Abstract
Aims
To investigate the characteristics and clinical significance of myocardial injury in patients with severe coronavirus disease 2019 (COVID-19).
Methods and results
We enrolled 671 eligible hospitalized patients with severe COVID-19 from 1 January to 23 February 2020, with a median age of 63 years. Clinical, laboratory, and treatment data were collected and compared between patients who died and survivors. Risk factors of death and myocardial injury were analysed using multivariable regression models. A total of 62 patients (9.2%) died, who more often had myocardial injury (75.8% vs. 9.7%; P < 0.001) than survivors. The area under the receiver operating characteristic curve of initial cardiac troponin I (cTnI) for predicting in-hospital mortality was 0.92 [95% confidence interval (CI), 0.87–0.96; sensitivity, 0.86; specificity, 0.86; P < 0.001]. The single cut-off point and high level of cTnI predicted risk of in-hospital death, hazard ratio (HR) was 4.56 (95% CI, 1.28–16.28; P = 0.019) and 1.25 (95% CI, 1.07–1.46; P = 0.004), respectively. In multivariable logistic regression, senior age, comorbidities (e.g. hypertension, coronary heart disease, chronic renal failure, and chronic obstructive pulmonary disease), and high level of C-reactive protein were predictors of myocardial injury.
Conclusion
The risk of in-hospital death among patients with severe COVID-19 can be predicted by markers of myocardial injury, and was significantly associated with senior age, inflammatory response, and cardiovascular comorbidities.
Keywords: Coronavirus disease 2019, Mortality, Myocardial injury, Risk
Introduction
Coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) remains pandemic with considerable morbidity and mortality. Similar to SARS in 2003, acute respiratory distress syndrome (ARDS) is usually considered to be an acute process confined to the lungs and results in a high mortality rate in COVID-19 patients. However, accumulating evidence points to myocardial injury as a COVID-19-related complication with an incidence ranging from 7.2% to 12%.1,2 Notably, an American College of Cardiology clinical bulletin has highlighted the cardiac implications of COVID-19. It suggests that patients with underlying cardiovascular disease face higher risks and recommends them to be triaged and treated with priority.3 However, according to limited information on cardiac complication of COVID-19, characteristics and clinical significance of myocardial injury for risk of death remain unclear. In this study, we determined the predictive value of myocardial indicators on in-hospital death, and investigated the features and potential causes of myocardial injury in cases of severe COVID-19.
Methods
Participants
All consecutive patients admitted to Renmin Hospital of Wuhan University with laboratory-confirmed COVID-19 were included in the present retrospective study. Renmin Hospital of Wuhan University, located in Wuhan, Hubei Province, was assigned responsibility for the treatment of patients with severe COVID-19 by the Wuhan government.
The patients with severe COVID-19 enrolled in this study were diagnosed according to the guidelines for diagnosis and treatment of COVID-19 (trial sixth edition) published by the Chinese National Health Commission on 18 February 2020. The cases in this study included severe and critically ill disease, which was defined by the presence of any of the following: respiratory rate >30/min; oxygen saturation ≤93%; PaO2/FiO2 ratio ≤ 300 mmHg; respiratory failure requiring mechanical ventilation; shock; or respiratory failure combined with other organ failure requiring ICU treatment. The cases under 18 years of age and missing cardiac biomarkers, including levels of cardiac troponin I (cTnI), were excluded. The clinical outcomes (i.e. discharges, mortality, and length of stay) of all included patients were monitored up to 23 February 2020, the final date of follow-up.
This study was approved by the National Health Commission of China and the institutional review board at Renmin Hospital of Wuhan University (Wuhan, China). Written informed consent was waived by the Ethics Commission of the designated hospital for emerging infectious diseases.
Data collection
The demographic characteristics (age and gender), clinical data (comorbidities, laboratory findings, treatments, complications, and outcomes), laboratory findings and results of cardiac examinations (cardiac biomarkers) for participants during hospitalization were extracted from electronic medical records by two investigators. All the data were independently reviewed and entered into a computer database by a trained team of physicians. Patients were grouped according to whether they died (death group) or survived (survivors group). The study outcome was rate of in-hospital death, and clinical outcomes were determined based on the information of our hospital’s real-time medical records system.
Myocardial injury was defined as blood levels of cardiac biomarkers cTnI increased above the 99th percentile upper reference limit.4 ARDS was defined according to the Berlin Definition.5 The date of disease onset was defined as the day when the symptom was noticed.
The detection principle of creatinine kinase-myocardial band (CK-MB), myoglobin (MYO), and cTnI is the double-antibody sandwich method of chemiluminescence immunotechnology, the detection instrument is Siemens ADVIA Centaur XP Immunoassay System, and the reagent is an original matching kit from Siemens. The normal reference range of CK-MB is 0–5 ng/mL; the normal reference range of MYO is 0–110 ug /L; the normal reference range of cTnI is 0–0.04 ng/mL, and the minimum detectable concentration (analytical sensitivity) is 0.006 ng/mL in our hospital. The detection method of N-terminal pro-B-type natriuretic peptide (NT-proBNP) is based on a double-antibody clip one-step enzyme immunoassay based on the heart detection principle. The detection instrument is a Dimension EXL with LM automatic biochemical analysis system. The reagent is a Siemens Dimension EXL with LM kit. The normal reference range is 0–900 pg/mL.
To confirm COVID-19, the Viral Nucleic Acid Kit (Health, Ningbo, China) was used to extract nucleic acids from clinical samples according to manufacturer’s instructions. A 2019-nCoV detection kit (Bioperfectus, Taizhou, China) was used to detect the ORF1ab gene (nCovORF1ab) and the N gene (nCoV-NP) according to the manufacturer’s instructions using real-time RT-PCR.6 An infection was considered as laboratory-confirmed if both the nCovORF1ab and nCoV-NP showed positive results.
Statistical analysis
Descriptive statistics were obtained for all study variables. Continuous data are expressed as median (interquartile range, IQR). Categorical data are expressed as proportions. All categorical variables were compared for the study outcome by using the Fisher’s exact test or Chi-square test, and continuous variables were compared using the Student t-test or the Mann–Whitney U test, if appropriate. A receiver operating characteristic (ROC) curve analysis was performed to establish a single cut-off point. Multivariable Cox regression models using all covariates available were performed to determine the risk factors for in-hospital mortality, with a time from admission to the end of follow-up. Logistics regression analysis was performed to determine the predictors of myocardial injury. The cases missing biomarker data were excluded listwise with statistics software. Data were analysed using SPSS 25.0 (IBM, Chicago, IL). Statistical charts were generated using Prism 5 (GraphPad), Minitab (Version 18), and Python. For all statistical analyses, P < 0.05 was considered significant.
Results
Patient characteristics and treatment
A total of 2253 cases with confirmed COVID-19 were screened initially, from 1 January to 23 February 2020, in a tertiary teaching hospital with two integrated hospital districts and a cabin hospital. Figure 1 depicts the flowchart for participant selection. Briefly, after excluding cases with mild COVID-19 (1095), duplicated cases (156), and cases without available core medical information (331), 671 cases (death, 62; survivors, 609) with severe COVID-19 were enrolled in final analysis. In these patients, the median age was 63 years (IQR, 50–72 years), 48% of patients were male, and the median time from symptom and admission to end of follow-up was 23 days and 17 days, respectively. The most common main comorbidity was hypertension (29.7%), followed by diabetes (14.5%), coronary heart disease (8.9%), chronic renal disease (4.2%), chronic obstructive pulmonary disease (3.4%), cancer (3.4%), chronic heart failure (3.3%), cerebrovascular disease (3.3%), and atrial fibrillation (1.0%). During hospitalization, 95.5% of patients were given oxygen treatment; however, use of extracorporeal membrane oxygenation and continuous renal replacement therapy was rare. The proportion of antiviral treatment use was 96.4% in included patients, and 59.5%, 56.5%, and 54.2% of patients, respectively, were treated with intravenous immunoglobulin, glucocorticoids, and antibiotics, Table 1.
Figure 1.
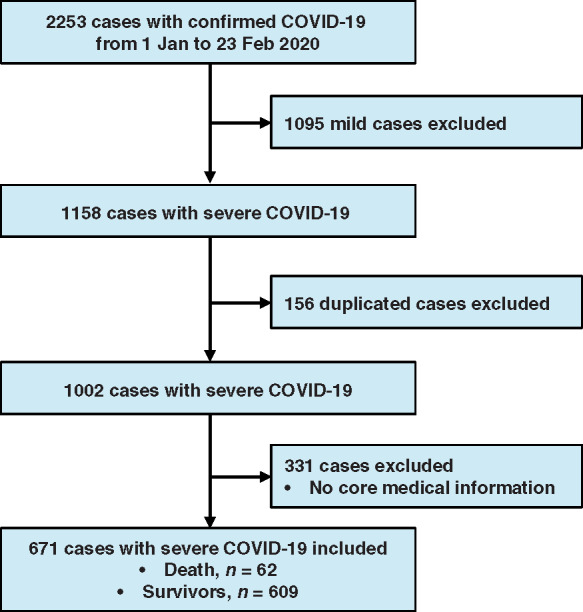
Flowchart of participant selection.
Table 1.
Baseline characteristics
| Characteristics | All patients | Death | Survivors | P-value |
|---|---|---|---|---|
| (n = 671) | (n = 62) | (n = 609) | ||
| Age (years), median (IQR) | 63 (50–72) | 74 (66–81) | 61 (49–70) | <0.001 |
| Gender (male), n (%) | 322 (48.0) | 35 (56.5) | 287 (47.1) | <0.001 |
| Time from symptom, days (IQR) | 23 (17–28) | 15 (10–18) | 24 (17–28) | <0.001 |
| Time from admission, days (IQR) | 17 (8–18) | 4 (3–7) | 17 (9–19) | <0.001 |
| Comorbidities, n (%) | ||||
| Hypertension | 199 (29.7) | 37 (59.7) | 162 (26.6) | <0.001 |
| Diabetes | 97 (14.5) | 17 (27.4) | 80 (13.1) | 0.004 |
| Coronary heart disease | 60 (8.9) | 21 (33.9) | 39 (6.4) | <0.001 |
| Chronic renal disease | 28 (4.2) | 12 (19.4) | 16 (2.6) | <0.001 |
| Chronic obstructive pulmonary disease | 23 (3.4) | 2 (3.2) | 21 (3.4) | 1.000 |
| Cancer | 23 (3.4) | 4 (6.5) | 19 (3.1) | 0.154 |
| Chronic heart failure | 22 (3.3) | 13 (21.0) | 9 (1.5) | <0.001 |
| Cerebrovascular disease | 22 (3.3) | 8 (12.9) | 14 (2.3) | <0.001 |
| Atrial fibrillation | 7 (1.0) | 2 (3.2) | 5 (0.8) | 0.130 |
| Treatment, n (%) | ||||
| Oxygen inhalation | 527 (78.5) | 16 (25.8) | 511 (83.9) | <0.001 |
| Non-invasive ventilation | 76 (11.3) | 17 (27.4) | 59 (9.7) | <0.001 |
| Invasive mechanical ventilation | 36 (5.4) | 29 (46.8) | 7 (1.1) | <0.001 |
| Extracorporeal membrane oxygenation | 2 (0.3) | 2 (3.2) | 0 (0.0) | 0.008 |
| Continuous renal replacement therapy | 4 (0.6) | 4 (6.5) | 0 (0.0) | <0.001 |
| Antiviral | 647 (96.4) | 58 (93.5) | 589 (96.7) | 0.267 |
| Immunoglobulin | 399 (59.5) | 55 (88.7) | 344 (56.5) | <0.001 |
| Glucocorticoids | 379 (56.5) | 53 (85.5) | 326 (53.5) | <0.001 |
| Antibiotic | 364 (54.2 ) | 49 (79.0) | 315 (51.7) | <0.001 |
IQR, interquartile range.
Comparison of clinical characteristics between death and survivors groups
The patients who died were older, more often male than the survivors (all P < 0.001, Table 1). Prevalence of hypertension, diabetes, coronary heart disease, chronic renal failure, chronic heart disease, and cerebrovascular disease was higher among the patients who died (all P < 0.01, Table 1); however, frequencies of other comorbidities including chronic obstructive pulmonary disease, cancer, and atrial fibrillation were similarly distributed between groups (all P > 0.05, Table 1). Compared with survivors, patients who died had more often received mechanical ventilation, extracorporeal membrane oxygenation, and continuous renal replacement therapy (all P < 0.001, Table 1). The proportion of antiviral treatment use was similarly distributed between groups; however, the usage of intravenous immunoglobulin, glucocorticoids, and antibiotics was higher among the patients who died (all P < 0.001, Table 1).
On admission, most patients who died presented abnormal laboratory results, such as higher leucocyte counts, neutrophil rate, and levels of aspartic transaminase, creatinine, C-reactive protein, and procalcitonin (PCT). Moreover, non-survivors had significant higher levels of all cardiac indices than survivors, including CK-MB, MYO, cTnI, and NT-proBNP (all P < 0.001, Table 2). However, lymphocyte rate, platelet counts, albumin level, and creatinine clearance were lower in non-survivors than those in survivors (all P < 0.001, Table 2).
Table 2.
Laboratory findings
| Characteristics | Normal range | All patients | Death | Survivors | P-value |
|---|---|---|---|---|---|
| (n = 671) | (n = 62) | (n = 609) | |||
| Leucocyte counts | 3.5–9.5 ×109/L | 5.8 (4.3–8.2) | 9.6 (7.8–14.0) | 5.2 (3.7–7.0) | <0.001 |
| Neutrophil rate | 40–75 % | 72 (62–84) | 91 (86–93) | 68 (59–76) | <0.001 |
| Lymphocyte rate | 20–50 % | 19 (10–26) | 6 (4–10) | 26 (16–28) | <0.001 |
| Platelet count | 125–350 ×109/L | 210 (155–270) | 153 (75–204) | 215 (172–284) | <0.001 |
| Erythrocyte count | 3.8–5.1 ×1012/L | 4.1 (3.6–4.5) | 4.0 (3.4–4.4) | 4.1 (3.8–4.5) | 0.050 |
| Haemoglobin | 115–150 g/L | 124 (111–135) | 128 (103–140) | 125 (118–135) | 0.545 |
| Alanine aminotransferase | 7–40 U/L | 27 (18–44) | 26 (19–39) | 22 (15–35) | 0.823 |
| Aspartic transaminase | 7–40 U/L | 29 (21–40) | 42 (29–64) | 25 (20–32) | <0.001 |
| Albumin | 40–55 g/L | 36 (33–38) | 33 (30–35) | 37 (35–39) | <0.001 |
| Creatinine | 41–73 μmol/L | 58 (48–70) | 87 (59–160) | 55 (48–63) | <0.001 |
| Creatinine clearance | > 90 mL/min | 97 (88–97) | 67 (35–94) | 99 (92–107) | <0.001 |
| C-reactive protein | < 10 mg/L | 41 (12–81) | 111 (64–191) | 30 (8–59) | <0.001 |
| Procalcitonin | < 0.1 ng/mL | 0.06 (0.04–0.13) | 0.46 (0.14–1.58) | 0.05 (0.03–0.09) | <0.001 |
| CK-MB | 0–5 ng/mL | 0.96 (0.63–1.82) | 3.6 (2.4–6.9) | 0.8 (0.6–1.2) | <0.001 |
| MYO | 0–110 μg/L | 42 (27–85) | 268 (92–768) | 32 (24–63) | <0.001 |
| cTnI* | 0–0.04 ng/mL | 0.006 (0.006–0.016) | 0.235 (0.042–1.996) | 0.006 (0.006–0.011) | <0.001 |
| NT-proBNP | 0–900 pg/mL | 189 (67–494) | 1819 (759–5164) | 132 (58–237) | <0.001 |
CK-MB, creatinine kinase-myocardial band; MYO, myoglobin; cTnI, cardiac troponin I; NT-proBNP, N-terminal pro-B-type natriuretic peptide.
indicated that the proportion of patients with cTnI levels >0.04 ng/mL in all included patients, dead patients, and survivors was 15.8%, 75.8%, and 9.7% on admission, respectively.
The rapid deterioration of the respiratory function was the main cause of death, followed by cardiovascular complications. Table 3 summarized the distribution of death-related complications in included patients, including ARDS (98.4%), acute respiratory failure (90.3%), acute myocardial injury (30.6%), acute heart failure (19.4%), multiple organ failure syndrome (9.7%), shock (6.5%), and sudden death (1.6%).
Table 3.
Cause of death in included patients
| Complications | No. (%) |
|---|---|
| Acute respiratory distress syndrome | 61 (98.4) |
| Acute respiratory failure | 56 (90.3) |
| Acute myocardial injury | 20 (30.6) |
| Acute heart failure | 12 (19.4) |
| Multiple organ failure syndrome | 6 (9.7) |
| Shock | 4 (6.5) |
| Sudden death | 1 (1.6) |
Predictive value of cardiac indicators for in-hospital mortality
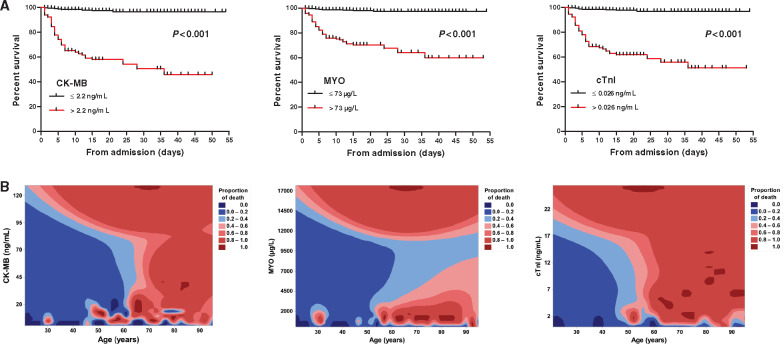
ROC curve analysis was performed to determine the value of CK-MB, MYO, and cTnI for predicting mortality during the time from admission to the end of follow-up in patients with severe COVID-19. As shown in Figure 2, the area under the ROC curve (AUC) for the outcome of mortality was 0.87 (95% CI, 0.81–0.93), 0.88 (95% CI, 0.83–0.93), and 0.92 (95% CI, 0.87–0.96), all with significant sensitivity and specificity (all P < 0.001). The single cut-off concentrations of CK-MB, MYO, and cTnI were 2.2 ng/mL, 73 μg/L, and 0.026 ng/mL, respectively.
Figure 2.
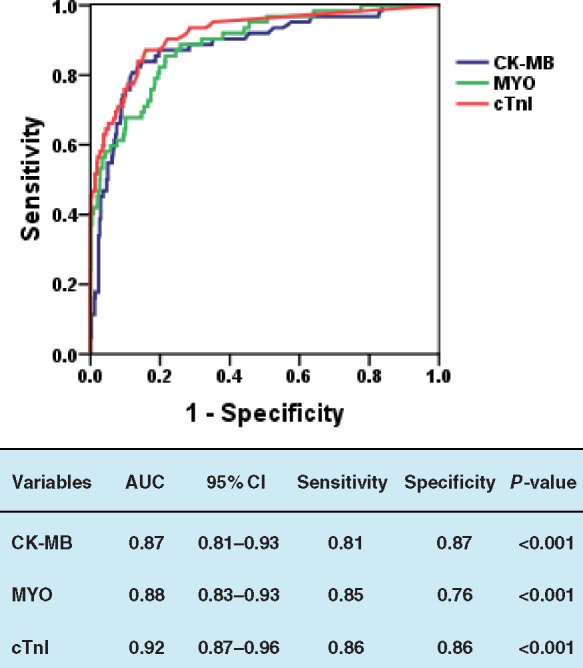
Receiver operating characteristic analysis of the clinical prediction model. Prediction of in-hospital mortality by levels of CK-MB, MYO, and cTnI; the area under the curve was 0.87, 0.88, and 0.92, respectively. All P < 0.001. AUC, area under the receiver operating characteristic curve; CI, confidence interval; CK-MB, creatinine kinase-myocardial band; MYO, myoglobin; cTnI, cardiac troponin I.
On Kaplan–Meier analysis, baseline CK-MB [49/117 (41.9%) vs. 13/554 (2.3%); P < 0.001], MYO [53/182 (29.1%) vs. 9/489 (1.8%); P < 0.001], and cTnI [51/133 (38.3%) vs. 11/538 (2.0%); P < 0.001] above these cut-offs were associated with markedly higher hospitalized death (Figure 3A). We performed a contour plot to determine the relationship between levels of cardiac biomarkers, age, and mortality. The results showed that the higher initial levels of CK-MB, MYO, and cTnI were consistently associated with higher mortality, which was pronounced in patients with senior age (Figure 3B).
Figure 3.
Kaplan–Meier plots and contour plot of survival probability in hospitalized patients with severe COVID-19. (A) Mortality was significantly higher in patients with CK-MB > 2.2 ng/mL, MYO > 73 μg/L, and cTnI > 0.026 ng/mL, all P <0.001 by log-rank test. (B) Higher initial levels of CK-MB, MYO, and cTnI were consistently associated with higher mortality, which was pronounced in patients with senior age. CK-MB, creatinine kinase-myocardial band; MYO, myoglobin; cTnI, cardiac troponin I.
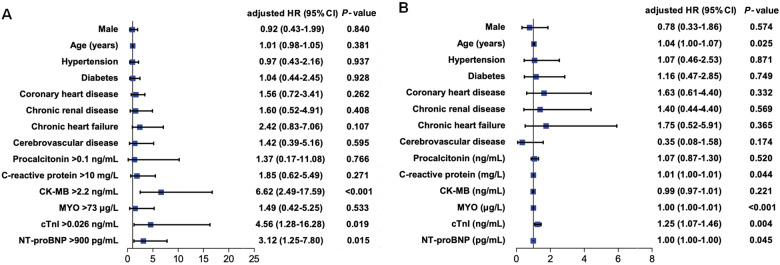
To analyse the risk factors for in-hospital mortality by multivariable Cox regression, the values of these biomarkers were transformed into categorical variables according to ROC cut-off points (CK-MB, MYO, and cTnI) and the 99th percentile upper reference limit (PCT, C-reactive protein, and NT-proBNP) in model 1 (Figure 4A), and all biomarkers remained as continuous variables in model 2 (Figure 4B), respectively. In multivariable Cox model 1, CK-MB > 2.2 ng/mL [hazard ratio (HR), 6.62; 95% confidence interval (CI), 2.49–17.59; P < 0.001], cTnI > 0.026 ng/mL (HR, 4.56; 95% CI, 1.28–16.28; P = 0.019), and NT-proBNP > 900 pg/mL (HR, 3.12; 95% CI, 1.25–7.80; P = 0.015) were risk factors, Figure 4A. In multivariable model 2, age (HR, 1.04; 95% CI, 1.00–1.07; P = 0.025), CRP (HR, 1.01; 95% CI, 1.00–1.01; P = 0.044), MYO (HR, 1.00; 95% CI, 1.00–1.01; P < 0.001), cTnI (HR, 1.25; 95% CI, 1.07–1.46; P = 0.004), and NT-proBNP (HR, 1.00; 95% CI, 1.00–1.00; P = 0.045) were risk factors for death, Figure 4B.
Figure 4.
Multivariable Cox regression analysis of clinical indicators of in-hospital mortality in patients with severe COVID-19. (A) In multivariable model 1, CK-MB > 2.2 ng/mL, cTnI > 0.026 ng/mL, and NT-proBNP > 900 pg/mL were risk factors; these cardiac indicators were transformed into categorical variables according to the cut-off points, and procalcitonin, C-reactive protein, and NT-proBNP were transformed into categorical variables according to the 99th percentile upper reference limit. (B) In multivariable model 2, senior age, C-reactive protein, MYO, cTnI, and NT-proBNP were risk factors; these biomarkers were performed as continuous variables. HR, hazard ratio; CI, confidence interval; CK-MB, creatinine kinase-myocardial band; MYO, myoglobin; cTnI, cardiac troponin I; NT-proBNP, N-terminal pro-B-type natriuretic peptide.
After these continuous variables were ln-transformed, the results of multivariable Cox regression model 2 showed PCT (HR, 1.72; 95% CI, 1.20–2.45) and cTnI (HR, 1.90; 95% CI, 1.44–2.49) were significantly associated with in-hospitalization mortality, but the other factors were no longer significant, including age, C-reactive protein, MYO, and NT-proBNP. In order to avoid the influence of collinearity among CK-MB, MYO, and cTnI, we excluded CK-MB and MYO in two multivariable Cox models, the results displayed that cTnI was still a significant factor in both the models, with 11.18 (95% CI, 3.23–38.75) and 1.27 (95% CI, 1.10–1.46) of HR in model 1 and model 2, respectively. Moreover, coronary heart disease (HR, 2.25; 95% CI, 1.04–4.87) became a new significant factor in model 1, and chronic heart disease (HR, 3.37; 95% CI, 1.24–9.18) was significant in model 2, respectively.
Incidence and predictors of myocardial injury
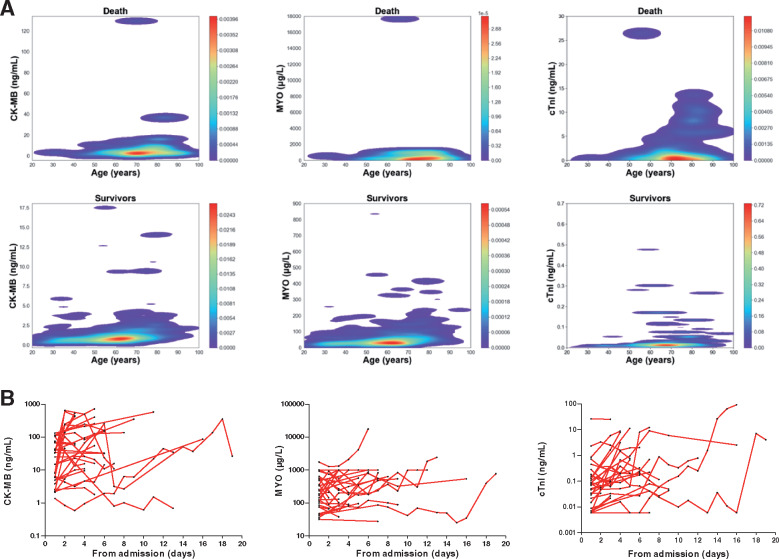
A total of 106 patients (15.8%) had myocardial injury in all included patients on admission. Patients with myocardial injury had older age, more comorbidities, and more abnormalities in laboratory findings than those patients without myocardial injury (Table 4). Patients who died had suffered more often from myocardial injury during hospitalization compared with survivors (75.8% vs. 9.7%; P < 0.001). A contour plot for the distribution characteristics of myocardial indicators showed that these biomarkers were higher among patients who died (Figure 5A). During the period from admission to death, cardiac indicators showed a dynamic change in the death group, especially in the levels of CK-MB and cTnI, Figure 5B. Because most patients were not continuously monitored during hospitalization, the serial biomarker results were based on a very small subcohort. In univariable logistic regression analysis, male, senior age, hypertension, diabetes, coronary heart disease, chronic renal failure, chronic heart failure, cerebrovascular disease, chronic obstructive pulmonary disease, and high levels of PCT and C-reactive protein were risk factors for myocardial injury (Table 5). In multivariable logistic regression analysis, male, diabetes, chronic heart failure, cerebrovascular disease, and PCT were no longer predictors (Table 5).
Table 4.
Baseline characteristics in patients with and without myocardial injury
| Characteristics | Myocardial injury | Without myocardial injury | P-value |
|---|---|---|---|
| (n = 106) | (n = 565) | ||
| Age (years), median (IQR) | 73 (66–80) | 57 (43–70) | <0.001 |
| Gender (male), n (%) | 58 (54.7) | 264 (46.7) | 0.139 |
| Comorbidities, n (%) | |||
| Hypertension | 63 (59.4) | 136 (24.1) | <0.001 |
| Diabetes | 25 (23.6) | 72 (12.7) | 0.006 |
| Coronary heart disease | 29 (27.4) | 31 (5.5) | <0.001 |
| Chronic renal disease | 17 (16.0) | 11 (1.9) | <0.001 |
| Chronic obstructive pulmonary disease | 10 (9.4) | 13 (2.3) | 0.001 |
| Cancer | 8 (7.5) | 15 (2.7) | 0.019 |
| Chronic heart failure | 13 (12.3) | 9 (1.6) | <0.001 |
| Cerebrovascular disease | 11 (10.4) | 11 (1.9) | <0.001 |
| Atrial fibrillation | 1 (0.9) | 6 (1.1) | 1.000 |
| Laboratory findings, median (IQR) | |||
| Leucocyte count, ×109/L | 9.0 (6.2–13.3) | 5.3 (4.0–7.3) | <0.001 |
| Neutrophil rate, % | 88 (82–92) | 68 (59–79) | <0.001 |
| Lymphocyte rate, % | 8 (4–12) | 21 (14–28) | <0.001 |
| Alanine aminotransferase, U/L | 29 (18–43) | 23 (16–36) | 0.527 |
| Aspartic transaminase, U/L | 36 (27–52) | 26 (20–34) | <0.001 |
| Creatinine, μmol/L | 67 (57–117) | 55 (48–65) | <0.001 |
| C-reactive protein, mg/L | 88 (60–147) | 29 (8–64) | <0.001 |
| Procalcitonin, ng/mL | 0.23 (0.09–0.69) | 0.05 (0.03–0.09) | <0.001 |
| CK-MB, ng/mL | 2.81 (1.45–5.72) | 0.85 (0.58–1.21) | <0.001 |
| MYO, μg/L | 125 (66–343) | 34 (24–62) | <0.001 |
| cTnI, ng/mL | 0.159 (0.075–0.695) | 0.006 (0.006–0.007) | <0.001 |
| NT-proBNP, pg/mL | 1346 (474–3018) | 133 (50–265) | <0.001 |
IQR, interquartile range; CK-MB, creatinine kinase-myocardial band; MYO, myoglobin; cTnI, cardiac troponin I; NT-proBNP, N-terminal pro-B-type natriuretic peptide.
Figure 5.
Distribution and dynamic profiles of myocardial indicators. (A) Contour plot showing the levels of CK-MB, MYO, and cTnI displayed a rising trend with age, and were significantly higher in the death group than those in the survivors group. (B) Line chart showing a dynamic growth of myocardial indicators in the death group (n = 31), these values were log-transformed for analysis. CK-MB, creatinine kinase-myocardial band; MYO, myoglobin; cTnI, cardiac troponin I.
Table 5.
Univariate and multivariate predictors of myocardial injury
| Characteristics | No. | Univariate |
Multivariate |
|||||
|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | P-value | OR | 95% CI | P-value | |||
| Male | 322 | 1.81 | 1.21–2.70 | 0.004 | 1.74 | 0.97–3.13 | 0.064 | |
| Age (per 10-year increase) | 671 | 2.23 | 1.84–2.69 | <0.001 | 1.64 | 1.28–2.10 | <0.001 | |
| Hypertension | 199 | 5.74 | 3.77–8.74 | <0.001 | 3.30 | 1.77–6.14 | <0.001 | |
| Diabetes | 97 | 2.00 | 1.21–3.28 | 0.006 | 0.63 | 0.29–1.34 | 0.231 | |
| Coronary heart disease | 60 | 6.79 | 3.90–11.83 | <0.001 | 2.92 | 1.32–6.48 | 0.008 | |
| Chronic renal disease | 28 | 11.33 | 4.98–25.75 | <0.001 | 9.03 | 2.43–33.59 | 0.001 | |
| Chronic heart failure | 22 | 7.32 | 3.05–17.55 | <0.001 | 2.19 | 0.54–8.92 | 0.276 | |
| Atrial fibrillation | 7 | 1.85 | 0.36–9.66 | 0.465 | 0.10 | 0.01–1.48 | 0.093 | |
| Cerebrovascular disease | 22 | 4.95 | 2.10–11.72 | <0.001 | 1.35 | 0.35–5.21 | 0.663 | |
| Chronic obstructive pulmonary disease | 23 | 4.08 | 1.72–9.69 | 0.001 | 4.01 | 1.28–12.61 | 0.018 | |
| Procalcitonin (ng/mL) | 671 | 1.28 | 1.08–1.52 | 0.005 | 0.99 | 0.79–1.23 | 0.900 | |
| C-reactive protein (mg/L) | 671 | 1.02 | 1.01–1.02 | <0.001 | 1.01 | 1.01–1.02 | <0.001 | |
OR, odds ratio; CI, confidence interval.
Discussion
The major findings of the present study are as follows: (i) myocardial injury is not uncommon among patients with severe COVID-19, especially among those who die; (ii) elevated levels of myocardial markers predict risk for in-hospital death; and (iii) senior age, inflammatory response, and underlying cardiovascular-related comorbidities are associated with myocardial injury in patients with COVID-19.
COVID-19 has managed to supersede the death toll of SARS and MERS. Severe respiratory distress is usually considered as a leading cause of coronavirus-induced death.7 In a recent study on the largest clinical sample in China, severe pneumonia was independently associated with admission to ICU, mechanical ventilation, or death. Other complications also were associated with higher risk of in-hospital death.8 Cheng et al. documented a 3.2% prevalence of kidney impairment in hospitalized COVID-19 patients in association with higher risk of mortality.9 Although information on cardiac complications is limited among COVID-19 patients, cardiac impairment has been demonstrated as a direct or underlying cause of death in 27% of pneumonia-associated deaths. Even after adjustment for baseline risk, cardiac complications are associated with a 60% increase in pneumonia-associated short-term mortality.10,11 However, there was insufficient evidence to suggest that patients with cardiovascular disease are more likely to develop COVID-19. Moreover, a larger and more representative sample is required to determine the exact mortality rate of COVID-19 in China, because mortality varies in different regions and time points. In the present study, patients with severe COVID-19 who died in hospital usually presented with a worse situation, such as older age, more comorbidities and abnormal laboratory findings, and more often required use of non-invasive or invasive ventilation. To analyse the potential risk factors for death, we found that several factors including older age, occurrence of chronic heart disease, elevated inflammatory response, and myocardial injury acted as co-effects on the risk for death. It is notable that in patients aged >60 years, the mortality seems independent of the elevation of myocardial indicator from the contour plot. Wu et al. reported that older age was associated with the development of ARDS and progression from ARDS to death among patients with COVID-19 pneumonia.12 In general, senior age has an important effect on complications and mortality in patients with COVID-19.
However, severe complications such as ARDS were not analysed in this multivariate model, since ARDS as the direct cause of mortality has always been identified in either MERS or SARS, and this time, in COVID-19.13 Consistently, we also found that ARDS and acute respiratory failure act as the main causes of mortality and present the highest proportion in dead cases. Moreover, ARDS usually acts as an endpoint in recent studies and its risk factors are similar to those for death. In consideration of the fact that ARDS may be more generally thought of as a pathway to mortality, we considered it would overshadow the other early indicators of mortality if it was included in the analysis of the multivariate model.
The elevation of cTnI and CK-MB might predict risk for death. Notably, these single cut-off points may include a number of individuals with a normal level of myocardial markers on admission.4 Although it is premature to say whether these patients are doomed with poor outcomes since only ∼30% of individuals died of myocardial injury, it plays a role as early warning for death in COVID-19 when these indicators exceed cut-off points.
Circulating inflammatory mediators (i.e. cytokines and/or endotoxins) or direct viral invasion of cardiomyocytes, or both mechanisms, may lead to myocardial injury in COVID-19. It was recently reported that the novel coronavirus uses the angiotensin-converting enzyme II (ACE2) as a cell entry receptor, and SARS-CoV was detected in the heart of 35% of the subjects suggesting that SARS-CoV is capable of infecting the myocardium through ACE2 receptors.14 Although one could posit that the direct invasion of cardiomyocytes by SARS-CoV-2 underlies cardiac impairment, a recent pathological study documented scarce interstitial mononuclear inflammatory infiltrates in heart tissue without substantial myocardial damage in a patient with COVID-19.15 Due to present limited evidence, whether SARS-CoV-2 can directly impair the heart remains to be further demonstrated.
A meta-analysis reported cumulative rates of new or worsening myocardial infarction or unstable angina (5%, range 1–11%) in patients with pneumonia.16 The serum concentrations of troponin increase during the acute phase of pneumonia, and the magnitude of this increase is associated with the severity and outcome of the infection.17 Recently, Huang et al. documented that patients with COVID-19 admitted to the ICU had higher plasma levels of cytokines.1 In the present study, we also found that levels of markers of inflammatory response such as C-reactive protein and PCT were significantly increased especially among patients who died. To further analyse related factors of myocardial injury by multivariable regression, senior age, comorbidities (e.g. hypertension, coronary heart disease, chronic renal failure, and chronic obstructive pulmonary disease), and inflammatory markers (e.g. PCT and C-reactive protein) were predictors of in-hospital myocardial injury in patients with severe COVID-19. The latter findings indicate that occurrence of cardiac impairment is possibly secondary to severe pneumonia-related systemic inflammatory response and closely related with underlying cardiovascular disease or potential risk in patients with COVID-19.
Consistently, previous evidence demonstrated that risk factors for pneumonia-associated cardiac complications include older age, pre-existing cardiovascular disease, and greater severity of pneumonia at presentation.18,19 Nonetheless, about one third of transient cardiac complications occur in patients without a history of cardiac disease.20 A study in SARS also found that reversible left ventricular impairment appears common even among those without underlying cardiac disease.21 However, sufficient evidence is needed to clarify whether the effects of COVID-19 and SARS on the myocardium are different. In the present study, patients who died had higher proportions of both myocardial injury and cardiovascular-related comorbidities, suggesting that cardiac complications with underlying cardiovascular disease or risk usually co-exist and develop to irreversible outcome. Alternatively, the systemic inflammatory response to pneumonia can also increase the inflammatory activity within coronary atherosclerotic plaques, rendering them unstable and prone to rupture.22 Therefore, the presence of pre-existing cardiovascular disease or related risk factors may aggravate myocardial injury, which therefore cannot be ignored in management of COVID-19. In general, patients with COVID-19 and myocardial injury are not actively recommended for emergency intervention strategies due to existing risk of cross-infection, and in most mid/moderate myocardial injury, the standard process of comprehensive management is generally based on risk stratification and patient classification.23
Limitations
Although this study included a large number of patients, it has several limitations. First, some data (such as echocardiography and dynamic measurements of myocardial markers) were lacking from clinical examinations of patients in isolation ward or ICU because of the logistical restriction at the onset of these emerging infections in Wuhan. Second, due to the existence of either unmeasured or unknown confounders, the causes of death or myocardial injury could be underestimated by analysis of multivariable regression. Third, because the clinical observation of patients is still ongoing at the time of manuscript submission, most patients have not reached the clinical endpoint. Finally, for the total number of 2 350 000 patients with COVID-19 worldwide, the current sample size is still small. In order to avoid statistical bias as much as possible, data from multiple centres and larger samples are warranted to further confirm the results of the present study.
Conclusion
Myocardial injury is not an uncommon complication among patients with severe COVID-19, especially among those who die. cTnI and CK-MB levels predict risk for in-hospital death, and myocardial injury is associated with senior age, inflammatory response, and cardiovascular-related comorbidities.
Acknowledgements
This research was supported by grants from the Natural Science Foundation of China (No: 81800447 and 81770324); the Natural Science Foundation of Hubei Province (No: 2017CFB204); and the Major Program of Technological Innovation of Hubei Province (No: 2016ACA153).
Conflict of interest: none declared.
References
- 1. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B.. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020;395:497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z.. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 2020;323:1061–1069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Madjid M, Solomon S, Vardeny O. ACC clinical bulletin addressing the cardiac implications of novel Wuhan Coronavirus (2019-nCoV). American College of Cardiology (ACC). https://www.acc.org/~/media/665AFA1E710B4B3293138D14BE8D1213.pdf.
- 4. Thygesen K, Alpert JS, Jaffe AS, Chaitman BR, Bax JJ, Morrow DA, White HD; ESC Scientific Document Group. Fourth universal definition of myocardial infarction (2018). Eur Heart J 2019;40:237–269. [DOI] [PubMed] [Google Scholar]
- 5.ARDS Definition Task Force, Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, Fan E, Camporota L, Slutsky AS. ARDS Definition Task Force. Acute respiratory distress syndrome: the Berlin Definition. JAMA 2012;307:2526–2533. [DOI] [PubMed] [Google Scholar]
- 6. Ming W, Qing Q, Wanzhou X, Bin Q, Jingwei W, Hongyun Z, Shupeng J, Junchi M, Zegang W, Yayun D, Fangyuan Z, Wei W, Yan Z, Zhihua L, Jingtao H, Xiaoqian G, Lina F, Zunen X, Di L, Zhiliang X, Tiangang L, Pingan Z, Yongqing T, Yan L. Clinical diagnosis of 8274 samples with 2019-novel coronavirus in Wuhan. medRxiv 2020.02.12.20022327.
- 7. Chen T, Wu D, Chen H, Yan W, Yang D, Chen G, Ma K, Xu D, Yu H, Wang H, Wang T, Guo W, Chen J, Ding C, Zhang X, Huang J, Han M, Li S, Luo X, Zhao J, Ning Q.. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ 2020;368:m1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS, for the. China Medical Treatment Expert Group for Covid-19. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. doi:10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Cheng Y, Luo R, Wang K, Zhang M, Wang Z, Dong L, Li J, Yao Y, Ge S, Xu G.. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney International 2020;97:829–838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Corrales-Medina VF, Musher DM, Wells GA, Chirinos JA, Chen L, Fine MJ.. Cardiac complications in patients with community-acquired pneumonia: incidence, timing, risk factors, and association with short-term mortality. Circulation 2012;125:773–781. [DOI] [PubMed] [Google Scholar]
- 11. Mortensen EM, Coley CM, Singer DE, Marrie TJ, Obrosky DS, Kapoor WN, Fine MJ.. Causes of death for patients with community-acquired pneumonia: results from the Pneumonia Patient Outcomes Research Team cohort study. Arch Intern Med 2002;162:1059–1064. [DOI] [PubMed] [Google Scholar]
- 12. Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S, Huang H, Zhang L, Zhou X, Du C, Zhang Y, Song J, Wang S, Chao Y, Yang Z, Xu J, Zhou X, Chen D, Xiong W, Xu L, Zhou F, Jiang J, Bai C, Zheng J, Song Y.. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. doi:10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Petrosillo N, Viceconte G, Ergonul O, Ippolito G, Petersen E.. COVID-19, SARS and MERS: are they closely related? Clin Microbiol Infect. doi:10.1016/j.cmi.2020.03.026. Published online ahead of print 28 March 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Oudit GY, Kassiri Z, Jiang C, Liu PP, Poutanen SM, Penninger JM, Butany J.. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur J Clin Invest 2009;39:618–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, Bai C, Gao T, Song J, Xia P, Dong J, Zhao J, Wang FS.. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. pii: S2213-2600(20)30076-X. doi:10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Dong M, Liu T, Li G.. Association between acute infections and risk of acute coronary syndrome: a meta-analysis. Int J Cardiol 2011;147:479–482. [DOI] [PubMed] [Google Scholar]
- 17. Moammar MQ, Ali MI, Mahmood NA, DeBari VA, Khan MA.. Cardiac troponin I levels and alveolar-arterial oxygen gradient in patients with community-acquired pneumonia. Heart Lung Circ 2010;19:90–92. [DOI] [PubMed] [Google Scholar]
- 18. Corrales-Medina VF, Serpa J, Rueda AM, Giordano TP, Bozkurt B, Madjid M, Tweardy D, Musher DM.. Acute bacterial pneumonia is associated with the occurrence of acute coronary syndromes. Medicine (Baltimore) 2009;88:154–159. [DOI] [PubMed] [Google Scholar]
- 19. Ramirez J, Aliberti S, Mirsaeidi M, Peyrani P, Filardo G, Amir A, Moffett B, Gordon J, Blasi F, Bordon J.. Acute myocardial infarction in hospitalized patients with community-acquired pneumonia. Clin Infect Dis 2008;47:182–187. [DOI] [PubMed] [Google Scholar]
- 20. Corrales-Medina VF, Musher DM, Shachkina S, Chirinos JA.. Acute pneumonia and the cardiovascular system. Lancet 2013;381:496–505. [DOI] [PubMed] [Google Scholar]
- 21. Li SS, Cheng CW, Fu CL, Chan YH, Lee MP, Chan JW, Yiu SF.. Left ventricular performance in patients with severe acute respiratory syndrome: a 30-day echocardiographic follow-up study. Circulation 2003;108:1798–1803. [DOI] [PubMed] [Google Scholar]
- 22. Madjid M, Vela D, Khalili-Tabrizi H, Casscells SW, Litovsky S.. Systemic infections cause exaggerated local inflammation in atherosclerotic coronary arteries: clues to the triggering effect of acute infections on acute coronary syndromes. Tex Heart Inst J 2007;34:11–18. [PMC free article] [PubMed] [Google Scholar]
- 23. Jing ZC, Zhu HD, Yan XW, Chai WZ, Zhang S.. Recommendations from the Peking Union Medical College Hospital for the management of acute myocardial infarction during the COVID-19 outbreak. Eur Heart J. doi:10.1093/eurheartj/ehaa258. [DOI] [PMC free article] [PubMed] [Google Scholar]