Abstract
Purpose of Review
This paper seeks to review the current literature and trends regarding use of hamstring autograft for lateral ankle instability.
Recent Findings
Reconstruction of the lateral ankle ligaments using hamstring autograft has been found to be an effective method to treat ankle instability in terms of patient-reported outcomes and objective measures. Biomechanically, reconstruction has been shown to be stronger (load to failure) when compared with the Broström procedure. Clinical studies have demonstrated non-inferiority when compared with the Broström procedure, with one synthetic reconstruction technique demonstrating superior outcomes.
Summary
Reconstruction of the lateral ankle ligaments using hamstring autograft is especially useful in patients who are at high risk of failure (insufficient soft tissue available for repair, ligamentous laxity, previous failed ligament repair, ossicle > 1 cm, or in the heavier, high-demand athletes).
Keywords: Lateral ankle instability, Hamstring autograft, Broström procedure
Introduction
Ankle sprains are the most common injury affecting the athlete, accounting for over 40% of all sports injuries depending on the sport [1–3]. Overall, it is estimated that there are over 2 million ankle sprains in the USA a year [4]. Majority of injuries to the ankle affect the lateral ligamentous structures, with the anterior talofibular ligament (ATFL) being the most commonly injured structure [5, 6]. Majority of ankle injuries respond well to non-operative treatment, with over 85% of individuals recovered at the 3-year time point [7]. However, a prospective cohort demonstrated that up to 40% of individuals with an acute ankle injury progress to symptoms of chronic ankle instability at 1 year out from injury [8]. Chronic ankle instability can occur due to repeated injuries to the same structures, ultimately leading to chronic laxity.
The lateral ligamentous complex of the ankle joint consists of three ligaments: anterior talofibular (ATFL), calcaneofibular ligament (CFL), and the posterior talofibular ligament (PTFL). The ATFL has been found to be the weakest of the three ligaments. The main function of the ATFL is to prevent inversion of the ankle and anterior translation of the talus. Injury to this structure occurs due to inversion of the ankle in a plantarflexed position [9]. The ATFL originates on the anterior portion of the fibular head and inserts just distal to the articular margin of the talus, with the center of this insertion approximately 18 mm proximal to the subtalar joint [10]. The calcaneofibular ligament is the second most commonly injured ligament of the lateral ligamentous complex. The main function of the CFL is to prevent excessive supination of the ankle and subtalar joints. The CFL can be injured during an inversion injury with the foot in a dorsiflexed position [9]. The CFL originates from the posterior portion of the lateral malleolus and inserts onto the calcaneus at a mean angle of 133° from the long axis of the distal fibula [10]. Finally, the posterior talofibular ligament has broad origins and insertions on the posterolateral aspect of the lateral malleolus and posterolateral talus [10]. The PTFL provides restraint to inversion and internal rotation of a loaded ankle joint [11].
Indications for operative intervention for lateral ankle instability include failure of non-operative treatment (i.e., bracing, physical therapy, activity modification) in the setting of chronic lateral ankle instability. Over 50 procedures have been described for lateral ankle instability [12]. Historically, broad categories of reconstruction included non-anatomic and anatomic. While historically, non-anatomic procedures were more commonly performed, more recent literature indicates that anatomic repairs result in more physiologic motion and good clinical outcomes when compared with non-anatomic procedures [13, 14]. A very commonly performed anatomic procedure is the Broström procedure with Gould modification [15]. This involves direct repair of the injured ligaments with augmentation using the inferior extensor retinaculum [16, 17]. Previous literature indicates good long-term results with this procedure [18–21].
However, even with good clinical results, the Broström procedure with Gould modification (MBG) has a failure rate of over 10% [20–23]. This number is higher in patients who are high-demand athletes, demonstrate ligamentous laxity, have previously failed a repair, or those with confounding factors predisposing to chronic laxity [24]. More recent literature has demonstrated improved patient-reported outcomes when performing anatomic reconstruction compared with anatomic repair (MBG) in a randomized clinical trial at 2- and 5-year follow-up [22, 25]. The gold standard for graft choice for this procedure has not been defined, and the use of autograft, allograft, and synthetic graft has been described [26–32].
The use of hamstring autograft for anatomic reconstruction of the lateral ankle ligamentous complex was first described in 2000 by Paterson et al. and has since been modified and described by other authors [28, 33, 34]. The purpose of this paper is to review the current literature and trends regarding use of hamstring autograft for lateral ankle instability.
Indications
Two general categories of instability exist: mechanical instability and functional instability. Mechanical instability is defined by motion that is beyond physiologic norms for the patient. Functional instability is defined by motion that is physiologic but the patient continues to have the perception that the ankle is giving way [35]. While these categories are not always mutually exclusive, it is important to understand them as it can guide treatment. The notion exists that patients with functional instability may have strength and proprioceptive deficits that are more likely to be benefited by physical therapy [36].
Surgical consideration is warranted in patients who fail conservative management including the use of a brace and physical therapy for proprioception and ankle stabilization through peroneal muscle training and strengthening. Previous studies have demonstrated similar return to pre-injury status when comparing non-operative treatment with acute surgery [37]. When surgery is indicated, anatomic repairs such as the Broström procedure with Gould modification remain the gold standard in treatment of lateral ankle instability. It results in good clinical outcomes, even with long-term follow-up [18, 19]. However, this procedure is relatively contraindicated in patients who have insufficient soft tissue available for repair, generalized ligamentous laxity (as defined by Beighton score > 5), previous failed ligament repair, ossicle > 1 cm, or in the heavier, high-demand athletes [30, 38, 39]. Consideration to anatomic reconstruction should be made in patients with severe ankle instability as indicated by a talar tilt > 20° and anterior translation of the talus > 15 mm as measured on stress radiographs [27]. In our own experience, the senior author (M.C.D) has used similar indications for anatomic reconstructions over anatomic repair.
Preoperative Planning
Assessment of a patient with concern for chronic lateral ankle instability should include a thorough history that focuses on the length, quantity, and energy level of inversion injuries. Physical exam should include assessment of patient’s lower mechanical axis, foot alignment (cavovarus), muscular strength (peroneals), and evaluation of ankle instability with a talar tilt test and anterior drawer test. Radiographic evaluation should include standard ankle series (AP, lateral, and mortise), as well as stress radiographs (talar tilt, anterior drawer). Anterior translation is measured as the distance the talus shifts anterior in relation to the most posterior portion of the tibial and talar articular surface with the ankle in 15° of dorsiflexion [40]. Instability is defined as a talus shifts greater than 10 mm (in isolation), or greater than 5 mm difference than the contralateral side, assuming no pathology contralaterally. Recent literature advocates for numbers as low as 4 mm of absolute shift being indicative of ankle instability [41]. Instability with talar tilt which is radiographic also represents CFL incompetence, should normally be less than 10°, or less than 5° different compared with the contralateral side. The talar tilt test is performed by inverting the calcaneus, stabilizing the tibia, and holding the foot in neutral position [40]. (Fig. 1) Advanced imaging, such as magnetic resonance imaging, can be helpful to look for other intra-articular pathology that can be addressed concomitantly. Previous literature indicates the incidence of concomitant chondral injury to be as high as 95% [42, 43]. However, due to its static nature, MRI often lacks utility when diagnosing symptomatic lateral ankle instability. Up to 60% of asymptomatic patients will have torn or attenuated ATFLs on MRI [44]. After an injury to the ATLF, it often never looks normal again on MRI; however, this does not mean the patient suffers from ankle instability. Stress exams may be a more effective modality to assess the competence of the lateral structures by using dynamic testing.
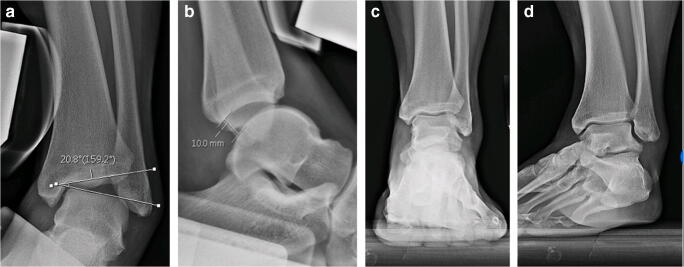
Fig. 1.
Preoperative radiographs including anteroposterior (a) and lateral (b) stress images showing talar tilt of 20.8° and anterior drawer of 10.0 mm as well as standard, standing anteroposterior (c) and oblique (d) views. Increased talar tilt and anterior drawer on stress radiographs indicate for operative treatment via lateral ligament reconstruction
Surgical Technique
The technique for reconstruction of lateral ligamentous structures by the senior author (M.C.D) has been previously published [27]. The patient is positioned supine with a large bump under the ipsilateral ischial tuberosity and a tourniquet on the affected thigh. The affected leg should be prepped and draped from above the knee down in a usual sterile fashion. The tourniquet is insufflated to 250 mmHg after exsanguination of the extremity. The ankle is placed in traction, and ankle arthroscopy is performed, addressing all intra-articular pathology.
Hamstring Autograft Harvest and Preparation
A linear incision measuring approximately 3 cm should be made halfway between the tibial tubercle and the posteromedial border of the tibia beginning at the top of the tibial tubercle and moving distally. Soft tissue dissection should continue using knife and electrocautery as desired until the sartorial fascia is encountered. The sartorial fascia is cleared of adipose tissue using blunt dissection with a lap sponge. The gracilis and semitendinosus should then be identified using manual palpation. The sartorial fascia is divided on the proximal aspect of the gracilis and semitendinosus taking care not to cut too deep, as fibers of the superficial medial collateral ligament are just deep to the gracilis and semitendinosus. Metzenbaum scissors can be used to extend the opening in the sartorial fascia as needed. At this time, the decision can be made as to whether the semitendinosus, gracilis, or both tendons are to be harvested, with size being the determining factor. Ideally, the graft should be a minimum of 4 mm. Our preference is to leave the tendon of choice attached distally. Metzenbaum scissors are used to remove any adhesions between the tendon and surrounding tissues. Care should be taken to ensure all adhesions are removed in order to prevent a short graft harvest. To ensure adhesion removal, tensioning the tendon of choice should result in no medial gastrocnemius movement. Manual palpation should also be used to ensure that the tendon is free up to its hiatus.
The Linvatec tendon stripper (ConMed, Utica, NY) is used to harvest the tendon. Care should be taken to appropriately point the tendon stripper along the axis of the tendon aiming for the ischial tuberosity. The tendon is then harvested, removed distally, and brought safely to the back table for preparation. A ruler, cobb, or elevator is used to remove muscle off of the tendon. The graft is tubularized with a 0 vicryl suture using a modified Krakow Stitch. The typical length of the tendon measures between 22 and 30 cm. The typical length of the graft required ranges from 12 to 15 cm, meaning that some or all portions of this graft can be doubled as the surgeon sees fit. It is important, however, to consider the diameter of the doubled tendon and whether there is enough bony support for tunnels in the fibula, calcaneus, and talus. Typically, the tendon is doubled over to get a 4.5–5 mm diameter graft.
Reconstruction of the Lateral Ligaments Using Hamstring Autograft
A 5-cm curvilinear incision is made over the distal fibula (Fig. 2). The CFL and ATFL are dissected off the distal fibula. The peroneal tendons are protected posterior during this portion of the case. In order to facilitate graft passage later in the case, a path should be created on the deep surface of the peroneal tendons, being careful not to violate the sural nerve as it passes posterior to the peroneal tendons. After this dissection is complete, a tunnel should be drilled in the fibula. This should be directed from anteriosuperior to posterioinferior with the anterior starting point being just distal to the ankle joint. The tunnel size should be approximately the same size as the graft. Care should be taken to protect the peroneals as the tunnel is drilled. A Huston suture passer is then used to pass a looped 0 vicryl suture that can be used for shuttling at a later point. Hemostat clamp should secure the stitch.
Fig. 2.
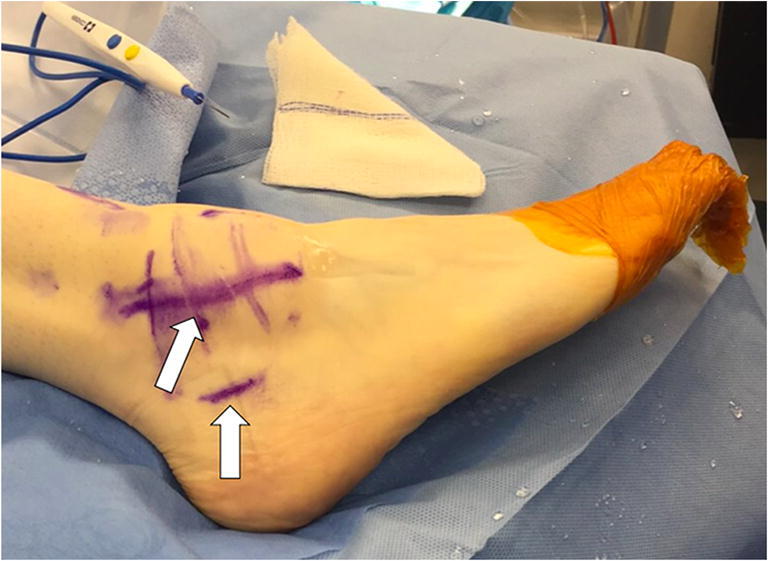
Sites of fibular and calcaneal incisions marked and indicated by arrows. An approximately 1-cm incision is made at the lateral aspect of the calcaneus, below the peroneal tendons and an approximately 5-cm curvilinear incision is made at the distal fibula
Attention is then turned to the calcaneus. Fluoroscopy is used to create a 1-cm incision at the calcaneal insertion of the CFL (Fig. 2). This should be located just posterior to the fibula, and approximately 1 cm distal to the superior edge of the calcaneus just posterior to the peroneals. Blunt dissection is carried down to the periosteum, being careful to not injure the peroneal tendons or sural nerve. A docking tunnel is drilled at this location approximately 0.5 mm larger than the graft. A pin is used to identify location first, and once confirmed on fluoroscopy, it can be reamed using the correct size drill. The socket should measure approximately 20 mm in depth.
Attention is then turned to the talus. Identification of the ATFL insertion on the talus is performed. This is located on the anterior process of the talus, just distal to the articular surface, and approximately 18 mm from the subtalar joint. A pin with eyelet is used to identify the correct position and drilled all the way through the talus and out the medial skin between the tibialis anterior and posterior tibialis tendon. Reamer is used over the pin, again measuring the same size as the graft. A looped 0 vicryl is then passed through the tunnel by pulling the pin out of the talus from the medial side. Passing stitch is held in place with a hemostat (Fig. 3).
Fig. 3.
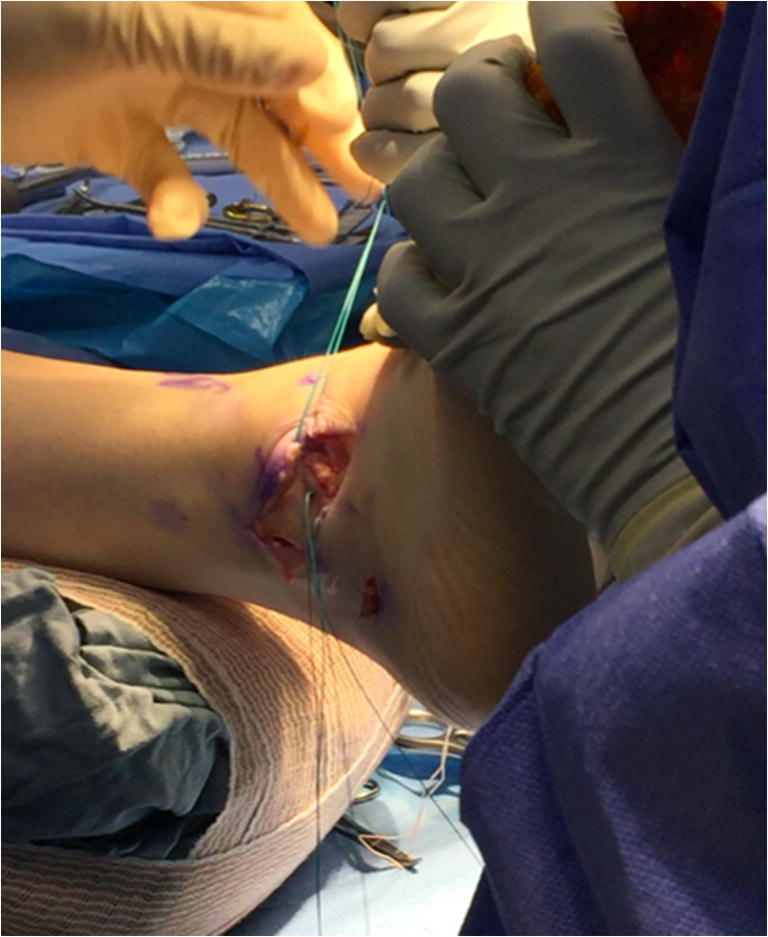
Passing sutures are in place and will be used to shuttle the hamstring autograft through each of the bone tunnels
Appropriate soft tissue plane is then created between the calcaneal incision and the fibular-based incision, insuring to keep this plan posterior (or deep) to the peroneal tendons. This is done with blunt dissection with a hemostat and periosteal elevator. Care is taken to avoid injury to the sural nerve. Passing stitched is placed through this appropriate soft tissue plane and held in place with hemostat (Fig. 3).
The graft is then docked in the tunnel in the calcaneus using a biotenodesis screw. Screw size should be the same or 0.25 mm larger than the tunnel based on what options one has available. Care should be taken to ensure the graft docks in the socket prior to placement of the screw. The graft is then shuttled in the appropriate soft tissue plane just posterior to the peroneal tendons. It is then passed through the fibula from posterior to anterior using the previously placed shuttling stitch. The graft is finally brought down to the talus, and through the tunnel created using the shuttle stitch (Fig. 4). A medial skin incision is made to pull the remainder of the graft out through the skin. The ankle is held in eversion and posterior translation, and the graft is fixed to the talus first and fibula using biotenodesis screw. The native ATFL and CFL are repaired and the wound is closed per the surgeon’s preference.
Fig. 4.

The graft has been pulled out of the medial aspect of the talus after being initially secured in the calcaneal bone tunnel and then passed, first, underneath the peroneal tendons, and then underneath the ATFL
The patient is made non-weight bearing with splint immobilization for 2 weeks, with aspirin for DVT prophylaxis unless other risk factors exist. The patient is then transitioned to a boot and gentle range of motion exercises is started. The patient begins advancement of weight-bearing at 4 weeks, with full weight-bearing at 6 weeks. At this time, physical therapy is initiated for proprioception, motion, and gradual strengthening. The patient returns to running at 3 months, and full clearance to sports at 6 months, depending on any other concomitant procedures performed. Postoperative stress radiographs can be performed to evaluate the reconstruction as well as tunnel position (Figs. 5 and 6).
Fig. 5.
Postoperative anteroposterior stress radiograph (a) indicating a decreased talar tilt of 5.4° and postoperative lateral stress radiograph (b) indicating a decreased anterior drawer of 7.2 mm
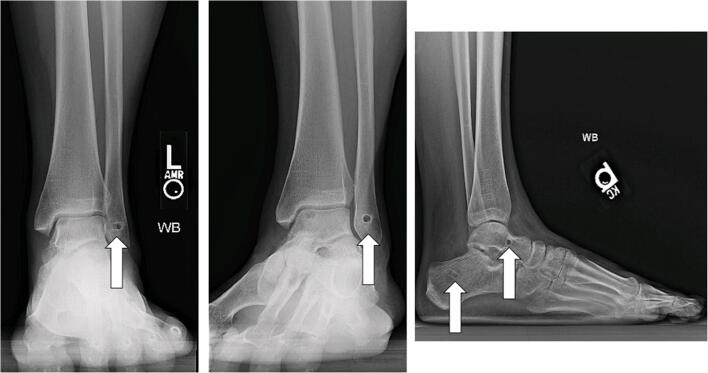
Fig. 6.
Postoperative anteroposterior (a), oblique (b), and lateral (c) weight-bearing radiographs show the 3 bone tunnels created in the fibula, calcaneus, and talus. Arrows indicate the sites of the bone tunnels. The fibular tunnel is seen on the anteroposterior and oblique views. Calcaneal and talar tunnels are seen on the lateral view
Current Literature (Outcomes, Complications)
Biomechanics
Previous biomechanical research indicates that the stiffness and ultimate load to failure of ATFL reconstruction using semitendinosus allograft was not significantly different than the native ATFL [45]. This data can also reasonably be assumed to be true for the use of autograft. Biomechanically, the Broström procedure was shown to have half the strength of the native ATFL [46]. This supports the current indications for anatomic reconstruction (high-demand patient, larger patient, previously failed repair, etc.). Previously, studies have also established that interference screws are superior to bone anchors in both graft elongation and mean load to failure for fixation in anatomic reconstruction of the lateral ligamentous complex [47].
Clinical Outcomes
To the authors’ knowledge, there have been 6 published studies on the use of hamstring autograft for the treatment of lateral ankle instability (Table 1). The current literature is composed of case series and comparative case series.
Table 1.
Published studies on the use of hamstring autograft for the treatment of lateral ankle instability
| Author, year | Study, level of evidence | Type of graft | No. of ankles (patients) | Average follow-up (range), months | Survivorship | Results |
|---|---|---|---|---|---|---|
| Coughlin et al. (2004) | Case series, IV | Gracilis tendon autograft | 29 (28) | 23 (12–52) | 100% |
86% (24 patients) excellent result, 14% (4 patients) good result AOFAS AHS Preop 57 Postop 98 (p < .01) |
| Takao, et al. (2005) | Case series, IV | Gracilis tendon autograft | 17 (17) ATLF reconstruction, 4 (4) ATFL+CFL reconstruction | 24 | 100% |
AOFAS AHS Preop 69.4 ± 4.0 Postop 97.0 ± 2.6 points (p < .01) |
| Ibrahim et al. (2010) | Case series, IV | Gracilis tendon autograft | 14 (14) | 33.5 (32–48) | 100% |
AOFAS AHS Preop 58 Postop 96 (p < .01) 69% (10 patients) excellent result, 31% (4 patients) good result |
| Xu et al. (2014) | Comparative case series, III | Semitendinosus autograft versus semitendinosus allograft | 32 (32) autograft, 36 (36) allograft |
Autograft 33.5 ± 6.7 Allograft 28.5 ± 6.7 |
100% (both groups) |
AOFAS AHS Autograft Preop 62.3 ± 8.2 Postop 95.1 ± 7.5 (p < .01) Allograft Preop 60.2 ± 8.4 Postop 94.8 ± 5.5 (p < .01) |
| Li et al. (2017) | Comparative case series, III | Semitendinosus autograft versus semitendinosus allograft | 10 (10) autograft, 16 (16) allograft |
Autograft 37.7 ± 20.8 Allograft 55.9 ± 35.5 |
100% (both groups) |
AOFAS AHS Autograft Preop 68.4 ± 10.0 Postop 94.7 ± 5.0 (p < .01) Allograft Preop 69.9 ± 13.3 Postop 94.8 ± 5.4 (p < .01) Postoperative T2 MRI demonstrated more signal in allograft compared with autograft (p = .027) |
| Song et al. (2017) | Comparative case series, III |
Broström group Semitendinosus autograft |
16 (16) Broström 12 (12) autograft |
Broström 16.31 ± 4.16 Autograft 19.33 ± 5.14 |
100% (both groups) |
AOFAS AHS Broström Preop 59.31 ± 7.15 6 months 86.19 ± 4.119 12 months 93.00 ± 3.950 Autograft Preop 61.25 ± 5.39 6 months 93.25 ± 7.338 12 months 94.42 ± 5.316 AOFAS score significantly different between groups at 6-month time point only |
Coughlin et al. published the first case series on the use of hamstring autograft for the treatment of lateral ankle instability. They reported 86% of patients with excellent results and 14% with good results, based on patient satisfaction. AOFAS increased in a clinically and statistically significant way when comparing preoperative to postoperative scores [48]. Two other case series have since been published with similar results in terms of their patient-reported outcome scores. No failures are noted in any group [49, 50].
Three comparative case series currently exist in the literature [34, 51, 52]. Xu et al. reported on reconstruction of the ATFL and CFL using semitendinosus autograft versus semitendinosus allograft. Both groups have clinically and statistically significant improvements in their AOFAS scores from preop to postop, and the groups did not differ from each other. No failures were reported in either group with a mean follow-up 33.5 ± 6.7 months in the autograft group, and 28.5 ± 6.7 months in the allograft group [51]. Li et al. corroborated these findings in a comparative case series comparing semitendinosus allograft and autograft. They noted similar findings with regard to AOFAS. In addition to this, the study investigated postoperative MRI, and found increased signal on T2 MRI in the allograft group compared with the autograft group that was statistically significant. While no clinical differences were found, this could indicate improved healing and vascularity with autograft compared with allograft [52]. Finally, Song et al. investigated results following a modified Broström Gould and semitendinosus autograft. Both groups improved with regard to their AOFAS. There was a statistically significant difference in AOFAS at 6 months, with the autograft group outperforming the Broström group; however, this difference was not seen at 12 months.
Currently, there are no randomized clinical trials exist comparing hamstring autograft reconstruction to the modified Broström Gould (considered gold standard for lateral ankle instability). However, a randomized clinical trial has been performed investigating the difference between the ligament augmentation reconstruction system (LARS AC 30 DB, LARS Surgical Implants and Devices, Arc-sur-Tille, France) and the modified Broström Gould. Clinical results have been published at 2 and 5 years [22, 25]. While this does not entirely represent findings you can expect with autograft, it does give insight into an anatomic reconstruction versus modified Broström Gould. Clinical findings demonstrated statistically significant differences in total FAOS score at the 1-year, 2-year, and 5-year time points favoring the LARS group. No clinical failures were noted in the LARS group, with 2 failures noted in the modified Broström Gould [25]. A study by Drakos et al. demonstrated that autograft was better when compared with allograft, even in patients with generalized ligamentous laxity [53].
Conclusion
Reconstruction of the lateral ankle can be performed using hamstring autograft in a reliable and reproducible fashion with short-term results that are comparable with the current gold standard (MBG). Further clinical research is required to investigate the long-term outcomes following lateral ankle ligamentous reconstruction using hamstring autograft. Anatomic reconstruction using hamstring autograft is a reliable procedure for the treatment of chronic ankle instability, especially in those patients that are at greater risk of failure (i.e., insufficient soft tissue available for repair, ligamentous laxity, previous failed ligament repair, ossicle > 1 cm, or in the heavier, high-demand athletes).
Compliance with Ethical Standards
Conflict of Interest
Karan A. Patel and Taylor Cabe declare that they have no conflict of interest. Mark C. Drakos declares that he is a paid consultant for Fast Form, Extremity Medical, and Pitusan 360.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Footnotes
This article is part of the Topical Collection on Management of Ankle Instability
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Karan A. Patel, Email: Karanapatel30@gmail.com
Taylor Cabe, Email: cabet@hss.edu, Email: taylorcabe29@gmail.com.
Mark Drakos, Email: DrakosM@hss.edu.
References
- 1.Milgrom C, Shlamkovitch N, Finestone A, Eldad A, Laor A, Danon YL, Lavie O, Wosk J, Simkin A. Risk factors for lateral ankle sprain: a prospective study among military recruits. Foot Ankle. 1991;12:26–30. doi: 10.1177/107110079101200105. [DOI] [PubMed] [Google Scholar]
- 2.DiGiovanni BF, Partal G, Baumhauer JF. Acute ankle injury and chronic lateral instability in the athlete. Clin Sports Med. 2004;23:1–19, v. doi: 10.1016/S0278-5919(03)00095-4. [DOI] [PubMed] [Google Scholar]
- 3.Fernandez WG, Yard EE, Comstock RD. Epidemiology of lower extremity injuries among U.S. high school athletes. Acad Emerg Med Off J Soc Acad Emerg Med. 2007;14:641–645. doi: 10.1197/j.aem.2007.03.1354. [DOI] [PubMed] [Google Scholar]
- 4.Waterman BR, Owens BD, Davey S, Zacchilli MA, Belmont PJ. The epidemiology of ankle sprains in the United States. J Bone Joint Surg Am. 2010;92:2279–2284. doi: 10.2106/JBJS.I.01537. [DOI] [PubMed] [Google Scholar]
- 5.Tummala SV, Hartigan DE, Makovicka JL, Patel KA, Chhabra A. 10-year epidemiology of ankle injuries in men’s and women’s collegiate basketball. Orthop J Sports Med. 2018;6:2325967118805400. doi: 10.1177/2325967118805400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Martin Robroy L., Davenport Todd E., Paulseth Stephen, Wukich Dane K., Godges Joseph J. Ankle Stability and Movement Coordination Impairments: Ankle Ligament Sprains. Journal of Orthopaedic & Sports Physical Therapy. 2013;43(9):A1–A40. doi: 10.2519/jospt.2013.0305. [DOI] [PubMed] [Google Scholar]
- 7.Shakked R, Sheskier S. Acute and chronic lateral ankle instability diagnosis, management, and new concepts. Bull Hosp Jt Dis 2013. 2017;75:71–80. [PubMed] [Google Scholar]
- 8.Doherty C, Bleakley C, Hertel J, Caulfield B, Ryan J, Delahunt E. Recovery from a first-time lateral ankle sprain and the predictors of chronic ankle instability: a prospective cohort analysis. Am J Sports Med. 2016;44:995–1003. doi: 10.1177/0363546516628870. [DOI] [PubMed] [Google Scholar]
- 9.de Asla RJ, Kozánek M, Wan L, Rubash HE, Li G. Function of anterior talofibular and calcaneofibular ligaments during in-vivo motion of the ankle joint complex. J Orthop Surg. 2009;4:7. doi: 10.1186/1749-799X-4-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Burks RT, Morgan J. Anatomy of the lateral ankle ligaments. Am J Sports Med. 1994;22:72–77. doi: 10.1177/036354659402200113. [DOI] [PubMed] [Google Scholar]
- 11.Stability of the loaded ankle. Relation between articular restraint and primary and secondary static restraints. - PubMed - NCBI [Internet]. [cited 2019 Jun 13]. Available from: https://www.ncbi.nlm.nih.gov/pubmed/4051085. [DOI] [PubMed]
- 12.Yasui Y, Shimozono Y, Kennedy JG. Surgical procedures for chronic lateral ankle instability. JAAOS - J Am Acad Orthop Surg. 2018;26:223–230. doi: 10.5435/JAAOS-D-16-00623. [DOI] [PubMed] [Google Scholar]
- 13.Cao Y, Hong Y, Xu Y, Zhu Y, Xu X. Surgical management of chronic lateral ankle instability: a meta-analysis. J Orthop Surg. 2018;13:159. doi: 10.1186/s13018-018-0870-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hennrikus WL, Mapes RC, Lyons PM, Lapoint JM. Outcomes of the Chrisman-Snook and modified-Broström procedures for chronic lateral ankle instability. A prospective, randomized comparison. Am J Sports Med. 1996;24:400–404. doi: 10.1177/036354659602400402. [DOI] [PubMed] [Google Scholar]
- 15.Repair of lateral ligament of ankle - Nathaniel Gould, 1987 [Internet]. [cited 2020 Mar 4]. Available from: https://journals.sagepub.com/doi/abs/10.1177/107110078700800111?journalCode=faia
- 16.Gould N, Seligson D, Gassman J. Early and late repair of lateral ligament of the ankle. Foot Ankle. 1980;1:84–89. doi: 10.1177/107110078000100206. [DOI] [PubMed] [Google Scholar]
- 17.Broström L. Sprained ankles. VI. Surgical treatment of “chronic” ligament ruptures. Acta Chir Scand. 1966;132:551–565. [PubMed] [Google Scholar]
- 18.Bell SJ, Mologne TS, Sitler DF, Cox JS. Twenty-six-year results after Broström procedure for chronic lateral ankle instability. Am J Sports Med. 2006;34:975–978. doi: 10.1177/0363546505282616. [DOI] [PubMed] [Google Scholar]
- 19.Maffulli N, Del Buono A, Maffulli GD, Oliva F, Testa V, Capasso G, et al. Isolated anterior talofibular ligament Broström repair for chronic lateral ankle instability: 9-year follow-up. Am J Sports Med. 2013;41:858–864. doi: 10.1177/0363546512474967. [DOI] [PubMed] [Google Scholar]
- 20.Lee KT, Park YU, Kim JS, Kim JB, Kim KC, Kang SK. Long-term results after modified Brostrom procedure without calcaneofibular ligament reconstruction. Foot Ankle Int. 2011;32:153–157. doi: 10.3113/FAI.2011.0153. [DOI] [PubMed] [Google Scholar]
- 21.So E, Preston N, Holmes T. Intermediate- to long-term longevity and incidence of revision of the modified Broström-Gould procedure for lateral ankle ligament repair: a systematic review. J Foot Ankle Surg Off Publ Am Coll Foot Ankle Surg. 2017;56:1076–1080. doi: 10.1053/j.jfas.2017.05.018. [DOI] [PubMed] [Google Scholar]
- 22.Porter M, Shadbolt B, Stuart R. Primary ankle ligament augmentation versus modified Brostrom-Gould procedure: a 2-year randomized controlled trial. ANZ J Surg. 2015;85:44–48. doi: 10.1111/ans.12837. [DOI] [PubMed] [Google Scholar]
- 23.Cho B-K, Kim Y-M, Kim D-S, Choi E-S, Shon H-C, Park K-J. Outcomes of the modified Brostrom procedure using suture anchors for chronic lateral ankle instability--a prospective, randomized comparison between single and double suture anchors. J Foot Ankle Surg Off Publ Am Coll Foot Ankle Surg. 2013;52:9–15. doi: 10.1053/j.jfas.2012.10.004. [DOI] [PubMed] [Google Scholar]
- 24.Park KH, Lee JW, Suh JW, Shin MH, Choi WJ. Generalized ligamentous laxity is an independent predictor of poor outcomes after the modified Broström procedure for chronic lateral ankle instability. Am J Sports Med. 2016;44:2975–2983. doi: 10.1177/0363546516656183. [DOI] [PubMed] [Google Scholar]
- 25.Porter M, Shadbolt B, Ye X, Stuart R. Ankle lateral ligament augmentation versus the modified Broström-Gould procedure: a 5-year randomized controlled trial. Am J Sports Med. 2019;47:659–666. doi: 10.1177/0363546518820529. [DOI] [PubMed] [Google Scholar]
- 26.Brambilla L, Bianchi A, Malerba F, Loppini M, Martinelli N. Lateral ankle ligament anatomic reconstruction for chronic ankle instability: allograft or autograft? A systematic review. Foot Ankle Surg Off J Eur Soc Foot Ankle Surg. 2018. [DOI] [PubMed]
- 27.Cody EA, Karnovsky SC, DeSandis B, Tychanski Papson A, Deland JT, Drakos MC. Hamstring autograft for foot and ankle applications. Foot Ankle Int. 2018;39:189–195. doi: 10.1177/1071100717738220. [DOI] [PubMed] [Google Scholar]
- 28.Jeys LM, Harris NJ. Ankle stabilization with hamstring autograft: a new technique using interference screws. Foot Ankle Int. 2003;24:677–679. doi: 10.1177/107110070302400904. [DOI] [PubMed] [Google Scholar]
- 29.Jung H-G, Shin M-H, Park J-T, Eom J-S, Lee D-O, Lee S-H. Anatomical reconstruction of lateral ankle ligaments using free tendon allografts and Biotenodesis screws. Foot Ankle Int. 2015;36:1064–1071. doi: 10.1177/1071100715584848. [DOI] [PubMed] [Google Scholar]
- 30.Michels F, Cordier G, Guillo S, Stockmans F, ESKKA-AFAS Ankle Instability Group Endoscopic ankle lateral ligament graft anatomic reconstruction. Foot Ankle Clin. 2016;21:665–680. doi: 10.1016/j.fcl.2016.04.010. [DOI] [PubMed] [Google Scholar]
- 31.Parks RM, Parks SM. Lateral ankle stabilization using acellular human dermal allograft augmentation. J Am Podiatr Med Assoc. 2015;105:209–217. doi: 10.7547/0003-0538-105.3.209. [DOI] [PubMed] [Google Scholar]
- 32.Richter J, Volz R, Immendörfer M, Schulz M. Reconstruction of the lateral ankle ligaments with hamstring tendon autograft in patients with chronic ankle instability. Oper Orthopadie Traumatol. 2012;24:50–60. doi: 10.1007/s00064-011-0126-7. [DOI] [PubMed] [Google Scholar]
- 33.Paterson R, Cohen B, Taylor D, Bourne A, Black J. Reconstruction of the lateral ligaments of the ankle using semi-tendinosis graft. Foot Ankle Int. 2000;21:413–419. doi: 10.1177/107110070002100510. [DOI] [PubMed] [Google Scholar]
- 34.Song B, Li C, Chen N, Chen Z, Zhang Y, Zhou Y, Li W. All-arthroscopic anatomical reconstruction of anterior talofibular ligament using semitendinosus autografts. Int Orthop. 2017;41:975–982. doi: 10.1007/s00264-017-3410-9. [DOI] [PubMed] [Google Scholar]
- 35.Delahunt E, Coughlan GF, Caulfield B, Nightingale EJ, Lin C-WC, Hiller CE. Inclusion criteria when investigating insufficiencies in chronic ankle instability. Med Sci Sports Exerc. 2010;42:2106–2121. doi: 10.1249/MSS.0b013e3181de7a8a. [DOI] [PubMed] [Google Scholar]
- 36.Freeman MA. Instability of the foot after injuries to the lateral ligament of the ankle. J Bone Joint Surg Br. 1965;47:669–677. [PubMed] [Google Scholar]
- 37.Pijnenburg ACM, Bogaard K, Krips R, Marti RK, Bossuyt PMM, van Dijk CN. Operative and functional treatment of rupture of the lateral ligament of the ankle. A randomised, prospective trial. J Bone Joint Surg Br. 2003;85:525–530. doi: 10.1302/0301-620x.85b4.13928. [DOI] [PubMed] [Google Scholar]
- 38.Schenck RC, Coughlin MJ. Lateral ankle instability and revision surgery alternatives in the athlete. Foot Ankle Clin. 2009;14:205–214. doi: 10.1016/j.fcl.2009.01.002. [DOI] [PubMed] [Google Scholar]
- 39.Messer TM, Cummins CA, Ahn J, Kelikian AS. Outcome of the modified Broström procedure for chronic lateral ankle instability using suture anchors. Foot Ankle Int. 2000;21:996–1003. doi: 10.1177/107110070002101203. [DOI] [PubMed] [Google Scholar]
- 40.Physical examination of the spine and extremities by Hoppenfeld, Stanley [Internet]. [cited 2019 Jun 15]. Available from: https://www.biblio.com/physical-examination-of-by-hoppenfeld-stanley/work/96680.
- 41.Dowling LB, Giakoumis M, Ryan JD. Narrowing the normal range for lateral ankle ligament stability with stress radiography. J Foot Ankle Surg. 2014;53:269–273. doi: 10.1053/j.jfas.2013.12.014. [DOI] [PubMed] [Google Scholar]
- 42.Komenda GA, Ferkel RD. Arthroscopic findings associated with the unstable ankle. Foot Ankle Int. 1999;20:708–713. doi: 10.1177/107110079902001106. [DOI] [PubMed] [Google Scholar]
- 43.Taga I, Shino K, Inoue M, Nakata K, Maeda A. Articular cartilage lesions in ankles with lateral ligament injury. An arthroscopic study. Am J Sports Med. 1993;21:120–126. doi: 10.1177/036354659302100120. [DOI] [PubMed] [Google Scholar]
- 44.Galli MM, Protzman NM, Mandelker EM, Malhotra AD, Schwartz E, Brigido SA. Examining the relation of osteochondral lesions of the talus to ligamentous and lateral ankle tendinous pathologic features: a comprehensive MRI review in an asymptomatic lateral ankle population. J Foot Ankle Surg Off Publ Am Coll Foot Ankle Surg. 2014;53:429–433. doi: 10.1053/j.jfas.2014.03.014. [DOI] [PubMed] [Google Scholar]
- 45.Clanton TO, Viens NA, Campbell KJ, Laprade RF, Wijdicks CA. Anterior talofibular ligament ruptures, part 2: biomechanical comparison of anterior talofibular ligament reconstruction using semitendinosus allografts with the intact ligament. Am J Sports Med. 2014;42:412–416. doi: 10.1177/0363546513509963. [DOI] [PubMed] [Google Scholar]
- 46.Waldrop Norman E., Wijdicks Coen A., Jansson Kyle S., LaPrade Robert F., Clanton Thomas O. Anatomic Suture Anchor Versus the Broström Technique for Anterior Talofibular Ligament Repair. The American Journal of Sports Medicine. 2012;40(11):2590–2596. doi: 10.1177/0363546512458420. [DOI] [PubMed] [Google Scholar]
- 47.Jeys L, Korrosis S, Stewart T, Harris NJ. Bone anchors or interference screws? A biomechanical evaluation for autograft ankle stabilization. Am J Sports Med. 2004;32:1651–1659. doi: 10.1177/0363546504265051. [DOI] [PubMed] [Google Scholar]
- 48.Coughlin MJ, Schenck RC, Grebing BR, Treme G. Comprehensive reconstruction of the lateral ankle for chronic instability using a free gracilis graft. Foot Ankle Int. 2004;25:231–241. doi: 10.1177/107110070402500407. [DOI] [PubMed] [Google Scholar]
- 49.Ibrahim SA, Hamido F, Al Misfer AK, Ghafar SA, Awad A, Salem HK, et al. Anatomical reconstruction of the lateral ligaments using Gracillis tendon in chronic ankle instability; a new technique. Foot Ankle Surg Off J Eur Soc Foot Ankle Surg. 2011;17:239–246. doi: 10.1016/j.fas.2010.07.006. [DOI] [PubMed] [Google Scholar]
- 50.Takao M, Oae K, Uchio Y, Ochi M, Yamamoto H. Anatomical reconstruction of the lateral ligaments of the ankle with a gracilis autograft: a new technique using an interference fit anchoring system. Am J Sports Med. 2005;33:814–823. doi: 10.1177/0363546504272688. [DOI] [PubMed] [Google Scholar]
- 51.Xu X, Hu M, Liu J, Zhu Y, Wang B. Minimally invasive reconstruction of the lateral ankle ligaments using semitendinosus autograft or tendon allograft. Foot Ankle Int. 2014;35:1015–1021. doi: 10.1177/1071100714540145. [DOI] [PubMed] [Google Scholar]
- 52.Li Q, Ma K, Tao H, Hua Y, Chen S, Chen S, Zhao Y. Clinical and magnetic resonance imaging assessment of anatomical lateral ankle ligament reconstruction: comparison of tendon allograft and autograft. Int Orthop. 2018;42:551–557. doi: 10.1007/s00264-018-3802-5. [DOI] [PubMed] [Google Scholar]
- 53.Drakos MC. Lateral ligament reconstruction with hamstring autograft for severe and revision cases of ankle instability. July 20th to July 23rd.