Abstract
Human coronaviruses, especially COVID-19, is an emerging pandemic infectious disease with high morbidity and mortality. Coronaviruses are associated with comorbidities, along with the symptoms of it. SARS-CoV-2 is one of the highly pathogenic coronaviruses that causes a high death rate compared to the SARS-CoV and MERS. In this review, we focused on the mechanism of coronavirus with comorbidities and impairment in multi-organ function. The main dysfunction upon coronavirus infection is damage to alveolar and acute respiratory failure. It is associated with the other organ damage such as cardiovascular risk via an increased level of hypertension through ACE2, gastrointestinal dysfunction, chronic kidney disease, diabetes mellitus, liver dysfunction, lung injury, CNS risk, ocular risks such as chemosis, conjunctivitis, and conjunctival hyperemia, cancer risk, venous thromboembolism, tuberculosis, aging, and cardiovascular dysfunction and reproductive risk. Along with this, we have discussed the immunopathology and coronaviruses at a molecular level and therapeutic approaches for the coronavirus infection. The comorbidities and multi-organ failure of COVID-19 have been explained at a molecular level along with the base of the SARS-CoV and MERS-CoV. This review would help us to understand the comorbidities associated with the coronaviruses with multi-organ damage.
Keywords: SARS-CoV-2, COVID-19, Comorbidities, Multi-organ failure and immunopathology
Abbreviations: ACE, angiotensin-converting enzyme; ACEi, angiotensin-converting enzyme inhibitors; AKI, acute kidney injury; ALT, alanine aminotransferase; ARBs, angiotensin type II receptor blockers; ARDS, acute respiratory distress syndrome; AST, aspartate aminotransferase; CCL2, C-C Motif Chemokine Ligand 2; CCL3, C-C Motif Chemokine Ligand 3; CCL5, C-C Motif Chemokine Ligand 5; CD4+, cluster of differentiation 4; CD8+, cluster of differentiation 8; CDK, chronic kidney disease; CNS, central nervous system; COVID-19, coronavirus disease-19; CRRT, continuous renal replacement therapy; CXCL10, C-X-C motif chemokine 10; DPP-4, Dipeptidyl peptidase 4; DWI, diffusion-weighted images; GGT, gamma-glutamyl transferase; IFN, interferons; IL, interleukin; MERS-CoV, Middle East Respiratory Syndrome associated coronavirus; Mig, gamma interferon; NF-kB, nuclear factor kappa-light-chain-enhancer of activated B cells; RSAD2, radical S-adenosyl methionine domain containing 2; RT-PCR, reverse transcriptase-polymerase chain reaction; SARS-CoV-2, Severe Acute Respiratory Syndrome associated coronavirus; TLR, toll-like receptors; TNF, tumor necrosis factor; TRIF, TIR-domain-containing adapter-inducing interferon-β; WHO, World health organization
Graphical abstract
1. Introduction
In December 2019, a severe pneumonia outbreak by Severe Acute Respiratory Syndrome associated coronavirus (SARS-CoV-2) had supervened in Hubei province, Wuhan, China [1,2]. Among the viruses that belong to Coronaviridae family and pathogenic to human beings, the SARS-CoV-2 has been identified as the third virus after SARS-CoV and Middle East Respiratory Syndrome associated coronavirus (MERS-CoV) to create a pandemic situation [3]. The SARS-CoV-2 belongs to the family Coronaviridae. Genealogical examination of the SARS-CoV-2 revealed that it belongs to the genera beta coronavirus and exhibited 96% homology sequence similarity with Bat SL-CoV; whereas, the bat is an intermediate host or not needs to be elucidated [2]. The diameter of SARS-CoV-2 ranges between 50 and 200 nm [4].
This virus possesses four distinct proteins that provide the virus its structure. The outermost layer consists of glycoprotein spikes followed by an envelope, a membrane, and a nucleocapsid layer towards the inner side. The nucleocapsid layer holds the viral genome, RNA. The nucleocapsid layer, envelope, and the spike membrane provide a protective layer for the viral genome [5]. The RNA of CoVs is a single-stranded positive-sense RNA (+ssRNA), which is ~30 kb possessing 5′-cap and 3′-poly-A tail [6]. After entering the host cell, the SARS-CoV-2 undergoes replication by using the host's machinery and synthesizes N, M, E, and S structural proteins. The negative strand of the RNA genome is produced using a positive-strand as a template. At the time of the process of replication in the host cell, the M protein attaches to the membranes of the endoplasmic reticulum, and the N protein binds to the genome. Nsp proteins help the RNA to assemble into a helical structure which enters the lumen of the endoplasmic reticulum. The viral progenies produced undergo exocytosis and enter the extracellular space [7].
Dr. TedrosAdhanom Ghebreyesus, Director-General of world health organization (WHO) on February 11, 2020, has declared the name of the pandemic condition as coronavirus disease-19 abbreviated as COVID-19. The symptoms of COVID-19 include infection to the lower respiratory tract, pneumonia ranging from mild to severe, dry cough, lymphopenia, fever, and fatigue [6,8]. Upon mild infection, the patients present dry cough, vomiting, diarrhea, fatigue, and fever as symptoms whereas in case of severe infection, one week later the disease onset the condition of the patient may worsen resulting in hypoxemia, septic shock, acute respiratory distress syndrome (ARDS), metabolic diseases and may also lead to death in some cases [9]. Apart from the epidemic condition created by the coronavirus, it has been found that the affected individuals, along with other comorbidities such as cardiovascular diseases, type 1 and type II diabetes, and renal diseases, are found to be severely ill compared to the individuals infected only by the coronavirus. The controversy has been raised when Lei Fang, a postdoctoral researcher, and his colleagues suggested that “Patients with cardiac diseases, hypertension or diabetes who are treated with ACE2-increasing drugs are at higher risk for severe COVID-19 infection and, therefore, should be monitored for ACE2-modulating medications, such as angiotensin-converting enzyme inhibitors (ACEi) or angiotensin receptor blockers” [10].
In response to the above statement, the European Society of Cardiology on March 13 and the American College of Cardiology, the American Heart Association, and the Heart Failure Society of America endowed a common statement on March 17 which mentioned that “since there is no scientific evidence behind ceasing the usage of ACEi and angiotensin type II receptor blockers (ARBs) for the patients affected by COVID-19 possessing other comorbidities such as high blood pressure, diabetes mellitus, cardiovascular problems and renal diseases” [11]. The pandemic condition caused by SARS-CoV-2 has brought a whole world to lockdown. To safeguard the human, from the deadly virus, a lot of research has been carried out.
2. Mechanism of the pathogenesis of COVID-19
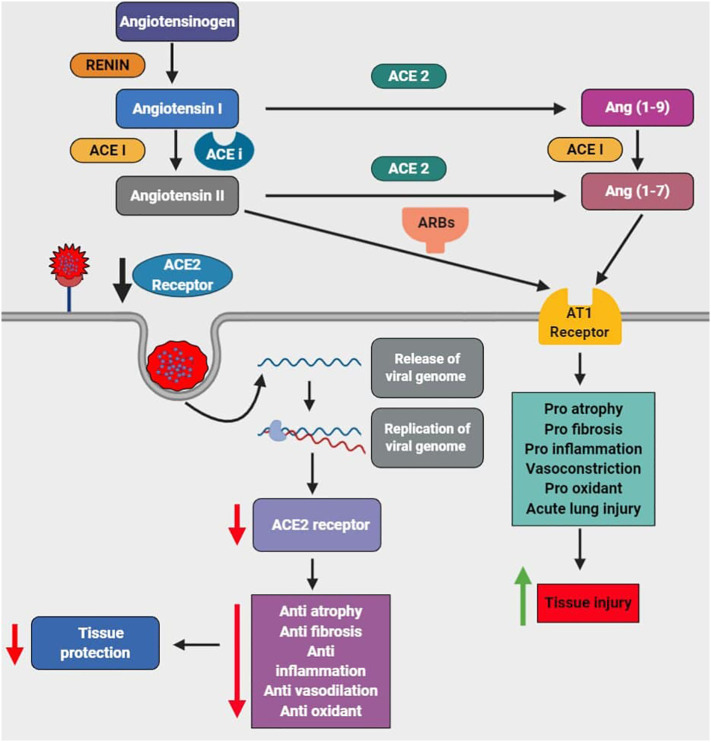
The ACE-2 enzyme is found bound to the cell membranes of the cells in the regions of the heart, lungs, kidney, intestines, and arteries [12]. This enzyme's function is to degrade the angiotensin II peptide, which acts as a vasoconstrictor [13]. Therefore, the ACE2 enzyme is considered to be a potential vasodilator. The SARS-CoV-2 virus binds with the ACE-2 receptor, which mediates the virus's entry into the host cells [14] via the plasma membrane or endocytosis through the release of proteases [2]. The activated immune cells secrete chemokines and inflammatory cytokines into the endothelial cells of pulmonary arteries [15]. The angiotensin-converting enzyme (ACE) is required to convert angiotensin I to angiotensin II [16]. The angiotensin II produced results in vasoconstriction upon the activation of angiotensin II receptors due to which there is an increase in blood pressure [17,18]. Therefore, ACEi and ARBs are used as anti-hypertensive treatment options [18] (Fig. 1 ).
Fig. 1.
The abnormal activation of the Renin-Angiotensin-Aldosterone system (RAAS) pathway via the ACE-AngII-AT1R axis results in vasoconstriction (hypertension) which is treated by the usage of angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs). Since ACE2 is also the binding receptor for SARS-CoV-2 and enhances the viral entry, replication decreases the level of the ACE2, leading to reduced anti-atrophy and anti-fibrosis anti-inflammation, anti-oxidant, and vasodilation further leads to decreased tissue protection. On the other hand, there is a pro-atrophy, pro-fibrosis, pro-inflammation, vasoconstriction, pro-oxidant, and acute lung injury via angiotensin II type 1 receptor which leads to tissue injury.
3. Endemic coronaviruses
There are seven types of HCoV identified till date out of which four of them usually results in mild acute respiratory illness [19]. The four types of HCoV are HCoV-229E (α-CoV), HCoV-NL63 (αCoV), HCoV-OC43 (β-CoV), and HCoV-HKU1 (β-CoV) [20]. The severity of HCoV induced upper respiratory tract infection may range from mild to severe, depending upon the individual's age. And also, those people already having problems related to lung and cardiac may exhibit severe infectious conditions [21]. The remaining three viruses SARS-CoV, MERS-CoV, and SARS-CoV-2, are found to be deadly pathogenic, causing ARDS, hepatic diseases, intestinal diseases, multiple organ failure, and eventually, may lead to death in severe cases [[22], [23], [24]].
4. Severe Acute Respiratory Syndrome associated coronavirus (SARS-CoV)
The SARS- epidemic occurred in the Guangdong Province of Southern China in November 2002 and had spread over >30 countries resulting in 774 deaths [25]. The disease transmission was most likely associated with a zoonotic occurrence in the wild animal markets in China. Bats are considered to be the reservoir hosts for coronavirus. It was reported that civet cats were the intermediate hosts that contacted bats before being sold in the animal market [25]. Soon after the isolation of SARS-CoV, SARS-CoV-like viruses were found in Himalayan palm civets and raccoon dogs, with 99.8% nucleotide homology for human SARS-CoV [26]. SARS-CoV transmission is mainly through direct contact person-to-person, through respiratory droplets, with an incubation time of 2 to 11 days after exposure [20].
Initially, the disease shows muscle aches, headache, diarrhea, chills, and fever as symptoms. After the incubation period, the patient will be present with shortness of breath, dry cough, and high fever, further resulting in severe pneumonia, which may lead to death in some cases. The severity of the disease depends upon factors such as age and other health problems such as cardiovascular diseases, hepatitis, and diabetes mellitus [27]. The SARS-CoV is a member of a β-CoVs group and binds with the ACE2 (zinc peptidase) to make its entry into the host cell. The ACE2 is present on the endothelial cells of respiratory tract epithelium, small intestinal epithelium, epithelia of respiratory tract, immune cells, arterial smooth muscle, arteries, and veins [[28], [29], [30]].
4.1. SARS-CoV – cardiovascular risk
It was reported that SARS induced myocardial infarction and acute coronary syndrome [31,32]. A small group of 75 patients infected with SARS had shown that 2 of the five patients died due to acute myocardial infarction [32]. In another study which was conducted among 46 patients infected with SARS indicated that there were no significant changes in the initial stages of infection and, after 30 days, didn't show any risks related to the cardiovascular system. In another study conducted on 121 patients, it was reported that patients had other comorbidities such as cardiovascular diseases, hypotension, and transient cardiomegaly, particularly tachycardia and transient paroxysmal atrial fibrillation and most importantly tachycardia [33] (represented in Table 1 ).
Table 1.
Differentiating SARS-CoV, MERS-CoV, and SARS-CoV-2 based on the impact of manifestations posed by them at various organ levels.
| Target | SARS-CoV |
MERS-CoV |
SARS-CoV-2 |
|---|---|---|---|
| ACE2 receptor | DPP4 receptor | ACE2 receptor | |
|
Hypotension, transient cardiomegaly, tachycardia, transient paroxysmal atrial fibrillation, myocardial infarction, acute coronary syndrome | Elevated troponin levels, a chronic severe 3-vessel disease with no acute vessel closure characterized by coronary angiography | Decreased levels of ACE2 receptor and increased troponin, resulting in cardiac arrest and cardiac injury. |
|
Hepatization, severe lung consolidation, edema with pleural effusion, mucopurulent material in the tracheobronchial tree, focal hemorrhage, alveolar and septal fibrosis, edema, alveolar hemorrhage, fibrin exudation in alveolar spaces, hyaline membrane formation, and extreme bronchiolar injury indicate the acute phase. | Acute respiratory distress syndrome, septic shock, the mild upper respiratory illness may result in swiftly growing pneumonitis, respiratory failure | Severe acute respiratory syndrome (SARS), acute respiratory distress syndrome (ARDS), focal reactive hyperplasia of pneumocytes with patchy inflammatory cellular infiltration, edema, multinucleated giant cells, protruding hyaline membranes, and proteinaceous exudate. |
|
Acute renal impairment | – | Proteinuria, hematuria, acute kidney injury (AKI) |
|
Hepatitis, liver injury characterized by elevated ALT, and AST levels | Disintegrated death of hepatocytes in the hepatic sinus, penetration of large amounts of activated macrophages and Kupffer cells, elevated pro-inflammatory cytokine responses, and lobular lymphocytes infiltrate, mild cellular hydropic degeneration in hepatic parenchyma and mild portal tract and liver damage indicated by elevated ALT and AST levels. | Liver injury characterized by elevated ALT, GGT, and AST levels. Decreased bilirubin levels. |
|
– | – | Viral load found in the cytoplasm of epithelia of duodenum and rectum, nausea, and diarrhea. |
|
Vision-related issues, the chemotactic attraction of immune cells by the infected brain cells, the presence of a chemokine called Mig found in the patient's brain | Fever, vomiting, confusion and ataxia, bilateral basal ganglia, deep white matter, large area of hypodensity in the proximal half of the corpus callosum up to the mid part of his body and interval multiple patchy hypodensities bilaterally in the periventricular, acute infarction, and bilaterally in the deep watershed and the parasagittal region as well as scattered foci in the cortical and subcortical regions of the temporal and parietal lobes | Nausea, vomiting, and headache |
4.2. SARS – diabetes mellitus
A study was carried out on 26 SARS affected patients. It revealed that the patient's age and other comorbidities such as diabetes, hyperglycemic condition, percentage of lymphocyte, the extent of lung infection, and liver damage were distinct to be taken into consideration. Most of the SARS affected patients who died were old and had other health issues, whereas young patients recovered and didn't exhibit any abnormal health conditions. Out of 26 patients, 11 patients were old and already had diabetes and secondary hyperglycemia. Whereas out of the 15 patients, three patients died, and the remaining 12 patients survived were young [34].
4.3. SARS – kidney risk
SARS virus sequences were found in the distal renal tubules of the epithelium, and aggregates of viral fragments were noticed in the cytoplasm of distal tubular epithelium. The focal hemorrhagic condition was observed in the kidneys of the patients infected by SARS [35] (represented in Table 1). In a study that was conducted among 536 patients infected with SARS, 6.7% of the affected individuals exhibited acute renal impairment 20 days after the infection. Most of the people who died were infected with SARS and had renal impairment than those without renal impairment. Acute renal impairment resulted in the disturbance of pre-renal factors, thereby resulting in multi-organ failure. Age and acute respiratory distress syndrome were the two risk factors determining the progression of acute renal impairment in SARS affected individuals [36].
4.4. SARS-CoV-liver risk
The elevated ALT and AST levels indicate liver damage, which was evident in SARS-CoV infected individuals right from the initial stages of infection. Also, the bilirubin levels were elevated, and the albumin levels decreased, indicating the hepatic damage. Also, the degree of hepatic damage depends on the severity of the disease [[37], [38], [39], [40], [41], [42]]. The ACE2 receptor, which is responsible for the SARS-CoV entry, is widely distributed on the hepatic tissue's endothelial cells. The existence of the viral genome in the vascular endothelial cells and parenchymal cells indicates that SARS-CoV infects the lungs and other organs. The presence of viral load in the hepatic cells was confirmed using RT-PCR technique [12,14,37,43]. Also, the SARS-CoV specific protein 7a has shown to be responsible for causing apoptosis not only in the lungs but also in the other organs, including the liver through the caspase-dependent pathway, thereby resulting in liver injury [44].
The levels of pro-inflammatory cytokines were increased, which indicates that SARS-CoV also results in the inflammation [42]. It was also noted that SARS-CoV patients with predisposed hepatitis were more vulnerable to severe SARS-CoV infection due to the reason that the hepatitis virus exhibits exaggerated replication during SARS-CoV infection [45]. Sometimes, the drugs used to treat viral infections may also lead to liver injury, which is termed as drug-induced liver injury [37].
4.5. SARS-CoV-lung injury
The lungs of SARS-CoV infected patients were examined for histological and pathological changes due to the viral infection [35]. The histological changes include hepatization, severe lung consolidation, edema with pleural effusion, mucopurulent material in the tracheobronchial tree, focal hemorrhage, which was presented by the SARS-CoV affected patients [35,[46], [47], [48]]. The pathological changes include vascular edema, vascular injury, including small vessel vasculitis [49], desquamation of squamous metaplasia, and epithelial cells [50], pulmonary infarcts and vascular fibrin thrombi [51]. BOOP-like lesions such as the proliferation of fiber granulation tissue in alveolar spaces located subpleural and small airways. Also, the severity of the clinical manifestations depends on the stage of disease or onset of the disease which has been classified into three stages., edema, alveolar hemorrhage, fibrin exudation in alveolar spaces, hyaline membrane formation, and extreme bronchiolar injury indicate the acute phase, the first phase during the infection [50] (represented in Table 1).
Secondly, the proliferative phase characterized by cellular fibromyxoid-organizing exudates, widening of septae, and pneumocyte hyperplasia [52]. Formation of alveolar and septal fibrosis, where the severity of the fibrosis depends on the onset and duration of the disease [49,53].
4.6. SARS-CoV-CNS risk
Coronaviruses share a common inclination in infecting the central nervous system in some cases. A case was reported where a 39 years old doctor who got infected with SARS-CoV exhibited usual symptoms and was hospitalized. After hospitalization, he was presented with eyesight problems and later started exhibiting central nervous system symptoms such as delirium and restlessness. After a month, he passed away. When his brain tissue was examined, this revealed the presence of SARS-CoV virus. The doctors reported that he might have got infected while treating the patients, and also the chemotactic attraction of immune cells by the infected brain cells might have injured the brain more than that of the viral infection. The chemotactic attraction was characterized by the presence of a chemokine called monokine induced by gamma interferon (Mig) found in the patient's brain and the bloodstream [54] (represented in Table 1).
4.7. SARS-CoV-immunopathology
The human coronaviruses connected with the viral infection, highly infiltration of inflammatory cells, an enhanced level of the pro-inflammatory, and cytokine storm response, resulting in lung injury at an acute level. This infection reduces the level of the anti-viral cytokines such as interferons (IFN-α/β); upregulation of the pro-inflammatory cytokines such as interleukin 6 (IL-6) and tumor necrosis factor (TNF) and upregulation of the inflammatory cytokines such as C—C Motif Chemokine Ligand 2 (CCL2), C—C Motif Chemokine Ligand 3 (CCL3), C—C Motif Chemokine Ligand 5 (CCL5) and C-X-C motif chemokine 10(CXCL10) [55,56]. Macrophage infected with the SARS-CoV showed an elevated level of the other pro-inflammatory cytokines and IFN. Also, the epithelial cells infected with the SARS-CoV have an enhanced CCL2, CCL3, CCL5, and CXCL10 [56,57].
Along with the chemokines and pro-inflammatory cytokine, there is an elevated level of the IFN inclusive of IFN-α and IFN-γ, stimulatory genes of IFN include CCL-2 and CXCL-10. There is an exacerbation of the IFN, chemokines such as TNF, IFN-γ, IL-6 and CCL5, pro-inflammatory cytokines and stimulatory genes of interferon such as an interferon-induced protein with tetratricopeptide repeats 1) IFIT1, radical S-adenosyl methionine domain containing 2 (RSAD2) and CXCL10 in the TIR-domain-containing adapter-inducing interferon-β (TRIF) and toll-like receptors 3 (TLR3) deficient mice [58]. The deregulation of the cytokine or chemokine response causes the cytokine storm, leading to the immunopathological changes and the SARS-CoV infection.
5. Middle East Respiratory Syndrome associated coronavirus (MERS-CoV)
In Saudi Arabia, in June 2012, a new virus outbreak took place, which caused fever, dry cough, and shortness of breath resulting in respiratory illness [59]. The disease spreads from person to person through direct contact with the infected person. It is believed that the MERS-CoV had originated from bats, and dromedary camels are intermediate hosts for transmitting the disease to humans [60]. The MERS-CoV belongs to the β group of coronaviruses, and it is a distinct virus from SARS and common flu viruses. The incubation period of this virus is 2–13 days, and it enters the human cells through serine peptidase, dipeptidyl peptidase4 receptors [28,61]. The genome of MERS-CoV consists of a single positive-stranded RNA comprising 30,119 nucleotides and 11 open reading frames [62]. The clinical risk factors determining the mortality rate among MERS-CoV infected patients were age, sex, and comorbidities such as cardiovascular diseases, chronic renal diseases, hypertension, diabetes mellitus, cancer, and respiratory diseases [63,64].
5.1. MERS-CoV infection along with other comorbidities
Metabolic disorder related conditions, for example, hypertension, diabetes, obesity, and cardiovascular diseases, together with their inclining conditions, can be connected etiologically to the pathogenesis of MERS-CoV. These conditions act to decline the innate immune system of the host, thereby the inability of the host to fight against the pathogenesis of MERS-CoV. A study conducted on 637 MERS-CoV patients revealed that 15% of them have diabetes, 16% of them are obese, 50% of them had hypertension, and 30% are predisposed to cardiovascular diseases [65]. A 57-year-old diabetic and the hypertensive patient exhibited myocardial ischemia besides edema in the pulmonary tract, elevated troponin levels which returned to normal after 48 h, a chronic severe 3-vessel disease with no acute vessel closure characterized by coronary angiography. Later, he didn't respond and became hypotensive with facial paralysis on the left side [66].
5.2. MERS-CoV-liver injury
The patients infected by MERS-CoV were also presented with alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels above normal. Besides the elevated liver enzymes, the albumin levels were decreased, which serves as an indicator of the severity of the disease [[67], [68], [69]]. The MERS-CoV enters the host cell through the DPP-4 receptor and spreads the infection [70]. The liver is the main organ that expresses high levels of Dipeptidyl peptidase 4 (DPP-4) [71]. This was experimentally proved by infecting the transgenic mice expressing hDPP4 with MERS-CoV which indicated that the liver injury had taken place on the 5th day of the infection as evidenced by the pathological manifestations such as disintegrated death of hepatocytes in the hepatic sinus, penetration of large amounts of activated macrophages and Kupffer cells. The changes related to fat metabolism were observed on the 9th day of infection, and the liver cell necrosis was quite less [72] (represented in Table 1).
The histopathological findings of the hepatic tissue during MERS-CoV infection include infiltration of lobular lymphocytes, mild cellular hydropic degeneration in hepatic parenchyma, and mild portal tract [73,74]. During the acute phase of infection, the levels of pro-inflammatory cytokines such as TNF-α, IFN-γ, IL-17, and IL-15 were significantly elevated [75]. But the question of whether liver injury initiated is due to the viral-mediated pathogenesis or due to the drugs that have been used during the viral infection remains elusive [37].
5.3. MERS-CoV-lung injury
The MERS-CoV viral infection mainly affects the lungs resulting in respiratory illness, which was confirmed by the clinical manifestations present in the affected patients which include acute respiratory distress syndrome, septic shock, the mild upper respiratory illness may result in swiftly growing pneumonitis, respiratory failure (represented in Table 1). All these events may lead to multi-organ failure, eventually producing lethal effects [60,[76], [77], [78], [79], [80]].
5.4. MERS-CoV-CNS risk
A report presented 3 cases that were infected by MERS-CoV and exhibited abnormal neurological findings along with other predisposed co-morbid conditions. A 74 years old patient with co-morbid medical conditions, including dyslipidemia, diabetes, and hypertension, exhibited fever, vomiting, confusion, and ataxia three days later. Computerized tomography exhibited no acute changes whereas presented chronic lacunar strokes. The chest radiograph revealed infiltration in the mid-right lung zone. Decreased motor activity on the left side was observed. He was tested positive for MERS-CoV exhibiting respiratory illness, and then he was admitted to ICU. On day 24, after hospitalization, his medical condition improved confirmed by tracheostomy, which confirmed an improvement in the respiratory status of the patient. The CSF examination revealed lymphopenia and was tested negative for MERS-CoV-2 RT-PCR. In 57 years old patient infected with MERS-CoV exhibited neurological findings apart from other cardiovascular abnormalities. The neurological findings include bilateral basal ganglia, deep white matter, a large area of hypodensity in the proximal half of the corpus callosum up to the mid part of his body, and interval multiple patchy hypodensities bilaterally in the periventricular. MRI examination revealed occipital lobes with a restriction on the diffusion-weighted images (DWI) consistent with acute infarction, and bilaterally in the deep watershed and the parasagittal region scattered foci in the cortical and subcortical regions of the temporal and parietal lobes (represented in Table 1).
The patient experienced multiple cardiac arrests, severe shock, and acute kidney injury and eventually expired. Forty-five-year-old patients with acute kidney injury, diabetes, and hypertension were tested positive for MERS-CoV, and CT scan findings revealed no acute abnormalities but exhibited similar findings consistent with encephalitis. The patient exhibited lymphocytopenia and was tested negative after a few days [66].
5.5. MERS-CoV-immunopathology
Like the SARS-CoV, MERS also has an infection in the airway epithelial cells, which enhances the level of pro-inflammatory cytokines and IFN in a delayed manner [81]. Even in the cells of THP-1, macrophage-derived from the human peripheral blood, dendritic cells showed an elevated level of the chemokines such as CCL3, CCL2, IL-2, IL-8, and CCL-5 and pro-inflammatory cytokines in a delayed manner during MERS-CoV infection [82,83]. The high level of the pro-inflammatory cytokine and chemokine associated with the enhanced level of the monocyte, neutrophil numbers in both the lung and peripheral blood, which leads to immune damage [84].
6. SARS-CoV-2
6.1. SARS-CoV-2 – heart disease
ACE2 plays a vital role in the cardiovascular system and is mainly involved in the functioning of the heart and development of hypertension and type II diabetes. Since ACE2 is the main target for the SARS-CoV-2 to enter inside the host cell and the ACE2 is also highly expressed in the heart apart from the lungs, indicating a chance of developing cardiac issues during SARS-CoV-2 infection. The anti-hypertensive therapy that has been prescribed for the patients is the usage of ACEi and ARBs should be carefully considered. The COVID-19 patients with comorbidities hypertension, taking the drug of ACEi or blocker of angiotensin receptor, need to switch off to other anti-hypertensive medications found to be controversial and require further studies this co-morbid conditions. The severity of the viral infection is higher in patients with comorbidities than those infected only by the virus without any predisposed health issue [85].
The interaction of the SARS-CoV-2 with ACE2 triggers a signal, which might be the reason to develop heart injury through the immune response triggered by T-helper cells creating a cytokine response. The SARS-CoV-2 has a structural similarity with that of SARS-CoV. SARS-CoV caused severe cardiovascular damage during its pandemic period. Therefore, it is essential to investigate SARS-CoV-2 treatment options and ensure a cardio-protective effect. The age and the predisposition of the acute coronary syndrome are the two critical factors which need to be considered in patients affected with COVID-19 [85].
Since the ACE2 is highly expressed in pericytes, the patients infected with SARS-CoV-2 and predisposed with cardiac failure are more susceptible to develop severe cardiac problems [86]. The usage of mineralocorticoids receptor blocker can attenuates oxidative stress, reduces the level of ACE with the augmented production of the angiotensin 1–7 and attenuates the angiotensin II generation and increases the activity of ACE2 The antagonist of angiotensin II type 1 receptor, have modulates the profilin-1 expression and ACE2 [87]. The selective of the angiotensin II activity or synthesis mediated the augmented level of ACE2 expression and its activity in cardiac. Still, the combination of the lisinopril and losartan increases the activity of ACE2 but not ACE2 mRNA. The inhibition of ACE results in reduced formation of angiotensin II and metabolism of angiotensin 1–7, the angiotensin II type 1 receptor antagonist increases the metabolism of angiotensin II via ACE2 [88]. SARS-CoV 2 enters into the cell via the ACE2 receptor; this causes the decreased level of the ACE2 due to the binding of the spike protein to the ACE2 and no changes in ACE. This process causes the virus's entry, replicates it, and causes tissue injury via decreasing anti-atrophy, anti0-fibrosis, anti-inflammation, anti-oxidant, and vasodilation. On the other hand, the angiotensin II type 1 receptor causes pro-atrophy, pro-fibrosis, pro-inflammation, pro-oxidant, severe myocardial remodeling, vasoconstriction and vascular permeability which leads to tissue injury [89] (represented in Table 1).
Therefore, the use of ACEi, ARBs, and mineralocorticoids has shown to have an impact on the cardiac patients affected by COVID-19, but the exact mechanism behind it is not clear. Aliskiren, a direct inhibitor of renin, resulted in reduced expression of ACE2 besides reducing the blood pressure that has been elucidated in diabetic nephropathy animal models [90]. Therefore, renin blockers, especially aliskiren, need to be further investigated as a treatment option for COVID-19 patients with comorbidities [91].
6.2. SARS-CoV-2 – gastrointestinal risk
The ACE2 is also highly expressed in the small intestine and large intestine of the gastrointestinal tract. The S protein present on the envelope of the SARS-CoV-2 ensures higher binding affinity, thereby mediating the entry of the virus into the host cell. The presence of SARS-CoV-2 in the gastrointestinal tract was confirmed by nucleocapsid staining of the virus. The virus was found to be visualized in the cytoplasm of epithelia of duodenum and rectum [92] (represented in Table 1). Apart from the pulmonary infection, the COVID-19 affected patients also exhibited vomiting, abdominal pain, and diarrhea, which was first reported in a 35 years old man from the United States. He was presented with nausea and diarrhea on 2nd day of the infection and was hospitalized. Stools examination revealed the presence of SARS-CoV-2 RNA, which was confirmed by using reverse transcriptase-polymerase chain reaction (RT-PCR) on day 7 of infection [93]. Out of 171 COVID-19 affected children, 6.4% of them have shown vomiting, and 8.8% of them and 3 out of ten affected children have shown diarrhea as symptoms [94]. Also, previously there were two COVID-19 young cases reported with vomiting and diarrhea.
In China, from 552 hospitals, 1099 cases were reported, of which 3.8% and 5% were diagnosed with diarrhea and vomiting as symptoms [95]. According to another report, out of 140 COVID-19 patients from Wuhan, 39.6% exhibited gastrointestinal symptoms apart from 5% exhibiting vomiting, 12.9% exhibiting diarrhea, and 17.3% exhibiting nausea [96]. It was reported that 17 patients had shown a negative result when tested using pulmonary samples, but when tested using rectal swabs have shown to have a positive result [96].
Similarly, 8 out of 10 children affected by SARS-CoV-2 had shown dual results, negative results when tested by nasopharyngeal samples, and positive results when tested using a rectal swab [97]. The percentage of patients exhibiting nausea, vomiting, and diarrhea as symptoms vary between the cohort groups, whereas, in the case of certain groups, these symptoms contributed very few percent. In contrast, in the case of the other groups, a high percentage of these gastrointestinal symptoms have been recorded.
6.3. SARS-CoV-2- kidney risk
It is suspected that whether the patients with predisposed kidney issues or COVID-19 induced acute kidney injury is fatal to the affected patients. In an article posted on March 19 by HospiMedica International staff writers, it was mentioned that patients affected by COVID-19 developed proteinuria estimated by the elevated levels of albumin in urine were diagnosed in the initial stages of infection. The majority of the affected members, i.e., 34% out of 59 patients, developed proteinuria where the elevated albumin levels serve as a biomarker for kidney damage, and 63% of the patients exhibited proteinuria and hematuria characterized by the presence of blood in the urine. In another study conducted on 710 patients hospitalized due to COVID-19. The impairment in the functioning of the kidney was observed in 27% of the 59 patients and 66% of the corona infected patients. In a later study conducted on COVID-19 affected patients from 710 hospitals, 26.7% exhibited only hematuria, and 44% exhibited proteinuria and hematuria. The impaired kidney function was found in 15% of the study population [98].
According to an article published by Wang L and his colleagues reported that out of 116 COVID-19 positive cases from Renmin Hospital of Wuhan University, out of whom 111 didn't have any kidney issues in their history and the remaining five patients had chronic kidney disease (CDK). Also, out of the 111 patients, only 10.8% of the patients exhibited a slight increase in creatine or blood urea nitrogen levels, and 7.2% of the patients had shown a minute raise in the albuminuria levels. None of the patients out of 111 didn't exhibit any acute kidney injury (AKI) after the COVID-19 infection. The remaining five patients who were already undergoing continuous renal replacement therapy (CRRT) also didn't show any fatal effects after COVID-19 infection confirmed by diagnosing the renal indicators [99]. Out of 701 patients, 2% of the patients were found to have CKD, and the average lymphocytic number was found to be reduced than the normal. High sensitivity C-reactive protein levels were significantly increased. In patients reported with high levels of serum, creatine indicated high levels of serum lactose dehydrogenase. Among hospitalized patients, patients with high levels of serum creatine were mostly old and male sex. Also, they exhibited lower platelet and lymphocytic count and elevated leukocytic count. The patients with elevated procalcitonin also exhibited elevated levels of aspartate aminotransferase and lactate dehydrogenase. Acute kidney injury was significantly higher in those patients with high serum creatine levels. AKI eventually results in mortality [100] (represented in Table 1). Therefore, advanced identification of kidney diseases in patients affected by COVID-19 may help the clinicians to reduce the mortality rate due to comorbidities such as chronic kidney disease, acute kidney injury, proteinuria, and hematuria.
6.4. SARS-CoV-2 – diabetes mellitus
Patients with diabetes mellitus exhibit high levels of ACE2, and treatment with the inhibitors of ACE and ARBs augments the expression of ACE2. Diabetes condition may lead to the suspicion that the excessive expression of ACE2 aids the entry of SARS-CoV-2 into the host cells resulting in COVID-19. Since diabetes mellitus and hypertension conditions are treated, ACE2 increasing drugs are having a severe risk of COVID-19 [10].
Simultaneously, the elevated expression of ACE2 ensures the entry of the SARS-CoV-2, attaching to the ACE2 enzyme with the help of S protein present on its envelope's surface. There forth, it became controversial whether COVID-19 affected patients who are predisposed with diabetes mellitus should be resumed with their treatment with ACEi and ARBs or not. Also, if the medication is abruptly ceased, there are chances that the patient may die due to hypertension instead of COVID-19. For this reason, it has been suggested that there is no need to stop the medication of a diabetic patient even though if he is affected by COVID-19 because there is no scientific evidence available conducted on human subjects regarding whether to use ACEi and ARBs [10].
6.5. SARS-CoV-2-liver injury
The SARS-CoV-2 infection has shown to increase the levels of ALT, AST, and bilirubin levels, indicating injury to the hepatic tissue [9,95,101,102]. Apart from these liver serum enzymes indicating hepatic damage, the levels of albumin were significantly decreased, indicating the severity of the infection [101]. The elated levels of gamma-glutamyl transferase (GGT) were observed only in patients with severe cases. In contrast, thealkaline phosphatize (AKP) levels remained unchanged irrespective of the severity of the infection [37] (represented in Table 1).
SARS-CoV and SARS-CoV-2 share the similarity in making an entry into the host cell through the ACE2 receptor. In addition to the type 2 cells in the lungs, both the bile duct cells and hepatocytes also express ACE2, which might be the reason that liver injury occurs during SARS-CoV-2 infection [103]. The bile duct epithelial cells are involved in immune responses and the regeneration of the hepatocytes [104]. Besides possessing a high number of ACE2 on its surface, bile duct cells are more prone to the SARS-CoV-2 infection resulting in liver injury, which indicates that the hepatocytes are not directly involved in the hepatic injury [37]. The histopathological manifestations of SARS-CoV-2 infected patients revealed mild lobular and portal activity and restrained microvascular steatosis [105]. The degree of liver damage is directly proportional to the severity of the SARS-CoV-2 infection. Still, the reason for the liver injury remains elusive. A recent study reported that the use of anti-viral medication lopinavir might result in hepatic damage [102]. Therefore, whether the liver injury is due to the virus entry or due to the drugs used as anti-viral medication needs to be elucidated.
6.6. SARS-CoV-2-lung injury
It was possible to study the lung pathology of COVID-19 patients diagnosed with COVID-19 retrospectively after undergoing lung lobectomy surgery for adenocarcinoma. Also, the patients didn't present pneumonia at the time of surgery, indicating the initial stages of COVID-19. A female patient who was 80 years old was hospitalized due to an irregularly shaped solid nodule in the right middle lobe. On a postoperative day 1, a CT scan was performed, which indicated a few clinical manifestations. Later, the patient was presented with increased leucocytes, decreased lymphocytic count, chest tightness, wheezing, dry cough, and difficulty in breathing. Later findings confirmed that SARS-CoV-2 infected her from a patient in the same room who was already affected. Another patient who was 73 years old and had a medical history of hypertension from 20 years who underwent surgery for adenocarcinoma was discharged on the 6th-day post-operation. On postoperative day 9, the patient exhibited chest tightness, muscle pain, dry cough, and decreased lymphocytic count. His CT-scan findings revealed for viral pneumonia, and he was also tested positive for the 20,189-nC0V test. He was admitted to the infectious disease ward. After, 20 days he was discharged as he recovered. Histopathological findings showed focal reactive hyperplasia of pneumocytes with patchy inflammatory cellular infiltration, edema, multinucleated giant cells, protruding hyaline membranes, and proteinaceous exudates [106] (represented in Table 1).
The patients, who are recovered from COVID-19, have a possibility of chronic lung damage. Out of 70 COVID-19 pneumonia patients, 66 patients have lung damage in a different manner, which was determined by CT. This damage is in a diverse range; in the alveoli, there is a hardened tissue with the dense clumps which block the vessels in the tiny air sacs, which absorb the oxygen and cause tissue lesions. This tissue lesion acts as a sign of the long-term lung disease. Similar reports have been found in both the SARS CoV and MERS. The patients admitted with lung pneumonia shows damage in both the lungs. The CT scan of the COVID-19 patients indicates that there is a tissue lesion found over the alveoli and which can develop into the scars. There are >2.9 million people have COVID-19 disease worldwide, according to April 27. 80% of the patients are having a high risk of problems, such as from breathing to respiratory failure [107].
6.7. SARS-CoV-2-CNS risk
Mathew reported that it might be due to the obstructions in the nervous system, which may restrict the entry of air into the lungs [108]. The virus enters the medullary neurons during the latency phase. A possible mechanism explained was that the SARS-CoV-2 might enter the olfactory lobe of the brain through nasal chambers upon inhalation and may lead to inflammation and demyelination. Later, it spreads to the whole brain due to its ability to cross the blood-brain barrier. Some neurological symptoms exhibited by the COVID-19 infection patients include nausea, vomiting, and headache (represented in Table 1). Since the SARS-CoV-2 possess the ability to cross the blood-brain barrier, it is important to design the drugs in such a way that they can cross the blood-brain barrier and scavenges the brain from viral infection. Also, it is essential to note that the respiratory illness accompanies neurological invasion by the virus, therapeutic care must be taken to prevent the viral entry into the central nervous system (CNS) [7].
6.8. SARS-CoV-2 – immunopathology
SARS-CoV-2 infection mediates immune response, both innate and adaptive. The enhanced level of the innate response and decreased level of adaptive causes tissue damage. Upon viral infection, there is an immune response. T cells, especially the cluster of differentiation 4 (CD4+) and a group of differentiation 8 (CD8+), have an anti-viral role. CD8+ T cells are highly cytotoxic, and they killed infected cells. This T cell induces the pro-inflammatory cytokines through nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB) signaling. This process further activates the cytokines and chemokines [7]. Similar to SARS-CoV, MERS, SARS-CoV-2 infection in the immune effector cells are also having the enhanced level of the cytokines such as IFN-γ, IFN-α, IL-6, IL-1β, IL-18, IL-12, TGFβ, IL-33 and TNF-α and chemokines such as CCL2, CCL5, CCL3, CXCL10, CXCL9, and CXCL8. Similar to this, severe MERS-CoV infected patient's serum shows an increased level of the cytokines and chemokines. This cytokine storm further causes multiple organ failures to further leads to the death of the SARS-CoV-2 patient [109].
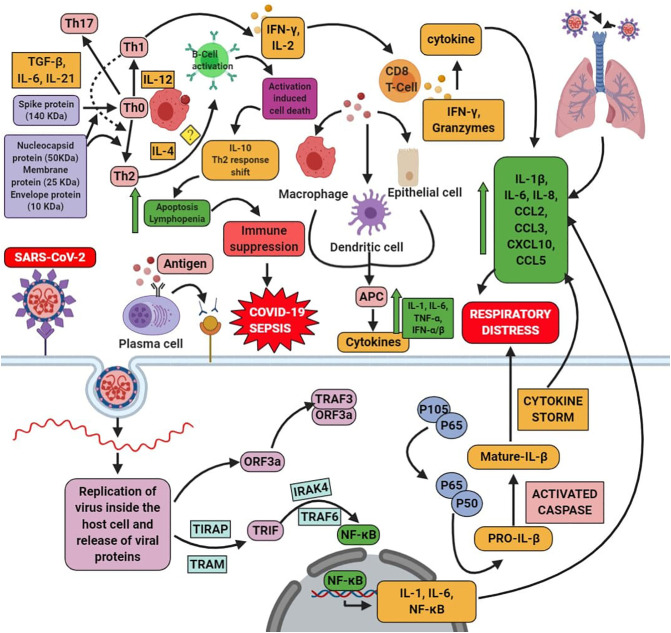
On the other hand, the T-cells are having four types of it, such as Th1, Th2, Th17 (inflammatory), and anti-inflammatory Tregs. These Th2 cells generate IL-4 cells. The compound which can block the Th1 (Boost immune system) tends to augments the level of the Th2 (anti-inflammatory response). IL-4 triggers the Th2 cells and obstructs the Th1 response. This Th2 helps to repair form tissue dysfunction and another type of malfunction. It is found to be good that the increased level of the Th1 and Th17 plays an important role in hyperactive immune response and during autoimmune conditions. It is showed that there is an induction of the Th1/Th17 and the production of antibodies upon COVID-19 condition [110]. Contradict to this; the patient has a high level of Th2 in need of intensive care [111]. During the MERS condition, there is a reduced level of both the Th1 and Th2, which further enhances the inflammatory cytokines and causes more infection and death [112]. SARS patients who died with an older age have higher cytokines of Th2, such as IL-4, IL-10, and IL-5 [113]. This incidence supports the increased Th2 level of COVID-19 patients who needs intensive care. The mechanism behind this is based on the molecular weight of the protein of SARS-CoV2; the activity of the immune system is changed. The recent research by the F. Javier Martín Oncina in 2020 showed that based on the antigen molecular weight, the host's immune response is varied. The proteins of the SARS-CoV2 are spike protein with 140 kDa, Envelope protein with 10 kDa, Membrane protein with 25 kDa, and nucleocapsid protein with 50 kDa. The protein with more than >70 kDa, such as spike protein, cannot be arrested by the B-cell, thereby it activates the inflammatory response through the Th1 response with the macrophages. This process occurs in the subcapsular sinus and through dendritic cells phagocytosis. The other proteins with <70 kDa are found in the protein of SARS-CoV2, which activates the receptor of B-cell and stimulates Th2 immune response. The unremittingviral particle overload, which is unrestricted by the viral cell lysing, will be terminated by the Th1 (pro-inflammatory response). Further, it increases the protein level with a weight of <70 kDa stimulates the B cells in large amounts. This induction of the B-cells leads to the condition called as Activated Induced Cell Death (AICD), which speediness the apoptosis and release of pro-inflammatory cytokines cause lymphopenia, thereby it produces IL-10, augments the shift of Th2 form Th1 response which causes further suppression of immune system, COVID-19 sepsis [114]. Correlative to that that COVID-19 patients with the high level of Th2 immune response need intensive care [111], which shows that they might be having apoptosis, lymphopenia, and suppression of immune system further causes COVID-19 sepsis [114]. Though the cell death is via Th2 immune response but the level of IL-4 is not elucidated yet. The role of IL-4 and its mechanism behind COVID-19 remain elusive (Represented in Fig. 2 ).
Fig. 2.
SARS-CoV-2 enters into the host cell via binding with the cellular receptor ACE-2. It undergoes the fusion with the joining of the plasma membrane and the virus. Then it undergoes the process of the proteolytic cleavage; further, it will undergo replication and lead to the formation of the proteins. This process activates the signaling pathway, such as the NF-kB pathway, via TRIF. The interaction between the cells and the virus activates many cytokine storms. On the other hand, once the virus enters into the cell, the antigen present in that would undergo the antigen presentation cells (APC); further, this stimulates the humoral and cellular immunity. COVID-19 infects the macrophage cells, which presents to the T cell, further, which leads to the activation, differentiation of T-cells, along with the production of cytokines. This shows the negative action on the activation of CD8 T cells. Thus the mediator produced by the CD8+ T cells clears the infection of SARS-CoV. Upon COVID-19 infection, there is a reduced CD4+ and CD8+ cell level, further increasing the cytokine level in the cells, which triggers the inflammation. This mediates the production of the cytokine storm via secreting chemokine and cytokines such as IL-1β, IL-6, TNF-α, IL-8, IL-21, CCL2, CCL3, CCL5, CXCL10, TNF-β, and MCP-1 and triggers the tissue injury. On the other hand, based on the weight of the protein of SARS-CoV2, there is an activation of Th1/Th17 (boosts immune system) when the spike protein is >70 kDa. In case of the Th2 (anti-inflammatory) is activated by the majority of the protein with <70 kDa, then the activation of the B-cell receptor, which causes activation-induced cell death such as apoptosis and lymphopenia which is by releasing IL-10, shifting of Th1 to Th2 immune response, suppression of the immune system and further leads to the COVID-19 sepsis. The role of IL-4 has not been elucidated yet. It remains unanswerable upon COVID-19 condition with the mechanism of Th2 and Th1/Th17.
The wet lung is also known as acute respiratory distress syndrome (ARDS). The patient with COVID-19 has lung edema, and it needs more attention clinically. Lung edema is found to be a symptom of acute lung injury, which development to hypoxemia condition and which leads to acute respiratory distress syndrome (ARDS). The patients who are found with ARDS have more mortality. Though there is no proper treatment or vaccine treatment, ventilation with the therapy of oxygen and ventilation mechanically is mandatory. Sometimes, the treatment with the glucocorticoids (systemic or locally) would help attenuate lung edema and pulmonary inflammation [115]. But there is a need for more research to correlate the cytokine storm with wet lung pneumonia in COVID-19 patients.
6.9. SARS-CoV-2 – ocular risk
The ACE2 receptors are also widely distributed in the superficial parts such as conjunctiva and cornea inferior parts such as iris, ciliary body, aqueous humor, trabecular meshwork, retina, and non-pigmented ciliary epithelium of the ocular globe which is mentioned out to be the intraocular renin-angiotensin system. Therefore, the distribution of ACE2 and TMPRSS2 protein must be taken into consideration to study the passage of infection through the ocular route [116]. Also, there is a chance for the virus to enter into the nasal cavity, which causes respiratory illness and the digestive system through the lacrimal canaliculi aided by the effectiveness of the tear film [117,118]. Out of 38 COVID-19 positive patients, 12 exhibited clinical manifestations, which include chemosis, epiphora, increased secretions, conjunctivitis, and conjunctival hyperemia. Researchers reported that patients exhibiting ocular symptoms would have elevated neutrophilic and lymphocytic counts, elated procalcitonin, lactate dehydrogenase, and C-reactive protein levels than those without ocular manifestations. The patients with ocular manifestations exhibited severe illness [119].
Even though there is no such evidence, the presence of ACE2 and TMPRSS2 protein in the corneal limbal stem cells suggests that the SARS-CoV-2 may enter the bloodstream and travel to the other parts including the brain [120]. Even though there is no viral load in the conjunctiva of the SARS-CoV-2 infected patients without conjunctivitis, the risk of transmission of the virus through tears is low [121]. The exact mechanism behind the ocular risk and SARS-CoV-2 need to be elucidated further.
6.10. SARS-CoV-2 – cancer risk
Cancer patients are highly vulnerable to SARS-CoV-2 infection. 18 out of 1590 COVID-19 positive cases exhibited cancer history. 5 out of 18 patients suffered from lung cancer, 4 out of 18 patients had no past information on medication and had undergone surgery or chemotherapy in the previous month, and the remaining 12 were cancer survivors. The COVID-19 was highly severe in cancer patients than non-cancer COVID-19 affected patients. Also, COVID-19 patients with cancer exhibited a severe chain of symptoms than those without a history of cancer. Also, patients with lung cancer did not exhibit severe symptoms when compared to those with other types of cancers [122].
In a retrospective study conducted among 1276 patients, 28 patients who have cancer were selected for the study. The mean age group people belonged to the elderly category, and 60.7% of them were males. Lung cancer was the most frequent type, followed by oesophageal and breast cancers among cancer affected patients. 10 out of 28 patients were presented with IV stage cancer. 28.6% of the patients developed COVID-19 while receiving antitumor therapy, whereas 71.4% of them developed due to the presence of infection in their vicinity. Besides, 39.2% of the patients exhibited chronic co-morbid conditions apart from cancer. 28.6% of patients died, and the remaining patients developed serious consequences even after receiving anti-viral and corticosteroids. The most common complication developed by the patients includes septic shock, acute myocardial infarction, and ARDS [123]. Therefore, more focus should be laid on the research for medication and treatment options for COVID-19 affected cancer patients. More studies are required to determine the exact mechanism behind cancer and SARS-CoV-2.
6.11. SARS-CoV-2 – tuberculosis
A study was conducted in February 2020, Shenyang, China, which involved 36 COVID-19 positive patients and was classified based on the severity of the symptoms as mild and critical. The results of these patients were compared with that of the patients affected by pneumonia caused by Mycobacterium tuberculosis from Shenyang chest hospital. The study was conducted to determine which one caused severe pneumonia, either the viral or the bacterial strain. Then the severity of tuberculosis was compared between the mild and severe COVID-19 cases and confirmed that tuberculosis due to mycobacterial strain affected patients are more susceptible to COVID-19 and enhance the severity of COVID-19 [124]. Patients with HIV undergoing treatment with antiretroviral medicines exhibit weak immune systems and are more susceptible to viral infections. Also, until now, only one HIV case had been reported with COVID-19 infection from china. The patient recovered from the infection the same as that of those without HIV. There is no evidence on whether HIV patients are more susceptible to COVID-19 infection [125].
The COVID-19 associated TB is found in 94th day of the nonexistence of public health interventions and 138th day in the occurrence of interventions. This occurs at the peak of an outbreak where the implementation of the intervention, out of it 11,066, is with the presence of interventions. 27,968 COVID-19 cases found in the absence of interventions, out of it 14,823, are with no interventions. This scenario shows that there is a need to take preventive measures of TB associated with COVID-19 patients. They need a prior diagnosis and proper management [126]. The main reasons for mortality are age, sex, and other co-morbid factors such as diabetes, cardiovascular diseases, and renal diseases are the main reasons for mortality.
6.12. SARS-CoV-2 – Venous thromboembolism
People affected with pneumonia are more prone to venous thromboembolism. Out of 1026 COVID-19 patients, 40% of them exhibited high risk, and 11% of them at high risk of developing venous thromboembolism without any prophylaxis. Prophylactic measures can prevent venous thromboembolism. Only 7 out of 140 patients at high risk for venous thromboembolism had recorded about the anticoagulation treatment. And 44 out of 407 exhibited a higher risk for bleeding. In these patients, the mechanical compressions such as intermittent pneumatic compression and elastic compression stockings are instructed, and the duration and dosage of anticoagulants should be adjusted. Also, the COVID-19 patients, along with other co-morbid conditions such as cardiovascular manifestations, respiratory failure, renal and hepatic problems, can affect the bleeding status and venous thromboembolism. Therefore, further investigations are required to reduce the risk due to venous thromboembolism and bleeding in the case of COVID-19 patients [127].
6.13. SARS-CoV-2- reproductive risk
Viral infections during the first 20 weeks of gestation have shown to be more severe. The infections, such as SARS and MERS, which occurred due to coronaviruses, had reported an increased risk for pregnancy-related mortality and morbidity. Such cases among COVID-19 affected patients are not yet reported. Nine women who are in their third semester and infected with COVID-19 were reported to have outcomes as that of non-pregnant COVID-19 affected adults. In another study, out of 33 newborn infants, three infants were shown to be tested positive for COVID-19 who got the disease transferred from their mother through intrauterine vertical transmission mode [128].
6.14. SARS-CoV-2 – mental illness
According to a report, out of a group of 50 COVID-19 cases from a psychiatric hospital in Wuhan, China has uplifted the worry about the role of mental disorder in coronavirus transmission. The conditions include incomplete or no awareness of the risk, inability to have personal protection, and cognitive impairment. The treatment has become more challenging for people with COVID-19 infection and health disorders as co-morbid conditions [129].
6.15. SARS-CoV-2 – aging and cardiovascular risk
It has been reported that the expression of ACE2 reduces with the increasing age. Since young patients possess high levels of ACE2, they are more prone to COVID-19 infection. Whereas, in old age people other than reduced ACE2 levels, other co-morbid conditions such as cardiovascular problems, diabetes, and hypertension might be present. The medication prescribed for these patients helps in elevating the ACE2 levels, which might enhance the viral entry. Though the treatment reduces the co-morbid severities, the viral infection is enhanced in those patients. Therefore, it is conclusive that young patients, even though they are more susceptible to the COVID-19 infection, they are less severely affected and exhibit elevated angiotensin II signaling. Whereas, the older patients who exhibit low levels of ACE2 and higher angiotensin II signaling leads to hypertension. The usage of ACEi and ARBs help in promoting the vasodilatory effects by elevating the ACE2 levels, thereby becoming susceptible to COVID-19 infection and are severely affected [130].
7. Therapeutics for coronaviruses
Ribavirin, an anti-viral agent, is a nucleoside analog and is used for the treatment of SARS-CoV infection. The mechanism of action of this drug includes that the drug inhibits the DNA and RNA replication, disrupting the synthesis of guanosine triphosphate by the inhibition of inosine monophosphate dehydrogenase enzyme [[131], [132], [133]]. 5′ triphosphate metabolite of ribavirin, which inhibits the virus's polymerase activity, viral capping inhibition, and induces lethal mutagenesis in the RNA genome of the virus, resulting in the anti-viral effect of ribavirin [134]. The route of drug administration includes aerosol, oral, and intravenous. This drug is used to treat SARS-CoV infection. It has resulted in severe adverse effects, including anemia in about 27–59% of tested patients, which further led to dreadful outcomes such as tissue hypoxia [[135], [136], [137], [138]]. Also, it elevated the levels of transaminases and resulted in hypocalcemia, hypomagnesemia, developed the risk of teratogenicity and bradycardia [139].
Lopinavir and ritonavir, protease inhibitors are used in combination to treat SARS-CoV as the drugs were effective against the virus [135]; whereas, this combination is usually used in HIV treatment. The ritonavir inhibits the CYP3A metabolism of lopinavir, thereby making the drug available for a longer time by increasing the serum concentration. The lopinavir and ritonavir exhibited very few adverse effects, and beneficial outcomes of this combination of drugs include decreased viral load, increase in the lymphocytic count, and a reduced need for “rescue” pulse steroid doses [140]. Viral infection activates the immune system. The corticosteroids have been used to eliminate and inhibit the cytokine storm and further, which has been initiated by the virus. The viral infection affects a cascade of the immune system, which leads to the production of cytokines such as tumor necrosis factor, IL-1, IL-6, and interferon-gamma, which results in injury to the tissue [141,142].
Another possible treatment is the usage of convalescent plasma where the plasma from a recovered patient is transferred to an infected patient, which results in the adaptive immune response against the infectious agent since the recovered patients have antibodies against the infectious agent. The convalescent plasma had been shown as a better treatment option for SARS-CoV affected patients. This treatment reduced the mortality rate, improved the medical condition of the patient [[143], [144], [145], [146]].
According to the Chinese report, in-vitro treatment of SARS-CoV-2 with hydroxychloroquine had resulted in the inhibition of SARS-CoV-2 growth. Along with hydroxychloroquine, azithromycin was also administered at a dosage of 500 mg, the patients treated with a combination of hydroxychloroquine and azithromycin exhibited 100% cure and tested negative for PCR results; whereas, the patients treated with hydroxychloroquine alone exhibited only 57.1% success rate. This combinational therapy might be an approachable way to cure COVID-19 and needs to be tested over a large sample size [147]. Another drug combination, which includes lopinavir and ritonavir, had no in-vitro evidence for COVID-19. Also, the clinical trials are being carried out on malarial drugs chloroquine and hydroxychloroquine (represented in Table 2 ). Some scientists say that these drugs are not recommendable against SARS-CoV-2. Since there are no scientific proofs available and it might be harmful, leading to heart arrhythmia.
Table 2.
Elucidating various drugs used to treat SARS-CoV, MERS-CoV, and SARS-CoV-2, along with their mechanism of action and adverse effects.
| S. no. | Drug | Effective against | Mechanism of action | Adverse effects | References |
|---|---|---|---|---|---|
| 1. | Ribavirin | SARS-CoV | Anti-viral nucleoside analog – inhibits the viral genomic replication | Anemia, hypocalcemia, hypomagnesemia, developed the risk of teratogenicity and bradycardia | [118]. |
| 2. | Lopinavir | SARS-CoV | Protease inhibitors | Diarrhea, nausea, vomiting, headache | [122] |
| 3. | Ritonavir | SARS-CoV | Protease inhibitors | Diarrhea, nausea, vomiting, headache | [122] |
| 4. | Methylprednisolone | SARS-CoV | Anti-inflammatory effect | The onset of diabetes, avascular necrosis, osteoporosis. | [132,133] |
| 5. | Corticosteroids | SARS-CoV | Inhibits pro-inflammatory effects | Aspergillosis and fungal superinfection | [49,134] |
| 6. | Methylprednisolone (0.5–1 mg/kg) followed by hydrocortisone (100 mg/8 h) and pulse doses of methylprednisolone (0.5 g for 3 d) | Lung diseases | – | – | [124] |
| 7. | Convalescent plasma | SARS-CoV, MERS-CoV, SARS-CoV-2 | Adaptive or passive immune response | – | [135,139] |
| 8. | Lopinavir/ritonavir | MERS-CoV, SARS-CoV-2 | Not effective against MERS, anti-viral therapy for pneumonia caused by SARS-CoV-2 | Diarrhea, nausea, vomiting, headache | [140] |
| 9. | Pegylated interferon-α | Hepatitis-C, MERS-CoV | Inhibits DNA synthesis | Diarrhea, nausea, vomiting, headache | [140] |
| 10. | Lopinavir/ritonavir and IFN-β | MERS-CoV | Improves pulmonary, reduce viral load, and necrosis in lungs and extrapulmonary tissues | Diarrhea, nausea, vomiting, headache | [143,144] |
| 11. | Hydroxychloroquine | SARS-CoV-2, Malaria | Blocks maturation of endosome, maturation and its function by inhibiting the release of virions in the infected cell | Diarrhea, nausea, vomiting, headache, dizziness | [112] |
7.1. SARS-CoV-2 and anti-TNFα treatment
The SARS-CoV-2 mild/moderately affected patients exhibit symptoms such as dry cough, fever, and shortness of breath. In the case of severely affected individuals, hyperactivation of immune cells leads to the cytokine storm, which is created upon SARS-CoV-2 infection, resulting in the inflammation at the targeted tissues. Therefore, inhibiting the pro-inflammatory candidates might be a potential therapeutic option to treat COVID-19 [148]. Anti-TNF therapy was approved and has been utilized for 20 years as a promising option to treat autoimmune diseases such as ankylosing spondylitis, rheumatoid arthritis, and inflammatory bowel disease [149]. TNF is the key enhancer of inflammation and also present in the blood of COVID-19 severely affected patients [150]. Therefore, prescribing anti-TNF therapy may help to prevent the aggression of the infection. It has been reported that patients with inflammatory bowel disease contracted with COVID-19. One hundred sixteen patients were on anti-TNF therapy, of whom 99 recovered, and only one patient died. Therefore, the usage of anti-TNF therapy may stand as a promising therapy to prevent the progression of the viral infection by inhibiting the key pro-inflammatory cytokine TNF [148].
7.2. SARS-CoV-2 and soluble ACE2 receptor
ACE2 is required for the degradation of Ang II to Ang (1–7), reducing the blood pressure through vasodilation. ACE2 exists as both membranous forms, which are found to be anchored to the plasma membrane and the soluble form, circulating in the bloodstream but at a lower proportion [151,152]. Besides serving as an entry point for SARS-CoV-2, the higher levels of ACE2 are required by the hypertension patient to reduce the blood pressure. Therefore, the ACE2 levels need to be maintained besides inhibiting the entry of SARS-CoV-2. Thus, the usage of human recombinant soluble ACE2 (hrsACE2) which exhibited positive results in the phase I and phased II clinical trials to elevate the soluble form of ACE2that competes with the membranous ACE2 for binding with SARS-CoV-2 and inhibits the viral entry and replication can be considered as a therapeutic option to treat COVID-19. Besides inhibiting the viral entry, it also sustains the ACE2 levels required for the degradation of Ang II [153,154].
Currently, hrsACE2 has been approved as a treatment option of ARDS. African green monkey kidney derived cell line; namely, Vero E6 was cultured and infected with SARS-CoV-2. Then the infected culture was added with hrsACE2 to estimate the therapeutic potential of hrsACE2. 15 h post-infection, the viral replication was found to be inhibited in a dose-dependent manner with reference to the concentration of hrsACE2 added. The addition of hrsACE2 to the embryonic stem cells derived kidney organoids comprised of proximal tubular cells and podocytes clusters infected with SARS-CoV-2 had shown to reduce the viral replication in a dose-dependent manner. No studies support the in-vivo and the therapeutic roles of hrsACE2 on the pulmonary cells upon SARS-CoV-2 infection [153,155]. Therefore, the usage of hrsACE2 can be a potential therapeutic option to inhibit the viral infection besides preserving the ACE2 levels and blood pressure in control.
7.3. FDA approved drug for SARS-CoV-2, remdesivir but not effective against it
The treatment of remdesivir in COVID-19 patients is not effective against it instead of FDA approved drugs. Out of 236 patients, 158 patients received remdesivir at a dosage of 200 mg on 1st day, followed by 100 mg from days 2–10 daily as a single infusion. 78 out of the 236 patients were assigned to the placebo group and were administered with placebo infusions. The presence and quantification of RNA were carried out on 1, 3, 5, 7, 10, 14, 21, and 28 days after administration of the drug, and the samples were collected as nasopharyngeal, fecal and anal swabs. One hundred two participants from the remdesivir group and 50 participants from the placebo group exhibited adverse outcomes. The adverse effects in the remdesivir group include hypokalaemia, hypoalbuminemia, thrombocytopenia, increased total bilirubin, anemia, and constipation. The placebo group's adverse effects include anemia, constipation, elated aspartate, aminotransferases, hypoalbuminemia, elevated total bilirubin, and blood lipids. Many participants discontinued from the remdesivir group due to the severe adverse effects such as respiratory failure or ARDS. Even though remdesivir has been declared an emergency anti-viral therapy by the FDA, it was found that it was not effective against SARS-CoV-2 [156].
8. Conclusion
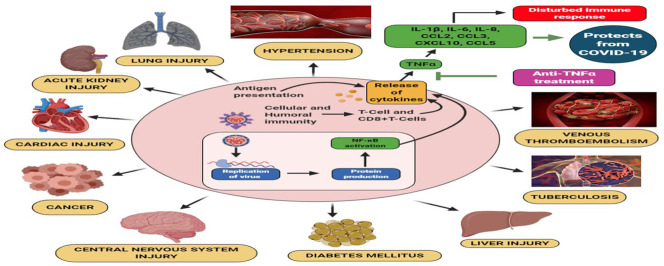
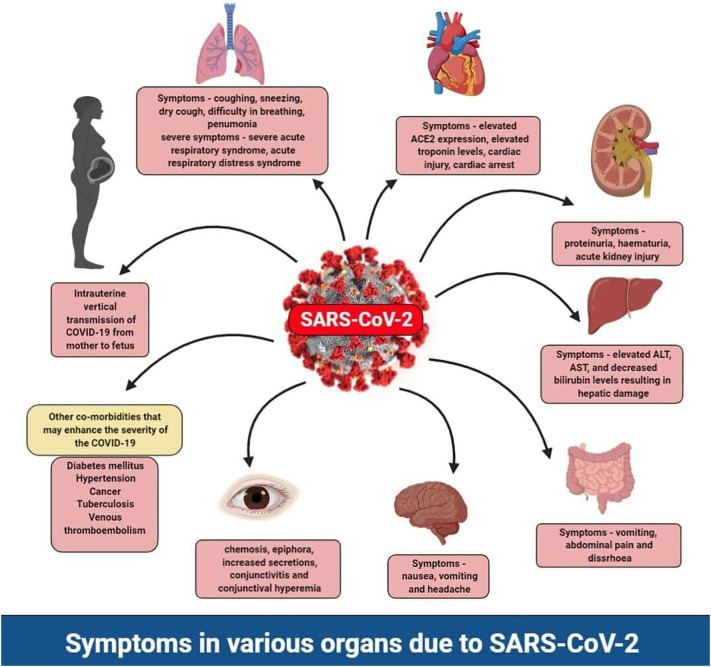
The COVID-19 outbreak in the world has 4,860,260 confirmed cases, 318,687 confirmed deaths, and it reached almost 215 countries around the globe, updated on May 18, 2020, by WHO [157]. In India, 100,340 confirmed cases, 3155 deaths, and 39,231 had been recovered from COVID-19 updated on May 18. This COVID-19 is highly associated with the comorbidities and multi-organ injuries [158], so, in this review, we focused on the COVID-19 associated with the risk factors of a different diseased condition. Also, we have given the multi-organ injuries and comorbidities in the SARS-CoV and MERS, which was helpful for the prediction of COVID-19 associated diseased conditions. In this review, we have given the comorbidities risk with the COVID-19 and multi-organ injuries along with the symptoms such as cardiovascular risk through the deregulation of ACE2 with the symptom of hypertension and vasodilation. COVID-19 associated gastrointestinal risks are with the symptoms of vomiting, nausea, and diarrhea. Kidney risk associated with COVID-19 is with the symptoms of proteinuria, hematuria, increased albumin, increased level of creatinine, BUN, procalcitonin, aspartate aminotransferase, lactate dehydrogenase leads to chronic kidney disease. Along with that, it is connected with diabetes mellitus with a high sugar level. It has a high level of liver injury, determined through the elevated level of the AST, ALT, GGT, and bilirubin. It has a high level of lung injury with the symptoms of chest tightness, wheezing, and dry cough. It is related to the elevated CNS risk through SARS-CoV-2 enters into the olfactory lobe of the brain via a nasal chamber, further causes inflammation and demyelination. It is connected with the ocular risk with the chemosis, epiphora, conjunctivitis, and conjunctival hyperemia with an increased level of neutrophilic, lymphocytic counts, procalcitonin, and C-reactive protein. It also has a high risk of lung cancer risk, tuberculosis, venous thromboembolism via an increased level of bleeding. It has a high level of reproductive risk, which indicates the baby has a high risk through intrauterine vertical transmission mode (Represented in Fig. 3 ). We have also given the immunopathological action of it along with COVID-19 and therapeutic action of it focused via anti-TNFα and soluble ACE2 receptor. Added data would further notify the handling of high-risk patients associated with SARS-CoV-2, clinicians necessitate being cognizant of the inadvertent penalty of impulsively discontinuing confirmed therapies in reaction to supposed concerns that could be based on deficient investigational confirmation.
Fig. 3.
The symptoms due to the SARS-CoV-2 in normal patients and patients with other co-morbid conditions. The symptoms found SARS-CoV-2 in normal patients and patients with other co-morbid conditions. The typical symptoms that emerged due to SARS-CoV-2 include dry cough, sneezing, and difficulty in breathing, which eventually leads to severe acute respiratory syndrome (SARS). The severity of the SARS not only depends upon the person's age and immunity but also various other co-morbid conditions such as cardiac issues, diabetes mellitus, hypertension, tuberculosis, cancer, and venous thromboembolism. The pre-existing co-morbid conditions deteriorate the patient health upon SARS-CoV-2 entry. Also, the SARS-CoV-2 have different effects on different organs which include elevated troponin levels in the cardiac tissue, acute kidney injury, proteinuria and hematuria in the kidney, elevated ALT, AST levels indicating liver damage, conjunctivitis and conjunctival hyperemia in the eye, the intrauterine transmission of the SARS-CoV-2 from mother to the fetus, vomiting, and diarrhea.
Funding
This work was supported by the “VIT SEED GRANT” and ICMR-National Task Force Project [F.No. 5/7/482/2010-RBMH&CH].
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
The authors thank the VIT, Vellore, Tamilnadu, India, for supporting this work. The author KaviyarasiRenu is grateful to ICMR for providing financial assistance in the form of a Senior Research Fellowship (SRF).
References
- 1.Calisher C., Carroll D., Colwell R., Corley R.B., Daszak P., Drosten C., Enjuanes L., Farrar J., Field H., Golding J. Statement in support of the scientists, public health professionals, and medical professionals of China combatting COVID-19. Lancet. 2020;395:e42–e43. doi: 10.1016/S0140-6736(20)30418-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ou X., Liu Y., Lei X., Li P., Mi D., Ren L., Guo L., Guo R., Chen T., Hu J. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020;11:1–12. doi: 10.1038/s41467-020-15562-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cascella M., Rajnik M., Cuomo A., Dulebohn S.C., Di Napoli R. StatPearls Publishing; 2020. Features, Evaluation and Treatment Coronavirus (COVID-19), StatPearls. Internet. [PubMed] [Google Scholar]
- 4.Ngandu T., Lehtisalo J., Solomon A., Levälahti E., Ahtiluoto S., Antikainen R., Bäckman L., Hänninen T., Jula A., Laatikainen T. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): a randomised controlled trial. Lancet. 2015;385:2255–2263. doi: 10.1016/S0140-6736(15)60461-5. [DOI] [PubMed] [Google Scholar]
- 5.Wu C., Liu Y., Yang Y., Zhang P., Zhong W., Wang Y., Wang Q., Xu Y., Li M., Li X. Analysis of therapeutic targets for SARS-CoV-2 and discovery of potential drugs by computational methods. Acta Pharm. Sin. B. 2020 doi: 10.1016/j.apsb.2020.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen Y., Liu Q., Guo D. Emerging coronaviruses: genome structure, replication, and pathogenesis. J. Med. Virol. 2020;92:418–423. doi: 10.1002/jmv.25681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vellingiri B., Jayaramayya K., Iyer M., Narayanasamy A., Govindasamy V., Giridharan B., Ganesan S., Venugopal A., Venkatesan D., Ganesan H. COVID-19: a promising cure for the global panic. Sci. Total Environ. 2020;725 doi: 10.1016/j.scitotenv.2020.138277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang T., Du Z., Zhu F., Cao Z., An Y., Gao Y., Jiang B. Comorbidities and multi-organ injuries in the treatment of COVID-19. Lancet. 2020;395:10228. doi: 10.1016/S0140-6736(20)30558-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fang L., Karakiulakis G., Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir. Med. 2020;8:4–E21. doi: 10.1016/S2213-2600(20)30116-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.H.C. today, cardiology societies recommend patients taking ACE inhibitors, ARBs who contract COVID-19 should continue treatment. March 17, 2020. https://www.healio.com/cardiology/vascular-medicine/news/online/%7Bfe7f0842-aecb-417b-9ecf-3fe7e0ddd991%7D/cardiology-societies-recommend-patients-taking-ace-inhibitors-arbs-who-contract-covid-19-should-continue-treatment
- 12.Hamming I., Timens W., Bulthuis M., Lely A., Navis G., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Keidar S., Kaplan M., Gamliel-Lazarovich A. ACE2 of the heart: from angiotensin I to angiotensin (1–7) Cardiovasc. Res. 2007;73:463–469. doi: 10.1016/j.cardiores.2006.09.006. [DOI] [PubMed] [Google Scholar]
- 14.Li W., Moore M.J., Vasilieva N., Sui J., Wong S.K., Berne M.A., Somasundaran M., Sullivan J.L., Luzuriaga K., Greenough T.C. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426:450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jiang F., Deng L., Zhang L., Cai Y., Cheung C.W., Xia Z. Review of the clinical characteristics of coronavirus disease 2019 (COVID-19) J. Gen. Intern. Med. 2020:1–5. doi: 10.1007/s11606-020-05762-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Golan D.E., Tashjian A.H., Armstrong E.J. Lippincott Williams & Wilkins; 2011. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. [Google Scholar]
- 17.Fyhrquist F., Metsärinne K., Tikkanen I. Role of angiotensin II in blood pressure regulation and in the pathophysiology of cardiovascular disorders. J. Hum. Hypertens. 1995;9:S19–S24. [PubMed] [Google Scholar]
- 18.Sanders G.D., Coeytaux R., Dolor R.J., Hasselblad V., Patel U.D., Powers B., Yancy W.S., Jr., Gray R.N., Irvine R.J., Kendrick A. 2011. Angiotensin-Converting Enzyme Inhibitors (ACEIs), Angiotensin II Receptor Antagonists (ARBs), and Direct Renin Inhibitors for Treating Essential Hypertension: An Update. [PubMed] [Google Scholar]
- 19.El-Sahly H.M., Atmar R.L., Glezen W.P., Greenberg S.B. Spectrum of clinical illness in hospitalized patients with “common cold” virus infections. Clin. Infect. Dis. 2000;31:96–100. doi: 10.1086/313937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Su S., Wong G., Shi W., Liu J., Lai A.C., Zhou J., Liu W., Bi Y., Gao G.F. Epidemiology, genetic recombination, and pathogenesis of coronaviruses. Trends Microbiol. 2016;24:490–502. doi: 10.1016/j.tim.2016.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Falsey A.R., Walsh E.E., Hayden F.G. Rhinovirus and coronavirus infection-associated hospitalizations among older adults. J. Infect. Dis. 2002;185:1338–1341. doi: 10.1086/339881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Peiris J., Lai S., Poon L., Guan Y., Yam L., Lim W., Nicholls J., Yee W., Yan W., Cheung M. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet. 2003;361:1319–1325. doi: 10.1016/S0140-6736(03)13077-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kupferschmidt K. Researchers scramble to understand camel connection to MERS. American Association for the Advancement of Science. 2013;341:702. doi: 10.1126/science.341.6147.702. [DOI] [PubMed] [Google Scholar]
- 24.Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., Zhao X., Huang B., Shi W., Lu R. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tesini B.L. Coronaviruses and acute respiratory syndromes (COVID-19, MERS, and SARS) April 2020. https://www.msdmanuals.com/professional/infectious-diseases/respiratory-viruses/coronaviruses-and-acute-respiratory-syndromes-covid-19,-mers,-and-sars
- 26.Berry M., Gamieldien J., Fielding B.C. Identification of new respiratory viruses in the new millennium. Viruses. 2015;7:996–1019. doi: 10.3390/v7030996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.M. Clinic Severe acute respiratory syndrome (SARS) https://www.mayoclinic.org/diseases-conditions/sars/symptoms-causes/syc-20351765
- 28.Li F. Structure, function, and evolution of coronavirus spike proteins. Annual Review of Virology. 2016;3:237–261. doi: 10.1146/annurev-virology-110615-042301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Imai Y., Kuba K., Rao S., Huan Y., Guo F., Guan B., Yang P., Sarao R., Wada T., Leong-Poi H. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436:112–116. doi: 10.1038/nature03712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gu J., Korteweg C. Pathol ogy and pathogenesis of Severe Acute Respiratory Syndrome. The American Journal of Patology. 2007;170:1136–1147. doi: 10.2353/ajpath.2007.061088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chong P.Y., Chui P., Ling A.E., Franks T.J., Tai D.Y., Leo Y.S., Kaw G.J., Wansaicheong G., Chan K.P., Ean Oon L.L. Analysis of deaths during the severe acute respiratory syndrome (SARS) epidemic in Singapore: challenges in determining a SARS diagnosis. Arch. Pathol. Lab. Med. 2004;128:195–204. doi: 10.5858/2004-128-195-AODDTS. [DOI] [PubMed] [Google Scholar]
- 32.Peiris J., Chu C., Cheng V., Chan K., Hung I., Poon L., Law K., Tang B., Hon T., Chan C., members of the HKU/UCH SARS Study Group Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet. 2003;361:1767–1772. doi: 10.1016/S0140-6736(03)13412-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Li S.S.-l., Cheng C.-w., Fu C.-l., Chan Y.-h., Lee M.-p., Chan J.W.-m., Yiu S.-f. Left ventricular performance in patients with severe acute respiratory syndrome: a 30-day echocardiographic follow-up study. Circulation. 2003;108:1798–1803. doi: 10.1161/01.CIR.0000094737.21775.32. [DOI] [PubMed] [Google Scholar]
- 34.Liao Z., Yao B., Guan K., Beng W., Hu G., Weng J. Clinical feature in patients of sever acute respiratory syndrome with type 2 diabetes and secondary hyperglycemia. Chinese Journal of Endocrinology and Metabolism. 2004;20:14–15. [Google Scholar]
- 35.Gu J., Gong E., Zhang B., Zheng J., Gao Z., Zhong Y., Zou W., Zhan J., Wang S., Xie Z. Multiple organ infection and the pathogenesis of SARS. J. Exp. Med. 2005;202:415–424. doi: 10.1084/jem.20050828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chu K.H., Tsang W.K., Tang C.S., Lam M.F., Lai F.M., To K.F., Fung K.S., Tang H.L., Yan W.W., Chan H.W. Acute renal impairment in coronavirus-associated severe acute respiratory syndrome. Kidney Int. 2005;67:698–705. doi: 10.1111/j.1523-1755.2005.67130.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xu L., Liu J., Lu M., Yang D., Zheng X. Liver injury during highly pathogenic human coronavirus infections. Liver Int. 2020;40:998–1004. doi: 10.1111/liv.14435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.LU Y.-c., YIN C.-b., TANG X.-p., YANG Z., LEI C.-l., CHEN W.-s., GONG L., JIA W.-d. Clinical characteristics and mechanism of liver function injury in 250 patients with severe acute respiratory syndrome. China Journal of Modern Medicine. 2004;14:121–123. [Google Scholar]
- 39.Wang J.-T., Sheng W.-H., Fang C.-T., Chen Y.-C., Wang J.-L., Yu C.-J., Chang S.-C., Yang P.-C. Clinical manifestations, laboratory findings, and treatment outcomes of SARS patients. Emerg. Infect. Dis. 2004;10:818. doi: 10.3201/eid1005.030640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chau T.N., Lee K.C., Yao H., Tsang T.Y., Chow T.C., Yeung Y.C., Choi K.W., Tso Y.K., Lau T., Lai S.T. SARS-associated viral hepatitis caused by a novel coronavirus: report of three cases. Hepatology. 2004;39:302–310. doi: 10.1002/hep.20111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wu K.-L., Lu S.-N., Changchien C.-S., Chiu K.-W., Kuo C.-H., Chuah S.-K., Liu J.-W., Lin M.-C., Eng H.-L., Chen S.-S. Sequential changes of serum aminotransferase levels in patients with severe acute respiratory syndrome. The American Journal of Tropical Medicine and Hygiene. 2004;71:125–128. [PubMed] [Google Scholar]
- 42.Duan Z., Chen Y., Zhang J., Zhao J., Lang Z., Meng F., Bao X. Clinical characteristics and mechanism of liver injury in patients with severe acute respiratory syndrome. Zhonghua gan zang bing za zhi= Zhonghua ganzangbing zazhi= Chinese journal of hepatology. 2003;11:493–496. [PubMed] [Google Scholar]
- 43.Ding Y., He L., Zhang Q., Huang Z., Che X., Hou J., Wang H., Shen H., Qiu L., Li Z. Organ distribution of severe acute respiratory syndrome (SARS) associated coronavirus (SARS-CoV) in SARS patients: implications for pathogenesis and virus transmission pathways. The Journal of Pathology: A Journal of the Pathological Society of Great Britain and Ireland. 2004;203:622–630. doi: 10.1002/path.1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tan Y.-J., Fielding B.C., Goh P.-Y., Shen S., Tan T.H., Lim S.G., Hong W. Overexpression of 7a, a protein specifically encoded by the severe acute respiratory syndrome coronavirus, induces apoptosis via a caspase-dependent pathway. J. Virol. 2004;78:14043–14047. doi: 10.1128/JVI.78.24.14043-14047.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.HUANG Y.-h., GAO Z.-l. Study of the relationship SARS and hepatitis virus B. Chinese Journal of Clinical Hepatology. 2003:10. [Google Scholar]
- 46.Tse G.M., To K., Chan P.K., Lo A., Ng K., Wu A., Lee N., Wong H., Mak S., Chan K. Pulmonary pathological features in coronavirus associated severe acute respiratory syndrome (SARS) J. Clin. Pathol. 2004;57:260–265. doi: 10.1136/jcp.2003.013276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ding Y., Wang H., Shen H., Li Z., Geng J., Han H., Cai J., Li X., Kang W., Weng D. The clinical pathology of severe acute respiratory syndrome (SARS): a report from China. The Journal of Pathology: A Journal of the Pathological Society of Great Britain and Ireland. 2003;200:282–289. doi: 10.1002/path.1440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nicholls J.M., Poon L.L., Lee K.C., Ng W.F., Lai S.T., Leung C.Y., Chu C.M., Hui P.K., Mak K.L., Lim W. Lung pathology of fatal severe acute respiratory syndrome. Lancet. 2003;361:1773–1778. doi: 10.1016/S0140-6736(03)13413-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hwang D.M., Chamberlain D.W., Poutanen S.M., Low D.E., Asa S.L., Butany J. Pulmonary pathology of severe acute respiratory syndrome in Toronto. Mod. Pathol. 2005;18:1–10. doi: 10.1038/modpathol.3800247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Franks T.J., Chong P.Y., Chui P., Galvin J.R., Lourens R.M., Reid A.H., Selbs E., Mcevoy C.P.L., Hayden C.D.L., Fukuoka J. Lung pathology of severe acute respiratory syndrome (SARS): a study of 8 autopsy cases from Singapore. Hum. Pathol. 2003;34:743–748. doi: 10.1016/S0046-8177(03)00367-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lang Z.W., Zhang L.J., Zhang S.J., Meng X., Li J.Q., Song C.Z., Sun L., Zhou Y.S., Dwyer D.E. A clinicopathological study of three cases of severe acute respiratory syndrome (SARS) Pathology. 2003;35:526–531. doi: 10.1080/00313020310001619118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lee N., Hui D., Wu A., Chan P., Cameron P., Joynt G.M., Ahuja A., Yung M.Y., Leung C., To K. A major outbreak of severe acute respiratory syndrome in Hong Kong. N. Engl. J. Med. 2003;348:1986–1994. doi: 10.1056/NEJMoa030685. [DOI] [PubMed] [Google Scholar]
- 53.Guo Y., Korteweg C., McNutt M.A., Gu J. Pathogenetic mechanisms of severe acute respiratory syndrome. Virus Res. 2008;133:4–12. doi: 10.1016/j.virusres.2007.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.America I.D.S.o. New study shows SARS can infect brain tissue. ScienceDaily. 2005 [Google Scholar]
- 55.Cheung C.Y., Poon L.L., Ng I.H., Luk W., Sia S.-F., Wu M.H., Chan K.-H., Yuen K.-Y., Gordon S., Guan Y. Cytokine responses in severe acute respiratory syndrome coronavirus-infected macrophages in vitro: possible relevance to pathogenesis. J. Virol. 2005;79:7819–7826. doi: 10.1128/JVI.79.12.7819-7826.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Law H.K., Cheung C.Y., Ng H.Y., Sia S.F., Chan Y.O., Luk W., Nicholls J.M., Peiris J.M., Lau Y.L. Chemokine up-regulation in sars-coronavirus–infected, monocyte-derived human dendritic cells. Blood. 2005;106:2366–2374. doi: 10.1182/blood-2004-10-4166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yen Y.-T., Liao F., Hsiao C.-H., Kao C.-L., Chen Y.-C., Wu-Hsieh B.A. Modeling the early events of severe acute respiratory syndrome coronavirus infection in vitro. J. Virol. 2006;80:2684–2693. doi: 10.1128/JVI.80.6.2684-2693.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Totura A.L., Whitmore A., Agnihothram S., Schäfer A., Katze M.G., Heise M.T., Baric R.S. Toll-like receptor 3 signaling via TRIF contributes to a protective innate immune response to severe acute respiratory syndrome coronavirus infection. MBio. 2015;6 doi: 10.1128/mBio.00638-15. (e00638-00615) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Mohd H.A., Al-Tawfiq J.A., Memish Z.A. Middle East respiratory syndrome coronavirus (MERS-CoV) origin and animal reservoir. Virol. J. 2016;13:87. doi: 10.1186/s12985-016-0544-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zumla A., Hui D.S., Perlman S. Middle East respiratory syndrome. Lancet. 2015;386:995–1007. doi: 10.1016/S0140-6736(15)60454-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Mackay I.M., Arden K.E. MERS coronavirus: diagnostics, epidemiology and transmission. Virol. J. 2015;12:222. doi: 10.1186/s12985-015-0439-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.van Boheemen S., de Graaf M., Lauber C., Bestebroer T.M., Raj V.S., Zaki A.M., Osterhaus A.D., Haagmans B.L., Gorbalenya A.E., Snijder E.J. Genomic characterization of a newly discovered coronavirus associated with acute respiratory distress syndrome in humans. MBio. 2012;3 doi: 10.1128/mBio.00473-12. (e00473-00412) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Park J.-E., Jung S., Kim A. MERS transmission and risk factors: a systematic review. BMC Public Health. 2018;18:574. doi: 10.1186/s12889-018-5484-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Matsuyama R., Nishiura H., Kutsuna S., Hayakawa K., Ohmagari N. Clinical determinants of the severity of Middle East respiratory syndrome (MERS): a systematic review and meta-analysis. BMC Public Health. 2016;16:1203. doi: 10.1186/s12889-016-3881-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Badawi A., Ryoo S.G. Prevalence of comorbidities in the Middle East respiratory syndrome coronavirus (MERS-CoV): a systematic review and meta-analysis. Int. J. Infect. Dis. 2016;49:129–133. doi: 10.1016/j.ijid.2016.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Arabi Y., Harthi A., Hussein J., Bouchama A., Johani S., Hajeer A., Saeed B., Wahbi A., Saedy A., AlDabbagh T. Severe neurologic syndrome associated with Middle East respiratory syndrome corona virus (MERS-CoV) Infection. 2015;43:495–501. doi: 10.1007/s15010-015-0720-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Assiri A., Al-Tawfiq J.A., Al-Rabeeah A.A., Al-Rabiah F.A., Al-Hajjar S., Al-Barrak A., Flemban H., Al-Nassir W.N., Balkhy H.H., Al-Hakeem R.F. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: a descriptive study. Lancet Infect. Dis. 2013;13:752–761. doi: 10.1016/S1473-3099(13)70204-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Al-Hameed F., Wahla A.S., Siddiqui S., Ghabashi A., Al-Shomrani M., Al-Thaqafi A., Tashkandi Y. Characteristics and outcomes of Middle East respiratory syndrome coronavirus patients admitted to an intensive care unit in Jeddah, Saudi Arabia. J. Intensive Care Med. 2016;31:344–348. doi: 10.1177/0885066615579858. [DOI] [PubMed] [Google Scholar]
- 69.Saad M., Omrani A.S., Baig K., Bahloul A., Elzein F., Matin M.A., Selim M.A., Al Mutairi M., Al Nakhli D., Al Aidaroos A.Y. Clinical aspects and outcomes of 70 patients with Middle East respiratory syndrome coronavirus infection: a single-center experience in Saudi Arabia. Int. J. Infect. Dis. 2014;29:301–306. doi: 10.1016/j.ijid.2014.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Raj V.S., Mou H., Smits S.L., Dekkers D.H., Müller M.A., Dijkman R., Muth D., Demmers J.A., Zaki A., Fouchier R.A. Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus-EMC. Nature. 2013;495:251–254. doi: 10.1038/nature12005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Boonacker E., Van Noorden C.J. The multifunctional or moonlighting protein CD26/DPPIV. Eur. J. Cell Biol. 2003;82:53–73. doi: 10.1078/0171-9335-00302. [DOI] [PubMed] [Google Scholar]
- 72.Zhao G., Jiang Y., Qiu H., Gao T., Zeng Y., Guo Y., Yu H., Li J., Kou Z., Du L. Multi-organ damage in human dipeptidyl peptidase 4 transgenic mice infected with Middle East respiratory syndrome-coronavirus. PLoS One. 2015;10 doi: 10.1371/journal.pone.0145561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Alsaad K.O., Hajeer A.H., Al Balwi M., Al Moaiqel M., Al Oudah N., Al Ajlan A., AlJohani S., Alsolamy S., Gmati G.E., Balkhy H. Histopathology of Middle East respiratory syndrome coronovirus (MERS-CoV) infection–clinicopathological and ultrastructural study. Histopathology. 2018;72:516–524. doi: 10.1111/his.13379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ng D.L., Al Hosani F., Keating M.K., Gerber S.I., Jones T.L., Metcalfe M.G., Tong S., Tao Y., Alami N.N., Haynes L.M. Clinicopathologic, immunohistochemical, and ultrastructural findings of a fatal case of Middle East respiratory syndrome coronavirus infection in the United Arab Emirates, April 2014. Am. J. Pathol. 2016;186:652–658. doi: 10.1016/j.ajpath.2015.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Mahallawi W.H., Khabour O.F., Zhang Q., Makhdoum H.M., Suliman B.A. MERS-CoV infection in humans is associated with a pro-inflammatory Th1 and Th17 cytokine profile. Cytokine. 2018;104:8–13. doi: 10.1016/j.cyto.2018.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.WHO Report, Middle East Respiratory Syndrome Coronavirus (MERS-CoV) 2019. [Google Scholar]
- 77.Kim J.-E., Heo J.-H., Kim H.-o., Song S.-h., Park S.-S., Park T.-H., Ahn J.-Y., Kim M.-K., Choi J.-P. Neurological complications during treatment of Middle East respiratory syndrome. Journal of Clinical Neurology. 2017;13:227–233. doi: 10.3988/jcn.2017.13.3.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kim K., Tandi T., Choi J.W., Moon J., Kim M. Middle East respiratory syndrome coronavirus (MERS-CoV) outbreak in South Korea, 2015: epidemiology, characteristics and public health implications. J. Hosp. Infect. 2017;95:207–213. doi: 10.1016/j.jhin.2016.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hui D.S., Memish Z.A., Zumla A. Severe acute respiratory syndrome vs. the Middle East respiratory syndrome. Curr. Opin. Pulm. Med. 2014;20:233–241. doi: 10.1097/MCP.0000000000000046. [DOI] [PubMed] [Google Scholar]
- 80.Memish Z.A., Perlman S., Van Kerkhove M.D., Zumla A. Middle East respiratory syndrome. Lancet. 2020;95:10229. doi: 10.1016/S0140-6736(19)33221-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lau S.K., Lau C.C., Chan K.-H., Li C.P., Chen H., Jin D.-Y., Chan J.F., Woo P.C., Yuen K.-Y. Delayed induction of proinflammatory cytokines and suppression of innate antiviral response by the novel Middle East respiratory syndrome coronavirus: implications for pathogenesis and treatment. J. Gen. Virol. 2013;94:2679–2690. doi: 10.1099/vir.0.055533-0. [DOI] [PubMed] [Google Scholar]
- 82.Tynell J., Westenius V., Rönkkö E., Munster V.J., Melén K., Österlund P., Julkunen I. Middle East respiratory syndrome coronavirus shows poor replication but significant induction of antiviral responses in human monocyte-derived macrophages and dendritic cells. The Journal of General Virology. 2016;97:344. doi: 10.1099/jgv.0.000351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zhou J., Chu H., Li C., Wong B.H.-Y., Cheng Z.-S., Poon V.K.-M., Sun T., Lau C.C.-Y., Wong K.K.-Y., Chan J.Y.-W. Active replication of Middle East respiratory syndrome coronavirus and aberrant induction of inflammatory cytokines and chemokines in human macrophages: implications for pathogenesis. J. Infect. Dis. 2014;209:1331–1342. doi: 10.1093/infdis/jit504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kim E.S., Choe P.G., Park W.B., Oh H.S., Kim E.J., Nam E.Y., Na S.H., Kim M., Song K.-H., Bang J.H. Clinical progression and cytokine profiles of Middle East respiratory syndrome coronavirus infection. J. Korean Med. Sci. 2016;31:1717–1725. doi: 10.3346/jkms.2016.31.11.1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Zheng Y.-Y., Ma Y.-T., Zhang J.-Y., Xie X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020:1–2. doi: 10.1038/s41569-020-0360-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Chen L., Li X., Chen M., Feng Y., Xiong C. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS-CoV-2. Cardiovasc. Res. 2020;116:1097–1100. doi: 10.1093/cvr/cvaa078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Keidar S., Gamliel-Lazarovich A., Kaplan M., Pavlotzky E., Hamoud S., Hayek T., Karry R., Abassi Z. Mineralocorticoid receptor blocker increases angiotensin-converting enzyme 2 activity in congestive heart failure patients. Circ. Res. 2005;97:946–953. doi: 10.1161/01.RES.0000187500.24964.7A. [DOI] [PubMed] [Google Scholar]
- 88.Ferrario C.M., Jessup J., Chappell M.C., Averill D.B., Brosnihan K.B., Tallant E.A., Diz D.I., Gallagher P.E. Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation. 2005;111:2605–2610. doi: 10.1161/CIRCULATIONAHA.104.510461. [DOI] [PubMed] [Google Scholar]
- 89.Zhang H., Penninger J.M., Li Y., Zhong N., Slutsky A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020:1–5. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ding W., Li X., Wu W., He H., Li Y., Gao L., Gan L., Wang M., Ou S., Liu J. Aliskiren inhibits angiotensin II/angiotensin 1-7 (Ang II/Ang1-7) signal pathway in rats with diabetic nephropathy. Xi bao yu fen zi mian yi xue za zhi= Chinese journal of cellular and molecular immunology. 2018;34:891–895. [PubMed] [Google Scholar]
- 91.Mourad J.-J., Levy B.I. Interaction between RAAS inhibitors and ACE2 in the context of COVID-19. Nat. Rev. Cardiol. 2020;17:313-313. doi: 10.1038/s41569-020-0368-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Zhao D., Yao F., Wang L., Zheng L., Gao Y., Ye J., Guo F., Zhao H., Gao R. A comparative study on the clinical features of COVID-19 pneumonia to other pneumonias. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Holshue M.L., DeBolt C., Lindquist S., Lofy K.H., Wiesman J., Bruce H., Spitters C., Ericson K., Wilkerson S., Tural A. First case of 2019 novel coronavirus in the United States. N. Engl. J. Med. 2020;382:929–936. doi: 10.1056/NEJMoa2001191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Lu X., Zhang L., Du H., Zhang J., Li Y.Y., Qu J., Zhang W., Wang Y., Bao S., Li Y. SARS-CoV-2 infection in children. N. Engl. J. Med. 2020;382:1663–1665. doi: 10.1056/NEJMc2005073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Guan W.-j., Ni Z.-y., Hu Y., Liang W.-h., Ou C.-q., He J.-x., Liu L., Shan H., Lei C.-l., Hui D.S. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.F. Xiao, M. Tang, X. Zheng, C. Li, J. He, Z. Hong, S. Huang, Z. Zhang, X. Lin, Z. Fang, Evidence for gastrointestinal infection of SARS-CoV-2, medRxiv, (2020). [DOI] [PMC free article] [PubMed]
- 97.Xu Y., Li X., Zhu B., Liang H., Fang C., Gong Y., Guo Q., Sun X., Zhao D., Shen J. Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding. Nat. Med. 2020:1–4. doi: 10.1038/s41591-020-0817-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.H.I.S. Writers . HospiMedica Daily Clinical News; March 2020. Novel Coronavirus Could be Targeting Kidneys, Suggests New Data. [Google Scholar]
- 99.Wang L., Li X., Chen H., Yan S., Li D., Li Y., Gong Z. Coronavirus disease 19 infection does not result in acute kidney injury: an analysis of 116 hospitalized patients from Wuhan, China. Am. J. Nephrol. 2020:1–6. doi: 10.1159/000507471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Cheng Y., Luo R., Wang K., Zhang M., Wang Z., Dong L., Li J., Yao Y., Ge S., Xu G. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020;97:829–838. doi: 10.1016/j.kint.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Chen N., Zhou M., Dong X., Qu J., Gong F., Han Y., Qiu Y., Wang J., Liu Y., Wei Y. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Fan Z., Chen L., Li J., Tian C., Zhang Y., Huang S., Liu Z., Cheng J. 2020. Clinical Features of COVID-19-Related Liver Damage. Available at SSRN 3546077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Chai X., Hu L., Zhang Y., Han W., Lu Z., Ke A., Zhou J., Shi G., Fang N., Fan J. 2020. Specific ACE2 Expression in Cholangiocytes May Cause Liver Damage after 2019-nCoV Infection, bioRxiv. [Google Scholar]
- 104.Banales J.M., Huebert R.C., Karlsen T., Strazzabosco M., LaRusso N.F., Gores G.J. Cholangiocyte pathobiology. Nat. Rev. Gastroenterol. Hepatol. 2019;16:269–281. doi: 10.1038/s41575-019-0125-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Xu Z. Lancet Respir. Med. 2020;8:4–E6. doi: 10.1016/S2213-2600(19)30476-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Tian S., Hu W., Niu L., Liu H., Xu H., Xiao S.-Y. Pulmonary pathology of early phase 2019 novel coronavirus (COVID-19) pneumonia in two patients with lung cancer. J. Thorac. Oncol. 2020;15:700–704. doi: 10.1016/j.jtho.2020.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Cox D. Some patients who survive COVID-19 may suffer lasting lung damage. Sci. News. 2020 [Google Scholar]
- 108.Yeager A. The Scientist Exploring Life, Inspiring Innovation. Mar 24, 2020. Lost smell and taste hint COVID-19 can target the nervous system. [Google Scholar]
- 109.Li X., Geng M., Peng Y., Meng L., Lu S. Molecular immune pathogenesis and diagnosis of COVID-19. Journal of Pharmaceutical Analysis. 2020;10:102–108. doi: 10.1016/j.jpha.2020.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Prompetchara E., Ketloy C., Palaga T. Immune responses in COVID-19 and potential vaccines: lessons learned from SARS and MERS epidemic. Asian Pac. J. Allergy Immunol. 2020;38:1–9. doi: 10.12932/AP-200220-0772. [DOI] [PubMed] [Google Scholar]
- 111.Roncati L., Nasillo V., Lusenti B., Riva G. Signals of Th2 immune response from COVID-19 patients requiring intensive care. Ann. Hematol. 2020:1. doi: 10.1007/s00277-020-04066-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Alosaimi B., Hamed M.E., Naeem A., Alsharef A.A., AlQahtani S.Y., AlDosari K.M., Alamri A.A., Al-Eisa K., Khojah T., Assiri A.M. MERS-CoV infection is associated with downregulation of genes encoding Th1 and Th2 cytokines/chemokines and elevated inflammatory innate immune response in the lower respiratory tract. Cytokine. 2020;126 doi: 10.1016/j.cyto.2019.154895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Li C.K.-f., Wu H., Yan H., Ma S., Wang L., Zhang M., Tang X., Temperton N.J., Weiss R.A., Brenchley J.M. T cell responses to whole SARS coronavirus in humans. J. Immunol. 2008;181:5490–5500. doi: 10.4049/jimmunol.181.8.5490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Martín J. 2020. B Cells Over-Activation by Viral Proteins <70 kDa Causes Th2 Immune Suppression in COVID-19 Sepsis. Preprints 2020050244. [Google Scholar]
- 115.Li L., Huang Q., Wang D.C., Ingbar D.H., Wang X. Acute lung injury in patients with COVID-19 infection. Clinical and Translational Medicine. 2020;10:20–27. doi: 10.1002/ctm2.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Napoli P.E., Nioi M., d'Aloja E., Fossarello M. The ocular surface and the coronavirus disease 2019: does a dual ‘ocular route’exist? Multidisciplinary Digital Publishing Institute. 2020;9:E1269. doi: 10.3390/jcm9051269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Sun Y., Liu L., Pan X., Jing M. Mechanism of the action between the SARS-CoV S240 protein and the ACE2 receptor in eyes. Int J Ophthalmol-Chi. 2006;6:783. [Google Scholar]
- 118.Sun K., Gu L., Ma L., Duan Y. Atlas of ACE2 gene expression in mammals reveals novel insights in transmission of SARS-Cov-2. BioRxiv. 2020 doi: 10.1016/j.heliyon.2020.e05850. 2020.03.30.015644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Sommer A. Humans, viruses, and the eye—An early report from the COVID-19 front line. JAMA Ophthalmology. 2020:E1–E2. doi: 10.1001/jamaophthalmol.2020.1294. [DOI] [PubMed] [Google Scholar]
- 120.Sungnak W., Huang N., Bécavin C., Berg M., Queen R., Litvinukova M., Talavera-López C., Maatz H., Reichart D., Sampaziotis F. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat. Med. 2020:1–7. doi: 10.1038/s41591-020-0868-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Xia J., Tong J., Liu M., Shen Y., Guo D. Evaluation of coronavirus in tears and conjunctival secretions of patients with SARS-CoV-2 infection. J. Med. Virol. 2020;92:589–594. doi: 10.1002/jmv.25725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Liang W., Guan W., Chen R., Wang W., Li J., Xu K., Li C., Ai Q., Lu W., Liang H. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. The Lancet Oncology. 2020;21:335–337. doi: 10.1016/S1470-2045(20)30096-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Zhang L., Zhu F., Xie L., Wang C., Wang J., Chen R., Jia P., Guan H., Peng L., Chen Y. Clinical characteristics of COVID-19-infected cancer patients: a retrospective case study in three hospitals within Wuhan, China. Ann. Oncol. 2020 doi: 10.1016/j.annonc.2020.03.296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Liu Y., Bi L., Chen Y., Wang Y., Fleming J., Yu Y., Gu Y., Liu C., Fan L., Wang X. 2020. Active or Latent Tuberculosis Increases Susceptibility to COVID-19 and Disease Severity, medRxiv. [Google Scholar]
- 125.WHO Report, Q&A on COVID-19, HIV and Antiretrovirals. 2020. https://www.who.int/news-room/q-a-detail/q-a-on-covid-19-hiv-and-antiretrovirals
- 126.Marimuthu Y., Nagappa B., Sharma N., Basu S., Chopra K.K. COVID-19 and tuberculosis: a mathematical model based forecasting in Delhi, India. Indian J. Tuberc. 2020 doi: 10.1016/j.ijtb.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Tao Wang R.C., Liu Chunli, Liang Wenhua, Guan Weijie, Tang Ruidi, Tang Chunli, Zhang Nuofu, Zhong Nanshan, Li Shiyue. Attention should be paid to venous thromboembolism prophylaxis in the management of COVID-19. Lancet Hematology. 2020;7:E362–363. doi: 10.1016/S2352-3026(20)30109-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.E.F.d.B. Andy Haines, Anita Berlin, M.J.H. David L Heymann, Centring sexual and reproductive health and justice in the global COVID-19 response, Lancet, (2020). [DOI] [PMC free article] [PubMed]
- 129.Yao H., Chen J.-H., Xu Y.-F. Patients with mental health disorders in the COVID-19 epidemic. Lancet Psychiatry. 2020;7:e21. doi: 10.1016/S2215-0366(20)30090-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.AlGhatrif M., Cingolani O., Lakatta E.G. The dilemma of coronavirus disease 2019, aging, and cardiovascular disease: insights from cardiovascular aging science. JAMA Cardiol. 2020 doi: 10.1001/jamacardio.2020.1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Cameron C.E., Castro C. The mechanism of action of ribavirin: lethal mutagenesis of RNA virus genomes mediated by the viral RNA-dependent RNA polymerase. Curr. Opin. Infect. Dis. 2001;14:757–764. doi: 10.1097/00001432-200112000-00015. [DOI] [PubMed] [Google Scholar]
- 132.Leyssen P., Balzarini J., De Clercq E., Neyts J. The predominant mechanism by which ribavirin exerts its antiviral activity in vitro against flaviviruses and paramyxoviruses is mediated by inhibition of IMP dehydrogenase. J. Virol. 2005;79:1943–1947. doi: 10.1128/JVI.79.3.1943-1947.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Shetty R., Ghosh A., Honavar S.G., Khamar P., Sethu S. Therapeutic opportunities to manage COVID-19/SARS-CoV-2 infection: present and future. Indian J. Ophthalmol. 2020;61:62. doi: 10.4103/ijo.IJO_639_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Hong Z., Cameron C.E. Prog. Drug Res. Springer; 2002. Pleiotropic mechanisms of ribavirin antiviral activities; pp. 41–69. [DOI] [PubMed] [Google Scholar]
- 135.Chu C., Cheng V., Hung I., Wong M., Chan K., Chan K., Kao R., Poon L., Wong C., Guan Y. Role of lopinavir/ritonavir in the treatment of SARS: initial virological and clinical findings. Thorax. 2004;59:252–256. doi: 10.1136/thorax.2003.012658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Choi K.W., Chau T.N., Tsang O., Tso E., Chiu M.C., Tong W.L., Lee P.O., Ng T.K., Ng W.F., Lee K.C. Outcomes and prognostic factors in 267 patients with severe acute respiratory syndrome in Hong Kong. Ann. Intern. Med. 2003;139:715–723. doi: 10.7326/0003-4819-139-9-200311040-00005. [DOI] [PubMed] [Google Scholar]
- 137.Sung J., Wu A., Joynt G., Yuen K., Lee N., Chan P., Cockram C., Ahuja A., Yu L., Wong V. Severe acute respiratory syndrome: report of treatment and outcome after a major outbreak. Thorax. 2004;59:414–420. doi: 10.1136/thx.2003.014076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Chan J., Ng C., Chan Y., Mok T., Lee S., Chu S., Law W., Lee M., Li P. Short term outcome and risk factors for adverse clinical outcomes in adults with severe acute respiratory syndrome (SARS) Thorax. 2003;58:686–689. doi: 10.1136/thorax.58.8.686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Knowles S.R., Phillips E.J., Dresser L., Matukas L. Common adverse events associated with the use of ribavirin for severe acute respiratory syndrome in Canada. Clin. Infect. Dis. 2003;37:1139–1142. doi: 10.1086/378304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Yu W., Hui D., Chan-Yeung M. BMJ Publishing Group Ltd; 2004. Antiviral Agents and Corticosteroids in the Treatment of Severe Acute Respiratory Syndrome (SARS) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Oba Y. The use of corticosteroids in SARS. N. Engl. J. Med. 2003;348:2034–2035. doi: 10.1056/NEJM200305153482017. (author reply 2034-2035) [DOI] [PubMed] [Google Scholar]
- 142.Cheung C., Poon L., Lau A., Luk W., Lau Y., Shortridge K., Gordon S., Guan Y., Peiris J. Induction of proinflammatory cytokines in human macrophages by influenza A (H5N1) viruses: a mechanism for the unusual severity of human disease? Lancet. 2002;360:1831–1837. doi: 10.1016/s0140-6736(02)11772-7. [DOI] [PubMed] [Google Scholar]
- 143.Yeh K.-M., Chiueh T.-S., Siu L., Lin J.-C., Chan P.K., Peng M.-Y., Wan H.-L., Chen J.-H., Hu B.-S., Perng C.-L. Experience of using convalescent plasma for severe acute respiratory syndrome among healthcare workers in a Taiwan hospital. J. Antimicrob. Chemother. 2005;56:919–922. doi: 10.1093/jac/dki346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Soo Y., Cheng Y., Wong R., Hui D., Lee C., Tsang K., Ng M., Chan P., Cheng G., Sung J. Retrospective comparison of convalescent plasma with continuing high-dose methylprednisolone treatment in SARS patients. Clin. Microbiol. Infect. 2004;10:676–678. doi: 10.1111/j.1469-0691.2004.00956.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Burnouf T., Radosevich M. Treatment of severe acute respiratory syndrome with convalescent plasma. Hong Kong Med. J. 2003;9:309. [PubMed] [Google Scholar]
- 146.Cheng Y., Wong R., Soo Y., Wong W., Lee C., Ng M., Chan P., Wong K., Leung C., Cheng G. Use of convalescent plasma therapy in SARS patients in Hong Kong. Eur. J. Clin. Microbiol. Infect. Dis. 2005;24:44–46. doi: 10.1007/s10096-004-1271-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Gautret P., Lagier J.-C., Parola P., Meddeb L., Mailhe M., Doudier B., Courjon J., Giordanengo V., Vieira V.E., Dupont H.T. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int. J. Antimicrob. Agents. 2020:105949. doi: 10.1016/j.ijantimicag.2020.105949. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 148.Feldmann M., Maini R.N., Woody J.N., Holgate S.T., Winter G., Rowland M., Richards D., Hussell T. Trials of anti-tumour necrosis factor therapy for COVID-19 are urgently needed. Lancet. 2020;395:1407–1409. doi: 10.1016/S0140-6736(20)30858-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Gerriets V., Bansal P., Khaddour K. StatPearls Publishing; 2019. Tumor Necrosis Factor (TNF) Inhibitors, StatPearls [Internet] [PubMed] [Google Scholar]
- 150.Wang L., He W., Yu X., Hu D., Bao M., Liu H., Zhou J., Jiang H. Coronavirus disease 2019 in elderly patients: characteristics and prognostic factors based on 4-week follow-up. J. Inf. Secur. 2020;80:639–645. doi: 10.1016/j.jinf.2020.03.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Menachery V.D., Yount B.L., Jr., Debbink K., Agnihothram S., Gralinski L.E., Plante J.A., Graham R.L., Scobey T., Ge X.-Y., Donaldson E.F. A SARS-like cluster of circulating bat coronaviruses shows potential for human emergence. Nat. Med. 2015;21:1508. doi: 10.1038/nm.3985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Wysocki J., Ye M., Rodriguez E., González-Pacheco F.R., Barrios C., Evora K., Schuster M., Loibner H., Brosnihan K.B., Ferrario C.M. Targeting the degradation of angiotensin II with recombinant angiotensin-converting enzyme 2: prevention of angiotensin II–dependent hypertension. Hypertension. 2010;55:90–98. doi: 10.1161/HYPERTENSIONAHA.109.138420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Alhenc-Gelas F., Drueke T.B. Blockade of SARS-CoV-2 infection by recombinant soluble ACE2. Kidney Int. 2020;97:1091–1093. doi: 10.1016/j.kint.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Batlle D., Wysocki J., Satchell K. Soluble angiotensin-converting enzyme 2: a potential approach for coronavirus infection therapy? Clin. Sci. 2020;134:543–545. doi: 10.1042/CS20200163. [DOI] [PubMed] [Google Scholar]
- 155.Fukao T., Fukuda Y., Kiga K., Sharif J., Hino K., Enomoto Y., Kawamura A., Nakamura K., Takeuchi T., Tanabe M. An evolutionarily conserved mechanism for microRNA-223 expression revealed by microRNA gene profiling. Cell. 2007;129:617–631. doi: 10.1016/j.cell.2007.02.048. [DOI] [PubMed] [Google Scholar]
- 156.Wang Y., Zhang D., Du G., Du R., Zhao J., Jin Y., Fu S., Gao L., Cheng Z., Lu Q. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020;95:P1569–1578. doi: 10.1016/S0140-6736(20)31022-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.WHO Report . 2020. Coronavirus Disease (COVID-19) Pandemic. [Google Scholar]
- 158.Cao X. 2020. COVID-19: Immunopathology and its Implications for Therapy. [DOI] [PMC free article] [PubMed] [Google Scholar]