also play a role in the defense responses during LRTI. Eosinophils can be recruited into the airways during viral infections and elicit a protective response against IAV [41], RSV [202], parainfluenza [203], rhinovirus [204] and the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2; COVID-19) [205]. Among the different eosinophil granule proteins, ribonucleic acid degrading enzymes (RNases) have antiviral properties and, together with the production of nitric oxide (NO) from eosinophils, can contribute to virus clearance in the lungs [206,207]. This protective role also extends for bacterial infections: eosinophils can recognize, ingest and kill the bacteria [208].
Eosinophil can extrude DNA extracellular traps to entrap bacteria and other extracellular pathogens [209]. However, their exact functions during an in vivo bacterial infection in the lungs are yet to be elucidated. Besides their roles in host defenses, eosinophils can also be detrimental during LRTI induced by RSV. The heightened eosinophilia observed in the lungs of RSV infected patients is correlated with both airway obstructions and severity of the disease. In this aspect, therapies targeting eosinophils are proposed to control airway hyperresponsiveness post-RSV infection [210]. Such strategies intend to control asthma symptoms that may arise after infection and were discussed before. Because of the limited knowledge on the contribution of eosinophils to disease development in most viral and bacterial lung infections, this granulocyte has not been considered as potential therapeutic target for the majority of LRTI.
A similar antimicrobial role is played by mast cells [145] and basophils [211]. Mast cells express several PRRs that enable their activation following viral or bacterial infections in the lungs [145]. The antimicrobial peptides of mast cells granules enhanced the killing of Klebsiella pneumoniae [212] and Streptococcus pneumoniae [213]. Interestingly, mast cells can indirectly execute phagocytosis of bacteria by secreting cytokines such as TNF-α and IL-6, which recruit neutrophils and macrophages during pulmonary infections [145]. Moreover, human peripheral blood-derived mast cells have been shown to be activated to release type 1 interferons by several viruses including the influenza virus [214]. Conversely, it was also shown that activation of mast cells may be linked to the immunopathology observed during IAV infection [215]. Considering the limited information of mast cells involvement in LTRI, more studies that assess mast cells role in severe pulmonary infections are needed.
Although many studies attempted to describe strategies to modulate granulocytic activation and function during LRTI, there is an unmet medical need for the development of therapeutic approaches that would promote effective pathogens clearance while minimizing unwanted bystander lung injury. The timing of administration, dosage and possible side effects are also parts to be discussed and evaluated carefully through clinical studies. By focusing on the host immune responses, new strategies to treat pulmonary infections may prevent microbial resistance while targeting a broader spectrum of pathogens.
4. Perspectives and conclusion
The early description of granulocytes dates back to the 18th century [216], and since then, there is much interest in understanding granulocytes functions and development. The different granule content of these immune cells is strongly related to their diverse role in host defense processes, where proteases, histamine and antimicrobial agents are released after activation of granulocytes; mediators that are important in killing the potential pathogens and induce repair mechanisms at the site of injury. However, exaggerated recruitment or activation of these granulocytes, and their consequent degranulation, leads to amplification of the inflammatory response and bystander tissue injury (Fig. 2). Therefore, the knowledge gathered regarding the role of granulocytes in health and disease has led to the development of therapeutic strategies that target specific features of granulocyte biology.
The enormous mucosal surface of the lungs allows an efficient gas exchange which is impaired by the intense recruitment of granulocytes that fill the alveoli during disease [165]. Allergens, pollutants, or potential pathogens are recognized by the resident pulmonary cells (e.g. epithelial cells and macrophages) and can initiate a significant inflammatory response that, if uncontrolled, leads to tissue damage and respiratory failure. Therapeutics targeting granulocyte recruitment and function are powerful strategies in controlling different respiratory diseases (Table 1 ). It is important to account the unique aspects of the pathophysiology of each respiratory disease during drug development. Of note, the immunosuppression of patients when treated with CSs increases susceptibility to secondary infections and impair tissue repair [217,218], but may be protective in some specific contexts [219,220]. There are different phenotypes of granulocytes that might be related to distinct pathological conditions or may not even be associated to the induction of disease. Thus, the effectiveness of any drug might rely on the responses of a given phenotype to that particular intervention and does not guarantee the same results in other inflammatory diseases.
Table 1.
Granulocyte-targeted therapies for respiratory diseases.
| Disease | Treatment | Cell target | Evidence |
|---|---|---|---|
| Asthma | Corticosteroids | Eosinophils/Neutrophils | Approved [62,96] |
| Leukotriene receptor antagonist | Eosinophils | Approved [100,102] | |
| IgE-neutralizing humanized Abs | Eosinophils | Approved [62,99] | |
| Basophils | |||
| Mast cells | |||
| IL-5/IL-5R neutralizing humanized Abs | Eosinophils | Approved [104,105,106,107,108] | |
| IL-4R neutralizing humanized Abs | Eosinophils | Preclinical/Clinical studies [113,114] | |
| IL-13 neutralizing humanized Abs | Eosinophils | Clinical studies [115,116,117] | |
| CXCR2 antagonist | Neutrophils | Clinical studies [91] | |
| Human anti-IL-17R monoclonal antibody | Neutrophils | Clinical studies [92] | |
| Macrolides | Neutrophils | Clinical studies [90] | |
| COPD | Inhibitor of the ErbB family of receptor tyrosine kinases | Neutrophils | Preclinical studies [131] |
| Matrine | Neutrophils | Preclinical studies [132] | |
| CXCR2 antagonist | Neutrophils | Clinical studies [134] | |
| Dipeptidyl Peptidase 1 Inhibitor | Neutrophils | Preclinical [137] | |
| IL-5R neutralizing humanized Abs | Eosinophils | Clinical studies [142,143] | |
| GATA3-specifc DNAzyme | Eosinophils | Clinical studies [144] | |
| Acute lower respiratory tract infections | HMG-CoA reductase inhibitors | Neutrophils | Preclinical/Clinical studies [173,174,175,176,177,178,179,180,181] |
| Corticosteroids | Neutrophils | Preclinical/Clinical studies [183,184,185,186,187,188] | |
| Macrolides | Neutrophils | Preclinical/Clinical studies [170,171] | |
| CXCR2 antagonist | Neutrophils | Preclinical/Clinical studies [189,190,191,192,193] | |
| Leukotriene receptor antagonist | Eosinophils | Clinical studies [210] |
The future directions of granulocyte-targeted therapies for airway diseases lie in the development of drugs that act on specific functions or selected phenotypes of granulocytes, while not affecting the resident granulocytic populations responsible for host defenses.
Transparency document
Declaration of Competing Interest
The authors declared no conflict of interests.
Acknowledgements
Francesca Levi-Schaffer's research is funded partly by the Israel Science Foundation, Rosetrees Trust (United Kingdom), Aimwell Charitable Trust (United Kingdom), the United States–Israel Binational Science Foundation (BSF), and the Israel Ministry of Science and Technology. Hong Yong Peh is a recipient of National University of Singapore Overseas Postdoctoral Fellowship and co-funded by Harvard Medical School and Brigham and Women's Hospital.
References
- 1.Collaborators, G.B.D.L.R.I Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory infections in 195 countries, 1990–2016: a systematic analysis for the global burden of disease study 2016. Lancet Infect. Dis. 2018;18(11):1191–1210. doi: 10.1016/S1473-3099(18)30310-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Collaborators, G.B.D.C.o.D Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: a systematic analysis for the global burden of disease study 2016. Lancet. 2017;390(10100):1151–1210. doi: 10.1016/S0140-6736(17)32152-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brooks C.R. Sputum basophils are increased in eosinophilic asthma compared with non-eosinophilic asthma phenotypes. Allergy. 2017;72(10):1583–1586. doi: 10.1111/all.13185. [DOI] [PubMed] [Google Scholar]
- 4.Rignault-Bricard R. IL-3-producing basophils are required to exacerbate airway hyperresponsiveness in a murine inflammatory model. Allergy. 2018;73(12):2342–2351. doi: 10.1111/all.13480. [DOI] [PubMed] [Google Scholar]
- 5.Minai-Fleminger Y., Levi-Schaffer F. Mast cells and eosinophils: the two key effector cells in allergic inflammation. Inflamm. Res. 2009;58(10):631–638. doi: 10.1007/s00011-009-0042-6. [DOI] [PubMed] [Google Scholar]
- 6.Mesnil C. Lung-resident eosinophils represent a distinct regulatory eosinophil subset. J. Clin. Invest. 2016;126(9):3279–3295. doi: 10.1172/JCI85664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cohen M. Lung single-cell signaling interaction map reveals basophil role in macrophage imprinting. Cell. 2018;175(4):1031–1044. doi: 10.1016/j.cell.2018.09.009. e18. [DOI] [PubMed] [Google Scholar]
- 8.Artuc M. Mast cells and their mediators in cutaneous wound healing--active participants or innocent bystanders? Exp. Dermatol. 1999;8(1):1–16. doi: 10.1111/j.1600-0625.1999.tb00342.x. [DOI] [PubMed] [Google Scholar]
- 9.Maizels R.M., Yazdanbakhsh M. Immune regulation by helminth parasites: cellular and molecular mechanisms. Nat. Rev. Immunol. 2003;3(9):733–744. doi: 10.1038/nri1183. [DOI] [PubMed] [Google Scholar]
- 10.Casanova-Acebes M. Neutrophils instruct homeostatic and pathological states in naive tissues. J. Exp. Med. 2018;215(11):2778–2795. doi: 10.1084/jem.20181468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bardoel B.W. The balancing act of neutrophils. Cell Host Microbe. 2014;15(5):526–536. doi: 10.1016/j.chom.2014.04.011. [DOI] [PubMed] [Google Scholar]
- 12.Geering B. Living and dying for inflammation: neutrophils, eosinophils, basophils. Trends Immunol. 2013;34(8):398–409. doi: 10.1016/j.it.2013.04.002. [DOI] [PubMed] [Google Scholar]
- 13.Franco C.B. Distinguishing mast cell and granulocyte differentiation at the single-cell level. Cell Stem Cell. 2010;6(4):361–368. doi: 10.1016/j.stem.2010.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rieger M.A., Schroeder T. Hematopoiesis. Cold Spring Harb. Perspect. Biol. 2012;4(12) doi: 10.1101/cshperspect.a008250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Grootens J. Deciphering the differentiation trajectory from hematopoietic stem cells to mast cells. Blood Adv. 2018;2(17):2273–2281. doi: 10.1182/bloodadvances.2018019539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ribatti D., Crivellato E. Mast cell ontogeny: an historical overview. Immunol. Lett. 2014;159(1–2):11–14. doi: 10.1016/j.imlet.2014.02.003. [DOI] [PubMed] [Google Scholar]
- 17.Mehta H.M., Glaubach T., Corey S.J. Systems approach to phagocyte production and activation: neutrophils and monocytes. Adv. Exp. Med. Biol. 2014;844:99–113. doi: 10.1007/978-1-4939-2095-2_6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ramirez G.A. Eosinophils from physiology to disease: a comprehensive review. Biomed Res. Int. 2018;2018:9095275. doi: 10.1155/2018/9095275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tian F. Pulmonary resident neutrophils regulate the production of GM-CSF and alveolar macrophages. FEBS J. 2016;283(8):1465–1474. doi: 10.1111/febs.13684. [DOI] [PubMed] [Google Scholar]
- 20.Weaver K.Ma.C. 9th edition ed. Garland Science; New York, NY, USA: 2017. Janeway’s Immunobiology. 928. [Google Scholar]
- 21.Campillo-Navarro M. Mast cells in lung homeostasis: beyond type I hypersensitivity. Curr. Respir. Med. Rev. 2014;10(2):115–123. doi: 10.2174/1573398X10666141024220151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cowland J.B., Borregaard N. Granulopoiesis and granules of human neutrophils. Immunol. Rev. 2016;273(1):11–28. doi: 10.1111/imr.12440. [DOI] [PubMed] [Google Scholar]
- 23.Aulakh G.K. Neutrophils in the lung: “the first responders”. Cell Tissue Res. 2018;371(3):577–588. doi: 10.1007/s00441-017-2748-z. [DOI] [PubMed] [Google Scholar]
- 24.Luster A.D. Chemokines--chemotactic cytokines that mediate inflammation. N. Engl. J. Med. 1998;338(7):436–445. doi: 10.1056/NEJM199802123380706. [DOI] [PubMed] [Google Scholar]
- 25.Hartl D. Infiltrated neutrophils acquire novel chemokine receptor expression and chemokine responsiveness in chronic inflammatory lung diseases. J. Immunol. 2008;181(11):8053–8067. doi: 10.4049/jimmunol.181.11.8053. [DOI] [PubMed] [Google Scholar]
- 26.Tregay N. Use of autologous (99m)Technetium-labelled neutrophils to quantify lung neutrophil clearance in COPD. Thorax. 2019;74(7):659–666. doi: 10.1136/thoraxjnl-2018-212509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Peiseler M., Kubes P. More friend than foe: the emerging role of neutrophils in tissue repair. J. Clin. Invest. 2019;129(7):2629–2639. doi: 10.1172/JCI124616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Coffelt S.B., Wellenstein M.D., de Visser K.E. Neutrophils in cancer: neutral no more. Nat. Rev. Cancer. 2016;16(7):431–446. doi: 10.1038/nrc.2016.52. [DOI] [PubMed] [Google Scholar]
- 29.Lien D.C. Physiological neutrophil sequestration in the lung: visual evidence for localization in capillaries. J Appl Physiol (1985) 1987;62(3):1236–1243. doi: 10.1152/jappl.1987.62.3.1236. [DOI] [PubMed] [Google Scholar]
- 30.Downey G.P. Retention of leukocytes in capillaries: role of cell size and deformability. J Appl Physiol (1985) 1990;69(5):1767–1778. doi: 10.1152/jappl.1990.69.5.1767. [DOI] [PubMed] [Google Scholar]
- 31.Cowburn A.S. Advances in neutrophil biology: clinical implications. Chest. 2008;134(3):606–612. doi: 10.1378/chest.08-0422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jasper A.E. Understanding the role of neutrophils in chronic inflammatory airway disease. F1000Res. 2019;8 doi: 10.12688/f1000research.18411.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pechous R.D. With friends like these: the complex role of neutrophils in the progression of severe pneumonia. Front. Cell. Infect. Microbiol. 2017;7:160. doi: 10.3389/fcimb.2017.00160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sur S. Sudden-onset fatal asthma. A distinct entity with few eosinophils and relatively more neutrophils in the airway submucosa? Am. Rev. Respir. Dis. 1993;148(3):713–719. doi: 10.1164/ajrccm/148.3.713. [DOI] [PubMed] [Google Scholar]
- 35.Fridlender Z.G. Polarization of tumor-associated neutrophil phenotype by TGF-beta: "N1" versus "N2" TAN. Cancer Cell. 2009;16(3):183–194. doi: 10.1016/j.ccr.2009.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Melo R.C.N., Weller P.F. Contemporary understanding of the secretory granules in human eosinophils. J. Leukoc. Biol. 2018;104(1):85–93. doi: 10.1002/JLB.3MR1217-476R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Swartzendruber J.A., Byrne A.J., Bryce P.J. Cutting edge: histamine is required for IL-4-driven eosinophilic allergic responses. J. Immunol. 2012;188(2):536–540. doi: 10.4049/jimmunol.1101795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rosenberg H.F., Phipps S., Foster P.S. Eosinophil trafficking in allergy and asthma. J. Allergy Clin. Immunol. 2007;119(6):1303–1310. doi: 10.1016/j.jaci.2007.03.048. quiz 1311–1312. [DOI] [PubMed] [Google Scholar]
- 39.Rothenberg M.E. Eosinophilia. N. Engl. J. Med. 1998;338(22):1592–1600. doi: 10.1056/NEJM199805283382206. [DOI] [PubMed] [Google Scholar]
- 40.Phipps S. Eosinophils contribute to innate antiviral immunity and promote clearance of respiratory syncytial virus. Blood. 2007;110(5):1578–1586. doi: 10.1182/blood-2007-01-071340. [DOI] [PubMed] [Google Scholar]
- 41.Samarasinghe A.E. Eosinophils promote antiviral immunity in mice infected with influenza a virus. J. Immunol. 2017;198(8):3214–3226. doi: 10.4049/jimmunol.1600787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rosenberg H.F., Dyer K.D., Foster P.S. Eosinophils: changing perspectives in health and disease. Nat. Rev. Immunol. 2013;13(1):9–22. doi: 10.1038/nri3341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Foster P.S. Targeting eosinophils in asthma. Curr. Mol. Med. 2008;8(6):585–590. doi: 10.2174/156652408785748013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kubo M. Mast cells and basophils in allergic inflammation. Curr. Opin. Immunol. 2018;54:74–79. doi: 10.1016/j.coi.2018.06.006. [DOI] [PubMed] [Google Scholar]
- 45.Schwartz C., Eberle J.U., Voehringer D. Basophils in inflammation. Eur. J. Pharmacol. 2016;778:90–95. doi: 10.1016/j.ejphar.2015.04.049. [DOI] [PubMed] [Google Scholar]
- 46.Chirumbolo S. State-of-the-art review about basophil research in immunology and allergy: is the time right to treat these cells with the respect they deserve? Blood Transfus. 2012;10(2):148–164. doi: 10.2450/2011.0020-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wakahara K. Basophils are recruited to inflamed lungs and exacerbate memory Th2 responses in mice and humans. Allergy. 2013;68(2):180–189. doi: 10.1111/all.12072. [DOI] [PubMed] [Google Scholar]
- 48.Karasuyama H. Newly discovered roles for basophils: a neglected minority gains new respect. Nat. Rev. Immunol. 2009;9(1):9–13. doi: 10.1038/nri2458. [DOI] [PubMed] [Google Scholar]
- 49.Robida P.A. Human eosinophils and mast cells: birds of a feather flock together. Immunol. Rev. 2018;282(1):151–167. doi: 10.1111/imr.12638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Komi D.E.A. The role of mast cells in IgE-Independent lung diseases. Clin. Rev. Allergy Immunol. 2020 doi: 10.1007/s12016-020-08779-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Migalovich-Sheikhet H. Novel identified receptors on mast cells. Front. Immunol. 2012;3:238. doi: 10.3389/fimmu.2012.00238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Elieh Ali Komi D., Wohrl S., Bielory L. Mast cell biology at molecular level: a comprehensive review. Clin. Rev. Allergy Immunol. 2019 doi: 10.1007/s12016-019-08769-2. [DOI] [PubMed] [Google Scholar]
- 53.Moon T.C., Befus A.D., Kulka M. Mast cell mediators: their differential release and the secretory pathways involved. Front. Immunol. 2014;5:569. doi: 10.3389/fimmu.2014.00569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Frossi B. Is it time for a new classification of mast cells? What do we know about mast cell heterogeneity? Immunol. Rev. 2018;282(1):35–46. doi: 10.1111/imr.12636. [DOI] [PubMed] [Google Scholar]
- 55.Varricchi G. Heterogeneity of human mast cells with respect to MRGPRX2 receptor expression and function. Front. Cell. Neurosci. 2019;13:299. doi: 10.3389/fncel.2019.00299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Irani A.M., Schwartz L.B. Mast cell heterogeneity. Clin. Exp. Allergy. 1989;19(2):143–155. doi: 10.1111/j.1365-2222.1989.tb02357.x. [DOI] [PubMed] [Google Scholar]
- 57.Bradding P., Arthur G. Mast cells in asthma--state of the art. Clin. Exp. Allergy. 2016;46(2):194–263. doi: 10.1111/cea.12675. [DOI] [PubMed] [Google Scholar]
- 58.Virk H., Arthur G., Bradding P. Mast cells and their activation in lung disease. Transl. Res. 2016;174:60–76. doi: 10.1016/j.trsl.2016.01.005. [DOI] [PubMed] [Google Scholar]
- 59.Kritikou E. Hypercholesterolemia induces a mast Cell-CD4(+) t cell interaction in atherosclerosis. J. Immunol. 2019;202(5):1531–1539. doi: 10.4049/jimmunol.1800648. [DOI] [PubMed] [Google Scholar]
- 60.Organization W.H. 2017. Asthma Fact Sheet.https://www.who.int/en/news-room/fact-sheets/detail/asthma [cited 2020 March 29]; Available from: [Google Scholar]
- 61.Asthma G.If. 2020. Global Strategy for Asthma Management and Prevention.https://ginasthma.org/wp-content/uploads/2020/04/GINA-2020-full-report_-final-_wms.pdf [cited 2020 April 14]; 2020:[Available from: [Google Scholar]
- 62.Fanta C.H. Asthma. N. Engl. J. Med. 2009;360(10):1002–1014. doi: 10.1056/NEJMra0804579. [DOI] [PubMed] [Google Scholar]
- 63.Kuruvilla M.E., Lee F.E., Lee G.B. Understanding asthma phenotypes, endotypes, and mechanisms of disease. Clin. Rev. Allergy Immunol. 2019;56(2):219–233. doi: 10.1007/s12016-018-8712-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wenzel S.E. Asthma phenotypes: the evolution from clinical to molecular approaches. Nat. Med. 2012;18(5):716–725. doi: 10.1038/nm.2678. [DOI] [PubMed] [Google Scholar]
- 65.Martinez F.D., Vercelli D. Asthma. Lancet. 2013;382(9901):1360–1372. doi: 10.1016/S0140-6736(13)61536-6. [DOI] [PubMed] [Google Scholar]
- 66.Galli S.J., Tsai M., Piliponsky A.M. The development of allergic inflammation. Nature. 2008;454(7203):445–454. doi: 10.1038/nature07204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Hammad H., Lambrecht B.N. Dendritic cells and epithelial cells: linking innate and adaptive immunity in asthma. Nat. Rev. Immunol. 2008;8(3):193–204. doi: 10.1038/nri2275. [DOI] [PubMed] [Google Scholar]
- 68.Busse W.W., Lemanske R.F. Advances in immunology - asthma. N. Engl. J. Med. 2001;344(5):350–362. doi: 10.1056/NEJM200102013440507. [DOI] [PubMed] [Google Scholar]
- 69.Larché M., Robinson D.S., Kay A.B. The role of T lymphocytes in the pathogenesis of asthma. J. Allergy Clin. Immunol. 2003;111(3):450–463. doi: 10.1067/mai.2003.169. [DOI] [PubMed] [Google Scholar]
- 70.Wenzel S.E. Asthma: defining of the persistent adult phenotypes. Lancet. 2006;368(9537):804–813. doi: 10.1016/S0140-6736(06)69290-8. [DOI] [PubMed] [Google Scholar]
- 71.Hamelmann E., Gelfand E.W. IL-5-induced airway eosinophilia – the key to asthma? Immunol. Rev. 2001;179(1):182–191. doi: 10.1034/j.1600-065x.2001.790118.x. [DOI] [PubMed] [Google Scholar]
- 72.Foster P.S. Interleukin 5 deficiency abolishes eosinophilia, airways hyperreactivity, and lung damage in a mouse asthma model. J. Exp. Med. 1996;183(1):195–201. doi: 10.1084/jem.183.1.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Barnes P.J. Elsevier Science; 2002. Asthma and COPD: Basic Mechanisms and Clinical Management. [Google Scholar]
- 74.Kariyawasam H.H., Robinson D.S. The role of eosinophils in airway tissue remodelling in asthma. Curr. Opin. Immunol. 2007;19(6):681–686. doi: 10.1016/j.coi.2007.07.021. [DOI] [PubMed] [Google Scholar]
- 75.Corry D.B., Kheradmand F. Induction and regulation of the IgE response. Nature. 1999;402(6760 Suppl):B18–23. doi: 10.1038/35037014. [DOI] [PubMed] [Google Scholar]
- 76.Kabesch M. IL-4/IL-13 pathway genetics strongly influence serum IgE levels and childhood asthma. J. Allergy Clin. Immunol. 2006;117(2):269–274. doi: 10.1016/j.jaci.2005.10.024. [DOI] [PubMed] [Google Scholar]
- 77.Theoharides T.C. Mast cells and inflammation. Biochim. Biophys. Acta. 2012;1822(1):21–33. doi: 10.1016/j.bbadis.2010.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Krishnaswamy G. The human mast cell: functions in physiology and disease. Front Biosci. 2001;6:D1109–27. doi: 10.2741/krishnas. [DOI] [PubMed] [Google Scholar]
- 79.Duffin R. Targeting granulocyte apoptosis: mechanisms, models, and therapies. Immunol. Rev. 2010;236(1):28–40. doi: 10.1111/j.1600-065X.2010.00922.x. [DOI] [PubMed] [Google Scholar]
- 80.Lambrecht B.N., Hammad H. Biology of lung dendritic cells at the origin of asthma. Immunity. 2009;31(3):412–424. doi: 10.1016/j.immuni.2009.08.008. [DOI] [PubMed] [Google Scholar]
- 81.Kawakami T., Galli S.J. Regulation of mast-cell and basophil function and survival by IgE. Nat. Rev. Immunol. 2002;2(10):773–786. doi: 10.1038/nri914. [DOI] [PubMed] [Google Scholar]
- 82.Jatakanon A. Neutrophilic inflammation in severe persistent asthma. Am. J. Respir. Crit. Care Med. 1999;160(5):1532–1539. doi: 10.1164/ajrccm.160.5.9806170. [DOI] [PubMed] [Google Scholar]
- 83.Sampson A. The role of eosinophils and neutrophils in inflammation. Clin. Exp. Allergy. 2000;30:22–27. doi: 10.1046/j.1365-2222.2000.00092.x. [DOI] [PubMed] [Google Scholar]
- 84.Monteseirin J. Neutrophils and asthma. J. Investig. Allergol. Clin. Immunol. 2009;19(5):340–354. [PubMed] [Google Scholar]
- 85.Al-Ramli W. T(H)17-associated cytokines (IL-17A and IL-17F) in severe asthma. J. Allergy Clin. Immunol. 2009;123(5):1185–1187. doi: 10.1016/j.jaci.2009.02.024. [DOI] [PubMed] [Google Scholar]
- 86.Atkinson J.J., Senior R.M. Matrix metalloproteinase-9 in lung remodeling. Am. J. Respir. Cell Mol. Biol. 2003;28(1):12–24. doi: 10.1165/rcmb.2002-0166TR. [DOI] [PubMed] [Google Scholar]
- 87.Vermaelen K., Pauwels R. Accelerated airway dendritic cell maturation, trafficking, and elimination in a mouse model of asthma. Am. J. Respir. Cell Mol. Biol. 2003;29(3):405–409. doi: 10.1165/rcmb.2003-0008OC. [DOI] [PubMed] [Google Scholar]
- 88.Ito K. Steroid-resistant neutrophilic inflammation in a mouse model of an acute exacerbation of asthma. Am. J. Respir. Cell Mol. Biol. 2008;39(5):543–550. doi: 10.1165/rcmb.2008-0028OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Maneechotesuwan K. LOss of control of asthma following inhaled corticosteroid withdrawal is associated with increased sputum interleukin-8 and neutrophils*. Chest. 2007;132(1):98–105. doi: 10.1378/chest.06-2982. [DOI] [PubMed] [Google Scholar]
- 90.Brusselle G.G. Azithromycin for prevention of exacerbations in severe asthma (AZISAST): a multicentre randomised double-blind placebo-controlled trial. Thorax. 2013;68(4):322–329. doi: 10.1136/thoraxjnl-2012-202698. [DOI] [PubMed] [Google Scholar]
- 91.O’Byrne P.M. Efficacy and safety of a CXCR2 antagonist, AZD5069, in patients with uncontrolled persistent asthma: a randomised, double-blind, placebo-controlled trial. Lancet Respir. Med. 2016;4(10):797–806. doi: 10.1016/S2213-2600(16)30227-2. [DOI] [PubMed] [Google Scholar]
- 92.Busse W.W. Randomized, double-blind, placebo-controlled study of brodalumab, a human anti-IL-17 receptor monoclonal antibody, in moderate to severe asthma. Am. J. Respir. Crit. Care Med. 2013;188(11):1294–1302. doi: 10.1164/rccm.201212-2318OC. [DOI] [PubMed] [Google Scholar]
- 93.Chung K.F. New treatments for severe treatment-resistant asthma: targeting the right patient. Lancet Respir. Med. 2013;1(8):639–652. doi: 10.1016/S2213-2600(13)70128-0. [DOI] [PubMed] [Google Scholar]
- 94.Rang H.P. 6th ed. Churchill Livingstone; 2007. Rang & Dale’s Pharmacology. [Google Scholar]
- 95.Beasley R. Call for withdrawal of LABA single-therapy inhaler in asthma. Lancet. 2010;376(9743):750–751. doi: 10.1016/S0140-6736(10)61158-0. [DOI] [PubMed] [Google Scholar]
- 96.Sovijärvi A.R.A. Sustained reduction in bronchial hyperresponsiveness with inhaled fluticasone propionate within three days in mild asthma: time course after onset and cessation of treatment. Thorax. 2003;58(6):500–504. doi: 10.1136/thorax.58.6.500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Barnes P.J. Efficacy of inhaled corticosteroids in asthma. J. Allergy Clin. Immunol. 1998;102(4 Pt 1):531–538. doi: 10.1016/s0091-6749(98)70268-4. [DOI] [PubMed] [Google Scholar]
- 98.Schuh S. High-dose inhaled fluticasone does not replace oral prednisolone in children with mild to moderate acute asthma. Pediatrics. 2006;118(2):644–650. doi: 10.1542/peds.2005-2842. [DOI] [PubMed] [Google Scholar]
- 99.Barnes P.J. Severe asthma: advances in current management and future therapy. J. Allergy Clin. Immunol. 2012;129(1):48–59. doi: 10.1016/j.jaci.2011.11.006. [DOI] [PubMed] [Google Scholar]
- 100.Reiss T.F. Montelukast, a once-daily leukotriene receptor antagonist, in the treatment of chronic asthma: a multicenter, randomized, double-blind trial. Arch. Intern. Med. 1998;158(11):1213–1220. doi: 10.1001/archinte.158.11.1213. [DOI] [PubMed] [Google Scholar]
- 101.Barnes P.J. Biochemical basis of asthma therapy. J. Biol. Chem. 2011;286(38):32899–32905. doi: 10.1074/jbc.R110.206466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Gao Z.G., Jacobson K.A. Purinergic signaling in mast cell degranulation and asthma. Front. Pharmacol. 2017;8:947. doi: 10.3389/fphar.2017.00947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ekaterini Tiligada K.G. Vol. 1. Springer; Cham: 2018. Francesca Levi-Schaffer. (Innovative Drugs for Allergies. Immunopharmacology and Inflammation). [Google Scholar]
- 104.Haldar P. Mepolizumab and exacerbations of refractory eosinophilic asthma. N. Engl. J. Med. 2009;360(10):973–984. doi: 10.1056/NEJMoa0808991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Haldar P. Outcomes after cessation of mepolizumab therapy in severe eosinophilic asthma: A 12-month follow-up analysis. J. Allergy Clin. Immunol. 2014;133(3):921–923. doi: 10.1016/j.jaci.2013.11.026. [DOI] [PubMed] [Google Scholar]
- 106.Ghazi A., Trikha A., Calhoun W.J. Benralizumab – a humanized mAb to IL-5Rα with enhanced antibody-dependent cell-mediated cytotoxicity – a novel approach for the treatment of asthma. Expert Opin. Biol. Ther. 2012;12(1):113–118. doi: 10.1517/14712598.2012.642359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Castro M. Benralizumab, an anti-interleukin 5 receptor α monoclonal antibody, versus placebo for uncontrolled eosinophilic asthma: a phase 2b randomised dose-ranging study. Lancet Respir. Med. 2014;2(11):879–890. doi: 10.1016/S2213-2600(14)70201-2. [DOI] [PubMed] [Google Scholar]
- 108.FitzGerald J.M. Benralizumab, an anti-interleukin-5 receptor α monoclonal antibody, as add-on treatment for patients with severe, uncontrolled, eosinophilic asthma (CALIMA): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2016;388(10056):2128–2141. doi: 10.1016/S0140-6736(16)31322-8. [DOI] [PubMed] [Google Scholar]
- 109.Schmid-Grendelmeier P. Eosinophils express functional IL-13 in eosinophilic inflammatory diseases. J. Immunol. 2002;169(2):1021–1027. doi: 10.4049/jimmunol.169.2.1021. [DOI] [PubMed] [Google Scholar]
- 110.Bao K., Reinhardt R.L. The differential expression of IL-4 and IL-13 and its impact on type-2 immunity. Cytokine. 2015;75(1):25–37. doi: 10.1016/j.cyto.2015.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Hart T.K. Preclinical efficacy and safety of pascolizumab (SB 240683): a humanized anti-interleukin-4 antibody with therapeutic potential in asthma. Clin. Exp. Immunol. 2002;130(1):93–100. doi: 10.1046/j.1365-2249.2002.01973.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Corren J. A randomized, controlled, phase 2 study of AMG 317, an IL-4Rα antagonist, in patients with asthma. Am. J. Respir. Crit. Care Med. 2010;181(8):788–796. doi: 10.1164/rccm.200909-1448OC. [DOI] [PubMed] [Google Scholar]
- 113.Wenzel S. Dupilumab efficacy and safety in adults with uncontrolled persistent asthma despite use of medium-to-high-dose inhaled corticosteroids plus a long-acting β2 agonist: a randomised double-blind placebo-controlled pivotal phase 2b dose-ranging trial. Lancet. 2016;388(10039):31–44. doi: 10.1016/S0140-6736(16)30307-5. [DOI] [PubMed] [Google Scholar]
- 114.Wenzel S. Dupilumab in persistent asthma with elevated eosinophil levels. N. Engl. J. Med. 2013;368(26):2455–2466. doi: 10.1056/NEJMoa1304048. [DOI] [PubMed] [Google Scholar]
- 115.Hanania N.A. Lebrikizumab in moderate-to-severe asthma: pooled data from two randomised placebo-controlled studies. Thorax. 2015;70(8):748–756. doi: 10.1136/thoraxjnl-2014-206719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Panettieri R.A., Jr. Tralokinumab for severe, uncontrolled asthma (STRATOS 1 and STRATOS 2): two randomised, double-blind, placebo-controlled, phase 3 clinical trials. Lancet Respir. Med. 2018;6(7):511–525. doi: 10.1016/S2213-2600(18)30184-X. [DOI] [PubMed] [Google Scholar]
- 117.Wollenberg A. Treatment of atopic dermatitis with tralokinumab, an anti–IL-13 mAb. J. Allergy Clin. Immunol. 2019;143(1):135–141. doi: 10.1016/j.jaci.2018.05.029. [DOI] [PubMed] [Google Scholar]
- 118.Levy B.D., Vachier I., Serhan C.N. Resolution of inflammation in asthma. Clin. Chest Med. 2012;33(3):559–570. doi: 10.1016/j.ccm.2012.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Levy B.D., Serhan C.N. Resolution of acute inflammation in the lung. Annu. Rev. Physiol. 2014;76:467–492. doi: 10.1146/annurev-physiol-021113-170408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Duvall M.G., Bruggemann T.R., Levy B.D. Bronchoprotective mechanisms for specialized pro-resolving mediators in the resolution of lung inflammation. Mol. Aspects Med. 2017;58:44–56. doi: 10.1016/j.mam.2017.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Planaguma A. Airway lipoxin A4 generation and lipoxin A4 receptor expression are decreased in severe asthma. Am. J. Respir. Crit. Care Med. 2008;178(6):574–582. doi: 10.1164/rccm.200801-061OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Karra L. Lipoxin B(4) promotes the resolution of allergic inflammation in the upper and lower airways of mice. Mucosal Immunol. 2015;8(4):852–862. doi: 10.1038/mi.2014.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Levy B.D. Cysteinyl maresins regulate the prophlogistic lung actions of cysteinyl leukotrienes. J. Allergy Clin. Immunol. 2020;145(1):335–344. doi: 10.1016/j.jaci.2019.09.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Butler A., Walton G.M., Sapey E. Neutrophilic inflammation in the pathogenesis of chronic obstructive pulmonary disease. Copd. 2018;15(4):392–404. doi: 10.1080/15412555.2018.1476475. [DOI] [PubMed] [Google Scholar]
- 125.Quaderi S.A., Hurst J.R. The unmet global burden of COPD. Glob Health Epidemiol Genom. 2018;3:e4. doi: 10.1017/gheg.2018.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Pandey K.C., De S., Mishra P.K. Role of proteases in chronic obstructive pulmonary disease. Front. Pharmacol. 2017;8:512. doi: 10.3389/fphar.2017.00512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Barnes P.J. Cellular and molecular mechanisms of asthma and COPD. Clin. Sci. 2017;131(13):1541–1558. doi: 10.1042/CS20160487. [DOI] [PubMed] [Google Scholar]
- 128.Crisford H., Sapey E., Stockley R.A. Proteinase 3; a potential target in chronic obstructive pulmonary disease and other chronic inflammatory diseases. Respir. Res. 2018;19(1):180. doi: 10.1186/s12931-018-0883-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Dey T. Proteases and their inhibitors in chronic obstructive pulmonary disease. J. Clin. Med. 2018;7(9) doi: 10.3390/jcm7090244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Wang Y. Role of inflammatory cells in airway remodeling in COPD. Int. J. Chron. Obstruct. Pulmon. Dis. 2018;13:3341–3348. doi: 10.2147/COPD.S176122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Rahman A. Inhibition of ErbB kinase signalling promotes resolution of neutrophilic inflammation. Elife. 2019;8 doi: 10.7554/eLife.50990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Yu X. Matrine reduces cigarette smoke-induced airway neutrophilic inflammation by enhancing neutrophil apoptosis. Clin. Sci. 2019;133(4):551–564. doi: 10.1042/CS20180912. [DOI] [PubMed] [Google Scholar]
- 133.Milara J. In vitro anti-inflammatory effects of AZD8999, a novel bifunctional muscarinic acetylcholine receptor antagonist /beta2-adrenoceptor agonist (MABA) compound in neutrophils from COPD patients. PLoS One. 2019;14(1):e0210188. doi: 10.1371/journal.pone.0210188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Pedersen F. Neutrophil extracellular trap formation is regulated by CXCR2 in COPD neutrophils. Eur. Respir. J. 2018;51(4) doi: 10.1183/13993003.00970-2017. [DOI] [PubMed] [Google Scholar]
- 135.Horio Y. Protective effect of Galectin-9 in murine model of lung emphysema: involvement of neutrophil migration and MMP-9 production. PLoS One. 2017;12(7):e0180742. doi: 10.1371/journal.pone.0180742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Che K.F. The neutrophil-mobilizing cytokine interleukin-26 in the airways of long-term tobacco smokers. Clin. Sci. 2018;132(9):959–983. doi: 10.1042/CS20180057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Palmer R. Dipeptidyl peptidase 1 inhibitor AZD7986 induces a sustained, exposure-dependent reduction in neutrophil elastase activity in healthy subjects. Clin. Pharmacol. Ther. 2018;104(6):1155–1164. doi: 10.1002/cpt.1053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Cicha I. From design to the clinic: practical guidelines for translating cardiovascular nanomedicine. Cardiovasc. Res. 2018;114(13):1714–1727. doi: 10.1093/cvr/cvy219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Vij N. Neutrophil targeted nano-drug delivery system for chronic obstructive lung diseases. Nanomedicine. 2016;12(8):2415–2427. doi: 10.1016/j.nano.2016.06.008. [DOI] [PubMed] [Google Scholar]
- 140.Bel E.H., Ten Brinke A. New anti-eosinophil drugs for asthma and COPD: targeting the trait! Chest. 2017;152(6):1276–1282. doi: 10.1016/j.chest.2017.05.019. [DOI] [PubMed] [Google Scholar]
- 141.Bafadhel M., Pavord I.D., Russell R.E.K. Eosinophils in COPD: just another biomarker? Lancet Respir. Med. 2017;5(9):747–759. doi: 10.1016/S2213-2600(17)30217-5. [DOI] [PubMed] [Google Scholar]
- 142.Bagnasco D. Targeting Interleukin-5 or Interleukin-5Ralpha: safety considerations. Drug Saf. 2017;40(7):559–570. doi: 10.1007/s40264-017-0522-5. [DOI] [PubMed] [Google Scholar]
- 143.Sridhar S. Modulation of blood inflammatory markers by benralizumab in patients with eosinophilic airway diseases. Respir. Res. 2019;20(1):14. doi: 10.1186/s12931-018-0968-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Greulich T. A GATA3-specific DNAzyme attenuates sputum eosinophilia in eosinophilic COPD patients: a feasibility randomized clinical trial. Respir. Res. 2018;19(1):55. doi: 10.1186/s12931-018-0751-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Arthur G., Bradding P. New developments in mast cell biology: clinical implications. Chest. 2016;150(3):680–693. doi: 10.1016/j.chest.2016.06.009. [DOI] [PubMed] [Google Scholar]
- 146.Shibata S. Basophils trigger emphysema development in a murine model of COPD through IL-4-mediated generation of MMP-12-producing macrophages. Proc Natl Acad Sci U S A. 2018;115(51):13057–13062. doi: 10.1073/pnas.1813927115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Cazzola M. An update on the pharmacotherapeutic management of lower respiratory tract infections. Expert Opin. Pharmacother. 2017;18(10):973–988. doi: 10.1080/14656566.2017.1328497. [DOI] [PubMed] [Google Scholar]
- 148.Muller-Redetzky H. Therapeutic strategies in pneumonia: going beyond antibiotics. Eur. Respir. Rev. 2015;24(137):516–524. doi: 10.1183/16000617.0034-2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Opitz B. Innate immune recognition in infectious and noninfectious diseases of the lung. Am. J. Respir. Crit. Care Med. 2010;181(12):1294–1309. doi: 10.1164/rccm.200909-1427SO. [DOI] [PubMed] [Google Scholar]
- 150.Hayashi F., Means T.K., Luster A.D. Toll-like receptors stimulate human neutrophil function. Blood. 2003;102(7):2660–2669. doi: 10.1182/blood-2003-04-1078. [DOI] [PubMed] [Google Scholar]
- 151.Rawat A., Bhattad S., Singh S. Chronic granulomatous disease. Indian J. Pediatr. 2016;83(4):345–353. doi: 10.1007/s12098-016-2040-3. [DOI] [PubMed] [Google Scholar]
- 152.Turner S.R., Nasir B.S. Infectious thoracic disease in patients with neutropenia. Curr. Probl. Cancer. 2015;39(5):287–291. doi: 10.1016/j.currproblcancer.2015.08.001. [DOI] [PubMed] [Google Scholar]
- 153.Lim K. Neutrophil trails guide influenza-specific CD8(+) T cells in the airways. Science. 2015;349(6252) doi: 10.1126/science.aaa4352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Bou Ghanem E.N. Extracellular adenosine protects against Streptococcus pneumoniae lung infection by regulating pulmonary neutrophil recruitment. PLoS Pathog. 2015;11(8):e1005126. doi: 10.1371/journal.ppat.1005126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Brandes M. A systems analysis identifies a feedforward inflammatory circuit leading to lethal influenza infection. Cell. 2013;154(1):197–212. doi: 10.1016/j.cell.2013.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Tang B.M. Neutrophils-related host factors associated with severe disease and fatality in patients with influenza infection. Nat. Commun. 2019;10(1):3422. doi: 10.1038/s41467-019-11249-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.McNamara P.S. Bronchoalveolar lavage cellularity in infants with severe respiratory syncytial virus bronchiolitis. Arch. Dis. Child. 2003;88(10):922–926. doi: 10.1136/adc.88.10.922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Yasui K. Neutrophil-mediated inflammation in respiratory syncytial viral bronchiolitis. Pediatr. Int. 2005;47(2):190–195. doi: 10.1111/j.1442-200x.2005.02039.x. [DOI] [PubMed] [Google Scholar]
- 159.Tate M.D. Neutrophils ameliorate lung injury and the development of severe disease during influenza infection. J. Immunol. 2009;183(11):7441–7450. doi: 10.4049/jimmunol.0902497. [DOI] [PubMed] [Google Scholar]
- 160.Haick A.K. Neutrophils are needed for an effective immune response against pulmonary rat coronavirus infection, but also contribute to pathology. J. Gen. Virol. 2014;95(Pt 3):578–590. doi: 10.1099/vir.0.061986-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Jingyuan Liu Y.L., Xiang Pan, Lin Pu, Xiong Haofeng, Li Chuansheng, Zhang Ming, Tan Jianbo, Yanli Xu, Song Rui, Song Meihua, Wang Lin, Zhang Wei, Han Bing, Li Yang, Wang Xiaojing, Zhou Guiqin, Zhang Ting, Li Ben, Wang Yanbin, Chen Zhihai, Wang Xianbo. Neutrophil-to-lymphocyte ratio predicts severe illness patients with 2019 novel coronavirus in the early stage. medRxiv. 2020 doi: 10.1186/s12967-020-02374-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Li X. Clinical characteristics of 25 death cases with COVID-19: a retrospective review of medical records in a single medical center, Wuhan, China. Int. J. Infect. Dis. 2020 doi: 10.1016/j.ijid.2020.03.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Shi Y. COVID-19 infection: the perspectives on immune responses. Cell Death Differ. 2020 doi: 10.1038/s41418-020-0530-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Camp J.V., Jonsson C.B. A role for neutrophils in viral respiratory disease. Front. Immunol. 2017;8:550. doi: 10.3389/fimmu.2017.00550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Ware L.B., Matthay M.A. The acute respiratory distress syndrome. N. Engl. J. Med. 2000;342(18):1334–1349. doi: 10.1056/NEJM200005043421806. [DOI] [PubMed] [Google Scholar]
- 166.Holden V.I. Klebsiella pneumoniae siderophores induce inflammation, bacterial dissemination, and HIF-1alpha stabilization during pneumonia. mBio. 2016;7(5) doi: 10.1128/mBio.01397-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Sohail I. Role of inflammatory risk factors in the pathogenesis of Streptococcus pneumoniae. Front. Immunol. 2018;9:2275. doi: 10.3389/fimmu.2018.02275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Pott G.B. Alpha-1 antitrypsin reduces severity of pseudomonas pneumonia in mice and inhibits epithelial barrier disruption and pseudomonas invasion of respiratory epithelial cells. Front. Public Health. 2013;1:19. doi: 10.3389/fpubh.2013.00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Tavares L.P., Teixeira M.M., Garcia C.C. The inflammatory response triggered by Influenza virus: a two edged sword. Inflamm. Res. 2017;66(4):283–302. doi: 10.1007/s00011-016-0996-0. [DOI] [PubMed] [Google Scholar]
- 170.Tsai W.C. Azithromycin blocks neutrophil recruitment in Pseudomonas endobronchial infection. Am. J. Respir. Crit. Care Med. 2004;170(12):1331–1339. doi: 10.1164/rccm.200402-200OC. [DOI] [PubMed] [Google Scholar]
- 171.Min J.Y., Jang Y.J. Macrolide therapy in respiratory viral infections. Mediators Inflamm. 2012;2012:649570. doi: 10.1155/2012/649570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Garnacho-Montero J. Severe community-acquired pneumonia: current management and future therapeutic alternatives. Expert Rev. Anti. Ther. 2018;16(9):667–677. doi: 10.1080/14787210.2018.1512403. [DOI] [PubMed] [Google Scholar]
- 173.Jain M.K., Ridker P.M. Anti-inflammatory effects of statins: clinical evidence and basic mechanisms. Nat. Rev. Drug Discov. 2005;4(12):977–987. doi: 10.1038/nrd1901. [DOI] [PubMed] [Google Scholar]
- 174.de Paula T.P. Treatment with atorvastatin provides additional benefits to imipenem in a model of gram-negative pneumonia induced by Klebsiella pneumoniae in mice. Antimicrob. Agents Chemother. 2018;62(5) doi: 10.1128/AAC.00764-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Atamna A. Statins and outcomes of hospitalized patients with laboratory-confirmed 2017–2018 influenza. Eur. J. Clin. Microbiol. Infect. Dis. 2019;38(12):2341–2348. doi: 10.1007/s10096-019-03684-y. [DOI] [PubMed] [Google Scholar]
- 176.Douglas I., Evans S., Smeeth L. Effect of statin treatment on short term mortality after pneumonia episode: cohort study. BMJ. 2011;342:d1642. doi: 10.1136/bmj.d1642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Mortensen E.M. Population-based study of statins, angiotensin II receptor blockers, and angiotensin-converting enzyme inhibitors on pneumonia-related outcomes. Clin. Infect. Dis. 2012;55(11):1466–1473. doi: 10.1093/cid/cis733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Nielsen A.G. The impact of statin use on pneumonia risk and outcome: a combined population-based case-control and cohort study. Crit Care. 2012;16(4):R122. doi: 10.1186/cc11418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Dublin S. Statin use and risk of community acquired pneumonia in older people: population based case-control study. BMJ. 2009;338:b2137. doi: 10.1136/bmj.b2137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Boyd A.R. Impact of oral simvastatin therapy on acute lung injury in mice during pneumococcal pneumonia. BMC Microbiol. 2012;12:73. doi: 10.1186/1471-2180-12-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Sapey E. Simvastatin improves neutrophil function and clinical outcomes in pneumonia. A pilot randomized controlled clinical trial. Am. J. Respir. Crit. Care Med. 2019;200(10):1282–1293. doi: 10.1164/rccm.201812-2328OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Trieu J. Rhabdomyolysis resulting from interaction of simvastatin and clarithromycin demonstrated by Tc-99m MDP scintigraphy. Clin. Nucl. Med. 2004;29(12):803–804. doi: 10.1097/00003072-200412000-00008. [DOI] [PubMed] [Google Scholar]
- 183.Blum C.A. Adjunct prednisone therapy for patients with community-acquired pneumonia: a multicentre, double-blind, randomised, placebo-controlled trial. Lancet. 2015;385(9977):1511–1518. doi: 10.1016/S0140-6736(14)62447-8. [DOI] [PubMed] [Google Scholar]
- 184.Feldman C., Anderson R. Corticosteroids in the adjunctive therapy of community-acquired pneumonia: an appraisal of recent meta-analyses of clinical trials. J. Thorac. Dis. 2016;8(3):E162–71. doi: 10.21037/jtd.2016.02.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Garcia-Vidal C. Effects of systemic steroids in patients with severe community-acquired pneumonia. Eur. Respir. J. 2007;30(5):951–956. doi: 10.1183/09031936.00027607. [DOI] [PubMed] [Google Scholar]
- 186.Huang J. Efficacy and safety of adjunctive corticosteroids therapy for patients with severe community-acquired pneumonia: a systematic review and meta-analysis. Medicine (Baltimore) 2019;98(13):e14636. doi: 10.1097/MD.0000000000014636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Stern A. Corticosteroids for pneumonia. Cochrane Database Syst. Rev. 2017;12:CD007720. doi: 10.1002/14651858.CD007720.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Ronchetti S. How glucocorticoids affect the neutrophil life. Int. J. Mol. Sci. 2018;19(12) doi: 10.3390/ijms19124090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Miller B.E. The pharmacokinetics and pharmacodynamics of danirixin (GSK1325756)--a selective CXCR2 antagonist --in healthy adult subjects. BMC Pharmacol. Toxicol. 2015;16:18. doi: 10.1186/s40360-015-0017-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Nair P. Safety and efficacy of a CXCR2 antagonist in patients with severe asthma and sputum neutrophils: a randomized, placebo-controlled clinical trial. Clin. Exp. Allergy. 2012;42(7):1097–1103. doi: 10.1111/j.1365-2222.2012.04014.x. [DOI] [PubMed] [Google Scholar]
- 191.Tavares L.P. CXCR1/2 antagonism is protective during influenza and post-influenza pneumococcal infection. Front. Immunol. 2017;8:1799. doi: 10.3389/fimmu.2017.01799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Washburn M.L. Therapeutically attenuating neutrophil recruitment with a CXCR2 antagonist in combination with oseltamivir ameliorates influenza-induced lung injury and disease. Open Forum Infect. Dis. 2019;6(4):ofz106. doi: 10.1093/ofid/ofz106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.GlaxoSmithKline . 2016. Study to Evaluate the Efficacy and Safety of Danirixin Co-administered With Oseltamivir in the Treatment of Adults Hospitalized With Influenza.https://www.clinicalstudydatarequest.com/SearchAllPostings.aspx?searchparam=201023 August 19, 2019 [cited 2020 April 4]; Available from: [Google Scholar]
- 194.Narasaraju T. Excessive neutrophils and neutrophil extracellular traps contribute to acute lung injury of influenza pneumonitis. Am. J. Pathol. 2011;179(1):199–210. doi: 10.1016/j.ajpath.2011.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Polverino E. The role of neutrophil elastase inhibitors in lung diseases. Chest. 2017;152(2):249–262. doi: 10.1016/j.chest.2017.03.056. [DOI] [PubMed] [Google Scholar]
- 196.Basil M.C., Levy B.D. Specialized pro-resolving mediators: endogenous regulators of infection and inflammation. Nat. Rev. Immunol. 2016;16(1):51–67. doi: 10.1038/nri.2015.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Abdulnour R.E. Aspirin-triggered resolvin D1 is produced during self-resolving gram-negative bacterial pneumonia and regulates host immune responses for the resolution of lung inflammation. Mucosal Immunol. 2016;9(5):1278–1287. doi: 10.1038/mi.2015.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Schloer S. The annexin A1/FPR2 signaling axis expands alveolar macrophages, limits viral replication, and attenuates pathogenesis in the murine influenza A virus infection model. FASEB J. 2019;33(11):12188–12199. doi: 10.1096/fj.201901265R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Machado M.G. The Annexin A1/FPR2 pathway controls the inflammatory response and bacterial dissemination in experimental pneumococcal pneumonia. FASEB J. 2020;34(2):2749–2764. doi: 10.1096/fj.201902172R. [DOI] [PubMed] [Google Scholar]
- 200.Sham H.P. 15-epi-Lipoxin A4, resolvin D2, and resolvin D3 induce NF-kappaB regulators in bacterial pneumonia. J. Immunol. 2018;200(8):2757–2766. doi: 10.4049/jimmunol.1602090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Planaguma A. Lovastatin decreases acute mucosal inflammation via 15-epi-lipoxin A4. Mucosal Immunol. 2010;3(3):270–279. doi: 10.1038/mi.2009.141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Percopo C.M. Activated mouse eosinophils protect against lethal respiratory virus infection. Blood. 2014;123(5):743–752. doi: 10.1182/blood-2013-05-502443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Drake M.G. Human and mouse eosinophils have antiviral activity against parainfluenza virus. Am. J. Respir. Cell Mol. Biol. 2016;55(3):387–394. doi: 10.1165/rcmb.2015-0405OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Handzel Z.T. Eosinophils bind rhinovirus and activate virus-specific T cells. J. Immunol. 1998;160(3):1279–1284. [PubMed] [Google Scholar]
- 205.Liu F. Patients of COVID-19 may benefit from sustained lopinavir-combined regimen and the increase of eosinophil may predict the outcome of COVID-19 progression. Int. J. Infect. Dis. 2020 doi: 10.1016/j.ijid.2020.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Rosenberg H.F., Domachowske J.B. Eosinophils, eosinophil ribonucleases, and their role in host defense against respiratory virus pathogens. J. Leukoc. Biol. 2001;70(5):691–698. [PubMed] [Google Scholar]
- 207.Su Y.C. Dual proinflammatory and antiviral properties of pulmonary eosinophils in respiratory syncytial virus vaccine-enhanced disease. J. Virol. 2015;89(3):1564–1578. doi: 10.1128/JVI.01536-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.DeChatelet L.R. Comparison of intracellular bactericidal activities of human neutrophils and eosinophils. Blood. 1978;52(3):609–617. [PubMed] [Google Scholar]
- 209.Mukherjee M., Lacy P., Ueki S. Eosinophil extracellular traps and inflammatory pathologies-untangling the web! Front. Immunol. 2018;9:2763. doi: 10.3389/fimmu.2018.02763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Kim C.K. A randomized intervention of montelukast for post-bronchiolitis: effect on eosinophil degranulation. J. Pediatr. 2010;156(5):749–754. doi: 10.1016/j.jpeds.2009.12.001. [DOI] [PubMed] [Google Scholar]
- 211.Yousefi S. Basophils exhibit antibacterial activity through extracellular trap formation. Allergy. 2015;70(9):1184–1188. doi: 10.1111/all.12662. [DOI] [PubMed] [Google Scholar]
- 212.Thakurdas S.M. The mast cell-restricted tryptase mMCP-6 has a critical immunoprotective role in bacterial infections. J. Biol. Chem. 2007;282(29):20809–20815. doi: 10.1074/jbc.M611842200. [DOI] [PubMed] [Google Scholar]
- 213.Cruse G. Human lung mast cells mediate pneumococcal cell death in response to activation by pneumolysin. J. Immunol. 2010;184(12):7108–7115. doi: 10.4049/jimmunol.0900802. [DOI] [PubMed] [Google Scholar]
- 214.Kulka M. Activation of mast cells by double-stranded RNA: evidence for activation through toll-like receptor 3. J. Allergy Clin. Immunol. 2004;114(1):174–182. doi: 10.1016/j.jaci.2004.03.049. [DOI] [PubMed] [Google Scholar]
- 215.Graham A.C. Inflammatory response of mast cells during influenza A virus infection is mediated by active infection and RIG-I signaling. J. Immunol. 2013;190(9):4676–4684. doi: 10.4049/jimmunol.1202096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Kay A.B. Paul Ehrlich and the early history of granulocytes. Microbiol. Spectr. 2016;4(4) doi: 10.1128/microbiolspec.MCHD-0032-2016. [DOI] [PubMed] [Google Scholar]
- 217.Confalonieri M. To use or not to use corticosteroids for pneumonia? A clinician’s perspective. Monaldi Arch. Chest Dis. 2012;77(2):94–101. doi: 10.4081/monaldi.2012.157. [DOI] [PubMed] [Google Scholar]
- 218.Jantz M.A., Sahn S.A. Corticosteroids in acute respiratory failure. Am. J. Respir. Crit. Care Med. 1999;160(4):1079–1100. doi: 10.1164/ajrccm.160.4.9901075. [DOI] [PubMed] [Google Scholar]
- 219.Meijvis S.C. Dexamethasone and length of hospital stay in patients with community-acquired pneumonia: a randomised, double-blind, placebo-controlled trial. Lancet. 2011;377(9782):2023–2030. doi: 10.1016/S0140-6736(11)60607-7. [DOI] [PubMed] [Google Scholar]
- 220.Confalonieri M. Hydrocortisone infusion for severe community-acquired pneumonia: a preliminary randomized study. Am. J. Respir. Crit. Care Med. 2005;171(3):242–248. doi: 10.1164/rccm.200406-808OC. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.