Abstract
Paracoccidioidomycosis is a granulomatous fungal infection, endemic to Latin America, which has great economic impact. Prostatic involvement is rare and can simulate other conditions, which poses a diagnostic challenge for the clinician. This article describes the report of a case of disseminated paracoccidioidomycosis with prostatic involvement, initially confused with prostatic adenocarcinoma. We hope to contribute to the knowledge of this presentation as a differential diagnosis and reinforce the importance of thorough clinical investigation.
Keywords: Paracoccidioides brasiliensis, Paracoccidiomycosis, Mycoses, Neoplasm, Prostate cancer
1. Introduction
Paracoccidioidomycosis (PCM) or South American blastomycosis is a granulomatous fungal infection, endemic to Latin America, caused by the thermodimorphic fungi Paracoccidioides brasiliensis and P. lutzii [1]. Approximately 75% of the adult Brazilian population has already been exposed to this fungus, and of these, 2% will develop the disease [2]. It is an important public health issue that has a substantial economic impact and high mortality rates when there is no early diagnosis and treatment [3].
The clinical presentation can be acute/subacute, predominant in young individuals, and chronic, in 90% of the cases [2]. The most common manifestations are pulmonary (50–100%) and mucocutaneous lesions, involving the nasal and oral region [1]. Moreover, like other systemic mycoses, PCM lesions can affect several organs depending on the host's immune status, through the dissemination of pathogens by lymphatic and hematogenic routes [4]. Systemic impairment can promote significant weight loss, asthenia, adynamia. It also has a differential diagnosis with neoplasias [5]. Therefore, given the several types of clinical presentations of PCM, similar to those of other diseases, early diagnosis of the condition can pose a challenge.
Few publications describe prostate involvement by PCM [[6], [7], [8]]. Even regarding North American blastomycosis, caused by the fungus Blastomyces dermatitidis, the descriptions in the literature are very limited. That is also the case with fungi like Coccidioidis immitis, whose more related extrapulmonary manifestations involve skin, joints and meninges [9], and Histoplasma capsulatum, which is mainly related to the reticuloendothelial organs [10].
That said, we report a case of disseminated PCM with prostatic involvement initially diagnosed as metastatic prostate adenocarcinoma, in order to emphasize the importance of gaining knowledge on the various presentations of the disease and strengthening the role of a thorough clinical investigation.
2. Case
Male patient aged 61 without evidence of immunosuppression, with previous diagnoses of controlled hypertension and chronic obstructive pulmonary disease (COPD) due to smoking without specific treatment. The patient reported weight loss of 17 kg in the last 3 months and, on investigation (on day 0), he was referred to an oncologist for investigation of neoplasia, due to images suggestive of infiltration in the lung parenchyma on chest CT (day 1). He was dyspneic, asthenic and emaciated. There were not respiratory, intestinal and urinary symptoms.
Positron emission tomography - computed tomography(PET/CT) showed an increase in glycolytic metabolism at the prostate and in the right seminal vesicle (Fig. 1), in addition to ground-glass pulmonary opacities and diffuse bilateral septal thickening (Fig. 2). Solid hypercapturing nodular lesions (right: 4.6 cm; left: 5.2 cm) in both adrenal glands and in the paravertebral muscles on the right side, suggestive of secondary neoplastic implants, according to the medical report (day 15). A diagnosis of metastatic prostate cancer in pulmonary and adrenal foci was suggested. Flexible bronchoscopy with bronchoalveolar lavage (day 17) was then requested, which obtained a negative result for the presence of fungi, mycobacteria or neoplastic cells.
Fig. 1.
PET-CT images (positron emission tomography - computed tomography) showing increased glycolytic metabolism at the prostate and in the right seminal vesicle.
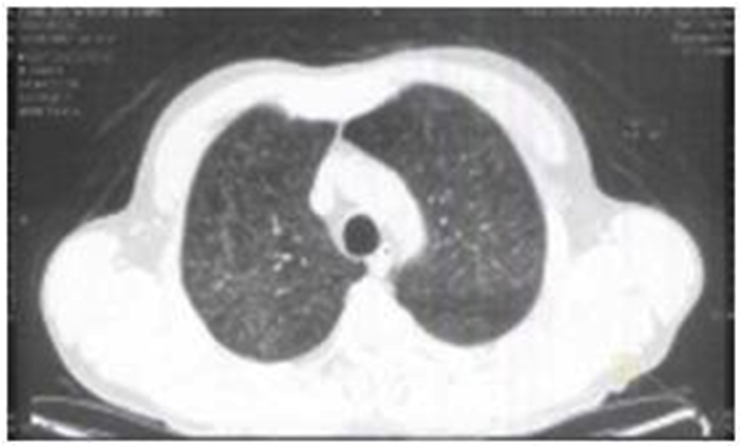
Fig. 2.
CT scan showing pulmonary opacity with ground-glass pattern and diffuse bilateral septal thickening.
After the exam, the patient experienced atrial flutter, malaise and poor peripheral perfusion, with capillary refill time greater than 3 seconds. He was transferred to an intensive care unit (ICU) due to possible post-procedure bacteremia (day 18). Electrocardiogram showed multifocal atrial rhythm with sudden conduction blocks characteristic of second-degree atrioventricular block (Mobitz 2). Drug therapy was then administered and the installation of a pacemaker was programmed.
Following the diagnostic investigation, the patient underwent lung biopsy with the use of video-assisted thoracic surgery (day 25). The material collected was sent to the laboratory for histopathology for the possible detection of the referred neoplasia. Direct tests and exam of the culture of mycobacteria and fungi were also requested and obtained negative results. The serological test was unavailable. Despite this, histopathological evaluation showed chronic granulomatous inflammation with multinucleate giant cells and necrosis. The use of the special Grocott-Gomori Methenamine Silver (GMS) staining technique to visualize fungus microscopically showed the presence of budding cells. These findings, given the local epidemiology, made it possible to confirm the diagnosis of PCM. In addition, the other foci captured by PET-CT were associated with the infectious process, data compatible with diagnosis of disseminated PCM (day 39). Other tests were conducted to investigate the causes of immunosuppression, such as HIV, use of corticosteroids, immunobiological drugs or chemotherapy and other conditions that might explain this state. All were negative.
A therapeutic scheme (day 39) for disseminated PCM was started with 5mg/kg/day of liposomal amphotericin for 15 days followed by itraconazole (400mg/day) during 12 months. Five months after the beginning of the treatment, the serological test could be performed and was not reagent for PCM. Until the submission of the paper, the patient had outpatient monitoring, being asymptomatic, with radiological remission of the lesions, and regained his usual weight.
3. Discussion
A small percentage of infected individuals have PCM, and the risk of developing the disease is related to patients' personal characteristics and lifestyle, including genetic background, age, sex, ethnicity, smoking, drinking and cellular immune dysfunction [1]. Among its clinical presentations, the chronic form is the most prevalent [2], producing respiratory symptoms such as dyspnea, cough, hemoptysis and mucocutaneous signs, such as granulomatous or ulcerated lesions in the mouth and nasopharynx [1,11]. However, the early clinical manifestations of the disease are usually nonspecific - weakness, weight loss - or absent, making early diagnosis difficult [11].
Cancer has also a multifactorial etiology. In the case described, the patient had several factors that were common to both conditions, such as old age, smoking history, pneumopathy and male gender. Therefore, he was more likely to manifest PCM, as well as to develop neoplasms. Moreover, although this is not frequent, diagnoses can coexist, and respiratory and digestive tract carcinomas are the most commonly reported [12].
Prostate cancer is the most common malignancy among the Brazilian male population, with the exception of non-melanoma skin tumors [13]. However, benign prostatic disorders should not be overlooked in differential diagnoses. Nevertheless, even when it comes to infectious etiologies, fungal prostatitis is uncommon and affects mainly immunocompromised patients [7,8]. In the case described, the patient did not have urinary symptoms. However, individuals with chronic urinary retention who are diagnosed with chronic bacterial prostatitis may be underdiagnosed, since screening for PCM is not performed [8].
Rejecting a diagnosis of lung cancer is also important in the case described, given its high incidence and lethality [13]. However, its investigation may delay the diagnosis of benign lesions that have a significant potential for cure when early identified [14]. Although the patient had several lung and extrapulmonary lesions, he was only dyspnoic, asthenic and emaciated. The presence of COPD may have masked the onset or intensification of respiratory manifestations, but a diagnosis of neoplasia is undeniably one of the first to be considered for this presentation, but neoplasia is undoubtedly one of the first diagnoses considered for this presentation. It can also be inferred that there was some degree of immunosuppression, since the disease was disseminated.
After the lungs and lymph nodes, the adrenal glands are the most affected organs. Most patients are asymptomatic, as in the case described, symptomatic adrenal insufficiency occurs when there is 80–90% involvement of the adrenal glands [15]. Thus, other fungal infections by Mycobacterium tuberculosis, Histoplasma capsulatum, Blastomyces dermatitidis or Cryptococcus neoformans should also be considered [4,16].
Imaging exams of the affected adrenal glands do not have specific characteristics, as the findings vary according to the stage of infection. However, the changes detected in this case, such as irregular shape of adrenal the gland and tendency to calcification [15], are common in the chronic involvement of the glands.
Clinical investigation of differential diagnoses is essential due to the suspicion of neoplasia, because some chronic diseases, with granulomatous inflammation or pseudoepitheliomatous hyperplasia, can mimic tumors or occur concomitantly. Thus, histological investigation through biopsy, as well as microbiological and immunological methods, are tools that significantly increase the chances of ratifying a correct diagnosis [17].
The findings of the anatomopathological examination of the patient in the present study corroborate the typical lesions found in the literature, such as chronic granulomatous inflammation and diagnosis of fungal infection using. The Grocott-Gomori Methenamine Silver (GMS) staining technique, with the presence of budding cells. Also, extensive areas of fibrosis are often found, both in the innermost layer and in the interstitial space, directly compromising the functionality of the affected structure [18]. In the referred patient, such histopathological pattern of fibrosis was not observed, which may be related to the patient's favorable prognosis, without residual organ dysfunction.
Regarding imaging exams, although CT scan allows better visualization of anatomical structures, a plain radiograph can be used in the initial investigation due to its low cost and easy access, although it expresses nonspecific patterns, because these patterns indicate the need to pursue the investigation [15]. Magnetic resonance imaging (MRI) has also been described as a potential diagnostic tool in systemic mycoses, allowing detailed evaluation and longitudinal monitoring, without exposure to ionizing radiation [19].
In view of the unspecific or obscure clinical conditions, clinical and laboratory investigation is key to diagnostic clarification. Epidemiological and occupational information, history of smoking, personal and family history, age, contact with endemic regions for systemic mycoses, especially men in rural areas; exposure to the tuberculosis bacillus; poor oral health, alcoholism and traumatic orofacial injuries are topics that should be covered in the interview questions [14]. Moreover, lung biopsy was essential to discard the initial hypothesis of neoplasia and focus on granulomatous infections of national prevalence that mimic the lesions found. In this case, prostate biopsy was not initially performed, and fungal involvement was inferred after the pulmonary result. However, the authors recognized that this fact could have delayed a diagnosis of cancer for the patient's cancer, if adenocarcinoma was concomitant.
It should be noted that the histopathological finding of epithelioid granulomas with a small amount of fungi is also common to several other conditions, and can be confused with other diseases such as Leishmaniasis, sarcoidosis and tuberculoid leprosy [20]. Thus, the experience and technical knowledge of the physician and the pathologist can be decisive for a correct diagnosis.
Although most cases of PCM evolve with residual fibrosis, restricting the functionality of the affected organs, active investigation and effective treatment provided a good recovery for the patient described here, who had no persistent limiting injuries.
PCM is the most common systemic mycosis in Brazil and has the potential to involve all organs. We therefore emphasize the need to actively investigate the symptoms and pay attention to all patient complaints, even those not associated with the most frequent sites of the disease.
Funding source
There are none.
Declaration of competing interest
The authors declare no potential conflicts of interest.
References
- 1.Martinez R. New trends in paracoccidioidomycosis epidemiology. J. Fungi. 2017;3:1–13. doi: 10.3390/jof3010001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Vermelho M.B.F., Correia A.S., Michailowsky T.C. de A., Suzart E.K.K., Ibanês A.S., Almeida L.A., Khoury Z., Barba M.F. Abdominal alterations in disseminated paracoccidioidomycosis : computed tomography findings *. Radiol. Bras. 2015;48:81–85. doi: 10.1590/0100-3984.2013.0025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shikanai-yasuda M.A., Mendes R.P., Colombo A.L., De Queiroz-telles F., Satie A., Kono G., Paniago A.M.M., Nathan A., Carlos A., Bagagli E. Brazilian guidelines for the clinical management of paracoccidioidomycosis. Rev. Soc. Bras. Med. Trop. 2017;50:715–740. doi: 10.1590/0037-8682-0230-2017. [DOI] [PubMed] [Google Scholar]
- 4.Tatagiba L.S., Pivatto L.B., Faccini-Martínez Á.A., Peçanha P.M., Velloso T.R.G., Gonçalves S.S., Rodrigues A.M., Camargo Z.P., Falqueto A. A case of paracoccidioidomycosis due to Paracoccidioides lutzii presenting sarcoid-like form. Med. Mycol. Case Rep. 2018;19:6–8. doi: 10.1016/j.mmcr.2017.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rocha-Silva F., Guimarães C.F., Júnior E.R. de O., de Figueiredo S.M., Caligiorne R.B. Disseminated paracoccidioidomycosis prediagnosticated as neoplasm : an important challenge in diagnosis using rt-PCR. Med. Mycol. Case Rep. 2018;19:1–5. doi: 10.1016/j.mmcr.2017.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Melo C.R., Melo I.S., Cersk C.T. Leukaemic infiltration , paracoccidioidomycosis and nodular hyperplasia of the prostate. Bristish J. Urol. 1992:329–330. doi: 10.1111/j.1464-410x.1992.tb15744.x. [DOI] [PubMed] [Google Scholar]
- 7.Lopes D.L., Araújo S.D.A., antos J.P.L.D.S., Lyon A.C., Dantas D.V., Reis B.S., De Góes A.M., Pedroso Ê.R.P. Prostatic paracoccidioidomycosis : differential diagnosis of prostate cancer. Mem. Inst. Oswaldo Cruz. 2009;104:33–36. doi: 10.1590/s0074-02762009000100005. [DOI] [PubMed] [Google Scholar]
- 8.de Arruda P.F.F., Gatti M., de Arruda J.G.F., Ayres D.C., Narvaes E.M., de Arruda L.F., de Godoy M.F. Prostatic paracoccidioidomycosis with a fatal outcome : a case report. J. Med. Case Rep. 2013;7:1–4. doi: 10.1186/1752-1947-7-126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Togashi R.H., Aguiar F.M.B., Ferreira D.B., de Moura C.M., Sales M.T.M., Rios N.X. Pulmonary and extrapulmonary coccidioidomycosis: three cases in an endemic area in the state of Ceará, Brazil*. J. Bras. Pneumol. 2009;35:275–279. doi: 10.1590/s1806-37132009000300013. [DOI] [PubMed] [Google Scholar]
- 10.Ferreira M.S., Borges A.S. Histoplasmose, Rev. Soc. Bras. Med. Trop. 2009;42:192–198. doi: 10.1590/s0037-86822009000200020. [DOI] [PubMed] [Google Scholar]
- 11.Canteros C.E. Paracoccidioidomicosis: crónica de una enfermedad olvidada. Med. (Buenos Aires) 2018;78:180–184. [PubMed] [Google Scholar]
- 12.Almeida J.N., Jr., Peçanha-Pietrobom P.M., Colombo A.L. Paracoccidioidomycosis in immunocompromised Patients : a literature review. J. Fungi. 2019;5:2–11. doi: 10.3390/jof5010002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bray F., Ferlay J., Soerjomataram I., Siegel R.L., Torre L.A., Jemal A. Global cancer statistics 2018 : GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Ca - Cancer J. Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 14.Homrich G.K., Andrade C.F., Marchiori R.C., Lidtke G.D.S., Martins F.P., Dos Santos J.W.A. Prevalence of benign diseases mimicking lung Cancer : experience from a university hospital of southern Brazil. Tuberc. Respir. Dis. (Seoul) 2015;78:72–77. doi: 10.4046/trd.2015.78.2.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Júnior M.R., Baldon I.V., Amorim A.F.C., Fonseca A.P.A., Volpato R., Lourenço R.B., Baptista R.M., de Mello R.A.F., Peçanha P., Falqueto A. Imaging paracoccidioidomycosis: a pictorial review from head to toe. Eur. J. Radiol. 2018 doi: 10.1016/j.ejrad.2018.03.026. [DOI] [PubMed] [Google Scholar]
- 16.Wagner G., Moertl D., Eckhardt A., Sagel U., Wrba F., Dam K., Willinger B. Chronic Paracoccidioidomycosis with adrenal involvement mimicking tuberculosis – a case report from Austria. Med. Mycol. Case Rep. 2016;14:12–16. doi: 10.1016/j.mmcr.2016.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rodrigues G. da S., Severo C.B., Oliveira F. de M., Moreira J. da S., Prolla J.C., Severo L.C. Association between paracoccidioidomycosis and cancer. J. Bras. Pneumol. 2010;36:356–362. doi: 10.1590/s1806-37132010000300014. [DOI] [PubMed] [Google Scholar]
- 18.Gazzoni F.F., Severo L.C., Marchiori E., Irion K.L., Guimarães M.D., Godoy M.C., Sartori A.P.G., Hochhegger B. Fungal diseases mimicking primary lung cancer: radiologic – pathologic correlation. Mycoses. 2014;57:197–208. doi: 10.1111/myc.12150. [DOI] [PubMed] [Google Scholar]
- 19.Marchiori E., Ferreira E.C., Zanetti G., Hochhegger B. Whole-body magnetic resonance imaging for the evaluation of thoracic involvement in disseminated paracoccidioidomycosis. J. Bras. Pneumol. 2013;39:248–250. doi: 10.1590/S1806-37132013000200019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.de Abreu M.A.M.M., Lastória J.C., Mattos A.L. de A., Alchorne M.M. de A. Paracoccidioidomycosis with sarcoid-like lesions : a diagnostic challenge. Rev. Soc. Bras. Med. Trop. 2017;50:273–276. doi: 10.1590/0037-8682-0246-2016. [DOI] [PubMed] [Google Scholar]