Introduction
Novel Corona Virus infection (COVID-19) is a highly infective disease with rapid global spread since January 2020 and was declared as pandemic by World Health Organization [1]. Angiotensin Converting Enzyme 2 (ACE 2), present on the cell surfaces of some important organs, has been identified as target receptor for COVID-19 virus entry into the host cells [2]. SPIKE protein present in the virus contains receptor-binding region, which attaches with ACE 2. The host protease TMPRSS2 breaks down SPIKE protein into subunits (S1 and S2) after binding with ACE 2 [1]. Eventually, virus fuses with the cell membrane of the host cell and gains entry via endocytosis [1]. Cells of lungs, heart, kidney, intestine and arteries predominantly harbor expression of ACE 2 on their surfaces and thus are the potential sites for virus entry and infectivity [3]. In addition, ACE 2 has been reported to be present at various locations in the oral cavity and hence, oral cavity is considered as a potential site for the COVID 19 virus [3].
ACE 2 is required for maintaining tissue homeostasis and is known for its anti-apoptotic, anti-inflammatory, anti-oxidant as well as anti-fibrotic role [4]. These protective effects are caused due to degradation of local Ang II and its product Ang1-7 via ACE2-Ang1-7-Mas pathway [5]. Disruption of this axis is the cause of various pathologies including fibrosis of the organs. Thus, down regulation of ACE 2 (decreased expression) has been reported to cause activation of ANG II, which acts as proinflammatory cytokines and facilitates fibrosis in many organs [6].
OSMF and COVID-19 interaction
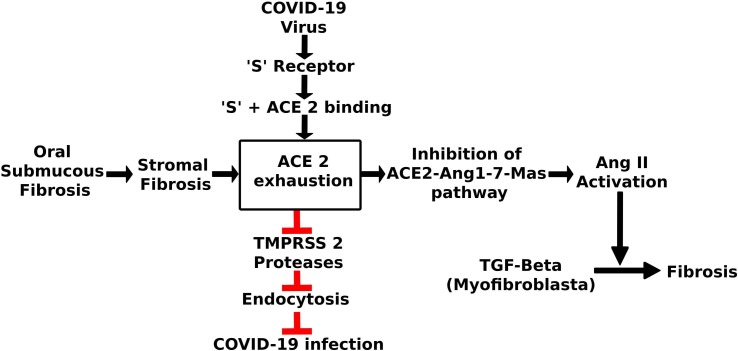
Oral submucous fibrosis (OSMF) is an insidious chronic disease characterized by increased fibrosis in the oral mucosa leading to reduced mouth opening. Numerous singling pathways causing increased collagen fiber deposition and reduced collagen degradation govern the fibrosis [7]. With aforementioned discussion in mind, it is quite conceivable to speculate down-regulation of ACE 2 in the oral cavity of OSMF patients. In this regard, ACE insertion/deletion polymorphism has been reported in OSMF tissues [8]. Thus, the less availability of ACE 2 in the oral cavity might have compromised the binding of COVID 19 virus. However, this has to be confirmed with appropriate epidemiological studies. The other way round, if OSMF patient is affected with COVID-19, then it may cause further exhaustion of ACE 2. The exhausted ACE 2 will unable to degrade the Ang II via ACE2-Ang1-7-Mas axis and lead to accentuation of fibrosis in OSMF patients. (Figure 1 )
Fig. 1.
Flowchart depicting the possible interactions between COVID-19 and oral submucous fibrosis.
Frontline workers and data collection
The frontline doctors and healthcare workers for management of COVID-19 patients belong to the medical fraternity. For them, monitoring of vital parameters is the primary task to effectively manage the severe complications of COVID-19 infection. Moreover, frontline doctors and healthcare workers are under tremendous stress, anxiety and depression because of fear of contacting infection and associated morbidity [9]. Under these circumstances, reporting of minor clinical details such are presence of OSMF is less imperative. However, if we add ‘presence or absence of OSMF’ in the checklist of case history format of COVID-19 patients, we would then be able to draw some interesting conclusions. Since diagnosis of OSMF is purely clinical in nature and no imaging or laboratory procedure is needed, it can be incorporated in the case history format without causing much burden to the frontline workers. Moreover, correlation of mouth opening (degree of severity of OSMF) with viral load will also give a nice outlook about this comorbidity. Since, majority of the frontline specialists belong to the medical field such as Emergency Medicine, General Medicine, Anesthesia etc., a brief training would be needed for clinically diagnosing and recording inter-incisal distance in COVID-19 positive OSMF patients.
The prevalence of OSMF is increasing every year with a current reported rate of 1.0–3.03% in China, 0.62–6.42% in India, 0.15–14.4% in Vietnam, and 0.086–17.6% in Taiwan [9]. Looking at the pandemic nature and magnitude of the spread of COVID-19 infection, health care workers and doctors are bound to come across the OSMF patients with the infection. In fact, such comorbidity might be present in the general population, which is not yet reported. We hereby strongly recommend that case history checklist for COVID-19 patients should have the OSMF related data entry.
In conclusion, ACE 2 exhaustion is the center stage of COVID-19 and OSMF pathogenesis. As ACE 2 is the binding site for COVID-19, it may compromise the infectivity of the virus. Moreover, COVID-19 infection in OSMF patient might aggravate the disease process by activation of Ang II. Nature of this complex interaction needs exploration in future studies so as to develop effective therapeutic strategies for this unique comorbidity.
Funding source: None declared.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.de Wit Emmie, van Doremalen Neeltje, Falzarano Darryl, Munster Vincent J. SARS and MERS: recent insights into emerging coronaviruses. Nat Rev Microbiol. 2016;14(8):523–534. doi: 10.1038/nrmicro.2016.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Xu H., Zhong L., Deng J. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int J Oral Sci. 2020;12(1): 8. Published. 2020;Feb:24. doi: 10.1038/s41368-020-0074-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Benigni A., Cassis P., Remuzzi G. Angiotensin II revisited: new roles in inflammation, immunology and aging. EMBO Mol Med. 2010;2(7):247–257. doi: 10.1002/emmm.201000080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Donoghue M., Hsieh F., Baronas E. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ Res. 2000;87(5):E1–E9. doi: 10.1161/01.res.87.5.e1. [DOI] [PubMed] [Google Scholar]
- 5.Oudit G.Y., Herzenberg A.M., Kassiri Z., Wong D., Reich H., Khokha R. Loss of angiotensin-converting enzyme-2 leads to the late development of angiotensin II-dependent glomerulosclerosis. Am J Pathol. 2006;168:1808–1820. doi: 10.2353/ajpath.2006.051091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ranganathan K., Devi M.U., Joshua E., Kirankumar K., Saraswathi T.R. Oral submucous fibrosis: a case-control study in Chennai. South India. J Oral Pathol Med. 2004;33(5):274–277. doi: 10.1111/j.0904-2512.2004.00116.x. [DOI] [PubMed] [Google Scholar]
- 7.Chung F.M., Yang Y.H., Chen C.H., Lin C.C., Shieh T.Y. Angiotensin-converting enzyme gene insertion/deletion polymorphism is associated with risk of oral precancerous lesion in betel quid chewers. Br J Cancer. 2005;93(5):602–606. doi: 10.1038/sj.bjc.6602746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lai Jianbo, Ma Simeng, Wang Ying, Cai Zhongxiang, Hu Jianbo, Wei Ning, Wu Jiang, Du Hui, Chen Tingting, Li Ruiting, Tan Huawei, Kang Lijun, Yao Lihua, Huang Manli, Wang Huafen, Wang Gaohua, Liu Zhongchun, Hu Shaohua. Factors Associated With Mental Health Outcomes Among Health Care Workers Exposed to Coronavirus Disease 2019. JAMA Netw Open. 2020;3(3):e203976. doi: 10.1001/jamanetworkopen.2020.3976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tilakaratne W.M., Klinikowski M.F., Saku T., Peters T.J., Warnakulasuriya S. Oral submucous fibrosis: review on aetiology and pathogenesis. Oral Oncol. 2006;42(6):561–568. doi: 10.1016/j.oraloncology.2005.08.005. [DOI] [PubMed] [Google Scholar]