Abstract
Objectives
The aim of this study was to evaluate the effectiveness of subtotal parathyroidectomy for patients with renal hyperparathyroidism.
Methods
We studied 25 patients with renal hyperparathyroidism who underwent subtotal parathyroidectomy from October 2002 to October 2017. We analyzed serum intact parathyroid hormone (iPTH), calcium, and inorganic phosphorus levels before and at multiple time points following surgery, and evaluated the surgical outcomes and complications.
Results
Of the 25 patients, 13 (52%) were male and 12 (48%) were female, and the mean age was 53.4±9.3 years. The mean duration of dialysis before parathyroidectomy was 156.8±79.5 months. Mean preoperative serum iPTH and calcium levels were 1,199.0±571.3 pg/mL and 10.5±1.0 mg/dL, respectively. At 6 months postoperatively, the mean iPTH and calcium levels decreased to 49.2±47.6 pg/mL (P<0.01) and 8.0±1.0 mg/dL (P<0.01), respectively. Recurrent hyperparathyroidism occurred in two patients: one subsequently underwent kidney transplantation and the other continued hemodialysis and maintained normal calcium levels. One patient developed postoperative permanent hypoparathyroidism.
Conclusion
Subtotal parathyroidectomy is a safe and effective surgical treatment for renal hyperparathyroidism.
Keywords: Secondary Hyperparathyroidism, Chronic Renal Insufficiency, Parathyroidectomy
INTRODUCTION
Secondary hyperparathyroidism is a state of overproduction of parathyroid hormone (PTH), occurring secondary to a chronic abnormal stimulus such as chronic kidney disease (CKD), longterm lithium therapy, gastrointestinal absorption dysfunction (e.g., celiac disease), vitamin D deficiency, liver diseases, and pseudohypoparathyroidism [1-3].
Renal hyperparathyroidism (rHPT) is a complication of CKD, and is characterized by elevated PTH levels secondary to de rangements in the homeostasis of calcium, phosphate, and vitamin D. Hyperphosphatemia, a decreased level of 1,25-dihydroxyvitamin D, and the resultant slight decrease in serum calcium levels are thought to be the main contributors to the pathogenesis of rHPT. Diffuse hyperplasia occurs at the beginning of this disease, but gradually this progresses into nodular hyperplasia as the expression of calcium and vitamin D receptors decreases [3,4].
Surgical treatment of rHPT is preceded by medical treatment, namely restriction of phosphorus intake and the use of dietary calcium supplements, phosphorus binding agents, active vitamin D, and calcimimetics, which maintains serum calcium and phosphorus levels within the normal ranges. Most patients respond to medical treatment, but if this fails or symptoms such as osteoporosis or bone fracture persist, surgical treatment may be considered [1,5].
The three most common surgical procedures used for rHPT are total parathyroidectomy alone, total parathyroidectomy with autotransplantation, and subtotal parathyroidectomy [6]. However, there is controversy as to which procedure is the most effective. Our current understanding of the best surgical approach for rHPT is limited by the small sample size of studies, a lack of randomized controlled trials, and a lack of nationwide data. The aim of this study was to evaluate the effectiveness of subtotal parathyroidectomy in the surgical treatment of rHPT.
MATERIALS AND METHODS
This study was approved by the Institutional Review Board of Hanyang University Guri Hospital (IRB No. 2018-06-016) and was conducted in compliance with the principles of the Declaration of Helsinki. We studied 25 patients with rHPT who underwent subtotal parathyroidectomy between October 2002 and October 2017. Patients with tertiary hyperparathyroidism were excluded from the study. Surgical indications included patients with persistent high serum levels of intact parathyroid hormone (iPTH; usually >500 pg/mL) and hypercalcemia despite adequate medical treatment and those with osteoporosis or subjective symptoms such as bone and joint pain, arthralgia, and pruritus.
Serum iPTH, calcium, and inorganic phosphorus levels were measured by radioimmunoassay preoperatively; immediately after surgery; 1, 3, and 7 days postoperatively; 1, 3, and 6 months postoperatively; and at 3–6-month intervals until the last followup. Preoperative diagnostic imaging such as 99mtechnetium-sestamibi parathyroid scanning (99mTc-sestamibi scanning), neck ultrasonography (US), and computed tomography (CT) was conducted for evaluating the localization of the lesions.
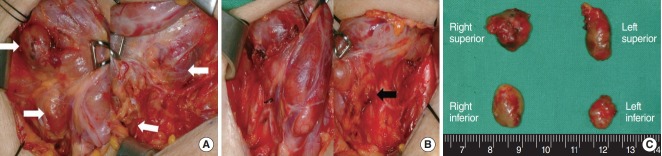
Subtotal parathyroidectomy was performed by making a conventional transcervical incision two finger’s breadth above the sternal notch. Bilateral exploration was conducted to identify all parathyroid glands before the resection of these glands. Subtotal parathyroidectomy involved the resection of three and a half parathyroid glands, leaving between 40 and 80 mg of the smallest gland intact in its original anatomical location after marking it with a metallic clip (Fig. 1).
Fig. 1.
The surgical procedures of subtotal parathyroidectomy. (A) Identification of the bilateral four parathyroid glands before the resection (white arrows). (B) Surgical view after subtotal parathyroidectomy, in which a small part of the left inferior parathyroid gland has been left in situ (black arrow). (C) Surgical specimen from subtotal parathyroidectomy.
In this study, persistent hyperparathyroidism was defined as an iPTH level three times the upper normal limit (>195 pg/mL) immediately following surgery. Recurrent hyperparathyroidism was defined as an iPTH level three times the upper normal limit (>195 pg/mL) 6 months postoperatively. Postoperative permanent hypoparathyroidism was defined as an iPTH level lower than the normal limit (<15 pg/mL) 6 months postoperatively.
Statistical analysis was carried out using SPSS ver. 18.0 (SPSS Inc., Chicago, IL, USA). Paired t-tests were used to analyze the differences between the preoperative and postoperative values of biochemical parameters. A P-value <0.05 was determined to be statistically significant.
RESULTS
Of the 25 patients, 13 (52%) were male and 12 (48%) female, and the mean age was 53.4±9.3 years. All patients had been receiving hemodialysis prior to surgery, and the mean duration of pre-surgical hemodialysis was 156.8±79.5 months. Renal failure was caused by hypertension in 11 patients (44%), glomerulonephritis in six (24%), and polycystic kidney disease in two (8%). Other causes such as diabetes mellitus, hydronephrosis, renal cell carcinoma, and systemic lupus erythematosus were noted. The cause of renal failure was unknown in two patients (Table 1).
Table 1.
Characteristics of patients with renal hyperparathyroidism
| Variable | Value (N=25) |
|---|---|
| Sex | |
| Male | 13 (52) |
| Female | 12 (48) |
| Age (yr) | 53.4 ±9.3 |
| Cause of chronic kidney disease | |
| Hypertension | 11 (44) |
| Glomerulonephritis | 6 (24) |
| Polycystic kidney disease | 2 (8) |
| Diabetes mellitus | 1 (4) |
| Hydronephrosis | 1 (4) |
| Renal cell carcinoma | 1 (4) |
| Systemic lupus erythematosus | 1 (4) |
| Unknown | 2 (8) |
| Symptom | |
| Fatigue | 5 (20) |
| Pruritus | 3 (12) |
| Diarrhea | 2 (8) |
| Arthritis | 2 (8) |
| Abdominal pain | 2 (8) |
| Asymptomatic | 11 (44) |
| Stage of chronic kidney disease | |
| Stage 5 | 25 (100) |
| Hemodialysis | 25 (100) |
| Duration of dialysis (mo) | 156.8 ±79.5 |
| Pathology | |
| Hyperplasia | 25 (100) |
Values are presented as number (%) or mean±standard deviation.
The most frequent clinical manifestations noted were fatigue (20%), pruritis (12%), diarrhea (8%), arthritis (8%), and abdominal pain (8%). Forty-four percent of patients were asymptomatic (Table 1). For preoperative localization of the lesions, 99mTc-sestamibi scanning was conducted in all patients (100%), US in 23 (92%), and CT in 12 (48%). More than three abnormal glands were identified in 69.5%, 50%, and 48% of patients using US, CT, and 99mTc-sestamibi scanning, respectively. When a combination of modalities was utilized, the identification rate of abnormal parathyroid glands increased–more than three abnormal glands were identified in 91.7% of patients using a combination of 99mTc-sestamibi scanning, US, and CT (Table 2).
Table 2.
Imaging modalities utilized for the localization of renal hyperparathyroidism
| Imaging modality | No. of parathyroid glands identified (%) |
|||
|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |
| 99mTc-sestamibi (n=25) | 8 (32.0) | 5 (20.0) | 6 (24.0) | 6 (24.0) |
| US (n=23) | 3 (13.1) | 4 (17.4) | 7 (30.4) | 9 (39.1) |
| CT (n=12) | 1 (8.3) | 5 (41.7) | 4 (33.3) | 2 (16.7) |
| 99mTc-sestamibi and US (n=23) | 2 (8.3) | 4 (20.8) | 7 (29.2) | 10 (41.7) |
| 99mTc-sestamibi and CT (n=11) | 0 | 2 (16.6) | 5 (41.7) | 4 (41.7) |
| US and CT (n=11) | 0 | 2 (16.6) | 4 (33.3) | 5 (50.0) |
| 99mTc-sestamibi, US and CT (n=12) | 0 | 1 (8.3) | 4 (33.4) | 7 (58.3) |
99mTc-sestamibi, 99mtechnetium-sestamibi parathyroid scan; US, ultrasonography; CT, computed tomography.
In terms of complications, no wound infections, hematomas or recurrent laryngeal nerve palsies were observed. Histopathological results showed hyperplasia in all patients. Mean preoperative serum iPTH, calcium, and inorganic phosphorus levels were 1,199.0±571.3 pg/mL (normal range, 15 to 65 pg/mL), 10.5± 1.0 mg/dL (normal range, 8.8 to 10.6 mg/dL), and 6.0±2.3 mg/dL (normal range, 2.5 to 4.5 mg/dL), respectively. No patients exhibited persistent disease immediately after surgery. Six months postoperatively, mean serum iPTH and calcium levels decreased significantly to 49.2±47.6 pg/mL (P<0.01) and 8.0±1.0 mg/dL (P<0.01), respectively.
Recurrent hyperparathyroidism occurred in two patients who exhibited serum iPTH levels of 210.8 and 251.7 pg/mL (Fig. 2). One of these patients underwent kidney transplantation 6 years after the subtotal parathyroidectomy. After transplantation, their iPTH and calcium levels returned to within normal ranges. The other patient has since been continuing hemodialysis and medical treatment, maintaining a normal calcium level.
Fig. 2.
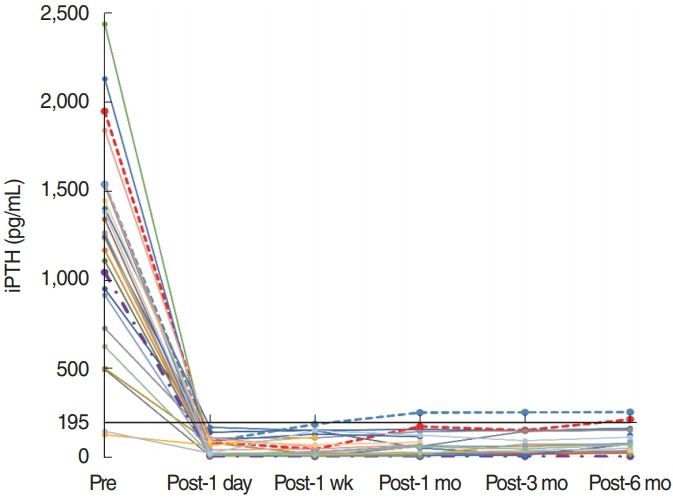
Changes in serum intact parathyroid hormone (iPTH) level before and after subtotal parathyroidectomy. Dashed lines indicate patients with recurrent hyperparathyroidism, and the dash-dotted line indicates one patient with permanent hypoparathyroidism. The solid black line indicates the reference value for recurrent hyperparathyroidism (195 pg/mL). Pre, preoperative; Post, postoperative.
Six months postoperatively, one patient exhibited permanent hypoparathyroidism with a serum iPTH level of 2.3 pg/mL. His serum calcium level was 10.0 mg/dL, which was within the normal range (Fig. 2). Postoperative hypocalcemia occurred in six patients without any hypocalcemic symptoms (Fig. 3). In these six patients, the mean calcium level was 7.1±0.3 mg/dL (range, 6.6 to 7.5 mg/dL), although serum iPTH level was within the normal range. Intermittent administration of oral calcium and vitamin D and hemodialysis with calcium-containing dialysate were performed in these patients.
Fig. 3.
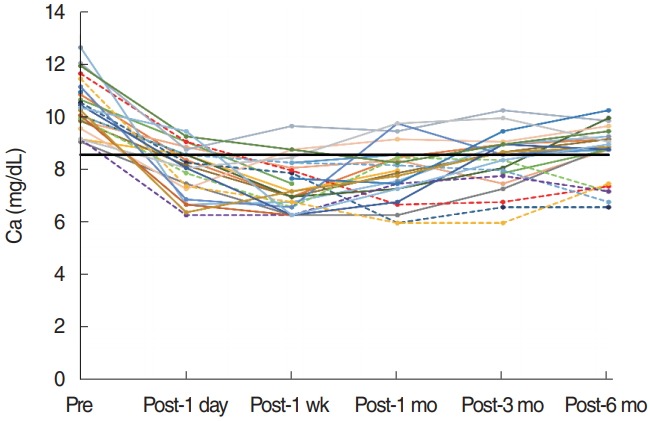
Changes in serum calcium level before and after subtotal parathyroidectomy. Dashed lines indicate patients with postoperative hypocalcemia. The solid black line indicates the reference value for postoperative hypocalcemia. Pre, preoperative; Post, postoperative.
DISCUSSION
The ultimate goals of rHPT treatment are to normalize mineral metabolism and prevent bone disease and extraskeletal manifestations of the altered biochemical processes. The early identification and treatment of rHPT can improve bone and mineral metabolism, thus it is important that all patients with a glomerular filtration rate <60 mL/min/1.73 m2 undergo evaluation of serum calcium, phosphorus, and iPTH levels [3,7].
The initial medical management of rHPT follows a stepwise approach with the goal of optimizing serum phosphorus and calcium levels through a combination of a low phosphorus diet and administration of phosphate binders, vitamin D derivatives, and calcimimetics [1,3,7].
Cinacalcet is a calcimimetic agent that exhibits allosteric modulation of calcium receptors on the parathyroid gland. Cinacalcet is effective even in patients with marked parathyroid hyperplasia, suggesting that it may serve as an alternative to parathyroidectomy for the treatment of severe rHPT. Indeed, the rate of parathyroidectomy has decreased dramatically following the introduction of cinacalcet [1,8].
However, surgical parathyroidectomy remains the definitive therapy for refractory rHPT, drastically decreasing PTH levels and ameliorating the symptoms of severe rHPT. Since the introduction of calcimimetics, approximately 1%–2% of patients with rHPT require parathyroidectomy each year [1,6].
The actual indications for parathyroidectomy for rHPT are poorly defined, but it should be considered when severe rHPT is resistant to medical and pharmacological therapy. The K/DOQI guidelines by the U.S. Kidney Foundation propose that parathyroidectomy should be recommended in patients with severe secondary hyperparathyroidism (persistent serum iPTH levels >800 pg/mL) which is associated with hypercalcemia or hyperphosphatemia and is refractory to medical therapy [9]. The Japanese Society for Dialysis Therapy guidelines recommend surgical treatment at an early stage of secondary hyperparathyroidism, given that progressive secondary hyperparathyroidism is known to influence mortality [10].
Surgical treatment for rHPT should aim for an adequate balance between the extent of parathyroid resection, prevention of persistent/recurrent disease, and avoidance of postoperative permanent hypoparathyroidism. The surgical management of rHPT involves standard bilateral exploration with or without bilateral cervical thymectomy. Since the first parathyroidectomy for secondary hyperparathyroidism was performed by Fergusson in 1967, several surgical approaches have been reported, namely total parathyroidectomy alone, total parathyroidectomy with autotransplantation, subtotal parathyroidectomy, and resection of only the enlarged parathyroid glands [11]. However, the optimal timing, extent, and type of surgery for rHPT remain a constant matter of debate.
Decision-making about the extent of surgery may depend on the surgeon’s preference and the clinical status of the patient, involving factors such as the patient’s age, whether they are a candidate for kidney transplantation, expected survival after parathyroidectomy, and their ability to obtain and comply with medication.
Total parathyroidectomy involves the resection of all four parathyroid and any supernumerary glands. Compared to other procedures, it is associated with a lower recurrence rate, but a higher rate of chronic hypocalcemia and adynamic bone disease [1]. In previous studies, the recurrence rate after total parathyroidectomy varied from 1.9% to 23.8% [12,13]. It is not recommended for patients with CKD stage 5D awaiting kidney transplantation, as the control of serum calcium level may be difficult following the transplantation procedure [14].
Total parathyroidectomy with autotransplantation involves the removal of all four parathyroid glands. For autotransplantation, the most normal-appearing or least nodular gland is identified, and 10–15 mg of this parathyroid tissue is implanted into the brachioradialis muscle or the subcutaneous fat of the forearm. This is advantageous as the autograft can be removed by a relatively simple reoperation without re-exploration of the neck in order to control recurrence [15]. However, Tominaga et al. [16] reported an increasing frequency of autograft excision over time for the treatment of recurrence in 2,260 patients over a period of 29 years—7.5% required removal of the autograft after 5 years, 17.4% after 10 years, and 25.8% after 20 years. Also, some authors have reported difficulty during autograft excision due to the infiltration of implanted parathyroid tissue into the muscular layers [13].
Subtotal parathyroidectomy involves the resection of three and a half parathyroid glands, leaving a small remnant in situ. This is advantageous as it exhibits a lower rate of hypoparathyroidism compared to total parathyroidectomy alone, and is considered a better choice for patients awaiting a kidney transplant [5]. Subtotal parathyroidectomy may also be the preferable approach in predialysis patients, as it results in less pronounced postoperative transient hypoparathyroidism, thereby causing lesser deterioration of kidney function than by total parathyroidectomy [17].
According to several retrospective case series and cohort studies, the rates of persistent or recurrent hyperparathyroidism and postoperative permanent hypoparathyroidism following subtotal parathyroidectomy range between 0 and 12% and 2 and 17%, respectively. For total parathyroidectomy with autotransplantation, these rates vary between 0 and 10% and 0 and 85%, respectively [17].
After subtotal parathyroidectomy, Dotzenrath et al. [18] reported recurrent hyperparathyroidism in only 3.7% of 190 patients. Among these patients, reoperation was performed in 75%, and more than one reoperation was performed in 37.5%. However, an alternative study reported recurrent hyperparathyroidism in 20.8% of 24 patients [19]. In the current study, we obtained satisfying results after subtotal parathyroidectomy. In the majority of patients, iPTH levels dropped dramatically immediately after surgery and were maintained within normal limits. No patients exhibited persistent hyperparathyroidism and only two patients (8.7%) exhibited recurrent hyperparathyroidism 6 months postoperatively, exhibiting normal calcium levels at the last follow-up without reoperation. Postoperative permanent hypoparathyroidism occurred in only one patient who exhibited a normal calcium level and no hypocalcemic symptoms. Schneider et al. [20] considered moderate postoperative hypoparathyroidism as a stable situation in which calcium levels are between 7.2 and 8.8 mg/dL without the administration of oral calcium and/or vitamin D, and no symptoms of hypocalcemia are exhibited. According to this, our patient therefore had moderate hypoparathyroidism.
At present, there is no standardized definition of persistent or recurrent hyperparathyroidism or postoperative permanent hypoparathyroidism which makes the interpretation of data difficult. Several studies, including ours, have defined recurrent hyperparathyroidism after parathyroidectomy as an iPHT level three times the upper normal limit [18,19]. Using this definition, subtotal parathyroidectomy was successful in 88% of patients in this study, and thus this seems to be a useful and effective surgical treatment for rHPT.
rHPT is generally characterized by multiglandular hyperplasia. Therefore, it is necessary to detect all hyperplastic glands and remove all lesions to avoid recurrent hyperparathyroidism. However, the role of preoperative imaging in rHPT is not well defined [21]. Wimmer et al. [22] reported that all glands were localized in 36.7% and 13.3% of patients using solely 99mTcsestamibi scanning and CT, respectively. Furthermore, Sakman et al. [23] reported that the number of glands detected by preoperative imaging increased when 99mTc-sestamibi scanning and US were utilized simultaneously. In this study, we also found that the identification rate of abnormal glands tended to increase when a combination of imaging modalities was utilized. Indeed, more than three abnormal parathyroid glands were localized in 91.7% of patients when 99mTc-sestamibi scanning, US, and CT were performed simultaneously.
There exist some inevitable limitations of this study. It is a non-randomized retrospective study and thus may involve some selection bias. In addition, the sample size utilized in this study is small. Therefore, the long-term surgical outcomes and effectiveness of subtotal parathyroidectomy need to be verified in further studies comprising a larger number of cases and a longterm follow-up period.
In conclusion, based on our results, subtotal parathyroidectomy seems to be a safe and effective surgical treatment for rHPT. Further prospective studies with a larger sample size are necessary to verify these results, and validate the long-term surgical outcomes and effectiveness of subtotal parathyroidectomy.
HIGHLIGHTS
▪ Optimal extent of parathyroidectomy for renal hyperparathyroidism remains controversial.
▪ We evaluated the efficacy of subtotal parathyroidectomy for renal hyperparathyroidism.
▪ Surgical outcomes were successful in most patients with renal hyperparathyroidism.
Footnotes
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS
Conceptualization: KT. Data curation: KT, GHK, CHL, JSP, JYL. Formal analysis: MSK, KT. Methodology: KT, MSK. Project administration: KT. Visualization: all authors. Writing - original draft: MSK. Writing - review & editing: all authors.
REFERENCES
- 1.Madorin C, Owen RP, Fraser WD, Pellitteri PK, Radbill B, Rinaldo A, et al. The surgical management of renal hyperparathyroidism. Eur Arch Otorhinolaryngol. 2012 Jun;269(6):1565–76. doi: 10.1007/s00405-011-1833-2. [DOI] [PubMed] [Google Scholar]
- 2.Cunningham J, Locatelli F, Rodriguez M. Secondary hyperparathyroidism: pathogenesis, disease progression, and therapeutic options. Clin J Am Soc Nephrol. 2011 Apr;6(4):913–21. doi: 10.2215/CJN.06040710. [DOI] [PubMed] [Google Scholar]
- 3.Saliba W, El-Haddad B. Secondary hyperparathyroidism: pathophysiology and treatment. J Am Board Fam Med. 2009 Sep-Oct;22(5):574–81. doi: 10.3122/jabfm.2009.05.090026. [DOI] [PubMed] [Google Scholar]
- 4.Jimenez Vibora E, Areste N, Salgueira M, del Toro N, Jimenez Garcia A, Villar JL, et al. Renal hyperparathyroidism’s control after subtotal parathyroidectomy. Nefrologia. 2005;25(4):416–21. [PubMed] [Google Scholar]
- 5.Konturek A, Barczynski M, Stopa M, Nowak W. Subtotal parathyroidectomy for secondary renal hyperparathyroidism: a 20-year surgical outcome study. Langenbecks Arch Surg. 2016 Nov;401(7):965–74. doi: 10.1007/s00423-016-1447-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.de Francisco AL, Fresnedo GF, Rodrigo E, Pinera C, Amado JA, Arias M. Parathyroidectomy in dialysis patients. Kidney Int Suppl. 2002 May;(80):161–6. doi: 10.1046/j.1523-1755.61.s80.27.x. [DOI] [PubMed] [Google Scholar]
- 7.Moe S, Drueke T, Cunningham J, Goodman W, Martin K, Olgaard K, et al. Definition, evaluation, and classification of renal osteodystrophy: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO) Kidney Int. 2006 Jun;69(11):1945–53. doi: 10.1038/sj.ki.5000414. [DOI] [PubMed] [Google Scholar]
- 8.Stracke S, Keller F, Steinbach G, Henne-Bruns D, Wuerl P. Long-term outcome after total parathyroidectomy for the management of secondary hyperparathyroidism. Nephron Clin Pract. 2009;111(2):c102–9. doi: 10.1159/000191200. [DOI] [PubMed] [Google Scholar]
- 9.National Kidney Foundation K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis. 2003 Oct;42(4 Suppl 3):S1–201. [PubMed] [Google Scholar]
- 10.Guideline Working Group. Japanese Society for Dialysis Therapy Clinical practice guideline for the management of secondary hyperparathyroidism in chronic dialysis patients. Ther Apher Dial. 2008 Dec;12(6):514–25. doi: 10.1111/j.1744-9987.2008.00648.x. [DOI] [PubMed] [Google Scholar]
- 11.Lim CT, Kalaiselvam T, Kitan N, Goh BL. Clinical course after parathyroidectomy in adults with end-stage renal disease on maintenance dialysis. Clin Kidney J. 2018 Apr;11(2):265–9. doi: 10.1093/ckj/sfx086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Liang Y, Sun Y, Ren L, Qi XW, Li Y, Zhang F. Short-term efficacy of surgical treatment of secondary hyperparathyroidism. Eur Rev Med Pharmacol Sci. 2015 Oct;19(20):3904–9. [PubMed] [Google Scholar]
- 13.Liu ME, Qiu NC, Zha SL, Du ZP, Wang YF, Wang Q, et al. To assess the effects of parathyroidectomy (TPTX versus TPTX+AT) for secondary hyperparathyroidism in chronic renal failure: a systematic review and meta-analysis. Int J Surg. 2017 Aug;44:353–362. doi: 10.1016/j.ijsu.2017.06.029. [DOI] [PubMed] [Google Scholar]
- 14.Bellorin-Font E, Ambrosoni P, Carlini RG, Carvalho AB, Correa-Rotter R, Cueto-Manzano A, et al. Clinical practice guidelines for the prevention, diagnosis, evaluation and treatment of mineral and bone disorders in chronic kidney disease (CKD-MBD) in adults. Nefrologia. 2013;33 Suppl 1:1–28. doi: 10.3265/Nefrologia.pre2013.Feb.11945. [DOI] [PubMed] [Google Scholar]
- 15.Xu D, Yin Y, Hou L, Dai W. Surgical management of secondary hyperparathyroidism: how to effectively reduce recurrence at the time of primary surgery. J Endocrinol Invest. 2016 May;39(5):509–14. doi: 10.1007/s40618-015-0410-8. [DOI] [PubMed] [Google Scholar]
- 16.Tominaga Y, Uchida K, Haba T, Katayama A, Sato T, Hibi Y, et al. More than 1,000 cases of total parathyroidectomy with forearm autograft for renal hyperparathyroidism. Am J Kidney Dis. 2001 Oct;38(4 Suppl 1):S168–71. doi: 10.1053/ajkd.2001.27432. [DOI] [PubMed] [Google Scholar]
- 17.Lorenz K, Bartsch DK, Sancho JJ, Guigard S, Triponez F. Surgical management of secondary hyperparathyroidism in chronic kidney disease: a consensus report of the European Society of Endocrine Surgeons. Langenbecks Arch Surg. 2015 Dec;400(8):907–27. doi: 10.1007/s00423-015-1344-5. [DOI] [PubMed] [Google Scholar]
- 18.Dotzenrath C, Cupisti K, Goretzki E, Mondry A, Vossough A, Grabensee B, et al. Operative treatment of renal autonomous hyperparathyroidism: cause of persistent or recurrent disease in 304 patients. Langenbecks Arch Surg. 2003 Jan;387(9-10):348–54. doi: 10.1007/s00423-002-0322-x. [DOI] [PubMed] [Google Scholar]
- 19.Rajeev P, Lee KY, Tang XJ, Goo TT, Tan WB, Ngiam KY. Outcomes of parathyroidectomy in renal hyperparathyroidism in patients with No access to renal transplantation in Singapore. Int J Surg. 2016 Jan;25:64–8. doi: 10.1016/j.ijsu.2015.11.005. [DOI] [PubMed] [Google Scholar]
- 20.Schneider R, Slater EP, Karakas E, Bartsch DK, Schlosser K. Initial parathyroid surgery in 606 patients with renal hyperparathyroidism. World J Surg. 2012 Feb;36(2):318–26. doi: 10.1007/s00268-011-1392-0. [DOI] [PubMed] [Google Scholar]
- 21.Alkhalili E, Tasci Y, Aksoy E, Aliyev S, Soundararajan S, Taskin E, et al. The utility of neck ultrasound and sestamibi scans in patients with secondary and tertiary hyperparathyroidism. World J Surg. 2015 Mar;39(3):701–5. doi: 10.1007/s00268-014-2878-3. [DOI] [PubMed] [Google Scholar]
- 22.Wimmer G, Profanter C, Kovacs P, Sieb M, Gabriel M, Putzer D, et al. CT-MIBI-SPECT image fusion predicts multiglandular disease in hyperparathyroidism. Langenbecks Arch Surg. 2010 Jan;395(1):73–80. doi: 10.1007/s00423-009-0545-1. [DOI] [PubMed] [Google Scholar]
- 23.Sakman G, Parsak CK, Balal M, Seydaoglu G, Eray IC, Saritas G, et al. Outcomes of total parathyroidectomy with autotransplantation versus subtotal parathyroidectomy with routine addition of thymectomy to both groups: single center experience of secondary hyperparathyroidism. Balkan Med J. 2014 Mar;31(1):77–82. doi: 10.5152/balkanmedj.2014.9544. [DOI] [PMC free article] [PubMed] [Google Scholar]