Abstract
In late December 2019 severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) first emerged in Wuhan, Hubei, China, resulting in the potentially fatal COVID-19. It went on to be officially recognised as a pandemic by the World Health Organisation on 11 March 2020. While many public health strategies have evolved, there has been little mention of the immune system and how this could be strengthened to help protect against viral infections such as SARS-CoV-2. The present paper evaluates the current evidence base relating to immunonutrition, with a particular focus on respiratory viruses. Within the nutrition sector a promising body of evidence studying inter-relationships between certain nutrients and immune competence already exists. This could potentially be an important player in helping the body to deal with the coronavirus, especially among elders. Evidence for vitamins C, D and zinc and their roles in preventing pneumonia and respiratory infections (vitamins C and D) and reinforcing immunity (zinc) appears to look particularly promising. Ongoing research within this important field is urgently needed.
Keywords: nutrient deficiencies, infectious disease, pulmonary disease, preventive counselling
Introduction
Coronaviruses are important pathogens to animals and humans and have been around for many years. Severe acute respiratory syndrome was first detected in Guangdong in southern China in 2002 and the Middle East respiratory syndrome followed and first emerged in Saudi Arabia in 2012.1 Both result in severe respiratory infections, which can be fatal.1 In late December 2019 a further coronavirus emerged, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), resulting in the potentially fatal COVID-19 disease.2 This novel coronavirus has rapidly become a global public health concern, spreading to most countries worldwide.
Most public health strategies being employed are currently reactive. However, immunonutrition could have an important preventative role—a form of ‘prehabilitation’ helping the body to cope with potentially lethal viruses such as coronavirus. Elsewhere, prehabilitation has been defined as ‘interventions that can help to improve patient’s health in advanced of being exposed to a physiological stressor so they are then better able to cope with that stress’.3 It is known that viral clearance and infection recovery require activation of the host’s immune response, and nutrition could be a means of achieving this.4 Suboptimal immune competence commonly occurs in later life, with undernutrition, protein-energy malnutrition and micronutrient deficiencies being age-related lifestyle factors that can further hamper this.5
Undoubtedly there are many nutritional myths of bolstering the immune system, including diet, fruit, vitamin C, antioxidants, vitamins, minerals and probiotics—all typically being mentioned most often on the infosphere.6 The present article extrapolates some of the most pertinent scientific evidence related to immunonutrition, focusing specifically on its potential roles in resisting respiratory viruses and diseases.
Severe acute respiratory syndrome coronavirus 2
In late December 2019 a novel coronavirus emerged in Wuhan, Hubei Province, China and is the most recent emerging pathogen of a potentially fatal disease that has rapidly become a global public health concern.2 In February 2020, the World Health Organisation (WHO) named the disease COVID-19.2 By 11 March 2020 the WHO went on to further recognise COVID-19 as a global pandemic.7 The virus itself has been named SARS-CoV-2 as it has been determined as the same species as SARS but a different strain of the species.
Coronaviruses are a family of viruses known to infect mammals and birds, belonging to the family Coronaviridae.8 Before the emergence of COVID-19, there were six different coronaviruses known to infect humans, four of which cause a mild common cold-type illness.9 The remaining two coronaviruses emerged in 2002 and 2012 and result in a more severe disease in humans. These were named severe acute respiratory syndrome (SARS) and the Middle East respiratory syndrome (MERS) and are highly pathogenic viruses.9 Both SARS and MERS are believed to have originated in bats, suggesting animal to human transmission.9
SARS-CoV-2 has emerged as the third known coronavirus that causes fatal respiratory diseases in humans. The causative pathogen was isolated by Chinese researchers in January 2020 and discovered to be a large enveloped virus with a positive sense, single-stranded RNA genome of about 26–33 kb.1 The SARS-CoV-2 shares 96% similarity to bat coronavirus at the whole genome level, which suggests, like SARS and MERS, bats were the original host, with an intermediate host prior to infecting humans.9 It will be vital to track the path of the virus in order to prevent future exposure and outbreaks. In terms of the development of vaccines and antibody-based therapies, it will be important to understand the mutation rate of SARS-CoV-2, which appears to be moderate to high and similar to the other coronaviruses.10
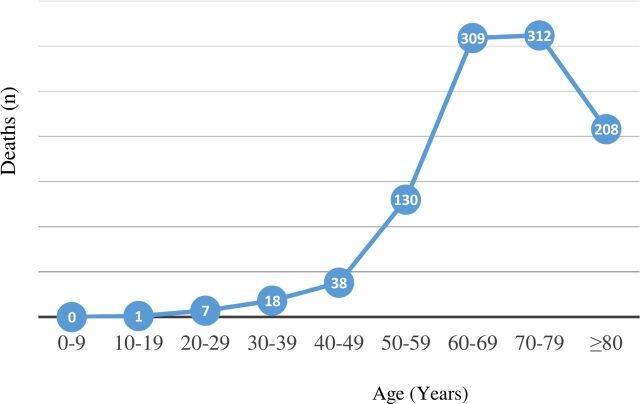
Unfortunately, no vaccines against coronaviruses have ever been developed, including for SARS or MERS, with MERS still remaining largely uncontrolled.11 Similarly, there is no single specific antiviral therapy. The ramification of SARS-CoV-2 is atypical viral pneumonia, with the main treatment strategy of supportive care, which may include oxygen or artificial ventilation.12 Antibiotics are of no use to treat viral pneumonia. The fatality rates in China (and now Italy) follow a distinct pattern, with the elderly having the highest death and case fatality rate (figure 1 and table 1).13 In particular, older patients (>65 years) with underlying health conditions, including acute respiratory distress syndrome, are at increased risk of dying.12
Figure 1.
Death rates in China, by age. Data extrapolated from Porcheddu et al.13
Table 1.
Case Fatality Rates in China, by Age
| Age (years) | Confirmed SARSCOV-2 n (%) | Case Fatality Rate |
| 0-9 | 416 (0.9) | - |
| 10-19 | 549 (1.2) | - |
| 20-29 | 3,619 (8.1) | - |
| 30-39 | 7,600 (17.0) | 0.2 |
| 40-49 | 8,571 (19.2) | 0.2 |
| 50-59 | 10,008 (22.4) | 0.2 |
| 60-69 | 8,583 (19.2) | 0.4 |
| 70-79 | 3,918 (8.8) | 1.3 |
| ≥80 | 1,408 (3.2) | 3.6 |
Data extrapolated from Porcheddu et al (2020)13
Immunity focus
The human immune system is comprised of four central components—T cells, B cells, the complement system and phagocytes which are vital in defending the organism against foreign intruders.14 The immune system is the body’s primary defence barrier against infections, and thus any weaknesses could be potentially detrimental to the host.15–17
It has been recognised that a ‘well-fed’ immune system is one way of helping to provide defence against pathogenic organisms.5 It has been elegantly stated that an ideal immune system should be ‘constantly alert and monitoring for signs of danger or invasion’.4 Professor Philip Calder is one of the leading experts in nutritional immunology and has published widely on this topic. In his article ‘Feeding the Immune System’, he explains that the immune system functions by acting as an exclusion barrier, identifying and eliminating pathogens and involving complex interplay between many different cell types and chemical mediators.5
Unfortunately a decline in immune function is usually inevitable with ageing, a process referred to scientifically as ‘immunosenescence’, which typically involves the deterioration of both innate and acquired immune systems.18 19 A number of potential mechanisms have been proposed, including (1) declining T cell function attributed to thymic involution and subsequent reduced output of naïve T cells20; (2) ageing-associated inflammation, often referred to as ‘inflammaging’, which paradoxically can reduce immunity and contribute to pathogenic age-related diseases21; and (3) poor micronutrient status – a bidirectional relationship between infection/immunity and nutrition whereby changes in one of these components can impact the other.22 It is also becoming apparent that ageing can modulate immune function and cellular composition in ways that are sex-specific, possibly due to differences in how menopause and andropause unfold.23
With regard to respiratory conditions, the combined effects of compromised immune function, frailty and length of exposure to pathogens mean that ageing populations are naturally predisposed to pulmonary disease.24 Nevertheless, there is hope—innate and acquired immune function may be supported by human nutrition, particularly in instances where this is suboptimal, that is, in clinical settings and the aged, potentially helping to lower the risk of infections and their severity and promote recovery from these.
Nutrition focus
As discussed, we are in the information era, and with this there are many online nutritional myths about how to bolster the immune system.6 However, a number of scientific reviews and research publications have been published focusing on the role of diet and specific nutrients in the immune system, some of which have focused on respiratory viral infections. Here we discuss some of the main findings.
One recent review25 identified that an array of micronutrients are required to meet the complex needs of the immune system, including vitamins A, D, C, E, B6, B12, folate, copper, iron, zinc and selenium, with many of these having potential synergistic relationships. Of the evidence, however, it was concluded that the largest body of evidence related to immune function existed for vitamin C, D and zinc.25 Hereon we subsequently focus on these nutrients with specific reference to the evidence on respiratory health.
Vitamin C
Vitamin C was first purified in the early 1930s, and soon after physicians proposed that vitamin C could be beneficial in the treatment of pneumonia.26 Historically, vitamin C has been used for immune support and is known to be acquired by neutrophils.27 This nutrient supports epithelial barrier function against pathogens, cellular functions of the adaptive and innate immune systems, and protects against oxidative stress.28
A review published in 201726 concluded that three controlled trials found that vitamin C prevented pneumonia and two controlled trials observed a treatment benefit of vitamin C for patients with pneumonia. An earlier Cochrane review,29 undertaken in 2013, collated evidence from three prophylactic trials,30–32 finding a statistically significant (80% or greater) reduction in pneumonia incidence in the vitamin C groups. It was subsequently concluded that therapeutic use of vitamin C supplementation could be reasonable for patients with pneumonia and low plasma vitamin C levels given its low cost and health risks.
With regard to dosages Glazebrook and Thomson30 monitored male students during World War II whose diets typically provided 10–15 mg vitamin C daily, and an additional 0.05–0.3 g/day ascorbic acid was added to morning cocoa or their evening glass of milk. Cases of pneumonia occurred in the control group but not in the test group, indicating a potential role in pneumonia prevention.30 Kimbarowski and Mokrow31 provided 0.3 g vitamin C daily to military recruits who were hospitalised with influenza type A. In this study pneumonia was reported in 2 subjects in the treatment group and 10 in the control group, again indicating a potential adjunctive role.31 Pitt and Costrini32 administered one of the highest dosages, 2 g/day, to US Marine recruits for 2 months with no reported symptoms attributed to the pills. In this study there was a reduced incidence of pneumonia among the recruits receiving vitamin C, but only 8 of 674 had pneumonia, indicating a need for larger sample sizes.32
In terms of potential mechanisms, it is well recognised that infections increase oxidative stress.26 Infections typically activate phagocytes which release reactive oxygen species, which are oxidising agents.26 Vitamin C is a renowned antioxidant which can counteract these effects.26 In one study a respiratory syncytial virus reduced the expression of antioxidant enzymes and subsequently increased oxidative damage.33
Vitamin D
A number of studies have investigated the inter-relationships between vitamin D and its effects on respiratory viruses and conditions such as community-acquired pneumonia.34–37
It is well appreciated that vitamin D is a powerful immunoregulator, with vitamin D receptors being expressed by the majority of immune cells (B and T lymphocytes, macrophages and monocytes).38 It has also been proposed that immune cells themselves can convert 25(OH)D3 into 1,25(OH)2D3, its active form.38
Zdrenghea et al 34 reported in their review that respiratory epithelial cells, macrophages and monocytes express vitamin D receptor, and concluded that vitamin D could act as a potential adjuvant in protecting and treating patients with respiratory viral infections who typically have lower vitamin D status. Work by Greiller and Martineau35 also reported that vitamin D deficiency is associated with a higher risk of viral acute respiratory infection and found that vitamin D metabolites modulated the expression and secretion of type 1 interferon, chemokines CXCL8 and CXCL10, and proinflammatory cytokines, including tumour necrosis factor and interleukin-6.
Another study,36 using human bronchial epithelial cells infected with respiratory syncytial and rhinoviruses (with or without exogenous D), showed that both viral strains reduced the number of vitamin D receptors and 24-hydroxylase. However, despite this, exogenous vitamin D was shown to increase antiviral defences and increase rhinovirus-induced interferon-stimulated genes and cathelicidin, implying a protective response.36 Other work37 focusing on pneumonia found a negative association between 1,25(OH)2D3 and pneumonia severity.
A systematic review and meta-analysis39 has collated evidence from 25 separate randomised controlled trials (n=11 321 participants) studying the effects of vitamin D supplementation on acute respiratory infections among those aged 0–95 years. Protective effects were seen among all participants, but particularly among those with baseline 25-hydroxyvitamin D levels <25 nmol/L, indicative of deficiency.39 Overall, the authors concluded that vitamin D appeared to be a safe strategy to protect against acute respiratory tract infections.39
Zinc
Zinc is regarded as a ‘gatekeeper’ of immune function: it is essential for the functioning of the immune system.40 Zinc ions play a role in the regulation of intracellular signalling pathways in adaptive and innate immune cells.40 Zinc is also involved in inflammation, elevating inflammatory responses and inducing cell-mediated immunity, and is a key component of pathogen-eliminating transduction pathways that contribute to neutrophil extracellular traps (networks which bind pathogens) formation.41
No meta-analysis or Cochrane reviews currently appear to have been undertaken in this field. Among paediatric populations a review of several studies42 concluded that zinc supplementation for more than 3 months could be effective in preventing pneumonia in children younger than 5 years of age, although the evidence was not robust enough to advocate prophylactic properties if given for shorter periods of time. Among the elderly it is recognised that inadequate zinc status impairs immune function, reduces pathogenic resistance, and is linked to an increased incidence and duration of pneumonia, along with overall mortality.43 Rigorous trials, however, are yet to determine the efficacy of zinc supplementation.
The need for more research
Given the rapid transmission of COVID-19, scientists are working against the clock to find potential vaccines, antiviral medications or other possible solutions. One intervention could potentially be the reinforcement of the immune function, a strategy that could be particularly worthwhile among vulnerable groups such as those aged >65, with underlying health conditions, such as asthma, chronic obstructive pulmonary disease, diabetes and heart disease, or who are immunosuppressed. Unfortunately, high-quality, rigorous trials are currently lacking. Most of what we know are based on observational evidence or older studies conducted on military populations.
Hopefully now more funds will be invested into nutritional immunology and its applications among ageing populations and those with viral respiratory conditions. This could be a viable approach given the relatively low cost of vitamin D, C and zinc supplements. What remains to be confirmed are the dosages and durations that these would need to be taken to be most beneficial and whether different combinations of micronutrients would be most effective in helping to fight against infection.
As previously mentioned, vitamins A, E, B6, B12, folate, copper, iron and selenium are other examples of nutrients that possess important role(s) in helping to meet the complex needs of the immune system.25 These may work individually or in synergy. Furthermore, it is highly likely that other dietary components could also play a role in the modulation of immunity, but are yet to be identified.
Given the recent shifts towards food shortages layered on top of the already pressing concerns about food insecurity in older adults, this could only further hamper the nutritional density and micronutrient profiles of diets. Further interventions and public health strategies should therefore consider the value of distributing both food and nutrition supplementation packages to the vulnerable.
Summary and perspective
Unfortunately, the transmission and speed at which COVID-19 is spreading globally appear to be surpassing the speed at which nutritional immunology studies can give firm answers. An insight into the evidence that is currently available indicates that from an immunological and respiratory stance, ageing populations are undoubtedly more ‘at risk’ from the harmful ramifications of such infections. Subsequently, a number of nutritional strategies could help them, taking on board what we know so far:
Public health strategies involving immunonutrition could be an alternative way to promote prehabilitation and reduce burdens on healthcare systems.
The general public and indeed the ageing population should be encouraged to follow guidance from Public Health England44 and continue taking supplements containing 10 μg of vitamin D daily, given the fact that low vitamin D status appears to correlate with reduced immune function. Family and friends should help communicate this information to older generations given that they may have limited awareness of these guidelines.
Foods that are naturally abundant in vitamin C such as broccoli (60 mg/100 g), blackcurrants (130 mg/100 g), fortified breakfast cereals (up to 134 mg/100 g) and oranges (37–52 mg/100 g)45 should be made accessible to older individuals who are most in need of their nutritional benefits.
In the UK 5.5% of men and 4% of women 65 years and over (around 1 in 20) presently have zinc intakes lower than the lower reference nutrient intake (the level below which deficiency could occur).46 The consumption of foods naturally abundant in zinc such as canned crab (5.7 mg/100 g), canned shrimps (3.7 mg/100 g), canned adzuki beans (≈2.3 mg/100 g) and boiled eggs (1.3 mg/100 g) should be encouraged as a supplementation strategy to reinforce immunity.
Among those with established respiratory conditions or pneumonia, specific nutrients such as vitamin C, D or zinc could be considered as potential adjuvants to conventional treatment pathways.
Tolerable upper intake levels (ULs) are intake levels which should not be surpassed as toxicity problems could appear.47 For vitamin D a UL of 50 µg/day is advised and for zinc a UL of 25 mg/day is recommended.47 There is insufficient evidence to establish UL for vitamin C, but available human data suggest that supplemental daily doses of up to about 1 g, in addition to normal dietary intake, are not associated with adverse gastrointestinal effects.47 Not having an adverse effect, however, is not necessarily indicative of a benefit either, and ongoing trials are warranted.
Every individual patient should be carefully assessed when providing nutrient intakes higher than the reference nutrient intake, and dosages should preferably be clinically validated for critically ill patients.
Now that a pandemic has arisen with SARS-CoV-2, and given the easy access, low cost and limited side effects of the discussed nutrients, now could be an appropriate time to trial and test their efficacy in relation to prevention, severity and recovery of such infections.
Ongoing research is urgently needed within the field of immunonutrition and, in particular, how this could benefit vulnerable, at risk groups.
bmjnph-2020-000071supp001.pdf (248.6KB, pdf)
Footnotes
Twitter: @drderbyshire
Contributors: ED, with her nutritional expertise, contributed to conceptualising and writing the main manuscript. JD, with her virology expertise, contributed to and edited the manuscript.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: ED consults for the Healthcare Supplement Information Service, British Summer Fruits and UK Food Advisory Board but did not receive funding for writing this article nor its open access fees. ED has a lung condition herself called bronchiectasis, and so was genuinely interested in the research underpinning this field and the importance of its communication. Open access fees were funded by Nutritional Insight for which ED is the Director.
Patient consent for publication: Not required.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: Data sharing not applicable as no data sets generated and/or analysed for this study. Data are available upon reasonable request. No data are available.
References
- 1. Lu R, Zhao X, Li J, et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet 2020;395:565–74. 10.1016/S0140-6736(20)30251-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. World Health Organization Director-General's remarks at the media briefing on 2019-nCoV, 2020. Available: https://www.who.int/dg/speeches/detail/who-director-general-s-remarks-at-the-media-briefing-on-2019-ncov-on-11-february-2020
- 3. Silver J. Prehabilitation could save lives in a pandemic. BMJ Opinion, 2020. Available: https://blogs.bmj.com/bmj/2020/03/19/julie-k-silver-prehabilitation-could-save-lives-in-a-pandemic/ [DOI] [PubMed]
- 4. Childs CE, Calder PC, Miles EA. Diet and immune function. Nutrients 2019;11:1933 10.3390/nu11081933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Calder PC. Feeding the immune system. Proc Nutr Soc 2013;72:299–309. 10.1017/S0029665113001286 [DOI] [PubMed] [Google Scholar]
- 6. Cassa Macedo A, Oliveira Vilela de Faria A, Ghezzi P. Boosting the immune system, from science to myth: analysis the Infosphere with Google. Front Med 2019;6:165 10.3389/fmed.2019.00165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. WHO WHO Director-General's opening remarks at the media briefing on COVID-19, 2020. Available: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19-11-march-2020
- 8. Cui J, Li F, Shi Z-L. Origin and evolution of pathogenic coronaviruses. Nat Rev Microbiol 2019;17:181–92. 10.1038/s41579-018-0118-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Zhou P, Yang X-L, Wang X-G, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020;579:270–3. 10.1038/s41586-020-2012-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Zhao Z, Li H, Wu X, et al. Moderate mutation rate in the SARS coronavirus genome and its implications. BMC Evol Biol 2004;4:21 10.1186/1471-2148-4-21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Shehata MM, Gomaa MR, Ali MA, et al. Middle East respiratory syndrome coronavirus: a comprehensive review. Front Med 2016;10:120–36. 10.1007/s11684-016-0430-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med 2020. 10.1016/S2213-2600(20)30079-5. [Epub ahead of print: 24 Feb 2020]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Porcheddu R, Serra C, Kelvin D, et al. Similarity in case fatality rates (CFR) of COVID-19/SARS-COV-2 in Italy and China. J Infect Dev Ctries 2020;14:125–8. 10.3855/jidc.12600 [DOI] [PubMed] [Google Scholar]
- 14. Geha RS. The immune system development and function. J Med Liban 1980;31:15–23. [PubMed] [Google Scholar]
- 15. Scully C, Georgakopoulou EA, Hassona Y. The immune system: basis of so much health and disease: 4. immunocytes. Dent Update 2017;44:436–42. 10.12968/denu.2017.44.5.436 [DOI] [PubMed] [Google Scholar]
- 16. Scully C, Georgakopoulou EA, Hassona Y. The immune system: basis of so much health and disease: 3. adaptive immunity. Dent Update 2017;44:322–7. 10.12968/denu.2017.44.4.322 [DOI] [PubMed] [Google Scholar]
- 17. Scully C, Georgakopoulou EA, Hassona Y. The immune system: basis of so much health and disease: 2. innate immunity. Dent Update 2017;44:246–52. 10.12968/denu.2017.44.3.246 [DOI] [PubMed] [Google Scholar]
- 18. Crooke SN, Ovsyannikova IG, Poland GA, et al. Immunosenescence: a systems-level overview of immune cell biology and strategies for improving vaccine responses. Exp Gerontol 2019;124:110632 10.1016/j.exger.2019.110632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Pawelec G. Age and immunity: What is “immunosenescence”? Exp Gerontol 2018;105:4–9. 10.1016/j.exger.2017.10.024 [DOI] [PubMed] [Google Scholar]
- 20. Berzins SP, Uldrich AP, Sutherland JS, et al. Thymic regeneration: teaching an old immune system new tricks. Trends Mol Med 2002;8:469–76. 10.1016/S1471-4914(02)02415-2 [DOI] [PubMed] [Google Scholar]
- 21. Franceschi C, Garagnani P, Parini P, et al. Inflammaging: a new immune–metabolic viewpoint for age-related diseases. Nat Rev Endocrinol 2018;14:576–90. 10.1038/s41574-018-0059-4 [DOI] [PubMed] [Google Scholar]
- 22. Maggini S, Pierre A, Calder P. Immune function and micronutrient requirements change over the life course. Nutrients 2018;10:1531 10.3390/nu10101531 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Gubbels Bupp MR, Potluri T, Fink AL, et al. The confluence of sex hormones and aging on immunity. Front Immunol 2018;9:1269 10.3389/fimmu.2018.01269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Haq K, McElhaney JE. Ageing and respiratory infections: the airway of ageing. Immunol Lett 2014;162:323–8. 10.1016/j.imlet.2014.06.009 [DOI] [PubMed] [Google Scholar]
- 25. Gombart AF, Pierre A, Maggini S. A review of micronutrients and the immune System–Working in harmony to reduce the risk of infection. Nutrients 2020;12:236 10.3390/nu12010236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Hemilä H. Vitamin C and infections. Nutrients 2017;9:339 10.3390/nu9040339 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Liugan M, Carr AC, Vitamin C. Vitamin C and neutrophil function: findings from randomized controlled trials. Nutrients 2019;11:2102 10.3390/nu11092102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Carr A, Maggini S. Vitamin C and immune function. Nutrients 2017;9:1211 10.3390/nu9111211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Hemila H, Louhiala P. Vitamin C for preventing and treating pneumonia. Cochrane Database Syst Rev 2013;8:CD005532. [DOI] [PubMed] [Google Scholar]
- 30. Glazebrook AJ, Thomson S. The administration of vitamin C in a large institution and its effect on general health and resistance to infection. J Hyg 1942;42:1–19. 10.1017/S0022172400012596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Kimbarowski JA, Mokrow NJ. [Colored precipitation reaction of the urine according to Kimbarowski (FARK) as an index of the effect of ascorbic acid during treatment of viral influenza]. Dtsch Gesundheitsw 1967;22:2413–8. [PubMed] [Google Scholar]
- 32. Pitt HA, Costrini AM. Vitamin C prophylaxis in marine recruits. JAMA 1979;241:908–11. 10.1001/jama.1979.03290350028016 [DOI] [PubMed] [Google Scholar]
- 33. Hosakote YM, Jantzi PD, Esham DL, et al. Viral-mediated inhibition of antioxidant enzymes contributes to the pathogenesis of severe respiratory syncytial virus bronchiolitis. Am J Respir Crit Care Med 2011;183:1550–60. 10.1164/rccm.201010-1755OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Zdrenghea MT, Makrinioti H, Bagacean C, et al. Vitamin D modulation of innate immune responses to respiratory viral infections. Rev Med Virol 2017;27:e1909 10.1002/rmv.1909 [DOI] [PubMed] [Google Scholar]
- 35. Greiller C, Martineau A. Modulation of the immune response to respiratory viruses by vitamin D. Nutrients 2015;7:4240–70. 10.3390/nu7064240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Telcian AG, Zdrenghea MT, Edwards MR, et al. Vitamin D increases the antiviral activity of bronchial epithelial cells in vitro. Antiviral Res 2017;137:93–101. 10.1016/j.antiviral.2016.11.004 [DOI] [PubMed] [Google Scholar]
- 37. Pletz MW, Terkamp C, Schumacher U, et al. Vitamin D deficiency in community-acquired pneumonia: low levels of 1,25(OH)2 D are associated with disease severity. Respir Res 2014;15:53 10.1186/1465-9921-15-53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Sassi F, Tamone C, D'Amelio P, et al. Hormone, and immunomodulator. Nutrients 2018;10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Martineau AR, Jolliffe DA, Hooper RL, et al. Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data. BMJ 2017;356:i6583 10.1136/bmj.i6583 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Wessels I, Maywald M, Rink L, et al. Zinc as a gatekeeper of immune function. Nutrients 2017;9:1286. 10.3390/nu9121286 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Gammoh NZ, Rink L. Zinc in infection and inflammation. Nutrients 2017;9:E624. 10.3390/nu9060624 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Sakulchit T, Goldman RD. Zinc supplementation for pediatric pneumonia. Can Fam Physician 2017;63:763–5. [PMC free article] [PubMed] [Google Scholar]
- 43. Barnett JB, Hamer DH, Meydani SN. Low zinc status: a new risk factor for pneumonia in the elderly? Nutr Rev 2010;68:30–7. 10.1111/j.1753-4887.2009.00253.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Public Health England PHE publishes new advice on vitamin D, 2016. Available: https://www.gov.uk/government/news/phe-publishes-new-advice-on-vitamin-d
- 45. Public Health England Composition of foods integrated dataset (CoFID), 2019. Available: https://www.gov.uk/government/publications/composition-of-foods-integrated-dataset-cofid
- 46. Public Health England NDNS: results from years 7 and 8 (combined). Results of the National diet and nutrition survey (NDNS) rolling programme for 2014 to 2015 and 2015 to 2016. Tables 5.1 and 5.2, 2018. Available: https://www.gov.uk/government/statistics/ndns-results-from-years-7-and-8-combined
- 47. EFSA Scientific Committee on food scientific panel on dietetic products, nutrition and allergies. Tolerable upper intake levels for vitamin and minerals. EFSA, 2006. Available: http://www.efsa.europa.eu/sites/default/files/efsa_rep/blobserver_assets/ndatolerableuil.pdf
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjnph-2020-000071supp001.pdf (248.6KB, pdf)