Emerging data from various countries most affected by coronavirus disease 2019 (COVID-19) reveal that hypertension is strongly associated with poor clinical outcomes.
Guan et al. reported data from 1099 patients with confirmed COVID-19, of which the single highest risk factor of infection was hypertension reported in 15% of patients. Among patients who developed severe disease (173 patients), the most common co-morbidity was hypertension (23.7%), and 35.8% of the patients requiring intensive care unit (ICU) admission or mechanical ventilation or who died also had hypertension.1 Zhang et al. studied 140 patients with COVID-19 and found 30% of all patients and 37.9% of those with severe disease had hypertension.2 Hypertension was reported in 23.7–30% of patients who were admitted with the infection and who were linked to more severe infection. Data from Italy looking at patients admitted to the ICU showed 49% (509/1043) had hypertension. These retrospective studies have limitations, given the rapidity of the data collection in the current climate. Italian data were obtained from an ad hoc questionnaire, and China’s data were taken from electronic medical records. Therefore, the timing of hypertension diagnosis, antihypertensive medication usage, adherence and control of hypertension are unknown, which increases the heterogeneity of the patient population. A meta-analysis pooling six studies with 1527 patients showed that hypertension was present in 17.1% of patients with the infection and that patients with severe symptoms had a twofold likelihood of being hypertensive compared to non-severe/non-ICU patients. In this study, the incidence of acute cardiac injury was 13-fold higher in ICU/severe patients.3
The reason for this apparent association remains unclear. Several explanations have been offered, including pre-existing hypertensive cardiac end-organ damage, interactions between COVID-19 and commonly used antihypertensive medications and simply due to the high prevalence of hypertension in older individuals.
Why would hypertension be a risk factor?
COVID-19 seems to follow a pattern seen with influenza and previous severe acute respiratory syndrome coronavirus (SARS-CoV) outbreaks: that the severity and mortality of the infection is higher in the elderly age group. As hypertension is strongly age related, the data could simply be confounded by age. However, an alternative explanation is end-organ damage in hypertensive patients. Hypertension results in a number of pathophysiological changes in the cardiovascular system such as left ventricular hypertrophy and fibrosis. This may make the hypertensive heart particularly susceptible to SARS-CoV-2.
The American College of Cardiology (ACC) produced a clinical bulletin on 6 March 2020 highlighting some of the acute cardiac complications of COVID-19, including acute-onset heart failure, myocardial infarction, myocarditis and cardiac arrest from anecdotal and published data. Wang et al. retrospectively reviewed 138 hospitalised patients with COVID-19, and showed 16.7% of patients developed dysrhythmia and 7.2% experienced acute cardiac injury, defined as serum levels of cardiac biomarkers (e.g. troponin I) above the 99th percentile upper reference limit or new changes on echocardiogram or electrocardiograph.4 Ruan et al. noted similar findings in 150 patients from Wuhan, China, and noted 7% of the 68 deaths occurred due to myocardial damage, with a further 33% dying from both myocardial and respiratory failure. They concluded that COVID-19 could cause fulminant myocarditis.5 This is supported by Huang et al.’s review of 41 patients with COVID-19, of whom 12% had acute cardiac injury with substantially increased hypersensitive troponin I.6 Further data from Zhou et al. indicated that those who did not survive had significantly raised troponin I and lactate dehydrogenase through their clinical course compared to survivors.7 A recent review of 187 patients in Wuhan showed that those with underlying cardiovascular disease and a raised troponin were at a higher risk of death than those with raised troponin and no underlying cardiovascular disease. Additionally, those patients with pre-existing cardiovascular disease were more at risk of developing a raised troponin, suggesting a causal link between underlying cardiovascular conditions (e.g. hypertension) and COVID-19 outcomes.8
COVID-19 and angiotensin-converting enzyme inhibitor/angiotensin receptor blocker controversy
The controversy regarding continuing or discontinuing angiotensin-converting enzyme inhibitors (ACEi) and angiotensin receptor blockers (ARBs) in COVID-19 patients arose after it became apparent that SARS-CoV uses angiotensin-converting enzyme 2 (ACE2) to gain entry in host cells.
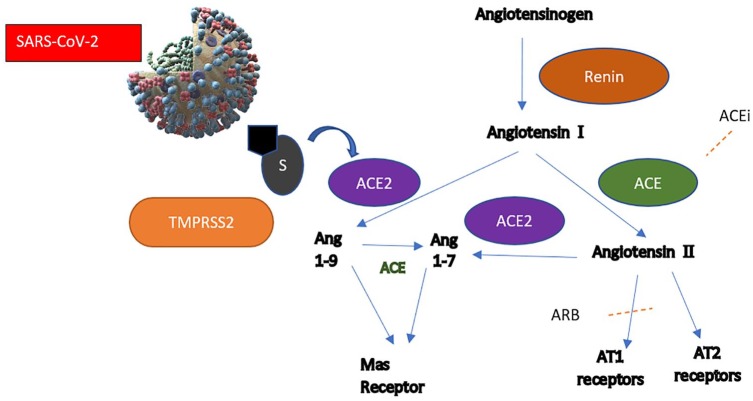
Receptor-binding domains on the spike protein (S) on the virus facilitate cellular entry. S1 of S protein attaches to target cells. This requires priming by cellular protease which cleaves at S1/S2 and the S2 subunit. SARS-CoV engages ACE2 as the entry receptor and employs cellular transmembrane protease serine 2 for priming, as shown in Figure 1.9 ACE2 is a membrane-associated aminopeptidase expressed in vascular endothelia, renal and cardiovascular tissue, and epithelia of the small intestine and testes.10 Efficiency of ACE2 usage was the key determinant in previous SARS infection. SARS-CoV-1 and SARS-CoV-2 share 76–82.3% total amino acid identity. The receptor-binding domains share 72% similarity. There are differences in distinct loops which are replaced in SARS-CoV-2 with flexible glycyl residues, giving it a higher affinity for ACE2 in comparison to SARS-CoV-1.11 ACEi and ARB use may stimulate increased ACE2 mRNA expression. However, the mechanisms by which these medications exert transcriptional control of the ACE2 remain unclear, as gene expression does not entirely correlate with ACE2 activity, as seen in previous mice studies.12 Consequently, the increased expression of ACE2 would facilitate an increased rate or susceptibility to infection with SARS-CoV-2 and further hypothesis that this may increase the risk of developing severe and fatal COVID-19. This comes also from information that undifferentiated cells expressing little ACE2 were poorly infected with coronaviruses, while well-differentiated cells expressing more ACE2 were readily infected.13 On the contrary, upregulation of pulmonary renin–angiotensin–aldosterone system (RAAS), as shown in Figure 1, with increased expression of angiotensin II (Ang2/AngII) is associated with morbidity. ACE and angiotensin receptor type 1 (AT1) lead to multisystem inflammation, and increased ACE and Ang 2 are poor prognostic factors for pneumonia and mortality. This has also been reported in a clinical study of COVID-19 patients, where plasma Ang2 correlated linearly with viral load and lung injury.14 The ACE2 paradox arises from evidence that it not only acts as the entry receptor of SARS-CoV, but also protects the lung from injury.15 Based on previous studies, excessive soluble ACE2, for example using recombinant ACE2, may bind SARS-CoV competitively, allowing preserved cellular ACE2 activity.16,17
Figure 1.
Traditional RAAS pathway, presumed ACE2 pathway and sites of ACEi and ARB action are highlighted. The ACE2 pathway is thought to increase vasodilatory peptides (Ang 1–9, Ang 1–7) and decrease downstream effects of RAAS activation. ACEi might upregulate ACE2 expression. The effect of ARB use is probably slightly different from ACEi, as angiotensin II and ACE levels remain unaffected. Viral entry is thought to be via ACE2 receptor in the presence of transmembrane serine protease.
RAAS: renin–angiotensin–aldosterone system; ACE: angiotensin-converting enzyme; AT1: angiotensin receptor type 1; AT2: angiotensin receptor type 2; ARB: angiotensin receptor blocker; ang: angiotensin; S: spike protein; Mas receptor: Mitochondrial assembly receptor.
The peptides cleaved by ACE2 reduce inflammation and have been suggested as a potential new therapy for inflammatory lung diseases, cancer, diabetes and hypertension. In preclinical studies, ARBs attenuate lipopolysaccharide-induced lung injury, and ACE2 knockout mice are predisposed to lung injury, acute respiratory distress syndrome (ARDS) and lung fibrosis in the long term. Exhaustion of ACE2 leads to reduced angiotensin (1–7) formation, which is a vasodilator, and is also associated with progression of ARDS.18,19 As such, ARBs were proposed as treatment strategies for 2002 SARS.
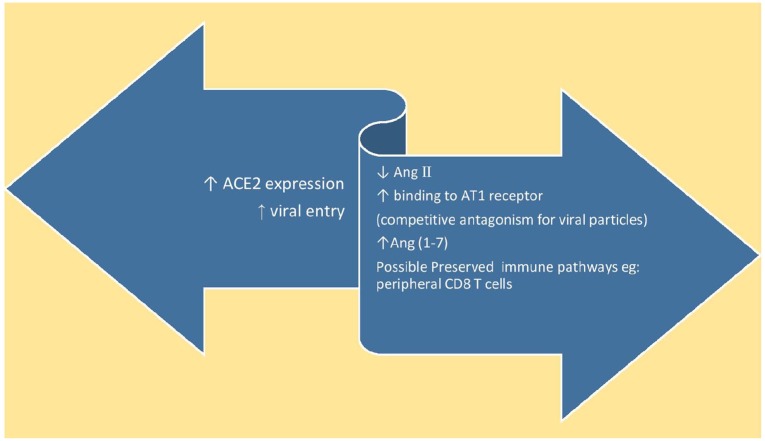
Ramchand et al. studied ACE2 levels in patients with coronary heart disease and did not find any correlation between RAAS blockers or age and plasma ACE2 levels. They found that AT1R and ACE2 physically interact to form complexes on the cell membrane, and Ang2 administration decreases this interaction and induces ubiquitination and ACE2 internalisation and lysosomal degradation.20 Theoretically, increased expression of ACE2 may help counteract the deleterious effects of Ang2, which would provide another mechanism by which RAAS inhibition may be beneficial. However, it is currently unknown if prevention of ACE2 internalisation could lead to clinical benefit in, SARS or COVID-19. In summary, these present opposing hypotheses, as summarised in Figure 2. This is made even more complex by the unknown correlation between plasma/soluble ACE2 and expression at the tissue level in individual organs. Genetic ACE2 deficiency is associated with enhanced atherogenesis in animal studies.21 Studies and subsequent meta-analysis have shown that ACE insertion/deletion polymorphisms increase the risk of ARDS and of mortality in patients with ARDS. They also highlight this polymorphism as being significant in Asian patients, as reported in studies in ICUs.22–25 The relationship between ACE genetic polymorphisms and ACE2 genetic polymorphisms remains unclear.
Figure 2.
Summary of the hypothesized unknown effects of ACEi/ARB use in patients affected by coronavirus disease 2019.
Ang II: angiotensin II.
There are very many questions that need to be answered urgently before these laboratory observations can be translated to the clinic, especially as the face of COVID-19 is a constantly changing one. In a small retrospective study of patients hospitalised in China (n=51), Meng et al. reported that ACEi/ARB use in patients with hypertension and COVID-19 (n=17) had better clinical outcomes and lower peak viral load in comparison to patients with hypertension not on ACEi/ARB (n=25). They report that ACEi/ARB therapy is associated with higher CD8 and CD3 T-cell counts and hence avoids peripheral T-cell depletion, which may play a role.26 More recently, a preprint version of a report on a cohort study conducted in London was made available on the medRixv website. The study tested the hypothesis of an increased risk of severe illness in COVID-19 with hypertension with ACEi use (on ACEi n=37; not on ACEi n=168) in admitted patients. Severe illness was defined as an increased rate of critical unit use for organ support or death within seven days of admission. Similar to the study done in China, they report that the rate of severe illness was lower in patients prescribed ACEi.27 Both of the above studies are relatively small, albeit with encouraging results. Further larger multi-centre, double-blind, placebo-controlled randomised controlled trials of ACEi/ARB in patients with COVID-19 may provide additional insight. As of 14 April 2020, a search on losartan and COVID-19 reveals seven registered studies on ClinicalTrials.Gov.28 Further focussed research is needed to explore the underlying mechanisms better, including the relationship between pulmonary ACE2 and circulating ACE2 and the relationship between COVID-19 spike protein and circulating or pulmonary ACE2.
Management of hypertension during COVID-19
As the interaction of ACEi and ARBs with COVID-19 is unclear, we would recommend a pragmatic approach to hypertension management in the coming months. For newly diagnosed hypertensive patients, we would suggest initiation of calcium channel blockers as first-line therapy in all new patients, irrespective of ethnicity or age. The initiation of ACEi or ARBS requires repeat blood tests to ensure renal function and potassium levels remain stable. As COVID-19 will cause pressure on both primary and secondary care systems, this could cause difficulties in attaining blood tests and the follow-up of results. Once health-care systems have less demand towards the end of the year, consideration of switching to an appropriate ACEi or ARB could be considered, given long-term outcome data. If patients are already on ACEi or ARB therapy, there are currently insufficient data to suggest cessation of this. This view is supported by the European Medicines Agency, the Medicines and Healthcare products Regulatory Agency (MHRA), the European Society of Cardiology (ESC) and the ACC. However, we should remind patients of ‘sick-day rules’, and if they are unable to drink sufficiently and have diarrhoea or vomiting or a persistently low blood pressure, then their ACEi or ARBs medications should be stopped whilst they recover to reduce the risk of acute kidney injury.
To reduce the number of patients attending primary or secondary care services, we would encourage all hypertensive patients to procure upper-arm home blood pressure monitors (BIHS recommended) and to encourage self-monitoring. This would allow for titration of medication through telemedicine clinics29 and would encourage patient-initiated review when required rather than a timed review. We should advise patients to ensure that they have at least two weeks of antihypertensive medications, as there may be delays in procuring repeat prescriptions. We should be open with patients and inform them that we do not fully understand the relationship between COVID-19 and hypertension, but that they should remain up-to-date with the government recommendations regarding hand washing, social distancing and appropriate isolation.
Footnotes
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
- 1. Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. Epub ahead of print 28 February 2020. DOI: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Zhang JJ, Dong X, Cao Y-Y, et al. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy. Epub ahead of print 19 February 2020. DOI: 10.1111/all.14238. [DOI] [PubMed] [Google Scholar]
- 3. Li B, Yang J, Zhao F, et al. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin Res Cardiol. Epub ahead of print 11 March 2020. DOI: 10.1007/s00392-020-01626-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. Epub ahead of print y February 2020. DOI: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Ruan Q, Yang K, Wang W, et al. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. Epub ahead of print 3 March 2020. DOI: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020;395:497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020;395:1054–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Guo T, Fan Y, Chen M, et al. Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID-19). JAMA Cardiol. Epub ahead of print 27 March 2020. DOI: 10.1001/jamacardio.2020.1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Li W, Moore MJ, Vasilieva N, et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003;426:450–454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Donoghue M, Hsief F, Baronas E, et al. , A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9. Circ Res 2000;87:E1–9. [DOI] [PubMed] [Google Scholar]
- 11. Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020;181:271–280.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Ferrario CM, VonCannon J, Jiao Y, et al. Cardiac angiotensin-(1-12) expression and systemic hypertension in rats expressing the human angiotensinogen gene. Am J Physiol Heart Circ Physiol 2016;310:H995–1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Jia HP, Look DC, Shi L, et al. ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia. J Virol 2005;79:14614–14621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Liu Y, Yang Y, Zhang C, et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci 2020;63:364–374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Zhang HA, Penninger JM, Li Y, et al. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med 2020;46:586–590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Khan A, Benthin C, Zeno B, et al. A pilot clinical trial of recombinant human angiotensin-converting enzyme 2 in acute respiratory distress syndrome. Crit Care 2017;21:234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Wosten-Van Asperen RM, Lutter R, Specht PA, et al. Acute respiratory distress syndrome leads to reduced ratio of ACE/ACE2 activities and is prevented by angiotensin-(1-7) or an angiotensin II receptor antagonist. J Pathol 2011;225:618–627. [DOI] [PubMed] [Google Scholar]
- 18. Sun ML, Yang JM, Sun YP, et al. [Inhibitors of RAS might be a good choice for the therapy of COVID-19 pneumonia]. Zhonghua Jie He He Hu Xi Za Zhi 2020;43:E014. [DOI] [PubMed] [Google Scholar]
- 19. Gurwitz D. Angiotensin receptor blockers as tentative SARS-CoV-2 therapeutics. Drug Dev Res. Epub ahead of print 4 March 2020. DOI: 10.1002/ddr.21656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Ramchand J, Yang JM, Sun YP, et al. Elevated plasma angiotensin converting enzyme 2 activity is an independent predictor of major adverse cardiac events in patients with obstructive coronary artery disease. PLoS One 2018;13:e0198144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Thomas MC, Pickering RJ, Tsorotes D, et al. Genetic Ace2 deficiency accentuates vascular inflammation and atherosclerosis in the ApoE knockout mouse. Circ Res 2010;107:888–897. [DOI] [PubMed] [Google Scholar]
- 22. Cardinal-Fernandez P, Ferruelo A, El-Assar M, et al. Genetic predisposition to acute respiratory distress syndrome in patients with severe sepsis. Shock 2013;39:255–260. [DOI] [PubMed] [Google Scholar]
- 23. Matsuda A, Kishi T, Jacob A, et al. Association between insertion/deletion polymorphism in angiotensin-converting enzyme gene and acute lung injury/acute respiratory distress syndrome: a meta-analysis. BMC Med Genet 2012;13:76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Tsantes AE, Kopterides P, Bonovas S, et al. Effect of angiotensin converting enzyme gene I/D polymorphism and its expression on clinical outcome in acute respiratory distress syndrome. Minerva Anestesiol 2013;79:861–870. [PubMed] [Google Scholar]
- 25. Deng X, Zhang S, Jin K, et al. Angiotensin-converting enzyme I/D polymorphism and acute respiratory distress syndrome. J Renin Angiotensin Aldosterone Syst 2015;16:780–786. [DOI] [PubMed] [Google Scholar]
- 26. Meng J, Xiao G, Zhang J, et al. Renin–angiotensin system inhibitors improve the clinical outcomes of COVID-19 patients with hypertension. Emerg Microbes Infect 2020;9:757–760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Bean D, Kraljevic Z, Searle T, et al. Treatment with ACE-inhibitors is associated with less severe disease with SARS-Covid-19 infection in a multi-site UK acute Hospital Trust. medRxiv. Epub ahead of print 11 April 2020. DOI: 10.1101/2020.04.07.20056788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. ClinicalTrials. Losartan COVID-19, https://clinicaltrials.gov/ct2/results?cond=COVID-19&term=losartan&cntry=&state=&city=&dist=&Search=Search&flds=aby (accessed 14 April 2020).
- 29. Hollander JE, Carr BG. Virtually perfect? Telemedicine for Covid-19. N Engl J Med. Epub ahead of print 11 March 2020. DOI: 10.1056/NEJMp2003539. [DOI] [PubMed] [Google Scholar]