Abstract
The current COVID-19 pandemic caused by SARS-CoV-2 has prompted investigators worldwide to search for an effective anti-viral treatment. A number of anti-viral drugs such as ribavirin, remdesivir, lopinavir/ritonavir, antibiotics such as azithromycin and doxycycline, and anti-parasite such as ivermectin have been recommended for COVID-19 treatment. In addition, sufficient pre-clinical rationale and evidence have been presented to use chloroquine for the treatment of COVID-19. Furthermore, Zn has the ability to enhance innate and adaptive immunity in the course of a viral infection. Besides, Zn supplement can favour COVID-19 treatment using those suggested and/or recommended drugs. Again, the effectiveness of Zn can be enhanced by using chloroquine as an ionophore while Zn inside the infected cell can stop SARS-CoV-2 replication. Given those benefits, this perspective paper describes how and why Zn could be given due consideration as a complement to the prescribed treatment of COVID-19.
Keywords: Anti-viral drugs, Chloroquine, SARS-CoV-2, RNA dependent RNA polymerase, Zinc transporter, Pneumocytes
Introduction
Coronaviruses (CoV), named after its crown-like appearance under electron microscope, were known to cause severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV) diseases with high mortality rate [1]. On March 11, 2020, the World Health Organization (WHO) declared Coronavirus Disease 2019 (COVID-19) outbreak as pandemic caused by another member of the Coronaviridae family—SARS-CoV-2.
SARS-CoV-2 primarily infects cells of the small air sacs known as alveoli consisting of alveolar cells and alveolar macrophages. There are two types of alveolar cells (type I and II) which are also known as pneumocytes. Type I cells provide 95% of the surface area of each alveoli and are flat hence are named squamous epithelial cells. Type II cells generally cluster in the corners of the alveoli and have a cuboidal shape. Infection by the SARS-CoV-2 causes an inflammatory condition also known as pneumonia affecting primarily alveoli [2]. Typically, symptoms include a combination of non-productive or dry cough, chest pain, fever, and difficulty in breathing. The pneumonic condition in COVID-19 is severe and is associated with its high mortality [3, 4].
The current COVID-19 pandemic caused by SARS-CoV-2 has prompted policy makers to take actions to prevent further spread of the virus. The same crisis also made investigators worldwide to search for an effective anti-viral treatment. The current review paper will highlight the potential of Zn supplement with the ongoing treatment modalities for COVID-19 patients. To start with, the paper will first briefly describe the virus and its mechanism of replication in the host cells (generally that is the pneumocytes of the lungs). A brief description will be added to highlight the changes in the host immune responses upon SARS-CoV-2 infection. Subsequently, a summary of the ongoing and recommended treatments will be presented. Finally, the paper will highlight how Zn can contribute to fight the battle with the SARS-CoV-2.
The SARS-COV-2 Virus and Its Replication in Host Pneumocytes (Cells of the Lungs)
SARS-CoV-2 is one of the seven types of coronavirus that are known to infect humans [4]. Based on the genetic properties, coronaviruses are grouped into four genera: α-CoV, β-CoV, γ-CoV, and δ-CoV [2], and the COVID-19 belongs to β-CoV [4]. Like other coronaviruses, SARS-CoV-2 is also an enveloped virus with a single-strand, positive-sense RNA genome [5].
At the beginning of the replicative cycle, the polycistronic viral genome uses a unique transcription mechanism to generate a nested set of subgenomic (sg) mRNAs after entry and uncoating inside the host cell. Using the 5′-proximal open reading frames (ORFs) of the genome, namely ORF1a and ORF1b, two large replicase polyproteins (pp1a and pp1ab) are translated. Eventually, 16 non-structural proteins (NSP) producing mature replicase proteins are released from pp1a and pp1ab attributed to the proteolytic cleavages by ORF1a-encoded proteases. The replicase proteins have a variety of functions that are required for viral RNA synthesis and capping, such as the RNA-dependent RNA polymerase (RdRp; NSP12), a helicase (NSP13), RNA cap-modifying methyltransferases (NSP14 and NSP16), and an exoribonuclease (NSP14). Using the host proteins, the coronavirus NSPs form membrane-associated replication and transcription complexes for viral membrane structures. For detail of the functions of each protein, please see the review by Cheng et al. [6].
Along with 16 non-structural proteins (NSPs) and four major structural proteins, namely spike (S), envelope (E), membrane (M), and nucleocapsid (N), SARS-CoV-2 contains eight accessory proteins [7]. The spike proteins have an S1 domain which is responsible for receptor binding and an S2 domain responsible for cell membrane fusion. In other words, S glycoproteins aid binding of the virus to the host cells. The receptor binding domain (RBD) of β-CoV is commonly located in the C-terminal domain of S1 [8]. The SARS-CoV-2 spike proteins were found to have 10- to 20-fold higher binding affinity to human angiotensin-converting enzyme 2 (ACE2) receptors than SARS-CoV does [9].
Using spike glycoproteins (S-glycoproteins), the SARS-CoV2 binds to human angiotensin-converting enzyme 2 (ACE2) receptors expressed on pneumocytes [10]. Notably, ACE2 receptors are widely expressed on the epithelial cells of alveoli, trachea, bronchi, bronchial serous glands [11], and alveolar monocytes and macrophages [12].
Binding to ACE2 receptors triggers conformational changes in the S-glycoprotein allowing cleavage by the transmembrane protease-serine 2 of the S-glycoprotein. The virus is then transported into the cytoplasm through a mechanism called endocytosis. The low pH inside the endosomes favours the host protease cathepsin-L to cleave the S-glycoprotein. This results in the fusion of the viral envelope and endosomal phospholipidic membrane to release the positive-strand viral genomic RNA (+RNA) into the cell cytoplasm.
Like other RNA viruses, SARS-CoV-2 genome–encoded RNA-dependent RNA polymerase (RdRp) is central to SARS-CoV-2 replicative cycle. Initially, a polyprotein precursor is formed from which the RdRp-containing subunit is proteolytically cleaved. Subsequently, the RdRp is integrated into a membrane associated viral enzyme complex that drives the synthesis of negative-strand RNA [13, 14]. The negative RNA strand is used as a template for the synthesis of viral mRNA (Fig. 1). SARS-CoV-2, MERS-CoV, and SARS-CoV have remarkably similar sequences and encode structurally similar RdRp [15]. The RdRp has a deep groove as an active site for the polymerization of RNA.
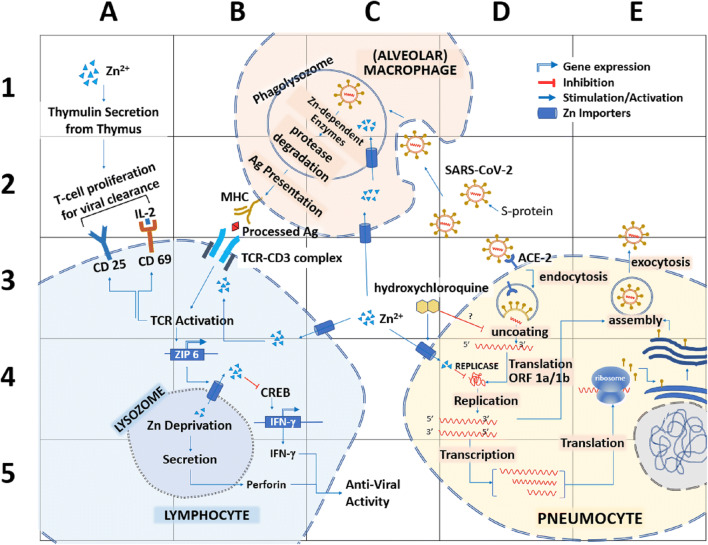
Fig. 1.
Potential sites of action of Zn to counter SARS-CoV-2 in pneumocytes. SARS-CoV uses spike (S) proteins to bind to angiotensin-converting enzyme 2 (ACE2) on pneumocytes (D3). Virus enters the host cell through endocytosis and releases therein the viral RNA (D3). The replicase enzyme complex is translated from the viral genome that mediates both replication and transcription (D4). Virions are shed from the infected cell through exocytosis (E3). The primary site of Zn2+ could be the inactivation of the viral replicase (D4). At the same time, the additional Zn supplement might initiate interferon-© (a common anti-viral agent) production by T lymphocytes (B5). However, Zn deprivation in the lysosome of the lymphocytes triggers to secrete perforin, which also exert anti-viral activity (B5). A pool of Zn importing inside the T lymphocytes activates T cell receptors as well as CD25 and CD69 to aid T cell proliferation and stabilization (A2–3). Added Zn also contributes to the production of thymulin from the thymus and triggers T lymphocyte production (A1–2). In alveolar macrophages, Zn can help to degrade the phagocytosed viral particle by the enzymes of the phagolysosome (B-C2). That in turn will help to present the processed An by the major histocompatibility complex (MHC) (B2)
Infected cells contain between 10 and 100 times more +RNA strands than −RNA strands. The polycistronic ribosome machinery of the infected cell synthesizes non-structural proteins of the SARS-CoV-2 and assembles these into the replicase-transcriptase complex to favour viral subgenomic mRNA synthesis. Following replication, the envelope proteins are translated and inserted into the endoplasmic reticulum of the host cells to finally enter into the Golgi compartment. Consequently, the viral genomic RNA is packaged into the nucleocapsid and then envelope proteins are incorporated during the budding step to form mature virions. The M protein plays an essential role during viral assembly by interacting with the other proteins of the virus. Following assembly, the newly formed viral particles are transported to the cell surface in vesicles and are released by exocytosis. A number of detailed reviews have summarized the replication and the assembly of the virus [13, 16].
Changes in Immune Response in COVID-19 Patients
Immunological profiles of COVID-19 patients seem to vary widely. In general, COVID-19 patients were shown to have normal or lower white blood cell counts, lymphopenia, or thrombocytopenia, with an increased C-reactive protein level [3, 8, 17–21]. In vitro studies of SARS-CoV infection of respiratory epithelial cells, dendritic cells (DCs), showed delayed release of cytokines and chemokines at the early stages. However, higher secretion anti-viral cytokines such as interferons (IFNs) and proinflammatory cytokines such as interleukins (IL) IL-1β, IL-6, and tumour necrosis factor (TNF)) and chemokines (C-C motif chemokine ligand CCL-2, CCL-3, and CCL-5) were recorded in the later stages [22–24].
In a retrospective, single-centre study, involving 99 COVID-19 patients treated in Wuhan Jinyintan Hospital, increased concentrations of C-reactive protein, IL-6, and serum ferritin were recorded along with an increased erythrocyte sedimentation rate [19]. The same group of patients also showed an increased number of neutrophils with decreased number of lymphocytes. Similar phenomena were observed with cytokine storms, with an overproduction of IL-7, IL-10, GCSF, IP10, MCP1, MIP1A, and TNF-α [3, 17].
Among the COVID-19 patients (n = 69)—admitted to Union Hospital in Wuhan between January 16 and January 29, 2020—only those who had SpO2 < 90% succumb to the infection [25]. Compared with the SpO2 ≥ 90% group, patients of the SpO2 < 90% group were older and showed more comorbidities and higher plasma levels of IL6, IL10, lactate dehydrogenase, and C-reactive protein [25].
Wang and colleagues [26] analysed 339 patients with COVID-19 (aged 71 ± 8 years). Among them, 80 (23.6%) were critical, 159 were severe (46.9%), and 100 were moderate (29.5%) cases admitted at the Renmin Hospital of Wuhan University. Compared with the normal values, the count of CD4+ and CD8+ cells was all significantly decreased in these patients. When compared between the survivor (n = 274) and non-survivor (n = 65) groups, the lymphocyte, monocyte, and platelet counts were significantly decreased in the non-survivor group, but the neutrophil counts were significantly higher. Furthermore, a high level of lymphocytes was found as a predictive better outcome (OR = 0.10, P < 0.001) for the patients who recovered [26].
Zn Regulation in Human Cells
Zinc is widely distributed in human tissues, where virtually all Zn is present in intracellular compartments such as the nucleus (30–40%), cytosol, and other organelles and specialized vesicles (50%), and the rest is bound with cell membrane proteins [27]. While cells need a constant supply of Zn, free Zn ions (Zn2+) can be toxic to the cells by inhibiting cytoplasmic enzymes such as adenylate cyclase [28].
In humans, plasma Zn level is maintained between 10 and 18 mol/L representing 0.1% of total body Zn [29]. The total zinc content that favours a typical fibroblast-like cell to grow in ordinary culture media is ~ 0.25 fmol per cell or ~ 200 μM. However, in vitro growth of the cells stops at cellular zinc levels below ~ 0.2 fmol per cell [30].
The intracellular homeostasis of Zn as well as exchange of Zn in and out of the cells is controlled by a large number of proteins belonging to two Zn transporter protein families, SLC39A (Zn importer protein, i.e. ZIP and ZRT/IRT-related protein, 14 ZIP) and SLC30A (Zn Transporter, i.e. ZnT, 10 ZnTs) [31]. ZnTs generally transport Zn2+ out of the cytosol, whereas ZIPs import them from cellular compartments or the extracellular space into the cytosol [32, 33]. Most ZnTs are present in intracellular compartments, such as endosomes, Golgi, or endoplasmic reticulum while only ZnT1 appears to be located at the plasma membrane as it is the primary regulator of cellular Zn efflux [30]. Most ZIPs are observed at the plasma membrane; however, Zip7 is located at the Golgi apparatus [34].
Zinc in Host Immune Mechanisms
Unlike other “first row” transition metals/elements, Zn does not participate in redox reactions but rather functions as a Lewis acid to accept a pair of electrons. This property makes Zn2+ a stable ion in a biological medium and an ideal metal cofactor for reactions that require a redox-stable ion such as proteolysis and the hydration of carbon dioxide. Metallothioneins—a cysteine-rich low molecular weight group of proteins—act as reservoir of the intracellular concentration of free Zn2+ [35–37]. Hence, Zn2+ can serve as intracellular second messenger and may trigger apoptosis or a decrease in protein synthesis at elevated concentrations [38–40].
A number of immunome activation pathways are activated by Zn such as NF-κB signalling pathway. NF-κB influences the expression of pro-inflammatory cytokines, namely IL-1b, IL-6, IL-8, TNF-α, and MCP-1, chemokines, acute phase proteins (CRP and fibrinogen), matrix metalloproteinases, adhesion molecules, growth factors, and other factors involved in inflammatory response, such as COX-2 and iNOS [41, 42].
Zinc administration in mixed lymphocyte cultures was shown to induce and stabilize CD4+CD25+Foxp3+ and CD4+CD25+CTLA-4+ T cells. These effects were attributed to zinc-induced upregulation of Foxp3 and KLF-10 and downregulation of IRF-1 whereas in resting lymphocytes zinc increases IRF-1 [43]. It is important to note that the number of CD4+ and CD8+ T cells is critical in anti-viral immunity [44, 45].
On the other hand, Zn depletion can cause a significant suppression of autophagy in cells (human hepatoma cells VL-17A). Conversely, in vitro Zn addition stimulated autophagy in the same cells. Thus, a critical role of Zn was suggested in autophagy under basal conditions [46]. In various pathological conditions including viral infection, autophagy plays an important protective role as host defence mechanism [47–49]. In autophagy, the intracellular components such as protein aggregates and damaged organelles are engulfed into a double-membrane structure called autophagosome and fuse with lysosome to form autolysosome to degrade the engulfed components using lysosomal enzymes [50, 51]. Lysosome contains more than 50 enzymes, including proteases, peptidases, phosphatases, nucleases, glycosidases, sulfatases, and lipases [52]. Essentially, the structural and functional integrity of many of these enzymes depends on Zn [53].
Immune Regulation of Zinc in Viral Infection
In vitro studies involving added Zn2+ in the presence of its cellular import stimulatory compounds, such as hinokitiol (HK), pyrrolidine dithiocarbamate (PDTC), and pyrithione (PT), were shown to inhibit the replication of various RNA viruses, including influenza virus [54], respiratory syncytial virus [55], and several picornaviruses [56–58]. These reports suggested inhibition of intracellular Zn2+ in the replicative cycle of these viruses. More particularly, Zn2+ was shown to inhibit polyprotein processing in cells infected with human rhinovirus and coxsackievirus B3 [58].
In addition to SARS-CoV, a number of other viruses, including HIV, HSV, and vaccinia virus, are known to be inhibited by Zn salts. Zn is known to inhibit the viral entry, blocking of polyprotein processing, or inhibition of viral RdRp activity [59–61]. Using Huh7 cells transfected with in vitro synthesized capped genomic RNA of a g-1 HEV, Kaushik et al. [62] showed that Zn but not Mg salts, namely Zn-sulfate and Zn-acetate, can inhibit viral sense and antisense RNA levels by approximately 50% at a working concentration of 10 μM. Thus, Zn salts were shown to directly inhibit the activity of viral RdRp thus inhibiting viral replication.
Recommended and Ongoing Treatments for COVID-19
The suggested treatments for COVID-19 are, but not limited to, the use of (i) convalescent plasma for COVID-19 treatment [63–65]; (ii) ribavirin, a nucleoside analogue in combination with recombinant interferon showed inhibition of MERS-CoV replication [66]; (iii) lopinavir/ritonavir—a combination of a protease inhibitor and a booster used for the treatment of human immunodeficiency virus infection [67]; (iv) remdesivir, a nucleotide analogue that inhibit RNA polymerase with a broad spectrum of anti-viral activities; in inhibition of human and zoonotic coronavirus [15, 68, 69]; (v) favipiravir (also known as T-705, Avigan or favilavir) is a pyrazinecarboxamide derivative known to inhibit RNA polymerase [70]. In addition, azithromycin and doxycycline—commonly used antibiotics to inhibit viral replication and IL-6 production [71] and drugs that suppress IL-1 or IL-1R [72]—were also suggested for the treatment and prevention of COVID-19 disease.
As of April 21, 2020, more than 500 clinical trials have been registered at the various international and national clinical trial registry sites [73]. The efficacy of a number of recommended anti-viral drugs has been investigated with various outcomes. Such randomized clinical trials on lopinavir-ritonavir did not show any benefit beyond standard care [74]; favipiravir, compared with Arbidol, did not significantly improve the clinical recovery rate at day 7 [75]. Besides, hydroxychloroquine versus hydroxychloroquine combined with azithromycin was investigated following non-randomized trials [76]. Other ongoing clinical trials include IL-6 inhibitors (tocilizumab and sarilumab), convalescent plasma therapy, stem-cell transfusion, candidate vaccines, and traditional Chinese medicines.
In a systematic review, Cortegiani et al. [77] argued that “there is sufficient pre-clinical rationale and evidence regarding the effectiveness of chloroquine for treatment of COVID-19 as well as evidence of safety from long-time use in clinical practice for other indications.” Chloroquine was shown to inhibit in vitro replication of SARS-CoV-2 in Vero E6 cells in an effective concentration EC90 of 6.90 μM that can be easily achieved with standard dosing, due to its favourable penetration into tissues, including in the lung [68]. This is substantially lower than the concentration detected in human plasma when the drug is prescribed to treat malaria at a dose of 25 mg/kg over 3 days [78]. However, for COVID-19 patients, a lower dose such as 3.6 mg/kg that are often prescribed to treat rheumatoid arthritis has been suggested for long-term prophylaxis as the dose is similar to IC50 for SARS-CoV [78, 79]. It is to be noted that hydroxychloroquine showed greater efficacy than chloroquine, at least based on in vitro studies [80]. Besides, hydroxychloroquine was shown to have minimal risk of toxicity such as retinopathy [81]. Other recent studies also showed that hydroxychloroquine is a less toxic metabolite of chloroquine, as it is more soluble, and causes less side effects and therefore is safer [76, 80, 82, 83].
It has been hypothesized that both hydroxychloroquine and chloroquine can interfere with ACE2 receptor glycosylation and prevents SARS-CoV-2 binding to pneumocytes. Chloroquine could also possibly inhibit sialic acid biosynthesis thus limiting cell surface binding of SARS-CoV-2. In case the viral particle is endocytosed, chloroquine has been hypothesized to modulate the acidification of endosomes thereby inhibiting formation of the autophagosome. Through reduction of cellular mitogen-activated protein (MAP) kinase activation, chloroquine may also inhibit virus replication. Moreover, chloroquine could alter M protein maturation and interfere with virion assembly and budding [16].
It is to be noted that both chloroquine and hydroxychloroquine are weak bases present in protonated form in the extracellular environment hence are incapable of crossing the plasma membrane. In the non-protonated form, chloroquine and hydroxychloroquine may enter the intracellular compartments and gradually become protonated according to the Henderson-Hasselbach law. This conversion allows both chloroquine and hydroxychloroquine to reside in acidic organelles such as the endosome, Golgi vesicles, and the lysosomes. [84].
Why Is Zn Critical for COVID-19 Treatment?
Zn Can Enhance Cell-Mediated and Adaptive Immunity in the Course of Infection
Zinc is thoroughly involved in cell-mediated immunity against any infectious agent such as bacteria and virus. Zinc is one of the major factors that control function and proliferation of neutrophils, NK cells, macrophages, and T and B lymphocytes as well as cytokine production by the immune cells. Zn also mediates protection from the adverse effect of ROS that are generally produced during inflammatory processes. Free intracellular Zn2+ is essential in extravasation to the site of the infection and uptake and killing of microorganisms by neutrophils [85]. For more detail on the functions of Zn in immunity, please see the review articles by [86–89].
Augmented Effect of Zn and Chloroquine to Stop SARS-CoV-2 Replication
Chloroquine was known to increase vacuolar pH when trapped in acidic organelles, such as lysosomes. This increase in pH disrupts lysosomal acidification leading to the impairment of autophagosome fusion and autophagic degradation [90, 91]. Using human ovarian carcinoma cell line A2780, Xue J. et al. [92] reported that chloroquine can act as ionophore for Zn. Chloroquine enhances uptake of Zn by the lysosomes, and the combination of Zn and chloroquine enhances chloroquine cytotoxicity and induces apoptosis in malignant cells. In their experiment [92], A2780 ovarian cancer cells were treated with 100–300 μM chloroquine in the presence of increased concentrations of ZnCl2 for 1 h. Intracellular basal Zn levels were barely detectable in control cells without any chloroquine. However, chloroquine addition to the culture medium caused significant increases of intracellular Zn in a dose-dependent manner.
Zn Can Directly Inhibit SARS-CoV-2 Replication
Using recombinant SARS-CoV nsp12, te Velthuis et al. [61] showed that Zn2+ directly inhibited the in vitro RdRp activity (Fig. 1). They also reported that specifically, Zn2+ was found to inhibit the SARS-CoV RdRp elongation and template binding. Earlier, it was also shown that Zn2+ inhibited the proteolytic processing of replicase polyproteins [93, 94].
Zn Enhances Efficiency of Anti-viral Drugs
A number of anti-viral drugs such as ribavirin, remdesivir, lopinavir/ritonavir, and antibiotics such as azithromycin and doxycycline have been recommended for the treatment of COVID-19. Zn supplement can favour COVID-19 treatment using some of these anti-viral drugs.
Zinc supplementation was suggested as a complementary therapy in chronic hepatitis C patients to increase the tolerance to IFN-α-2a and ribavirin [95]. However, a 24-week Zn supplementation reduced the incidence of abdominal discomfort without any additive effect on the anti-hepatitis C virus dual therapy of IFN-α-2 and ribavirin [96].
While evaluating the impact of ZnSO4 supplementation in HIV-infected individuals, the supplement was found useful in management of atazanavir-ritonavir-related unconjugated hyperbilirubinemia in selected patients [67].
Zinc Supplement: Limits and Risks
Zinc as an adjuvant therapy can be prescribed in different forms of Zn salt, such as Zn-gluconate, Zn-acetate, Zn-sulfate, and Zn-picolinate. However, the amount of elemental Zn in each salt varies. For example, Zn-sulfate contains about 23% elemental Zn; therefore, to have 50 mg of Zn, a 220 mg of Zn-sulfate tablet would be required for consumption. It is important to note that the recommended daily allowance of Zn will vary according to the age, sex, and health conditions of an individual. For healthy adults, the recommended daily allowance is typically 15–30 mg of elemental Zn. Despite the beneficial effects of Zn in immune response, long-term high-dose Zn consumption will cause a decrease of high-density lipoprotein cholesterol levels, anaemia, copper deficiency, and possible genitourinary complications [97].
Conclusion
Zinc plays crucial roles in many aspects of life. In the course of infection, the immunomodulatory role of Zn is well evident. In the current pandemic of SARS-CoV-2, Zn supplement could play an important role to treat COVID-19 patients such as (i) added immune boosting effects with anti-viral drugs and (ii) stopping SARS-CoV-2 replication in infected cells, if combined with chloroquine. In view of this discussion, oral Zn supplement can be given using a suitable form of Zn-salt.
Authors’ Contributions
SZI and MTR conceptualize the idea. MTR prepared the initial draft. SZI and MTR revised the manuscript.
Data Availability
I have not used any data that are required to be available for the readers.
Compliance with Ethical Standards
Conflict of Interest
The authors declare that they have no competing interests.
Ethics Approval
Not applicable.
Consent to Participate (Include Appropriate Statements)
Not applicable.
Code Availability
Not applicable.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Mohammad Tariqur Rahman, Email: m.tariqur.rahman@gmail.com, Email: tarique@um.edu.my.
Syed Zahir Idid, Email: syedzahir@iium.edu.my, Email: szidid@gmail.com.
References
- 1.Hui DS, Memish ZA, Zumla A. Severe acute respiratory syndrome vs. the Middle East respiratory syndrome. Curr Opin Pulm Med. 2014;20:233–241. doi: 10.1097/MCP.0000000000000046. [DOI] [PubMed] [Google Scholar]
- 2.Su S, Wong G, Shi W, Liu J, Lai ACK, Zhou J, Liu W, Bi Y, Gao GF. Epidemiology, genetic recombination, and pathogenesis of coronaviruses. Trends Microbiol. 2016;24:490–502. doi: 10.1016/j.tim.2016.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W, China Novel Coronavirus Investigating and Research Team A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Forni D, Cagliani R, Clerici M, Sironi M. Molecular evolution of human coronavirus genomes. Trends Microbiol. 2017;25:35–48. doi: 10.1016/j.tim.2016.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cheng VCC, Lau SKP, Woo PCY, Yuen KY. Severe acute respiratory syndrome coronavirus as an agent of emerging and reemerging infection. Clin Microbiol Rev. 2007;20:660–694. doi: 10.1128/CMR.00023-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wu A, Peng Y, Huang B, Ding X, Wang X, Niu P, Meng J, Zhu Z, Zhang Z, Wang J, Sheng J, Quan L, Xia Z, Tan W, Cheng G, Jiang T. Genome composition and divergence of the novel coronavirus (2019-nCoV) originating in China. Cell Host Microbe. 2020;27:325–328. doi: 10.1016/j.chom.2020.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lu R, Zhao X, Li J, et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395:565–574. doi: 10.1016/S0140-6736(20)30251-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wrapp D, Wang N, Corbett KS, Goldsmith JA, Hsieh CL, Abiona O, Graham BS, McLellan JS. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020;367:1260–1263. doi: 10.1126/science.abb2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ou X, Liu Y, Lei X, Li P, Mi D, Ren L, Guo L, Guo R, Chen T, Hu J, Xiang Z, Mu Z, Chen X, Chen J, Hu K, Jin Q, Wang J, Qian Z. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat Commun. 2020;11:1620. doi: 10.1038/s41467-020-15562-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liu L, Wei Q, Alvarez X, Wang H, du Y, Zhu H, Jiang H, Zhou J, Lam P, Zhang L, Lackner A, Qin C, Chen Z. Epithelial cells lining salivary gland ducts are early target cells of severe acute respiratory syndrome coronavirus infection in the upper respiratory tracts of rhesus macaques. J Virol. 2011;85:4025–4030. doi: 10.1128/JVI.02292-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, Huan Y, Yang P, Zhang Y, Deng W, Bao L, Zhang B, Liu G, Wang Z, Chappell M, Liu Y, Zheng D, Leibbrandt A, Wada T, Slutsky AS, Liu D, Qin C, Jiang C, Penninger JM. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med. 2005;11:875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23. doi: 10.1007/978-1-4939-2438-7_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Perlman S, Netland J. Coronaviruses post-SARS: update on replication and pathogenesis. Nat Rev Microbiol. 2009;7:439–450. doi: 10.1038/nrmicro2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Morse JS, Lalonde T, Xu S, Liu WR. Learning from the past: possible urgent prevention and treatment options for severe acute respiratory infections caused by 2019-nCoV. Chembiochem. 2020;21:730–738. doi: 10.1002/cbic.202000047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Devaux CA, Rolain J-M, Colson P, Raoult D. New insights on the antiviral effects of chloroquine against coronavirus: what to expect for COVID-19? Int J Antimicrob Agents. 2020;105938:105938. doi: 10.1016/j.ijantimicag.2020.105938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lescure F-X, Bouadma L, Nguyen D, Parisey M, Wicky PH, Behillil S, Gaymard A, Bouscambert-Duchamp M, Donati F, le Hingrat Q, Enouf V, Houhou-Fidouh N, Valette M, Mailles A, Lucet JC, Mentre F, Duval X, Descamps D, Malvy D, Timsit JF, Lina B, van-der-Werf S, Yazdanpanah Y (2020) Clinical and virological data of the first cases of COVID-19 in Europe: a case series. Lancet Infect Dis. 10.1016/S1473-3099(20)30200-0 [DOI] [PMC free article] [PubMed]
- 18.Zhou M, Zhang X, Qu J (2020) Coronavirus disease 2019 (COVID-19): a clinical update. Front Med. 10.1007/s11684-020-0767-8 [DOI] [PMC free article] [PubMed]
- 19.Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ma HY, Li H, Wang JC, Xu FS (2006) Expression and significance of metallothionein in the placenta of women with low level lead exposure during pregnancy. Zhonghua Fu Chan Ke Za Zhi 41(10):676–679 [PubMed]
- 21.Ye Q, Wang B, Mao J (2020) The pathogenesis and treatment of the ‘cytokine storm’ in COVID-19. J Inf Secur. 10.1016/j.jinf.2020.03.037 [DOI] [PMC free article] [PubMed]
- 22.Law HKW, Cheung CY, Ng HY, Sia SF, Chan YO, Luk W, Nicholls JM, Peiris JSM, Lau YL. Chemokine up-regulation in SARS-coronavirus-infected, monocyte-derived human dendritic cells. Blood. 2005;106:2366–2374. doi: 10.1182/blood-2004-10-4166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cheung CY, Poon LLM, Ng IHY, Luk W, Sia SF, Wu MHS, Chan KH, Yuen KY, Gordon S, Guan Y, Peiris JSM. Cytokine responses in severe acute respiratory syndrome coronavirus-infected macrophages in vitro: possible relevance to pathogenesis. J Virol. 2005;79:7819–7826. doi: 10.1128/JVI.79.12.7819-7826.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pedersen SF, Ho Y-C. SARS-CoV-2: a storm is raging. J Clin Invest. 2020;130:2202–2205. doi: 10.1172/JCI137647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wang Z, Yang B, Li Q, Wen L, Zhang R (2020) Clinical features of 69 cases with coronavirus disease 2019 in Wuhan. China Clin Infect Dis an Off Publ Infect Dis Soc Am. 10.1093/cid/ciaa272 [DOI] [PMC free article] [PubMed]
- 26.Wang L, He W, Yu X, Hu D, Bao M, Liu H, Zhou J, Jiang H (2020) Coronavirus disease 2019 in elderly patients: characteristics and prognostic factors based on 4-week follow-up. J Inf Secur. 10.1016/j.jinf.2020.03.019 [DOI] [PMC free article] [PubMed]
- 27.Vallee BL, Falchuk KH. The biochemical basis of zinc physiology. Physiol Rev. 1993;73:79–118. doi: 10.1152/physrev.1993.73.1.79. [DOI] [PubMed] [Google Scholar]
- 28.Klein C, Heyduk T, Sunahara RK. Zinc inhibition of adenylyl cyclase correlates with conformational changes in the enzyme. Cell Signal. 2004;16:1177–1185. doi: 10.1016/j.cellsig.2004.03.008. [DOI] [PubMed] [Google Scholar]
- 29.Foster M, Samman S. Zinc and regulation of inflammatory cytokines: implications for cardiometabolic disease. Nutrients. 2012;4:676–694. doi: 10.3390/nu4070676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Palmiter RD, Findley SD. Cloning and functional characterization of a mammalian zinc transporter that confers resistance to zinc. EMBO J. 1995;14:639–649. doi: 10.1002/j.1460-2075.1995.tb07042.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lichten LA, Cousins RJ. Mammalian zinc transporters: nutritional and physiologic regulation. Annu Rev Nutr. 2009;29:153–176. doi: 10.1146/annurev-nutr-033009-083312. [DOI] [PubMed] [Google Scholar]
- 32.Cousins RJ, Liuzzi JP, Lichten LA. Mammalian zinc transport, trafficking, and signals. J Biol Chem. 2006;281:24085–24089. doi: 10.1074/jbc.R600011200. [DOI] [PubMed] [Google Scholar]
- 33.Aydemir TB, Blanchard RK, Cousins RJ. Zinc supplementation of young men alters metallothionein, zinc transporter, and cytokine gene expression in leukocyte populations. Proc Natl Acad Sci U S A. 2006;103:1699–1704. doi: 10.1073/pnas.0510407103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Huang L, Kirschke CP, Zhang Y, Yu YY. The ZIP7 gene (Slc39a7) encodes a zinc transporter involved in zinc homeostasis of the Golgi apparatus. J Biol Chem. 2005;280:15456–15463. doi: 10.1074/jbc.M412188200. [DOI] [PubMed] [Google Scholar]
- 35.Pauwels M, van Weyenbergh J, Soumillion A, et al. Induction by zinc of specific metallothionein isoforms in human monocytes. Eur J Biochem. 1994;220:105–110. doi: 10.1111/j.1432-1033.1994.tb18603.x. [DOI] [PubMed] [Google Scholar]
- 36.Krȩzel A, Maret W. Dual nanomolar and picomolar Zn (II) binding properties of metallothionein. J Am Chem Soc. 2007;129:10911–10921. doi: 10.1021/ja071979s. [DOI] [PubMed] [Google Scholar]
- 37.Rahman MT, Haque N, Abu Kasim NH, De Ley M. Origin, function, and fate of metallothionein in human blood. Rev Physiol Biochem Pharmacol. 2017;173:41–62. doi: 10.1007/112_2017_1. [DOI] [PubMed] [Google Scholar]
- 38.Alirezaei M, Nairn AC, Glowinski J, Prémont J, Marin P. Zinc inhibits protein synthesis in neurons. Potential role of phosphorylation of translation initiation factor-2alpha. J Biol Chem. 1999;274:32433–32438. doi: 10.1074/jbc.274.45.32433. [DOI] [PubMed] [Google Scholar]
- 39.Frederickson CJ, Koh J-Y, Bush AI. The neurobiology of zinc in health and disease. Nat Rev Neurosci. 2005;6:449–462. doi: 10.1038/nrn1671. [DOI] [PubMed] [Google Scholar]
- 40.Lazarczyk M, Favre M. Role of Zn2+ ions in host-virus interactions. J Virol. 2008;82:11486–11494. doi: 10.1128/JVI.01314-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lawrence T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol. 2009;1:a001651. doi: 10.1101/cshperspect.a001651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hayden MS, Ghosh S. Regulation of NF-kappaB by TNF family cytokines. Semin Immunol. 2014;26:253–266. doi: 10.1016/j.smim.2014.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Maywald M, Rink L. Zinc supplementation induces CD4(+)CD25(+)Foxp3(+) antigen-specific regulatory T cells and suppresses IFN-gamma production by upregulation of Foxp3 and KLF-10 and downregulation of IRF-1. Eur J Nutr. 2017;56:1859–1869. doi: 10.1007/s00394-016-1228-7. [DOI] [PubMed] [Google Scholar]
- 44.Whitmire JK, Ahmed R. Costimulation in antiviral immunity: differential requirements for CD4(+) and CD8(+) T cell responses. Curr Opin Immunol. 2000;12:448–455. doi: 10.1016/s0952-7915(00)00119-9. [DOI] [PubMed] [Google Scholar]
- 45.Jansen JM, Gerlach T, Elbahesh H, Rimmelzwaan GF, Saletti G. Influenza virus-specific CD4+ and CD8+ T cell-mediated immunity induced by infection and vaccination. J Clin Virol. 2019;119:44–52. doi: 10.1016/j.jcv.2019.08.009. [DOI] [PubMed] [Google Scholar]
- 46.Liuzzi JP, Yoo C. Role of zinc in the regulation of autophagy during ethanol exposure in human hepatoma cells. Biol Trace Elem Res. 2013;156:350–356. doi: 10.1007/s12011-013-9816-3. [DOI] [PubMed] [Google Scholar]
- 47.Jiang P, Mizushima N. Autophagy and human diseases. Cell Res. 2014;24:69–79. doi: 10.1038/cr.2013.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Choi AMK, Ryter SW, Levine B. Autophagy in human health and disease. N Engl J Med. 2013;368:651–662. doi: 10.1056/NEJMra1205406. [DOI] [PubMed] [Google Scholar]
- 49.Meijer AJ, Codogno P. Autophagy: regulation and role in disease. Crit Rev Clin Lab Sci. 2009;46:210–240. doi: 10.1080/10408360903044068. [DOI] [PubMed] [Google Scholar]
- 50.Mizushima N. A brief history of autophagy from cell biology to physiology and disease. Nat Cell Biol. 2018;20:521–527. doi: 10.1038/s41556-018-0092-5. [DOI] [PubMed] [Google Scholar]
- 51.Levine B, Kroemer G. Biological functions of autophagy genes: a disease perspective. Cell. 2019;176:11–42. doi: 10.1016/j.cell.2018.09.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lubke T, Lobel P, Sleat DE. Proteomics of the lysosome. Biochim Biophys Acta. 2009;1793:625–635. doi: 10.1016/j.bbamcr.2008.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Maret W. Zinc biochemistry: from a single zinc enzyme to a key element of life. Adv Nutr. 2013;4:82–91. doi: 10.3945/an.112.003038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Uchide N, Ohyama K, Bessho T, et al. Effect of antioxidants on apoptosis induced by influenza virus infection: inhibition of viral gene replication and transcription with pyrrolidine dithiocarbamate. Antivir Res. 2002;56:207–217. doi: 10.1016/s0166-3542(02)00109-2. [DOI] [PubMed] [Google Scholar]
- 55.Suara RO, Crowe JEJ. Effect of zinc salts on respiratory syncytial virus replication. Antimicrob Agents Chemother. 2004;48:783–790. doi: 10.1128/aac.48.3.783-790.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Korant BD, Kauer JC, Butterworth BE. Zinc ions inhibit replication of rhinoviruses. Nature. 1974;248:588–590. doi: 10.1038/248588a0. [DOI] [PubMed] [Google Scholar]
- 57.Lanke K, Krenn BM, Melchers WJG, Seipelt J, van Kuppeveld FJM. PDTC inhibits picornavirus polyprotein processing and RNA replication by transporting zinc ions into cells. J Gen Virol. 2007;88:1206–1217. doi: 10.1099/vir.0.82634-0. [DOI] [PubMed] [Google Scholar]
- 58.Krenn BM, Gaudernak E, Holzer B, Lanke K, van Kuppeveld FJM, Seipelt J. Antiviral activity of the zinc ionophores pyrithione and hinokitiol against picornavirus infections. J Virol. 2009;83:58–64. doi: 10.1128/JVI.01543-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Katz E, Margalith E. Inhibition of vaccinia virus maturation by zinc chloride. Antimicrob Agents Chemother. 1981;19:213–217. doi: 10.1128/aac.19.2.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Haraguchi Y, Sakurai H, Hussain S, Anner BM, Hoshino H. Inhibition of HIV-1 infection by zinc group metal compounds. Antivir Res. 1999;43:123–133. doi: 10.1016/s0166-3542(99)00040-6. [DOI] [PubMed] [Google Scholar]
- 61.te Velthuis AJW, van den Worm SHE, Sims AC, Baric RS, Snijder EJ, van Hemert MJ. Zn(2+) inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture. PLoS Pathog. 2010;6:e1001176. doi: 10.1371/journal.ppat.1001176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kaushik N, Subramani C, Anang S, Muthumohan R, Shalimar, Nayak B, Ranjith-Kumar CT, Surjit M (2017) Zinc salts block hepatitis E virus replication by inhibiting the activity of viral RNA-dependent RNA polymerase. J Virol 91. 10.1128/JVI.00754-17 [DOI] [PMC free article] [PubMed]
- 63.Chen L, Xiong J, Bao L, Shi Y. Convalescent plasma as a potential therapy for COVID-19. Lancet Infect Dis. 2020;20:398–400. doi: 10.1016/S1473-3099(20)30141-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Casadevall A, Pirofski L-A. The convalescent sera option for containing COVID-19. J Clin Invest. 2020;130:1545–1548. doi: 10.1172/JCI138003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wong HK, Lee CK (2020) Pivotal role of convalescent plasma in managing emerging infectious diseases. Vox Sang. 10.1111/vox.12927 [DOI] [PMC free article] [PubMed]
- 66.Falzarano D, de Wit E, Martellaro C, Callison J, Munster VJ, Feldmann H. Inhibition of novel beta coronavirus replication by a combination of interferon-alpha2b and ribavirin. Sci Rep. 2013;3:1686. doi: 10.1038/srep01686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Moyle G, Else L, Jackson A, Back D, Yapa MH, Seymour N, Ringner-Nackter L, Karolia Z, Gazzard B, Boffito M. Coadministration of atazanavir-ritonavir and zinc sulfate: impact on hyperbilirubinemia and pharmacokinetics. Antimicrob Agents Chemother. 2013;57:3640–3644. doi: 10.1128/AAC.00357-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, Shi Z, Hu Z, Zhong W, Xiao G. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30:269–271. doi: 10.1038/s41422-020-0282-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Gordon CJ, Tchesnokov EP, Feng JY, Porter DP, Götte M. The antiviral compound remdesivir potently inhibits RNA-dependent RNA polymerase from Middle East respiratory syndrome coronavirus. J Biol Chem. 2020;295:4773–4779. doi: 10.1074/jbc.AC120.013056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Furuta Y, Takahashi K, Shiraki K, Sakamoto K, Smee DF, Barnard DL, Gowen BB, Julander JG, Morrey JD. T-705 (favipiravir) and related compounds: novel broad-spectrum inhibitors of RNA viral infections. Antivir Res. 2009;82:95–102. doi: 10.1016/j.antiviral.2009.02.198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Sargiacomo C, Sotgia F, Lisanti MP (2020) COVID-19 and chronological aging: senolytics and other anti-aging drugs for the treatment or prevention of corona virus infection? Aging (Albany NY). 10.18632/aging.103001 [DOI] [PMC free article] [PubMed]
- 72.Conti P, Gallenga CE, Tete G et al (2020) How to reduce the likelihood of coronavirus-19 (CoV-19 or SARS-CoV-2) infection and lung inflammation mediated by IL-1. J Biol Regul Homeost Agents 34 [DOI] [PubMed]
- 73.Thorlund K, Dron L, Park J, et al. Correspondence a real-time dashboard. Lancet. 2020;7500:2019–2020. doi: 10.1016/S2589-7500(20)30086-8. [DOI] [Google Scholar]
- 74.Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, Ruan L, Song B, Cai Y, Wei M, Li X, Xia J, Chen N, Xiang J, Yu T, Bai T, Xie X, Zhang L, Li C, Yuan Y, Chen H, Li H, Huang H, Tu S, Gong F, Liu Y, Wei Y, Dong C, Zhou F, Gu X, Xu J, Liu Z, Zhang Y, Li H, Shang L, Wang K, Li K, Zhou X, Dong X, Qu Z, Lu S, Hu X, Ruan S, Luo S, Wu J, Peng L, Cheng F, Pan L, Zou J, Jia C, Wang J, Liu X, Wang S, Wu X, Ge Q, He J, Zhan H, Qiu F, Guo L, Huang C, Jaki T, Hayden FG, Horby PW, Zhang D, Wang C. A trial of lopinavir–ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020;382:1787–1799. doi: 10.1056/NEJMoa2001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Chen C, Zhang Y, Jianying H et al (2020) Favipiravir versus Arbidol for COVID-19: a randomized clinical trial. medRxiv. 10.1101/2020.03.17.20037432
- 76.Gautret P, Lagier J-C, Parola P et al (2020) Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents:105949. 10.1016/j.ijantimicag.2020.105949 [DOI] [PMC free article] [PubMed] [Retracted]
- 77.Cortegiani A, Ingoglia G, Ippolito M, Giarratano A, Einav S (2020) A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care. 10.1016/j.jcrc.2020.03.005 [DOI] [PMC free article] [PubMed]
- 78.Savarino A, Boelaert JR, Cassone A, et al. Personal view antiviral effects of chloroquine effects of chloroquine on viral infections : an old drug against today ’ s diseases ? Personal view. 2003;3:722–727. doi: 10.1016/S1473-3099(03)00806-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Principi N, Esposito S. Correspondence Chloroquine or hydroxychloroquine for prophylaxis of COVID-19. Lancet Infect Dis. 2020;3099:30296. doi: 10.1016/S1473-3099(20)30296-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Yao X, Ye F, Zhang M, Cui C, Huang B, Niu P, Liu X, Zhao L, Dong E, Song C, Zhan S, Lu R, Li H, Tan W, Liu D (2020) In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin Infect Dis an Off Publ Infect Dis Soc Am. 10.1093/cid/ciaa237 [DOI] [PMC free article] [PubMed]
- 81.Finbloom DS, Silver K, Newsome DA, Gunkel R. Comparison of hydroxychloroquine and chloroquine use and the development of retinal toxicity. J Rheumatol. 1985;12:692–694. [PubMed] [Google Scholar]
- 82.Sahraei Z, Shabani M, Shokouhi S, Saffaei A. Aminoquinolines against coronavirus disease 2019 (COVID-19): chloroquine or hydroxychloroquine. Int J Antimicrob Agents. 2020;55:105945. doi: 10.1016/j.ijantimicag.2020.105945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Liu J, Cao R, Xu M, Wang X, Zhang H, Hu H, Li Y, Hu Z, Zhong W, Wang M (2020) Hydroxychloroquine, a less toxic derivative of chloroquine , is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov:6–9. 10.1038/s41421-020-0156-0 [DOI] [PMC free article] [PubMed]
- 84.Ohkuma S, Poole B. Cytoplasmic vacuolation of mouse peritoneal macrophages and the uptake into lysosomes of weakly basic substances. J Cell Biol. 1981;90:656–664. doi: 10.1083/jcb.90.3.656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Hasan R, Rink L, Haase H. Chelation of free Zn(2)(+) impairs chemotaxis, phagocytosis, oxidative burst, degranulation, and cytokine production by neutrophil granulocytes. Biol Trace Elem Res. 2016;171:79–88. doi: 10.1007/s12011-015-0515-0. [DOI] [PubMed] [Google Scholar]
- 86.Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14:353–357. doi: 10.2119/2008-00033.Prasad. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Haase H, Rink L. Zinc signals and immune function. Biofactors. 2014;40:27–40. doi: 10.1002/biof.1114. [DOI] [PubMed] [Google Scholar]
- 88.Bonaventura P, Benedetti G, Albarede F, Miossec P. Zinc and its role in immunity and inflammation. Autoimmun Rev. 2015;14:277–285. doi: 10.1016/j.autrev.2014.11.008. [DOI] [PubMed] [Google Scholar]
- 89.Rahman MT, Karim MM. Metallothionein: a potential link in the regulation of zinc in nutritional immunity. Biol Trace Elem Res. 2018;182:1–13. doi: 10.1007/s12011-017-1061-8. [DOI] [PubMed] [Google Scholar]
- 90.Solomon VR, Lee H. Chloroquine and its analogs: a new promise of an old drug for effective and safe cancer therapies. Eur J Pharmacol. 2009;625:220–233. doi: 10.1016/j.ejphar.2009.06.063. [DOI] [PubMed] [Google Scholar]
- 91.Mizushima N, Yoshimori T, Levine B. Methods in mammalian autophagy research. Cell. 2010;140:313–326. doi: 10.1016/j.cell.2010.01.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Xue J, Moyer A, Peng B, Wu J, Hannafon BN, Ding WQ. Chloroquine is a zinc ionophore. PLoS One. 2014;9:e109180. doi: 10.1371/journal.pone.0109180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Denison MR, Perlman S. Translation and processing of mouse hepatitis virus virion RNA in a cell-free system. J Virol. 1986;60:12–18. doi: 10.1128/JVI.60.1.12-18.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Denison MR, Zoltick PW, Hughes SA, Giangreco B, Olson AL, Perlman S, Leibowitz JL, Weiss SR. Intracellular processing of the N-terminal ORF 1a proteins of the coronavirus MHV-A59 requires multiple proteolytic events. Virology. 1992;189:274–284. doi: 10.1016/0042-6822(92)90703-r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ko W-S, Guo C-H, Hsu G-SW, Chiou YL, Yeh MS, Yaun SR. The effect of zinc supplementation on the treatment of chronic hepatitis C patients with interferon and ribavirin. Clin Biochem. 2005;38:614–620. doi: 10.1016/j.clinbiochem.2005.04.003. [DOI] [PubMed] [Google Scholar]
- 96.Suzuki H, Takagi H, Sohara N, Kanda D, Kakizaki S, Sato K, Mori M, Gunma Liver Study Group Triple therapy of interferon and ribavirin with zinc supplementation for patients with chronic hepatitis C: a randomized controlled clinical trial. World J Gastroenterol. 2006;12:1265–1269. doi: 10.3748/wjg.v12.i8.1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Saper RB, Rash R. Zinc: an essential micronutrient. Am Fam Physician. 2009;79:768–772. [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
I have not used any data that are required to be available for the readers.