Abstract
Pneumonia is very common and continues to exact a high burden on health. The Global Burden of Disease Study 2015 found lower respiratory infections (LRIs) were the leading infectious cause of death and the fifth leading cause of death overall. Pneumococcal pneumonia caused 55% of LRI deaths in all ages (1.5 million deaths).
Novel pathogens, particularly viruses, continue to emerge as causes of pneumonia. The rise of drug-resistance among common respiratory pathogens is a further challenge. Pneumonia is commonly classified according to patient location at the time of infection, leading to the categories of community-acquired, hospital-acquired and ventilator-acquired pneumonia.
Keywords: Bacterial pneumonia, Biomarkers, Community acquired pneumonia, Fungal pneumonia, Hospital-acquired pneumonia, Immunocompromised, Influenza, Legionella pneumonia, Lower respiratory tract infection, Streptococcus pneumoniae, Ventilator-acquired pneumonia, Viral pneumonia
Definitions
Pneumonia may be defined as an infection of the lung characteristically involving the alveolar space. The presence of microorganisms in the alveolar space without an accompanying inflammatory response represents colonization and does not constitute pneumonia. A range of other types of infection may also affect the lung and can be classified according to their principle site of infection (Fig. 1 ).
Fig. 1.
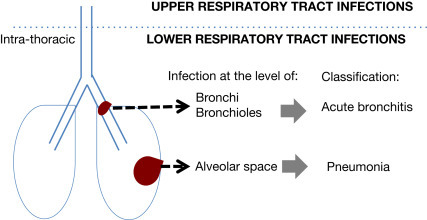
Schematic classification of infections of the lung.
Other Definitions
The term lower respiratory tract infection (LRTI) is often considered to include both acute bronchitis and pneumonia. However, it is sometimes used to designate non-pneumonic infections of the lower respiratory tract alone. In patients with chronic lung disease (e.g., chronic obstructive pulmonary disease (COPD)), an infection of the bronchi often results in an exacerbation of the underlying lung illness. In these circumstances, the illness is usually designated as an exacerbation of disease (e.g., exacerbation of COPD) rather than “acute bronchitis.”
In general, the more distal the infection within the respiratory tract, the greater the likelihood of bacterial infection and the greater the severity of illness. Exceptions to this include acute epiglottitis, diphtheria and pertussis which may present as severe bacterial infections of the upper respiratory tract without causing pneumonia.
Classification of Pneumonia
Pneumonia is further classified in various different manners. These are mainly clinical classifications that broadly describe differences in the likely range of pathogens involved (Table 1 ). The commonest grouping is according to patient location at the time of acquisition of infection. Infections arising within a hospital setting may involve more drug-resistant pathogens compared to infections arising in the community. Within the grouping of hospital-acquired pneumonia (HAP), further distinction is usually made according to whether the patient was on an intensive care unit, or intubated (ventilator-acquired pneumonia (VAP)) at the time of infection (Torres et al., 2017; Kalil et al., 2016). A specific category of Healthcare-associated pneumonia (HCAP) was previously advocated as describing pneumonia developing outside a hospital setting that shared features of pathogenesis, causative pathogens and antibiotic resistance patterns with nosocomial (hospital-acquired) pneumonia. This category was never fully adopted internationally and the latest evidence does not support the continued use of this classification.
Table 1.
Classification of pneumonia.
| Classification | Description/comments |
|---|---|
|
A. By Location 1. Community-acquired pneumonia 2. Hospital-acquired pneumonia 3. Ventilator-acquired pneumonia |
Infection acquired in the community Infection acquired following > 48 h stay in hospital or in someone recently discharged from hospital (within 7 days) Infection acquired by patient on ICU and mechanically ventilated for > 48 h |
|
B. By Immune status 1. Immunocompetent 2. Immunocompromised |
No immune dysfunction Immune dysfunction present– different definitions described, wide range in degree of immune dysfunction |
|
C. By Microbiology 1. Viral 2. Bacterial 3. Fungal 4. Mycobacterial 5. Parasitic |
This classification relies upon definitive microbiology which is usually unavailable at the time of first presentation, and may remain elusive even after investigation |
Host factors play an important role in the manifestation and management of pneumonia. Pneumonia arising in immunocompromised hosts usually warrants distinct treatment. In general, the greater the degree of immune compromise, the wider the range of potential pathogens. The classic symptoms of infection, which are partly related to the host immune response, may also be absent, altering the clinical presentation.
A third common classification is according to the causative pathogen(s). Until recently, a microbiological diagnosis used to take days to confirm. However, with the advance of rapid, point-of-care diagnostics, microbiological confirmation within minutes/hours of clinical presentation is becoming more realistic. Hence, this classification will hopefully become more clinically relevant in guiding patient management at the time of presentation.
Anatomically, pneumonias may be classified as bronchopneumonia or lobar pneumonia. Bronchopneumonia occurs when infection leads to multiple discrete foci of consolidation within the lung, whereas lobar pneumonia is described when the area of consolidation is confined to the affected lobe which is diffusely involved. It was once thought that the pattern of consolidation (whether described radiologically or pathologically) was indicative of the causative pathogen (e.g., lobar pneumonia caused by Streptococcus pneumoniae). However, it is now recognized that such discrimination is unreliable because of the large overlap in patterns caused by infecting pathogens.
Aspiration pneumonia refers to a specific situation when a patient who is manifestly at risk of aspiration develops pneumonia and anaerobic pathogens from the digestive tract are implicated, usually alongside multiple other microorganisms. An accompanying pleural reaction or lung abscess may develop. Micro-aspiration events are common and the aspiration of microorganisms into the lower airways likely accounts for how pneumonia develops in the majority of cases (see “Pathogenesis” section). Hence, the presence of micro-aspiration alone in a patient with pneumonia does not invariably denote aspiration pneumonia. Stroke-associated pneumonia has been advocated as the desired terminology for LRTI occurring within 7 days of acute stroke (Smith et al., 2015).
Pathogenesis
The lung is not a sterile environment. The normal lung microbiome includes bacterial species that may be implicated in the development of pneumonia, such as Streptococcus spp. and Mycoplasma spp. (Beck et al., 2015). These, and other microorganisms, are normally held in check by pulmonary host defenses. Disruption of these host defenses allow externally transposed pathogenic microorganisms to grow and displace the normal flora, or allow overgrowth of selected resident flora, leading to infection. There is growing interest in the role of antecedent viral respiratory tract infections as triggers for the disruption of the normal lung microbiome, providing an avenue for bacterial pathogens to take hold.
The acute inflammation generated by the host immune response to infection results in an influx of inflammatory cells into the alveolar space, giving rise to the radiological pattern of consolidation (Fig. 2 ). In most cases, the predominant inflammatory cell involved reflects the inciting pathogen; neutrophils in bacterial infections, lymphocytes in viral infections and granulomatous inflammation in Mycobacterial and fungal infections. The systemic cytokine response gives rise to many of the characteristic features of infection, such as fever, myalgia and a rise in C-reactive protein levels.
Fig. 2.
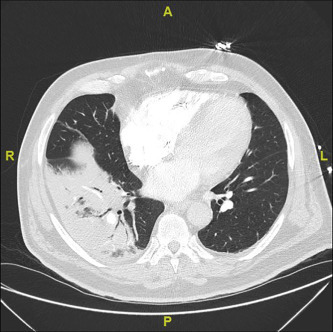
Consolidation on CT chest imaging.
The introduction of microorganisms to the lung is most commonly via micro-aspiration. Haematogenous spread from other sites in the body, and direct spread from a contiguous source are less common.
A range of host factors that predispose to pneumonia have been identified (Wunderink and Waterer, 2017; Almirall et al., 2017) (Table 2 ). These factors mostly increase the susceptibility to pneumonia through reducing host defenses. Some commonly used non-immunosuppressive drugs have been associated with pneumonia, but the mechanisms of action for all of these have not been fully described.
Table 2.
Risk factors for pneumonia.
| Condition | Example(s) |
|---|---|
| Age | Age > 65 years |
| Chronic co-morbid conditions | COPD, cancer, diabetes, chronic liver disease, renal impairment |
| Immunosuppressive disorders | HIV, Solid organ transplantation, immunosuppressive agents |
| Factors that increase the risk of aspiration | Placement of endotracheal tube, stroke, neurological disorder |
| Lifestyle factors | Smoking, high alcohol intake, malnutrition |
| Drugs | Proton-pump inhibitors, anti-psychotic medication, inhaled corticosteroids (in patients with COPD) |
Immunocompromised patients are not only at higher risk of developing pneumonia but the range of possible pathogens is also wider. As the number of therapeutic interventions that modify the immune system (such as monoclonal antibodies and tyrosine kinase inhibitors) expands, patients with widely differing levels of immune integrity are being described. In addition, multiple immune insults may exist together. For instance, the severe immune defects caused by hematological malignancies are often compounded by their treatments which may include cytotoxic drugs and/or total bone marrow ablation followed by hematopoietic stem cell transplantation.
Immunologically, defects can be broadly grouped into (a) cell-mediated defects, (b) antibody deficiencies and (c) neutrophil dysfunction, most commonly neutropenia. Understanding the type of immune defect facing a patient can aid as a guide to the likely range of pathogens involved, prior to confirmatory microbiological diagnosis.
Diagnosis
A definitive diagnosis of pneumonia comprises four aspects:
-
(i)
symptoms and signs of a respiratory tract infection,
-
(ii)
radiological changes,
-
(iii)
identification of a putative pathogen and
-
(iv)
a treatment response, or clinical course, consistent with pneumonia.
It is not always easy to make this diagnosis. In settings where investigations are not readily available, such as in primary care, a clinical (or working) diagnosis of pneumonia may be made without recourse to radiological or microbiological tests. The accuracy of a clinical diagnosis of pneumonia made in primary care is reasonable; 49–57% of patients clinically diagnosed with CAP have radiologically confirmed CAP. However, of patients with acute cough in whom a radiological diagnosis of CAP is made, only about 30% are clinically diagnosed as CAP (van Vugt et al., 2013). Even in secondary care, up to 25% of cases of pneumonia diagnosed in the Emergency Department are eventually discharged from hospital with an alternative diagnosis (Sikka et al., 2012).
The differential diagnosis of pneumonia includes other cardiac and pulmonary conditions that present acutely with features of cough and/or dyspnoea together with radiological abnormalities. (Table 3 ) In patients who are mechanically ventilated, diagnosing pneumonia amidst the wide range of differential diagnoses is challenging.
Table 3.
Differential diagnosis of pneumonia.
| Common | Less common |
|---|---|
| Cardiac failure/pulmonary oedema | Cryptogenic organizing pneumonia |
| Pulmonary embolism | Allergic bronchopulmonary aspergillosis |
| Cancer—primary lung/metastases | Acute pneumonitis—drugs, radiation |
| Adult Respiratory Distress Syndrome (ARDS) | Eosinophilic pneumonia—drugs, idiopathic |
| Pulmonary hemorrhage | |
| Pulmonary vasculitis |
Symptoms of Pneumonia
Patients with pneumonia usually present with a combination of (i) respiratory symptoms, specifically cough (∼ 75%), dyspnoea (∼ 65%), sputum production (∼ 30%) and chest pain (∼ 30%), and (ii) systemic symptoms including fever, rigors, myalgia and confusion. Confusion is commoner in older patients and those who are severely ill. Immunocompromised patients, and to a lesser extent, older patients, may not mount as rigorous an immune response and therefore may present with more subtle symptoms. About 10% of patients with CAP present to hospital with extra-pulmonary features alone; these include falls, generalized weakness and acute abdominal pain. A high index of suspicion is required in these circumstances.
Radiology
Until recently, identification of consolidation on a chest radiograph (CXR) has been regarded as the “gold standard” radiological investigation in the diagnosis of pneumonia. It is recognized that in patients with chronic lung abnormalities (e.g., pulmonary fibrosis, bronchiectasis, lung cancer) or in certain settings (e.g., intensive care unit), the sensitivity and specificity of a CXR for the identification of pneumonic changes can be limited. However, computed tomography (CT) scanning (Fig. 2) has raised further questions regarding the reliability of the CXR for the diagnosis of pneumonia more generally.
When evaluated against CT imaging, the CXR results in both over-diagnosis and under-diagnosis of pneumonia with up to a third of patients diagnosed with pneumonia on CXR having no infiltrate on CT scanning (Claessens et al., 2015). Ultrasonography is also being evaluated for the diagnosis of pneumonia with promising initial findings when compared against CXR (Orso et al., 2018). It remains necessary to determine if these imaging modalities will enable discrimination of patients suspected of having pneumonia into groups that warrant different management strategies.
Microbiology
Identification of a causative pathogen not only aids in the diagnosis and classification of pneumonia, it also guides antimicrobial therapy and infection control measures. A wide array of microbiological tests is available (Table 3). However, even with the use of modern molecular-based microbiological investigations (e.g., PCR, antigen detection tests) in patients with CAP, a pathogen is identified in only 50–75% of cases (Jain et al., 2015). In routine care where microbiology testing is still based mainly around cultural techniques (e.g., from blood and respiratory tract samples), a pathogen may be identified in only 5–10% of cases.
Conversely, in patients with suspected VAP, extensive colonization of airways creates difficulties in the interpretation of positive microbiology results. The use of highly sensitive PCR techniques can compound the difficulty. Similar challenges confront the management of immunocompromised patients. In these instances, careful attention to the coherence of microbiology test results with the clinico-radiological pattern is necessary to distinguish colonization from infection. Sometimes, a diagnosis of pneumonia can only be confirmed or refuted following review of the subsequent clinical course of illness, including the clinical response to empirically initiated antimicrobial agents.
Microbiological investigations are rarely performed in primary care settings due in part to a lack of access to laboratory facilities, low yield and delays in obtaining results in time to influence clinical management.
Rapid point-of-care tests
Advances in science and technology have come together to enable the development of rapid point-of-care (POC) tests that can provide microbiology test results within 15–60 min. The benefits of definitive identification of the causative pathogen (or in some cases, definitive exclusion of a specific pathogen, such as influenza) at the time of clinical presentation need to be weighed against the resource demands of these newer technologies and their costs. Because LRTIs can be caused by a range of pathogens, single-pathogen specific technologies are less helpful. Multi-array PCR platforms may overcome some of these hurdles, but come with increased costs. The cost-effectiveness of POC-driven management strategies compared to more empirical approaches remains to be proven in many circumstances.
Biomarkers
An alternative approach to the management of patients with LRTIs is to rapidly discriminate those who may have a bacterial infection and who would therefore require antibiotic therapy, versus those with a viral infection or a non-infectious condition. Several host-response biomarkers have been investigated in this regard (Table 4 ).
Table 4.
Biomarkers studied in respiratory tract infections.
| Biomarker | Description |
|---|---|
| C-reactive protein | Acute phase protein synthesized by hepatocytes |
| Procalcitonin | Prohormone of calcitonin which is induced by the activation and adherence of monocytes to the endothelial layer of blood vessels |
| Proadrenomedullin | Precursor for adrenomedullin which is involved in immuno-modulation |
| sTREM-1 | Soluble triggering receptor expressed on myeloid cells -1 (sTREM-1) levels rise following an increase of TREM-1 expression on neutrophils, granulocytes, monocytes and alveolar macrophages. TREM-1 expression is increased by microbial products |
| Pentraxin-3 | Acute inflammatory marker and a component of innate immunity |
| Neopterin | Produced in monocytes and macrophages. A marker of cell-mediated immunity. Levels rise in viral infections |
| Copeptin | Stable byproduct of vasopressin biosynthesis |
| Lipocalin-2 | Protein involved in innate immunity. It limits bacterial growth by sequestering iron-containing siderophores |
| Syndecan-4 | Receptor in intracellular signaling. Present in alveolar macrophages |
| Midregional proatrial natriuretic peptide (MR-proANP) | A byproduct of atrial natriuretic peptide (ANP) biosynthesis. ANP regulates macrophage activity in innate and acquired immunity |
C-reactive protein (CRP) and procalcitonin (PCT) are the two most extensively studied biomarkers in respiratory tract infections. Levels of these biomarkers increase more extensively in bacterial compared to viral infections. Procalcitonin has a more responsive kinetic profile than CRP, which means that levels of procalcitonin increase and decrease more swiftly in line with bacterial load. Apart from informing the decision whether or not to initiate antibiotics, the other role of CRP- and procalcitonin-guided treatment strategies in patients with pneumonia may be to guide the duration of antibiotic therapy through serial assessments of biomarker levels. A meta-analysis of individual participant data from 26 RCTs found that PCT-directed treatment in the management of acute respiratory tract infections (of varying types and severity, including CAP and HAP) was associated with a reduction in antibiotic exposure (5.0 vs. 7.0 days) composed of a decrease in both (a) the proportions initiating antibiotics and (b) the duration of antibiotics (6.0 vs. 8.0 days), as well as a reduction in 30-day mortality (8.6% vs. 10.0%) (Schuetz et al., 2017).
Proadrenomedullin, neopterin, sTREM-1 and pentraxin-3 are other biomarkers that have been found in a small number of early studies to be potentially of value in LRTIs (Saleh et al., 2018). Measuring Pentraxin-3 and sTREM-1 in bronchoalveolar lavage fluid samples has been found to be more discriminatory for bacterial versus viral infections than their levels in blood. Other biomarkers (e.g., copeptin, lipocalin-2, syndecan-4) display poorer performance characteristics. It may be that diagnostic performance can be further improved by combining different biomarkers (e.g., PCT and MR-proANP) or matching certain biomarkers to selected target patient populations (e.g., HAP vs. CAP).
Most of the investigated biomarkers aim to differentiate between bacterial or viral infections according to the host’s immunological response. The role of these biomarkers in patients with impairments of the immune system is therefore more limited, depending on the nature of the immune defect.
Epidemiology
Data on the epidemiology of pneumonia are drawn from two broad sources—(a) datasets that rely on the coding of pneumonia following clinical episodes, and (b) cohort studies of patients with radiology-confirmed pneumonia. The accuracy of clinical coding of pneumonia reflects the difficulties with making a firm diagnosis. In the UK, up to 20% of coded cases of hospitalized-pneumonia may have no radiographic evidence of infection (Daniel et al., 2017). Changes in the way pneumonia is coded can also affect the interpretation of data. In the US, there has been a shift in recent years from assigning a primary diagnosis code of “pneumonia” in patients hospitalized with severe CAP, towards a primary diagnosis code of “sepsis” with “pneumonia” as the secondary diagnosis (Lindenauer et al., 2012; Ruhnke et al., 2013). Prospective studies of radiology-confirmed pneumonia provide more robust data but are often less comprehensive in scope.
Community Acquired Pneumonia
Incidence
The Global Burden of Disease study estimated that in 2015, the incidence of LRTI in children aged < 5 years old was 0.15 episodes per child-year, and in all ages it was 0.04 episodes per person-year (Collaborators, 2017). Most episodes of LRTI/CAP are treated in community settings.
In the UK, CAP affects approximately 1% of the UK adult population each year, accounting for over 100,000 hospital admissions. The average length of stay is 6 days and estimated direct healthcare costs are £441 million. In the US, the annual incidence of CAP requiring hospitalization has been estimated at around 46 per 10,000 adults (Hayes et al., 2018). This compares with lower estimates from other parts of the world (Table 5 ). Incidence rises steeply in adults aged > 65 year (Table 6 ).
Table 5.
Estimates of the incidence of hospitalisations for CAP (Takahashi et al., 2013; Ewig et al., 2009; Trotter et al., 2008; Hayes et al., 2018).
| Country | Annual incidence (per 10,000 adults) |
|---|---|
| Vietnam | 8.1 |
| Germany | 29.6 |
| United Kingdom | 31.2 |
| United States | 46.5 |
Table 6.
Incidence of hospitalization for CAP according to age.
| Age (years) | Annual incidence (per 10,000 adults) |
|---|---|
| < 65 | 6.5–8.4 |
| 65–74 | 26.3–35.5 |
| 75–84 | 68–88 |
| 85 and above | 160–224 |
Data from Trotter CL, Stuart JM, George RC and Miller E (2008). Increasing hospital admission for pneumonia, England, Emerging Infectious Disease 14:727–33.
Changes in the prevalence of risk factors for pneumonia can be expected to influence the incidence of pneumonia. In the UK, a 34% increase in the incidence of CAP requiring hospitalization was observed from 1998 to 2005 (Trotter et al., 2008). Such trends of increasing incidence are thought to be explained by an aging population and a higher proportion of persons living with co-morbid illnesses, the latter encompassing an increase in persons with complex multi-morbidity. Some have implicated alterations in the epidemiology of causative pathogens (Quan et al., 2016). Changing patient expectations may also influence how healthcare is accessed and hospital admission policies. In contrast, in the US between 2001 and 2014, a decrease in the annual age-adjusted rate of pneumonia-associated hospitalisations was noted despite an increase in the proportion of co-existing immunocompromising conditions from 18.7% in 2001 to 29.9% in 2014 (Hayes et al., 2018).
In Sub-Saharan Africa, the incidence of CAP is dominated by the effect of HIV infection. The prevalence of HIV within cohorts of patients with CAP is 50–75%. In a community surveillance study in Kenya, the annual incidence of pneumococcal acute respiratory infection was 50 per 10,000 persons in HIV negative individuals compared to 670 per 10,000 persons in HIV positive individuals (Aston, 2017).
Effective vaccination against respiratory pathogens has the potential to prevent infection and decrease the incidence of pneumonia. National immunization programmes involving pneumococcal vaccines have contributed towards a reduction in overall pneumococcal infections and attendant mortality. However, in some countries, replacement disease, involving pneumococcal serotypes not covered by existing vaccines, has since begun to offset the reductions in vaccine serotype disease. Further vaccine development incorporating other serotypes, or effective against all pneumococcal serotypes/serogroups, will hopefully curtail this rise in replacement disease.
Mortality
Globally, LRTIs are the leading infectious cause of death and the fifth-leading cause of death overall. In 2015, LRTIs caused 2·74 million deaths in all ages, with children < 5 years of age bearing a disproportionate burden (704,000 deaths). Between 2005 and 2015, the number of deaths due to LRTI decreased by 36.9% in children younger than 5 years, and by 3.2% in all ages; most of these decreases occurred in countries with a low to middle socio-demographic Index (SDI). In high-SDI countries, the LRTI mortality rate in all ages increased by 9.6% over the same period (from 36.2 per 100,000 to 39.7 per 100,000) (Collaborators, 2017).
In the US, pneumonia remains the leading infectious cause of death, and the eighth commonest cause of death overall. Since 2000, mortality from pneumonia and influenza in the US has stayed below 20 deaths per 100,000 population. Most deaths occur in hospitalized patients. In the period 2001–14, in-hospital deaths occurred in 7.4% of pneumonia-associated hospitalisations (Hayes et al., 2018).
In Europe, mortality rates for hospitalized patients are mostly around 5–20% though a wide range is reported likely reflecting differences in healthcare systems, data sources and possibly microbiological patterns. In patients admitted to ICU with CAP, mortality rates are in the region of 20–30%. In Africa, an even wider range of mortality rates have been reported, from < 1% to nearly 50%. In most high-income countries, advancing age is associated with increasing mortality rates. However, in sub-Saharan Africa, this trend is not always evident; 55% of LRTI-related deaths in adults occur in persons under 70 years, including 22% in adults aged 15–49 years.
Rates of admission to ICU vary globally according to availability of resources and admission policies. A higher proportion of hospitalized patients are admitted to ICUs in North American (> 15%) compared to Europe (5–10%).
Hospital Acquired Pneumonia
Incidence
Hospital acquired pneumonia is the second commonest nosocomial infection after urinary tract infections. Estimated incidence rates are 5–20 cases per 1000 hospital admissions (Torres et al., 2017). These rates are influenced by the hospital ward setting and patient groups affected. The majority of HAP (65%) occurs on non-ICU wards, however, the incidence of HAP is greater among patients in ICU compared to patients on general wards. Higher rates are also observed in at-risk patient groups such as the elderly, those who have had surgery and immunocompromised hosts.
The incidence of VAP is 2–6 episodes per 1000 ventilator-days according to data from the US. During the first 5 days of mechanical ventilation (MV), the estimated risk of VAP is 3% per day, decreasing to 2% per day from days 5 to 10 of MV, and to 1% per day from day 10 of MV onwards. Patients with brain injury and trauma are at particularly high risk of VAP (50%) (Kalil et al., 2016).
Mortality
Hospital acquired pneumonia is the leading cause of death from nosocomial infections in critically ill patients. The crude mortality from HAP is high (up to 70%). However, patients who develop HAP are often already severely ill and the factors that predispose to HAP may also increase the risk of mortality. Accordingly, mortality is higher for patients with VAP than for patients with HAP on general wards. In some studies of VAP, 30–50% of VAP-related deaths are deemed to be a direct result of infection. The VAP-attributable mortality has been estimated at 13% with surgical patients carrying the highest associated risks (Melsen et al., 2013). Infection with Pseudomonas aeruginosa or Acinetobacter spp. is associated with higher mortality as well.
Etiology
An extensive range of pathogens can cause pneumonia. The respiratory pathogens commonly implicated in patients with CAP remain important aetiological agents in all other types of pneumonia, including HAP and pneumonia in the immunocompromised host (Table 8). Factors that modify (usually extend) the range of pathogens that might be implicated include alterations in immune status, exposure to specific environments/pathogens and prior exposure to antibiotics.
Table 8.
Pathogens commonly associated with specific environments or exposures.
| Exposure/high incidence areas | Pathogen |
|---|---|
| Bats | Histoplasma capsulatum |
| Birds | Chlamydia psittaci, Histoplasma capsulatum, Cryptococcus neoformans |
| Poultry, wild birds | Avian influenza |
| Rabbits, deer | Francisella tularensis |
| Farm animals (esp. birthing season) | Coxiella burnetii (Q fever) |
| Contaminated water sources, compost | Legionella spp. |
| Kingdom of Saudi Arabia—camels | Middle East Respiratory Syndrome (MERS) |
| East Asia, South America—consumption of infected crustaceans | Paragonimus westermani (lung fluke) |
| Thailand, Vietnam, northern Australia—contaminated soil | Burkholderia pseudomallaei |
| South-East Asia, East India, South China—immunocompromised | Talaromyces marneffei (previously Penicillium marneffei) |
S. pneumoniae is the predominant bacterial pathogen in pneumonia, especially CAP. Geographical differences are important in two broad regards: (a) the spectrum and frequency of likely pathogens, (b) the patterns of drug-resistance likely to be encountered. In countries where tuberculosis (TB) is endemic, acute TB pneumonia is a well-recognized cause of CAP in both immunocompetent and immunocompromised persons. In Thailand particularly, and to a less extent in some surrounding countries such as northern Australia, Burkholderia pseudomallei (a Gram-negative bacillus that is present as free-living saprophytes in soil and surface waters in endemic areas) is a common cause of fulminant CAP with high mortality.
Time from hospital admission to the development of HAP influences the likely pathogens encountered. Alterations in a patient’s normal flora increase with duration of stay in hospital, potentially modified by illness, medical procedures and drug (antibiotic) exposure. As a general guide, the risk of infection from drug-resistant pathogens increases with duration of hospital stay. The main additional pathogens to consider are Gram-negative enteric bacilli and methicillin-resistant S. aureus (MRSA) (Table 7 ).
Table 7.
Common microbial pathogens in pneumonia.
|
A. Community acquired pneumonia | |
| Bacterial | Streptococcus pneumoniae |
| Haemophilus influenzae | |
| Legionella spp. | |
| Mycoplasma pneumoniae | |
| Chlamydia pneumoniae | |
| Staphylococcus aureus | |
| Klebsiella pneumoniae | |
| Viral | Influenza virus |
| Respiratory Syncytial virus | |
| Metapneumovirus | |
| Coronavirus | |
| Rhinovirus | |
| Adenovirus | |
| Other | Mycobacterium tuberculosis—in TB endemic areas |
| B. Hospital acquired pneumonia | |
| In addition to pathogens implicated in CAP | Pseudomonas aeruginosa |
| Acinetobacter baumannii | |
| Stenotrophomonas maltophilia | |
| Enterobacteriaceae | |
| Methicillin-resistant S. aureus | |
| C. Immunocompromised host | |
| In addition to pathogens implicated in CAP and HAP: | |
| Fungal | Pneumocystis jirovecii |
| Aspergillus spp. | |
| Cryptococcus neoformans | |
| Endemic fungi e.g., histoplasmosis, cocciodiomycosis (geographically restricted) | |
| Viral | Cytomegalovirus |
| Varicella | |
| Herpes | |
| Parasitic | Strongyloides stercoralis |
| Toxoplasma gondii | |
| Bacterial | Nocardia asteroides |
| Rhodococcus eui | |
| Bordetella pertussis | |
| Pasteurella multocida | |
| Mycobacterial | Mycobacterium tuberculosis |
| Non-tuberculous mycobacteria | |
The additional pathogens to consider in immunocompromised patients with pneumonia depends on the degree of immune dysfunction at the time of infection. In patients with HIV, the risk of infection with S. pneumoniae (and subsequent bacteraemia) is increased over 10-fold even with normal CD4 counts (> 500 cells/μL). Similarly, the risk of infection by Mycobacterium tuberculosis is increased in early HIV infection. With CD4 counts < 200 cells/μL, the risk from “opportunisitc” infections increases vastly (Table 8 ) (Segal et al., 2011). Infections with fungal pathogens are of particular concern in patients with severely impaired immune defenses. Of note, some pathogens which commonly cause systemic infections in immunocompromised hosts rarely involve the lung (e.g., Candida spp.). Other pulmonary infections represent reactivation of disease as immune function declines rather than the acquisition of new disease (e.g., non-tuberculous mycobacteria, Toxoplasma gondii).
Certain pathogens are associated with specific environmental exposures, or in the case of zoonosis, exposure to animal reservoirs (Table 8). Some pathogens display seasonal patterns of higher incidence. For instance, infections with influenza, respiratory syncytial virus and S. pneumoniae are commoner in winter, whilst legionella infections are commoner in summer when the weather is hotter and more humid.
Over the years, the list of pathogens causing pneumonia has continued to increase through a combination of factors; advances in microbiological diagnostics, better recognition of clinical syndromes and the expansion of human populations into new territories. In particular, a number of viral pathogens have been recognized in the last two decades as implicated in the development of pneumonia. These include coronavirus, human metapneumovirus and enterovirus-D68.
Infection by more than one pathogen can occur in up to a third of patients with pneumonia. These are mostly viral-bacterial pathogen combinations and may reflect recognized associations between certain infections. For instance, influenza-related pneumonia is associated strongly with the bacterial pathogens S. aureus and S. pneumoniae. Particular attention is necessary with immunocompromised patients when multiple pathogens may co-exist to cause disease.
Drug-Resistant Pathogens
In 2017, the World Health Organization (WHO) published a global priority list of antibiotic-resistant bacteria to guide research, discovery and development of new antibiotics. The list includes a number of important respiratory pathogens, such as penicillin non-susceptible S. pneumoniae, ampicillin-resistant H. influenzae, methicillin and vancomycin-resistant S. aureus, carbapenem-resistant A. baumannii, carbapenem-resistant P. aeruginosa, and carbapenem- and third-generation-cephalosporin resistant Enterobacteriaceae (such as K. pneumoniae). Whilst many of these pathogens are mainly implicated in nosocomial infections, some are important in CAP as well.
Rates of drug-resistant S. pneumoniae (DRSP) vary globally. Northern European countries have tended to have lower numbers of DRSP (< 5% of pneumococcal isolates) while some other countries (Southern Europe, Japan and the United States) report figures of DRSP approximating 20–30% of isolates. The introduction of childhood PCV vaccination programmes in many countries has generally led to a reduction in rates of DRSP. The epidemiology of DRSP continues to change under vaccine pressure. In Europe, during the era of PCV vaccination, the most frequent serotypes to display resistance to penicillin (from samples taken in 2010 to 2011) were serotypes 14, 19A and 15A. Multi-drug resistance (defined as resistance to 3 or more classes of antimicrobial agents) was commonest in serotype 15A, a non-vaccine serotype (Yahiaoui et al., 2018).
Treatment
Severity Assessment
An assessment of illness severity is fundamental to the management of patients with pneumonia. Severity assessment guides decisions around (a) place of treatment (whether in the community, in hospital, or in intensive care), (b) depth of investigations, (c) speed of treatment and (d) type of treatment, including the choice of antimicrobial agents and route of administration. Various severity assessment tools have been developed for the management of patients presenting with CAP. The two most widely validated and adopted tools are the Pneumonia Severity Index (PSI) and the CURB65 score. Both of these were developed to predict short-term, 30-day mortality in patients presenting to hospital with CAP. Prognostic tools to predict ICU admission are not as widely used partly due to differences in ICU admission policies influencing the predictive accuracy of tools developed in one healthcare system and applied to a different healthcare system.
Severity assessment tools for HAP are less well validated. This reflects the much broader diversity of factors influencing prognosis in patients with HAP, including type of hospital ward, reason for hospital admission, time from hospital admission, medical interventions, exposure to nosocomial pathogens and preceding antibiotic exposure. Similarly, there are no pneumonia-specific tools for assessing severity in immunocompromised patients. In these patients, the degree of immune compromise is often the dominating factor in severity assessment. Patients who are severely immunocompromised may lack many of the symptoms or signs associated with severe illness. A high degree of vigilance is therefore important.
Time to Antimicrobial Treatment
Early treatment with appropriate antimicrobial agents is the goal. In patients who are severely ill, earlier treatment (measured in hours) is associated with improved clinical outcomes, such as mortality. In patients presenting with signs indicative of severe sepsis, antibiotic administration within an hour is advocated. The impetus for early antimicrobial treatment limits the window in which to complete investigations to confirm the diagnosis of pneumonia and associated microbiology, prior to antimicrobial administration. In many instances, empirical broad-spectrum treatment must be started whilst awaiting results.
In the case of influenza infection, time from onset of illness to antiviral treatment is the critical factor. Viral load peaks rapidly within the first 2 days of illness. In line with this, evidence of clinical benefit from neuraminidase inhibitors is greatest when treatment is started within 48 h of symptom onset.
Concepts such as “start smart, then focus” acknowledge the inevitable uncertainty that often exists at the time of commencement of antimicrobials, and emphasize the equally important role of reviewing the diagnosis and treatment plan in the light of emerging results and response to empirical treatment. This pertains not only to antibiotics, but also to antiviral and antifungal agents.
Duration of Antimicrobial Treatment
The optimal duration of antimicrobial therapy in the treatment of pneumonia has not been adequately studied. Traditionally, a 7-day course of antibiotics has been standard. Increasing awareness of the importance of good antimicrobial stewardship has led to efforts to refine the required duration of antimicrobial therapy. Shorter 5-day courses of treatment are gaining acceptance, as are biomarker-driven antibiotic prescribing strategies (see section on “Biomarkers”). Some infections, such as legionella pneumonia, are still treated with longer courses of antibiotics (10–14 days) based on clinical experience rather than evidence derived from clinical trials.
Adjuvant Therapy
In the treatment of pneumonia, adjuvant therapy refers broadly to interventions that are aimed at modulating the immune response to infection. The use of systemic corticosteroids has been extensively tested in patients with severe sepsis, a large proportion of whom have pneumonia. A meta-analysis of 42 trials involving > 10,000 patients found, with low certainty, a small mortality benefit in favor of low-dose corticosteroids (Rochwerg et al., 2018). Fewer trials have been conducted in patients with CAP and the existing debate around the value of corticosteroids for this specific indication reflects the weaker evidence base; evidence from 17 trials involving > 2000 patients indicate a mortality benefit in patients with severe pneumonia, but not in those with non-severe pneumonia (Stern et al., 2017).
Macrolide antibiotics have immunomodulatory properties apart from their antibiotic effects. Large observational cohort studies of patients with both all-cause pneumonia (i.e., involving a range of respiratory pathogens) and pneumococcal pneumonia suggest the combination of macrolide with beta-lactam antibiotics is associated with improved prognosis. In contrast, data from available randomized controlled trials are conflicting.
HMG-CoA reductase inhibitors (statins) have a range of immunomodulatory effects and in murine models of sepsis, pre-dosing with statins has been associated with improved outcomes. Studies in adults with pneumonia support the notion that patients already taking a statin at the time of infection have a better prognosis, but the value of statins started as adjuvant therapy in patients presenting with pneumonia has not been established.
Prevention
Immunization programmes against common respiratory pathogens are cost-effective public health interventions for the prevention of CAP in many countries. In relation to S. pneumoniae, two types of pneumococcal vaccines are commercially available. The current multivalent pneumococcal polysaccharide vaccine (PPV) was first introduced in 1983 and covers 23 of over 90 pneumococcal serotypes/serogroups (1,2,3,4,5,6B,7F,8,9N,9V, 10A, aa!,12F,14,15B,17F,18C,19A,19F,20,22F,23F,33F.). The 23-valent PPV offers good protection against invasive pneumococcal disease but relatively weak protection against pneumococcal pneumonia (Falkenhorst et al., 2017). In older persons who are most at risk of pneumonia, immunosenescence adversely influences the protective effect of these vaccines.
Pneumococcal conjugate vaccines (PCVs) promote a more robust immune response and are able to reduce nasopharyngeal carriage of vaccine-type S. pneumoniae strains, but cover a smaller range of pneumococcal serotypes. As children are the main carriers of S. pneumoniae, vaccination of children with PCVs has been associated not only with a reduction in the incidence of childhood pneumococcal infections, but also in the incidence of adult pneumococcal pneumonia (herd protection) (Tsaban and Ben-Shimol, 2017). Countries vary in the target groups for pneumococcal vaccination and the vaccines offered.
Trivalent influenza vaccines against 3 of the 4 main circulating influenza strains (2 influenza A strains and 1 influenza B strain) have been available for many years. Due to antigenic shifts within influenza viruses, the components of these vaccines are reviewed (and renewed) each year to maximize vaccine-pathogen “match.” Vaccines against all four of the common seasonal influenza viruses (quadrivalent vaccines covering 2 influenza A strains and 2 influenza B strains) are now available. In addition, and perhaps more importantly, is the development of newer conjugated vaccines and high-dose vaccines that promote stronger immune responses in older at-risk persons compared to previous “standard” influenza vaccines, hence offering greater protection. Influenza vaccines that are not strain-specific (so-called “universal” vaccines) and therefore less subject to mismatch are also being developed as are vaccines against respiratory syncytial virus.
The H. influenzae type b (Hib) vaccine is available for the prevention of childhood pneumonia. However, in adults, non-typeable H. influenzae (NTHi) is much more important and there are some data to suggest that childhood carriage of NTHi may be increasing. An effective vaccine for NTHi is not currently available.
Smoking cessation and a reduction in alcohol use are important modifiable lifestyle factors for the prevention of pneumonia. Current tobacco smokers are over two times more likely (pooled odds ratio 2.7, from meta-analysis) to develop CAP compared to adults who have never smoked, while people who consume alcohol (or in higher amounts) have a 83% increased risk of CAP compared to those who consume no (or less) alcohol (Simou et al., 2018). For every 10–20 g higher alcohol intake per day, there is an 8% increase in the risk of CAP.
A large number of specific interventions have been advocated in the prevention of VAP. These may be grouped as functional (e.g., semi-recumbent position), mechanical (e.g., silver-coated endotracheal tube) and pharmacological (e.g., selective decontamination of the digestive tract) interventions. The Institute of Health Improvement (IHI) developed the concept of “bundles” of care in the ICU to improve clinical outcomes. Implementation of a VAP prevention bundle may enable the ideal goal of “zero VAP” to be achieved (Álvarez-Lerma et al., 2018, Alvarez-Lerma et al., 2018b).
Recovery From Pneumonia
Recovery from pneumonia usually takes several weeks. At 4–6 weeks following discharge from hospital for an episode of CAP, one or more symptoms continue to be reported by 70% of patients, including cough, dyspnoea and fatigue in roughly equal proportions. Functional impairment is reported by 18–51% of patients at 4 weeks (Pick et al., 2019).
Cardiac complications (including heart failure, acute coronary syndrome and arrhythmias) have been observed in 18% of patients within 30 days following occurrence of CAP, with higher rates among patients who are hospitalized compared to those treated in the community (Corrales-Medina et al., 2011). The risk of cardiac complications remains high for at least the first 1–2 years following hospitalization (Corrales-Medina et al., 2015).
Higher rates of long-term mortality (17% at 1 year, 40% at 5 years) have been observed in patients following hospitalization for CAP compared to patients hospitalized for other reasons. The predominant causes of long-term mortality are vascular and respiratory in nature. Although studies have attempted to adjust for co-existing medical illnesses, it remains uncertain whether the association of pneumonia with long-term mortality is causal or whether pneumonic events are simply markers of poorer overall health (Wagenvoort et al., 2017).
A Need for Research
Many of the current treatment strategies for pneumonia have remained broadly unchanged over the last decade. Efforts at reducing the burden of disease through vaccination have been rewarded with some success but continued innovation is required to maintain gains made so far. The rise of antimicrobial resistance threatens our ability to treat infections that occur, urging a more judicious approach than the empirical antimicrobial strategies that characterize much of current clinical practice. The future expectation is a shift towards targeted treatments supported by rapid diagnostics, thus enabling the use of non-antibiotic pathogen-specific therapies (such as anti-toxins) and promising the eradiation of inappropriate antimicrobial use. Immune-modulating agents may offer further benefits in relation to short-term recovery, and improved post-pneumonia care could translate into better longer-term outcomes.
References
- Almirall J., Serra-Prat M., Bolibar I., Balasso V. Risk factors for community-acquired pneumonia in adults: A systematic review of observational studies. Respiration. 2017;94:299–311. doi: 10.1159/000479089. [DOI] [PubMed] [Google Scholar]
- Álvarez-Lerma F., Palomar-Martínez M., Sánchez-García M., et al. Prevention of Ventilator-Associated Pneumonia: The multimodal approach of the Spanish ICU “Pneumonia Zero” Program. Critical Care Medecine. 2018;46(2):181–188. doi: 10.1097/CCM.0000000000002736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Álvarez-Lerma F., Sanchez Garcia M., Task Force of Experts for Project “Zero VAP” in Spain “The multimodal approach for ventilator-associated pneumonia prevention”-requirements for nationwide implementation. Annals of Translational Medicine. 2018;6:420. doi: 10.21037/atm.2018.08.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aston S.J. Pneumonia in the developing world: Characteristic features and approach to management. Respirology. 2017;22:1276–1287. doi: 10.1111/resp.13112. [DOI] [PubMed] [Google Scholar]
- Beck J.M., Schloss P.D., Venkataraman A., Twigg H., 3rd, Jablonski K.A., Bushman F.D., Campbell T.B., Charlson E.S., Collman R.G., Crothers K., Curtis J.L., Drews K.L., Flores S.C., Fontenot A.P., Foulkes M.A., Frank I., Ghedin E., Huang L., Lynch S.V., Morris A., Palmer B.E., Schmidt T.M., Sodergren E., Weinstock G.M., Young V.B., Lung HIV Microbiome Project Multicenter comparison of lung and oral microbiomes of HIV-infected and HIV-uninfected individuals. American Journal of Respiratory and Critical Care Medicine. 2015;192:1335–1344. doi: 10.1164/rccm.201501-0128OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Claessens Y.E., Debray M.P., Tubach F., Brun A.L., Rammaert B., Hausfater P., Naccache J.M., Ray P., Choquet C., Carette M.F., Mayaud C., Leport C., Duval X. Early chest CT-scan to assist diagnosis and guide treatment decision for suspected community-acquired pneumonia. American Journal of Respiratory and Critical Care Medicine. 2015;192(8):974–982. doi: 10.1164/rccm.201501-0017OC. [DOI] [PubMed] [Google Scholar]
- Collaborators G.L. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory tract infections in 195 countries: A systematic analysis for the global burden of disease study 2015. The Lancet Infectious Diseases. 2017;17:1133–1161. doi: 10.1016/S1473-3099(17)30396-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corrales-Medina V.F., Suh K.N., Rose G., Chirinos J.A., Doucette S., Cameron D.W., Fergusson D.A. Cardiac complications in patients with community-acquired pneumonia: A systematic review and meta-analysis of observational studies. PLoS Medicine. 2011;8 doi: 10.1371/journal.pmed.1001048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corrales-Medina V.F., Alvarez K.N., Weissfeld L.A., Angus D.C., Chirinos J.A., Chang C.C., Newman A., Loehr L., Folsom A.R., Elkind M.S., Lyles M.F., Kronmal R.A., Yende S. Association between hospitalization for pneumonia and subsequent risk of cardiovascular disease. JAMA. 2015;313:264–274. doi: 10.1001/jama.2014.18229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daniel P., Bewick T., Welham S., Mckeever T.M., Lim W.S., British Thoracic S. Adults miscoded and misdiagnosed as having pneumonia: Results from the British Thoracic Society pneumonia audit. Thorax. 2017;72:376–379. doi: 10.1136/thoraxjnl-2016-209405. [DOI] [PubMed] [Google Scholar]
- Ewig S., Birkner N., Strauss R., Schaefer E., Pauletzki J., Bischoff H., Schraeder P., Welte T., Hoeffken G. New perspectives on community-acquired pneumonia in 388 406 patients. Results from a nationwide mandatory performance measurement programme in healthcare quality. Thorax. 2009;64:1062–1069. doi: 10.1136/thx.2008.109785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falkenhorst G., Remschmidt C., Harder T., Hummers-Pradier E., Wichmann O., Bogdan C. Effectiveness of the 23-valent pneumococcal polysaccharide vaccine (PPV23) against pneumococcal disease in the elderly: Systematic review and meta-analysis. PLoS One. 2017;12 doi: 10.1371/journal.pone.0169368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayes B.H., Haberling D.L., Kennedy J.L., Varma J.K., Fry A.M., Vora N.M. Burden of pneumonia-associated hospitalizations: United States, 2001-2014. Chest. 2018;153:427–437. doi: 10.1016/j.chest.2017.09.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jain S., Self W.H., Wunderink R.G., Fakhran S., Balk R., Bramley A.M., Reed C., Grijalva C.G., Anderson E.J., Courtney D.M., Chappell J.D., Qi C., Hart E.M., Carroll F., Trabue C., Donnelly H.K., Williams D.J., Zhu Y., Arnold S.R., Ampofo K., Waterer G.W., Levine M., Lindstrom S., Winchell J.M., Katz J.M., Erdman D., Schneider E., Hicks L.A., Mccullers J.A., Pavia A.T., Edwards K.M., Finelli L., CDC EPIC Study Team Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults. The New England Journal of Medicine. 2015;373:415–427. doi: 10.1056/NEJMoa1500245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalil A.C., Metersky M.L., Klompas M., Muscedere J., Sweeney D.A., Palmer L.B., Napolitano L.M., O’Grady N.P., Bartlett J.G., Carratala J., El Solh A.A., Ewig S., Fey P.D., File T.M., Jr., Restrepo M.I., Roberts J.A., Waterer G.W., Cruse P., Knight S.L., Brozek J.L. Management of Adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the infectious diseases Society of America and the American Thoracic Society. Clinical Infectious Diseases. 2016;63:e61–e111. doi: 10.1093/cid/ciw353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindenauer P.K., Lagu T., Shieh M.S., Pekow P.S., Rothberg M.B. Association of diagnostic coding with trends in hospitalizations and mortality of patients with pneumonia, 2003-2009. JAMA. 2012;307:1405–1413. doi: 10.1001/jama.2012.384. [DOI] [PubMed] [Google Scholar]
- Melsen W.G., Rovers M.M., Groenwold R.H., Bergmans D.C., Camus C., Bauer T.T., Hanisch E.W., Klarin B., Koeman M., Krueger W.A., Lacherade J.C., Lorente L., Memish Z.A., Morrow L.E., Nardi G., Van Nieuwenhoven C.A., O’Keefe G.E., Nakos G., Scannapieco F.A., Seguin P., Staudinger T., Topeli A., Ferrer M., Bonten M.J. Attributable mortality of ventilator-associated pneumonia: A meta-analysis of individual patient data from randomised prevention studies. The Lancet Infectious Diseases. 2013;13:665–671. doi: 10.1016/S1473-3099(13)70081-1. [DOI] [PubMed] [Google Scholar]
- Orso D., Guglielmo N., Copetti R. Lung ultrasound in diagnosing pneumonia in the emergency department: A systematic review and meta-analysis. European Journal of Emergency Medicine. 2018;25:312–321. doi: 10.1097/MEJ.0000000000000517. [DOI] [PubMed] [Google Scholar]
- Pick H.J., Bolton C.E., Lim W.S., Mckeever T.M. Patient reported outcome measures in the recovery of adults hospitalised with community-acquired pneumonia: A systematic review. The European Respiratory Journal. 2019;53(3) doi: 10.1183/13993003.02165-2018. pii: 1802165. [DOI] [PubMed] [Google Scholar]
- Quan T.P., Fawcett N.J., Wrightson J.M., Finney J., Wyllie D., Jeffery K., Jones N., Shine B., Clarke L., Crook D., Walker A.S., Peto T.E., Infections in Oxfordshire Research Database (IORD) Increasing burden of community-acquired pneumonia leading to hospitalisation, 1998-2014. Thorax. 2016;71(6):535–542. doi: 10.1136/thoraxjnl-2015-207688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rochwerg B., Oczkowski S.J., Siemieniuk R.A.C., Agoritsas T., Belley-Cote E., D’Aragon F., Duan E., English S., Gossack-Keenan K., Alghuroba M., Szczeklik W., Menon K., Alhazzani W., Sevransky J., Vandvik P.O., Annane D., Guyatt G. Corticosteroids in sepsis: An updated systematic review and meta-analysis. Critical Care Medicine. 2018;46:1411–1420. doi: 10.1097/CCM.0000000000003262. [DOI] [PubMed] [Google Scholar]
- Ruhnke G.W., Coca Perraillon M., Cutler D.M. Mortality reduction among pneumonia patients still substantial despite the impact of coding changes. The American Journal of Medicine. 2013;126:266–269. doi: 10.1016/j.amjmed.2012.08.006. [DOI] [PubMed] [Google Scholar]
- Saleh M.A.A., van de Garde E.M.W., van Hasselt J.G.C. Host-response biomarkers for the diagnosis of bacterial respiratory tract infections. Clinical Chemistry and Laboratory Medicine. 2018;57(4):442–451. doi: 10.1515/cclm-2018-0682. [DOI] [PubMed] [Google Scholar]
- Schuetz P., Wirz Y., Sager R., Christ-Crain M., Stolz D., Tamm M., Bouadma L., Luyt C.E., Wolff M., Chastre J., Tubach F., Kristoffersen K.B., Burkhardt O., Welte T., Schroeder S., Nobre V., Wei L., Bucher H.C., Bhatnagar N., Annane D., Reinhart K., Branche A., Damas P., Nijsten M., de Lange D.W., Deliberato R.O., Lima S.S., Maravic-Stojkovic V., Verduri A., Cao B., Shehabi Y., Beishuizen A., Jensen J.S., Corti C., van Oers J.A., Falsey A.R., de Jong E., Oliveira C.F., Beghe B., Briel M., Mueller B. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database of Systematic Reviews. 2017;10 doi: 10.1002/14651858.CD007498.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segal L.N., Methe B.A., Nolan A., Hoshino Y., Rom W.N., Dawson R., Bateman E., Weiden M.D. HIV-1 and bacterial pneumonia in the era of antiretroviral therapy. Proceedings of the American Thoracic Society. 2011;8:282–287. doi: 10.1513/pats.201006-044WR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sikka R., Tommaso L.H., Kaucky C., Kulstad E.B. Diagnosis of pneumonia in the ED has poor accuracy despite diagnostic uncertainty. The American Journal of Emergency Medicine. 2012;30:881–885. doi: 10.1016/j.ajem.2011.06.006. [DOI] [PubMed] [Google Scholar]
- Simou E., Britton J., Leonardi-Bee J. Alcohol and the risk of pneumonia: A systematic review and meta-analysis. BMJ Open. 2018;8 doi: 10.1136/bmjopen-2018-022344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith C.J., Kishore A.K., Vail A., Chamorro A., Garau J., Hopkins S.J., Di Napoli M., Kalra L., Langhorne P., Montaner J., Roffe C., Rudd A.G., Tyrrell P.J., van de Beek D., Woodhead M., Meisel A. Diagnosis of stroke-associated pneumonia: Recommendations from the pneumonia in Stroke Consensus Group. Stroke. 2015;46:2335–2340. doi: 10.1161/STROKEAHA.115.009617. [DOI] [PubMed] [Google Scholar]
- Stern A., Skalsky K., Avni T., Carrara E., Leibovici L., Paul M. Corticosteroids for pneumonia. Cochrane Database of Systematic Reviews. 2017;12 doi: 10.1002/14651858.CD007720.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi K., Suzuki M., Minh Le N., Anh N.H., Huong L.T., Son T.V., Long P.T., Ai N.T., Tho Le H., Morimoto K., Kilgore P.E., Anh D.D., Ariyoshi K., Yoshida L.M. The incidence and aetiology of hospitalised community-acquired pneumonia among Vietnamese adults: A prospective surveillance in Central Vietnam. BMC Infectious Diseases. 2013;13:296. doi: 10.1186/1471-2334-13-296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres A., Niederman M.S., Chastre J., Ewig S., Fernandez-Vandellos P., Hanberger H., Kollef M., Li Bassi G., Luna C.M., Martin-Loeches I., Paiva J.A., Read R.C., Rigau D., Timsit J.F., Welte T., Wunderink R. International ERS/ESICM/ESCMID/ALAT guidelines for the management of hospital-acquired pneumonia and ventilator-associated pneumonia: Guidelines for the management of hospital-acquired pneumonia (HAP)/ventilator-associated pneumonia (VAP) of the European Respiratory Society (ERS), European Society of Intensive Care Medicine (ESICM), European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and Asociación Latinoamericana del Tórax (ALAT) The European Respiratory Journal. 2017;50 doi: 10.1183/13993003.00582-2017. [DOI] [PubMed] [Google Scholar]
- Trotter C.L., Stuart J.M., George R.C., Miller E. Increasing hospital admission for pneumonia, England. Emerging Infectious Disease. 2008;14:727–733. doi: 10.3201/eid1405.071011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsaban G., Ben-Shimol S. Indirect (herd) protection, following pneumococcal conjugated vaccines introduction: A systematic review of the literature. Vaccine. 2017;35:2882–2891. doi: 10.1016/j.vaccine.2017.04.032. [DOI] [PubMed] [Google Scholar]
- van Vugt S.F., Verheij T.J., DE Jong P.A., Butler C.C., Hood K., Coenen S., Goossens H., Little P., Broekhuizen B.D., GRACE Project Group Diagnosing pneumonia in patients with acute cough: Clinical judgment compared to chest radiography. The European Respiratory Journal. 2013;42:1076–1082. doi: 10.1183/09031936.00111012. [DOI] [PubMed] [Google Scholar]
- Wagenvoort G.H., Sanders E.A., De Melker H.E., van der Ende A., Vlaminckx B.J., Knol M.J. Long-term mortality after IPD and bacteremic versus non-bacteremic pneumococcal pneumonia. Vaccine. 2017;35:1749–1757. doi: 10.1016/j.vaccine.2017.02.037. [DOI] [PubMed] [Google Scholar]
- Wunderink R.G., Waterer G. Advances in the causes and management of community acquired pneumonia in adults. BMJ. 2017;358:j2471. doi: 10.1136/bmj.j2471. [DOI] [PubMed] [Google Scholar]
- Yahiaoui R.Y., Bootsma H.J., Den Heijer C.D.J., Pluister G.N., John Paget W., Spreeuwenberg P., Trzcinski K., Stobberingh E.E. Distribution of serotypes and patterns of antimicrobial resistance among commensal Streptococcus pneumoniae in nine European countries. BMC Infectious Diseases. 2018;18:440. doi: 10.1186/s12879-018-3341-0. [DOI] [PMC free article] [PubMed] [Google Scholar]


