Abstract
In December 2019, an outbreak of coronavirus disease 2019 (COVID-19) was identified in Wuhan, China. The World Health Organization (WHO) declared this outbreak a significant threat to international health. COVID-19 is highly infectious and can lead to fatal comorbidities especially acute respiratory distress syndrome (ARDS). Thus, fully understanding the characteristics of COVID-19-related ARDS is conducive to early identification and precise treatment. We aimed to describe the characteristics of COVID-19-related ARDS and to elucidate the differences from ARDS caused by other factors. COVID-19 mainly affected the respiratory system with minor damage to other organs. Injury to the alveolar epithelial cells was the main cause of COVID-19-related ARDS, and endothelial cells were less damaged with therefore less exudation. The clinical manifestations were relatively mild in some COVID-19 patients, which was inconsistent with the severity of laboratory and imaging findings. The onset time of COVID-19-related ARDS was 8–12 days, which was inconsistent with ARDS Berlin criteria, which defined a 1-week onset limit. Some of these patients might have a relatively normal lung compliance. The severity was redefined into three stages according to its specificity: mild, mild-moderate, and moderate-severe. HFNO can be safe in COVID-19-related ARDS patients, even in some moderate-severe patients. The more likely cause of death is severe respiratory failure. Thus, the timing of invasive mechanical ventilation is very important. The effects of corticosteroids in COVID-19-related ARDS patients were uncertain. We hope to help improve the prognosis of severe cases and reduce the mortality.
Keywords: Coronavirus, COVID-19, Acute respiratory distress syndrome, Berlin criteria
Introduction
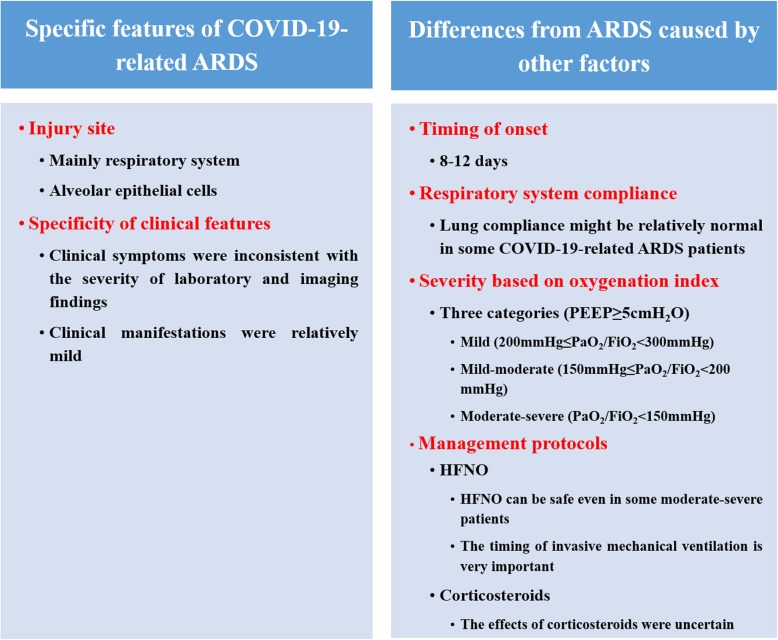
In December 2019, an outbreak of coronavirus disease 2019 (COVID-19), which was caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), broke out in Wuhan, China [1–3]. The World Health Organization (WHO) declared it a significant threat to international health [4]. COVID-19 was of clustering onset and mainly affected the respiratory system with some patients rapidly progressing to acute respiratory distress syndrome (ARDS); other organ functions were less involved [5, 6]. These patients were likely to be admitted to the intensive care unit (ICU) and might die. The elderly and those with comorbidities are at highest risk of death. The death appeared to be related to ARDS [7]. Although several studies have reported the clinical features of COVID-19 [1, 8–13], our understanding about it remains limited [14]. Can we consider all the cases of acute respiratory failure associated with COVID-19 as ARDS? The answer is probably no. Based on current reports and our experience in the management of COVID-19-related ARDS patients, we realized that there are many differences between COVID-19-related ARDS and ARDS caused by other factors as defined by Berlin criteria, and therefore differences in treatment. Thus, we aimed to describe the characteristics of COVID-19-related ARDS and to elucidate the differences (Fig. 1).
Fig. 1.
Summary of characteristics of COVID-19-related ARDS
Specific features of COVID-19-related ARDS
Injury site of COVID-19
ARDS occurs as a result of an acute systemic inflammatory response, which can be caused by insults to the lung, either direct or indirect. The early exudative stage presents diffuse alveolar damage with destruction of epithelial and endothelial cells. COVID-19 mainly affected the respiratory system with minor damage to other organs. Studies reported that acute myocardial injury (7.2–17%) and acute renal injury (2.9–15%) could occur in severe patients. The reported incidence of ARDS was 15.6–31%, higher than that of other organ injuries [1, 8–11].
The most common respiratory symptom of COVID-19 is dry cough (59.4–82%) [1, 8–11]. Sputum production was less. It suggested that injury to the alveolar epithelial cells was the main cause of COVID-19-related ARDS, and endothelial cells were less damaged with therefore less exudation. Endothelial cells line the inner surface of blood vessels in all organs. It was possible that due to less damage to the endothelial cells, other organ functions were less involved in COVID-19 patients.
Specificity of clinical features
The respiratory system was mainly involved in COVID-19 patients as mentioned above. Some patients had a low oxygenation index, indicating severe respiratory failure. Chest imaging findings suggested the involvement of both lungs. Chest computed tomography (CT) scans usually showed multifocal bilateral patchy shadows and/or ground-glass opacities; some patients showed a mixed pattern of ground-glass opacities and consolidation [15]. The CT results indicated diffuse and severe lung injury. However, the clinical manifestations were relatively mild in some patients. These patients might have no complaint of dyspnea, no significant increase in respiratory rate, and no respiratory distress. Hemodynamics and indexes of tissue perfusion such as lactate were also relatively stable. The clinical symptoms were inconsistent with the severity of laboratory and imaging findings. However, these patients may deteriorate rapidly and need to be monitored closely. Blood carbon dioxide levels may be a meaningful indicator for invasive mechanical ventilation.
Differences from ARDS caused by other factors
Timing of onset
The ARDS Berlin criteria defined that for a patient to be diagnosed as having ARDS, the onset must be within 1 week of a known clinical insult or new or worsening respiratory symptoms [16]. The reported onset of COVID-19-related ARDS was similar in different studies. Huang et al. [1] first reported 41 cases of COVID-19 in which the median time from onset of symptoms to ARDS was 9.0 days (8.0–14.0). Subsequently, Wang et al. [9] reported 138 cases of COVID-19 in which the median time from the first symptom to ARDS was 8.0 days (6.0–12.0). Zhou et al. [11] reported the median time from illness onset to ARDS was 12.0 days (8.0–15.0). Studies by Chen et al. [8] and Guan et al. [10] did not report the onset of ARDS. As the onset time of COVID-19-related ARDS was 8–12 days, it suggested that the 1-week onset limit defined by ARDS Berlin criteria did not apply to COVID-19-related ARDS. It reminded us to pay more attention to the development of ARDS in patients with the course of more than a week, so as to treat timely.
Respiratory system compliance
Not all the cases of acute respiratory failure caused by COVID-19 were ARDS. The typical CT findings of COVID-19 showed bilateral ground-glass shadow with a peripheral lung distribution [15]. Although there was consolidation and exudation, it was not a “typical” ARDS image. ARDS is a condition associated with many disease processes, resulting in reduced lung compliance and severe hypoxemia [16]. Lung compliance might be relatively normal in some COVID-19-related ARDS patients who met ARDS Berlin criteria. This was obviously inconsistent with ARDS caused by other factors. In addition, the lung compliance was relatively high in some COVID-19-related ARDS patients, which was inconsistent with the severity of hypoxemia.
Severity based on oxygenation index
According to the Berlin definition, ARDS is divided into three stages based on oxygenation index (PaO2/FiO2) on positive end-expiratory pressure (PEEP) ≥ 5 cmH2O: mild (200 mmHg < PaO2/FiO2 ≤ 300 mmHg), moderate (100 mmHg < PaO2/FiO2 ≤ 200 mmHg), and severe (PaO2/FiO2 ≤ 100 mmHg) [16]. ARDS classification determines both the severity of the disease and choice of treatment protocol. A previous study reported that more than 50% of patients with moderate and severe ARDS according to the Berlin definition did not show diffuse alveolar damage [17]. In fact, the specific ranges under which the hypoxemia is evaluated differ among clinicians. From the perspective of therapy, the intensity of adjunctive treatment varies according to the degree of hypoxemia. Thus, we need a more suitable classification of ARDS severity that can accurately identify patients for a specified therapy.
Till now, the clinical features of COVID-19-related ARDS are still unclear. There are no specific monitoring and implementation protocols. Under the current situation, a number of designated hospitals and dozens of medical teams from different provinces have participated in the treatment. The unified ARDS treatment standard is needed in order to improve the uniformity and thus reduce the mortality. Therefore, experts from the national health commission of China developed a standard treatment protocol for COVID-19 based on their experience. COVID-19-related ARDS was divided into three categories based on oxygenation index (PaO2/FiO2) on PEEP ≥ 5 cmH2O: mild (200 mmHg ≤ PaO2/FiO2 < 300 mmHg), mild-moderate (150 mmHg ≤ PaO2/FiO2 < 200 mmHg), and moderate-severe (PaO2/FiO2 < 150 mmHg) [18]. The new stratification for COVID-19-related ARDS determines personalized treatment for different patients. In fact, a number of ARDS treatments, including prone positioning and neuromuscular blockers, are recommended for patients with PaO2/FiO2 less than 150 mmHg. This indicates that Berlin classification is not suitable to define the severity of ARDS and accurately guide the corresponding treatments.
Management protocols
High-flow nasal oxygen in COVID-19-related ARDS patients
Hypoxemic respiratory failure in ARDS generally results from intrapulmonary ventilation-perfusion mismatch or shunt and usually requires mechanical ventilation. Compared to standard oxygen therapy, high-flow nasal oxygen (HFNO) reduces the need for endotracheal intubation in ARDS patients [19]. WHO recommended that HFNO should only be used in selected patients with hypoxemic respiratory failure [20]. Studies indicated that HFNO is more suitable for patients with mild ARDS. However, according to clinical situations, HFNO can be safe in both mild and mild-moderate COVID-related ARDS patients, and even some moderate-severe patients. Some patients with an oxygenation index of 100 mmHg can remain relatively stable with the support of HFNO. This is clearly inconsistent with the stratified treatment strategies of ARDS caused by other factors.
Although COVID-19 may be associated with myocardial injury and arrhythmia as reported [8, 9, 11], there is currently no evidence that myocarditis is the cause of death. The respiratory system is the most commonly involved for COVID-19, and some cases can rapidly progress to ARDS, which requires venous-venous extracorporeal membrane oxygenation (V-V ECMO) in the most severe cases. To date, no patients with severe arrhythmia or acute heart failure due to acute myocarditis have been reported to require venous-arterial ECMO (V-A ECMO) treatment. Cardiac injury was diagnosed by elevation of cardiac biomarkers in serum or new abnormalities in electrocardiography and echocardiography. However, serum lactate dehydrogenase (LDH) and creatine kinase-MB were elevated more commonly than hypersensitive troponin I in COVID-19 patients as reported [1, 9]. The pathological findings of a COVID-19-related ARDS patient by Xu et al. [21] indicated that there were no obvious histological changes seen in heart tissue. Therefore, the diagnosis of acute myocardial injury needs further consideration. Thus, the more likely cause of death is severe respiratory failure. Therefore, the timing of invasive mechanical ventilation is very important. Since severe COVID-19 patients may deteriorate rapidly, patients receiving HFNO should be closely monitored and cared for by experienced personnel capable of endotracheal intubation at any time. Currently published studies did not report the proportion of different respiratory support according to COVID-19-related ARDS classification. Further research is expected to provide more evidence for the use of HFNO in COVID-19-related ARDS patients.
Corticosteroids in COVID-19-related ARDS patients
Corticosteroids are considered a potential treatment for ARDS because of their role in reducing inflammation and fibrosis. Although the use of corticosteroids in ARDS patients remains controversial, treatment with corticosteroids is currently the only pharmacological intervention that may reduce morbidity and mortality. It is reported that treatment with high-dose corticosteroids for a prolonged period of time could accelerate the improvement of ARDS [22]. Furthermore, methylprednisolone shortened periods of need for invasive mechanical ventilation and lowered mortality in ARDS patients [23]. However, WHO recommended that systemic corticosteroids should not be routinely administered to COVID-19 or COVID-19-related ARDS patients [20]. Studies reported that less than half of the COVID-19 patients were given systemic corticosteroids, mostly in severely ill patients with ARDS [1, 9, 11]. Low-to-moderate dosage was administered depending on the severity of the disease, for as short time of treatment as possible. Although it has been reported that treatment with methylprednisolone may be beneficial for COVID-19-related ARDS patients [24], the effect of corticosteroids in such patients is still uncertain and needs to be further evaluated. In particular, the use of corticosteroids may affect virus clearance in COVID-19 patients. Further evidence is needed to evaluate the role of systemic corticosteroid therapy and its impact on long-term prognosis in this group of patients.
Conclusion
COVID-19 is highly infectious and can lead to fatal comorbidities especially ARDS. There are currently no recommended specific anti-COVID-19 treatments, so supportive treatment is important. Fully understanding the characteristics of COVID-19-related ARDS is conducive to early identification and precise treatment. We hope to help improve the prognosis of severe cases and reduce the mortality.
Acknowledgements
None.
Abbreviations
- ARDS
Acute respiratory distress syndrome
- COVID-19
Coronavirus disease 2019
- CT
Computed tomography
- HFNO
High-flow nasal oxygen
- ICU
Intensive care unit
- LDH
Lactate dehydrogenase
- PEEP
Positive end-expiratory pressure
- SARS-CoV-2
Severe acute respiratory syndrome coronavirus 2
- V-A ECMO
Venous-arterial extracorporeal membrane oxygenation
- V-V ECMO
Venous-venous extracorporeal membrane oxygenation
- WHO
World Health Organization
Authors’ contributions
Both authors had their substantial contributions to the conception or design of the work or the acquisition and interpretation of data. XL drafted the work. XCM revised it critically for important intellectual content. Both authors read and approved the final manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (81671936).
Availability of data and materials
Not applicable.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Huang C, Wang Y, Li X, et al. Clinical features of patients with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lu R, Zhao X, Li J, et al. Genomic characterization and epidemiology of 2019 novel coronavirus: implications of virus origins and receptor binding. Lancet. 2020;395(10224):565–574. doi: 10.1016/S0140-6736(20)30251-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.World Health Organization. Coronavirus disease (COVID-19) outbreak. https://www.who.int.
- 5.Chan JF-W, Yuan S, Kok K-H, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet. 2020;S0140-6736(20):30154–30159. doi: 10.1016/S0140-6736(20)30154-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Phan LT, Nguyen TV, Luong QC, et al. Importation and human-to-human transmission of a novel coronavirus in Vietnam. N Engl J Med. 2020;382(9):872–874. doi: 10.1056/NEJMc2001272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Burki TK. Coronavirus in China. Lancet Respir Med. 2020;8(3):238. doi: 10.1016/S2213-2600(20)30056-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020. 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed]
- 10.Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020. 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed]
- 11.Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020. 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed]
- 12.Xu XW, Wu XX, Jiang XG, et al. Clinical findings in a group of patients infected with the 2019 novel coronavirus (SARS-Cov-2) outside of Wuhan, China: retrospective case series. BMJ. 2020. 10.1136/bmj.m606. [DOI] [PMC free article] [PubMed]
- 13.Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020. 10.1016/S2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed]
- 14.Wu ZY, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020. 10.1001/jama.2020.2648. [DOI] [PubMed]
- 15.Chung M, Bernheim A, Mei XY, et al. CT imaging features of 2019 novel coronavirus (2019-nCoV) Radiology. 2020;295(1):202–207. doi: 10.1148/radiol.2020200230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Definition Task Force ARDS, Ranieri VM, Rubenfeld GD, et al. Acute respiratory distress syndrome: the Berlin definition. JAMA. 2012;307:2526–2533. doi: 10.1001/jama.2012.5669. [DOI] [PubMed] [Google Scholar]
- 17.Thille AW, Esteban A, Fernández-Segoviano P, et al. Comparison of the Berlin definition for acute respiratory distress syndrome with autopsy. Am J Respir Crit Care Med. 2013;187:761–767. doi: 10.1164/rccm.201211-1981OC. [DOI] [PubMed] [Google Scholar]
- 18.Zheng R, Hu M, Li R, et al. Respiratory treatment procedures in patients with severe novel coronavirus infected pneumonia: an expert opinion. Chin J Crit Care Intensive Care Med. 2020. 10.3877/cma.j.issn.2096-1537.2020.0004.
- 19.Ou X, Hua Y, Liu J, et al. Effect of high-flow nasal cannula oxygen therapy in adults with acute hypoxemic respiratory failure: a meta-analysis of randomized controlled trials. CMAJ. 2017;189:E260–E2E7. doi: 10.1503/cmaj.160570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.World Health Organization. (2020) . Clinical management of severe acute respiratory infection when novel coronavirus (2019-nCoV) infection is suspected: interim guidance, 28 January 2020. World Health Organization. https://apps.who.int/iris/handle/10665/330893.
- 21.Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020. 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed]
- 22.Meduri GU, Bridges L, Shih M-C, et al. Prolonged glucocorticoid treatment is associated with improved ARDS outcomes: analysis of individual patients’ data from four randomized trials and trial-level meta-analysis of the updated literature. Intensive Care Med. 2016;42:829–840. doi: 10.1007/s00134-015-4095-4. [DOI] [PubMed] [Google Scholar]
- 23.Meduri GU, Siemieniuk RAC, Ness RA, et al. Prolonged low-dose methylprednisolone treatment is highly effective in reducing duration of mechanical ventilation and mortality in patients with ARDS. J Intensive Care. 2018;6:53. doi: 10.1186/s40560-018-0321-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020. 10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.