Abstract
Purpose
To date, research characterizing swallowing changes in individuals with amyotrophic lateral sclerosis (ALS) has primarily relied on subjective descriptions. Thus, the degree to which swallowing physiology is altered in ALS, and relationships between such alterations and swallow safety and/or efficiency are not well characterized. This study provides a quantitative representation of swallow physiology, safety, and efficiency in a sample of individuals with ALS, to estimate the degree of difference in comparison to published healthy reference data and identify parameters that pose risk to swallow safety and efficiency. Secondary analyses explored the therapeutic effect of thickened liquids on swallowing safety and efficiency.
Method
Nineteen adults with a diagnosis of probable-definite ALS (El-Escorial Criteria–Revised) underwent a videofluoroscopic swallowing study, involving up to 15 sips of barium liquid (20% w/v), ranging in thickness from thin to extremely thick. Blinded frame-by-frame videofluoroscopy analysis yielded the following measures: Penetration–Aspiration Scale, number of swallows per bolus, amount of pharyngeal residue, degree of laryngeal vestibule closure (LVC), time-to-LVC, duration of LVC (LVCdur), pharyngeal area at maximum constriction, diameter of upper esophageal sphincter opening, and duration of UES opening (UESOdur). Measures of swallow physiology obtained from thin liquid trials were compared against published healthy reference data using unpaired t tests, chi-squared tests, and Cohen’s d effect sizes (adjusted p < .008). Preliminary relationships between parameters of swallowing physiology, safety, and efficiency were explored using nonparametric Cochrane’s Q, Friedman’s test, and generalized estimating equations (p < .05).
Results
Compared to healthy reference data, this sample of individuals with ALS displayed a higher proportion of swallows with partial or incomplete LVC (24% vs. < 1%), increased time-to-LVC (d = 1.09), reduced UESwidth (d = 0.59), enlarged pharyngeal area at maximum constriction, prolonged LVCdur (d = 0.64), and prolonged UESOdur (d = 1.34). Unsafe swallowing (i.e., PAS ≥ 3) occurred more frequently when LVC was partial/incomplete or time-to-LVC was prolonged. Pharyngeal residue was associated with larger pharyngeal areas at maximum constriction. Unsafe swallowing occurred less frequently with extremely thick liquids, compared to thin liquids. No significant differences in pharyngeal residue were observed based on liquid thickness.
Conclusions
Quantitative videofluoroscopic measurements revealed moderate-to-large differences in swallow physiology between this sample of individuals with ALS and healthy reference data. Increased time-to-LVC, noncomplete LVC, and enlarged pharyngeal area at maximum constriction were associated with impaired swallow safety or efficiency. Thickened liquids may mitigate the risk of acute episodes of aspiration in individuals with ALS. Further work is needed to corroborate these preliminary findings and explore how swallowing profiles evolve throughout disease progression.
amyotrophic lateral sclerosis (ALS) is a neurodegenerative condition characterized by progressive loss of upper and lower motor neurons, leading to muscle weakness throughout the body and loss of motor function. Although bulbar dysfunction is seen at disease onset for approximately 30% of individuals with ALS, more than 80% of individuals with ALS will experience bulbar dysfunction at some point in the disease process, including dysarthria and dysphagia (e.g., Briani et al., 1998; Makkonen et al., 2017; Ruoppolo et al., 2013). For many individuals, ALS progresses quickly, with average survival being 2–5 years following diagnosis (Pupillo et al., 2014; Talbot, 2009). Thus, timely identification of management needs and counseling toward supports/strategies (e.g., assistive devices, enteral nutrition) are of high importance.
Dysphagia in ALS has been characterized by changes in all stages of swallowing, which become progressively worse over time and contribute to reduced quality of life as well as impairments in swallowing safety and efficiency (e.g., Paris et al., 2012; Tabor et al., 2016; Tabor & Plowman, 2017; Waito et al., 2017). Identifying risk of dysphagia and addressing swallowing impairments as early as possible are imperative, as respiratory infections and malnutrition are linked to a heightened risk of death in individuals with ALS (e.g., Corcia et al., 2008; Desport et al., 1999). Based on current descriptions, a variety of swallow-related changes can be anticipated by clinicians and monitored over time, including (but not limited to) aspiration, pharyngeal residue, piecemeal swallowing, impaired closure of the laryngeal vestibule, reduced opening of the upper esophageal sphincter (UES), and impaired pharyngeal pressure/constriction (e.g., Higo et a., 2004; Lo Re et al., 2007; Robbins, 1987; Ruoppolo et al., 2013). However, our current understanding of these differences is primarily based upon subjective and vague descriptions of swallowing behavior and physiology (e.g., “impaired” tongue function, “reduced” hyolaryngeal excursion; Waito et al., 2017). Few studies have objectively measured swallowing physiology and function in individuals with ALS, leaving consulting clinicians with a challenge to detect and monitor subtle changes over time and estimate the degree of difference that indicates a risk to swallowing safety and/or efficiency in this clinical population.
In order to predict functional impairments and provide counseling toward long-term management strategies, clinicians must be equipped to identify differences at early disease stages and track those changes over time. In current practice, bulbar impairment (including dysphagia) in ALS is typically identified based on clinical evaluation (e.g., cranial nerve exam, clinical swallow assessment) and patient-reported symptoms using the ALS Functional Rating Scale–Revised (ALSFRS-R; Cedarbaum et al., 1999) to determine areas of impairment (Plowman et al., 2017). However, these approaches have their limitations. Clinical evaluations, including the cranial nerve exam, for example, have been criticized for being unstandardized and subjective (Yunusova et al., 2019). Furthermore, the ALSFRS-R evaluates symptoms of bulbar function based on a patient self-report questionnaire, which may not be sensitive to detect subtle changes in bulbar function (Green et al., 2013); in fact, measurable kinematic changes in the bulbar system have been shown to precede clinician judgments and patient reports of bulbar dysfunction (Allison et al., 2017), and identification of additional physiological markers that are able to detect early or subtle disease-related changes is a top priority in ALS research (Green et al., 2013; Turner et al., 2009). Although a great deal of work has been done to measure kinematic and temporal properties of speech and chewing (e.g., Shellikeri et al., 2016; Wilson et al., 2019; Yunusova et al., 2010), measurement of oropharyngeal swallowing physiology in individuals with ALS has received less attention.
Recent studies have begun measuring swallow physiology from videofluoroscopic swallowing studies to understand biomechanical and functional changes in swallowing related to ALS. Waito, Tabor-Gray, Steele, & Plowman (2018) measured maximum pharyngeal constriction in a sample of individuals with ALS and identified enlarged pharyngeal areas at maximum constriction, associated with swallow inefficiency. In a related study, the authors identified a reduction in anterior hyoid movement speed and distance during the swallow despite superior movement being relatively maintained (Waito, Peladeau-Pigeon, et al., 2018). These studies have expanded our understanding of the degree of change observed in individuals with ALS, pointing to potential mechanisms of unsafe or inefficient swallowing. However, a limitation to each of these studies is that they focused narrowly on a single physiological parameter. As the swallow is dynamic in nature, a more comprehensive picture of concurrent swallowing changes in individuals with ALS is needed.
Garand et al. (2018) used computational analysis of swallowing mechanics to evaluate swallowing movements more globally in a small sample of patients with ALS, comparing results to a neurotypical control group. The authors identified gestalt differences in swallow mechanics between the two groups, with specific changes including reduced tongue base retraction, reduced pharyngeal shortening, and increased hyolaryngeal excursion. However, computational of swallowing mechanics analysis reports results in terms of canonical variates, rather than raw measures of timing/kinematics, leaving the values inaccessible for clinicians to interpret and replicate. Similarly, measures of swallowing safety and efficiency were not reported in this study, making it difficult to understand the functional impact of the identified differences. The authors also note a great deal of heterogeneity in swallowing mechanics, suggesting additional physiological alterations may have gone undetected by the group-level analysis.
The current study aims to build on this previous work by expanding the quantification of swallow mechanics in individuals of ALS, capturing multiple features, and using metrics that are clinically interpretable and reported in relation to disease severity and swallowing ability. Following a hypothesis-driven approach, we selected six physiological measures involved in airway protection and bolus flow that have been previously described as impaired in individuals with ALS and/or are known to pose risk to swallowing safety or efficiency (see Method section for discussion of specific measures). Specifically, for Aim 1, we examined differences in physiological parameters of swallowing, comparing individuals with ALS to published healthy reference values (in Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019). We hypothesized that we would find large group differences, based on estimates of effect size (Vacha-Haase & Thompson, 2004), including increased time intervals between events (i.e., slowed movement), shortened event durations, and reduced movement of key swallowing structures. Furthermore, we hypothesized that individuals with ALS who report functional bulbar impairment on the ALSFRS-R (with or without symptoms of dysphagia) would present with measures of swallowing that fall outside the range of healthy reference values.
In addition to detecting differences from neurotypical controls, measures that can provide meaningful information related to dysphagia risk (e.g., unsafe or inefficient swallowing) are of high clinical value. Thus, for Aim 2, we performed a preliminary exploration to identify which of the selected measures (if any) were most associated with impaired safety or efficiency in this group. For this, we expected to find mechanisms of airway protection (e.g., timeliness, degree, and duration of airway closure) to be associated with swallow safety, while mechanisms of bolus clearance (e.g., pharyngeal constriction, UES opening) would be associated with swallow efficiency.
A secondary aim of this study was to provide additional guidance on the role of thickened liquids for dysphagia management in individuals with ALS. Although thickened fluids are discussed as a potential management option for individuals with ALS, few studies have empirically studied the ability of thickened liquids to reduce the occurrence of aspiration in this clinical population (Waito et al., 2017). Further complicating the therapeutic effect of thickened liquids in individuals with ALS are reports that thickened liquids may lead to increased pharyngeal residue, particularly in those with reduced pharyngeal pressure or constriction (Hind et al., 2012; Oliveira et al., 2017; Waito, Tabor-Gray, et al., 2018). Thus, Aim 3 for this study tested the effect of bolus thickness on acute swallowing safety and impact (if any) on efficiency.
Method
Data Collection
This study was reviewed and approved by research ethics boards, and all participants provided written informed consent prior to enrollment. Twenty individuals with a diagnosis of definite-probable ALS (El Escorial criteria–revised; Brooks et al., 2000) were recruited for this study, following a convenience sampling paradigm as part of a larger group of individuals with neurodegenerative conditions. Patients with and without subjective bulbar symptoms were eligible to participate, as recent research has demonstrated that individuals with ALS may present with kinematic changes that precede patient-reported symptoms (Allison et al., 2017). However, to explore preliminary mechanisms of swallow safety and efficiency (Aim 2), it was necessary for us to recruit a range of swallow safety/efficiency profiles; thus, patients who endorsed bulbar symptoms were preferentially approached for recruitment. Exclusionary criteria included a medical history or cancer, surgery, or radiation in the head and neck region; history of dysphagia prior to diagnosis of ALS; diagnosis of comorbid neuromuscular or neurological disorder (e.g., Parkinson's disease, stroke); and/or evidence of severe cognitive impairment (screened informally; e.g., unable to follow test instructions) or respiratory compromise (e.g., reliance on invasive mechanical ventilation).
A selection of demographic information was recorded or obtained from chart review, including year of birth, sex, diagnosis date (month, year), ALS onset type (i.e., bulbar, spinal, mixed), and time since onset of initial and bulbar-specific symptoms. To obtain functional severity scores, each participant completed the ALSFRS-R (Cedarbaum et al., 1999 on the day of their visit.
Each participant underwent a standardized videofluoroscopy exam seated upright in a lateral viewing plane using a properly collimated Philips BV Endura fluoroscopic C-arm unit (GE OEC 8800 Digital Mobile C-Arm System, Type 718074). Videofluoroscopies were performed using continuous fluoroscopy and recorded on a TIMS DICOM System (TIMS Medical by Foresight Imaging LLC) capturing at 30 frames per second. Participants were instructed to take comfortable sips of thin, slightly thick, and mildly thick liquids, as well as comfortable teaspoons of moderately and extremely thick liquids, defined by the International Dysphagia Diet Standardisation Initiative (IDDSI) framework (Cichero et al., 2017). Each cup was weighed before and after the participant took a sip to obtain measures of bolus weight, which were converted to bolus volume using published conversions (Steele, Peladeau-Pigeon, Barbon, Guida, Tapson, et al., 2019). All liquids were mixed following standardized recipes (Barbon & Steele, 2018) to a 20% weight-to-volume concentration of barium contrast (Bracco Diagnostics E-Z-PAQUE barium sulfate) and thickened with a xanthan gum thickener (Nestlé Resource ThickenUp Clear). Boluses were presented from thinnest (IDDSI Level 0) to thickest (IDDSI Level 4), and each participant was given three trials of each consistency, yielding up to 15 boluses, as tolerated.
For participant safety, we implemented the following bailout criteria: (a) If aspiration or penetration leaving residue in the laryngeal vestibule was observed on two trials of the same stimulus, the remaining trial of that stimulus was omitted and the examiner moved on to the next (thicker) IDDSI level; (b) if aspiration or penetration leaving residue in the laryngeal vestibule was observed on three occasions overall, or if severe levels of pharyngeal residue were observed and persisted despite cued compensatory maneuvers, the protocol was terminated and the patient was provided with standard management care.
Videofluoroscopy Ratings
Bolus trials were saved as separate video clips (one bolus, per video—including all clearing swallows associated with that bolus) and randomized for blinded rating. Each bolus clip was rated according to the Analysis of Swallowing Physiology: Events, Kinematics & Timing method to obtain measures of swallow safety and efficiency and quantify temporal and kinematic parameters of swallowing physiology (Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019). Following a hypothesis-driven approach, we purposefully selected parameters that we expected to illustrate change based either on previous descriptive accounts of swallowing in people with ALS (e.g., Ertekin et al., 2000; Higo et al., 2004; Lo Re et al., 2007; Murono et al., 2015; Robbins, 1987) or associations with swallowing function reported in other clinical populations (e.g., Leonard et al., 2004; Macrae et al., 2014; Molfenter et al., 2011; Molfenter & Steele, 2014; Stokely, Peladeau-Pigeon, Leigh, et al., 2015; Waito, Steele, et al., 2018), including:
time-to-laryngeal vestibule closure (time-to-LVC; notated as “laryngeal vestibule closure reaction time” in Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019),
degree of LVC,
duration of laryngeal vestibule closure (LVCdur),
pharyngeal area at maximum constriction,
UES opening duration (UESOdur), and
diameter of UES opening (UESMax).
The parameters of interest used in this study are summarized in Table 1. For additional details and complete operational definitions, please refer to the full protocol published by Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019.
Table 1.
Parameters of swallowing physiology and function included in the current study
| Category | Parameter | Acronym | Metric | Definition a |
|---|---|---|---|---|
| Safety | Penetration–Aspiration Scale | PAS | Nominal Scale | 8-point scale characterizing depth of and response to penetration–aspiration b |
| Efficiency | Number of swallow events | Count | The number of swallows generated for a single bolus (including attempted swallows where the UES failed to open) | |
| Vallecular residue | % C2–42 | Normalized pixel-based measure of residue located in the vallecular space | ||
| Pyriform sinus residue | % C2–42 | Normalized pixel-based measure of residue located in the pyriform sinuses | ||
| Total pharyngeal residue | % C2–42 | Normalized pixel-based measure of all residue located in the pharynx | ||
| Timing measures | Time-to-laryngeal vestibule closure | Time-to-LVC | ms | Interval from onset of hyoid burst until closure of the laryngeal vestibule |
| Laryngeal vestibule closure duration | LVCdur | ms | Interval from closure of the laryngeal vestibule until opening | |
| Upper esophageal sphincter opening duration | UESOdur | ms | Interval from onset of UES opening until first point of UES closure observed | |
| Degree of movement | Upper esophageal sphincter diameter | UESMax | % C2–4 | Normalized pixel-based measure of UES diameter at maximum distension during the swallow |
| Maximum pharyngeal constriction area | % C2–42 | Normalized pixel-based measure of pharyngeal area at maximum constriction during the swallow | ||
| Laryngeal vestibule closure | LVC | Nominal Scale | Degree of airway closure (three levels): complete, partial, and incomplete |
Note. PAS = Penetration–Aspiration Scale; UES = upper esophageal sphincter; LVC = laryngeal vestibule closure; LVCdur = duration of laryngeal vestibule closure; UESOdur = UES opening duration UESMax = diameter of UESopening.
For full description of the Analysis of Swallowing Physiology: Events, Kinematics & Timing method and operational definitions of each parameter included in the current study, please refer to Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al. (2019).
Source: Rosenbek et al. (1996).
All videofluoroscopic ratings were completed by trained and experienced raters, with 100% duplication (i.e., two raters) for evaluations of swallowing safety and event frame selection and 70% duplication for pixel-based measures of residue, UESMax, and maximum pharyngeal constriction area. Discrepancies between raters, which exceeded a priori thresholds (see Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019), were resolved through consensus. This included any difference in Penetration–Aspiration Scale (PAS; Rosenbek et al., 1996) score, LVC rating, or the number of swallow events; frame selections that were > 5 frames apart; and pixel-based measures that exceeded the following calculated cutoffs (in C2–4 units): maximum pharyngeal constriction area > 0.052, UESMax > 0.101, and total pharyngeal residue > 0.088.
Statistical Analysis
All statistical analyses were conducted using IBM SPSS Statistics Version 24 (IBM Corporation, 2016).
Descriptive data for all parameters were calculated at the bolus level to characterize the participant sample. As certain measures of swallow timing and kinematics are sensitive to changes in bolus volume (e.g., Chi-Fishman & Sonies, 2002; Hoffman et al., 2010; Kahrilas et al., 1996), physiological parameters were summarized based on the first swallow per trial to minimize bolus volume variability in clearing swallows. LVC timing (i.e., time-to-LVC and LVCdur) for swallows with partial or incomplete LVC was referenced to the frame of maximum arytenoid-to-epiglottic contact and analyzed separately from swallows with complete LVC.
Aim 1: We compared average measures of swallowing physiology from thin liquids against reference data from a recent report of healthy adults (< aged 60 years), matched by task (i.e., 20% w/v barium concentration; uncued swallows, “comfortable” sip volume; see Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019). Group comparisons were made using independent-samples t tests and chi-squared tests. For measures that differed significantly from reference data, we calculated Cohen's d as an estimate of the magnitude of difference between groups (Schuele & Justice, 2006; Vacha-Haase & Thompson, 2004). Statistical significance was set at p < .008 to adjust for multiple (six) comparisons.
At the participant level, the proportion of thin liquid trials that fell outside the healthy range was calculated. Measures from each thin liquid trial were compared against the reference data mean and standard deviation, applying a threshold of > 2 SDs above or below the reference mean, consistent with reference limits defined in medical laboratory tests (e.g., Ceriotti et al., 2009; Ozarda, 2016). Thresholds for measures with a positively skewed distribution were set at the 95th percentile of healthy reference values.
Aim 2: To investigate preliminary associations between parameters of swallowing physiology and metrics of swallowing safety and efficiency, we ran separate generalized estimating equation models with a repeated factor of bolus trial, exploring parameters of airway protection and bolus clearance against PAS (binary logit) and total pharyngeal residue (linear). For models exploring swallowing safety, bolus trials were categorized as “safe” (PAS < 3) or “unsafe” (PAS ≥ 3) based on the worst PAS score recorded for that trial.
Aim 3: To provide evidence toward the clinical use of thickened liquids for reducing aspiration in individuals with ALS, we compared the frequency of unsafe swallowing, observed during videofluoroscopy, across liquid thicknesses using Cochrane's Q tests. Friedman's tests were used to determine whether thicker liquids contributed to increased pharyngeal residue.
Missing Data and Outliers
All videofluoroscopy data from one participant (n = 15 bolus clips) and n = 10 additional bolus clips from separate participants were excluded due to synchronization issues during recording, which resulted in image replication and visual artifacts. Furthermore, five participants inconsistently displayed atypical patterns of LVC, such that the resulting time-to-LVC and/or LVCdur measures could not be merged with group-level data. Specifically, three participants (n = 12 thin bolus trials) inconsistently displayed premature LVC (i.e., complete arytenoid-to-epiglottic contact, which occurred prior to the onset of hyoid movement), whereas two other participants (n = 4 bolus trials) swallowed sequentially and maintained complete LVC until the subsequent swallow had been completed (i.e., no LVC offset during Swallow 1).
Results
Interrater Reliability
Interrater reliability was calculated based on videofluoroscopy ratings obtained prior to consensus resolution. Results are summarized in the Appendix. Overall results suggested excellent agreement for frame selection and good-to-excellent agreement for continuous pixel-based videofluoroscopy measures (Koo & Li, 2016). Agreement estimated by the kappa statistic for binary and nominal ratings of LVC and PAS were “poor” and “moderate,” respectively, likely due to an observed imbalance in LVC and PAS scores in the data set, which can result in a lower kappa value (Viera & Garrett, 2005). Of note, percentage of agreement for each of these parameters remained above 75%, and all discrepancies were resolved through consensus review, prior to analysis.
Descriptive Data
Nineteen adults (nine women) over the age of 18 years (M age = 62 years, range: 47–78 years) with a confirmed diagnosis of ALS were recruited from a university ALS clinic. Demographic details are presented in Table 2. Of note, the proportion of individuals with bulbar versus spinal-onset ALS in this study exceeded expected population prevalence rates (approximately 3:7; Swinnen & Robberecht, 2014), likely due to the aforementioned recruitment strategy. Bulbar symptoms were reported by 8 of 10 participants who initially presented with spinal-onset ALS; two participants in the study denied bulbar symptoms at the time of assessment.
Table 2.
Summary of participant demographics and baseline measures.
| Demographics | ALS (n = 19) | Reference a (n = 40) | |
|---|---|---|---|
| Age, x̄ (range) | 62 (47–78) | 34 (24–58) | |
| Male/female | 10/9 | 20/20 | |
| Bulbar-onset, n (%) | 9 (%) | — | |
| Spinal-onset, n (%) | 10 | — | |
| Mixed-onset, n (%) | 1 | — | |
| ALSFRS-R scores | |||
| Total (max: 48), x̄ (range) | 36.5 (23–44) | — | |
| Bulbar (max: 12), x̄ (range) | 9 (3–12) | — | |
| Swallowing (max: 4), x̄ (range) | 3 (1–4) | — | |
| Respiration (max: 12), x̄ (range) | 11 (4–12) | — | |
| Disease duration | |||
| Months since diagnosis b | 16.53 (SD = 15.6) | — | |
| Months since initial symptom b | 36.37 (SD = 26.68) | — | |
| Months since first bulbar symptom b | 16.74 (SD = 17.46) | — | |
| Sip size (thin liquids) | |||
| Bolus weight (g), x̄ (SD) | 12.1 (6.0) | 10.5 (5.8) | |
| Estimated bolus volume (ml) c | 14.0 (7.0) | 12.1 (6.7) | |
Note. ALS = amyotrophic lateral sclerosis; ALSFRS-R = ALS Functional Rating Scale–Revised; max = maximum.
Healthy control data reproduced, with permission, from Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al. (2019).
Disease/symptom duration not available for n = 1 participant with spinal-onset ALS.
Sip volume estimated from previous data characterizing the density of the barium liquids used in this study Steele, Peladeau-Pigeon, Barbon, Guida, Tapson, et al., 2019.
The videofluoroscopy protocol was terminated early for three patients due to bailout criteria being met (n = 2) or participant fatigue (n = 1). Thus, a total of 245 bolus trials were available for analysis, including n = 54 thin barium, n = 52 slightly thick, n = 45 mildly thick, n = 47 moderately thick, and n = 47 extremely thick.
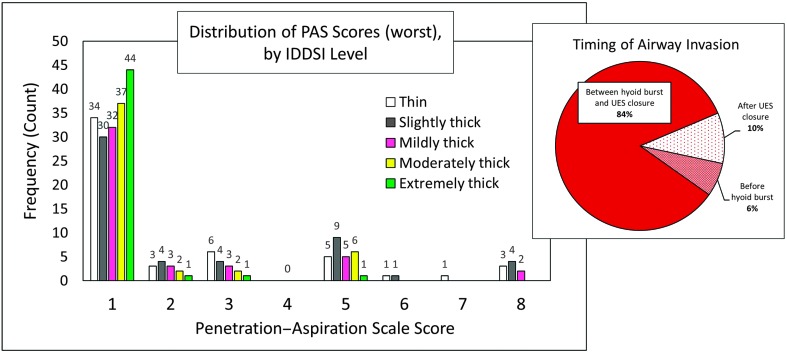
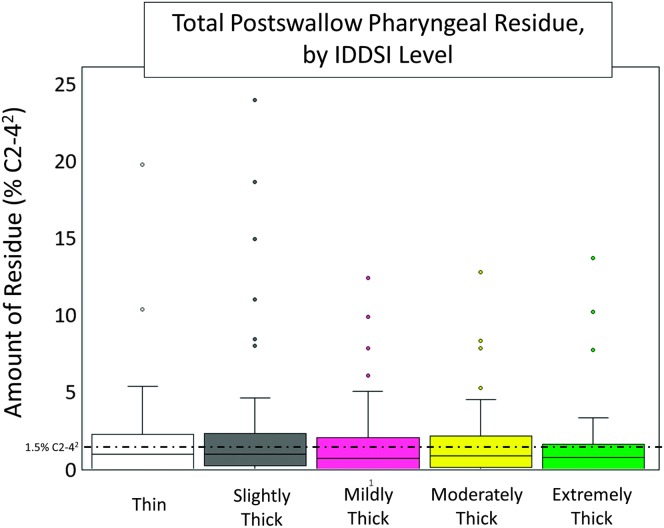
In terms of swallowing function, 14 of 19 participants had at least one bolus trial that was rated as unsafe. At the bolus level, 22% of all trials (n = 54) were deemed to be unsafe, with a total of 94 unsafe events recorded (i.e., n = 25 bolus trials had more than one instance of PAS ≥ 3). The distribution of PAS scores and timing of airway invasion at the bolus level, based on worst PAS score observed per bolus trial, are shown in Figure 1. Swallowing efficiency was impaired in 10 of 19 individuals with ALS in this study based on total pharyngeal residue and maximum number of swallows. Eight participants displayed above-reference residue on at least one trial, and eight participants (42% of sample) required three or more swallow events on at least one bolus trial (Mdn = 1, range: 1–10). At the bolus level, 13% of all trials (n = 32) displayed pharyngeal area at maximum constriction residue, which measured above the 95th percentile for healthy adults (Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019). Total pharyngeal residue, by IDDSI level, is presented in Figure 2.
Figure 1.
Distribution of PAS scores at the bolus level, based on worst PAS score (n = 244; n = 1 missing data). Raw frequency count of observations is displayed above each column. Timing of airway invasion for instances of PAS ≥ 3 at the swallow level is shown in the pie chart (right). PAS = Penetration–Aspiration Scale; IDDSI = International Dysphagia Diet Standardisation Initiative; UES = upper esophageal sphincter.
Figure 2.
Measures of total pharyngeal residue, expressed as a percentage of the squared C2–4 reference area. Median pharyngeal residue across all bolus tasks was 2.42% C2–42 (range: 0–7; 23.93% C2–42). A reference line placed at 1.5% C2–42 depicts the upper 95% confidence interval for pharyngeal residue in healthy individuals (see Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019). IDDSI = International Dysphagia Diet Standardisation Initiative.
Aim 1: Differences in Swallow Timing and Kinematics Between Individuals With ALS and Healthy Reference Data
Parameters of swallowing physiology and function are summarized in Table 3.
Table 3.
Summary of physiological measures of swallowing, calculated at the bolus level based on the first swallow per trial.
| Parameter | Unit of measure | IDDSI level | ALS mean (95% CI) | Ref data mean (thin boluses)
c
(95% CI) |
Interpretation for individuals with ALS | p value |
|---|---|---|---|---|---|---|
| Time-to-LVC a | ms | Thin | 298 [260, 337] | 179 [69, 148] | LVC occurred later | < .001 |
| Slightly | 272 [224, 319] | |||||
| Mildly | 296 [253, 339] | |||||
| Moderately | 321 [277, 366] | |||||
| Extremely | 331 [288, 373] | |||||
| LVCdur a | ms | Thin | 526 [471, 581] | 436 [412, 460] | LVC closed longer | .004 |
| Slightly | 485 [445, 526] | |||||
| Mildly | 463 [426, 500] | |||||
| Moderately | 440 [408, 472] | |||||
| Extremely | 438 [402, 474] | |||||
| UESOdur | ms | Thin | 589 [555, 623] | 458 [444, 472] | UES open longer | < .001 |
| Slightly | 558 [527, 590] | |||||
| Mildly | 583 [560, 606] | |||||
| Moderately | 499 [480, 519] | |||||
| Extremely | 486 [463, 509] | |||||
| UESMax | %C2–4 | Thin | 16.6 [14.6, 18.6] | 20.6 [19.3, 21.8] | UES opening narrower | .001 |
| Slightly | 17.8 [15.7, 19.9] | |||||
| Mildly | 18.5 [16.6, 20.4] | |||||
| Moderately | 17.4 [15.7, 19.1] | |||||
| Extremely | 20.0 [18.3, 21.8] | |||||
| Max. pharyngeal constriction area b | %C2–42 | Thin | 1.8 (0;44.5) | 0 (0;7.0) | Pharynx less constricted | < .001 |
| Slightly | 2.3 (0;54.8) | |||||
| Mildly | 2.2 (0;18.2) | |||||
| Moderately | 2.4 (0;18.2) | |||||
| Extremely | 1.6 (0;18.3) | |||||
| LVC | Nominal grading; shown as % of bolus trials with partial or incomplete LVC | Thin | 24% | < 1% | LVC more frequently not complete | < .001 |
| Slightly | 27% | |||||
| Mildly | 22% | |||||
| Moderately | 11% | |||||
| Extremely | 2% |
Note. Results from thin liquid trials compared to a published healthy data (Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019) set using unpaired t tests. IDDSI = International Dysphagia Diet Standardisation Initiative; ALS = amyotrophic lateral sclerosis; CI = confidence interval; LVC = laryngeal vestibule closure; LVCdur = duration of laryngeal vestibule closure; UESOdur = UES opening duration; UES = upper esophageal sphincter UESmax = UES opening diameter.
Based on swallows with complete LVC only.
Maximum pharyngeal constriction area reported using median (min;max) due to significant positive skew and presence of statistical outliers in the data.
Reference values reprinted from Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al. (2019) reference article, with permission.
Group Comparisons
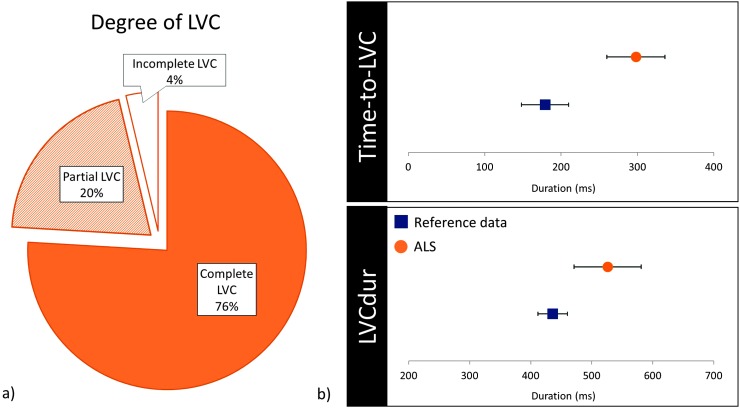
Compared to healthy reference data, this sample of individuals with ALS presented with longer time-to-LVC with a large effect size, t(66) = 5.396, p < .001, d = 1.09; longer LVCdur with a medium effect size, t(52) = 3.032, p = .004, d = 0.64; and a higher proportion of bolus trials with partial or incomplete LVC, χ2(1) = 30.930, p < .001. Swallows with partial LVC (20%, n = 11 boluses) presented with a mean time-to-max A–E approximation of 367 ms (95% confidence interval [307, 426]) and a mean duration at max A–E approximation of 356 ms (95% confidence interval [277, 435]).
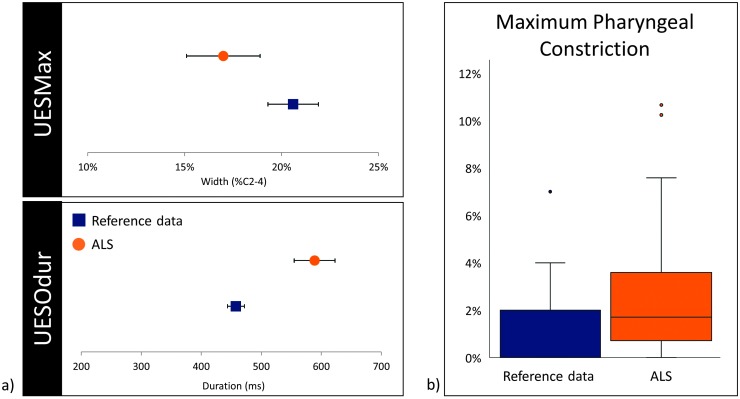
In terms of pharyngeal parameters, average UESOdur measured significantly longer with a large effect size, t(70) = 7.133, p < .001, d = 1.34; UESMax measured narrower with a medium effect size, t(89) = −3.401, p = .001, d = 0.59; and pharyngeal area at maximum constriction was significantly larger (i.e., less constricted; U = 1325.0, z = −5.248, p < .001). Of note, two participants displayed maximum pharyngeal constriction areas that measured as extreme outliers on at least one thin trial (i.e., greater than 3 times the interquartile range above the 75th percentile; Rosner, 2016). Exclusion of these extreme outliers had no influence on the results of the Mann–Whitney U test (U = 1325.0, z = −4.857, p < .001). Comparisons to reference data are presented in Figures 3 and 4.
Figure 3.
(a) Pie chart illustrating the proportion of cases (bolus level) with complete, partial, or incomplete LVC. (b) Error plots displaying means and 95% confidence intervals for time-to-LVC and LVCdur (includes swallows with complete LVC only), compared to healthy reference data. LVC = laryngeal vestibule closure; LVCdur = duration of laryngeal vestibule closure; ALS = amyotrophic lateral sclerosis.
Figure 4.
(a) Error plots displaying means and 95% confidence intervals for UESMax (top) and UESOdur (bottom), compared to healthy reference data. (b) Box and whisker plots comparing distribution of pharyngeal constriction area between healthy reference data and ALS cohort; extreme outliers (> 12.9% C2–42) are not displayed. UES = upper esophageal sphincter; UESMax = diameter of UES opening; UESOdur = UES opening duration; ALS = amyotrophic lateral sclerosis.
Participant-Level Comparisons
Table 4 summarizes swallow physiology at the participant level, sorted by patient-reported bulbar and swallowing symptoms on the ALSFRS-R. Time-to-LVC measured > 2 SDs longer than the healthy reference mean for seven participants, including two participants who denied experiencing any bulbar symptoms at the time of evaluation. Seven participants displayed LVCdur, which was > 2 SDs longer than the healthy reference mean, whereas only one participant displayed LVCdur, which was > 2 SDs shorter than the healthy reference mean. Eight individuals presented with partial LVC on at least one thin liquid trial, including n = 1 participant who denied bulbar symptoms at the time of evaluation. Only one participant displayed incomplete LVC, which was scored incomplete across all repeated trials.
Table 4.
Participant-level summary of swallow physiology in relation to symptom severity (ALSFRS-R) and swallow safety.
| Case no. | Age | Sex | Onset type | ALSFRS-R scores |
Maximum PAS (% unsafe) | LVC pattern (% not complete) | Time-to-LVC (ms) | LVCdur (ms) | UESOdur (ms) | Pharyngeal Area at Maximum Constriction (%C–42) | UESMax (%C2–4) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total /48 |
Bulbar /12 |
Swallow /4 |
|||||||||||
| 1 | 62 | F | Spinal | 44 | 12 | 4 | 1 (0) | Complete (0) | 133.3 (0%) | 683.3 (50%)↑ | 700.0 (100%)↑ | 0.74 (0%) | 19.14 (0%) |
| 2 | 68 | M | Spinal | 40 | 12 | 4 | 1 (0) | Complete (0) | 311.1 (33%)↑ | 544.4 (33%)↑ | 566.7 (33%)↑ | 2.55 (33%)↑ | 24.66 (0%) |
| 3 | 47 | F | Spinal | 39 | 12 | 4 | 3 (67) | Partial (33) | 400.0 (50%)↑ | 350.0 (0%) | 622.2 (100%)↑ | 0.44 (0%) | 13.69 (0%) |
| 4 | 48 | M | Spinal | 39 | 12 | 4 | 1 (0) | Complete (0) | 166.7 (0%) | 444.4 (0%) | 677.8 (100%)↑ | 0.18 (0%) | 25.32 (0%) |
| 5 | 67 | F | Spinal | 35 | 11 | 4 | 1 (0) | Partial (33) | 250.0 (0%) | 600.0 (50%)↑ | 733.3 (100%)↑ | 3.71 (0%) | 21.42 (0%) |
| 6 | 59 | M | Spinal | 32 | 11 | 4 | 1 (0) | Partial (100) | N/A a | N/A a | 444.4 (0%) | 0.34 (0%) | 24.76 (0%) |
| 7 | 68 | F | Spinal | 28 | 11 | 4 | 5 (33) | Partial (33) | 283.3 (0%) | 416.7 (0%) | 600.0 (67%)↑ | 3.19 (33%)↑ | 17.58 (0%) |
| 8 | 68 | F | Spinal | 29 | 9 | 4 | 5 (50) | Complete (0) | 183.3 (0%) | 750.0 (100%)↑ | 616.7 (100%)↑ | 0.25 (0%) | 26.24 (0%) |
| 9 | 59 | M | Bulbar | 44 | 9 | 3 | 1 (0) | Complete (0) | 250.0 (0%) | 550.0 (0%) | 466.7 (0%) | 3.14 (0%) | 15.28 (0%) |
| 10 | 77 | M | Mixed | 41 | 9 | 3 | 1 (0) | Complete (0) | 222.2 (0%) | 466.7 (0%) | 600.0 (67%)↑ | 1.66 (0%) | 12.58 (0%) |
| 11 | 49 | M | Bulbar | 36 | 8 | 3 | 1 (0) | Complete (0) | 350.0 (33%)↑ | 633.3 (67%)↑ | 844.4 (100%)↑ | 1.65 (0%) | 18.85 (0%) |
| 12 | 76 | M | Bulbar | 41 | 7 | 3 | 5 (33) | Partial (33) | 333.3 (0%) | 500.0 (0%) | 600.0 (100%)↑ | 4.07 (67%)↑ | 20.56 (0%) |
| 13 | 56 | F | Bulbar | 42 | 6 | 3 | 8 (67) | Partial (67) | 400.0 (100%)↑ | N/A c | 588.9 (33%)↑ | 3.14 (0%) | 6.00 (100%)↓ |
| 14 | 52 | M | Bulbar | 24 | 4 | 3 | 6 (100) | Partial (33) | 383.3 (50%)↑ | 700.0 (50%)↑ | 388.9 (33%)↓ | 12.41 (100%)↑ | 5.43 (100%)↓ |
| 15 | 72 | F | Bulbar | 34 | 3 | 3 | 1 (0) | Complete (0) | 322.2 (0%) | 500.0 (0%) | 433.3 (0%) | 2.33 (33%)↑ | 12.10 (0%) |
| 16 | 55 | M | Bulbar | 41 | 7 | 2 | 3 (100) | Partial (33) | 383.3 (50%)↑ | 333.3 (0%) | 533.3 (33%)↑ | 4.50 (67%)↑ | 8.97 (0%) |
| 17 | 70 | F | Bulbar | 37 | 6 | 2 | 1 (0) | Complete (0) | 322.2 (0%) | 700.0 (33%)↑ | 566.7 (33%)↑ | 0.87 (0%) | 9.38 (0%) |
| 18 | 78 | F | Spinal | 23 | 5 | 2 | 5 (33) | Complete (0) | 455.6 (100%)↑ | 266.7 (33%)↓ | 600.0 (67%)↑ | 1.58 (0%) | 14.42 (0%) |
| 19 | 49 | M | Bulbar | 34 | 5 | 1 | 8 (100) | Incomplete (100) | N/A b | N/A b | 483.3 (0%) | 43.74 (100%)↑ | 6.24 (100%)↓ |
Note. Percentage above threshold refers to the proportion of thin bolus trials that fell > 2 SDs above or below the reference data mean (see Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019. Arrows indicate the direction of the difference (i.e., ↑ indicates > 2 SDs greater than reference mean; ↓ indicates > 2 SDs less than reference mean). Mean time-to-LVC and LVCdur based on trials demonstrating complete LVC, only. Participants are sorted by self-reported swallow function (note that lower scores on the ALSFRS-R reflect greater functional impairment). Values displayed are mean and proportion of thin trials falling outside the reference threshold. ALSFRS-R = ALS Functional Rating Scale–Revised; PAS = Penetration–Aspiration Scale; LVC = laryngeal vestibule closure; LVCdur = duration of laryngeal vestibule closure; UESOdur = UES opening duration; UES = upper esophageal sphincter; F = female; M = male; N/A = not available.
Complete A–E contact achieved prior to swallow onset.
Incomplete LVC status on all trials.
Sequential swallow pattern (no LVC offset).
UESOdur measured > 2 SDs longer than the healthy reference mean for 14 participants, whereas only one participant displayed UESOdur, which measured > 2 SDs shorter than the healthy reference mean, on a single trial. Four participants displayed UESMax > 2 SDs narrower than healthy reference data. Maximum pharyngeal constriction area was measured above the 95th percentile for healthy reference data in seven participants.
Aim 2: Physiological Mechanisms Associated With Impaired Safety or Efficiency
Preliminary explorations revealed that bolus trials with airway invasion were more likely to display prolonged time-to-LVC, χ2(1) = 6.764, p = .009; partial/incomplete LVC, χ2(1) = 17.000, p < .001; or enlarged (i.e., less constricted) maximum pharyngeal constriction areas, χ2(1) = 21.459, p < .001. Enlarged maximum pharyngeal constriction area was also associated with higher total pharyngeal residue, χ2(1) = 307.255, p < .001. No other associations reached statistical significance.
Aim 3: The Influence of Thickened Liquids on Acute Swallowing Safety and Efficiency
Liquid thickness had a significant effect on acute swallow safety, χ2(4) = 10.720, p = .03, such that thin liquids had a higher proportion of unsafe swallows, compared to extremely thick liquids (p = .002). Bolus thickness did not influence the amount of pharyngeal residue, χ2(4) = 1.908, p = 0.753, nor number of swallow events per bolus, χ2(4) = 7.200, p = .126.
Discussion
The primary purpose of this study was to quantify various parameters of swallowing physiology and function across various liquid consistencies in a sample of individuals with ALS. Comparing results from thin barium liquid swallows to healthy reference data (Steele, Peladeau-Pigeon, Barbon, Guida, Namasivayam-MacDonald, et al., 2019), individuals with ALS presented with prolonged time to achieve airway closure, a higher proportion of swallows with partial or incomplete LVC, a narrower UES opening diameter, prolonged LVC duration and UES opening duration, and reduced maximum pharyngeal constriction areas. In some cases, differences in swallowing physiology preceded patient-reported swallowing problems, consistent with previous findings that subclinical changes may be occurring early in the disease process (Allison et al., 2017; Briani et al., 1998; Perry et al., 2018). We also identified a higher proportion of unsafe or inefficient swallows in individuals with ALS, with preliminary results implicating the timeliness and degree of airway closure (time-to-LVC, LVC) as mechanisms underlying swallowing safety and maximum pharyngeal constriction as a mechanism underlying swallowing efficiency. Airway safety ratings were variable within the same participant, even among swallows of the same bolus thickness. This finding is consistent with observations in other clinical populations (e.g., Hedström et al., 2017; Steele, Nagy, et al., 2015).
In this study, all included measures of swallowing physiology fell outside the boundaries of healthy reference data, even for some individuals who denied experiencing any swallowing problems or bulbar symptoms as reported on the ALSFRS-R. This was not surprising, as recent research has indicated that patient reports and clinician judgments lack sensitivity to detect early changes, when compared to objective measures of bulbar kinematics (Allison et al., 2017). Early, measurable differences such as those included in this study may equip clinicians to initiate discussions with patients regarding swallowing function and permit proactive counseling geared toward long-term management of dysphagia. Measures that are able to detect differences also hold great value in ALS research, as there is an ongoing need for the identification and development of biomarkers and clinical measures to estimate prognosis, monitor disease progression, and serve as outcome measures in clinical research trials (Taga & Maragakis, 2018; Turner et al., 2013).
We were surprised, however, to find that LVCdur and UESOdur measured longer in this group of individuals with ALS, compared to healthy adults. This may be in part due to inherent variability in swallow timing and kinematics, as measures of LVCdur and UESOdur have been reported to vary by bolus volume and age in healthy adults (Leonard et al., 2004; Molfenter & Steele, 2012; Namasivayam-MacDonald et al., 2018). Even still, prolonged UESOdur has also been described in patients with oculopharyngeal and myotonic muscular dystrophies (Leonard et al., 2001; Waito, Steele, et al., 2018) as well as adults with cricopharyngeal bar (Leonard et al., 2004), which the authors attributed to potential physiological compensation. Evidence for such a phenomenon in individuals with ALS has been seen in measures of speech kinematics, as patients with ALS who display reduced tongue movement often present with increases in jaw movement to maintain overall intelligibility (Shellikeri et al., 2016; Yunusova et al., 2010).
In the current study, the frequency of unsafe swallowing was lower for extremely thick liquids compared to thin liquids, without evidence of increased pharyngeal residue. These results are encouraging to provide support toward the recommendation of thickened liquids to reduce the immediate risk of aspiration in individuals with ALS, without compromising swallowing efficiency. However, these findings contrast results from another recent study involving patients with ALS, in which greater vallecular residue was observed for pudding-thick liquids, compared to thin liquids (Waito, Tabor-Gray, et al., 2018). This discrepancy may be related to group differences in overall disease severity, as well as differences in bolus composition (barium, thickener type) and volume in each study (Steele, Alsanei, et al., 2015; Vilardell et al., 2016). Owing to a variety of factors that may also impact the relationships between bolus texture and swallow safety/efficiency in individuals with ALS, individualized recommendations based on instrumental assessment continue to be encouraged. Furthermore, it is important to clarify that the findings from this analysis are limited to the short-term reduction of aspiration events and cannot be generalized to impact long-term health effects (e.g., aspiration pneumonia).
Limitations
As with any study, there are several limitations to note that impact generalization of the results. First, this study involved a small, heterogeneous cross-sectional sample of individuals with ALS, with a low overall occurrence of unsafe or inefficient swallowing. Thus, it is unknown whether the observations in this study translate to individuals experiencing more severe symptoms of dysphagia. Furthermore, the mean age of our participant group was older than the available reference data set, as reference data for older individuals across the IDDSI liquid framework are not yet available. Of note, two measures included in this study (i.e., UESOdur, UESMax) have been reported to change with age, such that UESOdur has measured longer in older adults and lateral view UESMax has measured narrower (e.g., Kern et al., 1999; Leonard et al., 2004). Still, it remains unclear at what age these differences are truly observed, due to variability in age stratification between studies (Namasivayam-MacDonald et al., 2018). In addition, cognitive ability was screened informally for purposes of inclusion, to ensure that participants understood the study risks and would be able to follow the study protocol, safely. Given that up to 50% of individuals with ALS may present with concomitant cognitive changes (e.g., Lomen-Hoerth et al., 2003; Ringholz et al., 2005), future studies may consider exploring the role of cognition on swallow physiology and function in ALS. Finally, this study focused on the first swallow per bolus trial to characterize swallow physiology, in an effort to minimize physiological variability associated with bolus volume on subsequent swallows. As such, we are unable to generalize these findings to clearing or piecemeal swallows. Similarly, we did not explore the risk of penetration–aspiration related to pharyngeal residue, as described by others (e.g., Goeleven et al., 2006; Lo Re et al., 2007; Solazzo et al., 2014), pointing to an area for future research.
Conclusions
Dysphagia is a complex and evolving complication of bulbar disease in individuals with ALS. This study characterizes some of the changes observed during the pharyngeal phase of swallowing using objective and quantitative measurements obtained from videofluoroscopic analysis. Key differences were identified in all aspects of the pharyngeal swallow, including metrics of airway protection, pharyngeal constriction, and UES opening. Despite observing few occurrences of unsafe swallowing in the sample, relationships were identified between swallowing safety and the timeliness and degree of LVC, as well as the degree of maximum pharyngeal constriction. Comparable to previous reports, reduced maximum pharyngeal constriction emerged as a mechanism contributing to pharyngeal residue. Thickened liquids (IDDSI, extremely thick) were efficacious in reducing the occurrence of penetration/aspiration in this sample; however, further research investigating the influence of bolus properties on parameters of swallowing physiology and function is needed, incorporating additional textures (e.g., solids) and bolus composition. Further work is also needed to identify patterns of change within individuals over the course of the disease. In particular, longitudinal study designs will be imperative to define patterns of clinical presentation, including potential compensatory relationships between parameters. Having a better understanding of the measurable changes of swallowing that are associated with ALS, particularly at early stages, will help clinicians detect and monitor changes over the course of the disease to inform timely counseling toward appropriate management options.
Ethics
The study was approved by the local institutional review boards.
Acknowledgments
Funding for this study was provided through an RO1 grant from the National Institute on Deafness and Other Communication Disorders DC011020 to the final author. The authors gratefully acknowledge the patients who participated in this study. The authors also extend thanks to additional members of the Swallowing Rehabilitation Research Lab, namely, Emily Barrett, Andrea Guran, Renata Mancopes, Sana Smaoui, and Teresa Valenzano, who were involved in videofluoroscopy ratings, and members of the Swallowing Systems Core, namely, Jennifer Chapin, Alessandra Gallastegui, Alycia Rivet, and Carol Smith, who supported data collection at the University of Florida. Ashley Waito was involved in project design, study administration, data collection, videofluoroscopy rating, statistical analysis, interpretation of results, and drafting of the final article. Emily Plowman was a co-principal investigator for the project and was involved in project design, study administration, participant recruitment, swallowing data collection, interpretation of results, and editing of the final article. Carly Barbon was involved in project design, barium stimuli development, videofluoroscopy rating, data and statistical analysis, and editing of the final article. Melanie Peladeau-Pigeon contributed essential tools to the project and was involved in project design, data analysis, and editing of the final article. Lauren Tabor-Gray was responsible for assisting with swallowing data collection, interpretation of results, and editing of the final article. Kelby Magennis and Raele Robison were responsible for study administration, participant recruitment, swallowing data collection, and review of the final article. Catriona Steele was the principal investigator for the project and was involved in project design, study administration, statistical analysis, interpretation of results, and editing of the final article.
Appendix
Interrater Agreement Statistics for PAS scores, Frame Selection, and Pixel-Based Measurements
| Parameter | Level of data | Agreement statistic | Value (95% CI) | Interpretation c | % Requiring consensus resolution |
|---|---|---|---|---|---|
| PAS | Nominal | Kappa | 0.515; 82% in agreement | Moderate | 18 |
| LVC (±) | Binary | Kappa | 0.198; 76% in agreement | Poor | 24 |
| Hyoid burst frame | Continuous | ICC a | 0.972 [0.966, 0.977] | Excellent | 8 |
| LVC frame | Continuous | ICC a | 0.982 [0.979, 0.986] | Excellent | 13 |
| LVC offset frame | Continuous | ICC a | 0.982 [0.978, 0.985] | Excellent | 2 |
| Maximum pharyngeal constriction frame | Continuous | ICC a | 0.980 [0.976, 0.984] | Excellent | 3 |
| UES opening frame | Continuous | ICC a | 0.984 [0.980, 0.987] | Excellent | 3 |
| Maximum UES distension frame | Continuous | ICC a | 0.999 [0.998, 0.999] | Excellent | 3 |
| UES closure frame | Continuous | ICC a | 0.980 [0.976, 0.983] | Excellent | 9 |
| Swallow rest frame | Continuous | ICC a | 0.966 [0.959, 0.972] | Excellent | 18 |
| UESMax (%C2–4) | Continuous | ICC a | 0.828 [0.783, 0.863] b | Good | 7 |
| Pharyngeal constriction area (%C2–42) | Continuous | ICC a | 0.903 [0.877, 0.932] b | Good to excellent | 2 |
| Total pharyngeal residue (%C2–42) | Continuous | ICC a | 0.885 [0.848, 0.912] | Good to excellent | 3 |
Note. PAS = Penetration–Aspiration Scale; CI = confidence interval; LVC = laryngeal vestibule closure; UES = upper esophageal sphincter.
ICC model = two-way random, absolute agreement.
n = 2 impossible values identified and removed from ICC calculations.
Qualitative interpretation of agreement statistics from following references: Viera & Garrett (2005) and Koo & Li (2016).
Funding Statement
Funding for this study was provided through an RO1 grant from the National Institute on Deafness and Other Communication Disorders DC011020 to the final author.
References
- Allison K. M., Yunusova Y., Campbell T. F., Wang J., Berry J. D., & Green J. R. (2017). The diagnostic utility of patient-report and speech-language pathologists' ratings for detecting the early onset of bulbar symptoms due to ALS. Amyotrophic Lateral Sclerosis and Frontotemporal Degeneration, 18(5–6), 358–366. https://doi.org/10.1080/21678421.2017.1303515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbon C. E. A., & Steele C. M. (2018). Characterizing the flow of thickened barium and non-barium liquid recipes using the IDDSI flow test. Dysphagia, 34(1), 73–79. https://doi.org/10.1007/s00455-018-9915-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briani C., Marcon M., Ermani M., Costantini M., Bottin R., Iurilli V., Zaninotto G., Primon D., Feltrin G., & Angelini C. (1998). Radiological evidence of subclinical dysphagia in motor neuron disease. Journal of Neurology, 245(4), 211–216. https://doi.org/10.1007/s004150050207 [DOI] [PubMed] [Google Scholar]
- Brooks B. R., Miller R. G., Swash M., & Munsat T. L. (2000). El Escorial revisited: Revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotrophic Lateral Sclerosis and Other Motor Neuron Diseases, 1(5), 293–299. [DOI] [PubMed] [Google Scholar]
- Cedarbaum J. M., Stambler N., Malta E., Fuller C., Hilt D., Thurmond B., & Nakanishi A. (1999). The ALSFRS-R: A revised ALS functional rating scale that incorporates assessments of respiratory function. Journal of the Neurological Sciences, 169(1–2), 13–21. https://doi.org/10.1016/s0022-510x(99)00210-5 [DOI] [PubMed] [Google Scholar]
- Ceriotti F., Hinzmann R., & Panteghini M. (2009). Reference intervals: The way forward. Annals of Clinical Biochemestry, 46(Pt. 1), 8–17. https://doi.org/10.1258/acb.2008.008170 [DOI] [PubMed] [Google Scholar]
- Chi-Fishman G., & Sonies S. C. (2002). Effects of systematic bolus viscosity and volume changes on hyoid movement kinematics. Dysphagia, 17(4), 278–287. https://doi.org/10.1007/s00455-002-0070-7 [DOI] [PubMed] [Google Scholar]
- Cichero J. A., Lam P., Steele C. M., Hanson B., Chen J., Dantas R. O., Duivestein J., Kayashita J., Lecko C., Murray J., Pillay M., Riquelme L., & Stanschus S. (2017). Development of international terminology and definitions for texture-modified foods and thickened fluids used in dysphagia management: The IDDSI Framework. Dysphagia, 32(2), 293–314. https://doi.org/10.1007/s00455-016-9758-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corcia P., Pradat P.-F., Salachas F., Bruneteau G., Forestier N., Seilhean D., Hauw J.-J., & Meininger V. (2008). Causes of death in a post-mortem series of ALS patients. Amyotrophic Lateral Sclerosis, 9(1), 59–62. https://doi.org/10.1080/17482960701656940 [DOI] [PubMed] [Google Scholar]
- IBM Corporation. (2016). IBM SPSS Statistics for Windows, Version 24.0 [Computer software].
- Desport J. C., Preux P. M., Truong T. C., Vallat J. M., Sautereau D., & Couratier P. (1999). Nutritional status is a prognostic factor for survival in ALS patients. Neurology, 53(5), 1059–1063. https://doi.org/10.1212/WNL.53.5.1059 [DOI] [PubMed] [Google Scholar]
- Ertekin C., Aydogdu I., Yüceyar N., Kiylioglu N., Tarlaci S., & Uludag B. (2000). Pathophysiological mechanism of oropharyngeal dysphagia in amyotrophic lateral sclerosis. Brain, 123(Pt. 1), 125–140. https://doi.org/10.1093/brain/123.1.125 [DOI] [PubMed] [Google Scholar]
- Garand K. L., Schwertner R., Chen A., & Pearson W. G. Jr. (2018). Computational analysis of pharyngeal swallowing mechanics in patients with motor neuron disease: A pilot investigation. Dysphagia, 33(2), 243–250. https://doi.org/10.1007/s00455-017-9853-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goeleven A., Robberecht W., Sonies B., Carbonez A., & Dejaeger E. (2006). Manofluorographic evaluation of swallowing in amyotrophic lateral sclerosis and its relationship with clinical evaluation of swallowing. Amyotrophic Lateral Sclerosis, 7(4), 235–240. https://doi.org/10.1080/17482960600664870 [DOI] [PubMed] [Google Scholar]
- Green J. R., Yunusova Y., Kuruvilla M. S., Wang J., Pattee G. L., Synhorst L., Zinman L., & Berry J. D. (2013). Bulbar and speech motor assessment in ALS: Challenges and future directions. Amyotrophic Lateral Sclerosis and Frontotemporal Degeneration, 14(7–8), 494–500. https://doi.org/10.3109/21678421.2013.817585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedström J., Tuomi L., Andersson M., Dotevall H., Osbeck H., & Finizia C. (2017). Within-bolus variability of the Penetration–Aspiration Scale across two subsequent swallows in patients with head and neck cancer. Dysphagia, 32(5), 683–690. https://doi.org/10.1007/s00455-017-9814-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higo R., Tayama N., & Nito T. (2004). Longitudinal analysis of progression of dysphagia in amyotrophic lateral sclerosis. Auris, Nasus, Larynx, 31(3), 247–254. https://doi.org/10.1016/j.anl.2004.05.009 [DOI] [PubMed] [Google Scholar]
- Hind J., Divyak E., Zielinski J., Taylor A., Hartman M., Gangnon R., & Robbins J. (2012). Comparison of standardized bariums with varying rheological parameters on swallowing kinematics in males. Journal of Rehabilitation Research and Development, 49(9), 1399–1404. https://doi.org/10.1682/JRRD.2011.09.0180 [DOI] [PubMed] [Google Scholar]
- Hoffman M. R., Ciucci M. R., Mielens J. D., Jiang J. J., & McCulloch T. M. (2010). Pharyngeal swallow adaptations to bolus volume measured with high resolution manometry. Laryngoscope, 120(12), 2367–2373. https://doi.org/10.1002/lary.21150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahrilas P. J., Lin S., Chen J., & Logemann J. A. (1996). Oropharyngeal accommodation to swallow volume. Gastroenterology, 111(2), 297–306. https://doi.org/10.1053/gast.1996.v111.pm8690194 [DOI] [PubMed] [Google Scholar]
- Kern M., Hofmann C., Bardan E., Ren J., Arndorfer R., & Shaker R. (1999). Comparison of upper esophageal sphincter opening in healthy asymptomatic young and elderly volunteers. Annals of Otology, Rhinology & Laryngology, 108(10), 982–989. https://doi.org/10.1177/000348949910801010 [DOI] [PubMed] [Google Scholar]
- Koo T. K., & Li M. Y. (2016). A guideline of selecting and reporting intraclass correlation coefficients for reliability research. Journal of Chiropractic Medicine, 15(2), 155–163. https://doi.org/10.1016/j.jcm.2016.02.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leonard R. J., Kendall K. A., Johnson R., & McKenzie S. (2001). Swallowing in myotonic muscular dystrophy: A videofluoroscopic study. Archives of Physical Medicine and Rehabilitation, 82(7), 979–985. https://doi.org/10.1053/apmr.2001.23962 [DOI] [PubMed] [Google Scholar]
- Leonard R., Kendall K., & McKenzie S. (2004). UES opening and cricopharyngeal bar in nondysphagic elderly and nonelderly adults. Dysphagia, 19(3), 182–191. https://doi.org/10.1007/s00455-004-0005-6 [DOI] [PubMed] [Google Scholar]
- Lo Re G., Galia M., La Grutta L., Russo S., Runza G., Taibbi A., D'Agostino T., Lo Greco V., Bartolotta T. V., Midiri M., Cardinale A. E., De Maria M., & Lagalla R. (2007). Digital cineradiographic study of swallowing in patients with amyotrophic lateral sclerosis. La Radiologia Medica, 112(8), 1173–1187. https://doi.org/10.1007/s11547-007-0214-9 [DOI] [PubMed] [Google Scholar]
- Lomen-Hoerth C., Murphy J., Langmore S., Kramer J. H., Olney R. K., & Miller B. (2003). Are amyotrophic lateral sclerosis patients cognitively normal? Neurology, 60(7), 1094–1097. https://doi.org/10.1212/01.WNL.0000055861.95202.8D [DOI] [PubMed] [Google Scholar]
- Macrae P., Anderson C., & Humbert I. (2014). Mechanisms of airway protection during chin-down swallowing. Journal of Speech, Language, and Hearing Research, 57(4), 1251–1258. https://doi.org/10.1044/2014_JSLHR-S-13-0188 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makkonen T., Ruottinen H., Puhto R., Helminen M., & Palmio J. (2017). Speech deterioration in amyotrophic lateral sclerosis (ALS) after manifestation of bulbar symptoms. International Journal of Language & Communication Disorders, 53(2), 385–392. https://doi.org/10.1111/1460-6984.12357 [DOI] [PubMed] [Google Scholar]
- Molfenter S. M., Cliffe Polacco R., & Steele C. M. (2011). The validity of multiple swallows per bolus as a sign of swallowing impairment. Paper presented at the European Society for Swallowing Disorder, Leiden, The Netherlands. [Google Scholar]
- Molfenter S. M., & Steele C. M. (2012). Temporal variability in the deglutition literature. Dysphagia, 27(2), 162–177. https://doi.org/10.1007/s00455-012-9397-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molfenter S. M., & Steele C. M. (2014). Kinematic and temporal factors associated with penetration–aspiration in swallowing liquids. Dysphagia, 29(2), 269–276. https://doi.org/10.1007/s00455-013-9506-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murono S., Hamaguchi T., Yoshida H., Nakanishi Y., Tsuji A., Endo K., Kondo S., Wakisaka N., Yamada M., & Yoshizaki T. (2015). Evaluation of dysphagia at the initial diagnosis of amyotrophic lateral sclerosis. Auris, Nasus, Larynx, 42(3), 213–217. https://doi.org/10.1016/j.anl.2014.10.012 [DOI] [PubMed] [Google Scholar]
- Namasivayam-MacDonald A. M., Barbon C. E. A., & Steele C. M. (2018). A review of swallow timing in the elderly. Physiology & Behavior, 184(1), 12–26. https://doi.org/10.1016/j.physbeh.2017.10.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveira D. L., Moreira E. A. M., de Freitas M. B., de Gonçalves J. A., Furkim A. M., & Clavé P. (2017). Pharyngeal residue and aspiration and the relationship with clinical/nutritional status of patients with oropharyngeal dysphagia submitted to videofluoroscopy. Journal of Nutrition, Health & Aging, 21(3), 336–341. https://doi.org/10.1007/s12603-016-0754-6 [DOI] [PubMed] [Google Scholar]
- Ozarda Y. (2016). Reference intervals: Current status, recent developments and future considerations. Biochema Medica, 26(1), 5–16. https://doi.org/10.11613/BM.2016.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paris G., Martinaud O., Hannequin D., Petit A., Cuvelier A., Guedon E., Ropenneck P., & Verin E. (2012). Clinical screening of oropharyngeal dysphagia in patients with ALS. Annals of Physical and Rehabilitation Medicine, 55(9–10), 601–608. https://doi.org/10.1016/j.rehab.2012.10.005 [DOI] [PubMed] [Google Scholar]
- Perry B. J., Martino R., Yunusova Y., Plowman E. K., & Green J. R. (2018). Lingual and jaw kinematic abnormalities precede speech and swallowing impairments in ALS. Dysphagia, 33(6), 840–847. https://doi.org/10.1007/s00455-018-9909-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plowman E. K., Tabor L. C., Wymer J., & Pattee G. (2017). The evaluation of bulbar dysfunction in amyotrophic lateral sclerosis: Survey of clinical practice patterns in the United States. Amyotrophic Lateral Sclerosis and Frontotemporal Degeneration, 18(5–6), 351–357. https://doi.org/10.1080/21678421.2017.1313868 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pupillo E., Messina P., Logroscino G., Beghi E., & SLALOM Group. (2014). Long-term survival in amyotrophic lateral sclerosis: A population-based study. Annals of Neurology, 75(2), 287–297. https://doi.org/10.1002/ana.24096 [DOI] [PubMed] [Google Scholar]
- Ringholz G. M., Appel S. H., Bradshaw M., Cooke N. A., Mosnik D. M., & Schulz P. E. (2005). Prevalence and patterns of cognitive impairment in sporadic ALS. Neurology, 65(4), 586–590. https://doi.org/10.1212/01.wnl.0000172911.39167.b6 [DOI] [PubMed] [Google Scholar]
- Robbins J. (1987). Swallowing in ALS and motor neuron disorders. Neurologic Clinics, 5(2), 213–229. https://doi.org/10.1016/S0733-8619(18)30924-1 [PubMed] [Google Scholar]
- Rosenbek J. C., Robbins J. A., Roecker E. B., Coyle J. L., & Wood J. L. (1996). Dysphagia, 11(2), 93–98. [DOI] [PubMed] [Google Scholar]
- Rosner B. (2016). Fundamentals of biostatistics (8th ed.). Brooks/Cole Cengage Learning. [Google Scholar]
- Ruoppolo G., Schettino I., Frasca V., Giacomelli E., Prosperini L., Cambieri C., Roma R., Greco A., Mancini P., De Vincentiis M., Silani V., & Inghilleri M. (2013). Dysphagia in amyotrophic lateral sclerosis: Prevalence and clinical findings. Acta Neurologica Scandinavica, 128(6), 397–401. https://doi.org/10.1111/ane.12136 [DOI] [PubMed] [Google Scholar]
- Schuele C. M., & Justice L. M. (2006). The importance of effect sizes in the interpretation of research. The ASHA Leader, 11(10), 14–27. https://doi.org/10.1044/leader.FTR4.11102006.14 [Google Scholar]
- Shellikeri S., Green J. R., Kulkarni M., Rong P., Martino R., Zinman L., & Yunusova Y. (2016). Speech movement measures as markers of bulbar disease in amyotrophic lateral sclerosis. Journal of Speech, Language, and Hearing Research, 59(5), 887–899. https://doi.org/10.1044/2016_JSLHR-S-15-0238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solazzo A., Monaco L., Vecchio L. D., Reginelli A., Iacobellis F., Capasso R., Tamburrini S., Berritto D., Barillari M. R., Monsurrò M. R., Di Martino N., & Grassi R. (2014). Earliest videofluoromanometric pharyngeal signs of dysphagia in ALS patients. Dysphagia, 29(5), 539–544. https://doi.org/10.1007/s00455-014-9542-9 [DOI] [PubMed] [Google Scholar]
- Steele C. M., Alsanei W. A., Ayanikalath S., Barbon C. E. A., Chen J., Cichero J. A. Y., Coutts K., Dantas R. O., Duivestein J., Giosa L., Hanson B., Lam P., Lecko C., Leigh C., Nagy A., Namasivayam A. M., Nascimento W. V., Odendaal I., Smith C. H., & Wang H. (2015). The influence of food texture and liquid consistency modification on swallowing physiology and function: A systematic review. Dysphagia, 30(1), 2–26. https://doi.org/10.1007/s00455-014-9578-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steele C. M., Nagy A., Tapson M., Peladeau-Pigeon M., Wolkin T., Brady S. L., Swigert N., Brinkman T. K., Riquelme L. F., Krisciunas G. P., Langmore S. E., & Blumenfeld L. (2015). Prevalence of impaired swallowing with thin and gum-thickened barium stimuli. Poster session presented at the 23rd Annual Dysphagia Research Society Meeting. Chicago, IL, United States. [Google Scholar]
- Steele C. M., Peladeau-Pigeon M., Barbon C. A. E., Guida B. T., Namasivayam-MacDonald A. M., Nascimento W. V., Smaoui S., Tapson M. S., Valenzano T. J., Waito A. A., & Wolkin T. (2019). Reference values for healthy swallowing across the range from thin to extremely thick liquids. Journal of Speech, Language, and Hearing Research., 62(5), 1338–1363. https://doi.org/10.1044/2019_JSLHR-S-18-0448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steele C. M., Peladeau-Pigeon M., Barbon C. A. E., Guida B. T., Tapson M. S., Valenzano T. J., Waito A. A., Wolkin T. S., Hanson B., Ong J. J., & Duizer L. M. (2019). Modulation of tongue pressure according to liquid flow properties in healthy swallowing. Journal of Speech, Language, and Hearing Research, 62(1), 22–33. https://doi.org/10.1044/2018_JSLHR-S-18-0229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stokely S. L., Peladeau-Pigeon M., Leigh C., Molfenter S. M., & Steele C. M. (2015). The relationship between pharyngeal constriction and post-swallow residue. Dysphagia, 30(3), 349–356. https://doi.org/10.1007/s00455-015-9606-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swinnen B., & Robberecht W. (2014). The phenotypic variability of amyotrophic lateral sclerosis. Nature Reviews Neurology, 10(11), 661–670. https://doi.org/10.1038/nrneurol.2014.184 [DOI] [PubMed] [Google Scholar]
- Tabor L., Gaziano J., Watts S., Robison R., & Plowman E. K. (2016). Defining swallowing-related quality of life profiles in individuals with amyotrophic lateral sclerosis. Dysphagia, 31(3), 376–382. https://doi.org/10.1007/s00455-015-9686-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabor L. C., & Plowman E. K. (2017). Dysphagia in amyotrophic lateral sclerosis. In Ekberg O. (Ed.), Dysphagia: Medical radiology (pp. 165–174). Springer; https://doi.org/10.1007/174_2017_137 [Google Scholar]
- Taga A., & Maragakis N. J. (2018). Current and emerging ALS biomarkers: Utility and potential in clinical trials. Expert Review of Neurotherapeutics, 18(11), 871–886. https://doi.org/10.1080/14737175.2018.1530987 [DOI] [PubMed] [Google Scholar]
- Talbot K. (2009). Motor neuron disease: The bare essentials. Practical Neurology, 9(5), 303–309. https://doi.org/10.1136/jnnp.2009.188151 [DOI] [PubMed] [Google Scholar]
- Turner M. R., Bowser R., Bruijn L., Dupuis L., Ludolph A., McGrath M., Manfredi G., Maragakis N., Miller R. G., Pullman S. L., Rutkove S. B., Shaw P. J., Shefner J., & Fischbeck K. H. (2013). Mechanisms, models and biomarkers in amyotrophic lateral sclerosis. Amyotrophic Lateral Sclerosis & Frontotemporal Degeneration, 14(Suppl. 1), 19–32. https://doi.org/10.3109/21678421.2013.778554 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner M. R., Kiernan M. C., Leigh P. N., & Talbot K. (2009). Biomarkers in amyotrophic lateral sclerosis. Lancet Neurology, 8(1), 94–109. https://doi.org/10.1016/S1474-4422(08)70293-X [DOI] [PubMed] [Google Scholar]
- Vacha-Haase T., & Thompson B. (2004). How to estimate and interpret various effect sizes. Journal of Counseling Psychology, 51(4), 473–481. https://doi.org/10.1037/0022-0167.51.4.473 [Google Scholar]
- Viera A. J., & Garrett J. M. (2005). Understanding interobserver agreement: The kappa statistic. Family Medicine, 37(5), 360–363. [PubMed] [Google Scholar]
- Vilardell N., Rofes L., Arreola V., Speyer R., & Clavé P. (2016). A comparative study between modified starch and xanthan gum thickeners in post-stroke oropharyngeal dysphagia. Dysphagia, 31(2), 169–179. https://doi.org/10.1007/s00455-015-9672-8 [DOI] [PubMed] [Google Scholar]
- Waito A. A., Peladeau-Pigeon M., Steele C. M., Tabor L. C., & Plowman E. K. (2018). Hyoid kinematics in patients with amyotrophic lateral sclerosis (ALS): A pilot analysis. Poster session presented at the 26th Annual Dysphagia Research Society Meeting, Baltimore, MD, United States. [Google Scholar]
- Waito A. A., Steele C. M., Peladeau-Pigeon M., Genge A., & Argov Z. (2018). A preliminary videofluoroscopic investigation of swallowing physiology and function in individuals with oculopharyngeal muscular dystrophy (OPMD). Dysphagia, 33(6), 789–802. https://doi.org/10.1007/s00455-018-9904-9 [DOI] [PubMed] [Google Scholar]
- Waito A. A., Tabor-Gray L. C., Steele C. M., & Plowman E. K. (2018). Reduced pharyngeal constriction is associated with impaired swallowing efficiency in amyotrophic lateral sclerosis (ALS). Neurogastroenterology & Motility, 30(12), e13450 https://doi.org/10.1111/nmo.13450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waito A. A., Valenzano T. J., Peladeau-Pigeon M., & Steele C. M. (2017). Trends in research literature describing dysphagia in motor neuron diseases (MND): A scoping review. Dysphagia, 32(6), 734–747. https://doi.org/10.1007/s00455-017-9819-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson E. M., Kulkarni M., Simione M., Rong P., Green J. R., & Yunusova Y. (2019). Detectin gbulbar motor involvement in ALS: Comparing speech and chewing tasks. International Journal of Speech-Language Pathology, 21(6), 564–571. https://doi.org/10.1080/17549507.2018.1557254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yunusova Y., Green J. R., Lindstrom M. J., Ball L. J., Pattee G. L., & Zinman L. (2010). Kinematics of disease progression in bulbar ALS. Journal of Communication Disorders, 43(1), 6–20. https://doi.org/10.1016/j.jcomdis.2009.07.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yunusova Y., Plowman E. K., Green J. R., Barnett C., & Bede P. (2019). Clinical measures of bulbar dysfunction in ALS. Frontiers in Neurology, 10, 106 https://doi.org/10.3389/fneur.2019.00106 [DOI] [PMC free article] [PubMed] [Google Scholar]



 This work is licensed under a
This work is licensed under a