Abstract
Background
The impact of postoperative complications (POCs) on the long-term prognosis of patients with colorectal carcinoma was analysed with respect to their severity according to the Clavien-Dindo classification (CDC).
Methods
The prospectively collected data of 2158 patients who underwent curative resection of a colorectal carcinoma (1168 rectal carcinomas, 990 colon carcinomas) without distant metastases from 1995 to 2014 were analysed. The POCs were documented in a standardized form and graded with the CDC. Patients who died postoperatively (CDC grade V, 1.7%) were excluded.
Results
In total, 467 patients (21.6%) had POCs: CDC I, 141 (6.5%); CDC II, 162 (7.5%); CDC III, 112 (5.2%); and CDC IV, 52 (2.4%). More POCs and higher CDC grades were found in men, ASA III-IV patients, rectal carcinoma patients, and patients who underwent abdominoperineal excisions or multivisceral resections. The 5-year locoregional recurrence rate was 5.3% in patients without POCs and 6.6% in patients with POCs. It was highest in CDC III patients (12.9%), which was confirmed in multivariate analysis (HR 2.2; p = 0.005). The 5-year distant metastasis rate was 15.9% in CDC 0 patients and 19.5% in CDC I–IV patients. In multivariate analysis, distant metastasis was highest in CDC III patients (HR 1.7; p = 0.020). The 5-year overall survival rate was 83.5% in patients without POCs and 73.5% in patients with POCs. It was worst in CDC IV patients (63.1%), which was confirmed by multivariate analysis (HR 1.9; p = 0.001).
Conclusion
Patients with POCs after colorectal surgery have a poor long-term prognosis. As the CDC grade increases, survival deteriorates.
Keywords: Colorectal carcinoma, Postoperative complications, Clavien-Dindo classification, Prognosis
Introduction
Quality management in colorectal carcinoma is usually divided into short- and long-term results. How closely these two are linked was first demonstrated with anastomotic leaks. In 1991 Akyol et al. [1] and in 2001 Merkel et al. [2] showed that anastomotic leaks in rectal carcinoma, a traditional short-term quality indicator, was associated with a poor long-term outcome, i.e., elevated locoregional recurrence rates and poor survival. Moreover, this has also been proven in meta-analyses [3]. Over the last few decades, the frequency of anastomotic leaks has decreased due to improvements in surgical techniques. Whether there is a connection between postoperative complications (POCs) in general and long-term prognosis is still under debate, particularly whether a classification of complications can gradually predict the long-term outcome of patients with colorectal cancer.
In 1992, Clavien et al. [4] classified negative outcomes by differentiating complications, sequelae, and failures. Twelve years later, the classification was re-evaluated and modified by one of the authors, focusing on the grading of life-threatening complications and long-term disabilities due to complications [5]. The use of therapeutic consequences as a basis for the classification of complications remained unchanged. Although new attempts to classify complications appeared, the Clavien-Dindo classification (CDC) has been validated and established in international studies across many fields of surgery. Recently, a study of 2266 patients reported that POCs are associated with adverse oncological outcomes, with an increased effect at higher CDC grades [6].
The Erlangen Registry for Colorectal Carcinoma (ERCRC) was established in 1978. Quality management and prognostic factor analysis are the main objectives of this prospective cancer registry. The aim of the current study was to analyse the impact of postoperative complications on long-term prognosis and to assess the severity of complications graded by the CDC and their influence on locoregional recurrence, distant metastasis, disease-free survival, and overall survival.
Methods
The study refers to 2215 consecutive patients who underwent colorectal carcinoma surgery at the Department of Surgery of the University Hospital Erlangen, Germany, between 1995 and 2014. We evaluated the prospectively collected database of the Erlangen Registry for Colorectal Carcinomas (ERCRC) according to the following inclusion criteria: solitary invasive colorectal carcinoma (at least into the submucosa); no appendiceal carcinoma; no other previous or synchronous malignancies; carcinomas not related with familial adenomatous polyposis, ulcerative colitis or Crohn’s disease; no distant metastases; radical elective surgery; and residual tumour classification R0 (no residual tumour at clinical and pathohistological examination). All patients underwent resection with regional lymph node dissection according to the standards of total or partial mesorectal excision (TME, PME) [7] or complete mesocolic excision (CME) [8]. Fifty-seven patients had to be excluded: 37 patients (1.7%) who died postoperatively (Clavien-Dindo classification V) and 20 patients (0.9%) with unknown follow-up information. Finally, we included 2158 patients in the analysis.
Carcinomas with a distal margin ≤ 16 cm to the anal verge were classified as rectal carcinomas and those with a distal margin > 16 cm were classified as colon carcinomas [9].
After preoperative staging, long-term preoperative chemoradiation was administered for locally advanced rectal carcinomas (cT3,4 or cN+) and for selected lower-third carcinomas (< 6 cm) to enable sphincter preservation. Of the 545 patients (46.7%) with neoadjuvant therapy, the majority of the patients (n = 250) received chemoradiation according to the protocol presented by the German CAO/ARO/AIO-94 study [10], with radiotherapy consisting of a total of 50.4 Gy apportioned in 28 fractions of 1.8 Gy, five times weekly and a continuous 120-h infusion of 5-fluorouracil with a dose of 1000 mg/m2 per day during the first and fifth week. A total of 91 patients received oral capecitabine instead of an infusion of 5-FU. In addition, 235 patients received oxaliplatin, 17 patients received irinotecan, and 8 patients received additional cetuximab. During the last few years, hyperthermia was provided to some patients within clinical trials. Six to eight weeks after the completion of preoperative chemoradiation, the patients underwent open TME surgery [7] or PME surgery in selected patients with carcinomas of the upper third (12–16 cm).
Patients with colon carcinomas were treated by CME. The extent of surgery was always determined by the localization of the tumour and its potential lymphatic spread. Carcinomas of the transverse colon, hepatic or splenic flexure were treated with extended hemicolectomies or subtotal colectomies. Adjuvant chemotherapy was recommended for stage III patients and for select stage II patients.
The detailed documentation allowed for a classification of the carcinomas according to the eighth edition of the Tumour Node Metastasis (TNM) classification system [11].
Postoperative complications (POCs) were documented in a standardized form. To assess the severity of these POCs, the Clavien-Dindo classification (CDC) [5, 12] was retrospectively applied. This classification consists of 5 different grades. Grade I comprises any deviation from the normal postoperative course without the need for pharmacological treatment or surgical, endoscopic and radiological interventions. The allowed therapeutic regimens are antiemetics, antipyretics, analgesics, diuretics, electrolytes and physiotherapy. This grade also includes wound infections opened at the bedside. Grade II complications require pharmacological treatment with drugs other than those applied for grade I complications, including blood transfusions and total parenteral nutrition. Grade III complications require surgical, endoscopic or radiological intervention (grade III a, intervention not under general anaesthesia; grade III b, intervention under general anaesthesia). Grade IV complications are life-threatening complications requiring intensive care/intermediate care management (grade IV a, single organ dysfunction including dialysis; grade IV b, multi-organ dysfunction). Grade V complications describe the postoperative death of the patient.
Anastomotic leaks were classified according to Rahbari et al. (grade A, no change in patients’ management; grade B, active therapeutic intervention without relaparotomy; grade C, requiring re-laparotomy [13]). Severe late anastomotic leaks diagnosed after readmission were also considered.
The American Society of Anaesthesiologists (ASA) classifies the comorbidities of the patients into 6 levels [14]. The levels were grouped into ASA I-II and ASA III-IV. The ASA classification was missing in 324 patients.
The patients were followed up for at least 5 years, every 3 months for the first 2 years and every 6 months thereafter; the follow-up included a physical examination, carcinoembryonic antigen (CEA) analysis, abdominal ultrasonography, chest X-ray, computed tomography (CT) of the pelvis and rectoscopy/colonoscopy, depending on localization (rectum or colon) and stage of the primary tumour. In 2004, the follow-up examinations were changed to semi-annually for the first 2 years and then yearly for a total of 5 years according to the first edition of the German S3-Guidelines for Colorectal Carcinoma [15]. A locoregional recurrence was defined as the presence of a recurrent tumour located at the anastomosis, in the tumour bed or in residual lymph nodes, as diagnosed by a clinical and/or pathological examination. Distant metastases were typically diagnosed using imaging techniques; histological confirmation was encouraged. Follow-up data were collected either at the university hospital or from written correspondence with the patients’ family doctors. Thereafter, at least the vital status of each patient was regularly monitored through inquiries at the patients` local registration office. The median follow-up time of all patients was 9 years (0–23 years).
Statistical analysis
The χ2 test and Fisher’s exact test were used to compare frequencies, and the Mann-Whitney U test was used to analyse continuous data. The Kaplan-Meier method was used to calculate the 5-year rates of locoregional recurrence, distant metastasis and survival. Survival curves were compared using a log-rank test. Disease-free survival was defined as the time to the first occurrence of locoregional recurrence, distant metastasis or death by any cause. The endpoint of overall survival was death from any cause. Factors that were found to be significant in the univariate analysis were included in a multivariate Cox regression model. A p value less than 0.05 was considered significant. Statistical analysis was carried out using the statistics software package SPSS® version 21 (IBM, Armonk, New York, USA).
Results
The characteristics of all 2158 patients and their tumours are shown in Table 1. A total of 467 (21.6%) patients had postoperative complications. These complications were distributed across the 4 CDC grades as follows: grade I, n = 141 (6.5%); grade II, n = 162 (7.5%); grade III, n = 112 (5.1%); and grade IV, n = 52 (2.4%). Table 2 shows typical examples of postoperative complications and their respective treatment according to the different CDC grades. The most frequent complications were: for CDC grade I complications, secondary healing (n = 28) and urinary bladder dysfunction (n = 40); for CDC grade II complications, urinary tract infection (n = 40) and central venous catheter infection (n = 23); for CDC grade III, anastomotic leak (n = 27), abdominal wall dehiscence (n = 14) and postoperative bleeding (n = 11); and for CDC grade IV, anastomotic leak (n = 23) and pulmonary complication (n = 12). Compared to other patients, male patients, patients with an ASA level > II, tumour localization in the rectum, abdominoperineal excision and multivisceral resection had significantly more postoperative complications and higher CDC grades.
Table 1.
Patients’ and tumour characteristics for 2158 patients
| Clavien-Dindo classification | All n = 2158 |
0 n = 1691 |
I n = 141 |
II n = 162 |
III n = 112 |
IV n = 52 |
p |
|---|---|---|---|---|---|---|---|
| N (%) | n (%) | n (%) | n (%) | n (%) | n (%) | ||
| Age median (range) (years) | 64.5 (17–94) | 64 (17–94) | 66 (21–87) | 67 (36–93) | 65 (35–88) | 71 (39–90) | < 0.001 |
| Sex | |||||||
| Male | 1351 (62.6) | 1038 (61.4) | 92 (65.2) | 99 (61.1) | 87 (77.7) | 35 (67) | |
| Female | 807 (37.4) | 653 (38.6) | 49 (34.8) | 63 (38.9) | 25 (22.3) | 17 (33) | 0.011 |
| ASA* | |||||||
| ASA I–II | 1498 (81.7) | 1200 (83.7) | 93 (77.5) | 114 (78.6) | 65 (69.9) | 26 (61) | |
| ASA III–IV | 336 (18.3) | 233 (16.3) | 27 (22.5) | 31 (21.4) | 28 (30.1) | 17 (40) | < 0.001 |
| Tumour site | |||||||
| Colon | 990 (45.9) | 807 (47.7) | 61 (43.3) | 62 (38.3) | 33 (29.5) | 27 (52) | |
| Rectum | 1168 (54.1) | 884 (52.3) | 80 (56.7) | 100 (61.7) | 79 (70.5) | 25 (48) | < 0.001 |
| Surgical procedure | |||||||
| (Low) anterior resection | 951 (44.1) | 747 (44.2) | 48 (34.0) | 77 (47.5) | 57 (50.9) | 22 (42) | |
| Abdominoperineal excision | 216 (10.0) | 138 (8.2) | 30 (21.3) | 23 (14.2) | 22 (19.6) | 3 (6) | |
| Colon standard resection | 787 (36.5) | 656 (38.8) | 40 (28.4) | 50 (30.9) | 26 (23.2) | 15 (29) | |
| Colon extended resection | 204 (9.5) | 150 (8.9) | 23 (16.3) | 12 (7.4) | 7 (6.3) | 12 (23) | < 0.001 |
| Multivisceral resection | 246 (11.4) | 170 (10.1) | 26 (18.4) | 23 (14.2) | 17 (15.2) | 10 (19) | 0.003 |
| Multimodal treatment | |||||||
| Neoadjuvant treatment | 550 (25.5) | 420 (24.8) | 37 (26.2) | 43 (26.5) | 39 (34.8) | 11 (21.2) | 0.187 |
| Adjuvant treatment | 658 (30.5) | 537 (31.8) | 44 (31.2) | 41 (25.3) | 28 (25.0) | 8 (15.4) | 0.032 |
| Adjuvant treatment for rectal carcinoma stage II,III | 162/342 (47.4) | 126/253 (49.8) | 10/29 (34) | 11/34 (32) | 10/17 (59) | 5/9 (56) | 0.150 |
| Adjuvant treatment for colon carcinoma stage III | 215/312 (68.9) | 186/257 (72.4) | 14/17 (82) | 10/15 (67) | 5/12 (42) | 0/11 (0) | < 0.001 |
| pT category | |||||||
| pT1,2/ypT0,1,2 | 998 (46.2) | 790 (46.7) | 53 (37.6) | 76 (46.9) | 59 (52.7) | 20 (38) | |
| pT3/ypT3 | 1028 (47.6) | 801 (47.4) | 80 (56.7) | 74 (45.7) | 45 (40.2) | 28 (54) | |
| pT4/ypT4 | 132 (6.1) | 100 (5.9) | 8 (5.7) | 12 (7.4) | 8 (7.1) | 4 (8) | 0.309 |
| pN category | |||||||
| pN0/ypN0 | 1491 (69.1) | 1166 (69.0) | 95 (67.4) | 119 (73.5) | 81 (72.3) | 30 (58) | |
| pN1,2/ypN1,2 | 667 (30.9) | 525 (31.0) | 46 (32.6) | 43 (26.5) | 31 (27.7) | 22 (42) | 0.290 |
| Stage (UICC) | |||||||
| Stage I | 573 (26.6) | 454 (26.8) | 32 (22.7) | 42 (25.9) | 33 (29.5) | 12 (23) | |
| Stage II | 535 (24.8) | 424 (25.1) | 37 (26.2) | 46 (28.4) | 17 (15.2) | 11 (21) | |
| Stage III | 500 (23.2) | 393 (23.2) | 35 (24.8) | 31 (19.1) | 23 (20.5) | 18 (35) | |
| Stage y0 | 73 (3.4) | 60 (3.5) | 2 (1.4) | 4 (2.5) | 7 (6.3) | 0 | |
| Stage yI | 164 (7.6) | 127 (7.5) | 6 (4.3) | 17 (10.5) | 12 (10.7) | 2 (4) | |
| Stage yII | 146 (6.8) | 101 (6.0) | 18 (12.8) | 10 (6.2) | 12 (10.7) | 5 (10) | |
| Stage yIII | 167 (7.7) | 132 (7.8) | 11 (7.8) | 12 (7.4) | 8 (7.1) | 4 (8) | 0.045 |
ASA American Society of Anesthesiologists Classification; *ASA missing in 324 patients
Table 2.
Examples of postoperative complications and treatment according to different CDC grades
| Postoperative complication | CDC I | CDC II | CDC III | CDC IV |
|---|---|---|---|---|
| Pulmonary complication | Mild infection requiring respiratory therapy | Pneumonia requiring antibiotics | Pleural effusion requiring drainage | Respiratory insufficiency requiring ventilation |
| Urologic complication | Urinary bladder dysfunction requiring leg bags or sanitary pads | Urinary tract infection requiring antibiotics; bladder dysfunction treated by anticholinergics or alpha-blocking agents | Urinary bladder dysfunction treated by suprapubic catheterization | Urosepsis requiring intensive care |
| Wound healing disorder | Wound infection opened at the bedside | Phlegmonous wound infection requiring antibiotics | Wound dehiscence requiring surgical abdominal wall closure | Wound infection leading to sepsis requiring intermediate or intensive care |
| Anastomotic leak | Anastomotic leak requiring observation | Anastomotic leak requiring antibiotics | Anastomotic leak requiring drainage or relaparatomy | Major leak with peritonitis and multi-organ dysfunction requiring relaparotomy and intensive care |
Quality indicators
The median number of regional lymph nodes examined in all specimens was 25 (1–145). In patients with rectal carcinoma, the median number of regional lymph nodes examined was 22 (2–76). In patients with primary surgery, it was 27 (4–76), and in those with neoadjuvant treatment, it was 18 (2–62). In patients with colon carcinoma, the median number of regional lymph nodes examined was 29 (1–145). In those who underwent standard colon resections, it was 27 (4–76), and in extended resections, it was 37 (12–145).
The pathological circumferential resection margin (pCRM) was positive (≤ 1 mm) in 20 (2.0%) of the 997 rectal cancer specimens who underwent respective examinations.
The quality of TME/PME [16] was evaluated as mesorectal plane or intramesorectal plane of surgery in 911 (98.3%) of 927 specimens, while 16 (1.7%) specimens were classified as muscularis propria plane.
Anastomotic leaks of grade B and C were observed in 45 (4.8%) of 942 patients after rectal resection and in 23 (2.3%) of 985 patients after colon resection.
Adjuvant treatment
Six hundred fifty-eight patients (30.5%) received adjuvant treatment. The higher the CDC grade, the less frequently adjuvant therapy was administered (31.8% to 15.4%; Table 1). While this was not observed for stage II and III rectal cancer patients without neoadjuvant therapy, it was highly significant for stage III colon carcinomas (p < 0.001).
Prognosis
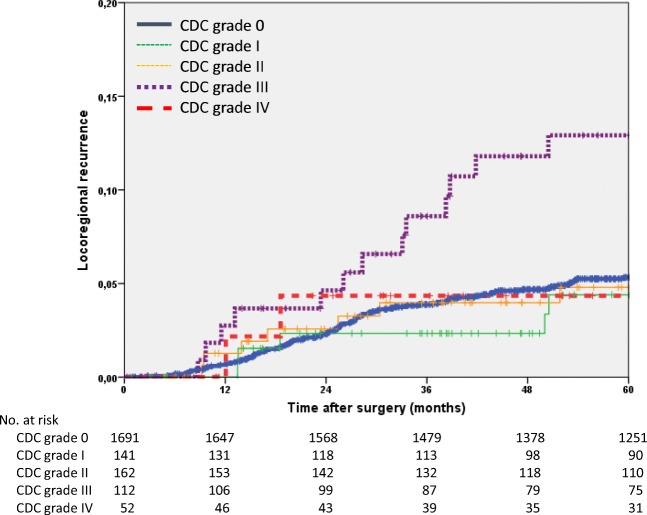
The 5-year locoregional recurrence rate of the entire study group was 5.6% (95% confidence interval 4.6–6.6). The rate was 8.5% for rectal carcinomas compared to 2.2% for colon carcinomas (p < 0.001). The locoregional recurrence rate was significantly higher in men than in women and increased with stage. The rate was higher in patients with POCs (6.6% vs 5.3%; p = 0.068) and was highest in patients with CDC grade III POCs (12.9%; p = 0.007). In the multivariate Cox regression analysis, sex (p = 0.050), tumour site (p < 0.001), stage III tumours (p = 0.005) and CDC grade III POCs were found to significantly influence the locoregional recurrence rate (HR 2.2; p = 0.005; Table 3, Fig. 1).
Table 3.
Univariate and multivariate Cox regression analyses of locoregional recurrences
| Univariate analysis | Multivariate model | ||||||
|---|---|---|---|---|---|---|---|
| n | 5-year rate | 95%CI | p** | Hazard ratio | 95% CI | p | |
| All | 2158 | 5.6 | 4.6–6.6 | ||||
| Age (years) | |||||||
| < 65 | 1079 | 5.7 | 4.3–7.1 | ||||
| ≥ 65 | 1079 | 5.6 | 4.2–7.0 | 0.718 | |||
| Sex | |||||||
| Male | 1351 | 6.5 | 5.1–7.9 | 1.0 | |||
| Female | 807 | 4.1 | 2.7–5.5 | 0.010 | 0.7 | 0.4–1.0 | 0.050 |
| ASA* | |||||||
| ASA I–II | 1498 | 4.8 | 3.6–6.0 | ||||
| ASA III–IV | 336 | 6.4 | 3.5–9.3 | 0.344 | |||
| Tumour site | |||||||
| Colon | 990 | 2.2 | 1.2–3.2 | 1.0 | |||
| Rectum | 1168 | 8.5 | 6.7–10.3 | < 0.001 | 5.3 | 3.2–8.8 | < 0.001 |
| Stage (UICC) | |||||||
| Stage I | 573 | 3.5 | 1.9–5.1 | 1.0 | |||
| Stage II | 535 | 4.6 | 2.8–6.4 | 2.2 | 1.3–4.0 | 0.006 | |
| Stage III | 500 | 7.7 | 5.2–10.2 | 2.7 | 1.5–4.6 | < 0.001 | |
| Stage y0 | 73 | 0 | 0 | 0.954 | |||
| Stage yI | 164 | 4.5 | 1.2–7.8 | 0.6 | 0.3–1.5 | 0.296 | |
| Stage yII | 146 | 9.6 | 4.5–14.7 | 1.7 | 0.9–3.3 | 0.118 | |
| Stage yIII | 167 | 10.2 | 5.3–15.1 | < 0.001 | 1.9 | 1.0–3.5 | 0.061 |
| Clavien-Dindo classification | |||||||
| 0 | 1691 | 5.3 | 4.1–6.5 | 1.0 | |||
| I | 141 | 4.4 | 0.7–8.1 | 0.9 | 0.4–1.8 | 0.683 | |
| II | 162 | 4.8 | 1.3–8.3 | 1.0 | 0.5–1.9 | 0.948 | |
| III | 112 | 12.9 | 6.2–19.6 | 2.2 | 1.3–3.9 | 0.005 | |
| IV | 52 | 4.3 | 0–10.2 | 0.007 | 0.8 | 0.2–3.1 | 0.699 |
ASA American Society of Anesthesiologists Classification; *ASA missing in 324 patients
**Log-rank test
Fig. 1.
Kaplan-Meier curves of the time to locoregional recurrence (n = 2158)
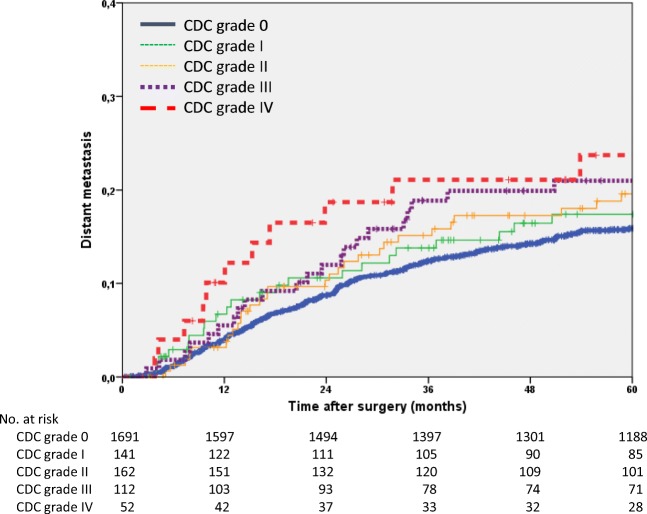
The 5-year distant metastasis rate of the entire study group was 16.7% (95% CI 15.1–18.3). The rate differed significantly between patients with and without POCs (19.5% vs 15.9%; p = 0.009). The univariate analysis also found that ASA classification (p = 0.028), tumour site (p < 0.001) and tumour stage (p < 0.001) were significant factors. The 5-year distant metastasis rate increased as the CDC grade advanced, but this association did not reach significance (p = 0.066). In the multivariate analysis, ASA classification (p = 0.045), tumour site (p < 0.001) and stage III tumours (p = 0.020) were found to be independent prognostic factors. In addition, CDC grade III POCs were again significantly associated with the highest risk for distant metastases (HR 1.7; p = 0.020; Table 4, Fig. 2).
Table 4.
Univariate and multivariate Cox regression analyses of distant metastases
| Univariate analysis | Multivariate model | ||||||
|---|---|---|---|---|---|---|---|
| n | 5-year rate | 95% CI | p** | Hazard ratio | 95% CI | p | |
| All | 2158 | 16.7 | 15.1–18.3 | ||||
| Age (years) | |||||||
| < 65 | 1079 | 16.5 | 14.3–18.7 | ||||
| ≥ 65 | 1079 | 16.9 | 14.5–19.3 | 0.598 | |||
| Sex | |||||||
| Male | 1351 | 17.9 | 15.7–20.1 | ||||
| Female | 807 | 14.6 | 12.1–17.1 | 0.051 | |||
| ASA* | |||||||
| ASA I-II | 1498 | 15.5 | 13.5–17.5 | 1.0 | |||
| ASA III-IV | 336 | 20.0 | 15.3–24.7 | 0.028 | 1.3 | 1.0–1.8 | 0.045 |
| Tumour site | |||||||
| Colon | 990 | 12.9 | 10.7–15.1 | 1.0 | |||
| Rectum | 1168 | 19.9 | 17.5–22.3 | < 0.001 | 1.8 | 1.4–2.4 | < 0.001 |
| Stage (UICC) | |||||||
| Stage I | 573 | 6.3 | 4.1–8.5 | 1.0 | |||
| Stage II | 535 | 13.1 | 10.2–16.0 | 2.5 | 1.7–3.8 | < 0.001 | |
| Stage III | 500 | 28.8 | 24.7–32.9 | 4.7 | 3.3–6.9 | < 0.001 | |
| Stage y0 | 73 | 1.4 | 0–4.1 | 0.1 | 0.0–1.0 | 0.047 | |
| Stage yI | 164 | 8.8 | 4.5–13.1 | 1.0 | 0.6–1.8 | 0.941 | |
| Stage yII | 146 | 14.8 | 7.7–21.9 | 3.3 | 2.1–5.2 | < 0.001 | |
| Stage yIII | 167 | 36.4 | 29.0–43.8 | < 0.001 | 4.8 | 3.2–7.4 | < 0.001 |
| Clavien-Dindo classification | |||||||
| 0 | 1691 | 15.9 | 14.1–17.7 | 1.0 | |||
| I | 141 | 17.4 | 10.7–24.1 | 1.0 | 0.7–1.6 | 0.878 | |
| II | 162 | 19.6 | 13.1–26.1 | 1.4 | 1.0–2.1 | 0.056 | |
| III | 112 | 21.0 | 13.2–28.8 | 1.7 | 1.1–2.5 | 0.020 | |
| IV | 52 | 23.7 | 11.4–36.0 | 0.066 | 1.2 | 0.6–2.5 | 0.543 |
ASA American Society of Anesthesiologists Classification; *ASA missing in 324 patients. **Log-rank test
Fig. 2.
Kaplan-Meier curves of the time to distant metastases (n = 2158)
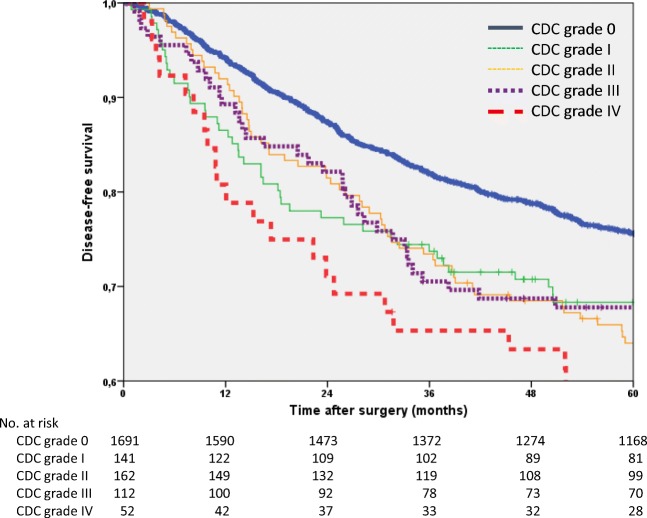
The 5-year disease-free survival rate of the entire study group was 73.4% (95% CI 71.4–75.4). The patients with POCs had a significantly worse disease-free survival (65.2%) than patients without POCs (75.6%; p < 0.001). In the univariate analysis, significant differences were found for age, sex, ASA classification, tumour site, tumour stage and CDC grade. In the multivariate analysis adjusted for age, all of these factors were found to have a significant influence on disease-free survival. Patients with CDC grade IV POCs were found to have the worst disease-free survival compared to patients without POCs (HR 1.8; p = 0.002; Table 5, Fig. 3).
Table 5.
Univariate and multivariate Cox regression analyses of disease-free survival
| Univariate analysis | Multivariate model adjusted for age | ||||||
|---|---|---|---|---|---|---|---|
| n | 5-year rate | 95% CI | p** | Hazard ratio | 95% CI | p | |
| All | 2158 | 73.4 | 71.4–75.4 | ||||
| Age (years) | |||||||
| < 65 | 1079 | 78.7 | 76.2–81.2 | ||||
| ≥ 65 | 1079 | 68.0 | 65.3–70.7 | < 0.001 | |||
| Sex | |||||||
| Male | 1351 | 71.6 | 69.2–74.0 | 1.0 | |||
| Female | 807 | 76.3 | 73.4–79.2 | 0.001 | 0.8 | 0.7–1.0 | 0.015 |
| ASA* | |||||||
| ASA I-II | 1498 | 77.4 | 75.2–79.6 | 1.0 | |||
| ASA III-IV | 336 | 57.1 | 51.8–62.4 | < 0.001 | 1.8 | 1.5–2.1 | < 0.001 |
| Tumour site | |||||||
| Colon | 990 | 76.8 | 74.1–79.5 | 1.0 | |||
| Rectum | 1168 | 70.5 | 68.0–73.0 | 0.047 | 1.4 | 1.2–1.7 | < 0.001 |
| Stage (UICC) | |||||||
| Stage I | 573 | 83.8 | 80.7–86.9 | 1.0 | |||
| Stage II | 535 | 74.4 | 70.7–78.1 | 1.4 | 1.1–1.7 | 0.001 | |
| Stage III | 500 | 61.0 | 56.7–65.3 | 1.9 | 1.5–2.3 | < 0.001 | |
| Stage y0 | 73 | 97.3 | 93.6–100 | 0.6 | 0.3–1.0 | 0.047 | |
| Stage yI | 164 | 85.7 | 80.2–91.2 | 0.8 | 0.6–1.1 | 0.171 | |
| Stage yII | 146 | 65.5 | 57.7–73.3 | 1.6 | 1.2–2.1 | 0.002 | |
| Stage yIII | 167 | 56.2 | 48.6–63.8 | <0.001 | 2.1 | 1.6–2.8 | < 0.001 |
| Clavien-Dindo classification | |||||||
| 0 | 1691 | 75.6 | 73.4–77.8 | 1.0 | |||
| I | 141 | 68.3 | 60.5–76.1 | 1.5 | 1.2–2.0 | 0.002 | |
| II | 162 | 64.0 | 56.6–71.4 | 1.4 | 1.1–1.8 | 0.002 | |
| III | 112 | 67.8 | 59.2–76.4 | 1.4 | 1.1–1.9 | 0.013 | |
| IV | 52 | 55.4 | 41.9–68.9 | <0.001 | 1.8 | 1.2–2.5 | 0.002 |
ASA American Society of Anesthesiologists Classification; *ASA missing in 324 patients. **Log-rank test
Fig. 3.
Kaplan-Meier curves of disease-free survival (n = 2158)
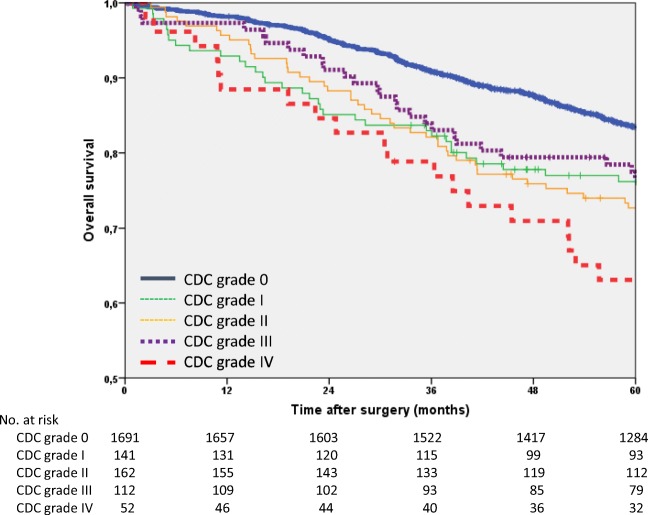
The 5-year overall survival rate of the entire study group was 81.3% (95% CI 79.7–82.9). The patients with POCs had a strikingly decreased overall survival (73.5%) compared to patients without POCs (83.5% p < 0.001). The univariate analysis was significant for age (p < 0.001), sex (p = 0.001), tumour stage (p < 0.001) and CDC grade (p < 0.001). In the multivariate analysis, all of these factors were found to be significant. Patients with CDC grade IV POCs had the worst overall survival (HR 1.9; p = 0.001; Table 6, Fig. 4).
Table 6.
Univariate and multivariate Cox regression analyses of overall survival
| Univariate analysis | Multivariate model adjusted for age | ||||||
|---|---|---|---|---|---|---|---|
| n | 5-year rate | 95% CI | p** | Hazard ratio | 95% CI | p | |
| All | 2158 | 81.3 | 79.7–82.9 | ||||
| Age (years | |||||||
| < 65 | 1079 | 88.3 | 86.3–90.3 | ||||
| ≥ 65 | 1079 | 74.4 | 71.9–76.9 | < 0.001 | |||
| Sex | |||||||
| Male | 1351 | 80.2 | 78.0–82.4 | 1.0 | |||
| Female | 807 | 83.2 | 80.7–85.7 | 0.001 | 0.8 | 0.7–0.9 | 0.002 |
| ASA* | |||||||
| ASA I-II | 1498 | 85.2 | 83.4–87.0 | 85.2 | 1.0 | ||
| ASA III-IV | 336 | 63.6 | 58.3–68.9 | 63.6 | 1.9 | 1.6–2.3 | < 0.001 |
| Tumour site | |||||||
| Colon | 990 | 82.5 | 80.1–84.9 | ||||
| Rectum | 1168 | 80.3 | 77.9–82.7 | 0.306 | |||
| Stage (UICC) | |||||||
| Stage I | 573 | 88.2 | 85.5–90.9 | 1.0 | |||
| Stage II | 535 | 81.7 | 78.4–85.0 | 1.2 | 1.0–1.5 | 0.120 | |
| Stage III | 500 | 70.9 | 66.8–75.0 | 1.7 | 1.3–2.0 | < 0.001 | |
| Stage y0 | 73 | 98.6 | 95.9–100 | 0.7 | 0.4–1.3 | 0.290 | |
| Stage yI | 164 | 94.4 | 90.9–97.9 | 0.9 | 0.6–1.3 | 0.504 | |
| Stage yII | 146 | 74.6 | 67.3–81.9 | 1.8 | 1.3–2.4 | < 0.001 | |
| Stage yIII | 167 | 73.5 | 66.8–80.2 | < 0.001 | 2.3 | 1.7–3.0 | < 0.001 |
| Clavien-Dindo classification | |||||||
| 0 | 1691 | 83.5 | 81.7–85.3 | 1.0 | |||
| I | 141 | 76.2 | 69.1–93.3 | 1.6 | 1.2–2.1 | < 0.001 | |
| II | 162 | 72.7 | 65.8–79.6 | 1.5 | 1.2–1.9 | 0.001 | |
| III | 112 | 76.5 | 68.7–84.3 | 1.4 | 1.1–2.0 | 0.022 | |
| IV | 52 | 63.1 | 50.0–76.2 | < 0.001 | 1.9 | 1.3–2.7 | 0.001 |
ASA American Society of Anesthesiologists Classification; *ASA missing in 324 patients
**Log-rank test
Fig. 4.
Kaplan-Meier curves of overall survival (n = 2158)
Results over time
If the study time is divided and the two periods, 1995–2004 (n = 1097) and 2005–2014 (n = 1061), are compared, we found fewer POCs in the second period (23.2% vs 20.0%; p = 0.066), in particular a decrease in POCs of CDC grade IV was observed (3.4% vs 1.4%; p = 0.003). With regard to long-term outcome, the 5-year locoregional recurrence rate decreased significantly from 7.3% (95%CI 5.7–8.9) to 3.8% (95% CI 2.6–5.0; p = 0.006), while improvements in distant metastasis (18.5% vs 14.8%; p = 0.082), disease-free survival (70.9% vs 76.0%; p = 0.092) and overall survival (79.5% vs 83.3%; p = 0.239) did not reach significance.
Discussion
The present study shows that men, ASA III–IV patients, patients with rectal carcinomas, patients who underwent abdominoperineal excisions and patients who underwent multivisceral resections have more POCs and higher CDC grades than other patients. In investigating the influence of postoperative complications on long-term prognosis, a worse prognosis was found in patients with postoperative complications, especially in patients with high CDC grades. In particular, CDC grade III patients were associated with an increased risk for locoregional recurrences and distant metastases. This corresponds to the recurrence rates in the study by Duraes et al. [6], in which CDC grade III patients also had the worst recurrence rate. In addition, patients with CDC grade IV POCs showed significantly worse disease-free and overall survival rates, which is also consistent with the results of the study mentioned above. Postoperative complications usually depend on the extent of the surgical intervention. Sigmoid resection of a small tumour is typically associated with fewer and less serious complications than total pelvic exenteration [17].
Most patients with CDC grade III complications had surgical complications. In our series, 89% (97 of 112 patients) of the patients with CDC grade III POCs had a surgical complication. These patients required endoscopic, radiological or surgical interventions. Anastomotic leak is a typical example of a CDC grade III complication frequently associated with increased rates of locoregional recurrences and even poor overall survival [2, 3]. This can be explained by exfoliated cancer cells that remain after tumour resection, which may lead to local recurrence induced by a pelvic infection with systemic inflammatory and immunomodulatory responses primarily caused by the anastomotic leak.
CDC grade IV complications are life-threatening and require the patient to be admitted to an intermediate or intensive care unit. The results of the present study show that these patients had the worst disease-free and overall survival rates compared to other patients. It should be noted that these severe complications are not necessarily surgical complications. In the present study, 40% of CDC IV patients had non-surgical complications. Preoperative comorbidities, such as chronic heart failure, chronic obstructive pulmonary disease (COPD) and renal failure, represent a particular risk for non-surgical complications. These comorbidities can lead to single- or multi-organ dysfunction after colorectal surgery, which affects survival outcomes regardless of the original oncological prognosis.
Artinyan et al. [18] showed that postoperative complications after the resection of colorectal carcinomas are an independent risk factor for poor long-term survival. In particular, patients with infectious complications, such as deep and superficial surgical site infections, pneumonia or urinary tract infections, have been associated with increased rates of locoregional recurrence and decreased long-term prognosis. This association is especially impressive in view of the fact that the affected patients in the study were young and had few preoperative comorbidities. In our study, severe infectious complications such as peritonitis, colon necrosis or pneumonia were found in 46% of the patients with CDC IV POCs, who had decreased disease-free and overall survival rates. Previous studies have shown that C-reactive protein (CRP) can predict the occurrence of complications [19] and even the severity of postoperative complications in colorectal cancer [20]. Among patients with anastomotic leaks, patients who required reoperations were found to have worse overall survival compared to those for whom conservative treatment was sufficient [21].
The ASA classification, which assesses the preoperative status of the patient, is an independent factor for morbidity and mortality. An older age is frequently associated with more comorbidities and thus with an increased ASA classification. The present study showed that patients with more serious comorbidities (ASA III or IV classification) were associated with more frequent complications and a higher CDC grade. This is reflected in the worse long-term prognosis with shorter disease-free and overall survival when patients with comorbidities do not recover properly from the complications of surgery. Patients who are more ill are more likely to have a higher complication rate and thus a lower survival rate. As a complication-free course is important for the overall prognosis, good preoperative preparation is essential, particularly for older and high-risk patients. In addition to ensuring appropriately treated comorbidities, such as good control of diabetes and hypertension, optimal preoperative and postoperative nutrition and physical activity play an important role. Malietzis et al. found an association between muscle mass and postoperative morbidity and mortality. They identified myopenia as an independent risk factor for disease-free and overall survival in patients with colorectal cancer [22]. It has also been found that physical exercise programs improve the symptoms of the side effects of chemotherapy and thus enable an increase in the completion rate of chemotherapy in patients with colorectal carcinoma [23], which ultimately affects disease-free and overall survival.
Patients with complications have an increased risk of not receiving adequate adjuvant therapy. The lack of receiving adjuvant therapy postoperatively is known to increase metastasis rate and to decrease survival. In addition, if adjuvant treatment is indicated after surgery, it should start on time; otherwise, the prognosis will deteriorate [24]. According to the German Evidenced-based Guideline for Colorectal Cancer, colon cancer patients should start adjuvant chemotherapy within 8 weeks [15]. This is supported by a complication-free postoperative course, while severe complications may prevent or delay the receipt of adjuvant chemotherapy [3]. In a systematic review and meta-analysis, Biagi et al. identified an inverse correlation between the time to adjuvant chemotherapy and survival [25]. This is supported by the study by Bayraktar et al. [26]. These results showed that starting adjuvant treatment in stage II and III colon carcinoma patients more than 60 days after surgery worsens the prognosis. Lima et al. [27] found no difference in survival between patients who started adjuvant treatment for stage III colon cancer within 8 weeks or 8 to 12 weeks. However, patients who received adjuvant chemotherapy after 12 to 16 weeks had worse survival, and those who started after more than 16 weeks had a prognosis similar to that of patients who did not receive adjuvant treatment. Interestingly, the group of patients who received adjuvant chemotherapy more than 12 weeks after surgery were associated with a poor socioeconomic level and more comorbidities. Nachiappan et al. [28] found that reoperation does not worsen survival if adjuvant chemotherapy can still begin without delay.
Nevertheless, the anatomical extent of the tumour (TNM classification) and presence of residual tumour (R classification) are still the most important prognostic factors for the oncologic outcome. High-quality surgery, as part of a multidisciplinary care strategy, is essential for a complication-free postoperative course and a favourable long-term outcome. The increasing specialization in colorectal surgery contributes to this conclusion [29].
Our study has some limitations, particularly the single-centre design and its retrospective nature. During the long study time of 20 years, treatment protocols have changed, and multimodal treatment has gained importance. For example, the administration of adjuvant chemotherapy for stage III colon cancer nearly doubled from 44% in 1995–1999 to 79% in 2010–2014.
In conclusion, this study identified postoperative complications as an inverse prognostic factor in colorectal cancer with a strong relationship to the Clavien-Dindo classification. It emphasizes the importance of a complication-free postoperative course, not only in view of the immediate postoperative problems and costs but also for the long-term course. This relevance should be known to the entire treating and caring team during the preoperative preparation, the actual surgery and in the postoperative course. It also should be discussed with the patient, so that he/she will definitely attend all follow-up examinations.
Acknowledgements
The present work was performed in fulfilment of the requirements for obtaining the degree “Dr. med.” at the Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU).
Funding Information
Open Access funding provided by Projekt DEAL.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflicts of interest.
Informed consent
According to the Clinical Ethics Committee of the University Hospital Erlangen, no written consent was required for this retrospective analysis of anonymous data.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Akyol AM, McGregor JR, Galloway DJ, Murray GD, George WD. Anastomotic leaks in colorectal cancer surgery: a risk factor for recurrence? Int J Color Dis. 1991;6(4):179–183. doi: 10.1007/BF00341385. [DOI] [PubMed] [Google Scholar]
- 2.Merkel S, Wang WY, Schmidt O, Dworak O, Wittekind C, Hohenberger W, Hermanek P. Locoregional recurrence in patients with anastomotic leakage after anterior resection for rectal carcinoma. Color Disease : the official journal of the Association of Coloproctology of Great Britain and Ireland. 2001;3(3):154–160. doi: 10.1046/j.1463-1318.2001.00232.x. [DOI] [PubMed] [Google Scholar]
- 3.Wang S, Liu J, Zhao H, Ge S, Wang W. Adverse effects of anastomotic leakage on local recurrence and survival after curative anterior resection for rectal cancer: a systematic review and meta-analysis. World J Surg. 2017;41(1):277–284. doi: 10.1007/s00268-016-3761-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Clavien PA, Sanabria JR, Strasberg SM. Proposed classification of complications of surgery with examples of utility in cholecystectomy. Surgery. 1992;111(5):518–526. [PubMed] [Google Scholar]
- 5.Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240(2):205–213. doi: 10.1097/01.sla.0000133083.54934.ae. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Duraes LC, Stocchi L, Steele SR, Kalady MF, Church JM, Gorgun E, Liska D, Kessler H, Lavryk OA, Delaney CP. The relationship between Clavien-Dindo morbidity classification and oncologic outcomes after colorectal cancer resection. Ann Surg Oncol. 2018;25(1):188–196. doi: 10.1245/s10434-017-6142-6. [DOI] [PubMed] [Google Scholar]
- 7.Heald RJ, Husband EM, Ryall RD. The mesorectum in rectal cancer surgery--the clue to pelvic recurrence? Br J Surg. 1982;69(10):613–616. doi: 10.1002/bjs.1800691019. [DOI] [PubMed] [Google Scholar]
- 8.Hohenberger W, Weber K, Matzel K, Papadopoulos T, Merkel S. Standardized surgery for colonic cancer: complete mesocolic excision and central ligation--technical notes and outcome. Color Disease : the official journal of the Association of Coloproctology of Great Britain and Ireland. 2009;11(4):354–364. doi: 10.1111/j.1463-1318.2008.01735.x. [DOI] [PubMed] [Google Scholar]
- 9.Soreide O, Norstein J (eds) (1997) rectal cancer surgery. Optimisation—standardisation—documentation. Springer, Berlin
- 10.Sauer R, Becker H, Hohenberger W, Rodel C, Wittekind C, Fietkau R, Martus P, Tschmelitsch J, Hager E, Hess CF, Karstens JH, Liersch T, Schmidberger H, Raab R. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004;351(17):1731–1740. doi: 10.1056/NEJMoa040694. [DOI] [PubMed] [Google Scholar]
- 11.Brierly JD, Gospodarowicz MK, Wittekind C, editors. TNM classification of malignant tumours. 8. Oxford: Wiley Blackwell; 2017. [Google Scholar]
- 12.Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, de Santibanes E, Pekolj J, Slankamenac K, Bassi C, Graf R, Vonlanthen R, Padbury R, Cameron JL, Makuuchi M. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250(2):187–196. doi: 10.1097/SLA.0b013e3181b13ca2. [DOI] [PubMed] [Google Scholar]
- 13.Rahbari NN, Weitz J, Hohenberger W, Heald RJ, Moran B, Ulrich A, Holm T, Wong WD, Tiret E, Moriya Y, Laurberg S, den Dulk M, van de Velde C, Buchler MW. Definition and grading of anastomotic leakage following anterior resection of the rectum: a proposal by the international study Group of Rectal Cancer. Surgery. 2010;147(3):339–351. doi: 10.1016/j.surg.2009.10.012. [DOI] [PubMed] [Google Scholar]
- 14.Dixon J. Consistency of the ASA classification. Anaesthesia. 1995;50(9):826. doi: 10.1111/j.1365-2044.1995.tb06156.x. [DOI] [PubMed] [Google Scholar]
- 15.Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft DK, AWMF) S3-Leitlinie Kolorektales Karzinom, Langversion 2.1, 2019, AWMF Registrierungsnummer: 021/007OL
- 16.Nagtegaal ID, van de Velde CJ, Marijnen CA, van Krieken JH, Quirke P. Low rectal cancer: a call for a change of approach in abdominoperineal resection. J Clin Oncol : official journal of the American Society of Clinical Oncology. 2005;23(36):9257–9264. doi: 10.1200/JCO.2005.02.9231. [DOI] [PubMed] [Google Scholar]
- 17.Collaborative TP. Surgical and survival outcomes following pelvic exenteration for locally advanced primary rectal Cancer: results from an international collaboration. Ann Surg. 2019;269(2):315–321. doi: 10.1097/SLA.0000000000002528. [DOI] [PubMed] [Google Scholar]
- 18.Artinyan A, Orcutt ST, Anaya DA, Richardson P, Chen GJ, Berger DH. Infectious postoperative complications decrease long-term survival in patients undergoing curative surgery for colorectal cancer: a study of 12,075 patients. Ann Surg. 2015;261(3):497–505. doi: 10.1097/SLA.0000000000000854. [DOI] [PubMed] [Google Scholar]
- 19.Selby J, Prabhudesai A. Can C-reactive protein predict the severity of a post-operative complication after elective resection of colorectal cancer? Int J Color Dis. 2014;29(10):1211–1215. doi: 10.1007/s00384-014-1977-9. [DOI] [PubMed] [Google Scholar]
- 20.McSorley ST, Ramanathan ML, Horgan PG, McMillan DC. Postoperative C-reactive protein measurement predicts the severity of complications following surgery for colorectal cancer. Int J Color Dis. 2015;30(7):913–917. doi: 10.1007/s00384-015-2229-3. [DOI] [PubMed] [Google Scholar]
- 21.Nachiappan S, Askari A, Malietzis G, Giacometti M, White I, Jenkins JT, Kennedy RH, Faiz O. The impact of anastomotic leak and its treatment on cancer recurrence and survival following elective colorectal cancer resection. World J Surg. 2015;39(4):1052–1058. doi: 10.1007/s00268-014-2887-2. [DOI] [PubMed] [Google Scholar]
- 22.Malietzis G, Currie AC, Athanasiou T, Johns N, Anyamene N, Glynne-Jones R, Kennedy RH, Fearon KC, Jenkins JT. Influence of body composition profile on outcomes following colorectal cancer surgery. Br J Surg. 2016;103(5):572–580. doi: 10.1002/bjs.10075. [DOI] [PubMed] [Google Scholar]
- 23.Oruc Z, Kaplan MA. Effect of exercise on colorectal cancer prevention and treatment. World J Gastro Oncol. 2019;11(5):348–366. doi: 10.4251/wjgo.v11.i5.348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gao Z, Parhar A, Gallant V, Heffernan C, Ahmed R, Egedahl ML, Long R. A population-based study of tuberculosis case fatality in Canada: do aboriginal peoples fare less well? Int J Tuberc Lung Disease : the official journal of the International Union against Tuberculosis and Lung Disease. 2015;19(7):772–779. doi: 10.5588/ijtld.14.0753. [DOI] [PubMed] [Google Scholar]
- 25.Biagi JJ, Raphael MJ, Mackillop WJ, Kong WD, King WD, Booth CM. Association between time to initiation of adjuvant chemotherapy and survival in colorectal cancer a systematic review and meta-analysis. Jama-J Am Med Assoc. 2011;305(22):2335–2342. doi: 10.1001/jama.2011.749. [DOI] [PubMed] [Google Scholar]
- 26.Bayraktar UD, Chen E, Bayraktar S, Sands LR, Marchetti F, Montero AJ, Rocha-Lima CMS. Does delay of adjuvant chemotherapy impact survival in patients with resected stage II and III colon adenocarcinoma? Cancer-Am Cancer Soc. 2011;117(11):2364–2370. doi: 10.1002/cncr.25720. [DOI] [PubMed] [Google Scholar]
- 27.Lima ISF, Yasui Y, Scarfe A, Winget M. Association between receipt and timing of adjuvant chemotherapy and survival for patients with stage III colon cancer in Alberta, Canada. Cancer-Am Cancer Soc. 2011;117(16):3833–3840. doi: 10.1002/cncr.25954. [DOI] [PubMed] [Google Scholar]
- 28.Nachiappan S, Askari A, Mamidanna R, Munasinghe A, Currie A, Stebbing J, Faiz O. Initiation of adjuvant chemotherapy within 8 weeks of elective colorectal resection improves overall survival regardless of reoperation. Color Disease : the official journal of the Association of Coloproctology of Great Britain and Ireland. 2016;18(11):1041–1049. doi: 10.1111/codi.13308. [DOI] [PubMed] [Google Scholar]
- 29.Oliphant R, Nicholson GA, Horgan PG, Molloy RG, McMillan DC, Morrison DS. Contribution of surgical specialization to improved colorectal cancer survival. Br J Surg. 2013;100(10):1388–1395. doi: 10.1002/bjs.9227. [DOI] [PubMed] [Google Scholar]