Abstract
Objectives
The aim of this study was to identify the parameters that predict the risk of future foot ulcer occurrence in patients with diabetes.
Research design and methods
1810 (male (M)/female (F): 1012/798) patients, with no foot ulcer at baseline, participated in this study. Data from a set of 28 parameters were collected at baseline. During follow-up, 123 (M/F: 68/55) patients ulcerated. Survival analyses together with logistic regression were used to identify the parameters that could predict the risk of future diabetic foot ulcer occurrence.
Results
A number of parameters (HR (95% CI)) including neuropathy (2.525 (1.680 to 3.795)); history of ulceration (2.796 (1.029 to 7.598)); smoking history (1.686 (1.097 to 2.592)); presence of callus (1.474 (0.999 to 2.174)); nail ingrowth (5.653 (2.078 to 15.379)); foot swelling (3.345 (1.799 to 6.218)); dry skin (1.926 (1.273 to 2.914)); limited ankle (1.662 (1.365 to 2.022)) and metatarsophalangeal (MTP) joint (2.745 (1.853 to 4.067)) ranges of motion; and decreased (3.141 (2.102 to 4.693)), highly decreased (5.263 (1.266 to 21.878)), and absent (9.671 (5.179 to 18.059)) sensation to touch; age (1.026 (1.010 to 1.042)); vibration perception threshold (1.079 (1.060 to 1.099)); duration of diabetes (1.000 (1.000 to 1.000)); and plantar pressure at the first metatarsal head (1.003 (1.001 to 1.005)), temperature sensation (1.019 (1.004 to 1.035)) and temperature tolerance (1.523 (1.337 to 1.734)) thresholds to hot stimuli and blood sugar level (1.027 (1.006 to 1.048)) were all significantly associated with increased risk of ulceration. However, plantar pressure underneath the fifth toe (0.990 (0.983 to 0.998)) and temperature sensation (0.755 (0.688 to 0.829)) and temperature tolerance (0.668 (0.592 to 0.0754)) thresholds to cold stimuli showed to significantly decrease the risk of future ulcer occurrence. Multivariate survival model indicated that nail ingrowth (4.42 (1.38 to 14.07)); vibration perception threshold (1.07 (1.04 to 1.09)); dry skin status (4.48 (1.80 to 11.14)); and temperature tolerance threshold to warm stimuli (1.001 (1.000 to 1.002)) were significant predictors of foot ulceration risk in the final model. The mean time to ulceration was significantly (p<0.05) shorter for patients with: dry skin (χ2=11.015), nail ingrowth (χ2=14.688), neuropathy (χ2=21.284), or foot swelling (χ2=16.428).
Conclusion
Nail ingrowth and dry skin were found to be strong indicators of vulnerability of patients to diabetic foot ulceration. Results highlight that assessments of neuropathy in relation to both small and larger fiber impairment need to be considered for predicting the risk of diabetic foot ulceration.
Keywords: foot biomechancis, foot ulcer, neuropathy, Africans
Significance of this study.
What is already known about this subject?
Insensitivity to a 10 g monofilament or one absent pedal pulse was reported to be adequate to identify patients with moderate risk of foot ulceration, while history of diabetic foot ulcer (DFU) or lower-extremity amputations was found to allow identifying those at high risk of developing DFU.
What are the new findings?
Presence of dry skin or nail ingrowth increases the likelihood of future incident of DFU by more than four times.
Impaired sensation to vibration, measured as vibration perception threshold, was significantly associated with increased risk of DFU.
Decreased sensation to touch was associated with future incidents of DFU and can be used for DFU risk stratification.
Increased tolerance thresholds to temperature stimuli were associated with increased likelihood of future ulceration.
How might these results change the focus of research or clinical practice?
Nail ingrowth and dry skin were found to be strong indicators for vulnerability of patients to future diabetic foot ulceration as presence of each showed to increase the ulceration risk by more than four times. Assessment of neuropathy in relation to both small and large fiber impairment needs to be considered for predicting the risk of diabetic foot ulceration.
Introduction
With a prevalence rate of between 15% and 34% in persons with diabetes,1 2 diabetic foot ulcer (DFU) is the main cause of non-traumatic lower limb amputation worldwide.1 Approximately 50% of diabetic ulcers become infected3 and 20% of moderate or severe diabetic foot infections lead to amputation.4 5 The presence of DFU in patient increase the risk of death at 5 years by 2.5 times.6 The prevalence of DFU in African countries is 4%–19%7 imposing heavy burden on health systems.8 While the clinical presentation of diabetic lesions in Tanzanian populations was previously reported,9 to decrease the socioeconomic cost associated with diabetic foot complications, a population-based DFU risk assessment tool needs to be developed that can identify patients vulnerable to future DFU occurrence.
There has been an abundance of studies focusing on the predictive factors for diabetic foot ulceration with the majority indicating the multifactorial nature of DFUs and their link to a number of different parameters. Systematic reviews of the literature indicated that impaired sensation, peripheral vascular disease indicated by ankle brachial index (ABI), vibration perception threshold (VPT), peak plantar pressure, foot deformities, and fasting blood sugar level were identified as risk factors for DFU.10 11 Furthermore, other easily identifiable parameters like age, duration of diabetes, height, body weight, and body mass index (BMI) have been associated with the risk of DFU occurrence.10 11
In addition, studies indicated that peak plantar pressure,12 ankle range of motion,13 and hallux limitus14 were associated with an increased risk of DFU occurrence. Although impaired thermal sensation, which is clinically associated with small fiber neuropathy has also been previously linked to the presence of neuropathic DFU,15 the value of this parameter in predicting the risk of DFU occurrence has not been previously established in a large cohort of patients.
A previous systematic review of the risk stratification systems for diabetic foot ulceration identified: (A) foot deformity, (B) peripheral neuropathy (VPT or cutaneous insensitivity to monofilament), (C) peripheral arterial disease (pulses and/or ABI), (D) previous amputation, (E) the presence of callus, (F) hemoglobin A1c, (G) tinea pedis, and (H) onychomycosis as factors that are commonly used to predict the risk of ulceration.10
A recent systematic review of literature and meta-analysis reported insensitivity to a 10 g monofilament or one absent pedal pulse as prognostic factors to identify patients with moderate or intermediate risk of foot ulceration.11 While history of DFUs or lower-extremity amputations was reported to be sufficient to identify those at high risk of developing DFU.11 Despite these, all reviewed studies in these systematic reviews were focused on western population.16 Hence there is a scarcity of studies which highlights the characteristics of patients who are vulnerable to future DFU occurrence in other populations. This is particularly evident for an African population. In addition, there is a dearth of studies in which a combination of foot-related and generic clinical parameters is investigated. Furthermore, the role of these parameters in identifying patients with further likelihood and higher risk of developing diabetic foot ulceration incident has not adequately been identified.
The overall aim of this study was to identify a set of risk factors that could be used to predict the future DFU occurrence in patients with diabetes from Tanzania. The first objective of this study is to identify the differences in the characteristics of patients with (against patients without) future DFU occurrence. The second objective of this study was to identify the parameters that identify the risk (hazard) and likelihood (odds) of future DFU occurrence and to propose a model that can predict the risk of future DFU occurrence in this group of patients.
Methods
Participants
Ethical approval was granted by the local ethics committee. Participants were recruited from patients who attended the clinic between January 2011 and December 2015.
All data were collected in a specialist clinic located within a city. This clinic had a comprehensive outpatient capacity and is one of the main diabetic foot clinics in the Eastern, Western and Central Africa with a focus on foot complication as a result of diabetes. The primary inclusion criteria was that the patient being diagnosed with diabetes. The main exclusion criterion was the presence of any DFU at baseline. DFU was defined as a full-thickness wound involving the foot or the ankle, distal to and including the malleoli.
The sample size was calculated as 1584 participants based on the diabetic foot ulceration rate of 7% in diabetes population in Africa17 with alpha level of 5% and power of 95%. Assuming a missing data in 1 out of 8 participants, an additional 227 participants were recruited.
Data collection
A set of 28 categorical and continuous parameters were collected from the patients during a single visit at baseline.
Categorical parameters
The general categorical parameters were: smoking (current smoker, never smoked, previous smoker), alcohol habits (currently drinks, never drunk, in the past), previous amputation, and history of ulceration according to protocols set by International Working Group on the Diabetic Foot (IWGDF).18
The foot-specific categorical parameters included: neuropathy and touch sensation tests. Neuropathy was assessed using 10 g monofilament19 on both feet at 10 sites including hallux, third toe, fifth toe, first metatarsal head (MTH), third MTH, fifth MTH, lateral midfoot, medial midfoot, center of the hindfoot, and dorsum of the foot.20 The touch sensation was assessed using Ipswich touch test that involved lightly touching/resting the tip of the index finger for 1–2 s on the tips of the first, third, and fifth toes.21 Touch sense status was defined: normal as 0 (zero) insensate sites, decreased as 1–3 insensate sites, highly decreased as 4–5 insensate sites, and absent as 6 insensate sites from the total 6 sites tested.
Foot deformity was assessed as structural abnormalities in the foot such as claw/hammer toe and hallux valgus or prominent MTHs, status after Charcot foot, amputations and other foot surgery were considered as having foot deformity.14 Skin status was considered as: dry when epidermis lacks moisture or sebum, and normal: well-balanced skin eudermic that is neither too oily nor too dry.22 23 Dry skin is very common in patients with diabetes where the skin lacks adequate moisture. On visual examination dry skin looks rough, shows signs of itching, have flaking, scaling or peeling. Fine lines and crack can be seen on the skin, which leads to deep cracks that may bleed and ulcers. In addition, the skin becomes gray and ashy. Mycosis was considered as fungal infection in between the toes and macerated skin.22
Nail ingrowth was considered as in-growing toe nail (also known as onychocryptosis) and it was considered as present when the nail grows so that it cuts into one or both sides of the paronychium or nail bed.22 Swelling was considered as present when swelling of foot sufficiently pronounced to leave a clear imprint of the pressure by a finger.22 Presence of callus was also considered to be present based on the protocol proposed in IWGDF guidelines.22 Specific categorical parameters for each participant were defined as if these occurred on either or both feet for each participant.
Continuous parameters
The general continuous parameters included: age, weight, height, duration of diabetes, blood glucose level, and BMI. The foot-specific continuous parameters were: ankle brachial pressure index, vibration perception threshold (VPT), temperature sensation threshold (TST) and temperature tolerance threshold (TTT), and plantar pressures during walking. VPT was measured using a clinically accepted device (Neuropathy Analyser—Vibrotherm—Dx; Diabetik Foot Care India, Chennai, India) at the wrist, knee, ankle, and big toe according to the previous protocol.24 This device was also used to measure the TST and TTT to both cold/warm stimuli at: hallux, third toe, fifth toe, underneath the arch and heel according to a previous protocol.25
A plantar pressure platform (EMED, Novel, Munich, Germany) was used to measure average peak plantar pressure during the stance phases of walking at 16 sites (hallux, second toe, third toe, fourth toe, fifth toe, first MTH, second MTH, third MTH, fourth MTH, fifth MTH, lateral midfoot, central midfoot, medial midfoot, lateral hindfoot, medial hindfoot, center of the hindfoot), based on a previous protocol.26 The participants were asked to walk over the platform using a two-step protocol27 after completing a number of familiarization trials. The mean of peak pressures from three stance phases from each foot was calculated based on which the overall and regional pressures were reported.28 All specific continuous parameters were averaged between the left and right feet.
Data analyses
All statistical tests were performed using IBM SPSSV.25.
Test of differences
Chi-square test for independence with Yates Continuity Correction was used to identify significant (p<0.05) association between categorical parameters and the future occurrence of DFU. For two-category parameters the effect sizes were determined based on Phi coefficient, where 0.01, 0.3, and 0.5 represent small, medium, and large effect sizes, respectively. For three-category parameters the effect sizes were determined based on the value of Cramer’s V coefficient, where 0.07, 0.21, and 0.35 represent small, medium, and large effect sizes, respectively. For four-category parameters the effect sizes were determined based on Cramer’s V coefficient, where 0.06, 0.17, and 0.29 represent small, medium, and large effect sizes, respectively.
Furthermore, given the non-normal distribution of the data which was established through the test of normality (Kolmogorov-Smirnov, p<0.05), Mann-Whitney U test was used to assess significant (p<0.05) differences in continuous parameters between the patients with and without future ulceration. Mann-Whitney U test identifies the effect size based on the values of r=z/(N1+N2)0.5 where 0.1, 0.3, and 0.5 represent small, medium, and larger effect sizes, respectively.
Assessment of the contribution of parameters to predicting the survival (ulcer-free status) and occurrence (incident) of DFUs
Cox univariate regression was used to assess the contribution of each of the categorical and continuous parameters to predicting the risk of DFU occurrence (HR). In addition, Kaplan-Meier survival analyses were used to compare the differences in survival time (ulcer-free time) for categorical parameters. Univariate logistic regression was also used to assess if each of the parameters contributed to predicting future DFU likelihood (OR) independent of the time of occurrence.
The logistic regression has the capability to identify the parameters that contribute to predicting the likelihood of ulcer occurrence. This is independent of the time of occurrence and is reported as ORs. This ratio indicates the likelihood of ulcer occurrence. On the other hand, the survival analyses identify the parameters that contribute to predicting the ulcer occurrence considering the time to occurrence. This analysis includes the observation time (from baseline) for the ulcer to occur or the observation time (from baseline) till the end of follow-up (ulcer-free survival). This is reported as HRs, which indicates the risk of ulceration.
Predicting the foot ulceration risk
Cox proportional hazard multiple regression analysis was used to find the minimum set of contributory factors that can predict the future diabetic foot ulceration occurrence with the highest prediction power.
To identify the effect of each set of parameters on the explanatory accuracy of the model, the covariates were entered through consecutive blocks of covariates as shown in our previous study.29
In each block of parameters, an automated backward stepwise selection algorithm (retaining variables with p<0.05 removal testing is based on the probability of the Wald statistic) was used to arrive at the Cox proportional hazard multiple regression model. The collinearity between independent parameters in the same block was taken care of by the automated backward stepwise selection algorithm.
Cox proportional hazard multiple regression was also used to calculate HRs for each of the independent variables that stayed in the model. Chi-square and significance level were used to indicate how worthwhile the model was in predicting the future ulceration. Furthermore, the area under the receiver operating characteristic curve with 95% confidence level was calculated and referred to as the diagnosis strength of the model.
Results
A total of 1810 (male (M)/female (F): 1012/798) patients with non-ulcerated feet were recruited and data were collected at the baseline. Then patients were followed for median of 133 days (range of 2904 days) until their first ulcer occurrence or until they were censored. One hundred and twenty-three patients (M/F: 68/55) ulcerated and 1687 (M/F: 944/743) were ulcer free by the end of follow-up.
Tables 1–4 represent the results related to the categorical and continuous parameters. Although there were no missing data for the categorical and for most of continuous parameters, the missing data were accounted for in all statistical analyses, as indicated in tables 1–4.
Table 1.
Categorical parameters including the previous ulceration, callus and amputation history along with the lifestyle factors like smoking and drinking habits for all participants and for each group with and with no future ulceration occurrence.
| Categorical variable | All (1810) | No ulcer occurrence (1687 (93.2%)) | With ulcer occurrence (123 (6.8%)) | Survival analysis Cox univariate regression | Univariate logistic regression | Chi-square test of independence | Kaplan-Meier survival analysis | ||||||
| Count | % | Count | % | Count | % | HR (95% CI)* |
P value Cox survival analysis * | OR (95% CI)† | P value univariate logistic regression† | P value for differences‡ | Estimated mean reduction in survival (days)§ | P value for reduction in survival § | |
| Male | 1012 | 55.9 | 944 | 56.0 | 68 | 53.3 | 1.023 (0.717 to 1.460) | 0.898 | 0.973 (0.673 to 1.406) | 0.885 | 0.959 | 2 | 0.899 |
| Previous ulceration | 14 | 0.8 | 10 | 0.6 | 4 | 3.3 | 2.796 (1.029 to 7.598) | 0.035 | 5.624 (1.738 to 18.198) | 0.004 | 0.007 | 869 | 0.035 |
| Amputation | 6 | 0.3 | 5 | 0.3 | 1 | 0.8 | 1.851 (0.258 to 13.263) | 0.534 | 2.757 (0.320 to 23.788) | 0.356 | 0.881 | 875 | 0.534 |
| Presence of callus | 297 | 16.4 | 260 | 15.4 | 37 | 30.1 | 1.474 (0.999 to 2.174) | 0.049 | 2.361 (1.571 to 3.549) | 0.000 | 0.000 | 191 | 0.049 |
| No smoking | 1404 | 77.6 | 1313 | 77.8 | 91 | 74.0 | 0.209 | ||||||
| Past smoker | 300 | 16.6 | 273 | 16.2 | 27 | 22.0 | 1.686 (1.097 to 2.592) | 0.017 | 1.427 (0.911 to 2.235) | 0.121 | 324 | 0.016 | |
| Current smoker | 106 | 5.9 | 101 | 6.0 | 5 | 4.1 | 0.754 (0.306 to 1.856) | 0.539 | 0.714 (0.284 to 1.797) | 0.475 | 57 | 0.546 | |
| Never consumed alcohol | 996 | 55.0 | 928 | 55.0 | 68 | 55.3 | 0.351 | ||||||
| Used to consume alcohol in the past | 568 | 31.4 | 525 | 31.1 | 43 | 35.0 | 1.319 (0.898 to 1.936) | 0.158 | 1.118 (0.752 to 1.662) | 0.582 | 90 | 0.161 | |
| Currently consume alcohol | 246 | 13.6 | 234 | 13.9 | 12 | 9.8 | 0.720 (0.389 to 1.330) | 0.294 | 0.700 (0.373 to 1.314) | 0.267 | 109 | 0.299 | |
| Dry skin | 1623 | 89.7 | 1506 | 89.3 | 117 | 95.1 | 1.926 (1.273 to 2.914) | 0.001 | 2.344 (1.017 to 5.400) | 0.046 | 0.076 | 470 | 0.001 |
P values <0.05 indicate a significant association and are shown in bold in the table.
*Based on univariate Cox survival analysis.
†Based on univariate logistic regression.
‡P values based on χ2 test of independence (with Yates continuity correction).
§Based on Kaplan-Meier survival analysis.
Table 2.
Categorical parameters indicating neuropathy, nail ingrowth, mycosis, foot deformity along with biomechanical factors like foot swelling and foot and ankle joint mobilities for all participants and for each group of with and with no future ulceration occurrence
| Categorical variable | All (1810) | No-ulcer occurrence (1687 (93.2%)) | With ulcer occurrence (123 (6.8%)) | Survival analysis Cox univariate regression | Univariate logistic regression | Chi-square test of independence | Kaplan-Meier survival analysis | ||||||
| Count | % | Count | % | Count | % | HR (95% CI)* |
P-value Cox survival analysis * | OR (95% CI)† | P-value univariate logistic regression† | P value for differences‡ | Estimated mean reduction in survival- days§ | P value for reduction in survival§ | |
| Neuropathy | 938 | 52.0 | 846 | 50.1 | 92 | 74.8 | 2.525 (1.680 to 3.795) | 0.000 | 2.926 (1.926 to 4.444) | 0.000 | 0.000 | 314 | 0.000 |
| Foot deformity | 23 | 1.3 | 22 | 1.3 | 1 | 0.8 | 0.797 (0.111 to 5.712) | 0.821 | 0.625 (0.084 to 4.677) | 0.647 | 0.965 | 557 | 0.821 |
| Mycosis | 42 | 2.3 | 38 | 2.3 | 4 | 3.3 | 1.003 (0.370 to 2.719) | 0.996 | 1.459 (0.512 to 4.156) | 0.480 | 0.689 | 241 | 0.996 |
| Nail ingrowth | 9 | 0.5 | 5 | 0.3 | 4 | 3.3 | 5.653 (2.078 to 15.379) | 0.000 | 11.308 (2.997 to 42.663) | 0.002 | 0.000 | 1393 | 0.000 |
| Foot swelling | 55 | 3.0 | 44 | 2.6 | 11 | 8.9 | 3.345 (1.799 to 6.218 | 0.000 | 3.667 (1.843 to 7.296) | 0.001 | 0.000 | 831 | 0.000 |
| Limited ankle mobility | 275 | 15.2 | 239 | 14.2 | 36 | 29.3 | 1.662 (1.365 to 2.022) | 0.000 | 2.552 (1.689 to 3.856) | 0.000 | 0.000 | 850 | 0.000 |
| Limited metatarsophalangeal (MTP) joint mobility | 273 | 15.1 | 237 | 14.0 | 36 | 29.3 | 2.745 (1.853 to 4.067) | 0.000 | 2.527(1.673 to 3.818) | 0.000 | 0.000 | 848 | 0.000 |
| Normal touch sensation | 1093 | 60.5 | 1057 | 62.8 | 36 | 29.3 | 0.000 | ||||||
| Decreased touch sensation | 631 | 34.9 | 560 | 33.3 | 71 | 57.7 | 3.141 (2.102 to 4.693) | 0.000 | 3.723 (2.461 to 5.631) | 0.000 | 468 | 0.000 | |
| Highly decreased touch sensation | 73 | 4.0 | 59 | 3.5 | 14 | 11.4 | 5.263 (1.266 to 21.878) | 0.022 | 6.967 (3.563 to 13.625) | 0.000 | 1442 | 0.000 | |
| Absent sensation | 10 | 0.6 | 8 | 0.5 | 2 | 1.6 | 9.671 (5.179 to 18.059) | 0.000 | 7.340 (1.505 to 35.804) | 0.014 | 1128 | 0.000 | |
P values <0.05 indicate a significant association and are shown in bold in the table.
*Based on univariate Cox survival analyses.
†Based on univariate logistic regression.
‡P values based on χ2 test of independence (with Yates continuity correction).
§Based on Kaplan-Meier survival analysis.
Table 3.
Continuous parameters including age, duration of diabetes, weight, height, BMI, blood sugar level, and ABI along with the parameters indicating neuropathy for all participants and for the groups with and with no future ulceration occurrence
| Continuous parameter | All | No-ulcer occurrence | With ulcer occurrence | Survival analysis Cox univariate regression | Mann-Whitney U test | Univariate logistic regression | ||||||||
| Median | Range | n‡ | Median | Range | n‡ | Median | Range | n‡ | HR (95% CI)* |
P- survival analysis * | P- differences† | OR (95% CI)‡ | P-value logistic regression‡ | |
| Age (years) | 52 | 90 | 1810 | 52 | 90 | 1687 | 54 | 60 | 123 | 1.026 (1.010 to 1.042) | 0.001 | 0.011 | 1.019 (1.003 to 1.035) | 0.019 |
| Height (m) | 1.6 | 0.8 | 1809 | 1.6 | 0.7 | 1686 | 1.6 | 0.6 | 123 | 0.928 (0.125 to 6.858) | 0.941 | 0.923 | 1.175 (0.157 to 8.775) | 0.875 |
| Weight (kg) | 73.2 | 120 | 1807 | 73.2 | 120 | 1684 | 73.0 | 77 | 123 | 0.992 (0.981 to 1.003) | 0.157 | 0.664 | 0.997 (0.985 to 1.008) | 0.582 |
| BMI (kg/m2) | 29.1 | 58.6 | 1810 | 29.1 | 58.6 | 1687 | 28.9 | 34.6 | 123 | 0.984 (0.955 to 1.014) | 0.293 | 0.760 | 0.995(0.966 to 1.024) | 0.722 |
| Ankle brachial index | 1.1 | 1.8 | 1810 | 1.1 | 1.8 | 1687 | 1.1 | 1.2 | 123 | 0.499 (0.142 to 1.750) | 0.531 | 0.205 | 0.247 (0.073 to 0.834) | 0.023 |
| Vibration perception threshold average (V) | 20 | 48 | 1810 | 20 | 48 | 1687 | 30 | 41 | 123 | 1.079 (1.060 to 1.099) | 0.000 | 0.000 | 1.073 (1.054 to 1.092) | 0.000 |
| Duration of diabetes (days) | 1095 | 18 980 | 1810 | 1095 | 18 980 | 1687 | 2555 | 10 949 | 123 | 1.000 (1.000 to 1.000) | 0.000 | 0.000 | 1.000 (1.000 to 1.000) | 0.000 |
| TST to cold probe average (°C) | 28.6 | 34.0 | 1725 | 28.7 | 34.0 | 1614 | 26.7 | 5.6 | 111 | 0.755 (0.688 to 0.829) | 0.000 | 0.000 | 0.654 (0.565 to 0.757) | 0.000 |
| TST to hot probe average (°C) | 37.2 | 166.7 | 1727 | 37.1 | 166.7 | 1615 | 39.5 | 9.8 | 112 | 1.019 (1.004 to 1.035) | 0.003 | 0.000 | 1.031 (0.996 to 1.067) | 0.033 |
| TTT to cold probe average (°C) | 20.7 | 89.7 | 1724 | 20.7 | 89.7 | 1612 | 18.5 | 8.4 | 112 | 0.668 (0.592 to 0.754) | 0.000 | 0.000 | 0.717 (0.640 to 0.803) | 0.000 |
| TTT to hot probe average (°C) | 44.9 | 15.5 | 1722 | 44.8 | 15.5 | 1612 | 46.1 | 6.6 | 112 | 1.523 (1.337 to 1.734) | 0.000 | 0.000 | 1.517 (1.342 to 1.714) | 0.000 |
| Vibration perception threshold wrist (V) | 11 | 46 | 1751 | 11 | 46 | 1635 | 13 | 17 | 116 | 1.068 (1.036 to 1.101) | 0.000 | 0.000 | 1.117 (1.060 to 1.177) | 0.000 |
| Vibration perception threshold knee (V) | 22 | 43 | 1749 | 22 | 43 | 1633 | 30 | 39 | 116 | 1.082 (1.059 to 1.107) | 0.000 | 0.000 | 1.081 (1.057 to 1.106) | 0.000 |
| Vibration perception threshold ankle (V) | 21 | 43 | 1751 | 20 | 43 | 1635 | 31 | 39 | 116 | 1.084 (1.063 to 1.105) | 0.000 | 0.000 | 1.077 (1.056 to 1.098) | 0.000 |
| Vibration perception threshold hallux (V) | 21 | 44 | 1751 | 21 | 44 | 1635 | 32 | 40 | 116 | 1.077 (1.057 to 1.098) | 0.000 | 0.000 | 1.072 (1.053 to 1.092) | 0.000 |
| Blood glucose level (mmol/L) | 14 | 31 | 1809 | 14 | 31 | 1686 | 16 | 28 | 123 | 1.027 (1.006 to 1.048) | 0.000 | 0.026 | 1.024 (1.003 to 1.045) | 0.031 |
P values <0.05 indicate a significant association and are shown in bold in the table.
*Based on univariate Cox survival analysis.
†Mann-Whitney U test.
‡Based on univariate logistic regression.
ABI, ankle brachial index; BMI, body mass index; TST, temperature sensation threshold; TTT, temperature tolerance threshold.
Table 4.
Peak plantar pressure during walking at different regions of the foot for all participants and for the groups with and with no future ulceration occurrence
| Continuous parameter | All | No-ulcer occurrence | With ulcer occurrence | Survival analysis Cox univariate regression | Mann-Whitney U test | Univariate logistic regression | ||||||||
| Median | Range | n‡ | Median | Range | n‡ | Median | Range | n‡ | HR (95% CI)* |
P- survival analysis * | P-differences† | OR (95% CI)‡ | P-value logistic regression‡ | |
| Plantar pressure at hallux (kPa) | 250 | 758 | 1810 | 250 | 758 | 1687 | 255 | 688 | 123 | 0.999 (0.998 to 1.001) | 0.446 | 0.966 | 1.000 (0.998 to 1.001) | 0.656 |
| Plantar pressure at second toe (kPa) | 130 | 695 | 1810 | 128 | 695 | 1687 | 135 | 415 | 123 | 1.001 (0.998 to 1.003) | 0.458 | 0.501 | 1.001 (0.999 to 1.003) | 0.324 |
| Plantar pressure at third toe (kPa) | 85 | 448 | 1810 | 85 | 448 | 1687 | 88 | 368 | 123 | 1.000 (0.997 to 1.003) | 0.825 | 0.892 | 1.000 (0.997 to 1.004) | 0.808 |
| Plantar pressure at fourth toe (kPa) | 58 | 355 | 1810 | 58 | 355 | 1687 | 57 | 223 | 123 | 0.996 (0.992 to 1.001) | 0.092 | 0.286 | 0.997 (0.993 to 1.002) | 0.269 |
| Plantar pressure at fifth toe (kPa) | 30 | 248 | 1810 | 30 | 248 | 1687 | 23 | 170 | 123 | 0.990 (0.983 to 0.998) | 0.011 | 0.008 | 0.991 (0.983 to 0.998) | 0.005 |
| Plantar pressure first MTH (kPa) | 158 | 758 | 1810 | 158 | 758 | 1687 | 158 | 475 | 123 | 1.003 (1.001 to 1.005) | 0.015 | 0.217 | 1.001 (0.999 to 1.003) | 0.366 |
| Plantar pressure at second MTH (kPa) | 203 | 605 | 1810 | 203 | 605 | 1687 | 208 | 543 | 123 | 1.001 (0.998 to 1.003) | 0.556 | 0.307 | 1.002 (0.999 to 1.004) | 0.219 |
| Plantar pressure at third MTH (kPa) | 225 | 911 | 1810 | 225 | 911 | 1687 | 223 | 665 | 123 | 1.000 (0.998 to 1.002) | 0.987 | 0.859 | 1.001 (0.999 to 1.004) | 0.326 |
| Plantar pressure at fourth MTH (kPa) | 213 | 883 | 1810 | 213 | 883 | 1687 | 208 | 645 | 123 | 1.000 (0.997 to 1.002) | 0.862 | 0.843 | 1.001 (0.998 to 1.003) | 0.595 |
| Plantar pressure at fifth MTH (kPa) | 183 | 1000 | 1810 | 183 | 788 | 1687 | 178 | 980 | 123 | 1.000 (0.999 to 1.002) | 0.707 | 0.439 | 1.000 (0.999 to 1.002) | 0.753 |
| Plantar pressure at lateral midfoot (kPa) | 78 | 410 | 1810 | 78 | 410 | 1687 | 76 | 305 | 123 | 0.999 (0.993 to 1.005) | 0.660 | 0.408 | 0.998 (0.992 to 1.003) | 0.425 |
| Plantar pressure at center of midfoot (kPa) | 105 | 365 | 1810 | 105 | 353 | 1687 | 98 | 353 | 123 | 0.997 (0.992 to 1.001) | 0.105 | 0.419 | 1.000 (0.996 to 1.004) | 0.871 |
| Plantar pressure at medial midfoot (kPa) | 68 | 215 | 1810 | 68 | 215 | 1687 | 65 | 140 | 123 | 0.998 (0.990 to 1.005) | 0.591 | 0.310 | 0.996 (0.989 to 1.003) | 0.284 |
| Plantar pressure at lateral hindfoot (kPa) | 110 | 293 | 1810 | 110 | 293 | 1687 | 100 | 215 | 123 | 0.998 (0.993 to 1.003) | 0.496 | 0.018 | 0.994 (0.989 to 1.000) | 0.034 |
| Plantar pressure at center of hindfoot (kPa) | 165 | 458 | 1810 | 165 | 458 | 1687 | 170 | 245 | 123 | 0.998 (0.994 to 1.002) | 0.430 | 0.896 | 1.000 (0.996 to 1.004) | 0.992 |
| Plantar pressure at medical hind foot (kPa) | 115 | 420 | 1810 | 115 | 420 | 1687 | 113 | 215 | 123 | 1.001 (0.997 to 1.005) | 0.635 | 0.279 | 0.997 (0.993 to 1.002) | 0.228 |
P values <0.05 indicate a significant association and are shown in bold in the table.
*Based on univariate Cox survival analysis.
†Mann-Whitney U test.
‡Based on univariate logistic regression.
MTH, metatarsal head.
Categorical parameters
Differences in categorical parameters between the groups with and without future ulcer incident
In comparing the categorical parameters between the two groups, it was found that neuropathy (χ2=26.46, Phi=0.123), history of ulceration (χ2=7.36, Phi=0.076), presence of callus (χ2=16.93, Phi=0.1), nail ingrowth (χ2=14.71, Phi=0.106), foot swelling (χ2=13.54, Phi=0.093), limited ankle (χ2=17.957, Phi=0.109), and metatarsophalangeal (MTP) joint (χ2=18.29, Phi=0.110) mobilities were all significantly (p<0.05) and with small effect size associated with incident of ulceration. Furthermore, insensitivity to touch (χ2=62.77, Cramer’s V=0.186) with medium effect size was significantly associated with future incident of ulceration.
Effect of categorical parameters on the risk and likelihood of DFU incident
The categorical parameters (HR (95% CI); OR (95% CI)) including neuropathy (2.525 (1.680 to 3.795); 2.926 (1.926 to 4.444)); history of ulceration (2.796 (1.029 to 7.598); 5.624 (1.738 to 18.198)); presence of callus (1.474 (0.999 to 2.174); 2.361 (1.571 to 3.549)); nail ingrowth (5.653 (2.078 to 15.379); 11.308 (2.997 to 42.663)); foot swelling (3.345 (1.799 to 6.218); 3.667 (1.843 to 7.296)); dry skin (1.926 (1.273 to 2.914)); 2.344 (1.017 to 5.400)); limited ankle (1.662 (1.365 to 2.022); 2.552 (1.689 to 3.856)) and MTP joint (2.745 (1.853 to 4.067); 2.527 (1.673 to 3.818)) ranges of motion; and decreased (3.141 (2.102 to 4.693); 3.723 (2.461 to 5.631)), highly decreased (5.263 (1.266 to 21.878); 6.967 (3.563 to 13.625)), and absent (9.671 (5.179 to 18.059); 7.340 (1.505 to 35.804)) sensation to touch were all significantly associated with higher risk and with higher likelihood of ulceration risk occurrence (tables 1 and 2). Furthermore, all patients with the characteristics described above showed significantly decreased ulcer-free survival time compared with the patients without those characteristics (Kaplan-Meier survival analyses) (tables 1 and 2).
All categorical parameters (except history of smoking) which were significant risk factors for DFU occurrence were also significantly associated with the likelihood of DFU occurrence (tables 1 and 2).
History of smoking was the only parameter that showed to be significantly associated with risk (1.686 (1.097 to 2.592)) of future DFU but was not significantly (p>0.05) associated with odds of ulceration occurrence and showed to significantly decrease the ulcer-free survival time by 324 days.
Continuous parameters
Differences in the continuous parameters between the group with and without future ulcer incident
While comparing the general continuous parameters between the two groups, the vulnerable group to future ulceration were significantly (p<0.05) older (U=89 544, Z=−2.53, r=0.06), had higher blood glucose level (U=91 269, Z=−2.223, r=0.05), and showed lower plantar pressure at the fifth toe (U=89 069, Z=−2.643, r=0.06) and lateral heel (U=90 531, Z=−2.363, r=0.06) regions during walking, all with small effect size, compared to their counterparts who were non-vulnerable to future ulceration. The corresponding p values are shown in tables 3 and 4.
Mann-Whitney U test further revealed that the group who incurred DFU during follow-up had significantly (p<0.05) longer duration of diabetes (U=74 974, Z=−5.15, r=0.12); higher VPT at the foot (U=60 198, Z=−7.79, r=0.18), wrist (U=68 458, Z=−5.021, r=0.12), knee (U=58 162, Z=−6.96, r=0.17), and ankle (U=54 774, Z=−7.615, r=0.18); higher TST (U=58 570, Z=−6.274, r=0.15) and TTT (U=55 662, Z=−6.799, r=0.16) to warm stimuli at their feet; lower TST (U=54 177, Z=−6.985, r=0.17) and TTT (U=61 673, Z=−5.624, r=0.14) to cold stimuli at their feet; all with small to medium effect size compared with the group that did not incur ulceration during follow-up. The corresponding p values are shown in table 3.
Effect of continuous parameters on the risk and likelihood of DFU incident
From the continuous parameters (HR (95% CI); OR (95% CI)): age (1.026 (1.010 to 1.042); 1.019 (1.003 to 1.035)); VPT (1.079 (1.060 to 1.099); 1.073 (1.054 to 1.092)); duration of diabetes in days (1.000 (1.000 to 1.000); 1.000 (1.000 to 1.000)); TST (1.019 (1.004 to 1.035); 1.031 (0.996 to 1.067)) and TTT (1.523 (1.337 to 1.734); 1.517 (1.342 to 1.714)) to hot stimuli; and blood glucose level (1.027 (1.006 to 1.048); 1.024 (1.003 to 1.045)) were all positively and significantly associated with increased risk (HR) and increase the likelihood (OR) of future foot ulcer occurrence (tables 3 and 4). However, plantar pressure underneath the fifth toe (0.990 (0.983 to 0.998); 0.991 (0.983 to 0.998)) (table 4) and TST (0.755 (0.688 to 0.829); 0.654 (0.565 to 0.757)) and TTT (0.668 (0.592 to 0.0754); 0.717 (0.640 to 0.803)) to cold stimuli showed to significantly decrease both the risk (HR) and likelihood (OR) of the future DFU occurrence (table 3).
Most continuous parameters (except plantar pressure at the first MTH) which were significantly associated with the risk (based on univariate Cox regression) of DFU occurrence were observed to significantly increase the likelihood (based on univariate logistic regression) of DFU occurrence.
Plantar pressure at the first MTH showed to only significantly increase the risk (HR= (1.003 (1.001 to 1.005)) but not the odds of the future DFU occurrence. On the other hand, there were only two continuous parameters including the plantar pressure at the lateral heel and ABI that showed to significantly decrease the likelihood (ORs (0.994 (0.989 to 1.000)) and (0.247 (0.073 to 0.834)), respectively, but not the hazard of future DFU occurrence (table 4).
The model to predict risk
Cox proportional hazard multiple regression analysis indicated a worthwhile model indicated by: χ2=94.545, p=0.000. The result of Cox proportional hazard multiple regression analysis indicated that only six (four categorical and two continuous) parameters stayed in the final risk prediction model. From those, only four parameters (two categorical and two continuous parameters) significantly contributed to predicting DFU risk (HR (95% CI)) as follows: nail ingrowth (4.42 (1.38 to 14.07)); dry skin (4.48 (1.80 to 11.14)); VPT (1.07 (1.04 to 1.09)); and TTT to warm stimuli (1.001 (1.000 to 1.002)). Together with two other parameters that were not significant (p>0.05) predictors of DFU risk including neuropathy (1.13 (0.71 to 1.82)) and foot swelling (1.79 (0.91 to 3.52)), the overall model showed to be a worthwhile model (indicated by: χ2=94.545, p=0.000) to predict ulceration risk. The area below the ROC curve as a measure of prognosis power of the model was 0.62 (0.57 to 0.67).
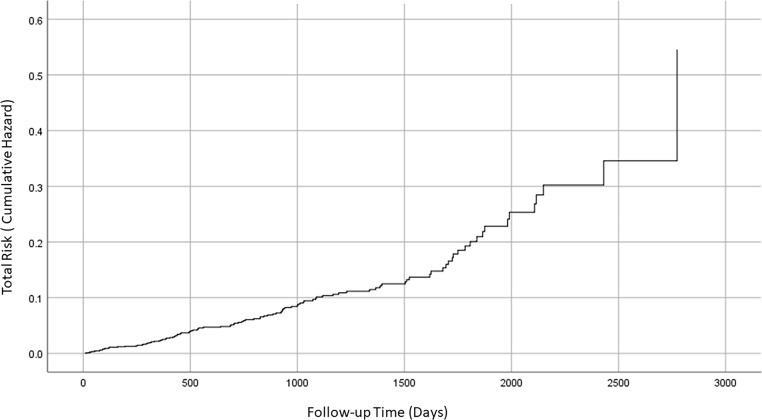
Figure 1 shows the cumulative hazard for DFU occurrence over time. This indicates how the risk of DFU increases over time. For example, the average risk of DFU is less than 0.1 (10%) at 1000 days follow-up. However, the risk increases to approximately 0.25 (25%) at 2000 days follow-up.
Figure 1.
The total risk (cumulative hazard) for diabetic foot ulcer incident as a function of follow-up time.
For further information about the differences in the average survival times (ulcer-free time) for patients with dry skin, nail ingrowth, neuropathy, and swelling, the readers are referred to the supplementary materials (online supplementary appendices 1–4).
bmjdrc-2019-001122supp001.pdf (866.9KB, pdf)
bmjdrc-2019-001122supp002.pdf (902.5KB, pdf)
bmjdrc-2019-001122supp003.pdf (308.1KB, pdf)
bmjdrc-2019-001122supp004.pdf (314.9KB, pdf)
As in online supplementary appendix 2, the proportion of population without ulcer at 2000 days follow-up was more than 0.8 in participants without neuropathy. However, this was just over 0.6 in participants with neuropathy. This indicates a significant decrease in the average ulcer-free survival as a result of neuropathy.
Discussion
Difference in parameters
The present study indicates that the patients with future DFU occurrence had distinctive characteristics in a set of parameters that were considered in this investigation. Significantly higher proportions of patients with any of the following characteristics: neuropathy, history of ulceration, presence of callus, nail ingrowth, foot swelling, and limited ankle and MTP joint mobilities were shown to have an incident of future DFU. However, the effect sizes were found to be small indicating that these criteria may not be used for effective patient stratification. On the other hand, we showed that a significantly higher proportions of patients with impaired sensitivity to touch ended up having future incident of DFU. To be more precise, the higher the degree of impairment to touch, the higher the proportion of patients with DFU incident in that group. With a medium effect size, this indicates that this parameter has the potential for stratifying patients into four groups based on the sensitivity level to touch where each group showed a significantly higher prevalence of future DFU proportionate to the sensitivity level to touch sensation.
The significantly higher proportion of participants with impaired sensitivity to monofilament or history of ulcer incurred future DFU that is in line with the previous studies for neuropathy30 or for history of ulcer30–32 in European populations.
Out of all biomechanical parameters, limited ankle mobility that can indicate excessive load on the forefoot during ambulation was found to be significantly more prevalent in the group with future DFU occurrence that is in line with the results of the previous study in Europe.33
The results of this study highlighting the significantly older age and longer duration of diabetes of the group vulnerable to future DFU occurrence are in line with the previous finding for age in European31 and for the duration of diabetes in European30 31 and Middle Eastern34 populations. This indicates similarities in the generic characteristics between the studied group and the patient population in previous studies.30 31 34
However, the results of the current study which indicate that significantly higher proportion of patients who incur future DFU had callus or swollen feet contradicts the previous studies on south European population for callus30 31 or swollen foot30 31 where no such observations were reported.
These distinct differences in foot characteristics of patients with future DFU that were found in the current study can indicate a different etiology of ulceration in the studied group. In addition to the fact that the previous studies reported on a different demographic (ie, European30 vs African population in the current study), in few a combination of patients recruited from a hospital and a community setting was studied.30 However, our study which had a focus on patients in a hospital setting indicates that with regards to identifying the patient group vulnerable to foot ulceration, attention need to be paid to patients with callus and swollen feet (edema).
Parameters associated with increased risk (hazard) and likelihood (odds) of DFU occurrence
The results of the current study in which association between older age and longer duration of diabetes on increasing the likelihood of DFU occurrence were found, are in line with the study conducted on the pooled patient data from Europe and North America.16 This can be the results of the deterioration in the soft tissue due to prolonged exposure to high blood sugar level, reflected in the significantly higher risk for future DFU occurrence for patients with higher duration of diabetes or higher blood glucose levels that were found in the current study. These reults of the current study are also in agreement with the findings from studies in North America.35 36 Although the observed association between swollen feet and increased risk of future DFU occurrence in present study is in line with the findings of previous studies in North America,35 contrary to that study35 in the current study presence of callus was associated with increased risk of DFU occurrence.
On the other hand, onychomycosis was previously reported to have strong association with nail ingrowth37; hence, the results of the current study where nail ingrowth was associated with the future DFU occurrence are in agreement with a previous study on North American population.35
The significantly higher risk and more likelihood of future DFU occurrence for patients with impaired sensation to monofilament or with history of ulceration that is found in the present study are in line with the studies in North America35 36 or Europe16 indicating the notion that DFU occurrence is linked to the damages in the soft tissue which patient does not recognize as a result of impaired sensation. This can be further backed up by the result of the current study where a significantly higher likelihood of future DFU occurrence for patients with increased VPT was observed in accordance with previous study in Europe.38
Furthermore in line with other findings of the current study with regards to neuropathy, impaired sensation to touch21 showed significant association with future DFU occurrence in the present study. While the value of Ipswich touch test to accurately assess loss of protective sensation was previously established,39 the present study indicates that the risk of future DFU occurrence significantly increases as the level of impairment to touch sensation increases. Furthermore, the ulcer-free survival time also showed to significantly decrease as the sensitivity to touch decreases. Hence, touch sense could be used to stratify patient based on the risk of future DFU occurrence.
The significant associations between impaired sensitivities and tolerances to temperature stimuli with the risk and likelihood of future DFU occurrence that observed in the present study indicate the importance of assessing small fiber function in identifying the risk of future DFU and are in line with previous study on small cohort of patients.40
With regards to the biomechanical parameters, the association between limited MTP joint mobility and increased likelihood of future DFU occurrence is in line with the results of the previous findings in large cohorts from North America.14 This could be affiliated to the increased pressure on the forefoot that is observed as the significant association between the peak plantar pressure at the first MTH during shod walking and the DFU occurrence were reported in North American population.41
The fact that the ulcerated participants showed higher plantar pressure at the fifth MTH can be attributed to some morphological changes as a result of motor nerve damage in this group of patients where this leads to applying less load at the outer side of the forefoot. Investigating the motor neurons in patients with diabetes warrants further analyses in future studies.
While several categorical and continuous parameters were shown to increase the risk and likelihood of DFU in the current study, it can be argued that collecting all these parameters may not be feasible in a clinical setting. Hence, development of a model using proportionate hazard multiple regression analysis deemed necessary to reveal the minimum number of parameters which can predict the DFU with highest accuracy discussed as follows.
Survival analysis (Cox regression model to identify ulcer-free survival)
The Cox regression model containing six parameters (including four categorical–out of which two were significant predictors and two continuous–both were significant predictors) was found to be worthwhile for predicting the incident of diabetic foot ulceration.
Two categorical parameters, nail ingrowth and dry skin indicate that patients with each of these characteristics have significantly higher (four times more) risk of future ulcer occurrence compared with those without these characteristics.
In the present study, VPT was found to be a significant contributor to predicting the future DFU occurrence, and it indicates that increase in VPT by 1 V would increase the chance of ulceration by 7%.
Cox proportional hazard multiple regression analyses indicated that the final prediction model of foot ulceration included four significant (p<0.05) parameters, including: nail ingrowth, dry skin, VPT, and TTT to warm stimuli, plus the two non-significant (p>0.05), including neuropathy and foot swelling. The overall model showed to be a worthwhile model that can predict ulceration risk with a prognosis power of 0.62.
Strength and limitations
The present study is unique as it reports on a wide range of foot-related parameters along with the clinical and lifestyle characteristics to identify the risk factors for future DFU occurrence in large cohort of patients with diabetes in Africa. The prognosis power of the model proposed in the current study (0.62) is lower than the values of 0.73 reported in other studies for western populations.31 However in the current study when, a prognostic model was developed using only the three parameters (namely: absent pedal pulse, history of ulcer, and monofilament insensitivity) that were suggested based on a systematic review,16 the prognosis accuracy was found to be much lower.
Despite the difference in the prevalence of diabetic foot ulceration across African countries,7 it can be argued that there are similarities in ethnicity, socioeconomic conditions, and culture between Tanzania and Eastern, Central, and Western Africa. Hence, a similar model to the one that was developed in the current study may be applied to patients from those populations.
Clinical implications and future directions
The results of this study indicated that a few parameters significantly associated with and contributed to predicting the risk of DFU occurrence, except the nail ingrowth, the rest of parameters were related to neuropathy. This highlights the importance of considering both the sensory and autonomic peripheral neuropathy associated with large and small fiber impairment together.
The results on VPT (HR: 1.07) indicate the importance of assessing the impairment in A-alpha/beta as the nerves that are functional in sensing touch and vibration sensations.42 The impairment in C-fibers that is responsible for input from high threshold stimuli43 44 indicated by the findings of the current study in which a high TTT to hot stimuli (HR: 1.001) are among the predictors of risk of future DFU occurrence. The results of the current study also indicated the importance of VPT and touch sensation in assessing the risk of DFU occurrence.
The impairment of small unmyelinated C-fibers of the peripheral nervous system that are responsible for cutaneous sensations and for mediating thermal sensations44 45 is reported in the current study, which is in line with the previous observations in patients with diabetic neuropathy and with DFU.15 46
The current study further indicates impairment of small fiber as mediator of autonomic functions such as sweating44 indicated by the importance of skin status (dry skin HR=4.48) in predicting the risk of future DFU occurrence. This is related to decreased activity of sweat glands and a dry skin,47 48 which make the dry skin vulnerable to cracks and fissures leading to ulceration.
Although previous studies indicate that the small fiber impairment precede large fiber complications in people with diabetes,48 the results of present study indicate that the measurements related to small fiber impairment provide as a strong prognostic tool to assess the risk of DFU occurrence.
Although in the current study a vast range of parameters were collected from each participant, the inclusion of further parameters to reflect the microcirculatory49 and mechanical properties of the plantar soft tissue50 could have resulted in achieving a higher prognosis power in predicting the DFU occurrence.
Conclusion
Participants who were vulnerable to future diabetic foot ulceration occurrence showed distinct characteristics in few foot-related parameters including higher VPT and lower sensitivity to touch, and lower sensitivity and higher tolerance to both warm and cold stimuli. The results of the current study indicate the need to assess small fiber neuropathy as a major risk factor for DFU occurrence in patients. The current study also highlighted that, the presence of nail ingrowth and dry skin each increase the likelihood of future DFU by more than four times.
In addition to the common measures of neuropathy associated with large fiber (ie, VPT and monofilament), measurements related to small fiber impairment (ie, TST and TTT; and touch sensation test) need to be included in the risk assessment for DFU occurrence.
Acknowledgments
We acknowledge the assistance from Novel GmbH (Munich, Germany) for providing plantar pressure platform. We also acknowledge Shabneez Gangji and other staff at Abbas Medical Centre for helping with data collection.
Footnotes
Contributors: RN conceived and rationalized the study and performed statistical analyses and wrote the first draft of the manuscript. NC contributed to the design of the study and revised the manuscript. JKL contributed to the study design and data analyses. ZGA designed the data collection process and oversaw the recruitment and data collection.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent for publication: Not required.
Ethics approval: This study used secondary anonymized data from a wider study conducted at Abbas Medical Centre, Dar es Salaam, Tanzania. It received ethical approval from an independent ethics committee constituted at Abbas Medical Centre (Ref: Ethics/StaffsUni/03-2016). All participants gave informed consent before taking part in the wider study.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: Data are available on reasonable request.
References
- 1.Singh N, Armstrong DG, Lipsky BA. Preventing foot ulcers in patients with diabetes. JAMA 2005;293:217. 10.1001/jama.293.2.217 [DOI] [PubMed] [Google Scholar]
- 2.Armstrong DG, Boulton AJM, Bus SA. Diabetic foot ulcers and their recurrence. N Engl J Med 2017;376:2367–75. 10.1056/NEJMra1615439 [DOI] [PubMed] [Google Scholar]
- 3.Prompers L, Huijberts M, Apelqvist J, et al. High prevalence of ischaemia, infection and serious comorbidity in patients with diabetic foot disease in Europe. baseline results from the Eurodiale study. Diabetologia 2007;50:18–25. 10.1007/s00125-006-0491-1 [DOI] [PubMed] [Google Scholar]
- 4.Lipsky BA, Berendt AR, Cornia PB, et al. Infectious diseases Society of America clinical practice guideline for the diagnosis and treatment of diabetic foot infections. Clin Infect Dis 2012;2012:e132–73. [DOI] [PubMed] [Google Scholar]
- 5.Lavery LA, Armstrong DG, Wunderlich RP, et al. Predictive value of foot pressure assessment as part of a population-based diabetes disease management program. Diabetes Care 2003;26:1069–73. 10.2337/diacare.26.4.1069 [DOI] [PubMed] [Google Scholar]
- 6.Walsh JW, Hoffstad OJ, Sullivan MO, et al. Association of diabetic foot ulcer and death in a population-based cohort from the United Kingdom. Diabet Med 2016;33:1493–8. 10.1111/dme.13054 [DOI] [PubMed] [Google Scholar]
- 7.Abbas ZG, Archibald LK. Epidemiology of the diabetic foot in Africa. Med Sci Monit 2005;11:RA262–70. [PubMed] [Google Scholar]
- 8.Abbas ZG, Archibald LK. Challenges for management of the diabetic foot in Africa: doing more with less. Int Wound J 2007;4:071027000841001 10.1111/j.1742-481X.2007.00376.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Morbach S, Lutale JK, Viswanathan V, et al. Regional differences in risk factors and clinical presentation of diabetic foot lesions. Diabet Med 2004;21:91–5. 10.1046/j.1464-5491.2003.01069.x [DOI] [PubMed] [Google Scholar]
- 10.Monteiro-Soares M, Boyko EJ, Ribeiro J, et al. Risk stratification systems for diabetic foot ulcers: a systematic review. Diabetologia 2011;54:1190–9. 10.1007/s00125-010-2030-3 [DOI] [PubMed] [Google Scholar]
- 11.Crawford F, Cezard G, Chappell FM, et al. A systematic review and individual patient data meta-analysis of prognostic factors for foot ulceration in people with diabetes: the International research collaboration for the prediction of diabetic foot ulcerations (PODUS). Health Technol Assess 2015;19:1–210. 10.3310/hta19570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pham H, Armstrong DG, Harvey C, et al. Screening techniques to identify people at high risk for diabetic foot ulceration: a prospective multicenter trial. Diabetes Care 2000;23:606–11. 10.2337/diacare.23.5.606 [DOI] [PubMed] [Google Scholar]
- 13.Zimny S, Schatz H, Pfohl M. The role of limited joint mobility in diabetic patients with an at-risk foot. Diabetes Care 2004;27:942–6. 10.2337/diacare.27.4.942 [DOI] [PubMed] [Google Scholar]
- 14.Ledoux W, Shofer J, DS-J of. Relationship between foot type, foot deformity, and ulcer occurrence in the high-risk diabetic foot. Available: https://archive-ouverte.unige.ch/unige:39927/ATTACHMENT01 [Accessed 11 Jul 2018]. [DOI] [PubMed]
- 15.Guy RJ, Clark CA, Malcolm PN, et al. Evaluation of thermal and vibration sensation in diabetic neuropathy. Diabetologia 1985;28:131–7. 10.1007/bf00273859 [DOI] [PubMed] [Google Scholar]
- 16.Crawford F, Cezard G, Chappell FM, et al. The development and validation of a multivariable prognostic model to predict foot ulceration in diabetes using a systematic review and individual patient data meta-analyses. Diabet Med 2018;35:1480–93. 10.1111/dme.13797 [DOI] [PubMed] [Google Scholar]
- 17.Zhang P, Lu J, Jing Y, et al. Global epidemiology of diabetic foot ulceration: a systematic review and meta-analysis †. Ann Med 2017;49:106–16. 10.1080/07853890.2016.1231932 [DOI] [PubMed] [Google Scholar]
- 18.Jeffcoate WJ, Bus SA, Game FL, et al. Reporting standards of studies and papers on the prevention and management of foot ulcers in diabetes: required details and markers of good quality. Lancet Diabetes Endocrinol 2016;4:781–8. 10.1016/S2213-8587(16)30012-2 [DOI] [PubMed] [Google Scholar]
- 19.Feng Y, Schlösser FJ, Sumpio BE. The Semmes Weinstein monofilament examination as a screening tool for diabetic peripheral neuropathy. J Vasc Surg 2009;50:675–82. 10.1016/j.jvs.2009.05.017 [DOI] [PubMed] [Google Scholar]
- 20.Frykberg RG, Zgonis T, Armstrong DG, et al. Diabetic foot disorders. A clinical practice guideline (2006 revision). J Foot Ankle Surg 2006;45:S1–66. 10.1016/S1067-2516(07)60001-5 [DOI] [PubMed] [Google Scholar]
- 21.Rayman G, Vas PR, Baker N, et al. The Ipswich touch test: a simple and novel method to identify inpatients with diabetes at risk of foot ulceration. Diabetes Care 2011;34:1517–8. 10.2337/dc11-0156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schaper NC, Van Netten JJ, Apelqvist J, et al. Prevention and management of foot problems in diabetes: a summary guidance for daily practice 2015, based on the IWGDF guidance documents. Diabetes Metab Res Rev 2016;32 Suppl 1:7–15. 10.1002/dmrr.2695 [DOI] [PubMed] [Google Scholar]
- 23.Bus SA, van Netten JJ, Lavery LA, et al. IWGDF guidance on the prevention of foot ulcers in at-risk patients with diabetes. Diabetes Metab Res Rev 2016;32 Suppl 1:16–24. 10.1002/dmrr.2696 [DOI] [PubMed] [Google Scholar]
- 24.Jayaprakash P, Bhansali A, Bhansali S, et al. Validation of bedside methods in evaluation of diabetic peripheral neuropathy. Indian J Med Res 2011;133:645–9. [PMC free article] [PubMed] [Google Scholar]
- 25.Ziegler D, Mayer P, Gries FA. Evaluation of thermal, pain, and vibration sensation thresholds in newly diagnosed type 1 diabetic patients. J Neurol Neurosurg Psychiatry 1988;51:1420–4. 10.1136/jnnp.51.11.1420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fernando M, Crowther RG, Cunningham M, et al. The reproducibility of acquiring three dimensional gait and plantar pressure data using established protocols in participants with and without type 2 diabetes and foot ulcers. J Foot Ankle Res 2016;9:4. 10.1186/s13047-016-0135-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bus SA, de Lange A, de LA. A comparison of the 1-step, 2-step, and 3-step protocols for obtaining barefoot plantar pressure data in the diabetic neuropathic foot. Clin Biomech 2005;20:892–9. 10.1016/j.clinbiomech.2005.05.004 [DOI] [PubMed] [Google Scholar]
- 28.Naemi R, Chevalier TL, Healy A, et al. The effect of the use of a walkway and the choice of the foot on plantar pressure assessment when using pressure platforms. Foot 2012;22:100–4. 10.1016/j.foot.2012.01.004 [DOI] [PubMed] [Google Scholar]
- 29.Naemi R, Chockalingam N, Lutale JK, et al. Can a combination of lifestyle and clinical characteristics explain the presence of foot ulcer in patients with diabetes? J Diabetes Complications 2019;33:437–44. 10.1016/j.jdiacomp.2019.02.006 [DOI] [PubMed] [Google Scholar]
- 30.Monteiro-Soares M, Ribas R, Pereira da Silva C, et al. Diabetic foot ulcer development risk classifications' validation: a multicentre prospective cohort study. Diabetes Res Clin Pract 2017;127:105–14. 10.1016/j.diabres.2017.02.034 [DOI] [PubMed] [Google Scholar]
- 31.Monteiro-Soares M, Vaz-Carneiro A. Validation and comparison of currently available stratification systems for patients with diabetes by risk of foot ulcer development. Eur J, 2012. Available: http://www.eje-online.org/content/167/3/401.short [Accessed 28 Sep 2016]. [DOI] [PubMed]
- 32.Hurley L, Kelly L, Garrow AP, et al. A prospective study of risk factors for foot ulceration: the West of Ireland diabetes foot study. QJM 2013;106:1103–10. 10.1093/qjmed/hct182 [DOI] [PubMed] [Google Scholar]
- 33.Francia P, Seghieri G, Gulisano M. The role of joint mobility in evaluating and monitoring the risk of diabetic foot ulcer. diabetes Res, 2015. Available: http://www.sciencedirect.com/science/article/pii/S0168822715001837 [Accessed 28 Sep 2016]. [DOI] [PubMed]
- 34.Yazdanpanah L, Shahbazian H, Nazari I, et al. Incidence and Risk Factors of Diabetic Foot Ulcer: A Population-Based Diabetic Foot Cohort (ADFC Study)-Two-Year Follow-Up Study. Int J Endocrinol 2018;2018:1–9. 10.1155/2018/7631659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Boyko EJ, Ahroni JH, Cohen V, et al. Prediction of diabetic foot ulcer occurrence using commonly available clinical information: the Seattle diabetic foot study. Diabetes Care 2006;29:1202–7. 10.2337/dc05-2031 [DOI] [PubMed] [Google Scholar]
- 36.Boyko EJ, Ahroni JH, Stensel V, et al. A prospective study of risk factors for diabetic foot ulcer. The Seattle diabetic foot study. Diabetes Care 1999;22:1036–42. 10.2337/diacare.22.7.1036 [DOI] [PubMed] [Google Scholar]
- 37.Vural S, Bostanci S, Koçyigit P, et al. Risk factors and frequency of ingrown nails in adult diabetic patients. J Foot Ankle Surg 2018;57:289–95. 10.1053/j.jfas.2017.10.006 [DOI] [PubMed] [Google Scholar]
- 38.Kästenbauer T, Sauseng S, Sokol G, et al. A prospective study of predictors for foot ulceration in type 2 diabetes. J Am Podiatr Med Assoc 2001;91:343–50. 10.7547/87507315-91-7-343 [DOI] [PubMed] [Google Scholar]
- 39.Sharma S, Kerry C, Atkins H, et al. The Ipswich touch test: a simple and novel method to screen patients with diabetes at home for increased risk of foot ulceration. Diabet Med 2014;31:1100–3. 10.1111/dme.12450 [DOI] [PubMed] [Google Scholar]
- 40.Carrington AL, Shaw JE, Van Schie CHM, et al. Can motor nerve conduction velocity predict foot problems in diabetic subjects over a 6-year outcome period? Diabetes Care 2002;25:2010–5. 10.2337/diacare.25.11.2010 [DOI] [PubMed] [Google Scholar]
- 41.Ledoux WR, Shofer JB, Cowley MS, et al. Diabetic foot ulcer incidence in relation to plantar pressure magnitude and measurement location. J Diabetes Complications 2013;27:621–6. 10.1016/j.jdiacomp.2013.07.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vinik AI, Maser RE, Mitchell BD, et al. Diabetic autonomic neuropathy. Diabetes Care 2003;26:1553–79. 10.2337/diacare.26.5.1553 [DOI] [PubMed] [Google Scholar]
- 43.Sène D. Small fiber neuropathy: diagnosis, causes, and treatment. Joint Bone Spine 2018;85:553–9. 10.1016/j.jbspin.2017.11.002 [DOI] [PubMed] [Google Scholar]
- 44.Themistocleous AC, Ramirez JD, Serra J, et al. The clinical approach to small fibre neuropathy and painful channelopathy. Pract Neurol 2014;14:368–79. 10.1136/practneurol-2013-000758 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Misra UK, Kalita J, Nair PP. Diagnostic approach to peripheral neuropathy. Ann Indian Acad Neurol 2008;11:89–97. 10.4103/0972-2327.41875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ali Z, Carroll M, Robertson KP, et al. The extent of small fibre sensory neuropathy in diabetics with plantar foot ulceration. J Neurol Neurosurg Psychiatry 1989;52:94–8. 10.1136/jnnp.52.1.94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Khalfallah K, Ayoub H, Calvet JH, et al. Noninvasive galvanic skin sensor for early diagnosis of sudomotor dysfunction: application to diabetes. IEEE Sens J 2012;12:456–63. 10.1109/JSEN.2010.2103308 [DOI] [Google Scholar]
- 48.Körei AE, Istenes I, Papanas N, et al. Small-Fiber neuropathy: a diabetic microvascular complication of special clinical, diagnostic, and prognostic importance. Angiology 2016;67:49–57. [DOI] [PubMed] [Google Scholar]
- 49.Körei AE, Istenes I, Papanas N, et al. Small-Fiber neuropathy. Angiology 2016;67:49–57. 10.1177/0003319715583595 [DOI] [PubMed] [Google Scholar]
- 50.Naemi R, Chatzistergos P, Suresh S, et al. Can plantar soft tissue mechanics enhance prognosis of diabetic foot ulcer? Diabetes Res Clin Pract 2017;126:182–91. 10.1016/j.diabres.2017.02.002 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjdrc-2019-001122supp001.pdf (866.9KB, pdf)
bmjdrc-2019-001122supp002.pdf (902.5KB, pdf)
bmjdrc-2019-001122supp003.pdf (308.1KB, pdf)
bmjdrc-2019-001122supp004.pdf (314.9KB, pdf)