Abstract
Chloroplast-to-nucleus retrograde signalling (RS) is known to impact plant growth and development. In Arabidopsis, we and others have shown that RS affects seedling establishment by inhibiting deetiolation. In the presence of lincomycin, a chloroplast protein synthesis inhibitor that triggers RS, Arabidopsis light-grown seedlings display partial skotomorphogenesis with undeveloped plastids and closed cotyledons. By contrast, RS in monocotyledonous has been much less studied. Here, we show that emerging rice seedlings exposed to lincomycin do not accumulate chlorophyll but otherwise remain remarkably unaffected. However, by using high red (R) and blue (B) monochromatic lights in combination with lincomycin, we have uncovered a RS inhibition of length and a reduction in the B light-induced declination of the second leaf. Furthermore, we present data showing that seedlings grown in high B and R light display different non-photochemical quenching capacity. Our findings support the view that excess B and R light impact seedling photomorphogenesis differently to photoprotect and optimize the response to high-light stress.
This article is part of the theme issue ‘Retrograde signalling from endosymbiotic organelles'.
Keywords: retrograde signalling, rice, photomorphogenesis, blue and red light, photoprotection, non-photochemical quenching
1. Introduction
Light is fundamental for plants as a source of energy and as an essential environmental cue. The quality, intensity, direction and duration of ambient light inform the plant about its immediate surroundings, time of day and season. To capture this information, plants possess multiple photoreceptors sensitive to different wavelengths, including the red (R) and far red (FR) sensing phytochromes (phy) and the blue (B)/UV-A sensing cryptochromes (CRY). Rice, a model monocotyledonous plant, contains three phytochromes (phyA, phyB and phyC) [1] and three cryptochromes (OsCRY1a, OsCRY1b and OsCRY2) [2,3]. The contribution of each photoreceptor to light-regulated development has been elucidated through the study of deficient and overexpression mutants, which have shown similar but also distinct functions compared with their Arabidopisis counterparts [3–7].
Light or its absence is especially critical during germination and seedling establishment [8]. Early seedling growth and development is first fuelled by the seed reserves, and this heterotrophic lifestyle can proceed in the dark for a few days. Seedlings in darkness exhibit skotomorphogenic development, characterized in rice by long coleoptiles, long first leaves and elongation of the second internode. Upon light illumination, elongation of coleoptiles, first leaves and internodes is inhibited, and seedlings switch to a photomorphogenesis pattern of growth with the development of fully functional chloroplasts and transition to autotrophy [5]. During this process, R, FR and B light contribute to the inhibition of elongation through the action of both phytochromes and cryptochromes. In contrast to Arabidopsis, rice phyA and phyC are both responsible for responses to continuous FR [4,5], whereas phyB and phyC are involved in the responses to continuous R, and CRY1a and CRY1b are the main cryptochromes involved in the deetiolation response to B [3]. Interestingly, B light promotes the declination of second leaf blades via cryptochrome function, and phytochromes (most prominently phyB) behave antagonistically [5]. Furthermore, supplemental B light has been shown to be essential for proper growth by enhancing photosynthesis and increasing the total nitrogen content of rice leaves [9].
In addition to providing environmental information, light can also be a source of stress for the plant. Whenever it is absorbed beyond photosynthetic capacity, excess energy becomes harmful, causing oxidation of the photosynthetic apparatus and eventually cell death. In response, plants induce a photoprotective mode known as non-photochemical quenching (NPQ), which dissipates the excess energy as heat [10]. NPQ has been shown to be particularly important for plant fitness and productivity in field conditions [11,12]. Mechanistically, NPQ comprises different components defined by their time-scales of induction and relaxation. The fastest component is qE, energy quenching in the antenna of photosystem II (PSII). qE relies on specific carotenoids from the xanthophyll group and on the qE protein effector PSBS [10]. PSBS is able to sense thylakoidal pH changes and activate the energy-dissipation mode by a mechanism that is not yet resolved [13,14]. The slowest NPQ component is qI, photoinhibitory quenching that comprises all processes directly related to photoinhibition of PSII [15].
Assembly of fully functional chloroplasts during deetiolation requires exquisite coordination between the nucleus and the chloroplast. Most of the chloroplast components are encoded in the nuclear genome [16], and need to be imported into the chloroplast following their synthesis in the cytosol, a process referred to as anterograde regulation [17]. The chloroplast can, in turn, communicate with the nucleus in a process called retrograde signalling (RS), which adjusts nuclear gene expression to chloroplast status [18,19]. This RS taking place during chloroplast biogenesis has been called ‘biogenic control', and involves tight regulation of the expression of nuclear-encoded photosynthetic genes (PhANGs) such as those from the LHCb gene family [20].
Activation of RS takes place when chloroplast are damaged under stress conditions like high light (HL), or with the use of chemicals like lincomycin that specifically inhibit plastid translation [21,22]. Activation of RS causes repression of PhANG expression through a process that is mediated by the plastid-localized protein genomes uncoupled 1 (GUN1) and involves repression of GOLDEN2-LIKE (GLK) gene expression [20,23–27]. In Arabidopsis, RS has been shown to optimize not only pigment accumulation and photosynthetic capacity but also morphogenic development to acclimate to HL stress [21,27,28]. Accordingly, the gun1 mutant is more sensitive to HL [21].
Our previous studies together with others [21,27], have established in Arabidopsis that GUN1-mediated RS blocks photomorphogenic development during seedling deetiolation, particularly the inhibition of cotyledon separation. Indeed, light-grown seedlings grown in the presence of lincomycin show a phenotype resembling dark-grown seedlings, with longer hypocotyls and appressed cotyledons with undeveloped plastids that do not green [27]. This response involves repression of GLK1 expression and has been proposed to minimize the area exposed to potentially damaging light [27]. By contrast, RS in monocots has been much less studied [29–31], and whether it impacts photomorphogenesis has not been addressed. Here, we aim to start to characterize the RS response in rice to better understand how RS has evolved as a photoprotective mechanism for plants. We show that emerging rice seedlings activate RS in response to chloroplast stress, but their early development is remarkably unaffected by lincomycin and they do not exhibit characteristics of dark-grown seedlings, in clear contrast to Arabidopsis. Interestingly, seedlings grown under R or B monochromatic lights respond differently to lincomycin, and we uncover an inhibition of the length and the declination angle of second leaf blades in B light. Furthermore, we present data that indicate that seedlings grown in B and R light have distinct capacity to induce NPQ and display different photoprotection mechanisms to optimize growth in HL environments.
2. Methods
(a). Plant material and growth conditions
Rice (Oryza sativa cv. Nipponbare) was used in all experiments. After harvesting, rice seeds were incubated at 37°C for two weeks to break dormancy. Before sowing, seeds were dehusked and sterilized for 1 min in 75% ethanol with shaking, followed by a 20 min treatment in 2.5% bleach and five washes with sterile water. Eight seeds were plated on 0.5 MS medium without sucrose in 14 cm high glass jars covered with one layer of transparent plastic film, except for the red light treatments where 7 cm high glass jars were used.
For constant light experiment, plants were grown at 25–28°C for 5 days in darkness (D), or in continuous blue (B) (450 nm) (provided by Philips GreenPower LED research module blue), red (R) (660 nm) (provided by Philips GreenPower LED research deep red) or white light (W) at the specified light intensities. Light spectra were measured with a Flame-S spectrometer (Ocean Optics), and fluence rates were measured with an LI-190R quantum sensor (LI-COR).
In the deetiolation experiments, plants were first grown in continuous dark at 25–28°C for 3 days, and then transferred to light for another 4 days. Lincomycin treatments were done by supplementing the medium with 2 mM lincomycin (Sigma-Aldrich, L2774). All the experiments were performed with at least two biological replicates as specified in each figure legend.
For seedling morphology measurements (see electronic supplementary material, figure S1), seedlings were photographed using a digital camera Nikon D8000 and measurements were made using the NIH Image software (ImageJ, National Institutes of Health) [32].
(b). RNA extraction and quantitative RT-PCR
Shoot tissue from 5-day-old rice seedlings was harvested and immediately macerated in liquid N2 in the dark prior to storage at −80°C. Frozen samples were mechanically ground with TissueLyser II (QIAGEN) into frozen powder, and extracted using the Maxwell® RSC Plant RNA kit (Promega, AS1500) according to the manufacturer's instructions. cDNA synthesis was done using the NZY First-Strand cDNA Synthesis kit (NZYtech, MB12502), and cDNA samples were used for real-time PCR (RT-PCR) (Light Cycler 480; Roche) with SYBR Green I Master (Roche Life Science) following previously described procedures [27]. For gene expression analyses, three independent biological replicates were assayed, each with two technical repeats. Primers are described in electronic supplementary material, table S1.
(c). Pigment extraction and quantification
For fresh weight chlorophyll (Chl) extraction, rice shoot samples were weighed, frozen and ground in liquid nitrogen. Samples were extracted in extraction buffer (45% ethanol, 45% acetone and 10% water) and then shaken for 3 h in the dark at 4°C. Samples were centrifuged at 12 000g for 5 min, and the supernatant absorbance was measured at 645 nm and 663 nm in a Spectramax M3 plate reader (Molecular Devices). Chl content was calculated according to Arnon [33], following the equations Chl A = 12.72 × A663 − 2.59 × A645; Chl B = 22.88 × A645 − 4.67 × A663.
For dry weight Chl and carotenoid extraction, samples were ground and freeze-dried overnight in the dark using a freeze-dryer ALPHA 2–4 LD machine (at −47°C and 0.055 mbar). Each sample was weighed and extracted in pre-cooled 80% acetone for 1 h at 4°C in the dark. Absorbance was measured at 663, 647 and 470 nm using a UV–Vis spectrophotometer (UV-2600, SHIMADZU). Chl and carotenoid content were calculated according to Lichtenthaler [34]: Chl A = 12.25 × A663 − 2.79 × A647; Chl B = 21.50 × A647 − 5.10 × A663; total Chl = 7.15 × A663 + 18.71 × A647 or Chl A + Chl B; carotenoids = (1000 × A470 − 1.82 × Chl A − 85.02 × Chl B)/198.
(d). Data analyses and statistics
R, Rstudio program and GraphPad Prism v. 7 for Mac (GraphPad Software, La Jolla, CA, USA) were used to perform the data analyses and visualization. Seedlings whose total length was less than 0.4 times the mean length of their biological replicate were eliminated as poorly germinating seeds. Morphological data were analysed by one-way ANOVA, and the mean difference of each treatment in the multiple comparisons was calculated using Tukey's test. For two-group comparisons, such as low and HL treatment, or with and without lincomycin treatment, Student's t-test was used. For gene expression and Chl content comparisons, data are the means ± s.e.m of biological triplicates (n = 3). Statistically significant differences were defined and labelled with *, ** and ***, corresponding to p-values < 0.05, <0.01 and <0.001, respectively.
(e). Fluorescence measurements
In vivo Chl fluorescence was measured at room temperature using a pulse modulated amplitude fluorimeter (MAXI-IMAGING-PAM, Heinz Waltz GmbH, Germany). Photosynthetic parameters including the ratio of variable to maximum fluorescence (Fv/Fm), and NPQ) were assessed as described elsewhere [35]. Briefly, seedlings were first dark-adapted for 30 min. Next, a blue measuring light (450 nm, 0.5 µmol photons m−2 s−1) was turned on and a very short saturating pulse (SP) (800 ms, 2700 µmol photons m−2 s− 1, 450 nm) was applied. Fluorescence signal before (Fo) and after (Fm) the SP was recorded to estimate the maximum quantum yield of photosystem II (PSII) (Fv/Fm), defined as Fv/Fm = (Fm − Fo)dark/Fmdark [36]. NPQ of Chl fluorescence was then measured by exposing dark-adapted seedlings to 800 µmol photons m−2 s−1 for 10 min, followed by a 70 min dark recovery period. NPQ was calculated as NPQ = (Fm − F′m)/F′m [37], where Fm corresponds to the maximum fluorescence from dark-adapted plants during a SP, and F'm to maximum fluorescence after SP applied every minute in the light as well as in the dark recovery period.
3. Results
(a). Lincomycin-induced retrograde signalling blocks pigment accumulation in rice and prevents growth and full separation of the second leaf
Rice seedlings were grown under continuous darkness (D), low (40 µmol m−2 s−1) red light (R), low (10 µmol m−2 s−1) blue light (B) and low (40 µmol m−2 s−1) white light (W) for 5 days (figure 1a,b). Light intensities were selected based on similar coleoptile lengths. Different photomorphogenic features (coleoptile length, coleoptile angle, first and second leaf length, and leaf declination) were quantified as described in electronic supplementary material, figure S1. Dark-grown seedlings exhibited vertical (angle approx. 6o) elongated coleoptiles (2.1 cm) around the first and second leaves, which were 3.4 and 4.3 cm long, respectively (figure 1a,b). Light dramatically inhibited coleoptile elongation (to approx. 0.6 cm) and the growth of the first leaf (to 1.6 cm) to a similar extent in all three different light conditions (figure 1a,b). By contrast, elongation of the second leaf was inhibited in R and W compared with D (to approx. 2.7 cm), but not in B. Light also promoted opening of the coleoptile in all light conditions tested, and B and W were more efficient than R (to 72°, 69° and 55°, respectively) (figure 1a,b). Additionally, we observed that light-induced Chl accumulation was approximately 2.6-fold higher in B compared with R (figure 1c). These data are in accordance with previous reports [3,4,38] and illustrate how light affects early rice seedling photomorphogenesis.
Figure 1.
Effect of lincomycin treatment on light-regulated development of rice seedlings under continuous light. (a) Representative 5-day-old rice seedlings grown under continuous dark (D), red light (R) (40 µmol m−2 s−1), blue light (B) (10 µmol m−2 s−1) and white light (W) (40 µmol m−2 s−1) at 25°C with or without lincomycin (linco., 2 mM) treatment. (b) Quantification of coleoptile angle, and coleoptile, first and second leaf length in 5-day-old rice seedlings (n = 16) under the conditions indicated in (a). (c) Chlorophyll content in R and B light-grown seedlings with or without lincomycin (d). GLK1 and LHCB and expression in seedlings grown as detailed in (a). Gene expression and chlorophyll data correspond to the means ± s.e.m. of independent biological triplicates. Multiple groups in (b) were analysed by one-way ANOVA. Different letters denote statistically significant differences among means of each group by Tukey's test. Student's t-test was used in (b), (c) and (d) for the analysis between lincomycin-treated and non-treated groups in each light condition. Asterisks denote statistically significant differences: p-values <0.05 (*), <0.01 (**) and <0.001 (***). Black dots in (c) and (d) denote individual data points.
To investigate how RS might affect this process, we first defined the optimal lincomycin concentration to use. Rice seedlings grown in W were treated from germination onwards with 0.25, 0.5, 1 and 2 mM lincomycin in 0.5 MS medium without sucrose. Seedlings showed a progressive decrease in pigmentation and were completely white in 2 mM lincomycin, which was chosen for further experiments (electronic supplementary material, figure S2). At this concentration, we confirmed the block of Chl accumulation in B and R (figure 1c), which correlated with gene expression repression of OsLHCB and OsGLK1 (figure 1d), hallmarks of RS [27], confirming that lincomycin induces RS in rice in both R and B light conditions.
We next compared the morphology of lincomycin-treated with non-treated control seedlings grown under continuous D, R, B and W light conditions (figure 1a,b). Besides the abovementioned blockage of pigment accumulation, lincomycin increased the coleoptile angle in light-grown seedlings, which was significant in B and W, with an increase of approximately 20°. The treatment also had an effect on the growth of the first and second leaves in R and B, but not in W. Interestingly, lincomycin increased leaf length in R and the effect was the opposite in B. Finally, RS did not affect coleoptile length in the dark or in any of the light treatments (figure 1a,b), which might be expected as the coleoptile does not develop functional chloroplasts.
To complete this characterization, we also analysed the effect of light and lincomycin in 3 day dark-grown seedlings transferred to R and B for 4 days (figure 2). Under these conditions, B light inhibited the elongation of the first and second leaves more efficiently than R, and lincomycin further repressed this growth (as observed above in continuous B conditions), whereas it did not have any significant effect in D or R light seedlings. We also observed separation of the second leaf specifically in B light, a response that has been described to increase with higher B intensities [5]. Indeed, we observed an increase in the second leaf angle declination when seedlings were transferred to high B (150 µmol m−2 s−1) compared with low B (10 µmol m−2 s−1). Interestingly, this increase was significantly affected in the presence of lincomycin (figure 2c; electronic supplementary material, figure S3).
Figure 2.
Effect of lincomycin treatment on light-regulated development of dark-grown rice seedlings transferred to light. (a) Representative 7-day-old rice seedlings grown under continuous dark (D), or under D for 3 days and then transferred to red light (40 µmol m−2 s−1) (DR) or blue light (10 µmol m−2 s−1) (DB) for 4 days at 25°C with or without lincomycin (linco., 2 mM). (b) Quantification of coleoptile angle, and coleoptile, first and second leaf length in rice seedlings (n = 16) grown under the conditions indicated in (a). (c) Representative 7-day-old rice seedlings grown under dark for 3 days and then transferred to low blue light (10 µmol m−2 s−1) (DB10) or high blue light (150 µmol m−2 s−1) (DB150) for 4 days at 28°C with or without lincomycin. (d) Quantification of second leaf angle and third leaf length in rice seedlings (n = 16) grown under the conditions indicated in (c). Multiple groups in (b) were analysed by one-way ANOVA. Different letters denote statistically significant differences among means of each group by Tukey's test. Student's t-test was used in (b) and (d) for mean comparison between two groups. Asterisks denote statistically significant differences: p-values <0.05 (*), <0.01 (**) and <0.001 (***).
Together, these results indicate that lincomycin-induced chloroplast damage impacts two main processes during rice seedling deetiolation: (1) it blocks pigment accumulation, and (2) although it has modest morphological effect in the overall growth of the emerging seedling, it prevents growth and full separation of the second leaf. These results contrast with Arabidopsis, where lincomycin-treated seedlings resemble dark-grown seedlings [27]. Interestingly, however, the observed inhibition of the second leaf growth and declination in rice, albeit of less importance, is reminiscent of the inhibition of cotyledon separation, the main developmental effect of RS in Arabidopsis seedlings [27,39].
(b). High light treatment uncovers distinct effects of high red and high blue light in the photomorphogenesis of rice seedlings
The above results investigate the effects of monochromatic light on RS in lincomycin-treated seedlings. However, because we wished to understand how each light quality by itself impacts development through RS, we decided to use high-intensity B and R light in untreated seedlings. To this end, we started by examining the effects on photomorphogenesis of rice seedlings subjected to continuous low (40 µmol m−2 s−1) and high (450 µmol m−2 s−1) R light, and low (10 µmol m−2 s−1) and high (150 µmol m−2 s−1) B light. High R and B intensities resulted in similar coleoptile length (figure 3a,b). High R blocked Chl accumulation as described before [40], whereas, remarkably, high B did not (figure 3a,c). Even seedlings grown in higher B intensities (300 and 450 µmol m−2 s−1) were green (electronic supplementary material, figure S4). This result suggested that high B and R might have different capacities to induce RS. Indeed, high R repressed the expression level of LHCB and GLK1, whereas high B did not significantly impact their expression compared with low B (figure 3d), indicating that high R light but not high B can induce chloroplast damage triggering RS under these conditions. Interestingly, high R did not produce any apparent morphological change other than lack of pigmentation, whereas high B induced clear inhibition of growth (figure 3a,b), an effect similar (albeit of weaker magnitude) to that induced by lincomycin (figures 1 and 2). This inhibition of growth was stronger in higher B light intensities (electronic supplementary material, figure S4). These results suggest that high B might induce a RS that selectively affects growth but not pigmentation. Alternatively, the inhibition of growth under these conditions might be independent of RS (e.g. only photoreceptor-mediated).
Figure 3.
Effect of continuous high-intensity red and blue light on seedling development. (a) Representative 5-day-old rice seedlings grown under low (40 µmol m−2 s−1) (R40) or high (450 µmol m−2 s−1) (R450) red (R) light, and low (10 µmol m−2 s−1) (B10) or high (150 µmol m−2 s−1) (B150) blue (B) light at 25°C. (b) Quantification of coleoptile angle, and coleoptile, first and second leaf length in rice seedlings (n = 16) grown under the conditions indicated in (a). Chlorophyll content in seedlings grown under the conditions indicated in (a). (d) GLK1 and LHCB and expression in seedlings grown as detailed in (a). Gene expression and chlorophyll data correspond to the means ± s.e.m. of independent biological triplicates. Student's t-test was used in (b) and (d) for mean comparison between two groups. Asterisks denote statistically significant differences: p-values <0.05 (*), <0.01 (**) and <0.001 (***). Black dots in (d) denote individual data points.
Again, to complete this characterization, we examined the phenotype of dark-grown seedlings transferred to HL (figure 4). In seedlings grown in the dark for 3 days, transfer to high R for 4 days prevented Chl accumulation, while transfer to high B did not (figure 4a,b), similar to what we observed in continuous HL (figure 3; electronic supplementary material, figure S4). No other phenotype was observed for high R-grown seedlings (figure 4a). In high B, we again observed inhibition of growth, resulting in shorter leaf lengths (figure 4b,c; electronic supplementary material, figure S3). As discussed above (figure 2c), second leaf declination was promoted by high B (figure 4b,c; electronic supplementary material, figure S3).
Figure 4.
Effect of high-intensity red and blue light on the development of dark-grown rice seedlings transferred to high light. (a) Representative 7-day-old rice seedlings grown under dark (D) for 3 days and then transferred to low (40 µmol m−2 s−1) (DR40) or high (450 µmol m−2 s−1) (DR450) red (R) light at 25°C. (b) Representative 7-day-old rice seedlings grown under dark (D) for 3 days and then transferred to low (10 µmol m−2 s−1) (DB10) or high (150 µmol m−2 s−1) (DB150) blue (B) light at 28°C. (c) Quantification of coleoptile and second leaf angle, and coleoptile, first, second and third leaf length in rice seedlings (n = 16) grown under the conditions indicated in (b). Student's t-test was used for mean comparison between two groups. Asterisks denote statistically significant differences: p-values <0.05 (*), <0.01 (**) and <0.001 (***).
Together, these results suggest that during rice seedling deetiolation, high R induces chloroplast damage triggering RS which results in a blockage of pigment accumulation while having little effect on morphology, whereas high B does not induce repression in the expression of the RS markers LHCB and GLK1 under these conditions, but clearly inhibits elongation growth. To our knowledge, this direct comparison between the effects of high R and high B in the accumulation of pigments and morphology is novel in rice or any other model plant.
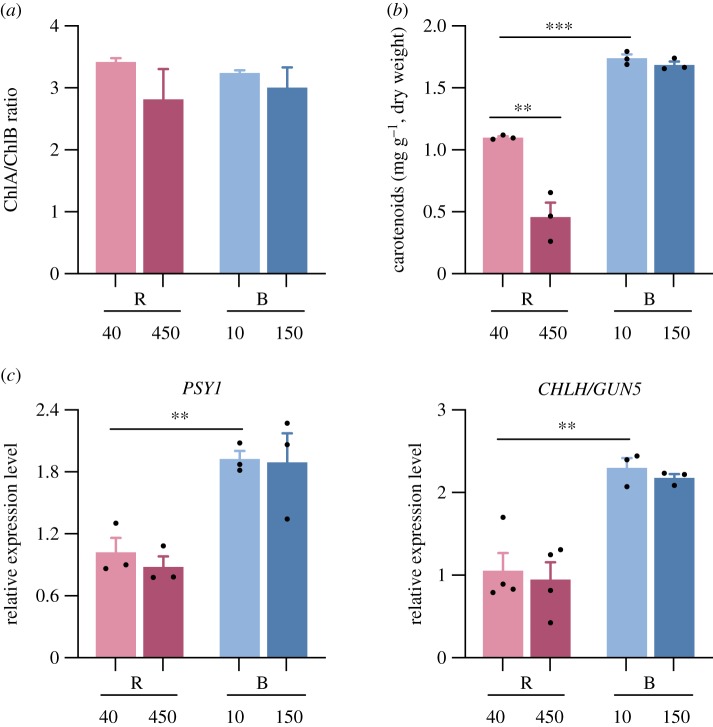
(c). Blue light triggers higher accumulation of chlorophylls and carotenoids in rice seedlings compared with red light
Our results above prompted us to investigate further the differences in pigment accumulation between seedlings grown in R and B. Interestingly, Chl levels were higher in B compared with R, in both low and high fluences (figures 1c and 3c), with similar Chl A/B ratio (figure 5a). Carotenoid quantification (figure 5b) revealed that carotenoid content in low B was also approximately 1.7-fold higher than that in low R, and did not change in high B, whereas high R induced blockage of carotenoid accumulation similar to what we observed for Chl (figure 3c). In accordance with high pigment levels in B light-grown seedlings compared with R, expression of genes encoding the enzymes phytoene synthase 1 (PSY1), which catalyses the carotenoid biosynthesis rate-limiting step, and Mg2-chelatase/genomes uncoupled 5 (CHLH/GUN5), involved in Chl synthesis [38,41], showed higher expression levels in B compared with R (figure 5c). These results are in general agreement with described light-specific transcriptional signatures of rice metabolism [42]. It is interesting to note that Nipponbare, the rice cultivar used in this work, lacks OsC1 function, an R2R3-MYB gene necessary for OsCHS1 expression and anthocyanin production [43]. Thus, our seedlings do not accumulate anthocyanin even in B light, conditions where flavonoid production in cotyledons and leaves is enhanced in Arabidopsis or commonly seen in wild rice varieties [44].
Figure 5.
Red and blue light differently affect pigment accumulation. Rice seedlings were grown for 5 days under low (40 µmol m−2 s−1) (R40) or high (450 µmol m−2 s−1) (R450) red (R) light, and low (10 µmol m−2 s−1) (B10) or high (150 µmol m−2 s−1) (B150) blue (B) light at 25°C. (a) Quantification of ChA/ChlB ratio (total content is shown in figure 3c). (b) Quantification of carotenoid content. (c) Relative gene expression of PSY1 and CHLH/GUN5. Data are the means ± s.e.m of independent biological triplicates. Means were compared by Student's t-test, and asterisks denote statistically significant differences: p-values <0.01 (**) and <0.001 (***). Black dots in (b) and (c) denote individual data points. (Online version in colour.)
(d). Photosynthesis efficiency and photoprotection in high red and high blue light
Given the differences in pigmentation between R and B light-grown seedlings (figures 3–5), we hypothesized that each light quality might have a different impact on the photosynthetic efficiency and photoprotective capacities of the seedling. We first measured the maximum quantum yield of PSII (Fv/Fm), as an indication of PSII functionality. Fv/Fm levels of high B light-grown seedlings were unaffected compared with low B treatments (figure 6a), even in higher B light intensities (average Fv/Fm of 0.6–0.8) (electronic supplementary material, figure S5). Interestingly, seedlings grown in low R displayed Fv/Fm levels similar to low and high B (figure 6a), even though the total Chl content and carotenoid content were significantly reduced in seedlings grown in low R compared with B (figures 1c, 3c and 5b). Finally, as expected owing to the lack of pigments, seedlings grown in high R displayed greatly reduced Fv/Fm levels, on average 0.3, indicative of photoinhibition (figure 6a).
Figure 6.
Photosynthesis efficiency and photoprotection capacities of rice seedlings grown in red and blue light. Rice seedlings were grown for 6 days under low (40 µmol m−2 s−1) (R40) or high (450 µmol m−2 s−1) (R450) red (R) light, and low (10 µmol m−2 s−1) (B10) or high (150 µmol m−2 s−1) (B150) blue (B) light at 28°C. (a) Maximum quantum yield of PSII (Fv/Fm). Values refer to four independent seedlings. Multiple groups were analysed by one-way ANOVA. Different letters denote statistically significant differences among means of each group by Tukey's test. (b) Representative coloured images of non-photochemical quenching (NPQ) in the second leaf data shown in (c). The colour scale is shown on the right. Left panel: NPQ induction after 5 min in high light. Right panel: NPQ relaxation after 20 min in the dark. (c) NPQ induction (light) and relaxation (dark). Values are the means ± s.d. for six independent seedlings grown under the specified light conditions. (d) Relative gene expression of PSBS1 and PSBS2. Data are the means ± s.e.m of independent biological triplicates. Means were compared by Student's t-test, and asterisks denote statistically significant differences: p-values <0.05 (*), and <0.001 (***). Black dots in (d) denote individual data points.
Because we detected a decrease of Fv/Fm specifically in high R but not in high B, suggestive of a differential capacity in activating photoprotective mechanisms, we next tested if light quality had an effect on the induction of NPQ, one of the main photoprotective strategies against HL in plants. Low B and R light-grown seedlings were dark-adapted for 30 min, then exposed to 800 µmol m−2 s−1 of actinic light for 10 min, and finally kept in the dark for 70 min to observe the induction and relaxation of NPQ. Strikingly, NPQ dynamics after exposure to HL were remarkably different in low B light-grown seedlings compared with low R light (figure 6b,c). First, B light-grown seedlings showed significantly higher maximum NPQ levels in the light compared with R, and they were also faster in their response. Second, dark relaxation of fluorescence quenching was incomplete in R light-grown seedlings compared with B, indicating again photoinhibition. These NPQ values in B light-grown seedlings were not significantly changed in seedlings grown in higher B light intensities (electronic supplementary material, figure S5). A more complete graph showing a longer dark relaxation period and levels of qE (energy quenching) and qI (photoinhibitory quenching) components is presented in electronic supplementary material, figure S6. These NPQ differences correlated with the observed differences in carotenoid content (figure 5b). Moreover, transcript levels of the qE effector PSBS appeared also to be light regulated. In rice, PSBS protein is encoded by two genes, PSBS1 and PSBS2 [45]. In agreement with a role in NPQ [45–47], expression of both genes was elevated in B compared with R light-grown seedlings (figure 6d).
4. Discussion
Our findings support a scenario in which RS in rice plays distinct roles under red and blue light conditions to fine-tune photomorphogenesis and photoprotection under potentially photodamaging conditions. Using monochromatic lights of different intensities and in combination with lincomycin treatment, we have uncovered a possible role for RS in rice to inhibit leaf growth and declination. Furthermore, our comparative data in R and B lights suggest that, whereas high R induces RS and can cause photobleaching, B light induces higher carotenoid and Chl contents and higher PSBS expression, and might equip plants with the capacity to withstand HL, as exemplified by increased NPQ levels compared with R.
We show that, remarkably, disruption of chloroplast function in rice seedlings by treatment with lincomycin from germination onwards only affected the rice photomorphogenic development modestly (figures 1a,b and 2). This is in clear contrast to Arabidopsis, where lincomycin blocks deetiolation and seedlings resemble dark-grown seedlings, with lack of pigmentation and appressed cotyledons [27]. However, our observation that in B light-grown seedlings lincomycin inhibited the length and the small increase in declination angle of the second leaf once it unfolded (figure 2c,d) indicates that RS can reduce the area exposed to light, which might protect the seedling from damage in an HL environment. Together with previous results in Arabidopsis showing RS inhibition of cotyledon separation [27], we suggest that a common role of biogenic RS in dicots and monocots might be to minimize the area of the seedling exposed to potentially damaging light, by inhibiting cotyledon separation or seedling growth and second leaf length and declination.
Our data showing that high B and R lights have different effects on seedling pigmentation and development suggest that both lights induce distinct processes and might have different capacity to induce chloroplast damage in rice. This effect is probably at least partially masked when using monochromatic light in the presence of lincomycin, as evident in experiments under continuous B light with or without lincomycin: B light by itself, even of intensities high enough to inhibit growth almost completely, did not seem to affect greening, whereas addition of lincomycin in low B completely blocked seedling greening (figures 1a, 2a,c, 3a,c and 4b; electronic supplementary material, figure S3). Therefore, the use of monochromatic HL has allowed us to dissect the ability of each wavelength to cause chloroplast damage and induce RS in rice. Interestingly, high R light inhibited pigment accumulation compared with low R, but seedlings otherwise looked unaffected (figures 3a,b and 4a). This suggests that RS induced by high R light specifically affects pigment accumulation and might not participate in tuning morphogenesis to the prevailing HL. By contrast, high B light-grown seedlings were able to accumulate pigments but were overall shorter (figures 3a,b and 4b,c). This elongation inhibition is similar to the effect that we saw with lincomycin (figures 1a,b and 2). However, because our results suggest that RS was probably not induced under this high B condition (figure 3d), and growth inhibition also took place in the presence of lincomycin (figures 1a,b and 2), our interpretation is that this effect might be photoreceptor-mediated and can take place whether RS is activated or not. Intriguingly, studies in Arabidopsis have suggested that plastid signals that affect photomorphogenesis are dependent on cryptochrome 1 [39].
Our data show a clear difference in the NPQ response when plants were grown in continuous B or R light. B light-grown seedlings were able to induce higher NPQ levels, which correlated with higher transcript levels of the qE effector protein PSBS (figure 6b–d). Indeed, higher PSBS levels have been shown to increase qE capacity in many photosynthetic organisms, including rice [46]. At the same time, B light-grown seedlings also showed increased levels of total carotenoids compared with R-grown plants (figure 5b). Whether the increased NPQ in B light compared with R is due to enhanced PSBS expression and/or enhanced carotenoid content in B remains to be further studied. Interestingly, in the green microalga Chlamydomonas reinhardtii, B light (and also UV-B independently) controls NPQ induction through photoreceptor-mediated regulation of the expression of the main qE effectors [48,49], which established for the first time a molecular link between photoreception and photoprotection. In this context, it is tempting to speculate that we have also uncovered a novel control of photoprotection by B light in higher plants that deserves further investigation. On the other hand, low R light-grown seedlings showed reduced NPQ levels, lower PBSB gene expression and lower carotenoid content compared with low B light-grown plants (figures 6b–d and 5b). When grown in high R light, seedlings showed a complete albino phenotype (figure 3a), possibly as a consequence of decreased photoprotective capacities. Interestingly, low B light treatment allowed Chl and carotenoid content restoration in high R-grown rice seedlings [40], suggesting a requirement for B light signalling in seedling photoprotection. Altogether, our results showing increased photoprotective capacities in B compared with R may explain why, under the conditions used, B light-grown rice seedlings may withstand HL without chloroplast damage and RS induction.
In Arabidopsis, we have previously shown that the RS regulating photomorphogenesis is mediated by GUN1, a plastid-localized member of the pentatricopeptide repeat family [25], and by Golden2-like 1 (GLK1), a transcription factor involved in the promotion of chloroplast development [27]. In rice, ectopic overexpression of OsGLK1 was shown to induce chloroplast biogenesis in non-green cells, but the work was mostly done in calli and did not address possible morphogenic implications [50]. As for GUN1, no rice mutants are available to date. Future work will address the molecular players of HL-induced RS in rice and how they mediate a different response to R and B light. Upcoming studies will also aim to decipher how B and R light-induced signals are mediated and interact to provide photoprotection, a relevant question in natural environments.
Supplementary Material
Acknowledgements
We thank Giovanni Finazzi (Université Grenoble Alpes) for his comments on the manuscript.
Data accessibility
This article has no additional data.
Authors' contributions
E.M., L.D. and M.A.R.-S. designed the work and analysed data. L.D., M.A.R.-S., A.C. and N.V. acquired the data. E.M., L.D. and M.A.R.-S. wrote the manuscript. All authors revised and approved the manuscript's content.
Competing interests
The authors declare no competing interests.
Funding
This work was supported by grants from FEDER/Ministerio de Ciencia, Innovación y Universidades – Agencia Estatal de Investigación (project references BIO2015-68460-P and PGC2018-099987-B-I00) and from the CERCA Programme/Generalitat de Catalunya (project reference 2017SGR-718) to E.M. We acknowledge financial support from the Spanish Ministry of Economy and Competitiveness, through the ‘Severo Ochoa Programme for Centres of Excellence in R&D' 2016–2019 (SEV-2015- 0533).
References
- 1.Mathews S, Sharrock RA. 1996. The phytochrome gene family in grasses (Poaceae): a phylogeny and evidence that grasses have a subset of the loci found in dicot angiosperms. Mol. Biol. Evol. 13, 1141–1150. ( 10.1093/oxfordjournals.molbev.a025677) [DOI] [PubMed] [Google Scholar]
- 2.Matsumoto N, Hirano T, Iwasaki T, Yamamoto N. 2003. Functional analysis and intracellular localization of rice cryptochromes. Plant Physiol. 133, 1494–1503. ( 10.1104/pp.103.025759) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhang YC, Gong SF, Li QH, Sang Y, Yang HQ. 2006. Functional and signaling mechanism analysis of rice CRYPTOCHROME 1. Plant J. 46, 971–983. ( 10.1111/j.1365-313X.2006.02753.x) [DOI] [PubMed] [Google Scholar]
- 4.Takano M, Kanegae H, Shinomura T, Miyao A, Hirochika H, Furuya M. 2001. Isolation and characterization of rice phytochrome A mutants. Plant Cell 13, 521–534. ( 10.1105/tpc.13.3.521) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Takano M, et al. 2005. Distinct and cooperative functions of phytochromes A, B, and C in the control of deetiolation and flowering in rice. Plant Cell 17, 3311–3325. ( 10.1105/tpc.105.035899) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hirose F, Shinomura T, Tanabata T, Shimada H, Takano M. 2006. Involvement of rice cryptochromes in de-etiolation responses and flowering. Plant Cell Physiol. 47, 915–925. ( 10.1093/pcp/pcj064) [DOI] [PubMed] [Google Scholar]
- 7.Takano M, Inagaki N, Xie X, Kiyota S, Baba-Kasai A, Tanabata T, Shinomura T.. 2009. Phytochromes are the sole photoreceptors for perceiving red/far-red light in rice. Proc. Natl Acad. Sci. USA 106, 14 705–14 710. ( 10.1073/pnas.0907378106). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gommers CMM, Monte E. 2018. Seedling establishment: a dimmer switch-regulated process between dark and light signaling. Plant Physiol. 176, 1061–1074. ( 10.1104/pp.17.01460) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Matsuda R, Ohashi-Kaneko K, Fujiwara K, Goto E, Kurata K. 2004. Photosynthetic characteristics of rice leaves grown under red light with or without supplemental blue light. Plant Cell Physiol. 45, 1870–1874. ( 10.1093/pcp/pch203) [DOI] [PubMed] [Google Scholar]
- 10.Muller P, Li XP, Niyogi KK. 2001. Non-photochemical quenching. A response to excess light energy. Plant Physiol. 125, 1558–1566. ( 10.1104/pp.125.4.1558) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kulheim C, Agren J, Jansson S. 2002. Rapid regulation of light harvesting and plant fitness in the field. Science (New York) 297, 91–93. ( 10.1126/science.1072359) [DOI] [PubMed] [Google Scholar]
- 12.Kromdijk J, Glowacka K, Leonelli L, Gabilly ST, Iwai M, Niyogi KK, Long SP. 2016. Improving photosynthesis and crop productivity by accelerating recovery from photoprotection. Science (New York) 354, 857–861. ( 10.1126/science.aai8878) [DOI] [PubMed] [Google Scholar]
- 13.Li XP, Bjorkman O, Shih C, Grossman AR, Rosenquist M, Jansson S, Niyogi KK. 2000. A pigment-binding protein essential for regulation of photosynthetic light harvesting. Nature 403, 391–395. ( 10.1038/35000131) [DOI] [PubMed] [Google Scholar]
- 14.Ruban AV. 2016. Nonphotochemical chlorophyll fluorescence quenching: mechanism and effectiveness in protecting plants from photodamage. Plant Physiol. 170, 1903–1916. ( 10.1104/pp.15.01935) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tikkanen M, Mekala NR, Aro EM. 2014. Photosystem II photoinhibition-repair cycle protects Photosystem I from irreversible damage. Biochim. Biophys. Acta 1837, 210–215. ( 10.1016/j.bbabio.2013.10.001) [DOI] [PubMed] [Google Scholar]
- 16.Li HM, Chiu CC. 2010. Protein transport into chloroplasts. Annu. Rev. Plant Biol. 61, 157–180. ( 10.1146/annurev-arplant-042809-112222) [DOI] [PubMed] [Google Scholar]
- 17.Jung HS, Chory J. 2010. Signaling between chloroplasts and the nucleus: can a systems biology approach bring clarity to a complex and highly regulated pathway? Plant Physiol. 152, 453–459. ( 10.1104/pp.109.149070) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kleine T, Voigt C, Leister D. 2009. Plastid signalling to the nucleus: messengers still lost in the mists? Trends Genet. 25, 185–192. ( 10.1016/j.tig.2009.02.004) [DOI] [PubMed] [Google Scholar]
- 19.Jarvis P, Lopez-Juez E. 2013. Biogenesis and homeostasis of chloroplasts and other plastids. Nat. Rev. Mol. Cell Biol. 14, 787–802. ( 10.1038/nrm3702) [DOI] [PubMed] [Google Scholar]
- 20.Waters MT, Wang P, Korkaric M, Capper RG, Saunders NJ, Langdale JA. 2009. GLK transcription factors coordinate expression of the photosynthetic apparatus in Arabidopsis. Plant Cell 21, 1109–1128. ( 10.1105/tpc.108.065250) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ruckle ME, DeMarco SM, Larkin RM. 2007. Plastid signals remodel light signaling networks and are essential for efficient chloroplast biogenesis in Arabidopsis. Plant Cell 19, 3944–3960. ( 10.1105/tpc.107.054312) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kindgren P, Kremnev D, Blanco NE, de Dios Barajas Lopez J, Fernandez AP, Tellgren-Roth C, Kleine T, Small I, Strand A. 2012. The plastid redox insensitive 2 mutant of Arabidopsis is impaired in PEP activity and high light-dependent plastid redox signalling to the nucleus. Plant J. 70, 279–291. ( 10.1111/j.1365-313X.2011.04865.x) [DOI] [PubMed] [Google Scholar]
- 23.Oelmuller R, Levitan I, Bergfeld R, Rajasekhar VK, Mohr H. 1986. Expression of nuclear genes as affected by treatments acting on the plastids. Planta 168, 482–492. ( 10.1007/BF00392267) [DOI] [PubMed] [Google Scholar]
- 24.Sullivan JA, Gray JC. 1999. Plastid translation is required for the expression of nuclear photosynthesis genes in the dark and in roots of the pea lip1 mutant. Plant Cell 11, 901–910. ( 10.1105/tpc.11.5.901) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Koussevitzky S, Nott A, Mockler TC, Hong F, Sachetto-Martins G, Surpin M, Lim J, Mittler R, Chory J. 2007. Signals from chloroplasts converge to regulate nuclear gene expression. Science (New York) 316, 715–719. ( 10.1126/science.1140516) [DOI] [PubMed] [Google Scholar]
- 26.Chan KX, et al. 2016. Sensing and signaling of oxidative stress in chloroplasts by inactivation of the SAL1 phosphoadenosine phosphatase. Proc. Natl Acad. Sci. USA 113, E4567–E4576. ( 10.1073/pnas.1604936113). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Martin G, Leivar P, Ludevid D, Tepperman JM, Quail PH, Monte E. 2016. Phytochrome and retrograde signalling pathways converge to antagonistically regulate a light-induced transcriptional network. Nat. Commun. 7, 11431 ( 10.1038/ncomms11431) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hills AC, Khan S, Lopez-Juez E. 2015. Chloroplast biogenesis-associated nuclear genes: control by plastid signals evolved prior to their regulation as part of photomorphogenesis. Front. Plant Sci. 6, 1078 ( 10.3389/fpls.2015.01078) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang Y, et al. 2011. A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods 7, 30 ( 10.1186/1746-4811-7-30) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Li RQ, Jiang M, Liu YH, Zheng YC, Huang JZ, Wu JM, Shu QY. 2017. The xantha marker trait is associated with altered tetrapyrrole biosynthesis and deregulated transcription of PhANGs in rice. Front. Plant Sci. 8, 901 ( 10.3389/fpls.2017.00901) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nishiyama T, et al. 2018. The Chara genome: secondary complexity and implications for plant terrestrialization. Cell 174, 448–464.e424. ( 10.1016/j.cell.2018.06.033) [DOI] [PubMed] [Google Scholar]
- 32.Schneider CA, Rasband WS, Eliceiri KW. 2012. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675. ( 10.1038/nmeth.2089) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Arnon DI. 1949. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 24, 1–15. ( 10.1104/pp.24.1.1) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lichtenthaler HK. 1987. Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol. 148, 350–382. ( 10.1016/0076-6879(87)48036-1) [DOI] [Google Scholar]
- 35.Maxwell K, Johnson GN. 2000. Chlorophyll fluorescence--a practical guide. J. Exp. Bot. 51, 659–668. ( 10.1093/jexbot/51.345.659) [DOI] [PubMed] [Google Scholar]
- 36.Genty B, Briantais JM, Baker NR. 1989. The relationship between the quantum yield of photosynthetic electron-transport and quenching of chlorophyll fluorescence. Biochim. Biophys. Acta 990, 87–92. ( 10.1016/S0304-4165(89)80016-9) [DOI] [Google Scholar]
- 37.Bilger W, Bjorkman O. 1990. Role of the xanthophyll cycle in photoprotection elucidated by measurements of light-induced absorbance changes, fluorescence and photosynthesis in leaves of Hedera canariensis. Photosynth. Res. 25, 173–185. ( 10.1007/BF00033159) [DOI] [PubMed] [Google Scholar]
- 38.Tran LH, Jung S. 2017. Effects of light-emitting diode irradiation on growth characteristics and regulation of porphyrin biosynthesis in rice seedlings. Int. J. Mol. Sci. 18, 641 ( 10.3390/ijms18030641) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ruckle ME, Larkin RM. 2009. Plastid signals that affect photomorphogenesis in Arabidopsis thaliana are dependent on genomes uncoupled 1 and cryptochrome 1. New Phytol. 182, 367–379. ( 10.1111/j.1469-8137.2008.02729.x) [DOI] [PubMed] [Google Scholar]
- 40.Roy A, Sahoo D, Tripathy BC. 2013. Involvement of phytochrome A in suppression of photomorphogenesis in rice seedling grown in red light. Plant Cell Environ. 36, 2120–2134. ( 10.1111/pce.12099) [DOI] [PubMed] [Google Scholar]
- 41.Jung KH, et al. 2008. Identification and functional analysis of light-responsive unique genes and gene family members in rice. PLoS Genet. 4, e1000164 ( 10.1371/journal.pgen.1000164) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lakshmanan M, Lim SH, Mohanty B, Kim JK, Ha SH, Lee DY. 2015. Unraveling the light-specific metabolic and regulatory signatures of rice through combined in silico modeling and multiomics analysis. Plant Physiol. 169, 3002–3020. ( 10.1104/pp.15.01379) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chin HS, Wu YP, Hour AL, Hong CY, Lin YR. 2016. Genetic and evolutionary analysis of purple leaf sheath in rice. Rice (New York) 9, 8 ( 10.1186/s12284-016-0080-y) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zheng J, et al. 2019. Determining factors, regulation system, and domestication of anthocyanin biosynthesis in rice leaves. New Phytol. 223, 705–721. ( 10.1111/nph.15807) [DOI] [PubMed] [Google Scholar]
- 45.Zulfugarov IS, et al. 2014. Production of superoxide from Photosystem II in a rice (Oryza sativa L.) mutant lacking PsbS. BMC Plant Biol. 14, 242 ( 10.1186/s12870-014-0242-2) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hubbart S, Ajigboye OO, Horton P, Murchie EH. 2012. The photoprotective protein PsbS exerts control over CO2 assimilation rate in fluctuating light in rice. Plant J. 71, 402–412. ( 10.1111/j.1365-313X.2012.04995.x) [DOI] [PubMed] [Google Scholar]
- 47.Hubbart S, Smillie IRA, Heatley M, Swarup R, Foo CC, Zhao L, Murchie EH. 2018. Enhanced thylakoid photoprotection can increase yield and canopy radiation use efficiency in rice. Commun. Biol. 1, 22 ( 10.1038/s42003-018-0026-6) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Allorent G, Lefebvre-Legendre L, Chappuis R, Kuntz M, Truong TB, Niyogi KK, Ulm R, Goldschmidt-Clermont M.. 2016. UV-B photoreceptor-mediated protection of the photosynthetic machinery in Chlamydomonas reinhardtii. Proc. Natl Acad. Sci. USA 113, 14 864–14 869. ( 10.1073/pnas.1607695114) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Petroutsos D, et al. 2016. A blue-light photoreceptor mediates the feedback regulation of photosynthesis. Nature 537, 563–566. ( 10.1038/nature19358) [DOI] [PubMed] [Google Scholar]
- 50.Nakamura H, Muramatsu M, Hakata M, Ueno O, Nagamura Y, Hirochika H, Takano M, Ichikawa H. 2009. Ectopic overexpression of the transcription factor OsGLK1 induces chloroplast development in non-green rice cells. Plant Cell Physiol. 50, 1933–1949. ( 10.1093/pcp/pcp138) [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
This article has no additional data.