Abstract
Background
Novel coronavirus-19 disease (COVID-19) is associated with significant cardiovascular morbidity and mortality. To date, there have not been reports of sinus node dysfunction (SND) associated with COVID-19. This case series describes clinical characteristics, potential mechanisms, and short-term outcomes of COVID-19 patients who experience de novo SND.
Case summary
We present two cases of new-onset SND in patients recently diagnosed with COVID-19. Patient 1 is a 70-year-old female with no major past medical history who was intubated for acute hypoxic respiratory failure secondary to COVID-19 pneumonia and developed new-onset sinus bradycardia without a compensatory increase in heart rate in response to relative hypotension. Patient 2 is an 81-year-old male with a past medical history of an ascending aortic aneurysm, hypertension, and obstructive sleep apnoea who required intubation for COVID-19-induced acute hypoxic respiratory failure and exhibited new-onset sinus bradycardia followed by numerous episodes of haemodynamically significant accelerated idioventricular rhythm. Two weeks following the onset of SND, both patients remain in sinus bradycardia.
Discussion
COVID-19-associated SND has not previously been described. The potential mechanisms for SND in patients with COVID-19 include myocardial inflammation or direct viral infiltration. Patients diagnosed with COVID-19 should be monitored closely for the development of bradyarrhythmia and haemodynamic instability.
Keywords: COVID-19, SARS-CoV-2, Bradycardia, Sinus node dysfunction, Case series
Learning points
Novel coronavirus disease-19 (COVID-19) may cause pathological sinus node dysfunction in affected patients.
The mechanism for COVID-19-induced sinus node dysfunction is likely to be secondary to cardiac inflammation or direct viral infiltration.
Patients with COVID-19 should be monitored closely for development of bradyarrhythmia and haemodynamic instability.
Introduction
Novel coronavirus disease-19 (COVID-19) is a global pandemic caused by the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2).1 Along with respiratory distress, COVID-19 causes significant cardiovascular dysfunction in patients with, and without, pre-existing cardiovascular disease.1,2 During the SARS-CoV epidemic that began in 2002, significant sinus bradycardia occurred in 15% of patients, with a mean heart rate of 43 beats per minute (b.p.m.) and an average duration of 2.6 days.3 Recent studies have demonstrated that COVID-19 is associated with direct myocardial injury, and affected patients are at risk for cardiovascular complications including acute myocardial infarction, myocarditis, heart failure, cardiogenic shock, and cardiac arrest.2,4 To date, persistent inappropriate sinus bradycardia associated with COVID-19 has not been reported.5 In this report, we describe two cases of COVID-19-associated pathological sinus node dysfunction (SND) (Table 1).
Table 1.
Patient characteristics
| Age (years) | Gender | Cardiovascular comorbidities | Historical HR (b.p.m.) | Nadir HR (b.p.m.) | PR (ms) | QRS (ms) | Rhythms developed | Admission/peak troponin-I (ng/mL)* | Admission/ peak D dimer (ng/mL)† | Admission/ peak C-reactive protein (mg/dL)‡ | Hospital day conduction disease developed | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient 1 | 70 | F | None | 65–75 | 38 | 158 | 86 | Sinus bradycardia | 0.00/0.02 | 648/3264 | 27.7/33.6 | 1 |
| Patient 2 | 81 | M | Ascending aortic aneurysm (5.1 cm), OSA, HTN | 70–90 | 51 | 234 | 118 | Sinus bradycardia AIVR | 0.04/0.09 | 5297/5297 | 13.8/14.9 | 4 |
Abbreviations: OSA = obstructive sleep apnoea; HTN = hypertension; AIVR = accelerated idioventricular rhythm.
Troponin-I reference range: 0.00–0.04 ng/mL
D-dimer reference range: 0–230 ng/mL
C-reactive protein reference range: 0–0.5 mg/dL.
Timeline
| Day(s) of hospita lization | Event |
|---|---|
| Patient 1 | |
| 0 | Patient presents with 10 days of fever, congestion, and dry cough. COVID-19 is diagnosed. |
| 1 | Intubated for acute hypoxic respiratory failure. |
| 2 | Patient develops new sinus bradycardia (rate 38 b.p.m.) with decrease in mean arterial blood pressure (MAP) to a nadir of 50 mmHg. |
| Transthoracic echocardiogram does not demonstrate any structural or functional abnormalities. Epinephrine infusion is started with improvement of MAP to 65 mmHg. | |
| Sedation is changed from propofol to midazolam without improvement in heart rate. | |
| 3 | Epinephrine is weaned but the patient becomes hypotensive and does not have a compensatory rise in heart rate. |
| Midazolam is changed to ketamine with improvement in MAP. Epinephrine is discontinued. | |
| 4–15 | Patient remains haemodynamically stable in sinus bradycardia. |
| 16 | Patient is extubated and remains in sinus bradycardia. |
| Patient 2 | |
| 0 | Patient presents with 7 days of fever, cough, and congestion after recent travel. COVID-19 is diagnosed.Intubated for acute hypoxic respiratory failure |
| 4 | Patient develops new sinus bradycardia (rate 51 b.p.m.) but remains haemodynamically stable. |
| Transthoracic echocardiogram does not demonstrate any structural or functional abnormalities. | |
| Sedation is changed from propofol to ketamine without improvement in heart rate. | |
| 6 | Patient develops numerous episodes of accelerated idioventricular rhythm with associated hypotension; however, he does not require initiation of vasopressors due to prompt conversion to haemodynamically stable sinus rhythm. |
| 7–14 | Patient continues to be in haemodynamically stable sinus bradycardia with intermittent episodes of accelerated idioventricular rhythm. |
| 15 | Patient is extubated and remains in stable sinus bradycardia. |
Case presentation
Patient 1
A 70-year-old female with no significant past medical history and on no home medications presented to the hospital with 10 days of fever, congestion, and dry cough. She had recent sick contacts. On admission, her cardiovascular exam revealed a regular rate and rhythm with no murmurs or elevated jugular venous pressure, and respiratory exam was notable for soft inspiratory crackles in her bilateral lower lobes. In the Emergency Department, she developed acute hypoxaemic respiratory failure, requiring 3 L/min of supplemental oxygen to maintain adequate oxygen saturations. Her heart rate was 68 b.p.m., and her historical heart rates ranged from 65 to 75 b.p.m. A chest radiograph showed bilateral peripheral airspace opacities. Nasopharyngeal PCR was positive for COVID-19.
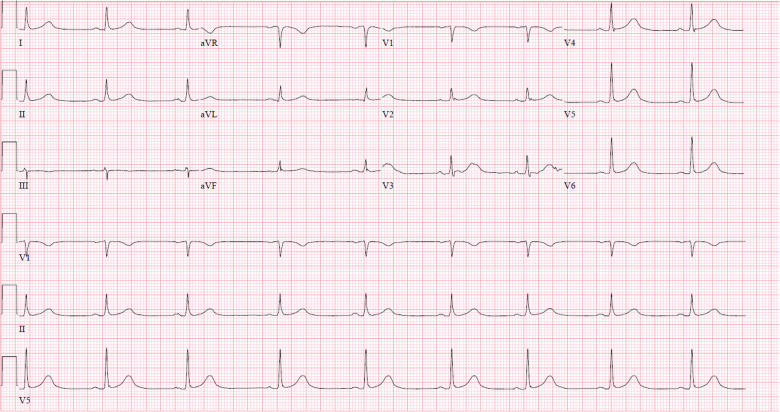
On day 1 of hospitalization, she developed worsening acute hypoxic respiratory failure, requiring 100% oxygen via a non-rebreather mask, and was transferred to the intensive care unit (ICU) where she was intubated and sedated with propofol and fentanyl. One day after intubation, she developed new sinus bradycardia on telemetry with a nadir heart rate of 38 b.p.m., associated with a mean arterial pressure (MAP) of 50 mmHg. An electrocardiogram confirmed sinus bradycardia (Figure 1). Transthoracic echocardiogram demonstrated no structural abnormalities and an ejection fraction of 65%. At this time, the patient was afebrile, and was producing 0.5 mL/kg/h of urine output. She had no electrolyte abnormalities, and had a normal troponin-I level. An epinephrine infusion was started, with improvement in her heart rate to 55 b.p.m. and MAP to 65 mmHg. Because her heart rate responded to epinephrine, temporary pacing was deferred. Her sedation was changed from propofol to midazolam; however, her heart rate did not increase. The patient was not taking atrioventricular (AV) node-blocking medications, nor was she being treated with any investigational drugs. Epinephrine was weaned on numerous occasions; however, the patient’s MAP decreased to <55 mmHg without a compensatory rise in her heart rate. Ultimately, midazolam was transitioned to ketamine, with improvement in her blood pressure. Epinephrine was discontinued, and her MAP remained stable above 65 mmHg. Two weeks following the onset of conduction disease, she has been extubated, and remains haemodynamically stable with average MAP >60 mmHg in persistent sinus bradycardia on continuous telemetry monitoring.
Figure 1.
Patient 1 ECG demonstrating new-onset sinus bradycardia.
Patient 2
An 81-year-old male with a past medical history of ascending aortic aneurysm, obstructive sleep apnoea, and hypertension presented with 7 days of fevers, myalgias, dry cough, and congestion after recent travel by aeroplane. On arrival to the hospital, he was hypoxic to 87% on room air and required 4 L/min of supplemental oxygen. The remainder of his pulmonary exam was notable for tachypnoea with coarse inspiratory crackles bilaterally. His cardiovascular exam revealed a heart rate of 98 b.p.m. and normal heart sounds without audible murmurs. Jugular venous pressure was not elevated. Per review of the medical record, historical heart rates ranged from 70 to 90 b.p.m. A chest radiograph demonstrated perihilar and retrocardiac airspace opacities. A respiratory viral panel was non-diagnostic for common respiratory viruses. PCR from a bronchoalveolar sample diagnosed COVID-19. The patient was intubated in the Emergency Department for rapidly increasing oxygen requirements.
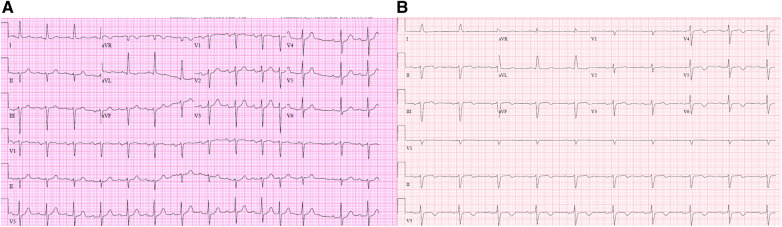
Four days after intubation and admission to the ICU, he developed new-onset sinus bradycardia and progressive first-degree AV block compared with an ECG performed 30 months previously (Figure 2). His nadir heart rate on telemetry was 51 b.p.m. A transthoracic echocardiogram demonstrated normal ejection fraction and no structural abnormalities. The patient was afebrile and producing 0.4 mL/kg/h of urine output. He had no electrolyte abnormalities. The day prior to onset of sinus bradycardia, his troponin-I was newly elevated to 0.09 ng/mL (reference range: 0.00–0.04 ng/mL); however, this value decreased to 0.04 ng/mL at the time that sinus bradycardia developed. He was not taking AV node-blocking medications, nor was he being treated with investigational drugs.
Figure 2.
Patient 2 ECG from 30 months prior to hospitalization (A) demonstrating normal sinus rhythm with first-degree AV block and premature atrial complexes. The patient’s ECG during his hospitalization for COVID-19 (B) demonstrates new-onset sinus bradycardia, stable advanced interatrial block, new inferolateral T wave inversions, progressive first-degree heart block, and stable left anterior fascicular block.
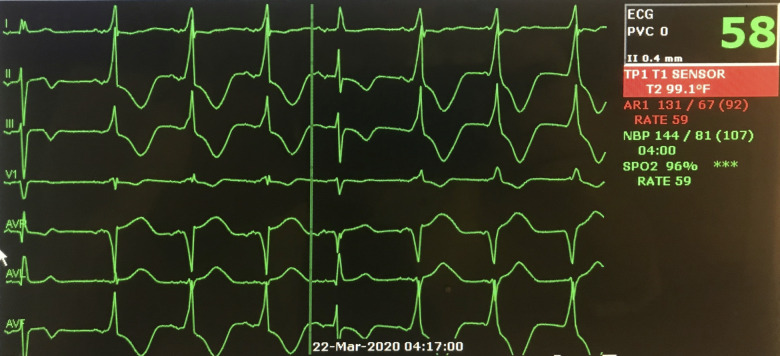
Prior to the onset of bradycardia, the patient was being sedated with propofol. After bradycardia developed, sedation was changed to ketamine without improvement in heart rate. Despite new-onset bradycardia, the patient’s MAP did not fall beneath 60 mmHg. Two days after the onset of sinus bradycardia, the patient developed numerous episodes of accelerated idioventricular rhythm (AIVR) (Figure 3). This rhythm was associated with hypotension; however the patient did not require initiation of vasopressors due to the brevity of these episodes and prompt conversion to a haemodynamically stable sinus rhythm. Two weeks following the onset of his arrhythmia, he has been extubated and remains haemodynamically stable with average MAP >65 mmHg in persistent sinus bradycardia on continuous telemetry monitoring.
Figure 3.
Telemetry from Patient 2 demonstrating interval development of accelerated idioventricular rhythm with isorhythmic atrioventricular dissociation and a single fusion beat.
Discussion
COVID-19-associated SND has not previously been described. In this case series of two patients with COVID-19, new SND was observed in the absence of provoking medications, structural heart disease, or prior arrythmia. The persistent nature of the sinus bradycardia in each patient made a classic vagally mediated response to the tracheal intubation, sedation, and/or critical illness unlikely. Patient 1 was unable to mount a chronotropic response to hypotension, suggesting development of an intrinsic deficit in her cardiac conduction system. Patient 2 developed haemodynamically insignificant sinus bradycardia, which subsequently progressed to haemodynamically significant AIVR.
The pathophysiology of cardiac disease secondary to SARS-CoV2 infection is postulated to be secondary to systemic inflammation and subsequent myocardial damage.4 Early descriptions of COVID-19-associated acute myocardial injury suggest that cardiovascular sequalae are secondary to a high systemic inflammatory response via cytokine release (interleukin-6 and tumour necrosis factor-α).6,7 Elevated troponin, as a surrogate for myocardial inflammation, has been associated with poor outcomes.8
Cardiac damage and conduction abnormalities were also observed during the original SARS-CoV outbreak (2002–2003). In vitro studies established that the angiotensin-converting enzyme 2 (ACE2) was responsible for viral uptake into host cells via spike protein and that this receptor is abundant in endothelial cells, intramyocardial smooth muscle cells, and cardiac myocytes.5 Autopsy studies of mice and rabbits infected with SARS-CoV demonstrated direct viral RNA inclusion in myocytes of specimens with cardiac injury and conduction system disease.9,10 Donoghue et al. demonstrated a correlation between ACE2 up-regulation and the severity of conduction disease in ACE2 transgenic mice.11 ACE2 is also the viral receptor for SARS-CoV2.5,12
While a mechanism for COVID-19-associated myocardial conduction abnormalities is currently speculative, it is possible that myocardial inflammation or direct viral infiltration via ACE2 affects myocardial conduction, leading to an impaired chronotropic response and progressive conduction system disease.5–7,12 While Patient 1 did not develop abnormal cardiac biomarkers, other markers of systemic inflammation such as D-dimer and C-reactive protein were significantly elevated (Table 1). Patient 2 also had elevated systemic markers of inflammation, along with elevated troponin on the day prior to developing sinus bradycardia, potentially suggesting an increase in myocardial inflammation predisposing to subsequent damage (Table 1). In Patient 2, myocardial inflammation may have progressed, causing the patient to intermittently lose sinus node dominance. His cardiovascular comorbidities of hypertension and obstructive sleep apnoea probably predisposed him to developing conduction system abnormalities.
Conclusion
COVID-19 may be associated with clinically relevant SND. Patients diagnosed with COVID-19 should be monitored closely for the development of bradyarrhythmia and haemodynamic instability. Potential mechanisms for SND include myocardial inflammation or direct viral infiltration. Future studies are needed to isolate this mechanism, and determine long-term outcomes of affected patients.
Lead author biography

Graham Peigh, MD, is a third-year internal medicine resident at McGaw Medical Center of Northwestern University. Originally from Chicago, IL, USA, Dr. Peigh completed his undergraduate education at Princeton University, and his medical education at Thomas Jefferson University. He will be a Chief Medical Resident at Northwestern before pursuing fellowship in Cardiovascular Disease.
Supplementary material
Supplementary material is available at European Heart Journal - Case Reports online.
Slide sets: A fully edited slide set detailing this case and suitable for local presentation is available online as Supplementary data.
Consent: The author/s confirm that consent for submission and publication of this case report including image(s) and associated text has been obtained from both patients in line with COPE guidance.
Conflict of interest: none declared.
Supplementary Material
Acknowledgements
The authors thank Jeremiah Wasserlauf, MD, MS, for his contributions to the manuscript.
References
- 1.WHO. World Health Organization Director-General’s Opening Remarks at the Media Briefing on COVID-19 March 11, 2020 https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020.
- 2. Clerkin KJ, Fried JA, Raikhelkar J, Sayer G, Griffin JM, Masoumi A, Jain SS, Burkhoff D, Kumaraiah D, Rabbani L, Schwartz A, Uriel N.. Coronavirus disease 2019 (COVID-19) and cardiovascular disease. Circulation 2020;doi: 10.1161/CIRCULATIONAHA.120.046941. [DOI] [PubMed] [Google Scholar]
- 3. Yu CM, Wong RS, Wu EB, Kong SL, Wong J, Yip GW, Soo YO, Chiu ML, Chan YS, Hui D, Lee N, Wu A, Leung CB, Sung JJ.. Cardiovascular complications of severe acute respiratory syndrome. Postgrad Med J 2006;82:140–144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Driggin E, Madhavan MV, Bikdeli B, Chuich T, Laracy J, Bondi-Zoccai G, Brown TS, Nigoghossian C, Zidar DA, Haythe J, Brodie D, Beckman JA, Kirtane AJ, Stone GW, Krumholz HM, Parikh SA.. Cardiovascular considerations for patients, health care workers, and health systems during the coronavirus disease 2019 (COVID-19) pandemic. J Am Coll Cardiol 2020;doi: 10.1016/j.jacc.2020.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Kochi AN, Tagliari AP, Forleo GB, Fassini GM, Tondo C.. Cardiac and arrhythmic complications in Covid-19 patients. J Cardiovasc Electrophysiol 2020;doi: 10.1111/jce.14479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Wang W, Ye L, Ye L, Li B, Gao B, Zeng Y, Kong L, Fang X, Zheng H, Wu Z, She Y.. Up-regulation of IL-6 and TNF-alpha induced by SARS-coronavirus spike protein in murine macrophages via NF-kappaB pathway. Virus Res 2007;128:1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Zheng YY, Ma YT, Zhang JY, Xie X.. COVID-19 and the cardiovascular system. Nat Rev Cardiol 2020;17:259–260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Lippi G, Lavie CJ, Sanchis-Gomar F. Cardiac troponin I in patients with coronavirus disease 2019 (COVID-19): evidence from a meta-analysis. Prog Cardiovasc Dis 2020;doi: 10.1016/j.pcad.2020.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Alexander LK, Keene BW, Small JD, Yount B Jr, Baric RS.. Electrocardiographic changes following rabbit coronavirus-induced myocarditis and dilated cardiomyopathy. Adv Exp Med Biol 1993;342:365–370. [DOI] [PubMed] [Google Scholar]
- 10. Oudit GY, Kassiri Z, Jiang C, Liu PP,, Poutanen SM, Penninger JM, Butany J.. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur J Clin Invest 2009;39:618–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Donoghue M, Wakimoto H, Maguire CT, Acton S, Hales P, Stagliano N, Fairchild-Huntress V, Xu J, Lorenz JN, Kadambi V, Berul CI, Breitbart RE.. Heart block, ventricular tachycardia, and sudden death in ACE2 transgenic mice with downregulated connexins. J Mol Cell Cardiol 2003;35:1043–1053. [DOI] [PubMed] [Google Scholar]
- 12. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B.. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020;395:1054–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.