Although the most common symptoms of COVID-19 are cough, fever, and anorexia, neurologic manifestations and complications have been observed, particularly among critically ill patients; these include headache, dizziness, encephalopathy, taste and smell impairment, seizures, and stroke [1, 2]. Here, we present two cases of patients with COVID-19 who developed catastrophic intracranial hemorrhage and cerebral edema.
Patient #1
A 62-year-old man with a remote history of colon cancer presented to NYU Langone Medical Center with 1 week of subjective fever, shortness of breath, fatigue, and cough in March 2020. In the emergency room, he was febrile to 38.1 °C, tachycardic, normotensive, tachypneic, and saturating 88% on 15L non-rebreather mask. Chest x-ray was notable for multifocal pneumonia. Nasopharyngeal swab was positive for COVID-19. He was started on ceftriaxone, azithromycin, and plaquenil, and was admitted to a medicine floor. At the time of admission, he was lethargic, but neurologically intact, with a normal mental status, intact cranial nerves, and grossly normal sensory and motor functions. On hospital day (HD) #2, he desaturated to 65% and was emergently intubated, started on vancomycin and zosyn, and transferred to the intensive care unit (ICU). There, he was proned, paralyzed, and sedated, which improved his oxygen saturation.
On HD #3, his D-dimer was > 10,000 ng/mL (from 501 ng/mL on admission). A heparin infusion was started empirically and titrated to an anti-Xa level of 0.3–0.5 IU/mL. With the exception of four anti-Xa levels that were slightly supratherapeutic (0.55–0.73 IU/mL), the level remained within the target range. On HD #5, he was enrolled in a clinical trial and was given sarilumab, an IL-6 receptor antibody, or placebo.
His subsequent hospital course was complicated by hypotension with increasing vasopressor requirements and acute kidney injury necessitating initiation of dialysis on HD #10. Paralytics were discontinued on HD #7, and all sedative infusions were weaned off by HD #10.
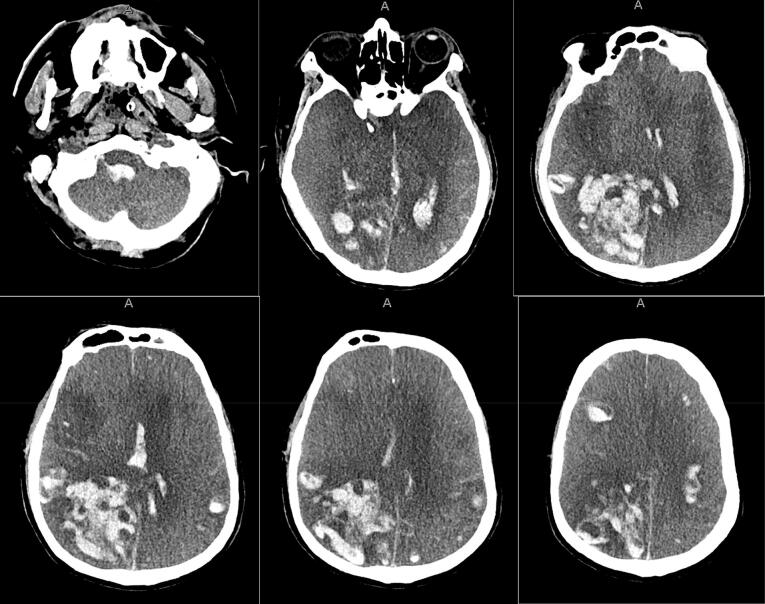
On HD #13, neurocritical care was consulted due to persistent encephalopathy and concern for absent brainstem reflexes. It was unclear when he last had brainstem reflexes. He was found to be comatose with absent brainstem reflexes, with the exception of the vestibulocochlear reflex. He was minimally breathing over the ventilator. Table 1 shows vitals and laboratory results. Non-contrast head CT (NCHCT) demonstrated multifocal intraparenchymal hemorrhage with intraventricular extension, mass effect, and evidence of global anoxic injury, cerebral edema, and downward herniation of the cerebellar tonsils (Fig. 1). Given his grave prognosis, the family decided to make the patient DNR, and the patient died on HD #13.
Table 1.
Blood pressure and laboratory results
| Patient #1 mean (range) | Patient #2 mean (range) | |
|---|---|---|
| Vital signs | ||
| Systolic blood pressure (BP) while on heparin (mmHg) | 113 (70–197) | 117 (47–204) |
| Diastolic BP while on heparin (mmHg) | 67 (40–104) | 58 (29–112) |
| Oxygen saturation | 94% (66–100%) | 94% (71–100%) |
| Coagulation/hematologic labs | ||
| Anti-Xa (IU/mL) | 0.42 (0.23–0.73) | 0.30 (0.01–0.62) |
| D-dimer (ng/mL) | 2997 (460- > 10,000) | 1608 (677–4488) |
| White blood cell count (WBC) (× 103/uL) | 7.75 (4.7–17.4) | 9.7 (4.6–18.3) |
| Platelets (× 103/uL) | 170 (123–216) | 385 (210–518) |
| Metabolic panels | ||
| Sodium (mmol/L) | 142 (137–148) | 148 (130–167) |
| BUN (mg/dL) | 67.5 (10–157) | 48 (11–80) |
| Creatinine (mg/dL) | 5.71 (0.75–12.36) | 1.33 (0.73–1.69) |
| Inflammatory markers | ||
| C-reactive protein (CRP) (mg/L) | 165 (22–370) | 232 (121–415) |
| Ferritin (ng/mL) | 3072 (103–5227) | 8530 (2477–40,000) |
| Procalcitonin (ng/mL) | 7.8 (0.28–23.33) | 2.12 (0.39–8) |
Fig. 1.
Patient #1 NCHCT. NCHCT obtained on HD #13, demonstrating multifocal intraparenchymal hemorrhage with intraventricular extension, mass effect, and evidence of global anoxic injury, cerebral edema, and downward herniation of the cerebellar tonsils
Patient #2
A 74-year-old man with history of tongue cancer, hypertension, and carotid stenosis status post right carotid stent presented to NYU Langone Medical Center with 3 days of extreme dyspnea in March 2020. In the emergency room, he was afebrile, tachycardic, normotensive, tachypneic, and saturating 70% on room air. Chest x-ray showed bilateral hazy airspace opacities, and nasopharyngeal COVID-19 swab was positive. He was emergently intubated, but prior to intubation, he was able to answer questions slowly and was oriented, was able to follow simple commands, and had intact motor function. He was started on ceftriaxone, azithromycin, and plaquenil, sedated with fentanyl and propofol, and admitted to the ICU. For several days, he required high doses of sedation to maintain vent synchrony. Despite this, he was intermittently hypertensive to systolic blood pressure 180–200 mm Hg. On HD #6, he was noted to have an uptrending D-dimer (2887 ng/L from 256 ng/L on admission) and was empirically started on a heparin infusion with the same anti-Xa goals described above in addition to his home dose of aspirin 81 mg daily. There were only two occasions when the anti-Xa level was mildly elevated to 0.52 and 0.62 IU/mL throughout his hospital course.
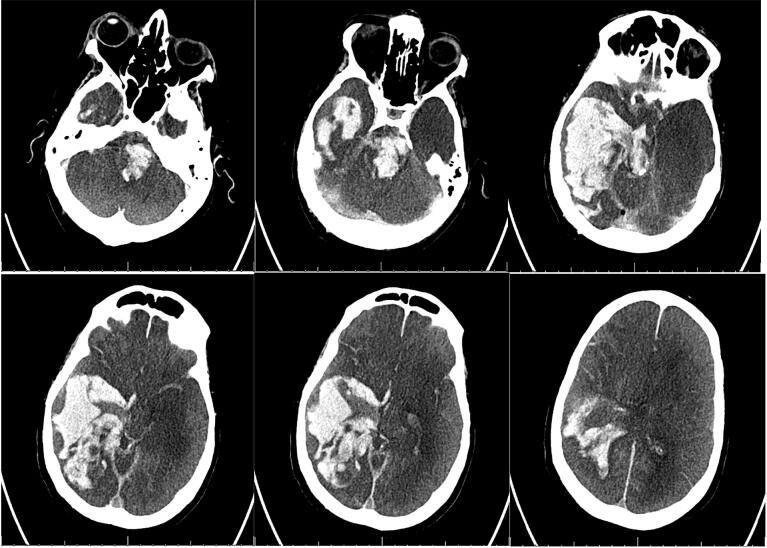
His ICU course was complicated by persistent septic shock necessitating escalating doses of vasopressors, acute kidney injury, and hyponatremia followed by hypernatremia (sodium range 130–167 mmol/L over his entire hospital course, but there was never an increase of more than 10 mmol/L within 24 hours). By HD #15, he was off sedation. At this time, he was noted to have fixed pupils. It was unclear when they were last reactive. Neurocritical care was consulted on HD #17 due to persistent coma; he was found to have no brainstem reflexes, but was taking occasional spontaneous breaths. Labs and vitals are noted in Table 1. NCHCT demonstrated a large intraparenchymal hemorrhage in the right temporal, occipital, and parietal lobes with intraventricular extension, as well as a large brainstem hemorrhage, with diffuse cerebral edema, evidence of global anoxic injury and impending herniation (Fig. 2). Given his grave prognosis, his family decided to withdraw life-sustaining treatment on HD #17, and the patient died.
Fig. 2.
Patient #2 NCHCT. NCHCT obtained on HD #17, demonstrating large intraparenchymal hemorrhage in the right temporal, occipital, and parietal lobes with intraventricular extension, and a large brainstem hemorrhage, with diffuse cerebral edema and evidence of global anoxic injury and impending herniation
Discussion
Although it has been noted that COVID-19 may increase risk for acute cerebrovascular events, including both ischemic and hemorrhagic stroke [2], to our knowledge, this is the first report of patients with COVID-19 who had catastrophic intracranial hemorrhages. Both patients were on anticoagulation at the time of their bleeds, but the fact that they had multifocal hemorrhages, severe diffuse cerebral edema, and evidence of anoxic brain injury suggests to us that their injuries were not simply the result of anticoagulation-induced hemorrhage or hemorrhagic transformation of multifocal ischemic strokes. This hypothesis is supported by speculation in the literature that ischemic brain injury can be worsened by the cytokine cascade triggered by COVID-19 [3, 4]. In considering the potential mechanisms for brain damage in these cases, it is also worth noting the possibly that there could have been direct intracranial invasion of COVID-19 secondary to a mechanism that has yet to be fully elucidated [5].
Autopsy could have helped explain the neuropathology in these cases. Unfortunately, it was not feasible to directly visualize and study the brain parenchyma of these patients; when they died, our hospital did not have the vacuum attachment for the saw used to open the calvarium that the Centers for Disease Control recommended to safely prevent aerosolization during the procedure. However, brain autopsies from other patients with COVID-19 have reportedly demonstrated hyperemic and edematous parenchyma [2].
It is important to acknowledge that both of our patients were on anticoagulation empirically. COVID-19 is known to cause coagulopathy, most likely as a result of the profound inflammatory response incited by the virus, which is associated with increased mortality [6]. Recent reports have highlighted the elevated risk of venous thromboemboli (VTE) in patients with COVID-19, even while on VTE prophylaxis, when compared to historic ICU VTE rates [6]. To address this, Thachil noted that use of anticoagulation in patients with elevated D-dimers may decrease mortality by both preventing thrombi and dampening the inflammatory response triggered by COVID-19 [7].
Thus, at our center, it was decided that therapeutic anticoagulation should be initiated empirically for patients with COVID-19 who have a D-dimer > 10,000 ng/mL and considered for patients who have a D-dimer between 2000 and < 10,000 ng/mL using treatment dose enoxaparin or heparin titrated to a low therapeutic anti-Xa goal of 0.3–0.5 U/mL.
However, the decision to use treatment dose anticoagulation in this patient population is controversial [6]. It is, of course, well-known that anticoagulation use is associated with risk of bleeding. Accordingly, Connors and Levy recommend that the decision to initiate treatment dose anticoagulation in patients with COVID-19 not be made based on D-dimer; rather, they suggest that VTE prophylaxis be administered to these patients just as it would be given to any other critically ill patient [6]. Further research is clearly needed to identify which, if any, patients with COVID-19 the risk/benefit assessment favors anticoagulation use.
Although patients with COVID-19 initially present with pulmonary pathology, this patient population is at risk for neurologic complications [1, 2]; as we have demonstrated with these two cases, these complications can be devastating. As such, we wish to emphasize the importance of performing neurologic assessments on patients with COVID-19. We recognize that there are a number of factors that may preclude performance of a thorough neurologic evaluation of these patients including need for sedation +/− paralytics to manage acute hypoxic respiratory failure, limited availability of personal protective equipment, and fear of exposure to the virus. However, at a minimum, the pupillary reflex should be monitored at least daily in patients who are intubated and sedated, especially if they are on systemic anticoagulation.
Further research is needed to better understand the reason our patients developed catastrophic bleeds and to determine if there are specific factors that predispose patients to this type of brain injury, or ways to prevent this type of brain injury.
Author contributions
The manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed.
Source of support
None.
Conflict of interest
No conflicts of interest.
Ethical approval/informed consent
This study has been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments. Given the nature of the case report, informed consent was not required.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Asadi-Pooya A, Simani L. Central nervous system manifestations of COVID-19: a systematic review. J Neurol Sci. 2020;413:1–4. doi: 10.1016/j.jns.2020.116832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mao L, Jin H, Wang M, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2019 doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wu Y, Xu X, Chen Z, et al. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav Immun. 2020 doi: 10.1016/j.bbi.2020.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Muhammad S, Haasbach E, Kotchourko M, et al. Influenza virus infection aggravates stroke outcome. Stroke. 2011;42(3):783–791. doi: 10.1161/STROKEAHA.110.596783. [DOI] [PubMed] [Google Scholar]
- 5.Li YC, Bai WZ, Hashikawa T. The neuroinvasive potential of SARS-CoV2 may be at least partially responsible for the respiratory failure of COVID-19 patients. J Med Virol. 2020 doi: 10.1002/jmv.25728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood. 2020 doi: 10.1182/blood.2020006000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Thachil J. The versatile heparin in COVID-19. J Thromb Haemost. 2020;18(5):1020–1022. doi: 10.1111/jth.14821. [DOI] [PMC free article] [PubMed] [Google Scholar]