Abstract
Background
There is limited knowledge on the influence of concurrent coinfections on the clinical presentation of Zika virus (ZIKV) disease.
Methods
To better understand the types, frequencies and clinical manifestations of ZIKV coinfections, we did a systematic review of four databases (PubMed, Embase, Web of Science, LILACS) without restrictions for studies on ZIKV coinfections confirmed by nucleic acid (quantitative real-time-PCR) testing of ZIKV and coinfecting pathogens. The review aimed to identify cohort, cross-sectional, case series and case report studies that described frequencies and/or clinical signs and symptoms of ZIKV coinfections. Conference abstracts, reviews, commentaries and studies with imprecise pathogen diagnoses and/or no clinical evaluations were excluded.
Results
The search identified 34 articles from 10 countries, comprising 2 cohort, 10 cross-sectional, 8 case series and 14 case report studies. Coinfections were most frequently reported to have occurred with other arthropod-borne viruses (arboviruses); out of the 213 coinfections described, ZIKV infections co-occurred with chikungunya in 115 cases, with dengue in 68 cases and with both viruses in 19 cases. Other coinfecting agents included human immunodeficiency, Epstein-Barr, human herpes and Mayaro viruses, Leptospira spp, Toxoplasma gondii and Schistosoma mansoni. ZIKV-coinfected cases primarily presented with mild clinical features, typical of ZIKV monoinfection; however, 9% of cases in cohort and cross-sectional studies were reported to experience complications.
Conclusion
Based on the evidence collated in this review, coinfections do not appear to strongly influence the clinical manifestations of uncomplicated ZIKV infections. Further research is needed to confirm whether risk of severe complications is altered when ZIKV infection co-occurs with other infections.
PROSPERO registration number
CRD42018111023.
Keywords: systematic review, arboviruses, epidemiology
Key questions.
What is already known?
As Zika virus (ZIKV) has been most prevalent in subtropical and tropical regions with high burdens of cocirculating infectious agents, a proportion of ZIKV infections occur simultaneously with infections by one or multiple other pathogens; however, it is uncertain whether coinfections may influence ZIKV-related pathology.
What are the new findings?
This systematic review collated the evidence on ZIKV coinfections as published in 34 studies in 10 countries. ZIKV coinfections were most frequently reported in the context of the arthropod-borne viruses, dengue and chikungunya, but were also described in relation to eight other pathogens.
While the findings of this review suggest that coinfections do not appear to strongly influence the clinical manifestations of uncomplicated ZIKV infections, this review did identify reports of neurological complications in the context of coinfection.
What do the new findings imply?
The findings of this review highlight a need for coordinated and rapid research efforts during future outbreaks to optimise diagnostic testing strategies for detecting coinfections and determining whether they may exacerbate the risk of severe ZIKV complications, such as Guillain-Barré syndrome and congenital Zika syndrome.
Introduction
Zika virus (ZIKV) is an Aedes mosquito-borne flavivirus that recently emerged in the Americas.1 First recognised in Brazil in early 2015, the ZIKV epidemic spread explosively, with autochthonous transmission reported in more than 86 countries and territories by 2018.1 Given the widespread circulation of this emerging infection of public health concern, it is critical that healthcare practitioners can readily recognise ZIKV disease across the full range of its clinical presentations.
Current evidence indicates that ZIKV infections typically present with no or mild clinical features.1 A 2018 meta-analysis of 23 studies by Haby and colleagues estimated a prevalence of asymptomatic ZIKV infections of 62% (95% CI 33% to 87%).2 For symptomatic ZIKV disease, the WHO describes a mild clinical presentation marked by fever, rash, conjunctivitis, myalgia, arthralgia, malaise and headache.1 Nevertheless, ZIKV is neurotropic and, in a subset of cases, infections have been associated with severe neurological complications, including the polyneuropathy Guillain-Barré syndrome (GBS) and congenital Zika syndrome (CZS), a constellation of congenital central nervous system malformations resulting from the vertical transmission of ZIKV during pregnancy.3 It has been estimated that GBS arises in approximately 2 per 10 000 ZIKV infections,1 4 and the absolute risk of adverse birth outcomes (ie, miscarriage, stillbirth, premature birth and CZS) has been reported to range between 7% and 46% in pregnancies with quantitative real-time PCR (qRT-PCR)-confirmed ZIKV infection.5–8
Although the clinical presentation of ZIKV monoinfections has been well characterised, one factor that may influence the clinical spectrum of ZIKV disease is coinfection. Given the high incidence of infectious diseases in the subtropical and tropical areas where ZIKV is prevalent, a proportion of all ZIKV infections occur concurrently with infections by one or multiple pathogens.9 ZIKV disease in the context of coinfection remains inadequately investigated, and it is uncertain whether specific coinfections may influence the presentation and severity of ZIKV-related signs and symptoms. A 2019 literature review by Vogels and colleagues hypothesised that coinfecting agents have the potential to enhance, inhibit, compete with or have no effect on ZIKV replication and the resulting clinical disease.10 To advance understanding on this topic, this systematic review aims to quantify how frequently ZIKV coinfections occur among ZIKV-infected populations and to investigate whether the clinical course of ZIKV disease in humans is altered in the context of coinfection.
Methods
Search
Four databases (PubMed, Web of Science, LILACs and EMBASE) were searched for publications up to 19 October 2019 using a comprehensive search strategy (online supplementary appendix 1). Keywords and Medical Subject Headings linked to ZIKV, bacterial, parasitic and other viral infectious diseases were used. The search included English, French, Spanish and Portuguese terms. No date or language restrictions were applied. The systematic review was registered in PROSPERO. All study titles and abstracts were screened based on eligibility criteria, and references of included studies were also screened to identify additional eligible articles.
bmjgh-2020-002350supp001.pdf (224.7KB, pdf)
Study selection and data extraction
Cohort studies, cross-sectional studies, case series and case reports describing coinfections of ZIKV with one or multiple other pathogens, confirmed by nucleic acid testing (eg, qRT-PCR) for ZIKV, and all coinfecting pathogens were eligible for inclusion in the review. Recovery of live pathogens was also considered to be indicative of acute coinfection. Of note, HIV-positive ZIKV cases with HIV suppression were not included in this review. Two reviewers (AR and LL) simultaneously screened studies for eligibility, and any discrepancies were resolved by a third reviewer (EBB). Conference abstracts, reviews, commentaries and studies without nucleic acid confirmation were excluded. Whereas cohort, cross-sectional and case series studies reporting on numbers of ZIKV coinfections without description of signs and symptoms were included to describe the frequency of ZIKV coinfections, studies with no reporting of signs and symptoms of ZIKV coinfections were otherwise excluded from the review. Data extraction was independently performed by two reviewers (AR, LL). From the full-text articles, information on study author, location, year, data source, age and sex of identified cases was extracted. Additional extracted information included frequencies of ZIKV cases with coinfection, types of coinfection, types of diagnostic testing, reported signs and symptoms, non-infectious comorbidities, and types and frequencies of complications. To investigate the frequency of ZIKV coinfections in cohort, cross-sectional and case series studies, the numbers of coinfections out of the total number of qRT-PCR-confirmed ZIKV cases were calculated for the eligible studies. The study quality assessment was conducted using the Oxford Centre for Evidence-based Medicine (OCEBM) Levels of Evidence, March 200911; see online supplementary appendix 2 for details.
Patient and public involvement
This research was done without patient or public involvement.
Results
Study selection
The search initially identified 12 253 titles, of which 12 050 titles were excluded after screening titles and abstracts and removing duplicates (figure 1). Full-text screening was completed for 203 publications, and, ultimately, 34 articles representing coinfections in 10 countries were included (tables 1–4 and figure 2).
Figure 1.
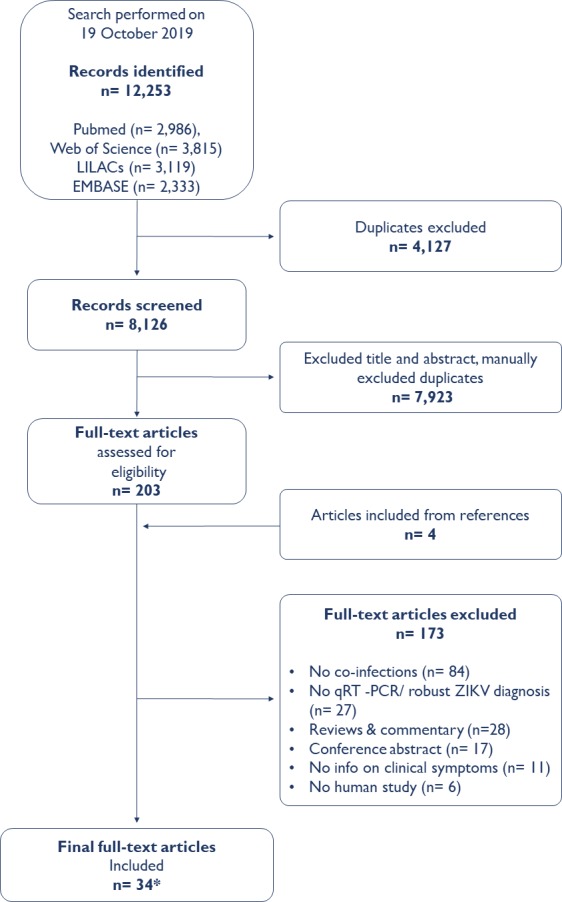
Study selection.
*Studies reporting on both clinical signs and symptoms and frequency of Zika virus coinfections (n=27); studies reporting only on Zika virus coinfection frequencies (n=7).qRT-PCR, quantitative real-time PCR; ZIKV, Zika virus.
Table 1.
ZIKV/CHIKV, ZIKV/DENV and ZIKV/CHIKV/DENV coinfection frequencies among qRT-PCR-confirmed ZIKV infected study population (n=11 studies)
| Author (year) | Country/ Territory | Region | Study year | Study design | Study population* | Coinfecting agent(s) | Coinfection cases (n) | qRT-PCR-confirmed ZIKV infected study population (n) | Frequency (%) |
Level of evidence† |
| Mercado-Reyes et al28 (2018) | Colombia | N/A‡ | Oct 2015– Dec 2016 |
Cross sectional | Suspected arbovirus infections | CHIKV | 28 | 10 118 | 0.3% | 2c |
| Brasil et al5 (2016) |
Brazil | South-East | Sept 2015– May 2016 |
Cohort | Pregnant women with rash | CHIKV | 3 | 182 | 1.7% | 2b |
| Magalhaes et al12 (2017) |
Brazil | North-East | May 2015– May 2016 |
Cross sectional | Suspected arbovirus infections | CHIKV | 2 | 26 | 7.7% | 2c |
| Waggoner et al13 (2016) | Nicaragua | N/A | Sept 2015– Apr 2016 |
Cross sectional | Suspected arbovirus infections | CHIKV | 16 | 75 | 21.3% | 2c |
| Carrillo- Hernández et al9 (2018) | Colombia | East | Aug 2015– Apr 2016 |
Cross sectional | Suspected arbovirus infections | CHIKV | 10 | 29 | 27.6% | 2c |
| Charlys da Costa et al14 (2017) | Brazil | North-East | Mar 2016– May 2016 |
Cross sectional | Suspected arbovirus infections with rash | CHIKV | 36 | 66 | 54.0% | 2c |
| Mercado-Reyes et al28 (2018) | Colombia | N/A‡ | Oct 2015– Dec 2016 |
Cross sectional | Suspected arbovirus infections | DENV | 3 | 10 118 | 0.03% | 2c |
| Chia et al15 (2017) |
Singapore | Singapore | Aug 2016– Sept 2016 |
Case series | Suspected ZIKV infections | DENV | 4 | 163 | 2.4% | 4 |
| Pessôa et al16 (2016) |
Brazil | North-East | May 2015 | Case series | Suspected arbovirus infections | DENV | 1 | 31 | 3.2% | 4 |
| Colombo et al17 (2017) |
Brazil | South-East | Jan 2016–Nov 2016 | Cross sectional | Suspected ZIKV infections | DENV | 4 | 100 | 4.0% | 2c |
| Estofolete et al18 (2018) |
Brazil | South-East | Jan 2016–Nov 2016 | Case series | Suspected arbovirus infections | DENV | 12 | 151 | 7.9% | 4 |
| Waggoner et al (2016)13 | Nicaragua | N/A | Sept 2015 –Apr 2016 | Cross sectional | Suspected arbovirus infections | DENV | 6 | 75 | 8.0% | 2c |
| Carrillo- Hernández et al9 (2018) | Colombia | East | Aug 2015– Apr 2016 |
Cross sectional | Suspected arbovirus infections | DENV | 12 | 29 | 34.4% | 2c |
| Azeredo et al19 (2018) |
Brazil | Central-West | Feb 2016– Mar 2016 |
Cross sectional | Suspected ZIKV infections | DENV | 18 | 38 | 47.7% | 2c |
| Waggoner et al13 (2016) | Nicaragua | N/A | Sept 2015– Apr 2016 |
Cross sectional | Suspected arbovirus infections | CHIKV/ DENV |
6 | 75 | 8.0% | 2c |
| Carrillo-Hernández et al9 (2018) | Colombia | East | Aug 2015– Apr 2016 |
Cross sectional | Suspected arbovirus infections | CHIKV/ DENV |
8 | 29 | 27.6% | 2c |
*Online supplementary table 2 gives details of all study populations in cohort studies and case reports.
†All articles were rated according to level of evidence using the Oxford Centre for Evidence-based Medicine's Levels of Evidence, March 2009.11
‡Cases originated from the National Surveillance System in Public Health from Colombia. Therefore, cases come from all over Colombia, with the condition of living in a place 2200 m above sea level.
CHIKV, chikungunya virus; DENV, dengue virus; N/A, not available; ZIKV, Zika virus.
Table 2.
Summary of cohort and cross-sectional studies reporting on signs and symptoms of qRT-PCR-confirmed ZIKV coinfections (n=6 studies)
| Author (year) |
Location | Study year | Study design | Study population* | % female | Mean age (years) | N, total study population | Coinfecting agent(s) | N, coinfection cases | Other pathogens tested (negative) |
Frequency of WHO ZIKV signs and symptoms (%) |
Frequency of other reported signs and symptoms (%) | Level of evidence† | ||||||
| Rash | Fever | Arthralgia | Conjunctivitis | Myalgia | Headache | URT symptoms | GI symptoms | ||||||||||||
| Mercado-Reyes et al28 (2018) | Colombia | 2015/2016 | Cross-sectional study | Suspected arbovirus infections | 74 | 28 | 23 871 | CHIKV | 28 | DENV | NR | NR | NR | NR | NR | NR | NR | NR | 2c |
| Ball et al20 (2018) | Haiti | 2014/2015 | Cohort study | AFI cases | 48 | 7.5 | 252 | CHIKV | 6 | DENV2/ MAYV |
0 | 33% | 67% | NR | 67% | 17% | NR | 17% | 2b |
| Azeredo et al19 (2018) | Brazil | 2016 | Cross-sectional study | Suspected arbovirus infections | NR | 34 | 134 | DENV‡ | 18§ | CHIKV | 53% | 73% | 87% | 60% | 93% | 67% | 13% | 40%¶ | 2c |
| Mercado-Reyes et al28 (2018) | Colombia | 2015/2016 | Cross-sectional study | Suspected arbovirus infections | 74 | 28 | 23 871 | DENV | 3 | CHIKV | NR | NR | NR | NR | NR | NR | NR | NR | 2c |
| Araújo et al38 (2019) | Brazil | 2014/2016 | Cross-sectional study | AFI cases | NR | NR | 9 | DENV | 1 | CHIKV | 100% | NR | 100% | NR | NR | 100% | NR | 100% | 2c |
| Alva-Urcia et al30 (2016) | Peru | 2016 | Cross-sectional study | AFI cases | 63 | NR | 139 | DENV | 1 | NR | NR | 100% | NR | NR | NR | NR | NR | NR | 2c |
| de Souza Costa et al42 (2019) |
Brazil | 2015/2016 | Cross-sectional study | AFI cases | 59 | NR | 453 | MAYV | 1 | DENV, CHIKV, YFV, SLEV, ILHV, ROCV, WNV, EEEV, WEEV, VEEV |
NR** | NR** | NR** | NR** | NR** | NR** | NR** | NR** | 2c |
URT symptoms: pharyngitis, sore throat, cough, pharyngeal congestion, adenopathy. GI symptoms: nausea, diarrhoea, vomiting, constipation, stomach ache.
*Online supplementary table 2 gives details of all study populations.
†All articles were rated according to level of evidence using the Oxford Centre for Evidence-based Medicine’s Levels of Evidence, March 2009.11
‡Seventeen cases of DENV-1 and one case of DENV-4.
§Signs and symptoms were only reported for 15 patients.
¶Forty per cent of cases presented with nausea, and 13% of cases with vomiting.
**Case was described to present with typical ZIKV signs for 3 days, but specific details were not reported.
AFI, acute febrile illness; CHIKV, chikungunya virus; DENV, dengue virus; EEV, east equine encephalitis virus; GI, gastrointestinal; ILHV, Ilheus virus; MAYV, Mayaro virus; NR, not reported; ROCV, Rocio virus; SLEV, Saint Louis encephalitis virus; URT, upper respiratory tract; VEEV, Venezuelan equine encephalitis virus; WEEV, west equine encephalitis virus; WNV, West Nile virus; YFV, yellow fever virus; ZIKV, Zika virus.
Table 3.
Summary of case series studies reporting on signs and symptoms of different qRT-PCR-confirmed ZIKV coinfections (n=7 studies)
| Author (year) |
Location | Study year | Study design | Study population* | % female | Mean age (years) | N, total study population | Coinfecting agent(s) | N, coinfection cases | Other pathogens tested (negative) | Frequency of WHO ZIKV signs and symptoms (%) |
Frequency of other reported signs and symptoms (%) | Level of evidence† | ||||||
| Rash | Fever | Arthralgia | Conjunctivitis | Myalgia | Headache | URT symptoms | GI symptoms | ||||||||||||
| Acevedo et al (2017)22 | Ecuador | 2016 | Case series |
Cases with neurological symptoms‡ | 38 | 42 | 16 | CHIKV | 3 | § | NR | 100% | 33% | NR | NR | 33% | NR | NR | 4 |
| Metha et al (2018)29 | Brazil | 2015/2016 | Case series |
Cases with neurological symptoms§ | 50 | 52 | 22 | CHIKV | 2 | DENV | 100% | 50% | 50% | NR | NR | NR | NR | NR | 4 |
| Sardi et al (2016)23 | Brazil | 2015 | Case series |
AVI and of qRT-PCR ZIKV+ infections | NR | NR | 15 | CHIKV | 2 | DENV | 50% | 100% | 50% | 50% | 100% | 50% | NR | 50% | 4 |
| Cabral-Castro et al (2016)21 | Brazil | 2015/2016 | Case series |
Suspected DENV infections | NR | NR | 30 | CHIKV | 1 | DENV | 100% | 100% | 0 | 0 | NR | NR | NR | 100% | 4 |
| Estofolete et al (2018)18 | Brazil | 2016 | Case series |
Suspected arbovirus infections | 42 | 46 | 1254 | DENV | 12 | CHIKV | 58% | 58% | 50% | 25% | 83% | 75% | NR | 17% | 4 |
| Chia et al (2017)15 |
Singapore | 2016 | Case series |
Suspected ZIKV infections | NR | NR | 163 | DENV | 4 | NR | 100% | 100% | 50% | 50% | 75% | 50% | 25% | 75% | 4 |
| Li et al (2017)31 | Singapore | 2016 | Case series |
Suspected ZIKV infections | 50 | 11 | 14 | DENV | 1 | CHIKV | 100% | 100% | 100% | 100% | 100% | 100% | 0 | NR | 4 |
| Acevedo et al (2017)22 | Ecuador | 2015/2016 | Case series |
Cases with neurological symptoms‡ | 38 | 42 | 16 | CHIKV, DENV |
4 | § | NR | 50% | NR | NR | NR | 25% | 25% | 25% | 4 |
| Acevedo et al (2017)22 | Ecuador | 2016 | Case series |
Cases with neurological symptoms‡ | 38 | 42 | 16 | CHIKV, HIV, Toxo |
1 | § | NR | 100% | NR | NR | NR | 100% | NR | NR | 4 |
URT symptoms: pharyngitis, sore throat, cough, pharyngeal congestion, adenopathy. GI symptoms: nausea, diarrhoea, vomiting, constipation, stomach ache.
*Online supplementary table 2 gives details of all study populations.
†All articles were rated according to level of evidence using the Oxford Centre for Evidence-based Medicine’s Levels of Evidence, March 200911
‡Associated with suspected arbovirus infection.
§Tested for DENV; gram stain, HSV1/2/6, CMV, EBV, VZ, Toxo, MTB, enterovirus.
AVI, acute viral illness; CHIKV, chikungunya virus; CMV, cytomegalovirus; DENV, dengue virus; GI, gastrointestinal; HSV, herpes simplex virus; MT, Mycobacterium tuberculosis; NR, not reported; Toxo, Toxoplasma gondii; URT, upper respiratory tract; VZ, varicella zoster.
Table 4.
Summary of case reports reporting on signs and symptoms of qRT-PCR-confirmed ZIKV coinfections (n=21 reports)
| Author (year) |
Location | Study year | Sex | Age (years) | Coinfecting agent(s) | Other pathogens tested (negative) |
WHO ZIKV signs or symptoms | Other reported signs or symptoms | Additional information | Level of evidence* | ||||||
| Rash | Fever | Arthralgia | Conjunctivitis | Myalgia | Headache | URT symptoms | GI symptoms | |||||||||
| Brito et al24 (2017) | Brazil | 2016 | M | 74 | CHIKV | DENV, HIV, CMV, HTLV, Schisto, HSVI/2 cystcercosis |
NR | † | † | NR | NR | NR | NR | † | Complications: Meningoencephalitis associated with Guillain-Barré syndrome (EMG confirmed) | 5 |
| Silva et al (2018) | Brazil | 2016 | M | 30 | CHIKV | DENV, bacterial/fungal and MTB infections | NR | † | † | NR | † | NR | NR | NR |
Comorbidities: Systemic lupus erythematosus Complications: Persistent fever for 5 weeks and renal dysfunction. 2 months postinfection severe arthralgia, requiring arthroplasty. 10 months postinfection acute deterioration in renal function, respiratory insufficiency and sepsis. Outcome: Death |
5 |
| Cherabuddi et al26 (2016) |
Colombia | 2016 | F | 40 | CHIKV | DENV | † | † | † | † | NR | NR | NR | NR | Back pain, retro-ocular pain Complications: Persistent severe arthralgia after 2 months Outcome: Sequelae |
5 |
| Zambrano et al27 (2016) | Ecuador | 2016 | M | 43 | CHIKV | DENV | † | † | † | † | NR | NR | † | NR | Outcome: Full recovery | 5 |
| Zambrano et al27 (2016) | Ecuador | 2016 | F | 43 | CHIKV | NR | ‡ | † | ‡ | † | † | † | NR | NR | Outcome: Full recovery | 5 |
| Zambrano et al27 (2016) | Ecuador | 2016 | F | 57 | CHIKV | DENV | NR | † | NR | NR | NR | † | NR | NR | Lumbar back pain Complications: Guillain-Barré syndrome (EMG confirmed) Outcome: Full recovery |
5 |
| Azeredo et al19 (2018) |
Brazil | 2016 | F | NR | DENV | CHIKV | † | † | NR | NR | NR | † | NR | † | Retro-orbital pain Complications: Suspected vertical transmission, pregnancy outcome alive newborn with functional plagiocephaly a flat spot on the back or side of newborn’s skull Gestational age at infection: 9 weeks |
5 |
| Azeredo et al19 (2018) |
Brazil | 2016 | F | NR | DENV | CHIKV | † | † | NR | † | NR | NR | NR | NR | Retro-orbital pain Complications: Suspected vertical transmission to deceased newborn with respiratory insuf-ficiency Gestational age at infection: 20 weeks |
5 |
| Dupont-Rouzeyrol et al32 (2015) | New Caledonia | NR | F | 38 | DENV 1 | NR | † | † | † | † | † | † | NR | † | Asthenia, retro-ocular pain Outcome: Full recovery |
5 |
| Dupont-Rouzeyrol et al32 (2015) | New Caledonia | NR | M | 14 | DENV 3 | NR | NR | † | † | NR | † | † | NR | NR | Asthenia Outcome: Full recovery |
5 |
| Iovine et al 33 (2017) | United States§ | 2016 | F | 26 | DENV 2 | CHIKV | † | † | NR | † | NR | NR | † | † | Retro-orbital pain, fatigue, malaise, facial flushing Outcome: Full recovery |
5 |
| Villamil-Gomez et al35 (2016) | Colombia | NR | F | 33 | CHIKV, DENV |
Plasmodium spp | † | NR | † | † | ‡ | † | NR | ‡ | Physical examination revealed cervical lymphadenopathy, bipalpebral oedema, and painful oedema in the lower limbs Outcome: Weekly obstetric ultrasounds from 14.6 to 29 weeks of gestation were normal |
5 |
| Gunturiz et al36 (2018) | Colombia | 2016 | F | 18 | CHIKV, Toxo |
NR | NR | NR | NR | NR | NR | NR | NR | NR | Complications: Suspected vertical transmission of ZIKV infections to the fetus with outcome of fetus diagnosed with CZS at 20 weeks of gestation, termination at 29 weeks of gestation | 5 |
| Villamil-Gomez et al34 (2018) | Colombia | 2015/2016 | M | 28 | HIV | NR | † | † | NR | † | NR | NR | NR | NR | Recently diagnosed with HIV (<1 year), Lymphocytes T CD4 count (cells/mm3): 450, HIV viral load (RNA copies/mL): 100 Complication: Demyelination was found (EMG findings) |
5 |
| Villamil-Gomez et al34 (2018) | Colombia | 2015/2016 | F | 49 | HIV | NR | NR | NR | NR | NR | NR | NR | NR | † | Recently diagnosed with HIV (<1 year) Lymphocytes T CD4 count (cells/mm3): 98 HIV viral load (RNA copies/mL): 1800 Hypotension, dysarthria, decreased muscle strength, relaxation of sphincters, areflexia and basal bilateral crackles in the lungs Complications: Sepsis |
5 |
| Villamil-Gomez et al34 (2018) | Colombia | 2015/2016 | F | 45 | HIV | NR | † | † | NR | † | NR | NR | NR | † | Recently diagnosed with HIV (1 year ago) Lymphocytes T CD4 count (cells/mm3): 380 HIV viral load (RNA copies/mL): 800 Complication: Demyelination was found (EMG findings) |
5 |
| Valdespino- Vazquez et al37 (2018) | Mexico | 2016 | F | 22 | EBV, HHV6 | DENV, CHIKV, WNV, VZ, HSV-I/2, HHV7, HHV8, CMV |
† | † | NR | NR | NR | † | NR | NR | Infection of pregnant women at 14 weeks of gestation Complications: Suspected vertical transmission of ZIKV infections to the foetus with outcome of diagnosed CZS and fetal death at 30 weeks of gestational age, 4 hours after birth |
5 |
| Araujo et al38 (2018) | Brazil | 2016 | M | 26 | HSV-1 | DENV, HSV, VZ, EBV, Toxo, HepC/B, Syphilis spp. |
NR | † | NR | NR | NR | † | NR | † |
Complications: Meningoencephalitis Outcome: Full recovery |
5 |
| Biron et al 39 (2016) | New Caledonia | NR | M | 19 | Leptospira spp | CHIKV, DENV |
NR | † | NR | NR | † | NR | † | NR |
Complications: Haemodynamic condition was unstable, sceptic shock Outcome: Full recovery |
5 |
| Neaterour et al (2017) | Puerto Rico | 2016 | M | 48 | Leptospira spp | DENV, CHIKV |
NR | † | NR | NR | † | † | NR | † |
Complications: Severe thrombocytopenia, persistent hypotension (BP=60/40), and onset of haematochezia suggestive of an acute gastrointestinal bleed. Haemodynamic instability and haemorrhagic manifestations. Cardiac arrest. Outcome: Death. |
5 |
| Alves et al41 (2017) | Brazil | NR | M | NR, a boy | Schistosoma mansoni | NR | † | † | NR | NR | NR | NR | NR | NR |
Complications: Inflammation of the right testicle and epididymis. The microscopic examination of the testis ruled out the possibility of cancer but confirmed the diagnosis of extensive loss of testicular structure and schistosome egg-induced granulomas. Outcome: Full recovery |
5 |
URT symptoms: pharyngitis, sore throat, cough, pharyngeal congestion, adenopathy. GI symptoms: nausea, diarrhoea, vomiting, constipation, stomach ache.
*All articles were rated according to level of evidence using the Oxford Centre for Evidence-based Medicine’s Levels of Evidence, March 2009.11
†Reported to be present.
‡Reported not to be present.
§Travel associated diagnosed from Haiti.
CHIKV, chikungunya virus; CMV, cytomegalovirus; CZS, congenital Zika syndrome; DENV, dengue virus; EMG, electromyography testing; GI, gastrointestinal; Hep, hepatitis virus; HHV, human herpes virus; HSV, herpes simplex virus; HTLV, human T-lymphotropic virus; MTB, Mycobacterium tuberculosis; NR, not reported; Toxo, Toxoplasma gondii; URT, upper respiratory tract; VZ, varicella zoster; WNV, West Nile virus.
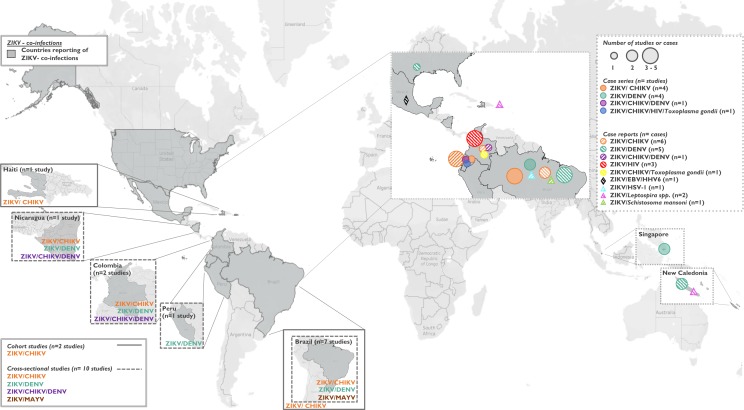
Figure 2.
Studies included in the systematic review: cohort studies (n=2), cross-sectional studies (n=10), case series studies (n=8) and case reports (n=21 reported in 14 case report studies). Two cohort studies on ZIKV/CHIKV coinfections were conducted in Haiti (n=1) and Brazil (n=1). Ten cross-sectional studies were conducted in Brazil (n=6), Colombia (n=2), Nicaragua (n=1) and Peru (n=1). Eight case series were reported from Brazil (n=5), Ecuador (n=1) and Singapore (n=2). Twenty-one case reports were reported from Brazil (n=6), Colombia (n=6), Ecuador (n=3), Mexico (n=1), New Caledonia (n=3), Puerto Rico (n=1) and the USA (n=1). CHIKV, chikungunya virus; DENV, dengue virus; EBV, Epstein-Barr virus; HSV, herpes simplex virus; MAYV, Mayaro virus; ZIKV, Zika virus.
ZIKV coinfection types
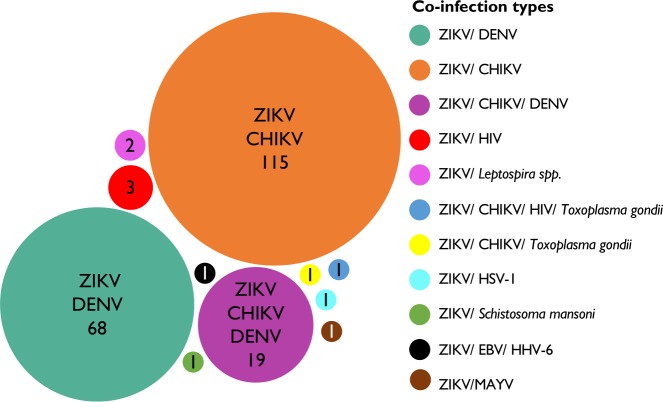
ZIKV infections were most frequently reported to occur concurrently with other arthropod-borne viruses (arboviruses). Out of the 213 coinfections examined, there were 115 ZIKV/chikungunya virus (CHIKV) coinfection cases, 68 ZIKV/dengue virus (DENV) coinfection cases and 19 cases coinfected with all three viruses. Other reported ZIKV coinfections included ZIKV/HIV (n=3), ZIKV/Leptospira spp (n=2), ZIKV/CHIKV/HIV/Toxoplasma gondii (n=1), ZIKV/CHIKV/Toxoplasma gondii (n=1), ZIKV/Epstein-Barr virus (EBV)/human herpes viruses-6 (HHV-6) (n=1), ZIKV/herpes simplex virus-1 (HSV-1) (n=1), ZIKV/Mayaro virus (MAYV) (n=1) and ZIKV/Schistosoma mansoni (n=1) (figure 3).
Figure 3.
Zika virus coinfection types identified in this systematic review. Size of circles represents the number of cases reported per coinfection type. In total, 213 coinfection cases were included, ie, ZIKV/CHIKV (n=115), ZIKV/DENV (n=68), ZIKV/CHIKV/DENV (n=19), ZIKV/HIV (n=3), ZIKV/Leptospira spp (n=2), ZIKV/HIV/Toxoplasma gondii (n=1), ZIKV/CHIKV/Toxoplasma gondii (n=1), ZIKV/HSV-1 (n=1), ZIKV/Schistosoma mansoni (n=1), ZIKV/EBV/HHV-6 (n=1), ZIKV/MAYV (n=1. CHIKV, chikungunya virus; DENV, dengue virus; EBV, Epstein-Barr virus; HHV, human herpes virus; HSV, herpes simplex virus; MAYV, Mayaro virus; ZIKV, Zika virus.
Frequencies of ZIKV coinfections
The frequencies of coinfections among ZIKV-infected populations were reported in 11 studies, including 1 cohort study, 7 cross-sectional studies and 3 case series (table 1, online supplementary table 2). Frequency estimates were reported only for coinfections with CHIKV and DENV and varied geographically and across study populations at risk. Among patients presenting with arbovirus-like symptoms, ZIKV/CHIKV coinfection frequencies were reported to range from 0.3% in a study in Colombia to 54% in a study in Brazil.5 9 12–14 Similarly, ZIKV/DENV coinfection frequencies in patients with arbovirus-like symptoms were reported to range from 0.03% in a study in Colombia to 47.4% in a study in Brazil.9 13 15–19 ZIKV/CHIKV/DENV coinfection frequencies ranged from 8% in a study in Nicaragua to 27.6% in a study in Colombia.9 13
Signs and symptoms of coinfections
In total, 27 studies, including 1 cohort study, 5 cross-sectional studies, 7 case series and 14 case report studies, reported the signs and symptoms of ZIKV coinfection across a total of 106 ZIKV-coinfected cases.
ZIKV/CHIKV coinfections
The clinical presentations of 48 cases with ZIKV/CHIKV coinfection were reported in 1 cohort study, 1 cross-sectional study, 4 case series and 6 case reports (tables 2–4, online supplementary tables 1 and 2).20–28 Within the cohort, cross-sectional and case series studies, cases were reported to present with the following signs and symptoms consistent with the WHO ZIKV clinical case definition1: fever (33%–100%), rash (0%–100%), conjunctivitis (0%–50%), myalgia (67%–100%), arthralgia (0%–67%) and headache (17%–50%) (tables 2 and 3). In addition, gastrointestinal (GI) symptoms were reported in 17% to 100% of cases in three studies (tables 2 and 3).20 21 23
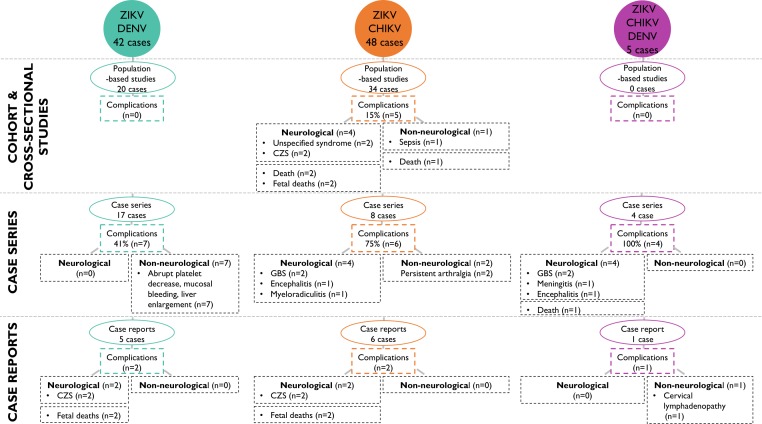
Complications were reported among 14.7% (5 cases) of ZIKV/CHIKV-coinfected cases in cohort and cross-sectional studies,22–27 of whom two adult cases presented with unspecified neurological complications that resulted in death (figure 4).28 Additionally, two coinfections in pregnancy were associated respectively with anencephaly and an absence of a heartbeat.28 A non-neurological complication reported was a case that died from multiorgan failure following haemorrhagic manifestations.23 28 The case series studies described that six out of eight ZIKV/CHIKV-coinfected cases developed complications, which included neurological manifestations, such as GBS in two cases,22 29 encephalitis in one case,22 myeloradiculitis in one case,29 as well as non-neurological complications, such as persistent severe arthralgia in one case.23 Additionally, four case reports described ZIKV/CHIKV coinfection-associated complications, including GBS in two cases,24 27 persistent severe arthralgia in one case26 and sepsis resulting in death in one case.25
Figure 4.
Complications resulting from Zika virus coinfections with CHIKV and DENV by study design. In cohort and cross-sectional studies, 15% of ZIKV/CHIKV coinfections resulted in complications. In case series, 41% of ZIKV/DENV, 75% of ZIKV/CHIKV and 100% of ZIKV/CHIKV/DENV cases resulted in in complications. In case reports, two ZIKV/DENV, two ZIKV/CHIKV and one ZIKV/CHIKV/DENV coinfections resulted in complications. CHIKV, chikungunya virus; CZS, congenital Zika syndrome; DENV, dengue virus; GBS, Guillain-Barré syndrome; n, number of complications; ZIKV, Zika virus.
ZIKV/DENV coinfections
The clinical features of 42 cases with ZIKV/DENV coinfection were described across four cross-sectional studies, three case series and five case reports (tables 2–4, online supplementary tables 1 and 2).15 18 19 30–33 Cases with ZIKV/DENV coinfection within the cross-sectional and case series studies were reported to present with the following signs and symptoms consistent with the WHO ZIKV clinical case definition1: fever (58%–100%), rash (53%–100%), conjunctivitis (25%–100%), myalgia (75%–100%), arthralgia (50%–100%) and headache (50%–100%) (tables 2 and 3). Other reported clinical features included GI symptoms in 17%–75% of cases and upper respiratory tract (URT) symptoms in 13%–25% of cases (tables 2 and 3).
Complications were reported among none of the ZIKV/DENV-coinfected individuals in cohort and cross-sectional studies (figure 4). However, seven cases with complications were reported in case series, which presented respectively with painful hepatomegaly, liver enlargement, mucosal bleeding, gingival bleeding, significant thrombocytopenia and abrupt platelet decrease.15 18 19 The only neurological complications resulting from ZIKV/DENV coinfection were reported in two case reports documenting infections in pregnancy, with one case resulting in a newborn with functional plagiocephaly and the other in fetal death (table 3).19
ZIKV/CHIKV/DENV coinfections
The clinical presentation of five cases with ZIKV/CHIKV/DENV coinfection were described in one case series (four cases) and one case report (tables 3 and 4, online supplementary tables 1 and 2).22 34 Similar to ZIKV/CHIKV and ZIKV/DENV-coinfected cases, ZIKV/CHIKV/DENV-coinfected cases presented with signs and symptoms consistent with the ZIKV WHO clinical case definition.1 All five cases were reported to have complications (figure 4). The case series reported GBS in two cases, one case of meningitis and one case of encephalitis, which resulted in death. Notably, the study’s population was selected to include only clinical patients presenting to hospital with neurological symptoms.22 The case report documented one case of cervical lymphadenopathy in pregnancy and full recovery.35
Other ZIKV coinfections
There is limited published evidence on ZIKV coinfections with other pathogens. To date, the clinical signs and symptoms of 10 cases with eight other ZIKV coinfection types have been documented in one cross-sectional study, one case series and seven case reports (tables 2–4, online supplementary tables 1 and 2).34 36–42
In addition to presenting with signs and symptoms consistent with the WHO ZIKV clinical case definition, almost all cases of ZIKV coinfections with pathogens other than DENV or CHIKV were reported to experience complications. Neurological complications were reported in two ZIKV/HIV coinfections, one ZIKV/CHIKV/HIV/Toxoplasma gondii coinfection and one ZIKV/HSV-1 coinfection. These neurological complications included meningitis, meningoencephalitis and demyelinations confirmed by electromyography.22 34 38 Further, one ZIKV/HIV-coinfected case developed sepsis, resulting in death.34 Two ZIKV/Leptospira spp-coinfected cases developed haemodynamic instability, one resulting in septic shock, and one in death.39 40 Additionally, one ZIKV/Schistosma mansoni-coinfected case experienced testicular inflammation with granulomas induced by schistosome eggs.41
Coinfections in pregnancy were described in three ZIKV coinfection types: ZIKV/MAYV, ZIKV/CHIKV/Toxoplasma gondii and ZIKV/EBV/HHV6 coinfections.36 37 42 In the latter two, vertical ZIKV transmission was suspected, as both fetuses were diagnosed with CZS. After diagnosis, one pregnancy was terminated at 29 weeks of gestation and one newborn died 4 hours after birth at 30 weeks of gestation due to respiratory distress syndrome.
Levels of evidence
The levels of evidence for the studies were assessed using the OCEBM Levels of Evidence (1=highest, 5=lowest). Two cohort studies with limited follow-up were graded evidence level 2b.5 20 Ten cross-sectional studies were graded evidence level 2c.9 12–14 17 19 28 30 42 43 Eight case series studies were graded evidence level 4.15 16 18 21–23 29 31 Fourteen case report studies were graded evidence level 5.19 24–27 32–41 Thus, most of the studies included in the systematic review are evidence level 4 or 5.
Discussion
This systematic review summarises the existing literature on ZIKV coinfections. Specifically, it describes the estimated frequencies of reported ZIKV coinfections and their clinical spectrum. The search identified 34 studies conducted between 2014 and 2019, which reported 213 cases of ZIKV coinfection with 10 different pathogens. ZIKV coinfections were detected across 10 countries, primarily in Latin America. CHIKV and DENV were the predominantly reported ZIKV coinfecting agents and the only ZIKV coinfections for which population frequencies were described. ZIKV coinfection frequencies among ZIKV-infected cases varied significantly between location and population type. The vast majority of ZIKV-coinfected cases were reported to present with the signs and symptoms described for uncomplicated ZIKV monoinfections and defined by the WHO.1 However, complications were reported to arise in 9% of ZIKV-coinfected cases in cohort and cross-sectional studies.
This is the first systematic review to study how frequently individuals with ZIKV infection have a coexisting infection of any kind. The variation in frequencies reported for ZIKV/arbovirus coinfections among the ZIKV-infected individuals reported in this study was likely influenced by differences in study design and the selected study population. Factors, such as study location, season and study period in relation to the ZIKV outbreak, will have additionally influenced ZIKV coinfection frequency estimates. As expected, ZIKV coinfections were relatively more common in studies conducted during concurrent arbovirus outbreaks.14 44 These differences in study design, timing and location make it difficult to generalise ZIKV coinfection frequency estimates, but provide important knowledge that arbovirus coinfections can occur in up to half of ZIKV-infected cases in certain contexts. Our findings are consistent with a systematic review of CHIKV/DENV coinfections, which found the frequency of CHIKV/DENV coinfections reported in 28 studies ranged from 1% to 36%.45 The heterogeneity across studies also reflects the difficulty in estimating the background level of ZIKV infections (ie, the denominator for assessing coinfection frequencies), given the diagnostic challenges in identifying acute ZIKV infections.46
Overall, the evidence identified in this review suggests that ZIKV coinfections appear to present with a mild clinical presentation similar to that previously described for ZIKV monoinfections. Of note, GI and URT symptoms, which are considered uncharacteristic for ZIKV, were reported to occur not infrequently in ZIKV/DENV, ZIKV/CHIKV and ZIKV/CHIKV/DENV-coinfected cases. While the evidence base from animal model studies of ZIKV coinfection is limited to date, two studies have compared ZIKV infection among rhesus macaque models with and without simian immunodeficiency virus or chimeric simian HIV.47 48 Whereas coinfected macaques were observed to have lower peak Zika viral loads with a longer clearance time in both investigations, the area under the viral load curves did not appear to differ substantively by coinfection status, potentially suggesting an overall limited impact of coinfection on disease progression but raising questions about the role of lentiviral coinfection in onward transmission.47 48
Although the existing reports suggest that coinfections do not appear to markedly alter the clinical presentation of uncomplicated ZIKV disease in humans, the findings from this review highlight a need for additional high quality research investigating whether coinfections may influence complication risks. Based on the limited available evidence, the complications described for ZIKV coinfections appear to be broadly similar to those reported for ZIKV monoinfections.49 However, 33% of the coinfection-related complications appeared to be atypical for ZIKV monoinfections, but were consistent with complications previously documented for the coinfecting pathogens (eg, bleeding in 10% of ZIKV/DENV cases and persistent arthralgia in 6% of ZIKV/CHIKV cases).50 51 In addition, among deaths of ZIKV-coinfected cases, three of the nine cases had immune deficiencies and one ZIKV/Leptospira spp-coinfected case died from complications established for Leptospira spp infections.40 The remaining five deaths reported from ZIKV coinfections were three fetal deaths, one case following multiorgan failure and one case following encephalitis.22 28 Additionally, some complications may have been missed, especially those that occurred after the acute infections, as the follow-up period of the individual studies may have not been adequate to detect late-onset complications. Further research (eg, an ongoing cohort study of ZIKV/HIV coinfections in pregnant women52) will be valuable for discerning the relative risk of complications of ZIKV coinfection versus monoinfections.
This review had strengths and limitations. ZIKV is an emerging infectious disease of significant public health concern, and this is the first systematic review of the frequency, types and clinical presentation of ZIKV coinfections. The study employed a broad search strategy including search terms for all potential coinfecting pathogens and using multiple languages to identify all available evidence. Most importantly, the review included only qRT-PCR-confirmed ZIKV coinfections, which is the most accurate way to diagnose acute coinfections (ie, due to the very short time window of qRT-PCR testing (<7 days)) and limits misdiagnosis, which is of particular importance with the high cross-reactivity reported from arbovirus serology testing. On the other hand, by focusing on concurrent infections, the current review was unable to appraise the potential impact of recent infections; for example, it has been previously reported that pre-existing immunity to DENV, which shares a common vector and circulates in most of the countries reporting ZIKV coinfection, may influence the clinical presentation of ZIKV infection.53 The additional limitations of this review mainly stem from the lack of available high-quality evidence on ZIKV coinfections. Notably, the majority of included studies were rated level 4 or 5 according to the OCEBM Levels of Evidence. Only seven studies were rated level 2 or above. Additionally, the reported ZIKV coinfection types may have been influenced by the underlying prevalence of coinfecting pathogens in the population and the applied diagnostic practices (ie, multiplex testing vs testing on clinician's suspicion). The use of specific case definitions in included cross-sectional and case series studies (eg, fever and rash15) may have also introduced a selection bias that potentially led to an over-representation of specific symptoms associated with ZIKV coinfection reported for a given study (eg, reporting 100% of cases as presenting with fever and rash).15 Finally, the studies selected for this systematic review only included symptomatic ZIKV-infected cases, which represent only approximately 40% of all ZIKV cases.2 It is likely that the actual frequency of ZIKV coinfections may be higher as many cases will be asymptomatic and therefore never seek medical attention. However, the recently implemented multiplex PCR assay, which tests for CHIKV, DENV and ZIKV simultaneously, will likely improve the detection of ZIKV/arbovirus coinfections and facilitate future assessment of the frequency of ZIKV coinfections.54
In conclusion, the findings of this review suggest that the cocirculating arboviruses, CHIKV and DENV, are the most common ZIKV coinfection types and may, in specific populations and epidemiological contexts, occur in up to half of ZIKV infections. The evidence collated in this systematic review suggests coinfections do not markedly alter the generally mild clinical presentation of uncomplicated ZIKV disease. However, additional and better quality evidence should be prioritised in future outbreaks to corroborate the estimates of the frequency of ZIKV coinfections and to interrogate the importance of ZIKV coinfections in the development of ZIKV-related complications, especially for ZIKV coinfections with CHIKV and DENV.
Footnotes
Handling editor: Alberto L Garcia-Basteiro
Twitter: @ebbrickley
Contributors: All authors contributed substantially to the design of the work and/or the acquisition, analysis and interpretation of the data, contributed meaningfully to the drafting and/or revision of the manuscript, provided final approval for the version published and share responsibility for the published findings.
Funding: This project was supported by the European Union’s Horizon 2020 research and innovation programme (https://ec.europa.eu/programmes/horizon2020/) under ZikaPLAN grant agreement No. 734 584 (https://zikaplan.tghn.org/), and the Wellcome Trust & the UK’s Department for International Development (205377/Z/16/Z; https://wellcome.ac.uk/). CMTM receives CNPq scholarship #308974/2018‐2.
Disclaimer: The funders had no role in the design and conduct of the study, the collection, management, analysis, and interpretation of the data, the preparation, review, or approval of the manuscript, or the decision to submit the manuscript for publication.
Competing interests: None declared.
Patient and public involvement: Patients and/or the public were not involved in the design, or conduct, or reporting or dissemination plans of this research.
Patient consent for publication: Not required.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: All data relevant to the study are included in the article or uploaded as supplementary information. This systematic review is based on published articles. All abstracted data are provided in the text and supplementary materials.
References
- 1.World Health Organization Zika virus: Factsheet, 2018. Available: https://www.who.int/en/news-room/fact-sheets/detail/zika-virus
- 2.Haby MM, Pinart M, Elias V, et al. Prevalence of asymptomatic Zika virus infection: a systematic review. Bull World Health Organ 2018;96:402–13. 10.2471/BLT.17.201541 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.de Araújo TVB, Rodrigues LC, de Alencar Ximenes RA, et al. Association between Zika virus infection and microcephaly in Brazil, January to may, 2016: preliminary report of a case-control study. Lancet Infect Dis 2016;16:1356–63. 10.1016/S1473-3099(16)30318-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mier-Y-Teran-Romero L, Delorey MJ, Sejvar JJ, et al. Guillain-Barré syndrome risk among individuals infected with Zika virus: a multi-country assessment. BMC Med 2018;16:67. 10.1186/s12916-018-1052-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brasil P, Pereira JP, Moreira ME, et al. Zika virus infection in pregnant women in Rio de Janeiro. N Engl J Med 2016;375:2321–34. 10.1056/NEJMoa1602412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hoen B, Schaub B, Funk AL, et al. Pregnancy outcomes after ZIKV infection in French territories in the Americas. N Engl J Med 2018;378:985–94. 10.1056/NEJMoa1709481 [DOI] [PubMed] [Google Scholar]
- 7.Nogueira ML, Nery Júnior NRR, Estofolete CF, et al. Adverse birth outcomes associated with Zika virus exposure during pregnancy in São José do Rio Preto, Brazil. Clin Microbiol Infect 2018;24:646–52. 10.1016/j.cmi.2017.11.004 [DOI] [PubMed] [Google Scholar]
- 8.Rodriguez-Morales AJ, Cardona-Ospina JA, Ramirez-Jaramillo V, et al. Diagnosis and outcomes of pregnant women with Zika virus infection in two municipalities of Risaralda, Colombia: second report of the ZIKERNCOL study. Travel Med Infect Dis 2018;25:20–5. 10.1016/j.tmaid.2018.06.006 [DOI] [PubMed] [Google Scholar]
- 9.Carrillo-Hernández MY, Ruiz-Saenz J, Villamizar LJ, et al. Co-circulation and simultaneous co-infection of dengue, Chikungunya, and Zika viruses in patients with febrile syndrome at the Colombian-Venezuelan border. BMC Infect Dis 2018;18:61. 10.1186/s12879-018-2976-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vogels CBF, Rückert C, Cavany SM, et al. Arbovirus coinfection and co-transmission: a neglected public health concern? PLoS Biol 2019;17:e3000130. 10.1371/journal.pbio.3000130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Phillips B. Oxford Centre for Evidence-based Medicine - Levels of Evidence, 2009. Available: http://www.cebm.net/index.aspx?o=1025
- 12.Magalhaes T, Braga C, Cordeiro MT, et al. Zika virus displacement by a Chikungunya outbreak in Recife, Brazil. PLoS Negl Trop Dis 2017;11:e0006055. 10.1371/journal.pntd.0006055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Waggoner JJ, Gresh L, Vargas MJ, et al. Viremia and clinical presentation in Nicaraguan patients infected with Zika virus, Chikungunya virus, and dengue virus. Clin Infect Dis 2016;63:1584–90. 10.1093/cid/ciw589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Charlys da Costa A, Thézé J, Komninakis SCV, et al. Spread of Chikungunya virus East/Central/South African genotype in northeast Brazil. Emerg Infect Dis 2017;23:1742–4. 10.3201/eid2310.170307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chia PY, Yew HS, Ho H, et al. Clinical features of patients with Zika and dengue virus co-infection in Singapore. J Infect 2017;74:611–5. 10.1016/j.jinf.2017.03.007 [DOI] [PubMed] [Google Scholar]
- 16.Pessôa R, Patriota JV, Lourdes de Souza Mde, et al. Investigation into an outbreak of dengue-like illness in Pernambuco, Brazil, revealed a cocirculation of Zika, Chikungunya, and dengue virus type 1. Medicine 2016;95:e3201. 10.1097/MD.0000000000003201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Colombo TE, Estofolete CF, Reis AFN, et al. Clinical, laboratory and virological data from suspected ZIKV patients in an endemic arbovirus area. J Clin Virol 2017;96:20–5. 10.1016/j.jcv.2017.09.002 [DOI] [PubMed] [Google Scholar]
- 18.Estofolete CF, Terzian ACB, Colombo TE, et al. Co-Infection between Zika and different dengue serotypes during DENV outbreak in Brazil. J Infect Public Health 2019;12:178–81. 10.1016/j.jiph.2018.09.007 [DOI] [PubMed] [Google Scholar]
- 19.Azeredo EL, Dos Santos FB, Barbosa LS, et al. Clinical and laboratory profile of Zika and dengue infected patients: lessons learned from the Co-circulation of dengue, Zika and Chikungunya in Brazil. PLoS Curr 2018;10. 10.1371/currents.outbreaks.0bf6aeb4d30824de63c4d5d745b217f5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ball JD, Elbadry MA, Telisma T, et al. Clinical and epidemiologic patterns of Chikungunya virus infection and coincident arboviral disease in a school cohort in Haiti, 2014-2015. Clin Infect Dis 2019;68:919–26. 10.1093/cid/ciy582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cabral-Castro MJ, Cavalcanti MG, Peralta RHS, et al. Molecular and serological techniques to detect co-circulation of DENV, ZIKV and CHIKV in suspected dengue-like syndrome patients. J Clin Virol 2016;82:108–11. 10.1016/j.jcv.2016.07.017 [DOI] [PubMed] [Google Scholar]
- 22.Acevedo N, Waggoner J, Rodriguez M, et al. Zika virus, Chikungunya virus, and dengue virus in cerebrospinal fluid from adults with neurological manifestations, Guayaquil, Ecuador. Front Microbiol 2017;8:42. 10.3389/fmicb.2017.00042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sardi SI, Somasekar S, Naccache SN, et al. Coinfections of Zika and Chikungunya viruses in Bahia, Brazil, identified by metagenomic next-generation sequencing. J Clin Microbiol 2016;54:2348–53. 10.1128/JCM.00877-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brito CAA, Azevedo F, Cordeiro MT, et al. Central and peripheral nervous system involvement caused by Zika and Chikungunya coinfection. PLoS Negl Trop Dis 2017;11:e0005583. 10.1371/journal.pntd.0005583 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Silva KR, Bica BERG, Pimenta ES, et al. Fatal human case of Zika and Chikungunya virus co-infection with prolonged viremia and viruria. Diseases 2018;6:53. 10.3390/diseases6030053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cherabuddi K, Iovine NM, Shah K, et al. Zika and Chikungunya virus co-infection in a traveller returning from Colombia, 2016: virus isolation and genetic analysis. JMM Case Rep 2016;3:e005072–e72. 10.1099/jmmcr.0.005072 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zambrano H, Waggoner JJ, Almeida C, et al. Zika virus and Chikungunya virus coinfections: a series of three cases from a single center in Ecuador. Am J Trop Med Hyg 2016;95:894–6. 10.4269/ajtmh.16-0323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mercado-Reyes M, Acosta-Reyes J, Navarro-Lechuga E, et al. Dengue, Chikungunya and Zika virus coinfection: results of the National surveillance during the Zika epidemic in Colombia. Epidemiol Infect 2019;147:e77. 10.1017/S095026881800359X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mehta R, Soares CN, Medialdea-Carrera R, et al. The spectrum of neurological disease associated with Zika and Chikungunya viruses in adults in Rio de Janeiro, Brazil: a case series. PLoS Negl Trop Dis 2018;12:e0006212. 10.1371/journal.pntd.0006212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Alva-Urcia C, Aguilar-Luis MA, Palomares-Reyes C, et al. Emerging and reemerging arboviruses: a new threat in eastern Peru. PLoS One 2017;12:e0187897–e97. 10.1371/journal.pone.0187897 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Li J, Chong CY, Tan NW, et al. Characteristics of Zika virus disease in children: clinical, hematological, and virological findings from an outbreak in Singapore. Clin Infect Dis 2017;64:1445–8. 10.1093/cid/cix137 [DOI] [PubMed] [Google Scholar]
- 32.Dupont-Rouzeyrol M, O'Connor O, Calvez E, et al. Co-Infection with Zika and dengue viruses in 2 patients, new Caledonia, 2014. Emerg Infect Dis 2015;21:381–2. 10.3201/eid2102.141553 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Iovine NM, Lednicky J, Cherabuddi K, et al. Coinfection with Zika and dengue-2 viruses in a traveler returning from Haiti, 2016: clinical presentation and genetic analysis. Clin Infect Dis 2017;64:72–5. 10.1093/cid/ciw667 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Villamil-Gómez WE, Sánchez-Herrera Álvaro Rosendo, Hernández-Prado H, et al. Zika virus and HIV co-infection in five patients from two areas of Colombia. J Formos Med Assoc 2018;117:856–8. 10.1016/j.jfma.2018.05.019 [DOI] [PubMed] [Google Scholar]
- 35.Villamil-Gómez WE, Rodríguez-Morales AJ, Uribe-García AM, et al. Zika, dengue, and Chikungunya co-infection in a pregnant woman from Colombia. Int J Infect Dis 2016;51:135–8. 10.1016/j.ijid.2016.07.017 [DOI] [PubMed] [Google Scholar]
- 36.Gunturiz ML, Cortés L, Cuevas EL, et al. Congenital cerebral toxoplasmosis, Zika and Chikungunya virus infections: a case report. Biomedica 2018;38:144–52. 10.7705/biomedica.v38i0.3652 [DOI] [PubMed] [Google Scholar]
- 37.Valdespino-Vázquez MY, Sevilla-Reyes EE, Lira R, et al. Congenital Zika syndrome and Extra-Central nervous system detection of Zika virus in a pre-term newborn in Mexico. Clin Infect Dis 2019;68:903–12. 10.1093/cid/ciy616 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Araújo PSRde, Silva Júnior MLdeM, Tenório M, et al. Co-Infection ZIKV and HSV-1 associated with meningoencephalitis: case report and literature review. J Infect Public Health 2019;12:97–100. 10.1016/j.jiph.2018.04.010 [DOI] [PubMed] [Google Scholar]
- 39.Biron A, Cazorla C, Amar J, et al. Zika virus infection as an unexpected finding in a leptospirosis patient. JMM Case Rep 2016;3:e005033–e33. 10.1099/jmmcr.0.005033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Neaterour P, Rivera A, Galloway RL, et al. Fatal Leptospira spp./Zika Virus Coinfection-Puerto Rico, 2016. Am J Trop Med Hyg 2017;97:1085–7. 10.4269/ajtmh.17-0250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Alves LS, Estanislau C, Barreto L, et al. Concomitant testicular infection by Zika virus and Schistosoma mansoni in a Brazilian young boy. Rev Assoc Med Bras 2017;63:500–3. 10.1590/1806-9282.63.06.500 [DOI] [PubMed] [Google Scholar]
- 42.de Souza Costa MC, Siqueira Maia LM, Costa de Souza V, et al. Arbovirus investigation in patients from Mato Grosso during Zika and Chikungunya virus introdution in Brazil, 2015-2016. Acta Trop 2019;190:395–402. 10.1016/j.actatropica.2018.12.019 [DOI] [PubMed] [Google Scholar]
- 43.Beltrán-Silva SL, Chacón-Hernández SS, Moreno-Palacios E, et al. Clinical and differential diagnosis: dengue, Chikungunya and Zika. Revista Médica del Hospital General de México 2018;81:146–53. 10.1016/j.hgmx.2016.09.011 [DOI] [Google Scholar]
- 44.Tanabe ELdeL, Tanabe ISB, Santos ECD, et al. Report of East-Central South African Chikungunya virus genotype during the 2016 outbreak in the Alagoas state, Brazil. Rev Inst Med Trop Sao Paulo 2018;60:e19. 10.1590/s1678-9946201860019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Furuya-Kanamori L, Liang S, Milinovich G, et al. Co-distribution and co-infection of Chikungunya and dengue viruses. BMC Infect Dis 2016;16:84. 10.1186/s12879-016-1417-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ximenes RAA, Miranda-Filho DB, Brickley EB, et al. Zika virus infection in pregnancy: establishing a case definition for clinical research on pregnant women with rash in an active transmission setting. Plos Ntd 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bidokhti MRM, Dutta D, Madduri LSV, et al. SIV/SHIV-Zika co-infection does not alter disease pathogenesis in adult non-pregnant rhesus macaque model. PLoS Negl Trop Dis 2018;12:e0006811. 10.1371/journal.pntd.0006811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Vinton CL, Magaziner SJ, Dowd KA, et al. Simian immunodeficiency virus infection of rhesus macaques results in delayed Zika virus clearance. mBio 2019;10:e02790–19. 10.1128/mBio.02790-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Carod-Artal FJ. Neurological complications of Zika virus infection. Expert Rev Anti Infect Ther 2018;16:399–410. 10.1080/14787210.2018.1466702 [DOI] [PubMed] [Google Scholar]
- 50.World Health Organization Chikungunya virus: Factsheet, 2017. Available: https://wwwwhoint/news-room/fact-sheets/detail/chikungunya
- 51.World Health Organization Dengue guidelines for diagnosis, treatment, prevention and control : new edition. Available: https://apps.who.int/iris/handle/10665/441882009 [PubMed]
- 52.National Institute of Health Available: https://clinicaltrials.gov/ct2/show/NCT03263195
- 53.Rodriguez-Barraquer I, Costa F, Nascimento EJM, et al. Impact of preexisting dengue immunity on Zika virus emergence in a dengue endemic region. Science 2019;363:607–10. 10.1126/science.aav6618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Centers for Disease Control and Prevention Diagnostic test for Zika Virus-Trioplex real time RT-PCR assay instructions. Centre for Disease Control and Prevention, 2016. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjgh-2020-002350supp001.pdf (224.7KB, pdf)