Abstract
Background and Purpose—
We compared the rates of death or disability, defined by modified Rankin Scale score of 4 to 6, at 3 months in patients with intracerebral hemorrhage according to post-treatment systolic blood pressure (SBP)–attained status.
Methods—
We divided 1000 subjects with SBP ≥180 mm Hg who were randomized within 4.5 hours of symptom onset as follows: SBP <140 mm Hg achieved or not achieved within 2 hours; subjects in whom SBP <140 mm Hg was achieved within 2 hours were further divided: SBP ≥140 mm Hg for 21 to 22 hours (reduced and maintained) or SBP was ≥140 mm Hg for at least 2 hours during the period between 2 and 24 hours (reduced but not maintained).
Results—
Compared with subjects without reduction of SBP <140 mm Hg within 2 hours, subjects with reduction and maintenance of SBP <140 mm Hg within 2 hours had a similar rate of death or disability (relative risk of 0.98; 95% confidence interval, 0.74–1.29). The rates of neurological deterioration within 24 hours were significantly higher in reduced and maintained group (10.4%; relative risk, 1.98; 95% confidence interval, 1.08–3.62) and in reduced but not maintained group (11.5%; relative risk, 2.08; 95% confidence interval, 1.15–3.75) compared with reference group. The rates of cardiac-related adverse events within 7 days were higher among subjects with reduction and maintenance of SBP <140 mmHg compared to subjects without reduction (11.2% versus 6.4%).
Conclusions—
No decline in death or disability but higher rates of neurological deterioration and cardiac-related adverse events were observed among intracerebral hemorrhage subjects with reduction with and without maintenance of intensive SBP goals.
Keywords: acute hypertensive response, blood pressure, cerebral hemorrhage, clinical trial, humans
An acute hypertensive response is seen in two thirds of patients with intracerebral hemorrhage (ICH),1 and acute aggressive reduction of systolic blood pressure (SBP) has been investigated in clinical trials to reduce the rate of hematoma expansion and death or disability.2 In the INTERACT2 (Intensive BP Reduction in Acute Cerebral Hemorrhage Trial),3 the rate of death or disability in subjects who were randomized to intensive SBP reduction, with a target SBP of <140 mm Hg within 1 hour, was nonsignificantly lower than that in those assigned to treatment with a target SBP of <180 mm Hg (absolute difference, 3.6 percentage points; P=0.06). In the ATACH 2 trial (Antihypertensive Treatment of Acute Cerebral Hemorrhage),4 the rate of death or disability was not different between subjects randomized to achieve a target SBP of 110 to 139 mm Hg compared with standard reduction to a target of 140 to 179 mm Hg (absolute difference, 1.0 percentage points; P=0.72) in intention-to-treat analysis. However the intention-to-treat analysis may be confounded by compliance to therapeutic intervention,5 with the probability that the benefit of intervention may be obscured if a proportion of subjects did not achieve the SBP goals specified for the intended randomized group.6 Therefore, the analysis based on actual blood pressure (BP) attained may be a more efficient method to evaluate the effect of rapid intensive lowering of SBP on various outcome measures.7–9 We present the results of analysis based on SBP attained using data from the ATACH 2 trial.
Methods
The data that support the findings of this study are available from the corresponding author on reasonable request.
Study Design
The ATACH 2 trial was a randomized multinational trial designed to determine the efficacy of intensive SBP reduction initiated within 4.5 hours of symptom onset in subjects with spontaneous supratentorial ICH with SBP ≥180 mm Hg.4,10 Briefly, subjects with a Glasgow Coma Scale score of ≥5 at the time of emergency department arrival and intraparenchymal hematoma <60 mm3 on initial computed tomographic scan were randomized. The goal of treatment was to reduce within 2 hours of randomization and maintain through 24 hours each subject’s hourly minimum SBP of 140 to 179 mm Hg in the standard treatment group and 110 to 139 mm Hg in the intensive treatment group.
Intervention
After randomization, intravenous nicardipine infusion was initiated at 5 mg/h and then increased by 2.5 mg/h every 15 minutes as needed, up to a maximum of 15 mg/h. Once the target SBP level was reached, the infusion rate was adjusted by 1 to 2.5 mg/h to maintain SBP in the specified range. If SBP fell below the target value, intravenous nicardipine was reduced by 2.5 mg/h every 15 minutes until the infusion was discontinued. Infusion was not restarted unless SBP rose >180 mm Hg or 140 mm Hg for standard and intensive treatment groups, respectively. If SBP was greater than the target, despite infusion of the maximum nicardipine dose for 30 minutes, a prespecified second agent, intravenous labetalol (5–20 mg intravenous bolus every 15 minutes), was applied for another hour. In countries where labetalol was not available, diltiazem (initial infusion rate of 5 mg/h ≤15 mg/h) or uradipil (12.5 mg every 15 minutes to a maximum dose of 50 mg) was used.
BP Measurements
SBP measurements were acquired every 5 minutes for the first 15 minutes after initiation of nicardipine infusion followed by every 15 minutes for the remainder of the first hour or every 5 minutes if the dose was being adjusted. SBP was measured at least every 30 minutes while receiving nicardipine or more frequently if prominent SBP changes were observed as determined by the treating physician. For the first 24 hours, minimum and maximum SBPs were recorded for each 15-minute period during the first hour and for each subsequent hour through 24 hours.
Data Collection
The SBP was collected at least twice hourly for the first 24 hours after randomization. A noncontrast head computed tomographic scan was performed at baseline and 24 hours after initiation of treatment and analyzed by a reader who was blinded to the treatment assignment, clinical findings, and time points of image acquisition. Parenchymal hematoma volume was measured using automated delineation (with manual correction) by computer software on each slice containing the hematoma.
Postdischarge follow-up included telephone contact at 1 month and in-person clinical evaluation at 3 months. The telephone interview by the site staff collected information on serious adverse events (SAEs) and deaths. The 3-month assessments were conducted by a qualified investigator who did not participate in the randomization, treatment, or in-hospital clinical management of the subject. The data collection consisted of disability assessment using modified Rankin Scale and quality-of-life assessment by EQ-5D. SAEs were ascertained throughout 3 months post-randomization and, non-SAEs were ascertained only within the first 5 days or on discharge (whichever came first). All adverse events and SAEs were coded with the Medical Dictionary for Regulatory Activities. The analysis was performed after grouping the related events (events that represent the same condition of interest according to body system) to determine the comprehensive occurrence rate of an event with relationship to SBP lowering.
Statistical Analysis
Study participants were grouped based on achieved minimum SBP, regardless of randomized allocation, as follows: first, based on whether or not SBP <140 mm Hg was achieved within 2 hours (based on 5 recordings acquired within 2 hours of randomization); subjects in whom SBP <140 mm Hg was achieved within 2 hours were further divided into those in whom SBP <140 mm Hg was maintained for 21 to 22 hours (reduced and maintained) and those in whom SBP was ≥140 mm Hg for at least 2 hours during the period between 2 and 24 hours (reduced but not maintained). If missing, minimum SBP was imputed using last observation carried forward.
The outcomes of interest in these 2 groups were compared against a reference group comprising those who did not achieve SBP <140 mm Hg within 2 hours. The primary outcome was the proportion of subjects with death or disability (modified Rankin Scale score of 4–6) at 3 months. Secondary outcomes were (1) death within 3 months, (2) the proportion of subjects with hematoma expansion of >33% in volume on the computed tomographic scan obtained at 24 hours compared with the baseline scan, and (3) scores on the EQ-5D utility index at 3 months. Safety outcomes were (1) neurological deterioration, defined as a decrease from baseline of ≥2 points on Glasgow Coma Scale or increase of ≥4 points on the National Institutes of Health Stroke Scale that was not related to sedation/hypnotic use and was sustained for at least 8 hours within 24 hours; (2) brain infarction-, cardiac-, and renal-related adverse events within 7 days; and (3) any SAE within 3 months. The comparison was adjusted for baseline age, Glasgow Coma Scale category (0–11, 12–14, and 15), and the presence of intraventricular hemorrhage determined by the central reader, consistent with the published primary analysis.4
For consistency with the published primary analysis,4 the analysis of the dichotomized outcomes (all except EQ-5D) was based on the generalized linear model with log link function with Poisson distribution. ED-5D analysis was based on the generalized linear model with normal distribution and identity link. Statistical significance was defined at an a of 0.05, recognizing that these are exploratory analyses that require caution in their interpretation. All analyses were performed using SAS, version 9.4, software.
Results
Among the 1000 subjects randomized, 731 subjects had achieved reduction of the minimum SBP to <140 mm Hg within 2 hours of randomization. Among those, the minimum SBP <140 mm Hg was reduced within 2 hours and maintained for 21 to 22 hours through 24 hours after randomization in 357 subjects (reduced and maintained group), and 374 achieved SBP <140 but did not maintain this level (reduced but not maintained group). Of the 267 subjects without reduction within 2 hours (reference group), the proportion of subjects who did not maintain SBP <140 mm Hg was 89.5% (n=239). Two subjects were excluded from the analysis because of completely missing SBP measurements. The baseline characteristics were generally well balanced among the groups (Table 1). Per Table 2, differences, if any, among the groups were not unexpected because of the nature of the post hoc classification. The SBP profiles of the 3 groups are provided in Figure 1.
Table 1.
Baseline Characteristics of Subjects by Blood Pressure-Attained Groups
| Subjects in Whom SBP Remained ≥140 mm Hg Within 2 h Post-Randomization | Subjects in Whom SBP Was Reduced <140 mm Hg Within 2 h Post-Randomization | |||
|---|---|---|---|---|
| Maintained <140 mm Hg 2–22 h | Not Maintained <140 mm Hg 2–22 h | |||
| Age, y; mean (SD) | 58 (12.7) | 63.4 (12.6) | 63.4 (13.4) | |
| Sex (men), n/total (%) | 189/267 (70.8) | 209/357 (58.5) | 220/374 (58.8) | |
| Ethnicity (Hispanic/Latino), n/total (%) | 24/267 (9.0) | 23/357 (6.4) | 32/374 (8.6) | |
| Race, n/total (%) | Asian | 149/267 (55.8) | 226/357 (63.3) | 185/374 (49.5) |
| Black | 55/267 (20.6) | 30/357 (8.4) | 46/374 (12.3) | |
| White | 58/267 (21.7) | 97/357 (27.2) | 132/374 (35.3) | |
| Other/unknown | 5/267 (1.9) | 4/357 (1.1) | 11/374 (2.9) | |
| Baseline Glasgow Coma Scale score category, n/total (%) | 3–11 | 40/267 (15.0) | 48/357 (13.4) | 59/374 (15.8) |
| 12–14 | 85/267 (31.8) | 104/357 (29.1) | 103/374 (27.5) | |
| 15 | 142/267 (53.2) | 205/357 (57.4) | 212/374 (56.7) | |
| Baseline NIHSS score, median (min-max) | 10 (0–37) | 10.5 (0–37) | 11 (0–37) | |
| Baseline SBP at presentation, mm Hg; mean (SD) | 204.4 (27.3) | 198 (26.3) | 200.2 (27.0) | |
| Baseline ICH volume, cm3; median (min–max) | 10.5 (0.4–57.3) | 9.5 (0.2–57.3) | 10.8 (0.1–57.3) | |
| Baseline ICH volume >30 cm3, n/total (%) | 24/264 (9.1) | 30/354 (8.5) | 42/368 (11.4) | |
| History of hypertension, n/total (%) | 211/267 (79.0) | 288/357 (80.7) | 292/374 (78.1) | |
| Prescribed antihypertensive medication,* n/total (%) | 127/266 (47.7) | 171/355 (48.2) | 195/370 (52.7) | |
| History of atrial fibrillation, n/total (%) | 6/267 (2.2) | 12/357 (3.4) | 18/374 (4.8) | |
| History of coronary artery disease, n/total (%) | 10/267 (3.7) | 14/357 (3.9) | 20/374 (5.3) | |
| History of diabetes mellitus type II, n/total (%) | 53/267 (19.9) | 58/357 (16.2) | 63/374 (16.8) | |
| History of hyperlipidemia, n/total (%) | 59/267 (22.1) | 83/357 (23.2) | 99/374 (26.5) | |
| History of stroke, n/total (%) | 47/267 (17.6) | 52/357 (14.6) | 65/374 (17.4) | |
| Location of ICH, n/total (%) | Basal ganglia | 141/264 (53.4) | 179/354 (50.6) | 185/368 (50.3) |
| Thalamus | 95/264 (36.0) | 138/354 (39.0) | 139/368 (37.8) | |
| Lobar | 27/264 (10.2) | 37/354 (10.5) | 44/368 (12.0) | |
| Pons/cerebellum | 1/264 (0.4) | 0/354 (0) | 0/368 (0) | |
Two subjects were excluded from the analysis because of completely missing SBP measurements. The denominator (total) reflects all subjects with nonmissing values for each categorical variable. ICH indicates intracerebral hemorrhage; max, maximum; min, minimum; NIHSS, National Institutes of Health Stroke Scale; and SBP, systolic blood pressure.
In 30 d before hospitalization.
Table 2.
Treatment Characteristics of Subjects by BP-Attained Groups
| Subjects in Whom SBP Remained ≥140 mm Hg Within 2 h Post-Randomization | Subjects in Whom SBP Was Reduced <140 mm Hg Within 2 h Post-Randomization | ||
|---|---|---|---|
| Maintained <140 mm Hg 2–22 h | Not Maintained <140 mm Hg 2–22 h | ||
| Randomized to intensive treatment, n/total (%) | 61/267 (22.8%) | 342/357 (95.8%) | 97/374 (25.9%) |
| Time interval between symptom onset and nicardipine infusion, min; mean (SD) | 158 (84.8) | 150.1 (66.0) | 162.9 (100.3) |
| Time interval between symptom onset and randomization, min; mean (SD) | 184.9 (58.2) | 181.1 (57.3) | 184.7 (55.7) |
| Mean minimum systolic BP (1–2 h), mm Hg; mean (SD) | 154.4 (8.9) | 124.1 (12.2) | 131.6 (11.8) |
| Mean minimum systolic BP (3–24 h), mm Hg; mean (SD) | 142.2 (12.5) | 119.9 (4.8) | 138.7 (10.6) |
| Received intraventricular catheter, n/total (%) | 12/266 (4.5) | 22/354 (6.2) | 36/371 (9.7) |
| Received mechanical ventilation, n/total (%) | 36/266 (13.5) | 39/354 (11.0) | 62/371 (16.7) |
| Received surgical evacuation/decompression, n/total (%) | 8/266 (3.0) | 13/354 (3.7) | 23/371 (6.2) |
| Length of hospitalization, d; mean (SD) | 20.0 (17.6) | 19.7 (15.1) | 17.4 (14.9) |
| In-hospital mortality, n/total (%) | 8/267 (3.0) | 7/357 (2.0) | 20/374 (5.3) |
BP indicates blood pressure; and SBP, systolic blood pressure.
Figure 1.
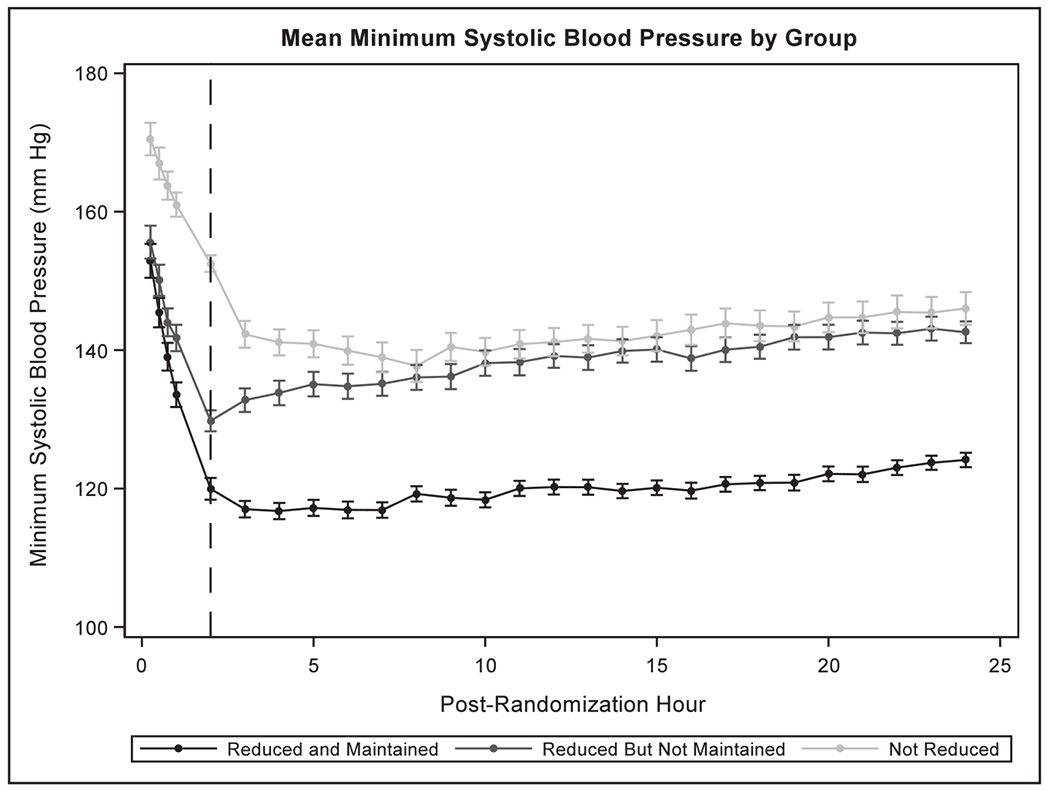
Systolic blood profile for 24 h post-randomization according to blood pressure-attained groups.
Figure 2 shows the outcomes analyses results. Death or disability at 3 months was observed in 279 of 708 (39.4%) subjects with reduction of SBP <140 mm Hg (reduced and maintained and reduced but not maintained groups combined) and 87 of 252 (34.5%) subjects in reference group. Compared with reference group, reduced but not maintained group had a higher rate, although not significantly, of death or disability at 3 months (43.6% versus 34.5%; adjusted relative risk [aRR], 1.13; 95% confidence interval [CI], 0.87–1.47). There was no difference in the rate of death or disability at 3 months in reduced and maintained group compared with those in the reference group (35.0% versus 34.5%; relative risk, 0.98; 95% CI, 0.74–1.29).
Figure 2.

Outcomes in blood pressure-attained groups. RR indicates relative risk.
The rates of hematoma expansion within 24 hours were not significantly different among the 3 groups. However, the rates of neurological deterioration within 24 hours were significantly higher in reduced and maintained group (10.4%; aRR, 1.98; 95% CI, 1.08–3.62) and in reduced but not maintained group (11.5%; aRR, 2.08; 95% CI, 1.15–3.75) compared with reference group (5.6%). There was no difference in the median values of EQ-5D utility index in the 3 groups. Although not statistically significant, a higher rate of any SAE within 3 months was observed in reduced but not maintained group compared with the reference group (27.0% versus 17.6%; aRR, 1.40; 95% CI, 0.98–1.99). Also compared with the reference group (6.4%), the rate of cardiac-related adverse events within 7 days was nonsignificantly higher in reduced but not maintained group (11.2%; aRR, 1.70; 95% CI, 0.96–3.01) and significantly higher in reduced and maintained group (11.2%; aRR, 1.84; 95% CI, 1.04–3.27; Table I in the online-only Data Supplement for detailed listing of cardiac adverse events). The rates of renal-related adverse events within 7 days were similar among the 3 groups (Figure 2).
Discussion
We observed no difference in the rate of death or disability at 3 months among subjects in whom SBP <140 mm Hg was achieved within 2 hours compared with those in whom this SBP goal was not met within the first 2 hours. The SBP profiles in Figure 1 demonstrate a clear separation of SBPs within 2 hours and for 21 to 22 hours through 24 hours after randomization between subjects classified as reduced and maintained and reference groups. Despite a clear separation in SBP profiles when defined by actual SBP values attained, the results of the analysis are consistent with the results of previous intention-to-treat analysis that the intensive SBP reduction does not result in reduction in death or disability at 3 months. A higher rate of neurological deterioration was observed in subjects with reduction with and without maintenance of SBP. A higher rate of cardiac-related adverse events was also observed in subjects with reduction and maintenance of SBP. If the safety profiles of the 3 groups were put into perspective, subjects in whom SBP values were >140 mm Hg appeared to have the best profile.
There are several methods to analyze the effect of actual SBP achieved on safety and efficacy end points. Our analysis focused on determining whether optimally achieving SBP <140 mm Hg (based on actually attaining target SBP <140 mm Hg) reduced the rate of death or disability and not directed toward identifying the optimal SBP for therapeutic benefit. In an analysis of the INTERACT 2 trial,8 subjects with an achieved SBP of 130 to 139 mm Hg defined by a mean of SBP values measured at 1, 6, 12, 18, and 24 hours had the lowest rate of death or disability. However, the prerandomization SBP was lower in patients who achieved SBP of 130 mm Hg compared with those who achieved a mean SBP of 140 to 149, 150 to 159, 160 to 169, or 170 to 179 mm Hg. Despite the small proportion of patients who achieved an SBP <130 mm Hg in the study (268/2794), there was an increase in the risk of death or major disability in patients with a mean SBP of 120 mm Hg (odds ratio, 1.30; 95% CI, 0.92–1.83; P=0.144). In another analysis of INTERACT 2,7 larger magnitude reduction in SBP was associated with the lowest rates of death or disability at 3 months with the maximum benefit observed in subjects with an initial mean SBP of 186 mm Hg (±16 mm Hg) and reduction of ≥20 mm Hg within the first hour (average SBP between 15 and 60 minutes) after randomization, which would be similar to the group in our analysis in whom SBP goals were not met within the first 2 hours (and at later intervals).
Another important finding is the safety of intensive SBP reduction. There appeared to be higher rates of neurological deterioration within 24 hours and cardiac-related SAEs within 7 days among subjects in whom SBP was reduced to <140 mm Hg. In the intention-to-treat analysis reported previously, there was a higher rate of SAEs within 3 months and renal-related adverse events within 7 days in subjects randomized to intensive treatment. When classified by actual SBP achieved, there was a nonsignificantly higher rate of SAEs in subjects who achieved initial reduction of SBP <140 mm Hg (within 2 hours) but not maintained between 2 and 24 hours compared with subjects who did not achieve reduction (reference group). A higher rate of neurological deterioration and cardiac-related adverse events within 7 days was observed in subjects with reduction with and without maintenance of SBP. The most likely explanation would be direct effects of rapid reduction in SBP resulting in regional hypoperfusion of target organs, such as brain and heart. These organs were more vulnerable in study subjects because of existing arterial disease secondary to chronic hypertension. We did not acquire magnetic resonance imaging as part of a standardized protocol and thus cannot determine whether ischemic injury to the brain was the underlying cause for the neurological deterioration. The rates of adverse events classified as brain infarction within 7 days were low and similar among the 3 groups (Figure 2). The cardiac adverse events were minor, and rates of adverse events classified as myocardial infarction within 7 days were similar among the 3 groups (Table I in the online-only Data Supplement).
Our analysis was not directed toward identifying or quantitating within individual BP fluctuations but indirectly addressed the issue. The detrimental consequences of SBP fluctuations have been identified in previous studies in patients with ICH.11–13 They have used variability as defined by the SD of achieved SBP and showed that it was associated with higher rates of death or disability at 3 months. Previous studies have shown that regional cerebral blood flow is maintained within a constant range by compliance and resistance of medium-sized artery mechanism independent of modulation of arteriolar tone (autoregulation).14–16 Such compensatory response is more vulnerable to alteration by intracranial mass lesions, such as ICH, and may increase risk of cerebral hypoperfusion with BP fluctuations.17,18 Another aspect that requires consideration is the SBP profile may be related to underlying pathogenesis and severity of disease. It is possible that certain patients with lobar ICH may have cerebral amyloid angiopathy (rather than chronic hypertension) as pathogenesis and thus may be more likely to respond to antihypertensive medication. Patients with larger hematoma volumes may be less likely to respond to antihypertensive medication because of confounding effects of intracranial hypertension. However, we did not observe any differences in location and volume of hematomas among the 3 groups (Table 1).
The practical implications of our analysis are that caution is required in how intensive SBP reduction is defined, and how it is achieved and maintained. SBP may need to be considered as a more complex continuous time-varying parameter, rather than as a static threshold target. An assumption that SBP reduction to value <140 mm Hg independent of pretreatment SBP and actual SBP achieved and maintained is beneficial and without any adverse events is not supported by the results of our analysis.19 The current guidelines from the American Heart Association/American Stroke Association state that for patients with ICH presenting with SBP between 150 and 220 mm Hg and without contraindication to acute BP treatment, acute lowering of SBP to 140 mm Hg is safe (class I; level of evidence A) and can be effective for improving functional outcome (class IIa; level of evidence B).20 Our analysis highlights the need for careful interpretation of the current guidelines and perhaps updating the guidelines to highlight the safety considerations when such BPs are actually achieved. Additional combined analysis of data from ATACH 2 and INTERACT 2 (already planned) may be necessary to better define the SBP targets where risk and benefit ratio could be optimized and thus provide a higher level of evidence. Another implication is the potential to improve SBP control through close monitoring and appropriate titration to reduce adverse events associated with intravenous antihypertensive medication. Several studies have demonstrated frequent dose adjustments are required to adequately maintain SBP in the desired target range,21–23 and perhaps more rigorous strategies are required to ensure timely response to change in BP in acute period of ICH.
Conclusions
The BP-attained analysis of ATACH 2 trial did not support efficacy but appeared to identify safety concerns related to intensive SBP reduction. Caution in the interpretation is advised because the statistical criterion for significance was not adjusted for multiple testing. Research into more precise definitions of BP-lowering quality parameters for medical management of acute hypertensive response in patients with ICH24,25 is needed. A combined analysis of data from ATACH 2 and INTERACT 2 has the potential to identify patient subgroups defined by SBP attained, rate and magnitude of SBP reduction, and other pertinent stratifications that may be more vulnerable to occurrence of adverse events with intensive SBP reduction.
Supplementary Material
Acknowledgments
Dr A.I. Qureshi confirms that he had full access to all the data in the study and had the final responsibility for the decision to submit the manuscript for publication.
Sources of Funding
This study was supported by grants (U01-NS062091, to Dr A.I. Qureshi; U01-NS061861 and U01-NS059041, to Dr Palesch; and U01-NS056975, to Dr Barsan) from the National Institute of Neurological Disorders and Stroke and by a grant (H23-4-3, to Dr Toyoda) from the Intramural Research Fund for Cardiovascular Diseases of the National Cerebral and Cardiovascular Center. Statistical analysis was conducted by the Department of Public Health Sciences, Medical University of South Carolina, Charleston.
Disclosures
L.D. Foster received a research grant from the National Institute of Neurological Disorders and Stroke—statistical and data coordinating center for the ATACH 2 (Antihypertensive Treatment of Acute Cerebral Hemorrhage; 5U01NS061861). She also receives compensation as the Significant (>$10k or 5%). Dr Goldstein receives research grant from Pfizer (Significant; >$10k or 5%), Boehringer Ingelheim (Modest; <$10k or 5%), and Portola (Significant; >$10k or 5%); serves as a consultant or advisory board member for CSL Behring; and also receives compensation (Modest; <$10k or <5%). Dr Hsu was supported by the Ministry of Health and Welfare Clinical Trial Center for Stroke (MOHW104-TDU-B-212–113002) and the Ministry of Science and Technology Taiwan Clinical Trial Consortium for Stroke (MOST 105–2325-B-039 −006). Dr Silbertgleit receives research grant support from the National Institutes of Health (NIH; the University of Michigan received grants from the NIH to perform the clinical trial results reported here). Dr Hanley is a principal investigator at MISTIE trial (Minimally Invasive Surgery and rtPA for Intracerebral Hemorrhage Evacuation; Significant; >$10k or 5%), receives research grant support from MISTIE trial (NIH funded; Significant; >$10k or 5%), and is an expert witness/legal consultant (Modest; <$10k or <5%). The other authors report no conflicts.
Footnotes
The online-only Data Supplement is available with this article at http://stroke.ahajournals.Org/lookup/suppl/doi:10.1161/STROKEAHA.117.019845/-/DC1.
References
- 1.Qureshi AI, Suri MF, Nasar A, Kirmani JF, Ezzeddine MA, Divani AA, et al. Changes in cost and outcome among US patients with stroke hospitalized in 1990 to 1991 and those hospitalized in 2000 to 2001. Stroke. 2007;38:2180–2184. doi: 10.1161/STROKEAHA.106.467506. [DOI] [PubMed] [Google Scholar]
- 2.Qureshi AI. The importance of acute hypertensive response in ICH. Stroke. 2013;44(6 suppl 1):S67–S69. doi: 10.1161/STROKEAHA.111.000758. [DOI] [PubMed] [Google Scholar]
- 3.Anderson CS, Heeley E, Huang Y, Wang J, Stapf C, Delcourt C, et al. ; INTERACT2 Investigators. Rapid blood-pressure lowering in patients with acute intracerebral hemorrhage. N Engl J Med. 2013;368:2355–2365. doi: 10.1056/NEJMoa1214609. [DOI] [PubMed] [Google Scholar]
- 4.Qureshi AI, Palesch YY, Barsan WG, Hanley DF, Hsu CY, Martin RL, et al. ; ATACH-2 Trial Investigators and the Neurological Emergency Treatment Trials Network. Intensive blood-pressure lowering in patients with acute cerebral hemorrhage. N Engl J Med. 2016;375:1033–1043. doi: 10.1056/NEJMoa1603460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Qureshi AI, Hutson AD, Harbaugh RE, Stieg PE, Hopkins LN; North American Trial of Unruptured and Ruptured Aneurysms Planning Committee. Methods and design considerations for randomized clinical trials evaluating surgical or endovascular treatments for cerebrovascular diseases. Neurosurgery. 2004;54:248–264; discussion 264-247. [DOI] [PubMed] [Google Scholar]
- 6.Qureshi AI, Palesch YY, Martin R, Toyoda K, Yamamoto H, Wang Y, et al. Interpretation and Implementation of Intensive Blood Pressure Reduction in Acute Cerebral Hemorrhage Trial (INTERACT II). J Vasc Interv Neurol. 2014;7:34–40. [PMC free article] [PubMed] [Google Scholar]
- 7.Wang X, Arima H, Heeley E, Delcourt C, Huang Y, Wang J, et al. ; INTERACT2 Investigators. Magnitude of blood pressure reduction and clinical outcomes in acute intracerebral hemorrhage: intensive blood pressure reduction in acute cerebral hemorrhage trial study. Hypertension. 2015;65:1026–1032. doi: 10.1161/HYPERTENSIONAHA.114.05044. [DOI] [PubMed] [Google Scholar]
- 8.Arima H, Heeley E, Delcourt C, Hirakawa Y, Wang X, Woodward M, et al. ; INTERACT2 Investigators. Optimal achieved blood pressure in acute intracerebral hemorrhage: INTERACT2. Neurology. 2015;84:464–471. doi: 10.1212/WNL.0000000000001205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Arima H, Anderson CS, Wang JG, Huang Y, Heeley E, Neal B, et al. ; Intensive Blood Pressure Reduction in Acute Cerebral Haemorrhage Trial Investigators. Lower treatment blood pressure is associated with greatest reduction in hematoma growth after acute intracerebral hemorrhage. Hypertension. 2010;56:852–858. doi: 10.1161/HYPERTENSIONAHA.110.154328. [DOI] [PubMed] [Google Scholar]
- 10.Qureshi AI, Palesch YY. Antihypertensive Treatment of Acute Cerebral Hemorrhage (ATACH) II: design, methods, and rationale. Neurocrit Care. 2011;15:559–576. doi: 10.1007/s12028-011-9538-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lattanzi S, Cagnetti C, Provinciali L, Silvestrini M. Blood pressure variability and clinical outcome in patients with acute intracerebral hemorrhage. J Stroke Cerebrovasc Dis. 2015;24:1493–1499. doi: 10.1016/j.jstrokecerebrovasdis.2015.03.014. [DOI] [PubMed] [Google Scholar]
- 12.Tanaka E, Koga M, Kobayashi J, Kario K, Kamiyama K, Furui E, et al. Blood pressure variability on antihypertensive therapy in acute intracerebral hemorrhage: the stroke acute management with urgent risk-factor assessment and improvement-intracerebral hemorrhage study. Stroke. 2014;45:2275–2279. doi: 10.1161/STROKEAHA.114.005420. [DOI] [PubMed] [Google Scholar]
- 13.Manning L, Hirakawa Y, Arima H, Wang X, Chalmers J, Wang J, et al. ; INTERACT2 Investigators. Blood pressure variability and outcome after acute intracerebral haemorrhage: a post-hoc analysis of INTERACT2, a randomised controlled trial. Lancet Neurol. 2014;13:364–373. doi: 10.1016/S1474-4422(14)70018-3. [DOI] [PubMed] [Google Scholar]
- 14.Chan GS, Ainslie PN, Willie CK, Taylor CE, Atkinson G, Jones H, et al. Contribution of arterial Windkessel in low-frequency cerebral hemodynamics during transient changes in blood pressure. J Appl Physiol (1985). 2011;110:917–925. doi: 10.1152/japplphysiol.01407.2010. [DOI] [PubMed] [Google Scholar]
- 15.Zhang R, Behbehani K, Levine BD. Dynamic pressure-flow relationship of the cerebral circulation during acute increase in arterial pressure. J Physiol. 2009;587(pt 11):2567–2577. doi: 10.1113/jphysiol.2008.168302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang R, Zuckerman JH, Giller CA, Levine BD. Transfer function analysis of dynamic cerebral autoregulation in humans. Am J Physiol. 1998;274(1 pt 2):H233–H241. [DOI] [PubMed] [Google Scholar]
- 17.Oeinck M, Neunhoeffer F, Buttler KJ, Meckel S, Schmidt B, Czosnyka M, et al. Dynamic cerebral autoregulation in acute intracerebral hemorrhage. Stroke. 2013;44:2722–2728. doi: 10.1161/STROKEAHA.113.001913. [DOI] [PubMed] [Google Scholar]
- 18.Reinhard M, Neunhoeffer F, Gerds TA, Niesen WD, Buttler KJ, Timmer J, et al. Secondary decline of cerebral autoregulation is associated with worse outcome after intracerebral hemorrhage. Intensive Care Med. 2010;36:264–271. doi: 10.1007/s00134-009-1698-7. [DOI] [PubMed] [Google Scholar]
- 19.Majidi S, Suarez JI, Qureshi AI. Management of acute hypertensive response in intracerebral hemorrhage patients after ATACH-2 trial. Neurocrit Care. 2017;27:249–258. doi: 10.1007/s12028-016-0341-z. [DOI] [PubMed] [Google Scholar]
- 20.Hemphill JC III, Greenberg SM, Anderson CS, Becker K, Bendok BR, Cushman M, et al. ; American Heart Association Stroke Council; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2015;46:2032–2060. doi: 10.1161/STR.0000000000000069. [DOI] [PubMed] [Google Scholar]
- 21.Aronson S, Dyke CM, Stierer KA, Levy JH, Cheung AT, Lumb PD, et al. The ECLIPSE trials: comparative studies of clevidipine to nitroglycerin, sodium nitroprusside, and nicardipine for acute hypertension treatment in cardiac surgery patients. Anesth Analg. 2008;107:1110–1121. doi: 10.1213/ane.0b013e31818240db. [DOI] [PubMed] [Google Scholar]
- 22.Roitberg BZ, Hardman J, Urbaniak K, Merchant A, Mangubat EZ, Alaraj A, et al. Prospective randomized comparison of safety and efficacy of nicardipine and nitroprusside drip for control of hypertension in the neu-rosurgical intensive care unit. Neurosurgery. 2008;63:115–20; discussion 120. doi: 10.1227/01.NEU.0000335078.62599.14. [DOI] [PubMed] [Google Scholar]
- 23.Neutel JM, Smith DH, Wallin D, Cook E, Ram CV, Fletcher E, et al. A comparison of intravenous nicardipine and sodium nitroprusside in the immediate treatment of severe hypertension. Am J Hypertens. 1994;7(7 pt 1):623–628. [DOI] [PubMed] [Google Scholar]
- 24.Qureshi AI, Majidi S, Chaudhry SA, Qureshi MH, Suri MF. Validation of intracerebral hemorrhage-specific intensity of care quality metrics. J Stroke Cerebrovasc Dis. 2013;22:661–667. doi: 10.1016/j.jstrokecerebrovasdis.2012.03.015. [DOI] [PubMed] [Google Scholar]
- 25.Qureshi AI. Intracerebral hemorrhage specific intensity of care quality metrics. Neurocrit Care. 2011;14:291–317. doi: 10.1007/s12028-010-9453-z. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


