Abstract
The recent emergence of COVID‐19 has resulted in a worldwide crisis, with large populations locked down and transportation links severed. While approximately 80% of infected individuals have minimal symptoms, around 15–20% need to be hospitalized, greatly stressing global healthcare systems. As of March 10, the death rate appears to be about 3.4%, although this number is highly stratified among different populations. Here, we focus on those individuals who have been exposed to nicotine prior to their exposure to the virus. We predict that these individuals are ‘primed’ to be at higher risk because nicotine can directly impact the putative receptor for the virus (ACE2) and lead to deleterious signaling in lung epithelial cells.
Keywords: addiction, infectious Disease, pandemic, public Health, tobacco
Coronavirus disease 2019 (COVID‐19), an infectious respiratory disease caused by severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2), originated in Wuhan in China in December 2019. Within months, the disease had spread globally leading to a worldwide pandemic with devastating consequences for communities and public healthcare services. The degree of severity associated with the disease varies widely among different populations. Here, James Olds and Nadine Kabbini discuss the role of smoking as a primary risk factor for severe infection. Drawing on the conclusions of studies of an earlier outbreak (SARS) and analysis of recent data on COVID‐19, the authors propose that some high‐risk individuals are ‘primed’ because of exposure to nicotine, which can directly impact the putative receptor for the virus (ACE2) in lung epithelial cells.

Abbreviations
- ACE2
angiotensin‐converting enzyme‐2
- COVID‐19
Coronavirus disease 2019
- e‐cig
electronic cigarette
- RAS
renin–angiotensin system
- SARS
severe acute respiratory syndrome
- SARS‐CoV‐2
severe acute respiratory syndrome coronavirus 2
Our theory for how nicotine consumption represents a special risk factor in coronavirus disease 2019 (COVID‐19) was spurred by an earlier outbreak. In 2002, reports emerged from the Guangdong Province in southern China of a new fatal atypical pneumonia termed severe acute respiratory syndrome (SARS). Its rapid expansion throughout Southeast Asia prompted scientific efforts that identified a phylogenetically distinct coronavirus (SARS‐CoV) through genomic sequencing [1]. To date, there is no treatment for SARS, and scientists are now grappling for potential vaccines and therapeutics that can target SARS‐CoV—or its host cell components involved in viral replication—as both short‐ and long‐term therapeutic strategies.
In order to gain access to cells, CoV binds host cell receptors via their viral envelope and are internalized into the cell through what appear to be traditional clathrin‐mediated processes [2, 3]. The ACE2 gene encodes angiotensin‐converting enzyme‐2 (ACE2), which has been found to be the target receptor for both SARS‐CoV and the human respiratory coronavirus NL63. Studies indicate that ACE2 is now likely to be the host receptor for the coronavirus 2019‐nCoV/SARS‐CoV‐2 (COVID‐19) [4, 5]. ACE2 is a relatively newly described type I transmembrane metallo‐carboxypeptidase with overall homology to more classical ACE enzymes that regulate vascular tone and hormone secretion within the renin–angiotensin system (RAS) [6]. ACE2 appears to play both protective and pathogenic roles within RAS pathways, and its direct mechanisms of function in cells remain less understood [7, 8]. ACE2 is a critical mediator of RAS signaling throughout the body but particularly in the heart, lung, kidney, and gastrointestinal tract [9], which are known sites for SARS‐CoV infection. Findings now suggest that common ACE inhibitors used in the treatment of disease such as diabetes can upregulate ACE2 expression and that ACE2 can also be increased by chronic use of drugs such as thiazolidinediones and ibuprofen [10]. Thus, it is not a coincidence that many of the same symptoms that account for COVID‐19‐related illnesses and fatalities parallel those that emerge from RAS dysfunction in humans and animal models, including congestive heart failure, acute and chronic lung diseases, and cardiorenal metabolic syndrome [11, 12, 13].
In the absence of long‐term immunization or effective therapies for COVID‐19, public health management must rely on rapid responses for the identification, treatment, and management of the infection and extra care for vulnerable (high‐risk) populations. Emergent evidence supports the involvement of smoking as a key predisposing factor for COVID‐19‐related illness severity and mortality based on a recent study of 1,590 patients from 575 hospitals in 31 province/autonomous regions/provincial municipalities across China [14]. Age‐ and sex‐matched comparisons indicate that mortality and symptom severity are higher in smokers and former smokers. These findings may begin to shed light on mechanisms that account for responses of infected individuals such as the old vs. young and males vs. females in China and now elsewhere. In a recent report based on 1099 patients with COVID‐19 from 552 hospitals in 30 provinces in China, 58% of the patients were men, indicating that there might be a sex predisposition to COVID‐19, with men more prone to being affected. However, it is more likely that this sex predisposition reflects the higher smoking rate in men than in women in China (288 million men and 12.6 million women were smokers in 2018) [15].
Smoking has long been known to be a key causative agent of cardiovascular and pulmonary illnesses through its direct actions on various types of nicotinic receptors expressed in cardiac tissue, lungs, and blood vessels [16, 17, 18]. Smoking is also significantly associated with high mortality rates in infections of various respiratory viruses including those that underlie annual (seasonal) influenza [19, 20]. Interaction between nicotine exposure, nicotinic receptor signaling, and modulation of the RAS has been recognized, yet remains understudied. In this case, however, smoking appears to participate in a direct cellular process that effects COVID‐19 infection and possible outcome, in a mechanism involving the ability of the nicotinic receptor to regulate ACE2 protein expression in cells [21, 22, 23]. Smoking is also known to cause lung damage through the activation of inflammatory cytokines and programmed cell death in the pulmonary tissue and direct actions on circulating immune cells such as T cell [24]. Of particular interest, lung AT2 cells exposed to nicotine show altered expression of the ACE2 protein that may underlie enhanced exposure of the putative receptor to COVID‐19 spike protein, and recent analysis of a large dataset from RNA‐seq and DNA microarray supports the finding that smoking is associated with increased ACE2 expression in the lung [25]. Prolonged nicotine exposure systemically—through various kinds of smoking habits—may thus provide a cellular mechanism for viral susceptibility and illness severity during the course of the infection in the lungs as well as other organ systems (Figure 1).
Fig. 1.

A schematic model for how nicotine exposure augments risk of COVID‐19 entry into the human host lung. (A) Pulmonary and immune responses to COVID‐19 infection in epithelial cells of smokers (right) and nonsmokers (left). (B) Cellular mechanisms of nicotinic receptor activity that promotes COVID‐19 entry and proliferation in epithelial cells through co‐expression of ACE2. Nicotine activation of nicotinic receptors can lead to enhanced protease activation, cell death (apoptosis), and inflammatory signaling through mechanisms that converge on ACE2 regulation and signaling.
Tobacco formulations are not just nicotine and often contain a varied mixture of >5000 chemicals, with potential carcinogenic, cardiovascular, and respiratory properties [26, 27]. In addition to nicotine, cigarettes contain toxins such as carbon monoxide and polycyclic aromatic hydrocarbons, which also perturb the function of the cardiovascular, pulmonary, and immune systems, and at this point, such toxins may also contribute to COVID‐19 disease outcome. Chronic nicotine use, systemically through cigarette and other tobacco products or indirectly possibly through secondhand smoke, now presents an important factor in COVID‐19 vulnerability in various populations. Alarming increases in nicotine consumption and mortalities throughout Asia (Figure 2) and rises in the distribution of electronic cigarette (e‐cig) formulations worldwide point to augmenting vulnerability to respiratory infecting viruses such as COVID‐19 [28]. With more data rapidly emerging in various countries and locations, it will be important to critically consider evidence between virus‐related infection and illness as well as resilience in relation to nicotine consumption. These findings aim to prioritize public health efforts of identifying populations at risk and aid in venues for critical intervention.
Fig. 2.
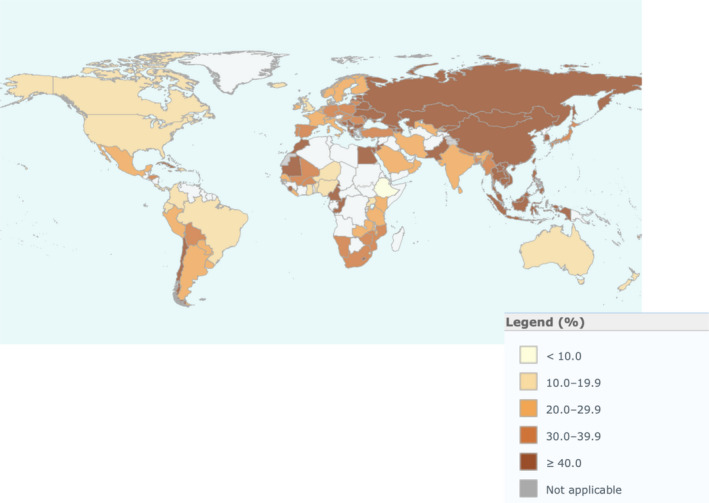
World Health Organization data on global smoking prevalence (percentage of smokers of overall population) segmented by country. Note how China, South Korea, and Italy represent local ‘hot spots’ for smoking of nicotine cigarettes. These nations were also early epicenters for COVID‐19. If our hypothesis is correct, nicotine consumption heterogeneities in Latin America and Africa predict future health challenges for at‐risk populations as the epidemic proceeds.
Summary
Cigarette smoking or nicotine inhalation in volunteers correlates with an acute increase in systolic and diastolic blood pressure as well as increased plasma ACE activity.
In many lung cells (including bronchial epithelial cells, alveolar macrophages, pulmonary endothelial cells, and interstitial fibroblasts), nicotinic receptors are co‐expressed with most components of the RAS.
ACE2‐mediated activation of the JAK/STAT pathway drives epigenetic changes that underlie lung damage through inflammation and protease activation. These pathways are also activated by nicotinic receptors.
Studies on the effects of e‐cigs on RAS are inconclusive, and thus, it is not yet clear how the use of these devices will impact infection and prognosis in COVID‐19 cases.
Interaction sites between ACE2 and SARS‐CoV have been identified at the atomic level and may provide a promising target site for antibodies or small molecules.
Conflict of interest
The authors declare no conflict of interest.
References
- 1. Li W, Kuhn JH, Moore MJ, Wong SK, Huang IC, Farzan M, Zhang C, Luo S, Xu K, Guan Y et al. (2005) Receptor and viral determinants of SARS‐coronavirus adaptation to human ACE2. EMBO J 24, 1634–1643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Inoue Y, Tanaka N, Tanaka Y, Inoue S, Morita K, Zhuang M, Hattori T & Sugamura K (2007) Clathrin‐dependent entry of severe acute respiratory syndrome coronavirus into target cells expressing ACE2 with the cytoplasmic tail deleted. J Virol 81, 8722–8729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Wang H, Yang P, Liu K, Guo F, Zhang Y, Zhang G & Jiang C (2008) SARS coronavirus entry into host cells through a novel clathrin‐ and caveolae‐independent endocytic pathway. Cell Res 18, 290–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Yan R, Zhang Y, Guo Y, Xia L & Zhou Q (2020) Structural basis for the recognition of the 2019‐nCoV by human ACE2. bioRxiv. 10.1101/2020.02.19.956946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Liu Z, Xiao X, Wei X, Li J, Yang J, Tan H, Zhu J, Zhang Q, Wu J & Liu L (2020) Composition and divergence of coronavirus spike proteins and host ACE2 receptors predict potential intermediate hosts of SARS‐CoV‐2. J Med Virol. 10.1002/jmv.25726 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Crackower MA, Sarao R, Oliveira‐dos‐Santos AJ, Da Costa J & Zhang L (2002) Angiotensin‐converting enzyme 2 is an essential regulator of heart function. Nature 417, 822–828. [DOI] [PubMed] [Google Scholar]
- 7. Kuba K, Imai Y & Penninger JM (2006) Angiotensin‐converting enzyme 2 in lung diseases. Curr Opin Pharmacol 6, 271–276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, Huan Y, Yang P, Zhang Y, Deng W et al. (2005) A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus‐induced lung injury. Nat Med 11, 875–879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Imai Y, Kuba K, Rao S, Huan Y, Guo F, Guan B, Yang P, Sarao R, Wada T, Leong‐Poi H et al. (2005) Angiotensin‐converting enzyme 2 protects from severe acute lung failure. Nature 5, 3594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Fang L, Karakiulakis G & Roth M (2020) Are patients with hypertension and diabetes mellitus at increased risk for COVID‐19 infection? Lancet Respir. 10.1016/S2213-2600(20)30116-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Guo Y, Korteweg C, McNutt MA & Gu J (2008) Pathogenetic mechanisms of severe acute respiratory syndrome. Virus Res 133, 4–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Yang P, Gu H, Zhao Z, Wang W, Cao B, Lai C, Yang X, Zhang LY, Duan Y, Zhang S et al. (2014) Angiotensin‐converting enzyme 2 (ACE2) mediates influenza H7N9 virus‐induced acute lung injury. Sci Rep 4, 7027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Mao C, Wu J, Xiao D, Lv J, Ding Y, Xu Z & Zhang L (2009) The effect of fetal and neonatal nicotine exposure on renal development of AT1 and AT2 receptors. Reprod Toxicol 27, 149–154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Guan W, Liang W, Zhao Y, Liang H, Chen Z, Li Y, Liu X, Chen R, Tang C, Wang T et al. (2020) Comorbidity and its impact on 1,590 patients with COVID‐19 in China: a nationwide analysis. medRxiv. 10.1101/2020.02.25.20027664 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Cai H (2020) Sex difference and smoking predisposition in patients with COVID‐19. Lancet Respir Med. 10.1016/S2213-2600(20)30117-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Changeux J‐P (2010) Nicotine addiction and nicotinic receptors: lessons from genetically modified mice. Nat Rev Neurosci 11, 389–401. [DOI] [PubMed] [Google Scholar]
- 17. Freitas K, Ghosh S, Ivy Carroll F, Lichtman AH & Imad Damaj M (2013) Effects of alpha 7 positive allosteric modulators in murine inflammatory and chronic neuropathic pain models. Neuropharmacology 65, 156–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Harwani SC, Ratcliff J, Sutterwala FS, Ballas ZK, Meyerholz DK, Chapleau MW & Abboud FM (2016) Nicotine mediates CD161a+ renal macrophage infiltration and premature hypertension in the spontaneously hypertensive rat. Circ Res 119, 1101–1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Altzibar JM, Tamayo‐Uria I, De Castro V, Aginagalde X, Albizu MV, Lertxundi A, Benito J, Busca P, Antepara I, Landa J et al. (2015) Epidemiology of asthma exacerbations and their relation with environmental factors in the Basque Country. Clin Exp Allergy 45, 1475. [DOI] [PubMed] [Google Scholar]
- 20. Watanabe T (2013) Renal complications of seasonal and pandemic influenza A virus infections. Eur J Pediatr 172, 15–22. [DOI] [PubMed] [Google Scholar]
- 21. Ferrari MFR, Raizada MK & Fior‐Chadi DR (2007) Nicotine modulates the renin‐angiotensin system of cultured neurons and glial cells from cardiovascular brain areas of Wistar Kyoto and spontaneously hypertensive rats. J Mol Neurosci 33, 284–293. [DOI] [PubMed] [Google Scholar]
- 22. Oakes JM, Fuchs RM, Gardner JD, Lazartigues E & Yue X (2018) Nicotine and the renin‐angiotensin system. Am J Physiol Regul Integr Comp Physiol 315, R895–R906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Yue X, Basting TM, Flanagan TW, Xu J, Lobell TD, Gilpin NW, Gardner JD & Lazartigues E (2018) Nicotine downregulates the compensatory angiotensin‐converting enzyme 2/angiotensin type 2 receptor of the renin‐angiotensin system. Ann Am Thorac Soc 15, S126–S127. [Google Scholar]
- 24. Nordman JC, Muldoon P, Clark S, Damaj MI & Kabbani N (2014) The α4 nicotinic receptor promotes CD4+ T‐Cell proliferation and a helper T‐cell immune response. Mol Pharmacol 85, 50–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Cai G (2020) Tobacco‐Use Disparity in Gene Expression of ACE2, the Receptor of 2019‐nCov. medRxiv, Preprints. 10.20944/preprints202002.0051.v1 [DOI] [Google Scholar]
- 26. Czogala J, Goniewicz ML, Fidelus B, Zielinska‐Danch W, Travers MJ & Sobczak A (2014) Secondhand exposure to vapors from electronic cigarettes. Nicotine Tob Res 16, 655–662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Anderson R, Meyer PWA, Ally MMTM & Tikly M (2016) Smoking and air pollution as pro‐inflammatory triggers for the development of rheumatoid arthritis. Nicotine Tob Res 18, 1556–1565. [DOI] [PubMed] [Google Scholar]
- 28. Garcia‐Arcos I, Geraghty P, Baumlin N, Campos M, Dabo AJ, Jundi B, Cummins N, Eden E, Grosche A, Salathe M et al. (2016) Chronic electronic cigarette exposure in mice induces features of COPD in a nicotine‐dependent manner. Thorax 71, 1119–1129. [DOI] [PMC free article] [PubMed] [Google Scholar]


