Abstract
Background
The aim of current study was to (1) construct and validate a novel hepatocellular carcinoma (HCC)-specific inflammatory index; (2) compare the performances of the Integrated Liver Inflammatory Score (ILIS) to existing 4 inflammatory indices in HCC; (3) explore the association between the inflammatory indices and systemic/intratumoral inflammatory markers.
Methods
Two cohorts from Hong Kong (HK; n = 1,315) and Newcastle (n = 574) were studied. A novel index was constructed from the HK training set (n = 627). The index was constructed from the training set by combing independent prognostic circulating parameters, followed by validating in the validation set of HK cohort (n = 688) and the Newcastle cohort. Its prognostic performance was compared to 4 inflammatory indices, namely, the neutrophil to lymphocyte ratio, platelet-to-lymphocyte ratio, prognostic nutrition index, and systemic immune-inflammation index, were compared in the HK cohort. Circulating cytokines and intratumoral gene expression were analyzed in a subset of patients with available samples and correlated with the inflammatory indices.
Results
In the training set of the HK cohort, the ILIS, was generated: −0.057 × albumin (g/L) + 0.978 × log (Bilirubin, µmol/L) + 1.341 × log (alkaline phosphatase, IU/L) + 0.086 × Neutrophil (10<sup>9</sup>/L) + 0.301 × log (alpha-fetoprotein, µg/L). With cutoff of 2.60 and 3.87, the ILIS could categorize patients into 3 risk groups in the both validation cohorts. ILIS outperforms other inflammatory indices and remains an independent prognosticator for overall survival after adjustment with Barcelona Clinic Liver Cancer (hazard ratio 31.90, p < 0.001). The ILIS had the best prognostic performances as compared to other inflammatory indices. In exploratory analyses, the ILIS correlated with circulating inflammatory cytokines (e.g., IL-8) but not with any intratumoral inflammatory gene expression.
Conclusions
ILIS is an HCC-specific prognostic index built on 5 readily available blood parameters. Its versatility is validated both Eastern and Western population of HCC. The score is correlated with levels of circulating cytokines.
Keywords: Inflammation, Liver cancer, Prognosis, Tumor markers
Introduction
The presence of tumor-associated inflammation represents enabling tumor characteristics, which promote tumorigenesis and progression [1]. Circulating inflammatory cytokines and markers could reflect the underlying systemic inflammation and prognoses for hepatocellular carcinoma (HCC) [2, 3]. A number of inflammation-based indexes (IBI) were derived from peripheral blood counts (e.g., neutrophil, lymphocyte, and platelet) and acute-phase proteins (e.g., C-reactive protein [CRP] and albumin) for prognostic purposes, with examples including IBI, neutrophil to lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), prognostic nutrition index (PNI), systemic immune-inflammation index (SII) [3, 4, 5, 6].
In HCC, NLR is the most widely studied IBI [7, 8], and is shown to be predictive of sorafenib response in an exploratory pooled analysis of a randomized clinical trial [9]. Clinical application of inflammation-based indices in HCC is limited by 2 factors. First, most of the previous studies have sample size in a scale of few 100 patients, which are statistically inadequate for studying a robust number of prognostic parameters and comparing different indices in patients with HCC [8, 10]. Second, except the PNI and IBI, inflammatory indices were not specifically developed for HCC [3, 4, 6], and as a result, HCC-specific prognosticators such as alpha-fetoprotein (AFP) and alkaline phosphatase (ALP), were not incorporated in the indices [11, 12, 13, 14].
To address the above 2 limitations, there is a need to develop an HCC-specific prognostic index from a robust sample of patients. Therefore, we conducted a study with following 3 objectives: the first objective was to derive and validate a novel HCC-specific IBI from a robust sample of patients with HCC. The second objective was to compare the prognostic performance of this novel index to existing inflammation-based prognostic indices. The third objective was to explore the potential clinical application of the novel index on aspects of therapeutic implications and association with tumor-related inflammation.
Patients and Methods
Patients
Two patient cohorts were included in the study. The first cohort consisted of HCC patients treated at the Prince of Wales Hospital, Hong Kong between January, 2001 and December, 2014 [2, 4]. The second cohort was composed of 583 HCC patients managed by the Newcastle HCC multidisciplinary team between the years 2000 and 2010, studying data collected as part of a Newcastle upon Tyne Hospitals Foundation Trust registered audit project [15]. The Hong Kong Cohort was used to comprehensively analyze the prognostic performance of existing inflammatory indices. It was later randomly split into half − into training and internal validation cohorts for constructing and testing of the novel HCC-specific inflammatory score. The Newcastle cohort was used as the independent external validation cohort to examine the novel HCC-specific inflammatory score.
All clinical and laboratory parameters were collected and reviewed from patients' records. Patients with incomplete data were excluded. Albumin-Bilirubin (ALBI) score, Platelet-ALBI, NLR, PLR, PNI and SII were calculated and categorized in accordance with original publications [4, 5, 16, 17]. Patients in Hong Kong cohort were staged according to the 7th edition of American Joint Committee on Cancer (AJCC) [18], Barcelona Clinic Liver Cancer (BCLC) [19], Chinese University Prognostic Index [12], Japan Integrated Staging [20], and Okuda systems [21]. Overall survival (OS) was defined as the date of first clinical diagnosis of HCC to the time of death, or last follow-up if death had not occurred.
Analysis of Cytokines in Circulation and Gene Expression in the Tumor
The serum cytokine concentrations of serum inflammatory markers were determined by the Human Cytokine Array 41-plex and Human Neurodegenerative Disease 1-plex assays (Eve Technologies, Alberta, Canada), with detailed methodologies described previously [22]. The gene expression in the tumor was analyzed by the NanoString analysis. The archival formalin-fixed paraffin-embedded tumor tissue was retrieved and manually macrodissected. Total mRNA was isolated from the macrodissected tumor tissues using Qiagen miRNeasy Kit (Qiagen, Valencia, CA, USA) according to the manufacturer's instructions. The RNA sample was quantified by NanoDrop (Thermo Scientific, Wilmington, DE, USA), and regarded as an adequate sample if it contained 400 ng at minimum. The sample was subsequently analyzed by the nCounter PanCancer Immune Profiling Panel (NanoString, Seattle, WA, USA) according to the manufacturer's instructions [23].
Statistical Analyses
All statistical analyses were done by R version 3.32 (R Foundation for Statistical Computing, Vienna, Austria). To construct a novel IBI, multivariable Cox regression model with stepwise forward selection together with bootstrap resampling was constructed (R package bootStepAIC 1.20) [24]. Only clinical parameters with consistent selection in 200 bootstrap samples and demonstration of same direction of hazard ratio (HR) (i.e., positive vs. negative) were used to construct the novel index. The novel score was the weighted sum of those selected parameters, of which the weights were β-coefficients from the multivariable Cox regression analysis. Three risk groups (low, intermediate, and high) were generated by employing the 50th and 85th centile of the score as cutoff values [25].
Prognostic performances of different indices were examined by survival analyses using Kaplan-Meier method and Cox proportional hazards model. The comparison of prognostic performance was evaluated by homogeneity, discriminatory ability, and monotonicity of gradients [11]. Homogeneity and monotonicity of gradients were assessed by the likelihood ratio chi-square test. A system with better homogeneity and monotonicity is indicated by a larger value of the likelihood ratio chi-square test. Discriminatory ability was evaluated by the corrected Akaike information criterion (AICc). A system with higher discriminatory ability was signified by a smaller value of AICc.
Raw data of the NanoString analysis were analyzed by nSolver 3.0 (NanoString, Seattle, WA, USA). Raw count values were first subtracted by a background count level (2 SDs above the mean count of internal negative controls). Data were then normalized by geometric mean of internal positive controls. p values of multiple comparisons among circulatory inflammatory indices, serum cytokines and gene expressions by NanoString were adjusted by controlling false discovery rate [26].
Results
Construction of an Integrated Liver Inflammatory Score
The baseline characteristics of patients in the Hong Kong cohort are summarized in Table 1. To build a blood-based and HCC-specific prognostic score integrated with an inflammatory component, the Hong Kong Cohort was randomly divided into training and internal validation cohorts. Both cohorts had similar clinical characteristics (online suppl. Table 1; for all online suppl. material, see www.karger.com/doi/10.1159/000504252) and OS (online suppl. Fig. 1d). In the training cohort (n = 657), blood parameters including liver biochemistry (albumin, bilirubin, alanine transaminase and ALP), INR, peripheral blood counts (neutrophil, lymphocyte and platelet) and AFP were significant prognosticators by the univariate Cox regression (Table 2). Only albumin, bilirubin, ALP, neutrophil, and AFP were selected as stable independent prognostic factors by forward selection along with bootstrap resampling.
Table 1.
Characteristics of Hong Kong cohort
| Hong Kong cohort (n = 1,315), n (%) | NLR, n (%) |
p value | PLR, n (%) |
p value | PNI, n (%) |
p value | SII, n (%) |
p value | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| <5(n = 1,043) | ≥5(n = 272) | ≤150 (n = 826) | ≥150 (n = 489) | ≥45(n = 717) | <45(n = 598) | <330 (n = 457) | ≥330 (n = 860) | ||||||
| Patient factors | |||||||||||||
| Gender, male | 1,140 (86.7) | 902 (86.5) | 238 (87.5) | 0.660 | 713 (86.3) | 427 (87.3) | 0.605 | 626 (87.3) | 514 (86.0) | 0.471 | 385 (84.4) | 755 (87.9) | 0.078 |
| Age, years, mean ± SD | 59.2±11.2 | 59.2±11.2 | 59.2±11.5 | 0.988 | 59.7±10.5 | 58.4±12.4 | 0.045 | 58.1±10.9 | 60.6±11.4 | <0.001 | 60.3±10.2 | 58.7±11.7 | 0.012 |
| Etiology | |||||||||||||
| Hepatitis B | 1,060 (80.6) | 843 (80.8) | 217 (79.8) | 0.698 | 679 (82.2) | 381 (77.9) | 0.057 | 577 (80.5) | 483 (80.8) | 0.893 | 373 (81.8) | 687 (80.0) | 0.427 |
| Hepatitis C | 86 (6.5) | 78 (7.5) | 8 (2.9) | 0.007 | 72 (8.7) | 14 (2.9) | <0.001 | 45 (6.3) | 41 (6.9) | 0.672 | 49 (10.7) | 37 (4.3) | <0.001 |
| Hepatitis B & C | 7 (0.5) | 4 (0.4) | 3 (1.1) | 0.159 | 3 (0.4) | 4 (0.8) | 0.435 | 1 (0.1) | 6 (1.0) | 0.052 | 3 (0.7) | 4 (0.5) | 0.699 |
| Non-B/Non-C | 162 (12.3) | 118 (11.3) | 44 (16.2) | 0.030 | 72 (8.7) | 90 (18.4) | <0.001 | 94 (13.1) | 68 (11.4) | 0.339 | 31 (6.8) | 131 (15.3) | <0.001 |
| Child-Pugh grade | 0.036 | ||||||||||||
| A | 1,097 (83.4) | 937 (89.8) | 160 (58.8) | 729 (88.3) | 368 (75.3) | 708 (98.7) | 389 (65.1) | 396 (86.8) | 701 (81.6) | ||||
| B | 197 (15.0) | 99 (9.5) | 98 (36.0) | 89 (10.8) | 108 (22.1) | 9 (1.3) | 188 (31.4) | 56 (12.3) | 141 (16.4) | ||||
| C | 21 (1.6) | 7 (0.7) | 14 (5.1) | 8 (1.0) | 13 (2.7) | 0(0) | 21 (3.5) | 4(0.9) | 17 (2.0) | ||||
| ALBI grade | <0.001 | <0.001 | <0.001 | 0.257 | |||||||||
| 1 | 622 (47.3) | 574 (55.0) | 48 (17.6) | 442 (53.5) | 180 (36.8) | 569 (79.4) | 389 (65.1) | 229 (50.2) | 393 (45.8) | ||||
| 2 | 619 (47.1) | 437 (41.9) | 182 (66.9) | 345 (41.8) | 274 (56.0) | 148 (20.6) | 188 (31.4) | 205 (45.0) | 414 (48.2) | ||||
| 3 | 74 (5.6) | 32 (3.1) | 42 (15.4) | 39 (4.7) | 35 (7.2) | 0(0) | 21 (3.5) | 22 (4.8) | 52 (6.1) | ||||
| Performance status >0 | 337 (25.6) | 195 (18.7) | 142 (52.2) | <0.001 | 128 (15.5) | 209 (42.7) | <0.001 | 105 (14.6) | 232 (38.8) | <0.001 | 50 (11.0) | 287 (33.4) | <0.001 |
| Tumor characteristics | |||||||||||||
| Tumor size, mm, | |||||||||||||
| median (IQR) | 51 (30–95) | 45 (27–80) | 90 (48–136) | <0.001 | 40 (25–68) | 92 (50–130) | <0.001 | 43 (26–80) | 65 (35–110) | <0.001 | 35 (25–52) | 70 (35–112) | <0.001 |
| Multiple tumors | 585 (44.5) | 422 (40.4) | 163 (59.7) | <0.001 | 313 (37.9) | 272 (55.6) | <0.001 | 266 (37.1) | 319 (53.3) | <0.001 | 176 (38.6) | 409 (47.6) | 0.002 |
| Vascular invasion | 263 (20.0) | 156 (14.9) | 108 (39.6) | <0.001 | 112 (13.6) | 151 (30.9) | <0.001 | 95 (13.2) | 168 (28.1) | <0.001 | 49 (10.7) | 214 (24.9) | <0.001 |
| Extrahepatic spread | 159 (12.1) | 78 (7.5) | 81 (29.8) | <0.001 | 50 (6.1) | 109 (22.3) | <0.001 | 48 (6.7) | 111 (18.6) | <0.001 | 17 (3.7) | 142 (16.5) | <0.001 |
| BCLC stage | <0.001 | <0.001 | <0.001 | ||||||||||
| 0 | 73 (5.6) | 66 (6.3) | 7 (2.6) | 61 (7.4) | 12 (2.5) | 51 (7.1) | 22 (3.7) | 39 (8.6) | 34 (4.0) | ||||
| A | 533 (40.5) | 480 (46.0) | 54 (19.8) | 404 (48.9) | 129 (26.4) | 361 (50.3) | 172 (28.8) | 228 (50.0) | 305 (35.5) | ||||
| B | 237 (18.0) | 197 (18.9) | 40 (14.7) | 150 (18.2) | 87 (17.8) | 132 (18.4) | 105 (17.6) | 95 (20.8) | 142 (16.5) | ||||
| C | 425 (32.3) | 284 (27.2) | 142 (52.0) | 194 (23.5) | 231 (47.2) | 172 (24.0) | 253 (42.3) | 87 (19.1) | 338 (39.3) | ||||
| D | 47 (3.6) | 17 (1.6) | 30 (11.0) | 17 (2.1) | 30 (6.1) | 4 (0.6) | 67 (11.2) | 7 (1.5) | 40 (4.7) | ||||
| Serum AFP, µg/L, median (IQR) | 90 (8–1,860) | 60 (7–772) | 898 (39–23,470) | <0.001 | 48 (8–484) | 429 (13–16,730) | <0.001 | 47 (6–748) 178 (15–4,318) | <0.001 | 38 (9–343) 169 (8–4,385) | <0.001 | ||
| Treatment modality | <0.001 | <0.001 | <0.001 | <0.001 | |||||||||
| Surgical resection | 538 (40.9) | 485 (46.5) | 55 (20.1) | 392 (47.5) | 146 (29.9) | 388 (54.1) | 150 (25.1) | 198 (43.4) | 340 (39.6) | ||||
| Local ablative therapy | 128 (9.7) | 114 (10.9) | 14 (5.1) | 115 (13.9) | 13 (2.7) | 79 (11.0) | 49 (8.2) | 83 (18.2) | 45 (5.2) | ||||
| Transarterial therapy | 318 (24.2) | 256 (24.5) | 62 (22.7) | 195 (23.6) | 123 (25.2) | 142 (19.8) | 176 (29.4) | 124 (27.2) | 194 (22.6) | ||||
| Systemic agent | 122 (9.3) | 80 (7.7) | 42 (15.4) | 39 (4.7) | 83 (17.0) | 60 (8.4) | 62 (10.4) | 14 (3.1) | 108 (12.6) | ||||
| Best supportive care | 209 (15.9) | 109 (10.4) | 100 (36.6) | 85 (10.3) | 124 (25.4) | 48 (6.7) | 161 (26.9) | 37 (8.1) | 172 (20.0) | ||||
AFP, alpha-fetoprotein; ALBI, albumin-bilirubin score; BCLC, Barcelona Clinic Liver Cancer; IQR, interquartile range; NLR, neutrophil-lymphocyte ratio; PLR, platelet-lymphocyte ratio; PNI, prognostic nutritional index; SII, systemic immune-inflammation index.
Table 2.
Cox regression models of laboratory parameters in the training cohort
| Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|
| ß-coefficient | HR | p value | ß-coefficient | HR | p value | |
| Albumin | -0.103 | 0.90 | <2×10−16 | -0.057 | 0.95 | 9.5×10−7 |
| Bilirubin | 0.010 | 1.01 | <2×10−16 | |||
| log (Bilirubin) | 2.115 | 8.29 | <2×10−16 | 0.978 | 2.66 | 4.6×10−8 |
| ALT | 0.004 | 1.00 | 5.8×10−8 | |||
| log (ALT) | 0.931 | 2.54 | 4.5×10−7 | |||
| ALP | 0.003 | 1.00 | <2×10−16 | |||
| log (ALP) | 2.637 | 13.97 | <2×10−16 | 1.341 | 3.82 | 6.1×10−8 |
| INR | 1.501 | 4.49 | 6.7×10−15 | |||
| Neutrophil count | 0.161 | 1.18 | <2×10−16 | 0.086 | 1.09 | 2.7×10−5 |
| Lymphocyte count | -0.674 | 0.51 | 3.7×10−12 | |||
| Platelet count | 0.002 | 1.00 | 7.5×10−6 | |||
| log (platelet count) | 0.716 | 2.05 | 0.004 | |||
| log (AFP) | 0.382 | 1.47 | <2×10−16 | 0.301 | 1.35 | 1.5×10−15 |
AFP, alpha-fetoprotein; ALP, alkaline phosphatase; ALT, alanine aminotransferase; HR, hazard ratio.
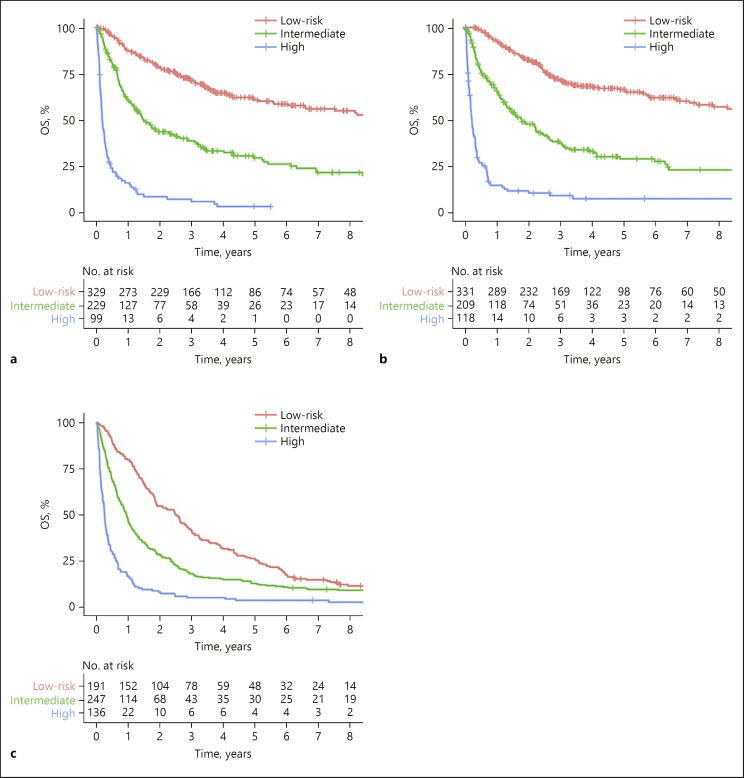
A prognostic model, Integrated Liver Inflammatory Score (ILIS), was generated: −0.057 × albumin (g/L) + 0.978 × log (Bilirubin, µmol/L) + 1.341 × log (ALP, IU/L) + 0.086 × Neutrophil (109/L) + 0.301 × log (AFP, µg/L). Using 50% (2.60) and 85% (3.87) of ILIS as cutoff values, 3 prognostically distinct groups were classified (Fig. 1a).
Fig. 1.
Kaplan-Meier survival plots comparing OS stratified by ILIS in the (a) training cohort (log rank p < 0.001), (b) internal validation cohort (log rank p < 0.001) and (c) external validation cohort (log rank p < 0.001). OS, overall survival.
Validation of the ILIS
Using aforementioned cutoff values, namely, 2.60 and 3.87, the ILIS could categorize patients into 3 different risk groups in both internal validation cohort of Hong Kong (n = 658; Fig. 1b). In the Newcastle cohort, among the 583 patients, 574 of them had all 5 parameters required by the ILIS (i.e., 9 patients were excluded). Similarly, the ILIS was validated in this external validation cohort (Fig. 1c). When compared to other inflammatory indices, ILIS outperformed other inflammatory indices, liver function assessment tools, serum AFP, and selected staging systems (the AJCC, Chinese University Prognostic Index, Japan Integrated Staging, and Okuda system; online suppl. Table 2).
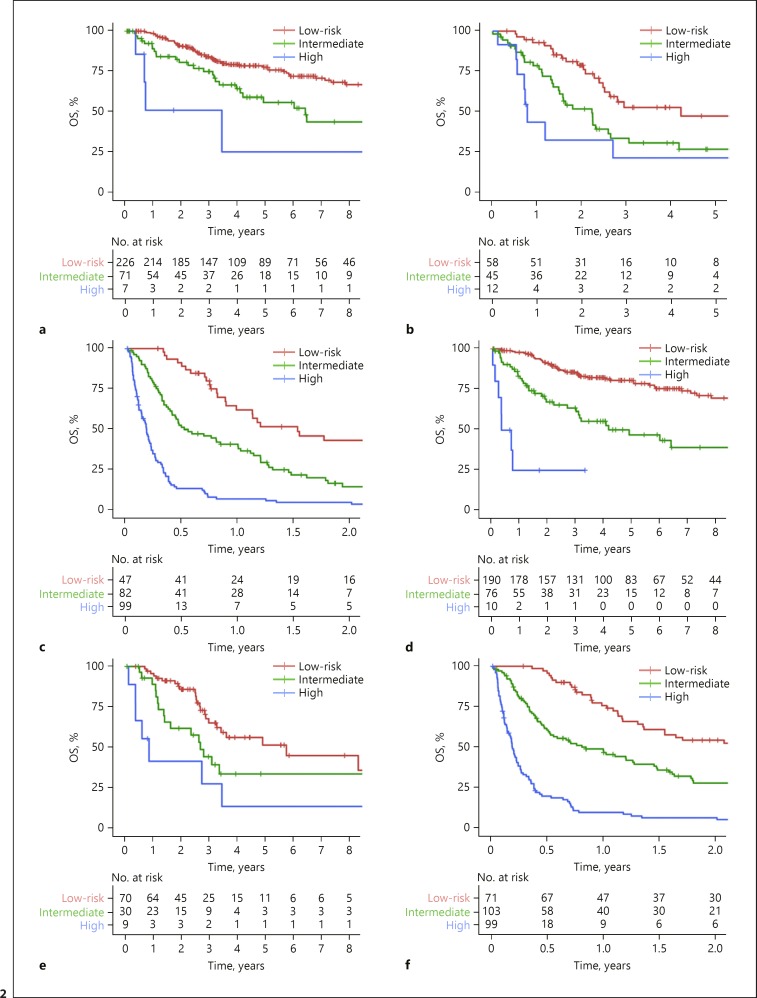
The ability of ILIS to stratify the widely adopted HCC staging system, BCLC, was studied in the internal validation cohort. Patients in each BCLC stage were stratified into 3 ILIS risk groups, namely the low, intermediate and high risks (Fig. 2a–c). ILIS could also further classify subgroups of patients according to AJCC stage (Fig. 2d–f), liver function and treatment intents (online suppl. Fig. 2, 3). In the multivariate analysis, the ILIS remained independent prognostic factor for OS after adjustment with BCLC (adjusted HR 31.90, p < 0.001) and AJCC TNM stage (adjusted HR 2.04, p < 0.001).
Fig. 2.
Kaplan-Meier survival plots comparing OS for patients stratified by ILIS in the internal validation cohort: (a) BCLC stage 0/A (log rank p < 0.001), (b) BCLC stage B (log rank p = 0.003), (c) BCLC stage C/D (log rank p < 0.001), (d) AJCC stage I (log rank p < 0.001), (e) AJCC stage II (log rank p < 0.001) and (f) AJCC stage III/IV (log rank p < 0.001). OS, overall survival.
Finally, the association between the ILIS and tumor features was investigated in the internal validation cohort. The median ILIS was higher in the presence of multiple tumors (median 3.16 vs. 2.31 p < 0.001), vascular invasion (median 3.80 vs. 2.43, p < 0.001) and extrahepatic spread (median 4.15 vs. 2.50, p < 0.001). The ILIS was also positively correlated with tumor size (Pearson R 0.431, p < 0.001) and stage (online suppl. Fig. 4). Even among patients with low serum AFP (<20 µg/L), ILIS remained higher in the presence of multiple tumors (p < 0.001), vascular invasion (p = 0.004), and extrahepatic spread (p = 0.009), and positively correlated with tumor size (R = 0.398, p < 0.001).
Prognostic Performances of Existing Inflammation-Based Prognostic Indices and ILIS
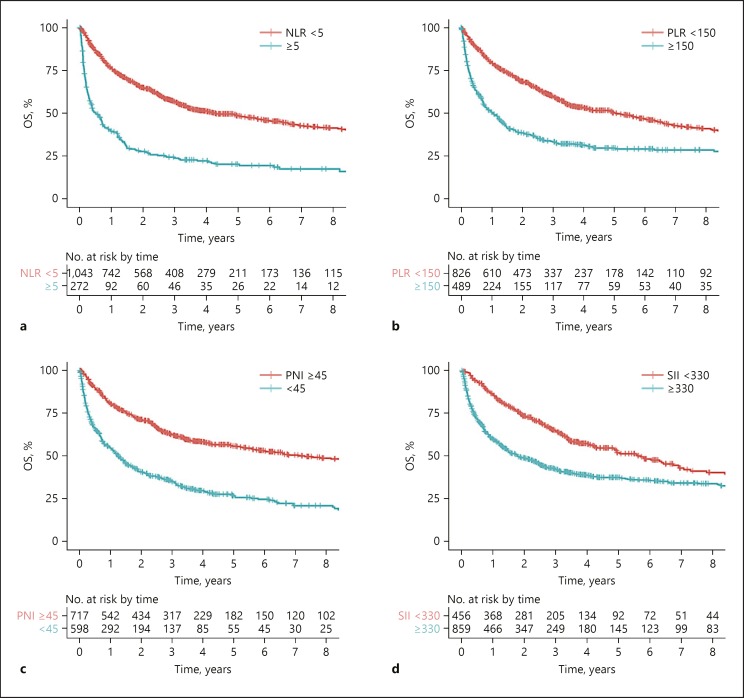
First, 4 existing indices, namely, the NLR, PLR, PNI, and SII were compared. Higher risk-category of NLR, PLR, PNI, and SII were associated with worse OS (Fig. 3). In the overall population, PNI had the best prognostic performance as evidenced by the highest homogeneity likelihood ratio chi-square and the lowest AICc, followed by NLR (Table 3). To determine how these tumor burden and hepatic dysfunction impact the prognostic performances of inflammation-based indices, subgroup analyses stratified by BCLC stages and ALBI grades were conducted (Table 3): In patients with early and intermediate-stage HCC (i.e., BCLC stage 0 to B), PNI has the best prognostic performance, while among patients with advanced stage HCC (i.e., BCLC stage C), NLR has the best performance. For patients with optimal hepatic function, defined as ALBI grade 1, PLR has the best performance, while NLR has the best prognostic performance in patients with less optimal liver function (i.e., ALBI grade 2 and 3). Second, ILIS was compared to the 4 above indices. As compared to NLR, PLR, PNI, and SII, the ILIS has the best prognostic performance in the Hong Kong cohort and subgroups of patients with different ALBI scores and BCLC stages (Table 3). Among patient subgroups with chronic hepatitis B, hepatitis C, non-B-non-C viral etiology, ILIS also has the best prognostic performance (Table 3).
Fig. 3.
Kaplan-Meier survival plots comparing OS for patients stratified by (a) NLR (logrank p < 0.001), (b) PLR (log rank p < 0.001), (c) PNI (log rank p < 0.001) and (d) SII (log rank p < 0.001). NLR, neutrophil to lymphocyte ratio; OS, overall survival; PLR, platelet-to-lymphocyte ratio; PNI, prognostic nutrition index; SII, systemic immune-inflammation index.
Table 3.
Prognostic performance of NLR, PLR, PNI, SII, and ILIS risk groups in the Hong Kong cohort
| HR | 95% CI | p value | Homogeneity likelihood ratio | ||
|---|---|---|---|---|---|
| χ2 test | AICc | ||||
| Entire cohort (n = 1,315) | |||||
| NLR | 2.88 | 2.443–3.389 | <0.001 | 135.35 | 9,240.77 |
| PLR | 2.07 | 1.782–2.398 | <0.001 | 88.26 | 9,287.86 |
| PNI | 2.47 | 2.125–2.868 | <0.001 | 140.63 | 9,235.48 |
| SII | 1.75 | 1.488–2.060 | <0.001 | 48.76 | 9,327.35 |
| ILIS | 3.63 | 3.261–4.038 | <0.001 | 525.84 | 8,849.78 |
| BCLC 0/A (n = 606) | |||||
| NLR | 1.54 | 1.019–2.325 | 0.041 | 3.77 | 2,289.61 |
| PLR | 1.09 | 0.785–1.505 | 0.615 | 0.25 | 2,293.13 |
| PNI | 2.29 | 1.724–3.030 | <0.001 | 31.23 | 2,262.15 |
| SII | 0.89 | 0.670–1.175 | 0.098 | 0.70 | 2,292.68 |
| ILIS | 2.47 | 1.936–3.160 | <0.001 | 44.97 | 2,281.82 |
| BCLC B (n = 237) | |||||
| NLR | 1.55 | 0.994–2.408 | 0.053 | 3.43 | 1,133.15 |
| PLR | 1.21 | 0.836–1.747 | 0.313 | 1.00 | 1,135.57 |
| PNI | 1.52 | 1.057–2.171 | 0.024 | 5.08 | 1,131.50 |
| SII | 1.31 | 0.899–1.896 | 0.161 | 2.01 | 1,134.57 |
| ILIS | 1.98 | 1.490–2.620 | <0.001 | 21.15 | 1,084.97 |
| BCLC C (n = 425) | |||||
| NLR | 2.31 | 1.848–2.891 | <0.001 | 49.23 | 3,628.31 |
| PLR | 1.76 | 1.419–2.181 | <0.001 | 26.87 | 3,650.68 |
| PNI | 1.45 | 1.171–1.803 | 0.001 | 11.76 | 3,665.78 |
| SII | 2.17 | 1.646–2.865 | <0.001 | 34.98 | 3,642.57 |
| ILIS | 2.33 | 1.983–2.735 | <0.001 | 109.13 | 3,568.15 |
| ALBI grade 1 (n = 622) | |||||
| NLR | 1.95 | 1.306–2.908 | 0.001 | 8.99 | 2,862.52 |
| PLR | 1.74 | 1.339–2.256 | <0.001 | 16.23 | 2,855.27 |
| PNI | 1.14 | 0.734–1.763 | 0.564 | 0.32 | 2,871.18 |
| SII | 1.47 | 1.115–1.926 | 0.006 | 7.84 | 2,863.67 |
| ILIS | 2.97 | 2.305–3.832 | <0.001 | 58.45 | 2,812.94 |
| ALBI grade 2/3 (n = 693) | |||||
| NLR | 2.17 | 1.800–2.624 | <0.001 | 60.31 | 5,399.42 |
| PLR | 1.95 | 1.624–2.342 | <0.001 | 50.26 | 5,409.48 |
| PNI | 1.45 | 1.146–1.836 | 0.002 | 10.33 | 5,449.40 |
| SII | 2.12 | 1.727–2.594 | <0.001 | 57.11 | 5,402.62 |
| ILIS | 3.31 | 2.853–3.843 | <0.001 | 261.25 | 5,198.05 |
| Hepatitis B (n = 1,060) | |||||
| NLR | 3.018 | 2.513–3.623 | <0.001 | 117.612 | 7,157.37 |
| PLR | 2.310 | 1.956–2.728 | <0.001 | 92.186 | 7,121.66 |
| PNI | 2.606 | 2.202–3.084 | <0.001 | 125.453 | 7,147.08 |
| SII | 1.884 | 1.569–2.263 | <0.001 | 49.649 | 7,113.82 |
| ILIS | 3.844 | 3.417–4.324 | <0.001 | 475.292 | 7,189.62 |
| Hepatitis C (n = 86) | |||||
| NLR | 0.83 | 0.255–2.714 | 0.761 | 0.097 | 300.62 |
| PLR | 1.76 | 0.806–3.832 | 0.156 | 1.794 | 298.92 |
| PNI | 1.72 | 0.921–3.214 | 0.089 | 2.917 | 297.80 |
| SII | 1.91 | 0.629–2.254 | 0.591 | 0.285 | 300.43 |
| ILIS | 1.57 | 1.022–2.408 | 0.039 | 4.003 | 296.71 |
| Non-B/non-C (n = 162) | |||||
| NLR | 2.71 | 1.771–4.145 | <0.001 | 18.74 | 849.78 |
| PLR | 1.18 | 0.787–1.766 | 0.425 | 0.640 | 867.88 |
| PNI | 2.01 | 1.346–3.007 | 0.001 | 11.436 | 857.08 |
| SII | 1.22 | 0.718–2.059 | 0.466 | 0.553 | 867.96 |
| ILIS | 2.72 | 2.051–3.618 | <0.001 | 44.606 | 823.91 |
ALBI, albumin-bilirubin score; BCLC, Barcelona clinic liver cancer; ILIS, integrated liver inflammatory score; NLR, neutrophil-lymphocyte ratio; PLR, platelet-lymphocyte ratio; PNI, prognostic nutritional index; SII, systemic immune-inflammation index; HR, hazard ratio.
ILIS, Serum Cytokines, and Tumor Microenvironment
A subset of cohort from Hong Kong cohort previously showed that selected circulating cytokines, namely, interleukin-6 (IL-6), IL-8, IL-10, and IL-17A, were prognostic of OS [22]. Hence, we studied the association between inflammatory indices and those 4 circulating cytokines. It was found that serum IL6, IL8, and IL10 were correlated with all inflammatory indices, whereas serum IL17A was correlated with ILIS, PNI, and SII (Table 4). Compared to other inflammatory indices, ILIS demonstrated the strongest association with all 4 cytokines, particularly IL8 (rho 0.553, p < 0.001; moderate correlation). At the same time, all 5 components of ILIS were significantly and weakly-to-moderately correlated with serum IL8, albumin (Spearman rho −0.305, p < 0.001), bilirubin (rho 0.330, p < 0.001), ALP (rho 0.480, p < 0.001), neutrophil count (rho 0.369, p < 0.001) and AFP (rho 0.352, p < 0.001).
Table 4.
Correlation of blood-based inflammatory indices with serum cytokines and tumor microenvironment
| ILIS | NLR | PLR | PNI | SII | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| spearman rho | adjusted p value | spearman rho | adjusted p value | spearman rho | adjusted p value | spearman rho | adjusted p value | spearman rho | adjusted p value | |
| Serum cytokines (n = 458) | ||||||||||
| IL6 | 0.395 | <0.001 | 0.287 | <0.001 | 0.167 | <0.001 | -0.237 | <0.001 | 0.279 | <0.001 |
| IL8 | 0.553 | <0.001 | 0.336 | <0.001 | 0.285 | <0.001 | -0.266 | <0.001 | 0.361 | <0.001 |
| IL10 | 0.204 | <0.001 | 0.108 | 0.023 | 0.076 | 0.142 | -0.098 | 0.047 | 0.119 | 0.015 |
| IL17A | 0.174 | <0.001 | 0.042 | 0.360 | 0.053 | 0.256 | -0.077 | 0.101 | 0.074 | 0.116 |
| Inflammatory cells (n = 71) | ||||||||||
| CD8A (cytotoxic T cell) | 0.005 | 0.970 | -0.083 | 0.861 | -0.121 | 0.890 | 0.057 | 0.970 | -0.023 | 0.851 |
| CD4 (T-helper cell) | -0.122 | 0.985 | -0.031 | 0.985 | -0.043 | 0.985 | -0.022 | 0.985 | -0.002 | 0.985 |
| FOXP3 (regulatory T cell) | -0.059 | 0.731 | -0.087 | 0.694 | -0.251 | 0.244 | 0.082 | 0.694 | -0.179 | 0.471 |
| CD19 (B cell) | -0.277 | 0.137 | -0.081 | 0.587 | -0.085 | 0.587 | 0.154 | 0.463 | 0.035 | 0.772 |
| CD163 (macrophage M2) | -0.202 | 0.640 | 0.075 | 0.747 | 0.105 | 0.747 | -0.010 | 0.937 | 0.121 | 0.747 |
| NCAM1 (natural killer cell) | -0.011 | 0.928 | 0.140 | 0.515 | 0.047 | 0.814 | -0.154 | 0.515 | 0.126 | 0.515 |
| Immune checkpoints (n = 71) | ||||||||||
| PD1 | 0.031 | 0.801 | 0.111 | 0.498 | 0.113 | 0.498 | -0.142 | 0.498 | 0.139 | 0.498 |
| PD-L1 | 0.092 | 0.579 | 0.166 | 0.289 | 0.176 | 0.289 | -0.082 | 0.579 | 0.216 | 0.289 |
| CTLA4 | 0.089 | 0.890 | -0.037 | 0.890 | -0.084 | 0.890 | -0.045 | 0.890 | -0.002 | 0.984 |
| LAG3 | -0.012 | 0.920 | 0.053 | 0.920 | 0.018 | 0.920 | -0.088 | 0.920 | 0.048 | 0.920 |
| TIM3 | 0.058 | 0.629 | 0.090 | 0.629 | 0.061 | 0.629 | -0.192 | 0.629 | 0.083 | 0.629 |
| Sorafenib-related genes (n = 71) | ||||||||||
| PDGFRB | -0.420 | 0.002 | -0.047 | 0.986 | 0.024 | 0.986 | 0.115 | 0.986 | 0.031 | 0.986 |
| KIT | -0.341 | 0.027 | -0.286 | 0.055 | -0.127 | 0.342 | 0.185 | 0.170 | -0.187 | 0.170 |
| FLT3 | -0.198 | 0.115 | -0.226 | 0.082 | -0.281 | 0.052 | 0.271 | 0.052 | -0.227 | 0.082 |
| VEGFA | -0.402 | 0.004 | -0.192 | 0.252 | -0.068 | 0.575 | 0.267 | 0.085 | -0.112 | 0.495 |
| CD34 | -0.348 | 0.022 | -0.235 | 0.168 | -0.147 | 0.321 | 0.108 | 0.370 | -0.164 | 0.321 |
CD, cluster of differentiation; CTLA4, cytotoxic T-lymphocyte-associated protein 4; FLT3, FMS-like tyrosine kinase 3; FOXP3, Forkhead Box P3; IL, interleukin; ILIS, integrated liver inflammatory score; KIT, KIT proto-oncogene receptor tyrosine kinase; LAG3, lymphocyte-activation gene 3; NCAM1, neural cell adhesion molecule 1; NLR, neutrophil-lymphocyte ratio; PD-1, programmed cell death protein 1; PD-L1, programmed cell death protein ligand 1; PDGFRB, platelet derived growth factor-beta; PLR, platelet-lymphocyte ratio; PNI, prognostic nutritional index; SII, systemic immune-inflammation index; TIM-3, T-cell immunoglobulin and mucin domain-3; VEGFA, vascular endothelial growth factor-A.
NanoString PanCancer Profiling Panel was performed on tumorous tissue of 71 surgically resected HCC. None of inflammatory indices was associated with gene expression levels of tumor-infiltrating inflammatory cells and immune checkpoints (Table 4). In the NanoString panel, genes related to the targets of sorafenib were also evaluated [27]. ILIS was negatively correlated with the intratumoral expression of PDGFRB(rho −0.420, p = 0.002), KIT (rho −0.341, p = 0.027), VEGFA (rho −0.402, p = 0.004) and vascular marker CD34 (rho −0.348, p = 0.022), whereas NLR showed borderline negative correlation with KIT (rho −0.286, p = 0.055) and FLT3 (rho −0.226, p = 0.082).
Discussion
In this study, a large cohort of 1,315-HCC patient was employed to comprehensively review the performances of current inflammation-based indices. All inflammatory indices, namely, the NLR, PNI, PLR, and SII, are prognostic of clinical outcomes. However, the prognostic performances of an inflammatory index vary remarkably according to the composition of patient subgroups. This finding could explain the inconsistent results on the performance of circulatory inflammatory index observed in different studies on patients with HCC [2, 3, 4, 5, 6, 8, 9, 10, 28, 29]. The absence of a single reliable inflammatory index across subgroups of HCC patients may limit the application of inflammatory indices in clinical practice.
We established a novel HCC-specific inflammatory score, ILIS, and validated its prognostic ability in both internal and external cohorts. There are unique features about the ILIS: First, it is a model that is built on objective blood-based parameters. In the literature, there is another similar blood-based HCC prognostic model, BALAD-2, which combines 5 parameters including bilirubin, albumin, AFP, AFP-L3% and des-gamma carboxyprothrombin (DCP) [30, 31]. However, AFP-L3% and DCP are not routinely measured in most centers, while the parameters of ILIS, namely, albumin, bilirubin, neutrophils, and AFP, are readily available from routine blood testing. Further, in a subset of cohort from Hong Kong with AFP-L3% and DCP data, the ILIS has better prognostic performance than the BALAD-2 (n = 225; homogeneity likelihood ratio chi-square 160.57 vs. 32.54; AICc 1,265.25 vs. 1,393.27) [30]. Second, in both cohorts from Hong Kong and Newcastle, the ILIS has consistently better prognostic performance than other existing inflammatory indices. Its superiority in predicting clinical outcome also persists among different subgroups of patients. This feature highlights the versatility of the ILIS in different populations of HCC globally. Third, in terms of clinical application, the ILIS is able to further classify the AJCC TNM and BCLC stages, suggesting that ILIS could provide additional prognostic information to currently used staging systems. The ILIS may also potentially provide references for clinicians in treatment decision in which ILIS-low risk has higher chance (72.8%) of undergoing treatment of curative intent and 53.4% of the ILIS-high-risk patients are put on supportive care (online suppl. Table 3).
The inflammation occurs in both systemic circulation or within tumor environment. For systemic inflammation, there are scanty data on the association between serum cytokines and the inflammatory indices [32, 33]. In this study, we examine 4 serum cytokines, namely, the IL6, IL8, IL-10, and IL17A, which were previously demonstrated to be important prognosticators [22]. It is shown that the ILIS is strongly correlated with all 4 cytokines, particularly with the serum level of IL8. These findings showed that ILIS is a potential surrogate for circulating inflammatory cytokines in HCC, which are congruent with existing observation that systemic inflammation is a complex interplay of host immune system with tumor and underlying chronic liver disease [34]. For intratumoral inflammation, there are mixed results on its association with inflammatory indices. On the one hand, Motomura et al. [7] reported that NLR was associated with peritumoral rather than intratumoral CD163 (tumor-associated macrophage M2) gene expression in HCC. On the other hand, Wang et al. [35] did not demonstrate any association between inflammatory indices (NLR, PLR, and PNI) and intratumoral/peritumoral gene expression of CD4 (T-helper cell) and CD8 (cytotoxic T-cell) in the tumors. Results of the current study do not demonstrate any association between inflammatory indices and intratumoral gene expression. The exact reasons are unclear, but it is likely that both systemic and intratumoral inflammation may not be simplistically linked by tissue and blood inflammatory cell counts in HCC as is the case with colorectal cancer [36, 37].
Inflammatory indices are potentially informative of treatment outcomes in cancers. NLR was recently shown to be negative predictive and prognostic factors of sorafenib-treated HCC patients [9]. We explore the relationship between circulatory inflammatory markers and expression levels of potential target genes of sorafenib. The results show that NLR is marginally negatively correlated with expression levels of KIT and FLT3 gene, while ILIS is significantly and negatively correlated with expression levels of PDGFRB, KIT, VEGFA, and CD34. Therefore, it is valuable to study whether ILIS is potentially prognostic or even predictive of clinical outcomes in sorafenib treatment in HCC [9]. Apart from sorafenib, NLR was also reported to be correlated with intratumoral PD-L1 expression in HCC [35], and prognosticators for patients undergoing check-point inhibitors for melanoma and non-small cell lung cancer [38, 39]. Sia et al. [40] studied the transcriptomes of HCC and identified 2 subtypes of HCC with different gene expressions and implications on immunotherapies. In the current study, we did not find any association between the inflammatory indices and intratumoral mRNA levels of immune checkpoint inhibitors in HCC. More studies are required to clarify the association between inflammatory indices and gene expression of check points as well as outcomes of immunotherapeutics in HCC.
There are few limitations about this study. First, although all 5 parameters of ILIS are readily obtained from routine blood tests, calculation of ILIS may be too complicated for daily practice. To facilitate the utility of ILIS, we designed a user-friendly online calculator available at http://goo.gl/2orPPY. Second, it may be noted that the ILIS does not incorporate serum CRP or its derivative scores, which are shown to be prognosticators for HCC [2, 6]. We decided not to incorporate CRP because it is not routinely evaluated in clinical practice of HCC. Due to the same reason, the inflammation-based prognostic score (GPS), another index with component of CRP, was not studied and compared to ILIS in the current paper. Third, although the prognostic performance of the ILIS persists in patients with hepatitis viral infection, the impact of antiviral therapy for chronic hepatitis B virus on prognostication of the ILIS is unclear because individual data on antiviral therapy for hepatitis B are not available in both cohorts. However, both cohorts were constructed after 2000, and the use of antiviral therapy is expected to be high. Finally, the robustness of data on tumor microenvironment may be limited by a small subset, of viral etiology, being analyzed for gene expression levels in a pre-selected NanoString panel. Future study on a larger cohort of patients receiving both surgical and non-surgical therapies will enhance our understanding on the relationship between tumor environment and systemic inflammation.
In conclusion, we developed a novel ILIS, by incorporating liver function, systemic inflammation and tumor factor. The ILIS is an externally validated, powerful prognosticator with consistent performance across subgroups of patients with different tumor burden and liver dysfunction.
Statement of Ethics
The study was conducted ethically in accordance with the World Medical Association Declaration of Helsinki. The assembly of databases and related protocols were approved by the Ethic Committees of both institutes (in Hong Kong and in the United Kingdom).
Disclosure Statement
The authors declare that they have no conflicts of interest to disclose.
Funding Sources
No funding was received for this study.
Author Contributions
S.L.C. and A.W.-H.C.: design of study. L.-L.W., C.C.-N.C., C.-M.C., S.L.C., A.W.-H.C., and H.L.R.: collection of clinical data. K.-C.A.C., C.C., J.H.-M.T., P.-H.L., K.-F.T., and A.W.-H.C.: contribution to laboratory data. T.C.-F.Y., G.L.-H.W., V.W.-S.W., S.L.C., and A.W.-H.C.: statistical analysis and interpretation of data. S.L.C. and A.W.-H.C.: drafting of manuscript. All authors: approval of manuscript.
Supplementary Material
Supplementary data
Acknowledgment
None.
References
- 1.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011 Mar;144((5)):646–74. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 2.Chan SL, Chan AW, Chan AK, Jian P, Mo F, Chan CM, et al. Systematic evaluation of circulating inflammatory markers for hepatocellular carcinoma. Liver Int. 2017 Feb;37((2)):280–9. doi: 10.1111/liv.13218. [DOI] [PubMed] [Google Scholar]
- 3.Pinato DJ, North BV, Sharma R. A novel, externally validated inflammation-based prognostic algorithm in hepatocellular carcinoma: the prognostic nutritional index (PNI) Br J Cancer. 2012 Apr;106((8)):1439–45. doi: 10.1038/bjc.2012.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chan AW, Chan SL, Wong GL, Wong VW, Chong CC, Lai PB, et al. Prognostic Nutritional Index (PNI) Predicts Tumor Recurrence of Very Early/Early Stage Hepatocellular Carcinoma After Surgical Resection. Ann Surg Oncol. 2015 Dec;22((13)):4138–48. doi: 10.1245/s10434-015-4516-1. [DOI] [PubMed] [Google Scholar]
- 5.Hu B, Yang XR, Xu Y, Sun YF, Sun C, Guo W, et al. Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma. Clin Cancer Res. 2014 Dec;20((23)):6212–22. doi: 10.1158/1078-0432.CCR-14-0442. [DOI] [PubMed] [Google Scholar]
- 6.Pinato DJ, Stebbing J, Ishizuka M, Khan SA, Wasan HS, North BV, et al. A novel and validated prognostic index in hepatocellular carcinoma: the inflammation based index (IBI) J Hepatol. 2012 Nov;57((5)):1013–20. doi: 10.1016/j.jhep.2012.06.022. [DOI] [PubMed] [Google Scholar]
- 7.Motomura T, Shirabe K, Mano Y, Muto J, Toshima T, Umemoto Y, et al. Neutrophil-lymphocyte ratio reflects hepatocellular carcinoma recurrence after liver transplantation via inflammatory microenvironment. J Hepatol. 2013 Jan;58((1)):58–64. doi: 10.1016/j.jhep.2012.08.017. [DOI] [PubMed] [Google Scholar]
- 8.Xiao WK, Chen D, Li SQ, Fu SJ, Peng BG, Liang LJ. Prognostic significance of neutrophil-lymphocyte ratio in hepatocellular carcinoma: a meta-analysis. BMC Cancer. 2014 Feb;14((1)):117. doi: 10.1186/1471-2407-14-117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bruix J, Cheng AL, Meinhardt G, Nakajima K, De Sanctis Y, Llovet J. Prognostic factors and predictors of sorafenib benefit in patients with hepatocellular carcinoma: analysis of two phase III studies. J Hepatol. 2017 Nov;67((5)):999–1008. doi: 10.1016/j.jhep.2017.06.026. [DOI] [PubMed] [Google Scholar]
- 10.Zhao Y, Si G, Zhu F, Hui J, Cai S, Huang C, et al. Prognostic role of platelet to lymphocyte ratio in hepatocellular carcinoma: a systematic review and meta-analysis. Oncotarget. 2017 Apr;8((14)):22854–62. doi: 10.18632/oncotarget.15281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ueno S, Tanabe G, Sako K, Hiwaki T, Hokotate H, Fukukura Y, et al. Discrimination value of the new western prognostic system (CLIP score) for hepatocellular carcinoma in 662 Japanese patients. Cancer of the Liver Italian Program. Hepatology. 2001 Sep;34((3)):529–34. doi: 10.1053/jhep.2001.27219. [DOI] [PubMed] [Google Scholar]
- 12.Chan SL, Johnson PJ, Mo F, Berhane S, Teng M, Chan AW, et al. International validation of the Chinese university prognostic index for staging of hepatocellular carcinoma: a joint United Kingdom and Hong Kong study. Chin J Cancer. 2014 Oct;33((10)):481–91. doi: 10.5732/cjc.014.10133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chan AW, Chan SL, Mo FK, Wong GL, Wong VW, Cheung YS, et al. Albumin-to-alkaline phosphatase ratio: a novel prognostic index for hepatocellular carcinoma. Dis Markers. 2015;2015:564057. doi: 10.1155/2015/564057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chan AW, Chong CC, Mo FK, Wong J, Yeo W, Johnson PJ, et al. Incorporating albumin-bilirubin grade into the cancer of the liver Italian program system for hepatocellular carcinoma. J Gastroenterol Hepatol. 2017 Jan;32((1)):221–8. doi: 10.1111/jgh.13457. [DOI] [PubMed] [Google Scholar]
- 15.Margetts J, Ogle LF, Chan SL, Chan AW, Chan KC, Jamieson D, et al. Neutrophils: driving progression and poor prognosis in hepatocellular carcinoma? Br J Cancer. 2018 Jan;118((2)):248–57. doi: 10.1038/bjc.2017.386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Johnson PJ, Berhane S, Kagebayashi C, Satomura S, Teng M, Reeves HL, et al. Assessment of liver function in patients with hepatocellular carcinoma: a new evidence-based approach-the ALBI grade. J Clin Oncol. 2015 Feb;33((6)):550–8. doi: 10.1200/JCO.2014.57.9151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liu PH, Hsu CY, Hsia CY, Lee YH, Chiou YY, Huang YH, et al. ALBI and PALBI grade predict survival for HCC across treatment modalities and BCLC stages in the MELD Era. J Gastroenterol Hepatol. 2017 Apr;32((4)):879–86. doi: 10.1111/jgh.13608. [DOI] [PubMed] [Google Scholar]
- 18.Chun YH, Kim SU, Park JY, Kim DY, Han KH, Chon CY, et al. Prognostic value of the 7th edition of the AJCC staging system as a clinical staging system in patients with hepatocellular carcinoma. Eur J Cancer. 2011;47:2568–2575. doi: 10.1016/j.ejca.2011.07.002. [DOI] [PubMed] [Google Scholar]
- 19.Chan AW, Kumada T, Toyoda H, Tada T, Chong CC, Mo FK, et al. Integration of albumin-bilirubin (ALBI) score into Barcelona Clinic Liver Cancer (BCLC) system for hepatocellular carcinoma. J Gastroenterol Hepatol. 2016 Jul;31((7)):1300–6. doi: 10.1111/jgh.13291. [DOI] [PubMed] [Google Scholar]
- 20.Chan AW, Chong CC, Mo FK, Wong J, Yeo W, Johnson PJ, et al. Applicability of albumin-bilirubin-based Japan integrated staging score in hepatitis B-associated hepatocellular carcinoma. J Gastroenterol Hepatol. 2016 Oct;31((10)):1766–72. doi: 10.1111/jgh.13339. [DOI] [PubMed] [Google Scholar]
- 21.Okuda K, Ohtsuki T, Obata H, Tomimatsu M, Okazaki N, Hasegawa H, et al. Natural history of hepatocellular carcinoma and prognosis in relation to treatment. Study of 850 patients. Cancer. 1985 Aug;56((4)):918–28. doi: 10.1002/1097-0142(19850815)56:4<918::aid-cncr2820560437>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- 22.Chan SL, Chan AW, Chan AK, Jian P, Mo F, Chan CM, Mok K, Liu C, Chong CC, Chan AT, Mok T, Yeo W. Systematic evaluation of circulating inflammatory markers for hepatocellular carcinoma. Liver Int. 2017 Feb;37((2)):280–9. doi: 10.1111/liv.13218. [DOI] [PubMed] [Google Scholar]
- 23.Cesano A. nCounter(®) PanCancer Immune Profiling Panel (NanoString Technologies, Inc., Seattle, WA) J Immunother Cancer. 2015 Dec;3((1)):42. doi: 10.1186/s40425-015-0088-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Austin PC, Tu JV. Automated variable selection methods for logistic regression produced unstable models for predicting acute myocardial infarction mortality. J Clin Epidemiol. 2004 Nov;57((11)):1138–46. doi: 10.1016/j.jclinepi.2004.04.003. [DOI] [PubMed] [Google Scholar]
- 25.Royston P, Altman DG. External validation of a Cox prognostic model: principles and methods. BMC Med Res Methodol. 2013 Mar;13((1)):33. doi: 10.1186/1471-2288-13-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Reiner A, Yekutieli D, Benjamini Y. Identifying differentially expressed genes using false discovery rate controlling procedures. Bioinformatics. 2003 Feb;19((3)):368–75. doi: 10.1093/bioinformatics/btf877. [DOI] [PubMed] [Google Scholar]
- 27.Hahn O, Stadler W. Sorafenib. Curr Opin Oncol. 2006 Nov;18((6)):615–21. doi: 10.1097/01.cco.0000245316.82391.52. [DOI] [PubMed] [Google Scholar]
- 28.Fu YP, Ni XC, Yi Y, Cai XY, He HW, Wang JX, et al. A Novel and Validated Inflammation-Based Score (IBS) Predicts Survival in Patients With Hepatocellular Carcinoma Following Curative Surgical Resection: A STROBE-Compliant Article. Medicine (Baltimore) 2016 Feb;95((7)):e2784. doi: 10.1097/MD.0000000000002784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yang T, Zhu J, Zhao L, Mai K, Ye J, Huang S, et al. Lymphocyte to monocyte ratio and neutrophil to lymphocyte ratio are superior inflammation-based predictors of recurrence in patients with hepatocellular carcinoma after hepatic resection. J Surg Oncol. 2017 May;115((6)):718–28. doi: 10.1002/jso.24549. [DOI] [PubMed] [Google Scholar]
- 30.Berhane S, Toyoda H, Tada T, Kumada T, Kagebayashi C, Satomura S, Schweitzer N, Vogel A, Manns MP, Benckert J, Berg T, Ebker M, Best J, Dechene A, Gerken G, Schlaak JF, Weinmann A, Worns MA, Galle P, Yeo W, Mo F, Chan SL, Reeves H, Cox T, Johnson P. Role of the GALAD and BALAD-2 Serologic Models in Diagnosis of Hepatocellular Carcinoma and Prediction of Survival in Patients. Clin Gastroenterol Hepatol. 2016 Jun;14((6)):875–86.e6. doi: 10.1016/j.cgh.2015.12.042. [DOI] [PubMed] [Google Scholar]
- 31.Chan SL, Mo F, Johnson P, Li L, Tang N, Loong H, et al. Applicability of BALAD score in prognostication of hepatitis B-related hepatocellular carcinoma. J Gastroenterol Hepatol. 2015 Oct;30((10)):1529–35. doi: 10.1111/jgh.13005. [DOI] [PubMed] [Google Scholar]
- 32.Guthrie GJ, Roxburgh CS, Richards CH, Horgan PG, McMillan DC. Circulating IL-6 concentrations link tumour necrosis and systemic and local inflammatory responses in patients undergoing resection for colorectal cancer. Br J Cancer. 2013 Jul;109((1)):131–7. doi: 10.1038/bjc.2013.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ock CY, Nam AR, Lee J, Bang JH, Lee KH, Han SW, et al. Prognostic implication of antitumor immunity measured by the neutrophil-lymphocyte ratio and serum cytokines and angiogenic factors in gastric cancer. Gastric Cancer. 2017 Mar;20((2)):254–62. doi: 10.1007/s10120-016-0613-5. [DOI] [PubMed] [Google Scholar]
- 34.Estevez J, Chen VL, Podlaha O, Li B, Le A, Vutien P, et al. Differential Serum Cytokine Profiles in Patients with Chronic Hepatitis B, C, and Hepatocellular Carcinoma. Sci Rep. 2017 Sep;7((1)):11867. doi: 10.1038/s41598-017-11975-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wang Q, Blank S, Fiel MI, Kadri H, Luan W, Warren L, et al. The Severity of Liver Fibrosis Influences the Prognostic Value of Inflammation-Based Scores in Hepatitis B-Associated Hepatocellular Carcinoma. Ann Surg Oncol. 2015 Dec;22((S3 Suppl 3)):S1125–32. doi: 10.1245/s10434-015-4598-9. [DOI] [PubMed] [Google Scholar]
- 36.Richards CH, Flegg KM, Roxburgh CS, Going JJ, Mohammed Z, Horgan PG, et al. The relationships between cellular components of the peritumoural inflammatory response, clinicopathological characteristics and survival in patients with primary operable colorectal cancer. Br J Cancer. 2012 Jun;106((12)):2010–5. doi: 10.1038/bjc.2012.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Turner N, Wong HL, Templeton A, Tripathy S, Whiti Rogers T, Croxford M, et al. Analysis of local chronic inflammatory cell infiltrate combined with systemic inflammation improves prognostication in stage II colon cancer independent of standard clinicopathologic criteria. Int J Cancer. 2016 Feb;138((3)):671–8. doi: 10.1002/ijc.29805. [DOI] [PubMed] [Google Scholar]
- 38.Ferrucci PF, Gandini S, Battaglia A, Alfieri S, Di Giacomo AM, Giannarelli D, et al. Baseline neutrophil-to-lymphocyte ratio is associated with outcome of ipilimumab-treated metastatic melanoma patients. Br J Cancer. 2015 Jun;112((12)):1904–10. doi: 10.1038/bjc.2015.180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bagley SJ, Kothari S, Aggarwal C, Bauml JM, Alley EW, Evans TL, et al. Pretreatment neutrophil-to-lymphocyte ratio as a marker of outcomes in nivolumab-treated patients with advanced non-small-cell lung cancer. Lung Cancer. 2017 Apr;106:1–7. doi: 10.1016/j.lungcan.2017.01.013. [DOI] [PubMed] [Google Scholar]
- 40.Sia D, Jiao Y, Martinez-Quetglas I, Kuchuk O, Villacorta-Martin C, Castro de Moura M, et al. Identification of an Immune-specific Class of Hepatocellular Carcinoma, Based on Molecular Features. Gastroenterology. 2017 Sep;153((3)):812–26. doi: 10.1053/j.gastro.2017.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary data