Introduction
The case for chloroquine and hydroxychloroquine, as treatment of the coronavirus disease 2019 (COVID-19) pandemic, has been voiced not only in medical journals1 but also in the popular press.2 Not only is the efficacy of these antimalarial and anti-inflammatory compounds as antiviral drugs uncertain, but there is also an ongoing debate about their safety.3 In particular, the magnitude of the proarrhythmic potential of these drugs, specifically related to their QT prolongation effects, is questioned: On the one hand, the fact that chloroquine and hydroxychloroquine block the rapid component of the delay rectifier potassium current (Ikr) potassium channel at the myocyte level, therefore causing QT prolongation, is well documented.4 , 5 On the other hand, the Malaria Policy Advisory Committee of the World Health Organization6 affirms that “despite hundreds of millions of doses administered in the treatment of malaria, there have been no reports of sudden unexplained death associated with chloroquine.” By the same token, both the American and European rheumatology societies do not recommend electrocardiographic (ECG) surveillance for their large number of patients with systemic lupus erythematosus and rheumatoid arthritis who receive long-term treatment with hydroxychloroquine. It is on this background, as chloroquine and hydroxychloroquine are being administered to huge numbers of patients with COVID-19,7 , 8 that we report a case of drug-induced torsades de pointes (TdP) due to chloroquine therapy for COVID-19.
Case report
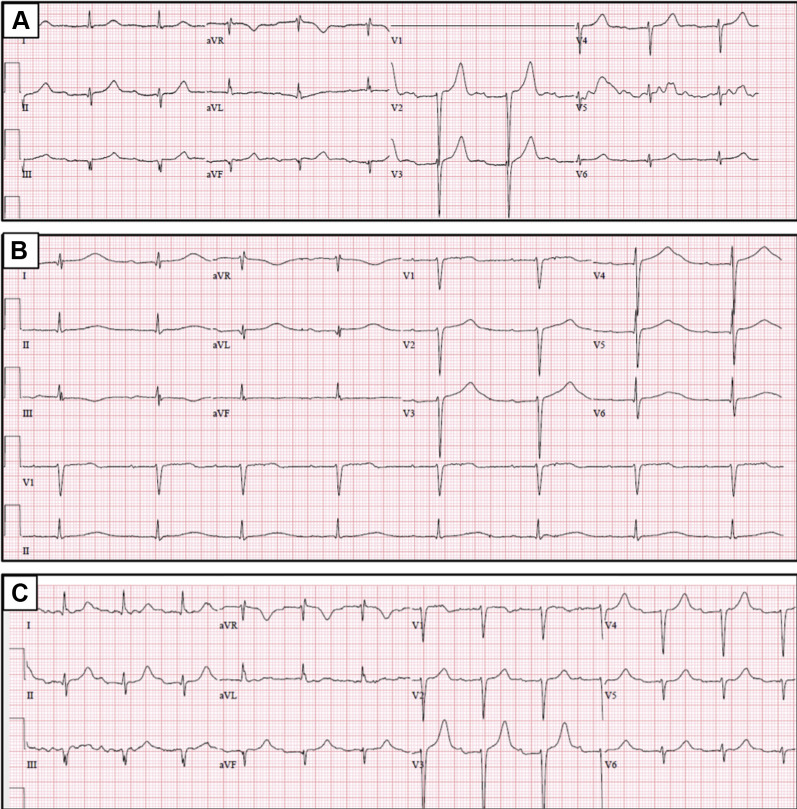
An 84-year-old woman with COVID-19 was admitted to a designated ward. Her medical history includes metastatic breast cancer and a remote history of pulmonary embolism. She also has treated arterial hypertension and evidence of left ventricular hypertrophy but is otherwise free of heart disease. Her medications include bisoprolol, letrozole (an aromatase inhibitor prescribed for breast cancer), memantine (an N-methyl-d-aspartate receptor antagonist used to treat Alzheimer disease), and apixaban. On admission, she was in no obvious distress but her chest radiograph revealed typical bilateral opacities. Her ECG (Figure 1A ) showed sinus rhythm at 63 beats/min, a PR interval of 320 ms, and a narrow QRS complex with small Q waves in the inferior leads. Her QT and corrected QT (QTc) intervals were 450 and 462 ms, respectively. Her laboratory results were significant for positive severe acute respiratory syndrome coronavirus 2 polymerase chain reaction from throat and nasal swabs, lymphopenia, a C-reactive protein level of 14 mg/L, a troponin I level of 22 ng/L (range 0–50 ng/L), a brain natriuretic peptide level of 105 pg/mL, a creatinine level of 0.81 mg/dL, a potassium level of 4.51 mmol/L, and a magnesium level of 1.84 mg/dL, along with normal venous blood gases. Her COVID-19 modified early warning score was 3. The next day, her ambient O2 saturation declined to 93% and her COVID-19 modified early warning score was updated to 5. According to our institutional COVID-19 therapeutic approach, oral therapy with chloroquine diphosphate (Remedica Ltd, Limassol, Cyprus) 500 mg twice daily was initiated. After 5 days of treatment, there was no significant clinical change. A follow-up ECG showed sinus rhythm of 47 beats/min with an extremely prolonged QT interval (QT interval 720 ms and QTc interval 627 ms) (Figure 1B). Chloroquine was discontinued, and other known or suspected QT-prolonging medications as well as nonessential medications were withheld, including memantine and letrozole. Bisoprolol was also discontinued owing to bradycardia. She was placed under continuous ECG monitor and was given additional oral potassium supplements. Her blood potassium level was 4.17 mmol/L. Six hours later, multiple episodes of typical pause-dependent TdP were noted (Figure 2 ). The patient remained asymptomatic and alert. She was treated with intravenous magnesium (2 g of magnesium sulfate injected over 10 minutes), potassium (3 g diluted in 1 L of 0.9% normal saline administered over 5 hours), and lidocaine (100 mg in slow bolus followed by a 2 mg/min infusion). Also, to prevent the pauses precipitating TdP, isoproterenol (1 mg/50 cm3 glucose 5%) was administered intravenously at an infusion rate titrated to raise her heart rate to 80–90 beats/min. This led to immediate resolution of all ventricular arrhythmias. The repeated blood potassium level was 4.43 mmol/L, and the magnesium level was 2.55 mg/dL. Isoproterenol infusion was discontinued after 18 hours. The patient remained free of arrhythmias with no apparent clinical change. Subsequent ECGs showed gradual normalization of her QT interval (Figure 1C). She was discharged 2 weeks later.
Figure 1.
A: Baseline electrocardiogram (ECG). B: Follow-up ECG depicting significant QT interval prolongation. C: Follow-up ECG depicting QT interval normalization.
Figure 2.
Multiple episodes of torsades de pointes as recorded in the remote electrocardiographic monitoring area outside the designated coronavirus disease 2019 ward.
Discussion
We present the first case of TdP in a patient with COVID-19 treated with chloroquine. As in many institutions worldwide, our patient was admitted to a designated COVID-19 ward and chloroquine therapy was initiated. After 5 days on this therapy, she had major QT prolongation and recurrent TdP. As most cases of drug-induced TdP, our patient is female and elderly.9
The fact that our patient was receiving memantine, a drug rated as a medication with a “possible risk of TdP,” likely contributed to the proarrhythmic effects of chloroquine. However, such use of “contributory drugs” in drug-induced long QT syndrome is the rule rather than the exception.9 Furthermore, her QTc interval at the time of hospitalization was only borderline high despite long-term therapy with this drug. Yet, the QTc interval increased from 462 to 627 ms after chloroquine was initiated, clearly pointing to chloroquine as the culprit drug of her TdP. Letrozole, an aromatase inhibitor, is breast cancer therapy that is actually considered safer, from the QT interval point of view, than selective estrogen receptor modulators.10 Both drugs, as well as chloroquine, were discontinued when QT prolongation was noted. Despite this measure, and despite oral potassium supplementation, the patient had multiple episodes of TdP 6 hours later.
Recommendations to help coping with the dilemmas surrounding the use of QT-prolonging medications during the COVID-19 pandemic have recently been published.11, 12, 13, 14 Based on our patient’s baseline QT interval (QTc interval 462 ms), it was permissible to treat her with chloroquine according to each of these 4 documents.11, 12, 13, 14 However, all 4 documents emphasize the importance of discontinuing all QT-prolonging drugs at the onset of chloroquine treatment rather than when excessive QT prolongation is first recognized (as done in our case). Furthermore, all 4 documents emphasize the need for repeated QT assessment at shorter time intervals (rather than after 5 days, as done here). Our patient developed an arrhythmic storm of recurrent TdP 6 hours after discontinuation of chloroquine despite potassium supplementation, probably reflecting the relatively long half-life of chloroquine.15
A huge number of patients are being treated with chloroquine during the COVID-19 pandemic. Excessive QT prolongation, defined as prolongation to a QTc interval of >500 ms, has already been reported for 11%–25% of patients with COVID-19 treated with hydroxychloroquine/azithromycin.7 Many of these patients are elderly and have comorbidities that increase the risk of drug-induced long QT syndrome.
Conclusions
Given its questionable efficacy in the treatment of COVID-19 and its risk of QT prolongation, chloroquine treatment must be considered thoroughly and reviewed on a regular basis. Clinicians should monitor patients with COVID-19 when treating them with chloroquine or other QT-prolonging drugs, with special attention to women, patients with structural heart disease, baseline QT interval on the ECG, concomitant use of other QT-prolonging medications, potassium or magnesium abnormalities, and bradycardia.
Acknowledgments
We thank Morine Fibelman, MD, Albert Kolomansky, MD, and Yoel Angel, MD.
References
- 1.Colson P., Rolain J.-M., Raoult D. Chloroquine for the 2019 novel coronavirus SARS-CoV-2. Int J Antimicrob Agents. 2020;55:105923. doi: 10.1016/j.ijantimicag.2020.105923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Baker P., Rogers K., Enrich D., Haberman M. Trump’s aggressive advocacy of malaria drug for treating coronavirus divides medical community. The New York Times Web site. https://www.nytimes.com/2020/04/06/us/politics/coronavirus-trump-malaria-drug.html
- 3.Ferner R.E., Aronson J.K. Chloroquine and hydroxychloroquine in covid-19. BMJ. 2020;369:m1432. doi: 10.1136/bmj.m1432. [DOI] [PubMed] [Google Scholar]
- 4.Haeusler I.L., Chan X.H.S., Guerin P.J., White N.J. The arrhythmogenic cardiotoxicity of the quinoline and structurally related antimalarial drugs: a systematic review. BMC Med. 2018;16:200. doi: 10.1186/s12916-018-1188-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Risk categories for drugs that prolong QT & induce torsades de pointes (TdP). CredibleMeds Web site. https://crediblemeds.org/index.php/?cID=328
- 6.World Health Organization The cardiotoxicity of antimalarials. Malaria Policy Advisory Committee Meeting, 22–24 March 2017, Geneva, Switzerland, page 36. www.who.int/malaria/mpac/mpac-mar2017-erg-cardiotoxicity-report-session2.pdf
- 7.Chorin E., Dai M., Shulman E. The QT interval in patients with COVID-19 treated with hydroxychloroquine and azithromycin. Nat Med. 2020;26:808–809. doi: 10.1038/s41591-020-0888-2. [DOI] [PubMed] [Google Scholar]
- 8.Borba M.G.S., Val FdA., Sampaio V.S. Chloroquine diphosphate in two different dosages as adjunctive therapy of hospitalized patients with severe respiratory syndrome in the context of coronavirus (SARS-CoV-2) infection: preliminary safety results of a randomized, double-blinded, phase IIb clinical trial (CloroCovid-19 Study) [published online ahead of print April 16, 2020]. medRxiv. https://doi.org/10.1101/2020.04.07.20056424
- 9.Tisdale J.E., Jaynes H.A., Kingery J.R. Development and validation of a risk score to predict QT interval prolongation in hospitalized patients. Circ Cardiovasc Qual Outcomes. 2013;6:479–487. doi: 10.1161/CIRCOUTCOMES.113.000152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Barber M., Nguyen L.S., Wassermann J., Spano J.-P., Funck-Brentano C., Salem J.-E. Cardiac arrhythmia considerations of hormone cancer therapies. Cardiovasc Res. 2019;115:878–894. doi: 10.1093/cvr/cvz020. [DOI] [PubMed] [Google Scholar]
- 11.Giudicessi J.R., Noseworthy P.A., Friedman P.A., Ackerman M.J. Urgent guidance for navigating and circumventing the QTc-prolonging and torsadogenic potential of possible pharmacotherapies for coronavirus disease 19 (COVID-19) Mayo Clin Proc. 2020;95:1213–1221. doi: 10.1016/j.mayocp.2020.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wu C.-I., Postema P.G., Arbelo E. SARS-CoV-2, COVID-19 and inherited arrhythmia syndromes. Heart Rhythm. 2020;17:1456–1462. doi: 10.1016/j.hrthm.2020.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ventricular arrhythmia risk due to hydroxychloroquine-azithromycin treatment for COVID-19 Cardiology Magazine. American College of Cardiology Web site. https://www.acc.org/latest-in-cardiology/articles/2020/03/27/14/00/ventricular-arrhythmia-risk-due-to-hydroxychloroquine-azithromycin-treatment-for-covid-19
- 14.Roden D.M., Harrington R.A., Poppas A., Russo A.M. Considerations for drug interactions on QTc in exploratory COVID-19 (coronavirus disease 2019) treatment. Circulation. 2020;141:e906–e907. doi: 10.1161/CIRCULATIONAHA.120.047521. [DOI] [PubMed] [Google Scholar]
- 15.Smit C., Peeters M.Y.M., van den Anker J.N., Knibbe C.A.J. Chloroquine for SARS-CoV-2: implications of its unique pharmacokinetic and safety properties. Clin Pharmacokinet. 2020;59:659–669. doi: 10.1007/s40262-020-00891-1. [DOI] [PMC free article] [PubMed] [Google Scholar]