Highlights
-
•
Covid-19 disease show gastrointestinal symptoms in some patients hinting at a role of gut-lung axis.
-
•
Gut microbiota diversity and its role in immunity highlighted.
-
•
Possible role of gut microbiota in Covid-19 discussed.
-
•
Implications of gut dysbiosis in Covid-19 analysed.
-
•
Role of diet in strengthening the gut microbiota and in the context of Covid-19 discussed.
-
•
Suggestions on therapy and prophylaxis based on gut microbiota in Covid-19.
Keywords: Covid-19, Gut microbiome, SARS-CoV-2, Immunity, Dysbiosis, Lung microbiota, Diet
Abstract
Covid-19 is a major pandemic facing the world today caused by SARS-CoV-2 which has implications on our understanding of infectious diseases. Although, SARS-Cov-2 primarily causes lung infection through binding of ACE2 receptors present on the alveolar epithelial cells, yet it was recently reported that SARS-CoV-2 RNA was found in the faeces of infected patients. Interestingly, the intestinal epithelial cells particularly the enterocytes of the small intestine also express ACE2 receptors. Role of the gut microbiota in influencing lung diseases has been well articulated. It is also known that respiratory virus infection causes perturbations in the gut microbiota. Diet, environmental factors and genetics play an important role in shaping gut microbiota which can influence immunity. Gut microbiota diversity is decreased in old age and Covid-19 has been mainly fatal in elderly patients which again points to the role the gut microbiota may play in this disease. Improving gut microbiota profile by personalized nutrition and supplementation known to improve immunity can be one of the prophylactic ways by which the impact of this disease can be minimized in old people and immune-compromised patients. More trials may be initiated to see the effect of co-supplementation of personalized functional food including prebiotics/probiotics along with current therapies.
1. Introduction
Coronavirus disease 2019 or Covid-19 is a new public health crisis threatening the humanity. Although, it originated in the Hubei province of China in late 2019 yet it has spread to many countries in the world (Wang et al., 2020). This pandemic disease is caused by the novel betacoronavirus, now named SARS-Cov-2(Lake, 2020). Covid-19 has very important clinical features such as high rates of transmission, mild to moderate clinical manifestation with more serious radiological abnormalities seen in the elderly (Yuen et al., 2020).
Coronaviruses are positive sense RNA viruses with spike like projections on its enveloped surface giving it a crown like appearance hence the term Coronavirus (Singhal, 2020). Phylogenetic diversity studies have highlighted that SARS-Cov-2 shares 79% nucleotide sequence identity with another virus of the same family SARS-Cov which caused major epidemic in 2002–2003 that resulted in 8000 cases in 26 countries (De Wit et al., 2016). Additionally, SARS-Cov-2 also displays sequence identities of 96% and 89.6%, for the envelope and nucleocapsid proteins respectively with SARS-Cov (Zhou et al., 2020b). Middle East respiratory syndrome or MERS disease in 2012 was also caused by another coronavirus called MERS-CoV. All these viruses utilize the ACE-2 receptor for their cellular entry (Zhou et al., 2020a). Interestingly, ACE-2 receptors are also reported to be expressed in the kidney and gastrointestinal tract, tissues known to harbour SARS-CoV (Harmer et al., 2002; Leung et al., 2003). Earlier evidence suggested that SARS coronavirus viral RNA was detectable in the respiratory secretions and stool of some patients after onset of illness for more than one month but live virus could not be cultured after third week (Chan et al., 2004). Now, recent report also suggests that SARS-Cov2 RNA can be detected in stool of some patients of Covid-19 (Wu et al., 2020). This along with the fact that some patients of this disease have diarrhoea points out towards a distinct possibility of involvement of gut-lung axis and may be the gut microbiota (Chan et al., 2020).
2. Gut microbiota and the gut-lung axis
The human gut microbiota consists of 1014 resident microorganisms which include bacteria, archae, viruses and fungi (Gill et al., 2006). Primarily, the gut bacteria in healthy individuals is dominated by four phyla Actinobacteria, Firmicutes, Proteobacteria, and Bacteroidetes (Villanueva-Millán et al., 2015). The colon harbours an extremely high density of bacteria in the families Bacteroidaceae, Prevotellaceae, Rikenellaceae, Lachnospiraceae and Ruminococcaceae (Hall et al., 2017). The gut microbiota plays a key role in health through its protective, trophic and metabolic actions.
While the microbes get a habitat and nourishment from the host, these microbes in turn help the host by regulating various host physiological functions, including dietary digestion, and imparting protective immunity against pathogens. Alterations of gut microbiota sometimes collectively called as “gut dysbiosis” have been shown to be associated with various diseases and disorders like IBD (Khan et al., 2019), type 2 diabetes (Gurung et al., 2020), depression (Zalar et al., 2018), cardiovascular disease (Tang et al., 2017).
Like the gut microbiota, there are evidences now that suggest the presence of distinct microorganisms in the lung (Bingula et al., 2017). In the gut Bacteroidetes and Firmicutes are predominant while Bacteroidetes, Firmicutes, and Proteobacteria preponderate in the lung (Zhang et al., 2020). Interestingly, the gut microbiota has been shown to affect pulmonary health through a vital cross-talk between the gut microbiota and the lungs which is referred to as the “gut-lung axis”(Keely et al., 2012). The gut-lung axis is supposed to be bidirectional, meaning the endotoxins, microbial metabolites can impact the lung through blood and when inflammation occurs in the lung, it can affect the gut microbiota as well (Dumas et al., 2018). This raises an interesting possibility that novel SARS-Cov2 might also have an impact on the gut microbiota. In fact, several studies have demonstrated that respiratory infections are associated with a change in the composition of the gut microbiota (Groves et al., 2020). One of the serious clinical manifestations of Covid-19 is pneumonia and progression to acute respiratory distress syndrome (ARDS) especially in elderly, immune-compromised patients (Lake, 2020). Numerous experimental and clinical observations have suggested that the gut microbiota plays a key role in the pathogenesis of sepsis and ARDS (Dickson and Arbor, 2017). Loss of gut bacteria diversity can lead to dysbiosis which may then be associated with many diseases (Mosca et al., 2016). In fact, elderly people have less diverse gut microbiota and beneficial microorganisms like bifidobacterium lose ground (Nagpal et al., 2018). Since, many elderly and immune-compromised patients progress to serious adverse clinical outcomes, it is therefore tempting to speculate that in Covid-19, there is a possible cross-talk taking place between the lung and the gut microbiota which might influence the outcome of the clinical manifestation.
2.1. Gut microbiota – role in immunity
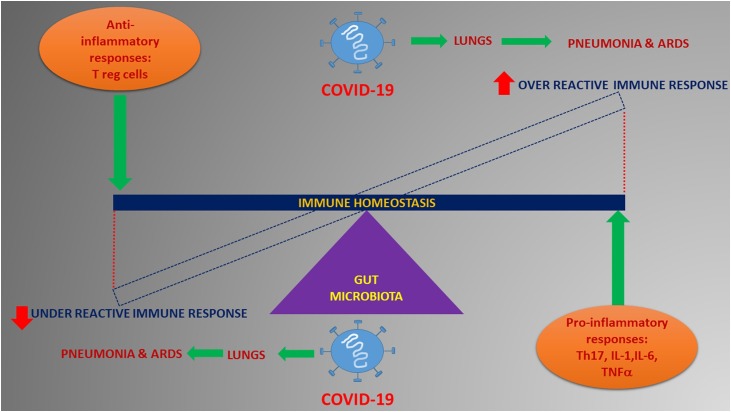
The interactions of the host with the microbiota are complex, numerous and bidirectional. The gut microbiota are supposed to significantly regulate the development and function of the innate and adaptive immune system (Negi and Das, 2019). Intestinal commensals secrete antimicrobial peptides, compete for the nutrients and the habitat site thereby aiding in the state of homeostasis (Moens and Veldhoen, 2012). The gut microbiota and immune homeostasis seem to have a back and forth relationship and is also a field of great interest and intense research investigation in the field of infectious diseases. Also, gut microbiota derived signals are known to tune the immune cells for pro and anti-inflammatory responses thereby affecting the susceptibility to various diseases (Negi and Pahari, 2019). The immune gut homeostasis is orchestrated by the fine tuning of the regulatory balance of pro-inflammatory responses such as Th17 versus inflammatory regulatory T cells (Tregs) that is ultimately controlled by the commensal microorganisms (Round and Mazmanian, 2010). In circumventing a response to pathogenic infections like coronavirus, a healthy gut microbiome essentially could be pivotal in maintaining an optimal immune system to prevent an array of excessive immune reactions that eventually become detrimental to lungs and vital organ systems. In such circumstances, it becomes imperative to have a balanced immune response wherein an over reactive one or an under reactive one can equally be consequential to aggravate clinical complications like pneumonia and ARDS in a viral disease like Covid-19 (Fig. 1 ).
Fig. 1.
Possible role of the gut microbiota in modulating immune response in Covid-19.
Gut microbiota can influence immune response thereby affecting the disease progression. Both over-active and under active immune response possibly mediated by the gut microbiota can lead to serious clinical adverse events
Microorganisms serve as a source of microorganism-associated molecular patterns (MAMPs) as well as pathogen-associated molecular patterns (PAMPs). The two are recognizable on the host’s cells through pattern recognition receptors (PRRs), which include toll-like receptors (TLRs) and nucleotide-binding receptors (NODs) (Ivanov and Honda, 2012). TLRs recognize the MAMPS and PAMPs among other molecules and evoke different immunological reactions depending on the type of cell, ligand or receptor. Training of PRRs expressing innate cells with gut microbial/non-microbial ligands is needed as a protective mechanism independent of adaptive immunity during secondary infection/pathogenic exposures. Importantly, the gut microbiota secreted metabolites and immunomodulatory signals that comprise the short chain fatty acids (SCFA) like the butyrate, acetate and propionate and secondary bile acids secreted by commensals like bacteroides, lactobacillus, and bifidobacteria bind to their receptors in innate cells such as DCs and macrophages thereby modulating their metabolism and functions (Rooks and Garrett, 2016; Jia et al., 2018). In fact, introduction of probiotic strains such as Bifidobacterium lactis into healthy elderly volunteers resulted in a significant increase in the proportion of mononuclear leukocytes, and the tumoricidal activity of NK cells (Gill et al., 2001). The composition of balanced gut microbiota is known to have a major influence on the effectiveness of lung immunity (Bingula et al., 2017). Germ free mice (GF mice), devoid of their intestinal microbiota have been shown to have impaired pathogen clearance capability in the lung (Fagundes et al., 2012). Disruption of gut microbiota with widespread antibiotic use can also have similar effect as was observed in population studies showing that increased use of penicillins, cephalosporins, macrolides, and quinolones correlated with an increased risk of lung cancer in humans (Boursi et al., 2015). Interestingly, influenza viral infection in the respiratory tract in mice increases Enterobacteriaceae as well reduces Lactobacilli and Lactococci in the intestinal microbiota (Looft and Allen, 2012). Taken together, it can be suggested that since the gut microbiota play such an important role in immunity, infection caused by SARS-Cov2 need to be properly studied with respect to the role played by the intestinal and lung commensal microorganisms.
3. Nutrition and gut microbiota – strengthening the reservoir
Diet plays an important role in shaping the composition of the gut microbiota thereby influencing the host’s health status. Various diet forms are found to influence the specific compositional patterns of the gut microbiota like, for e.g., the different composition of the microbiota with animal fat and protein-based diets versus vegetable-based diets has been reported (De Filippis et al., 2016). It is interesting to note that the gut microbiota of animals fed with a high fat or high sugar diet are more prone to circadian rhythm disruption (Voigt et al., 2014). Conversely, systemic stress, tissue injury and sustained inflammation can also produce acute changes in the gut microbiota thereby proving that environmental factors along with diet can modulate the composition of the gut microbiome (Earley et al., 2015). Encouragingly, various components of food have been shown to have differential effects on the gut microbiota. For instance, the consumption of the protein extracts of whey and pea increases gut-commensal bacteria bifidobacterium and lactobacillus, while whey is additionally shown to decrease the pathogenic bacteria Bacteroides fragilis and Clostridium perfringens (Dominika et al., 2011). Similarly, it was noted that consumption of a low-fat diet led to increased faecal abundance of bifidobacterium. On the other hand, a high saturated fat diet increased the relative proportion of Faecalibacterium prausnitzii (Singh et al., 2017). In contrast to the digestible carbohydrates, non-digestible carbohydrates such as fibre and resistant starch are known to undergo fermentation by the resident microorganisms in the intestine (De Filippis et al., 2016). Dietary fibres are good source of microbiota accessible carbohydrates which provide the host with energy and improves intestinal health. Prebiotics have been studied in the context of modification of the human gut microbiota. Prebiotic compounds such as inulin, polydextrose, maize fibre have been shown to improve the immunity, gut diversity, digestion etc. in humans and especially in elderly people (Kleessen et al., 1997; Bouhnik et al., 2007). In addition to the effects on the composition of the microbiota, prebiotics also produce notable shifts in immune and metabolic markers. Example, it was observed that consumption of non-digestible carbohydrates present in whole grains led to reductions in the proinflammatory cytokine IL-6 and insulin resistance (Keim and Martin, 2014). Similarly, increased plasma levels of anti-inflammatory cytokine, IL10, was observed with the intake of butyrylated high amylose maize starch (West et al., 2013). It is to be noted that beneficial effects of the prebiotics are thought to be mediated mostly by increased production of SCFAs and strengthening of the gastro-intestinal associated lymphoid tissue (GALT) (Schley and Field, 2002). Thought-provoking results have shown that a fibre rich diet changes not only the intestinal microbiota, but can also affect the lung microbiota, indicating influence of nutrition on lung immunity (Trompette et al., 2014). Like prebiotics, role of probiotics which is generally defined as “live microorganisms, which, when administered in adequate amounts, confer a health benefit on the host” have been shown to have profound effect on health of the host. In the intestine the probiotics mainly refer to the genera Lactobacillus and Bifidobacterium and include many different strains such as L. johnsonii, L. fermentum, L. reuteri, L. paracasei, L. rhamnosus, L. acidophilus, L. plantarum, B. longum, B. breve, B. bifidum, and B. animalis subsp. Lactis (Bingula et al., 2017). Fermented foods such as cultured milk products and yogurt are enriched in probiotics. Probiotic-containing yogurt has been found to significantly reduce counts of the enteropathogens E. coli and Helicobacter pylori (Yang and Sheu, 2012). Fascinatingly, lactobacilli and bifidobacteria have been used successfully for the prophylactic prevention of traveller’s diarrhoea (McFarland, 2007). Probiotics have shown good results in improving inflammatory conditions as well as regulating innate immunity using toll-like receptors and the corresponding signalling pathways (West et al., 2017). Mice model-based research has shown that Treg cells, which down-regulate the allergic response, can be induced by the administration of probiotic bacteria like Lactobacillus rhamnosus, Bifidobacterium lactis, and Bifidobacterium breve (Feleszko et al., 2007). Overall, it is apparent that diet mediated modulation of gut microbiota and to some extent even lung microbiota can influence immunity. Therefore, diet especially, personalized, may improve prophylaxis and can be thoughtfully administered to patients affected with Covid-19 to accelerate recovery and improve clinical outcomes.
4. Conclusion and future perspective
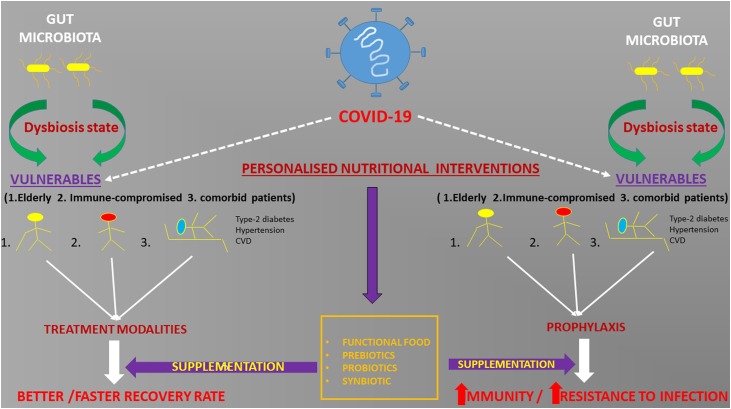
Covid-19 has pushed the world to the brink. The faster we understand this disease the better we will be prepared the next time. Research in gut microbiota has propelled our knowledge in the field of chronic and infectious diseases. The presence of SARS Cov2 RNA in the stool of some patients and diarrhoea in few suggest a subtle link between the lung and the intestine. Although, no faecal-oral transmission is reported, yet it can be assumed that many asymptomatic children and adults may shed infectious virus particles in the stool leading to infection in others. Gut microbiota diversity and the presence of beneficial microorganisms in the gut may play an important role in determining the course of this disease. Elderly, immune-compromised patients and patients with other co-morbidities like type-2 diabetes, cardiovascular disorders fare poorly in combating Covid-19. It is interesting to note that a general imbalance of gut microbiota called “dysbiosis” is implicated in such patients and the elderly. Interestingly, it is known that in murine models, removal of certain gut bacteria by antibiotic leads to increased susceptibility to influenza virus infection in lungs (Looft and Allen, 2012). Also, several bacterial metabolites and bacterial fragments can modulate lung immune response (Trompette et al., 2014). So, it is quite a possibility that gut dysbiosis may be influencing the clinical manifestation in Covid-19 as well. Microbial action on dietary fibre is known to increase short chain fatty acids (SCFA) in blood thereby protecting against allergic inflammation in the lungs (Trompette et al., 2014). In fact, prebiotics such as wheat bran and fructo-oligosachharides (Fos), galactosachharides (Gos) are known to increase butyrate levels thereby reducing inflammation and improving conditions in asthma and cystic fibrosis (Anand and Mande, 2018). Similarly, many probiotics have been shown to improve or alleviate lung disease conditions. These probiotics show the effect by modulating the immune system. Mice model studies have shown that introduction of probiotic bacteria like Lactobacillus rhamnosus, Bifidobacterium lactis and Bifidobacterium breve can down regulate allergic response (Feleszko et al., 2007). Likewise, administration of Lactobacillus casei Shirota or Lactobacillus rhamnosus GG in cystic fibrosis patients leads to improvement in their conditions (West et al., 2017). Since, gut microbiota is malleable and is modulated by diet, it is imperative that personalized diet strategies may be implemented as a supplement to current routine therapies. This can be done by profiling gut microbiota of the individual patients and recommending effective diet including specialized pre/probiotics such as FOS, GOS and various lactobacilli strains to improve gut dysbiosis and thereby improving overall immune response in such patients. This may improve and fasten recovery in patients especially the elderly and the immune-compromised who are infected with SARS-Cov2 virus (Fig. 2 ). Another line of action may be to prophylactically provide specialized supplements including prebiotics, probiotics to care givers who are in the front line in tackling this disease. Thus, effective nutritional strategy and specific functional foods aiming at the microbiota for specific population group may be the need of the hour. Research may be conducted to look at the effect of Covid-19 on the gut microbiota profile and vice versa. Secondly, it would be interesting to dissect the role of not only bacteria but other microorganisms such as fungi and phages that form a very niche ecosystem in the intestine in Covid-19. Finally, role of the lung microbiota may also be investigated. Insights from such studies will add new dimensions to understand infectious diseases and can help in taking decisive actions in future.
Fig. 2.
Personalized nutritional strategies as prophylaxis and in treatment supplementation.
Personalized nutritional strategies may be adopted for the Covid-19 vulnerable sections both as prophylaxis and supplementation to current treatment modalities.
Declaration of Competing Interest
Debojyoti Dhar is the Director of Leucine Rich Bio Pvt Ltd.; There is no financial disclosure
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Contributor Information
Debojyoti Dhar, Email: ddhar@leucinerichbio.com.
Abhishek Mohanty, Email: pro@rgcirc.org.
References
- Anand S., Mande S.S. Diet, microbiota and gut-lung connection. Front. Microbiol. 2018;9(September) doi: 10.3389/fmicb.2018.02147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bingula R. Desired turbulence? Gut-lung Axis, immunity, and lung Cancer. J. Oncol. 2017:2017. doi: 10.1155/2017/5035371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouhnik Y. Four-week short chain fructo-oligosaccharides ingestion leads to increasing fecal bifidobacteria and cholesterol excretion in healthy elderly volunteers. Nutrition Journal. BioMed Central. 2007;6:42. doi: 10.1186/1475-2891-6-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boursi B. Recurrent antibiotic exposure may promote cancer formation-Another step in understanding the role of the human microbiota? Eur. J. Cancer. 2015;51(17):2655–2664. doi: 10.1016/j.ejca.2015.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan K.H. Detection of SARS coronavirus in patients with suspected SARS. Emerging Infect. Dis. 2004;10(2):294–299. doi: 10.3201/eid1002.030610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan J.F.W. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet. 2020;395(10223):514–523. doi: 10.1016/S0140-6736(20)30154-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Filippis F. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut. 2016;65(11):1–10. doi: 10.1136/gutjnl-2015-309957. [DOI] [PubMed] [Google Scholar]
- De Wit E. SARS and MERS: recent insights into emerging coronaviruses. Nat. Rev. Microbiol. 2016;14(8):523–534. doi: 10.1038/nrmicro.2016.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickson R.P., Arbor A. The microbiome and critical illness. Lancet Respir. Med. 2017;4(1):59–72. doi: 10.1016/S2213-2600(15)00427-0.The. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dominika Ś. The study on the impact of glycated pea proteins on human intestinal bacteria. Int. J. Food Microbiol. 2011;145(1):267–272. doi: 10.1016/j.ijfoodmicro.2011.01.002. [DOI] [PubMed] [Google Scholar]
- Dumas A. The role of the lung microbiota and the gut–lung axis in respiratory infectious diseases. Cell. Microbiol. 2018 doi: 10.1111/cmi.12966. [DOI] [PubMed] [Google Scholar]
- Earley Z.M. Burn injury alters the intestinal microbiome and increases gut permeability and bacterial translocation. PLoS One. 2015;10(7):e0129996. doi: 10.1371/journal.pone.0129996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fagundes C.T. Transient TLR activation restores inflammatory response and ability to control pulmonary bacterial infection in Germfree Mice. J. Immunol. 2012;188(3):1411–1420. doi: 10.4049/jimmunol.1101682. [DOI] [PubMed] [Google Scholar]
- Feleszko W. Probiotic-induced suppression of allergic sensitization and airway inflammation is associated with an increase of T regulatory-dependent mechanisms in a murine model of asthma. Clin. Exp. Allergy. 2007;37(4):498–505. doi: 10.1111/j.1365-2222.2006.02629.x. [DOI] [PubMed] [Google Scholar]
- Gill H.S. Enhancement of immunity in the elderly by dietary supplementation with the probiotic Bifidobacterium lactis HN019. Am. J. Clin. Nutr. 2001;74(6):833–839. doi: 10.1093/ajcn/74.6.833. [DOI] [PubMed] [Google Scholar]
- Gill S.R. Metagenomic analysis of the human distal gut microbiome. Science. 2006;312(5778):1355–1359. doi: 10.1126/science.1124234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groves H.T. Respiratory viral infection alters the gut microbiota by inducing inappetence. mBio. 2020;11(1):1–17. doi: 10.1128/mBio.03236-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurung M. Role of gut microbiota in type 2 diabetes pathophysiology. EBioMedicine. 2020;51:102590. doi: 10.1016/j.ebiom.2019.11.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall A.B., Tolonen A.C., Xavier R.J. Human genetic variation and the gut microbiome in disease. Nat. Rev. Genet. 2017;18(11):690–699. doi: 10.1038/nrg.2017.63. [DOI] [PubMed] [Google Scholar]
- Harmer D. Quantitative mRNA expression profiling of ACE 2, a novel homologue of angiotensin converting enzyme. FEBS Lett. 2002;532(1–2):107–110. doi: 10.1016/S0014-5793(02)03640-2. John Wiley & Sons, Ltd. [DOI] [PubMed] [Google Scholar]
- Ivanov I.I., Honda K. Intestinal commensal microbes as immune modulators. Cell Host Microbe. 2012:496–508. doi: 10.1016/j.chom.2012.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia Wei, Xie G., Jia Weiping. Bile acid–microbiota crosstalk in gastrointestinal inflammation and carcinogenesis. Nat. Rev. Gastroenterol. Hepatol. 2018:111–128. doi: 10.1038/nrgastro.2017.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keely S., Talley N.J., Hansbro P.M. Pulmonary-intestinal cross-talk in mucosal inflammatory disease. Mucosal Immunol. 2012;5(1):7–18. doi: 10.1038/mi.2011.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keim N.L., Martin R.J. Dietary whole grain–microbiota interactions: insights into mechanisms for human health. Adv. Nutr. 2014;5(5):556–557. doi: 10.3945/an.114.006536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan I. Alteration of gut microbiota in inflammatory bowel disease (IBD): cause or consequence? IBD treatment targeting the gut microbiome. Pathogens. 2019;8(3):1–28. doi: 10.3390/pathogens8030126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleessen B. Effects of inulin and lactose on fecal microflora, microbial activity, and bowel habit in elderly constipated persons. Am. J. Clin. Nutr. 1997;65(5):1397–1402. doi: 10.1093/ajcn/65.5.1397. [DOI] [PubMed] [Google Scholar]
- Lake M.A. What we know so far: COVID-19 current clinical knowledge and research. Clin. Med. Lond. (Lond) 2020:124–127. doi: 10.7861/clinmed.2019-coron. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung W.K. Enteric involvement of severe acute respiratory syndrome - Associated coronavirus infection. Gastroenterology. 2003;125(4):1011–1017. doi: 10.1016/j.gastro.2003.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Looft T., Allen H.K. Collateral effects of antibiotics on mammalian gut microbiomes. Gut Microbes. 2012;3(5):463. doi: 10.4161/gmic.21288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFarland L.V. Meta-analysis of probiotics for the prevention of traveler’s diarrhea. Travel Med. Infect. Dis. 2007;5(2 SPEC. ISS.):97–105. doi: 10.1016/j.tmaid.2005.10.003. [DOI] [PubMed] [Google Scholar]
- Moens E., Veldhoen M. Epithelial barrier biology: good fences make good neighbours. Immunology. 2012:1–8. doi: 10.1111/j.1365-2567.2011.03506.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosca A., Leclerc M., Hugot J.P. Gut microbiota diversity and human diseases: should we reintroduce key predators in our ecosystem? Front. Microbiol. 2016;7(March):1–12. doi: 10.3389/fmicb.2016.00455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagpal R. Gut microbiome and aging: physiological and mechanistic insights. Nutr. Healthy Aging. 2018;4(4):267–285. doi: 10.3233/NHA-170030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Negi S., Das D.K. Potential role of gut microbiota in induction and regulation of innate immune memory. Front. Immunol. 2019;10(October):1–12. doi: 10.3389/fimmu.2019.02441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Negi S., Pahari S. Gut microbiota regulates mincle mediated activation of lung dendritic cells to protect against mycobacterium tuberculosis. Front. Immunol. 2019;10(MAY):1142. doi: 10.3389/fimmu.2019.01142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rooks M.G., Garrett W.S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 2016:341–352. doi: 10.1038/nri.2016.42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Round J.L., Mazmanian S.K. Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc. Natl. Acad. Sci. U.S.A. 2010;107(27):12204–12209. doi: 10.1073/pnas.0909122107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schley P.D., Field C.J.* The immune-enhancing effects of dietary fibres and prebiotics. Br. J. Nutr. 2002;87(6):221–230. doi: 10.1079/bjnbjn/2002541. [DOI] [PubMed] [Google Scholar]
- Singh R.K. Influence of diet on the gut microbiome and implications for human health. J. Transl. Med. 2017;15(1):1–17. doi: 10.1186/s12967-017-1175-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singhal T. A review of coronavirus Disease-2019 (COVID-19) Indian J. Pediatr. 2020;87(April):281–286. doi: 10.1007/s12098-020-03263-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang W.H.W., Kitai T., Hazen S.L. Gut microbiota in cardiovascular health and disease. Circ. Res. 2017;120(7):1183–1196. doi: 10.1161/CIRCRESAHA.117.309715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trompette A. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat. Med. 2014;20(2):159–166. doi: 10.1038/nm.3444. [DOI] [PubMed] [Google Scholar]
- Villanueva-Millán M.J., Pérez-Matute P., Oteo J.A. Gut microbiota: a key player in health and disease. A review focused on obesity. J. Physiol. Biochem. 2015;71(3):509–525. doi: 10.1007/s13105-015-0390-3. [DOI] [PubMed] [Google Scholar]
- Voigt R.M. Circadian disorganization alters intestinal microbiota. PLoS One. 2014;9(5):e97500. doi: 10.1371/journal.pone.0097500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C. A novel coronavirus outbreak of global health concern. Lancet. 2020:470–473. doi: 10.1016/S0140-6736(20)30185-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- West N.P. Butyrylated starch increases colonic butyrate concentration but has limited effects on immunity in healthy physically active individuals. Exerc. Immunol. Rev. 2013;19:102–119. [PubMed] [Google Scholar]
- West C.E. Bugging allergy; role of pre-, pro- and synbiotics in allergy prevention. Allergol. Int. 2017:529–538. doi: 10.1016/j.alit.2017.08.001. [DOI] [PubMed] [Google Scholar]
- Wu Y. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol. Hepatol. 2020;1253(20):20–21. doi: 10.1016/S2468-1253(20)30083-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y.-J., Sheu B.-S. Probiotics-containing yogurts suppress Helicobacter pylori load and modify immune response and intestinal microbiota in the Helicobacter pylori-infected children. Helicobacter. 2012;17(4):297–304. doi: 10.1111/j.1523-5378.2012.00941.x. [DOI] [PubMed] [Google Scholar]
- Yuen K.-S. SARS-CoV-2 and COVID-19: the most important research questions. Cell Biosci. 2020;10(1):40. doi: 10.1186/s13578-020-00404-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zalar B., Haslberger A., Peterlin B. The role of microbiota in depression - A Brief review. Psychiatr. Danub. 2018;30(2):136–141. doi: 10.24869/spsih.2018.136. [DOI] [PubMed] [Google Scholar]
- Zhang D. The cross-talk between gut microbiota and lungs in common lung diseases. Front. Microbiol. 2020;11(February):1–14. doi: 10.3389/fmicb.2020.00301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou P. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y. Network-based drug repurposing for novel coronavirus 2019-nCoV/SARS-CoV-2. Cell Discov. 2020;6(1):1–18. doi: 10.1038/s41421-020-0153-3. [DOI] [PMC free article] [PubMed] [Google Scholar]