Highlights
-
•
The new severe acute respiratory syndrome- coronavirus 2 affects the nervous system
-
•
A few documented specific processes reported to explain the neurological signs
-
•
A COVID-19 associated encephalitis patient is introduced here
-
•
The natural history is presented clinically and paraclinically
-
•
Immunotherapy seemed to hasten the improvement in this case
Keywords: COVID-19, SARS-Cov-2, encephalitis, post-infectious, natural history, immunotherapy
Abstract
The new severe acute respiratory syndrome- coronavirus 2 is reported to affect the nervous system. Among the reports of the various neurological manifestations, there are a few documented specific processes to explain the neurological signs. We report a para-infectious encephalitis patient with clinical, laboratory, and imaging findings during evolution and convalescence phase of coronavirus infection. This comprehensive overview can illuminate the natural history of similar cases. As the two previously reported cases of encephalitis associated with this virus were not widely discussed regarding the treatment, we share our successful approach and add some recommendations about this new and scarce entity.
1. Introduction
As the new emerging coronavirus, severe acute respiratory syndrome- coronavirus 2 (SARS- CoV 2), spreads all over the world, new features of this coronavirus disease 2019 (COVID- 19) is being reported. It is reported that there might be some mechanisms by which the disease can affect the central nervous system (CNS) (Li et al. 2020). Despite various neurological signs and symptoms, which have been reported (Mao et al. 2020), there are only a few reported cases with documented specific neurological processes to explain the CNS signs (Liu et al. 2020). To our knowledge, no cases of post infectious encephalitis (PIE) and its close follow up have been reported. Herein we report a case with clinical (including respiratory and neurological), laboratory, chest Computed Tomography and Brain Magnetic Resonance Imaging (B-MRI) findings during evolution and convalescence phase which can illuminate the natural history of similar cases.
2. Case report
A 39-year-old female presented to emergency department (ED) with fever, myalgias, anorexia, drowsiness and dry cough. Fever and myalgias had been present for nine days; she did not experience any improvement with rest and anti-inflammatory drug (NSAIDS). She had prolonged exposure to a covid-19 patient for prior 2 weeks. Past medical history was otherwise insignificant.
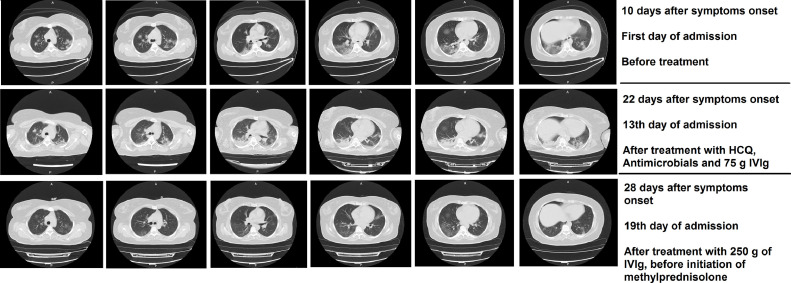
After presentation to ED (day 10), she demonstrated a decline in consciousness along with respiratory distress, ultimately needed endotracheal intubation. Chest CT showed multiple peripheral patchy ground-glass opacities with typical covid-19 distribution (day 10); these opacities later formed bilateral peripheral consolidation (day 22) (figure 1 ). She was transferred to intensive care unit (ICU) and treated with broad-spectrum IV antibiotics (meropenem 1g tid, levofloxacin 750 mg daily, linezolide 600 bid), hydroxychloroqine 400 mg bid for the first day, then 200 mg bid, atazanavir 400 mg daily and intravenous immunoglobulin (IVIg) 25 g/day for 3 days. RT-polymerase chain reaction (PCR) of a nasopharyngeal swab was negative for SARS-CoV 2 yet she was admitted with a diagnosis of Covid-19, based on clinical history and chest CT scan findings.
Figure 1.
Axial chest CT scans of the patient at days 10 (upper row), 22 (middle row) and 28 (bottom row) after onset of symptoms.
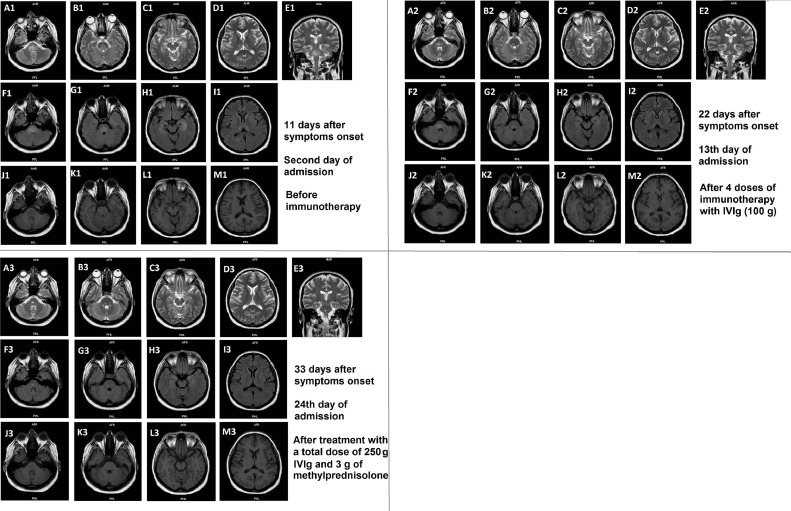
The patient experienced an episode of self-limited generalized tonic-clonic seizure (GTCS) on the following day (day 11) treated with intravenous (IV) levetiracetam 500 mg bid. She experienced another episode of GTCS on the same day, managed with increment in levetiracetam dosage (500 mg tid). After improvement of respiratory function, she was extubated (day 15). Despite improvement in inflammatory and metabolic tests, she had a fluctuating level of consciousness. During investigation for decreased level of consciousness, B-MRI revealed T2- fluid attenuated inversion recovery (FLAIR) high signal intensities in bilateral thalami, medial temporal and pons (figure 2 ). Corresponding areas in T1 images were hypo-signal and without gadolinium enhancement or restriction on T1 post-contrast and diffusion weighted imaging (DWI) sequences, respectively. Cerebrospinal fluid (CSF) analysis showed normal protein (19 mg/dl) and glucose (61 mg/dl) levels with no white or red blood cells. PCR tests for detecting viruses including SARS-CoV 2 and herpes simplex virus in CSF were negative. No oligoclonal bands (OCB) was detected. Autoimmune serologic markers including SS-A/SS-B and Anti-phospholipid antibodies were unremarkable. There was a borderline positive anti-nuclear antibody (ANA=2.7, positive>1.2) result. Other laboratory tests and their changes during disease course are listed in table 1 . These results led to the diagnosis of para- infectious encephalitis associated with COVID-19 and treatment with IVIg continued to a total dosage of 3g/kg of body weight (250g total) which resulted in considerable improvement in consciousness, but discontinued because of headaches (day 28). She complained of diplopia and was drowsy, even though the orientation seemed fully recovered (day 28). At this point, we continued the treatment with high dose methylprednisolone (500mg/day) IV for 6 days, after which the diplopia resolved and she regained complete consciousness. Subsequent B-MRIs (figure 2) showed marked resolution of medial temporal and thalami involvements as well as partial improvement in pontine lesions after reinstitution of IVIg (day 22) and administration of methylprednisolone (day 33). Results of SARS-CoV 2 serology tests taken during admission became available after discharge and were positive for both IgG and IgM.
Figure 2.
Three sets of axial (A-M except E) and coronal (E) brain MRIs obtained at days 11 (planes A1-M1), 22 (planes A2-M2) and 33 (planes A3-M3) after symptoms onset, show the evolution of brain lesions over time. There are T2-FLAIR high signal lesions in bilateral pons (A1, B1, E1, F1 & G1), medial temporal lobes (C1 & H1) and thalami (D1 & I1) and with low signal in T1 sequences (J1-M1). Corresponding images in second and third series show considerable resolution of signal intense lesions. There was neither gadolinium enhancement nor diffusion restriction in post-contrast T1 and diffusion weighted imagings (not shown here).
Table 1.
The sequence of clinical, laboratory and imaging findings during the natural history of our patient's disease.
| Days from symptoms onset | Day 1 | Day 10 | Day 11 | Day 22 | Day 28 | Day 33 |
|---|---|---|---|---|---|---|
| Days after admission | - | First admission day | Second admission day | 13th admission day | 19th admission day | 24th admission day |
| Clinical symptoms and treatments | Fever, Myalgia | Fever, Dry cough, Myalgia, Anorexia, Drowsiness Starting antibiotics, hydroxychloroqine, atazanavir and IVIg (75 g in the next three days) |
Fever, Myalgia (?), Anorexia (?), Respiratory distress, Coma, GTCS Intubation (for four days) |
Myalgia (?), Anorexia (?), No respiratory distress, Lethargy Reinstitution of IVIg (175 g in the next seven days; total=250 g) |
Drowsiness, diplopia, headache (improved with IVIg discontinuation) IV methylprednisolone started (total dose of 3 g in the next six days) |
Normal consciousness, No diplopia or other abnormal findings |
| Laboratory findings (in blood unless specified) | - | CBC (WBC=20000, Neut=15000, Lym=800, Plt=168000), CPK=540, Cr=1.6, CRP=48, ESR=31, LDH=1744, O2Sat=76%, Pro-BNP=1458, PCT≥200, Tn=296, VBG (pH=7.3, pCO2=79, HCO3=18.9) | CBC (WBC=14600, Neut=12200, Lym=1460, Plt=180000), CPK=N/A, Cr=1.6, CRP=N/A, ESR=N/A, LDH=938, O2Sat=81%, Pro-BNP=N/A, PCT=14.96, VBG (pH=7.34, pCO2=32, HCO3=18) | CBC (WBC=4600, Neut=2990, Lym=1380, Plt=199000), CPK=N/A, Cr=0.8, CRP<6, ESR=89, LDH=394, O2Sat=92%, Pro-BNP=N/A, PCT=N/A, VBG (pH=7.39, pCO2=64, HCO3=28) | CBC (WBC=4100, Neut=2660, Lym=1230, Plt=241000), CPK=N/A, Cr=0.8, CRP<6 CRP<6, ESR=33, LDH=466, O2Sat=88%, Pro-BNP=N/A, PCT=0.11, Tn=N/A, VBG (pH=N/A, pCO2=N/A, HCO3=N/A) | CBC (WBC=15800, Neut=10400, Lym=4700, Plt=349000), CPK=N/A, Cr=0.7, CRP<6, ESR=N/A, LDH=584, O2Sat=96%, Pro-BNP=N/A, PCT=N/A, Tn=N/A, (pH=N/A, pCO2=N/A, HCO3=N/A) |
| Chest CT findings | - | Multiple peripheral patchy ground-glass opacities | - | Obvious peripheral consolidations in both lunges | Marked resolution of consolidations, hazy opacities | - |
| Brain MRI findings | - | T2 high signal abnormalities in bilateral thalami, Pons and medial temporal lobes | T2 high signal abnormalities in bilateral Pons | - | Residual T2 signal abnormalities in lower pons |
CBC: complete blood count; CPK: creatin phosphokinase; Cr: creatinine; CRP: C- reative protein (mg/L, negative<6); CT: computed tomography; ESR: erythrocyte sedimentation rate (mm/hr); GTCS: generalized tonic clonic seizure; HCO3: bicarbonate (meq/L); IVIg: intravenous immunoglobulins; LDH: lactate dehydrogenase; Lym: lymphocytes (per mm3); MRI: magnetic resonance imaging; N/A: not available; Neut: neutrophils (per mm3); O2Sat: oxygen saturation; pCO2: partial pressure of carbon dioxide (mmHg); pH: power of hydrogen; Plt: platelets (per mm3); PRO-BNP: pro b-type natriuretic peptide (pg/ml, abnormal>125); PCT: Procalcitonin (ng/ml, normal<0.5); Tn=troponin- n (ng/ml, normal<19);VBG: venous blood gas; WBC: white blood cells (per mm3); ?: unknown because of decreased consciousness
3. Discussion
Brain involvement during COVID-19 pandemic has been reported in various forms, but the prevalence and proportion of each type of involvement is unknown (Mao et al. 2020, Liu et al. 2020). Stroke and encephalitis are specific CNS disorders reported in COVID-19 but there are many reports of neurological findings without obvious pathophysiologic explanations. The most common reported neurological manifestations have been headache, dizziness, encephalopathy and delirium. Some of these patients have been undergone a neurological workup, yet despite reports of CNS symptoms in up to 40% of these patients, there are only few cases where distinct etiology has been discovered (Liu et al. 2020). Among these there is only one established cases of encephalitis with evidence of virus in CNS (Moriguchi et al. 2020) and another case with acute necrotizing encephalitis (ANE) but without evidence for direct viral invasion (Poyiadji et al. 2020). The distribution of cerebral lesions in MRI for our patient was similar to both aforementioned cases but with more extensive and symmetrical involvement of thalami and medial temporal lobes. The involvement of pons has not been present in previous reported cases. Contrary to the ANE case, in our patient there was no evidence for hemorrhage or necrosis in serial B-MRIs. A recent paper has described a number of B-MRI changes in COVID-19 patients, but none of them have been the same as those in our patient (Kandemirl et al. 2020). Although in ANE the authors mentioned treatment with IVIg, but exact dosage, protocol and response to treatment has not been described. Our patient before diagnosis of neurologic involvement had received IVIg (25 g/day for three days) as a part of treatment for COVID-19 severe pulmonary involvement; and after the CNS lesions were established, it was reinstituted and due to very good clinical and radiological response, we decided to continue IVIg therapy until complete recovery, unless there is a complication. After administration of a total 250 g (3 g/kg) IVIg, it was stopped due to occurrence of headaches that seemed related to drug infusion. After discontinuation of IVIg, the headaches resolved completely and due to presence of diplopia and drowsiness treatment was then continued with methylprednisolone (500 mg/day for six days), which led to complete resolution of neurological symptoms. Upon discharge from the hospital, the patient was completely asymptomatic. We consider this type of brain involvement, a para- infectious autoimmune encephalitis, as we believe was the case for ANE patient, and recommend prompt immunotherapy upon diagnosis of similar cases. The use of IVIg in severe COVID-19 patients may prevent or ameliorate CNS involvement, as seemed to happen for our patient compared to ANE case.
4. Conclusion
Neurological involvement in COVID-19 is increasingly reported and the investigations regarding etiology, natural history and appropriate treatment are essential. As for our patient, neurological symptoms not correlated with respiratory and metabolic conditions should be an alarm for the possibility of CNS involvement. Similar to many other viral agents, SARS-CoV 2 seems capable of causing encephalitis. The nature of etiology regarding encephalitis remains the matter of debate as both direct invasion and para-infectious mechanisms seem possible. We recommend prompt assessment of CSF for overt signs of viral infection (marked cellular reaction, increased protein and positive PCR test) and considering immunotherapy with IVIg in cases compatible with para-infectious origin. The usefulness of prophylactic IVIg in severe COVID-19 and the role of serology in CSF for diagnosing neuroinvasion, remain interesting discussions that we are unable to comment about at this moment.
Disclosures
Funding sources
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Compliance with ethical standards
This study was approved by the ethics committee of Iran University of Medical Sciences, Tehran, Iran and also have been conducted in accordance with the ethical standards of Declaration of Helsinki.
Informed consent was obtained from the patient for publication of this report.
CRediT authorship contribution statement
Hale Afshar: Conceptualization, Investigation, Methodology, Supervision, Writing - review & editing. Zeynab Yassin: Investigation, Methodology, Writing - review & editing. Saeed Kalantari: Investigation, Resources, Writing - review & editing. Oldooz Aloosh: Resources, Writing - review & editing. Tayebeh Lotfi: Investigation, Writing - original draft. Mehdi Moghaddasi: Conceptualization, Investigation, Supervision, Writing - review & editing. Alireza Sadeghipour: Investigation, Resources. Maziar Emamikhah: Conceptualization, Data curation, Investigation, Methodology, Project administration, Resources, Software, Supervision, Writing - original draft, Writing - review & editing.
Declaration of Competing Interest
None.
Acknowledgement
None.
References
- Li YC, Bai WZ, Hashikawa T. The neuroinvasive potential of SARS‐CoV2 may play a role in the respiratory failure of COVID‐19 patients. Journal of medical virology. 2020 doi: 10.1002/jmv.25728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao L, Jin H, Wang M, Hu Y, Chen S, He Q. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA neurology. 2020 doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu K, Pan M, Xiao Z, Xu X. Neurological manifestations of the coronavirus (SARS-CoV-2) pandemic 2019–2020. Journal of Neurology, Neurosurgery & Psychiatry. 2020;91(6):669–670. doi: 10.1136/jnnp-2020-323177. [DOI] [PubMed] [Google Scholar]
- Moriguchi T, Harii N, Goto J, Harada D, Sugawara H, Takamino J. A first Case of Meningitis/Encephalitis associated with SARS-Coronavirus-2. International Journal of Infectious Diseases. 2020;94:55–58. doi: 10.1016/j.ijid.2020.03.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poyiadji N, Shahin G, Noujaim D, Stone M, Patel S, Griffith B. COVID-19–associated acute hemorrhagic necrotizing encephalopathy: CT and MRI features. Radiology. 2020:201187. [DOI] [PMC free article] [PubMed]
- Kandemirli SG, Dogan L, Sarikaya ZT, Kara S, Akinci C, Kaya D. Brain MRI Findings in Patients in the Intensive Care Unit with COVID-19 Infection. Radiology. 2020 doi: 10.1148/radiol.2020201697. [DOI] [PMC free article] [PubMed] [Google Scholar]