Pandemic spread of the novel coronavirus, severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), the causative agent of the new disease Coronavirus disease 2019 (COVID-19), has been associated with a case fatality rate of 2.3% [1], approaching the >2.5% rate estimated for the 1918 Influenza pandemic [2]. However, information regarding the precise cause of death in patients expiring with COVID-19 has been limited. We report the finding of pulmonary thromboembolism as the cause of death in the initial two COVID-19 patients examined postmortem at our institution.
The first patient, a middle-aged male with well-controlled hypertension, was admitted with nine days of fever, chills, myalgia, dry cough, and dyspnea. On examination, he was found to have a temperature of 39.4 ⁰C and SpO2 of 92% on room air. A nasopharyngeal swab was positive for SARS-CoV-2 by real-time reverse-transcription-polymerase-chain-reaction amplification for both the SARS-CoV-2 ORF1 a/b and pan-Sarbecovirus E-gene (RT-PCR, cobasⓇ 6800 system, RocheDiagnostics), but the initial chest radiograph showed only mild bilateral pulmonary vascular congestion. He was treated with ceftriaxone and azithromycin for presumed bacterial superinfection with improvement in fever and leukocytosis, but continued to require supplemental nasal oxygen. The serum inflammatory markers ferritin (752 ng/ml, reference range 30-400ng/ml) and C-reactive protein (43.74 mg/L, reference range <5.1 mg/L) were noted to be elevated. Repeat chest radiograph on hospital day 8 revealed new dense patchy left mid-lung and retrocardiac opacities, and a hazy opacity in the right lower lung. On day 9, the patient complained of increasing left inspiratory chest pain and tenderness; 30 minutes later the patient was found to have pulseless electrical activity.
The second, a middle-aged male with asthma, hypertension and human immunodeficiency virus infection controlled on highly-active antiretroviral therapy, was admitted with fever, chills, productive cough and dyspnea worsening over 2 days. A nasopharyngeal swab obtained during an evaluation the previous day was positive for SARS-CoV-2 by the Roche cobasⓇ 6800 RT-PCR assay. Examination at admission was notable for a temperature of 38.4 ⁰C and SpO2 of 93% on room air; multiple bilateral lung opacities were noted on chest radiography. Elevated serum ferritin (411 ng/ml) and C-reactive protein (271.6 mg/L) were noted, and these values continued to rise despite treatment with broad-spectrum antibiotics. Of note, prothrombin time/INR was measured on multiple days and was not prolonged. His condition deteriorated, requiring intubation and ventilator support; over the next 3 days he became hemodynamically unstable requiring multiple pressors. Eight days following admission he developed pulseless electrical activity.
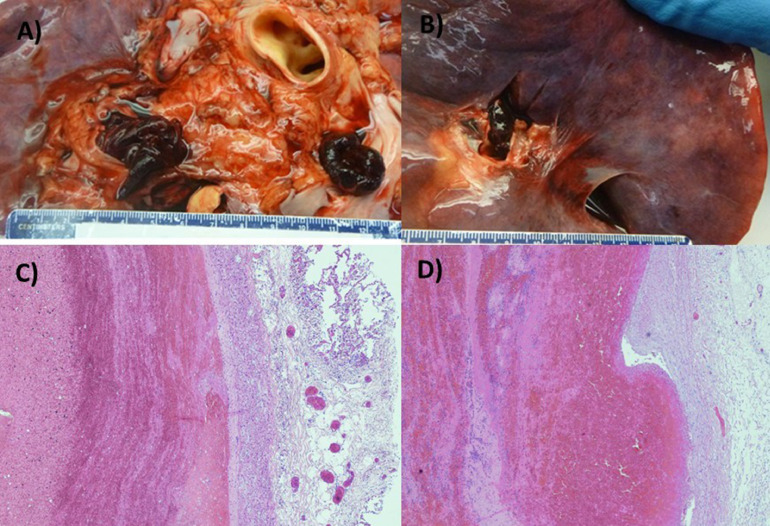
At autopsy, a common notable finding was pulmonary thromboembolism, with occlusion of the right main pulmonary artery in one case, and occlusion of both left and right main pulmonary arteries in the second (Fig. 1 A-B). Deep venous thrombosis was found in both cases. Other findings in both cases were multiple foci of pulmonary parenchymal firmness consistent with bilateral pulmonary consolidation, cardiomegaly, and left ventricular hypertrophy consistent with hypertensive cardiovascular disease.
Fig. 1.
Pulmonary Emboli Occluding the Main Pulmonary Arteries (A) Gross autopsy image showing pulmonary thromboembolism occluding right and left main pulmonary arteries (Patient 1) and (B) right main pulmonary artery (Patient 2). Pulmonary embolism: alternating areas of pale pink (fibrin) and red (erythrocytes) layers forming "lines of Zahn" in patient 1 (C) and 2 (D).
Histological examination of the lungs revealed characteristic pale pink and red areas (Zhan lines) representing alternating strata of platelets and fibrin with admixed layers of erythrocytes, consistent with pulmonary thromboembolism (Fig. 1 C-D).
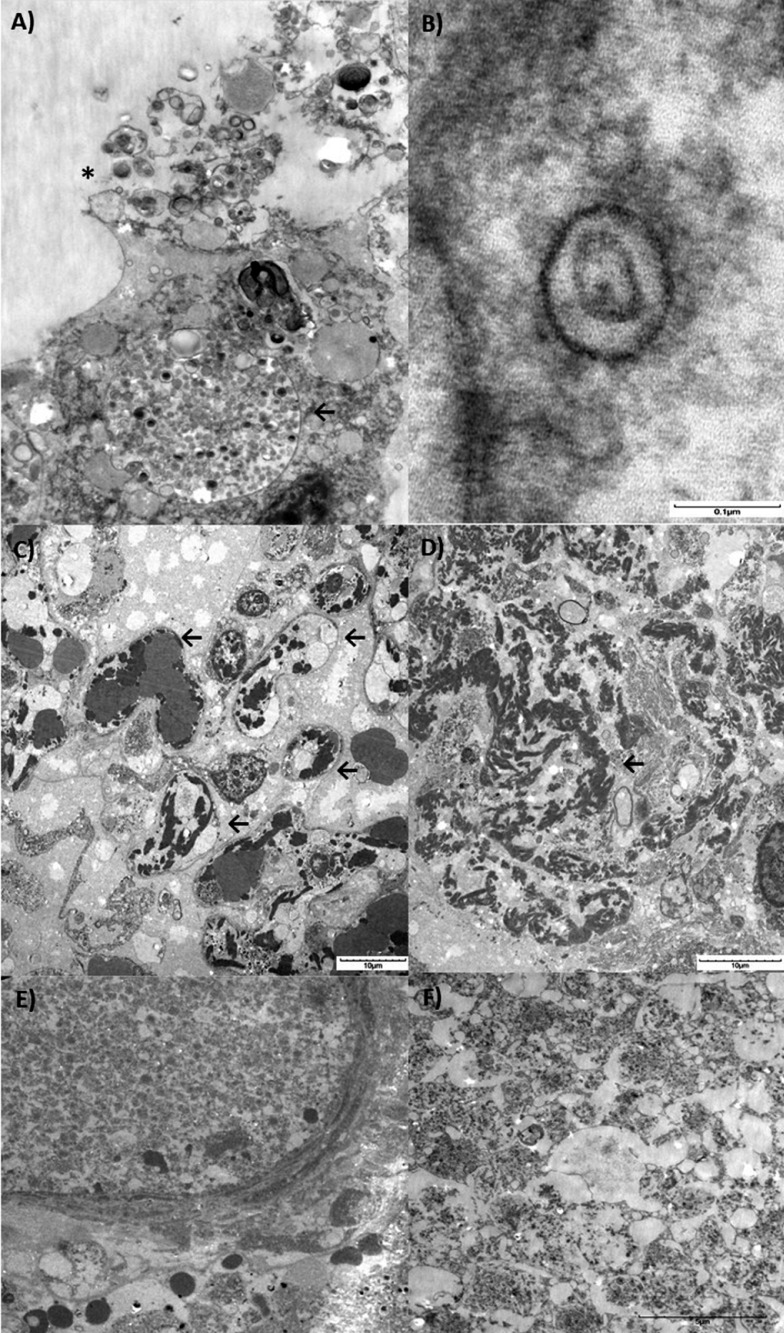
Ultrastructural examination demonstrated pleomorphic viral-like particles ranging from 60 to 120 nm in distended cytoplasmic vacuoles within the pneumocytes in lung specimens from both cases (Fig. 2 A-B). Rupture of the cytoplasmic vacuoles with release of the virus-like particles into the alveolar lumen is shown in Fig. 2A. The individual spherical viral-like particles display distinctive projections with mature particles exhibiting electron dense centers, consistent with the appearance of coronavirus (Fig. 2B). Fibrin deposition within and outside capillaries (Fig. 2 C-D), as well as pure platelet thrombi was also noted (Fig. 2 E-F).
Fig. 2.
Electron microscopy and histopathological lung findings of SARS-CoV-2. Lung sections were initially placed in neutral buffered formalin and later transferred to 3% buffered glutaraldehyde. Following post-fixation in 1% osmium tetroxide, tissues were serially dehydrated and embedded in epoxy resin in standard fashion. One micron toluidine-stained scout sections were prepared for light microscopic orientation and 80nm ultrathin sections for electron microcopy were stained with uranyl acetate and lead citrate and examined in an Hitachi 7650 transmission electron microscope at 80kV. (A) Transmission electron microscopy image showing an infected pneumocyte. Note the cytoplasmic vacuole loaded with viral particles in various stages of bud formation (←), and shedding into the luminal compartment (*) (3000x). (B) Typical morphology of Coronavirus (SARS-CoV-2) showing a symmetrical arrangement, outer envelope and surrounding electron dense shell with central zone. Note the distinctive projections conferring a solar corona appearance. (C) Multiple capillaries showing deposits of fibrin deposition (←). (D) Coarse electron dense areas of cytoplasmic fibrin deposition. (E) Pure platelet thrombus within the arterial lumen and (F) Detail on platelet thrombi. Scale bars are shown at the bottom right of each figure.
In contrast to the majority of patients with SARS-CoV-2 infection, who are asymptomatic or pauci-symptomatic, some patients will develop severe illness progressing to acute respiratory distress syndrome (ARDS), followed by death in a significant subset [3]. Recent reports from the People's Republic of China have associated severe pneumonia and fatal outcomes in SARS CoV-2 with elevated levels of D-dimer and fibrin degradation products (FDP), suggesting derangement of coagulation activation and consequently a potential for predisposition to thromboembolic events [4,5].
Notably, several viral infections have been associated with coagulation disorders that may lead to thrombosis and disseminated intravascular coagulation [6], including influenza virus and SARS which have been associated with pulmonary intravascular thrombi formation and fibrin deposition likely as a result of disseminated intravascular coagulation and microthrombosis [7,8]. In SARS coronavirus infection, increased production of a novel procoagulant by infected cells due to increased hfg12 gene transcription induced by viral nucleocapsid (N) protein has been proposed as mechanism contributing to thrombosis [9].
As reported previously for SARS-CoV [10], our findings suggest that SARS-CoV-2 infected patients similarly may be at increased risk for pulmonary thromboembolic events, due to a baseline hypercoagulable state, and may benefit from evaluation and management for coagulopathy early in the course of disease.
Acknowledgment
The authors thank Carlos Cordon-Cardo MD, PhD for his consistent encouragement and support.
References
- 1.Porcheduu R., Serra C., Kelvin D., Kelvin N., Rubino S. Similarity in case fatality rates (CFR) of COVID-19/SARS-COV-2 in Italy and China. J Infect Dev Ctries. 2020;14:125–128. doi: 10.3855/jidc.12600. [DOI] [PubMed] [Google Scholar]
- 2.Taukenberger J.K., Morens D.M. 1918 Influenza: the mother of all pandemics. Emerg Infect Dis. 2006;12:15–22. doi: 10.3201/eid1201.050979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Huang C., Wang Y., Li X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Han H., Yang L., Liu R., Liu F., Wu K.L., Li J., Liu X.H., Zhu C.L. Prominent changes in blood coagulation of patients with SARS-CoV-2 infection. Clin Chem Lab Med. 2020 doi: 10.1515/cclm-2020-0188. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 5.Yin S., Huang M., Li D., Tang N. Difference of coagulation features between severe pneumonia induced by SARS-CoV2 and non-SARS-CoV2. J Thromb Thrombolysis. 2020 doi: 10.1007/s11239-020-02105-8. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Goeijenbier M.1, van Wissen M., van de Weg C., Jong E., Gerdes V.E. Review: Viral infections and mechanisms of thrombosis and bleeding. J Med Virol. 2012;84:1680–1696. doi: 10.1002/jmv.23354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Muramoto Y., Ozaki H., Takada A., Park C.H., Sunden Y. Highly pathogenic H5N1 influenza virus causes coagulopathy in chickens. Microbiol Immunol. 2006;50:73–81. doi: 10.1111/j.1348-0421.2006.tb03764.x. [DOI] [PubMed] [Google Scholar]
- 8.Hwang D.M., Chamberlain D.W., Poutanen S.M., Low D.E., Asa S.L., Butany J. Pulmonary pathology of severe acute respiratory syndrome in Toronto. Mod Pathol. 2005;18:1–10. doi: 10.1038/modpathol.3800247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Han M., Yan W., Huang Y., Yao H., Wang Z. The nucleocapsid protein of SARS-CoV induces transcription of hfgl2 prothrombinase gene dependent on C/EBP alpha. J Biochem. 2008;144:51–62. doi: 10.1093/jb/mvn042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ng K.H., Wu A.K., Cheng V.C. Pulmonary artery thrombosis in a patient with severe acute respiratory syndrome. Postgrad Med J. 2005;81(956):e3. doi: 10.1136/pgmj.2004.030049. [DOI] [PMC free article] [PubMed] [Google Scholar]