The definitive champion of the 92nd Academy Awards, best known as the 2020 Oscar’s was the Korean film 기생충; Gisaengchung, translated to English as Parasite, directed by Bong Joon-ho, that premiered earlier, also with a win, at the 2019 Cannes Film Festival (Figure 1). More good things are recently blooming from South Korea, including K-pop, the latest cell phones with ultra cameras, and last but not least, respiratory research.
Figure 1.
기생충; Gisaengchung (Parasite) 2019, film directed by Bong Joon-ho.
In this issue of Annals of Translational Medicine, the group of Yang B, and colleagues (1), reported the burden associated with two common respiratory conditions, bronchiectasis and chronic obstructive pulmonary disease (COPD). They analyzed data from the Korea National Health and Nutrition Examination Survey (KNHANES), an annual survey conducted to investigate the health and nutrition levels of the Korean population aimed to establish and evaluate national health policies. KNHANES is a landmark study, that mirrors its US namesake, which is now running in its 14th iteration (2), and other nationally representative surveys elsewhere. KNHANES is embedded within their National Health Insurance system, which is compulsory and universal, and collects all medical claim data for all Koreans. Therefore, KNHANES is having a significant advantage for epidemiological research, previously observed in the Scandinavian environment only.
Yang and colleagues (1), report that the frequency of self-reported diagnosis of bronchiectasis (0.8%) was ten times less common than spirometrically-confirmed COPD (12.6%), but the burden and costs associated with both were substantial, even though on average their bronchiectasis patients were five years younger than COPD patients. Interestingly, the observed increase with ageing seen in COPD prevalence by decade, that is of 4.2% in 40–49 yrs. old, 9.9% in 50–59 yrs. old, and 24.8% in those older than 60 yrs. was not observed in bronchiectasis, with 0.2%, 1.1% and 1.2% respectively. Indeed, a major limitation of this study is related with the universal underdiagnosis that we see in all chronic respiratory conditions, which is magnified in here by the absence of imaging to identify bronchiectasis, specially in a population with high rates of untreated chest infections and smoking. This problem is even of greater concern in men, with an estimated population prevalence of smoking in 2015 in South Korea of 35.5%, and even greater (38.5%) in North Korean men (3). The prevalence of bronchiectasis is very variable by country and region, not only attributable to demographic factors associated with the age- and sex-pyramid distribution of every population, as it is an age-related condition, or with variations in past infection, tuberculosis, and climate determinants, but likely due to local variations in incidence, all beyond diagnostic practices and health development. A recent literature review reported extraordinary heterogeneity in its worldwide distribution, ranging from 42 to 566 cases per 100,000 per year (4), greater in women and in the elderly, although most likely they are all underestimates. A high prevalence of bronchiectasis is reported in South Korea (4) and in one particular study of respiratory patients, 9% were deemed to have bronchiectasis, with a higher prevalence in females (5).
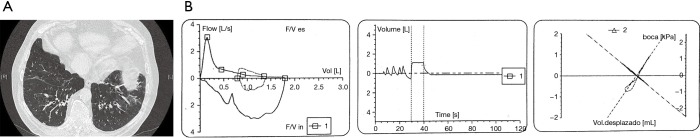
With all likelihood, the coexistence of airflow imitation compatible with COPD and bronchiectasis identified by high- or low-dose computer tomography in the same patient (Figure 2) is not testimonial. A report from Spain found a 57.6% prevalence of bronchiectasis in patients with moderate to severe COPD, and this was associated with greater severe airflow obstruction, isolation of a potentially pathogenic microorganism from sputum, and at least one hospital admission for exacerbations in the previous year (6). Very recently, a systematic review meta-analyzed six observational studies by pooling 881 COPD patients, and reported a mean prevalence of bronchiectasis in patients with COPD of 54.3%, ranging from 25.6% to 69% (7).
Figure 2.
Lung function (A) and CT-scan (B) of a patient with overlapping bronchiectasis and COPD. Male, 78 yr. old, height 156 cm, weight 84 kg, with a tobacco smoking exposure of 50 pack-yr.; Spirometry post-bronchodilator: FVC: 1,790 mL (68.5%); FEV1: 790 mL (40.2%); FEV1/FVC: 0.44; TLC: 5,300 mL (98.5%); RV: 3,320 mL (131.4%); RV/TLC: 62.6 (141%); CT scan: large, cylindrical bronchiectasis in both lower lobes, and areas of emphysema.
In South Korea in particular, as well as it is seen in other countries in Asia, Africa and South America, bronchiectasis affects mostly post-infection patients, and at a younger age than those seen in Europe (4) Further, non-compliance with vaccinations during childhood, poor tuberculosis control, toxic inhalants, substandard treatment of other respiratory infections in adulthood due to drug shortages, non-compliance and lack of access to health systems, likely causes a greater impact for bronchiectasis incidence.
The finding of a low socioeconomic status associated with both respiratory conditions highlights the unmet burden that vulnerable populations worldwide suffer in their lungs. In this study, three quarters of participants with bronchiectasis and two thirds of those with COPD had a low family income, compared with less than half in those in the control group. Worldwide, respiratory disease is on the increase, in part due to aging and population growth (8); but they are becoming an even higher toll for the poor, who overall endure more exposure to active and passive smoking, live in more polluted areas, work with unsafe air and often with poor hygiene, and lack access to preventive actions than their more priviledged counterparts (9). Recently, the Global Alliance for Chronic Disease (GACD) called for further research on the burden of all respiratory conditions and their multimorbidity (10).
A comment on spirometry in the KNHANES framework is granted. We are not told about quality control, devices, and other standards. Indeed, having spirometry measured in the general population is better than not having it, and it is an accepted KNHANES limitation they used preBD spirometry only, which likely inflates somehow the observed frequency of airflow limitation. Further, given that the control group of participants without respiratory disease had only percent predicted FVC of 92.0% and FEV1 of 93.3%, when they should be closer to 100%, and that they also report frequently respiratory symptoms linked with bronchitis (6.4% cough and 12.8% sputum), it is unknown how sensible are lung measurements. Au contraire, a plus of this study is expressing predicted values according to local reference equations (11), which is a recommendation from GOLD and should be copied/exported in other countries. Constructively, it might be considered in future iterations after this KNHANES wave 2007 to 2009, that they lobby to include postBD spirometry and imaging by low-dose computer tomography, the latter at the least in a subsample of participants.
Another interesting finding by Yang B, and colleagues (1), is on comorbidities: depression was two-fold increased in bronchiectasis than in COPD patients, although we must bear in mind it was again self-reported based on previous physician diagnosis, just like in all other comorbidities. The Global Alliance Against Chronic Respiratory Diseases (GARD) initiative recommends that anxiety and depression, among other conditions, should be actively explored in respiratory patients, due to their higher frequency, correlation with higher dyspnoea, and eventual treatment.
The bronchiectasis bacteriome in the Asia-pacific region identifies predominance of P. aeruginosa and H. influenzae. It appears that South Korea has a different distribution of bacteria in bronchiectasis patients, with high non-tuberculosis mycobacteria (44.5%), similar to that of the US and lower rates of P. aeruginosa (18.1%), and also had a significant prevalence of K. pneumoniae (12). To date, our current understanding of the virome and mycobiome in bronchiectasis in this world region is limited, beyond the exploratory search of viruses and fungi in a few studies there.
Although tobacco is indeed the most relevant causal factor for COPD development, it is not yet described as an etiological cause of bronchiectasis, although tobacco alters the mucociliary clearance system and this will favor more infections. Regrettably, respiratory and non-respiratory smoking-related disease remain a significant health risk in Korea and elsewhere, Wraping up, we can envisage that the only “parasite”, even a predator parasite, in bronchiectasis and COPD patients, either in Korea and elsewhere, is tobacco and all types of smoking, from cigarettes (13) to new forms of heat-not-burn technologies and vaping (3). They should be periodically monitored well within this 21st century to capture the changing landscape of risk factors for all chronic respiratory ailments (14). Eventually, and beyond «Parasite» we may claim: “The Oscar goes to…” anti-tobacco advocates in their fight for a world with no tobacco and therefore with less respiratory disease.
Supplementary
The article’s supplementary files as
Acknowledgments
Funding: None.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Provenance and Peer Review: This article was commissioned by the Editorial Office, Annals of Translational Medicine. The article did not undergo external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/atm.2020.03.40). The authors have no conflicts of interest to declare.
References
- 1.Yang B, Choi H, Lim JH, et al. The disease burden of bronchiectasis in comparison with chronic obstructive pulmonary disease: a national database study in Korea. Ann Transl Med 2019;7:770. 10.21037/atm.2019.11.55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.National Health and Nutrition Examination Survey - NHANES 2019-2020 Available online: https://wwwn.cdc.gov/nchs/nhanes/continuousnhanes/default.aspx?BeginYear=2019, accessed 13 February 2020.
- 3.GBD 2015 Tobacco Collaborators. Smoking prevalence and attributable disease burden in 195 countries and territories, 1990-2015: a systematic analysis from the Global Burden of Disease Study 2015. Lancet 2017;389:1885-906. 10.1016/S0140-6736(17)30819-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chandrasekaran R, Mac Aogáin M, Chalmers JD, et al. Geographic variation in the aetiology, epidemiology and microbiology of bronchiectasis. BMC Pulm Med 2018;18:83. 10.1186/s12890-018-0638-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kwak HJ, Moon JY, Choi YW, et al. High prevalence of bronchiectasis in adults: analysis of CT findings in a health screening program. Tohoku J Exp Med 2010;222:237-42. 10.1620/tjem.222.237 [DOI] [PubMed] [Google Scholar]
- 6.Martínez-García MÁ, Soler-Cataluña JJ, Donat Sanz Y, et al. Factors associated with bronchiectasis in patients with COPD. Chest 2011;140:1130-7. 10.1378/chest.10-1758 [DOI] [PubMed] [Google Scholar]
- 7.Ni Y, Shi G, Yu Y, et al. Clinical characteristics of patients with chronic obstructive pulmonary disease with comorbid bronchiectasis: a systemic review and meta-analysis. Int J Chron Obstruct Pulmon Dis 2015;10:1465-75. 10.2147/COPD.S83910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Soriano JB, Kendrick P, Paulson K, et al. Prevalence and Attributable Health Burden of Chronic Respiratory Diseases from 1990 -2017: A systematic analysis from the Global Burden of Disease Study 2017. Lancet Respir Med 2020 (in press. 20 February 2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tabyshova A, Hurst JR, Soriano JB, et al. Evidence implementation gaps in COPD guidelines: systematic comparison of low- and middle-income versus highincome countries 2020. Eur Respir J 2020 (in press. 2 March 2020). [Google Scholar]
- 10.Hurst JR, Dickhaus J, Maulik PK, et al. Global Alliance for Chronic Disease researchers' statement on multimorbidity. Lancet Glob Health 2018;6:e1270-1. 10.1016/S2214-109X(18)30391-7 [DOI] [PubMed] [Google Scholar]
- 11.Choi JK, Paek D, Lee JO. Normal predictive values of spirometry in Korean population. Tuberc Respir Dis (Seoul) 2005;58:230-42. 10.4046/trd.2005.58.3.230 [DOI] [Google Scholar]
- 12.Park J, Kim S, Lee YJ, et al. Factors associated with radiologic progression of non-cystic fibrosis bronchiectasis during long-term follow-up. Respirology 2016;21:1049-54. 10.1111/resp.12768 [DOI] [PubMed] [Google Scholar]
- 13.The Cigarette Century: The Rise, Fall, and Deadly Persistence of the Product That Defined America. Allan M Brandt. Basic Books, Cambridge, MA; 2009. [Google Scholar]
- 14.Christiani DC. Vaping-Induced Lung Injury. N Engl J Med 2020;382:960-2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The article’s supplementary files as