Abstract
Middle East respiratory syndrome (MERS) is a respiratory disease caused by a zoonotic coronavirus (MERS-CoV). Camel handlers, including slaughterhouse workers and herders, are at risk of acquiring MERS-CoV infections. However, there is limited evidence of infections among camel handlers in Africa. The purpose of this study was to determine the presence of antibodies to MERS-CoV in high-risk groups in Kenya. Sera collected from 93 camel handlers, 58 slaughterhouse workers and 35 camel herders, were screened for MERS-CoV antibodies using ELISA and PRNT. We found four seropositive slaughterhouse workers by PRNT. Risk factors amongst the slaughterhouse workers included being the slaughterman (the person who cuts the throat of the camel) and drinking camel blood. Further research is required to understand the epidemiology of MERS-CoV in Africa in relation to occupational risk, with a need for additional studies on the transmission of MERS-CoV from dromedary camels to humans, seroprevalence and associated risk factors.
Keywords: coronavirus, camels, Republic of Kenya, slaughterhouses
1. Introduction
Middle East respiratory syndrome (MERS) is caused by an emerging beta-coronavirus (MERS-CoV). It is a zoonotic respiratory disease that was first reported in the Kingdom of Saudi Arabia in 2012 [1]. Dromedary camels are the reservoir of MERS-CoV [2] and contact with camels and their products is considered to be a risk factor for human MERS-CoV infection [3]. Occupational exposure has been reported in camel handlers, including camel farm workers and camel slaughterhouse workers [4], in the Middle East. It is hypothesized that the disease is transmitted from camels to people and person-to-person via respiratory secretions [5].
Previous research in Kenya has demonstrated a high MERS-CoV seropositivity in camels [6]. However, so far MERS-CoV neutralizing antibodies have only been detected in two non-camel-livestock keepers in Kenya [7]. In a targeted surveillance, none of the camel herders exposed to seropositive camels had MERS-CoV neutralizing antibodies [8].
In Kenya, populations living in semi-arid to arid environments have adopted camel rearing for drought resilience. Livestock keepers have a close relationship with their animals and cultural, animal husbandry and consumption practices may expose them to zoonotic agents. Poor hygienic conditions at farms and slaughterhouses, a lack of adequate infrastructure, the slaughtering and consumption of sick animals and weak monitoring and surveillance systems may contribute to an increased risk of exposure to MERS-CoV. There are limited data on MERS-CoV human infections in relation to occupational risk in Africa, with a need for additional studies on the transmission of MERS-CoV from dromedary camels to humans, seroprevalence and associated risk factors. The aims of this study were to determine the presence of antibodies to MERS-CoV in people in contact with camels and identify the risk factors associated with seropositivity.
2. Materials and Methods
The study was conducted in Isiolo, Laikipia and Machakos counties, Kenya, from October to November 2016 (Supplementary Figure S1). Slaughterhouse workers and camel herders were recruited in Isiolo and Laikipia and slaughterhouse workers only in Machakos. These areas represent rural arid and semi-arid regions in Kenya where camels are kept and/or where camel slaughterhouses are located. Isiolo and Laikipia counties are in the arid and semi-arid northern region of Kenya and are inhabited by pastoral communities who keep camels as part of their livelihoods. Camels are transported to slaughterhouses in Isiolo, Laikipia and Machakos counties.
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved on August 17th 2016 by the Institutional Research Ethics Committee of the International Livestock Research Institute (IREC 2016-07), a committee approved by the Kenya National Commission for Science, Technology and Innovation. Permission to work in the slaughterhouses was granted by the Directorate of Veterinary Services for the Ministry of Agriculture, Livestock Development and Fisheries in Kenya. Written informed consent was sought from all the participants who agreed to take part in the study and anonymity and confidentiality were adhered to by using randomly assigned barcodes to label samples.
Data were collected from participants on their demographics and occupational and consumption practices using structured questionnaires from October to November 2016. Trained personnel collected clotted blood samples in 10 ml vacutainer tubes (Becton–Dickinson). The serum was separated by centrifugation at 1150× g for 20 minutes using a Beckman Coulter Avanti J-E centrifuge. The sera samples were stored in duplicate at −20 °C until testing.
All the sera were tested for the presence of MERS-CoV specific antibodies using the commercial Euroimmun Anti-MERS-CoV ELISA at the International Livestock Research Institute (ILRI) laboratory in Nairobi. The positive and negative controls were human sera supplied by the manufacturer. The tests were carried out as per the manufacturers’ instructions. The extinction value or optical density of each analyzed sample was divided by the extinction value of the calibrator (supplied by the manufacturer) to calculate an extinction ratio. Samples with an extinction ratio of 0.3 were considered positive as previously described [7].
The sera were tested for the presence of MERS-CoV spike specific antibodies using an in-house S1 ELISA at the Erasmus MC Viroscience Laboratory in Rotterdam according to the previously validated protocol [9].
Twenty-one samples, including all Euroimmun and in-house S1 ELISA positive samples and a random selection of negative samples, were tested for the presence of MERS-CoV neutralizing antibodies using PRNT as described earlier with some modifications [9]. The positive and negative controls were the same as those described previously [9]. The testing was performed at the Erasmus MC Viroscience Laboratory in Rotterdam. Heat-inactivated sera were serially diluted in an RPMI1640 medium supplemented with penicillin, streptomycin and 1% fetal bovine serum (starting 1:10), mixed 1:1 with MERS-CoV (EMC/2012; 400 PFU) and incubated at 37 °C for one hour. Following that, the mix was transferred to a monolayer of HuH-7 cells in 96-well plates and incubated at 37 °C for one hour. The mix was then removed and the cells were further incubated at 37 °C for eight hours. The cells were then fixed and stained using an anti-MERS-CoV N protein mouse monoclonal antibody (1:5000, Sino Biological) and a secondary peroxidase-labelled goat anti-mouse IgG1 (1:2000, SouthernBiotech). The signal was developed using a precipitate forming TMB substrate (True Blue, KPL). The numbers of infected cells per well were counted using the ImmunoSpot® Image analyzer (CTL Europe GmbH). The neutralization titer of each serum sample was determined as the reciprocal of the highest dilution resulting in a ≥50% reduction in the number of infected cells (PRNT50). A titer of ≥20 was considered to be positive.
The statistical analysis was performed in R (http://CRAN.R-project.org/). To identify the risk factors associated with MERS-CoV seropositivity, a univariable analysis was conducted using Pearson’s chi-square with a Monte Carlo simulation of 10,000 repetitions to account for the small sample size. The results of the PRNT were used as a final determination of sero-reactivity. The statistical significance was set at p = 0.05 and confidence intervals were determined using a standard error of 1.96.
3. Results
A total of 58 slaughterhouse workers were sampled from three slaughterhouses; 5 were from Machakos, 16 from Laikipia and 37 from Isiolo (Supplementary Table S2). The majority of workers were male (n = 47) and the mean age was 37 years (range 17–73 years). In Machakos, 8–10 camels were slaughtered daily, in Laikipia 5 camels were slaughtered one day per week and in Isiolo 10–20 camels were slaughtered two days per week.
Thirty-five camel herders were sampled—33 in Isiolo and 2 in Laikipia. The majority of herders were male (n = 30) and the mean age was 45 years (range 2–82 years). The mean number of camels owned was 36 (range 0–149). The ratio of juvenile camels (less than 12 months) to adults in herds was 1:1.4.
The results of the three serological tests are presented in Table 1. Samples from five slaughterhouse workers (8.6%; 95% CI 3.8%–19.0%) and three camel herders (8.6%; 95% CI 8.6%–22.1%) were seropositive for MERS-CoV when tested using the Euroimmun ELISA. The samples were retested with the S1 ELISA and four slaughterhouse workers (6.9%; 95% CI 2.8%–16.3%) demonstrated antibodies to MERS-CoV compared to one camel herder (2.9%; 95% CI 0.7%–14.2%).
Table 1.
The proportion of slaughterhouse workers and camel herders who were positive for MERS-CoV antibodies when tested by the Euroimmun ELISA, S1 ELISA and PRNT.
| Cohort | Number of Samples | Euroimmun S1 ELISA Positive Number (%) | In-house S1 ELISA Positive Number (%) | PRNT50 Positive Number |
|
|---|---|---|---|---|---|
| Commercial S1 ELISA positive | In-House S1 ELISA Positive | ||||
| Slaughterhouse workers | 58 | 5 (8.6) | 4 (6.8) | 2/5 | 4/4 |
| Camel herders | 35 | 3 (8.6) | 1 (2.9) | 0/3 | 0/1 |
| Total | 93 | 8 (8.6) | 5 (5.4) | 2/8 | 4/5 |
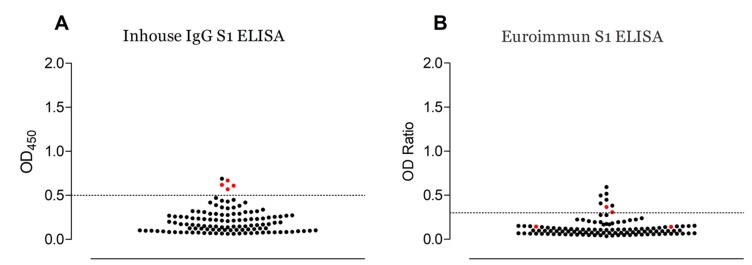
Four of the twenty-one samples tested using PRNT were positive. The agreement between the tests is shown in Supplementary Table S1 and Figure 1. All the PRNT positive samples originated from slaughterhouse workers in Isiolo, where the apparent prevalence was 10.8% (95% CI 4.4%–24.6%).
Figure 1.
The testing of human serum samples for MERS-CoV antibodies, Kenya, (2016). The reactivities of individual serum samples to MERS-CoV S1—tested using in-house (A) and commercial (B) S1 ELISAs—are plotted. PRNT positive serum samples are shown in red. The dotted line indicates the cut-off of each assay [7,9].
The proportion of PRNT positive samples at the Isiolo slaughterhouse is demonstrated in Supplementary Table S3. A risk factor analysis was conducted on samples from the Isiolo slaughterhouse only. Only PRNT positive samples were considered in the analysis. The proportion of positive male slaughterhouse workers (3/26) was not significantly different to the proportion of positive female workers (1/11) (Chi2 = 0.04, p = 1). There was a higher but not significant proportion of positive workers aged 40 years and over (2/12) compared to those less than 40 years (2/25) (Chi2 = 0.49, p = 0.60). The three positive men were slaughtermen, meaning they were responsible for the slaughtering event, and the positive female worker had another role in the slaughterhouse. The proportion of positive slaughtermen (3/8) was significantly different to that of other roles in the slaughterhouse (1/29) (Chi2 = 5.2, p = 0.05). Drinking camel blood was also significantly associated with seropositivity (3/6) compared to those who did not drink camel blood (1/31) (Chi2 = 7.3, p = 0.03). Other non-significant exposures included drinking camel milk (3/22) compared to not consuming the milk (1/15) (Chi2 = 0.37, p = 0.64) and milking camels (3/14) compared to not milking camels (1/23) (Chi2 = 2.05, p = 0.28).
4. Discussion
This is the first report of MERS-CoV neutralizing antibodies in camel handlers in Kenya. We detected an apparent prevalence of 10.8% MERS-CoV seropositivity by PRNT in slaughterhouse workers working in a camel-keeping area of the country. The detection of MERS-CoV in dromedary camels in Kenya in recent years [10] has highlighted the potential for transmission of the virus to people in close contact with camels as reported in the Middle East [4], but previous research investigating high-risk groups failed to detect individuals who were seropositive by virus neutralization [8]. The clinical significance of detecting MERS-CoV seropositivity by PRNT in our population is unknown. This was a cross-sectional serosurvey of healthy workers; we highlight that these individuals did not show clinical signs of disease and we are unable to determine when the exposures took place. The low virus neutralizing antibody titers may suggest asymptomatic infections [4], and clinical infections, which most likely present as transient infections, may be misdiagnosed as other endemic diseases [11].
Studies investigating the potential for transmission of MERS-CoV from camels to high-risk groups in other regions have had variable findings. Despite virus detection in camels at slaughterhouses in Nigeria, neutralizing antibodies were not detected in slaughterhouse workers [12]. Similarly, neutralizing antibodies have not been detected in slaughterhouse workers in the Kingdom of Saudi Arabia [13]. In contrast, virus neutralizing antibodies were detected in slaughterhouse workers in Qatar, where active MERS-CoV shedding has been demonstrated in slaughter animals [4]. A substantial pool of susceptible animals is necessary to support virus transmission and result in a risk to people [2]. Susceptible animals brought together for slaughter from different regions may drive virus amplification and zoonotic transmission [14].
Our previous research in Kenya has demonstrated a high seroprevalence of MERS-CoV antibodies in camels [6]. However, research in Kenya and elsewhere has demonstrated that juvenile camels have a higher rate of viral RNA positivity than adult animals [10,15]. In Kenya, the primary purpose of camel keeping is for milk production and therefore only mature animals (greater than three years) are presented for slaughter. This may limit the potential for transmission of the virus to slaughterhouse workers and explain the small number of positive samples in this study. Further studies targeting camel handlers who work with younger animals are required.
The currently available commercial ELISA is the Euroimmun Anti-MERS-CoV. This study demonstrates that even when using a low cut-off as recommended [7], the test is less specific and sensitive in detecting MERS-CoV seropositives compared to our in-house S1 ELISA, as previously observed [9]. However, there was a good correlation between the results of the in-house S1 ELISA and the PRNT. A sample reactive in both the S1 ELISA and PRNT was considered to be positive. Having a sensitive assay is crucial to avoid errors in the estimation of prevalence in seroepidemiological studies.
In this study, the small sample size makes it difficult to draw conclusions about risk factors for MERS-CoV seropositivity. The sample size is limited because the population of camel slaughterhouse workers is small. The statistical analysis accounted for the small sample size, but the results should be interpreted with caution. Potential risk factors that might be investigated in future studies include being a slaughterman (the person who cuts the animal’s throat). This has been reported for other zoonotic viruses transmitted by the respiratory route, including Rift Valley fever (RVF) [16].
Other risk factors that should be investigated include drinking camel blood [3]. This has not been significantly associated with MERS-CoV seropositivity but has been reported for RVF, another zoonotic RNA virus [16]. The milking of camels has also been reported by other studies as a risk factor for MERS-CoV and this needs further evaluation [3]. Further targeted studies investigating these and other risk factors in larger populations over longer periods of time are required.
Acknowledgments
We thank ILRI colleague Fredrick Amanya and Robert Rono, for their involvement in sampling of human serum.
Supplementary Materials
The following are available online at https://www.mdpi.com/1999-4915/12/4/396/s1, Figure S1: Map of sites where camel handlers were sampled in Kenya in 2016. Counties are indicated in light green and the locations of the slaughterhouses in dark green; Table S1: Results of testing selected sera from slaughterhouse workers and camel herders for antibodies to MERS-CoV by Euroimmun ELISA, S1 ELISA and PRNT. Positive results are shaded; Table S2: Demographics and practices of camel slaughterhouse workers in three counties, Machakos, Laikipia and Isiolo, Kenya, in 2016; Table S3: Univariable risk factor analysis for MERS-CoV seropositivity in slaughterhouse workers at Isiolo Camel Slaughterhouse, Kenya in 2016.
Author Contributions
Conceptualization, A.N.K. and E.M.F.; Methodology, A.N.K., E.A.J.C., N.M.A.O., V.K. and B.L.H.; Formal Analysis, A.N.K., E.C. and N.M.A.O.; Investigation, A.N.K.; Data Curation, E.C.; Writing—Original Draft Preparation, A.N.K. and E.A.J.C.; Writing—Review and Editing, All authors.; Funding Acquisition, E.M.F., C.R. and B.L.H. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Biotechnology and Biological Sciences Research Council, the Department for International Development, the Economic and Social Research Council, the Medical Research Council, the Natural Environment Research Council and the Defense Science and Technology Laboratory, under the Zoonoses and Emerging Livestock Systems (ZELS) programme, grant reference BB/L019019/1. This work was part-funded by the Global Challenges Research Fund (GCRF) One Health Regional Network for the Horn of Africa (HORN) Project, from UK Research and Innovation (UKRI) and Biotechnology and Biological Sciences Research Council (BBSRC) (project number BB/P027954/1). It also received support from the CGIAR Research Program on Agriculture for Nutrition and Health (A4NH), led by the International Food Policy Research Institute (IFPRI). We acknowledge the CGIAR Fund Donors (http://www.cgiar.org/funders). This work was supported by the Zoonoses Anticipation and Preparedness Initiative (ZAPI project; IMI grant agreement no.115760), with the assistance and financial support of IMI and the European Commission, in-kind contributions from EFPIA partners.
Conflicts of Interest
We know of no conflict of interest associated with this publication, and there has been no significant financial support for this work that could influence its outcome.
References
- 1.Zaki A.M., van Boheemen S., Bestebroer T.M., Osterhaus A.D.M.E., Fouchier R.A.M. Isolation of a Novel Coronavirus from a Man with Pneumonia in Saudi Arabia. N. Engl. J. Med. 2012;367:1814–1820. doi: 10.1056/NEJMoa1211721. [DOI] [PubMed] [Google Scholar]
- 2.Dudas G., Carvalho L.M., Rambaut A., Bedford T. MERS-CoV spillover at the camel-human interface. Elife. 2018;7:1–23. doi: 10.7554/eLife.31257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Alraddadi B.M., Watson J.T., Almarashi A., Abedi G.R., Turkistani A., Sadran M., Housa A., Almazroa M.A., Alraihan N., Banjar A., et al. Risk factors for primary middle east respiratory syndrome coronavirus illness in humans, Saudi Arabia, 2014. Emerg. Infect. Dis. 2016;22:49–55. doi: 10.3201/eid2201.151340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Reusken C.B.E.M., Farag E.A.B.A., Haagmans B.L., Mohran K.A., Godeke G.J., Raj V.S., Alhajri F., Al-Marri S.A., Al-Romaihi H.E., Al-Thani M., et al. Occupational exposure to dromedaries and risk for MERS-CoV infection, Qatar, 2013–2014. Emerg. Infect. Dis. 2015;21:1422–1425. doi: 10.3201/eid2108.150481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Aly M., Elrobh M., Alzayer M., Aljuhani S., Balkhy H. Occurrence of the Middle East Respiratory Syndrome Coronavirus (MERS-CoV) across the Gulf Corporation Council countries: Four years update. PLoS ONE. 2017;12:1–11. doi: 10.1371/journal.pone.0183850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Deem S.L., Fèvre E.M., Kinnaird M., Browne A.S., Muloi D., Godeke G.J., Koopmans M., Reusken C.B. Serological evidence of MERS-CoV antibodies in dromedary camels (camelus dromedaries) in laikipia county, Kenya. PLoS ONE. 2015;10:11–15. doi: 10.1371/journal.pone.0140125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liljander A., Meyer B., Jores J., Müller M.A., Lattwein E., Njeru I., Bett B., Drosten C., Corman V.M. MERS-CoV Antibodies in Humans, Africa, 2013-2014. Emerg. Infect. Dis. 2016;22:1086–1089. doi: 10.3201/eid2206.160064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Munyua P., Corman V.M., Bitek A., Osoro E., Meyer B., Müller M.A., Lattwein E., Thumbi S.M., Murithi R., Widdowson M.A., et al. No serologic evidence of middle east respiratory syndrome coronavirus infection among camel farmers exposed to highly seropositive camel herds: A household linked study, Kenya, 2013. Am. J. Trop. Med. Hyg. 2017;96:1318–1324. doi: 10.4269/ajtmh.16-0880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Okba N.M.A., Raj V.S., Widjaja I., GeurtsvanKessel C.H., de Bruin E., Chandler F.D., Park W.B., Kim N.-J., Farag E.A.B.A., Al-Hajri M., et al. Sensitive and Specific Detection of Low-Level Antibody Responses in Mild Middle East Respiratory Syndrome Coronavirus Infections. Emerg. Infect. Dis. 2019;25:1868–1877. doi: 10.3201/eid2510.190051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kiambi S., Corman V.M., Sitawa R., Githinji J., Ngoci J., Ozomata A.S., Gardner E., von Dobschuetz S., Morzaria S., Kimutai J., et al. Detection of distinct MERS-Coronavirus strains in dromedary camels from Kenya, 2017. Emerg. Microbes Infect. 2018;7:195. doi: 10.1038/s41426-018-0193-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Roess A., Carruth L., Lahm S., Salman M. Camels, MERS-CoV, and other emerging infections in east Africa. Lancet Infect. Dis. 2016;16:14–15. doi: 10.1016/S1473-3099(15)00471-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.So Ray T.Y., Perera Ranawaka A.P.M., Oladipo Jamiu O., Chu Daniel K.W., Kuranga Sulyman A., Kin-Ho C., Lau Eric H.Y., Cheng Samuel M.S., Poon Leo L.M., Webby Richard J., et al. Lack of serological evidence of middle east respiratory syndrome coronavirus infection in virus exposed camel abattoir workers in Nigeria, 2016. Eurosurveillance. 2018;23:2–5. doi: 10.2807/1560-7917.ES.2018.23.32.1800175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Aburizaiza A.S., Mattes F.M., Azhar E.I., Hassan A.M., Memish Z.A., Muth D., Meyer B., Lattwein E., Müller M.A., Drosten C. Investigation of anti-middle east respiratory syndrome antibodies in blood donors and slaughterhouse workers in Jeddah and Makkah, Saudi Arabia, Fall 2012. J. Infect. Dis. 2014;209:243–246. doi: 10.1093/infdis/jit589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hemida M.G., Al-Naeem A., Perera R.A.P.M., Chin A.W.H., Poon L.L.M., Peiris M. Lack of Middle East Respiratory Syndrome Coronavirus Transmission from Infected Camels. Emerg. Infect. Dis. 2015;21:699–701. doi: 10.3201/eid2104.141949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dighe A., Jombart T., Kerkhove M.D.v., Ferguson N. A systematic review of MERS-CoV seroprevalence and RNA prevalence in dromedary camels: Implications for animal vaccination. Epidemics. 2019;29:100350. doi: 10.1016/j.epidem.2019.100350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nicholas D.E., Jacobsen K.H., Waters N.M. Risk factors associated with human Rift Valley fever infection: systematic review and meta-analysis. Trop. Med. Int. Health. 2014;19:1420–1429. doi: 10.1111/tmi.12385. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.