Abstract
Influenza A virus (IAV) infection is perennially one of the leading causes of death worldwide. Effective therapy and vaccination are needed to control viral expansion. However, current anti-IAV drugs risk inducing drug-resistant virus emergence. Although intranasal administration of whole inactivated virus vaccine can induce efficient protective immunity, formalin and β-propiolactone are the currently used and harmful inactivating agents. Here, we analyzed the antiviral activity of hibiscus (Hibiscus sabdariffa L.) tea extract against human IAV and evaluated its potential as a novel anti-IAV drug and a safe inactivating agent for whole inactivated vaccine. The in vitro study revealed that the pH of hibiscus tea extract is acidic, and its rapid and potent antiviral activity relied largely on the acidic pH. Furthermore, the mouse study showed that the acidic extract was not effective for either therapeutic or vaccination purposes. However, hibiscus tea extract and protocatechuic acid, one of the major components of the extract, showed not only potent acid-dependent antiviral activity but also weak low-pH-independent activity. The low-pH-independent activity did not affect the conformation of immunodominant hemagglutinin protein. Although this low-pH-independent activity is very limited, it may be suitable for the application to medication and vaccination because this activity is not affected by the neutral blood environment and does not lose antigenicity of hemagglutinin. Further study of the low-pH-independent antiviral mechanism and attempts to enhance the antiviral activity may establish a novel anti-IAV therapy and vaccination strategy.
Keywords: Acidic pH, Anti-human influenza A viral activity, Antiviral drug therapy, Hibiscus tea extract, Natural substance, Whole inactivated vaccine
Introduction
Outbreaks of seasonal influenza caused by influenza A virus (IAV) or B virus occur all over the world and lead to approximately 3 to 5 million severe cases and 290,000 to 650,000 deaths every year (World Health Organization 2018). Moreover, novel IAV strains have emerged repeatedly in the past, leading to a large number of deaths by pandemics in the twentieth century (Frederick 1958; Saunders and Krewski 2016; World Health Organization 2009). To combat IAV, there is a relentless need to develop more effective antiviral therapies and vaccine strategies.
The calyces of Hibiscus sabdariffa L. is an ingredient in hibiscus tea. Hibiscus tea contains abundant bioactive compounds including anthocyanins, polyphenols, organic acids, and flavonoids (Da-Costa-Rocha et al. 2014). Earlier reports have shown various biological and pharmacological activities of hibiscus tea and hibiscus tea-derived compounds like anti-inflammatory, anti-oxidant, anti-cholesterol, anti-hypertensive, anti-bacterial, and antiviral activities (D’Souza et al. 2016; Da-Costa-Rocha et al. 2014; Hassan et al. 2017; Joshi et al. 2015). We previously reported the antiviral activity of hibiscus tea extract against H5 subtype avian influenza viruses (Baatartsogt et al. 2016). Hibiscus tea extract inactivated the avian influenza viruses, showing activity in 10 min. Based on these results, we hypothesized that these potent and rapid antiviral activities could be used to develop a novel anti-IAV drug and a safe inactivating agent for the whole inactivated vaccine.
A number of anti-IAV drugs have been in practice or under development (Li et al. 2015; Noshi et al. 2018). Although these drugs elicit the therapeutic effect, viral strains resistant against these drugs tend to appear after prolonged use (Hurt 2014; Li et al. 2015), and some of these drugs also exhibit adverse effects. RNA polymerase inhibitor favipiravir showed teratogenicity and embryotoxicity in animal experiments. Therefore, its clinical use is strictly restricted in Japan (Nagata et al. 2015). In addition, the possibility of insufficient existing drug supply is a concern during pandemics. Finding novel anti-IAV substances would be a promising approach for treatment to deal with these problems.
Vaccination is also important to control IAV expansion. One of the effective vaccination strategies is intranasal (i.n.) administration of whole inactivated vaccine. Whole inactivated vaccine can prime potent protective immunity because it preserves virus particle integrity (Soema et al. 2015). In addition, i.n. vaccination can induce systemic IgG production and mucosal IgA production, the latter of which is indispensable to prevent viral infection establishment (Rose et al. 2012; Tamura et al. 2016). Formalin and β-propiolactone (β-PL) are widely used as inactivating agents for whole-virus vaccines (Delrue et al. 2012). However, removal of these substances is essential due to their chemical toxicity, making the procedure time consuming and expensive. Finding a safe anti-IAV compound that can be used as a novel inactivating agent for a whole-virus vaccine may solve critical problems.
Here, we attempted to clarify the mechanism of antiviral activity against H1N1 human IAV strain A/Puerto Rico/8/1934 (PR8 virus) and identify the antiviral compounds present in hibiscus tea extract. We focused on three compounds including protocatechuic acid (PCA), ferulic acid, and (2S,3R)-tetrahydro-3-hydroxy-5-oxo-2,3-furandicarboxylic acid (hibiscus acid). PCA and ferulic acid showed antiviral activity against several virus species (Hassan et al. 2017; Joshi et al. 2015). Hibiscus acid is one of the major organic acids in hibiscus calyces (Da-Costa-Rocha et al. 2014). In addition, we assessed hibiscus tea extract’s potential as a candidate for novel anti-IAV drug and as an inactivating agent for whole-virus vaccines.
Materials and Methods
Viruses, Cells, and Mice
PR8 virus (ATCC® Catalog No. VR-95™) was purchased from ATCC (Manassas, VA), and propagated in the allantoic fluid of 10-day-old embryonated chicken eggs. Madin-Darby canine kidney (MDCK) cells were kindly provided by Dr. H. Nagano (Hokkaido Institute of Public Health, Sapporo, Japan). For passaging, MDCK cells were cultured in Dulbecco’s modified Eagle’s minimal essential medium (DMEM) (Nissui Pharmaceutical Co., Ltd., Tokyo, Japan) supplemented with 10% fetal bovine serum, 20 μM l-glutamine (Wako Pure Chemical Industries, Ltd., Osaka, Japan), 0.15% NaHCO3 (Wako Pure Chemical Industries, Ltd.), 2 µg/ml amphotericin B (Bristol-Myers Squibb Co., New York, NY), and 100 µg/ml kanamycin (Meiji Seika Pharma Co., Ltd., Tokyo, Japan). After viral inoculation, MDCK cells were cultured in viral growth medium composed of DMEM supplemented with 0.2% bovine serum albumin (Wako Pure Chemical Industries, Ltd.), 0.01% glucose (Wako Pure Chemical Industries, Ltd.), 0.15% NaHCO3, 2.5 mM HEPES (Wako Pure Chemical Industries, Ltd.), and 0.0006% trypsin (Wako Pure Chemical Industries, Ltd.). Ten to 13-week-old female BALB/c mice were purchased from CLEA Japan Inc. (Tokyo, Japan) or generated in our laboratory. All animal experiments were approved by the Institutional Animal Care and Use Committee of Obihiro University of Agriculture and Veterinary Medicine and performed in compliance with institutional guidelines.
Reagents
PCA was purchased from Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan). Ferulic acid was purchased from Cayman Chemical Co. (An Arbor, MI). Formalin and β-PL were purchased from Wako Pure Chemical Industries, Ltd. Hibiscus tea (Roselle) was purchased from Nakazen Corp. (Nanjo, Japan). To prepare acidic crude hibiscus tea extract (Hib[crude]), 2 g of hibiscus tea powder was boiled in 100 ml ultrapure water until the volume reached 50 ml. To isolate the small molecular weight (< 3 kDa) fraction (frHibis), Hib[crude] was fractionated using Vivaspin® 6 (3000 MWCO PES) (Sartorius Ag., Göttingen, Germany) (Baatartsogt et al. 2016).
Isolation of Hibiscus Acid
Hibiscus acid was isolated as previously described (Ibnusaud et al. 2002; Ibrahim et al. 2000). In brief, hibiscus tea powder was soaked in a sufficient volume of ultrapure water at 24 °C for 24 h, and this process was repeated several times. The combined extract was concentrated under reduced pressure to obtain a viscous syrup. Methanol was added to the syrup to precipitate insoluble materials. The precipitate was removed by filtering, and the filtrate was concentrated to obtain a second viscous syrup that was extracted several times with acetone. Acetone was removed under reduced pressure, and the extract was further extracted several times with ether. The ether was removed under reduced pressure, and a crude acid was obtained which was recrystallized (ether + chloroform) to obtain pure hibiscus acid (molecular weight: 190.1, 0.1 g, 1%) in the form of colorless crystal. The structure and purity of isolated hibiscus acid was confirmed using previously reported spectroscopic and other physical analyses (Ibnusaud et al. 2002; Ibrahim et al. 2000).
Synthesis of Disodium (2S,3R)-Tetrahydro-3-hydroxy-5-oxo-2,3-furandicarboxylate (Hibiscus Acid-Na)
Hibiscus acid-Na was synthesized as previously described (Ibnusaud et al. 2002; Ibrahim et al. 2000). In brief, saturated aqueous sodium bicarbonate was added dropwise into an aqueous hibiscus acid solution (0.1 g, 0.53 mmol, in 1 ml water) at 0 °C until the pH of the solution was neutral. After removing water under reduced pressure, the residue was triturated and washed using dry acetone. The product was finally dried under reduced pressure to obtain a colorless solid (molecular weight: 234.1, 0.1 g, 81%).
Virus Treatment and Inoculation into MDCK Cells
105 50% tissue culture infective dose (TCID50)/ml of PR8 virus propagated in allantoic fluid was mixed with an equal amount of phosphate-buffered saline (PBS), Hib[crude], frHibis, PCA, ferulic acid, hibiscus acid, or hibiscus acid-Na. The effective concentrations of PCA, ferulic acid, hibiscus acid, and hibiscus acid-Na in the mixture were 2.5 mg/ml, 1 mg/ml, 2.5 mg/ml, and 2.5 mg/ml, respectively. pH of the mixture was adjusted within a range of acidic to around neutral by adding HCl (Wako Pure Chemical Industries, Ltd.) or NaOH (Wako Pure Chemical Industries, Ltd.). pH of the mixture was determined by measuring the pH of a virus-free mixture as a substitute for the virus-containing mixture. The mixture was placed at 26 °C for 10 min, 24 h, or 48 h. After each reaction time, the mixture was inoculated into MDCK cells and a tenfold serial dilution was performed. Three days after inoculation, the viral titer was evaluated by observing the cytopathic effect (CPE) on MDCK cells. We calculated log10 TCID50/ml using the Behrens-Kärber method (Kärber 1931).
Hemagglutination (HA) Test
PR8 virus propagated in allantoic fluid was mixed with an equal amount of neutral and acidic pH PBS, Hib[crude], frHibis, or PCA. The effective concentration of PCA in the mixture was 2.5 mg/ml. The mixture was placed at 26 °C for 10 min or 48 h. A twofold serial dilution was performed after each reaction time. An HA test was conducted using 0.5% chicken blood cells (Nippon Bio-Test Laboratories Inc., Asaka, Japan) according to the WHO Manual on Animal Influenza Diagnosis and Surveillance (WHO Manual, World Health Organization 2002). The HA test was also conducted with formalin-, β-PL-, or acidic Hib[crude]-inactivated purified PR8 virus.
Evaluation of Anti-PR8 Virus Activity of Acidic Hib[crude] in Mice
Mice were intranasally given 101 TCID50/50 μl PR8 virus propagated in allantoic fluid. Eight hours after the viral inoculation, 200 μl of neutral PBS or acidic Hib[crude] was administered once orally (p.o.) (day 0). From day 1 to 4, PBS or Hib[crude] was administered p.o. twice a day. Each group had 5 mice. The change in the body weight of each mouse was monitored daily. Mice were euthanized when the body weight decreased to 75% or 2 weeks after the viral inoculation. To determine the lung viral titer, mice were euthanized at days 3 and 5 and lungs were harvested from the euthanized mice. The lungs were homogenized in 20% (w/v) PBS supplemented with 5 μg/ml amphotericin B, 1 mg/ml kanamycin, and 100 μg/ml gentamicin (MSD K.K., Tokyo, Japan). The lung homogenate supernatant was added to MDCK cells, and a tenfold serial dilution was performed. Three days after inoculation, the viral titer (log10 TCID50/g tissue) was evaluated.
Evaluation of Acidic Hib[crude]-Inactivated Vaccine Efficacy
To prepare formalin-inactivated whole-virus vaccine, 1 mg/ml of purified PR8 virus was mixed with 0.1% formalin and kept at 4 °C for 48 h. After viral inactivation, 0.6% Na2S2O4 (Wako Pure Chemical Industries, Ltd.) was added and the mixture was placed at 4 °C overnight. Similarly, to prepare β-PL-inactivated whole-virus vaccine, 1 mg/ml of purified PR8 virus was mixed with 0.1% β-PL and placed at 4 °C for 48 h. Afterward, formalin and β-PL were removed using membrane dialysis with Spectra/Por® (8,000 MWCO) (Spectrum Laboratories Inc., Los Angeles, CA). The concentration of formalin- or β-PL-inactivated PR8 virus was measured using the Micro BCA protein assay kit (Pierce, Rockford, IL) and adjusted to 300 μg/ml. To prepare acidic Hib[crude]-inactivated whole-virus vaccine, 600 μg/ml of purified PR8 virus was mixed with an equal amount of acidic Hib[crude] and placed at 26 °C for 10 min. The inactivated virus was directly used to vaccinate mice. Viral inactivation was confirmed based on the absence of viral infectivity by egg inoculation followed by an HA test. 50 μl PBS, formalin-, β-PL-, or acidic Hib[crude]-inactivated PR8 virus vaccine was intranasally administered in mice (first vaccination) under light anesthesia with isolflurane (Intervet K.K., Tokyo, Japan). The second vaccination was performed 2 weeks after the first vaccination. There were 5 mice in each group. Six weeks after the second vaccination, the mice were intranasally inoculated with 102 TCID50/50 μl of PR8 virus propagated in allantoic fluid (day 0). Changes in body weight were monitored daily after PR8 viral inoculation. The mice were euthanized when the body weight decreased 75%. All surviving mice were euthanized on day 14. Blood samples were collected on day 0 (pre-inoculation) and day 14 (post-inoculation) to evaluate anti-PR8 virus antibody (Ab) titer using the hemagglutination inhibition (HI) test. The obtained serum samples were treated with receptor-destroying enzyme (Denka Seiken Co., Ltd., Japan) to remove non-specific HA inhibitors. The serum was mixed with chicken blood cells (the final concentration was 10%), and the mixture was rotated at room temperature for 1 h to remove any non-specific HA factors. The supernatant was collected after centrifugation, and the HI test was conducted in the presence of PR8 viral antigen according to the WHO manual. The serum anti-PR8 virus Ab titer was determined as log2 HI titer.
Statistical Analysis
P values were calculated using the following statistical analyses. Student’s t test or Student’s t test with Bonferroni’s test were performed for the viral titer. Student’s t test, Student’s t test with Bonferroni’s test, or Kluskal-Wallis test with Dunn’s multiple comparison were performed for the HA titer and HI titer. One-way analysis of variance (ANOVA) with Tukey’s multiple comparison test or Kluskal-Wallis test with Dunn’s multiple comparison test was performed for changes in body weight. Log-rank test with Bonferroni’s test was performed for the survival rate.
Results
Evaluation of Relationship Between Hibiscus Tea Extract Anti-PR8 Virus Activity and Acidic pH
Since Hib[crude] is acidic, we assessed whether the anti-PR8 virus activity of Hib[crude] depends on its acidic pH. PR8 virus was mixed with non-pH-adjusted and pH-adjusted PBS or Hib[crude] (pH of the mixture ranged from 3.0 to 7.2), and the titer of the treated virus was determined. The pH of the mixture containing non-pH-adjusted Hib[crude] was around 3.0. After a 10-min reaction, Hib[crude] and PBS of pH ≤ 4.0 strongly inactivated the PR8 virus, but the effect of Hib[crude] at pH 4.0 seemed to be slightly higher than PBS at pH 4.0. On the other hand, both PBS and Hib[crude] of pH ≥ 5.0 did not inactivate PR8 virus. After a 24-h reaction, PR8 virus was inactivated using both PBS and Hib[crude] at pH ≤ 5. On the other hand, the viral titer of Hib[crude]-treated PR8 virus was 101-times lower than PBS-treated PR8 virus under the conditions of pH ≥ 6.0 (Fig. 1a). The differences were statistically significant (p < 0.05 or 0.01). We also evaluated viral titers of acidic and neutral (pH-adjusted) frHibis-treated PR8 virus. The pH of the mixture containing non-pH-adjusted frHibis was around 3.0. Acidic frHibis inactivated PR8 virus after a 10-min reaction, but neutral frHibis did not. After a 48-h reaction, the viral titer of neutral frHibis-treated PR8 virus was consistent with neutral Hib[crude]-treated PR8 virus and 102-times lower (p < 0.01) than PBS-treated PR8 virus (Fig. 1b). These results show that frHibis has two different anti-PR8 virus activities: one is a rapid and potent activity largely dependent on acid, and the other is a slow and weak low-pH-independent activity.
Fig. 1.
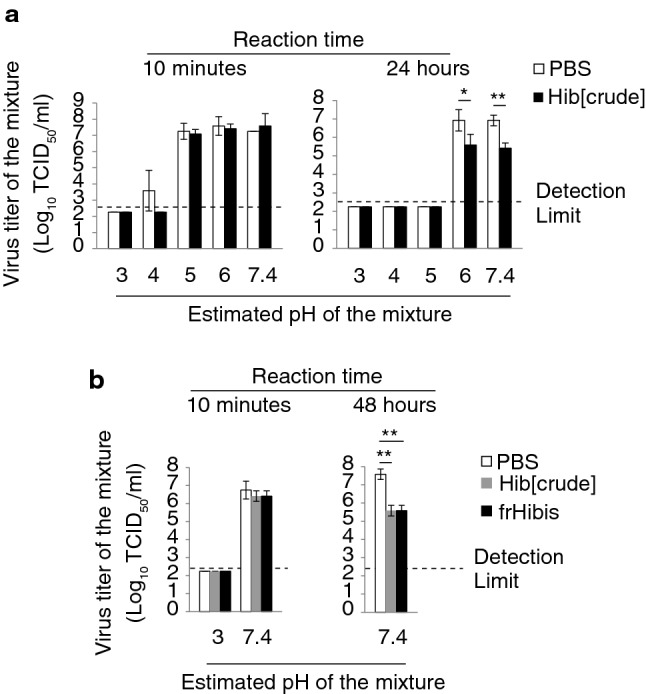
Hibiscus tea components show both acid-dependent antiviral activity and low-pH-independent antiviral activity against PR8 virus. a PR8 virus was mixed with an equal volume of PBS or Hib[crude]. The pH of PBS and Hib[crude] was adjusted from acidic to approximately neutral. The pH shown was an approximate value with a range ± 0.3. The mixture which was placed at 26 °C for 10 min or 24 h and inoculated in MDCK cells. After a 3-day incubation, the viral titer of the mixture was evaluated using CPE observation. b The mixture containing acidic and neutral PBS, Hib[crude], or frHibis was placed at 26 °C for 10 min or 48 h. The mixture was inoculated to MDCK cells. After a 3-day incubation, the viral titer of the mixture was evaluated. Error bars indicate mean ± SD; n = 3 per group. (a, b) Student’s t test (a), and Student’s t test with Bonferroni’s test (b) were performed to analyze statistical significance; *p < 0.05, **p < 0.01. The results represent three independent experiments
Evaluation of Anti-PR8 Virus Activity of frHibis Compounds
Next, we evaluated the anti-PR8 virus activity of PCA, ferulic acid, and hibiscus acid. These compounds are acidic in solution. PR8 virus was treated with PBS or each of these solutions, and the titer of the virus was measured. Both acidic (non-pH-adjusted) and neutral (pH-adjusted) solutions were tested to correlate between acidic pH of the solutions and the anti-PR8 virus activity. The estimated pH values of the mixtures containing acidic PCA, ferulic acid, and hibiscus acid were approximately 5.0, 6.5, and 3.0, respectively. The mixtures containing acidic PBS (pH 5.0, 6.5, and 3.0) were also tested as acidic solution controls. After a 10-min reaction, the titers of acidic and neutral PCA-treated PR8 virus were similar to a PBS-treated PR8 virus. Acidic PBS and PCA completely inactivated PR8 virus after a 48-h reaction. On the other hand, the viral titer of PCA-treated PR8 virus was 101.5 -times lower (p < 0.01) than PBS-treated PR8 virus under neutral conditions (Fig. 2a). Neither a 10-min nor 48-h reaction in acidic or neutral conditions showed anti-PR8 viral activity for ferulic acid (Fig. 2b). Finally, anti-PR8 viral activity of hibiscus acid was analyzed. Hibiscus acid-Na is a disodium salt of hibiscus acid, and its pH was approximately neutral. Acidic hibiscus acid inactivated PR8 virus after a 10-min or 48-h reaction, but neutral hibiscus acid and hibiscus acid-Na did not do so (Fig. 2c). These results show that PCA has weak low-pH-independent anti-PR8 virus activity.
Fig. 2.
Neutral PCA shows antiviral activity against PR8 virus. a–c PR8 virus was mixed with equal volume of PBS, PCA (a), ferulic acid (b), hibiscus acid, or hibiscus acid-Na (c). Acidic (non-pH-adjusted) PCA, ferulic acid, and hibiscus acid, and neutral (pH-adjusted) solutions of these compounds were prepared. Acidic PBS was also prepared. An estimated pH of the mixture was determined using pH measurement of a virus-free mixture as a substitute for a virus-containing mixture. The pH shown was an approximate value with a range ± 0.3. The mixture was placed at 26 °C for 10 min or 48 h and was inoculated into MDCK cells. After a 3-day incubation, the viral titer of the mixture was evaluated using CPE observation. Error bars indicate mean ± SD; n = 3 per group. (a–c) Student’s t test (a, b), and Student’s t test with Bonferroni’s test (c) were performed to analyze statistical significance; **p < 0.01. The results represent more than three independent experiments
Evaluation of Hibiscus Tea Extract Effect on HA Activity
Next, we assessed whether acidic and neutral hibiscus tea extracts affect the HA activity of PR8 virus. PR8 virus was mixed with acidic or neutral PBS, Hib[crude], or frHibis. HA titer of PR8 virus was measured after a 10-min or 48-h reaction. Acidic PBS-, Hib[crude]-, and frHibis-treated PR8 virus lost HA activity in 10 min. It was confirmed earlier that the acidic Hib[crude] did not directly affect chicken blood cell agglutination (data not shown). After a 48-h reaction, the HA titer of neutral Hib[crude]- and frHibis-treated PR8 virus did not decrease compared to PBS-treated PR8 virus (Fig. 3a). HA titers of acidic and neutral PCA-treated PR8 virus were also measured. Acidic PBS- and PCA-treated PR8 virus lost HA activity after a 48-h reaction. On the other hand, the HA titer of neutral PCA-treated PR8 virus did not decrease compared to PBS-treated PR8 virus (Fig. 3b). These results show that neutral Hib[crude], frHibis, and PCA does not affect viral HA activity.
Fig. 3.
Neutral hibiscus tea components do not induce conformational change in HAin. (a, b) PR8 virus was mixed with an equal volume of PBS, Hib[crude], frHibis (a), or PCA (b). The pH of each aqueous solution was adjusted to acidic or approximately neutral. The pH shown was an approximate value with a range ± 0.3. The mixture was placed at 26 °C for 10 min or 48 h and mixed with an equal volume of chicken blood cells. HA titer of the mixture was evaluated after a 30-min reaction. Error bars indicate mean ± SD; n = 3 to 6 per group. Student’s t test with Bonferroni’s test (a), and Student’s t test (b) were performed to analyze statistical significance. The results represent more than two independent experiments
Evaluation of Therapeutic Efficacy of Acidic Hib[crude]-Administration in PR8 Virus-Infected Mice
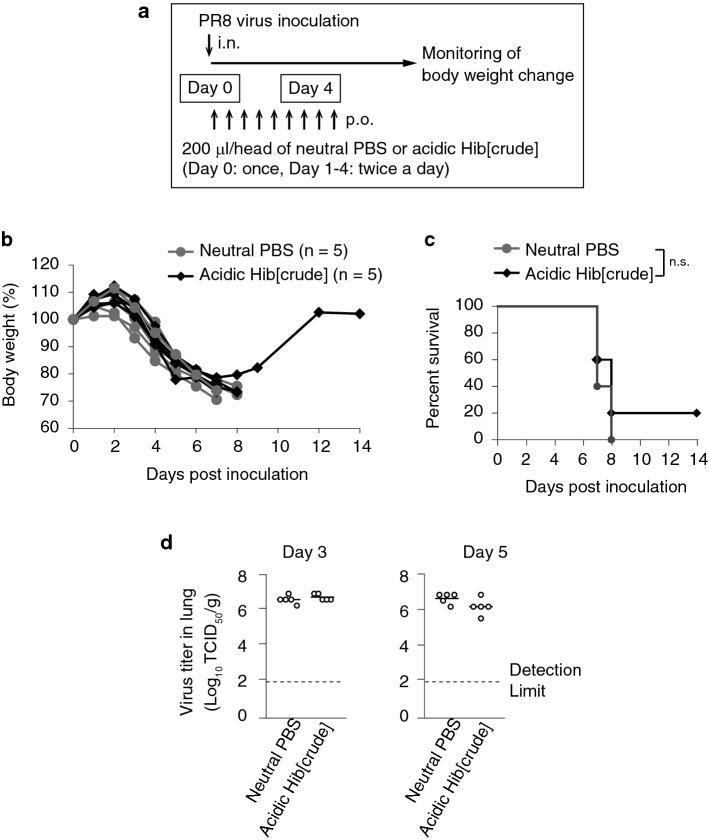
Next, we assessed the therapeutic efficacy of p.o. administration of acidic hibiscus tea extract in PR8 virus-infected mice. Neutral PBS or acidic Hib[crude] was administered p.o. to mice twice a day for the first 4 days after PR8 virus inoculation (Fig. 4a). A significant loss in body weight was observed in all treated mice (Fig. 4b). All PBS-treated mice and 80% (4 out of 5) Hib[crude]-treated mice died within 8 days post-infection. Although only one Hib[crude]-administered mouse survived and recovered from the disease, there was no significant difference in the survival rate between the PBS- and Hib[crude]-treated groups (Fig. 4c). The viral titer in the lungs was determined on days 3 and 5, and it was found to be similar to levels in both the PBS and Hib[crude] groups (Fig. 4d). These results show that p.o. administration of acidic Hib[crude] does not exert a therapeutic effect in PR8 virus-infected mice.
Fig. 4.
Acidic Hib[crude] administration does not have a therapeutic effect on PR8 virus infection. a PR8 virus inoculation scheme and therapy schedule is shown. b Changes in body weight after viral inoculation were evaluated in each mouse. n = 5 per group. c The survival percentage is shown. d Mice were euthanized, and the lungs were collected on days 3 and 5. The viral titer in 1 g of lung was evaluated. n = 5 per group. (b–d) Student’s t test (b, d), and Log-rank test (c) were performed to analyze statistical significance. n.s: not significant. The results represent three similar experiments
Evaluation of the Efficacy of Acidic Hib[crude]-Inactivated Whole-Virus Vaccine in PR8 Virus-Infected Mice
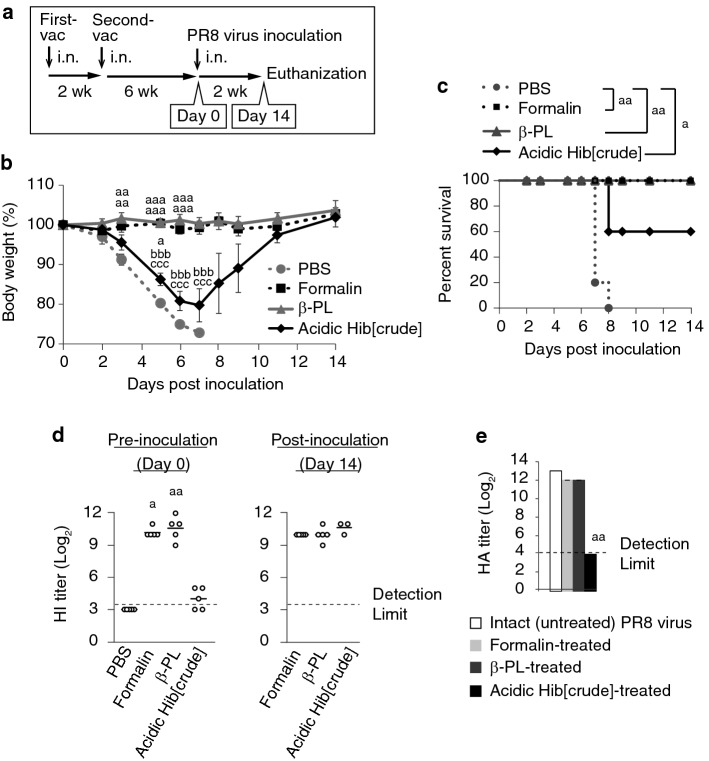
Next, we assessed the efficacy of acidic hibiscus tea extract as an inactivating agent for whole virus. The PR8 virus inactivated by formalin, β-PL, or acidic Hib[crude] was intranasally administered to mice twice before administering a lethal dose of PR8 virus (Fig. 5a). Non-vaccinated mice receiving PBS served as a control group. Non-vaccinated mice rapidly lost body weight after the viral inoculation, and all mice in the group died by day 7. On the other hand, the body weight in the formalin- and β-PL-treated groups did not change, and all the mice survived until day 14. The mice lost body weight in the Hib[crude]-treated group, but the degree of weight loss was ameliorated compared to the non-vaccinated group. The vaccine efficacy was much lower in the Hib[crude]-treated group, but the survival rate was significantly improved (Fig. 5b, c). The HI-active Ab titer in serum was also measured pre-inoculation on day 0 and post-inoculation on day 14. Prior to inoculation, the HI titers were below the detection limit in all non-vaccinated mice, but the titers were in the range of 29 to 212 in the formalin and β-PL groups. In the Hib[crude] group, the titers were less than 25 in all mice and below the detection limit in two mice (Fig. 5d). Although the HI titers were low in the Hib[crude] group, the HI-positive mice survived until day 14 (data not shown). Post-inoculation HI titer did not differ from the pre-inoculation titer in the formalin and β-PL groups. In the Hib[crude] group, the HI titer increased to the same level as the formalin and β-PL groups (Fig. 5d). These results indicate that formalin- and β-PL-inactivated vaccine completely prevented viral infection, but acidic Hib[crude]-inactivated vaccine did not prevent infection. The viral particle structure integrity in the whole inactivated vaccine relates to protective activity against IAV infection. We assessed whether the conformation of hemagglutinin (HAin) was preserved in each vaccine using the HA assay. HA activity of IAV is lost by a change in HAin conformation (Sriwilaijaroen and Suzuki 2012). Intact (untreated) or inactivated PR8 virus was mixed with chicken blood cells and the HA titer was measured. The HA titer of acidic Hib[crude]-treated PR8 virus was below the detection limit, but the HA titer of formalin- and β-PL-treated PR8 virus was similar to the intact PR8 virus (Fig. 5e). These results show that formalin and β-PL treatments but not acidic Hib[crude] preserve HAin conformation.
Fig. 5.
Acidic Hib[crude]-inactivated whole-virus vaccine induces weak anti-PR8 virus immunity. a The vaccination scheme and inoculation schedule is shown. 15 μg/50 μl of formalin-, β-PL-, or acidic Hib[crude]-inactivated PR8 virus was i.n. administered to mice twice for vaccination. b Body weight change after a viral inoculation was evaluated in each group. Error bars indicate mean ± SEM; n = 5 per group. c The survival percentage is shown. d HI titer of serum collected at pre-inoculation (day 0) and post-inoculation (day 14) stages was evaluated. e Formalin-, β-PL-, and acidic Hib[crude]-inactivated PR8 virus was mixed with an equal volume of chicken blood cells. HA titer was evaluated after a 30-min reaction. Intact (untreated) PR8 virus was used as a negative control. Error bars indicate mean ± SD; n = 3 per group. (b–e) One-way analysis of variance (ANOVA) with Tukey’s multiple comparison test or Kluskal-Wallis test with Dunn’s multiple comparison test (b), Log-rank test with Bonferroni’s test (c), and Kluskal-Wallis test with Dunn’s multiple comparison test (d, e) were performed to analyze statistical significance; compared with PBS (b–d) or untreated group (e), ap < 0.05, aap < 0.01, aaap < 0.001; compared with formalin group, bbbp < 0.001; compared with β-PL group, cccp < 0.001. A single experiment was performed
Discussion
Earlier studies indicated that the extract derived from Hibiscus sabdariffa L. calyces has antiviral activity against the H5 subtype of highly pathogenic and low-pathogenic avian influenza viruses, herpes simplex virus-2, feline calicivirus, murine norovirus-1, hepatitis A virus, and Aichi virus (Baatartsogt et al. 2016; D’Souza et al. 2016; Hassan et al. 2017; Joshi et al. 2015). Moreover, the leaves of Hibiscus sabdariffa L. also showed anti-measles virus activity (Sunday et al. 2010). However, antiviral activity mechanism against these viruses is still unclear. Since many acidic compounds in hibiscus calyces contribute to the low pH, we focused on correlating anti-PR8 viral activity and the acidic pH of hibiscus tea components. We found that not only Hib[crude] but also PBS of pH ≤ 4.0 potently inactivated PR8 virus in a short period of time (10 min) (Fig. 1). This finding indicates that the anti-PR8 virus activity of hibiscus tea extract is largely dependent on acid. However, we could not deny a possibility that there are unknown antiviral compounds which show its activity only in the acidic condition. This unknown mechanism might partially contribute to the viral inactivation. We further analyzed the anti-PR8 virus activity of PCA, ferulic acid, and hibiscus acid, which are all components of hibiscus tea extract. In the previous reports, PCA showed antiviral activity against feline calicivirus, murine norovirus-1, and herpes simplex virus-2 (Hassan et al. 2017; Joshi et al. 2015). Ferulic acid showed antiviral activity against feline calicivirus and murine norovirus-1 (Joshi et al. 2015). Here, acidic PCA and hibiscus acid showed anti-PR8 virus activity. Non-pH-adjusted ferulic acid solution with a relatively higher pH (pH 6.5) did not inactivate PR8 virus. When the IAV particle is exposed to acidic pH conditions, viral HAin changes its conformation and irreversibly loses binding affinity for sialic acid (Quan et al. 2011; Sriwilaijaroen and Suzuki 2012). Here, all PR8 virus particles treated with acidic PBS-, Hib[crude], frHibis, or PCA lost their HA activity (Fig. 3). It is also known that low-pH treatment of IAV induces M1 protein layer loss and coagulation of ribonucleoprotein complexes (Fontana et al. 2012). These comprehensive structural changes due to acid could have resulted in rapid and potent viral inactivation from acidic hibiscus component treatment. Many IAV strains are susceptible to acid, but the degree of sensitivity varies between strains (Puri et al. 1990; Ruigrok et al. 1984). Some highly pathogenic strains of avian H5 influenza virus like H5N1 strain isolated from Thailand were resistant to pH 3.0 (treated for 10 min at room temperature) treatment (Wanaratana et al. 2010). Although acidic Hib[crude] and frHibis inactivated highly pathogenic and low-pathogenic avian H5 influenza viruses in 10 min in our previous study (Baatartsogt et al. 2016), we have yet to determine whether all potent and rapid antiviral activity against these H5 viral strains is completely acid dependent.
Here, frHibis showed potent acid-dependent antiviral activity and weak low-pH-independent antiviral activity against PR8 virus (Fig. 1). The neutral PCA also showed anti-PR8 virus activity (Fig. 2). The anti-PR8 virus activity by both neutral frHibis and PCA did not affect HAin activity (Fig. 3). Since PCA accounts for 24% of Hibiscus sabdariffa water extract (Da-Costa-Rocha et al. 2014), a major part of the anti-PR8 virus activities of neutral hibiscus extract may be due to PCA. In addition, frHibis also contains abundant bioactive compounds such as phenolic compounds (Da-Costa-Rocha et al. 2014). Several studies reported that polyphenol-rich plant extracts and plant-derived phenolic compounds have anti-IAV activities (Droebner et al. 2007; Kim et al. 2013; Yamada et al. 2009). These phenolic compounds in frHibis may also contribute to antiviral activity. An attempt to identify the mechanism of low-pH-independent anti-PR8 virus activity of PCA and to search for other low-pH-independent anti-PR8 virus constituents is currently ongoing.
We assessed the therapeutic efficacy of p.o. administration of acidic Hib[crude] in PR8 virus-infected mice. However, no therapeutic effect was observed (Fig. 4). In the case of p.o. administration, Hib[crude] is first absorbed from the digestive tract and distributed into systemic blood. Subsequently, it reaches the respiratory organs where IAV resides. The neutralized Hib[crude] in the blood loses potent anti-IAV activity due to acid, and the low-pH-independent antiviral activity is inadequate to inactivate virus in vivo. The low content, weak antiviral activity, and short biological half-life of low-pH-independent anti-IAV compounds in Hib[crude] could contribute to therapeutic failure. To achieve a successful p.o. administration therapy using hibiscus tea components such as PCA, it would be necessary to chemically modify them to extend their biological half-life and enhance antiviral activity or use them in combination with a carrier to retain them in blood.
In IAV vaccination, i.n. vaccine administration induces effective anti-IAV immunity. We previously showed that i.n. co-administration of innate immune receptor adjuvants with split vaccine efficiently induced both systemic and mucosal anti-IAV immunity (Takaki et al. 2018, 2017; Takeda et al. 2018). On the other hand, whole inactivated vaccine comprises viral structural components with immune priming activity like immunostimulatory viral RNA. We assessed the utility of acidic Hib[crude] as a safe inactivating agent for whole-virus vaccine. However, unlike the viruses inactivated by formalin and β-PL, the viruses inactivated by acidic Hib[crude] failed to induce efficient protective immunity (Fig. 5). As shown in other reports (Pawar et al. 2015), formalin- and β-PL-treated virus but not acidic Hib[crude] showed intact HA activity (Fig. 5e). HAin is an immunodominant protein (Kaminski and Lee 2011; Liu et al. 2018; Sautto et al. 2018). The acidic pH of Hib[crude] could cause the loss of antigenicity by inducing a change in HAin conformation. These results show that the acidic hibiscus components cannot be used as inactivating agents for vaccine preparation. On the other hand, low-pH-independent activity of frHibis and PCA which does not affect HAin conformation is suitable for vaccine application. However, these weak virucidal effects have not yet been sufficient for use as an inactivating agent. Further study of the low-pH-independent anti-IAV mechanism and enhancement of its antiviral activity may contribute to IAV vaccine strategy development.
In conclusion, we showed the rapid and potent anti-PR8 virus activity of acidic hibiscus tea extract, which relies largely on acid. This acidic extract is not suitable for use in therapeutics and vaccine development. However, frHibis and PCA were found to have weak low-pH-independent anti-PR8 virus activity, which does not affect HAin conformation. Hence, further study of low-pH-independent antiviral compounds may enable us to establish a novel anti-IAV therapy and vaccination strategy.
Acknowledgements
We thank Sachiko Matsuda for her technical assistance. This work was partially supported by JSPS KAKENHI Grant Number 17K19878 from the Japan Society for the Promotion of Science, Japan, and the grant from Mishima Kaiun Memorial Foundation. We would like to thank Editage (www.editage.jp) for English language editing.
Abbreviations
- Ab
Antibody
- β-PL
β-Propiolactone
- CPE
Cytopathic effect
- HA
Hemagglutination
- HAin
Hemagglutinin
- HI
Hemagglutination inhibition
- IAV
Influenza A virus
- i.n.
Intranasal
- PBS
Phosphate-buffered saline
- PCA
Protocatechuic acid
- p.o.
Orally
- TCID50
50% Tissue culture infective dose
Author Contributions
YT, YO, HN, YY, KM, HO, and KI conceived and designed the experiments. YT performed viral experiments. YO isolated hibiscus acid and made hibiscus acid-Na. YT, YO, HN, YY, KM, HO, and KI analyzed the data. YT, YO, HN, HO, and KI contributed to writing the manuscript.
Compliance with Ethical Standards
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Baatartsogt T, Bui VN, Trinh DQ, Yamaguchi E, Gronsang D, Thampaisarn R, et al. High antiviral effects of hibiscus tea extract on the H5 subtypes of low and highly pathogenic avian influenza viruses. Journal of Veterinary Medical Science. 2016 doi: 10.1292/jvms.16-0124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D’Souza DH, Dice L, Davidson PM. Aqueous extracts of hibiscus sabdariffa calyces to control aichi virus. Food and Environmental Virology. 2016 doi: 10.1007/s12560-016-9229-5. [DOI] [PubMed] [Google Scholar]
- Da-Costa-Rocha I, Bonnlaender B, Sievers H, Pischel I, Heinrich M. Hibiscus sabdariffa L.—A phytochemical and pharmacological review. Food Chemistry. 2014 doi: 10.1016/j.foodchem.2014.05.002. [DOI] [PubMed] [Google Scholar]
- Delrue I, Verzele D, Madder A, Nauwynck HJ. Inactivated virus vaccines from chemistry to prophylaxis: Merits, risks and challenges. Expert Review of Vaccines. 2012 doi: 10.1586/erv.12.38. [DOI] [PubMed] [Google Scholar]
- Droebner K, Ehrhardt C, Poetter A, Ludwig S, Planz O. CYSTUS052, a polyphenol-rich plant extract, exerts anti-influenza virus activity in mice. Antiviral Research. 2007 doi: 10.1016/j.antiviral.2007.04.001. [DOI] [PubMed] [Google Scholar]
- Fontana J, Cardone G, Heymann JB, Winkler DC, Steven AC. Structural changes in influenza virus at low pH characterized by cryo-electron tomography. Journal of Virology. 2012 doi: 10.1128/JVI.06698-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunn Frederick L. Pandemic influenza in 1957; review of international spread of new Asian strain. The Journal of the American Medical Association. 1958 doi: 10.1001/jama.1958.02990100028006. [DOI] [PubMed] [Google Scholar]
- Hassan STS, Švajdlenka E, Berchová-Bímová K. Hibiscus sabdariffa L. and its bioactive constituents exhibit antiviral activity against HSV-2 and anti-enzymatic properties against urease by an ESI-MS based assay. Molecules. 2017 doi: 10.3390/molecules22050722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurt AC. The epidemiology and spread of drug resistant human influenza viruses. Current Opinion in Virology. 2014 doi: 10.1016/j.coviro.2014.04.009. [DOI] [PubMed] [Google Scholar]
- Ibnusaud I, Thomas PT, Rani RN, Sasi PV, Beena T, Hisham A. Chiral γ-butyrolactones related to optically active 2-hydroxycitric acids. Tetrahedron. 2002 doi: 10.1016/S0040-4020(02)00431-3. [DOI] [Google Scholar]
- Ibrahim, I., Rani, R., Teena, P., & Salini, T. (2000). Convenient method for large-scale isolation of hibiscus acid. U.S. Patent 6,127,553.
- Joshi SS, Dice L, D’Souza DH. Aqueous extracts of hibiscus sabdariffa calyces decrease hepatitis a virus and human norovirus surrogate titers. Food and Environmental Virology. 2015 doi: 10.1007/s12560-015-9209-1. [DOI] [PubMed] [Google Scholar]
- Kaminski DA, Lee FEH. Antibodies against conserved antigens provide opportunities for reform in influenza vaccine design. Frontiers in Immunology. 2011 doi: 10.3389/fimmu.2011.00076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kärber G. Beitrag zur kollektiven Behandlung pharmakologischer Reihenversuche. Naunyn-Schmiedebergs Archiv für experimentelle Pathologie und Pharmakologie. 1931 doi: 10.1007/BF01863914. [DOI] [Google Scholar]
- Kim EH, Pascua PNQ, Song MS, Baek YH, Kwon H. Il, Park SJ, et al. Immunomodulaton and attenuation of lethal influenza A virus infection by oral administration with KIOM-C. Antiviral Research. 2013 doi: 10.1016/j.antiviral.2013.04.006. [DOI] [PubMed] [Google Scholar]
- Li TCM, Chan MCW, Lee N. Clinical Implications of Antiviral Resistance in Influenza. Viruses. 2015 doi: 10.3390/v7092850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu STH, Behzadi MA, Sun W, Freyn AW, Liu WC, Broecker F, et al. Antigenic sites in influenza H1 hemagglutinin display species-specific immunodominance. The Journal of Clinical Investigation. 2018 doi: 10.1172/JCI122895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagata T, Lefor AK, Hasegawa M, Ishii M. Favipiravir: A new medication for the ebola virus disease pandemic. Disaster Medicine and Public Health Preparedness. 2015 doi: 10.1017/dmp.2014.151. [DOI] [PubMed] [Google Scholar]
- Noshi T, Kitano M, Taniguchi K, Yamamoto A, Omoto S, Baba K, et al. In vitro characterization of baloxavir acid, a first-in-class cap-dependent endonuclease inhibitor of the influenza virus polymerase PA subunit. Antiviral Research. 2018 doi: 10.1016/j.antiviral.2018.10.008. [DOI] [PubMed] [Google Scholar]
- Pawar SD, Murtadak VB, Kale SD, Shinde PV, Parkhi SS. Evaluation of different inactivation methods for high and low pathogenic avian influenza viruses in egg-fluids for antigen preparation. Journal of Virological Methods. 2015 doi: 10.1016/j.jviromet.2015.05.004. [DOI] [PubMed] [Google Scholar]
- Puri A, Booy FP, Doms RW, White JM, Blumenthal R. Conformational changes and fusion activity of influenza virus hemagglutinin of the H2 and H3 subtypes: Effects of acid pretreatment. Journal of Virology. 1990 doi: 10.1109/ICCCNT.2015.7395209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quan FS, Li ZN, Kim MC, Yang D, Compans RW, Steinhauer DA, Kang SM. Immunogenicity of low-pH treated whole viral influenza vaccine. Virology. 2011 doi: 10.1016/j.virol.2011.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose MA, Zielen S, Baumann U. Mucosal immunity and nasal influenza vaccination. Expert Review of Vaccines. 2012 doi: 10.1586/erv.12.31. [DOI] [PubMed] [Google Scholar]
- Ruigrok RW, Cremers AF, Beyer WE, de Ronde-Verloop FM. Changes in the morphology of influenza particles induced at low pH. Archives of Virology. 1984 doi: 10.1007/BF01311162. [DOI] [PubMed] [Google Scholar]
- Saunders-Hastings P, Krewski D. Reviewing the history of pandemic influenza: understanding patterns of emergence and transmission. Pathogens. 2016 doi: 10.3390/pathogens5040066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sautto GA, Kirchenbaum GA, Ross TM. Towards a universal influenza vaccine: Different approaches for one goal. Virology Journal. 2018 doi: 10.1186/s12985-017-0918-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soema PC, Kompier R, Amorij JP, Kersten GFA. Current and next generation influenza vaccines: Formulation and production strategies. European Journal of Pharmaceutics and Biopharmaceutics. 2015 doi: 10.1016/j.ejpb.2015.05.023. [DOI] [PubMed] [Google Scholar]
- Sriwilaijaroen N, Suzuki Y. Molecular basis of the structure and function of H1 hemagglutinin of influenza virus. Proceedings of the Japan Academy, Series B. 2012 doi: 10.1111/j.1360-0443.1992.tb03126.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sunday OA, Munir AB, Akeeb OO, Adesanya Bolanle A, Badaru SO. Antiviral effect of Hibiscus sabdariffa and Celosia argentea on measles virus. African Journal of Microbiology Research. 2010;4(4):293–296. [Google Scholar]
- Takaki H, Kure S, Oshiumi H, Sakoda Y, Suzuki T, Ainai A, et al. Toll-like receptor 3 in nasal CD103 + dendritic cells is involved in immunoglobulin A production. Mucosal Immunology. 2018 doi: 10.1038/mi.2017.48. [DOI] [PubMed] [Google Scholar]
- Takaki H, Takashima K, Oshiumi H, Ainai A, Suzuki T, Hasegawa H, et al. cGAMP promotes germinal center formation and production of IgA in nasal-associated lymphoid tissue. Medical Sciences. 2017 doi: 10.3390/medsci5040035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeda Y, Takaki H, Fukui-Miyazaki A, Yoshida S, Matsumoto M, Seya T. Vaccine adjuvant ARNAX promotes mucosal IgA production in influenza HA vaccination. Biochemical and Biophysical Research Communications. 2018 doi: 10.1016/j.bbrc.2018.10.166. [DOI] [PubMed] [Google Scholar]
- Tamura SI, Ainai A, Suzuki T, Kurata T, Hasegawa H. Intranasal inactivated influenza vaccines: A reasonable approach to improve the efficacy of influenza vaccine? Japanese Journal of Infectious Diseases. 2016 doi: 10.7883/yoken.JJID.2015.560. [DOI] [PubMed] [Google Scholar]
- Wanaratana S, Tantilertcharoen R, Sasipreeyajan J, Pakpinyo S. The inactivation of avian influenza virus subtype H5N1 isolated from chickens in Thailand by chemical and physical treatments. Veterinary Microbiology. 2010 doi: 10.1016/j.vetmic.2009.07.008. [DOI] [PubMed] [Google Scholar]
- World Health Organization. (2002). WHO Manual on Animal Influenza Diagnosis and Surveillance. Retrieved Dec 4, 2018 from https://apps.who.int/iris/bitstream/handle/10665/68026/WHO_CDS_CSR_NCS_2002.5.pdf?sequence=1&isAllowed=y
- World Health Organization. (2009). World now at the start of 2009 influenza pandemic. Retrieved Dec 4, 2018 from https://www.who.int/mediacentre/news/statements/2009/h1n1_pandemic_phase6_20090611/en/
- World Health Organization. (2018). Influenza (Seasonal). Retrieved Dec 4, 2018 from https://www.who.int/en/news-room/fact-sheets/detail/influenza-(seasonal)
- Yamada K, Ogawa H, Hara A, Yoshida Y, Yonezawa Y, Karibe K, et al. Mechanism of the antiviral effect of hydroxytyrosol on influenza virus appears to involve morphological change of the virus. Antiviral Research. 2009 doi: 10.1016/j.antiviral.2009.03.002. [DOI] [PubMed] [Google Scholar]