Abstract
Background
A digital version of the clock drawing test (dCDT) provides new latency and graphomotor behavioral measurements. These variables have yet to be validated with external neuropsychological domains in non-demented adults.
Objective
The current investigation reports on cognitive constructs associated with selected dCDT latency and graphomotor variables and compares performances between individuals with mild cognitive impairment (MCI) and non-MCI peers.
Methods
202 non-demented older adults (age 68.79 ± 6.18, 46% female, education years 16.02 ± 2.70) completed the dCDT and a comprehensive neuropsychological protocol. dCDT variables of interest included: total completion time (TCT), pre-first hand latency (PFHL), post-clock face latency (PCFL), and clock face area (CFA). We also explored variables of percent time drawing (i.e., ‘ink time’) versus percent time not drawing (i.e., ‘think time’). Neuropsychological domains of interest included processing speed, working memory, language, and declarative memory.
Results
Adjusting for age and premorbid cognitive reserve metrics, command TCT positively correlated with multiple cognitive domains; PFHL and PCFL negatively associated with worse performance on working memory and processing speed tests. For Copy, TCT, PCFL, and PFHL negatively correlated with processing speed, and CFA negatively correlated with language. Between-group analyses show MCI participants generated slower command TCT, produced smaller CFA, and required more command ‘think’ (%Think) than ‘ink’ (%Ink) time.
Conclusion
Command dCDT variables of interest were primarily processing speed and working memory dependent. MCI participants showed dCDT differences relative to non-MCI peers, suggesting the dCDT may assist with classification. Results document cognitive construct validation to digital metrics of clock drawing.
Keywords: Attention, cognition, cognitive aging, neuropsychology, processing speed
INTRODUCTION
The clock drawing test (CDT) is a classic neuropsychological test first introduced in the 19th century [1] and has become a standard screener to assess for cognitive impairment [2]. The classic CDT involves two conditions: drawing a de novo clock to command followed by copying a pre-drawn clock. In the command condition, patients are asked to ‘draw the face of a clock, put in all the numbers and set the hands for ten after eleven.’ The command condition involves calling into mind the semantic attributes associated with a clock. Simultaneously, the individual must maintain mental set for test instructions, process the linguistic content regarding time setting, and effectively plan the execution of the test while ignoring distracting information. The copy condition also involves a wide variety of cognitive demands [3], but is more reliant upon visuoconstruction and executive function constructs at least within Alzheimer’s disease (AD) and vascular dementia participants [4, 5].
A digital version of the clock drawing test (dCDT) [3] with command and copy conditions provides new latency and graphomotor outcomes; however, these variables have yet to be validated with external neuropsychological domains in non-demented adults. The most frequently discussed variables in the literature for the dCDT are latency based and include total clock drawing completion time (TCT), and more decision-oriented latencies such as a) time (in seconds) to place the first hand (pre-first-hand latency; PFHL), and b) time to the next pen stroke after drawing the face of a clock (post-clock face latency; PCFL). These latencies increase in duration with age [5] and may be differentially compromised by dementia type [6]; individuals with small vessel vascular dementia display significantly longer dCDT TCT, PCFL, and PFHL relative to individuals with AD, while individuals with AD are slower on TCT and PFHL relative to non-demented peers [6].
Clock face area (CFA) is another relevant dCDT feature that may provide useful insight into cognitive functions. Significantly smaller clock drawings along with micrographia have been observed in subcortical diseases such as Parkinson’s disease. These individuals are characterized by reduced processing speed, but also reduced planning and graphomotor output associated with compromised frontostriatal functions [7]. Hypothetically, the size of the clock face may provide additional insight into higher cortical function that involve language and reasoning abilities.
The current investigation had two aims. First, to provide insight into neuropsychological constructs underlying published dCDT latency variables as well as clock face area in non-demented adults. We examined hypothesized associations between the dCDT variables of interest and constructs of processing speed, working memory, declarative memory, and language domains in non-demented older adults. Based on cognitive theories of normal aging involving the foundational changes in processing speed [8], we hypothesized the latency variables of interest would be primarily associated with processing speed metrics. Over and above processing speed, however, we anticipated the time it takes to draw a clock, put in all the numbers, and set the hands to 10 after 11 would also inversely associate with working memory, language, and declarative memory. As for the decision-oriented latencies of PFHL and PCFL whereby a participant has to recall the examiner’s instructions and accurately set short versus long hand placement, we hypothesized increased latency would also associate with working memory. Finally, given previous reports that individuals with subcortical diseases [9] produced smaller clock drawings and known reductions in processing speed and reasoning for these individuals, we hypothesized clock face area would associate with measures of planning and reasoning. For the second aim, we examined group differences in the selected dCDT latency and graphomotor parameters between participants with and without Mild Cognitive Impairment (MCI). Individuals with MCI often display cognitive slowing evidenced by reduced psychomotor speed and focused attention [10] which could be partly attributed to reductions in white matter tracts integrity [11, 12]. Therefore, we hypothesized MCI participants would display slower TCT, PFHL, PCFL, and smaller CFA. To assess the hypothesis that latency differences are due to decision making rather than motor output, we examined latency differences in time spent thinking versus drawing.
METHODS
Participants
The University of Florida’s Institutional Review Board approved the research investigation. Participants provided written informed consent and studies were conducted in accordance with the Declaration of Helsinki. Participants were recruited via brochures and community mailings, memory screenings, and locally posted fliers.
Inclusion criteria: age ≥60, English as primary language, and intact activities of daily living (ADLs) per Lawton & Brody’s Activity of Daily Living Scale completed by both the participant and their caregiver [13]. Exclusion criteria: evidence of a major neurocognitive disorder at baseline per the Diagnostic and Statistical Manual of Mental Disorders – Fifth Edition [14], significant medical illness potentially limiting lifespan, major psychiatric disorders, history of head trauma/neurodegenerative illness, documented learning disorder, seizure disorder or other significant neurological illness, less than a 6th grade education, substance abuse in the last year, major cardiac disease, chronic medical illness thought to induce encephalopathy. Participants were screened for dementia over the phone using the Telephone Interview for Cognitive Status (TICS; [15]) and during an in-person interview with a neuropsychologist and trained research coordinator assessing comorbidity rating [16], anxiety, depression, ADLs, neuropsychological functioning, and clock drawing [17]. The same examiner administered all test items and trained raters scored and double entered behavioral data. Two neuropsychologists reviewed all baseline data to confirm the absence of any dementias.
Digital clock drawing test (dCDT)
Digital clock drawing was obtained using digital pen technology from Anoto, Inc. which measures pen positioning 75 times/second at a spatial resolution of ± 0.002 inches. The dCDT software was used to classify each pen stroke (e.g., as a clock face, clock hand, digit, etc.) with over 84% accuracy [18]. The digital pen, which works as a conventional ballpoint pen, possesses a high spatial resolution making it possible to study minute drawing elements. Digital variables were scored using Massachusetts Institute of Technology in-house software. An external rater replayed and deconstructed each drawing to ensure appropriate scoring. dCDT variables of interest are listed below:
Total Completion Time (TCT)
Total time taken in seconds to complete all elements of the clock drawing, from first pen-paper contact until completion of the last stroke.
Pre-First-Hand Latency (PFHL)
Time taken between drawing the first clock hand and the previous stroke.
Post-Clock Face Latency (PCFL)
Time taken between completing the clock face and setting the first number.
Clock Face Area (CFA)
Computed as the area of a circle (πr2) using the average radius of horizontal and vertical radii in millimeters [19].
‘Think’ versus ‘Ink’ Time [5]
Percent ‘ink time’ measures the percent of total time the pen is in contact with the paper for each test condition. Percent ‘ink time’ was computed as: (total ‘ink time’/total time to completion × 100). This variable provides a means to operationally define time spent drawing while considering individual differences in total drawing time. Percent ‘think time’ measures the percent of total time the pen is not in contact with the paper, measured from the completion of the first pen stroke to the beginning of the last pen stroke. Percent ‘think time’ time was computed as: (1 – percent ink time). Percent ‘think time’ provides a means to operationally define non-drawing cognitive activity. These variables allowed us to examine the hypothesis that individuals with MCI have a longer latency response due to longer decision-making time rather than due to longer motor output drawing time.
Neuropsychological domains of interest
The neuropsychological measures were chosen based on their respective domain of cognition [21]. Raw scores for all tests were converted to z-scores using available published norms. Cognitive domain composites were created by averaging standardized z-scores in the categories below.
Processing speed
Digit Symbol, Wechsler Adult Intelligence Scale – 3rd edition (WAIS-III)
Requiring transposition of digits to a corresponding symbol and has been associated with the integrity of frontal, parietal, and temporal white matter regions [11]. Variable of interest: total correct in 120 seconds.
Stroop Color Word Test, Reading condition
Requiring rapid reading of words and associated with frontostriatal function [20]. Variable of interest: total correct in 45 seconds.
Trail Making Test Part A
Requiring divided attention and visual scanning. Sensitive to frontal pathology [21]. Variable of interest: total time in seconds.
Working memory
Letter Number Sequencing, WAIS-III
Requiring mental manipulation involving alphabetical and numerical ordering associated with functional activation of bilateral orbital frontal, dorsolateral prefrontal, and posterior parietal regions. Variable of interest: total correct sequences.
Digit Span Backward Span, WAIS-III
Requiring backward serial order recall and more challenging for individuals with frontal lesions [22]. Functional imaging [23] and transcranial magnetic stimulation [24] studies demonstrate right dorsolateral prefrontal cortex, bilateral inferior parietal regions, anterior cingulate, and medial occipital cortex involvement suggesting mental visualization strategies [24]. Variable of interest: longest correct span.
Spatial Span Backward Span, WAIS-III
Requiring spatial working memory. Poorer and more variable performance is observed in participants with right hemisphere lesions and right temporal lobectomy [25]. The task associates with involvement of bilateral parietal regions (Brodmann Area (BA) 6, BA7) and frontal regions (BA44, BA45, BA47, BA9), and increased activation in the left dorsolateral prefrontal cortex as test difficulty increases [26]. Variable of interest: longest correct span.
Language
Boston Naming Test
Confrontation naming test relying heavily on semantic knowledge [27]. Variable of interest: total correct/60 items.
Controlled Oral Word Association (Animal) Test
Requires rapid semantic word production. Associated with bilateral grey matter volume in the medial perirhinal cortex and entorhinal cortex/hippocampal head and also associates with fractional anisotropy values in the left anterior inferior longitudinal fasciculus [28]. Variable of interest: total correct in 60 seconds.
Declarative memory
Logical Memory Story I, II, Wechsler Memory Scale, third edition (WMS-III)
Requiring episodic memory and performance is associated with left subiculum and hippocampal volume [29] Decreased performance, particularly immediate recall, is observed in temporal lobe resection patients [30]. Variable of interest: total delay recall.
Hopkins Verbal Learning Test, Revised (HVLT-R)
List learning, delayed recall. Bilateral hippocampal volume associates with Delay Recall and Recognition Discrimination performance in non-demented older adults [31]. Delay Recall associates with hippocampal volume and mean diffusivity of the cingulum and fornix in AD and MCI samples [32]. Variable of interest: total words recalled.
MCI classification
MCI was defined using the comprehensive criteria suggested by Jak and colleagues [33] using age-adjusted normative data. Individuals met classification for single domain MCI when at least two measures within one cognitive domain fell below one standard deviation using normative data. Individuals met classification for multi-domain MCI when at least two measures involving more than one cognitive domain fell below one standard deviation relative to normative means and standard deviations.
Covariates of interest
We considered age since increasing age affects motor and processing speed performance as well as reasoning and memory abilities [34]. We also considered anxiety which correlates highly with processing speed [35], and depression given its association with psychomotor slowing in older adults [36] resulting in slower clock drawing time [37]. We considered how situational (State) and dispositional (Trait) anxiety, and depression symptoms (BDI-II) may affect dCDT performance. Premorbid cognitive reserve, which refers to the brain’s ability to offset pathological attacks by relying on previously acquired skills to maintain cognitive functioning [38] was also considered and operationalized as a composite encompassing: word reading ability using either Wide Range Achievement Test (WRAT; n = 145) or Wechsler Test of Adult Reading (WTAR; n = 57), vocabulary knowledge, and years of education; all considered estimates of premorbid intelligence [39].
Statistical analyses
Statistical analyses were performed using SPSS v.25 with significance set at p≤0.05. Covariates were analyzed using Spearman correlations between dCDT variables and participant demographics. Normality was achieved via square root transformation. MCI and non-MCI groups were compared using independent samples t-test for continuous demographic variables, and with Chi-Square test for categorical variables. Groups were also compared on clock face accuracy, measured by the amount of deviation from a perfectly circular clock face, calculated using the ellipse residual provided by the dCDT scoring program. To address the first aim, we conducted partial correlations between dCDT variables and neuropsychological domains on the whole sample controlling for age and premorbid cognitive reserve and correcting for multiple comparisons. We addressed the second aim using separate Multivariate Analysis of Covariance (MANCOVA) for both clock drawing conditions given the number of dependent variables, the multicollinearity amongst them, and to limit the number of statistical analyses performed. We compared groups on %Think time across both clock drawing conditions using a repeated measures ANOVA.
RESULTS
Participants
Two hundred and four participants completed baseline neuropsychological assessment and dCDT. Two participants were excluded based on concerns for learning disorder, thus retaining data from 202 participants. Table 1 illustrates demographic characteristics for the entire sample and the MCI and non-MCI groups. Age correlated with slower TCT in both dCDT conditions; premorbid cognitive reserve inversely associated with command TCT and CFA in both dCDT conditions. Correlations between dCDT variables and measures of anxiety and depression were not significant (Table 2).
Table 1.
Participant Characteristics for the Total Sample and Separated by MCI and Non-MCI Group
| Demographic | Total (n = 202) Mean±SD (range) |
Non-MCI (n = 171) Mean±SD (range) |
MCI (n = 31) Mean±SD (range) |
p |
|---|---|---|---|---|
| Age | 68.79 ± 6.18 (55–85) | 68.55 ± 5.95 (55–85) | 70.10 ± 7.30 (60–85) | 0.200 |
| Sex (Male:Female) | 110 : 92 | 94 : 77 | 16 : 15 | 0.920 |
| Race (White:Non-White) | 189 : 13 | 163 : 8 | 26 : 5 | 0.017 |
| Education | 16.02 ± 2.70 (9–24) | 16.23 ± 2.57 (9–24) | 14.89 ± 3.16 (10–22) | 0.031 |
| TICS | 37.8173 ± 3.73 (26–47) | 38.38 ± 3.53 (26–47) | 34.59 ± 3.29 (27–40) | <0.001 |
| Charlson Comorbidity Index | 0.422 ± 0.69 (0–4) | 0.424 ± 0.711 (0–4) | 0.414 ± 0.57 (0–2) | 0.944 |
| Premorbid cognitive reserve | 0.50 ± 0.62 (−1.25–1.90) | 0.56 ± 0.59 (−1.25–1.90) | 0.16 ± 0.64 (−0.9–1.58) | <0.001 |
| STAI-State | 28.24 ± 7.99 (20–59) | 27.86 ± 8.028 (20–59) | 30.63 ± 7.04 (20–44) | 0.094 |
| STAI-Trait | 30.23 ± 7.86 (20–55) | 29.83 ± 7.91 (20–55) | 32.74 ± 7.22 (21–48) | 0.074 |
| BDI-II | 4.6 ± 5.17 (0–30) | 4.40 ± 5.25 (0–30) | 5.85 ± 4.55 (0–17) | 0.176 |
TICS, Telephone Interview for Cognitive Status; STAI, State-Trait Anxiety Inventory; BDI-II, Beck Depression Inventory 2nd Edition.
Table 2.
Spearman Correlations between dCDT Metrics, Age, and Premorbid Cognitive Reserve
| Age p |
Premorbid cognitive reserve p |
STAI State p |
STAI Trait p |
BDI-II p |
|
|---|---|---|---|---|---|
| Command | |||||
| TCT | 0.194 | −0.206 | −0.004 | −0.038 | −0.90 |
| 0.006 | 0.004 | 0.959 | 0.604 | 0.215 | |
| PFHL | −0.580 | −0.102 | 0.059 | 0.029 | −0.044 |
| 0.418 | 0.152 | 0.414 | 0.623 | 0.543 | |
| PCFL | 0.045 | −0.980 | 0.134 | 0.11 | −0.029 |
| 0.528 | 0.168 | 0.064 | 0.128 | 0.688 | |
| CFA | 0.05 | 0.227 | −0.116 | −0.183 | −0.210 |
| 0.146 | 0.001 | 0.107 | 0.011 | 0.003 | |
| Copy | |||||
| TCT | 0.197 | −0.124 | 0.083 | 0.131 | 0.015 |
| 0.005 | 0.08 | 0.351 | 0.069 | 0.839 | |
| PFHL | 0.137 | −0.720 | −0.041 | 0.025 | −0.023 |
| 0.053 | 0.313 | 0.571 | 0.73 | 0.753 | |
| PCFL | 0.149 | −0.087 | 0.12 | 0.15 | 0.114 |
| 0.035 | 0.224 | 0.098 | 0.038 | 0.115 | |
| CFA | −0.077 | 0.189 | 0.031 | −0.046 | −0.088 |
| 0.278 | 0.007 | 0.668 | 0.53 | 0.227 |
TCT, total completion time; PFHL, pre-first-hand latency; PCFL, post-clock face latency.
dCDT variables and neuropsychological composites
Total Completion Time
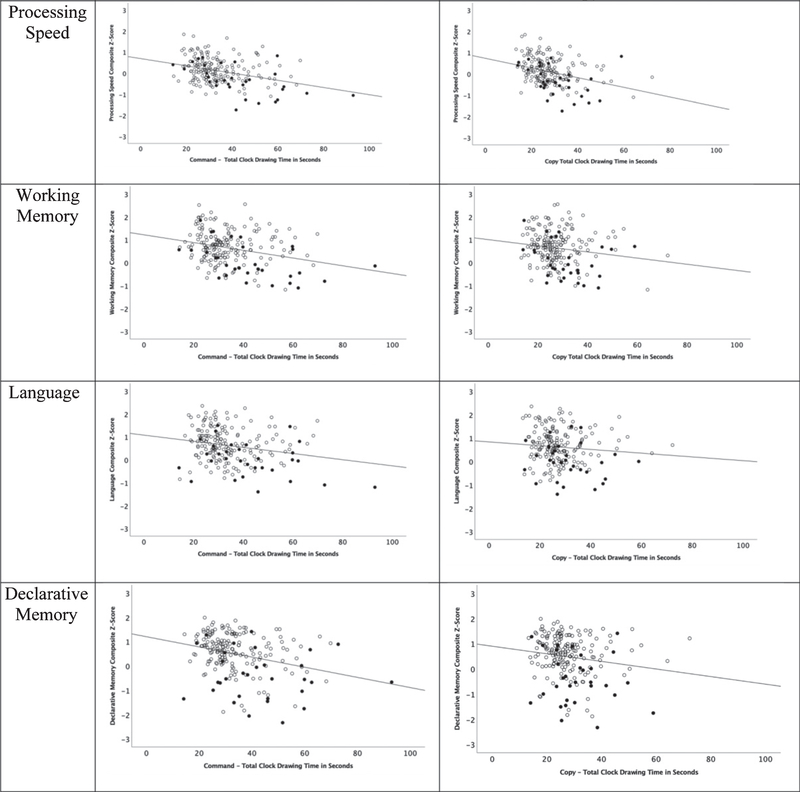
In the command condition, slower TCT was associated with slower performance on tests measuring processing speed (r = −0.284, p < 0.001), and worse performance on language (r = −0.201, p = 0.005), working memory (r = −0.240, p = 0.001), and declarative memory (r = −0.241, p < 0.001) measures. In the copy condition, a moderate and negative correlation was found between TCT and processing speed (r = −0.315, p < 0.001), while a weaker negative correlation was found between TCT and working memory (r = −0.140, p = 0.049). TCT remained negatively correlated with processing speed in both conditions after correcting for multiple comparisons. A follow up linear regression revealed that processing speed β = −0.172, t(−2.259), p = 0.025 and declarative memory β = −0.152, t(−2.078), p = 0.039 were significant predictors of command TCT with TCT accounting for 21.3% of the variance (Fig. 1).
Fig. 1.
Scatterplots of cognitive domain composites relative to Total Completion Time (in seconds) for all participants with group shown for comparison reference (solid black = MCI participants).
Pre-First-Hand Latency
In the command condition, PFHL was negatively correlated with working memory (r = −0.148, p = 0.039). In the copy condition PFHL was and negatively correlated with processing speed (r = −0.180, p = 0.011). Effect sizes were small and were not present after correcting for multiple comparisons.
Post-Clock Face Latency
In both conditions, negative correlations were found with small effect sizes between PCFL and processing speed (command: r = −0.161, p = 0.023; copy: r = −0.163, p = 0.022); these relationships were not present after correcting for multiple comparisons correction.
Graphomotor variable: Clock Face Area
In the copy, but not in the command condition, a negative correlation was found between CFA and language (command: r = −0.079, p = 0.278; copy: r = −0.200, p = 0.006) with small effect sizes. The relationship was not significant after multiple comparisons correction. CFA accuracy did not correlate with cognitive domains.
MCI and dCDT performance
Of the final sample (n = 202), 171 participants were cognitively well (non-MCI), while 31 met criteria for MCI [33] (15 amnestic MCI: 7 single-domain, 8 multi-domain; 16 non-amnestic MCI: 12 single-domain, 4 multi-domain). Groups differed on race as Non-Hispanic Whites were over represented in the MCI group (Table 1). Also, the MCI group was significantly less educated, had lower premorbid cognitive reserve, and scored lower on the TICS. Since slower time to completion is a ubiquitous cognitive aging phenomenon [34], therefore, our models included age as a covariate (Table 1). The homogeneity of variance-covariance assumption was met for the command (Box’s M value = 20.218, p = 0.04) and copy (Box’s M value = 20.019, p = 0.04).
Latency variables
Command
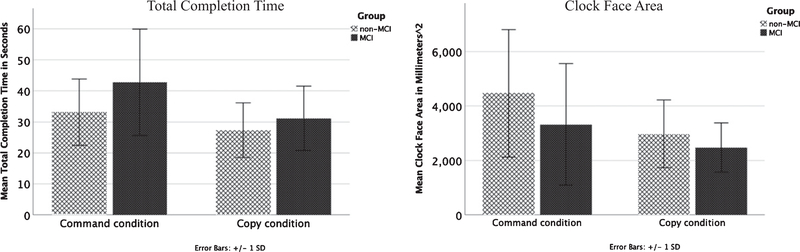
Overall multivariate omnibus was significant [Wilk’s Lambda = 0.938, F(4,185) = 3.050, p = 0.018]. Univariate analyses of MCI status on dCDT metrics indicated a significant difference in TCT, with the MCI group demonstrating slower total time to completion F(1,188) = 6.281, p = 0.013, η2 = 0.032 (Fig. 2).
Fig. 2.
MCI and non-MCI group comparisons on Total Completion Time and Clock Face Area for the command and copy conditions. MCI, mild cognitive impairment.
Copy
Overall multivariate omnibus was non-significant [Wilk’s Lambda = 0.963, F(4,192) = 1.852, p = 0.121]. Univariate analyses suggest an effect of MCI status on PFHL F(1,195) = 5.211, p = 0.024, η2 = 0.026, where the MCI group was slower to draw the first clock hand. We found no significant differences on TCT, and PCFL. Given the non-significant multivariate omnibus, univariate statistics for the copy condition should be interpreted with caution.
Graphomotor variable
Command
Univariate analyses show the MCI group produced smaller clocks F(1,188) = 4.501, p = 0.035, η2 = 0.023. Groups did not significantly differ in CFA ellipse residual suggesting similar clock face circle accuracy across groups (Fig. 2).
Copy
No significant differences were found between groups.
The repeated measures analysis of %Think time across conditions, revealed participants spent on average more time ‘thinking’ in the command relative to the copy condition [command %Think M = 0.55, SD = 0.82; copy %Think M = 0.52, SD = 0.83]; F(1,199) = 27.305, p < 0.001, η2 = 0.121. Trending strongly towards significance, there was a group by condition interaction such that the MCI group spent more time ‘thinking’ in the command relative to the copy condition F(1,199) = 3.556, p = 0.061, η2 = 0.018. The MCI group spent on average 9 seconds longer ‘thinking’ in the command than copy condition, while the non-MCI spent 4 seconds longer ‘thinking’ in the command versus copy.
DISCUSSION
The current research sought to augment our understanding of neuropsychological domains associated with specific clock drawing behavior, and to assess between-group differences comparing MCI to non-MCI adults age 60 and older. Among the important findings obtained in the current research were the differential relationships that appear to underlie TCT to command versus copy as related to traditional neuropsychological test performance. For example, TCT in the command condition with hands to “10 after 11” was significantly associated with multiple cognitive domains (i.e., processing speed, working memory, language, and declarative memory). Broadly speaking this finding is consistent with prior research described by Cosentino and colleagues [3] and Libon and colleagues [40] suggesting that a wide array of neuropsychological abilities must be properly coordinated in order to achieve maximum command test performance.
Command TCT most certainly has a foundational speed component; however, other cognitive operations are also necessary for optimal performance. For example, TCT in the command condition requires several subordinate cognitive operations involving graphomotor and non-motor behavior. Thus, slower PCFL (non-motor behavior) was associated with slower processing speed performance, a skill that is well known to be reduced with age [36]. These findings also suggest that slower TCT may indicate emergent degraded frontostriatal integrity beyond age and premorbid cognitive reserve. This is consistent with neuroimaging findings that myelination of white matter tracts predicts variability in processing speed performance. Indeed, dCDT TCT has also been observed to increase with age [41] and to be associated with decreased estimated myelin content involving white matter tracts [42]. Thus, digitally obtained latency variables such as TCT and PCFL may provide a quick and efficient indication of white matter integrity as related to proper frontal systems operations. In the command condition, cognitive domain correlates to PFHL were specifically related to working memory and the ability to disambiguate the syntactically complex proposition to set the clock hands for ‘10 after 11’. More effortful disambiguation of this syntactic proposition, reflected by a slower PFHL, may be indicative of greater working memory resources needed to achieve correct hand placement.
Another important finding in the current research were the more restricted relationships between TCT in the copy test condition involving only processing speed and working memory. This differential pattern of behavior seen in the current research is, again, consistent with prior research [3, 40] to the extent that clock drawing to command calls upon diverse cognitive operations whereas the domains associated with clock drawing to copy are more restricted, i.e., related to processing speed and working memory.
Though no significant relationship was identified between cognitive domains and clock face area to command, language was identified as an important underlying cognitive construct for clock face size in the copy condition, such that smaller clocks were observed in individuals with poorer performance on language abilities. Although these findings need to be further explored, smaller clocks may reflect relationships between semantics and planning appropriately sized clock face necessary for accurate number placement. Future research should examine how clock face area foretells number anchoring and spatial planning of numbers in MCI and individuals with dementia such as AD.
Indeed, the second goal of the current research was to assess for between-group differences in MCI and non-MCI peers. In the command test condition, MCI participants took longer to draw their clocks and were more likely to draw smaller clocks relative to non-MCI peers. Between-group differences were also found in percent ‘think time’. In the command test condition, the MCI group required 10 seconds longer on average to draw their clock from memory suggesting that differences between persons with and without MCI may lie in marshalling and coordinating necessary cognitive resources rather than in gross drawing errors. Although our entire sample spent more time ‘thinking’ than ‘inking’ (i.e., drawing), the MCI group displayed a larger discrepancy in %Think time between command and copy than non-MCI peers. Furthermore, individuals with MCI displayed more planning rather than conceptual deficits [43]; a phenomenon observed in Huntington’s disease patients who generally draw smaller clocks and exhibit planning deficits in laying out numbers [41].
Chiu and colleagues [42] observed that participants with early dementia also draw smaller circles relative to individuals with AD and healthy controls, a phenomenon that was interpreted as constructional difficulties. Consistent with this literature, our findings show that in the command condition MCI participants draw significantly smaller clock faces (on average 526.61 mm2, or 18% smaller). Although our MCI group consisted of different subtypes, some degree of executive functioning deficit is present in many MCI subtypes [44]. Smaller clock faces produced by the MCI group may, perhaps, indicate disruptions in frontostriatal and frontoparietal circuitry engendering executive dysfunctions above and beyond the product of cognitive aging. Our findings add to the existing literature documenting the advantage of incorporating digital technology into more traditional neuropsychological paradigms to capture nuanced behaviors, understand normal aging, and to detect early signs of cognitive deficits seen in MCI populations.
The current research study is not without limitations. First, our sample was primarily composed of well-educated non-Hispanic White older adults. Attempts to explore how ethnoracial and educational differences affected our results were not appropriate given the low representation of underrepresented groups in our study. Future studies exploring the underlying cognitive mechanisms involved in dCDT need to incorporate racial, ethnic, and educational diversity. Second, given the known contributions of cardiovascular disease risk on cognition, future investigations should consider its implications in clock drawing. Third, our MCI sample was small, limiting statistical power. While the overall multivariate omnibus test for the copy condition was nonsignificant, univariate analyses suggests that PFHL differed between groups. Thus, MCI group analyses may have been too underpowered to detect meaningful differences in the copy condition. Given the small MCI sample, we did not investigate differences across MCI subtypes. This should be considered in future studies. The MCI group was less educated, likely playing a role in premorbid cognitive reserve group differences, therefore, we controlled for it statistically. Finally, we used published normative references for creation of standardized cognitive composites. This is a limitation in that some of the published test norms come from different cohorts and may contain internal variance which we could not control for in our final analyses.
Nonetheless, the current research is notable for several strengths. First, a comprehensive neuropsychological protocol was used to characterize a large group of non-demented individuals. Second, our MCI group was classified according to rigorous published criteria that have been well validated through imaging and pathology reports [33]. Third, we considered covariates of accuracy in our analyses. Overall, digital clock drawing technology offers precise capture and scoring of latency, graphomotor output, and other neuropsychological behavior associated with specific neuropsychological constructs within a single task. Detecting and accurately quantifying nuanced behaviors within a single task provides clinically relevant information for differentiating healthy older adults from those with cognitive impairment.
In sum, future studies should expand upon our findings and investigate digital clock drawing variables relative to traditional neuropsychology measures in ethnoracially and educationally diverse samples and in the context of comorbid health conditions. Evaluating the utility of digital variables in identifying MCI subtypes may help to elucidate the underlying neurobiological mechanisms of clock drawing behaviors in older adults. Systematically quantifying objective measurements of size, angle, latency, and spacing between clock elements may be clinically useful in detecting subtle cognitive changes occurring in early stages of a disease process helping to differentiate between disease states. Finally, these research findings should be examined with other digital clock drawing platforms so that the scientific findings are not constrained to specific digital technology companies.
Table 3.
Partial Correlations between dCDT Metrics and Neuropsychological Domains
| Processing Speed p |
Language p |
Working Memory p |
Declarative Memory P |
|
|---|---|---|---|---|
| Command | ||||
| TCT | −0.284 | −0.201 | −0.240 | −0.241 |
| <0.001 | 0.005 | 0.001 | <0.001 | |
| PFHL | −0.084 | −0.083 | −0.148 | −0.004 |
| 0.242 | 0.246 | 0.039 | 0.953 | |
| PCFL | −0.161 | −0.048 | −0.042 | −0.137 |
| 0.023 | 0.498 | 0.559 | 0.055 | |
| CFA | 0.097 | −0.079 | 0.13 | −0.031 |
| 0.181 | 0.278 | 0.071 | 0.666 | |
| Copy | ||||
| TCT | −0.315 | −0.070 | −0.140 | −0.118 |
| <0.001 | 0.327 | 0.049 | 0.098 | |
| PFHL | −0.180 | −0.066 | −0.127 | −0.098 |
| 0.011 | 0.358 | 0.075 | 0.171 | |
| PCFL | −0.163 | −0.052 | −0.039 | −0.105 |
| 0.022 | 0.466 | 0.583 | 0.143 | |
| CFA | 0.065 | −0.200 | 0.027 | 0.045 |
| 0.365 | 0.006 | 0.703 | 0.534 |
TCT, total completion time; PFHL, pre-first-hand latency; PCFL, post-clock face latency.
ACKNOWLEDGMENTS
This work was supported by the National Institute of Health (grant no. R01AG055337, CP; P50AG047266, CP; R01NR014181; R01NS082386, CP; UL1R001427, CP), the National Science Foundation (grant no. 1404333, RD, DP, CP), the National Institute of Aging (grant no. T32-AG04963, FA).
Footnotes
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/19-1089r2).
REFERENCES
- [1].Libon D, Kaplan E, Swenson R, Penney D (2010) Clock drawing In The Encyclopedia of Clinical Neuropsychology,Kreutzer J, DeLuca J, Caplan B, eds. Springer, New York. [Google Scholar]
- [2].Kaplan E (1990) The process approach to neuropsychological assessment of psychiatric patients. J Neuropsychiatry Clin Neurosci 2, 72–87. [DOI] [PubMed] [Google Scholar]
- [3].Cosentino S, Jefferson A, Chute DL, Kaplan E, Libon DJ (2004) Clock drawing errors in dementia: Neuropsychological and neuroanatomical considerations. Cogn Behav Neurol 17, 74–84. [DOI] [PubMed] [Google Scholar]
- [4].Penney D, Davis R, Libon D, Lamar M, Price C, Swenson R, Garrett K (2011) The Digital Clock Drawing Test (dCDT) – II: A new computerized quantitative system. J Int Neuropsychol Soc 17, 274. [Google Scholar]
- [5].Piers RJ, Devlin KN, Ning B, Liu Y, Wasserman B, Massaro JM, Lamar M, Price CC, Swenson R, Davis R, Penney DL, Au R, Libon DJ (2017) Age and graphomotor decision making assessed with the digital Clock Drawing Test: The Framingham Heart Study. J Alzheimers Dis 60, 1611–1620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Libon D, Penney D, Lamar M, Swenson R, Garrett K, Davis R (2011) Digital Clock Drawing Test (dCDT) – V: Using total drawing time and latencies to assess decision making and capacity to shift mental set in patient with Alzheimer’s disease and subcortical vascular dementia. J Int Neuropsychol Soc 17, 271. [Google Scholar]
- [7].Eknoyan D, Hurley RA, Taber KH (2012) The Clock Drawing Task: Common errors and functional neuroanatomy. J Neuropsychiatry Clin Neurosci 24, 260–265. [DOI] [PubMed] [Google Scholar]
- [8].Salthouse TA (1996) The processing-speed theory of adult age differences in cognition. Psychol Rev 103, 403–428. [DOI] [PubMed] [Google Scholar]
- [9].Inzelberg R, Harpaz NK, Flash T (2016) Micrographia, much beyond the writer’s hand. Parkinsonism Relat Disord 26, 1–9. [DOI] [PubMed] [Google Scholar]
- [10].Levinoff EJ, Saumier D, Chertkow H (2005) Focused attention deficits in patients with Alzheimer’s disease and mild cognitive impairment. Brain Cogn 57, 127–130. [DOI] [PubMed] [Google Scholar]
- [11].Turken U, Whitfield-Gabrieli S, Bammer R, Baldo JV, Dronkers NF, Gabrieli JDE (2008) Cognitive processing speed and the structure of white matter pathways: Convergent evidence from normal variation and lesion studies. Neuroimage 42, 1032–1044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Filley C (2012) The Behavioral Neurology of White Matter, Oxford University Press, USA. [Google Scholar]
- [13].Lawton MP, Brody EM (1969) Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontologist 9, 8. [PubMed] [Google Scholar]
- [14].American Psychiatric Association (2013) Diagnostic and Statistical Manual of Mental Disorders (DSM-5®), American Psychiatric Publishing, Washington, DC. [Google Scholar]
- [15].Welsh KA, Breitner JC, Magruder-Habib KM (1993) Detection of dementia in the elderly using telephone screening of cognitive status. Neuropsychiatry Neuropsychol Behav Neurol 6, 103–110. [Google Scholar]
- [16].Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J Chronic Dis 40, 373–383. [DOI] [PubMed] [Google Scholar]
- [17].Davis R, Penney DL, Pittman D, Libon DJ, Swenson R, Kaplan E (2011) The Digital Clock Drawing Test (dCDT) I: Development of a new computerized quantitative system. J Int Neuropsychol Soc 17, 273. [Google Scholar]
- [18].Souillard-Mandar W, Davis R, Rudin C, Au R, Libon DJ, Swenson R, Price CC, Lamar M, Penney DL (2016) Learning classification models of cognitive conditions from subtle behaviors in the digital Clock Drawing Test. Mach Learn 102, 393–441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Hizel LP, Warner ED, Wiggins ME, Tanner JJ, Parvataneni H, Davis R, Penney DL, Libon DJ, Tighe P, Garvan CW, Price CC (2018) Clock drawing performance slows for older adults after total knee replacement surgery. Anesth Analg 129, 212–219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Demakis GJ (2004) Frontal lobe damage and tests of executive processing: A meta-analysis of the category test, Stroop test, and trail-making test. J Clin Exp Neuropsychol 26, 441–450. [DOI] [PubMed] [Google Scholar]
- [21].Stuss DT, Bisschop SM, Alexander MP, Levine B, Katz D, Izukawa D (2001) The trail making test: A study in focal lesion patients. Psychol Assess 13, 230–239. [PubMed] [Google Scholar]
- [22].Leskelä M, Hietanen M, Kalska H, Ylikoski R, Pohjasvaara T, Mäntylä R, Erkinjuntti T (1999) Executive functions and speed of mental processing in elderly patients with frontal or nonfrontal ischemic stroke. Eur J Neurol 6, 653–661. [DOI] [PubMed] [Google Scholar]
- [23].Gerton BK, Brown TT, Meyer-Lindenberg A, Kohn P, Holt JL, Olsen RK, Berman KF (2004) Shared and distinct neurophysiological components of the digits forward and backward tasks as revealed by functional neuroimaging. Neuropsychologia 42, 1781–1787. [DOI] [PubMed] [Google Scholar]
- [24].Aleman A, van’t Wout M (2008) Repetitive transcranial magnetic stimulation over the right dorsolateral prefrontal cortex disrupts digit span task performance. Neuropsychobiology 57, 44–48. [DOI] [PubMed] [Google Scholar]
- [25].Canavan AGM, Passingham RE, Marsden CD, Quinn N, Wyke M, Polkey CE (1989) Sequencing ability in Parkinsonians, patients with frontal lobe lesions and patients who have undergone unilateral temporal lobectomies. Neuropsychologia 27, 787–798. [DOI] [PubMed] [Google Scholar]
- [26].Toepper M, Gebhardt H, Beblo T, Thomas C, Driessen M, Bischoff M, Blecker CR, Vaitl D, Sammer G (2010) Functional correlates of distractor suppression during spatial working memory encoding. Neuroscience 165, 1244–1253. [DOI] [PubMed] [Google Scholar]
- [27].Kaplan E, Goodglass H, Weintraub S (2001) Boston Naming Test, Lippincott Williams & Wilkins. [Google Scholar]
- [28].Hirni DI, Kivisaari SL, Monsch AU, Taylor KI (2013) Distinct neuroanatomical bases of episodic and semantic memory performance in Alzheimer’s disease. Neuropsychologia 51, 930–937. [DOI] [PubMed] [Google Scholar]
- [29].Trifilio E, Tanner JJ, Butterfield L, Mangal P, Maye JE, Marsiske M, Price CC, Bowers D (2020) A tale of two stories: Validity of an alternative story memory test in a sample of older adults. Clin Neuropsychol 34, 158–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Lee TMC, Yip JTH, Jones-Gotman M (2002) Memory deficits after resection from left or right anterior temporal lobe in humans: A meta-analytic review. Epilepsia 43, 283–291. [DOI] [PubMed] [Google Scholar]
- [31].Bonner-Jackson A, Mahmoud S, Miller J, Banks SJ (2015) Verbal and non-verbal memory and hippocampal volumes in a memory clinic population. Alzheimers Res Ther 7, 61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Sexton CE, Mackay CE, Lonie JA, Bastin ME, Terrière E, O’Carroll RE, Ebmeier KP (2010) MRI correlates of episodic memory in Alzheimer’s disease, mild cognitive impairment, and healthy aging. Psychiatry Res Neuroimaging 184, 57–62. [DOI] [PubMed] [Google Scholar]
- [33].Jak AJ, Bondi MW, Delano-Wood L, Wierenga C, Corey- Bloom J, Salmon DP, Delis DC (2009) Quantification of five neuropsychological approaches to defining mild cognitive impairment. Am J Geriatr Psychiatry 17, 368–375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Salthouse TA, Fristoe N, Rhee SH (1996) How localized are age-related effects on neuropsychological measures? Neuropsychology 10, 272. [Google Scholar]
- [35].Beaudreau SA, O’Hara R (2009) The association of anxiety and depressive symptoms with cognitive performance in community-dwelling older adults. Psychol Aging 24, 507–512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Schrijvers D, Hulstijn W, Sabbe BGC (2008) Psychomotor symptoms in depression: A diagnostic, pathophysiological and therapeutic tool. J Affect Disord 109, 1–20. [DOI] [PubMed] [Google Scholar]
- [37].Cohen J, Penney DL, Davis R, Libon DJ, Swenson RA, Ajilore O, Kumar A, Lamar M (2014) Digital Clock Drawing: Differentiating “thinking” versus “doing” in younger and older adults with depression. J Int Neuropsychol Soc 20, 920–928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Stern Y (2009) Cognitive reserve. Neuropsychologia 47, 2015–2028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Lezak MD, Howieson DB, Bigler ED, Tranel D (2012) Neuropsychological Assessment, Oxford University Press, USA. [Google Scholar]
- [40].Libon DJ, Malamut B, Swenson R, Prouty Sands L, Cloud BS (1996) Further analyses of clock drawings among demented and nondemented older subjects. Arch Clin Neuropsychol 11, 193–205. [PubMed] [Google Scholar]
- [41].Rouleau I, Salmon DP, Butters N, Kennedy C, McGuire K (1992) Quantitative and qualitative analyses of clock drawings in Alzheimer’s and Huntington’s disease. Brain Cogn 18, 70–87. [DOI] [PubMed] [Google Scholar]
- [42].Chopra S, Shaw M, Shaw T, Sachdev PS, Anstey KJ, Cherbuin N (2018) More highly myelinated white matter tracts are associated with faster processing speed in healthy adults. NeuroImage 171, 332–340. [DOI] [PubMed] [Google Scholar]
- [43].Yamamoto S, Mogi N, Umegaki H, Suzuki Y, Ando F, Shimokata H, Iguchi A (2004) The Clock Drawing Test as a valid screening method for mild cognitive impairment. Dement Geriatr Cogn Disord 18, 172–179. [DOI] [PubMed] [Google Scholar]
- [44].Reinvang I, Grambaite R, Espeseth T (2012) Executive dysfunction in MCI: Subtype or early symptom. Int J Alzheimers Dis 2012, 1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]