Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a member of the coronaviridae that causes respiratory disorders. After infection, large amounts of inflammatory cytokines are secreted, known as the cytokine storm. These cytokines can cause pulmonary damage induced by inflammation resulting in acute respiratory distress syndrome (ARDS), and even death.
One of the therapeutic approaches for treatment of ARDS is a mesenchymal stem cell (MSC). MSCs suppress inflammation and reduce lung injury through their immunomodulatory properties. MSCs also have the potential to prevent apoptosis of the lung cells and regenerate them. But our suggestion is using MSCs-derived exosomes. Because these exosomes apply the same immunomodulatory and tissue repair effects of MSCs and they don’t have problems associated to cell maintenance and injections.
For investigation the hypothesis, MSCs should be isolated from tissues and characterized. Then, the exosomes should be isolated from the supernatants and characterized. These exosomes should be injected into a transgenic animal for COVID-19. In the final section, lung function assessment, histological examination, micro-CT, differential leukocyte, viral load analysis, cytokine assay, and CRP level analysis can be investigated.
COVID-19 treatment is currently focused on supportive therapies and no vaccine has been developed for it. So, numerous researches are needed to find potential therapies. Since the pathogenesis of this disease was identified in previous studies and can cause lung injury with ARDS, investigation of the therapeutic approaches that can suppress inflammation, cytokine storm and ARDS can be helpful in finding a novel therapeutic approach for this disease.
Keywords: COVID-19, SARS-CoV-2, Acute respiratory distress syndrome, Mesenchymal stem cell, Exosomes
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a member of the family coronaviridae that, like two other members of the family, severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV), causes respiratory disorders [1], [2]. The virus was first detected in Wuhan, China, on December 2019, and the disease caused by the virus, coronavirus disease 2019 (COVID-19), was characterized as a pandemic on 11 March 2020 by the World Health Organization (WHO) [3], [4].
SARS-CoV-2 is an RNA virus and has a variable number of open reading frames (ORF) that can encode a number of structural proteins such as M (matrix), E (envelope), N (nucleocapsid) and S (spike) protein as well as a number of non-structural proteins. Among these structural proteins, the S protein binds to the angiotensin-converting enzymes 2 (ACE2) present on the surface of human cells, especially the alveolar type II cells that highly express this receptor, and then, the virus enters the host lung cells [5], [6]. Effective immune responses against viral infections depend on type I interferons (IFN-I) production from innate immune cells and subsequently cell mediated immunity through T helper 1 and cytotoxic T cells [5]. So, as the virus enters the cell, its genome is recognized by various pattern recognition receptors (PRRs) such as toll-like receptor-3 (TLR-3), TLR7-9, retinoic acid-inducible gene-I (RIG-I), Melanoma-differentiation-associated gene 5 (MDA5), and cyclic GMP-AMP synthase (cGAS), then, signaling pathways are initiated. Following these incidences, large amounts of IFN-I and inflammatory cytokines are secreted [7], [8], [9], [10]. Following these inflammatory conditions in the lungs, which also increase with the proliferation of the virus, a sharp increase in inflammatory cytokines such as interleukin-2 (IL-2), IL-7, IL-10, granulocyte-colony stimulating factor (G-CSF), interferon-γ-inducible protein 10 (IP-10), monocyte chemoattractant protein-1 (MCP-1), macrophage inflammatory protein- 1 alpha (MIP-1A), and tumor necrosis factor alpha (TNFα) have been reported in patients with COVID-19, known as the cytokine storm [11]. This cytokine storm is similar to that seen in SARS and MERS and is caused by the lack of proper regulation of inflammatory immune responses [12], [13].
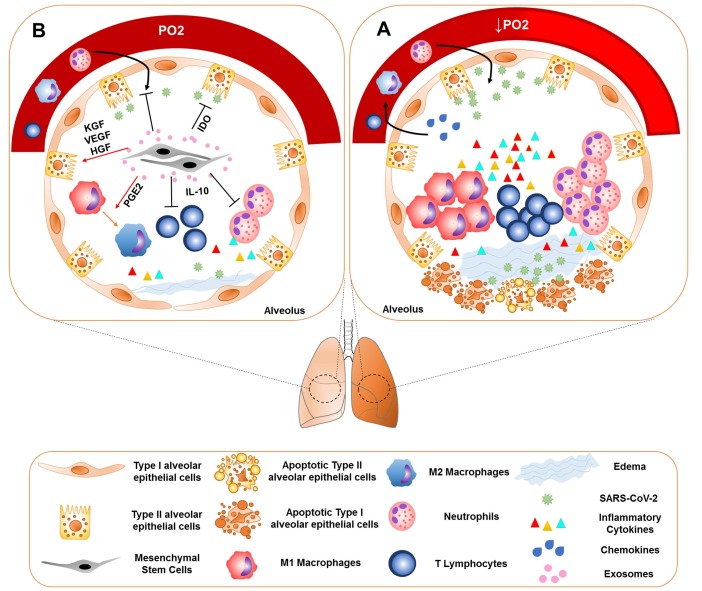
Clinical evidences demonstrated that this cytokine storm is the main cause of acute respiratory distress syndrome (ARDS) in COVID-19 [5], [12], [13], [14]. Pathogenic T cells produce inflammatory cytokines after activation and subsequently inflammatory monocytes and other leukocytes migrate to the lung [15]. Increased neutrophil counts have been reported as a symptom in a number of patients [2]. These activated immune cells may cause inflammation-induced pulmonary damage leading to complications such as pneumonia, loss of lung function, ARDS, and even death (Fig. 1 A) [5], [12], [13], [14]. Since ARDS is recognized as one of the leading causes of mortality in patients and appears to be caused by cytokine storm, suppression of this inflammation may decrease the inflammatory cytokine production and consequently reduce the inflammation and lung injury. As it has been shown, the use of anti-IL6 in patients with COVID-19-induced pneumonia can be useful and has been approved in China [16]. Currently, there is no approved treatment for SARS-CoV-2, and usually supportive treatments such as muscle relaxants and ventilators were used for these patients [6], [16], [17]. Numerous therapeutic interventions have been performed to treat ARDS, but since the mechanisms of damage in ARDS are complex and diverse, it seems that therapeutic strategies that target a single pathway or mediator can not be useful in achieving therapeutic outcome [17]. Therefore, interventions that target several aspects of immunopathogenesis and associated injuries, can be helpful to find the appropriate treatment strategy.
Fig. 1.
Immunopathogenesis of the SARS-CoV-2 and the therapeutic potential of the MSC exosomes. (A) SARS-CoV-2 infects type II alveolar epithelial cells or other target cells, which express ACE2. The influx of neutrophils, monocytes and T cells is induced by secretion of the chemokines. Accumulation of the inflammatory cells leads to the production of large amounts of pro-inflammatory cytokines, that is known as cytokine storm. Cytokine storm is the main cause of ARDS in COVID-19. These inflammatory responses may cause the apoptosis of the alveolar cells, lung fibrosis, edema, and organ failure. (B) MSCs and their exosomes suppress inflammation and reduce inflammation-induced lung injury through their immunomodulatory properties. These cells affect the macrophages and alter the phenotype of macrophages from M1 to M2 by releasing PG-E2. MSCs also reduce the production of the inflammatory cytokines and increase IL-10 production. IL-10 reduces neutrophils recruitment to the lungs. MSCs have also the tissue repair potential by producing KGF, VEGF, and HGF. These cells are involved in the pathogen clearance through expressing IDO.
Hypothesis
Numerous infectious agents, including bacteria (Streptococcus pneumoniae, Staphylococcus aureus, Hemophilus influenzae), viruses (SARS-CoV, MERS and SARS-CoV-2), fungi (Aspergillus species and Cryptococcus species), parasites (Pneumocystis jiroveci), as well as non-infectious agents such as pancreatitis, trauma, etc. can cause pneumonia and ARDS [18]. One of the therapeutic approaches for the treatment of ARDS is mesenchymal stem cell (MSC) therapy that previous studies have shown satisfactory results about it [19], [20], [21], [22], [23], [24]. MSCs are multipotent cells that can be obtained from different tissues such as, adipose tissue and bone marrow. These cells have a self-renewing capacity and can be differentiated into chondrogenic, osteogenic and adipogenic lineages [25]. Several features of these cells have made them an appealing candidate for the treatment of many diseases, such as ARDS.
-
•
Immunomodulatory properties: MSCs are able to alter the function of the immune cells and modulate the immune response [17]. For example, in LPS-induced ARDS animal models, it has been shown that, MSCs suppress inflammation and reduce inflammation-induced lung injury through their immunomodulatory properties. These cells affect the macrophages by releasing prostaglandin E2 (PG-E2), and reduce the production of inflammatory cytokines and increase IL-10 production. IL-10 reduced neutrophils recruitment to the lungs and attenuated inflammatory cytokine production [20], [22], [24]. MSCs can also increase regulatory T cells and alter the phenotype of macrophages from M1 to M2 [17].
-
•
Tissue repair characteristic: MSCs have also the tissue repair potential and can prevent apoptosis of the lung cells and regenerate them, especially type II alveolar cells, by producing growth factors such as keratinocyte growth factor (KGF), vascular endothelial growth factor (VEGF), and hepatocyte growth factor (HGF). This characteristic is important because people with ARDS develop pulmonary fibrosis even after surviving the acute phase of the disease [19], [21], [26]. MSCs can restore the epithelial protein permeability by secretion of angiopoietin-1. These cells can increase phagocytosis capacity of the macrophages by transferring the microvesicles [17].
-
•
Some studies shown that MSCs could imply anti-viral activity. It was shown that gammaherpesvirus-infected MSCs could limit the replication of the virus after sensing its DNA by cGAS and initiation of the STING-TBK1 signaling pathway. This pathway, that is resulted in IFN-γ production, along with the IFN-γ independent manner could be responsible for the anti-viral response of the MSCs [27]. In addition, some studies shown that IDO, which is expressed by MSCs, could be responsible for the anti-viral activity of the MSCs [28], [29], [30]. IDO-positive MSCs triggered by inflammatory cytokines demonstrated antimicrobial effector function against pathogens such as viruses [28]. In a study performed in 2011, it was shown that IFN-γ stimulated the expression of the IDO in MSCs and decreased the replication of the CMV and HSV-1 [29].
In summary, it can be suggested that MSCs can be attractive candidates for improving COVID-19-induced ARDS and lung injury, due to their immunomodulatory, tissue repair and anti-viral characteristics (Fig. 1B).
But our suggestion is using the MSC-derived exosomes. Because it has been shown that MSC-derived exosomes apply the same effects of MSCs [31]. Khatri et al. performed an in vitro study and demonstrated that the MSC-derived extracellular vesicles (MSC-EVs) transferred the mRNAs and miRNAs into the lung epithelial cells and reduced the apoptosis of these cells and inhibited influenza virus replication in them. For the confirmation of the results, they used a pig model of the influenza virus. It was shown that after MSC-EVs administration, the replication of the virus in the lung, pro-inflammatory cytokines production and virus-induced lung lesion were significantly decreased [32]. In a study performed in 2018, intravenous injection of MSC-exosomes reduced the TNF-α, IL-1β, NF-κB, and matrix metallopeptidase 9 in the lung. Another study mentioned that these exosomes could decrease endothelial cell apoptosis and IL-6 production. An increase in IL-10 production was also observed [33]. In addition to this, due to the specific structure of the exosomes, miRNAs as well as various drugs can be inserted into them. These miRNAs or drugs can target the specific molecules inside the infected cells and diminish the local inflammation or prevent apoptosis of the lung cells [34]. In a Song et al. study, it was showed that pre-treatment of the MSCs with IL-1β augmented their immunomodulatory effects, because their exosomes transfer the miR-146a to the target cells [35]. Other advantages of the exosomes include ease of access, lack of problems associated to cell maintenance and injections, proper size, and finally the nature of their phospholipid membranes, which enable them to fuse with the target cell membrane and transfer the cargo into the cytoplasm. Additionally, this special structure protects the contents of the exosomes from degradation [36].
Testing the hypothesis
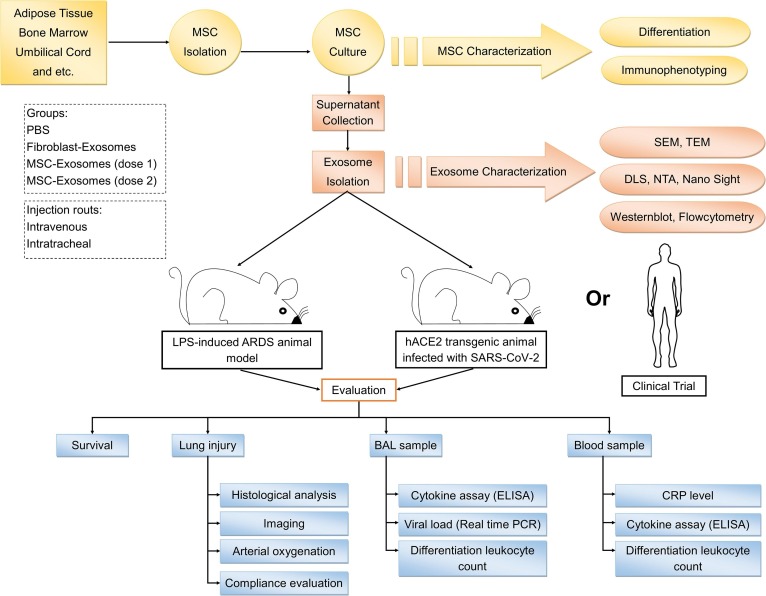
To investigate the effects of exosomes derived from MSCs, a study can be performed according to the following steps:
At the first section of the study, MSCs should be isolated from tissues. Different sources can be chosen such as adipose tissue, bone marrow, Wharton jelly, and etc. Second, these cells will be cultured. Then, the differentiation potential of homogenous population of spindle-shaped MSCs will be determined using in vitro adipogenic and osteogenic induction. In addition, the immunophenotype of MSCs will be confirmed by the measurement of cell surface markers such as CD73, CD90, CD105, CD14, CD45, CD34, and HLA-DR using flowcytometry. After the adaptation of the cells with serum-free medium, the supernatants of them can be collected. Then, a good manufacturing practice (GMP)-grade method should be used for the isolation of the exosomes from the supernatants, as a medicinal product [37]. The next step is the characterization of the exosomes. For this purpose, the size distribution of the isolated exosomes will be evaluated using dynamic light scattering (DLS) NanoSight, and nanoparticle tracking analysis (NTA). The morphology of these vesicles will be determined using scanning electron microscopy (SEM) and transmission electron microscopy (TEM) analysis. In addition, the expression of exosome membrane specific markers including CD9, CD63 and CD81 will be assessed using western blot or flowcytometry.
The second section of the study is related to animal model preparation and exosome injection to them. It is better to choose two ARDS models that are induced by different conditions and compare them with each other. For example, a hACE2 transgenic animal infected with SARS-CoV-2 [38] and an LPS-induced ARDS animal [24]. Next, the isolated exosomes should be injected into these animals. Intravenous or intratracheal injection can be performed. It is suggested to use both of these routes and compare them with each other. Injection program can be designed like this: PBS, exosomes derived from other sources such as fibroblasts, and MSC-exosomes (different doses).
In the final section of the study, the effects of exosome injection on different groups should be measured. Lung function assessment can be investigated through arterial oxygenation and static lung compliance evaluation [22]. In addition to these, histological examination or micro-CT can be done for assessment of the lung injury. Bronchoalveolar lavage (BAL) is an important sample for evaluating the effects of MSC-exorcisms' on lung inflammation. After collection of the BAL sample, differential leukocyte count can be done. For the monitoring of the viral load, real time PCR is a beneficial technique. For evaluating the cytokine profile, enzyme-linked immunosorbent assay (ELISA) can be useful. Another sample is the blood, that differential leukocyte count, inflammatory cytokines and C-reactive protein (CRP) level are the important data for evaluating MSC-exosomes effects on ARDS and cytokine storm. All of these procedures can be performed in clinical trial studies after confirmation in animal models (Fig. 2 ).
Fig. 2.
Testing the hypothesis. MSC, mesenchymal stem cell; SEM, scanning electron microscopy; TEM, transmission electron microscopy; DLS, dynamic light scattering. NTA, nanoparticle tracking analysis; ARDS, acute respiratory distress syndrome; BAL, bronchoalveolar lavage; CRP, C-reactive protein; ELISA, enzyme-linked immunosorbent assay.
Discussion
Since COVID-19 treatment is currently focused on supportive therapies and no vaccine has been developed for it [6], numerous research studies are needed to find potential therapies to control the virus as soon as possible. Because SARS-CoV-2 has a high transmission potential and spreads rapidly throughout the world and causes many deaths. Since the pathogenesis of this disease was identified partially in previous studies and there are similarities between its pathogenesis with those of other viruses in this family and they can cause lung injury with ARDS [1], [5], [14], investigation of the therapeutic approaches that can suppress inflammation, cytokine storm and ARDS can be helpful in finding a novel therapeutic approach for this disease. Therefore, the use of MSCs as one of the immunomodulating and tissue regenerating cells that have previously shown satisfactory effects on ARDS and cytokine storms [19], [21], [22], [23], [24], [39] could be considered. Since these cells exert their effects in a variety of ways, such as exosomes secretion, the use of their exosomes can be considered as a cell-free therapy [31], because these exosomes can have anti-inflammatory effects on lung tissue by transferring mRNAs, miRNAs, and various proteins from its secretory cell to the target cell [40]. As previously shown, these exosomes can reduce inflammatory cytokines, increase regulatory cytokines and suppress inflammation [34]. Monsel et al. also showed that MSC-exosomes could increase the survival of alveolar type II cells by increasing intracellular ATP levels [41]. So we introduced this hypothesis in order to perform further studies in the future and hope to see satisfactory results.
Financial support
No financial support was used for the development or writing of this manuscript.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Yin Y., Wunderink R.G. MERS, SARS and other coronaviruses as causes of pneumonia. Respirology. 2018;23:130–137. doi: 10.1111/resp.13196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen N., Zhou M., Dong X., Qu J., Gong F., Han Y. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.World Health Organization. Rolling updates on coronavirus disease (COVID-19) [Internet] 2020 [cited 2020 May 3]. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen.
- 4.Phelan A.L., Katz R., Gostin L.O. The novel coronavirus originating in Wuhan, China: challenges for global health governance. JAMA – J Am Med Assoc. 2020;323:709–710. doi: 10.1001/jama.2020.1097. [DOI] [PubMed] [Google Scholar]
- 5.Prompetchara E., Ketloy C., Palaga T. Immune responses in COVID-19 and potential vaccines: Lessons learned from SARS and MERS epidemic. Asian Pacific J Allergy Immunol. 2020;38:1–9. doi: 10.12932/AP-200220-0772. [DOI] [PubMed] [Google Scholar]
- 6.Li X., Geng M., Peng Y., Meng L., Lu S. Molecular immune pathogenesis and diagnosis of COVID-19. J Pharm Anal. 2020;19:1–7. doi: 10.1016/j.jpha.2020.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wu J., Chen Z.J. Innate immune sensing and signaling of cytosolic nucleic acids. Annu Rev Immunol. 2014;32:461–488. doi: 10.1146/annurev-immunol-032713-120156. [DOI] [PubMed] [Google Scholar]
- 8.Wu J., Sun L., Chen X., Du F., Shi H., Chen C. Cyclic GMP-AMP is an endogenous second messenger in innate immune signaling by cytosolic DNA. Science. 2013;339(6121):826–830. doi: 10.1126/science.1229963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Takeuchi O., Akira S. Innate immunity to virus infection. Immunol Rev. 2009;227:75–86. doi: 10.1111/j.1600-065X.2008.00737.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Guo Y.-R., Cao Q.-D., Hong Z.-S., Tan Y.-Y., Chen S.-D., Jin H.-J. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak - an update on the status. Mil Med Res. 2020;7:11. doi: 10.1186/s40779-020-00240-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;6736:1–9. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mahallawi W.H., Khabour O.F., Zhang Q., Makhdoum H.M., Suliman B.A. MERS-CoV infection in humans is associated with a pro-inflammatory Th1 and Th17 cytokine profile. Cytokine. 2018;104:8–13. doi: 10.1016/j.cyto.2018.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wong C.K., Lam C.W.K., Wu A.K.L., Ip W.K., Lee N.L.S., Chan I.H.S. Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome. Clin Exp Immunol. 2004;136:95–103. doi: 10.1111/j.1365-2249.2004.02415.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nicholls J.M., Poon L.L., Lee K.C. Lung pathology of fatal severe acute respiratory syndrome. Lancet. 2003;361(9371):1773–1778. doi: 10.1016/s0140-6736(03)13413-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhou Y.F., Zheng X., Wang D., Zhao C., Qi Y., Sun R. Aberrant pathogenic GM-CSF T cells and inflammatory CD14CD16 monocytes in severe pulmonary syndrome patients of a new coronavirus. BioRxiv. 2020 doi: 10.1101/2020.02.12.945576. [DOI] [Google Scholar]
- 16.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Laffey J.G., Matthay M.A. Cell-based therapy for acute respiratory distress syndrome: Biology and potential therapeutic value. Am J Respir Crit Care Med. 2017;196:266–273. doi: 10.1164/rccm.201701-0107CP. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee K.Y. Pneumonia acute respiratory distress syndrome, and early immune-modulator therapy. Int J Mol Sci. 2017;18 doi: 10.3390/ijms18020388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hu S., Li J., Xu X., Liu A., He H., Xu J. The hepatocyte growth factor-expressing character is required for mesenchymal stem cells to protect the lung injured by lipopolysaccharide in vivo. Stem Cell Res Ther. 2016;7:1–13. doi: 10.1186/s13287-016-0320-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gupta N., Su X., Popov B., Lee J.W., Serikov V., Matthay M.A. Intrapulmonary delivery of bone marrow-derived mesenchymal stem cells improves survival and attenuates endotoxin-induced acute lung injury in mice. J Immunol. 2007;179:1855–1863. doi: 10.4049/jimmunol.179.3.1855. [DOI] [PubMed] [Google Scholar]
- 21.Yang Y., Hu S., Xu X., Li J., Liu A., Han J. The vascular endothelial growth factors-expressing character of mesenchymal stem cells plays a positive role in treatment of acute lung injury in vivo. Mediators Inflamm. 2016;2016 doi: 10.1155/2016/2347938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Devaney J., Horie S., Masterson C., Elliman S., Barry F., O’Brien T. Human mesenchymal stromal cells decrease the severity of acute lung injury induced by E. Coli in the rat. Thorax. 2015;70:625–635. doi: 10.1136/thoraxjnl-2015-206813. [DOI] [PubMed] [Google Scholar]
- 23.Curley GF, Jerkic M, Dixon S, Hogan G, Masterson C, O’toole D, et al. Cryopreserved, xeno-free human umbilical cord mesenchymal stromal cells reduce lung injury severity and bacterial burden in rodent Escherichia coli -induced acute respiratory distress syndrome. Crit Care Med 2017;45:e202–12. https://doi.org/10.1097/CCM.0000000000002073. [DOI] [PubMed]
- 24.Lee J.W., Fang X., Gupta N., Serikov V., Matthay M.A. Allogeneic human mesenchymal stem cells for treatment of E. coli endotoxin-induced acute lung injury in the ex vivo perfused human lung. Proc Natl Acad Sci U S A. 2009;106:16357–16362. doi: 10.1073/pnas.0907996106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Taghavi-Farahabadi M., Mahmoudi M., Hashemi S.M., Rezaei N. Evaluation of the effects of mesenchymal stem cells on neutrophils isolated from severe congenital neutropenia patients. Int Immunopharmacol. 2020;83 doi: 10.1016/j.intimp.2020.106463. [DOI] [PubMed] [Google Scholar]
- 26.Harrell C.R., Sadikot R., Pascual J., Fellabaum C., Jankovic M.G., Jovicic N. Mesenchymal Stem Cell-Based Therapy of Inflammatory Lung Diseases: Current Understanding and Future Perspectives. Stem Cells Int. 2019;2019 doi: 10.1155/2019/4236973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yang K., Wang J., Wu M., Li M., Wang Y., Huang X. Mesenchymal stem cells detect and defend against gammaherpesvirus infection via the cGAS-STING pathway. Sci Rep. 2015;5:1–9. doi: 10.1038/srep07820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Meisel R., Zibert A., Laryea M., Göbel U., Däubener W., Dilloo D. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood. 2004;103:4619–4621. doi: 10.1182/blood-2003-11-3909. [DOI] [PubMed] [Google Scholar]
- 29.Meisel R., Brockers S., Heseler K., Degistirici O., Bülle H., Woite C. Human but not murine multipotent mesenchymal stromal cells exhibit broad-spectrum antimicrobial effector function mediated by indoleamine 2,3-dioxygenase. Leuk Off J Leuk Soc Am Leuk Res Fund, UK. 2011;25:648–654. doi: 10.1038/leu.2010.310. [DOI] [PubMed] [Google Scholar]
- 30.Thanunchai M, Hongeng S, Thitithanyanont A. Mesenchymal Stromal cells and viral infection. Stem Cells Int 2015;2015. https://doi.org/10.1155/2015/860950. [DOI] [PMC free article] [PubMed]
- 31.Mahmoudi M., Taghavi Farahabadi M., Hashemi S.M. Exosomes: Mediators of Immune Regulation. Immunoregulation. 2018:121–126. doi: 10.32598/Immunoregulation.1.3.121. [DOI] [Google Scholar]
- 32.Khatri M., Richardson L.A., Meulia T. Mesenchymal stem cell-derived extracellular vesicles attenuate influenza virus-induced acute lung injury in a pig model. Stem Cell Res Ther. 2018;9:1–13. doi: 10.1186/s13287-018-0774-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lo Chang C, Chen H.H., Chen K.H., Chiang J.Y., Li Y.C., Lin H.S. Adipose-derived mesenchymal stem cell-derived exosomes markedly protected the brain against sepsis syndrome induced injury in rat. Am J Transl Res. 2019;11:3955–3971. [PMC free article] [PubMed] [Google Scholar]
- 34.Shah T.G., Predescu D., Predescu S. Mesenchymal stem cells-derived extracellular vesicles in acute respiratory distress syndrome: a review of current literature and potential future treatment options. Clin Transl Med. 2019;8:4–9. doi: 10.1186/s40169-019-0242-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Song Y., Dou H., Li X., Zhao X., Li Y., Liu D. Exosomal miR-146a Contributes to the Enhanced Therapeutic Efficacy of Interleukin-1β-Primed Mesenchymal Stem Cells Against Sepsis. Stem Cells. 2017;35:1208–1221. doi: 10.1002/stem.2564. [DOI] [PubMed] [Google Scholar]
- 36.Stremersch S., De Smedt S.C., Raemdonck K. Therapeutic and diagnostic applications of extracellular vesicles. J Control Release. 2016;244:167–183. doi: 10.1016/j.jconrel.2016.07.054. [DOI] [PubMed] [Google Scholar]
- 37.Mendt M., Kamerkar S., Sugimoto H., McAndrews K.M., Wu C.C., Gagea M. Generation and testing of clinical-grade exosomes for pancreatic cancer. JCI. Insight. 2018;3 doi: 10.1172/jci.insight.99263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bao L., Deng W., Huang B., Gao H., Liu J., Ren L. The Pathogenicity of SARS-CoV-2 in hACE2 Transgenic Mice. BioRxiv. 2020;2020(02) doi: 10.1101/2020.02.07.939389. [DOI] [PubMed] [Google Scholar]
- 39.Chen J., Hu C., Chen L., Tang L., Zhu Y., Xu X. Clinical study of mesenchymal stem cell treatment for acute respiratory distress syndrome induced by Epidemic Influenza A (H7N9) infection: a hint for COVID-19 treatment. Engineering. 2020 doi: 10.1016/j.eng.2020.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.El Andaloussi S., Mäger I., Breakefield X.O., Wood M.J.A., EL Andaloussi S., Mäger I. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013;12:347–357. doi: 10.1038/nrd3978. [DOI] [PubMed] [Google Scholar]
- 41.Monsel A., Zhu Y.G., Gennai S., Hao Q., Hu S., Rouby J.J. Therapeutic effects of human mesenchymal stem cell-derived microvesicles in severe pneumonia in mice. Am J Respir Crit Care Med. 2015;192:324–336. doi: 10.1164/rccm.201410-1765OC. [DOI] [PMC free article] [PubMed] [Google Scholar]