Abstract
Children’s development is largely dependent on caregiving; when caregiving is disrupted, children are at increased risk for numerous poor outcomes, in particular psychopathology. Therefore, determining how caregivers regulate children’s affective neurobiology is essential for understanding psychopathology etiology and prevention. Much of the research on affective functioning uses fear learning to map maturation trajectories, with both rodent and human studies contributing knowledge. Nonetheless, as no standard framework exists through which to interpret developmental effects across species, research often remains siloed, thus contributing to the current therapeutic impasse. Here, we propose a developmental ecology framework that attempts to understand fear in the ecological context of the child: their relationship with their parent. By referring to developmental goals that are shared across species (to attach to, then, ultimately, separate from the parent), this framework provides a common grounding from which fear systems and their dysfunction can be understood, thus advancing research on psychopathologies and their treatment.
Keywords: fear, development, stress, parental buffering, ecology, human, rodent
INTRODUCTION
Psychopathologies are a leading cause of morbidity across the developed world (Kessler & Greenberg 2002), and it is now appreciated that most mental illness is the result of a neurodevelopmental process (Leonardo & Hen 2008). For example, the median age of onset of clinical anxiety is just 11 years (Kessler et al. 2005), and the impact of the illness is compounded when emerging in early life; early onset profiles are associated with anxiety chronicity (Woodward & Fergusson 2001). These findings provide an impetus for researchers to examine the developmental pathways and early-life risk factors that may underlie the emergence of mental illness.
One factor that greatly influences emotional functioning and anxiety across development is the family unit. As recognized long ago by theorists such as Urie Bronfenbrenner (1977) and John Bowlby (1969), caregivers are the essential unit driving children’s behavioral as well as neural development, especially within emotion domains (for reviews see Callaghan & Tottenham 2016a, Gunnar et al. 2015, Perry et al. 2018). For example, parents both buffer (i.e., downregulate) and amplify (i.e., upregulate) children’s fear responses, depending on their own emotional output (Eisenberg et al. 1998, Gunnar et al. 2015). Such regulatory relationships become established during the very earliest stages of postnatal and, arguably, even in prenatal, life, with several studies noting the role of parents in altering fetal and infant emotional, biological, and neural responses to stressors (DiPietro 2012, Papousek & Papousek 1990). Indeed, such relationships appear to set the stage for typical brain development and may act as a species-expected stimulus (Tottenham 2012), which is a cue that can provide a scaffold for development by virtue of its ubiquitous presence across the evolution of the species. Given the important role of parents in structuring emotional maturation, it is not surprising that natural variations in caregiving are important predictors of their offspring’s mental and physical health (Callaghan et al. 2017, Champagne et al. 2003). Moreover, when such early caregiving relationships are profoundly disturbed, such as in cases of neglect, abuse, trauma, and institutional care, the consequences for affective development and mental illness can be severe (Bos et al. 2011). Such studies highlight the need to examine affective system maturation within the postnatal context of caregiving during development.
Basic research efforts to understand affective illnesses have typically focused on fear/threat responding. Fear is an emotional response or reaction to a real or imagined threat that is essential to survival, driving self-preservation reactions. In maturity, the fear system enables the individual to independently learn about the sources and predictors of safety and danger and to change his or her behaviors in accordance. When environmental threats are ambiguous, these emotional reactions can depend greatly on context as well as on an individual’s history, including trauma. This variability can generate inappropriate fear responses to benign stimuli or responses that are exaggerated; in some cases, these responses become maladaptive, leading to anxiety and other emotional disorders (Mineka & Zinbarg 2006). While much is known about the fear/threat circuit in maturity (Myers & Davis 2007), our understanding of the development of this system is limited. This in adequate understanding of fear development may help to explain why we have now reached a therapeutic impasse. Specifically, while basic research into fear/threat behavior and circuitry has been increasing, the clinical application of this knowledge lags behind. Moreover, current gold-standard treatments for psychopathology (particularly anxieties) typically involve some form of fear inhibition or extinction learning, which (although successful for many people) comes with a high rate of nonresponse (McNally 2007). To bridge that impasse, the need for a greater understanding of neurodevelopment and a wider use of animal models to uncover the molecular, cellular, and systems pathways involved in fear maturation has been suggested (Thompson & Levitt 2010).
Animal models have undeniably increased our access to the neurobiology of fear learning, leading to a detailed understanding of the mechanisms of fear learning, expression, and inhibition (Myers & Davis 2007), especially in adults. Such data have revealed great cross-species convergence in fear neurocircuitry, in its regulation by different contexts, and in the periods of developmental plasticity (Callaghan et al. 2014, Casey et al. Lee 2015). In addition, the fact that animals can be used to model causal pathways linking early experiences of adversity and dysregulated emotional outcomes—a level of analysis nearly impossible in human populations—has aided our understanding of factors that directly increase anxiety risk. However, effectively utilizing the information derived from animal systems for clinical purposes requires understanding the ways in which the findings apply to the human condition. In particular, for the field of developmental psychopathology, anchoring cross-species comparisons on developmental stages (when), as well as neural systems and behavior (what/how), is essential. The challenges inherent in identifying such developmental anchor points (e.g., What is the human equivalent of a postnatal day–17 rat? What is the rat equivalent of a 7-year-old child?) have resulted in few cross-species translations because there is no standardized approach for making such comparisons. Consequently, until a consensus on how to best bridge the species divide is reached, the perceived inconsistencies between human and rodent research are likely to lead to a decrease in the utilization of such important mechanistic animal findings.
In this review, we propose a means of making cross-species comparisons that considers fear neurobiology within its expected and ubiquitous developmental context: parental care and ultimate separation from the parent. This approach anchors the development of fear/threat circuits to maturational goals relevant to virtually all altricial mammals (particularly rodents and humans): attaching to and, subsequently, separating from the parent. By utilizing developmental goals that are universal to altricial mammals, we can leverage neuroconstructivist theories that have been supported in human developmental work (Karmiloff-Smith 2009) to create a developmental ecology framework of fear neurobiology. We argue that this developmental ecological approach provides a common language—rooted in behavior, environmental context, and neurobiology— through which fear systems and their disorder can be understood. After outlining the developmental ecology framework, we use it to structure the literature on fear/threat development in rodents and humans. In doing so, we highlight areas of consistency across species, supporting further translational work, as well as areas of discrepancy, reflecting either a lack of knowledge that can drive future research efforts or that identifies a cross-species incompatibility. By setting the lens through which cross-species findings can be viewed, we aim to motivate focused research efforts to fill in the gaps in our understanding of fear/threat neurodevelopment and, in the process, advance our understanding of mental illness etiology and treatment.
UTILITY OF THE CROSS-SPECIES APPROACH TO FEAR LEARNING AND EXPRESSION
Studies of learned fear in rodents typically employ Pavlovian (classical) conditioning, which involves learning about cues and contexts that predict danger and which appears to approximate the development of anxiety disorders (Mineka & Zinbarg 2006). Using classical conditioning, rodent and human research has implicated the amygdala in the development, expression, and storage of fear memories in adults, and the prefrontal cortex (PFC) and hippocampus (HPC) in contextually gating and inhibiting the expression of fear (i.e., extinction; LeDoux 2000, Phelps et al. 2004, Quirk & Mueller 2008). This neurobiological research illustrates the high conservation of fear learning systems across species, lending support to cross-species comparisons being made within this domain.
TRANSLATING DEVELOPMENT ACROSS SPECIES
Cross-species translation in a developmental context necessitates ontogenetic alignment of the neural circuits and behaviors involved in fear learning. There is no single correct approach to making such a developmental alignment, and several options exist (see Clancy et al. 2007a for a review of such approaches). For example, when using rules of thumb, rat postnatal life begins at approximately 24 weeks of human gestation (Andrews & Fitzgerald 1997); when using neurogenesis, rats at 5 days postconception are equivalent to a human of about 4–5 postnatal weeks (Bayer et al. 1993); or when using embryological development to determine maturational stage at birth, rats of about 10 days old approximate humans who are 2 months old (Butler & Juurlink 1987). Similar alignments (although with slightly different estimates for cortical and subcortical regions) are found using statistical models derived from 10 different species (Clancy et al. 2007b). As can be seen, there is considerable heterogeneity in the relative positioning of species across these models. Moreover, such models use a preponderance of physical and neural metrics as anchor points for neural development, heavily focus on embryonic and very early postnatal development, and largely neglect the social and emotional lives of the animals whose developmental trajectories are being aligned. While each of these approaches likely has utility within a specific field (e.g., neurogenesis or embryology), the efficacy of using such methods to align species at the level of fear circuits and behavior, and across a wide swath of development (infancy, childhood, and adolescence), is limited.
In contrast to the methods discussed above, neuroconstructivist frameworks (which have experienced much success in human developmental research fields) stage human maturation by referring to the social and emotional goals or tasks of a developing individual, for example, attaching to a caregiver (Havighurst 1948, Karmiloff-Smith 2009). By recognizing that individuals may have a unique series of goals, specific to their environment and moment in development, this approach steps away from a chronological understanding of age and, in doing so, provides a framework for understanding atypical trajectories of development. Specifically, when developmental goals are shifted due to a change in species-expected environments (e.g., disruptions in caregiving), neural and cognitive development will be altered to meet those goals. In the next section, we leverage such neuroconstructivist ideas to describe life goals that are shared across rodent and human species and that may conceivably act as anchors for the maturation of fear neurobiology.
A DEVELOPMENTAL ECOLOGY APPROACH TO FEAR NEUROBIOLOGY
The experience of fear (or the induction of threat responses) involves the independent assessment of sources of threat, often based on prior threat experiences occurring during our earliest stages of life. As such, a complete understanding of fear behavior must include a deep appreciation of the role caregivers have in building a scaffold for their child’s fear development. Indeed, as recognized many years ago by Bowlby (1969), a major reason for a child to attach to a caregiver is because the caregiver can modulate the child’s fear. Reflecting the prime importance of the parent, it has also been suggested that the parent–child dyad (rather than the child alone) is the fundamental unit of analysis early in development (Winnicott 1960). Considered in this way, parental absence (or disruptions in the caregiving relationship), rather than representing a mere lack of input, is likely to signal the presence of an imminent survival threat to the developing individual. Moreover, parental presence and the emotion-modulation capacities of the parent provide scaffolding for the development of a child’s emotional behaviors and the neural circuits that support them. Thus, the dynamics of the parent–child relationship are a relevant ecological context in which to view fear maturation. Based on those ideas, the developmental ecology framework uses the progression from parental dependence to physical independence as the scaffolding upon which the maturation of the fear system can be aligned across species (Figure 1a). Moreover, by examining deviations from the canonical trajectories of caregiving, the developmental ecology framework is useful in building an understanding of the conditions under which fear development goes awry, resulting in psychopathology.
Figure 1.
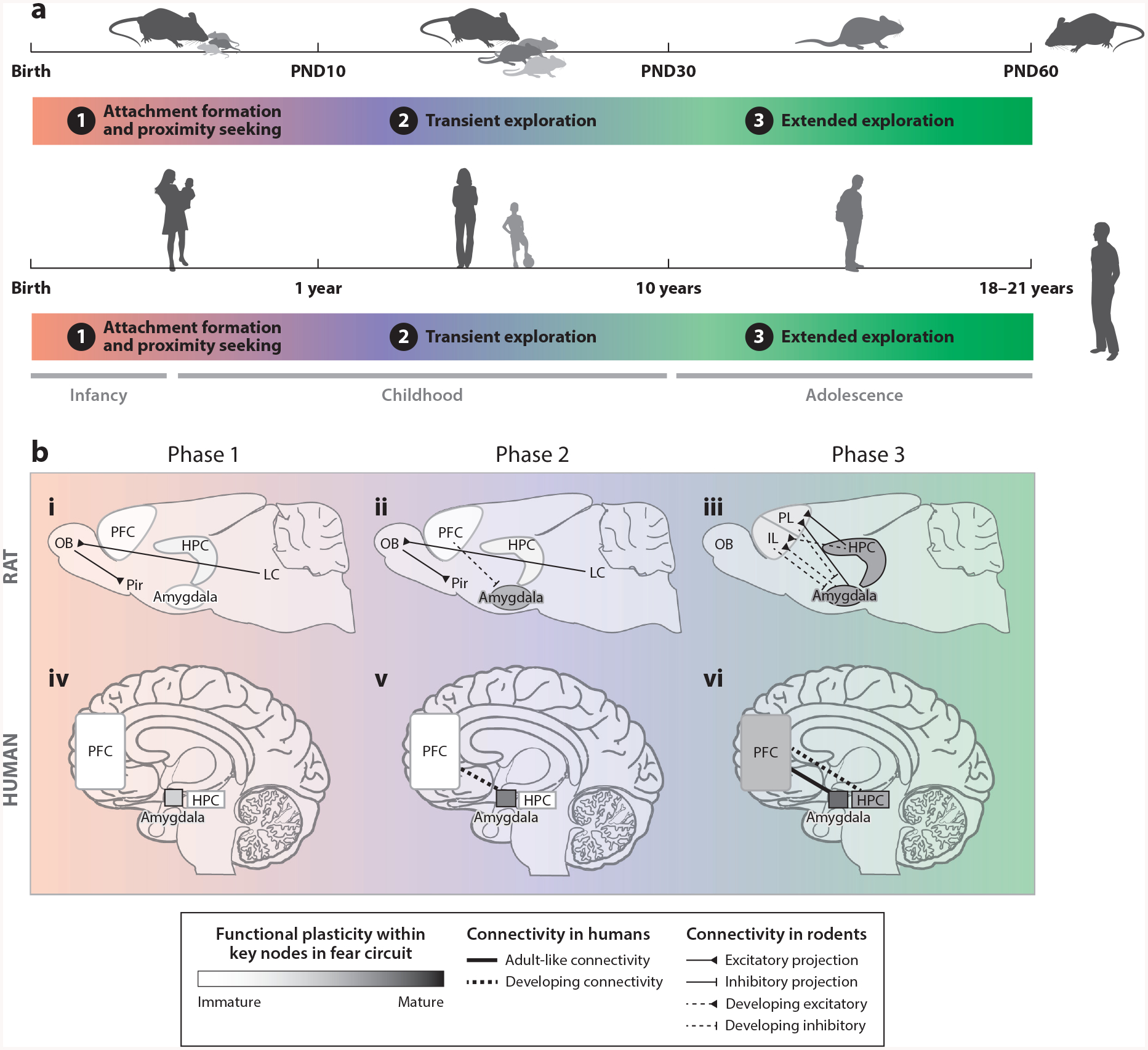
(a) Graphical depiction of cross-species alignment of fear learning and the relevant neural circuits across typical maturation within the developmental ecology framework. (b) Summary of fear learning circuitry across the three phases of typical development in early life for rodents (i–iii) and humans (iv–vi). Gray shading indicates the presumed functional maturity of different structures based on the evidence. Functional connectivity in humans is depicted by lines connecting structures, and the presumed maturity of those structures is illustrated by whether the line is solid (mature) or broken (immature). The same scheme is used for rodent connectivity, with the additional complexity of indicating whether the connection is an excitatory (triangle) or inhibitory (perpendicular) projection, a distinction afforded by the greater specificity of the evidence from rodents. Abbreviations: HPC, hippocampus; IL, infralimbic region of the PFC; LC, locus coeruleus; OB, olfactory bulb; Pir, piriform cortex; PFC, prefrontal cortex; PL, prelimbic region of the PFC; PND, postnatal day.
For all altricial mammals, reaching maturity and gaining physical independence from a caregiver progresses along a canonical pathway (albeit across different timescales), which is marked by milestones that are common across species and anchor fear development. In the remainder of this review we use the developmental ecology framework to outline three phases that lead from caregiver dependence to a state of physical independence, first describing the features that identify the phase in each species and the typical development of the fear system during the phase. We then describe how parents act as regulators of children’s emotional functioning during circumscribed developmental epochs in a manner consistent with the developmental goals of the individual. In the final section, we bring this research together to explore how a shift in developmental goals arising from the experience of caregiving adversity alters emotion behaviors and circuitry in a manner consistent with evolving ecology and dissociated from chronological age, which may place individuals at risk for long-term psychopathology. Importantly, rather than impose a new terminology on developmental fear research, we hope to push forward the hypothesis that the maturing fear network is best understood in the ecological context of the developing individual—that is, the parent—thus opening the door for more meaningful cross-species comparisons. Note that transitions between each of these phases are assumed to be gradual, particularly in the human, and chronological ages may not always map perfectly onto the literature nor are they intended to.
Phase 1: Sensitive Period for Attachment Formation and Proximity Seeking
We identify the earliest periods of postnatal life as the first developmental ecology phase, attachment formation and proximity seeking, during which there is complete reliance on the caregiver for nutrition, warmth, protection, and shelter. This is also the stage during which the infant is completely dependent on the caregiver for emotional navigation. In rodents, chronological age during this stage of development typically falls between postnatal days 0 and 9, and it is characterized by sensory immaturity (closed eyes and ears), motoric slowness and lack of coordination, and incomplete fur coverage, necessitating parental and littermate control of thermoregulation. In humans, this phase is similarly characterized by motoric immaturity, consumption of maternal milk (or liquid substitute) as the sole source of nutrition, and inability to thermoregulate, and it is chronologically thought of as infancy (Figure 1a).
During phase 1, high levels of parental caregiving are characteristic of both humans and rodents, and the routine presence of the parent promotes attachment formation (Anisfeld et al. 1990; Bowlby 1969, 1977), which increases survival potential by ensuring infants remain close to the parent, who is their primary vehicle for survival. The nature of this strong bond can be characterized by its security, which is hypothesized to reflect the degree of sensitive and responsive care by the attachment figure (Ainsworth & Bell 1970). Throughout this phase, the offspring learns that the parent is available and will respond directly to his or her needs. Given the importance of this attachment bond for survival (regardless of its security), the infant brain has evolved in such a way as to guarantee attachment formation to the parent. The activity of the amygdala seems to interfere with the formation of attachments, and, accordingly, the ability to learn amygdala-dependent fear is largely absent during phase 1 of development because the ability to form attachments to the caregiver takes precedence. It should be noted that some modulation of infant behavior toward threats exists, such as the suppression of ultrasonic vocalizations in the presence of an intruding adult male (via dopamine), but it does not involve the amygdala fear system. As the infant grows and motor refinements enable independent locomotion (e.g., crawling, toddling), a secure attachment helps to provide a reliable base from which the maturing individual can begin to explore (discussed in the section titled Phase 2: Developmental Emergence of Transient Exploration). As such, parent–child relations during phase 1 set the foundations upon which threat systems are built during subsequent developmental epochs.
As mentioned above, the developmental niche of phase 1 promotes attachment formation over fear learning. Indeed, research has shown that amygdala-dependent learning does not occur in infant rats younger than 10 days of age (Sullivan et al. 2000) (Figure 1b, subpanel i). Importantly, the inability of classical conditioning [i.e., paired presentations of a conditioned stimulus (CS) with an unconditioned stimulus (US), in this case, cue-shock] to produce fear learning is not due to pups’ inability to feel pain or detect the aversive stimulus, as noxious stimuli readily elicit pup escape responses (Barr 1995, Collier & Bolles 1980). Instead, it appears that fear behaviors are not learned or expressed because the amygdala is not engaged in contingency learning during this early phase of rodent development (Sullivan et al. 2000), likely to support pup–mother attachment. Indeed, when 8-day-old rat pups received odor–shock pairings, they learned to approach the odor predicting the shock rather than show the typical adult behavior of freezing to the odor. The lack of amygdala involvement in fear learning in postnatal day–8 pups is due to a quiescent stress response—the so-called stress hyporesponsive period (Dallman 2000)—that results in insufficient release of corticosterone (CORT) to the shock US during fear conditioning, which thus fails to activate amygdala plasticity. In the absence of amygdala-dependent fear learning, competing systems instead produce preference behaviors for the learned associations. Those competing systems are the same as those engaged when pups are learning their mother’s odor (Perry et al. 2016). Specifically, these learned odors stimulated copious norepinephrine from the locus coeruleus onto the mitral cells of the olfactory bulb, preventing these cells from habituating to the odor. This cellular cascade is conserved across many species to support attachment behaviors (Sullivan et al. 1990) (Figure 1b, subpanel i). Thus, even an odor predicting a shock can support nipple attachment and social interactions with the mother, effectively replacing the learned maternal odor. These data show that, remarkably, only during developmental phase 1, threat conditioning fails to engage the neural substrates for learning fear responses; instead it engages the mechanisms for forming an attachment to a caregiver. This early bias toward attachment learning may be adaptive for altricial rats that must learn to approach a caregiver for crucial resources, even when this care is compromised or painful (Opendak & Sullivan 2016). It should be noted that odor aversions can be induced in fetal and newborn pups using illness induced by lithium chloride or very strong shock (Camp & Rudy 1988, Haroutunian & Campbell 1979, Smotherman 1982). This infant learning emerges during the fetal period well before the development of the amygdala and depends upon the olfactory bulb and piriform cortex (Raineki et al. 2009), as well as developmental changes in the categorization of reward at the behavioral level (Camp & Rudy 1988, Haroutunian & Campbell 1979).
Due to ethical concerns surrounding experimentally inducing fear early in life, the aversive learning literature is scarce for phase 1 of human development. However, it is clear that infants readily learn attachments to their parents during the course of postnatal life, regardless of the quality of care, and it is not unusual for a child to form a strong attachment even when parents are maltreating the child. Empirically, it has been shown that threat-predictive contingencies can be learned soon after birth, with apparent extinction and relapse effects also observed at that time (Wickens & Wickens 1940); the mechanisms underlying this learning have not been characterized. Phase 1 is also a stage of rapid forgetting of fear (and appetitive) associations, a phenomenon known as infantile amnesia; this also occurs in rodents (discussed in the section titled Phase 2: Developmental Emergence of Transient Exploration). Such rapid forgetting of fear may be beneficial for infants who continue to live in the safe confines of the home during this vulnerable stage of physical development. In other words, investing in long-lasting retention of threat associations may not be necessary at this early developmental stage. Finally, although not a learned response, it is certainly true that phase 1 individuals are more indiscriminant in terms of their reactivity to adults and will exhibit low fear toward a variety of potential caregivers, including extended family and strangers (Schaffer et al. 1972). Such behaviors are reflective of the rodent literature and indicate that threat systems are maturing throughout phase 1 (Figure 1b, subpanel iv).
Although neural studies on fear development in humans during phase 1 are sparse, it has been shown that at least one structure, the amygdala (which is critical for learned fear responding), is not responsive to threat cues during this time (Graham et al. 2013) compared with what is seen in adults. This amygdala immaturity is consistent with the behavioral data on low fear and developing memory capacity during phase 1. However, the behavioral findings of infantile amnesia also implicate ongoing maturation of the HPC and PFC during phase 1, in addition to maturation of the amygdala. These gaps in our understanding should compel future studies to examine the functional activity and connectivity of the amygdala, HPC, and PFC during phase 1 of human development.
In sum, when reviewing these limited data across species, we see that phase 1 of development appears to be a period of typically high parental presence, and it is characterized by low fear in children and an immature neural circuit that supports attachment learning over fear responding (i.e., the amygdala, medial PFC, and HPC are not yet integrated into an effective, functional network). The ability of painful stimuli to engage attachment circuitry rather than fear learning circuitry in rodents suggests that processing of threat in early life is fundamentally different from that in adulthood.
Phase 2: Developmental Emergence of Transient Exploration
The attachment processes that are established during phase 1 anticipate the initial explorations of phase 2 children and ensure that return to the parent continues in these newly exploring offspring. For both humans and rodents, the transition from the goals of attachment formation and proximity seeking to transient exploration, producing brief separations from the attachment figure, occurs with the onset of motoric independence. This independence can be seen in young rodents making small excursions out of the nest and into the immediate surrounds (Bolles & Woods 1964) and in humans using skilled crawling and walking (Adolph & Berger 2006). A motoric aptitude paired with continuing reliance on the parent for food, shelter, and emotional support affords the young an opportunity to explore very short distances from the parent before returning for the satisfaction of their remaining developmental needs. Thus, this phase represents an initial transition from the parent navigating the emotional world of the offspring toward a time of increasingly independent navigation. As phase 2 progresses, the distance from the parent progressively increases and is phasic in nature as children explore new contexts and social networks that exist beyond the confines of the home environment (e.g., formal day-care settings, extended family and friends) but regularly return to the home. In rodents, phase 2 begins at approximately postnatal day 10 and ends sometime between postnatal days 21 and 30, although considerable changes in the role of the caregiver emerge from postnatal days 16 to 30. This age range is often still called infancy, although terms such as preweanling or juvenile have been used. In humans, developmental phase 2 is typically called childhood, and it lasts from approximately age 3 years to 10 years (Figure 1a).
Fear learning without the parent.
The phasic nature of parent–child interactions during phase 2, characterized by bouts of parental presence then absence, coincides with a fear system that is differentially responsive in parental and nonparental contexts. We first describe fear learning that occurs during phase 2 without parents present.
At rat postnatal day 10, in the absence of the mother, pups exhibit rising endogenous levels of CORT, resulting in the functional emergence of the amygdala in fear learning (Moriceau & Sullivan 2006, Sullivan et al. 2000). Pups’ increase in stress hormone at this age permits the amygdala to show learning-induced plasticity, allowing odor–pain pairings to activate the fear learning circuit and avoid stimuli that signal threat and danger (Figure 1b, subpanel ii). In this way, CORT acts as a switch that can enable amygdala functional plasticity to support the acquisition of cues associated with threat.
As pups continue to grow and motoric aptitude increases, they will consistently learn fear associations to cues, and while pups this age are capable of freezing, the full complement of the adult like behavioral fear response is only partially available to them (Hunt et al. 1994), and retention of threat associations is shorter compared with adults (Campbell & Spear 1972). Phase 2 pups do not show hippocampal-dependent context learning (Raineki et al. 2010a), even though there are reports of retained context learning that remains for a minute or two (Robinson-Drummer & Stanton 2015, Rudy & Morledge 1994). This indicates a notable dissociation between cued and contextual fear during phase 2, which is likely due to immature neural circuitry (Figure 1b, subpanels i,ii), and is in sharp contrast to the important role of the HPC in adult fear learning. Crucially, other brain regions shown to be critical in adult fear learning are not engaged by fear conditioning during phase 2. For example, rats younger than postnatal day 21 can express fear associations independently of activity within the prelimbic (PL) region of the PFC (Li et al. 2012), but PL activity is required at postnatal day 21, which might help in the longer-lasting retention of cued associations at this age.
In humans, although fear development during phase 2 is drastically understudied, research suggests that such learning occurs and involves the amygdala. For example, in one study of 18-month-old children, it was shown that they could learn threat associations by observing an aversive social interaction between two adults in reference to a toy (i.e., the emotional eavesdropping procedure; Repacholi & Meltzoff 2007). Gao et al. (2010) demonstrated that children as young as 3 years of age exhibited conditioned responses (i.e., skin conductance) following pairing of an auditory CS with a louder aversive auditory US. However, conditioned responding increased from age 3 to 8 years, suggesting that the underlying fear learning process matures throughout this period. From ages 6 to 16 years, avoidance learning to threat cues is reliably seen, and in some studies it is associated with amygdala activation that occurs independently of age (Block et al. 1970, Glenn et al. 2012, Jovanovic et al. 2014, Schlund et al. 2010, Silvers et al. 2016a) (Figure 1b, subpanel v).
Phase 2 is also associated with changes in fear behaviors that indicate ongoing hippocampal maturation. Specifically, increasing age was associated with better discrimination between two fear-predicting cues (i.e., a form of learning that typically relies on the HPC CA3 region and is important in context learning; De Shetler & Rissman 2017), but there were no overall differences in fear conditioning in children aged 5–10 years (Michalska et al. 2016). These behaviors are consistent with a slowly developing hippocampal learning system that leads to a dissociation between cued and context learning, consistent with what is seen in the rat. Indeed, during this stage of development, the amygdala is relatively mature (Pugh et al. 1997, Thompson & Levitt 2010, Upton & Sullivan 2010), but large developmental changes are still occurring at the level of the HPC and PFC (Giedd 2004, Uematsu et al. 2012) (Figure 1b, subpanel v). Moreover, the functional connections between HPC, PFC, and amygdala continue to develop throughout phase 2 (Gabard-Durnam et al. 2014, 2016; Gee et al. 2013a; Perlman & Pelphrey 2011; Silvers et al. 2016a,b; Swartz et al. 2014). Together with the data from rodent studies, these human studies suggest that in early life, fear circuitry is heavily bottom up (i.e., amygdala dependent), with the network increasing in complexity by integrating PFC and HPC connections across the course of phase 2.
Stepping away from fear expression and toward fear inhibition, it is also clear that this behavior changes dramatically throughout phase 2, with all of the data coming from rodents. Specifically, while adult rats exhibit several forms of contextually mediated relapse following extinction training (i.e., renewal, reinstatement, and spontaneous recovery), rats younger than postnatal day 21 do not: Instead, they exhibit relapse-resistant extinction (Kim et al. 2009, Yap & Richardson 2007). Again, the neural bases for these behaviors have been shown to change across development; the infralimbic region (IL) of the PFC is required for fear extinction only after postnatal day 21, although the amygdala appears to be similarly involved in fear extinction across phase 2 (Kim et al. 2009). This suggests a change in the connectivity between the amygdala and IL that emerges after postnatal day 21 to support adult-like, relapse-prone extinction learning. Indeed, recent studies have shown that structural connections between the amygdala and medial PFC begin to emerge only at postnatal day 15 in the mouse, but they strengthen significantly after postnatal day 21 (Arruda-Carvalho et al. 2017). Mature expressions of relapse behaviors rely on the integration of contextual information provided by the HPC, allowing memory to be expressed in a context dependent manner (Wilson et al. 1995). Hence, the absence of these behaviors in rats younger than postnatal day 21 suggests that the role of the HPC during extinction also changes dramatically during phase 2.
Parental modulation of fear neurobiology.
In contrast to the stress hyporesponsive period seen in phase 1 rats, during phase 2, baseline suppression of stress hormone levels is released, and pups exhibit an increase in CORT when confronted with the shock used in fear conditioning and innate threat cues (Hennessy et al. 2006, Kikusui et al. 2006). It is at this stage of development, when baseline CORT increases, that fear behaviors and ensuing amygdala plasticity can be blocked by maternal presence (Moriceau & Sullivan 2006, Stanton et al. 1987, Suchecki et al. 1993) (Table 1), which permits CORT to enable amygdala plasticity and support fear learning in pups (Moriceau et al. 2006). For example, at postnatal day 12, pups will learn to avoid odors that are paired with shock, but will still approach odors that were paired with shock in the presence of the mother (Moriceau et al. 2006). That is, the presence of the mother temporarily reverts the neurobiology underlying fear learning to the phase 1 stage. This maternal suppression of fear learning disappears at postnatal day 16, and pups can learn to avoid an odor paired with shock even if the mother is present during conditioning (Al Aïn et al. 2017, Barr et al. 2009, Sullivan & Holman 2010, Upton & Sullivan 2010), despite the mother’s continued ability to reduce hypothalamic–pituitary–adrenal (HPA) axis activity after postnatal day 16 (Stanton & Levine 1990, Suchecki et al. 1993, Upton & Sullivan 2010). Indeed, maternal presence can no longer attenuate amygdala function even if buffering is supplemented with a CORT blocker to produce almost full blockade of CORT release (Landers & Sullivan 2012, Perry & Sullivan 2014, Sullivan & Holman 2010, Upton & Sullivan 2010), suggesting that parental modulation of the amygdala is limited to the early part of phase 2 (see Table 1 for a summary). This phase may even represent two shorter substages in the rodent during which maternal control of the stress system switches the fear system on and off, while maternal presence has a more modulatory role in older phase 2 pups.
Table 1.
Summary of changes in fear circuitry and behavior as a function of parental presence and early caregiving adversity
| Role of parental presence | Early caregiving adversity | |||
|---|---|---|---|---|
| Rodent | Human | Rodent | Human | |
| 1 | No amygdala buffering by parents due to SHRP (Dallman 2000) | Parent can buffer HPA axis (Feldman et al. 2010) | Accelerated involvement of amygdala in learning (Raineki et al. 2010b) | Increased functional connectivity between amygdala and PFC (Graham et al. 2015, 2016) |
| 2 | Parental buffering of amygdala before postnatal day 15 (Moriceau & Sullivan 2006) and PFC (Al Aïn et al. 2017, Hennessy et al. 2006, 2015) | Parent can buffer HPA axis (Ahnert et al. 2004), decrease amygdala reactivity, and induce negative connectivity between amygdala and PFC (Gee et al. 2014) | Accelerated maturation of amygdala-, PFC- and HPC-dependent behaviors (Callaghan & Richardson 2011, 2012b) | Accelerated maturation of amygdala-PFC functional connectivity (Gee et al. 2013a); increased HPC involvement in learning (Lambert et al. 2017, McLaughlin et al. 2016, Silvers et al. 2016b) |
| 3 | No data | No parental buffering of amygdala (Gee et al. 2014) | Accelerated maturation of fear and extinction behaviors (Bath et al. 2016, Callaghan & Richardson 2012a); accelerated maturation of HPC (Bath et al. 2016); decreased activity of the amygdala; decreased amygdala-PFC plasticity (Koppensteiner et al. 2014, Marco et al. 2012) | No data |
Abbreviations: HPA, hypothalamic–pituitary–adrenal; HPC, hippocampus; PFC, prefrontal cortex; SHRP, stress hyporesponsive period.
For humans, there is no clearly defined period of parent-independent stress hyporesponsivity as there is in the rodent. Rather, the buffering of fear behaviors and stress responding may occur during the latter part of phase 1 and into phase 2. For example, several stressors encountered when parents are absent (such as novel events and approach by a stranger) do produce elevations in salivary cortisol levels in children (Nachmias et al. 1996, Spangler & Schieche 1998). However, parental stimuli, such as touch, can reduce the child’s HPA axis response to psychosocial stressors, as seen in the still-face procedure during phase 1 (Feldman et al. 2010) or in child care during phase 2 (Ahnert et al. 2004). Parental buffering is also seen in school-age girls during the Trier Social Stress Test (TSST; Seltzer et al. 2012). Those who had physical or auditory (i.e., over the phone) contact with their mother after the test showed stronger cortisol dampening than those girls whose mothers were in contact only via instant messaging (i.e., no physical or auditory contact). Similar cortisol buffering was seen in children who were able to prepare for a modified version of the TSST in the presence of their parent relative to those preparing in the presence of a stranger (Hostinar et al. 2015a). The neurobiological basis for such parental buffering during phase 2 appears to be the amygdala, as children exhibit less amygdala reactivity when viewing pictures of their parent than when viewing pictures of a stranger (Gee et al. 2014). In the study by Gee et al. (2014), parental buffering of the amygdala in childhood was associated with greater affective behavioral regulation when the parents remained nearby during an emotional Go/No-Go task, providing strong support for the amygdala as the neurobiological basis for parental buffering in humans (Table 1).
Phase 3: Extended Exploration
The final stage in the developmental ecology framework for understanding the maturation of threat systems is phase 3, or extended exploration. In this phase, the focus on parents as the supportive unit begins to wane as individuals gain increasing physical independence and sexual maturity. In rodents, phase 3 occurs after weaning from the mother, at a time when there is a surge in pubertal maturation (postnatal days 30–45; Spear 2000) (Figure 1a). Typically, this phase ends in young adulthood for the rodent (i.e., approximately postnatal day 60), when sexual maturity is firmly established. Arguably, this phase may even represent two shorter substages in the rodent: one that begins at weaning and ends before puberty and another that is tightly linked to pubertal development (although we discuss both together here).
In humans, the beginning of this stage of development roughly coincides with the onset of puberty and continues until full sexual maturation (much like in the rodent) and the attainment of adult roles and responsibilities (Spear 2000), corresponding to the ages of approximately 11 to 17 years. Importantly, while rodents completely break parental contact during phase 3, humans (in most societies) may remain living with their parents or engaging in high levels of contact with their parents well beyond the time when they are emotionally and financially independent and sexually mature.
Recent evidence from rodents indicates that fear learning and expression change dramatically throughout phase 3 of development. Rodents in this phase can be conditioned to freeze to a CS (see, e.g., Pattwell et al. 2012), but exhibit diminished extinction learning relative to younger and older individuals (Johnson & Casey 2015a, McCallum et al. 2010, Pattwell et al. 2012), as well as impaired retention of extinction memories (Baker et al. 2016, Kim et al. 2011, McCallum et al. 2010). Interestingly, these effects can be mitigated by additional extinction trials or by pharmacological intervention (e.g., administration of d-cycloserine; McCallum et al. 2010) (Figure 1b, subpanel iii), suggesting that the deficits in extinction may reflect quantitative, rather than qualitative, differences in learning. Moreover, unlike cued fear learning, contextual conditioning can result in unusual behaviors during phase 3 (Pattwell et al. 2011) whereby learned fear behaviors to contexts can be temporarily suppressed. In all, the rodent data are consistent with the idea that phase 3 is a period during which fear can be learned to cues, is suppressed to contexts, and may be difficult to inhibit once acquired.
In terms of phase 3 neural circuitry, protracted development of the prefrontal regions (PL andIL), which continues well into the phase (Casey et al. 2005, Giedd et al. 1999, Gogtay et al. 2004) (Figure 1b, subpanel iii), may limit the capacity for feedback mechanisms that mediate fear responding by regulating activity in the amygdala and HPC. In support of this hypothesis, amygdala potentiation is observed following cued fear conditioning during phase 3 in mice (Pattwell et al. 2011), but amygdala innervation of the PFC progresses slowly (see, e.g., Cunningham et al. 2002). Interestingly, connectivity of the amygdala and the PL, a prefrontal region known to be important for fear expression, is enhanced (above the adult baseline) specifically during phase 3, coinciding with a surge in dendritic spine formation in the PL (Pattwell et al. 2016). This phase-specific amygdala–PL connectivity pattern may help to explain the impaired fear extinction seen during this period. In terms of the neural mechanisms behind the temporary suppression of contextual memories that is also seen during phase 3, likely candidates are the transient decreases in amygdala signaling as well as a lack of retrieval-associated signaling in the HPC (Pattwell et al. 2011).
Like rodents, humans in phase 3 exhibit diminished extinction learning relative to younger and older individuals (Johnson & Casey 2015a). In terms of the neural circuitry behind this behavioral difference, amygdala activation is probably not the locus of the effects because its mature role of discerning threat from safety cues is in place by phase 3 (Monk et al. 2003), as it is in rodents. In contrast, there is good evidence that protracted prefrontal development leads to less modulation of amygdala reactivity at this stage (Casey et al. 2005, Giedd et al. 1999, Gogtay et al. 2004) (Figure 1b, subpanel vi), much like in the rodent. Interestingly, when methods of attenuating conditioned fear rely less on the PFC (e.g., reconsolidation), longer-term fear reduction is achieved than when prefrontal-dependent (e.g., extinction) methods are used (Johnson & Casey 2015b). Indeed, increased activity in the PFC elicited by the presentation of aversive faces (Yurgelun-Todd & Killgore 2006) and a regulatory inverse pattern of connectivity between the PFC and amygdala (Gee et al. 2013b) are positively correlated with age (Figure 1b, subpanel vi), indicating increased prefrontal involvement in fear regulation as individuals progress through phase 3.
DISRUPTION OF THE PARENT–CHILD DYAD
As shown above, a developmental ecology framework aids in the understanding of typical fear system development, its alignment across species, and the role of parents in providing scaffolding for such development. As we will see in this section, by examining deviations from canonical trajectories of caregiving, a developmental ecology framework is useful in building an understanding of the conditions under which fear development goes awry, resulting in either poor behavioral regulation or psychopathology. Such deviations in caregiving may include experiences as diverse as physical and emotional abuse, neglect (in which the parent is either physically or emotionally absent, such as in parental depression), parental death or incarceration, and institutionalization of the child. While each of these experiences varies widely in its causes and in psychological meaning to the child, they share a commonality of signaling to the child the presence of potential danger by virtue of the absence of invested and committed parental care. In human populations, caregiving adversities such as those described are observed most frequently (although not exclusively) during phases 1 and 2. Regardless of the phase of development during which adversity begins, such deviations in caregiving oppose the developmental goals of the growing child of being able to depend upon a healthy attachment to the parent and to use this attachment figure as a safe and secure base to transiently explore the environment while knowing there is a protector nearby if a threat is detected. In cases of abuse, neglect, and early separation from the parent, these experiences place developing children in goal conflict, a situation in which their developmental needs diverge from their chronological age to reflect those of a more mature organism, that is, in extended exploration, looking toward nonparents and other social groups to meet their emotional and safety needs. Interestingly, the evidence suggests that for those with disrupted early caregiver relations, the fear system also diverges from chronological age to match the developmental goals of the individual (Table 1).
For rats, disruptions in maternal care occurring during phase 1 that are outside the range of species-typical variation result in a premature shift in threat learning biases away from forming caregiver attachments and toward learning fear (Moriceau et al. 2009), thus effectively ending phase 1 prematurely. In rodents, such early caregiving adversities (ECAs) are studied using several procedures, including maternal separation (i.e., the daily removal of pups from the mother for periods of approximately 1–3 hours) and low bedding (i.e., a low-resources procedure that involves restricting the availability of nest building materials to model scarcity and induces abusive behavior in dams, including rough handling, dragging, and stepping on pups) (Roth & Sullivan 2005, Walker et al. 2017) (see Figure 2 for a graphical summary of several rodent paradigms of ECA). Although maternal separation and low bedding are very different models of caregiving adversity, both result in premature elevations in CORT levels in phase-1-aged pups that are typically stress hyporesponsive (Raineki et al. 2010b, van Oers et al. 1998).
Figure 2.
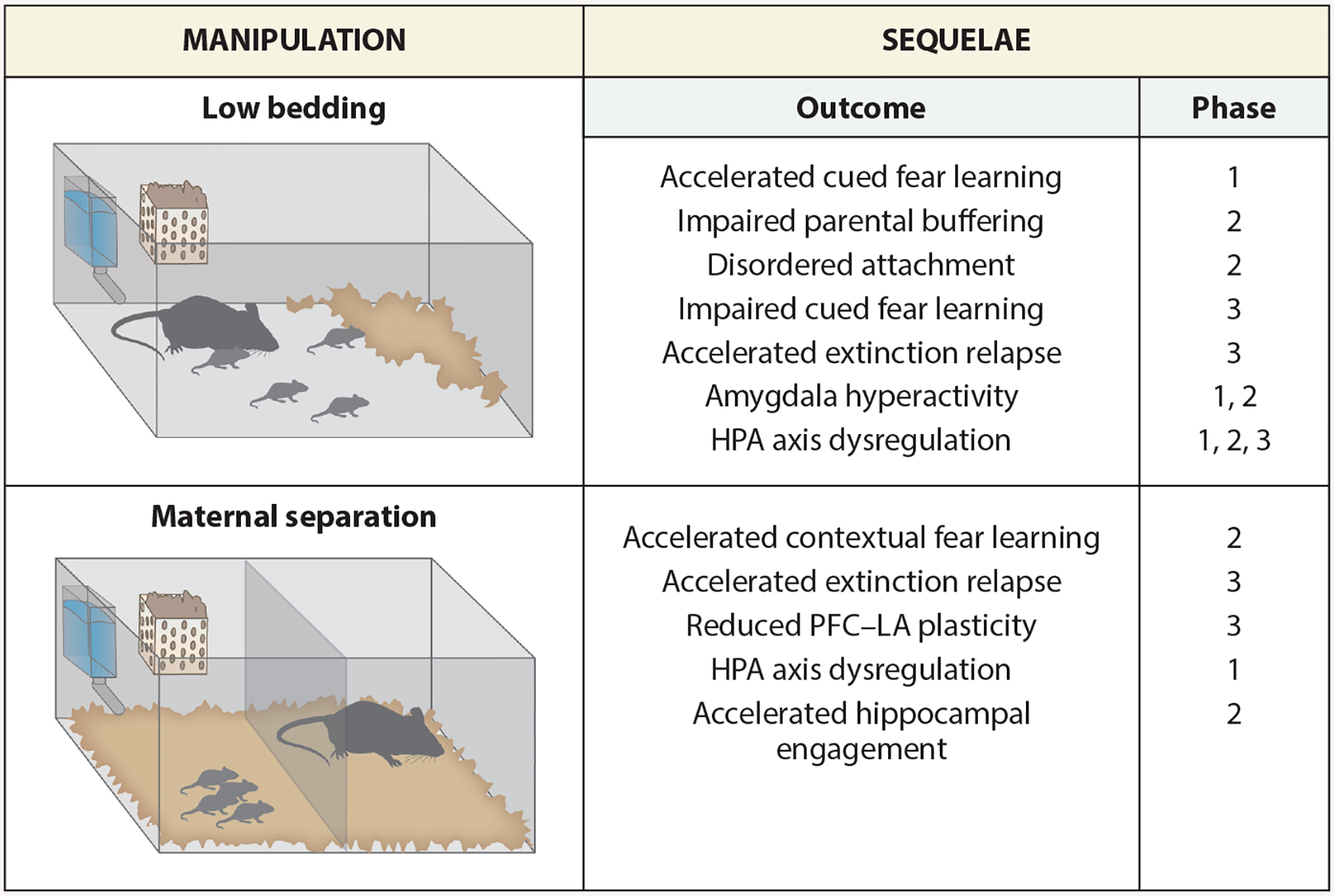
Select rodent models of early caregiving adversities discussed in this review. The fear behavior and circuitry that have been associated with these early caregiving adversities are listed under the heading Outcome, and the developmental ecology phase at which those outcomes have been reported is listed under Phase. Abbreviations: HPA, hypothalamic–pituitary–adrenal; LA, lateral amygdala; PFC, prefrontal cortex.
In humans, caregiving adversity in phase 1 affects the early development of neural circuits important for fear. For example, adversities that fall within the more normative range during infancy, such as interparental conflict, change connectivity patterns between the amygdala and posterior cingulate cortex (Graham et al. 2015). Similarly, in another study, maternal prenatal depressive symptomatology was associated with altered amygdala connectivity patterns in resting 6-montholds (Qiu et al. 2015). Importantly, intrinsic function of the amygdala in newborns predicts fear and cognitive development at a 6-month follow-up (Graham et al. 2016), suggesting that parent based adversities during phase 1 alter the foundational circuitry on which fear behaviors are built during later phases of development.
Phase 1 caregiving adversities are also associated with ongoing consequences for neural circuitry and fear behavior during phase 2. In rats, the effects of earlier disruptions in parental care often become evident when stress hormones are elevated. For example, rats that were exposed to early adversity during phase 1 (e.g., odor–shock pairings or more naturalistic maternal abuse) showed typical neurobehavioral responses to the mother. However, maladaptive programing could be uncovered by administering CORT injections during phase 2: Pups exhibited altered social behavior toward the mother and amygdala hyperactivity (Raineki et al. 2010b, 2012). In addition, rats exposed to the stressful procedure of fear conditioning during phase 2 (which increases pups’ stress hormone levels) showed accelerated development of fear behaviors if they had a history of maternal separation during phase 1. Specifically, a daily 3-hour maternal separation in phase 1 rats (postnatal days 2–14) has been shown to produce better context learning at postnatal day 17 than is seen in nonstressed same-aged peers (Callaghan & Richardson 2011) and also better retention of cued and context threat associations (i.e., for weeks as opposed to days; Callaghan & Richardson 2012b). In addition, the maternal-separation postnatal day–17 rats exhibit more relapse of threat responding after extinction than their nonstressed peers (Callaghan & Richardson 2011), a behavior that is characteristic of adulthood. In other words, fear behaviors of rats that are chronologically aligned in age at postnatal day 17 diverge as a function of caregiving adversity, with postnatal day–17 rats who have endured maternal separation looking more mature, that is, similar to phase-3 animals, whereas nonstressed postnatal day–17 rats behave in a manner consistent with the developmental goals of phase 2.
Emerging evidence in humans also suggests that ECA exposure may be associated with earlier maturation of fear-related neural circuitry during phase 2. In two separate studies, children in phase 2 with histories of ECA showed more adult-like amygdala–PFC connectivity during phase 2 relative to a same-aged comparison group (Gee et al. 2013a, Thijssen et al. 2017). In the study by Gee et al. (2013a), this amygdala–PFC phenotype was related to concurrent cortisol levels during a magnetic resonance imaging session, suggesting that similar to findings in the rodent literature, HPA axis function is involved in ECA-related changes in amygdala reactivity during phase 2. In humans, amygdala structure during phase 2 is also altered following ECA. Specifically, amygdala volume was larger in children that had been exposed to maternal depression (Lupien et al. 2011) or to prior institutionalization (Tottenham et al. 2010). It is hypothesized that the altered amygdala development observed in ECA-exposed children likely reflects the chronic effects of stress that reduced earlier parental buffering opportunities, which would contribute to altered circuit wiring (Callaghan & Tottenham 2016a). In support of this hypothesis, children in phase 2 who were exposed to prior institutional care were shown to exhibit less parental buffering of the HPA axis in response to a laboratory stressor than noninstitutionalized youths (Hostinar et al. 2015b). Whether previously institutionalized children also experience less parental buffering of the amygdala is under investigation.
In terms of human contextual learning and the HPC during phase 2, some changes have also been associated with exposure to ECAs. Specifically, youths exposed to early caregiver violence showed poor context encoding of scenes paired with angry faces, suggesting that heightened neural attention to threat may interfere with memory encoding during phase 2 (Lambert et al. 2017). Importantly, this deficit in performance coincided with increased hippocampal engagement, which is crucial for context encoding, and stronger hippocampal connectivity with the ventrolateral PFC, which might suggest accelerated development. However, those effects were consistent across all ages, which makes the examination of accelerated development difficult to ascertain. In phase 2, children with histories of maltreatment also show differences in hippocampal volume that correspond to impairments in aversive learning but that are not clearly associated with age (McLaughlin et al. 2016). In a separate study, although stressed youths did not show behavioral impairments in aversive learning, they exhibited greater HPC reactivity and connectivity with prefrontal regions relative to the comparison group (Silvers et al. 2016b). As with the study by McLaughlin et al. (2016), this finding was consistent across all ages, precluding an analysis of accelerated development in the stressed youth. Nevertheless, greater involvement of the HPC in threat learning in ECA-exposed youths is consistent with the idea that HPC-mediated behaviors (such as context learning) might show accelerated development following stress. One study that seems to support this idea has shown that autobiographical memories, a hippocampal-dependent phenomenon, emerge earlier in life following parental divorce (Artioli & Reese 2014). However, that study relied on retrospective reports of autobiographical memories, which are not always reliable, and was not examining the fear system per se. Nonetheless, the data clearly support further investigation into HPC development following ECA, especially as it relates to fear learning.
Accelerated development following phase 1 maternal separation is also seen in rats that are chronologically in phase 3 (i.e., postnatal day 30; Figure 1a). For example, maternal separation stress during phase 1 resulted in a leftward (i.e., accelerated) shift in the developmental trajectory of fear extinction behavior whereby impaired fear extinction typically seen post–pubertal onset was observed earlier within phase 3 (Callaghan & Richardson 2012a). A similar shift has been observed for the window of contextual fear suppression in mice exposed to phase 1 stress due to the low bedding procedure; although normally observed only postpuberty, contextual fear was instead suppressed earlier in phase 3 (Bath et al. 2016). This shift was associated with an accelerated profile of hippocampal maturation, including an earlier arrival and peak in interneuron development in the HPC, as well as earlier expression of markers of myelination and synaptic maturity (Bath et al. 2016). In line with these findings, maternal separation during phase 1 has also been shown to induce alterations to hippocampal functioning throughout phase 3 of fear development (Marco et al. 2012) and, additionally, has been linked to reduced neural plasticity in the PFC and lateral amygdala at the same time (Danielewicz & Hess 2014, Marco et al. 2012), disrupting communication in both the cortico–amygdala and thalamo–amygdala pathways. Taken together, this rodent literature suggests that a variety of early adversities are associated with the accelerated development evinced during phase 3 (Table 1).
Unlike for rodents, the human literature on phase 3 fear outcomes that are associated with ECAs is scarce, perhaps because maltreatment by parents is most likely to occur during phases 1 and 2 (CDC 2018). Importantly, whether ECA-induced changes in the fear system are seen during phase 3 of development may depend on the context in which individuals are examined. Specifically, one recent study in adults showed that non-fear-related working memory, which is typically impaired in ECA-exposed individuals, was actually enhanced in those individuals under contexts that were higher in uncertainty (Young et al. 2018). In other words, ECA may accelerate the development of fear neurobiology during earlier stages of life to prepare individuals to function in particular affective contexts in adulthood. Such ideas have been discussed extensively in the literature (see, e.g., Callaghan et al. 2014, Callaghan & Tottenham 2016a,b), but remain to be tested empirically for fear learning across phases 1 to 3.
ALTERED DEVELOPMENTAL ECOLOGIES AND PATHOLOGICAL BEHAVIORS
Considering that caregiving adversities are associated with altered trajectories of fear system development, it is not surprising that such adversities are also predictive of psychopathology in both the developing, as well as the adult, individual. For example, rodents that are exposed to the low bedding stressor exhibit altered social behavior in phase 2, which is followed by an increase in depressive behavior (as measured by the forced swim test) during phase 3 (Raineki et al. 2012). Moreover, maternal separation is commonly used as a model of depressive behavior (Matthews & Robbins 2003), anxiety, and substance abuse (Huot et al. 2001) in adulthood. Similarly, in humans, exposure to caregiving adversity is a leading cause of mental health problems (Green et al. 2010), being associated with a constellation of mental health issues that can emerge early in life (i.e., in phases 1 and 2) or also later in development (during phase 3 or adulthood), including internalizing and externalizing, as well as substance abuse disorders (Green et al. 2010, McLaughlin et al. 2012). Hence, many studies in both rodents and humans that have examined the consequences of ECAs on mental health have converged on the finding that such adversities are associated with a variety of mental illnesses.
Considering the scope of psychopathologies that are associated with ECAs, one interpretation is that ECA exposure may promote the emergence of a syndrome-like constellation of symptoms rather than discrete disorders that fit neatly within diagnostic categories (e.g., major depressive disorder or generalized anxiety disorder). Indeed, the fact that a range of different caregiving adversities, which are present across species, have converging effects on fear system development (namely, accelerated maturation) suggests that disrupted limbic neurobiology may be the core mechanism underlying this syndromic state. For this reason, paying attention to commonalities in limbic development across ECA-exposed individuals may be the most effective means of building successful treatments for the psychopathological sequelae of ECA. Moreover, examining how the constellation of symptoms changes across development might provide additional information on when and how best to intervene.
CROSS-SPECIES CONSISTENCIES EMERGING FROM A DEVELOPMENTAL ECOLOGY APPROACH TO FEAR
In this review, we have described how the environment constructs fear in parallel with major developmental milestones in rodent and human populations. In particular, we have used the relationship with the caregiver and how it changes throughout development as the critical factors upon which species maturation can be aligned, enabling comparisons of fear learning behavior and circuitry across a large time frame that includes chronological phases typically referred to as infancy, childhood, and adolescence. Using this approach, we have shown that the development of fear behaviors and the neural circuitry that supports them appear to follow the maturational goals of the organism as they relate to the parent–child relationship. In phase 1 of development, during which the goal is to attach to and seek proximity to the parent, we see tonic inhibition of threat behavior (rodent) or buffered fear responses (human) mediated through immature neural structures that would enable the individual to engage in attachment behaviors, regardless of the quality of the care received. At the next stage of development—phase 2—increased motoric independence and brief excursions from the home environment give rise to the onset of conditioned fear behaviors in the rodent and a potent form of parental buffering of fear behaviors and circuitry in both the rodent and human. These behaviors are also reflective of the goals of this developmental stage: to engage in transient exploration using the parent as a secure base that signals safety and, thus, low fear. This phasic nature of fear behaviors, guided by parents, has been hypothesized to help teach the neural circuit to engage in mature regulatory interactions during phase 2 (Callaghan & Tottenham 2016a). Finally, in phase 3 of development, when the goal is to explore and eventually break from parental dependence, both rodents and humans exhibit increased sensitivity to cues predictive of danger, a decreased inhibition of fear responses, and a generalization of fear responses across similar cues. Such sensitivity would presumably enhance fitness as individuals transition out of the safety of the home and begin to navigate the world independently. Interestingly, in rodents a temporary suppression of fear responding to contexts has been observed during this time, which could feasibly facilitate the process of finding a new home environment while remaining vigilant to danger cues.
CONCLUSIONS
Rather than impose a new terminology on developmental epochs or stages in fear research, what we hope to have done in this article is to push forward the hypothesis that the fear network is best understood in the ecological context of the developing individual, which opens the door to more meaningful cross-species comparisons. Considering the importance of parenting for child development, and the strong associations that exist between caregiving adversities and mental health across the life span, it is critical to understand the development of the fear system across species to improve mental illness prevention and treatment throughout the life span.
SUMMARY POINTS.
Studying the development of fear behavior and neurocircuitry can aid our understanding of mental illnesses.
Human and nonhuman animal studies have contributed to the literature on fear/threat development, but it is difficult to translate findings across species due to differing metrics for aligning maturation across species.
The developmental ecology framework uses developmental goals relating to caregiving relationships that are ubiquitous across species as the standard upon which to align development across species.
Within a developmental ecology framework, it becomes clear that the developing neurobiology that supports fear-related behaviors matures slowly postnatally and in concert with the caregiving environment.
The effect of caregiver disruptions on fear behaviors appears to be a good model for the development of psychopathologies and is compatible across species, encouraging future translation in this area.
FUTURE ISSUES.
Human studies of fear behaviors (particularly those that use innovative and noninvasive methods) occurring in phase 1 of development and the early part of phase 2 are needed.
Understanding whether the buffering of stress reactivity and fear responding transfers to peers during adolescence in both rodents and humans is an understudied, but important, area.
Longitudinal analyses of fear behaviors are lacking in both the human and rodent literatures.
Cross-cultural studies are needed to understand the degree of universality of these processes.
ACKNOWLEDGMENTS
This review was born out of a cross-species research cooperative spanning three different basic emotional development labs both in rodents—Regina Sullivan’s group at NYU, Francis Lee’s group at Weill Cornell—and in humans—Nim Tottenham’s group at Columbia—who came together with the aim of synthesizing the human and animal research on fear and threat development to identify areas of convergence and divergence and to highlight limits in our understanding, thus paving the way for future studies. The development and writing of this paper was supported by grants to N.T. from the US National Institute of Mental Health (R01MH091864), the Dana Foundation, and the National Science Foundation Conference Grant (BCS-1439258) (co-investigator); to B.C. from the Australian National Health and Medical Research Council (1091571), the US National Institutes of Mental Health (NIMH) (K99MH113821-01), and the US Brain Behavior Research Foundation (24739); to H.M. from the National Center for Advancing Translational Science of the US National Institutes of Health (NIH) (1TL1TR0002386-01); to A.L. from the Medical Scientist Training Program of the NIH (GM07739); to F.S.L. from the New York-Presbyterian Youth Anxiety Center; to M.O. from the NIH (F32MH112232 and T32MH01952); to R.M.S. from the NIH (DC009910, MH091451, HD083217, R37HD083217); to C.H. from the National Science Foundation (DGE1644869); and to M.V.T from the National Science Foundation (DGE1644869), NIMH National Research Service Award (F31MH115686-01A1), and the American Psychological Foundation Elizabeth Munsterberg Koppitz Child Psychology Fellowship.
Glossary
- Species-expected stimulus
a stimulus ubiquitous across the evolution of a species that has come to guide elements of development
- Therapeutic impasse
a barrier to the success of psychotherapies
- Extinction
a decrement in responding to a conditioned stimulus after its repeated presentation in the absence of the unconditioned stimulus
- Altricial mammal
species that are born requiring parental care for survival (e.g., most mammals)
- Neuroconstructivist
theories of how the brain shapes itself into a specialized organ via interactions with the environment
- Classical conditioning
a procedure whereby two stimuli are paired until a response elicited by one is eventually elicited by the other alone
- Attachment
the process of forming an emotional bond between caregiver and child that comes to guide maturation
- Conditioned stimulus (CS)
an initially neutral stimulus that comes to elicit a response through its association with a second stimulus
- Unconditioned stimulus (US)
a stimulus that can directly elicit a response without prior pairing with an outcome
- Stress hyporesponsive period
a time of suppressed stress axis functioning occurring in early development
- Infantile amnesia
the more rapid rate of forgetting observed in the younger members of a species compared with the older members
- Parental buffering
a decrease in stress reactivity and/or emotionality as a result of parental presence
- Institutionalization
internment of children in orphanages, group homes, hospitals, or other (usually government) facilities, characterized by high ratios of children to caregivers
- Early caregiving adversity (ECA)
a disruption in a child’s relationship with attachment figures, not necessarily involving physical separation from caregivers
- Accelerated development
a hastened rate of emotional development seen following adversity that matches a child’s emotional needs, rather than their chronological age
Footnotes
DISCLOSURE STATEMENT
The authors are not aware of any affiliations, memberships, funding, or financial holdings that might be perceived as affecting the objectivity of this review. N.T. is a member of the Child Mind Institute’s Scientific Research Council, is a board member of the Flux Congress and the Society for Social Neuroscience, has received funding from the US National Institutes of Health and the Mindset Scholars Network Foundation, and is a member of the Scientific Advisory Board of the Leipzig Research Center for Early Child Development.
LITERATURE CITED
- Adolph KE, Berger SE. 2006. Motor development In Handbook of Child Psychology, Vol. 2: Cognition, Perception, and Language, ed. Kuhn D, Siegler RS, pp. 161–213. New York: Wiley; 6th ed. [Google Scholar]
- Ahnert L, Gunnar MR, Lamb ME, Barthel M. 2004. Transition to child care: associations with infant–mother attachment, infant negative emotion, and cortisol elevations. Child Dev. 75:639–50 [DOI] [PubMed] [Google Scholar]
- Ainsworth MD, Bell SM. 1970. Attachment, exploration, and separation: illustrated by the behavior of one year-olds in a strange situation. Child Dev. 41:49–67 [PubMed] [Google Scholar]
- Al Aïn S, Perry RE, Kayser K, Hochman C, Brehman E, et al. 2017. Neurobehavioral assessment of maternal odor in developing rat pups: implications for social buffering. Soc. Neurosci 12:32–49 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews K, Fitzgerald M. 1997. Barriers to optimal pain management in infants, children, and adolescents: biological barriers to paediatric pain management. Clin. J. Pain 13:138–43 [DOI] [PubMed] [Google Scholar]
- Anisfeld E, Casper V, Nozyce M, Cunningham N. 1990. Does infant carrying promote attachment? An experimental study of the effects of increased physical contact on the development of attachment. Child Dev. 61:1617–27 [DOI] [PubMed] [Google Scholar]
- Arruda-Carvalho M, Wu W-C, Cummings KA, Clem RL. 2017. Optogenetic examination of prefrontal— amygdala synaptic development. J. Neurosci 37:2976–85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Artioli F, Reese E. 2014. Early memories in young adults from separated and non-separated families. Memory 22:1082–102 [DOI] [PubMed] [Google Scholar]
- Baker KD, Bisby MA, Richardson R. 2016. Impaired fear extinction in adolescent rodents: behavioural and neural analyses. Neurosci. Biobehav. Rev 70:59–73 [DOI] [PubMed] [Google Scholar]
- Barr GA. 1995. Ontogeny of nociception and antinociception. NIDA Res. Monogr 158:172–201 [PubMed] [Google Scholar]
- Barr GA, Moriceau S, Shionoya K, Muzny K, Gao P, et al. 2009. Transitions in infant learning are modulated by dopamine in the amygdala. Nat. Neurosci 12:1367–69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bath KG, Manzano-Nieves G, Goodwill H. 2016. Early life stress accelerates behavioral and neural maturation of the hippocampus in male mice. Horm. Behav 82:64–71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayer SA, Altman J, Russo R, Zhang X. 1993. Timetables of neurogenesis in the human brain based on experimentally determined patterns in the rat. Neurotoxicology 14:83–144 [PubMed] [Google Scholar]
- Block JD, Sersen EA, Wortis J. 1970. Cardiac classical conditioning and reversal in the Mongoloid, encephalopathic, and normal child. Child Dev. 41:771–85 [Google Scholar]
- Bolles RC, Woods PJ. 1964. The onotogeny of behavior in the albino rat. Anim. Behav 12:425–41 [Google Scholar]
- Bos K, Zeanah CH, Fox NA, Drury SS, McLaughlin KA, Nelson CA. 2011. Psychiatric outcomes in young children with a history of institutionalization. Harv. Rev. Psychiatry 19:15–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowlby J 1969. Attachment and Loss, Vol. 1: Attachment. New York: Basic Books [Google Scholar]
- Bowlby J 1977. The making and breaking of affectional bonds. I. Aetiology and psychopathology in the light of attachment theory. An expanded version of the Fiftieth Maudsley Lecture, delivered before the Royal College of Psychiatrists, 19 November 1976. Br. J. Psychiatry 130:201–10 [DOI] [PubMed] [Google Scholar]
- Bronfenbrenner U 1977. Toward an experimental ecology of human development. Am. Psychol 32:513–31 [Google Scholar]
- Butler H, Juurlink B. 1987. An Atlas for Staging Mammalian and Chick Embryos. Boca Raton, FL: CRC Press [Google Scholar]
- Callaghan BL, Dandash O, Simmons J, Schwartz O, Byrne ML, et al. 2017. Amygdala resting connectivity mediates association between maternal aggression and adolescent major depression: a 7-year longitudinal study. J. Am. Acad. Child Adolesc. Psychiatry 56:983–991 [DOI] [PubMed] [Google Scholar]
- Callaghan BL, Richardson R. 2011. Maternal separation results in early emergence of adult-like fear and extinction learning in infant rats. Behav. Neurosci 125:20–28 [DOI] [PubMed] [Google Scholar]
- Callaghan BL, Richardson R. 2012a. Early-life stress affects extinction during critical periods of development: an analysis of the effects of maternal separation on extinction in adolescent rats. Stress 15:671–79 [DOI] [PubMed] [Google Scholar]
- Callaghan BL, Richardson R. 2012b. The effect of adverse rearing environments on persistent memories in young rats: removing the brakes on infant fear memories. Transl. Psychiatry 2:e138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callaghan BL, Sullivan RM, Howell B, Tottenham N. 2014. The International Society for Developmental Psychobiology Sackler Symposium: early adversity and the maturation of emotion circuits—a crossspecies analysis. Dev. Psychobiol 56:1635–50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callaghan BL, Tottenham N. 2016a. The neuro-environmental loop of plasticity: a cross-species analysis of parental effects on emotion circuitry development following typical and adverse caregiving. Neuropsychopharmacology 41:163–76 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callaghan BL, Tottenham N. 2016b. The stress acceleration hypothesis: effects of early-life adversity on emotion circuits and behavior. Curr. Opin. Behav. Sci 7:76–81 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camp LL, Rudy JW. 1988. Changes in the categorization of appetitive and aversive events during postnatal development of the rat. Dev. Psychobiol 21:25–42 [DOI] [PubMed] [Google Scholar]
- Campbell BA, Spear NE. 1972. Ontogeny of memory. Psychol. Rev 79:215–36 [DOI] [PubMed] [Google Scholar]
- Casey BJ, Glatt CE, Lee FS. 2015. Treating the developing versus developed brain: translating preclinical mouse and human studies. Neuron 86:1358–68 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casey BJ, Tottenham N, Liston C, Durston S. 2005. Imaging the developing brain: What have we learned about cognitive development? Trends Cogn. Sci 9:104–10 [DOI] [PubMed] [Google Scholar]
- CDC (Cent. Dis. Control Prev.). 2018. Child abuse and neglect: risk and protective factors. Cent. Dis. Control Prev https://www.cdc.gov/violenceprevention/childabuseandneglect/riskprotectivefactors.html [Google Scholar]
- Champagne FA, Francis DD, Mar A, Meaney MJ. 2003. Variations in maternal care in the rat as a mediating influence for the effects of environment on development. Physiol. Behav 79:359–71 [DOI] [PubMed] [Google Scholar]
- Clancy B, Finlay BL, Darlington RB, Anand KJS. 2007a. Extrapolating brain development from experimental species to humans. Neurotoxicology 28:931–37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clancy B, Kersh B, Hyde J, Darlington RB, Anand K, Finlay BL. 2007b. Web-based method for translating neurodevelopment from laboratory species to humans. Neuroinformatics 5:79–94 [DOI] [PubMed] [Google Scholar]
- Collier AC, Bolles RC. 1980. The ontogenesis of defensive reactions to shock in preweanling rats. Dev. Psychobiol 13:141–50 [DOI] [PubMed] [Google Scholar]
- Cunningham MG, Bhattacharyya S, Benes FM. 2002. Amygdalo–cortical sprouting continues into early adulthood: implications for the development of normal and abnormal function during adolescence. J. Comp. Neurol 453:116–30 [DOI] [PubMed] [Google Scholar]
- Dallman MF. 2000. Moments in time—the neonatal rat hypothalamo–pituitary–adrenal axis. Endocrinology 141:1590–92 [DOI] [PubMed] [Google Scholar]
- Danielewicz J, Hess G. 2014. Early life stress alters synaptic modification range in the rat lateral amygdala. Behav. Brain Res 265:32–37 [DOI] [PubMed] [Google Scholar]
- De Shetler NG, Rissman J. 2017. Dissociable profiles of generalization/discrimination in the human hippocampus during associative retrieval. Hippocampus 27:115–21 [DOI] [PubMed] [Google Scholar]
- DiPietro JA. 2012. Maternal stress in pregnancy: considerations for fetal development. J. Adolesc. Health 51(Suppl.):S3–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberg N, Cumberland A, Spinrad TL. 1998. Parental socialization of emotion. Psychol. Inq 9:241–47 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldman R, Singer M, Zagoory O. 2010. Touch attenuates infants’ physiological reactivity to stress. Dev. Sci 13:271–78 [DOI] [PubMed] [Google Scholar]
- Gabard-Durnam LJ, Flannery J, Goff B, Gee DG, Humphreys KL, et al. 2014. The development of human amygdala functional connectivity at rest from 4 to 23 years: a cross-sectional study. Neuroimage 95:193–207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gabard-Durnam LJ, Gee DG, Goff B, Flannery J, Telzer E, et al. 2016. Stimulus-elicited connectivity influences resting-state connectivity years later in human development: a prospective study. J. Neurosci 36:4771–84 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao Y, Raine A, Venables PH, Dawson ME, Mednick SA. 2010. The development of skin conductance fear conditioning in children from ages 3 to 8 years. Dev. Sci 13:201–12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gee DG, Gabard-Durnam LJ, Flannery J, Goff B, Humphreys KL, et al. 2013a. Early developmental emergence of human amygdala–prefrontal connectivity after maternal deprivation. PNAS 110:15638–43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gee DG, Gabard-Durnam LJ, Telzer EH, Humphreys KL, Goff B, et al. 2014. Maternal buffering of human amygdala–prefrontal circuitry during childhood but not during adolescence. Psychol. Sci 25:2067–78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gee DG, Humphreys KL, Flannery J, Goff B, Telzer EH, et al. 2013b. A developmental shift from positive to negative connectivity in human amygdala–prefrontal circuitry. J. Neurosci 33:4584–93 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giedd JN. 2004. Structural magnetic resonance imaging of the adolescent brain. Ann. N. Y. Acad. Sci 1021:77–85 [DOI] [PubMed] [Google Scholar]
- Giedd JN, Blumenthal J, Jeffries NO, Castellanos FX, Liu H, et al. 1999. Brain development during childhood and adolescence: a longitudinal MRI study. Nat. Neurosci 2:861–63 [DOI] [PubMed] [Google Scholar]
- Glenn CR, Klein DN, Lissek S, Britton JC, Pine DS, Hajcak G. 2012. The development of fear learning and generalization in 8–13 year-olds. Dev. Psychobiol 54:675–84 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gogtay N, Giedd JN, Lusk L, Hayashi KM, Greenstein D, et al. 2004. Dynamic mapping of human cortical development during childhood through early adulthood. PNAS 101:8174–79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham AM, Buss C, Rasmussen JM, Rudolph MD, Demeter DV, et al. 2016. Implications of newborn amygdala connectivity for fear and cognitive development at 6-months-of-age. Dev. Cogn. Neurosci 18:12–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham AM, Fisher PA, Pfeifer JH. 2013. What sleeping babies hear: a functional MRI study of interparental conflict and infants’ emotion processing. Psychol. Sci 24:782–89 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham AM, Pfeifer JH, Fisher PA, Carpenter S, Fair DA. 2015. Early life stress is associated with default system integrity and emotionality during infancy. J. Child Psychol. Psychiatry 56:1212–22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green JG, McLaughlin KA, Berglund PA, Gruber MJ, Sampson NA, et al. 2010. Childhood adversities and adult psychiatric disorders in the National Comorbidity Survey Replication (NCS-R) I: associations with first onset of DSM-IV disorders. Arch. Gen. Psychiatry 67:113–23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunnar MR, Hostinar CE, Sanchez MM, Tottenham N, Sullivan RM. 2015. Parental buffering of fear and stress neurobiology: reviewing parallels across rodent, monkey, and human models. Soc. Neurosci 10:474–78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haroutunian V, Campbell BA. 1979. Emergence of interoceptive and exteroceptive control of behavior in rats. Science 205:927–29 [DOI] [PubMed] [Google Scholar]
- Havighurst RJ. 1948. Developmental Tasks and Education. Chicago: Chicago Univ. Press [Google Scholar]
- Hennessy MB, Hornschuh G, Kaiser S, Sachser N. 2006. Cortisol responses and social buffering: a study throughout the life span. Horm. Behav 49:383–90 [DOI] [PubMed] [Google Scholar]
- Hennessy MB, Schiml PA, Willen R, Watanasriyakul W, Johnson J, Garrett T. 2015. Selective social buffering of behavioral and endocrine responses and Fos induction in the prelimbic cortex of infants exposed to a novel environment. Dev. Psychobiol 57:50–62 [DOI] [PubMed] [Google Scholar]
- Hostinar CE, Johnson AE, Gunnar MR. 2015a. Parent support is less effective in buffering cortisol stress reactivity for adolescents compared to children. Dev. Sci 18:281–97 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hostinar CE, Johnson AE, Gunnar MR. 2015b. Early social deprivation and the social buffering of cortisol stress responses in late childhood: an experimental study. Dev. Psychol 51:1597–1609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt PS, Richardson R, Campbell BA. 1994. Delayed development of fear-potentiated startle in rats. Behav. Neurosci 108:69–80 [DOI] [PubMed] [Google Scholar]
- Huot RL, Thrivikraman K, Meaney MJ, Plotsky PM. 2001. Development of adult ethanol preference and anxiety as a consequence of neonatal maternal separation in Long Evans rats and reversal with antidepressant treatment. Psychopharmacology 158:366–73 [DOI] [PubMed] [Google Scholar]
- Johnson DC, Casey BJ. 2015a. Easy to remember, difficult to forget: the development of fear regulation. Dev. Cogn. Neurosci 11:42–55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson DC, Casey BJ. 2015b. Extinction during memory reconsolidation blocks recovery of fear in adolescents. Sci. Rep 5:8863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jovanovic T, Nylocks KM, Gamwell KL, Smith A, Davis TA, et al. 2014. Development of fear acquisition and extinction in children: effects of age and anxiety. Neurobiol. Learn. Mem 113:135–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karmiloff-Smith A 2009. Nativism versus neuroconstructivism: rethinking the study of developmental disorders. Dev. Psychol 45:56–63 [DOI] [PubMed] [Google Scholar]
- Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. 2005. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch. Gen. Psychiatry 62:593–602 [DOI] [PubMed] [Google Scholar]
- Kessler RC, Greenberg PE. 2002. The economic burden of anxiety and stress disorders. Neuropsychopharmacology 67:982–92 [Google Scholar]
- Kikusui T, Winslow JT, Mori Y. 2006. Social buffering: relief from stress and anxiety. Philos. Trans. R. Soc. B 361:2215–28 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JH, Hamlin AS, Richardson R. 2009. Fear extinction across development: the involvement of the medial prefrontal cortex as assessed by temporary inactivation and immunohistochemistry. J. Neurosci 29:10802–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JH, Li S, Richardson R. 2011. Immunohistochemical analyses of long-term extinction of conditioned fear in adolescent rats. Cereb. Cortex 21:530–38 [DOI] [PubMed] [Google Scholar]
- Koppensteiner P, Aizawa S, Yamada D, Kabuta T, Boehm S, et al. 2014. Age-dependent sensitivity to glucocorticoids in the developing mouse basolateral nucleus of the amygdala. Psychoneuroendocrinology 46:64–77 [DOI] [PubMed] [Google Scholar]
- Lambert HK, Sheridan MA, Sambrook KA, Rosen ML, Askren MK, McLaughlin KA. 2017. Hippocampal contribution to context encoding across development is disrupted following early-life adversity. J. Neurosci 37:1925–34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landers MS, Sullivan RM. 2012. The development and neurobiology of infant attachment and fear. Dev. Neurosci 34:101–14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- LeDoux JE. 2000. Emotion circuits in the brain. Annu. Rev. Neurosci 23:155–84 [DOI] [PubMed] [Google Scholar]
- Leonardo ED, Hen R. 2008. Anxiety as a developmental disorder. Neuropsychopharmacology 33:134–40 [DOI] [PubMed] [Google Scholar]
- Li S, Kim J, Richardson R. 2012. Updating memories: changing the involvement of the prelimbic cortex in the expression of an infant fear memory. Neuroscience 222:316–25 [DOI] [PubMed] [Google Scholar]
- Lupien SJ, Parent S, Evans AC, Tremblay RE, Zelazo PD, et al. 2011. Larger amygdala but no change in hippocampal volume in 10-year-old children exposed to maternal depressive symptomatology since birth. PNAS 108:14324–29 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marco EM, Valero M, de la Serna O, Aisa B, Borcel E, et al. 2012. Maternal deprivation effects on brain plasticity and recognition memory in adolescent male and female rats. Neuropharmacology 68:223–31 [DOI] [PubMed] [Google Scholar]
- Matthews K, Robbins TW. 2003. Early experience as a determinant of adult behavioural responses to reward: the effects of repeated maternal separation in the rat. Neurosci. Biobehav. Rev 27:45–55 [DOI] [PubMed] [Google Scholar]
- McCallum J, Kim JH, Richardson R. 2010. Impaired extinction retention in adolescent rats: effects of dcycloserine. Neuropsychopharmacology 35:2134–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin KA, Green JG, Gruber MJ, Sampson NA, Zaslavsky AM, Kessler RC. 2012. Childhood adversities and first onset of psychiatric disorders in a national sample of US adolescents. Arch. Gen. Psychiatry 69:1151–60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin KA, Sheridan MA, Gold AL, Duys A, Lambert HK, et al. 2016. Maltreatment exposure, brain structure, and fear conditioning in children and adolescents. Neuropsychopharmacology 41:1956–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNally RJ. 2007. Mechanisms of exposure therapy: how neuroscience can improve psychological treatments for anxiety disorders. Clin. Psychol. Rev 27:750–59 [DOI] [PubMed] [Google Scholar]
- Michalska KJ, Shechner T, Hong M, Britton JC, Leibenluft E, et al. 2016. A developmental analysis of threat/safety learning and extinction recall during middle childhood. J. Exp. Child Psychol 146:95–105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mineka S, Zinbarg R. 2006. A contemporary learning theory perspective on the etiology of anxiety disorders: It’s not what you thought it was. Am. Psychol 61:10–26 [DOI] [PubMed] [Google Scholar]
- Monk CS, Grillon C, Baas JMP, McClure EB, Nelson EE, et al. 2003. A neuroimaging method for the study of threat in adolescents. Dev. Psychobiol 43:359–66 [DOI] [PubMed] [Google Scholar]
- Moriceau S, Shionoya K, Jakubs K, Sullivan RM. 2009. Early-life stress disrupts attachment learning: the role of amygdala corticosterone, locus ceruleus corticotropin releasing hormone, and olfactory bulb norepinephrine. J. Neurosci 29:15745–55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moriceau S, Sullivan RM. 2006. Maternal presence serves as a switch between learning fear and attraction in infancy. Nat. Neurosci 9:1004–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moriceau S, Wilson DA, Levine S, Sullivan RM. 2006. Dual circuitry for odor–shock conditioning during infancy: corticosterone switches between fear and attraction via amygdala. J. Neurosci 26:6737–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myers KM, Davis M. 2007. Mechanisms of fear extinction. Mol. Psychiatry 12:120–50 [DOI] [PubMed] [Google Scholar]
- Nachmias M, Gunnar M, Mangelsdorf S, Parritz RH, Buss K. 1996. Behavioral inhibition and stress reactivity: the moderating role of attachment security. Child Dev. 67:508–22 [PubMed] [Google Scholar]
- Opendak M, Sullivan RM. 2016. Unique neurobiology during the sensitive period for attachment produces distinctive infant trauma processing. Eur. J. Psychotraumatology 7:31276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papousek M, Papousek H. 1990. Excessive infant crying and intuitive parental care: buffering support and its failures in parent–infant interaction. Early Child Dev. Care 65:117–26 [Google Scholar]
- Pattwell SS, Bath KG, Casey BJ, Ninan I, Lee FS. 2011. Selective early-acquired fear memories undergo temporary suppression during adolescence. PNAS 108:1182–87 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pattwell SS, Duhoux S, Hartley CA, Johnson DC, Jing D, et al. 2012. Altered fear learning across development in both mouse and human. PNAS 109:16318–23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pattwell SS, Liston C, Jing D, Ninan I, Yang RR, et al. 2016. Dynamic changes in neural circuitry during adolescence are associated with persistent attenuation of fear memories. Nat. Commun 7:11475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perlman SB, Pelphrey KA. 2011. Developing connections for affective regulation: age-related changes in emotional brain connectivity. J. Exp. Child Psychol 108:607–20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perry RE, Al Aïn S, Raineki C, Sullivan RM, Wilson DA. 2016. Development of odor hedonics: experience dependent ontogeny of circuits supporting maternal and predator odor responses in rats. J. Neurosci 36:6634–50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perry RE, Finegood ED, Braren SH, Dejoseph ML, Putrino DF, et al. 2018. Developing a neurobehavioral animal model of poverty: Drawing cross-species connections between environments of scarcityadversity, parenting quality, and infant outcome. Dev. Psychopathol In press. 10.1017/S095457941800007X [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perry RE, Sullivan RM. 2014. Neurobiology of attachment to an abusive caregiver: short-term benefits and long-term costs. Dev. Psychobiol 56:1626–34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phelps EA, Delgado MR, Nearing KI, LeDoux JE. 2004. Extinction learning in humans: role of the amygdala and vmPFC. Neuron 43:897–905 [DOI] [PubMed] [Google Scholar]
- Pugh CR, Tremblay D, Fleshner M, Rudy JW. 1997. A selective role for corticosterone in contextual-fear conditioning. Behav. Neurosci 111:503–11 [PubMed] [Google Scholar]
- Qiu A, Anh TT, Li Y, Chen H, Rifkin-Graboi A, et al. 2015. Prenatal maternal depression alters amygdala functional connectivity in 6-month-old infants. Transl. Psychiatry 5:e508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quirk GJ, Mueller D. 2008. Neural mechanisms of extinction learning and retrieval. Neuropsychopharmacology 33:56–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raineki C, Cortés MR, Belnoue L, Sullivan RM. 2012. Effects of early-life abuse differ across development: Infant social behavior deficits are followed by adolescent depressive-like behaviors mediated by the amygdala. J. Neurosci 32:7758–65 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raineki C, Holman PJ, Debiec J, Bugg M, Beasley A, Sullivan RM. 2010a. Functional emergence of the hippocampus in context fear learning in infant rats. Hippocampus 20:1037–46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raineki C, Moriceau S, Sullivan RM. 2010b. Developing a neurobehavioral animal model of infant attachment to an abusive caregiver. Biol. Psychiatry 67:1137–45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raineki C, Shinoya K, Sander K, Sullivan RM. 2009. Ontogeny of odor-LiCl vs. odor-shock learning: similar behaviors but divergent ages of functional amygdala emergency. Learn. Mem 16:114–21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Repacholi BM, Meltzoff AN. 2007. Emotional eavesdropping: Infants selectively respond to indirect emotional signals. Child Dev. 78:503–21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson-Drummer PA, Stanton ME. 2015. Using the context preexposure facilitation effect to study long term context memory in preweanling, juvenile, adolescent, and adult rats. Physiol. Behav 148:22–28 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth TL, Sullivan RM. 2005. Memory of early maltreatment: neonatal behavioral and neural correlates of maternal maltreatment within the context of classical conditioning. Biol. Psychiatry 57:823–31 [DOI] [PubMed] [Google Scholar]
- Rudy JW, Morledge P. 1994. Ontogeny of contextual fear conditioning in rats: implications for consolidation, infantile amnesia, and hippocampal system function. Behav. Neurosci 108:227–34 [DOI] [PubMed] [Google Scholar]
- Schaffer HR, Greenwood A, Parry MH. 1972. The onset of wariness. Child Dev. 43:165–75 [Google Scholar]
- Schlund MW, Siegle GJ, Ladouceur CD, Silk JS, Cataldo MF, et al. 2010. Nothing to fear? Neural systems supporting avoidance behavior in healthy youths. Neuroimage 52:710–19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seltzer LJ, Prososki AR, Ziegler TE, Pollak SD. 2012. Instant messages versus speech: hormones and why we still need to hear each other. Evol. Hum. Behav 33:42–45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silvers JA, Insel C, Powers A, Franz P, Helion C, et al. 2016a. vlPFC–vmPFC–amygdala interactions underlie age-related differences in cognitive regulation of emotion. Cereb. Cortex 27:3502–14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silvers JA, Lumian DS, Gabard-Durnam L, Gee DG, Goff B, et al. 2016b. Previous institutionalization is followed by broader amygdala–hippocampal–PFC network connectivity during aversive learning in human development. J. Neurosci 36:6420–30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smotherman WP. 1982. Odor aversion learning by the rat fetus. Physiol. Behav 29:769–71 [DOI] [PubMed] [Google Scholar]
- Spangler G, Schieche M. 1998. Emotional and adrenocortical responses of infants to the strange situation: the differential function of emotional expression. Int. J. Behav. Dev 22:681–706 [Google Scholar]
- Spear LP. 2000. The adolescent brain and age-related behavioral manifestations. Neurosci. Biobehav. Rev 24:417–63 [DOI] [PubMed] [Google Scholar]
- Stanton ME, Levine S. 1990. Inhibition of infant glucocorticoid stress response: specific role of maternal cues. Dev. Psychobiol 23:411–26 [DOI] [PubMed] [Google Scholar]
- Stanton ME, Wallstrom J, Levine S. 1987. Maternal contact inhibits pituitary–adrenal stress responses in preweanling rats. Dev. Psychobiol 20:131–45 [DOI] [PubMed] [Google Scholar]
- Suchecki D, Rosenfeld P, Levine S. 1993. Maternal regulation of the hypothalamic–pituitary–adrenal axis in the infant rat: the roles of feeding and stroking. Brain Res. Dev. Brain Res 75:185–92 [DOI] [PubMed] [Google Scholar]
- Sullivan RM, Holman PJ. 2010. Transitions in sensitive period attachment learning in infancy: the role of corticosterone. Neurosci. Biobehav. Rev 34:835–44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan RM, Landers M, Yeaman B, Wilson DA. 2000. Good memories of bad events in infancy. Nature 407:38–39 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan RM, Wilson DA, Wong R, Correa A, Leon M. 1990. Modified behavioral and olfactory bulb responses to maternal odors in preweanling rats. Brain Res. Dev. Brain Res 53:243–47 [DOI] [PubMed] [Google Scholar]
- Swartz JR, Carrasco M, Wiggins JL, Thomason ME, Monk CS. 2014. Age-related changes in the structure and function of prefrontal cortex–amygdala circuitry in children and adolescents: a multi-modal imaging approach. Neuroimage 86:212–20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thijssen S, Muetzel RL, Bakermans-Kranenburg MJ, Jaddoe VW, Tiemeier H, et al. 2017. Insensitive parenting may accelerate the development of the amygdala–medial prefrontal cortex circuit. Dev. Psychopathol 29:505–18 [DOI] [PubMed] [Google Scholar]
- Thompson BL, Levitt P. 2010. The clinical–basic interface in defining pathogenesis in disorders of neurodevelopmental origin. Neuron 67:702–12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tottenham N 2012. Human amygdala development in the absence of species-expected caregiving. Dev. Psychobiol 54:598–611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tottenham N, Hare TA, Quinn BT, McCarry TW, Nurse M, et al. 2010. Prolonged institutional rearing is associated with atypically large amygdala volume and difficulties in emotion regulation. Dev. Sci 13:46–61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uematsu A, Matsui M, Tanaka C, Takahashi T, Noguchi K, et al. 2012. Developmental trajectories of amygdala and hippocampus from infancy to early adulthood in healthy individuals. PLOS ONE 7:e46970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Upton KJ, Sullivan RM. 2010. Defining age limits of the sensitive period for attachment learning in rat pups. Dev. Psychobiol 52:453–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Oers HJ, de Kloet ER, Levine S. 1998. Early versus late maternal deprivation differentially alters the endocrine and hypothalamic responses to stress. Dev. Brain Res 111:245–52 [DOI] [PubMed] [Google Scholar]
- Walker CD, Bath KG, Joels M, Korosi A, Larauche M, et al. 2017. Chronic early life stress induced by limited bedding and nesting (LBN) material in rodents: critical considerations of methodology, outcomes and translational potential. Stress 20:421–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wickens DD, Wickens CL. 1940. A study of conditioning in the neonate. J. Exp. Psychol 26:94–102 [Google Scholar]
- Wilson A, Brooks DC, Bouton ME. 1995. The role of the rat hippocampal system in several effects of context in extinction. Behav. Neurosci 109:828–36 [DOI] [PubMed] [Google Scholar]
- Winnicott DW. 1960. The theory of the parent–infant relationship. Int. J. Psychoanal 41:585–95 [PubMed] [Google Scholar]
- Woodward LJ, Fergusson DM. 2001. Life course outcomes of young people with anxiety disorders in adolescence. J. Am. Acad. Child Adolesc. Psychiatry 40:1086–93 [DOI] [PubMed] [Google Scholar]
- Yap CS, Richardson R. 2007. Extinction in the developing rat: an examination of renewal effects. Dev. Psychobiol 49:565–75 [DOI] [PubMed] [Google Scholar]
- Young ES, Griskevicius V, Simpson JA, Waters TEA, Mittal C. 2018. Can an unpredictable childhood environment enhance working memory? Testing the sensitized-specialization hypothesis. J. Personal. Soc. Psychol 114:891–908 [DOI] [PubMed] [Google Scholar]
- Yurgelun-Todd DA, Killgore WD. 2006. Fear-related activity in the prefrontal cortex increases with age during adolescence: a preliminary fMRI study. Neurosci. Lett 406:194–99 [DOI] [PubMed] [Google Scholar]


