Abstract
In the present study, the essential oils obtained by hydrodistillation of the organs of Eryngium maritimum and E. alpinum were analyzed by GC-FID-MS. The dominant constituents assessed in the essential oil of E. maritimum were germacrene D (45.2%) in the fruits; hexadecanoic acid (18.5%), menthol (16.8%), and menthone (10.9%) in the roots; 2,3,4-trimethylbenzaldehyde (11.3%) and germacrene D (10.5%) in the leaves; 2,3,4-trimethylbenzaldehyde (11.3%) in the shoot culture. In the case of E. alpinum, the main components of the leaf oil were: β-elemenone (10.3%), germacrone (5.8%), two selinadienes (7.1% and 6.7%), and 1,8-cineole (5.3%), which were not found in the oil from shoot culture, whereas the shoot culture oil was dominated by hexadecanoic acid (15.5%), spathulenol (7.5%), (E)-β-farnesene (4.9%), germacra-4(15),5,10(14)-trien-1α-ol (4.7%), and falcarinol (4.3%). The essential oils obtained from E. maritimum fruits and leaves of the intact plants, measured by the broth microdilution method, were the most active against T. mentagophytes and S. aureus. Moreover, the essential oil of leaves had the moderate activity against C. albicans and E. coli. The results showed that the chemical compositions of the essential oils differed decidedly between the two studied species and between the organs. Furthermore, the essential oil of E. maritimum may play an important role as antimicrobial agent.
Keywords: Sea holly, Alpine eryngo, essential oil composition, germacrene D, organs from intact plant, shoot in vitro cultures, antimicrobial activity of the essential oil
1. Introduction
The genus Eryngium L., which belongs to the subfamily Saniculoideae of the Apiaceae family, is represented by 230–250 species widespread in Central Asia, America, and Central and Southeast Europe [1]. Among them, there are some species such as E. campestre, E. maritimum, E. foetidum, and E. creticum that have been used in the traditional medicines worldwide [2]. The pharmacological activities of Eryngium species depend mainly on the presence of triterpenoid saponins, flavonoids, phenolic acids, coumarin derivatives, acetylenes, and the essential oils [3,4].
E. maritimum L. (Sea holly) is a rare perennial under strict law protection in Poland and some European countries. It grows on coastal dunes of the Baltic Sea, the Mediterranean basin, and the Black Sea [5]. The phytochemical investigations of E. maritimum revealed the presence of secondary metabolites—polyhydroxylated oleanene triterpenoid saponins [6,7], phenolic acids, mostly rosmarinic and chlorogenic acids [8,9], flavonoids containing mainly kaempferol, astragalin, isoquercitrin, rutin, apigenin, and luteolin [8,9,10], coumarins [9], 20-hydroxyecdysone and polypodine B as the main phytoecdysteroids [11], betaines [12], and the essential oil [13,14,15,16,17,18,19]. Several studies concerning the essential oil composition of E. maritimum generally investigated aerial parts of the intact plant [13,14,15,16,17,18,19] and no other research dealt with biomass from in vitro cultures. Moreover, the Polish individuals have been examined on the basis of the essential oil characterization for the first time.
E. alpinum L. (Alpine eryngo) is also under strict law protection in the European countries. It is a perennial species that can be found across the European Alps, at the altitudes between 1500 and 2000 m a.s.l. [20]. The phytochemical investigations of E. alpinum revealed the presence of secondary metabolites—triterpenoid saponins [21], phenolic acids mainly caffeic acid, rosmarinic acid and its derivative R-(+)-3’-O-β-d-glucopyranosyl rosmarinic acid, chlorogenic, iso- and neochlorogenic acids, and 3,4-dihydroxyphenylacetic acid [21,22,23], flavonoids—kaempferol, quercetin, and isoquercetin [21,23,24], and the essential oil [25]. The analysis of the essential oil of the E. alpinum aboveground part performed by Dunkić et al. [25], showed the presence of oxygenated sesquiterpenes, sesquiterpene hydrocarbons, oxygenated monoterpenes, and monoterpene hydrocarbons. Falcarinol was also found by Hegenauer [26,27]. To our knowledge, basal leaves as well as biomass from in vitro shoot culture of this species have never been investigated for the presence and the composition of the essential oil.
The availability of several plant species is limited due to their protection status. Increasing pollution and adverse changes occurring in the natural environment result in the shrinkage of plant resources, and the collection of the raw materials from fields in such regions becomes questionable. The alternative solution to these restrictions may be production of plant biomass of the endangered species applying the biotechnological methods. Rapid production of homogeneous and renewable biomass capable of biosynthesis of substances with the pharmacological activity, and most importantly the possibility of strict control of this process, indicate the opportunity for wider application of the biotechnological methods in the pharmaceutical and cosmetic industry [28,29].
The aim of this work was to study the chemical composition of the essential oils obtained from different parts of the intact plants as well as in vitro shoot cultures of E. maritimum L. and E. alpinum L., and to screen the antimicrobial activity of the essential oils obtained from mature fruits and basal leaves of the E. maritimum intact plant against Gram-positive bacterium Staphylococcus aureus, Gram-negative bacterium Escherichia coli, yeast Candida albicans, and dermatophyte fungus Trichophyton mentagrophytes. Our study on the essential oil composition of E. maritimum has completed the existing knowledge on this subject and supplemented the raw materials not yet tested, and thus not characterized, especially those obtained by the biotechnological methods under controlled conditions. In addition, our study has provided the first preliminary results of the analysis of the E. alpinum essential oil from the raw materials obtained from the intact plant and biomass from in vitro culture. Thus, in vitro cultures of these endangered and protected species can be the alternative source of the raw material for phytochemical examination and the biological activity studies. In addition, this has been the first report on the content and the composition of the essential oils from Eryngium species growing in Poland—E. maritimum on Polish beaches and E. alpinum in a botanical garden.
2. Results and Discussion
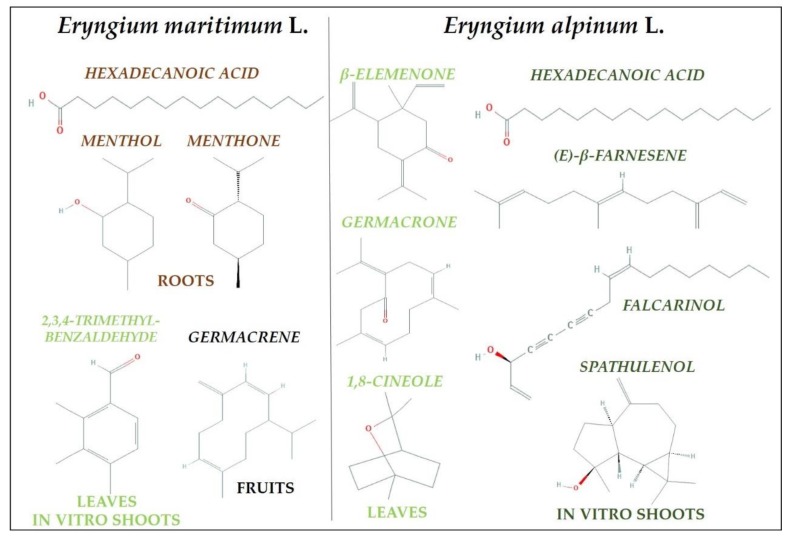
The main compounds of E. maritimum and E. alpinum essential oils are illustrated in Figure 1.
Figure 1.
The main compounds of the essential oil from different organs of the intact plants and in vitro shoot cultures of E. maritimum and E. alpinum.
Hydrodistillation of the dried parts (fruits, leaves, and roots) and in vitro-derived shoot culture of E. maritimum gave the essential oils in the yield of 0.30%, 0.06%, 0.01%, and 0.01%, respectively. These values have been in accordance with the earlier data that reported 0.31–0.93% for the fruit [18] and 0.06–0.13% for the aerial part [17] of the essential oil of this species.
The composition of the essential oils was analyzed by GC-FID-MS. The identified compounds of E. maritimum oils are presented in Table 1.
Table 1.
The chemical composition (%) of the essential oil from different organs of the intact plant and in vitro shoot culture of E. maritimum.
| Constituent | RIexp | RIlit | R | F | L | S |
|---|---|---|---|---|---|---|
| Hexanal | 775 | 777 | - | - | 0.1 | - |
| 2-Butylfuran | 870 | 869 | - | t | 0.1 | t |
| Heptanal | 879 | 882 | 0.3 | - | t | - |
| α-Pinene | 932 | 936 | 0.2 | t | 0.2 | 0.5 |
| Thuja-2,4(10)-diene | 946 | 946 | 0.1 | - | 0.1 | 0.6 |
| β-Pinene | 973 | 978 | 0.2 | - | t | t |
| Mesitylene | 977 | 980 | 0.6 | 0.1 | 0.9 | 3.5 |
| Octanal | 978 | 0981 | 1.1 | - | 0.1 | - |
| α-Phellandrene | 1000 | 1002 | 0.1 | - | - | - |
| 1,2,3-Trimethylbenzene | 1003 | 1005 | 0.2 | - | 0.4 | 1.0 |
| p-Cymene | 1013 | 1015 | t | t | t | 0.2 |
| 1,8-Cineole | 1021 | 1024 | 0.3 | t | t | t |
| Limonene | 1025 | 1025 | 0.4 | - | t | 0.3 |
| 1-Methoxy-2-(1-methylethenyl)benzene | 1076 | 1074 | - | - | 0.1 | 0.3 |
| 6-Camphenone | 1079 | 1082 | - | - | 0.2 | 0.5 |
| α-Campholenal | 1103 | 1105 | t | t | 0.1 | t |
| cis-Verbenol | 1128 | 1132 | - | t | 0.2 | 0.9 |
| Menthone | 1133 | 1136 | 10.9 | - | - | - |
| trans-Verbenol | 1133 | 1137 | - | 0.1 | 0.6 | 3.6 |
| p-Mentha-1,5-dien-8-ol | 1144 | 1142 | 0.2 | t | 0.3 | 0.9 |
| Isomenthone | 1145 | 1146 | 1.8 | - | - | - |
| Neomenthol | 1152 | 1156 | 0.8 | - | - | - |
| Terpinen-4-ol | 1161 | 1164 | - | - | - | 0.2 |
| Menthol | 1170 | 1172 | 16.8 | - | - | - |
| Myrtenal | 1169 | 1172 | 0.3 | - | t | 0.3 |
| α-Terpineol | 1176 | 1178 | - | - | - | 0.1 |
| Verbenone | 1180 | 1183 | - | - | - | 0.4 |
| Pulegone | 1214 | 1215 | 1.1 | - | - | - |
| Piperytone | 1228 | 1226 | 0.3 | - | - | - |
| cis-Chrysanthenyl acetate | 1248 | 1253 | 0.2 | 2.3 | 0.7 | 0.7 |
| Undecan-2-one | 1275 | 1273 | - | - | 0.1 | - |
| Menthyl acetate | 1278 | 1280 | 5.3 | - | - | - |
| Dihydroedulan II | 1280 | 1284 | 0.2 | t | 0.5 | 0.4 |
| 2,3,6-Trimethylbenzaldehyde | 1288 | 1287 | 1.5 | 0.2 | 2.0 | 1.8 |
| 2,3,4-Trimethylbenzaldehyde | 1330 | 1331 | 5.6 | 1.1 | 11.3 | 11.3 |
| Bicycloelemene | 1338 | 1338 | - | 0.9 | - | - |
| α-Cubebene | 1353 | 1355 | - | 0.2 | 0.1 | 0.1 |
| (E)-β-Damascenone | 1363 | 1363 | - | - | 0.1 | 0.2 |
| α-Ylangene | 1376 | 1376 | - | 0.1 | - | - |
| α-Copaene | 1380 | 1379 | t | 0.8 | 0.9 | 0.4 |
| β-Bourbonene | 1385 | 1387 | 0.3 | 0.2 | 1.1 | 0.3 |
| β-Elemene | 1387 | 1389 | 1.3 | 1.7 | 0.9 | 0.5 |
| β-Ylangene | 1418 | 1420 | - | 4.0 | 1.2 | t |
| β-Caryophyllene | 1421 | 1421 | 2.9 | 0.2 | 0.4 | 0.8 |
| γ-Elemene | 1432 | 1429 | 0.2 | 6.9 | t | 0.9 |
| β-Copaene | 1429 | 1431 | 0.2 | 2.4 | 1.2 | t |
| γ-Decanolide | 1432 | 1433 | 0.3 | - | - | - |
| Isogermacrene D | 1443 | 1445 | - | 0.8 | 0.2 | t |
| (E)-β-Famesene | 1447 | 1447 | 0.5 | t | t | 0.6 |
| Selina-4(15),6-diene | 1450 | 1450 | - | 0.9 | t | 0.2 |
| α-Humulene | 1451 | 1453 | 0.2 | 0.7 | 0.7 | t |
| β-Ionone | 1468 | 1468 | - | - | 0.3 | t |
| ar-Curcumene | 1470 | 1473 | 0.4 | - | - | - |
| γ-Muurolene | 1473 | 1475 | t | t | t | 0.3 |
| Germacrene D | 1476 | 1480 | 2.6 | 45.2 | 10.5 | 3.8 |
| Aristolochene | 1484 | 1486 | 0.2 | - | 0.4 | 2.2 |
| cis-β-Guaiene | 1488 | 1488 | 0.6 | - | - | - |
| Eremophila-1(10),7-diene | 1489 | 1488 | - | - | - | 5.7 |
| Valencene | 1493 | 1494 | - | - | 1.9 | - |
| Bicyclogermacrene | 1494 | 1494 | 0.3 | 1.5 | - | - |
| α-Muurolene | 1495 | 1496 | - | - | 0.3 | t |
| ε-Amorphene | 1498 | 1498 | - | 0.8 | - | - |
| (E,E)-α-Farnesene | 1501 | 1498 | 0.2 | - | t | 0.2 |
| Eremophila-1(10),8,11-triene | 1507 | 1504 | - | - | - | 1.2 |
| γ-Cadinene | 1509 | 1507 | 0.2 | 1.3 | 0.4 | t |
| Calamenene | 1510 | 1517 | 0.1 | 0.1 | t | 0.2 |
| δ-Cadinene | 1515 | 1520 | 0.4 | 2.3 | 1.0 | 2.0 |
| α-Calacorene | 1531 | 1527 | - | 0.3 | 0.3 | t |
| Selina-4(15),7(11)-diene | 1531 | 1534 | - | - | - | 2.0 |
| Elemol | 1537 | 1541 | - | 1.0 | - | - |
| Selina-3,7(11)-diene | 1539 | 1542 | - | - | - | 1.6 |
| Salviadienol | 1543 | 1545 | - | 0.6 | 0.7 | 0.5 |
| Germacrene B | 1552 | 1552 | - | 1.8 | - | - |
| (E)-Nerolidol | 1552 | 1553 | - | - | - | 1.9 |
| Mintoxide | 1561 | 1565 | - | 0.4 | 0.5 | 1.0 |
| Spathulenol | 1564 | 1572 | 0.6 | - | 4.9 | 1.6 |
| Germacrene D-4-ol | 1570 | 1576 | - | 4.2 | - | - |
| Caryophyllene oxide | 1570 | 1578 | 0.6 | - | 0.3 | t |
| β-Elemenone | 1586 | 1589 | - | - | - | 2.4 |
| Salvial-4(14)-en-1-one | 1586 | 1592 | 0.1 | 1.0 | 2.4 | 1.3 |
| Globulol | 1589 | 1592 | 0.6 | - | - | - |
| Torilenol | 1597 | 1599 | - | 0.9 | 2.1 | 2.1 |
| T-muurolol | 1629 | 1633 | 0.6 | 1.2 | - | - |
| α-Cadinol | 1642 | 1642 | 1.0 | 1.8 | - | - |
| Cadina-4,10(14)-dien-1α-ol | 1664 | 1662 | - | - | - | 0.5 |
| Eudesma-4(15),7-dien-1β-ol | 1673 | 1671 | - | 2.8 | - | - |
| 4βH-Cadin-9-en-15-al 4βH-Muurol-9-en-15-al |
1686 | 1684 | - | 2.3 | 11.5 | 3.0 |
| 1686 | 1684 | |||||
| Ferulyl angelate | 1680 | 1682 | 0.2 | - | - | 0.6 |
| Germacrone | 1680 | 1684 | - | - | - | 1.5 |
| Tetradecanoic acid | 1749 | 1748 | 0.4 | - | - | - |
| Nootkatone | 1781 | 1782 | - | - | 1.5 | 4.0 |
| Neophytadiene 2 | 1836 | 1830 | 0.5 | 0.7 | 5.2 | 1.7 |
| Hexadecanoic acid | 1967 | 1968 | 18.5 | 0.8 | 0.5 | 4.0 |
| Falcarinol | 2000 | 2000 | 1.3 | - | - | - |
| γ-Hexadecanolide | 2070 | 2081 | 0.4 | - | - | 0.4 |
| Phytol | 2101 | 2114 | - | - | 2.2 | 2.3 |
| Total identified | - | - | 84.2 | 94.6 | 71.7 | 79.5 |
| Monoterpene hydrocarbons (MH) | - | - | 1.2 | 0 | 0.7 | 2.6 |
| Oxygenated monoterpenes (MO) | - | - | 38.0 | 2.4 | 2.1 | 7.6 |
| Sesquiterpene hydrocarbons (SH) | - | - | 10.6 | 73.1 | 21.5 | 23.0 |
| Oxygenated sesquiterpenes (SO) | - | - | 3.5 | 16.2 | 23.9 | 19.8 |
| Others (O) | - | - | 30.9 | 2.9 | 23.5 | 26.5 |
| The essential oil yield | - | - | 0.01 | 0.30 | 0.06 | 0.01 |
RIexp—Experimental Retention Index, RIlit—Literature Retention Index, R—Roots of Intact Plant, F—Fruits of Intact Plant, L—Basal Leaves of Intact Plant, S—In Vitro Shoot Culture, t—Trace (percentage value less than 0.05%).
In total, more than fifty constituents were identified in each oil. The composition of the intact plant essential oils differed significantly both between plant parts and from the earlier data. However, some similarities were observed in both regards. The main constituents of the leaf and fruit oils were sesquiterpenes and this is in good agreement with the previous reports [14,15,16,17,18]. The content of the dominant compound of this group, germacrene D, amounted to 45.2% in the fruit oil and 10.5% in the leaf oil. Other important constituents were γ-elemene (6.9%) and β-ylangene (4.0%) in the fruit oil, as well as 2,3,4-trimethylbenzaldehyde (11.3%), spathulenol (4.9%), and neophytadiene (5.2%) in the leaf oil. In the leaf oil, the mixture of two sesquiterpene aldehydes 4βH-cadin-9-en-15-al and 4βH-muurol-9-en-15-al (11.5%) was tentatively identified on the basis of the retention index (RI) because mass spectra were not available. These compounds, together with the corresponding alcohols, were previously isolated from the E. maritimum oil and identified by NMR [16]. Darriet et al., [17] analyzed the essential oil of leaves, flowers, stems, and roots and compared the composition of the several oil samples of E. maritimum from Corsica and Sardinia. They observed small differences in the composition of the essential oils isolated from aerial parts of the plant and stated that the dominant components were germacrene D (32.1–42.5%), 4βH-cadin-9-en-15-al (18.4–27.6%), 4βH-cadin-9-en-15-ol (2.2–10.5%), and 4βH-muurol-9-en-15-al (4.3–9.3%). The main difference between 12 oil samples was the higher content of germacrene D in the Corsican samples (32.2–45.9%) than in the Sardinian ones (13.7–23.8%) [17]. Moreover, the high content of germacrene D (40%) was found in the essential oil of the Portuguese sample [13,14]. In the aerial part oil from the Italian species, the content of germacrene D was similar (10.4%) to the presented study (10.5%) [14]. The high amount of germacrene D (13.6–31.7%) was also noticed previously in the fruit essential oil of E. maritimum growing on the Tunisian shoreline. The major individual compounds, except germacrene D, were 15-hydroxy-α-muurolene (12.0–18.6%) and germacrene B (6.8–15.0%) [18].
The root essential oil differed significantly from the fruit and leaf oils. It contained mainly oxygenated monoterpenes menthol (16.8%), menthone (10.9%), and menthyl acetate (5.6%), as well as 2,3,4-trimethylbenzaldehyde (5.6%). According to the research of Darriet et al., [17], the root oil of this species also differed entirely from the essential oil of the aerial part. However, its composition was different from the presented oil. It contained 2,4,5- (39.8%) and 2,3,6-trimethylbenzaldehyde (29.0%), α-muurolene (23.5%), and germacrene D (2%). Various trimethylbenzaldehyde isomers (2,3,4-trimethylbenzaldehyde, 2,4,5-trimethylbenzaldehyde, 2,3,6-trimethylbenzaldehyde, and 2,4,6-trimethylbenzaldehyde) were reported in high concentrations in the essential oil from different parts of E. maritimum [16,17,18,19] and many other Eryngium species [30]. Unambiguous identification of these isomers is difficult because of the similarity of their mass spectra and different literature data of RIs. In this paper, identification was done on the basis of MassFinder 4.1.
The composition of the in vitro shoot essential oil was very similar to the leaf oil. The majority of the identified compounds were found in both oils. What is more, the content of the constituents was similar, with the exception of germacrene D, the content of which was lower in the shoot oil than in the leaf oil (3.8% versus 10.5%). The most important feature that differentiated these oils was the presence of the pronounced amounts (1.2–5.7%) of some sesquiterpenes in the shoot oil, which were found neither in the leaf oil nor in the other two oils, namely hydrocarbons with eremophilane and selinane skeleton, (E)-nerolidol, and two ketones β-elemenone and germacrone. The absence of hexanal, octanal, and undecan-2-one was noticed in the shoot oil in contrast to the in vitro shoot oil.
The essential oils from basal leaves of the intact plant and in vitro shoot culture of E. alpinum were obtained with the yield of 0.01%. The constituents of these oils are presented in Table 2.
Table 2.
The chemical composition (%) of the essential oil from basal leaves of the intact plant and in vitro shoot culture of E. alpinum.
| Constituent | RIexp | RIlit | L | S |
|---|---|---|---|---|
| Hexanal | 777 | 777 | - | 0.2 |
| Heptanal | 878 | 881 | - | 0.3 |
| α-Pinene | 932 | 936 | 1.4 | 0.1 |
| Camphene | 945 | 950 | 0.2 | - |
| β-Pinene | 975 | 978 | 0.2 | 0.1 |
| 2-Pentylfuran | 977 | 981 | 0.2 | 0.3 |
| Octanal | 978 | 981 | - | 3.0 |
| Myrcene | 984 | 987 | 0.4 | - |
| α-Phellandrene | 999 | 1002 | 0.2 | - |
| p-Cymene | 1012 | 1015 | 0.5 | - |
| 1,8-Cineole | 1019 | 1024 | 5.3 | - |
| Limonene | 1023 | 1025 | 1.1 | 0.2 |
| γ-Terpinene | 1048 | 1051 | 0.2 | - |
| Octanol | 1060 | 1063 | - | 0.1 |
| Nonan-2-one | 1072 | 1074 | - | 0.2 |
| Nonanal | 1079 | 1076 | - | 0.5 |
| Fenchol | 1097 | 1099 | 0.1 | - |
| cis-Verbenol | 1130 | 1132 | 0.1 | 0.2 |
| Menthone | 1133 | 1136 | 0.6 | - |
| Borneol | 1147 | 1150 | 0.5 | - |
| Neomenthol | 1157 | 1156 | 0.6 | - |
| Octanoic acid | 1168 | 1166 | 0.1 | 0.3 |
| Terpinen-4-ol | 1161 | 1164 | 0.9 | - |
| α-Terpineol | 1172 | 1176 | 1.1 | - |
| Citronellol | 1213 | 1213 | 0.5 | - |
| cis-Chrysanthenyl acetate | 1248 | 1253 | 0.6 | 2.1 |
| Bornyl acetate | 1267 | 1270 | 0.4 | 0.2 |
| Menthyl acetate | 1277 | 1280 | 0.2 | - |
| Dihydroedulan II | 1282 | 1280 | 0.2 | 0.7 |
| 2,3,4-Trimethylbenzaldehyde | 1327 | 1331 | 2.5 | 1.0 |
| Terpinyl acetate | 1331 | 1335 | 1.7 | 0.2 |
| Citronellyl acetate | 1334 | 1337 | 0.2 | - |
| β-Damascenone | 1361 | 1363 | t | 0.1 |
| α-Copaene | 1376 | 1379 | 0.7 | 0.6 |
| β-Bourbonene | 1385 | 1387 | 0.1 | 0.3 |
| β-Elemene | 1386 | 1389 | 0.5 | 1.7 |
| cis-α-Bergamotene | 1409 | 1411 | - | 0.5 |
| β-Caryophyllene | 1419 | 1421 | 1.4 | 1.7 |
| γ-Elemene | 1427 | 1429 | 1.4 | 1.6 |
| trans-α-Bergamotene | 1432 | 1434 | t | 0.6 |
| Erythrodiene | 1444 | 1446 | 0.3 | 2.1 |
| (E)-β-Farnesene | 1447 | 1446 | 0.7 | 4.9 |
| trans-β-Ionone | 1464 | 1468 | 0.5 | 0.4 |
| ar-Curcumene | 1470 | 1473 | - | 2.2 |
| γ-Muurolene | 1472 | 1475 | 0.3 | 0.2 |
| Germacrene D | 1475 | 1479 | 1.7 | 2.5 |
| (Z,E)-α-Farnesene | 1481 | 1480 | 0.8 | 1.2 |
| α-Muurolene | 1494 | 1496 | 0.5 | 1.9 |
| (E,E)-α-Farnesene | 1498 | 1498 | - | 1.8 |
| γ-Cadinene | 1505 | 1507 | 0.3 | 0.9 |
| Calamenene | 1509 | 1517 | 0.7 | - |
| δ-Cadinene | 1514 | 1520 | 3.8 | 1.8 |
| Selina-4(15),7(11)-diene | 1530 | 1534 | 7.1 | - |
| Selina-3,7(11)-diene | 1538 | 1542 | 6.7 | - |
| Salviadienol | 1541 | 1545 | t | 1.5 |
| 1.5(E)-Nerolidol | 1550 | 1553 | 2.5 | - |
| Spathulenol | 1568 | 1572 | 1.7 | 7.5 |
| Caryophyllene oxide | 1574 | 1578 | 1.9 | 2.2 |
| Elemenone | 1583 | 1589 | 10.3 | - |
| Salvial-4(14)-en-1-one | 1588 | 1592 | 1.1 | 1.3 |
| Torilenol | 1596 | 1597 | 0.8 | 2.0 |
| 1,10-di-epi-Cubenol | 1615 | 1615 | 1.5 | - |
| β-Bisabolol | 1656 | 1659 | - | 1.5 |
| Germacra-4(15), 5,10(14)-trien-1α-ol | 1669 | 1680 | 1.7 | 4.7 |
| Germacrone | 1675 | 1684 | 5.8 | - |
| Tetradecanoic acid | 1749 | 1748 | 1.3 | - |
| Hexahydrofarnesyl acetone | 1829 | 1817 | 2.6 | 3.9 |
| Neophytadiene 2 | 1835 | 1836 | 1.8 | 1.4 |
| Hexadecanoic acid | 1951 | 1968 | 4.3 | 15.5 |
| Falcarinol | 1998 | 2000 | 3.1 | 4.3 |
| Phytol | 2099 | 2114 | 2.0 | 1.9 |
| Pentacosane | 2499 | 2500 | 0.3 | 1.0 |
| Hexacosane | 2599 | 2600 | 0.1 | 0.3 |
| Heptacosane | 2696 | 2700 | 0.4 | 0.3 |
| Nonacosane | 2890 | 2900 | 0.9 | 0.3 |
| Total Identified | 91.6 | 86.3 | ||
| Monoterpene hydrocarbons (MH) | - | - | 4.2 | 0.4 |
| Oxygenated monoterpenes (MO) | - | - | 13.0 | 3.5 |
| Sesquiterpene hydrocarbons (SH) | - | - | 27.0 | 26.5 |
| Oxygenated sesquiterpenes (SO) | - | - | 27.3 | 20.7 |
| Others (O) | - | - | 20.1 | 35.2 |
| The essential oil yield | - | - | 0.01 | 0.01 |
RIexp—Experimental Retention Index, RIlit—Literature Retention Index, L—Basal Leaves of Intact Plant, S—In Vitro Shoot Culture, t—Trace (percentage value less than 0.05%).
The dominant constituents of the E. alpinum essential oils were sesquiterpenes, both hydrocarbons and oxygenated derivatives. Although leaf and in vitro shoot oils have numerous common components, they differed significantly. The main components of the leaf oil were β-elemenone (10.3%), germacrone (5.8%), two selinadienes (7.1% and 6.7%), and 1,8-cineole (5.3%) that were not found in the in vitro shoot oil. On the other hand, the in vitro shoot oil was dominated by hexadecanoic acid (15.5%), spathulenol (7.5%), germacra-4(15), 5,10(14)-trien-1α-ol (4.7%), (E)-β-farnesene (4.9%), and falcarinol (4.3%). Each of these components was present in the leaf oil, however, in a smaller amount. In respect to the presence of the four mentioned sesquiterpenes (ketones and selinadienes), (E)-nerolidol, and the low content of germacrene D (1.7%), the leaf oil of E. alpinum resembled the in vitro shoot oil of E. maritimum (Table 1). The composition of the leaf essential oil is quite different than that previously reported for the aerial part oil, which contained caryophyllene oxide (21.6%), bicyclogermacrene (11.8%), germacrene D (10.3%), and α-bisabolol (7.8%) [25] as the major constituents.
The study on the essential oil composition is a part of a bigger project aiming at phytochemical screening of Eryngium species: protected E. maritimum and E. alpinum as well as rare E. planum and E. campestre. In comparison with our previous investigation on the essential oils from different parts (inflorescence, stalk leaves, rosette leaves, and roots) as well as shoot in vitro culture of E. planum, some differences were observed between the aerial part, root, and in vitro shoot oils [30]. This is the first time the essential oils of the intact plant and shoot in vitro culture of E. maritimum and E. alpinum have been comparatively analyzed. It should be pointed out that the composition of the essential oil produced by in vitro shoot culture of E. maritimum (Table 1) was similar to the composition of the basal leaf oil. On the contrary, E. alpinum (Table 2) and E. planum [30] shoot cultures produced different oils than any aerial part. The essential oil composition of E. campestre aerial parts at the flowering stage [31,32,33,34] and roots [34] was the object of many studies.
The activity of the essential oils obtained from E. maritimum basal leaves and fruits was evaluated by the broth microdilution method. Both oils were the most active against T. mentagrophytes (Minimal Inhibitory Concentration MIC = 1.56 ± 0.0 mg/mL and 7.5 ± 0.0 mg/mL, respectively) and S. aureus (MIC = 12.5 ± 0.0 mg/mL and 60 ± 0.0 mg/mL, respectively). The essential oil from basal leaves had also the moderate activity against C. albicans (MIC = 12.5 ± 0.0 mg/mL) and E. coli (MIC = 25 ± 0.0 mg/mL) (Table 3).
Table 3.
The activity of the essential oils from E. maritimum leaves and fruits against standard microbial strains.
| Microorganism | Leaf Oil | Fruit Oil | Amikacin | Nystatin | ||||
|---|---|---|---|---|---|---|---|---|
| MIC ± SD [mg/mL] |
MBC ± SD [mg/mL] |
MIC ± SD [mg/mL] |
MBC ± SD [mg/mL] |
MIC ± SD [µg/mL] |
MBC ± SD [µg/mL] |
MIC ± SD [µg/mL] |
MFC ± SD [µg/mL] |
|
|
S. aureus ATCC 25923 |
12.5 ± 0.0 | >25 | 60 ± 0.0 | >120 | 2.0 ± 0.0 | 2.0 ± 0.0 | - | - |
|
E. coli ATCC 25922 |
25 ± 0.0 | >25 | >120 | >120 | 2.0 ± 0.0 | 2.0 ± 0.0 | - | - |
|
C. albicans ATCC 10231 |
12.5 ± 0.0 | 25 ± 0.0 | >120 | >120 | - | - | 8.0 ± 0.0 | 16.0 ± 0.0 |
|
T. mentagrophytes ATCC 9533 |
1.56 ± 0.0 | 3.13 ± 0.0 | 7.5 ± 0.0 | 11.25 ± 5.3 | - | - | 8.0 ± 0.0 | 64.0 ± 0.0 |
MIC—Minimal Inhibitory Concentration; MBC—Minimal Bactericidal Concentration; MFC—Minimum Fungicidal Concentration; SD—Standard Deviation.
The studies on the antimicrobial activity of the essential oil and its oxygenated sesquiterpene fractions from the aerial part of E. maritimum estimated by the agar diffusion method against several bacteria revealed the significant effect of 4βH-cadin-9-en-15-al, germacrene D, 4βH-cadin-9-en-15-ol (8.3%), and 4βH-muurol-9-en-15-al mixture against Escherichia coli, Enterococcus faecalis, and Listeria monocytogenes [15]. Moreover, the essential oil from the aerial part of this species growing in Portugal showed the moderate activity against Leishmania infantum promastigotes growth and no activity against Giardia lamblia [13,14].
The interest in the essential oils obtained from the Eryngium taxa has been increasing since some of the constituents and the essential oils showed the antimicrobial activities, for example E. creticum, E. campestre, E. thorifolium, and E. duriaei [4]. In the disc diffusion method, the essential oil of E. thorifolium, rich in α-pinene, showed the antibacterial activity against S. aureus with the inhibition zone ranging from 13 to 19 mm, which referred to sensitivity of the test microorganisms [32]. Moreover, the essential oil of E. duriaei revealed the antifungal activity against T. mentagrophytes, which was probably related to the caryophyllene derived compounds [35].
The antimicrobial activity of E. maritimum as well as E. planum and E. campestre has been already studied [36,37]. However, it was the extracts and their fractions that were examined, not the essential oils. The crude ethanolic extracts of both leaves and roots of investigated E. maritimum showed the significant antifungal activity against T. mentagrophytes and the moderate antibacterial activity against S. aureus [36]. This observation was in a great agreement with the study of Kholkhal [8], who showed the moderate antibacterial activity of the methanolic extract from the E. maritimum root against S. aureus. The methanolic extract of roots and saponin-phenolic acid fraction of this extract were the most active against C. albicans [37]. The results of Meot-Duros studies on the E. maritimum antimicrobial activity of the leaf hydromethanolic extracts, measured by microdilution method, showed that apolar fractions were more active than polar fractions. The antibacterial activity of apolar fraction against S. aureus showed MIC value of 10 µg/mL and the antifungal activity of apolar fraction against C. albicans showed MIC value of 100 µg/mL, while polar fractions had no antimicrobial activity. Moreover, both apolar and polar fractions were not sensitive to E. coli [38].
3. Materials and Methods
3.1. Intact Plants
The primary explants of E. maritimum and E. alpinum were collected from Adam Mickiewicz Botanical Garden in Poznań, Poland in September 2017. The voucher specimens have been deposited in the Department of Pharmaceutical Botany and Plant Biotechnology of Poznan University of Medical Sciences under the numbers: H-M-2017-101 (E. maritimum) and H-AP-2017-102 (E. alpinum). The permission for the collection of the organs from the protected species in order to use them for scientific research was granted by the Regional Director for Environmental Protection in Poznan.
3.2. In Vitro Shoot Culture
For the initiation of aseptic culture of E. maritimum and E. alpinum, the shoot fragments with axillary buds isolated from plantlets were used as explants. The isolated primary explants were rinsed in distilled water for 5 min and dipped in 70% (v/v) ethanol-water solution for 30 s followed by rinsing in 1.33% (E. maritimum) or 2.5% (E. alpinum) sodium hypochloride solution, containing two drops of surfactant Tween 80 for 8 min. They were finally rinsed three times in sterilized double-distilled water.
The explants of both the species were placed in a Erlenmeyer flask with 50 mL of the solidified MS medium (Murashige and Skoog [39]) with plant growth regulators—benzylaminopurine (BAP; Sigma-Aldrich, Saint Louis, MO, USA), indole-3-acetic acid (IAA; Sigma-Aldrich, Saint Louis, MO, USA), and gibberellic acid (GA3; Sigma-Aldrich, Saint Louis, MO, USA), each at the concentration of 1.0 mg/L [23,40]. The culture vessels were placed in a growth chamber (21 ± 2 °C; with a 16 h light/8 h dark photoperiod; 55 µmol/m2∙s light) and subcultured every 5 weeks. Multiplication of shoots via the axillary branching method on MS medium was repeated many times, using at least 10 explants per repetition. Shoots were air dried.
3.3. Isolation and the Analysis of the Essential Oil
In the present study, mature fruits, basal leaves, and roots of the intact plant, as well as biomass from in vitro shoot culture of E. maritimum, were chosen for the analysis of the essential oil. Furthermore, the preliminary investigation of the chemical composition of the essential oil from basal leaves and in vitro shoot culture was conducted for E. alpinum.
The essential oils were obtained by three-hour hydrodistillation of the dried plant material using the glass Clevenger-type apparatus, according to European Pharmacopoeia 5.0. [30].
The GC-FID-MS analyses were performed using the Trace GC Ultra apparatus (Thermo Electron Corporation, Waltham, MA, USA) with FID and MS DSQ II detectors and the FID-MS splitter (SGE). Operating conditions: apolar capillary column Rtx-1ms (Restek), 60 m × 0.25 mm i.d., film thickness 0.25 µm; temperature program, 50–300 °C at 4 °C/min; SSL injector temperature 280 °C; FID temperature 300 °C; split ratio 1:20; carrier gas helium at a regular pressure 200 kPa. Mass spectra were acquired over the mass range 30–400 Da, ionization voltage 70 eV; ion source temperature 200 °C.
Identification of components was based on the comparison of their MS spectra with those of laboratory-made MS library, commercial libraries (NIST 98.1, Wiley Registry of Mass Spectral Data, 8th Ed. and MassFinder 4.1, Hamburg, Germany) and with the literature data [41,42] along with the retention indices (Rtx-1, MassFinder 4.1) associated with a series of alkanes with linear interpolation (C8-C26). The quantitative analysis (expressed as percentages of each component) was carried out by peak area normalization measurements without correction factors.
3.4. The Antimicrobial Assay
The essential oils from basal leaves and fruits of the intact plant of E. maritimum were tested against selected bacteria and fungi strains.
3.5. Preparing of the Essential Oils
The essential oils were dissolved in DMSO and then diluted in MHB (bacteria) or SDB (fungi) to a concentration of 50 mg/mL—the leaf oil, and 240 mg/mL—the fruit oil. The final concentration of DMSO not excided 0.1%. The solutions of the essential oils (100 µL) were two-fold serially diluted with MHB (bacteria) and SDB (fungi) in 96 well microtiter plates to the concentrations ranging from 50 mg/mL to 0.10 mg/mL—the leaf oil, and from 240 mg/mL to 0.47 mg/mL—the fruit oil. Simultaneously, a test was carried out determining the antimicrobial activity of DMSO used for the preparation of the stock solution of the essential oils.
3.6. Microorganisms
The following microorganisms were tested: Gram-positive strain S. aureus ATCC 25923; Gram-negative strain E. coli ATCC 25922; yeast C. albicans ATCC 10231 and dermatophyte T. mentagrophytes ATCC 9533. All the strains were purchased from the American Type Culture Collection. Amikacin (Sigma-Aldrich, USA; A0368000) and nystatin (Sigma-Aldrich, USA; N4014) were used as reference substances for bacteria and fungi respectively (amikacin—breakpoint for Enterobacterales and S. aureus according to EUCAST: S ≤ 8, R > 8; nystatin—there is no breakpoint limit range for microdilution tests).
The bacteria were grown in the Brain Heart Infusion broth (BHI, Oxoid, UK) and C. albicans in Sabouraud dextrose broth (SDB; Oxoid, UK, company, city, state if USA, country) at 34 °C for 18 h. After incubation, 2 mL of each culture was harvested by centrifugation (3000 rpm for 15 min), re-suspended in 1.5 mL of 10 mM phosphate buffered saline (PBS, pH 7.0, Sigma-Aldrich, USA), and then diluted in a suitable medium (bacteria in Mueller-Hinton broth, MHB OXOID, UK; fungi in SDB).
Filamentous fungi were inoculated on the Sabouraud dextrose agar (SDA; OXOID, UK) and incubated at 34 °C for adequate formation of conidia. The culture of the T. mentagrophytes were covered with a sterile 0.9% NaCl solution supplemented with 0.1% Tween 80. Then the conidia were carefully rubbed with a cotton swab and transferred to a sterile flask. The suspension was homogenized, vortexed, and filtered. The number of conidia (macroconidia and microconidia) in the suspension was determined using haemocytometer chamber as well as the serial dilution agar plating method. Inoculum was examined for the presence of hyphae, and both macroconidia and microconidia were observed.
Bacterial and fungal suspensions were diluted in suitable broth (bacteria in Mueller–Hinton broth; MHB OXOID, UK; fungi in SDB) to obtain the final suspension containing about 1 × 106 CFU/mL (bacteria), 1 × 105 CFU/mL (C. albicans), and 5 × 105 CFU/mL (T. mentagrophytes).
3.7. Evaluation of Minimal Inhibitory Concentration and Minimal Bactericidal/Fungicidal Concentratioin
The minimal inhibitory concentration (MIC) of the essential oils was determined by employing the broth microdilution method in 96-well plates following EUCAST with some modifications for essential oils [43]. One hundred microliters of each dilution of the essential oil and sterile nutrient broth (for growth control) were distributed into the wells of the micro titer plates. Each well was inoculated with 100 µL of a microbial suspension. The final concentration of microorganisms was determined for all the strains by subculture of the growth control on TSA (bacteria) and SDA (fungi) plates.
All the microtiter plates were incubated at 34 ± 1 °C for 18 h (bacteria and C. albicans) and for 72 h (T. mentagrophytes). The MICs values were recorded as the lowest concentration of the essential oils that inhibited visible growth of the tested microorganisms. In order to determine the minimal bactericidal/fungicidal concentration (MBC/MFC), after recording the MIC endpoint, the concentrations equal to and higher than MIC were subcultured (10 µL) on Trypticase Soy Agar (TSA; OXOID, UK)—bacteria, and SDA—fungi. The MBC was defined as the lowest concentration at which no growth was observed. All the tests were performed in duplicate and the antimicrobial activity was expressed as mean values ± standard deviation (SD).
4. Conclusions
Our study showed that different parts of Eryngium species from intact plants and in vitro shoot cultures showed different yields and compositions of the essential oils. Comparing the volatile constituents of E. maritimum, E. alpinum, and other Eryngium species, it can be stated that there is a group of compounds that are common for the majority of the essential oils. Among them there are mono- and sesquiterpene hydrocarbons, and especially germacrene D, widespread as the components of the essential oils as well as compounds rarely found in the essential oils such as cis-chrysanthenyl acetate, trimethylbenzaledydes, and falcarinol. Our study on the essential oil composition of E. maritimum and E. alpinum has completed the existing knowledge on this subject. It was shown that in vitro shoot culture of E. maritimum produced the essential oil with the composition similar to the composition of the intact plant and E. alpinum basal leaves and shoot culture gave significantly different essential oils. Thus, in vitro cultures of these endangered and protected species can be the alternative source of the raw material for phytochemical examination and the biological activity studies. In addition, this has been the first report on the content and the composition of the essential oils from Eryngium species growing in Poland. The essential oils obtained from E. maritimum fruits and basal leaves of the intact plant were the most active against dermatophyte fungus T. mentagophytes and Gram-positive bacterium S. aureus. Moreover, the essential oil of basal leaves had also the moderate activity against C. albicans and Gram-negative E. coli.
To sum up, it is necessary to take into account in the above comparative analyses not only the ecotype and geographical localization of the plants but also the type of part of the plant analyzed, as well as the fact whether the oils come from the ground plant or in vitro cultures.
Author Contributions
Conceptualization, M.K.; methodology, M.K., D.K., J.D.; software, M.K.; formal analysis, M.K., D.K., J.D.; investigation, M.K., D.K., J.D.; resources, M.K., B.T., D.K., J.D.; writing—original draft preparation, M.K., D.K., B.T.; writing—review and editing, M.K., D.K., J.D.; visualization, M.K., D.K.; supervision, B.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Sample Availability: Samples of biomass are available from the authors.
References
- 1.Wörz A. On the distribution and relationships of the south-west Asian species of Eryngium L. (Apiaceae-Saniculoideae) Turk. J. Bot. 2004;28:85–92. [Google Scholar]
- 2.Kupeli E., Kartal M., Aslan S., Yesilada E. Comparative evaluation of the anti-inflammatory and antinociceptive activity of Turkish Eryngium species. J. Ethnopharmacol. 2006;107:32–37. doi: 10.1016/j.jep.2006.02.005. [DOI] [PubMed] [Google Scholar]
- 3.Wang P., Su Z., Yuan W., Deng G., Li S. Phytochemical constituents and pharmacological activities of Eryngium L. (Apiaceae) Pharm. Crops. 2012;3:99–120. doi: 10.2174/2210290601203010099. [DOI] [Google Scholar]
- 4.Erdem S.A., Nabavi S.F., Orhan I.E., Daglia M., Izadi M., Nabavi S.M. Blessings in disguise: A review of phytochemical composition and antimicrobial activity of plants belonging to the genus Eryngium. Daru J. Pharm. Sci. 2015;23:53. doi: 10.1186/s40199-015-0136-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kikowska M., Thiem B. In Vitro Systems of Selected Eryngium Species (E. planum, E. campestre, E. maritimum, and E. alpinum) for Studying Production of Desired Secondary Metabolites (Phenolic Acids, Flavonoids, Triterpenoid Saponins, and Essential Oil) In: Ramawat K., Ekiert H., Goyal S., editors. Plant Cell and Tissue Differentiation and Secondary Metabolites. Reference Series in Phytochemistry. Springer; Basel, Switzerland: 2020. pp. 1–33. [Google Scholar]
- 6.Hiller K., von Mach B., Franke P. Saponins of Eryngium maritimum L. Part 25. Contents of various Saniculoideae. Pharmazie. 1976;31:53. [PubMed] [Google Scholar]
- 7.Kowalczyk M., Masullo M., Thiem B., Piacente S., Stochmal A., Oleszek W. Three new triterpene saponins from roots of Eryngium planum. Nat. Prod. Res. 2014;28:653–660. doi: 10.1080/14786419.2014.895722. [DOI] [PubMed] [Google Scholar]
- 8.Kholkhal W., Ilias F., Bekhechi C., Bekkara F.A. Eryngium maritimum: A rich medicinal plant of polyphenols and flavonoids compounds with antioxidant, antibacterial and antifungal activities. Curr. Res. J. Biol. Sci. 2012;4:437–443. [Google Scholar]
- 9.Mejri H., Tir M., Feriani A., Ghazouani L., Allaqui M.S., Saidani-Tounsi M. Does Eryngium maritimum seeds extract protect against CCl4 and cisplatin induced toxicity in rats: Preliminary phytochemical screening and assessment of its in vitro and in vivo antioxidant activity and antifibrotic effect. J. Funct. Foods. 2017;37:363–372. doi: 10.1016/j.jff.2017.07.054. [DOI] [Google Scholar]
- 10.Hiller K., Pohl B., Franke P. Flavonoid spectrum of Eryngium maritimum L. Part 35. Components of some Saniculoideae. Pharmazie. 1981;36:451–452. [Google Scholar]
- 11.Dinan L., Savchenko T., Whiting P. On the distribution of phytoecdysteroids in plants. CMLS, Cell. Mol. Life Sci. 2001;58:1121–1132. doi: 10.1007/PL00000926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Adrian-Romero M., Wilson S.J., Blunden G., Yang M.-H., Carabot-Cuervo A., Bashir A.K. Betaines in coastal plants. Biochem. Syst. Ecol. 1998;26:535–543. doi: 10.1016/S0305-1978(98)00013-1. [DOI] [Google Scholar]
- 13.Machado M., Santoto G., Sousa M.C., Salgueiro L., Cavaleiro C. Activity of essential oils on the growth of Leishmania infantum promastigotes. Flavour Fragr. J. 2010;25:156–160. doi: 10.1002/ffj.1987. [DOI] [Google Scholar]
- 14.Machado M., Sousa M.C., Salgueiro L., Cavaleiro C. Effects of essential oils on the growth of Giardia lamblia trophozoites. Nat. Prod. Commun. 2010;5:137–141. doi: 10.1177/1934578X1000500132. [DOI] [PubMed] [Google Scholar]
- 15.Darriet F., Znini M., Majidi L., Muselli B., Hammouti B., Bouyanzer A., Costa J. Evaluation of Eryngium maritimum essential oil as environmentally friendly corrosion inhibitor for mild steel in hydrochloric acids solution. Int. J. Electrochem. Sci. 2013;8:4328–4345. [Google Scholar]
- 16.Darriet F., Bendahou M., Desjobert J.-M., Costa J., Muselli A. Bicyclo[4.4.0]decane oxygenated sesquiterpenes from Eryngium maritimum essential oil. Planta Medica. 2012;78:386–389. doi: 10.1055/s-0031-1298157. [DOI] [PubMed] [Google Scholar]
- 17.Darriet F., Andreani S., De Cian M.-C., Costa J., Muselli A. Chemical variability and antioxidant activity of Eryngium maritimum L. essential oils from Corsica and Sardinia. Flavour Fragr. J. 2014;29:3–13. doi: 10.1002/ffj.3160. [DOI] [Google Scholar]
- 18.Maggio A., Bruno M., Formisano C., Rigano D., Senatore F. Chemical composition of the essential oils of three species of Apiaceae growing wild in Sicily: Bonannia graeca, Eryngium maritimum and Opopanax chironium. Nat. Prod. Commun. 2013;8:841–844. doi: 10.1177/1934578X1300800640. [DOI] [Google Scholar]
- 19.Lajnef H.B., Ferioli F., Pasini F., Politowicz J., Khaldi A., D’Antuono F., Caboni M.F., Nasri N. Chemical composition and antioxidant activity of the volatile fraction extracted from air-dried fruits of Tunisian Eryngium maritimum L. ecotypes. J. Sci. Food Agric. 2017;98:635–643. doi: 10.1002/jsfa.8508. [DOI] [PubMed] [Google Scholar]
- 20.Gygax A., Bernhardt K.G., Jogan N., Montagnani C., Gigot G. Eryngium Alpinum. [(accessed on 2 March 2020)];2011 Available online: https://www.iucnredlist.org/species/162328/5574460.
- 21.Kikowska M., Kruszka D., Derda M., Thiem B. Phytochemical screening and acanthamoebic activity of shoots from in vitro cultures and in vivo plants of Eryngium alpinum L.—An endangered and protected species. Molecules. 2020;25:1416. doi: 10.3390/molecules25061416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Le Clarie E., Schwaiger S., Banaigs B., Stuppner H., Gafner F. Distribution of a new rosmarinic acid derivative in Eryngium alpinum L. and another Apiaceae. J. Agric. Food Chem. 2005;53:4367–4372. doi: 10.1021/jf050024v. [DOI] [PubMed] [Google Scholar]
- 23.Kikowska M., Thiem B., Szopa A., Klimek-Szczykułowicz M., Rewers M., Sliwinska E., Ekiert H. Comparative analysis of phenolic acids and flavonoids in shoot cultures of Eryngium alpinum L.—An endangered and protected species with medicinal value. Plant Cell Tiss. Organ Cult. 2019;139:167–175. doi: 10.1007/s11240-019-01674-8. [DOI] [Google Scholar]
- 24.Crowden R.K., Harborne J.B., Heywood V.H. (1969) Chemosystematics of the Umbelliferae—A general survey. Photochemistry. 1969;8:1963–1984. doi: 10.1016/S0031-9422(00)88084-X. [DOI] [Google Scholar]
- 25.Dunkić V., Vuko E., Bezic N., Kremer D., Ruscic M. Composition and antiviral activity of the essential oils of Eryngium alpinum and E. amethystinum. Chem. Biodivers. 2013;10:1894–1902. doi: 10.1002/cbdv.201300061. [DOI] [PubMed] [Google Scholar]
- 26.Hegnauer R. Chemotaxonomie der Pflanzen. Volume 6. Birkhäuser Verlag; Berlin, Germany: 1973. pp. 563–627. [Google Scholar]
- 27.Hegnauer R. Chemotaxonomie der Pflanzen. Volume 9. Birkhäuser Verlag; Berlin, Germany: 1990. pp. 678–679. [Google Scholar]
- 28.Verpoorte R., Contin A., Memelnik J. Biotechnology for the production of plant secondary metabolites. Phytochem. Rev. 2002;1:13–25. doi: 10.1023/A:1015871916833. [DOI] [Google Scholar]
- 29.Tasheva K., Kosturkova G. Role of biotechnology for protection of endangered medicinal plants. In: Petre M., editor. Environmental Biotechnology—New Approaches and Prospective Applications. IntechOpen Limited; London, UK: 2013. [Google Scholar]
- 30.Thiem B., Kikowska M., Kurowska A., Kalemba D. Essential oil composition of the different parts and in vitro shoot cultures of Eryngium planum L. Molecules. 2011;16:7115–7124. doi: 10.3390/molecules16087115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pala-Paul J., Usano-Alemany J., Soria A.C., Perez-Alonso M.J., Brophy J.J. Essential oil composition of Eryngium campestre L. growing in different soil types. A preliminary study. Nat. Prod. Commun. 2008;3:1121–1126. [Google Scholar]
- 32.Celik A., Aydinlik N., Arslan I. Phytochemical constituents and inhibitory activity towards methicillin-resistant Staphylococcus aureus strains of Eryngium species (Apiaceae) Chem. Biodivers. 2011;8:454–459. doi: 10.1002/cbdv.201000124. [DOI] [PubMed] [Google Scholar]
- 33.Cianfaglione K., Cianfaglione K., Blomme E.E., Quassinti L., Bramucci M., Lupidi G., Dall′Acqua S., Maggi F. Cytotoxic essential oils from Eryngium campestre and Eryngium amethystinum (Apiaceae) growing in central Italy. Chem. Biodivers. 2017;14:e1700096. doi: 10.1002/cbdv.201700096. [DOI] [PubMed] [Google Scholar]
- 34.Matejić J.S., Stojanović-Radić Z.Z., Ristić M.S., Veselinović J.B., Zlatković B.K., Marin P.D., Džamić A.M. Chemical characterization, in vitro biological activity of essential oils and extracts of three Eryngium L. species and molecular docking of selected major compounds. J. Food Sci. Technol. 2018;55:2910–2925. doi: 10.1007/s13197-018-3209-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Abou-Jawdah Y., Sobh H., Salameh A. Antimycotic activities of selected plant flora, growing wild in Lebanon, against phytopathogenic fungi. J. Agric. Food Chem. 2012;50:3208–3213. doi: 10.1021/jf0115490. [DOI] [PubMed] [Google Scholar]
- 36.Thiem B., Goślińska O., Kikowska M., Budzianowski J. Antimicrobial activity of three Eryngium L. species (Apiaceae) Herba Pol. 2010;56:52–59. [Google Scholar]
- 37.Kikowska M., Długaszewska J., Kubicka M.M., Kędziora I., Budzianowski J., Thiem B. In vitro antimicrobial activity of extracts and their fractions from three Eryngium L. species. Herba Pol. 2016;62:67–77. doi: 10.1515/hepo-2016-0012. [DOI] [Google Scholar]
- 38.Meot-Duros L., Le Floch G., Magne C. Radical scavenging, antioxidant and antimicrobial activities of halophytic species. J. Ethnopharmacol. 2008;116:258–262. doi: 10.1016/j.jep.2007.11.024. [DOI] [PubMed] [Google Scholar]
- 39.Murashige T., Skoog F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 1962;15:473–497. doi: 10.1111/j.1399-3054.1962.tb08052.x. [DOI] [Google Scholar]
- 40.Kikowska M., Thiem B., Szopa A., Ekiert H. Accumulation of valuable secondary metabolites: Phenolic acids and flavonoids in different in vitro systems of shoot cultures of the endangered plant species—Eryngium alpinum L. Plant Cell Tiss. Organ Cult. 2020:1–11. doi: 10.1007/s11240-020-01795-5. [DOI] [Google Scholar]
- 41.Adams R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy. 4th ed. Allured Publishing Corporation; Carol Stream, IL, USA: 2007. [Google Scholar]
- 42.Joulain D., König W.A. Journal of Natural Products. Volume 62. E.B.-Verlag; Hamburg, Germany: 1998. The Atlas of Spectral Data of Sesquiterpene Hydrocarbons; pp. 1212–1213. [Google Scholar]
- 43.Rodriques-Tudela J.L., Donnelly J.P., Arendrup M.C., Arikan-Akdagli S., Barchiesi F., Bille J., Chryssanthou E., Cuenca-Estrella M., Dannaoui E., Denning D., et al. EUCAST Technical Note on the method for the determination of broth dilution minimum inhibitory concentrations of antifungal agents for conidia–forming moulds. Clin. Microb. Infection. 2008;14:982–984. doi: 10.1111/j.1469-0691.2008.02086.x. [DOI] [PubMed] [Google Scholar]