Abstract
The novel coronavirus disease COVID-19 originates in the lungs, but it may extend to other organs, causing, in severe cases, multiorgan damage, including cardiac injury and acute kidney injury. In severe cases, the presence of kidney injury is associated with increased risk of death, highlighting the relevance of this organ as a target of SARS-CoV-2 infection. COVID-19–associated tissue injury is not primarily mediated by viral infection, but rather is a result of the inflammatory host immune response, which drives hypercytokinemia and aggressive inflammation that affect lung parenchymal cells, diminishing oxygen uptake, but also endothelial cells, resulting in endotheliitis and thrombotic events and intravascular coagulation. The complement system represents the first response of the host immune system to SARS-CoV-2 infection, but there is growing evidence that unrestrained activation of complement induced by the virus in the lungs and other organs plays a major role in acute and chronic inflammation, endothelial cell dysfunction, thrombus formation, and intravascular coagulation, and ultimately contributes to multiple organ failure and death. In this review, we discuss the relative role of the different complement activation products in the pathogenesis of COVID-19–associated tissue inflammation and thrombosis and propose the hypothesis that blockade of the terminal complement pathway may represent a potential therapeutic option for the prevention and treatment of lung and multiorgan damage.
Keywords: complement activation, complement terminal pathway, COVID-19, C5 inhibition, kidney injury, vascular injury
“If these folks are not dying of lung failure, they’re dying of renal failure.”
—Jennifer Frontera1
The outbreak of a novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), in December 2019 in Wuhan, China, has become a global pandemic.2
Although the virus initially infects the respiratory epithelium, this is likely merely a portal of entry, and the important steps in alveolar damage are mediated primarily by endothelial injury, resulting in the release of cytokines and chemokines, the recruitment of immune cells, and the activation of coagulation and thrombosis. These events originate in the lungs but may extend to other organs, as documented by reports that patients with severe coronavirus disease 2019 (COVID-19) often develop multiorgan damage, including cardiac injury and acute kidney injury (AKI).3 , 4 In a very recent published article, Cheng et al. 5 state that among 701 consecutive COVID-19 patients hospitalized in Wuhan, 44% had proteinuria, and about 27% had hematuria on admission. After adjustment for age, sex, disease severity, and leukocyte count, patients with elevated baseline serum creatinine or elevated baseline proteinuria had a 2- and 5-fold higher risk of in-hospital death, respectively, than COVID-19 patients with normal kidney parameters.5
Another preprint study6 reports that among 85 cases of COVID-19, 27% developed AKI. Severe acute tubular necrosis and lymphocyte and macrophage infiltration were observed in postmortem kidney tissues from 6 patients who had developed AKI. SARS-CoV-2 nucleocapsid (N) protein was identified in kidney tubules in 2 independent studies,6 , 7 and clusters of coronavirus particles were observed in tubular epithelium and podocytes, 7, 8, 9 suggesting that SARS-CoV-2 directly infects the kidney (Figure 1 ).6 This hypothesis is consistent with the fact that angiotensin-converting enzyme 2—the main SARS-CoV-2 receptor—is highly expressed in the kidneys.10 In the autopsies of COVID-19 patients, angiotensin-converting enzyme 2 expression was upregulated in proximal tubular cells, and strong focal staining was also observed on glomerular parietal epithelial cells.7 There are also nonviral causes of kidney damage in COVID-19 patients, including drug-related nephropathy of antiviral compounds, poor blood oxygenation, and plummeting blood pressure that is also favored by the massive release of cytokines in blood (Figure 1).11
Figure 1.
Coronavirus disease 2019 (COVID-19) and the kidney. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) initially infects the lungs, where it replicates and causes cytolysis of alveolar cells and an inflammatory burst, diminishing oxygen uptake, but the virus can injure many other organs. Kidney dysfunction is common in patients with severe forms of COVID-19 and is an important risk factor for death. SARS-CoV-2 may bind directly to angiotensin-converting enzyme 2 (ACE2) expressed on kidney cells, cause cell injury, and activate the inflammatory response and the complement cascade locally (green box). Kidney damage and dysfunction may also derive from drug nephrotoxicity or systemic events like poor blood oxygenation, lowered blood pressure, or cytokine storms (orange box). AKI, acute kidney injury.
The pathophysiology of COVID-19–associated tissue injury is not well understood, but the therapeutic effect of steroids, used to block progression, indicates that it is not primarily mediated by viral infection, but rather is the result of the inflammatory host immune response, which drives hypercytokinemia and aggressive inflammation.4 , 12
The complement system is the host immune system’s first response to pathogens. However, unrestrained complement activation contributes to acute and chronic inflammation, intravascular coagulation, and cell injury, and it ultimately leads to multiple organ failure and death.13
In a preprint article recently published online,14 an immunohistochemistry analysis of lung tissue from patients who died of COVID-19 revealed strong staining for the complement components mannose-binding lectin (MBL), C4, C3, and the terminal membrane attack complex C5b-9, in alveolar epithelial cells, as well as inflammatory cells, some pneumocytes, and exudates in alveolar spaces. In the same article, the authors described increased serum C5a levels in COVID-19 patients, particularly in severe cases. In another online report,6 an analysis of autoptic kidney tissues from 6 patients revealed strong C5b-9 deposition on tubules in all 6 cases, and low levels of glomerular C5b-9 deposits in 2 of these, demonstrating that viral infection induced complement activation in the kidney as well, which could contribute to tissue injury and organ dysfunction (Figure 1).
Collectively, these results indicate that complement is strongly activated in the lungs, in the circulation, and in the kidneys of COVID-19 patients. Based on the above, in this review, we discuss the double role of the complement system in the immune response against SARS-CoV-2 but also in the pathogenesis of COVID-19–associated tissue inflammation and thrombosis, and propose the hypothesis that blockade of the terminal complement pathway represents a potential therapeutic option for the prevention and treatment of lung and multiorgan damage, for which there is an urgent need to develop effective therapies.
Complement: yin and yang in SARS-CoV infections
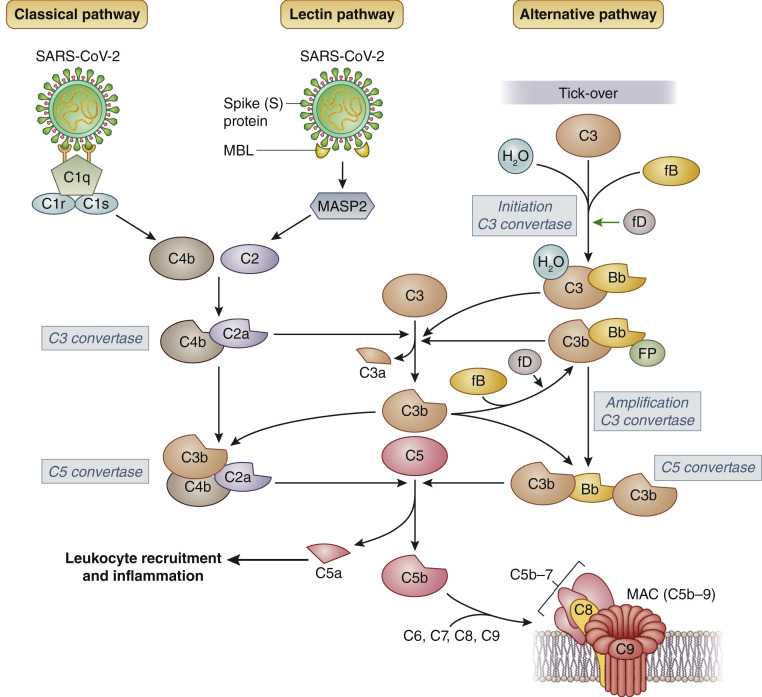
The complement system is made up of over 30 proteins organized in the 3 activation pathways—the classical, the lectin, and the alternative (Figure 2 )—that converge on the cleavage of central component C3 by enzyme complexes—the classical/lectin and the alternative C3 convertases—that generate activation products (C3a, C4a, C3b, and C4b) that function to eliminate pathogens through opsonization, attract and activate neutrophils and macrophages, and enhance humoral immunity and T-cell response.13
Figure 2.
The 3 complement pathways and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The classical pathway is activated by the binding of natural IgM or induced IgG antibodies, which form immune complexes with viral antigens, to the complement component (C) 1 complex, comprising C1q, C1r, and C1s molecules. The lectin pathway is triggered by the binding of mannose-binding lectin (MBL) with SARS-CoV spike (S) protein, which leads to the activation of MBL-associated serine protease 2 (MASP-2). The classical and lectin pathways converge into the cleavage of C2 and C4, leading to the formation of the C3 convertase (C4bC2a) of the classical/lectin pathways. The alternative pathway is continuously activated in plasma by low-grade hydrolysis (tick-over) of C3 that forms C3(H2O). The latter binds to factor B (fB), which, in turn, is cleaved by factor D (fD) to form the alternative pathway fluid-phase initiation C3 convertase. The C3 convertases cleave C3 into C3a, an anaphylotoxin, and C3b, which deposits on cell surfaces. C3b produced by any of the 3 pathways contributes to the formation of the alternative pathway amplification C3 convertase, which cleaves additional C3 molecules, resulting in an amplification loop. In addition, C3b contributes to the formation of the C5 convertases that cleave C5, producing the anaphylatoxin C5a that attracts and activates inflammatory leukocytes, and C5b. C5b initiates the late events of complement activation, leading to the formation of the membrane-attack complex (MAC or C5b-9 complex). Bb, bb fragment of factor B; FP, properdin.
Studies in knockout mice demonstrated that the activation of C3 is required for protection against infection by highly pathogenetic viruses and for proper viral clearance.15 C3–/– mice—which cannot activate any of the 3 complement pathways— infected with the fatal 2009 H1N1 or the Asian Avian H5N1 influenza A viruses had significantly higher viral titers postinfection than wild-type mice, developed more severe bronchitis, bronchiolitis, and vasculitis, and had higher numbers of macrophages infiltrating the lung parenchyma.15
On the other hand, in a mouse model of infection with the SARS-CoV that caused the 2003 coronavirus epidemic, C3–/– mice did not undergo weight loss and exhibited less respiratory dysfunction and fewer lung pathology changes, including less perivascular cuffing and thickening of the interstitial membrane, and fewer neutrophils and macrophages in the lung, compared with wild-type mice, in spite of having an equivalent viral load.16
The contrasting results from the above studies are likely related to differences in the mouse models; however, they highlight the complex role of complement in severe viral infections. Complement is therefore a double-edged sword of our immune system: it may be protective by favoring viral clearance, but unrestrained activation results in acute and chronic inflammation, tissue injury, and the activation of coagulation.
Complement inhibition is being proposed as a potential target in treating COVID-19,17 , 18 but great care should be taken in choosing among the drugs that are currently available or in advanced clinical development.19 In this context, C3 inhibitors do not appear to be a safe option for COVID-19 patients, as these drugs will prevent the activation of all 3 complement pathways in response to viral infections. C5 inhibitors block the formation of the terminal products that are common to the 3 complement pathways, but these drugs will allow C3 activation and opsonization of viral particles.
More studies are required to fully elucidate the relative role of each complement component and pathway in the pathophysiology of COVID-19, but relevant evidence is emerging that could be of help in orienting anti-complement therapy.
SARS-CoV interacts with MBL,20 a C-type lectin that activates the mannose-binding protein–associated serine protease 2 to cleave C4 and C2 and initiate the complement lectin pathway (Figure 2).13
MBL binding to SARS-CoV–infected cells results in enhanced deposition of C4-activation fragments, demonstrating that MBL activates the complement lectin pathway on virus-infected cells.20 A preprint paper showed that the SARS-CoV-2 N protein activates mannose-binding protein–associated serine protease 2 and potentiates C4b deposition in vitro, and mannose-binding protein–associated serine protease 2 and C4 deposits were seen in the lung tissue of COVID-19 patients.14 These findings indicated that both MBL opsonization and deposition of C3 and C4 onto virions are required for SARS-CoV-2 neutralization.
On the other hand, studies on other pandemic viruses suggest that the lectin pathway may contribute to the excessive inflammatory response associated with viral infection. Following inoculation with influenza A viruses H1N1 or H9N2/G1, MBL knockout mice developed less-severe disease, as underlined by less weight loss and lower proinflammatory cytokine and chemokine production compared with wild-type mice, demonstrating that MBL may upregulate inflammatory responses that worsen tissue damage.21
Both natural and induced antibodies form immune complexes with viral antigens that activate C1 and initiate complement activation via the classical pathway. This process was associated with C3b deposition on the viral envelope that interferes with virus attachment to susceptible cells and favors the phagocytosis by macrophages.22 The genetic C3, C4, or C1q deficiency in mice resulted in increased mortality following infection with West Nile virus and impaired antiviral IgG response, whereas antibody response to West Nile virus was normal in mice deficient for factor B or factor D, 2 components of the alternative complement pathway.23 It has also been postulated that the classical pathway plays a pathogenic role in tissue injury. Patients with SARS-CoV infection in phase 2 or 3 of the disease develop autoantibodies against human pulmonary epithelial cells and endothelial cells, and these antibodies can mediate complement-dependent cytotoxicity.24 This phenomenon may derive from cryptic autoantigens exposed on damaged cells or presented by phagocytes that infiltrated the area surrounding the lesion, and it could cause further postinfection cell damage through apoptosis and complement-mediated cytotoxicity.
Further studies are required to establish the relationship between the protective role of the lectin and classical complement pathways in virus clearance versus their potential pathogenetic roles in sustaining inflammatory response and tissue injury. Such studies will be instrumental in conduting clinical studies with specific inhibitors such as anti–mannose-binding protein–associated serine protease 2 antibodies,19 in COVID-19 patients.
The role of the terminal complement pathway in SARS-CoV-2–induced inflammation
A more plausible player in COVID-19–associated tissue inflammatory injury may be the terminal pathway, which represents the common endpoint of the 3 complement activation cascades (Figure 2).25 The binding of C3b to either the classical/lectin or the alternative C3 convertases forms the C5 convertases that cleave C5, generating C5a and C5b. C5a is a potent anaphylatoxin that is involved in exacerbating inflammatory reactions,26 and C5b participates in the formation of C5b-9, which inserts itself into cell membranes, causing cell injury and dysfunction.27
When administered to mice infected with the H1N1 virus, the arthropod-derived inhibitor of C5 activation, OmCI, significantly inhibited leukocyte infiltration in the airways, lung epithelial injury, and overall lung damage,28 suggesting that targeting C5 could be a promising approach to reducing acute lung injury associated with severe forms of viral infections.
In the lungs of mice challenged with Middle East respiratory syndrome coronavirus (MERS-CoV)—which in 2012 caused progressive severe pneumonia with a 35% mortality rate29—the deposition of C5b-9 was observed on the epithelial cells of bronchioles and on pneumocytes and infiltrating leukocytes, and the expression of the C5a receptor C5aR was upregulated in pneumocytes and inflammatory leukocytes.30 Furthermore, serum levels of C5a increased postinfection.30 These data document that MERS-CoV infection rapidly induces both local and systemic C5 activation.
Increased levels of C5a were also found in the plasma of avian influenza virus H7N9–infected monkeys, in serum from patients with H1N1 infection,31 as well as in the bronchoalveolar lavage fluid of mice infected with the highly pathogenic H5N1 virus A but not in mice challenged with the seasonal influenza virus A.15 , 28
C5a promotes monocyte and neutrophil attraction, aggregation, and activation to generate an oxidative burst with the release of reactive oxygen species that exert a critical role in virus-induced lung damage and mortality.32 The interaction between C5a and C5aR on endothelial cells induces the upregulation of adhesion molecules that favor the adhesion of leukocytes and promote their transmigration into lung parenchyma.25 C5a has also been shown to initiate mast cell degranulation, initiate cytokine storms, and increase vascular permeability,33 events that are observed following SARS-CoV infection.34
Results from studies involving pharmacologic manipulation of the C5a–C5aR axis in mouse models support the hypothesis that the C5a plays a role in the inflammatory response associated with fatal viral infections. Treating mice infected with the highly pathogenetic H5N1 virus with an anti-C5a antibody immediately following the challenge significantly reduced neutrophil infiltration in the lungs, attenuated acute lung injury, and increased survival rates.35
In mice, treatment with an anti-C5aR antibody prevented MERS-CoV–induced increase in proinflammatory cytokines interleukin-1β, tumor necrosis factor–α, interferon-gamma, and interleukin-12 in serum and decreased the expression of interferon-gamma receptor and the infiltration of macrophages, neutrophils, and lymphocytes in the lung tissue to a significant degree, compared with vehicle-treated mice,30 , 36 demonstrating that C5a–C5aR interaction mediates local and systemic cytokine release and inflammation following coronavirus infection. In the same model, C5aR blockade limited the thickening of alveolar septa and prevented the development of edema and hemorrhage in the interstitial space in the lungs.30
An anti-human C5a antibody (IFX-1) injected i.v. into African green monkeys infected with the avian influenza A virus H7N9 consistently reduced histopathologic lung injury to a remarkable degree, including endothelial detachment and damage to the basement membrane, and decreased the number of macrophages and neutrophils infiltrating the lung.37 The treatment also mitigated the intensity of systemic inflammatory reaction, as demonstrated by lower plasma levels of inflammatory cytokines and chemokines, compared with that in untreated monkeys.37
The role that C5a plays in human lung disease induced by SARS-CoV-2 has been suggested by a preliminary preprint report on 2 patients who received multiple injections of the C5a antibody BDB-001 in an open label 2-cohort trial in patients with severe and critical COVID-19 from Hubei, China.14 In both patients, clinical conditions improved within a few days, with the normalization of body temperature, increased oxygenation index, decreased C-reactive protein level, and the alleviation of cough, dyspnea, and oppression in the chest.
Coagulation, thrombosis, and vasculitis
Patients with severe COVID-19 infection often develop fulminant activation of coagulation, resulting in widespread microvascular thrombosis and the consumption of coagulation factors. This is reflected by the thrombocytopenia, prolongation of the prothrombin time, elevation of D-dimer, and decreased fibrinogen levels observed in these patients.38 Microangiopathy with schistocytes may be observed on the peripheral smear.39
In a webinar,40 Bin Cao of the National Clinical Research Center for Respiratory Diseases in Beijing described diffuse thrombi in the small vessels of all organs—not only the lungs, but also the heart, liver, and kidney in an autopsy review of COVID-19 deceased patients.
This statement is consistent with published histologic examinations of autoptic tissues from COVID-19 cases. Fibrin thrombi of small arterial vessels were observed in postmortem lung tissue from 33 of 38 Italian patients, half of whom had >25% of tissue involvement, and was associated with high levels of D-dimer in the blood.41
Congested and widened blood vessels of the alveolar septum, with infiltration of monocytes and lymphocytes within and around blood vessels and thrombi in the microvessels, were described in the lungs of 3 deceased patients from China. Furthermore, damage and necrosis of parenchymal cells and thrombi in small vessels were observed in the heart, blood vessels, liver, kidney, and other organs.42 The involvement of microvascular thrombosis in the kidney has not been definitely established. In a recent renal histopathologic analysis of autopsies of COVID-19 patients in China, few fibrin thrombi were identified in glomerular areas associated with severe endothelial injury.7
Microvascular injury and thrombosis with vasculopathy in septal capillaries of the lungs, and permeation by neutrophils and monocytes in the damaged capillaries, were reported in histology examinations of 5 US patients.43 Interestingly, the observations are reminiscent of the vasculitis-like lesions reported in the autopsies of patients with the SARS-CoV infection in 2003, as evidenced by edema of perivascular tissue and vascular walls, with thrombosis, localized necrosis, and infiltration of lymphocytes and monocytes in many organs, including the heart, liver, kidney, and the stroma of striated muscle.44 Relevant to this point is a paper about a 6-month-old baby with COVID-19, who was diagnosed with and treated for Kawasaki disease, a rare form of childhood acute vasculitis affecting vessels throughout the body.45 Other children with COVID-19 and Kawasaki disease–like manifestations are being reported in press releases from clinicians in the United Kingdom, the United States, and Italy.
Altogether, the above evidence demonstrates that SARS-CoV-2–associated thrombotic microangiopathy may progress to proliferative vasculitis with thrombosis of the arterioles and medium and large arterial and venous vessels that can evolve toward disseminated intravascular coagulation. Several factors may contribute to activation of coagulation in COVID-19 patients. Endothelial injury results in upregulation of the expression of tissue factor, which activates the extrinsic coagulation pathway, and lowered expression of the anticoagulant thrombomodulin. Cytokines, neutrophil-derived products, and pathogen-derived molecules increase the synthesis of procoagulant proteins and suppress fibrinolysis.46 Complement-activation products induce tissue factor expression and secretion of von Willebrand factor, and promote the secretion of platelet factor V and the assembly of the functional FXa/FVa complex.47
All the above pathologic changes contribute to respiratory deterioration and multiorgan failure and are important causes of morbidity and mortality, as demonstrated by data showing that among 191 COVID-19 patients seen at 2 hospitals in Wuhan, D-dimer levels over 1 μg/l at admission predicted an 18-fold increase in the odds of dying before discharge.48 For these reasons, the use of anticoagulants has recently been suggested as potentially beneficial in patients with severe COVID-19, although its efficacy and safety have not been demonstrated.49
Endothelial injury and the role of C5 activation products
The pathogenetic mechanism of COVID-19–associated thrombosis and vasculopathy is uncertain, but evidence is emerging that endothelial cell injury and dysfunction play a central role. This possibility is supported by a letter that appeared in The Lancet,50 describing extensive endotheliitis in the lung, heart, liver, kidneys, and small bowel, with an accumulation of inflammatory cells associated with the endothelium and apoptotic bodies in 3 patients with severe COVID-19. Postmortem electron microscopy examination of the kidney graft of a renal transplant patient revealed SARS-CoV-2 viral inclusion structures in endothelial cells.50 In another study, endothelial cell swelling with foam-like changes, subendothelial expansion, and endothelial proliferation were observed in glomerular capillary loops of kidneys from deceased COVID-19 patients.7 The above findings, together with evidence that endothelial cells express angiotensin-converting enzyme 2, which is used by SARS-CoV-2 to infect cells, supports the hypothesis that the virus binding to endothelial cells directly damages them.51 Various immune-mediated disruptive processes triggered by viral infection have also been postulated,52 but there is no evidence-based pathogenetic explanation yet.
Here, we put forward the hypothesis that the products of the terminal pathway—C5a and C5b-9—that are generated following activation of the lectin and classical pathways on cells infected with and damaged by SARS-CoV-2 (Figure 3 ) are key mediators of COVID-19–associated endothelial dysfunction.
Figure 3.
Complement activation and vasculopathy in coronavirus disease 2019 (COVID-19). Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) binds to angiotensin-converting enzyme 2 (ACE2) on endothelial cells and activates the complement lectin pathway first and then the classical pathway, leading to complement (C) 3b deposition. C3b formation fuels the alternative pathway and participates in the formation of the C5 convertases that cleave C5 into the terminal complement products C5a and C5b-9 (upper box). The terminal complement components promote vascular inflammation through multiple processes: the anaphylatoxin C5a recruits neutrophils (polymorphonuclear neutrophils [PMN]) and monocyte/macrophages. C5b-9 and C5a induce the release of interleukin (IL)-8 and monocyte chemoattractant protein (MCP-1) by endothelial cells and stimulates the expression of adhesion molecules (ICAM-1, E-selectin, VCAM-1) that favor recruitment, adhesion, and transendothelial migration of neutrophils and macrophages. Thereafter, infiltrating leukocytes release proteases, cytokines, and reactive oxygen species (ROS) that contribute to inflammation, subendothelial matrix disruption, and remodeling, causing vasculitis-like lesions. C5a and the membrane attack complex (MAC) also induce the exocytosis of P-selectin and von Willebrand factor (VWF) multimers from endothelial cells that promote platelet adhesion, and the shedding of thrombomodulin (TM) from the endothelial cell surface, which triggers the coagulation cascade. Finally, C5b-9 directly activates platelets, causing platelet aggregation and the release of procoagulant microparticles (PMP). The results of all these events are vascular injury and dysfunction, with extensive formation of blood clots.
In addition to promoting inflammation by recruiting and activating neutrophils and macrophages (Figure 3), C5a directly interacts with its C5aR on endothelial cells.53 C5a causes profound perturbations of the physiologically thromboresistant endothelium, including upregulation of tissue factor and loss of thrombomodulin, which activates the coagulation and exocytosis of P-selectin and of ultra-large multimers of von Willebrand factor, which favors platelet adhesion and aggregation.54 Also, C5b-9 induces endothelial activation and dysfunction, including the expression of tissue factor and adhesion molecules,55 and the release of chemokines and platelet-activating factor,56 which together promote inflammation, increase vascular permeability, and trigger the coagulation process. C5b-9 also can be a powerful platelet agonist by inducing the secretion of storage granules and the release of procoagulant platelet microparticles (Figure 3).57
Studies done by our group on atypical hemolytic uremic syndrome (aHUS) and thrombotic thrombocytopenic purpura (TTP)—rare thrombotic microangiopathies characterized by extensive formation of thrombi in the microcirculation of the kidney, brain, and other organs—contributed to highlighting the role of C5 activation products in microvascular thrombosis. In an ex vivo test in which microvascular endothelial cells were incubated with serum from aHUS or TTP patients or healthy controls, both aHUS and TTP serum, but not control serum, induced intense C5b-9 deposits.58 , 59 When the cell monolayer, preexposed to aHUS or TTP serum, was perfused in a flow chamber with normal whole blood, massive thrombus formation occurred.60 Thrombus formation was completely normalized by adding, to aHUS or TTP serum, the humanized monoclonal anti-C5 eculizumab, which blocks C5 cleavage, thus preventing the formation of C5a and C5b-9. A C5aR antagonist significantly reduced but did not fully normalize the thrombus area induced by TTP serum,60 demonstrating that both terminal complement products, C5a and C5b-9, play a role in the loss of endothelial anti-thrombogenic properties.
The role of C5 activation products in COVID-19–associated vasculopathy is supported by a number of pieces of evidence from a recent study43 and unpublished observations (Paola Rizzo, personal communication, May 8, 2020): (i) prominent deposition of C5b-9 was observed within the microvasculature of the interalveolar septa as well as in larger-caliber vessels of the lung parenchyma of 2 deceased COVID-19 patients43; (ii) in the same patients, C5b-9 deposits in septal capillaries colocalized with the SARS-CoV-2 spike and envelope proteins43; (iii) extensive deposits of C5b-9 in the microvasculature, with marked deposition in occluded arteries, were observed in skin biopsies of 3 COVID-19 patients who exhibited purpuric rushes43; and (iv) preliminary autoptic examination of kidneys of 7 COVID-19 patients from Azienda Socio-Sanitaria Territoriale (ASST) Papa Giovanni XXIII, Bergamo in Italy revealed strong C5b-9 staining in peritubular capillaries, and in glomerular afferent and efferent arterioles. Moderate C5b-9 staining was also found in medium and large vessels, and C5b-9 traces were observed in glomeruli (Paola Rizzo, personal communication, May 8, 2020). At variance with these results, C3 staining was faint or absent in all kidney structures, consistent with data regarding Chinese patients.7
The overall alterations induced by the terminal complement pathway may account for what clinicians and pathologists are observing in COVID-19 patients, that is, “although the lungs are ground zero, the virus’ reach can extend to many organs, including the heart and blood vessels, kidneys, gut and brain.”1
The therapeutic perspective of C5 inhibition
Complement C5 inhibition with eculizumab has been shown to be an effective therapeutic tool in thrombotic, hematological, and inflammatory diseases.61 In several trials, as well as in clinical practice, eculizumab protected against microvascular thrombosis and radically improved outcomes for aHUS patients62 , 63; it also prevented hemolysis and reduced thrombotic risk in patients with paroxysmal nocturnal hemoglobinuria, a rare form of complement-mediated hemolytic anemia.64 Eculizumab is also approved for myasthenia gravis, an inflammatory autoimmune disease caused by antibodies that block or destroy nicotinic acetylcholine receptors at the junction between the nerve and muscle.65
In patients with COVID-19, eculizumab, by preventing the cleavage of C5, could exert a favorable effect by blocking the proinflammatory and prothrombotic actions of the terminal products of the complement cascade activated by SARS-CoV-2, while preserving the activity of early complement components that are important for viral clearance and activation of the adaptive immune response.13 , 22 We hypothesize that C5 blockade could also have beneficial effects in countering AKI by preventing C5b-9 accumulation in tubuli. Indeed, experimental models of AKI have shown that tubular epithelial cells are primary targets of C5b-9, which contributes to cell necrosis, exacerbates adenosine triphosphate depletion in hypoxic cells, and triggers inflammatory cell adhesion.66 In a rat model of ischemia/reperfusion-induced AKI, an anti-C5 antibody decreased tubular necrosis and improved the glomerular filtration rate.67
Based on this background, we activated 2 compassionate use protocols for an expanded access program to C5 blockade with eculizumab therapy in patients with mild/moderate (in need of high-flow nasal oxygenation) or advanced (in need of continuous positive airway pressure ventilation) COVID-19 pneumonia, with the objective of stopping complement-mediated lung damage and preventing thromboembolic events. In addition, 3 clinical trials have been registered for off-label compassionate use of eculizumab for the treatment of patients with COVID-19 (ClinicalTrials.gov Identifiers: NCT04288713, NCT04346797, and NCT04355494).68, 69, 70
A preliminary report published recently describes 4 COVID-19 patients with pneumonia requiring oxygen supplementation who were treated off-label with eculizumab.71 The inflammatory markers dropped after eculizumab and the patients recovered in a mean of 13 days. However, the clinical course of the patients is poorly described, and the study is limited by a lack of comparison with COVID-19 controls with the same disease severity and taking the same medications, including anticoagulant and antiviral therapies, hydroxychloroquine, and antibiotics, which could have demonstrated the add-on value of eculizumab in clinical outcomes.
Another therapeutic option could be avacopan, an orally administered C5aR inhibitor that, in a randomized clinical trial in antineutrophil cytoplasmic antibody–associated vasculitis, was effective in replacing high-dose glucocorticoids.72 However, to the best of our knowledge, no clinical trials involving avacopan for COVID-19 patients have been registered yet. A double-blind randomized study has been initiated to evaluate the therapeutic potential of an anti-C5aR monoclonal antibody, avdoralimab, in severe COVID-19 pneumonia (ClinicalTrials.gov Identifier: NCT04371367).73
The results of ongoing controlled studies are needed to establish the therapeutic potential of C5 inhibition in COVID-19, and to discover which patients may benefit the most.
Disclosure
MN has received honoraria from Alexion Pharmaceuticals for giving lectures and participating in advisory boards and research grants from Omeros and Chemocentryx. None of these activities has had any influence on the results or interpretation in this article. AB has received honoraria from Janssen Research & Development, LLC., for consulting activity. GR has consultancy agreements with AbbVie, Alexion Pharmaceuticals, Bayer Healthcare, Reata Pharmaceuticals, Novartis Pharma, AstraZeneca, Otsuka Pharmaceutical Europe, and Concert Pharmaceuticals. No personal remuneration is accepted for these agreements; compensation is paid to AB’s institution for research and educational activities.
Acknowledgments
MN is supported by a grant from the Italian Ministry of Health (project number RF-2016-02361720). The authors thank Kerstin Mierke for editing the manuscript, Dr. Antonella Piccinelli for her contribution in figure drawing, and Fondazione Aiuti per la Ricerca sulle Malattie Rare (ARMR) for the generous support to our work.
References
- 1.Wadman M., Couzin-Frankel J., Kaiser J., Matacic C. A rampage through the body. Science. 2020;368:356–360. doi: 10.1126/science.368.6489.356. pages 356, 358. [DOI] [PubMed] [Google Scholar]
- 2.Remuzzi A., Remuzzi G. COVID-19 and Italy: what next? Lancet. 2020;395:1225–1228. doi: 10.1016/S0140-6736(20)30627-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Guan W.J., Ni Z.Y., Hu Y. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Huang C., Wang Y., Li X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cheng Y., Luo R., Wang K. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020;97:829–838. doi: 10.1016/j.kint.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Diao B, Wang C, Wang R, et al. Human kidney is a target for novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. medRxiv. Available at: 10.1101/2020.03.04.20031120. Accessed June 17, 2020. [DOI] [PMC free article] [PubMed]
- 7.Su H., Yang M., Wan C. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020;98:219–227. doi: 10.1016/j.kint.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Farkash EA, Wilson AM, Jentzen JM. Ultrastructural evidence for direct renal infection with SARS-CoV-2 [e-pub ahead of print]. J Am Soc Nephrol.https://doi.org/10.1681/ASN.2020040432. Accessed May 5, 2020. [DOI] [PMC free article] [PubMed]
- 9.Abbate M., Rottoli D., Gianatti A. COVID-19 attacks the kidney. Ultrastructural evidence for the presence of virus in the glomerular epithelium. Nephron. 2020 doi: 10.1159/000508430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Williams V.R., Scholey J.W. Angiotensin-converting enzyme 2 and renal disease. Curr Opin Nephrol Hypertens. 2018;27:35–41. doi: 10.1097/MNH.0000000000000378. [DOI] [PubMed] [Google Scholar]
- 11.Ronco C., Reis T. Kidney involvement in COVID-19 and rationale for extracorporeal therapies. Nat Rev Nephrol. 2020;16:308–310. doi: 10.1038/s41581-020-0284-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen G., Wu D., Guo W. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130:2620–2629. doi: 10.1172/JCI137244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Noris M., Remuzzi G. Overview of complement activation and regulation. Semin Nephrol. 2013;33:479–492. doi: 10.1016/j.semnephrol.2013.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gao T, Hu M, Zhang X, et al. Highly pathogenic coronavirus N protein aggravates lung injury by MASP-2-mediated complement overactivation. medRxiv. Available at: 10.1101/2020.03.29.20041962. Accessed June 17, 2020. [DOI]
- 15.O'Brien K.B., Morrison T.E., Dundore D.Y. A protective role for complement C3 protein during pandemic 2009 H1N1 and H5N1 influenza A virus infection. PLoS One. 2011;6 doi: 10.1371/journal.pone.0017377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gralinski L.E., Sheahan T.P., Morrison T.E. Complement activation contributes to severe acute respiratory syndrome coronavirus pathogenesis. mBio. 2018;9 doi: 10.1128/mBio.01753-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Risitano A.M., Mastellos D.C., Huber-Lang M. Complement as a target in COVID-19? Nat Rev Immunol. 2020;20:343–344. doi: 10.1038/s41577-020-0320-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Campbell C.M., Kahwash R. Will complement inhibition be the new target in treating COVID-19 related systemic thrombosis. Circulation. 2020;141:1739–1741. doi: 10.1161/CIRCULATIONAHA.120.047419. [DOI] [PubMed] [Google Scholar]
- 19.Mastellos D.C., Ricklin D., Lambris J.D. Clinical promise of next-generation complement therapeutics. Nat Rev Drug Discov. 2019;18:707–729. doi: 10.1038/s41573-019-0031-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ip W.K., Chan K.H., Law H.K. Mannose-binding lectin in severe acute respiratory syndrome coronavirus infection. J Infect Dis. 2005;191:1697–1704. doi: 10.1086/429631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ling M.T., Tu W., Han Y. Mannose-binding lectin contributes to deleterious inflammatory response in pandemic H1N1 and avian H9N2 infection. J Infect Dis. 2012;205:44–53. doi: 10.1093/infdis/jir691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stoermer K.A., Morrison T.E. Complement and viral pathogenesis. Virology. 2011;411:362–373. doi: 10.1016/j.virol.2010.12.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mehlhop E., Diamond M.S. Protective immune responses against West Nile virus are primed by distinct complement activation pathways. J Exp Med. 2006;203:1371–1381. doi: 10.1084/jem.20052388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yang Y.H., Huang Y.H., Chuang Y.H. Autoantibodies against human epithelial cells and endothelial cells after severe acute respiratory syndrome (SARS)-associated coronavirus infection. J Med Virol. 2005;77:1–7. doi: 10.1002/jmv.20407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wang R., Xiao H., Guo R. The role of C5a in acute lung injury induced by highly pathogenic viral infections. Emerg Microbes Infect. 2015;4:e28. doi: 10.1038/emi.2015.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Huber-Lang M., Sarma V.J., Lu K.T. Role of C5a in multiorgan failure during sepsis. J Immunol. 2001;166:1193–1199. doi: 10.4049/jimmunol.166.2.1193. [DOI] [PubMed] [Google Scholar]
- 27.Buras J.A., Rice L., Orlow D. Inhibition of C5 or absence of C6 protects from sepsis mortality. Immunobiology. 2004;209:629–635. doi: 10.1016/j.imbio.2004.09.004. [DOI] [PubMed] [Google Scholar]
- 28.Garcia C.C., Weston-Davies W., Russo R.C. Complement C5 activation during influenza A infection in mice contributes to neutrophil recruitment and lung injury. PLoS One. 2013;8 doi: 10.1371/journal.pone.0064443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zaki A.M., van Boheemen S., Bestebroer T.M. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med. 2012;367:1814–1820. doi: 10.1056/NEJMoa1211721. [DOI] [PubMed] [Google Scholar]
- 30.Jiang Y., Zhao G., Song N. Blockade of the C5a-C5aR axis alleviates lung damage in hDPP4-transgenic mice infected with MERS-CoV. Emerg Microbes Infect. 2018;7:77. doi: 10.1038/s41426-018-0063-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ohta R., Torii Y., Imai M. Serum concentrations of complement anaphylatoxins and proinflammatory mediators in patients with 2009 H1N1 influenza. Microbiol Immunol. 2011;55:191–198. doi: 10.1111/j.1348-0421.2011.00309.x. [DOI] [PubMed] [Google Scholar]
- 32.Akaike T., Ando M., Oda T. Dependence on O2-generation by xanthine oxidase of pathogenesis of influenza virus infection in mice. J Clin Invest. 1990;85:739–745. doi: 10.1172/JCI114499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Woodruff T.M., Nandakumar K.S., Tedesco F. Inhibiting the C5-C5a receptor axis. Mol Immunol. 2011;48:1631–1642. doi: 10.1016/j.molimm.2011.04.014. [DOI] [PubMed] [Google Scholar]
- 34.Franks T.J., Chong P.Y., Chui P. Lung pathology of severe acute respiratory syndrome (SARS): a study of 8 autopsy cases from Singapore. Hum Pathol. 2003;34:743–748. doi: 10.1016/S0046-8177(03)00367-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sun S., Zhao G., Liu C. Inhibition of complement activation alleviates acute lung injury induced by highly pathogenic avian influenza H5N1 virus infection. Am J Respir Cell Mol Biol. 2013;49:221–230. doi: 10.1165/rcmb.2012-0428OC. [DOI] [PubMed] [Google Scholar]
- 36.Jiang Y., Li J., Teng Y. Complement receptor C5aR1 inhibition reduces pyroptosis in hDPP4-transgenic mice infected with MERS-CoV. Viruses. 2019;11:39. doi: 10.3390/v11010039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sun S., Zhao G., Liu C. Treatment with anti-C5a antibody improves the outcome of H7N9 virus infection in African green monkeys. Clin Infect Dis. 2015;60:586–595. doi: 10.1093/cid/ciu887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tang N., Li D., Wang X. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lee AYY, Connors JM, Baumann Kreuziger L, et al. Online COVID-19 resources of the American Society of Hematology. Available at: https://www.hematology.org/covid-19/covid-19-and-coagulopathy. Accessed June 17, 2020.
- 40.Cao B. Chinese Cardiovascular Association and American College of Cardiology, cosponsors. https://youtu.be/CjEhV68GcD8 Available at: Accessed March 19, 2020.
- 41.Carsana L, Sonzogni A, Nasr A, et al. Pulmonary post-mortem findings in a large series of COVID-19 cases from Northern Italy. medRxiv. Available at: 10.1101/2020.04.19.20054262. Accessed June 17, 2020. [DOI] [PMC free article] [PubMed]
- 42.Yao X.H., Li T.Y., He Z.C. A pathological report of three COVID-19 cases by minimally invasive autopsies. Zhonghua Bing Li Xue Za Zhi. 2020;49:411–417. doi: 10.3760/cma.j.cn112151-20200312-00193. [DOI] [PubMed] [Google Scholar]
- 43.Magro C., Mulvey J.J., Berlin D. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res. 2020;220:1–13. doi: 10.1016/j.trsl.2020.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ding Y.Q., Wang H.J., Shen H. Study on etiology and pathology of severe acute respiratory syndrome. Zhonghua Bing Li Xue Za Zhi. 2003;32:195–200. [PubMed] [Google Scholar]
- 45.Jones V.G., Mills M., Suarez D. COVID-19 and Kawasaki disease: novel virus and novel case. Hosp Pediatr. 2020;10:537–540. doi: 10.1542/hpeds.2020-0123. [DOI] [PubMed] [Google Scholar]
- 46.Goeijenbier M., van Wissen M., van de Weg C. Review: viral infections and mechanisms of thrombosis and bleeding. J Med Virol. 2012;84:1680–1696. doi: 10.1002/jmv.23354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Keragala C.B., Draxler D.F., McQuilten Z.K. Haemostasis and innate immunity—a complementary relationship: a review of the intricate relationship between coagulation and complement pathways. Br J Haematol. 2018;180:782–798. doi: 10.1111/bjh.15062. [DOI] [PubMed] [Google Scholar]
- 48.Zhou F., Yu T., Du R. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tang N., Bai H., Chen X. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020;18:1094–1099. doi: 10.1111/jth.14817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Varga Z., Flammer A.J., Steiger P. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.American College of Cardiology/Chinese Cardiovascular Association Myocardial injury with coronavirus. https://youtu.be/CjEhV68GcD8 Available at: Accessed March 19, 2020.
- 52.Bikdeli B., Madhavan M.V., Jimenez D. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up. J Am Coll Cardiol. 2020;75:2950–2973. doi: 10.1016/j.jacc.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zwirner J., Fayyazi A., Gotze O. Expression of the anaphylatoxin C5a receptor in non-myeloid cells. Mol Immunol. 1999;36:877–884. doi: 10.1016/s0161-5890(99)00109-1. [DOI] [PubMed] [Google Scholar]
- 54.Ikeda K., Nagasawa K., Horiuchi T. C5a induces tissue factor activity on endothelial cells. Thromb Haemost. 1997;77:394–398. [PubMed] [Google Scholar]
- 55.Tedesco F., Pausa M., Nardon E. The cytolytically inactive terminal complement complex activates endothelial cells to express adhesion molecules and tissue factor procoagulant activity. J Exp Med. 1997;185:1619–1627. doi: 10.1084/jem.185.9.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kilgore K.S., Ward P.A., Warren J.S. Neutrophil adhesion to human endothelial cells is induced by the membrane attack complex: the roles of P-selectin and platelet activating factor. Inflammation. 1998;22:583–598. doi: 10.1023/a:1022362413939. [DOI] [PubMed] [Google Scholar]
- 57.Sims P.J., Faioni E.M., Wiedmer T. Complement proteins C5b-9 cause release of membrane vesicles from the platelet surface that are enriched in the membrane receptor for coagulation factor Va and express prothrombinase activity. J Biol Chem. 1988;263:18205–18212. [PubMed] [Google Scholar]
- 58.Noris M., Galbusera M., Gastoldi S. Dynamics of complement activation in aHUS and how to monitor eculizumab therapy. Blood. 2014;124:1715–1726. doi: 10.1182/blood-2014-02-558296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Galbusera M., Noris M., Gastoldi S. An ex vivo test of complement activation on endothelium for individualized eculizumab therapy in hemolytic uremic syndrome. Am J Kidney Dis. 2019;74:56–72. doi: 10.1053/j.ajkd.2018.11.012. [DOI] [PubMed] [Google Scholar]
- 60.Bettoni S., Galbusera M., Gastoldi S. Interaction between multimeric von Willebrand factor and complement: a fresh look to the pathophysiology of microvascular thrombosis. J Immunol. 2017;199:1021–1040. doi: 10.4049/jimmunol.1601121. [DOI] [PubMed] [Google Scholar]
- 61.Wijnsma K.L., Ter Heine R., Moes D. Pharmacology, pharmacokinetics and pharmacodynamics of eculizumab, and possibilities for an individualized approach to eculizumab. Clin Pharmacokinet. 2019;58:859–874. doi: 10.1007/s40262-019-00742-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Legendre C.M., Licht C., Muus P. Terminal complement inhibitor eculizumab in atypical hemolytic-uremic syndrome. N Engl J Med. 2013;368:2169–2181. doi: 10.1056/NEJMoa1208981. [DOI] [PubMed] [Google Scholar]
- 63.Noris M., Mescia F., Remuzzi G. STEC-HUS, atypical HUS and TTP are all diseases of complement activation. Nat Rev Nephrol. 2012;8:622–633. doi: 10.1038/nrneph.2012.195. [DOI] [PubMed] [Google Scholar]
- 64.Van Bijnen S.T., Van Heerde W.L., Muus P. Mechanisms and clinical implications of thrombosis in paroxysmal nocturnal hemoglobinuria. J Thromb Haemost. 2012;10:1–10. doi: 10.1111/j.1538-7836.2011.04562.x. [DOI] [PubMed] [Google Scholar]
- 65.Barnett C., Tabasinejad R., Bril V. Current pharmacotherapeutic options for myasthenia gravis. Expert Opin Pharmacother. 2019;20:2295–2303. doi: 10.1080/14656566.2019.1682548. [DOI] [PubMed] [Google Scholar]
- 66.McCullough J.W., Renner B., Thurman J.M. The role of the complement system in acute kidney injury. Semin Nephrol. 2013;33:543–556. doi: 10.1016/j.semnephrol.2013.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zilberman-Itskovich S., Abu-Hamad R., Stark M. Effect of anti-C5 antibody on recuperation from ischemia/reperfusion-induced acute kidney injury. Ren Fail. 2019;41:967–975. doi: 10.1080/0886022X.2019.1677248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Eculizumab (Soliris) in covid-19 infected patients (SOLID-C19). Available at: https://clinicaltrials.gov/ct2/show/NCT04288713. Published February 28, 2020. Updated March 30, 2020. Accessed June 17, 2020.
- 69.Trial evaluating efficacy and safety of eculizumab (Soliris) in patients with COVID-19 infection, nested in the CORIMUNO-19 cohort (CORIMUNO19-ECU). Available at: https://clinicaltrials.gov/ct2/show/NCT04346797. Published April 15, 2020. Updated April 27, 2020. Accessed June 17, 2020.
- 70.SOLIRIS (Eculizumab) treatment of participants with COVID-19. Available at: https://clinicaltrials.gov/ct2/show/NCT04355494. Published April 21, 2020. Accessed June 17, 2020.
- 71.Diurno F., Numis F.G., Porta G. Eculizumab treatment in patients with COVID-19: preliminary results from real life ASL Napoli 2 Nord experience. Eur Rev Med Pharmacol Sci. 2020;24:4040–4047. doi: 10.26355/eurrev_202004_20875. [DOI] [PubMed] [Google Scholar]
- 72.Jayne D.R.W., Bruchfeld A.N., Harper L. Randomized trial of C5a receptor inhibitor avacopan in ANCA-associated vasculitis. J Am Soc Nephrol. 2017;28:2756–2767. doi: 10.1681/ASN.2016111179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Avdoralimab an anti-C5aR antibody, in patients with COVID-19 severe pneumonia (FORCE). Available at: https://clinicaltrials.gov/ct2/show/NCT04371367. Published May 1, 2020. Accessed June 17, 2020.