Abstract
Purpose
To explore contemporary clincial case management of patients with Ebola virus disease.
Methods
A narrative review from a clinical perspective of clinical features, diagnostic tests, treatments and outcomes of patients with Ebola virus disease.
Results
Substantial advances have been made in the care of patients with Ebola virus disease (EVD), precipitated by the unprecedented extent of the 2014–2016 outbreak. There has been improved point-of-care diagnostics, improved characterization of the clinical course of EVD, improved patient-optimized standards of care, evaluation of effective anti-Ebola therapies, administration of effective vaccines, and development of innovative Ebola treatment units. A better understanding of the Ebola virus disease clinical syndrome has led to the appreciation of a central role for critical care clinicians—over 50% of patients have life-threatening complications, including hypotension, severe electrolyte imbalance, acute kidney injury, metabolic acidosis and respiratory failure. Accordingly, patients often require critical care interventions such as monitoring of vital signs, intravenous fluid resuscitation, intravenous vasoactive medications, frequent diagnostic laboratory testing, renal replacement therapy, oxygen and occasionally mechanical ventilation.
Conclusion
With advanced training and adherence to infection prevention and control practices, clinical interventions, including critical care, are feasible and safe to perform in critically ill patients. With specific anti-Ebola medications, most patients can survive Ebola virus infection.
Keywords: Ebola, Critical care, Intensive care
Take-home messages
|
The clinical spectrum of Ebola virus disease ranges from mild to severe illness and is often associated with life-threatening complications that require advanced supportive and critical care. Training, preparation and strict adherence to infection prevention and control practices by health workers have demonstrated the feasibility and safety of the provision of advanced critical care. Recent Ebola-specific care advances including vaccine prevention, point-of-care diagnostics, effective anti-Ebola treatments, a wider consensus on provision of optimized care and an increased acceptance and capacity to conduct well-designed research are leading to improved clinical outcomes for patients. |
Introduction
Ebola virus disease (EVD) has long been perceived as a rare Equatorial viral illness that leads to hemorrhagic fever and near certain death. Until recently there was a common view that little could be done for patients other than isolation and limited supportive care—a reasoning premised on the expected futility of care for patients with an illness of historically high mortality rate and a concern for risk of infection to healthcare workers [1–3]. However, over the past 5 years, alongside the West African outbreak, there has been improved characterization of the clinical course, recognition that Ebola frequently leads to a unique critical illness with multisystem organ failure [1–8], and that patients with Ebola virus that can be supported, treated and cured [5, 9–11].
Historical case-weighted mortality fell from approximately 70% in outbreaks prior to 2014, to 39% in the West African outbreak and 18.5% in patients with EVD who were treated in Europe and USA [12, 13]. Mortality has remained over 50% in the most recent outbreaks in the Democratic Republic of the Congo (DRC) [13]. Treatment has shifted from a focus upon isolation and oral rehydration (‘minimal-touch’) to one of optimized care involving rapid diagnostic testing, frequent or continuous monitoring of vital signs, individualized enteral and intravenous fluid treatment, intravenous vasoactive medications, supplemental oxygen, occasional mechanical ventilation, renal replacement therapy and now, specific and effective antiviral therapies [9–12, 14–18].
In this review, we highlight the need for clinicians to be able to recognize and manage Ebola-related organ dysfunction, and highlight that advanced supportive care is necessary of treatment for severely ill patients with EVD.
Epidemiology
Ebolaviruses fall under the order Mononegavirales and the filoviridae family, and whose viral genome comprises a single negative stranded RNA with negative polarity. Within the genus of Ebolavirus, 6 species have been identified, though only 4—Zaire Ebola virus, Bundibugyo virus, Sudan virus and Tai Forest virus—are known to cause disease in humans; no serious illness has been reported from Bombali or Reston ebolavirus [19, 20]. Non-human primates (NHP) and bats are considered the natural reservoirs of Ebola virus, with the virus capable of causing zoonotic outbreaks.
Since the first recognized EVD outbreak in 1976, approximately 30 others have occurred [13, 19], with the majority happening in equatorial sub-Saharan Africa. Previously, most outbreaks have occurred in rural and remote areas, and have been of relatively short duration. However, in 2014–2016, EVD spread throughout West Africa (predominantly Guinea, Liberia and Sierra Leone), fueled in part by a fragile pre-existing health system and lack of human resource capacity, and led to the world’s largest outbreak (28,610 cases with 11,308 deaths and a case fatality ratio of 39%) [13]. Since the West African outbreak, the Democratic Republic of the Congo (DRC) has faced three more EVD outbreaks. In the most recent, centered in Northeastern DRC, as of January 04, 2020, there have been 2233 deaths out of 3386 cases—the second largest Ebola outbreak in history [17, 21]. Now characterized as a public health emergency of international concern [17] there is a need for clinicians to be prepared to treat patients in both endemic or exported regions.
Clinical presentation
Demographics
Ebola virus (EV) can infect people of all ages—from infants to elderly—and infection can affect fetal development and viability. In previous outbreaks, 10-year age group-incidence for EVD increased linearly from infancy peaking at the 35–45 years age group and falling thereafter [22]. Attack rates have traditionally been higher among women and middle-aged people and lower in children and the elderly, probably due to differences in exposure risks. The current DRC outbreak has been epidemiologically notable for higher than previously recognized infections among children [17].
Transmission
Most human-to-human Ebola transmission is by direct exposure of the mucous membranes or non-intact skin to infectious body fluids such as blood, vomitus, stool, or with contaminated materials [19]. Inoculation of the virus into the body via needle stick or sharps injuries can occur in healthcare settings. Droplet or aerosol transmission is unlikely except under specific conditions such as when carrying out procedures in non-ventilated rooms on patients with high viral loads that increase the risk respiratory spread, such as bronchoscopy [23]. Survivors from previous outbreaks have shown persistent Ebola virus in some bodily fluids, such as semen in males, up to 500 days after recovery [24]; however, the viability and infectivity of the virus isolated from these sites is less certain.
Pathophysiology
Ebola virus gains entry into the body mainly via the mucous membranes (eyes, nose, and mouth). Its viral glycoprotein enhances receptor binding and attachment to the endosomes of cells [23]. The virus attacks dendritic cells, monocytes and macrophages, which then migrate to the lymph nodes where early viral replication and dissemination occurs before the onset of symptoms. Ebola virus disseminates to infect a broad range of cells including endothelial cells, hepatocytes, fibroblasts, and the adreno-cortical cells, among others. Ebola virus uses its structural and membrane proteins through a number of mechanisms to evade the host immune response [23, 25, 26]. Tissue damage happens via numerous but closely related mechanisms such as direct viral cytopathy, endothelial dysfunction, coagulation disruption, and likely, the host’s own inflammatory response [23]. Infected patients undergo a cell-mediated immune activation, however, patients that succumb to EVD may have a more limited functional T-cell-mediated response [4, 27].
Clinical presentation
Clinical suspicion of EVD should arise when considering a patient’s clinical syndrome and recent likelihood of exposure in an area where the virus is known to circulate [28]. An epidemiological history should include any recent (within 21 days) travel to an area with ongoing Ebola transmission, or contact history with infected persons or dead or possibly infected animals.
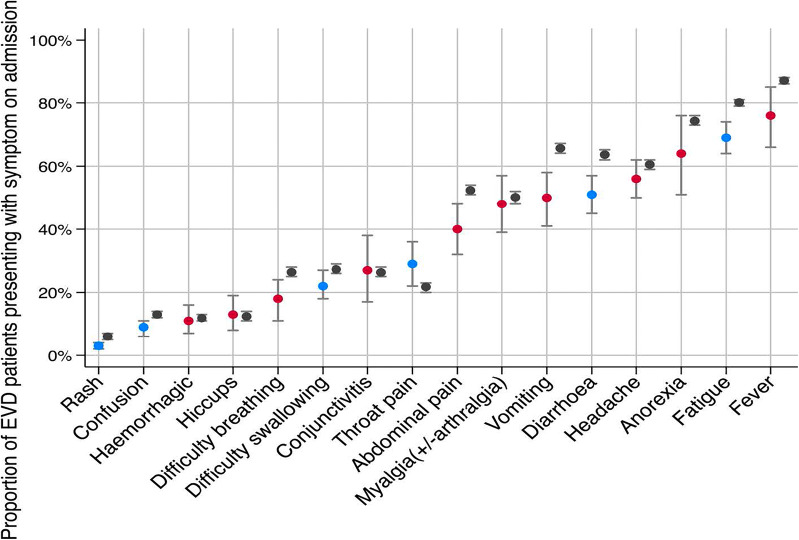
After exposure to Ebola virus, an incubation period of 3–21 days (mean 12.7, SD 4.3 days) [29] ensues where symptoms (Fig. 1) might present [30]. It is not uncommon for viral RNA to be undetectable by reverse transcriptase-polymerase chain reaction (RT-PCR) in the first 1–3 days after symptom onset. This necessitates re-testing all patients in whom clinical suspicion persists even after initial plasma RT-PCR results are negative if symptoms have been present for fewer than a few days [14].
Fig. 1.
A meta-analysis of proportion of 6168 EVD patients presenting with symptoms compared to reference data. Blue indicates a meta-analysis approximate with low or moderate heterogeneity, red is for a high heterogeneity of pooled estimates, and grey is the WHO reference data.
Adapted from Rojek AM et al. [30]
Several observational studies [12, 16, 31, 32], have grouped the progression of EVD illness into three stages (Table 1). In the initial phase, most patients’ symptoms include a non-specific febrile illness with headache, fever, myalgia and general malaise, with or without a maculopapular rash. These early symptoms often overlap with those of common diseases in the tropics (such as malaria, typhoid fever, and other bacterial and arboviral infections), complicating clinical diagnosis [7].
Table 1.
| Time since symptom onset | Clinical features | Typical patient | |
|---|---|---|---|
| Stage 1: early febrile or mild phase | 0–3 days | Non-specific features: fever, weakness, lethargy, and myalgia | Ambulatory, able to compensate for fluid losses; no indication for fluid administration |
| Stage 2: gastrointestinal phase | 3–10 days | Same as early stage plus diarrhea, vomiting, or both, or abdominal pain | Unable to compensate for fluid losses because of emesis or large volume losses; frequent indication for intravenous fluid administration |
| Stage 3: complications phase | 7–12 days | Same as gastrointestinal phase plus hemorrhage, shock, organ failure, and neurological complications | Critically ill, often with multisystem organ dysfunction such as acute kidney injury, coagulopathy, hemorrhage, encephalopathy, altered mental status, seizures |
Some patients will progress to the second phase with asthenia and gastro-intestinal illness, including nausea, anorexia, abdominal pain, odynophagia, emesis and then watery diarrhea. This stage can range from mild symptoms to a severe state of illness characterized by intravascular volume depletion and electrolyte derangement [6, 32], hypotension, metabolic acidosis, hepatitis, pancreatitis, hypoglycemia and renal injury. Patients are typically with a high viral load and are correspondingly infectious in this phase of illness [14].
In the third phase of illness [4], multi-organ failure may occur, with acute kidney failure, hepatic failure, cardiac dysfunction and shock, altered level of consciousness, seizures, coagulopathy (petechiae, ecchymoses, at venipuncture sites and bleeding from mucosal membranes and peri-partum) or death [30–33].
Diagnostic testing for Ebola
The WHO recommends confirmation of EVD by RT-quantitative PCR [34]. There are over a dozen RT-PCR processing platforms, but only a few have had their limits of detection (LOD) verified by independent assessors, including the WHO endorsed Cepheid Xpert® assay, which targets nucleoprotein and glycoprotein antigens on the Ebola virus [34–36]. For screening suspected EVD patients in remote settings without access to PCR testing, WHO recommends use of highly sensitive and specific rapid diagnostic tests (RDTs). Table 2 outlines various RDTs available, with only a few, such as ReEBOV, Ebola eZYSCREEN and OraQuick, having undergone clinical validation [35, 37–40]. Laboratory testing for alternative or co-infections such as malaria and bacterial infections should be considered [8, 12, 23, 31], since these are often assumed to co-exist with Ebola in severely ill patients and may lead to over-prescription of antimalarials or antibiotics unless rapid tests (for malaria) and cultures (for bacterial infections) can rule these out.
Table 2.
RDTs for the detection of Ebola virus.
Adapted from Tembo et al. [35]
| RDT type | Manufacturer | Target | Turnaround time (min) | Storage temperature (°C) | Clinical validation status | Sample |
|---|---|---|---|---|---|---|
| Ebola eZYSCREEN | BioMérieux and Institut des sciences du vivant Frédéric Joliot, France | EBOV GP | 15 | 4–30 | Yes | Whole blood or plasma |
| ReEBOV antigen rapid test | Zalgen Labs (previously Corgenix Ltd), USA | EBOV, BDBV, and SUDV VP40 protein antigen | 15 | 2–8 | Yes | Whole blood or plasma |
| ELISA antigen capture | National Institute of Infectious Diseases, Japan | RESTV NP protein antigen | Hours | 2–8 | NR | Serum |
| Ebola-specific IgM and Ig G antibody detection | Corgenix USA | EBOV and SUDV- specific antibodies against VP40 antigen | Hours | 2–8 | Yes | Serum |
| OraQuick® | OraSure Technologies Inc, USA | Detects Ebola VP40 antigen (EBOV, SUDV and BDBV) | 20–30 | 4–30 | Yes | Whole blood and saliva (post mortem samples) |
| DSTL EVD RDT | UK’s Defence Science Technology and Laboratory | EBOV antigens (unspecified) | 20 | NR | Yes | Whole blood |
| Matrix Multiplexed diagnostic | MMDX | EBOV GP | 10 | NR | NR | Plasma and serum |
| SD Q Line Zaire Ag assay | SD Biosensor Inc, Republic of Korea | EBOV protein antigens (GP, NP and VP40) | 30 | 1–40 | Yes | Whole blood, plasma and serum |
| QuickNavi-Ebola | Denka Seiken, Japan | EBOV, TAFV, BDBV, SUDV NP protein antigen | 10 | 4–45 | Yes | Whole blood, plasma and serum |
| iSENSE | University College London | EBOV, SUDV, and BDBV Ig G | 40 | NR | NR | Venous blood |
Clinical validation refers to field testing for specificity and sensitivity on blood specimens containing varying amounts of Ebola virus, with the specific RDT results compared to the polymerase chain reaction gold standard test [37–40]
BDBV, Bundibugyo ebolavirus; GP, glycoprotein; Ig G, immunoglobulin G; Ig M, immunoglobulin M; VP, matrix protein; NP, nucleoprotein; NR, not reported; RESTV, Reston ebolavirus; SUDV, Sudan ebolavirus; TAFV, Taï Forest ebolavirus; EBOV, Zaire ebolavirus
Laboratory abnormalities
Early in the course of EVD illness, leukocyte and lymphocyte counts often fall due to impaired immune response, while hemoglobin and hematocrit levels increase along with intravascular volume depletion [23]. Elevation in aspartate amino transferase (AST), alanine aminotransferase (ALT), and total serum bilirubin is common, generally believed to be due to direct viral cytopathic effect on hepatocytes [4, 12, 23]. Contributory pathology from rhabdomyolysis or myositis leads to high serum creatine kinase and AST levels [12, 23]. Elevated prothrombin time and activated partial thromboplastin time and increased d-dimers occur among patients with progressive illness [4, 8, 12, 23].
During the most severe phase of illness (Table 3), patients might have hyper- or hyponatremia, hyper- or hypokalemia, hypocalcemia and hypomagnesemia as a result severe vomiting and diarrhea; hypoglycemia secondary to reduced oral intake or depleted glycogen stores, and hypoalbuminemia, acute kidney injury, and both anion gap (lactate- or urea-associated) or non-anion gap (diarrhea-related bicarbonate losses) metabolic acidosis. [4, 8, 12, 23].
Table 3.
Laboratory abnormalities observed in EVD patients by stage.
Adapted with permission [23]
| Timing | Laboratory findings |
|---|---|
| Stage 1: early febrile or mild phase and Stage 2: gastrointestinal phase | Leukopenia, lymphopenia, and thrombocytopenia |
| Elevated hemoglobin, hematocrit, urea, creatinine (due to intravascular volume depletion) | |
| Elevated aspartate aminotransferase and alanine aminotransferase (ratio ≥ 3:1) | |
| Elevated prothrombin time, activated partial thromboplastin time, and D-dimers | |
| Stage 3: complications phase | Leukocytosis, neutrophilia, and anemia |
| Hyponatremia, hypo/hyperkalemia, hypomagnesemia, hypocalcemia, hypoalbuminemia, and hypoglycemia | |
| Elevated creatine phosphokinase and serum amylase | |
| Elevated blood urea nitrogen and serum creatinine and decreased urine output | |
| Metabolic acidosis, both anion gap (elevated serum lactate due to tissue hypoperfusion) and non-anion gap (low serum bicarbonate due to diarrhea and renal injury) | |
| Recovery phase | Thrombocytosis |
| Occasional residual virus detection in cerebrospinal fluid (CSF), semen, breast milk, vitreous humor and certain other body fluids |
Prevention
An overriding principle in providing EVD care is to ensure healthcare worker (HW) safety through strict infection prevention and control (IPC) practices. HWs involved in EVD care should adhere to both standard and transmission-based (contact and droplet) precautions for their safety [14]. Recommended personal protective equipment (PPE) when providing direct patient care includes: face shield/googles; fluid-resistant medical/surgical gown, surgical masks or N-95 respirators that maintain their shape and consistency in hot and humid environments; two pairs of gloves; waterproof apron; fluid-impermeable protective feet covering and a head cover [14, 41]. A buddy (two person) system while providing care, and PPE adherence supervisors are both advisable. Vaccination of frontline HWs should be a standard of clinical practice [16, 42].
Pre-exposure prophylaxis
Preliminary data on vaccination of populations at increased risk of infection, such as HWs, contacts of patients and contacts of contacts, with the rVSV ZEBOV-Gp, while utilizing the ring vaccination strategy in West Africa has shown a vaccine efficacy of 97.5% in people vaccinated at least 10 days before potential exposure, and an 88.1% efficacy in all the other analyses of EVD onset irrespective of timing [43]. Subsequently, a 26 vectored glycoprotein/MVA-BN (Ad26.ZEBOV/MVA-BN) vaccine developed by Johnson & Johnson has been deployed for evaluation in the current outbreak in DRC.
Treatment
The care for critically ill EVD patients is best offered in individual patient treatment rooms inside a facility that has the following features: a sectioned unit with separate areas for low risk patients in whom EVD has not been confirmed and who are not demonstrating ‘wet’ symptoms (vomiting and diarrhea); patients who have wet symptoms but as yet unconfirmed infection; and a section for confirmed EVD patients. Each section should have waste disposal areas; unidirectional flow of materials and personnel from the lowest risk zones to higher risk zones; and capacity to accommodate medical technologies such as oxygen delivery systems, mechanical ventilators and renal replacement systems [41]. Diagnostic laboratory testing should be either at the point of care and with a well-coordinated and timely system of safe transportation of samples to a dedicated laboratory equipped to process such specimens. There should be physical barriers to limit access to Ebola assessment and treatment areas to essential and trained personnel. However, there should be visual accessibility to patients by family and friends who provide important psychological support to patients.
Heath workers’ use of impermeable PPE while caring for EVD patients with infectious “wet’ symptoms puts them at increased risk for heat strain especially in hot and humid areas. This limitation can have the unintended consequence of reducing the time HWs can spend with patients and can limit the quality of care [2, 10, 16]. Thus, temperature and humidity control for HWs is an important element of caring for patients. This has been accomplished in the field through caring for patients in climate controlled hospitals, or through novel engineering solutions with individual patient rooms such as found in the (Alliance for International Medical Action—ALIMA) biosecure cubes (Fig. 2) that consist of transparent and air conditioned patient rooms, and come fitted with specialized ports to enable patient access and monitoring while in minimal PPE [10, 44].
Fig. 2.

A medical advisor for the Alliance for International Medical Action demonstrating a Biosecure Emergency Care Unit for outbreaks of infectious diseases.
Photo credit: Etinosa Yvonne/ALIMA [44]
Basic supportive care
Following the 2014–2016 EVD outbreak, a number of observational studies postulated that delivery of improved supportive care would reduce mortality from EVD [1–9, 12, 31, 32]. Basic supportive care recommendations [11] include: systematic monitoring and documentation of clinical signs and symptoms; a clinician to patient ratio of 1:4 but with emphasis placed on maximizing patient contact time; patient–family audio-visual communication; psychosocial care; provision of oral rehydration therapy to correct or prevent hypovolemia; parenteral fluids for patients unable to achieve sufficient enteral hydration; measurement of serum biochemistry with correction of identified electrolyte abnormalities; oxygen administration to achieve normal oxygen saturation; intravenous vasoactive medications for patients with fluid-resistant hypotension and organ hypoperfusion; pain management, antiemetics, and anxiolytics; nutritional support customized to patient assessment including treatment of hypoglycemia with intravenous glucose when necessary; and microbiological analysis to guide antimicrobial use, and in absence of such capacity, a low threshold for empirical use of broad-spectrum antibiotics and antimalarials, depending on the geographical context and symptoms [9–12, 14, 44, 45]. Provision of supportive care to patients will inevitably require a multi-disciplinary team of HWs: nurses, physicians, IPC personnel, laboratory personnel, and ideally, nutritionists, social and community workers, and psychologists. These recommendations derived from observational studies that generally found improved outcomes later in the outbreak compared to the beginning, attributed to improvements in multiple domains of care, including clinical monitoring and fluid administration [46].
Advanced supportive care
Data collected from patient cohorts admitted in Ebola Treatment Centres in Africa [4, 30] and in Europe and the USA [12] posited that approximately 50% of patients in either setting were critically ill and required advanced supportive care interventions. Described below is the management of common complications observed in patients with EVD.
Metabolic disturbance and renal injury has been observed as a complication of EVD in over 50% of patients in cohorts of patients during the West African outbreak [6–8, 12, 16, 23] and is likely multifactorial in etiology, including hypovolemia, hypotension and hypoperfusion, direct viral damage to the endothelium and renal tubular cells [6, 12–14, 23, 31], and occasional toxicity due to rhabdomyolysis or medications. Interventions include careful monitoring of urine output, maintaining adequate renal perfusion with fluids and vasoactive agents, avoiding nephrotoxic drugs and diuretics in hypovolemic patients, and treatment of other underlying cause(s) [10–12, 16]. Electrolyte disturbances such as hyperkalemia have commonly been reported in the context of Ebola-related renal injury. Temporizing measures for complications of acute kidney injury should be available, such as bicarbonate for metabolic acidosis, intravenous short-acting insulin and dextrose for hyperkalemia, and a diuretic trial in oliguric volume-replete patients. Where facilities and expertise are available, renal replacement therapy should be offered [11, 12, 16], as feasibility of delivering dialysis has been demonstrated in both resource-rich and resource-limited settings [12, 16, 47].
Cardiovascular complications have included arrhythmias and may often respond to normalization of serum electrolytes. Hypotension due to hypovolemia, vasodilation or impaired myocardial performance unresponsive to fluid resuscitation require intravenous vasopressor or inotropic support.
Coagulation abnormalities have included thrombocytopenia, vitamin K deficiency or liver-injury-related effects to the intrinsic and extrinsic pathways of coagulation, and rarely, disseminated intravascular coagulation. Clinical monitoring and measurement of blood counts and coagulation profiles can help to guide treatment with vitamin K and tranexamic acid, transfusion with red blood cells, platelets or plasma [11, 23, 44]. Early placement of a durable intravenous catheter of sufficient diameter to allow blood sampling (e.g., a peripherally inserted central catheter) facilitates safe blood sampling and administration of intravenous fluids or medications [12, 16].
Clinicians should anticipate that a proportion of patients will develop hypoxic or hypercapneic respiratory failure. Etiologies may be diverse, relating to pulmonary vascular leak and inflammatory insults, volume overload, impaired myocardial performance, pulmonary infections, decreased level of consciousness and aspiration, severe metabolic acidosis or hypoventilation syndromes. Over 70% of patients with EVD treated in Europe or USA required some form of respiratory support—invasive or non-invasive mechanical ventilation [12] or supplemental oxygen [1, 9–12, 16, 23].
Neurological complications such as altered mental status (confusion or coma), seizures, agitation and encephalopathy were reported in over 30% of patients treated in Europe, USA and West Africa [12, 30]. Interventions include treating underlying causes such as hypoglycemia, electrolyte imbalance, hepatic dysfunction, uraemia and viral or bacterial encephalitis or meningitis; and use of anticonvulsants, sedatives and antipsychotics as appropriate [11, 12, 14, 15].
Medical treatments
Supportive care remains the cornerstone of patient treatment. A number of direct acting anti-Ebola agents were proposed and evaluated during the West African Ebola outbreak. Most were tested using methodologically weak study designs that precluded determination of efficacy, and await further evaluation [44, 45]. A systematic review [48] evaluated the potential effect of different anti-Ebola therapies on clinical outcomes of EVD patients. All but one of the studies were limited by their non-randomized designs. ZMapp (a cocktail of three monoclonal antibodies), was the only anti-Ebola therapy whose efficacy was tested in a randomized control trial against standard care. In the 72 enrolled patients, mortality in the intervention group was 22% vs 37% in the control group (confidence interval for risk difference, − 36 to 7%). Although ZMapp achieved a 91.2% posterior probability of superiority over standard care, it failed to reach the pre-set threshold of 97.5% probability to establish efficacy.
Health authorities and the WHO R&D blueprint under the ethical framework of monitored emergency use of unregistered and investigational interventions (MEURI) recommended expanded access to investigational therapies, including three monoclonal antibodies (MAb114, ZMapp, and REGN-EB3) and one antiviral agent (remdesivir) in the 2018–2019 DRC Ebola outbreak (Table 4) [44, 49].
Table 4.
Current anti-Ebola therapies being implemented under compassionate use in the DRC
| Drug | Source | Description, mode of action, and strain targeted | Study site, dates, clinical trial registration and status | Study design and primary outcome | Results | Adverse events | General concerns, and comments |
|---|---|---|---|---|---|---|---|
| MAb114 [51] | Developed by VRC, and manufactured at Cook pharmica (Bloomington, IN, USA) |
Human IgG1 monoclonal antibody that targets the Ebola Virus GP Strain targeted: Zaire ebolavirus |
Conducted at the US National Institutes of Health (NI) Bethesda, USA. Enrolled healthy subjects outside an outbreak setting Dose Group 1: Infusion of 5 mg/kg, one dose Group 2: Infusion of 25 mg/kg, one dose Group 3: Infusion of 50 mg/kg, one dose Long term follow-up |
Open label phase 1, dose escalation trial Primary outcome was safety and tolerability Group 1: n = 3 Group 2: n = 5 Group 3: n = 10 |
All mAb114 infusions were safe and well tolerated. A dose-dependent linear pharmacokinetic relationship between dosing groups was noted Half-life was 24.2 days (Standard Error of Mean 0.2) Mean max serum concentration in the 50 mg/kg group: 1961 μg/ml (SD 340) achieved within 2.75 h (SD 1.63) of drug infusion No anti-drug antibody responses were detected |
Only 4 subjects had mild systemic symptoms (malaise, myalgia, joint pains and nausea) |
Tested in a small human population Ease and speed of administration in an outbreak setting; its formulation is a freeze powder that does not require freezer storage Storage in its lyophilized form remains stable at 40 C for up to 6 months Targets highly conserved epitope on the viral glycoprotein with reduced likelihood for mutation |
|
Remdesivir GS-5734 [53] |
Developed by Gilead Sciences (Foster City, CA, USA) |
Small molecule nucleotide prodrug with broad spectrum anti-filovirus activity Strains targeted: Zaire ebolavirus, Sudan ebolavirus, Bundibugyo ebolavirus |
Dosage: in the 1st case report (Adult patient) 150 mg/kg on day 0 and then dose was increased to 225 mg/kg after availability of additional pre-clinical safety data Efficacy was tested in a placebo-controlled randomized trial in Ebola virus infected NHP |
2 Case reports (a 39 year old adult in the UK with a late relapse case of Meningoencephalitis, and in a neonate born to an Ebola positive mother in Guinea) Infected NHP were given IM or IV 3 or 10 mg/kg of the GS-5734 once daily for 12 days or the placebo Primary outcome: Day 28 survival |
In both patients, viremia was suppressed after institution of Remdesivir despite earlier failure with MAbs Treatment initiated on day 3 after detection of systemic viremia, survival rates were 50% for the IM 3 mg/kg group, 100% for the 10 mg/kg group and 0% for the placebo group |
Slightly elevated serum amylase levels No adverse side effects reported |
Promise of efficacy is premised on strong pre-clinical data, anecdotal (2 case reports [54–55]) evidence and robust data from NHP Need for monitoring AST/ALT levels Single high doses offer rapid bioavailability and prolonged higher concentrations that neutralize high Ebola viraemia Effective against persistent virions in immune-protected sites |
| REGN-3470-3471-3479 [50] | Regeneron pharmaceuticals, Illinois, USA |
Cocktail of 3 human monoclonal antibodies targeting non-overlapping epitopes on the Ebola virus Strain targeted Zaire ebolavirus |
Conducted in Evansville IL, USA, Drug given to healthy adult subjects outside an outbreak setting. Study dates May 27, 2016 to April 26, 2017 NCT002777151 Dose; (each of the MAbs given in 1:1:1) Cohort 1: IV 3 mg/kg OR placebo Cohort 2: IV 15 mg/kg OR placebo Cohort 3: IV 60 mg/kg OR placebo Cohort 4: IV 150 mg/kg OR placebo |
Randomized phase 1, placebo-controlled, dose-escalation trial Primary outcome; Incidence and severity of TEAEs up to 169 days Participants were sequentially enrolled to one of the cohorts and randomized to either the placebo or the investigational drug N = 18 participants were assigned to the intervention group and n = 6 were in the placebo |
The antibody pharmacokinetics were linear, corresponding to the different dosage groups Mean half-life was 27.3 days for REGN 3471, 21.7 days, 23. 3 days for REGN 3479 and 21.7 days for REGN-3470 REGN 3470-3471-34479 was well tolerated and did not lead to any immunogenicity Anti-drug antibodies for all the monoclonal antibodies were negative in all patients |
19 emergent treatment adverse effects (all mild to moderate in severity) occurred in the treatment group vs 4 in the placebo group. The most common was headache 33% (in 6 out of the 18 participants, who were all in the treatment group) with a probable linear dose response effect |
Limited human safety data available from the phase 1 study (only 18 participants) Results from animal models were promising Stable for up to 6 months at a temperatures of 25 °C and up to 3 months at temperatures of 45 °C, similar storage conditions of routine vaccines Single daily dosing thus reduced HW time by patient bedside 3 antibodies bind simultaneously to 3 non-overlapping epitopes on the viral GP potentially reducing risk for mutations |
| ZMapp [45, 48] | Collaboration between MappBio, Leafbio (San Diego CA, USA) and Defyrus Inc (Toronto, Canada) |
A cocktail of 3 chimerized monoclonal antibodies (c13C6, 2G4 and 4G7) chosen from ZMab and MB-003 antibody cocktails They target specific surface epitopes on the Ebola virus glycoprotein Strain targeted Ebolavirus/Zaire ebolavirus |
PREVAIL II trial-was based in 4 countries, i.e., Liberia, Sierra Leone, Guinea and USA (11 centers) March-November 2015; study closed before target recruitment due to low patient numbers as countries were nearly declared Ebola free Dosage: IV 50 mg/kg by slow infusion, administered every third day, a total of 3 doses |
An open label, multicenter randomized safety and efficacy controlled trial with an adaptive design to update standard of care with the investigation drug. The standard of care included Favipiravir for the group in Guinea Randomized and analyzed patient data along different strata, i.e., PCR Ct value (≤ 22 vs > 22) and location (Liberia/USA/Sierra Leone vs Guinea) Primary outcome: 28 day mortality |
Fell short of the targeted population size (72 participants vs 200 required) Mortality was lower in patients with a high viral load (i.e., Ct ≤ 22) in the intervention group vs the control group [47% (7/15) vs 60% (9/15)], failing to meet pre-specified posterior probability 28 day mortality [intervention group 22% (8/36) vs control group 37% (13/35)]; although it achieved a 91,2% posterior probability of superiority over standard care, it failed to reach the set threshold of 97.5% probability to establish efficacy |
1 patient developed a serious side effect (hypertension) possibly attributed to ZMapp infusion 8 out of the 93 infusions were stopped due to adverse events (SAEs) 31% (11/36) in the intervention group vs 37% (13/35) in the control group |
The PREVAIL study offered the most robust evidence for efficacy of an Ebola therapy. Although study findings were promising, they were not statistically conclusive on the efficacy of ZMapp Concerns include the complexity of administering the drug; side effects; the need for committed staff to monitor patients; and the cold chain requirement 2 of the antibodies of ZMapp have overlapping epitopes thus competing for same binding site, a possible source of vulnerability in case of mutations |
|
Current trial in the DRC Title: investigational therapies for the treatment of people with Ebola |
Democratic Republic of Congo, patient enrollment started in November 2018 and stopped in August 2019. The first phase was an interventional trial |
A multi-centre, open label randomized controlled trial Primary outcome- 28 day mortality Patients randomized to any one of the 3 drugs (MAb114 + oSOC or Remdesivir + oSOC or REGN-EB3 + oSOC) vs control (ZMapp + oSOC) Number of patients enrolled at 681 expected number is 725 |
Overall 28 day mortality was lower in MAb 114 and REGN-EB3 groups than Remdesivir or ZMapp groups Mortality was lower in patients with low (i.e., PCR Ct-value > 22)-as compared to those with high viral loads (Ct-value ≤ 22) |
Although 29 SAEs were noted by the trial investigators, an independent pharmacovigilance committee attributed only 4 SAEs to the 2 drugs (ZMapp and Remdesivir) The 4 SAEs occurred in 3 patients (2 of whom were from the ZMapp group [1 with worsening gastrointestinal symptoms and the other with pre-infusional hypotension], and 1 patient from the Remdesivir group developed hypotension and subsequent cardiac arrest); all 3 patients died |
After an interim analysis of 499 patients, the independent DSMB on August 9, 2019 recommended that the trial be modified so that all subsequent patients be given either mAb114 or REGN-EB3. This was on the basis that REGN-EB3 had met criterion for early protocol stopping (reached the efficacy boundary for the surrogate end point of the primary outcome including 10-day follow-up outcomes in all patients). Patients receiving either MAb114 or REGN-EB3 had improved survival outcomes than those receiving ZMapp or remedesivir | ||
| MAb114: (NIAID) | See description above | Dosage: 1 dose of 50 mg/kg administered on day 1 as a single infusion |
Overall mortality in the MAb114 group was 35.1% (61/174) vs 49.7% (84/169) in the ZMapp subgroup, p = 0.007 In the MAb114 group, mortality in patients with a Ct-value > 22 was 9.9% (10/101) vs 69.9% (51/73) in patients with a Ct-value ≤ 22 |
||||
|
Remdesivir: GS-5734 (Gilead Sciences) |
See description above | Dosage: IV Loading dose 200 mg for patients > 40 kg and for children < 40 kg 5 mg/kg, then 100 mg in those > 40 kg and 2.5 mg/kg daily for 9–13 days (depending on viral load) |
Overall mortality in the Remdesivir group was 53.1% (93/175) vs 49.7% (84/169) in the ZMapp group In the Remdesivir group, mortality in patients with a Ct-value > 22 was 29.0% (29/100) vs 85.3% (64/75) in patients with a Ct-value ≤ 22 |
||||
|
REGN-EB3 (REGN-3470-3471-3479) (Regeneron Pharmaceuticals) |
See description above | Dosage: 1 dose of 150 mg/kg administered on day 1 as a single infusion |
REGN-EB3 group overall mortality, 33.5% (52/155) vs 51.3% (79/154) in the ZMapp group, p = 0.002 In the REGN-EB3, mortality in patients with a Ct-value > 22 was 11.2% (10/89) vs 63.6% (42/66) in patients with a Ct-value ≤ 22 |
||||
| ZMapp (Mapp Biopharma-ceutical) | See description above | Dosage: 3 doses of 50 mg/kg/body weight administered intravenously every third day |
See comparison to other 3 drugs above In the ZMapp group, mortality in patients with a Ct-value > 22 was 24.5% (24/98) vs 84.5% (60/70) in patients with a Ct-value ≤ 22 |
AST, Aspartate aminotransferase; ALT, alanine aminotransferase; DSMB, data and safety monitoring board; GP, glycoprotein; IM, intramuscular; HW, healthcare worker; Ig G, immunoglobulin G; IV, intravenous; MAbs, monoclonal antibodies; NIAID, National Institute of Allergy and Infectious Diseases; NHP, non-human primates; PCR Ct-value, polymerase chain reaction-Cycle threshold value; oSOC, optimized standard of care; SAEs, Serious Adverse Events; SD, standard deviation; TEAEs, treatment-emergent adverse effects
REGN-EB3 is a cocktail of three monoclonal antibodies (REGN-3470, -3471, and -3479, all humanized from mice) targeting non-overlapping epitopes of Ebola virus. REGN-EB3 previously showed promise in reducing mortality in infected NHPs even when single doses of up to 150 mg/kg were administered 5 days post EVD infection. REGN-EB3 was also found to be well tolerated when it was tested in randomized placebo-controlled phase 1 trial involving 18 human participants [50].
VRC-EBOMAB092-00-AB (MAb114) is a single human monoclonal antibody (isolated from a human survivor) that targets a highly conserved epitope on the receptor-binding domain of the Ebola virus glycoprotein, thus preventing its interaction with the host cell receptor protein and blocking viral entry. In pre-clinical studies involving NHPs challenged with lethal doses of Ebola virus, single 50 mg/kg or 30 mg/kg doses of MAb114 given 5 days after viral exposure offered complete protection. In a subsequent human phase 1, open-label, dose escalation trial, MAb114 was given to 18 participants. MAb114 was well tolerated in all participants, with no reported site infusion reactions or serious adverse effects [51].
In a subsequent randomized controlled trial (n = 681) conducted under challenging field conditions, the safety and effectiveness of three drugs (MAb114, remdesivir, and REGN-EB3) was compared to the control drug ZMapp in Ebola patients receiving optimized standard of care (consisting of intravenous fluid resuscitation, daily clinical laboratory testing, correction of hypoglycemia and other electrolyte abnormalities, and use of broad-spectrum antibiotics and antimalarials, whenever indicated) [49, 52]. The trial was stopped early after MAb114 and REGN-EB3 were found to be superior to ZMapp in reducing 28-day mortality, with event rates of 35.1% for MAb114 [risk difference (RD) vs. ZMapp, − 14.6%, 95% CI − 25.2 to − 1.7%) and 33.5% for REGN-EB3 (RD vs. ZMapp, 17.8%, 95% CI − 28.9 to − 2.9%). Mortality among patients receiving ZMapp was 49.7% and among those receiving remdesivir was 53.1%. However, mortality rates in patients with high viral loads or those with organ dysfunction (high creatinine or transaminases) remained high (> 60% for all interventions), even in a trial setting of optimized standard of care. This observation validates the need for advanced supportive interventions tailored to patient-specific critical care needs whenever feasible, and continued research into refining more effective anti-Ebola therapeutics, either newer drug compounds or combinations of the current ones [52].
Conclusion
Management of patients with Ebola has evolved substantially over the past decade, from a clinical perspective of isolation and provision of oral rehydration therapy to one of treating the syndromic illness, specific patterns of organ dysfunction with oral and intravenous volume repletion, management of life-threatening electrolyte disturbances, support of renal dysfunction with dialysis, cardiovascular dysfunction with intravenous vasoactive medications, and oxygen and mechanical ventilation for respiratory failure. With attention to the provision of care for patients, mortality has dropped from 70 to 40% in resource-challenged Ebola Treatment Centre context and is under 20% in resource-rich environments. With the promise of effective vaccine and specific anti-Ebola virus medications, we now understand that most patients can be safely supported, treated and cured.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Fowler RA, Fletcher T, Fischer WA, Lamontagne F, Jacob S, Brett-Major D, Lawler JV, Jacquerioz FA, Houlihan C, O’Dempsey T, Ferri M. Caring for critically ill patients with Ebola virus disease. Perspectives from West Africa. Am J Respir Crit Care Med. 2014;190(7):733–737. doi: 10.1164/rccm.201408-1514CP. [DOI] [PubMed] [Google Scholar]
- 2.Leligdowicz A, Fischer WA, Uyeki TM, Fletcher TE, Adhikari NK, Portella G, Lamontagne F, Clement C, Jacob ST, Rubinson L, Vanderschuren A. Ebola virus disease and critical illness. Crit Care. 2016;20(1):217. doi: 10.1186/s13054-016-1325-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lamontagne F, Clément C, Fletcher T, Jacob ST, Fischer WA, Fowler RA. Doing today's work superbly well—treating Ebola with current tools. N Engl J Med. 2014;371(17):1565–1566. doi: 10.1056/NEJMp1411310. [DOI] [PubMed] [Google Scholar]
- 4.Malvy D, McElroy AK, de Clerck H, Günther S, van Griensven J. Ebola virus disease. Lancet. 2019;393(10174):936–948. doi: 10.1016/S0140-6736(18)33132-5. [DOI] [PubMed] [Google Scholar]
- 5.Lamontagne F, Clément C, Kojan R, Godin M, Kabuni P, Fowler RA. The evolution of supportive care for Ebola virus disease. Lancet. 2019;393(10172):620–621. doi: 10.1016/S0140-6736(19)30242-9. [DOI] [PubMed] [Google Scholar]
- 6.Rojek A, Horby P, Dunning J. Insights from clinical research completed during the west Africa Ebola virus disease epidemic. Lancet Infect Dis. 2017;17(9):e280–e292. doi: 10.1016/S1473-3099(17)30234-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Beeching NJ, Fenech M, Houlihan CF. Ebola virus disease. BMJ. 2014;349:g7348. doi: 10.1136/bmj.g7348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hunt L, Gupta-Wright A, Simms V, Tamba F, Knott V, Tamba K, Heisenberg-Mansaray S, Tamba E, Sheriff A, Conteh S, Smith T. Clinical presentation, biochemical, and haematological parameters and their association with outcome in patients with Ebola virus disease: an observational cohort study. Lancet Infect Dis. 2015;15(11):1292–1299. doi: 10.1016/S1473-3099(15)00144-9. [DOI] [PubMed] [Google Scholar]
- 9.Lamontagne F, Fowler RA, Adhikari NK, Murthy S, Brett-Major DM, Jacobs M, Uyeki TM, Vallenas C, Norris SL, Fischer WA, 2nd, Fletcher TE. Evidence-based guidelines for supportive care of patients with Ebola virus disease. Lancet. 2018;391(10121):700–708. doi: 10.1016/S0140-6736(17)31795-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Clément C, Adhikari NK, Lamontagne F. Evidence-based clinical management of Ebola virus disease and epidemic viral hemorrhagic fevers. Infect Dis Clin. 2019;33(1):247–264. doi: 10.1016/j.idc.2018.10.013. [DOI] [PubMed] [Google Scholar]
- 11.World Health Organization (2019) Optimized supportive care for ebola virus disease: clinical management standard operating procedures. https://apps.who.int/iris/bitstream/handle/10665/325000/9789241515894-eng.pdf?sequence=1. Accessed 17 Nov 2019
- 12.Uyeki TM, Mehta AK, Davey RT, Jr, Liddell AM, Wolf T, Vetter P, Schmiedel S, Grünewald T, Jacobs M, Arribas JR, Evans L. Clinical management of Ebola virus disease in the United States and Europe. N Engl J Med. 2016;374(7):636–646. doi: 10.1056/NEJMoa1504874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Centers for Disease Prevention and Control: Ebola (Ebola Virus Disease). Outbreaks. https://www.cdc.gov/vhf/ebola/outbreaks/index-2018.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fvhf%2Febola%2Foutbreaks%2Findex.html. Accessed 17 Nov 2019
- 14.World Health Organization. Clinical management of patients with viral hemorrhagic fever: a pocket guide for front-line health workers: interim emergency guidance for country adaptation. https://apps.who.int/iris/bitstream/handle/10665/205570/?sequence=1. Accessed 17 Nov 2019
- 15.National Institute of Allergy and Infectious Diseases (2019) Independent monitoring board recommends early termination of Ebola therapeutics trial in DRC because of favorable results with two of four candidates. https://www.niaid.nih.gov/news-events/independent-monitoring-board-recommends-early-termination-ebola-therapeutics-trial-drc. Accessed 17 Nov 2019
- 16.Dickson SJ, Clay KA, Adam M, Ardley C, Bailey MS, Burns DS, Cox AT, Craig DG, Espina M, Ewington I, Fitchett G. Enhanced case management can be delivered for patients with EVD in Africa: experience from a UK military Ebola treatment centre in Sierra Leone. J Infect. 2018;76(4):383–392. doi: 10.1016/j.jinf.2017.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.World Health Organization (2019). Ebola in the DRC. Health Emergency update. https://www.who.int/emergencies/diseases/ebola/drc-2019. Accessed 04 Jan 2020
- 18.Ilunga Kalenga O, Moeti M, Sparrow A, Nguyen VK, Lucey D, Ghebreyesus TA. The ongoing Ebola epidemic in the Democratic Republic of Congo, 2018–2019. N Engl J Med. 2019;381:373–383. doi: 10.1056/NEJMsr1904253. [DOI] [PubMed] [Google Scholar]
- 19.World Health Organization. Ebola virus disease. Key facts. https://www.who.int/news-room/fact-sheets/detail/ebola-virus-disease. Accessed 17 Nov 2019
- 20.Kuhn JH, Adachi T, Adhikari NK, Arribas JR, Bah IE, Bausch DG, Bhadelia N, Borchert M, Brantsæter AB, Brett-Major DM, Burgess TH. New filovirus disease classification and nomenclature. Nat Rev Microbiol. 2019;5:261. doi: 10.1038/s41579-019-0187-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.World Health Organization. Ebola situation reports. https://www.who.int/publications-detail/ebola-virus-disease-democratic-republic-of-congo-external-situation-report-67-2019. Accessed 17 Nov 2019
- 22.Glynn JR. Age-specific incidence of Ebola virus disease. Lancet. 2015;386(9992):432. doi: 10.1016/S0140-6736(15)61446-5. [DOI] [PubMed] [Google Scholar]
- 23.Baseler L, Chertow DS, Johnson KM, Feldmann H, Morens DM. The pathogenesis of Ebola virus disease. Annu Rev Pathol. 2017;12:387–418. doi: 10.1146/annurev-pathol-052016-100506. [DOI] [PubMed] [Google Scholar]
- 24.Diallo B, Sissoko D, Loman NJ, Bah HA, Bah H, Worrell MC, Conde LS, Sacko R, Mesfin S, Loua A, Kalonda JK. Resurgence of Ebola virus disease in Guinea linked to a survivor with virus persistence in seminal fluid for more than 500 days. Clin Infect Dis. 2016;63(10):1353–1356. doi: 10.1093/cid/ciw601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Munoz-Fontela C, McElroy AK. Ebola virus disease in humans: pathophysiology and immunity. Curr Top Microbiol Immunol. 2017;411:141–169. doi: 10.1007/82_2017_11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.McElroy AK, Mühlberger E, Munoz-Fontela C. Immune barriers of Ebola virus infection. Curr Opin Virol. 2018;28:152–160. doi: 10.1016/j.coviro.2018.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Feldmann H, Bugany H, Mahner F, Klenk HD, Drenckhahn D, Schnittler HJ. Filovirus-induced endothelial leakage triggered by infected monocytes/macrophages. J Virol. 1996;70(4):2208–2214. doi: 10.1128/jvi.70.4.2208-2214.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.World Health Organization (2017) Ebola key technical documents. https://www.who.int/ebola/22-5-17-Ebola-Key-technical-documents-EN.pdf?ua=1. Accessed 17 Nov 2019
- 29.Eichner M, Dowell SF, Firese N. Incubation period of Ebola hemorrhagic virus subtype Zaire. Osong Public Health Res Perspect. 2011;2(1):3–7. doi: 10.1016/j.phrp.2011.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rojek AM, Salam A, Ragotte RJ, Liddiard E, Elhussain A, Carlqvist A, Butler M, Kayem N, Castle L, Lang’o O, Stepniewska K. A systematic review and meta-analysis of patient data from the west Africa (2013–16) Ebola virus disease epidemic. Clin Microbiol Infect. 2019;25(11):1307–1314. doi: 10.1016/j.cmi.2019.06.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lado M, Walker NF, Baker P, Haroon S, Brown CS, Youkee D, Studd N, Kessete Q, Maini R, Boyles T, Hanciles E. Clinical features of patients isolated for suspected Ebola virus disease at Connaught Hospital, Freetown, Sierra Leone: a retrospective cohort study. Lancet Infect Dis. 2015;15(9):1024–1033. doi: 10.1016/S1473-3099(15)00137-1. [DOI] [PubMed] [Google Scholar]
- 32.Chertow DS, Kleine C, Edwards JK, Scaini R, Giuliani R, Sprecher A. Ebola virus disease in West Africa—clinical manifestations and management. N Engl J Med. 2014;371(22):2054–2057. doi: 10.1056/NEJMp1413084. [DOI] [PubMed] [Google Scholar]
- 33.Khalafallah MT, Aboshady OA, Moawed SA, Ramadan MS. Ebola virus disease: essential clinical knowledge. Avicenna J Med. 2017;7(3):96. doi: 10.4103/ajm.AJM_150_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.World Health Organization (2015) Interim Guidance on the use of rapid antigen detection tests. https://apps.who.int/iris/bitstream/handle/10665/160265/WHO_EVD_HIS_EMP_15.1_eng.pdf?sequence=1. Accessed 17 Nov 2019
- 35.Tembo J, Simulundu E, Changula K, Handley D, Gilbert M, Chilufya M, Asogun D, Ansumana R, Kapata N, Ntoumi F, Ippolito G. Recent advances in the development and evaluation of molecular diagnostics for Ebola virus disease. Expert Rev Mol Diagn. 2019;19(4):325–340. doi: 10.1080/14737159.2019.1595592. [DOI] [PubMed] [Google Scholar]
- 36.World Health Organization (2015) Selection and use of Ebola in vitro diagnostic assays. Emergency guidance. https://apps.who.int/iris/bitstream/handle/10665/175554/WHO_EVD_HIS_EMP_15.2_annex_eng.pdf?sequence=2. Accessed 17 Nov 2019
- 37.Gallais F, Gay-Andrieu F, Picot V, Magassouba N, Mély S, Peyrefitte CN, Bellanger L. Field assessment of the new rapid diagnostic test Ebola eZYSCREEN®. Bull Soc Pathol Exot (1990) 2017;110(1):38–48. doi: 10.1007/s13149-016-0540-z. [DOI] [PubMed] [Google Scholar]
- 38.Walker NF, Brown CS, Youkee D, Baker P, Williams N, Kalawa A, Russell K, Samba AF, Bentley N, Koroma F, King MB. Evaluation of a point-of-care blood test for identification of Ebola virus disease at Ebola holding units, Western Area, Sierra Leone, January to February 2015. Eurosurveillance. 2015;20(12):21073. doi: 10.2807/1560-7917.ES2015.20.12.21073. [DOI] [PubMed] [Google Scholar]
- 39.Engwa GA. Ebola virus disease: progress so far in the management of the disease. Curr Top Trop Emerg Dis Travel Med. 2018;19:129. [Google Scholar]
- 40.Makiala S, Mukadi D, De Weggheleire A, Muramatsu S, Kato D, Inano K, Gondaira F, Kajihara M, Yoshida R, Changula K, Mweene A. Clinical evaluation of QuickNaviTM-Ebola in the 2018 outbreak of Ebola virus disease in the Democratic Republic of the Congo. Viruses. 2019;11(7):589. doi: 10.3390/v11070589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kiiza P, Adhikari NK, Mullin S, Teo K, Fowler RA. Principles and practices of establishing a hospital-based Ebola treatment unit. Crit Care Clin. 2019;35(4):697–710. doi: 10.1016/j.ccc.2019.06.011. [DOI] [PubMed] [Google Scholar]
- 42.Henao-Restrepo AM, Camacho A, Longini IM, Watson CH, Edmunds WJ, Egger M, Carroll MW, Dean NE, Diatta I, Doumbia M, Draguez B. Efficacy and effectiveness of an rVSV-vectored vaccine in preventing Ebola virus disease: final results from the Guinea ring vaccination, open-label, cluster-randomised trial (Ebola Ça Suffit!) Lancet. 2017;389(10068):505–518. doi: 10.1016/S0140-6736(16)32621-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.World Health Organization (2019) Vaccination. Preliminary results on the efficacy of rVSV-ZEBOV-GP Ebola vaccine using the ring vaccination strategy in the control of an Ebola outbreak in the Democratic Republic of the Congo: an example of integration of research into epidemic response. https://www.who.int/csr/resources/publications/ebola/ebola-ring-vaccination-results-12-april-2019.pdf?ua=1. Accessed 17 Nov 2019
- 44.Damon IK, Rollin PE, Choi MJ, Arthur RR, Redfield RR. New tools in the Ebola arsenal. N Engl J Med. 2018;379(21):1981–1983. doi: 10.1056/NEJMp1811751. [DOI] [PubMed] [Google Scholar]
- 45.Prevail II Writing Group A randomized, controlled trial of ZMapp for Ebola virus infection. New Engl J Med. 2016;375(15):1448–56. doi: 10.1056/NEJMoa1604330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ansumana R, Jacobsen KH, Idris MB, Bangura H, Boie-Jalloh M, Lamin JM, Sesay S, Sahr F. Ebola in Freetown area, Sierra Leone—a case study of 581 patients. N Engl J Med. 2015;372(6):587–588. doi: 10.1056/NEJMc1413685. [DOI] [PubMed] [Google Scholar]
- 47.Connor MJ, Kraft C, Mehta AK, Varkey JB, Lyon GM, Crozier I, Ströher U, Ribner BS, Franch HA. Successful delivery of RRT in Ebola virus disease. J Am Soc Nephrol. 2015;26(1):31–37. doi: 10.1681/ASN.2014111057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lee JS, Adhikari NK, Kwon HY, Teo K, Siemieniuk R, Lamontagne F, Chan A, Mishra S, Murthy S, Kiiza P, Hajek J, et al. Anti-Ebola therapy for patients with Ebola virus disease: a systematic review. BMC Infect Dis. 2019;19(1):376. doi: 10.1186/s12879-019-3980-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.World Health Organization (2018) WHO R&D Blueprint—Ad-hoc Expert Consultation on clinical trials for Ebola Therapeutics. https://www.who.int/ebola/drc-2018/treatments-approved-for-compassionate-use-update/en/. Accessed 17 Nov 2019
- 50.Sivapalasingam S, Kamal M, Slim R, Hosain R, Shao W, Stoltz R, Yen J, Pologe LG, Cao Y, Partridge M, Sumner G. Safety, pharmacokinetics, and immunogenicity of a co-formulated cocktail of three human monoclonal antibodies targeting Ebola virus glycoprotein in healthy adults: a randomized, first-in-human phase 1 study. Lancet Infect Dis. 2018;18(8):884–893. doi: 10.1016/S1473-3099(18)30397-9. [DOI] [PubMed] [Google Scholar]
- 51.Gaudinski MR, Coates EE, Novik L, Widge A, Houser KV, Burch E, Holman LA, Gordon IJ, Chen GL, Carter C, Nason M. Safety, tolerability, pharmacokinetics, and immunogenicity of the therapeutic monoclonal antibody mAb114 targeting Ebola virus glycoprotein (VRC 608): an open-label phase 1 study. Lancet. 2019;393(10174):889–898. doi: 10.1016/S0140-6736(19)30036-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Mulangu S, Dodd LE, Davey RT, Jr, Tshiani Mbaya O, Proschan M, Mukadi D, Lusakibanza Manzo M, Nzolo D, Tshomba Oloma A, Ibanda A, Ali R. A randomized, controlled trial of Ebola virus disease therapeutics. N Engl J Med. 2019;381(24):2293–2303. doi: 10.1056/NEJMoa1910993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Warren T, Jordan R, Lo M, Soloveva V, Ray A, Bannister R, Mackman R, Perron M, Stray K, Feng J, Xu Y (2015) Nucleotide prodrug GS-5734 is a broad-spectrum filovirus inhibitor that provides complete therapeutic protection against the development of Ebola virus disease (EVD) in infected non-human primates. Infectious Diseases Society of America Open Forum Infectious Diseases 2(1) LB-2. https://academic.oup.com/ofid/article/2/suppl_1/LB-2/2633814. Accessed 17 Nov 2019
- 54.Jacobs M, Rodger A, Bell DJ, Bhagani S, Cropley I, Filipe A, Gifford RJ, Hopkins S, Hughes J, Jabeen F, Johannessen I. Late Ebola virus relapse causing meningoencephalitis: a case report. Lancet. 2016;388(10043):498–503. doi: 10.1016/S0140-6736(16)30386-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Dörnemann J, Burzio C, Ronsse A, Sprecher A, De Clerck H, Van Herp M, Kolié MC, Yosifiva V, Caluwaerts S, McElroy AK, Antierens A. First newborn baby to receive experimental therapies survives Ebola virus disease. J Infect Dis. 2017;215(2):171–174. doi: 10.1093/infdis/jiw493. [DOI] [PMC free article] [PubMed] [Google Scholar]