Introduction
The Infectious Diseases Society of America (IDSA) produced guidelines for community-acquired pneumonia (CAP) in immunocompetent adults in 1998 and again in 2000 [1, 2]. Because of evolving resistance to antimicrobials and other advances, it was felt that an update should be provided every few years so that important developments could be highlighted and pressing questions answered.
We addressed those issues that the committee believed were important to the practicing physician, including suggestions for initial empiric therapy for CAP. In some cases, only a few paragraphs were needed, whereas, in others, a somewhat more in-depth discussion was provided. Because many physicians focus on the tables rather than on the text of guidelines, it was decided that all of the information dealing with the initial empiric treatment regimens should be in tabular format with footnotes (tables 1–3). The topics selected for updating have been organized according to the headings used in the August 2000 CAP guidelines published in Clinical Infectious Diseases [2]. The major headings were “Epidemiology,” “Diagnostic Evaluation,” “Special Considerations,” “Management,” “Prevention,” and “Performance Indicators,” and each section had a number of subentries. Our current topics are either updates of specific subheadings or are new contributions, and the committee's recommendations are given at the beginning of each section. A summary of prior IDSA recommendations presented in 2000 and the updated and new recommendations can be found in table 4. Ratings of the strength of the supporting evidence and the quality of the data are given in parentheses after each recommendation, and the grading system used to categorize them is in table 5.
Table 1.
Initial empiric therapy for suspected bacterial community-acquired pneumonia (CAP) in immunocompetent adults.
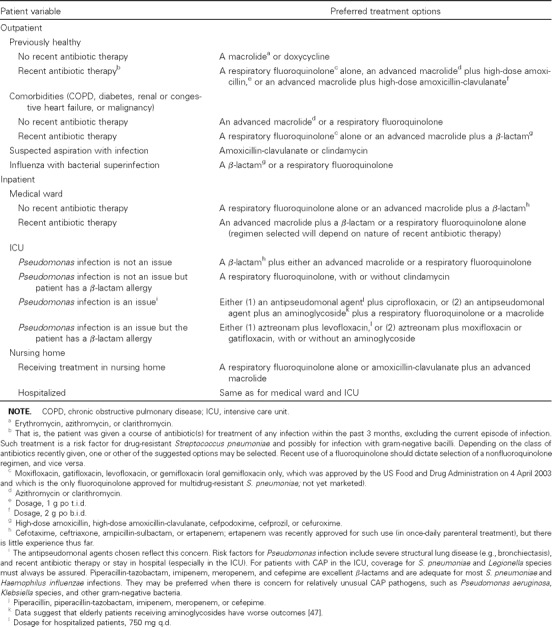
Table 3.
Susceptibility of Streptococcus pneumoniae isolates to commonly used antimicrobial agents, stratified by susceptibility to penicillin, according to 2001 data from the Center for Disease Control's Active Bacterial Core Surveillance (n = 3418) and 2002 NCCLS susceptibility definitions.
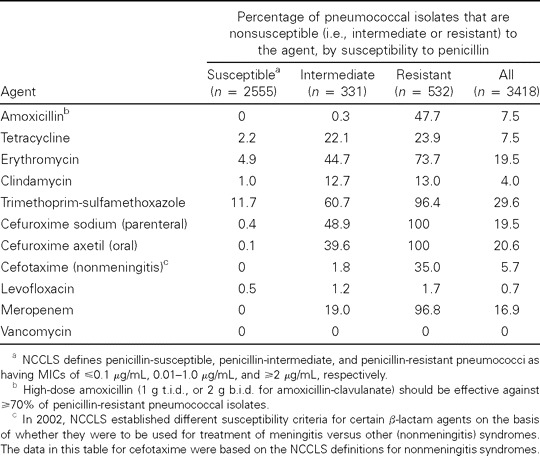
Table 4.
Recommendations for management of community-acquired pneumonia (CAP) in immunocompetent adults: summary of prior Infectious Diseases Society of America (IDSA) recommendations of 2000 and updated and new recommendations for 2003 (in bold).

Table 5.
Infectious Diseases Society of America—United States Public Health Service grading system for rating recommendations in clinical guidelines.

The next guidelines for the treatment of CAP will be a joint effort by the IDSA and the American Thoracic Society (ATS). A working group representing both societies has been formed and is already at work on the next CAP treatment guidelines.
Update on The Initial Site of Treatment Decision
Recommendation 1. The initial site of treatment should be based on a 3-step process: (1) assessment of preexisting conditions that compromise safety of home care; (2) calculation of the pneumonia PORT (Pneumonia Outcomes Research Team) Severity Index (PSI) with recommendation for home care for risk classes I, II, and III; and (3) clinical judgment (A-II).
Recommendation 2. For discharge criteria, during the 24 h prior to discharge to the home, the patient should have no more than 1 of the following characteristics (unless this represents the baseline status): temperature, >37.8°C; pulse, >100 beats/min; respiratory rate, >24 breaths/min; systolic blood pressure, <90 mm Hg; blood oxygen saturation, <90%; and inability to maintain oral intake (B-I).
Comment. Selection of the initial site of treatment, whether home or hospital, continues to be one of the most important clinical decisions made in the treatment of patients with CAP, often determining the selection and route of administration of antibiotic agents, intensity of medical observation, and use of medical resources. This decision is often made in the emergency department, the portal of entry for 75% of the 1 million annual pneumonia admissions in the United States.
Two recent articles suggest that the initial site of treatment decision be selected using a systematic 3-step process [3, 4]. Step 1 involves assessment of any preexisting conditions that compromise the safety of home care, including severe hemodynamic instability, active coexisting conditions that require hospitalization, acute hypoxemia or chronic oxygen dependency, and inability to take oral medications. The second step involves calculation of the pneumonia PSI, with a recommendation for home care for patients in risk classes I, II, or III. A description of how the PSI is derived is shown in Appendix A. The third step involves clinical judgment regarding the overall health of the patient and the suitability for home care. Mitigating factors for step 3 include frail physical condition, severe social or psychiatric problems compromising home care (including a history of substance abuse), and an unstable living situation or homelessness. Clinical judgment should supercede decisions made on the basis of PSI alone.
At the present time, 3 North American medical practice guidelines advocate use of the PSI as an objective measure of risk stratification to help determine the initial site of treatment for CAP [2, 5, 6]. Preliminary results from the Emergency Department Triage of Community-Acquired Pneumonia Study indicate that implementation of the PSI significantly increases the proportion of low-risk patients with pneumonia managed in the emergency department who are treated as outpatients without compromising outcomes, as measured by short-term mortality or subsequent hospitalization [7]. In this randomized, controlled study that involved 32 hospital emergency departments and >3200 patients with CAP, implementation of the PSI with high- and moderate-intensity implementation strategies resulted in a statistically significantly greater proportion of low-risk patients being treated in the outpatient setting.
The committee continues to support use of the PSI as a means of risk stratification and urges that this process be combined with careful assessment of the patient and use of clinical judgment.
Although discharge criteria are not part of the initial site of treatment decision, there are data showing that appropriate use of recommended criteria can reduce mortality [8]. The recommended discharge criteria are that, during the 24 h before discharge to the home, the patient should have no more than 1 of the following characteristics (unless this represents the baseline status): temperature, >37.8°C; pulse, >100 beats/min; respiratory rate, >24 breaths/min; systolic blood pressure, <90 mm Hg; blood oxygen saturation, <90%; and inability to maintain oral intake.
Update on Diagnosis of Chlamydophilia Pneumoniae
Recommendation. Acceptable diagnostic methods for C. pneumoniae pulmonary infections are the demonstration of a 4-fold increase in IgG titer or a single IgM titer of ⩾1 : 16 using a microimmunofluorescence (MIF) serologic test, isolation in tissue culture, or a PCR assay of respiratory secretions using reagents that satisfy optimal criteria for validation (B-II).
Comment. C. pneumoniae is an important respiratory pathogen, but it has also been associated with chronic conditions, such as atherosclerotic cardiovascular disease. Unfortunately, a “gold standard” for the diagnosis of infection with this organism is lacking; this accounts for the wide variation in the reported incidence and prevalence rates for C. pneumoniae.
To get a better idea of the significance of this pathogen, it is imperative that there is agreement with regard to standard diagnostic tests. The IDSA CAP Committee supports recommendations of the Centers for Disease Control and Prevention (CDC) in the United States and the Laboratory Center for Disease Control in Canada [9], which are that potential methods include serologic testing, culture, PCR, and tissue diagnostics or immunohistochemistry (IHC).
For the short-term treatment of patients with CAP, either the detection of IgM by MIF testing or the identification of the organism by culture or PCR of respiratory secretions is most likely to be useful. Four-fold increases in IgG antibody titers, determined by MIF testing, and tissue diagnostic tests may also provide an accurate diagnosis, but they are likely to be more useful in research or epidemiological settings.
For serologic testing, only MIF is acceptable. To document acute infection using serologic methods, a 4-fold increase in the IgG titer or an IgM titer of ⩾1 : 16 must be demonstrated. Use of a single elevated IgG titer is discouraged. Because the reading may vary by 2- to 4-fold from day to day, acute- and convalescent-phase serum samples should be studied in the same run on the same ELISA plate.
Culture methods are important to document the viability of the organism and to provide samples for susceptibility testing. Documentation of a positive culture result requires either propagation of the isolate by means of subsequent passage or confirmation with the use of PCR. There are currently 18 PCR assays available for detection of C. pneumoniae in clinical specimens. Each has its own advantages and disadvantages, but only 4 satisfy the optimal criteria for a validated assay. There is no commercial assay that has been cleared by the US Food and Drug Administration (FDA), and, therefore, PCR is essentially not available except in research laboratories.
A variety of methods have been used to detect C. pneumoniae in tissue specimens, including immunofluorescence, in situ hybridization, and IHC. The main advantage of tissue diagnostic methods is that they allow localization of the pathogen to specific areas and cells within the tissue.
Pneumococcal Urinary Antigen Test: New Addition
Recommendation. The pneumococcal urinary antigen assay is an acceptable test to augment the standard diagnostic methods of blood culture and sputum Gram stain and culture, with the potential advantage of rapid results similar to those for sputum Gram stain (B-II).
Comment. An assay that has recently been cleared by the FDA for pneumococcal antigen detection using a urine sample is now available as a method for the diagnosis of pneumococcal pneumonia in adults. The assay is an immunochromatographic membrane test (ICT) used to detect pneumococcal cell-wall polysaccharide, which is common to all serotypes. Its main advantages are its rapidity (∼15 min using unconcentrated urine samples) and simplicity.
When results of the ICT are compared with the results attained using conventional diagnostic methods for pneumococcal pneumonia in adults, the sensitivity ranges from 50% to 80%, and the specificity is ∼90% depending on the standard of comparison [10–15]. The sensitivity in defining bacteremic pneumococcal disease in adults has been reported to be 70%–90%. In one of the largest published studies to date, Gutierrez et al. [15] performed ICT on concentrated urine samples obtained from 452 adults with CAP. Pneumococcal antigen was detected in 19 (70%) of 27 patients with proven pneumococcal pneumonia. Importantly, of the 269 patients who had pneumonia with no pathogen identified, antigen was detected in 69 (26%), suggesting that a significant percentage of cases that are not diagnosed by standard microbiological tests can be identified with ICT. However, 16 (10%) of 156 samples obtained from patients with pneumonia due to other causes were positive, indicating problems with specificity.
Studies involving children have documented the lack of specificity of ICT [16–18]. Dowell et al. [18] reported that the test result was no more likely to be positive among 88 children with pneumonia than among 198 control subjects, and it was significantly more likely to be positive among those who were nasopharyngeal carriers of pneumococci. Thus, this test is not likely to be useful for distinguishing children with pneumococcal pneumonia from those who are merely colonized, and although the specificity appears to be higher for adults, the colonization status has not been systematically evaluated. Other possible limitations include the possibility of a positive test result for patients with bacteremia due to Streptococcus oralis or Streptococcus mitis, because these pathogens contain a cell-wall polysaccharide antigen similar to that of Streptococcus pneumoniae, and the potential for variable interpretation of a weakly positive test result [19].
For adults, the ICT should increase the yield of identified pathogens for CAP, and a positive result of this test may allow administration of more-focused therapy directed against S. pneumoniae. This test may be particularly helpful for patients receiving antimicrobial therapy at the time of evaluation. However, it should not be considered a substitute for culture, because susceptibility testing will be required to detect specific antimicrobial activity. Additional studies are required to establish the full clinical impact of ICT and to determine its effectiveness in clinical practice. Clinicians should be aware that false-positive results may result from detection of pneumococcal colonization in a patient with pneumonia caused by another agent.
On the basis of the present information, the panel considers this a possibly useful addition to blood culture and other standard tests for identifying pneumococcal pneumonia in adults. The committee believes that, at times, Gram staining of expectorated sputum may yield equally good results in the same time frame.
New Breakpoints for Cefotaxime and Ceftriaxone for S. PNEUMONIAE: New Addition
Recommendation 1. Susceptibility of S. pneumoniae isolates to cefotaxime and ceftriaxone in nonmeningeal infections should be defined as an MIC of ⩽1 ∼g/mL, intermediate should be defined as an MIC of 2 ∼g/mL, and resistant should be defined as an MIC of ⩾4 ∼g/mL (A-III).
Recommendation 2. Cefotaxime or ceftriaxone are the preferred parenteral agents for treatment of pneumococcal pneumonia without meningitis for strains with reduced susceptibility to penicillin but with MICs of cefotaxime or ceftriaxone of <2 ∼g/mL (B-III).
Recommendation 3. Amoxicillin is the preferred antibiotic for oral treatment of pneumococcal pneumonia involving susceptible strains (B-II).
Comment. As of January 2002, the NCCLS increased the MIC breakpoints for cefotaxime and ceftriaxone. The new breakpoints apply to treatment of nonmeningeal infections caused by S. pneumoniae and state that isolates with MICs of ⩽1 ∼g/mL are now considered to be susceptible, those with MICs of 2 ∼g/mL are intermediate, and those with MICs of ⩾4 ∼g/mL are resistant. This is the first time that the NCCLS has provided different interpretive standards for isolates recovered from CSF and non-CSF isolates and should permit more cases of pneumococcal pneumonia to be treated with these agents.
Historically, MIC interpretive standards for pneumococci were derived largely from considerations for treating meningitis (table 3) [20]. Because the level of antibiotic in CSF is only a fraction of that in serum, to be considered susceptible, an organism must have a much lower MIC. The new breakpoints acknowledge that nonmeningeal infections caused by strains formerly considered to be intermediately susceptible and even some that were regarded as resistant can be treated successfully with the usual doses of β-lactam drugs [21–26]. Cefotaxime or ceftriaxone are the preferred agents for pneumococcal pneumonia involving the 95% of strains with an MIC of <2 ∼g/mL. The same could be said for use of amoxicillin for outpatients.
The IDSA committee endorses these changes and encourages clinicians to apply the new interpretive breakpoints as appropriate for the clinical setting. Similar considerations should be applied in the future to breakpoints for penicillin and other effective β-lactam antibiotics.
Special Considerations: Severe Acute Respiratory Syndrome (Sars)—New Addition
Recommendation 1. Health care workers must be vigilant in recognizing SARS because of important epidemiologic implications, which include the potential for rapid spread to close contacts, including health care workers and household contacts (A-III).
Recommendation 2. Diagnostic criteria include clinical and epidemiologic features and may include diagnostic studies for the coronavirus (A-I). Recommended virologic studies for laboratory confirmation are (1) culture for SARS coronavirus, (2) detection of antibody during the acute phase of illness or any time after onset, or (3) detection of SARS coronavirus RNA confirmed by second PCR assay by using a second aliquot of the specimen or a different set of primers. (The panel considers it premature to rate the use of virologic tests.)
Recommendation 3. The major therapeutic intervention is supportive care (B-III).
Recommendation 4. Preventive efforts include proper precautions in patients with suspected or established SARS. These include standard precautions (hand hygiene), contact precautions (use of gowns, goggles, and gloves), and airborne precautions (use of negative-pressure rooms and fit-tested N95 respirators) (A-I).
Comment. “SARS” is the term used to describe outbreaks of pneumonia that were first recognized in Guangdong province in Southern China in late 2002 and that subsequently spread worldwide during March—June of 2003. As of July 2003, >8000 probable cases have been reported from >28 countries worldwide [27]. The heaviest concentrations of cases were identified in mainland China, Hong Kong, and Taiwan, with Singapore, Hanoi, and Toronto also experiencing severe outbreaks.
Transmission and infection control. A majority of early cases occurred among health care workers and family members reporting direct contact with patients who had SARS, and children were relatively spared. Subsequently, rapid international spread by infected airline passengers brought the disease to most continents. Most transmission has been from ill patients to their close contacts, with a relatively high degree of communicability. Direct contact with respiratory secretions and spread via respiratory droplets have been presumed to be the most important modes of transmission, and barrier nursing precautions (with hand washing) have been advocated as the mainstay of control measures. However, the transmission to 13 persons staying in a Hong Kong hotel, to airline passengers, to >200 residents of a single apartment block, and to a number of other persons without recognized close contact with a known case has raised the likelihood of more-remote transmission, whether by fomite or by airborne routes [28, 29].
Health care workers encountering a possible case of SARS should take meticulous safety precautions and should seek immediate advice from an expert in SARS infection control [30]. Protective measures should include standard precautions (hand washing and eye protection), contact precautions (use of gown and gloves), and airborne precautions (isolating the patient in a negative-pressure room and use of well-sealed N95 or greater respirators for all who enter the room). Additional precautions are advised for aerosol-generating procedures, which include many procedures routinely performed on patients undergoing ventilatory support, because of the evidence for transmission to health care workers in these settings, despite the routine use of airborne precautions [31]. This additional protection may include higher levels of respirators (N100 filters or powered air-purifying respirators), full-body isolation suits, and an outer disposable layer of equipment that can be discarded to reduce possible fomite spread [32]. Infection-control precautions should be continued for at least the duration of symptoms, and some precautions may be warranted for a longer period because of the possibility of more-prolonged viral shedding. Updated information should be sought from active reliable Web sites, such as those of the CDC (http://www.cdc.gov) and the World Health Organization (WHO; http://www.who.int/csr/sars/en).
Pathogen. Although a number of potential pathogens were initially identified in patients with SARS, including C. pneumoniae, influenza virus B, and human metapneumovirus, it is now clear that a novel coronavirus is the etiologic agent. Several different laboratories identified an identical strain of this novel coronavirus in patients with SARS by culture of respiratory secretions and lung tissue specimens, electron microscopy, RT-PCR, and seroconversion [33, 34]. Inoculation of macaques with the novel coronavirus, but not with human metapneumovirus, produced a severe respiratory illness akin to SARS in humans [35]. The findings of preliminary reports of detection of the SARS coronavirus in civet cats and a number of other species are provocative, and a number of investigators are attempting to confirm the findings. The sequence of the viral genome has been completed, placing the agent in the coronavirus family and either as a distant member of 1 of the 3 previously described antigenic groups or in a fourth antigenic group [36].
Diagnosis. For surveillance purposes, using clinical and epidemiologic criteria, SARS has been categorized as suspect or probable cases, and the working definitions proposed by WHO have been modified for applicability to particular countries. To meet the CDC criteria for a suspected case, a patient must have fever (temperature, >38°C) and ⩾1 clinical finding of moderate respiratory illness (e.g., cough, shortness of breath, and hypoxia), as well as epidemiologic criteria (travel within 10 days before onset of symptoms to an area with community transmission of SARS, or close contact within 10 days before onset of symptoms with a person known or suspected of having SARS infection) [37, 38]. A probable case is one that meets the definition for suspected cases and, in addition, has either radiographic evidence of pneumonia, respiratory distress syndrome, or autopsy findings consistent with pneumonia or respiratory distress syndrome without an identifiable cause. Current versions of these definitions have changed as new information has become available; in particular, the updated definition of “areas with community transmission of SARS” should be obtained from the CDC or WHO Web sites [37, 39].
As of July 2003, the CDC case definition [37, 38] also incorporates laboratory criteria, although most cases reported in the United States and internationally have been defined using clinical and epidemiologic criteria alone [39]. Culture of the SARS coronavirus is considered solid evidence of infection. However, the various generations of RT-PCR assays have had problems, both with false-positive results and with inconsistent detection of viral genome in both the first days of illness and in the convalescent phase [40]. Because antibodies to SARS coronavirus have not been found in the general population, background SARS coronavirus antibodies do not appear to be a substantial concern [33, 34]. However, the current serologic assays (both ELISA and IFA formats) do not reliably detect antibodies until the titers increase substantially after the second week of illness [40]. According to the CDC, suspect or probable cases are considered to be laboratory confirmed if SARS coronavirus is isolated, if antibody to SARS coronavirus is detected, or if 2 different RT-PCR assays performed with different specimen aliquots identify the coronavirus RNA. Because of the possibility of false-negative results of cultures and RT-PCR assays, only the absence of antibody in a serum specimen obtained >28 days after symptom onset is considered by the CDC to be a negative laboratory test result for SARS coronavirus [37, 38].
These diagnostic tests are not yet available for routine use in clinical laboratories. Clinicians should conduct thorough diagnostic testing to rule out other etiologies in patients suspected of having SARS. Respiratory specimens and blood, serum, and stool samples should be saved for additional testing until a specific diagnosis has been made, and convalescent-phase serum samples should be obtained from patients whose cases meet the SARS case definition and forwarded to state and local health departments for testing at the CDC.
Clinical features. There is a characteristic clinical picture associated with the SARS in several well-described studies, although distinguishing SARS from other causes of pneumonia remains a challenge [39, 41, 42]. After an incubation period of ∼2–10 days (median, 4 days), the most characteristic initial symptom is fever, with or without cough or dyspnea. Chills, myalgia, and progressive respiratory distress often accompany the persisting fever during the first week of illness, and mild gastrointestinal symptoms are present in some patients. The typical fever and the observation that pharyngitis, rhinorrhea, sneezing and conjuctivitis are unusual may help to distinguish patients with SARS from persons with more-common viral upper respiratory tract infections.
On initial presentation, there are typically few physical findings, with a normal chest examination or mild crackles without wheezing, and no rash. The chest radiograph may appear normal or show only mild abnormalities during the first few days, but progression to a bilateral lower lobe interstitial infiltrate is most characteristic. Other radiographic findings are also described, including lobar pneumonia, shifting atelectasis, and multiple focal areas of consolidation, particularly in the periphery of the lungs [43]. Routine laboratory findings include normal-to-low leukocyte counts, with absolute lymphopenia in approximately one-half the patients. Platelet counts are also normal to low. Mild to moderately elevated transaminase, lactate dehydrogenase, and creatinine phosphokinase levels are seen in 30%–70% of cases.
In most probable SARS cases, symptoms resolve spontaneously after the first week. In ⩾20% of patients, symptoms progress over 2–3 weeks to the more-severe respiratory distress syndrome, and the patients require intensive care and ventilatory support. Approximately 10%–15% of cases have died of progressive respiratory failure. Mortality is strongly age-dependent, with mortality of >50% for patients older than 65 years. Patients with underlying chronic heart or lung disease also appear to be at elevated risk for severe disease, although previously healthy younger adults have also died. The most prominent pathological findings in lung tissue samples on autopsy have been diffuse alveolar damage with hyaline membrane formation, interstitial mononuclear infiltration, and desquamation of pneumocytes; in some cases, tissues have shown intra-alveolar hemorrhage, necrotic debris within small airways, organizing pneumonia, or the presence of multinucleated giant cells without viral inclusions [33].
Therapy. A variety of treatments have been attempted, but there are no data from controlled studies, and the available anecdotal evidence is not persuasive that any of the treatment approaches thus far have demonstrated efficacy. Most patients have been treated throughout the illness with supplemental oxygen, intravenous fluids, and other supportive measures; broad-spectrum antibacterial agents have also been given, but these would not be expected to have any effect on the coronavirus infection itself. Early in vitro testing of ribavirin and other antiviral compounds against the novel coronavirus has not produced persuasive evidence of in vitro activity [44, 45]. Corticosteroids and a number of antiviral compounds, including the neuraminidase inhibitors and ribavirin, have been used empirically, but, in the future, use of antiviral compounds for SARS should be done within the context of a controlled clinical trial, because of the importance of identifying efficacious treatments and the lack of evidence of efficacy for any treatment to date.
SARS progressed rapidly from a localized outbreak in southern China to an epidemic with global reach. As of this writing, the epidemic has waned in most of the heavily affected areas, in association with vigorous public health interventions, including community mobilization and quarantine measures on a scale not seen during the past half-century or more. A second wave of infections in Toronto and isolated clusters of new cases elsewhere highlight the dangers of complacency as the acute phase of the epidemic passes. The impact of the epidemic on regional economies, international travel, and medical care is only beginning to be recognized, and the future of SARS is uncertain. It seems possible that SARS will be an important cause of pneumonia in the future, and the screening of outpatients at risk for SARS may become part of the pneumonia evaluation. Infectious diseases physicians will need to ensure that they maintain awareness and that triage procedures adequately provide for the standard, contact, and airborne precautions necessary to protect their fellow workers from infection.
Special Considerations: Treatment of Bacteremic Pneumococcal Pneumonia—New Addition
Recommendation 1. Initial empiric therapy prior to availability of culture data for a patient ill enough to require admission to a hospital ward can be with a β-lactam plus macrolide combination or a respiratory fluoroquinolone alone (A-I). If sufficiently ill to need intensive care unit (ICU) management and if Pseudomonas infection is not a concern, a combination of a β-lactam plus either a macrolide or a respiratory fluoroquinolone should be used (B-III).
Recommendation 2. Once culture data are available and it is known that the patient has pneumococcal pneumonia with bacteremia without evidence to support infection with a copathogen, treatment will depend upon in vitro susceptibility results. If the isolate is penicillin susceptible, a β-lactam (penicillin G or amoxicillin) alone may be used (B-II). If the isolate is penicillin resistant, cefotaxime, ceftriaxone, or a respiratory fluoroquinolone or other agent indicated by in vitro testing may be used (A-III).
Comment. The mortality rate for bacteremic pneumococcal pneumonia is 6%–20%, yet it was always assumed that monotherapy in such cases was sufficient. The guidelines from the IDSA, the ATS, and the Canadian Infectious Diseases Society and Canadian Thoracic Society recommend a macrolide and β-lactam regimen or a fluoroquinolone alone for empiric treatment of patients admitted to a hospital ward and a macrolide or fluoroquinolone plus a β-lactam for patients admitted to the ICU for whom Pseudomonas aeruginosa infection has been excluded. In the former instance, therapy was based on the results of studies that showed that such a regimen was associated with a shorter length of stay and reduced mortality [46, 47]. For ICU patients, this recommendation was based on a lack of efficacy data about fluoroquinolones as monotherapy for severe CAP, as well as concerns about infection with a resistant pathogen.
Three retrospective studies have suggested that dual therapy that included a macrolide given empirically reduced mortality associated with bacteremic pneumococcal pneumonia [48–50]. However, the fact that they were neither prospective nor randomized studies meant that they had significant design limitations. It is important to note that these studies evaluated the effects of initial empiric therapy before the results of blood cultures were known. They did not examine effects of pathogen-specific therapy after the results of blood cultures were available, and the panel believes that the results of these studies do not contradict the principles of pathogen-directed therapy.
Two possible explanations for the improved results with a macrolide are the concurrent presence of atypical pathogens (Mycoplasma pneumoniae, C. pneumoniae, or Legionella species) and the immunomodulating effects of macrolides [51]. A prospective, randomized trial is ultimately needed to determine the best regimen without bias or confounding variables distorting the answer.
A retrospective analysis of Medicare data involving >700 patients aged ⩾65 years with severe pneumococcal pneumonia by the Fine criteria showed that monotherapy with a third-generation cephalosporin was as effective as any other regimen involving a single drug or combination therapy. The end points were in-hospital mortality and 30-day mortality (P. Houck, personal communication).
Empiric therapy for patients with CAP admitted to a hospital ward can be with either a β-lactam plus macrolide regimen or a respiratory fluoroquinolone alone. For those ill enough to require admission to the ICU and in whom Pseudomonas infection is not an issue, initial empiric treatment started before any culture data are available should be with a β-lactam plus either a macrolide or a fluoroquinolone. However, if blood cultures subsequently reveal a pathogen such as S. pneumoniae and there is no evidence of infection with a copathogen, the decision to continue with combination therapy or to switch to a single agent is probably best determined on an individual basis [52]. Variables to consider include the patient's age and any comorbid conditions, as well as the clinical, bacteriological, and radiographic response to therapy.
If a single agent is to be used, the committee believes that bacteremic pneumococcal pneumonia should be treated with penicillin G or ampicillin if the pathogen is penicillin susceptible, and it should be treated with cefotaxime, ceftriaxone, a respiratory fluoroquinolone, or other agent indicated by in vitro testing, if the pathogen is penicillin resistant.
Special Considerations: Update on Legionnaires' Disease
Recommendation 1. Preferred diagnostic tests are the urinary antigen assay and culture of respiratory secretions on selective media (A-II).
Recommendation 2. Testing for Legionella species is appropriate for any patient hospitalized with enigmatic pneumonia (C-II). This test is recommended in patients with enigmatic pneumonia sufficiently severe to require care in the ICU, in the presence of an epidemic, or if there is failure to respond to a β-lactam (A-III).
Recommendation 3. Treatment for legionnaires' disease is appropriate when there is epidemiologic evidence of this disease, despite negative diagnostic test results (B-III).
Recommendation 4. The preferred treatment for legionnaires' disease for hospitalized patients is azithromycin or a fluoroquinolone (moxifloxacin, gatifloxacin, and levofloxacin; gemifloxacin is only available as an oral formulation) (B-II). For patients who do not require hospitalization, acceptable antibiotics include erythromycin, doxycycline, azithromycin, clarithromycin, or a fluoroquinolone (A-II). Treatment should be initiated as rapidly as is feasible (A-II).
Comment. Legionella is implicated in 0.5%–6% of CAP cases in most hospital-based series [53–57]. Risk is related to exposure, increasing age, smoking, and compromised cell-mediated immunity, such as occurs in transplant recipients [58]. Epidemiologic risk factors include recent travel with an overnight stay outside of the home, exposure to spas, recent changes in domestic plumbing, renal or hepatic failure, diabetes, and systemic malignancy [58–60]. Mortality rates are 5%–25% among immunocompetent hosts [57, 59, 61]. Legionella was 1 of 2 major respiratory tract pathogens in patients with CAP who required admission to the ICU, according to 7 of 9 recent reviews [62].
Some authorities feel that the following constellation of clinical features suggests this diagnosis: high fever, hyponatremia, CNS manifestations, lactate dehydrogenase levels of >700 U/mL, or severe disease [57]. However, several studies have demonstrated difficulty in distinguishing individual cases of legionnaires' disease from other causes of CAP on the basis of initial clinical findings, nonspecific laboratory findings, or radiograph findings [63–65]. A clinical scoring system that uses a combination of clinical and nonspecific laboratory findings is neither sufficiently specific nor sensitive to enable accurate diagnosis, although a high score may help direct cost-effective specific laboratory testing [57].
Methods of laboratory detection include culture, serologic tests, direct fluorescent antibody (DFA) staining, urinary antigen assay, and PCR [66]. DFA stains require substantial expertise for interpretation, and selection of reagents is critical. PCR is expensive, and there are no FDA-cleared reagents. The 2 recommended tests are the urinary antigen assay and culture of respiratory secretions. The urinary antigen assay for Legionella pneumophila serogroup 1 is not technically demanding and reliably and rapidly detects up to 80%–95% of community-acquired cases of legionnaires' disease, but it is substantially less sensitive for nosocomial cases because of frequent involvement of serogroups other than serogroup 1 [60]. Culture on selective media detects all but very rare strains but is technically demanding and requires 3–7 days [58, 67]. Testing for Legionella species is appropriate for any patient hospitalized with enigmatic pneumonia; testing is recommended for patients with enigmatic pneumonia sufficiently severe to require hospitalization in an ICU, pneumonia in a compromised host, in the presence of an epidemic, and failure to respond to treatment with a β-lactam. It should also be emphasized that no laboratory test for legionnaires' disease detects all patients with the disease. In the appropriate clinical and epidemiologic settings, therapy for legionnaires' disease should be given or continued even if the results of Legionella-specific tests are negative [58, 67].
The preferred therapy for legionnaires' disease depends upon the severity of illness, the underlying health of the patient, and patient drug tolerance. Otherwise healthy patients with mild pneumonia not requiring hospitalization may be treated with a wide variety of antimicrobial agents, including erythromycin, tetracycline, doxycycline, azithromycin, clarithromycin, levofloxacin, gatifloxacin, moxifloxacin, and gemifloxacin [57, 68, 69]. Azithromycin or a fluoroquinolone (moxifloxacin, gatifloxacin, or levofloxacin) are recommended for severe disease (gemifloxacin is only available in an oral formulation). A delay in therapy is associated with an increased mortality rate, and treatment should be started as soon as possible [70]. The duration of treatment should be 10–21 days, but it should be less for azithromycin because of its long half-life [57, 68].
Special Considerations: Viral Causes Of Cap—New Addition
Recommendation 1. Respiratory syncytial virus (RSV) antigen detection tests are readily available but are insensitive for detecting infections in adults and are not generally recommended for adults (C-III).
Recommendation 2. A rapid antigen detection assay for influenza virus is recommended for rapid detection of this pathogen for epidemiologic purposes and/or treatment (C-II). Tests that distinguish between influenza A and B are generally preferred (C-III).
Recommendation 3. Early treatment (within 48 h after onset of symptoms) is effective in the treatment of influenza A using amantadine, rimantadine, oseltamivir, or zanamivir and is effective in the treatment influenza B using oseltamivir and zanamivir (B-I). Use of these drugs is not recommended for uncomplicated influenza with a duration of symptoms of >48 h (D-I), but these drugs may be used to reduce viral shedding in hospitalized patients or for influenza pneumonia (C-III).
Recommendation 4. Empiric treatment of suspected bacterial superinfection of influenza should provide activity against S. pneumoniae, Staphylococcus aureus, and Haemophilus influenzae with antibiotics such as amoxicillin-clavulanate, cefpodoxime, cefprozil, cefuroxime, or a respiratory fluoroquinolone (B-III).
Recommendation 5. Pneumonia caused by varicella zoster virus (VZV) or herpes simplex virus (HSV) should be treated with parenteral acyclovir (A-II).
Recommendation 6. There is no antiviral agent with established efficacy for the treatment of adults with pulmonary infections involving parainfluenza virus, RSV, adenovirus, metapneumovirus, the SARS agent, or Hantavirus (D-I).
Comment. Respiratory tract viruses are common causes of often serious cases of pneumonia, particularly in elderly patients, patients with chronic obstructive lung disease, and patients with comorbidities. One prospective study of 1029 chronically ill adults found respiratory viral infections in 35%–48% (depending on age) of those hospitalized for an acute respiratory condition (i.e., pneumonia, tracheobronchitis, exacerbations of asthma, or chronic obstructive lung disease) and that influenza, RSV, or parainfluenza virus accounted for 75% of these viral infections [71]. A review of influenza and RSV for the 1976–1977 through 1998–1999 seasons suggested that influenza was responsible for an average of 36,155 respiratory- and circulatory-associated deaths per year in the United States. Particularly vulnerable were persons with cardiopulmonary disease and persons aged >65 years, especially the “elderly elderly,” defined as persons >85 years of age [72]. RSV was implicated in an average of 11,321 cardiopulmonary deaths per year, with most deaths occurring among elderly persons and persons with chronic cardiac or pulmonary diseases. Other viral causes of respiratory tract infections are parainfluenza virus and, less commonly, adenovirus, metapneumovirus, HSV, VZV, and measles. (SARS is discussed in a separate section in this guideline.) Metapneumovirus is a recently described paramyxovirus, which appears to be a potentially important viral respiratory tract pathogen, causing pneumonia in both children and adults [73, 74].
The clinical presentations of viral pneumonias and the spectrum of associated agents are highly dependent on patient age, comorbidities, and immune status. Approximately 10% of immunocompetent adults hospitalized with CAP have evidence of viral infection, but this varies from 4%–39% in different studies [75]. A recent report from the United Kingdom showed serologic evidence of a viral infection in 23% of 267 patients hospitalized with CAP, with influenza and RSV in 20% and 4%, respectively, of the total [76].
Influenza and other viruses can cause primary viral pneumonias; secondary bacterial infections are common in hospitalized adults, and the reported frequency has ranged widely, from 26% to 77% in different studies [75]. The most common cause of bacterial superinfection is S. pneumoniae, but S. aureus has been found in up to one-quarter of patients in earlier studies. In the absence of a characteristic exanthem, no clinical or radiographic criteria are able to reliably distinguish persons with viral infection from persons with bacterial infection. Cultures for respiratory viruses (except for shell vial methods, which can yield a diagnosis the next day) and serologic studies are usually too slow to be useful in individual patient treatment. Rapid antigen detection aimed at influenza can provide a diagnosis in 15–30 min, but test performance varies with the specific test used, sample type, duration of illness, and patient age. Sensitivity is ∼50%–70% in adults [77, 78], so that negative test results do not exclude the diagnosis; these tests have not generally proven to be superior to physician diagnosis based on the presence of fever and typical symptoms in the presence of an epidemic [79], but some rapid tests can distinguish between influenza A and B strains, which may have therapeutic implications. Antigen tests for RSV detection are insensitive (<15%) with use of upper respiratory samples from adults. One nucleic acid detection assay for multiple respiratory viruses is commercially available, and, in general, such assays offer the possibility of a rapid, highly sensitive means of a specific viral diagnosis.
No prospective controlled studies of antiviral treatment of viral pneumonias involving adults have been reported, but antiviral therapy is warranted for infection with influenza virus, VZV, HSV, and other viruses in selected circumstances. The M2 inhibitors amantadine and rimantadine are active only for influenza A virus, whereas the neuraminidase inhibitors are inhibitory for both influenza A and B viruses [80]. Amantadine appears to be as effective as the other agents for influenza A infections and is less expensive, but it is associated with higher rates of toxicity. All are effective for chemoprophylaxis and early treatment (<2 days) of uncomplicated influenza A, but their relative efficacies have not been directly compared, except in 1 study, which found that inhaled zanamivir (which has not been approved by the FDA for prophylaxis) was more effective than oral rimantadine in protection of nursing home residents during influenza A outbreaks, in part because of emergence of resistance to the M2 inhibitor [81]. Early treatment of influenza in ambulatory adults with inhaled zanamivir or oral oseltamivir may reduce the likelihood of lower respiratory tract complications [82–84]. The use of influenza antiviral medications may reduce the likelihood of respiratory tract complications, as reflected by reduced use rates of antibacterial agents in ambulatory patients with influenza. In hospitalized adults with influenza, a minority of whom had radiographically documented pneumonia, no obvious benefit was found in one retrospective study of amantadine treatment [85]. Because such patients often have recoverable virus (median duration, 4 days after hospitalization) after hospitalization, antiviral treatment seems reasonable. Because of its broad influenza spectrum, low risk of resistance emergence, and lack of bronchospasm risk, oseltamivir is an appropriate choice for hospitalized patients. For severely ill persons with influenza viral pneumonia, combined antiviral therapy with an M2 inhibitor and neuraminidase inhibitor deserves consideration, but this approach has not yet been shown to improve clinical outcomes in such a scenario [86].
Parenteral acyclovir is indicated for treatment of VZV [87] or HSV pneumonia. No antiviral treatment of proven value is available for other viral pneumonias in immunocompetent adults. Intravenous ribavirin has been used in adenovirus infection, but its efficacy has not been established; this drug appears ineffective for Hantavirus infection [88, 89]. Pleconaril is available for compassionate use for management of picornavirus pneumonias in immunocompromised patients.
Special Considerations: Update On Pneumonia in The Context Of Bioterrorism
Recommendation 1. Physicians should know the clues to bioterrorism and the appropriate mechanisms to alert public health officials in cases of suspected bioterrorism (A-III).
Recommendation 2. Recommended diagnostic tests and management guidelines are those of the Johns Hopkins Center for Biodefense Strategies and of the CDC, as modified for the specific outbreak (A-I). Means of diagnosis for category A agents of bioterrorism: for inhalation anthrax, blood culture (A-I) and chest CT scan (A-I); for pneumonic plague, blood culture and Gram stain and culture of sputum samples (A-I); and for tularemic pneumonia, culture of blood and sputum or pharynx in biocontainment level 3 (BL-3) laboratory (A-I).
Comment. A number of microbes can be disseminated by aerosol as biological weapons that can potentially afflict thousands of people. The etiologic agents most likely to cause severe pulmonary infection are Bacillus anthracis, Franciscella tularensis, and Yersinia pestis [90, 91]. Inhalation anthrax always indicates bioterrorism; pneumonic tularemia and pneumonic plague might or might not be associated with bioterrorism.
The greatest experience with inhalation anthrax was the 11 cases that followed established or suspected exposure to contaminated mail in 2001 in the United States [92, 93]. Clinical clues to facilitate the distinction of inhalation anthrax from CAP have been reported [94]. Features of this disease included a median incubation period of 4 days (range, 4–6 days), nonspecific initial symptoms (fever, gastrointestinal complaints, and cough without coryza) and some highly characteristic epidemiologic clues and laboratory findings (a wide mediastinum on chest radiograph, hyperdense mediastinal nodes on chest CT scan, and bloody pleural effusions). Blood cultures were positive for 8 of 8 untreated patients, usually within 18 h [92, 93]. The mortality rates in this and prior inhalation anthrax cases in the antibiotic era were 45%–80% [95]. The most important therapeutic interventions are rapid institution of antibiotic treatment and adequate drainage of pleural effusions. Antibiotic selection should be based on the epidemic strain, which may have an unusual resistance pattern due to genetic modification. Treatment and prophylaxis should be prolonged, because animal studies have shown in vivo persistence of spores [95]. Prophylaxis in the 2001 epidemic consisted of 60–100-day courses of oral doxycycline or ciprofloxacin for 10,000 persons with suspected exposure; none subsequently developed anthrax [93]. It should be emphasized that the last case of naturally occurring inhalation anthrax in the United States occurred in 1976, so any case of established or suspected inhalation anthrax should prompt notification of public health authorities [95].
F. tularensis causes <200 infections per year in the United States but caused hundreds of thousands of infections in Europe in World War II [96]. Its potential as a biological weapon was substantiated by extensive studies performed by the US biological weapons program in the 1960s [97]. The most common form of bioterrorism with F. tularensis after aerosol exposure is “typhoidal” or “pneumonic” tularemia. Clinical features include an incubation period of 3–5 days, nonspecific symptoms (fever, malaise, pleurisy, and nonproductive cough), and a chest radiograph showing pneumonia, often with mediastinal adenopathy. If tularemia is suspected, the organism may be cultured from blood samples, sputum samples, or pharyngeal exudates, but only with difficulty, using media containing cysteine or other sulfhydryl compounds, such as thioglycolate broth or charcoal-yeast agar. This organism represents a hazard to laboratory personnel, and culture should be attempted only in a BL-3 laboratory [98]. There are multiple diagnostic methods, including antigen detection, PCR, EIA, immunoblot assay, and pulsed-field gel electrophoresis; these are generally available only in research or public health laboratories. Standard treatment is streptomycin, which is preferred, although gentamicin is more generally available, can be given intravenously, and is an acceptable alternative [99–101]. Tetracycline and chloramphenicol are also alternatives, but treatment failures and relapses are more common with these agents [99]. Ciprofloxacin is not FDA-approved for tularemia, but has been used successfully in animals and people [99, 102]. The usual duration of treatment is 14 days. Compared with inhalation plague or anthrax, tularemia progresses more slowly and has a lower mortality rate. The experience with 1409 cases reported during 1985–1992 showed that mortality was 1.4% [99]. There is minimal risk of person-to-person spread, and the recommendation for prophylaxis for exposed persons is ciprofloxacin or doxycycline for 2 weeks.
Y. pestis is also a potential biological weapon of great concern because it has a fulminant course, causes death in the absence of antibiotic treatment, and can be spread from person to person [103]. Clinical features of infection include high fever, chills, headache, cough, bloody sputum, prominent gastrointestinal symptoms, leukocytosis, and radiographic changes that show bilateral pneumonia. There is rapid progression to septic shock and death. The acutely swollen, tender lymph node or bubo that is characteristic of bubonic plague is unlikely to be present with aerosol dissemination. A review of 390 cases of plague in the United States for 1947–1996 showed that only 6 cases (2%) were of the pneumonic form [104]. The diagnosis is established with culture of sputum or blood samples; sputum Gram stain shows typical safety-pin—shaped, bipolar-staining, gram-negative coccobacilli. Growth occurs within 24–48 h, but identification often takes up to 6 days; if this diagnosis is suspected, the specimen should be split in half for incubation at 28°C for one half (for rapid growth) and at 37°C for the other (for identification of the capsular antigen). Health care workers are at risk of aerosol exposure, so respiratory precautions should be taken until patients have undergone therapy for 48 h. The standard treatment for plague pneumonia is administration of streptomycin or gentamicin in standard doses for 10 days [105]. Doxycycline may be given for treatment or prophylaxis, although resistance has been described elsewhere [106]. Ciprofloxacin appears to be as effective as aminoglycosides in mice with experimental pneumonic plague [107] and may be given for treatment or prophylaxis. Administration of tetracyclines or fluoroquinolones for 7 days is the preferred prophylaxis when face-to-face contact has occurred or exposure is suspected.
Pneumonia in Elderly Persons—New Addition
Recommendation. Antimicrobial selection for elderly patients with CAP is the same as for all adults with CAP (B-III).
Note. Recommendations for pneumococcal and influenza vaccines in the elderly population are included as part of the recommendations given in the following section, Update on Prevention of CAP.
Comment. In the United States, CAP is the fifth-leading cause of death in people aged ⩾65 years, and an estimated 60,000 seniors die annually [108]. Residents of long-term care facilities, a distinct subpopulation of elderly people, are at particularly high risk of developing pneumonia [109].
Etiology. Determining the relative importance of the various etiologic agents of pneumonia in older adults is challenging. In a Finnish study involving 345 CAP episodes, S. pneumoniae was the etiologic agent in 48% of patients aged ⩾60 years, C. pneumoniae was detected in 12%, M. pneumoniae in 10%, H. influenzae in 4%, and respiratory viruses in 10% [110]. The incidence of gram-negative bacterial pneumonia in elderly persons living in the community is uncertain, but it is greater in those with comorbidities [111]. For nursing home residents, data are even more scant. The proportion of cases of pneumonia in long-term care facilities that are attributable to pneumococcus is 0%–39% [109]. The proportions of cases due to gram-negative bacteria and S. aureus were 0%-55% and 0%–33%, respectively. Legionella and Mycoplasma species were infrequently detected.
Risk factors. In another Finnish study, independent risk factors for pneumonia included alcoholism (relative risk [RR], 9.0; 95% CI, 5.1–16.2), bronchial asthma (RR, 4.2; 95% CI, 3.3–5.4), immunosuppression (RR, 3.1; 95% CI, 1.9–5.1), lung disease (RR, 3.0; 95% CI, 2.3–3.9), heart disease (RR, 1.9; 95% CI, 1.7–2.3), institutionalization (RR, 1.8; 95% CI, 1.4–2.4), and increasing age (for age of ⩾70 vs. 60–69 years: RR, 1.5; 95% CI, 1.3–1.7) [112]. In a cohort study to assess risk factors for pneumonia in residents of long-term care facilities, older age (OR, 1.7; 95% CI, 1.1–2.6 per 10-year interval; P = .01), male sex (OR, 1.9; 95% CI, 1.1–3.5; P = .03), difficulty swallowing (OR, 2.0; 95% CI, 1.2–3.3; P = .01), and the inability to take oral medications (OR, 8.3; 95% CI, 1.4–50.3; P = .02) were found to be significant [113].
Clinical presentation. The clinical presentation of CAP has frequently been described as being more subtle in elderly individuals; however, there have been relatively few systematic evaluations to confirm this. A study of 1812 patients found that persons aged 65–74 years and ⩾75 years had 2.9 and 3.3 fewer symptoms, respectively, than did those aged 18–44 years [114]. The reduced prevalence of symptoms was most pronounced for those related to febrile response (chills and sweats) and pain (chest, headache, and myalgia). When 71 long-term care facility residents admitted to hospital with pneumonia were compared with 93 seniors admitted with CAP, it was noted that nursing home residents were less likely to experience chills, pleuritic chest pain, headache, anorexia, myalgia, and productive cough [115].
Management. Guidelines for the antibacterial management of CAP in the elderly population have not been assessed in randomized, controlled trials. Antimicrobial selection recommendations for elderly patients with CAP are the same as for all adults with CAP (table 1).
A discussion of immunoprophylaxis of elderly persons against influenza and pneumococcus infection is included in the following section, Update on Prevention of CAP.
Update on Prevention of Cap
Recommendation 1. All persons >50 years, others at risk for influenza complications, and household contacts of high-risk persons should receive inactivated influenza vaccine, as recommended by the Advisory Committee on Immunization Practices (ACIP) (A-I). The injected inactivated vaccine is the preferred formulation for most persons at risk of complications associated with influenza, for household contacts of high-risk persons, and for health care workers (A-1). The intranasally administered live, attenuated vaccine (FluMist; Aventis) is an alternative vaccine formulation for some persons aged 5–49 years without chronic underlying diseases, including immunodeficiency, asthma, and chronic medical conditions (C-I). Influenza vaccine should be offered to persons at hospital discharge or during outpatient treatment during the fall and winter (C-III). Health care workers in inpatient and outpatient settings and long-term care facilities should receive annual influenza immunization (A-I).
Recommendation 2. Pneumococcal polysaccharide vaccine (Pneumovax; MedImmune [marketed by Wyeth in the United States]) is recommended for use, according to current ACIP guidelines, including use for persons aged >65 years and for those with selected high-risk concurrent diseases (B-II). Vaccination may be done either at hospital discharge or during outpatient treatment (C-III).
Comment. Vaccination against influenza and pneumococcus infection is the mainstay of prevention against pneumonia for older adults. A systematic review that included 1 randomized trial and 20 cohort studies showed that, for frail older adults, influenza vaccine had an efficacy (1-OR) of 53% for preventing pneumonia, 50% for preventing hospitalization, and 68% for preventing death [116]. A recent large observational study of adults >65 years found that vaccination against influenza was associated with a reduction in the risk of hospitalization for cardiac disease (19% reduction), cerebrovascular disease (16%–23% reduction), and pneumonia or influenza (29–32% reduction), as well as a reduction in the risk of death due to all causes (48%–50% reduction) [117]. In long-term care facilities, vaccination of health care workers with influenza vaccine is an important preventive health measure. Data from 2 cluster randomized trials have shown benefit [118, 119]. Potter et al. [118] randomized 12 long-term facilities to either the offer of vaccination of health care workers or to no offer of vaccination. Vaccination of health care workers was associated with a reduction in total patient mortality rate, from 17% to 10%. Carman et al. [119] conducted a randomized trial involving 20 geriatric care hospitals in which they compared influenza vaccination of health care workers with no vaccination. Vaccination of health care workers significantly reduced mortality among elderly people who had a stay of >6 months in hospitals where health care workers were vaccinated, compared with hospitals where they were not (OR, 0.58; 95% CI, 0.04–0.84; P = .014). Influenza vaccine effectiveness varies among influenza seasons, with effectiveness being higher when the vaccine antigens are more closely matched to the circulating strains.
Pneumococcal polysaccharide vaccine has not been consistently effective in randomized, double-blind, controlled trials involving elderly individuals. Results of one randomized clinical trial suggested that the polysaccharide vaccine provided some protection against pneumococcal pneumonia among high-risk elderly persons [120]; 2 other trials did not demonstrate efficacy against pneumonia or bronchitis without bacteremia [121, 122], although the use of nonspecific diagnostic methods may have limited the studies' ability to find an effect [123]. Two open-label trials have suggested protection against pneumococcal pneumonia among elderly residents of long-term care facilities [124, 125].
Postlicensure epidemiological studies, including a recent large observational study, involving elderly persons and younger adults with certain chronic medical conditions have documented effectiveness of pneumococcal polysaccharide vaccines for prevention of invasive infection (bacteremia and meningitis) but not for prevention of pneumonia without bacteremia [126–130]. The overall effectiveness against invasive pneumococcal disease among immunocompetent persons aged ⩾65 years is 75% [126], although efficacy may decrease with advancing age [128].
Older adults may be benefiting from vaccination of children against pneumococcal disease because of decreased pneumococcal transmission. In 2000, a protein-polysaccharide conjugate vaccine targeting 7 pneumococcal serotypes (Prevnar; Wyeth Lederle Vaccines) was licensed for use in young children in the United States. According to data from the CDC's Active Bacterial Core Surveillance (ABCS), rates of invasive pneumococcal disease (e.g., primary bacteremia, pneumonia with bacteremia, and meningitis) among children aged <2 years—the vaccine's target population—were 69% lower in 2001, compared with baseline [131]. Invasive disease rates decreased by 18% among persons ⩾65 years of age (49.5 cases per 100,000 persons vs. 60.1 cases per 100,000 persons) and 32% among adults aged 20–39 years (7.6 cases per 100,000 persons vs. 11.2 cases per 100,000 persons). To date, the pneumococcal conjugate vaccine is only licensed for children; the vaccine's safety and performance have not been adequately studied in adults.
If indicated, patients with CAP should receive pneumococcal and influenza vaccines as recommended by the CDC's ACIP. The optimal time for influenza vaccination is October and November, although vaccination in December and later is recommended for those who were not vaccinated earlier. Influenza and pneumococcal vaccines can be given at the same time in different arms. The vaccines should be provided either at hospital discharge or at the conclusion of outpatient treatment; standing orders can be used to simplify the process of ensuring that patients are vaccinated [132].
Recent ACIP influenza recommendations state that inactivated influenza vaccine should be given (by intramuscular administration) to all people >6 months of age who are at increased risk for complications from influenza [133]. Target groups for vaccination include persons aged ⩾50 years; persons of any age who reside in a nursing home or other long-term care facility, who have a chronic disorder of the pulmonary or cardiovascular systems, including asthma, or who have a chronic illness that required regular outpatient follow-up or hospitalization in the prior year, such as chronic metabolic diseases (including diabetes mellitus), renal dysfunction, hemoglobinopathies, or immunosuppression (including immunosuppression caused by medications or by HIV); and women who will be in the second or third trimester of pregnancy during the influenza season. All health care workers or others whose work involves any patient contact, including contact with nursing home residents, should receive influenza vaccine annually to prevent possible transmission to patients. In addition, vaccination of all children 6–23 months and their caregivers is encouraged.
An intranasally administered, live, attenuated, influenza virus vaccine was approved in 2003 by the FDA. ACIP guidelines on use were published in September 2003 [134]. The live, attenuated vaccine is approved for use and is currently recommended as an option for vaccination of healthy persons aged 5–49 years. Advantages of the new vaccine include the potential to induce both mucosal and systemic immune responses and the acceptability of administration using the intranasal rather than intramuscular route. Because it is made from live, attenuated virus, however, care should be taken to avoid administering it to certain persons. Inactivated influenza vaccine (the injected formulation) rather than the intranasally administered, live, attenuated virus vaccine should be given to persons aged <5 years or ⩾50 years; persons with asthma; persons with reactive airways disease or other chronic disorders of the pulmonary or cardiovascular systems; persons with other underlying medical conditions, including metabolic diseases, such as diabetes, renal dysfunction, and hemoglobinopathies; or persons who have immunodeficiency diseases or who are receiving immunosuppressive therapies; children or adolescents receiving aspirin or other salicylates (because of the association of Reye syndrome with wild-type influenza infection); and pregnant women. Because data are lacking on transmission of live vaccine virus from vaccinated persons to immunocompromised persons, use of inactivated vaccine is preferred for vaccinating household members, health care workers, and others who have close contact with immunosuppressed people.
Chemoprophylaxis can be used as an adjunct to vaccination for prevention and control of influenza. Both amantadine and rimantadine have FDA indications for treatment and chemoprophylaxis of influenza A infection, and oseltamivir is indicated for prevention and treatment of both influenza A and B [133]. (Zanamivir is FDA-approved for the treatment of both influenza A and B but is not approved for prophylaxis). Developing an adequate immune response to the inactivated influenza vaccine takes ∼2 weeks in adults; chemoprophylaxis may be useful during this period for persons with household exposure to influenza, persons who reside or work in institutions with an influenza outbreak, and other persons at high risk for influenza-associated complications in the setting of a community outbreak. Chemoprophylaxis also may be considered for persons with contraindications to influenza vaccine or may be given in addition to vaccination to persons in whom the vaccine may not be effective. The use of influenza antiviral medications for treatment or chemoprophylaxis should not affect response to the inactivated vaccine. Because it is unknown whether administration of influenza antiviral medications affects the performance of the new live, attenuated, intranasally administered vaccine, the live, attenuated vaccine should not be administered until 48 h after the end of any influenza antiviral therapy, and influenza antiviral medications should not be administered for 2 weeks after the receipt of the live, attenuated vaccine.
Pneumococcal polysaccharide vaccine is recommended by the ACIP for all adults >65 years of age and for younger adults with certain chronic diseases (such as diabetes, cardiovascular disease, lung disease, alcohol abuse, liver disease, CSF leaks, or renal failure) or immune system disorders (such as sickle cell disease, nephrotic syndrome, HIV infection, hematologic malignancies, or long-term use of immunosuppressive medications) [135]. A second dose is recommended after 5 years for persons with immune system disorders and for persons aged >65 years whose first dose was received before the age of 65 years. The efficacy of revaccination is unknown. A recent model suggested that it may be cost-effective to vaccinate all adults aged ⩾50 years, especially African American persons and those with comorbid conditions [136]. The ACIP is considering changes to the vaccine recommendations that would include vaccinating all adults aged ⩾50 years and listing smokers among those with chronic illnesses who should be vaccinated at an earlier age.
Update on Macrolides
Recommendation 1. A macrolide is recommended as monotherapy for selected outpatients, such as those who were previously healthy and not recently treated with antibiotics (A-I).
Recommendation 2. A macrolide plus a β-lactam is recommended for initial empiric treatment of outpatients in whom resistance is an issue and for hospitalized patients (A-I).
Comment. The macrolides constitute one of the most popular and long-standing classes of antibiotics in clinical use. The class includes 3 drugs in North America: erythromycin, azithromycin, and clarithromycin and has played a significant role in the management of CAP because of its activity against S. pneumoniae and the atypical pathogens. Although erythromycin is the least expensive of these 3 drugs, it is not used as often because of gastrointestinal intolerance and lack of activity against H. influenzae.
In the United States, pneumococci were uniformly susceptible to macrolides until the late 1980s [137]. As the result of a steady increase in the rate of resistance, at present, in the United States, ∼25% of all pneumococci show some level of resistance to macrolides [138–140], ranging from 17% in the Northeast to 35% in the Southeast [138]. There are 2 principal mechanisms of resistance: (1) an alteration of the macrolide binding site by methylation in the 23S rRNA, encoded by erm(B), and (2) an efflux pump, encoded by mef(A), by which bacteria expel macrolides [141, 142]. The methylase causes high level resistance (MIC of erythromycin, 128 mg/mL), whereas the efflux pump produces lower-level resistance (MIC of erythromycin, 1–64 mg/mL) that some experts believe can be overcome by increasing antibiotic concentrations. Rarer mechanisms of (high-level) resistance include alterations of ribosomal proteins L4 or L22 that are adjacent to domain V [143, 144].
In the United States, one-third of macrolide-resistant strains carry erm(B), and the other two-thirds carry mef(A) [138, 140]. The level of resistance among mef(A) strains has steadily increased in the past few years [139, 145]. In other words, even those organisms that historically had lower-level resistance have become increasingly resistant to achievable levels of macrolides [139, 146]. In Europe, a higher proportion of pneumococci are macrolide resistant, and erm(B) is responsible in the majority of isolates [147]. Rates of resistance are lower in Canada than in the United States, and they are higher in the Far East than in Europe [148].
Despite the reports of increasing resistance in vitro, the number of clinical failures has not kept pace. Reports of clinical failures in pneumococcal pneumonia by Dixon [149], Fogarty et al. [150], Kelley et al. [151], and Lonks et al. [152] have failed to provide convincing numbers to match the laboratory phenomena. Why is this?
There are a number of possible answers. First of all, mortality may be a relatively insensitive measure of the impact of resistance. Also, to detect treatment failures, one would have to use monotherapy with a drug to which the etiologic agent is known to be resistant.
In support of the IDSA approach is the relatively small number of reported failures and the fact that, when patients such as those described by Kelley et al. [151] and Lonks et al. [152] were hospitalized and treated with a β-lactam and a macrolide, they all survived.
What then is the role for macrolides in 2003? For outpatients, we believe that, for those who have previously been healthy and who have not been treated with antibiotics for any reason within the preceding 3 months, a macrolide alone is adequate (table 1). An advanced macrolide, such as azithromycin or clarithromycin, may be used alone for patients with comorbidities, such as chronic obstructive pulmonary disease, diabetes, renal or congestive heart failure, or malignancy, who have not been previously treated with antibiotics. For selected outpatients and inpatients, it is clear that, given together with a β-lactam, the macrolides still play an important role. If the infection is caused by macrolide-resistant S. pneumoniae, it is highly likely that the β-lactam will still be effective, and, if caused by one of the atypical pathogens, the macrolide will certainly have a role to play.
The Ketolides—New Addition
Recommendation. Telithromycin may have a role as an alternative to macrolides for treatment of patients with CAP. At this time, however, it is not yet FDA approved.
Comment. The ketolides, which are semisynthetic derivatives of 14-membered macrolides, were developed specifically to be effective against macrolide-resistant, gram-positive cocci. Structural modifications at the positions of 3, 6, and 11–12 have altered and improved the pharmacokinetic and antimicrobial activity of the parent compounds, and pharmaceutical manufacturers are seeking approval for their use in CAP, acute exacerbation of chronic bronchitis, and acute sinusitis.
The antibacterial activity of macrolides and ketolides is dependent on inhibition of bacterial protein synthesis. The main differences between them, however, are that, although macrolides bind to only 1 contact site within the 23S ribosomal subunit (domain V), ketolides bind more avidly to domain V and, in addition, bind to a second site on the 23S subunit (domain II). Telithromycin also has some affinity for the efflux pump [153–155]. These differences explain why ketolides remain active against pathogens with both erm- and mef-mediated resistance.
In vitro, telithromycin is active against S. pneumoniae, including macrolide-resistant strains, as well as H. influenzae and Moraxella catarrhalis [156, 157]. The drug also inhibits Legionella, Mycoplasma, and Chlamydophilia species [158, 159].
The drug is given once daily at a dose of 800 mg and appears to be well tolerated while achieving ratios of tissue to plasma of ⩾500 and 16.8 in alveolar macrophages and epithelial lining fluid, respectively [160, 161].
Data from 3 randomized, controlled, double-blind CAP trials comparing telithromycin with amoxicillin, clarithromycin, and trovafloxacin suggest that the ketolide is as effective as the comparators [162–164]. Data available to date suggest that the ketolides may have an important role to play in the treatment of CAP caused by macrolide-resistant S. pneumoniae, but more studies involving sicker patients are required before its full value can be appreciated. The drug has not yet been approved by the FDA.
S. Pneumoniae With Reduced Susceptibility to Fluoroquinolones in North America—New Addition
Recommendation 1. Fluoroquinolones (gatifloxacin, gemifloxacin, levofloxacin, and moxifloxacin) are recommended for initial empiric therapy of selected outpatients with CAP (A-I). Other options (macrolides and doxycycline) are generally preferred for uncomplicated infections in outpatients (A-I).
Recommendation 2. Fluoroquinolones (gatifloxacin, gemifloxacin, levofloxacin, and moxifloxacin) may be used as monotherapy for patients with CAP who are admitted to a hospital ward (A-I). With the exception of gemifloxacin (no intravenous formulation), they may be used as part of a combination for patients with CAP admitted to an ICU (C-III).
Comment. Since publication of the 2000 guidelines, fluoroquinolone agents have been more widely used to treat pneumonia, yet, at the same time, several compounds have been withdrawn because of serious safety concerns, and resistance to this class of drugs has been increasing. Emergence of S. pneumoniae with reduced susceptibility to the fluoroquinolones has been described in Canada, Spain, Hong Kong, eastern and central Europe, and, to a lesser extent, the United States [147, 165–171]. In some countries, resistance has been due to multiple serotypes, whereas, in others, it has resulted predominantly from a single serotype, such as the 23F clone in Hong Kong [165, 167]. Fluoroquinolone resistance in S. pneumoniae is primarily due to mutations in the genes encoding the target topoisomerase enzymes, namely parC, which encodes the A subunit of DNA topoisomerase IV, and/or gyrA, which encodes the A subunit of DNA gyrase. Resistance occurs in a stepwise fashion, with first-step mutations in one target gene (either parC or gyrA) resulting in low-level resistance and second-step mutations in the other target genes (either parC or gyrA) leading to higher levels of resistance. In Canada, Chen et al. [165] found that the prevalence of ciprofloxacin-resistant pneumococci (MIC, ⩾4 ∼g/mL) increased from 0% in 1993 to 1.7% in 1997–1998 (P = .01). In adults, the prevalence increased from 0% in 1993 to 3.7% in 1998. In addition to the increase in the prevalence of pneumococci with reduced susceptibility to fluoroquinolones, the degree of resistance also increased. From 1994 to 1998, there was a statistically significant increase in the proportion of isolates with an MIC of ciprofloxacin of ⩾32 ∼g/mL (P = .04). In 2002, the Canadian Bacterial Surveillance Network reported that the prevalence of levofloxacin-resistant pneumococci (MIC, 8 ∼g/mL) was 4% in sputum isolates recovered from patients >65 years of age [172].
Rates of resistance in the United States are <2% [173–176]. Doern et al. [177] reported ciprofloxacin resistance (MIC, ⩾4 ∼g/mL) rates of 1.4%, and the CDC's ABCS program performed during 1995–1999 reported levofloxacin nonsusceptibility rates of 0.2% [174]. The PROTEKT study (2000–2001), a surveillance study that examined the susceptibility of respiratory pathogens to a variety of antimicrobials, including levofloxacin, reported in vitro susceptibility test results for 10,103 respiratory tract isolates of S. pneumoniae recovered from patients in 154 cities and 44 states [178]. Overall, the study found that 0.8% of isolates were resistant to levofloxacin (MIC, 8 ∼g/mL); however, the resistance rates varied in some states (from 0% to 4.8%) and cities (from 0% to 22%). This wide divergence in the prevalence of resistance is similar to what occurred with β-lactam resistance to pneumococci in the 1980s, when overall resistance rates were <2% [179]. Extrapolating from what is known about the emergence and dissemination of resistance to β-lactams among pneumococci, the fluoroquinolones could possibly suffer the same fate, unless these agents are used appropriately. Local monitoring of susceptibility patterns of important pathogens to fluoroquinolones and, in fact, to all classes of antibiotics is important.
Clinical failures secondary to pneumococcal resistance to levofloxacin were recently reported [180]. It was shown that such resistance can develop de novo secondary to a point mutation while the patient is receiving therapy. Fluoroquinolone resistance appears to be more common among patients with pneumococcal pneumonia admitted from long-term care facilities [181].
The committee is concerned about misuse and overuse of fluoroquinolones and feels that, if abuse of this class of drugs continues unabated, we may see the demise of fluoroquinolones as useful antibiotics within the next 5–10 years.
Update on Performance Indicators
Recommendation 1. Antibiotic therapy should be initiated within 4 h after registration for hospitalized patients with CAP (B-III).
Recommendation 2. Smoking cessation should be a goal for persons hospitalized with CAP who smoke (B-II).
Comment. Timely antimicrobial therapy is important for patients who require hospitalization for acute pneumonia. The previous IDSA guidelines recommended initial administration within 8 h after arrival at the hospital. This recommendation was based on a retrospective analysis of 14,000 Medicare hospitalizations for pneumonia in 1994–1995 [182].
A more recent analysis of Medicare hospitalizations demonstrated an association between initiation of antimicrobial therapy within 4 h after arrival and improved outcomes [183]. In fact, the time to initiation of antibiotic therapy had a greater influence on patient outcome than did antibiotic selection itself. This study included >13,000 patients with pneumonia who were hospitalized in 1998 and 1999 and who had not received antibiotics before admission. Initial therapy within 4 h after arrival at the hospital was associated with reduced mortality in the hospital (severity-adjusted OR, 0.85; 95% CI, 0.76–0.95). Mean length of stay was 0.4 days shorter among patients who received antimicrobials within 4 h than among those whose initial therapy was given later. Improved outcomes were associated with timely therapy independent of PSI class and the presence of congestive heart failure. These findings are consistent with those of several previous studies [182, 184–187]. The committee supports the early initiation of antibiotic therapy in patients requiring hospitalization for CAP.
Smoking has a well-established association with morbidity and mortality, especially in the form of chronic lung disease and cancer. It is also associated with a substantial risk of pneumococcal bacteremia; one report showed that smoking was the strongest of multiple risks for invasive pneumococcal disease in immunocompetent, nonelderly adults [188]. Smoking is also identified as a risk for Legionella infection [58]. Smoking cessation should be attempted when smokers are hospitalized; this is particularly important and relevant when these patients are hospitalized for pneumonia.
Conflict of Interest Disclosure
Lionel A. Mandell has received research funding from Bayer, Bristol-Myers Squibb, and Pharmacia; has been a consultant for Bayer, Pfizer, Aventis, Ortho-McNeil, and Janssen-Ortho; and has been on the speakers' bureau for Pfizer, Aventis, Wyeth, Ortho-McNeil, and Bayer. Thomas M. File, Jr., has received research funding from Abbott, AstraZeneca, Bayer, Bristol-Myers Squibb, Cubist, GlaxoSmithKline, Pfizer, and Wyeth; has been a consultant for Aventis, Bayer, Cubist, GlaxoSmithKline, Ortho-McNeil, Pfizer, and Wyeth; and has been on the speakers' bureau for Abbott, Aventis, Bayer, GlaxoSmithKline, Merck, Ortho-McNeil, Pfizer, and Wyeth.
Table 2.

Empiric antibacterial selection for community-acquired pneumonia (CAP): advantages and disadvantages.
Acknowledgments
We thank Paul Edelstein, Michael Fine, Fred Hayden, Peter Houck, Mark Loeb, and Donald Low.
Appendix A
How The Pneumonia PORT Severity Index (PSI) is Derived
Patients are stratified into 5 severity classes by means of a 2-step process.
Step 1. Determination of whether patients meet the following criteria for class I: age <50 years, with 0 of 5 comorbid conditions (i.e., neoplastic disease, liver disease, congestive heart failure, cerebrovascular disease, and renal disease), normal or only mildly deranged vital signs, and normal mental status.
Step 2. Patients not assigned to risk class I are stratified into classes II–V on the basis of points assigned for 3 demographic variables (age, sex, and nursing home residency), 5 comorbid conditions (listed above), 5 physical examination findings (pulse, ⩾125 beats/min; respiratory rate, ⩾30 breaths/min; systolic blood pressure, <90 mm Hg; temperature, <35°C or ⩾40°C; and altered mental status), and 7 laboratory and/or radiographic findings (arterial pH, <7.35; blood urea nitrogen level, ⩾30 mg/dL; sodium level, <130 mmol/L; glucose level, ⩾250 mg/dL; hematocrit, <30%; hypoxemia by O2 saturation, <90% by pulse oximetry or <60 mm Hg by arterial blood gas; and pleural effusion on baseline radiograph).
For classes I–III, hospitalization is usually not required. For classes IV and V, the patient will usually require hospitalization.
It should be noted that social factors, such as outpatient support mechanisms and probability of adherence to treatment, are not included in this assessment.
Footnotes
These guidelines were developed and issued on behalf of the Infectious Diseases Society of America.
A conflict of interest disclosure can be found at the end of the text.
Members of the Infectious Diseases Society of American Community-Acquired Pneumonia Committee; L.A.M., chair.
References
- 1.Bartlett JG, Breiman RF, Mandell LA, File TM., Jr Community-acquired pneumonia in adults: guidelines for management. Infectious Diseases Society of America. Clin Infect Dis. 1998;26:811–38. doi: 10.1086/513953. [DOI] [PubMed] [Google Scholar]
- 2.Bartlett JG, Dowell SF, Mandell LA, File TM, Jr, Musher DM, Fine MJ. Practice guidelines for the management of community acquired pneumonia in adults. Clin Infect Dis. 2000;31:347–82. doi: 10.1086/313954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Metlay JP, Fine MJ. Testing strategies in the initial management of patients with community-acquired pneumonia. Ann Intern Med. 2003;138:109–18. doi: 10.7326/0003-4819-138-2-200301210-00012. [DOI] [PubMed] [Google Scholar]
- 4.Halm EA, Teirstein AS. Management of community-acquired pneumonia. N Engl J Med. 2002;347:2039–45. doi: 10.1056/NEJMcp020499. [DOI] [PubMed] [Google Scholar]
- 5.Mandell LA, Marrie TJ, Grossman RF, Chow AW, Hyland RH. Canadian guidelines for the initial management of community-acquired pneumonia: an evidence-based update by the Canadian Infectious Diseases Society and the Canadian Thoracic Society. The Canadian Community-Acquired Pneumonia Working Group. Clin Infect Dis. 2000;31:383–421. doi: 10.1086/313959. [DOI] [PubMed] [Google Scholar]
- 6.American College of Emergency Physicians Clinical policy for the management and risk stratifications of community acquired pneumonia in adults in the emergency department. Ann Emerg Med. 2001;38:107–13. doi: 10.1067/mem.2001.115880. [DOI] [PubMed] [Google Scholar]
- 7.Fine MJ, Yealy DM, Auble TE, et al. Translating the pneumonia severity index into practice: a trial to influence the admission decision. J Gen Intern Med. 2002;17((Suppl 1)):192. [Google Scholar]
- 8.Halm EA, Fine MJ, Kapoor WN, et al. Instability on hospital discharge and the risk of adverse outcome in patients with pneumonia. Arch Intern Med. 2002;162:1278–84. doi: 10.1001/archinte.162.11.1278. [DOI] [PubMed] [Google Scholar]
- 9.Dowell SF, Peeling RW, Boman J, et al. Standardizing Chlamydia pneumoniae assays: recommendations from the Centers for Disease Control and Prevention (USA) and the Laboratory Centre for Disease Control (Canada) Clin Infect Dis. 2001;33:492–503. doi: 10.1086/322632. [DOI] [PubMed] [Google Scholar]
- 10.Domínguez J, Galí N, Blanco S, et al. Detection of Streptococcus pneumoniae antigen by a rapid immunochromatographic assay in urine samples. Chest. 2001;119:243–9. doi: 10.1378/chest.119.1.243. [DOI] [PubMed] [Google Scholar]
- 11.Burel E, Dufour P, Gauduchon V, Jarraud S, Etienne J. Evaluation of a rapid immunochromatographic assay for detection of Streptococcus pneumoniae antigen in urine samples. Eur J Clin Microbiol Infect Dis. 2001;20:840–1. doi: 10.1007/s100960100614. [DOI] [PubMed] [Google Scholar]
- 12.Murdoch DR, Laing RT, Mills GD, et al. Evaluation of a rapid immunochromatographic test for detection of Streptococcus pneumoniae antigen in urine samples from adults with community-acquired pneumonia. J Clin Microbiol. 2001;39:3495–8. doi: 10.1128/JCM.39.10.3495-3498.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Farina C, Arosio M, Vailati F, Moioli F, Goglio A. Urinary detection of Streptococcus pneumoniae antigen for diagnosis of pneumonia. New Microbiol. 2002;25:259–63. [PubMed] [Google Scholar]
- 14.Yu VL, Kellog JA, Plouffe JF, et al. Program and abstracts of the 38th Annual Meeting of the Infectious Diseases Society of America (New Orleans) Alexandria, VA: Infectious Diseases Society of America; 2000. Evaluation of the Binax urinary, Gram stain and sputum culture for Streptococcus pneumoniae in patients with community-acquired pneumonia [abstract 262] [Google Scholar]
- 15.Gutierrez F, Rodriguez JC, Ayelo A, et al. Evaluation of the immunochromatographic Binax NOW assay for detection of Streptococcus pneumoniae urinary antigen in a prospective study of community-acquired pneumonia in Spain. Clin Infect Dis. 2003;36:286–92. doi: 10.1086/345852. [DOI] [PubMed] [Google Scholar]
- 16.Fadan H, Heimerl M, Varma C, Goodman G, Winkelstein P. Urinary excretion of pneumococcal cell wall polysaccharide in children. Pediatr Infect Dis J. 2002;21:791–3. doi: 10.1097/00006454-200208000-00020. [DOI] [PubMed] [Google Scholar]
- 17.Adegbola RA, Obaro SK, Biney E, Greenwood BM. Evaluation of Binax NOW Streptococcus pneumoniae urinary antigen test in children in a community with a high carriage rate of pneumococcus. Pediatr Infect Dis J. 2001;20:718–9. doi: 10.1097/00006454-200107000-00018. [DOI] [PubMed] [Google Scholar]
- 18.Dowell SF, Garman RL, Liu G, Levine O, Yang Y-H. Evaluation of Binax NOW, an assay for the detection of pneumococcal antigen in urine samples, performed among pediatric patients. Clin Infect Dis. 2001;32:824–5. doi: 10.1086/319205. [DOI] [PubMed] [Google Scholar]
- 19.Pesola GR. The urinary antigen test for the diagnosis of pneumococcal pneumonia. Chest. 2001;119:9–11. doi: 10.1378/chest.119.1.9. [DOI] [PubMed] [Google Scholar]
- 20.Musher DM, Bartlett JG, Doern GV. A fresh look at the definition of susceptibility of Streptococcus pneumoniae to beta lactam antibiotics. Arch Intern Med. 2001;161:2538–44. doi: 10.1001/archinte.161.21.2538. [DOI] [PubMed] [Google Scholar]
- 21.Friedland IR. Comparison of the response to antimicrobial therapy of penicillin-resistant and penicillin-susceptible pneumococcal disease. Pediatr Infect Dis J. 1995;14:885–90. doi: 10.1097/00006454-199510000-00013. [DOI] [PubMed] [Google Scholar]
- 22.Pallares R, Linares J, Vadillo M, et al. Resistance to penicillin and cephalosporin and mortality from severe pneumococcal pneumonia in Barcelona, Spain. N Engl J Med. 1995;333:474–80. doi: 10.1056/NEJM199508243330802. [DOI] [PubMed] [Google Scholar]
- 23.Choi EH, Lee HJ. Clinical outcome of invasive infections by penicillin-resistant Streptococcus pneumoniae in Korean children. Clin Infect Dis. 1998;26:1346–54. doi: 10.1086/516340. [DOI] [PubMed] [Google Scholar]
- 24.Turret G, Blum S, Fazal B, Justman J, Telzak E. Penicillin resistance and other predictors of mortality in pneumococcal bacteremia in a population with high HIV seroprevalence. Clin Infect Dis. 1999;29:321–7. doi: 10.1086/520209. [DOI] [PubMed] [Google Scholar]
- 25.Feikin D, Schuchat A, Kolczak M, et al. Mortality from invasive pneumococcal pneumonia in the era of antibiotic resistance, 1995–1997. Am J Public Health. 2000;90:223–9. doi: 10.2105/ajph.90.2.223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Heffelfinger JD, Dowel SF, Jorgensen JH, et al. Management of community acquired pneumonia in the era of pneumococcal resistance: a report from the Drug-Resistant Streptococcus pneumoniae Therapeutic Working Group. Arch Intern Med. 2000;160:1399–408. doi: 10.1001/archinte.160.10.1399. [DOI] [PubMed] [Google Scholar]
- 27.World Health Organization (WHO) Cumulative number of reported probable cases of severe acute respiratory syndrome (SARS) Geneva: WHO; 2003. [Google Scholar]
- 28.Outbreak of severe acute respiratory syndrome—worldwide, 2003. JAMA. 2003;289:1775–6. doi: 10.1001/jama.289.14.1775. [DOI] [PubMed] [Google Scholar]
- 29.Update: outbreak of severe acute respiratory syndrome—worldwide, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:241–6. 248. [PubMed] [Google Scholar]
- 30.Outbreak of severe acute respiratory syndrome—worldwide, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:226–8. [PubMed] [Google Scholar]
- 31.Cluster of severe acute respiratory syndrome cases among protected health-care workers—Toronto, Canada, April 2003. MMWR Morb Mortal Wkly Rep. 2003;52:433–6. [PubMed] [Google Scholar]
- 32.Centers for Disease Control and Prevention Interim domestic guidance on the use of respirators to prevent transmission of SARS. 6 May. 2003. Available at: http://www.cdc.gov.
- 33.Ksiazek TG, Erdman D, Goldsmith C, et al. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953–66. doi: 10.1056/NEJMoa030781. [DOI] [PubMed] [Google Scholar]
- 34.Drosten C, Gunther S, Preiser W, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med. 2003;348:1967–76. doi: 10.1056/NEJMoa030747. [DOI] [PubMed] [Google Scholar]
- 35.Fouchier RA, Kuiken T, Schutten M, et al. Aetiology: Koch's postulates fulfilled for SARS virus. Nature. 2003;423:240. doi: 10.1038/423240a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rota PA, Oberste MS, Monroe SS, et al. Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science. 2003;300:1394–9. doi: 10.1126/science.1085952. [DOI] [PubMed] [Google Scholar]
- 37.Centers for Disease Control and Prevention Updated interim US case definition of severe acute respiratory syndrome (SARS). 18 July. 2003 Available at: http://www.cdc.gov/ncidod/sars/casedefinition/htm. [Google Scholar]
- 38.Updated interim surveillance case definition for severe acute respiratory syndrome (SARS) April 29, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:391–3. [PubMed] [Google Scholar]
- 39.World Health Organization Case definitions for surveillance of severe acute respiratory syndrome (SARS). 1 May. 2003 Available at: http://www.who.int/csr/sars/casedefinition/en. [Google Scholar]
- 40.Peiris JS, Chu CM, Cheng VC, et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003;361:1767–72. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tsang KW, Ho PL, Ooi GC, et al. A cluster of cases of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348:1977–85. doi: 10.1056/NEJMoa030666. [DOI] [PubMed] [Google Scholar]
- 42.Poutanen SM, Low DE, Henry B, et al. Identification of severe acute respiratory syndrome in Canada. N Engl J Med. 2003;348:1995–2005. doi: 10.1056/NEJMoa030634. [DOI] [PubMed] [Google Scholar]
- 43.Wong KT, Antonio GE, Hui DS, et al. Severe acute respiratory syndrome: radiographic appearances and pattern of progression in 138 patients. Radiology. 2003;228:401–6. doi: 10.1148/radiol.2282030593. [DOI] [PubMed] [Google Scholar]
- 44.Centers for Disease Control and Prevention Severe acute respiratory syndrome (SARS) diagnosis/evaluation. 18 July. 2003. http://www.cdc.gov/ncidod/sars/diagnosis.htm.
- 45.Severe acute respiratory syndrome (SARS) and coronavirus testing—United States, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:297–302. [PubMed] [Google Scholar]
- 46.Stahl JE, Barza M, DesJardin J, et al. Effect of macrolides as part of initial empiric therapy on length of stay in patients hospitalized with community-acquired pneumonia. Arch Intern Med. 1999;159:2576–80. doi: 10.1001/archinte.159.21.2576. [DOI] [PubMed] [Google Scholar]
- 47.Gleason PP, Meehan TP, Fine JM, et al. Association between initial antimicrobial therapy and medical outcomes for hospitalized elderly patients with pneumonia. Arch Intern Med. 1999;159:2562–72. doi: 10.1001/archinte.159.21.2562. [DOI] [PubMed] [Google Scholar]
- 48.Mufson MA, Stanek RJ. Bacteremic pneumococcal pneumonia in one American city: a 20-year longitudinal study, 1978–1997. Am J Med. 1999;107((Suppl)):34–43. doi: 10.1016/s0002-9343(99)00098-4. [DOI] [PubMed] [Google Scholar]
- 49.Waterer GW, Somes GW, Wunderink RG. Monotherapy may be suboptimal for severe bacteremic pneumococcal pneumonia. Arch Intern Med. 2001;161:1837–42. doi: 10.1001/archinte.161.15.1837. [DOI] [PubMed] [Google Scholar]
- 50.Martinez JA, Horcajada JP, Almeld M, et al. Addition of a macrolide to a β-lactam based empirical antibiotic regimen is associated with lower in-hospital mortality for patients with bacteremic pneumococcal pneumonia. Clin Infect Dis. 2003;36:396–8. doi: 10.1086/367541. [DOI] [PubMed] [Google Scholar]
- 51.Culic O, Erakovic V, Parnham MJ. Anti-inflammatory effects of macrolide antibiotics. Eur J Pharmacol. 2001;429:209–29. doi: 10.1016/s0014-2999(01)01321-8. [DOI] [PubMed] [Google Scholar]
- 52.File TM, Mandell LA. What is optimal antimicrobial therapy for bacterial pneumococcal pneumonia. Clin Infect Dis. 2003;36:396–8. doi: 10.1086/367545. [DOI] [PubMed] [Google Scholar]
- 53.Marston BJ, Plouffe JF, File TM, et al. Incidence of community acquired pneumonia requiring hospitalizations: results of a population-based active surveillance study in Ohio. Community-Based Pneumonia Incidence Study Group. Arch Intern Med. 1997;157:1709–18. [PubMed] [Google Scholar]
- 54.Fang GD, Fine M, Orloff J, et al. New and emerging etiologies for community acquired pneumonia with implications for therapy: a prospective Multicenter study of 359 cases. Medicine (Baltimore) 1990;69:307–16. doi: 10.1097/00005792-199009000-00004. [DOI] [PubMed] [Google Scholar]
- 55.Mundy LM, Auwaerter PG, Oldach D, et al. Community acquired pneumonia: impact of immune status. Am J Respir Crit Care Med. 1995;152:1309–15. doi: 10.1164/ajrccm.152.4.7551387. [DOI] [PubMed] [Google Scholar]
- 56.Keller DW, Lipman HB, Marston BJ, et al. Program and abstracts of the 35th Interscience on Antimicrobial Agents and Chemotherapy (San Francisco) Washington, DC: American Society for Microbiology; 1995. Clinical diagnosis of legionnaires' disease (LD) using a multivariate model [abstract K55] p. 297. [Google Scholar]
- 57.Stout JE, Yu VL. Legionellosis. N Engl J Med. 1997;337:682–7. doi: 10.1056/NEJM199709043371006. [DOI] [PubMed] [Google Scholar]
- 58.Marston BJ, Lipman HB, Breiman RF. Surveillance for legionnaires' disease: risk factors for morbidity and mortality. Arch Intern Med. 1994;154:2417–22. [PubMed] [Google Scholar]
- 59.Benin AL, Benson RF, Arnold KE, et al. An outbreak of travel-associated legionnaires disease and Pontiac fever: the need for enhanced surveillance of travel-associated legionellosis in the United States. J Infect Dis. 2002;185:237–43. doi: 10.1086/338060. [DOI] [PubMed] [Google Scholar]
- 60.Helbig JH, Uldum SA, Bernander S, et al. Clinical utility of urinary antigen detection for diagnosis of community-acquired, travel-associated, and nosocomial legionnaires' disease. J Clin Microbiol. 2003;41:838–40. doi: 10.1128/JCM.41.2.838-840.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Den Boer JW, Yzerman EP, Schellekens J, et al. A large outbreak of Legionnaires' disease at a flower show, the Netherlands, 1999. Emerg Infect Dis. 2002;8:37–43. doi: 10.3201/eid0801.010176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Vergis EN, Yu VL. New directions for future studies of community-acquired pneumonia: optimizing impact on patient care. Eur J Clin Microbiol Infect Dis. 1999;18:847–51. doi: 10.1007/s100960050418. [DOI] [PubMed] [Google Scholar]
- 63.Roig J, Aguilar X, Ruiz J, et al. Comparative study of Legionella pneumophila and other nosocomial-acquired pneumonias. Chest. 1991;99:344–50. doi: 10.1378/chest.99.2.344. [DOI] [PubMed] [Google Scholar]
- 64.Sopena N, Sabria-Leal M, Pedro-Botet ML, et al. Comparative study of the clinical presentation of Legionella pneumonia and other community-acquired pneumonias. Chest. 1998;113:1195–200. doi: 10.1378/chest.113.5.1195. [DOI] [PubMed] [Google Scholar]
- 65.Gupta SK, Imperiale TF, Sarosi GA. Evaluation of the Winthrop-University Hospital criteria to identify Legionella pneumonia. Chest. 2001;120:1064–71. doi: 10.1378/chest.120.4.1064. [DOI] [PubMed] [Google Scholar]
- 66.Murdoch DR. Diagnosis of Legionella infection. Clin Infect Dis. 2003;36:64–9. doi: 10.1086/345529. [DOI] [PubMed] [Google Scholar]
- 67.Centers for Disease Control and Prevention Case definitions for infectious conditions under public health surveillance. MMWR Morb Mortal Wkly Rep. 1997;46(RR-10):1–55. [PubMed] [Google Scholar]
- 68.Roig J, Rello J, Yu VL. Legionnaires' disease: a rational approach to therapy. J Antimicrob Chemother. 2003;51:1119–29. doi: 10.1093/jac/dkg191. [DOI] [PubMed] [Google Scholar]
- 69.Edelstein PH. Antimicrobial chemotherapy for Legionnaires disease: time for a change. Ann Intern Med. 1998;129:328–30. doi: 10.7326/0003-4819-129-4-199808150-00012. [DOI] [PubMed] [Google Scholar]
- 70.Heath CH, Grove DI, Looke DFM. Delay in appropriate therapy of Legionella pneumonia associated with increased mortality. Eur J Clin Microbiol Infect Dis. 1996;15:286–90. doi: 10.1007/BF01695659. [DOI] [PubMed] [Google Scholar]
- 71.Glezen WP, Greenberg SB, Atmar RL, et al. Impact of respiratory virus infections on persons with chronic underlying conditions. JAMA. 2000;283:499–505. doi: 10.1001/jama.283.4.499. [DOI] [PubMed] [Google Scholar]
- 72.Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA. 2003;289:179–86. doi: 10.1001/jama.289.2.179. [DOI] [PubMed] [Google Scholar]
- 73.Stockton J, Stephenson I, Fleming D, Zambon M. Human metapneumovirus as a cause of community-acquired respiratory illness. Emerg Infect Dis. 2002;8:897–901. doi: 10.3201/eid0809.020084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Falsey AR, Erdman D, Anderson LJ, Walsh EE. Human metapneumovirus infections in young and elderly adults. J Infect Dis. 2003;187:785–90. doi: 10.1086/367901. [DOI] [PubMed] [Google Scholar]
- 75.Greenberg SB. Viral pneumonia. Infect Dis Clin North Am. 1991;5:603–21. [PubMed] [Google Scholar]
- 76.Lim WS, Macfarlane JT, Boswell TC, et al. Study of community acquired pneumonia aetiology (SCAPA) in adults admitted to hospital: implications for management guidelines. Thorax. 2001;56:296–301. doi: 10.1136/thorax.56.4.296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kaiser L, Briones MS, Hayden FG. Performance of virus isolation and Directigen Flu A to detect influenza A virus in experimental human infection. J Clin Virol. 1999;14:191–7. doi: 10.1016/s1386-6532(99)00058-x. [DOI] [PubMed] [Google Scholar]
- 78.Bellei N, Benfica D, Perosa AH, et al. Evaluation of a rapid test (QuickVue) compared with the shell vial assay for detection of influenza virus clearance after antiviral treatment. J Virol Methods. 2003;109:85–8. doi: 10.1016/s0166-0934(03)00050-8. [DOI] [PubMed] [Google Scholar]
- 79.Monto AS, Gravenstein S, Elliott M, et al. Clinical signs and symptoms predicting influenza infection. Arch Intern Med. 2000;160:3243–7. doi: 10.1001/archinte.160.21.3243. [DOI] [PubMed] [Google Scholar]
- 80.Gubareva LV, Kaiser L, Hayden FG. Influenza virus neuraminidase inhibitors. Lancet. 2000;355:827–35. doi: 10.1016/S0140-6736(99)11433-8. [DOI] [PubMed] [Google Scholar]
- 81.Gubareva LV, Kaiser L, Matrosovich MN, et al. Selection of influenza virus mutants in experimentally infected volunteers treated with oseltamivir. J Infect Dis. 2001;183:523–31. doi: 10.1086/318537. [DOI] [PubMed] [Google Scholar]
- 82.Kaiser L, Keene ON, Hammond JM, et al. Impact of zanamivir on antibiotic use for respiratory events following acute influenza in adolescents and adults. Arch Intern Med. 2000;160:3234–40. doi: 10.1001/archinte.160.21.3234. [DOI] [PubMed] [Google Scholar]
- 83.Treanor JJ, Hayden FG, Vrooman PS, et al. Efficacy and safety of the oral neuraminidase inhibitor oseltamivir in treating acute influenza: a randomized controlled trial. US Oral Neuraminidase Study Group. JAMA. 2000;283:1016–24. doi: 10.1001/jama.283.8.1016. [DOI] [PubMed] [Google Scholar]
- 84.Hayden FG, Treanor JJ, Fritz RS, et al. Use of the oral neuraminidase inhibitor oseltamivir in experimental human influenza: randomized controlled trials for prevention and treatment. JAMA. 1999;282:1240–6. doi: 10.1001/jama.282.13.1240. [DOI] [PubMed] [Google Scholar]
- 85.Kaiser L, Hayden FG. Hospitalizing influenza in adults. Curr Clin Top Infect Dis. 1999;19:112–34. [PubMed] [Google Scholar]
- 86.Oliveira EC, Marik PE, Colice G. Influenza pneumonia: a descriptive study. Chest. 2001;119:1717–23. doi: 10.1378/chest.119.6.1717. [DOI] [PubMed] [Google Scholar]
- 87.Haake DA, Zakowski PC, Haake DL, Bryson YJ. Early treatment with acyclovir for varicella pneumonia in otherwise healthy adults: retrospective controlled study and review. Rev Infect Dis. 1990;12:788–98. doi: 10.1093/clinids/12.5.788. [DOI] [PubMed] [Google Scholar]
- 88.Chapman LE, Mertz GJ, Peters CJ, et al. Intravenous ribavirin for Hantavirus pulmonary syndrome: safety and tolerance during 1 year of open-label experience. Ribavirin Study Group. Antivir Ther. 1999;4:211–9. doi: 10.1177/135965359900400404. [DOI] [PubMed] [Google Scholar]
- 89.Chapman LE, Ellis BA, Koster FT, et al. Discriminators between Hantavirus-infected and -uninfected persons enrolled in a trial of intravenous ribavirin for presumptive Hantavirus pulmonary syndrome. Clin Infect Dis. 2002;34:293–304. doi: 10.1086/324619. [DOI] [PubMed] [Google Scholar]
- 90.Henderson DA. The looming threat of bioterrorism. Science. 1999;283:1279. doi: 10.1126/science.283.5406.1279. [DOI] [PubMed] [Google Scholar]
- 91.Eitzen E, Pavlin J, Cieslak T, et al. Medical management of biological casualties handbook. 3rd ed. Frederick, MD: US Army Medical Research Institute of Infectious Diseases; 1998. pp. 15–21. 40–51. [Google Scholar]
- 92.Bartlett JG, Inglesby TV, Jr, Borio L. Management of anthrax. Clin Infect Dis. 2002;35:851–8. doi: 10.1086/341902. [DOI] [PubMed] [Google Scholar]
- 93.Jernigan JA, Stephens DS, Ashford DA, et al. Bioterrorism-related inhalational anthrax: the first 10 cases reported in the United States. Emerg Infect Dis. 2001;7:933–44. doi: 10.3201/eid0706.010604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Kuehnert MJ, Doyle TJ, Hill HA, et al. Clinical features that discriminate inhalational anthrax from other acute respiratory illnesses. Clin Infect Dis. 2003;36:328–36. doi: 10.1086/346035. [DOI] [PubMed] [Google Scholar]
- 95.Inglesby TV, O'Toole T, Henderson DA, et al. Anthrax as a biological weapon, 2002: updated recommendations for management. JAMA. 2002;287:2236–52. doi: 10.1001/jama.287.17.2236. [DOI] [PubMed] [Google Scholar]
- 96.Tularemia—United States, 1990–2000. MMWR Morb Mortal Wkly Rep. 2002;51:181–4. [PubMed] [Google Scholar]
- 97.Dennis DT, Inglesby TV, Henderson DA, et al. Tularemia as a biological weapon: medical and public health management. JAMA. 2001;285:2763–73. doi: 10.1001/jama.285.21.2763. [DOI] [PubMed] [Google Scholar]
- 98.Shapiro DS, Schwartz DR. Exposure of laboratory workers to Francisella tularensis despite a bioterrorism procedure. J Clin Microbiol. 2002;40:2278–81. doi: 10.1128/JCM.40.6.2278-2281.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Enderlin G, Morales L, Jacobs RF, Cross JT. Streptomycin and alternative agents for the treatment of tularemia: review of the literature. Clin Infect Dis. 1994;19:42–7. doi: 10.1093/clinids/19.1.42. [DOI] [PubMed] [Google Scholar]
- 100.Evans ME, Gregory DW, Schaffner W, McGee ZA. Tularemia: a 30-year experience with 88 cases. Medicine (Baltimore) 1985;64:251–69. [PubMed] [Google Scholar]
- 101.Mason WL, Eigelsbach HT, Little SF, Bates JH. Treatment of tularemia, including pulmonary tularemia, with gentamicin. Am Rev Respir Dis. 1980;121:39–45. doi: 10.1164/arrd.1980.121.1.39. [DOI] [PubMed] [Google Scholar]
- 102.Limaye AP, Hooper CJ. Treatment of tularemia with fluoroquinolones: two cases and review. Clin Infect Dis. 1999;29:922–4. doi: 10.1086/520458. [DOI] [PubMed] [Google Scholar]
- 103.Inglesby TV, Dennis DT, Henderson DA, et al. Plague as a biological weapon: medical and public health management. Working Group on Civilian Biodefense. JAMA. 2000;283:2281–90. doi: 10.1001/jama.283.17.2281. [DOI] [PubMed] [Google Scholar]
- 104.Fatal human plague—Arizona and Colorado, 1996. MMWR Morb Mortal Wkly Rep. 1997;46:617–20. [PubMed] [Google Scholar]
- 105.Rasoamanana B, Coulanges P, Michel P, Rasolofonirina N. Sensitivity of Yersinia pestis to antibiotics: 277 strains isolated in Madagascar between 1926 and 1989. Arch Inst Pasteur Madagascar. 1989;56:37–53. [PubMed] [Google Scholar]
- 106.Bonacorsi SP, Scavizzi MR, Guiyoule A, et al. Assessment of a fluoroquinolone, three beta lactams, two aminoglycosides, and a cycline in treatment of murine Yersinia pestis infection. Antimicrob Agents Chemother. 1994;38:481–6. doi: 10.1128/aac.38.3.481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Russell P, Eley SM, Green M, et al. Efficacy of doxycycline and ciprofloxacin against experimental Yersinia pestis infection. J Antimicrob Chemother. 1998;41:301–5. doi: 10.1093/jac/41.2.301. [DOI] [PubMed] [Google Scholar]
- 108.Pneumonia and influenza death rates, United States, 1979–1994. MMWR Morb Mortal Wkly Rep. 1995;44:535–7. [PubMed] [Google Scholar]
- 109.Muder R. Pneumonia in residents of long-term care facilities: epidemiology, etiology, management, and prevention. Am J Med. 1998;105:319–30. doi: 10.1016/s0002-9343(98)00262-9. [DOI] [PubMed] [Google Scholar]
- 110.Joikinen C, Heiskanem L, Juvonen H, et al. Microbial etiology of community-acquired pneumonia in the adult population of 4 municipalities in Eastern Finland. Clin Infect Dis. 2001;32:1141–54. doi: 10.1086/319746. [DOI] [PubMed] [Google Scholar]
- 111.Ruiz M, Ewig S, Marcos M, et al. Etiology of community-acquired pneumonia: impact of age, comorbidity, and severity. Am J Resp Crit Care Med. 1999;160:397–405. doi: 10.1164/ajrccm.160.2.9808045. [DOI] [PubMed] [Google Scholar]
- 112.Koivula I, Sten M, Makela PH. Risk factors for pneumonia in the elderly. Am J Med. 1994;96:313–20. doi: 10.1016/0002-9343(94)90060-4. [DOI] [PubMed] [Google Scholar]
- 113.Loeb M, McGeer A, McArthur M, Walter S, Simor AE. Risk factors for pneumonia and other lower respiratory tract infections in elderly residents of long-term care facilities. Arch Intern Med. 1999;159:2058–64. doi: 10.1001/archinte.159.17.2058. [DOI] [PubMed] [Google Scholar]
- 114.Metlay JP, Schulz R, Li YH, et al. Influence of age on symptoms at presentation in patients with community-acquired pneumonia. Arch Intern Med. 1997;157:1453–9. [PubMed] [Google Scholar]
- 115.Marrie TJ, Haldane EV, Faulkner RS, et al. Community-acquired pneumonia requiring hospitalization: is it different in the elderly? J Am Geriatr Soc. 1985;33:671–80. doi: 10.1111/j.1532-5415.1985.tb01775.x. [DOI] [PubMed] [Google Scholar]
- 116.Gross PA, Hermogenes AW, Sacks HS, Lau J, Levandowski RA. The efficacy of influenza vaccine in elderly persons: a meta-analysis and review of the literature. Ann Intern Med. 1995;123:518–27. doi: 10.7326/0003-4819-123-7-199510010-00008. [DOI] [PubMed] [Google Scholar]
- 117.Nichol KL, Nordin J, Mullooly J, Lask R, Fillbrandt K, Iwane M. Influenza vaccination and reduction in hospitalizations for cardiac disease and stroke among the elderly. N Engl J Med. 2003;348:1322–32. doi: 10.1056/NEJMoa025028. [DOI] [PubMed] [Google Scholar]
- 118.Potter J, Stott DJ, Roberts MA, et al. Influenza vaccination of healthcare workers in long-term care hospitals reduces the mortality of elderly patients. J Infect Dis. 1997;175:1–6. doi: 10.1093/infdis/175.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Carman WF, Elder AG, Wallace LA, et al. Effects of influenza vaccination of healthcare workers on mortality of elderly people in long term care: a randomized controlled trial. Lancet. 2000;355:93–7. doi: 10.1016/S0140-6736(99)05190-9. [DOI] [PubMed] [Google Scholar]
- 120.Koivula I, Stén M, Leinonen M, Mäkelä PH. Clinical efficacy of pneumococcal vaccine in the elderly: a randomized, single-blind population-based trial. Am J Med. 1997;103:281–90. doi: 10.1016/s0002-9343(97)00149-6. [DOI] [PubMed] [Google Scholar]
- 121.Simberkoff MS, Cross AP, Al-Ibrahim M, et al. Efficacy of pneumococcal vaccine in high-risk patients: results of a Veterans Administration Cooperative Study. N Engl J Med. 1986;315:1318–27. doi: 10.1056/NEJM198611203152104. [DOI] [PubMed] [Google Scholar]
- 122.örtqvist Å, Hedlund J, Burman LÅ, et al. Randomized trial of 23-valent pneumococcal capsular polysaccharide vaccine in prevention of pneumonia in middle-age and elderly people. Lancet. 1998;351:399–403. doi: 10.1016/s0140-6736(97)07358-3. [DOI] [PubMed] [Google Scholar]
- 123.Musher DM, Mediwala R, Phan HM, Chen G, Baughn RE. Nonspecificity of assaying for IgG to pneumolysin in circulating immune complexes as a means to diagnose pneumococcal pneumonia. Clin Infect Dis. 2001;32:534–8. doi: 10.1086/318709. [DOI] [PubMed] [Google Scholar]
- 124.Kaufman P. Pneumonia in old age: active immunization against pneumonia with pneumococcus polysaccharide; results of a six year study. Arch Intern Med. 1947;79:518–31. doi: 10.1001/archinte.1947.00220110058004. [DOI] [PubMed] [Google Scholar]
- 125.Gaillat J, Zmirou D, Mallaret MR, et al. Essai clinique du vaccin antipneumococcique chez des personnes âgées vivant en institution. Rev Epidémiol Santé Publique. 1985;33:437–44. [PubMed] [Google Scholar]
- 126.Butler JC, Freiman RF, Campbell JF, Lipman HB, Broome CV, Facklam RR. Polysaccharide pneumococcal vaccine efficacy: an evaluation of current recommendations. JAMA. 1993;270:1826–31. [PubMed] [Google Scholar]
- 127.Sims RV, Steinmann WC, McConville JH, King LR, Zwich WC, Schwartz JS. The clinical effectiveness of pneumococcal vaccine in the elderly. Ann Intern Med. 1988;108:653–7. doi: 10.7326/0003-4819-108-5-653. [DOI] [PubMed] [Google Scholar]
- 128.Shapiro ED, Berg AT, Austrian R, et al. The protective efficacy of polyvalent pneumococcal polysaccharide vaccine. N Engl J Med. 1991;325:1453–60. doi: 10.1056/NEJM199111213252101. [DOI] [PubMed] [Google Scholar]
- 129.Farr BM, Johnston BL, Cobb DK, et al. Preventing pneumococcal bacteremia in patients at risk: results of a matched case-control study. Arch Intern Med. 1995;155:2336–40. [PubMed] [Google Scholar]
- 130.Jackson LA, Neuzil KM, Yu O, et al. Effectiveness of pneumococcal polysaccharide vaccine in older adults. New Engl J Med. 2003;348:1747–55. doi: 10.1056/NEJMoa022678. [DOI] [PubMed] [Google Scholar]
- 131.Whitney CG, Farley MM, Hader J, et al. Decline in invasive pneumococcal disease following the introduction of protein-polysaccharide conjugate vaccine. N Engl J Med. 2003;348:1737–46. doi: 10.1056/NEJMoa022823. [DOI] [PubMed] [Google Scholar]
- 132.Centers for Disease Control and Prevention Facilitating influenza and pneumococcal vaccination through standing orders programs. MMWR Morb Mortal Wkly Rep. 2003;52:68–9. [PubMed] [Google Scholar]
- 133.Bridges CB, Harper SA, Fukuda K, et al. Prevention and control of influenza: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2003;52((RR-8)):1–34. [PubMed] [Google Scholar]
- 134.Harper SA, Fukuda K, Cox J, Bridges CB. Using live, attenuated influenza vaccine for prevention and control of influenza: supplemental recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2003;52((RR-13)):1–8. [PubMed] [Google Scholar]
- 135.Centers for Disease Control and Prevention Prevention of pneumococcal disease: recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Morb Mortal Wkly Rep. 1997;46:1–24. [Google Scholar]
- 136.Sisk JE, Whang W, Butler JC, Sneller V-P, Whitney CG. Cost-effectiveness of vaccination against invasive pneumococcal disease among people 50 through 64 years of age: role of comorbid conditions and race. Ann Intern Med. 2003;138:960–8. doi: 10.7326/0003-4819-138-12-200306170-00007. [DOI] [PubMed] [Google Scholar]
- 137.Lynch JP, Martinez FJ. Clinical relevance of macrolide-resistant Streptococcus pneumoniae for community-acquired pneumonia. Clin Infect Dis. 2002;34((Suppl)):27–46. doi: 10.1086/324527. [DOI] [PubMed] [Google Scholar]
- 138.Thornsberry C, Sahm DF, Kelly LJ, et al. Regional trends in antimicrobial resistance among clinical isolates of Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis in the United States: results from the TRUST Surveillance Program, 1999–2000. Clin Infect Dis. 2002;34((Suppl 1)):4–16. doi: 10.1086/324525. [DOI] [PubMed] [Google Scholar]
- 139.Hyde TB, Gay K, Stephens DS, et al. Macrolide resistance among invasive Streptococcus pneumoniae isolates. JAMA. 2001;286:1857–62. doi: 10.1001/jama.286.15.1857. [DOI] [PubMed] [Google Scholar]
- 140.Doern GV, Heilmann KP, Huynh HK, Rhomberg PR, Coffman SL, Brueggemann AB. Antimicrobial resistance among clinical isolates of Streptococcus pneumoniae in the United States during 1999–2000, including a comparison of resistance rates since 1994–1995. Antimicrob Agents Chemother. 2001;45:1721–9. doi: 10.1128/AAC.45.6.1721-1729.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Corso A, Severina EP, Petruk VF, Mauriz YR, Tomasz A. Molecular characterization of penicillin-resistant Streptococcus pneumoniae isolates causing respiratory disease in the United States. Microb Drug Resist. 1998;4:325–37. doi: 10.1089/mdr.1998.4.325. [DOI] [PubMed] [Google Scholar]
- 142.Shortridge V, Doern G, Brueggeman A, Beyer J, Flamm R. Prevalence of macrolide resistance mechanisms in Streptococcus pneumoniae isolates from a multicenter antibiotic resistance surveillance study conducted in The United States in 1994–1995. Clin Infect Dis. 1999;29:1186–8. doi: 10.1086/313452. [DOI] [PubMed] [Google Scholar]
- 143.Tait-Kamradt A, Davies T, Appelbaum PC, et al. Two new mechanisms of macrolide resistance in clinical strains of Streptococcus pneumoniae from Eastern Europe and North America. Antimicrob Agents Chemother. 2000;44:3395–401. doi: 10.1128/aac.44.12.3395-3401.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Musher DM, Dowell ME, Shortridge VD, et al. Emergence of macrolide resistance during treatment of pneumococcal pneumonia. New Engl J Med. 2002;346:630–1. doi: 10.1056/NEJM200202213460820. [DOI] [PubMed] [Google Scholar]
- 145.McCaig LF, Besser RE, Hughes JM. Antimicrobial drug prescription in ambulatory care settings, United States, 1992–2000. Emerg Infect Dis. 2003;9:432–7. doi: 10.3201/eid0904.020268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Gay K, Baughman W, Miller Y, et al. The emergence of Streptococcus pneumoniae resistant to macrolide antimicrobial agents: a 6-year population based assessment. J Infect Dis. 2000;182:1417–24. doi: 10.1086/315853. [DOI] [PubMed] [Google Scholar]
- 147.Perez-Trallero E, Fernandez-Mazarrasa C, Garcia-Rey C, et al. Antimicrobial susceptibilities of 1,684 Streptococcus pneumoniae and 2,039 Streptococcus pyogenes isolates and their ecological relationships: results of a 1-year (1998–1999) multicenter surveillance study in Spain. Antimicrob Agents Chemother. 2001;45:3334–40. doi: 10.1128/AAC.45.12.3334-3340.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Hoban DJ, Doern GV, Fluit AC, Roussel-Delvallez M, Jones RN. Worldwide prevalence of antimicrobial resistance in Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis in the SENTRY Antimicrobial Surveillance Program, 1997–1999. Clin Infect Dis. 2001;32((Suppl 2)):81–93. doi: 10.1086/320181. [DOI] [PubMed] [Google Scholar]
- 149.Dixon JMS. Pneumococcus resistant to erythromycin and lincomycin. Lancet. 1967;1:573. doi: 10.1016/s0140-6736(67)92803-6. [DOI] [PubMed] [Google Scholar]
- 150.Fogarty C, Goldschmidt R, Bush K. Bacteremic pneumonia due to multidrug-resistant pneumococci in 3 patients treated unsuccessfully with azithromycin and successfully with levofloxacin. Clin Infect Dis. 2000;31:613–5. doi: 10.1086/313976. [DOI] [PubMed] [Google Scholar]
- 151.Kelley MA, Weber DJ, Gilligan P, Cohen MS. Breakthrough pneumococcal bacteremia in patients being treated with azithromycin and clarithromycin. Clin Infect Dis. 2000;31:1008–11. doi: 10.1086/318157. [DOI] [PubMed] [Google Scholar]
- 152.Lonks JR, Garau J, Gomez L, et al. Failure of macrolide antibiotic treatment in patients with bacteremia due to erythromycin-resistant Streptococcus pneumoniae. Clin Infect Dis. 2002;35:556–64. doi: 10.1086/341978. [DOI] [PubMed] [Google Scholar]
- 153.Bertho G, Gharbi-Benarous J, Delaforge M, et al. Conformational analysis of ketolide, conformations of RU 004 in solution and bound to bacterial ribosomes. J Med Chem. 1998;27:3373–86. doi: 10.1021/jm970852i. [DOI] [PubMed] [Google Scholar]
- 154.Hansen LH, Mauvais P, Douthwaite S. The macrolide-ketolide antibiotic binding site is formed by structures in domains II and V of 23S ribosomal RNA. Mol Microbiol. 1999;31:623–31. doi: 10.1046/j.1365-2958.1999.01202.x. [DOI] [PubMed] [Google Scholar]
- 155.Leclercq R. Will resistance to ketolides develop in Streptococcus pneumoniae? Antimicrob Agents Chemother. 2002;46:2727–34. doi: 10.1128/AAC.46.9.2727-2734.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Panduch GA, Visalli MR, Jacobs MR, Appelbaum PC. Susceptibilities of penicillin and erythromycin susceptible and resistant pneumococci to MHR 3647 (RU 66647), a new ketolide, compared with susceptibilities to 17 other agents. Antimicrob Agents Chemother. 1998;42:624–30. doi: 10.1128/aac.42.3.624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Wooton M, Bowker KE, Janowska A, Holt HA, MacGowan AP. In-vitro activity of HMR 3647 against Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis and beta-haemolytic streptococci. J Antimicrob Chemother. 1999;44:445–53. doi: 10.1093/jac/44.4.445. [DOI] [PubMed] [Google Scholar]
- 158.Edelstein PH, Edelstein MA. In-vitro activity of the ketolide HMR 3647 (RU 6647) for Legionella spp., its pharmacokinetics in guinea pigs, and use of the drug to treat guinea pigs with Legionella pneumophila pneumonia. Antimicrob Agents Chemother. 1999;43:90–5. doi: 10.1128/aac.43.1.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Miyashita N, Fukano H, Niki Y, Matsushima T. In vitro activity of telithromycin, a new ketolide, against Chlamydia pneumoniae. J Antimicrob Chemother. 2001;48:403–5. doi: 10.1093/jac/48.3.403. [DOI] [PubMed] [Google Scholar]
- 160.Pascual AA, Ballesta S, Garcia I, et al. Uptake and intracellular activity of Ketolide HMR 3647 in human phagocytic and non phagocytic cells. Clin Microbiol Infect. 2001;7:65–9. doi: 10.1046/j.1469-0691.2001.00213.x. [DOI] [PubMed] [Google Scholar]
- 161.Kadota J, Ishimatsu Y, Iwashita T, et al. Intrapulmonary pharmacokinetics of telithromycin, a new ketolide, in healthy Japanese volunteers. Antimicrob Agents Chemother. 2002;46:917–21. doi: 10.1128/AAC.46.3.917-921.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Hagberg L, Carbon C, vanRensburg DJ, et al. Telithromycin in the treatment of community-acquired pneumonia: a pooled analysis. Respir Med. 2003;97:625–33. doi: 10.1053/rmed.2003.1492. [DOI] [PubMed] [Google Scholar]
- 163.Hagberg L, Torres A, VanRensburg DJ, et al. Efficacy and tolerability of once daily telithromycin compared with high-dose amoxicillin in the treatment of community-acquired pneumonia. Infection. 2002;30:378–86. doi: 10.1007/s15010-002-2096-z. [DOI] [PubMed] [Google Scholar]
- 164.Pullman J, Champlin J, Vrooman PS., Jr Efficacy and tolerability of once daily oral therapy with telithromycin compared with trovafloxacin in the treatment of community-acquired pneumonia. Int J Clin Pract. 2003;57:377–84. [PubMed] [Google Scholar]
- 165.Chen D, McGeer A, de Azevedo J, Low DE. Decreased susceptibility of Streptococcus pneumoniae to fluoroquinolones in Canada. Canadian Bacterial Surveillance Network. N Engl J Med. 1999;341:233–9. doi: 10.1056/NEJM199907223410403. [DOI] [PubMed] [Google Scholar]
- 166.Ferraro MJ. The rise of fluoroquinolone resistance: fact or fiction? J Chemother. 2002;14((Suppl 3)):31–41. doi: 10.1080/1120009x.2002.11782350. [DOI] [PubMed] [Google Scholar]
- 167.Ho Pl, Yung RW, Tsang DN, et al. Increasing resistance of Streptococcus pneumoniae to fluoroquinolones: results of a Hong Kong multicentre study in 2000. J Antimicrob Chemother. 2001;48:659–65. doi: 10.1093/jac/48.5.659. [DOI] [PubMed] [Google Scholar]
- 168.Low DL, de Azevedo J, Weiss CA, et al. Antimicrobial resistance among clinical isolates of Streptococcus pneumoniae in Canada during 2000. Antimicrob Agents Chemother. 2002;46:1295–301. doi: 10.1128/AAC.46.5.1295-1301.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Nagai K, Appelbaum PC, Davies TA, et al. Susceptibilities to telithromycin and six other agents and prevalence of macrolide resistance due to L4 ribosomal protein mutation among 992 pneumococci from 10 central and eastern European countries. Antimicrob Agents Chemother. 2002;46:371–7. doi: 10.1128/AAC.46.2.371-377.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Pankuch GA, Bozdogan B, Nagai K, et al. Incidence, epidemiology and characteristics of quinolone-nonsusceptible Streptococcus pneumoniae in Croatia. Antimicrob Agents Chemother. 2002;46:2671–5. doi: 10.1128/AAC.46.8.2671-2675.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Thornsberry C, Sahm DF, Kelly LJ, et al. Regional trends in antimicrobial resistance among clinical isolates of Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis in the United States: results from the TRUST Surveillance Program, 1999–2000. Clin Infect Dis. 2002;34((Suppl 1)):4–16. doi: 10.1086/324525. [DOI] [PubMed] [Google Scholar]
- 172.Tang P, Green K, McGeer A, et al. Program and abstracts of the 42nd Interscience Conference on Antimicrobial Agents and Chemotherapy (San Diego) Washington, DC: American Society for Microbiology; 2002. Emerging resistance in respiratory tract isolates of Streptococcus pneumoniae (SP) in Canada [abstract L992] p. 360. [Google Scholar]
- 173.Brueggemann AB, Coffman SL, Rhomberg P, et al. Fluoroquinolone resistance in Streptococcus pneumoniae in United States since 1994–1995. Antimicrob Agents Chemother. 2002;46:680–8. doi: 10.1128/AAC.46.3.680-688.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Resistance of Streptococcus pneumoniae to fluoroquinolones—United States, 1995–1999. MMWR Morb Mortal Wkly Rep. 2001;50:800–4. [PubMed] [Google Scholar]
- 175.Sahm DF, Karlowsky JA, Kelly LJ, et al. Need for annual surveillance of antimicrobial resistance in Streptococcus pneumoniae in the United States: 2-year longitudinal analysis. Antimicrob Agents Chemother. 2001;45:1037–42. doi: 10.1128/AAC.45.4.1037-1042.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Sahm DF, Peterson DE, Critchley IA, Thornsberry C. Analysis of ciprofloxacin activity against Streptococcus pneumoniae after 10 years of use in the United States. Antimicrob Agents Chemother. 2000;44:2521–4. doi: 10.1128/aac.44.9.2521-2524.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Doern G. Antimicrobial use and the emergence of antimicrobial resistance with Streptococcus pneumniae in the United States. Clin Infect Dis. 2001;33:187–92. doi: 10.1086/321847. [DOI] [PubMed] [Google Scholar]
- 178.Hsueh PR, Teng LJ, Wu TL, et al. Telithromycin- and fluoroquinolone-resistant Streptococcus pneumoniae in Taiwan with high prevalence of resistance to macrolides and beta-lactams: SMART program 2001 data. Antimicrob Agents Chemother. 2003;47:2145–51. doi: 10.1128/AAC.47.7.2145-2151.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Spika JS, Facklam RR, Plikaytis BD, Oxtoby MJ. Antimicrobial resistance of Streptococcus pneumoniae in the United States, 1979–1987. The Pneumococcal Surveillance Working Group. J Infect Dis. 1991;163:1273–8. doi: 10.1093/infdis/163.6.1273. [DOI] [PubMed] [Google Scholar]
- 180.Davidson R, Cavalcanti R, Brunton JL, et al. Resistance to levofloxacin and failure of treatment of pneumococcal pneumonia. N Engl J Med. 2002;346:747–50. doi: 10.1056/NEJMoa012122. [DOI] [PubMed] [Google Scholar]
- 181.Kupronis BA, Richards CL. Whitney CG, and the Active Bacterial Core Surveillance Team. Invasive pneumococcal disease in older adults residing in long term care facilities and in the community. J Amer Ger Society. 2003;51:1520–5. doi: 10.1046/j.1532-5415.2003.51501.x. [DOI] [PubMed] [Google Scholar]
- 182.Meehan TP, Fine MJ, Krumholz HM, et al. Quality of care, process and outcomes in elderly patients with pneumonia. JAMA. 1997;278:2080–4. [PubMed] [Google Scholar]
- 183.Houck PM, Bratzler DW, Nsa W, Ma A, Bartlett JG. Timing of antibiotic administration and outcomes for Medicare patients hospitalized with pneumonia. Arch Intern Med (in press). [DOI] [PubMed]
- 184.Kahn KL, Rogers WH, Rubenstein LV, et al. Measuring quality of care with explicit process criteria before and after implementation of the DRG-based prospective payment system. JAMA. 1990;264:1969–73. [PubMed] [Google Scholar]
- 185.McGarvey RN, Harper JJ. Pneumonia mortality reduction and quality improvement in a community based hospital. Quality Rev Bull. 1993;19:124–30. doi: 10.1016/s0097-5990(16)30605-4. [DOI] [PubMed] [Google Scholar]
- 186.Rosenstein AH, Hanel JB, Martin C. Timing is everything: impact of emergency department care on hospital length of stay. J Clinical Outcomes Management. 2000;7:31–6. [Google Scholar]
- 187.Battleman DS, Calahan M, Thaler HT. Rapid antibiotic delivery and appropriate antibiotic selection reduce length of hospital stay with community acquired pneumonia: link between quality of care and resource utilization. Arch Intern Med. 2002;162:682–8. doi: 10.1001/archinte.162.6.682. [DOI] [PubMed] [Google Scholar]
- 188.Nuorti JP, Butler JC, Farley MM, et al. Cigarette smoking and invasive pneumococcal disease. Active Bacterial Core Surveillance Team. N Engl J Med. 2000;342:681–9. doi: 10.1056/NEJM200003093421002. [DOI] [PubMed] [Google Scholar]
- 189.Rodvold KA, Gotfried MH, Danziger LH, Servi RJ. Intrapulmonary steady-state concentrations of clarithromycin and azithromycin in healthy adult volunteers. Antimicrob Agents Chemother. 1997;41:1399–402. doi: 10.1128/aac.41.6.1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Tessier PR, Kim MK, Zhou W, et al. Pharmacodynamic assessment of clarithromycin in a murine model of pneumococcal pneumonia. Antimicrob Agents Chemother. 2002;46:1425–34. doi: 10.1128/AAC.46.5.1425-1434.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Gotfried MH, Dattani D, Riffer E, et al. A controlled, double-blind, multicenter study comparing clarithromycin extended-release tablets and levofloxacin tablets in the treatment of community-acquired pneumonia. Clin Ther. 2002;24:736–51. doi: 10.1016/s0149-2918(02)85148-7. [DOI] [PubMed] [Google Scholar]
- 192.Vergis EN, Indorf A, File TM, Jr, et al. Azithromycin vs-cefuroxime plus erythromycin for empirical treatment of community-acquired pneumonia in hospitalized patients: a prospective, randomized, multicenter study. Arch Intern Med. 2000;160:1294–300. doi: 10.1001/archinte.160.9.1294. [DOI] [PubMed] [Google Scholar]
- 193.Frank E, Lin J, Kinasewitz G, et al. A multicenter, open-label, randomized comparison of levofloxacin and azithromycin plus ceftriaxone in hospitalized adults with moderate to severe community acquired pneumonia. Clin Ther. 2002;24:1292–308. doi: 10.1016/s0149-2918(02)80034-0. [DOI] [PubMed] [Google Scholar]
- 194.Contopoulos-Ioannidis DG, Ioannidis JP, Chow P, Lau J. Meta-analysis of randomized controlled trials of the comparative efficacy and safety of azithromycin against other antibiotics for lower respiratory tract infections. J Antimicrob Chemother. 2001;48:691–703. doi: 10.1093/jac/48.5.691. [DOI] [PubMed] [Google Scholar]
- 195.Ewig S, Ruiz M, Torres A, et al. Pneumonia acquired in the community through drug-resistant Streptococcus pneumoniae. Am J Respir Crit Care Med. 1999;159:1835–42. doi: 10.1164/ajrccm.159.6.9808049. [DOI] [PubMed] [Google Scholar]
- 196.Van Kerkhoven D, Peetermans WE, Verbist L, Verhaegen J. Breakthrough pneumococcal bacteraemia in patients treated with clarithromycin or oral beta-lactams. J Antimicrob Chemother. 2003;51:691–6. doi: 10.1093/jac/dkg116. [DOI] [PubMed] [Google Scholar]
- 197.Waterer GW, Wunderink RG, Jones CB. Fatal pneumococcal pneumonia attributed to macrolide resistance and azithromycin monotherapy. Chest. 2000;118:1839–40. doi: 10.1378/chest.118.6.1839-a. [DOI] [PubMed] [Google Scholar]
- 198.Brueggeman AB, Phaller MA, Doern GV. Use of penicillin MICs to predict in vitro activity of other β-lactam antimicrobial agents against Streptococcus pneumoniae. J Clin Microbiol. 2001;39:367–9. doi: 10.1128/JCM.39.1.367-369.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Aubier M, Verster R, Regamey C, et al. Once-daily sparfloxacin versus high dosage amoxicillin in the treatment of community-acquired, suspected pneumococcal pneumonia in adults. Clin Infect Dis. 1998;26:1312–20. doi: 10.1086/516366. [DOI] [PubMed] [Google Scholar]
- 200.File TJ, Jr, Tan JS. International guidelines for treatment of community-acquired pneumonia in adults: the role of macrolides. Drugs. 2003;63:181–205. doi: 10.2165/00003495-200363020-00005. [DOI] [PubMed] [Google Scholar]
- 201.Fernandez-Sabe N, Carratala J, Dorca J. Efficacy and safety of sequential amoxicillin-clavulanate in the treatment of anaerobic lung infections. Eur J Clin Microbiol Infect Dis. 2003;22:185–7. doi: 10.1007/s10096-003-0898-2. [DOI] [PubMed] [Google Scholar]
- 202.Roson B, Carratala J, Tuban F, et al. Usefulness of beta-lactam therapy for community-acquired pneumonia in the era of drug resistant Streptococcus pneumoniae a randomized study of amoxicillin-clavulanate and ceftriaxone. Microb Drug Resist. 2001;7:85–96. doi: 10.1089/107662901750152864. [DOI] [PubMed] [Google Scholar]
- 203.Fogarty CM, Cyganowski M, Palo WA, et al. A comparison of cefditoren pivoxil and amoxicillin/clavulanate in the treatment of community-acquired pneumonia: a multicenter, prospective, randomized, investigator-blinded, parallel-group study. Clin Ther. 2002;24:1854–70. doi: 10.1016/s0149-2918(02)80084-4. [DOI] [PubMed] [Google Scholar]
- 204.Van Zyl L, le Roux JG, LaFata JA, et al. Cefditoren pivoxil vs-cefpodoxime proxetil for community-acquired pneumonia: results of a multi-center, prospective, randomized, double blind study. Clin Ther. 2002;24:1840–53. doi: 10.1016/s0149-2918(02)80083-2. [DOI] [PubMed] [Google Scholar]
- 205.Clark CL, Nagai K, Dewasse BE, et al. Activity of cefditoren against respiratory pathogens. J Antimicrob Chemother. 2002;50:33–41. doi: 10.1093/jac/dkf076. [DOI] [PubMed] [Google Scholar]
- 206.Johnson JR. Doxycycline for treatment of community-acquired pneumonia. Clin Infect Dis. 2002;35:632. doi: 10.1086/341983. [DOI] [PubMed] [Google Scholar]
- 207.Malcolm C, Marrie TJ. Antibiotic therapy for ambulatory patients with community-acquired pneumonia in an emergency department setting. Arch Intern Med. 2003;163:797–802. doi: 10.1001/archinte.163.7.797. [DOI] [PubMed] [Google Scholar]
- 208.White RL, Enzweiler KA, Friedrich LV, Wagner D, Hoban D, Bosso JA. Comparative activity of gatifloxacin and other antibiotics against 4009 clinical isolates of Streptococcus pneumoniae in the United States during 1999–2000. Diag Microbiol Infect Dis. 2002;43:207–17. doi: 10.1016/s0732-8893(02)00391-7. [DOI] [PubMed] [Google Scholar]
- 209.Scheld WM. Maintaining fluoroquinolone class efficacy: review of influencing factors. Emerg Infect Dis. 2003;9:1–9. doi: 10.3201/eid0901.020277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Jones RN. Worldwide antimicrobial susceptibility patterns and pharmacodynamic comparisons of gatifloxacin and levofloxacin against Streptococcus pneumoniae: Report from the Antimicrobial Resistance Rate Epidemiology Study Team. Antimicrob Agents Chemother. 2003;47:292–6. doi: 10.1128/AAC.47.1.292-296.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Gotfried M, Quinn TC, Gothelf S, et al. Oral gatifloxacin in outpatient community-acquired pneumonia: results from Tea CES, a community-based, open-label multicenter study. Diagn Microbiol Infect Dis. 2002;44:85–91. doi: 10.1016/s0732-8893(02)00447-9. [DOI] [PubMed] [Google Scholar]
- 212.Marrie TJ, Lau CY, Wheeler SL, et al. A controlled trial of a critical pathway for treating community-acquired pneumonia. JAMA. 2000;283:749–55. doi: 10.1001/jama.283.6.749. [DOI] [PubMed] [Google Scholar]
- 213.Finch R, Schurmann D, Collins O, et al. Randomized controlled trial of sequential intravenous and oral moxifloxacin compared to sequential IV and oral co-amoxiclav with or without clarithromycin in patients with community acquired pneumonia requiring initial parenteral treatment. Antimicrob Agents Chemother. 2002;46:1746–54. doi: 10.1128/AAC.46.6.1746-1754.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.File TM, Jr, Segreti J, Dunbar L, et al. A multicenter, randomized study comparing the efficacy and safety of intravenous and/or oral levofloxacin versus ceftriaxone and/or defuroxime axetil in treatment of adults with community-acquired pneumonia. Antimicrob Agents Chemother. 1997;41:1965–72. doi: 10.1128/aac.41.9.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Salkind AR, Cuddy PG, Foxworth JW. Fluoroquinolone treatment of community-acquired pneumonia: a meta-analysis. Ann Pharmacother. 2002;36:1938–43. doi: 10.1345/aph.1C167. [DOI] [PubMed] [Google Scholar]
- 216.Weiss K, Restieri C, Gauthier R, et al. A nosocomial outbreak of fluoroquinolone-resistant Streptococcus pneumoniae. Clin Infect Dis. 2001;33:517–22. doi: 10.1086/322658. [DOI] [PubMed] [Google Scholar]
- 217.Ho PL, Tse WS, Tsang KW, et al. Risk factors for acquisition of levofloxacin-resistant Streptococcus pneumoniae a case control study. Clin Infect Dis. 2001;32:701–7. doi: 10.1086/319222. [DOI] [PubMed] [Google Scholar]
- 218.Klugman KP. The role of clonality in the global spread of fluoroquinolone-resistant bacteria. Clin Infect Dis. 2003;36:783–5. doi: 10.1086/367935. [DOI] [PubMed] [Google Scholar]
- 219.Quale J, Landman D, Ravishankar J, et al. Streptococcus pneumoniae, Brooklyn, New York: fluoroquinolone resistance at our doorstep. Emerg Infect Dis. 2002;8:594–7. doi: 10.3201/eid0806.010275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Perez-Trallero E, Marimon JM, Gonzalez A, Iglesias L. Spain (14-5) international multiresistant Streptococcus pneumoniae clone resistant to fluoroquinolones and other families of antibiotics. J Antimicrob Chemother. 2003;51:715–9. doi: 10.1093/jac/dkg106. [DOI] [PubMed] [Google Scholar]
- 221.Levison ME, Mangura CT, Lorber B, et al. Clindamycin compared with penicillin for the treatment of anaerobic lung abscess. Ann Intern Med. 1983;98:466–71. doi: 10.7326/0003-4819-98-4-466. [DOI] [PubMed] [Google Scholar]
- 222.Gudiol F, Manressa F, Pallares R, et al. Clindamycin vs-enicillin for anaerobic lung infections: high rate of penicillin failures associated with penicillin-resistant Bacteroides melaninogenicus. Arch Intern Med. 1990;150:2525–9. [PubMed] [Google Scholar]
- 223.Muller MP, Low DE, Green KA, et al. Clinical and epidemiologic features of group A streptococcal pneumonia in Ontario Canada. Arch Intern Med. 2003;163:467. doi: 10.1001/archinte.163.4.467. [DOI] [PubMed] [Google Scholar]
- 224.McCormick AW, Whitney CG, Farley MM, et al. Geographic diversity and temporal trends of antimicrobial resistance in Streptococcus pneumoniae in the United States. Nat Med. 2003;9:390–2. doi: 10.1038/nm839. [DOI] [PubMed] [Google Scholar]
- 225.Niederman MS, Mandell LA, Anzueto A, et al. Guidelines for the management of adults with community-acquired pneumonia: diagnosis, assessment of severity, antimicrobial therapy, and prevention. Am J Respir Crit Care Med. 2001;163:1730–54. doi: 10.1164/ajrccm.163.7.at1010. [DOI] [PubMed] [Google Scholar]
- 226.Yu VL, Chiou CC, Feldman C, et al. An international prospective study of pneumococcal bacteremia correlation with in vitro resistance, antibiotics administered, and clinical outcome. Clin Infect Dis. 2003;37:230–7. doi: 10.1086/377534. [DOI] [PubMed] [Google Scholar]
- 227.Fuchs PC, Barry AL, Brown SD. In vitro activity of telithromycin against Streptococcus pneumoniae resistant to other antimicrobials including cefotaxime. J Antimicrob Chemother. 2002;49:399–401. doi: 10.1093/jac/49.2.399. [DOI] [PubMed] [Google Scholar]
- 228.Fogarty CM, Kohno S, Kuchanan P, et al. Community-acquired respiratory tract infections caused by resistant pneumococci: clinical and bacteriological efficacy of the ketolide telithromycin. J Antimicrob Chemother. 2003;51:947–55. doi: 10.1093/jac/dkg153. [DOI] [PubMed] [Google Scholar]
- 229.Ball P, File TM, Twynholm M, Henkel T. Efficacy and safety of gemifloxacin 320 mg once daily for 7 days in the treatment of adult lower respiratory tract infections. Int J Antimicrob Agents. 2001;18:19–27. doi: 10.1016/s0924-8579(01)00359-4. [DOI] [PubMed] [Google Scholar]
- 230.File TM, Jr, Schelmmer R, Garau J, et al. Efficacy and safety of gemifloxacin in the treatment of community-acquired pneumonia: a randomized, double-blind comparison with trovafloxacin. J Antimicrob Chemother. 2001;48:67–74. doi: 10.1093/jac/48.1.67. [DOI] [PubMed] [Google Scholar]
- 231.Lode H, File TM, Jr, Mandell L, et al. Oral gemifloxacin versus sequential therapy with intravenous ceftriaxone/oral cefuroxime with or without a macrolide in the treatment of patients hospitalized with community-acquired pneumonia: a randomized, open-label, multicenter study of clinical efficacy and tolerability. Clin Ther. 2002;24:1915–36. doi: 10.1016/s0149-2918(02)80088-1. [DOI] [PubMed] [Google Scholar]
- 232.Vetter N, Cambroncro-Hernandez E, Rohlf J, et al. A prospective randomized double-blind multicenter comparison of parenteral ertapenem and ceftriaxone for the treatment of hospitalized adults with community-acquired pneumonia. Clin Ther. 2002;24:1770–85. doi: 10.1016/s0149-2918(02)80078-9. [DOI] [PubMed] [Google Scholar]
- 233.Ortiz-Ruiz G, Caballero-Lopez J, Friedland IR, et al. A study evaluating the efficacy, safety, and tolerability of ertapenem versus ceftriaxone for the treatment of community-acquired pneumonia in adults. Clin Infect Dis. 2002;34:1076–83. doi: 10.1086/339543. [DOI] [PubMed] [Google Scholar]
- 234.Moellering RC. Linezolid: the first oxazolidinones antimicrobial. Ann Intern Med. 2003;138:135–42. doi: 10.7326/0003-4819-138-2-200301210-00015. [DOI] [PubMed] [Google Scholar]
- 235.San Pedro GS, Cammarata SK, Oliphant TH, et al. Linezolid vs-ceftriaxone/cefpodoxime in patients hospitalized for the treatment of Streptococcus pneumoniae pneumonia. Scand J Infect Dis. 2002;34:720–8. doi: 10.1080/0036554021000026933. [DOI] [PubMed] [Google Scholar]


