Abstract
Cell surface macromolecules play a crucial role in the biology and pathobiology of flaviviruses, both as receptors for virus entry and as signaling molecules for cell–cell interactions in the processes of vascular permeability and inflammation. This review examines the cell tropism and pathogenesis of flaviviruses from the standpoint of cell surface molecules, which have been implicated as receptors in both virus–cell as well as cell–cell interactions. The emerging picture is one that encompasses extensive regulation and interplay among the invading virus, viral immune complexes, Fc receptors, major histocompatibility complex antigens, and adhesion molecules.
I. Introduction
Flaviviruses comprise a rich and diverse family of agents that infect a variety of hosts and cause a wide spectrum of disease. Three disease types are recognized for flaviviruses, namely encephalitis, hemorrhagic fever, and fever–arthralgia–rash. Disease distinctions are not absolute, and overlapping pathologies among various flavivirus members are often observed. The ability of flaviviruses to cause such divergent clinical syndromes, associated with virus replication in a number of different organs, has profound implications for the types of cell surface molecules the virus recognizes as receptors. Mutational analyses of the flaviviral E protein have demonstrated a striking ability of flaviviruses to adapt to different cells and receptors. Given the considerable homologies among them, flaviviruses show a remarkable capacity to cause vastly different diseases with a minimum of alterations in the E protein.
The cell surface molecules, which act as receptors for flaviviruses, are only starting to be identified. In addition to providing the molecules involved in virus attachment and penetration, the host cell erects a battery of surface structures that mediate communication with other cells and trigger host defense and pathological processes. Many of these are modulated by flavivirus infection and contribute to the overall picture of pathogenesis.
II. The Flavivirus Receptor Binding Protein
The flavivirus E protein is a multifunctional protein involved in cell receptor binding (Anderson 1992, Chen 1996, He 1995) and virus entry via fusion with a host cell membrane (Rice, 1996). Some of the functional activities of the E protein, notably membrane fusion, are regulated by interaction with a second viral protein, prM. It is believed that the association of prM with E stabilizes certain pH-sensitive epitopes on the E protein, thereby preventing the conformational changes that normally occur at acidic pH and activate the fusogenic activity of the E protein (Allison 1995, Guirakhoo 1992, Heinz 1994). In addition to its normal role in flavivirus assembly, the prM protein has also been included in novel recombinant formulations in which it is generally coexpressed with the E protein; the resultant E⧸prM complexes have been shown to be immunogenic and protective as vaccines against challenge with several flaviviruses, including Japanese encephalitis virus (Mason et al., 1991), yellow fever virus (Pincus et al., 1992), dengue virus (Fonseca et al., 1994), and tick-borne encephalitis (TBE) virus (Heinz et al., 1995).
In TBE virus, the majority of extracellular virus is largely free of prM protein due to a late intracellular processing event that generates a carboxy-terminal fragment designated M and which together with the E and C proteins are believed to constitute the protein components of the mature virus particle (Heinz et al., 1994). Cleavage of prM to M enhances low pH–dependent virus–cell fusion (Guirakhoo et al., 1991) and infectivity (Guirakhoo 1992, Heinz 1994, Randolph 1990, Shapiro 1972, Wengler 1989). Dengue virions containing prM are still infectious (Randolph et al., 1990) and bind to permissive cells in a manner that can be blocked using E-specific antibodies (He 1995, Wang 1999). Virus particles containing mainly E and prM also show antibody-enhanced binding to Fc receptor-bearing K562 cells as well as to platelets (Wang et al., 1995). Thus, in addition to being requisite precursors to mature virus particles, virus particles containing prM possess many properties associated with mature virus particles.
Flaviviruses appear to gain entry to the cell by the endocytic pathway (Rice, 1996). At low pH, the E protein undergoes a conformational change (Allison et al., 1995) involving dissociation of the E dimer (Stiasny et al., 1996), thereby exposing a hidden fusion peptide, followed by reorganization of E into a trimer (Allison et al., 1995), in which the fusion peptide is brought close to the membrane-anchoring carboxy terminus (Ferlenghi et al., 2001). Remarkably similar structural features and conformational rearrangements have been noted between the flavivirus E protein and the alphavirus E1 (Heinz 2001, Lescar 2001, Pletnev 2001, Strauss 2001), suggesting a common evolutionary origin for these two virion surface proteins.
Considerable homology exists among flaviviral E proteins, raising the possibility that different flaviviruses may have similar receptor-binding motifs. For example, many mosquito-borne flaviviruses contain an RGD sequence (e.g., residues 388–390 of the Murray Valley encephalitis virus E protein), which has been implicated in virulence (Lobigs et al., 1990) and receptor binding by analogy with integrin-binding motifs (Rey et al., 1995). Mutagenesis studies of the yellow fever virus (Van der Most et al., 1999) and Murray Valley encephalitis virus (Hurrelbrink and McMinn, 2001) RGD motifs, however, have cast doubt on the role of integrins in flavivirus attachment or entry.
Studies with TBE virus have identified important determinants for pathogenicity within the suspected receptor-binding site on the upper-lateral surface of domain III (Mandl et al., 2000). Acquisition of heparan sulfate-binding mutations by passaging TBE in cell culture has also implicated amino acids in this region in receptor binding (Mandl et al., 2001). The selection of virus mutants on the basis of weak binding to brain membranes has been used with several neurotropic flaviviruses (Holbrook 2001, Ni 1998, Ni 2000) and has identified a variety of mutations within domain III as well as other regions of E. For dengue virus, blocking of virus cell binding correlates more closely to virus neutralization for mAb 3H5 than for mAb 1B7 (Wang et al., 1999). This may suggest that mAb 3H5 neutralizes dengue virus predominantly by blocking virus–cell attachment, whereas mAb 1B7 neutralizes dengue virus largely by a postattachment mechanism. The mAb 3H5-binding site on the dengue viral E protein has been partly characterized (Hiramatsu 1996, Megret 1992, Trirawatanapong 1992) and probably encompasses, at a minimum, residues 383–385 (Hiramatsu et al., 1996) within domain III. More recent data involving a larger number of monoclonal antibodies indicate that mAbs that interact with domain III are in fact the most effective blockers of virus–cell attachment (Crill and Roehrig, 2001). A putative heparan sulfate-binding site on the dengue-2 E protein is also located within this region (Chen et al., 1997), and comparative sequencing of dengue type 2 genomes has implicated amino acid 390 of the E protein as a major determinant of pathogenicity (Leitmeyer et al., 1999). The pH-dependent conformational “hinge” region (between domains I and II) of the E protein has also been implicated in virulence, receptor interaction, and⧸or membrane fusion (Hurrelbrink 2001, Lee 1997, Monath 2002). Further mutagenesis studies will undoubtedly help define the sites of the E protein involved in flavivirus–cell macromolecule recognition.
III. Cell Targets for Flaviviruses
A. Dendritic Cells
Transmission of flaviviruses to humans generally occurs via the bite of an infected mosquito or tick. In the case of dengue, inoculated virus is thought to first replicate in skin Langerhans (dendritic) cells (Palucka 2000, Taweechaisupapong 1996a, Taweechaisupapong 1996b, Wu 2000). Dendritic cells have also been shown to be involved in the transport of intradermally inoculated West Nile virus to local draining lymph nodes, with a subsequent accumulation of leukocytes (Johnston et al., 2000). It is likely that dendritic cells will prove to be efficient carriers of a wide number of flaviviruses from their cutaneous site of infection to lymphoid and possibly other tissues.
Given the importance of dendritic cells in initiating immune responses (Banchereau et al., 2000), they probably play a pivotal role in stimulating host defense against invading flaviviruses. Dengue virus infection of immature myeloid dendritic cells has been shown to induce their maturation accompanied by the expression of major histocompatibility complex (MHC) class I and II antigens; the costimulatory molecules CD40, CD80, and CD86; and the dendritic cell marker CD83 (Libraty et al., 2001). Such changes were seen in both dengue-infected and bystander cells, indicating that upregulation of cell surface molecules could be a consequence of virus infection as well as virus-induced cytokine expression. Similarly, Langerhans cells infected with West Nile virus, as well as an alphavirus, Semliki Forest virus, express increased cell surface MHC class II and appear to undergo maturation to a cell type similar to lymphoid dendritic cells (Johnston et al., 1996). The efficient presentation of both MHC class I– and II–associated viral peptides on the surface of dendritic cells permits the generation of potent cytotoxic and helper T cell responses (see also Section V,A).
B. Monocytes and Macrophages
Monocytes and macrophages have long been recognized as major targets of flavivirus replication in the human host (Halstead 1989, Halstead 1977b, Scott 1980). They are also important host cells for the antibody-enhanced replication of certain flaviviruses (see Section IV,C). Because of their presence in the circulation, blood monocytes may be particularly important to the pathogenesis of hemorrhagic viruses, such as dengue. Because most of the pathological changes associated with dengue virus are hemostatic in nature, it is suspected that blood cells, particularly virus-infected blood monocytes, orchestrate many of these effects.
Dengue virus–infected human monocytes have been shown to be potent sources of vasoactive cytokines such as tumor necrosis factor (TNF)-α (Anderson et al., 1997) and interleukin (IL)-1β (Chang and Shaio, 1994). Monocytes are also known producers of several other vasoactive mediators, including IL-6, platelet-activating factor (PAF), prostaglandins, thromboxanes, leukotrienes, and nitric oxide (Bulger 2000, Funk 2001, Lefer 1989, Maruo 1992, Montrucchio 2000, Szabo 1999), any of which could have powerful effects on endothelial cell physiology. A crucial aspect in understanding dengue pathogenesis will be the identification of additional vasoactive mediators, which trigger the key dysfunctional events in vascular integrity.
Various tissue macrophages are undoubtedly important in the pathogenesis of flaviviral diseases but have, to date, not received much attention. Skin mononuclear cells, pulmonary, splenic, and thymic macrophages and liver Kupffer cells have been recognized carriers of viral antigen (Halstead, 1989). In the liver, virus or viral antigen has been found in Kupffer cells and hepatocytes in infections with yellow fever (Monath et al., 1989) and dengue (Bhamarapravati 1967, Hall 1991, Halstead 1989, Rosen 1989). Destruction of Kupffer cells, possibly by apoptosis, has been reported in the liver of some patients with fatal dengue (Huerre et al., 2001). Primary cultures of Kupffer cells apparently undergo an abortive infection with dengue virus in which viral antigen but no progeny virus is produced (Marianneau et al., 1999).
C. Endothelial Cells
Many flaviviruses invade either visceral or central nervous system tissues following initial replication in dendritic cells, monocytes, or macrophages. Often this necessitates a transfer of virus across blood vessel endothelial layers.
For neurotropic flaviviruses, endothelial cells of the cerebral microvasculature constitute a barrier that must be overcome in order to gain access to the central nervous system. How this occurs remains uncertain. Transendothelial passage of virus may direct infection of cerebral microvascular endothelial cells, may transport across the endothelial layer, or both (Dropulic and Masters, 1990). Japanese encephalitis virus has been observed electron microscopically to traverse mouse cerebral endothelial cells by transcytosis (Liou and Hsu, 1998). Alternatively, virus may spread from blood vessels to the olfactory neuroepithelium and from there to olfactory neurons (McMinn 1996, Monath 1983).
Even normally nonneurotropic flaviviruses may occasionally invade the central nervous system under certain conditions. Modulation of the blood–brain barrier by anesthetics (Ben-Nathan et al., 2000) or lipopolysaccharide (Lustig et al., 1992) has been reported to facilitate neuroinvasion by a normally noninvasive strain of West Nile virus. Flaviviruses may also trigger the production of soluble factors that perturb the integrity of the blood–brain barrier, leading to increased leakage of proteins and cells into the central nervous system (Chaturvedi et al., 1991). These studies indicate that even nonneurotropic flaviviruses may infect tissues of the central nervous system or otherwise affect the integrity of the blood–brain barrier under special circumstances.
Transendothelial migration of individual leukocytes (e.g., lymphocytes, monocytes, neutrophils, eosinophils) is regulated in a highly specific manner by the differential expression of selected adhesion molecules on endothelial cells (reviewed in Crockett 1998, Lowell 1999. Flaviviruses, including dengue (Anderson et al., 1997) and West Nile (Shen et al., 1997) viruses, activate endothelial cell adhesion molecule expression by either direct (virus-mediated) or indirect (cytokine-mediated) mechanisms (see Section V,C). In the presence of leukocyte-attracting chemokines, such virus-triggered activation of the vascular endothelium may contribute toward the migration of leukocytes into extravascular tissues. In addition to being a mechanism for virus dissemination, this process may also be a factor in phenomena such as leukopenia and particularly neutropenia (loss of circulating leukocytes, neutrophils) often observed in flavivirus, particularly dengue, infection (reviewed in Halstead, 1989). Due to the lack of suitable animal models for severe dengue disease, i.e., dengue hemorrhagic fever (DHF) or dengue shock syndrome (DSS), there are difficulties in assessing the roles of such events, particularly the identification of adhesion molecules mediating the transendothelial migration of neutrophils using blocking antibodies against specific integrins, as has been performed for other disease states (Doerschuk 1990, Gao 1994, Issekutz 1993, Laberge 1995, Springer 1995).
The hallmark feature of increased vascular permeability in hemorrhagic flavivirus (e.g., dengue) infection suggests that vascular endothelial cells may mediate the fluid leakage and hemorrhaging that occur in DHF⧸DSS. Endothelial cells line the inner surface of blood vessels and play essential roles in maintaining an antithrombogenic surface and regulating vascular permeability. Increased vascular permeability can arise from a variety of mediators associated with acute inflammation and shock (Bulger 2000, Funk 2001, Lefer 1989, Michel 1988, Montrucchio 2000, Schnittler 1990). It is thought that vascular permeability is largely controlled by changes in endothelial cell–cell contact, which result in gap formation, thus allowing for fluid exchange between blood and interstitial tissue fluid (Michel, 1988). An electron microscopic study of endothelium from DHF biopsy samples revealed the occasional presence of gaps (Sahaphong et al., 1980), thus providing evidence that endothelial cell features may indeed be perturbed during DHF⧸DSS.
Although dengue virus infects endothelial cells in vitro (Andrews 1978, Avirutnan 1998, Killen 1993), there is no evidence that endothelial cell infection occurs clinically, as neither virus particles nor viral antigen has been detected in the endothelium of tissue specimens (Halstead 1988, Halstead 1989, Sahaphong 1980), in contrast to that seen in cases of ebola (Zaki et al., 1999) or hantaan hemorrhagic fever (Gavrilovskaya 1999, Wang 1997). It is likely that dengue virus mediates endothelial cell activation via an indirect route, involving blood monocytes, which are a major cell target for dengue virus infection (Halstead 1977b, Scott 1980). A major candidate event in such a route is the activation of endothelial cell adhesion molecules by a factor(s) (particularly TNF-α) produced by dengue virus–infected blood monocytes (Anderson et al., 1997).
TNF is a key cytokine in a variety of normal and pathological immune responses, including immunoregulation, regulation of cell proliferation, cytotoxicity, and in the mediation of endotoxic shock (Fiers 1991, Tartaglia 1992, Tracey 1993, Vassalli 1992). Monocyte-derived TNF-α appears to play a pivotal role in dengue-associated endothelial cell activation (Anderson et al., 1997) and may be an important effector in the manifestation of DHF⧸DSS. Support for the clinical significance of this observation comes from observations of elevated TNF levels in the sera of patients with severe dengue disease (Green 1999b, Hober 1993, Vitarana 1991, Yadav 1991). Taken together, current evidence indicates that dengue virus represents a rather unique group of viruses that target monocytes, thereby triggering the production of factors such as TNF-α, which in turn affect other cell targets, including endothelial cells. While the overall picture of endothelial cell dysfunction in DHF⧸DSS is obviously more complex than can be explained by any single factor, the role of TNF in dengue pathogenesis would seem to merit particular attention.
Current knowledge of endothelial cell responses observed in endotoxic shock may be instructive for the understanding of vascular leakage in DHF⧸DSS. Plasma leakage induced by endotoxin (lipopolysaccharide, LPS) from gram-negative bacteria encompasses a complex cascade of processes, including activation and functional alteration of endothelial cells. Major mediators of endothelial cell perturbation in endotoxic shock are LPS itself, as well as cytokines such as TNF-α and IL-1β (Bevilacqua, 1993). These factors can modulate endothelial cell function to varying degrees by activating cytokine and vasoactive factor release (Rink 1996, Shanley 1995), upregulating adhesion molecule expression (Bevilacqua 1993, Luscinskas 1991, Moser 1989, Smith 1989), and mediating transendothelial migration of specific leukocytes (Issekutz 1995, Luscinskas 1991, Morzycki 1990, Moser 1989, Smith 1989). Additional factors, particularly lipid mediators such as PAF, leukotrienes, thromboxanes, and prostaglandins, may contribute to further endothelial cell dysfunction, including vascular leakage (Bulger 2000, Funk 2001, Lefer 1989, Montrucchio 2000). While the involvement of these vasoactive mediators is recognized in endotoxic shock, more needs to be learned of their role in the vascular dysfunction that occurs in severe dengue disease.
D. Lymphocytes
Although lymphocytes are potently involved in the host response and immunopathology of flavivirus (especially dengue) diseases, their role as virus-permissive host cells is unclear. Dengue virus has been identified in circulating B cells from acutely ill dengue patients by immunocytochemistry and by recovery of infectious virus after passage in mosquitoes (King et al., 1999). In vitro studies showed that cells and cultured cell lines of both B and T cell derivation could be infected with dengue virus (Bielefeldt-Ohmann 2001, Kurane 1990, Marchette 1978, Mentor 1997, Sung 1975, Takasaki 2001, Theofilopoulos 1976). Continued passage of dengue virus in lymphoblastoid (Raji) cells can give rise to dengue virus variants capable of replication in human lymphocytes (Brandt et al., 1979). Interestingly, lymphocytes do not appear to undergo antibody-enhanced dengue virus infection (Brandt 1979, Kurane 1990), even though B cells do have Fc receptors (Dijstelbloem et al., 2001; see Section IV,C).
E. Neural Cells
The initial stages of pathogenesis for neurotropic flaviviruses appear to be common for flaviviruses in general in that the virus progresses from the subcutaneous site of inoculation to lymph nodes, followed by viremia and replication in extraneural tissues. Invasion into the central nervous system is marked by high virus titers in the brain and detectable virus or viral antigen in neurons (Albrecht, 1968). Cell destruction in tick-borne encephalitis may be less extensive than that seen in herpes simplex type 1 encephalitis (Studahl et al., 2000), although this is variable and may involve considerable inflammation (Chu 1999, Matthews 2000, Suzuki 2000). Susceptible cell types include both neurons and glial cells (Chu 1999, Ramos 1998, Steele 2000).
F. Basophils⧸Mast Cells
As notorious producers of vasoactive mediators, mast cells have been a source of controversial speculation for years in dengue pathogenesis. Cells resembling degranulated mast cells have been reported in skin perivascular infiltrates from DHF⧸DSS cases (Bhamarapravati et al., 1967). Dengue patients showed elevated levels of urinary histamine (a major granule product of mast cells), which correlated with disease severity (Tuchinda et al., 1977), suggesting that mast cells may have a contributory role in the pathogenesis of dengue. Although antihistamine treatment does not resolve shock in severely dengue-diseased patients (Halstead, 1989), histamine is only one of several potent vasoactive factors produced by mast cells (Benyon 1991, Bradding 1993, Galli 1984, Grabbe 1994, Marshall 1994, Moller 1991, Moller 1993, Moller 1998, Nilsson 1995, Schwartz 1984), some of which could cause vascular dysfunction in dengue infection. DHF⧸DSS patients have been reported to have elevated serum levels of IgE (Pavri et al., 1979), which has been speculated to relate to IgE-triggered histamine release in the manifestation of shock (Pavri and Prasad, 1980).
Mast cells reside mainly in the tissues, often closely associated with blood vessels (Alving 1991, Anton 1998, Pesci 1996, Pulimood 1998, Selye 1966, Selye 1968). They are present in large numbers in the skin (Marshall et al., 1987), where transmission of insect-borne flaviviruses occurs. Basophils, however, comprise about 1% of total circulating cells and would be accessible to virus in the blood. Dengue virus infects basophil⧸mast cell–like KU812 cells in an antibody-enhanced manner, coupled with the release of vasoactive cytokines, IL-1β and IL-6 (King 2000, King 2002). This cell line, which can be differentiated easily toward either a basophil or mast cell phenotype (Saito et al., 1995), may provide further insights into potential roles for basophils and mast cells in dengue disease.
Dengue patients show increased serum levels of anaphylatoxins C3a and C5a (Malasit, 1987), which can attract (Nilsson et al., 1996) and activate (Kownatzki, 1982) mast cells. Among the expected mast cell secretion products would be vasoactive factors, including histamine, which has been detected in elevated amounts in the urine of dengue patients (Tuchinda et al., 1977).
G. Platelets
Evidence for platelet involvement in dengue pathogenesis comes from at least two (probably related) sources. First, thrombocytopenia (loss of circulating platelets) is one of the most consistent clinical features of severe dengue infection (Halstead, 1989). Second, viral immune complexes have been detected on platelets from dengue patients (Boonpucknavig 1979, Phanichyakarn 1977a). Functional studies on platelets in dengue-diseased individuals have been sparse, but include a markedly reduced half-life (Mitrakul et al., 1977), deficient ADP release (Mitrakul et al., 1977), increased adhesiveness (Doury et al., 1976), increased tagging by complement fragments (Malasit, 1987), and increased release of β-thromboglobulin and platelet factor 4 (Srichaikul et al., 1989). There is also evidence for platelet activation in dengue patients (Doury 1976, Krishnamurti 2001, Srichaikul 1989). Although these results relate to a variety of platelet functions, they do indicate a general alteration in platelet physiology, which is consistent with platelet involvement and triggering of thrombocytopenia in dengue disease.
Dengue virus has been recovered from washed patient platelets (Scott et al., 1978), and virus has been reported to bind to platelets in the absence of antibody as assayed using immunofluorescence and immunoperoxidase techniques (Funahara et al., 1987). However, the levels of antibody-independent bound virus are very low compared to the levels of virus bound in the presence of dengue-specific antibodies (Wang et al., 1995). As noted earlier, dengue immune complexes have been demonstrated on platelets from dengue patients (Boonpucknavig 1979, Phanichyakarn 1977a). Weiss and Halstead (1965) originally proposed the possibility that dengue virus interactions with platelets might be involved in the thrombocytopenia observed in severe dengue disease. The finding that dengue virus binding to platelets is dependent on a virus-specific antibody is consistent with epidemiological and experimental data linking preexisting host antibodies to an increased risk of DHF⧸DSS (reviewed in Halstead, 1990).
Several other viruses have been shown to bind directly to platelets (Bik 1982, Danon 1959, Forghani 1983, Larke 1970, Lee 1993, Zucker-Franklin 1990). Platelet association may stabilize or protect blood-borne viruses (Larke and Wheelock, 1970) and may function as a mechanism of hematogenous dissemination (Forghani and Schmidt, 1983). Virus binding to platelets has been suggested to be a contributing mechanism to thromobocytopenia arising from infections with vaccinia (Bik et al., 1982), chikungunya (Larke and Wheelock, 1970), and rubella (Bayer et al., 1965). Thrombocytopenia in these virus infections is generally much milder than that observed in severe dengue disease.
Levels of dengue virus in the blood can exceed 107 infectious units⧸ml (Gubler 1988, Monath 1994). Such high viremic titers are likely necessary to ensure infection and transmission of the obligate mosquito intermediary host (Monath, 1994). Assuming a reasonable particle:infectivity ratio of 100:1, virus particle titers in blood may rival normal platelet counts (3 × 108⧸ml). Such parity between numbers of virus particles and platelets suggests that antibody-enhanced binding of virus to platelets may have a profound effect on platelets. Circulating virus-immune complexes are detected in DHF⧸DSS, and levels of immune complexes have been correlated with severity of disease (Ruangjirachuporn et al., 1979) and some of these are platelet associated (Boonpucknavig 1979, Phanichyakarn 1977a). These observations suggest that sufficient binding of virus immune complexes to platelets may occur to tag the majority of circulating platelets. Such an event could lead to immune clearance by the reticuloendothelial system, thereby precipitating the thrombocytopenia frequently associated with severe dengue disease.
It is likely that molecules other than Fc receptors on the platelet surface may mediate antibody-enhanced binding of dengue virus (Wang et al., 1995). Drug-induced thrombocytopenias provide interesting examples in this regard. It is known that given the appropriate accessory ligand (i.e., drug), IgG can bind to platelets through either the Fc receptor or other surface proteins. A variety of clinical thrombocytopenias are known that involve an immune component in pathogenesis. Many of these reflect activities of host antibodies, which react with proteins on the surface of platelets. These antibodies may be autoimmune in nature (i.e., antibodies that bind to platelet surface molecules) or dependent on a third party ligand (drug or protein), which then induces binding of the antibody–ligand complex to either the platelet Fcγ receptor or to another surface protein. For example, a number of individuals are susceptible to drug-dependent thrombocytopenia when administered drugs such as heparin or quinine⧸quinidine (Aster 1989, Hackett 1982). While heparin-dependent antibodies bind to the platelet Fcγ receptor (Adelman 1989, Chong 1989a, Chong 1989b, Kelton 1988), quinine⧸quinidine-dependent antibodies bind to platelet protein heterodimers GPIIb⧸IIIa and GPIa⧸IX (Berndt 1985, Chong 1983, Christie 1987, Devine 1995). This latter category of immune-mediated thrombocytopenia may be relevant to the understanding of dengue-associated thrombocytopenia, as patient antibodies mediate dengue virus binding to platelets via a platelet surface protein other than the Fcγ receptor (Wang et al., 1995).
Communication between platelets and endothelial cells is a frequent intermediate step in certain events such as platelet adhesion, aggregation, and regulation of vascular permeability. How this occurs in dengue infection and what the effects are on endothelial cell function are unknown. Binding of viruses to platelets can have potentially profound immunological effects [e.g., the stimulation of TGF-β release by platelets bound by Epstein–Barr virus (Ahmad and Menezes, 1997)]. In light of reports of altered platelet function in dengue patients, discussed earlier, there is a tantalizing need to determine the immunological consequences of antibody-enhanced dengue virus binding to platelets in terms of platelet as well as endothelial cell physiological responses.
Many products of complement activation can also be deposited on platelets (Devine, 1992). In view of evidence for complement activation in severe dengue disease (Halstead 1989, Malasit 1987), binding of complement products might play a role in the immune destruction of platelets leading to thrombocytopenia. Platelets display surface receptors, e.g., C1q receptor (Peerschke 1987, Peerschke 1998), membrane cofactor protein (Seya et al., 1986), and decay-accelerating factor (Devine et al., 1987), for specific components of complement activation. In addition, the platelet surface can act as a substrate for the deposition of C3dg and C5b-9 (Devine, 1992). Fragments of C3 have been detected on the platelets of DHF⧸DSS patients (Malasit, 1987).
In addition to immune complex deposition on platelets, thrombocytopenia associated with DHF⧸DSS might also arise by the immune destruction of platelets through antiplatelet autoantibodies. Antiplatelet autoantibodies have been reported in the sera of dengue patients (Lin et al., 2001), although they have also been detected in patients recovering from a variety of viral infections (Imbach, 1994). Antiplatelet antibodies are strongly linked to the pathogenesis of immune-mediated thrombocytopenias, such as idiopathic thrombocytopenic purpura (Winkelstein and Kiss, 1997).
H. Cell Targets: An Overview
While this brief discussion of cell targets for flaviviruses is by no means complete, it highlights some of the major interactions as they relate to pathogenesis. Because pathogenesis is probably best understood for dengue, Fig. 1 illustrates the interactions of hemorrhagic flavivirus (e.g., dengue) with cell targets both within and outside the vascular system.
Fig 1.
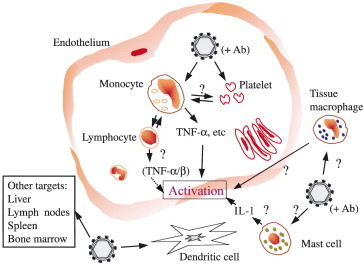
Model showing surface interactions of hemorrhagic flavivirus (dengue) with extra- and intravascular cell targets. Intravascularly, the presence of subneutralizing levels of antivirus antibodies stimulates virus attachment to platelets and infection of monocytes. This results in immune complex deposition on platelets and secretion of vasoactive factors from virus-infected monocytes. Among such vasoactive factors are cytokines, particularly TNF-α, which activates increased surface expression of adhesion molecules on endothelial cells. Extravascularly, virus infection of tissue macrophages, mast cells, and dendritic cells may result in the release of additional factors, which contribute to endothelial cell perturbation.
IV. Cell Surface Macromolecules Involved in Flavivirus Attachment
A. Glycosaminoglycans
Glycosaminoglycans and proteoglycans (i.e., proteins bearing glycosaminoglycans) are important cell surface molecules involved in a variety of ligand recognition and cell signaling processes (Gallo, 2000). Because glycosaminoglycans are widely distributed on cells, they are attractive candidates as virus receptors. Some degree of specificity (i.e., virus tropism) may arise from the compositional heterogeneity of glycosaminoglycans, as well as quantitative differences in the degree of expression on various cell types.
Flaviviruses seem to share, with a large number of virus families, the ability to bind glycosaminoglycans (Birkmann 2001, Dechecchi 2000, Dechecchi 2001, Duisit 1999, Feldman 1999, Feldman 2000, Giroglou 2001, Goodfellow 2001, Heil 2001, Hsiao 1999, Hulst 2000, Hulst 2001, Lin 2000, Liu 2002, Patel 1993, Rue 2002, Shukla 1999, Shukla 2001). Glycosaminoglycans such as heparin and its structural analogues have been investigated for their ability to bind dengue virus and thereby to gain insights as to the structural requirements for dengue receptors. Potential glycosaminoglycan-binding motifs have been identified on the dengue viral E protein at two sites, the best characterized of which appears to be composed of amino acids 188, 284–295, and 305–310 and which may also play a role in virus–cell attachment (Chen et al., 1997). Heparin (minimum of 10 carbohydrates) and an uncharacterized highly sulfated heparin sulfate isolated from bovine liver were found to show the best binding to dengue E protein (Chen et al., 1997). Attachment of dengue virus to human hepatoma cells has also been reported to be inhibited by heparin (Hilgard and Stockert, 2000). A further study involving a panel of natural and synthetic polyanionic, sulfated compounds suggested that binding of the dengue E protein required a highly sulfated (and highly charged) oligosaccharide with a minimum size of 39Å and a high degree of structural flexibility (Marks et al., 2001).
The role of glycosaminoglycans in natural (i.e., nontissue culture-adapted) strains of flaviviruses needs to be studied further. It has long been recognized that dengue virus passaged in various host cell types can give rise to virus variants with altered cell specificity (Brandt 1979, Halstead 1984a, Halstead 1984b, Halstead 1984c). Passage-dependent mutations of the dengue virus E protein at a number of different amino acid residues have been documented (Lee et al., 1997). Following passage of TBE virus in cultured BHK-21 cells, virus mutants were selected that contained more positively charged amino acids in the putative receptor-binding region of the E protein, resulting in dependence on cell surface heparan sulfate (Mandl et al., 2001). Such mutants were diminished in their neurovirulence in mice as well as in their replication in primary chicken cells and plaque formation in porcine kidney cells (Mandl et al., 2001). A large number of other viruses have also been shown to undergo loss of virulence upon adaptation to cell culture associated with heparan sulfate utilization (Bernard 2000, Byrnes 2000, Klimstra 1998, Klimstra 1999, Lee 2000, Neff 1998, Sa-Carvalho 1997).
B. CD14
CD14 and the Toll-like receptor (TLR) pattern recognition receptors are involved in the innate response to lipopolysaccharide and other microbial products (Diamond 2000, Imler 2000). A role for CD14 and TLR4 has been found for respiratory syncytial virus (RSV) (Kurt-Jones et al., 2000), suggesting that these receptors may have a broader involvement in host response than previously thought. A possible role for CD14 in dengue infection has been postulated on the basis of inhibition of dengue virus infection of human monocytes with bacterial lipopolysaccharide (Chen et al., 1999). However, this has been disputed (Bielefeldt-Ohmann et al., 2001) and requires further investigation.
C. Fc Receptors
As indicated earlier, flaviviruses are capable of initiating infection of appropriate host cells through as yet largely unidentified primary receptors. In addition, a number of flaviviruses are capable of using subneutralizing levels of virus-specific antibodies to attach to and gain entry to cells bearing Fc and⧸or complement receptors (Cardosa 1983, Halstead 1982, Halstead 1977a, Schlesinger 1981a) by a process known as antibody-dependent enhancement (ADE) of infection (Table I ). ADE has been documented for dengue (Halstead et al., 1980), West Nile (Peiris and Porterfield, 1979), yellow fever (Schlesinger and Brandriss, 1981b), tick-borne encephalitis (Phillpotts et al., 1985) and Japanese encephalitis (Cecilia and Ghosh, 1988) viruses. Early work with dengue virus and monocytes differentiated between trypsin-sensitive and trypsin-resistant cell surface molecules as the putative receptors for antibody-independent and antibody-dependent infection, respectively (Daughaday et al., 1981).
TABLE I.
FcRs for Antibody-Enhanced Infection of Dengue Virus
| Dengue virus replication | ||||
|---|---|---|---|---|
| Cell | FcγRa | Ab independent | Ab enhanced | FcγR for ADE |
| Monocyte | I,II,III | Yesb | Yesb | I, IIf |
| Dendritic cells | II | Yesc | Noc | None |
| Mast cell⧸basophil | I,II | Nod | Yesd | Unknown |
| Kupffer cell | I,II,III | Noe | Unknown | Unknown |
Compiled from van de Winkel 1991, Dijstelbloem 2001, Okayama 2000, Anselmino 1989, Tuijnman 1993.
From Wu et al. (2000) and Libraty et al. (2001).
From King et al. (2000).
Abortive infection, but expressing viral antigen (Marianneau et al., 1999).
From Littaua et al. (1990) and Kontny et al. (1988).
To date, dengue virus appears to be the only flavivirus in which strong evidence exists for antibody-dependent enhancement as a major contributing factor to severe disease (Halstead 1980, Thein 1997). Severe dengue disease, encompasing conditions known as dengue hemorrhagic fever⧸dengue shock syndrome, involves several well-defined hemostatic abnormalities, including the leakage of plasma into interstitial spaces, as well as thrombocytopenia and bleeding (Halstead 1990, Kurane 1994). The potential to cause severe hemorrhagic disease is a general property of dengue viruses and is not limited to any one viral serotype (Gubler 1998, Rigau-Perez 1998). Although different strains of dengue may influence the severity of hemorrhagic symptoms (Leitmeyer 1999, Rico-Hesse 1997), it is also generally accepted that pathogenesis depends on immunopathological processes (Rothman and Ennis, 1999). Thus the roles of prior immunity, antibody-enhanced virus infection, and immune-mediated pathologic effects on the vascular system are key points in understanding the pathogenesis of dengue hemorrhagic disease.
While the pathogenesis of severe dengue disease is not completely understood, it is clear from laboratory and epidemiological studies that a considerable risk factor is prior immunity. Severe dengue disease, DHF⧸DSS, rarely occurs in seronegative individuals suffering their first dengue infection, but instead occurs in individuals who have preexisting dengue viral antibodies, either from a previous infection or from passive antibody transfer, e.g., following maternal transmission of antibodies to the fetus (Kliks 1988, Kliks 1989). Estimates suggest that 99% of children suffering from DHF⧸DSS have preexisting immunity from a prior dengue virus infection (Halstead, 1988). Consequently, from this and other studies, it has been calculated that prior exposure to dengue increases the risk for hemorrhagic disease in a second dengue infection by at least 15-fold (Halstead 1980, Thein 1997). Preexisting serum antibodies can potentiate virus infection by the mechanism of antibody-dependent enhancement, giving rise to amplified virus replication and to an increased potential for the development of hemorrhagic symptoms (Halstead, 1989). Viremic titers are higher in secondary dengue infections in both humans (Gubler et al., 1979) and experimental monkeys (Halstead et al., 1973). Antibody-enhanced dengue virus infection of human blood monocytes is necessary for the production of endothelial cell activators (Anderson et al., 1997), thereby providing a link between antibody-dependent enhancement and alteration of endothelial cell properties, which might contribute to vascular permeability in dengue infection.
For certain other viruses, e.g., influenza (Tamura et al., 1993) and HIV (Takeda 1990, Takeda 1992), distinct “neutralizing” and “antibody-enhancing” epitopes have been identified on the respective viral attachment proteins. Surprisingly, no systematic approach has yet been undertaken to identify regions on the E protein that are essential for ADE, even though this issue was raised as a challenge to research on dengue many years ago (Halstead, 1988).
Human Fcγ receptors are currently categorized into three classes: FcγRI (CD64), FcγRII (CD32), and FcγRIII (CD16). While FcγRI shows high affinity for monomeric IgG, FcγRII and FcγRIII bind monomeric IgG poorly and are more likely involved in binding immune complexes (Dijstelbloem et al., 2001). FcγRII is the most widely distributed, being expressed on most circulating leukocytes (van de Winkel and Anderson, 1991). Monocytes express all three FcγRs to varying degrees (van de Winkel and Anderson, 1991), although FcγRI and FcγRII predominate, whereas FcγRIII appears to be limited to a subpopulation (∼10%) of monocytes (Anderson 1990, Passlick 1989). FcγRIII constitutes the major FcγR on macrophages (Fanger et al., 1989), although FcγRI and FcγRII are also present (Tuijnman 1993, van de Winkel 1991). It is also important to recognize that FcR expression on cells, including macrophages, can vary depending on the microenvironment (Tomita et al., 1994).
Although strong evidence exists for FcγR involvement in ADE of dengue virus, the participating FcγRs in vivo have not yet been identified rigorously. In cultured cell lines (monocytic U937 or erythroleukemic K562 cells), FcγRI (Kontny et al., 1988) and FcγRII (Littaua et al., 1990) have been shown to mediate ADE of dengue virus infection. That FcγRI has the ability to mediate ADE of dengue has been demonstrated using COS cells transfected with FcγRI (Schlesinger and Chapman, 1999).
Dengue and DHF patients show elevated serum levels of interferon (IFN)-γ (Kurane et al., 1991). Because IFN-γ can upregulate both MHC class I and II molecules as well as FcγR (particularly FcγRI) expression in monocytes (Erbe 1990, Perussia 1983), the chances for ADE may be increased, thereby creating a vicious cycle involving positive cytokine feedback and virus amplification (Kurane and Ennis, 1992). IFN-γ has been shown to enhance ADE of dengue virus infection of human monocytic U937 cells (Kontny et al., 1988), although any enhancing effect on dengue infection of peripheral blood monocytes may be negated by the antiviral properties of IFN-γ (Sittisombut et al., 1995).
Mast cells and basophils express mainly FcγRII (Anselmino 1989, Okayama 2001a, Wedi 1996) and some (IFN-γ–inducible) FcγRI (Okayama 2000, Okayama 2001b) as well as the high-affinity FcϵRI for IgE (Guo 1992, Sperr 1994). As noted previously, the basophil⧸mast cell KU812 cell line exhibits antibody-enhanced dengue virus infection and produces vasoactive cytokines (King et al., 2000).
Although FcγR-mediated ADE of flaviviruses has been examined extensively as a mechanism for virus amplification, the biological consequences for the participating host cell are not well understood. Because FcγR-mediated cell signaling is complex, the functional effects of virus–antibody interactions with cell surface FcγRs need to be investigated. Monocytes infected with dengue virus in the presence of antibody release cytokines such as TNF-α (Anderson et al., 1997). Induction of TNF-α requires infectious virus (Anderson et al., 1997), suggesting that virus replication (or perhaps expression of one or more crucial viral genes) is responsible for the stimulation of TNF-α release. Therefore, in this case, the FcγR is likely facilitating antibody-enhanced virus replication rather than providing a signal triggered by virus binding to the FcγR. Similarly, antibody-enhanced dengue virus infection of KU812 basophil⧸mast cells produces IL-1β, IL-6 (King et al., 2000, 2002), and selected chemokines (King et al., 2002). Suppressive effects of antibody-enhanced flavivirus or alphavirus infection on monocyte cytokine secretion have also been reported (Lidbury 2000, Yang 2001).
Both activating (FcγRI, FcγRIIa, and FcγRIIIa) and inhibitory (FcγRIIb) forms of FcγRs exist, which mediate signal transduction via a cytoplasmic immunoreceptor tyrosine-based activation motif (ITAM) or inhibitory (ITIM) motif, respectively (Dijstelbloem et al., 2001). The ITAM and associated molecules are necessary for the endocytosis of FcR-bound immune complexes (Amigorena and Bonnerot, 1999) and therefore play a likely role in the initiating events of antibody-enhanced flavivirus infection. Although not necessary for FcγRII, an accessory subunit (homo- or heterodimeric γ or ζ chains) is required for signaling through FcγRI and FcγRIIIa (Ravetch, 1994). A further FcγR (FcγRIIIb) lacks transmembrane and cytoplasmic domains and is instead anchored to the cell surface membrane via a glycosylphosphatidylinositol (GPI) linkage (Selvaraj 1988, Simmons 1988). It apparently does not participate in signal transduction and has been speculated to sequester and accumulate immune complexes at specific sites on the cell surface (Huizinga 1988, Selvaraj 1988).
The roles of activating and inhibitory FcRs in viral ADE have not yet been ascertained. Activating FcRs are expressed on monocytes, macrophages, granulocytes, natural killer (NK) cells, and platelets but not on most lymphocytes (Dijstelbloem et al., 2001). Inhibitory FcRs, however, are found on B cells, dendritic cells, and macrophages (Dijstelbloem et al., 2001). Interestingly, ADE of dengue virus is best documented for monocytes⧸macrophages and related cell lines (Halstead, 1989). In contrast, lymphocytic cells (Brandt 1979, Kurane 1990) and dendritic cells (Wu et al., 2000) do not appear to support antibody-enhanced dengue virus infection. Whether this is due to differential expression of activating versus inhibitory FcRs remains to be investigated.
FcRs for IgE (primarily the high-affinity FcϵRI) are expressed on cells such as monocytes, macrophages, mast cells, basophils, and dendritic cells and are structurally related to FcγRs (Ravetch, 1994). Their role in binding IgE and⧸or immune-complexed flaviviruses, such as dengue, remains unexplored. Similarly unexplored is the potential role of the neonatal Fc IgG receptor (FcRn), structurally related to MHC class I and involved in IgG transport across cells (Ghetie and Ward, 2000). In addition to being expressed on certain epithelial and endothelial cells, FcRn is also expressed functionally on monocytes, macrophages, and dendritic cells (Zhu et al., 2001).
D. Complement Receptors
In addition to the FcγR, the antibody-complexed flavivirus has been shown to be taken up by a macrophage cell line using the complement receptor-3 (Cardosa et al., 1983). In the case studied—West Nile virus infection of mouse P388D1 macrophages—ADE was mediated by the presence of antiviral IgM and was inhibited with a CR3-blocking antibody. This mode of ADE was, however, found to be quantitatively less productive than the more commonly studied route of ADE, i.e., involving FcγR-mediated uptake route of IgG–virus complexes (Cardosa et al., 1983).
E. Virus Binding Proteins Identified on Cells
The recent demonstration of DC-SIGN as a functional dengue virus receptor on human dendritic cells represents an important advance in the definitive identification of flavivirus receptors (Navarro_Sanchez 2003, Tassaneetrithep 2003). Several studies have identified cell surface proteins that bind flaviviruses, generally assayed by virus overlay blots of SDS–PAGE-resolved cell proteins (Table II ). Further work is required to confirm the involvement of these and other proteins as receptors in flavivirus infection.
TABLE II.
Flavivirus Binding Proteins on Cells
| Cell | Virus | Binding protein(s) | Reference |
|---|---|---|---|
| Human erythroleukemic K562 cells | Dengue-2 | 100 kDa | Rothwell et al. (1996) |
| Human and mouse neuroblastoma cells | Dengue-2 | 65 kDa | Ramos-Castaneda et al. (1997) |
| Human monocytic, B and T cell lines | Dengue-2 | 32, 45, 72 kDa | Bielefeldt-Ohmann 1998, Bielefeldt-Ohmann 2001 |
| Monkey kidney Vero cells | Dengue-4 | 44, 74 kDa | Martinez-Barragan and del Angel (2001) |
| Mosquito C6⧸36 cells | Dengue-4 | 40, 45 kDa | Salas-Benito and del Angel (1997) |
| Mosquito C6⧸36 cells | Dengue-2 | 65, 80 kDa | Munoz et al. (1998) |
| Human hepatoma HuH-7 cells | Dengue-1 | 33- and 37-kDa proteoglycans | Hilgard and Stockert (2000) |
| Pig kidney PS cells | TBE | 35 kDa | Kopecky et al. (1999) |
| Human dendritic cells | Dengue | DC-SIGN | Hilgard 2000, Navarro_Sanchez 2003, Navarro_Sanchez 2003 |
| Vero cells; mouse neuroblastoma cells | West Nile | 105 kDa | Kopecky 1999, Chu 2003 |
V. Cell Surface Macromolecules Modulated by Flavivirus Infection
A number of flaviviruses are able to stimulate the expression of cell surface molecules. Notable among these are adhesion molecules and major histocompatibility antigens. Multiple mechanisms appear to be involved, including virus- and cytokine-dependent pathways.
A. MHC Class I
Flavivirus infection of a number of cell types causes an increase in cell surface MHC class I expression (King 1988, King 1989, Libraty 2001, Liu 1989, Lobigs 1996, Shen 1995a, Shen 1997). Evidence for both virus-dependent (Lobigs et al., 1996) and cytokine-dependent (Libraty 2001, Shen 1997) mechanisms has been reported. One process appears to be driven by the amount of flaviviral peptides generated by proteolysis and imported into the transporter associated with antigen processing (TAP), which results in increased cell surface expression of peptide-loaded MHC class I (Momburg et al., 2001). The upregulation of MHC class I molecules by flaviviruses is perhaps reminiscent of that observed in infections by coronaviruses (Suzumura et al., 1986) but stands in contrast to the virus-manipulated downregulation of MHC class I by viruses such as herpesviruses (Jennings 1985, Ploegh 1998), adenoviruses (Sparer and Gooding, 1998), poxviruses (Boshkov et al., 1992), and HIV (Scheppler et al., 1989). Although enhanced MHC class I expression would be expected to lead to greater cytotoxic T (Tc) cell–mediated cytolysis, it would render cells less susceptible to recognition by NK cells. Evidence has been presented that flavivirus-infected cells in fact show reduced susceptibility to NK cells at the cost of enhanced Tc cell–mediated lysis (Lobigs et al., 1996). It has been suggested that such a response may permit flaviviruses to evade an early NK cell response and thereby allow for substantial amplification of virus during the viremic phase of infection (Momburg et al., 2001). Nevertheless, evidence shows that NK cells are activated during dengue infection (Green et al., 1999a), and NK cell–mediated cytotoxicity has been reported to correlate with the severity of disease (Homchampa et al., 1988).
Dendritic cells also undergo upregulation of MHC class I molecules following infection with dengue virus (Libraty et al., 2001). Compared to other antigen-presenting cells, dendritic cells have superior T cell–stimulating activities (McKinney 1989, Timares 1998). Because antigen presentation via dendritic cell MHC class I can provoke exceptionally strong proliferation in CD8-bearing T cells (Bhardwaj 1994, Elbe 1994, McKinney 1989), much of the overall cytotoxic T cell response arising in flavivirus infection may be dictated at the level of the dendritic cell.
B. MHC Class II
West Nile virus infection induces MHC class II expression in mouse macrophages (Shen et al., 1995a), mouse astrocytes (Liu et al., 1989), rat Schwann cells (Argall et al., 1991), and human myoblasts (Bao et al., 1992). Upregulation of dendritic cell MHC class II occurs in response to dengue (Libraty et al., 2001) and West Nile (Johnston et al., 1996) virus infection. Given the potent ability of dendritic cells to activate T cells (Banchereau et al., 2000), the communication between dendritic cell MHC class II–peptide complexes and recognition molecules on CD4-expressing T cells should provide insights into some of the molecular processes underlying T cell activation.
C. Adhesion Molecules
Adhesion molecules are expressed on a variety of cells and mediate a spectrum of processes (Ley 2001, Roebuck 1999, Springer 1995). From the standpoint of flaviviruses, the most significant processes likely concern adhesion molecules on vascular endothelial cells, as these cells regulate permeability as well as transendothelial migration of leukocytes (Springer, 1995). Of particular importance are intercellular adhesion molecule 1 (ICAM-1; CD54), vascular cell adhesion molecule-1 (VCAM-1; CD106), and E-selectin (CD 62E), which are upregulated on the surface of the endothelium by inflammatory cytokines, cellular stress, and virus infection (Roebuck and Finnegan, 1999).
In the case of dengue, activation of endothelial cells occurs in vitro via TNF-α released from antibody-enhanced dengue virus infection of monocytes (Anderson et al., 1997). Such activation involves upregulation of adhesion molecules E-selectin, ICAM-1, and VCAM-1. Evidence that similar activation processes occur in vivo comes from clinical studies showing elevated serum levels of TNF-α (Green 1999b, Hober 1993, Vitarana 1991, Yadav 1991) and soluble VCAM-1 (Murgue et al., 2001) in dengue- and DHF⧸DSS-infected patients. Surprisingly, serum levels of soluble ICAM-1 were actually found to be lower than those of control subjects, although this may reflect plasma protein loss through leakage (Bethell et al., 1998). Moreover, the function of soluble forms of ICAM-1 remains unclear, and their expression appeazrs to be regulated differently from that of membrane-bound ICAM-1 (Komatsu 1997, van Den Engel 2000).
Two phases of ICAM-1 upregulation have been noted in West Nile and Kunjin virus infection of human embryonic fibroblasts, namely an early (∼2 h postinfection) virus-dependent process and a later (∼24 h postinfection) event that is mediated by type 1 interferon (Shen et al., 1995b).
For neurotropic flaviviruses, such as West Nile virus in the mouse, the development of encephalitis has been correlated with viremia (Weiner et al., 1970), suggesting virus penetration of the blood–brain barrier. The endothelium of the brain microvasculature normally represents a block between circulating virus and the central nervous system. Expression of endothelial cell adhesion molecules, thereby facilitating leukocyte adherence and diapedesis through the endothelium, may be an important mode of dissemination of virus-infected monocytes or other leukocytes into the brain. West Nile virus infection of human endothelial cells causes the upregulation of E-selectin, ICAM-1, and VCAM-1 (Shen et al., 1997), which could mediate the transendothelial migration of leukocytes. Upregulation of these adhesion molecules was observed to occur early (2–4 h) in infection and appeared to be triggered by the virus rather than by cytokines (Shen et al., 1997).
Further studies are required to clarify the role of endothelial cell adhesion molecule expression in the neuroinvasion of certain flaviviruses. Assuming such a role is confirmed, it will be incumbent to identify the mechanisms by which either free or cell-borne flaviviruses are stimulated to cross the vascular endothelial layer. For virus-infected leukocytes, such stimulation likely arises, at least in part, from chemokines produced by cells of the central nervous system. Astrocytes infected with JE virus have been reported to release chemokines (RANTES and MCP-1), which may play a role in the transendothelial migration of leukocytes (including those possibly carrying virus) across the blood–brain barrier (Chen et al., 2000). Thus, once neural infection is initiated, the process could be amplified by the production of leukocyte-attracting chemokines at the site of infection.
VI. Other Cell Surface Macromolecular Modifications Triggered by Flavivirus Infection
A. Complement Deposition
Complement activation is well documented in dengue disease (Nishioka 1974, Phanichyakarn 1977b, Russell 1969), with peak activation and the production of C3a and C5a occurring at the time of vascular leakage and⧸or shock (Malasit, 1987). Complement activation is likely to be largely mediated by immune complexes consisting of IgG and virus (Bokisch 1973a, Bokisch 1973b, Shaio 1992, Sobel 1975), although the low levels of circulating immune complexes detected in patients have stimulated thought as to other possible mechanisms (Malasit, 1987). Receptors for C3a and C5a are found on a wide variety of cells, including many human peripheral blood leukocytes (Chenoweth 1978, Fureder 1995, Kretzschmar 1993, Nilsson 1996, van Epps 1984). C5a receptors have been reported on endothelial cells, although at lower levels than myeloid cells (Zwirner et al., 1999).
Although endothelial cells do not appear to be major targets for dengue virus in vivo (Halstead 1988, Halstead 1989, Sahaphong 1980), endothelial cells infected with dengue virus in vitro can become a substrate for deposition of C3dg and C5b-9, provided the dengue antibody is present (Avirutnan et al., 1998). The presence of complement activation products on the endothelial cell surface could be a contributing factor to vascular permeability (Saadi et al., 1995). Furthermore, anaphylotoxins and⧸or deposition of sublytic C5b-9 on the endothelial cell surface has the potential to activate the expression of adhesion molecules (Foreman et al., 1994), cytokines (Saadi et al., 2000), chemokines (Selvan et al., 1998), cyclooxygenase-2 (Bustos et al., 1997), tissue factor (Saadi and Platt 1995), heparan sulfate proteoglycan proteinases (Ihrcke and Platt, 1996), and even functional or morphological changes such as permeability loss and gap formation (Saadi et al., 1995).
Thus, in addition to being activated by leukocyte-derived cytokines (Anderson et al., 1997), endothelial cells may also be coaxed toward a more permeability-enhancing state by virus infection and virus-mediated complement deposition. At present, the lack of evidence for in vivo infection of endothelial cells by dengue virus would suggest that the cytokine-mediated pathway is dominant. Figure 2 shows a model illustrating the potential role of endothelial cell perturbation by monocyte-derived cytokines and complement activation products in initiating vascular permeability and leukocyte extravasation in severe hemorrhagic flavivirus disease.
Fig 2.
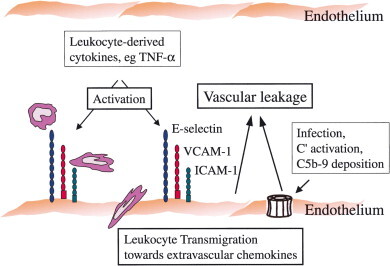
Model depicting possible events in endothelial cell surface perturbation during hemorrhagic flavivirus (dengue) infection. Endothelial cell activation, leading to upregulation of adhesion molecules (E-selectin, VCAM-1, ICAM-1), can be triggered by monocyte-derived cytokines (Anderson et al., 1997) or by deposition of C5b-9 and other products of complement activation (Avirutnan et al., 1998). C5b-9 is represented as a membrane attack complex pore structure, although the deposition of C5b-9 on dengue-infected cells appears associated with sublytic, rather than lytic, responses (Avirutnan et al., 1998). Increased adhesion molecule expression, along with uncharacterized vasoactive factors, can lead to endothelial leakage and can mediate rolling, adhesion, and transendothelial migration of leukocytes into extravascular tissues. Similar processes may also contribute to the invasion of cell-borne neurotropic flaviviruses through the endothelial blood–brain barrier.
VII. Conclusions
Much remains to be learned about the primary receptors for flaviviruses, though much knowledge has been gained about the initial interactions of flaviviruses with cell surface structures. The ability of flaviviruses to affect cell entry through heparan sulfate–type proteoglycans, as well as their dexterity to adjust mutationally to different receptors, depending on host cell type, illustrates the plasticity of the viral E protein to adapt to changing conditions and to ensure successful virus replication. Beyond this, certain flaviviruses, notably dengue virus, are masters at exploiting host antibody and Fc receptor–bearing cells to dramatically amplify viral replication. Flavivirus replication is coupled to altered cellular expression of cytokines, chemokines, and cell surface molecules, which shape the host response and immunopathogenesis associated with flavivirus infections. Ongoing and future characterization of the cell surface structures that mediate these events will be helpful in understanding the mechanisms of flavivirus-induced disease and in developing therapeutic and⧸or preventive strategies.
Acknowledgements
Research in the author's laboratory was supported by the Canadian Institutes of Health Research and the Natural Sciences and Engineering Research Council of Canada. The author is grateful for the collaboration and advice of many past and present colleagues, including Y. Huang, C. King, A. Issekutz, J. Marshall, C. Osiowy, R. He, S. Wang, B. Innis, A. King, D. Vaughn, K. S. Myint, T. Endy, and M. Mammen.
References
References
- Adelman B., Sobel M., Fujimura Y., Ruggeri Z.M., Zimmerman T.S. Heparin-associated thrombocytopenia: Observations on the mechanism of platelet aggregation. J. Lab. Clin. Med. 1989;113:204–210. [PubMed] [Google Scholar]
- Ahmad A., Menezes J. Binding of the Epstein–Barr virus to human platelets causes the release of transforming growth factor-beta. J. Immunol. 1997;159:3984–3988. [PubMed] [Google Scholar]
- Albrecht P. Pathogenesis of neurotropic arbovirus infections. Curr. Top. Microbiol. Immunol. 1968;43:44–91. doi: 10.1007/978-3-642-46118-7_2. [DOI] [PubMed] [Google Scholar]
- Allison S.L., Schalich J., Stiasny K., Mandl C.W., Kunz C., Heinz F.X. Oligomeric rearrangement of tick-borne encephalitis virus envelope proteins induced by an acidic pH. J. Virol. 1995;69:695–700. doi: 10.1128/jvi.69.2.695-700.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alving K. Airways vasodilation in the immediate allergic reaction: Involvement of inflammatory mediators and sensory nerves. Acta Physiol. Scand. Suppl. 1991;597:1–64. [PubMed] [Google Scholar]
- Amigorena S., Bonnerot C. Fc receptor signaling and trafficking: A connection for antigen processing. Immunol. Rev. 1999;172:279–284. doi: 10.1111/j.1600-065x.1999.tb01372.x. [DOI] [PubMed] [Google Scholar]
- Anderson C.L., Looney R.J., Culp D.J., Ryan D.H., Fleit H.B., Utell M.J., Frampton M.W., Manganiello P.D., Guyre P.M. Alveolar and peritoneal macrophages bear three distinct classes of Fc receptors for IgG. J. Immunol. 1990;145:196–201. [PubMed] [Google Scholar]
- Anderson R., King A.D., Innis B.L. Correlation of E protein binding with cell susceptibility to dengue 4 virus infection. J. Gen. Virol. 1992;73:2155–2159. doi: 10.1099/0022-1317-73-8-2155. [DOI] [PubMed] [Google Scholar]
- Anderson R., Wang S., Osiowy C., Issekutz A.C. Activation of endothelial cells via antibody-enhanced dengue virus infection of peripheral blood monocytes. J. Virol. 1997;71:4226–4232. doi: 10.1128/jvi.71.6.4226-4232.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews B.S., Theofilopoulos A.N., Peters C.J., Loskutoff D.J., Brandt W.E., Dixon F.J. Replication of dengue and Junin viruses in cultured rabbit and human endothelial cells. Infect. Immun. 1978;20:776–781. doi: 10.1128/iai.20.3.776-781.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anselmino L.M., Perussia B., Thomas L.L. Human basophils selectively express the Fc gamma RII (CDw32) subtype of IgG receptor. J. Allergy Clin. Immunol. 1989;84:907–914. doi: 10.1016/0091-6749(89)90388-6. [DOI] [PubMed] [Google Scholar]
- Anton F., Morales C., Aguilar R., Bellido C., Aguilar E., Gaytan F. A comparative study of mast cells and eosinophil leukocytes in the mammalian testis. Zentralbl. Veterinarmed. A. 1998;45:209–218. doi: 10.1111/j.1439-0442.1998.tb00819.x. [DOI] [PubMed] [Google Scholar]
- Argall K.G., Armati P.J., King N.J., Douglas M.W. The effects of West Nile virus on major histocompatibility complex class I and II molecule expression by Lewis rat Schwann cells in vitro. J. Neuroimmunol. 1991;35:273–284. doi: 10.1016/0165-5728(91)90181-6. [DOI] [PubMed] [Google Scholar]
- Aster R.H. In: Platelet Immunobiology, Molecular and Clinical Aspects. Kunicki T.J., George G.N., editors. Lippincott; Philadelphia: 1989. p. 387. [Google Scholar]
- Avirutnan P., Malasit P., Seliger B., Bhakdi S., Husmann M. Dengue virus infection of human endothelial cells leads to chemokine production, complement activation, and apoptosis. J. Immunol. 1998;161:6338–6346. [PubMed] [Google Scholar]
- Banchereau J., Briere F., Caux C., Davoust J., Lebecque S., Liu Y.J., Pulendran B., Palucka K. Immunobiology of dendritic cells. Annu. Rev. Immunol. 2000;18:767–811. doi: 10.1146/annurev.immunol.18.1.767. [DOI] [PubMed] [Google Scholar]
- Bao S., King N.J.C., Dos Remedios C.G. Flavivirus induces MHC antigen on human myoblasts: A model of autoimmune myositis. Muscle Nerve. 1992;15:1271–1277. doi: 10.1002/mus.880151109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayer W.L., Sherman F.E., Michaels R.H., Szeto I.L.F., Lewis J.H. Purpura in congenital and acquired rubella. N. Engl. J. Med. 1965;273:1362–1366. doi: 10.1056/NEJM196512162732504. [DOI] [PubMed] [Google Scholar]
- Ben-Nathan D., Kobiler D., Rzotkiewicz S., Lustig S., Katz Y. CNS penetration by noninvasive viruses following inhalational anesthetics. Ann N. Y. Acad. Sci. 2000;917:944–950. doi: 10.1111/j.1749-6632.2000.tb05460.x. [DOI] [PubMed] [Google Scholar]
- Benyon R.C., Bissonnette E.Y., Befus A.D. Tumor necrosis factor-alpha dependent cytotoxicity of human skin mast cells is enhanced by anti-IgE antibodies. J. Immunol. 1991;147:2253–2258. [PubMed] [Google Scholar]
- Bernard K.A., Klimstra W.B., Johnston R.E. Mutations in the E2 glycoprotein of Venezuelan equine encephalitis virus confer heparan sulfate interaction, low morbidity, and rapid clearance from blood of mice. Virology. 2000;276:93–103. doi: 10.1006/viro.2000.0546. [DOI] [PubMed] [Google Scholar]
- Berndt M.C., Chong B.H., Bull H.A., Zola H., Castaldi P.A. Molecular characterization of quinine⧸quinidine drug-dependent antibody platelet interaction using monoclonal antibodies. Blood. 1985;66:1292–1301. [PubMed] [Google Scholar]
- Bethell D.B., Flobbe K., Cao X.T., Day N.P., Pham T.P., Buurman W.A., Cardosa M.J., White N.J., Kwiatkowski D. Pathophysiologic and prognostic role of cytokines in dengue hemorrhagic fever. J. Infect. Dis. 1998;177:778–782. doi: 10.1086/517807. [DOI] [PubMed] [Google Scholar]
- Bevilacqua M.P. Endothelial-leukocyte adhesion molecules. Annu. Rev. Immunol. 1993;11:767–804. doi: 10.1146/annurev.iy.11.040193.004003. [DOI] [PubMed] [Google Scholar]
- Bhamarapravati N., Tuchinda P., Boonyapaknavik V. Pathology of Thailand haemorrhagic fever: A study of 100 autopsy cases. Ann Trop. Med. Parasitol. 1967;61:500–510. doi: 10.1080/00034983.1967.11686519. [DOI] [PubMed] [Google Scholar]
- Bhardwaj N., Bender A., Gonzalez N., Bui L.K., Garrett M.C., Steinman R.M. Influenza virus-infected dendritic cells stimulate strong proliferative and cytolytic responses from human CD8+ T cells. J. Clin. Invest. 1994;94:797–807. doi: 10.1172/JCI117399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bielefeldt-Ohmann H. Analysis of antibody-independent binding of dengue viruses and dengue virus envelope protein to human myelomonocytic cells and B lymphocytes. Virus Res. 1998;57:63–79. doi: 10.1016/s0168-1702(98)00087-2. [DOI] [PubMed] [Google Scholar]
- Bielefeldt-Ohmann H., Meyer M., Fitzpatrick D.R., Mackenzie J.S. Dengue virus binding to human leukocyte cell lines: Receptor usage differs between cell types and virus strains. Virus Res. 2001;73:81–89. doi: 10.1016/s0168-1702(00)00233-1. [DOI] [PubMed] [Google Scholar]
- Bik T., Sarov I., Livne A. Interaction between vaccinia virus and human blood platelets. Blood. 1982;59:482–487. [PubMed] [Google Scholar]
- Birkmann A., Mahr K., Ensser A., Yaguboglu S., Titgemeyer F., Fleckenstein B., Neipel F. Cell surface heparan sulfate is a receptor for human herpesvirus 8 and interacts with envelope glycoprotein K8.1. J. Virol. 2001;75:11583–11593. doi: 10.1128/JVI.75.23.11583-11593.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bokisch V.A., Muller-Eberhard H.J., Dixon F.J. The role of complement in hemorrhagic shock syndrome (dengue) Trans. Assoc. Am. Physicians. 1973;86:102–110. [PubMed] [Google Scholar]
- Bokisch V.A., Top F.H.J., Russell P.K., Dixon F.J., Muller-Eberhard H.J. The potential pathogenic role of complement in dengue hemorrhagic shock syndrome. N. Engl. J. Med. 1973;289:996–1000. doi: 10.1056/NEJM197311082891902. [DOI] [PubMed] [Google Scholar]
- Boonpucknavig S., Vuttiviroj O., Bunnag C., Bhamarapravati N., Nimmanitya S. Demonstration of dengue antibody complexes on the surface of platelets from patients with dengue hemorrhagic fever. Am. J. Trop. Med. Hyg. 1979;28:881–884. [PubMed] [Google Scholar]
- Boshkov L.K., Macen J.L., McFadden G. Virus-induced loss of class I MHC antigens from the surface of cells infected with myxoma virus and malignant rabbit fibroma virus. J. Immunol. 1992;148:881–887. [PubMed] [Google Scholar]
- Bradding P., Feather I.H., Wilson S., Bardin P.G., Heusser C.H., Holgate S.T., Howarth P.H. Immunolocalization of cytokines in the nasal mucosa of normal and perennial rhinitic subjects: The mast cell as a source of IL-4, IL-5, and IL-6 in human allergic mucosal inflammation. J. Immunol. 1993;151:3853–3865. [PubMed] [Google Scholar]
- Brandt W.E., McCown J.M., Top F.H., Bancroft W.H., Russell P.K. Effect of passage history on dengue-2 virus replication in subpopulations of human leukocytes. Infect. Immun. 1979;26:534–541. doi: 10.1128/iai.26.2.534-541.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bulger E.M., Maier R.V. Lipid mediators in the pathophysiology of critical illness. Crit. Care Med. 2000;28:N27–N36. doi: 10.1097/00003246-200004001-00004. [DOI] [PubMed] [Google Scholar]
- Bustos M., Coffman T.M., Saadi S., Platt J.L. Modulation of eicosanoid metabolism in endothelial cells in a xenograft model: Role of cyclooxygenase-2. J. Clin. Invest. 1997;100:1150–1158. doi: 10.1172/JCI119626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Byrnes A.P., Griffin D.E. Large-plaque mutants of Sindbis virus show reduced binding to heparan sulfate, heightened viremia, and slower clearance from the circulation. J. Virol. 2000;74:644–651. doi: 10.1128/jvi.74.2.644-651.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cardosa M.J., Porterfield J.S., Gordon S. Complement receptor mediates enhanced flavivirus replication in macrophages. J. Exp. Med. 1983;158:258–263. doi: 10.1084/jem.158.1.258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cecilia D., Ghosh S.N. Antibody dependent plaque enhancement by monoclonal antibodies against Japanese encephalitis virus. Indian J. Med. Res. 1988;87:521–525. [PubMed] [Google Scholar]
- Chang D.-M., Shaio M.-F. Production of interleukin-1 (IL-1) and IL-1 inhibitor by human monocytes exposed to dengue virus. J. Infect. Dis. 1994;170:811–817. doi: 10.1093/infdis/170.4.811. [DOI] [PubMed] [Google Scholar]
- Chaturvedi U.C., Dhawan R., Khanna M., Mathur A. Breakdown of the blood-brain barrier during dengue virus infection of mice. J. Gen. Virol. 1991;72:859–866. doi: 10.1099/0022-1317-72-4-859. [DOI] [PubMed] [Google Scholar]
- Chen C.J., Liao S.L., Kuo M.D., Wang Y.M. Astrocytic alteration induced by Japanese encephalitis virus infection. Neuroreport. 2000;11:1933–1937. doi: 10.1097/00001756-200006260-00025. [DOI] [PubMed] [Google Scholar]
- Chen Y., Maguire T., Hileman R.E., Fromm J.R., Esko J.D., Linhardt R.J., Marks R.M. Dengue virus infectivity depends on envelope protein binding to target cell heparan sulfate. Nature Med. 1997;3:866–871. doi: 10.1038/nm0897-866. [DOI] [PubMed] [Google Scholar]
- Chen Y., Maguire T., Marks R.M. Demonstration of binding of dengue virus envelope protein to target cells. J. Virol. 1996;70:8765–8772. doi: 10.1128/jvi.70.12.8765-8772.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y.C., Wang S.Y., King C.C. Bacterial lipopolysaccharide inhibits dengue virus infection of primary human monocytes⧸macrophages by blockade of virus entry via a CD14-dependent mechanism. J. Virol. 1999;73:2650–2657. doi: 10.1128/jvi.73.4.2650-2657.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chenoweth D.E., Hugli T.E. Demonstration of specific C5a receptor on intact human polymorphonuclear leukocytes. Proc. Natl. Acad. Sci. USA. 1978;75:3943–3947. doi: 10.1073/pnas.75.8.3943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chong B.H., Berndt M.C., Koutts J., Castaldi P.A. Quinidine-induced thrombocytopenia and leukopenia: Demonstration and characterization of distinct antiplatelet and antileukocyte antibodies. Blood. 1983;62:1218–1223. [PubMed] [Google Scholar]
- Chong B.H., Castaldi P.A., Berndt M.C. Heparin-induced thrombocytopenia: Effects of rabbit IgG, and its Fab and FC fragments on antibody-heparin-platelet interaction. Thromb. Res. 1989;55:291–295. doi: 10.1016/0049-3848(89)90447-7. [DOI] [PubMed] [Google Scholar]
- Chong B.H., Fawaz I., Chesterman C.N., Berndt M.C. Heparin-induced thrombocytopenia: Mechanism of interaction of the heparin-dependent antibody with platelets. Br. J. Haematol. 1989;73:235–240. doi: 10.1111/j.1365-2141.1989.tb00258.x. [DOI] [PubMed] [Google Scholar]
- Christie D.J., Mullen P.C., Aster R.H. Quinine- and quinidine platelet antibodies can react with GPIIb⧸IIIa. Br. J. Haematol. 1987;67:213–219. doi: 10.1111/j.1365-2141.1987.tb02329.x. [DOI] [PubMed] [Google Scholar]
- Chu C.T., Howell D.N., Morgenlander J.C., Hulette C.M., McLendon R.E., Miller S.E. Electron microscopic diagnosis of human flavivirus encephalitis: Use of confocal microscopy as an aid. Am. J. Surg. Pathol. 1999;23:1217–1226. doi: 10.1097/00000478-199910000-00006. [DOI] [PubMed] [Google Scholar]
- Chu J.J., Ng M.L. Characterization of a 105-kDa plasma membrane associated glycoprotein that is involved in West Nile Virus binding and infection. Virology. 2003;312:458–469. doi: 10.1016/s0042-6822(03)00261-7. [DOI] [PubMed] [Google Scholar]
- Crill W.D., Roehrig J.T. Monoclonal antibodies that bind to domain III of dengue virus E glycoprotein are the most efficient blockers of virus adsorption to Vero cells. J. Virol. 2001;75:7769–7773. doi: 10.1128/JVI.75.16.7769-7773.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crockett T.E. Selectins and mechanisms of signal transduction. J. Leukocyte Biol. 1998;63:1–14. [PubMed] [Google Scholar]
- Danon D., Jerushalmy Z., DeVries A. Incorporation of influenza virus in human blood platelets in vitro: Electron microscopical observation. Virology. 1959;9:719–722. doi: 10.1016/0042-6822(59)90168-0. [DOI] [PubMed] [Google Scholar]
- Daughaday C.C., Brandt W.E., McCown J.M., Russell P.K. Evidence for two mechanisms of dengue virus infection of adherent human monocytes: Trypsin-sensitive virus receptors and trypsin-resistant immune complex receptors. Infect. Immun. 1981;32:469–473. doi: 10.1128/iai.32.2.469-473.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dechecchi M.C., Melotti P., Bonizzato A., Santacatterina M., Chilosi M., Cabrini G. Heparan sulfate glycosaminoglycans are receptors sufficient to mediate the initial binding of adenovirus types 2 and 5. J. Virol. 2001;75:8772–8780. doi: 10.1128/JVI.75.18.8772-8780.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dechecchi M.C., Tamanini A., Bonizzato A., Cabrini G. Heparan sulfate glycosaminoglycans are involved in adenovirus type 5 and 2-host cell interactions. Virology. 2000;268:382–390. doi: 10.1006/viro.1999.0171. [DOI] [PubMed] [Google Scholar]
- Devine D.V. The effects of complement activation on platelets. Curr. Top. Microbiol. Immunol. 1992;178:101–113. doi: 10.1007/978-3-642-77014-2_7. [DOI] [PubMed] [Google Scholar]
- Devine D.V., Rosse W.F. Identification of platelet proteins that bind alloantibodies and autoantibodies. Blood. 1995;64:1240–1245. [PubMed] [Google Scholar]
- Devine D.V., Siegel R.S., Rosse W.F. Interactions of the platelets in paroxysmal nocturnal hemoglobinuria with complement: Relationship to defects in the regulation of complement and to platelet survival in vivo. J. Clin. Invest. 1987;79:131–137. doi: 10.1172/JCI112773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diamond G., Legarda D., Ryan L.K. The innate immune response of the respiratory epithelium. Immunol. Rev. 2000;173:27–38. doi: 10.1034/j.1600-065x.2000.917304.x. [DOI] [PubMed] [Google Scholar]
- Dijstelbloem H.M., van de Winkel J.G., Kallenberg C.G. Inflammation in autoimmunity: Receptors for IgG revisited. Trends. Immunol. 2001;22:510–516. doi: 10.1016/s1471-4906(01)02014-2. [DOI] [PubMed] [Google Scholar]
- Doerschuk C.M., Winn R.K., Coxson H.O., Harlan J.M. CD18-dependent and -independent mechanisms of neutrophil emigration. J. Immunol. 1990;144:2327–2333. [PubMed] [Google Scholar]
- Doury J.C., Teyssier J., Forcain A., Doury F. Modifications de l'adhesivite plaquettaire au cours de la dengue a form hemorrhagique. Bull. Soc. Pathol. Exot. Filiales. 1976;69:493–495. [PubMed] [Google Scholar]
- Dropulic B., Masters C.L. Entry of neurotropic arboviruses into the central nervous system: An in vitro study using mouse brain endothelium. J. Infect. Dis. 1990;161:685–691. doi: 10.1093/infdis/161.4.685. [DOI] [PubMed] [Google Scholar]
- Duisit G., Saleun S., Douthe S., Barsoum J., Chadeuf G., Moullier P. Baculovirus vector requires electrostatic interactions including heparan sulfate for efficient gene transfer in mammalian cells. J. Gene Med. 1999;1:93–102. doi: 10.1002/(SICI)1521-2254(199903/04)1:2<93::AID-JGM19>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- Elbe A., Schleischitz S., Strunk D., Stingl G. Fetal skin-derived MHC class I+, MHC class II-dendritic cells stimulate MHC class I-restricted responses of unprimed CD8+ T cells. J. Immunol. 1994;153:2878–2889. [PubMed] [Google Scholar]
- Erbe D.V., Collins J.E., Shen L., Graziano R.F., Fanger M.W. The effect of cytokines on the expression and function of Fc receptors for IgG on human myeloid cells. Mol. Immunol. 1990;27:57–67. doi: 10.1016/0161-5890(90)90060-d. [DOI] [PubMed] [Google Scholar]
- Fanger M.W., Shen L., Graziano R.F., Guyre P.M. Cytotoxicity mediated by human Fc receptors for IgG. Immunol. Today. 1989;10:92–99. doi: 10.1016/0167-5699(89)90234-X. [DOI] [PubMed] [Google Scholar]
- Feldman S.A., Audet S., Beeler J.A. The fusion glycoprotein of human respiratory syncytial virus facilitates virus attachment and infectivity via an interaction with cellular heparan sulfate. J. Virol. 2000;74:6442–6447. doi: 10.1128/jvi.74.14.6442-6447.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldman S.A., Hendry R.M., Beeler J.A. Identification of a linear heparin binding domain for human respiratory syncytial virus attachment glycoprotein G. J. Virol. 1999;73:6610–6617. doi: 10.1128/jvi.73.8.6610-6617.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferlenghi I., Clarke M., Ruttan T., Allison S.L., Schalich J., Heinz F.X., Harrison S.C., Rey F.A., Fuller S.D. Molecular organization of a recombinant subviral particle from tick-borne encephalitis virus. Mol. Cell. 2001;7:593–602. doi: 10.1016/s1097-2765(01)00206-4. [DOI] [PubMed] [Google Scholar]
- Fiers W. Tumor necrosis factor: Characterization at the molecular, cellular and in vivo level. FEBS Lett. 1991;285:199–212. doi: 10.1016/0014-5793(91)80803-b. [DOI] [PubMed] [Google Scholar]
- Fonseca B.A.L., Pincus S., Shope R.E., Paoletti E., Mason P.W. Recombinant vaccinia viruses coexpressing dengue-1 glycoproteins prM and E induce neutralizing antibodies in mice. Vaccine. 1994;12:279–285. doi: 10.1016/0264-410x(94)90206-2. [DOI] [PubMed] [Google Scholar]
- Foreman K.E., Vaporciyan A.A., Bonish B.K., Jones M.L., Johnson K.J., Glovsky M.M., Eddy S.M., Ward P.A. C5a-induced expression of P-selectin in endothelial cells. J. Clin. Invest. 1994;94:1147–1155. doi: 10.1172/JCI117430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forghani B., Schmidt N.J. Association of herpes simplex virus with platelets of experimentally infected mice. Arch. Virol. 1983;76:269–274. doi: 10.1007/BF01311111. [DOI] [PubMed] [Google Scholar]
- Funahara Y., Ogawa K., Fujita N., Okuno Y. Three possible triggers to induce thrombocytopenia in dengue virus infection. SE. Asian J. Trop. Med. Publ. Hlth. 1987;18:351–355. [PubMed] [Google Scholar]
- Funk C.D. Prostaglandins and leukotrienes: Advances in eicosanoid biology. Science. 2001;294:1871–1875. doi: 10.1126/science.294.5548.1871. [DOI] [PubMed] [Google Scholar]
- Fureder W., Agis H., Willheim M., Bankl H.C., Maier U., Kishi K., Muller M.R., Czerwenka K., Radaszkiewicz T., Butterfield J.H. Differential expression of complement receptors on human basophils and mast cells: Evidence for mast cell heterogeneity and CD88⧸C5aR expression on skin mast cells. J. Immunol. 1995;155:3152–3160. [PubMed] [Google Scholar]
- Galli S.J., Dvorak A.M., Dvorak H.F. Basophils and mast cells: Morphologic insights into their biology, secretory patterns, and function. Prog. Allergy. 1984;34:1–141. [PubMed] [Google Scholar]
- Gallo R.L. Proteoglycans and cutaneous vascular defense and repair. J. Invest. Dermatol. Symp. Proc. 2000;5:55–60. doi: 10.1046/j.1087-0024.2000.00008.x. [DOI] [PubMed] [Google Scholar]
- Gao J.X., Issekutz A.C., Issekutz T.B. Neutrophils migrate to delayed-type hypersensitivity reactions in joints but not in skin: Mechanism is LFA-1⧸MAC-1-independent. J. Immunol. 1994;153:5689–5697. [PubMed] [Google Scholar]
- Gavrilovskaya I.N., Brown E.J., Ginsberg M.H., Mackow E.R. Cellular entry of hantaviruses which cause hemorrhagic fever with renal syndrome is mediated by beta3 integrins. J. Virol. 1999;73:3951–3959. doi: 10.1128/jvi.73.5.3951-3959.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghetie V., Ward E.S. Multiple roles for the major histocompatibility complex class I-related receptor FcRn. Annu. Rev. Immunol. 2000;18:739–766. doi: 10.1146/annurev.immunol.18.1.739. [DOI] [PubMed] [Google Scholar]
- Giroglou T., Florin L., Schafer F., Streeck R.E., Sapp M. Human papillomavirus infection requires cell surface heparan sulfate. J. Virol. 2001;75:1565–1570. doi: 10.1128/JVI.75.3.1565-1570.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodfellow I.G., Sioofy A.B., Powell R.M., Evans D.J. Echoviruses bind heparan sulfate at the cell surface. J. Virol. 2001;75:4918–4921. doi: 10.1128/JVI.75.10.4918-4921.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grabbe J., Welker P., Moller A., Dippel E., Ashman L.K., Czarnetzki B.M. Comparative cytokine release from human monocytes, monocyte-derived immature mast cells, and a human mast cell line (HMC-1) J. Invest. Dermatol. 1994;103:504–508. doi: 10.1111/1523-1747.ep12395649. [DOI] [PubMed] [Google Scholar]
- Green S., Pichyangkul S., Vaughan D.W., Kalayanarooj S., Nimmannitya S., Nisalak A., Kurane I., Rothman A.L., Ennis F.A. Early CD69 expression on peripheral blood lymphocytes from children with dengue hemorrhagic fever. J. Infect Dis. 1999;180:1429–1435. doi: 10.1086/315072. [DOI] [PubMed] [Google Scholar]
- Green S., Vaughn D.W., Kalayanarooj S., Nimmannitya S., Suntayakorn S., Nisalak A., Lew R., Innis B.L., Kurane I., Rothman A.L., Ennis F.A. Early immune activation in acute dengue illness is related to development of plasma leakage and disease severity. J. Infect. Dis. 1999;179:755–762. doi: 10.1086/314680. [DOI] [PubMed] [Google Scholar]
- Gubler D.J. In: Monath T.P., editor. Vol. II. CRC; Boca Raton, FL: 1988. pp. 223–260. (The Arboviruses: Epidemiology and Ecology). [Google Scholar]
- Gubler D.J. Dengue and dengue hemorrhagic fever. Clin. Microbiol. Rev. 1998;11:480–496. doi: 10.1128/cmr.11.3.480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gubler D.J., Suharyono W., Lubis I., Eram S., Sulianti S.J. Epidemic dengue hemorrhagic fever in rural Indonesia: I. Virological and epidemiological studies. Am. J. Trop. Med. Hyg. 1979;28:701–710. [PubMed] [Google Scholar]
- Guirakhoo F., Bolin R.A., Roehrig J.T. The Murray Valley encephalitis virus prM protein confers acid resistance to virus particles and alters the expression of epitopes within the R2 domain of E glycoprotein. Virology. 1992;191:921–931. doi: 10.1016/0042-6822(92)90267-S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guirakhoo F., Heinz F.X., Mandl C.W., Holzmann H., Kunz C. Fusion activity of flaviviruses: Comparison of mature and immature (prM-containing) tick-borne encephalitis virions. J. Gen. Virol. 1991;72:1323–1329. doi: 10.1099/0022-1317-72-6-1323. [DOI] [PubMed] [Google Scholar]
- Guo C.B., Kagey-Sobotka A., Lichtenstein L.M., Bochner B.S. Immunophenotyping and functional analysis of purified human uterine mast cells. Blood. 1992;79:708–712. [PubMed] [Google Scholar]
- Hackett T., Kelton J.G., Powers P. Drug-induced platelet destruction. Semin. Thromb. Hemost. 1982;8:116–137. doi: 10.1055/s-2007-1005047. [DOI] [PubMed] [Google Scholar]
- Hall W.C., Crowell T.P., Watts D.M., Barros V.L., Kruger H., Pinheiro F., Peters C.J. Demonstration of yellow fever and dengue antigens in formalin-fixed paraffin-embedded human liver by immunohistochemical analysis. Am. J. Trop. Med. Hyg. 1991;45:408–417. doi: 10.4269/ajtmh.1991.45.408. [DOI] [PubMed] [Google Scholar]
- Halstead S.B. In: The togaviruses: Biology, Structure, Replication. Schlesinger R.W., editor. Academic Press; New York: 1980. pp. 107–173. [Google Scholar]
- Halstead S.B. Immune enhancement of viral infection. Prog. Allergy. 1982;31:301–364. [PubMed] [Google Scholar]
- Halstead S.B. Pathogenesis of dengue: Challenges to molecular biology. Science. 1988;239:476–481. doi: 10.1126/science.3277268. [DOI] [PubMed] [Google Scholar]
- Halstead S.B. Antibody, macrophages, dengue virus infection, shock, and hemorrhage: A pathogenetic cascade. Rev. Infect. Dis. 1989;11(Suppl. 4):S830–S839. doi: 10.1093/clinids/11.supplement_4.s830. [DOI] [PubMed] [Google Scholar]
- Halstead S.B. In: Tropical and Geographical Medicine. 2nd Ed. Warren K.S., Mahmoud A.A.F., editors. McGraw-Hill; New York: 1990. pp. 675–685. [Google Scholar]
- Halstead S.B., Diwan A.R., Marchette N.J., Palumbo N.E., Srisukonth L. Selection of attenuated dengue 4 viruses by serial passage in primary kidney cells: I. Attributes of uncloned virus at different passage levels. Am. J. Trop. Med. Hyg. 1984;33:654–665. doi: 10.4269/ajtmh.1984.33.654. [DOI] [PubMed] [Google Scholar]
- Halstead S.B., Marchette N.J., Diwan A.R., Palumbo N.E., Putvatana R. Selection of attenuated dengue 4 viruses by serial passage in primary kidney cells: II. Attributes of virus cloned at different dog kidney passage levels. Am. J. Trop. Med. Hyg. 1984;33:666–671. doi: 10.4269/ajtmh.1984.33.666. [DOI] [PubMed] [Google Scholar]
- Halstead S.B., Marchette N.J., Diwan A.R., Palumbo N.E., Putvatana R., Larsen L.K. Selection of attenuated dengue 4 viruses by serial passage in primary kidney cells: III. Reversion to virulence by passage of cloned virus in fetal rhesus lung cells. Am. J. Trop. Med. Hyg. 1984;33:672–678. doi: 10.4269/ajtmh.1984.33.672. [DOI] [PubMed] [Google Scholar]
- Halstead S.B., O'Rourke E.J. Dengue viruses and mononuclear phagocytes: I. Infection enhancement by non-neutralizing antibody. J. Exp. Med. 1977;146:201–217. doi: 10.1084/jem.146.1.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halstead S.B., O'Rourke E.J., Allison A.C. Dengue viruses and mononuclear phagocytes: II. Identity of blood and tissue leukocytes supporting in vitro infection. J. Exp. Med. 1977;146:218–229. doi: 10.1084/jem.146.1.218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halstead S.B., Porterfield J.S., O'Rourke E.J. Enhancement of dengue virus infection in monocytes by flavivirus antisera. Am. J. Trop. Med. Hyg. 1980;29:638–642. doi: 10.4269/ajtmh.1980.29.638. [DOI] [PubMed] [Google Scholar]
- Halstead S.B., Shotwell H., Casals J. Studies on the pathogenesis of dengue infection in monkeys: II. Clinical laboratory responses to heterologous infection. J. Infect. Dis. 1973;128:15–22. doi: 10.1093/infdis/128.1.15. [DOI] [PubMed] [Google Scholar]
- He R.T., Innis B.L., Nisalak A., Usawattanakul W., Wang S., Kalayanarooj S., Anderson R. Antibodies that block virus attachment to Vero cells are a major component of the human neutralizing antibody response against dengue virus type 2. J. Med. Virol. 1995;45:451–461. doi: 10.1002/jmv.1890450417. [DOI] [PubMed] [Google Scholar]
- Heil M.L., Albee A., Strauss J.H., Kuhn R.J. An amino acid substitution in the coding region of the E2 glycoprotein adapts Ross River virus to utilize heparan sulfate as an attachment moiety. J. Virol. 2001;75:6303–6309. doi: 10.1128/JVI.75.14.6303-6309.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinz F.X., Allison S.L. The machinery for flavivirus fusion with host cell membranes. Curr. Opin. Microbiol. 2001;4:450–455. doi: 10.1016/s1369-5274(00)00234-4. [DOI] [PubMed] [Google Scholar]
- Heinz F.X., Allison S.L., Stiasny K., Schalich J., Holzmann H., Mandl C.W., Kunz C. Recombinant and virion-derived soluble and particulate immunogens for vaccination against tick-borne encephalitis. Vaccine. 1995;13:1636–1642. doi: 10.1016/0264-410x(95)00133-l. [DOI] [PubMed] [Google Scholar]
- Heinz F.X., Stiasny K., Puschner-Auer G., Holzmann H., Allison S.L., Mandl C.W., Kunz C. Structural changes and functional control of the tick-borne encephalitis virus glycoprotein E by the heterodimeric association with protein prM. Virology. 1994;198:109–117. doi: 10.1006/viro.1994.1013. [DOI] [PubMed] [Google Scholar]
- Hilgard P., Stockert R. Heparan sulfate proteoglycans initiate dengue virus infection of hepatocytes. Hepatology. 2000;32:1069–1077. doi: 10.1053/jhep.2000.18713. [DOI] [PubMed] [Google Scholar]
- Hiramatsu K., Tadano M., Men R., Lai C.-J. Mutational analysis of a neutralization epitope on the dengue type 2 virus (DEN2) envelope protein: Monoclonal antibody resistant DEN2⧸DEN4 chimeras exhibit reduced mouse neurovirulence. Virology. 1996;224:437–445. doi: 10.1006/viro.1996.0550. [DOI] [PubMed] [Google Scholar]
- Hober D., Poli L., Roblin B., Gestas P., Chungue E., Granic G., Imbert P., Pecarere J.L., Vergez-Pascal R., Wattre P., Maniez-Montreuil M. Serum levels of tumor necrosis factor-alpha (TNF-alpha), interleukin-6 (IL-6) and interleukin-1 beta (IL-1 beta) in dengue-infected patients. Am. J. Trop. Med. Hyg. 1993;48:324–331. doi: 10.4269/ajtmh.1993.48.324. [DOI] [PubMed] [Google Scholar]
- Holbrook M.R., Ni H., Shope R.E., Barrett A.D. Amino acid substitution(s) in the stem-anchor region of langat virus envelope protein attenuates mouse neurovirulence. Virology. 2001;286:54–61. doi: 10.1006/viro.2001.0959. [DOI] [PubMed] [Google Scholar]
- Homchampa P., Sarasombath S., Suvatte V., Vongskul M. Natural killer cells in dengue hemorrhagic fever⧸dengue shock syndrome. Asian Pac. J. Allergy Immunol. 1988;6:95–102. [PubMed] [Google Scholar]
- Hsiao J.C., Chung C.S., Chang W. Vaccinia virus envelope D8L protein binds to cell surface chondroitin sulfate and mediates the adsorption of intracellular mature virions to cells. J. Virol. 1999;73:8750–8761. doi: 10.1128/jvi.73.10.8750-8761.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huerre M.R., Lan N.T., Marianneau P., Hue N.B., Khun H., Hung N.T., Khen N.T., Drouet M.T., Huong V.T., Ha D.Q., Buisson Y., Deubel V. Liver histopathology and biological correlates in five cases of fatal dengue fever in Vietnamese children. Virch. Arch. 2001;438:107–115. doi: 10.1007/s004280000329. [DOI] [PubMed] [Google Scholar]
- Huizinga T.W.J., van der Schoot C.E., Jost C., Klaassen R., Kleijer M., Von dem Borne A.E.G.K., Roos D., Tetteroo P.A.T. The PI-linked receptor FcRIII is released on stimulation of neutrophils. Nature. 1988;333:667–669. doi: 10.1038/333667a0. [DOI] [PubMed] [Google Scholar]
- Hulst M.M., van Gennip H.G., Moormann R.J. Passage of classical swine fever virus in cultured swine kidney cells selects virus variants that bind to heparan sulfate due to a single amino acid change in envelope protein E(rns) J. Virol. 2000;74:9553–9561. doi: 10.1128/jvi.74.20.9553-9561.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hulst M.M., van Gennip H.G., Vlot A.C., Schooten E., de Smit A.J., Moormann R.J. Interaction of classical swine fever virus with membrane-associated heparan sulfate: Role for virus replication in vivo and virulence. J. Virol. 2001;75:9585–9595. doi: 10.1128/JVI.75.20.9585-9595.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurrelbrink R.J., McMinn P.C. Attenuation of Murray Valley encephalitis virus by site-directed mutagenesis of the hinge and putative receptor-binding regions of the envelope protein. J. Virol. 2001;75:7692–7702. doi: 10.1128/JVI.75.16.7692-7702.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ihrcke N.S., Platt J.L. Shedding of heparan sulfate proteoglycan by stimulated endothelial cells: Evidence for proteolysis of cell-surface molecules. J. Cell Physiol. 1996;168:625–637. doi: 10.1002/(SICI)1097-4652(199609)168:3<625::AID-JCP15>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- Imbach P.A. Harmful and beneficial antibodies in immune thrombocytopenic purpura. Clin. Exp. Immunol. 1994;97(Suppl 1):25–30. [PMC free article] [PubMed] [Google Scholar]
- Imler J.L., Hoffmann J.A. Toll and Toll-like proteins: An ancient family of receptors signaling infection. Rev. Immunogenet. 2000;2:294–304. [PubMed] [Google Scholar]
- Issekutz A.C., Chuluyan H.E., Lopes N. CD11⧸CD18-independent transendothelial migration of human polymorphonuclear leokocytes and monocytes: Involvement of distinct and unique mechanisms. J. Leukocyte. Biol. 1995;57:553–561. doi: 10.1002/jlb.57.4.553. [DOI] [PubMed] [Google Scholar]
- Issekutz A.C., Issekutz T.B. A major portion of polymorphonuclear leukocyte and T lymphocyte migration to arthritis in the rat is via LFA-1⧸MAC-1 independent mechanisms. Clin. Immunol. Immunopathol. 1993;67:257–263. doi: 10.1006/clin.1993.1073. [DOI] [PubMed] [Google Scholar]
- Jennings S.R., Rice P.L., Kloszewski E.D., Anderson R.W., Thompson D.L., Tevethia S.S. Effect of herpes simplex virus types 1 and 2 on surface expression of class I major histocompatibility complex antigens on infected cells. J. Virol. 1985;56:757–766. doi: 10.1128/jvi.56.3.757-766.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston L.J., Halliday G.M., King N.J. Phenotypic changes in Langerhans' cells after infection with arboviruses: A role in the immune response to epidermally acquired viral infection? J. Virol. 1996;70:4761–4766. doi: 10.1128/jvi.70.7.4761-4766.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston L.J., Halliday G.M., King N.J. Langerhans cells migrate to local lymph nodes following cutaneous infection with an arbovirus. J. Invest. Dermatol. 2000;114:560–568. doi: 10.1046/j.1523-1747.2000.00904.x. [DOI] [PubMed] [Google Scholar]
- Kelton J.G., Sheridan D., Santos A., Smith J., Steeves K., Smith C., Brown C., Murphy W.G. Heparin-induced thrombocytopenia: Laboratory studies. Blood. 1988;72:925–930. [PubMed] [Google Scholar]
- Killen H., O'Sullivan M.A. Detection of dengue virus by in situ hybridization. J. Virol. Methods. 1993;41:135–146. doi: 10.1016/0166-0934(93)90121-7. [DOI] [PubMed] [Google Scholar]
- King A.D., Nisalak A., Kalayanrooj S., Myint K.A., Pattanapanyasat K., Nimmannitya S., Innis B.L. B cells are the principal circulating mononuclear cells infected by dengue virus. SE. Asian J. Trop. Med. Public Health. 1999;30:718–728. [PubMed] [Google Scholar]
- King C., Anderson R., Marshall J.S. Dengue virus selectively induces human mast cell chemokine production. J. Virol. 2002;76:8408–8419. doi: 10.1128/JVI.76.16.8408-8419.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King C.A., Marshall J.S., Alshurafa H., Anderson R. Release of vasoactive cytokines by antibody-enhanced dengue virus infection of a human mast cell⧸basophil line. J. Virol. 2000;74:7146–7150. doi: 10.1128/jvi.74.15.7146-7150.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King N.J., Kesson A.M. Interferon-independent increases in class I major histocompatibility complex antigen expression follow flavivirus infection. J. Gen. Virol. 1988;69:2535–2543. doi: 10.1099/0022-1317-69-10-2535. [DOI] [PubMed] [Google Scholar]
- King N.J., Maxwell L.E., Kesson A.M. Induction of class I major histocompatibility complex antigen expression by West Nile virus on gamma interferon-refractory early murine trophoblast cells. Proc. Natl. Acad. Sci. USA. 1989;86:911–915. doi: 10.1073/pnas.86.3.911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kliks S.C., Nimmanitya S., Nisalak A., Burke D.S. Evidence that maternal dengue antibodies are important in the development of dengue hemorrhagic fever in infants. Am. J. Trop. Med. Hyg. 1988;38:411–419. doi: 10.4269/ajtmh.1988.38.411. [DOI] [PubMed] [Google Scholar]
- Kliks S.C., Nisalak A., Brandt W.E., Wahl L., Burke D.S. Antibody-dependent enhancement of dengue virus growth in human monocytes as a risk factor for dengue hemorrhagic fever. Am. J. Trop. Med. Hyg. 1989;40:444–451. doi: 10.4269/ajtmh.1989.40.444. [DOI] [PubMed] [Google Scholar]
- Klimstra W.B., Ryman K.D., Bernard K.A., Nguyen K.B., Biron C.A., Johnston R.E. Infection of neonatal mice with sindbis virus results in a systemic inflammatory response syndrome. J. Virol. 1999;73:10387–10398. doi: 10.1128/jvi.73.12.10387-10398.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klimstra W.B., Ryman K.D., Johnston R.E. Adaptation of Sindbis virus to BHK cells selects for use of heparan sulfate as an attachment receptor. J. Virol. 1998;72:7357–7366. doi: 10.1128/jvi.72.9.7357-7366.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komatsu S., Flores S., Gerritsen M.E., Anderson D.C., Granger D.N. Differential up-regulation of circulating soluble and endothelial cell intercellular adhesion molecule-1 in mice. Am. J. Pathol. 1997;151:205–214. [PMC free article] [PubMed] [Google Scholar]
- Kontny U., Kurane I., Ennis F.A. Gamma interferon augments Fcγ receptor-mediated dengue virus infection of human monocytic cells. J. Virol. 1988;62:3928–3933. doi: 10.1128/jvi.62.11.3928-3933.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kopecky J., Grubhoffer L., Kovar V., Jindrak L., Vokurkova D. A putative host cell receptor for tick-borne encephalitis virus identified by anti-idiotypic antibodies and virus affinoblotting. Intervirology. 1999;42:9–16. doi: 10.1159/000024954. [DOI] [PubMed] [Google Scholar]
- Kownatzki E. Triggering of mast cells. Mol. Immunol. 1982;19:1297–1300. doi: 10.1016/0161-5890(82)90296-6. [DOI] [PubMed] [Google Scholar]
- Kretzschmar T., Jeromin A., Gietz C., Bautsch W., Klos A., Kohl J., Rechkemmer G., Bitter-Suermann D. Chronic myelogenous leukemia-derived basophilic granulocytes express a functional active receptor for the anaphylatoxin C3a. Eur. J. Immunol. 1993;23:558–561. doi: 10.1002/eji.1830230239. [DOI] [PubMed] [Google Scholar]
- Krishnamurti C., Kalayanarooj S., Cutting M.A., Peat R.A., Rothwell S.W., Reid T.J., Green S., Nisalak A., Endy T.P., Vaughn D.W., Nimmannitya S., Innis B.L. Mechanisms of hemorrhage in dengue without circulatory collapse. Am. J. Trop. Med. Hyg. 2001;65:840–847. doi: 10.4269/ajtmh.2001.65.840. [DOI] [PubMed] [Google Scholar]
- Kurane I., Ennis F.E. Immunity and immunopathology in dengue virus infections. Semin. Immuno. 1992;4:121–127. [PubMed] [Google Scholar]
- Kurane I., Innis B.L., Nimmannitya S., Nisalak A., Meager A., Janus J., Ennis F.A. Activation of T lymphocytes in dengue virus infections: High levels of soluble interleukin 2 receptor, soluble CD4, soluble CD8, interleukin 2, and interferon-gamma in sera of children with dengue. J. Clin. Invest. 1991;88:1473–1480. doi: 10.1172/JCI115457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurane I., Kontny U., Janus J., Ennis F.A. Dengue-2 virus infection of human mononuclear cell lines and establishment of persistent infections. Arch. Virol. 1990;110:91–101. doi: 10.1007/BF01310705. [DOI] [PubMed] [Google Scholar]
- Kurane I., Rothman A.L., Livingston P.G., Green S., Gagnon S.J., Janus J., Innis B.L., Nimmannitya S., Nisalak A., Ennis F.A. Immunopathologic mechanisms of dengue hemorrhagic fever and dengue shock syndrome. Arch. Virol. 1994;136(Suppl. 9):59–64. doi: 10.1007/978-3-7091-9326-6_7. [DOI] [PubMed] [Google Scholar]
- Kurt-Jones E.A., Popova L., Kwinn L., Haynes L.M., Jones L.P., Tripp R.A., Walsh E.E., Freeman M.W., Golenbock D.T., Anderson L.J., Finberg R.W. Pattern recognition receptors TLR4 and CD14 mediate response to respiratory syncytial virus. Nature Immunol. 2000;1:398–401. doi: 10.1038/80833. [DOI] [PubMed] [Google Scholar]
- Laberge S., Rabb H., Issekutz T., Martin J.G. Role of VLA-4 and LFA-1 in allergen-induced airway inflammation in the rat. Am. J. Resp. Crit. Care Med. 1995;151:822–829. doi: 10.1164/ajrccm.151.3.7881677. [DOI] [PubMed] [Google Scholar]
- Larke R.P.B., Wheelock F. Stabilization of chikungunya virus infectivity by human blood platelets. J. Infect. Dis. 1970;122:523–531. doi: 10.1093/infdis/122.6.523. [DOI] [PubMed] [Google Scholar]
- Lee E., Lobigs M. Substitutions at the putative receptor-binding site of an encephalitic flavivirus alter virulence and host cell tropism and reveal a role for glycosaminoglycans in entry. J. Virol. 2000;74:8867–8875. doi: 10.1128/jvi.74.19.8867-8875.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee E., Weir R.C., Dalgarno L. Changes in the dengue virus major envelope protein on passaging and their localization on the three-dimensional structure of the protein. Virology. 1997;232:281–290. doi: 10.1006/viro.1997.8570. [DOI] [PubMed] [Google Scholar]
- Lee T.-H., Stromberg R.R., Henrard D., Busch M.P. Effect of platelet-associated virus on assays of HIV-1 in plasma. Science. 1993;262:1585–1585. doi: 10.1126/science.8248811. [DOI] [PubMed] [Google Scholar]
- Lefer A.M. Trends in shock research: Significance of lipid mediators in shock states. Circ. Shock. 1989;27:3–12. [PubMed] [Google Scholar]
- Leitmeyer K.C., Vaughn D.W., Watts D.M., Salas R., Villalobos I., de C., Ramos C., Rico-Hesse R. Dengue virus structural differences that correlate with pathogenesis. J. Virol. 1999;73:4738–4747. doi: 10.1128/jvi.73.6.4738-4747.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lescar J., Roussel A., Wien M.W., Navaza J., Fuller S.D., Wengler G., Rey F.A. The fusion glycoprotein shell of Semliki Forest virus: An icosahedral assembly primed for fusogenic activation at endosomal pH. Cell. 2001;105:137–148. doi: 10.1016/s0092-8674(01)00303-8. [DOI] [PubMed] [Google Scholar]
- Ley K. Pathways and bottlenecks in the web of inflammatory adhesion molecules and chemoattractants. Immunol. Res. 2001;24:87–95. doi: 10.1385/IR:24:1:87. [DOI] [PubMed] [Google Scholar]
- Libraty D.H., Pichyangkul S., Ajariyakhajorn C., Endy T.P., Ennis F.A. Human dendritic cells are activated by dengue virus infection: Enhancement by gamma interferon and implications for disease pathogenesis. J. Virol. 2001;75:3501–3508. doi: 10.1128/JVI.75.8.3501-3508.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lidbury B.A., Mahalingam S. Specific ablation of antiviral gene expression in macrophages by antibody-dependent enhancement of Ross River virus infection. J. Virol. 2000;74:8376–8381. doi: 10.1128/jvi.74.18.8376-8381.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin C.F., Lei H.Y., Liu C.C., Liu H.S., Yeh T.M., Wang S.T., Yang T.I., Sheu F.C., Kuo C.F., Lin Y.S. Generation of IgM anti-platelet autoantibody in dengue patients. J. Med. Virol. 2001;63:143–149. [PubMed] [Google Scholar]
- Lin C.L., Chung C.S., Heine H.G., Chang W. Vaccinia virus envelope H3L protein binds to cell surface heparan sulfate and is important for intracellular mature virion morphogenesis and virus infection in vitro and in vivo. J. Virol. 2000;74:3353–3365. doi: 10.1128/jvi.74.7.3353-3365.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liou M.L., Hsu C.Y. Japanese encephalitis virus is transported across the cerebral blood vessels by endocytosis in mouse brain. Cell Tissue Res. 1998;293:389–394. doi: 10.1007/s004410051130. [DOI] [PubMed] [Google Scholar]
- Littaua R., Kurane I., Ennis F.A. Human IgG Fc receptor II mediates antibody-dependent enhancement of dengue virus infection. J. Immunol. 1990;144:3183–3186. [PubMed] [Google Scholar]
- Liu J., Thorp S.C. Cell surface heparan sulfate and its roles in assisting viral infections. Med. Res. Rev. 2002;22:1–25. doi: 10.1002/med.1026. [DOI] [PubMed] [Google Scholar]
- Liu Y., King N., Kesson A., Blanden R.V., Mullbacher A. Flavivirus infection up-regulates the expression of class I and class II major histocompatibility antigens on and enhances T cell recognition of astrocytes in vitro. J. Neuroimmunol. 1989;21:157–168. doi: 10.1016/0165-5728(89)90171-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lobigs M., Blanden R.V., Mullbacher A. Flavivirus-induced up-regulation of MHC class I antigens: Implications for the induction of CD8+ T-cell-mediated autoimmunity. Immunol. Rev. 1996;152:5–19. doi: 10.1111/j.1600-065X.1996.tb00908.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lobigs M., Usha R., Nestorowicz A., Marshall I.D., Weir R.C., Dalgarno L. Host cell selection of Murray Valley encephalitis virus variants altered at an RGD sequence in the envelope protein and in mouse virulence. Virology. 1990;176:587–595. doi: 10.1016/0042-6822(90)90029-q. [DOI] [PubMed] [Google Scholar]
- Lowell C.A., Berton G. Integrin signal transduction in myeloid leukocytes. J. Leukocyk Biol. 1999;65:313–320. doi: 10.1002/jlb.65.3.313. [DOI] [PubMed] [Google Scholar]
- Luscinskas F.W., Cybulsky M.I., Kiely J.M., Peckins C.S., Davis V.M., Gimbrone-MA J. Cytokine-activated human endothelial monolayers support enhanced neutrophil transmigration via a mechanism involving both endothelial-leukocyte adhesion molecule-1 and intercellular adhesion molecule-1. J. Immunol. 1991;146:1617–1625. [PubMed] [Google Scholar]
- Lustig S., Danenberg H.D., Kafri Y., Kobiler D., Ben-Nathan D. Viral neuroinvasion and encephalitis induced by lipopolysaccharide and its mediators. J. Exp. Med. 1992;176:707–712. doi: 10.1084/jem.176.3.707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malasit P. Complement and dengue hemorrhagic fever⧸shock syndrome. SE. Asian J. Trop. Med. Publ. Hlth. 1987;18:316–320. [PubMed] [Google Scholar]
- Mandl C.W., Allison S.L., Holzmann H., Meixner T., Heinz F.X. Attenuation of tick-borne encephalitis virus by structure-based site-specific mutagenesis of a putative flavivirus receptor binding site. J. Virol. 2000;74:9601–9609. doi: 10.1128/jvi.74.20.9601-9609.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mandl C.W., Kroschewski H., Allison S.L., Kofler R., Holzmann H., Meixner T., Heinz F.X. Adaptation of tick-borne encephalitis virus to BHK-21 cells results in the formation of multiple heparan sulfate binding sites in the envelope protein and attenuation in vivo. J. Virol. 2001;75:5627–5637. doi: 10.1128/JVI.75.12.5627-5637.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchette N.J., Halstead S.B. Phytohemagglutinin enhancement of dengue-2 virus replication in nonimmune rhesus monkey peripheral blood leukocytes. Infect. Immun. 1978;19:40–45. doi: 10.1128/iai.19.1.40-45.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marianneau P., Steffan A.M., Royer C., Drouet M.T., Jaeck D., Kirn A., Deubel V. Infection of primary cultures of human Kupffer cells by Dengue virus: No viral progeny synthesis, but cytokine production is evident. J. Virol. 1999;73:5201–5206. doi: 10.1128/jvi.73.6.5201-5206.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marks R.M., Lu H., Sundaresan R., Toida T., Suzuki A., Imanari T., Hernaiz M.J., Linhardt R.J. Probing the interaction of dengue virus envelope protein with heparin: Assessment of glycosaminoglycan-derived inhibitors. J. Med. Chem. 2001;44:2178–2187. doi: 10.1021/jm000412i. [DOI] [PubMed] [Google Scholar]
- Marshall J.S., Bienenstock J. The role of mast cells in inflammatory reactions of the airways, skin and intestine. Curr. Opin. Immunol. 1994;6:853–859. doi: 10.1016/0952-7915(94)90004-3. [DOI] [PubMed] [Google Scholar]
- Marshall J.S., Ford G.P., Bell E.B. Formalin sensitivity and differential staining of mast cells in human dermis. Br. J. Dermatol. 1987;117:29–36. doi: 10.1111/j.1365-2133.1987.tb04087.x. [DOI] [PubMed] [Google Scholar]
- Martinez-Barragan J.J., del Angel R.M. Identification of a putative coreceptor on vero cells that participates in dengue 4 virus infection. J. Virol. 2001;75:7818–7827. doi: 10.1128/JVI.75.17.7818-7827.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maruo N., Morita I., Shirao M., Murota S.I. IL-6 increases endothelial permeability in vitro. Endocrinology. 1992;131:710. doi: 10.1210/endo.131.2.1639018. [DOI] [PubMed] [Google Scholar]
- Mason P.W., Pincus S., Fournier M.J., Mason T.L., Shope R.E., Paoletti E. Japanese encephalitis virus-vaccinia recombinants produce particulate forms of the structural membrane proteins and induce high levels of protection against lethal JEV infection. Virology. 1991;180:294–305. doi: 10.1016/0042-6822(91)90034-9. [DOI] [PubMed] [Google Scholar]
- Matthews V., Robertson T., Kendrick T., Abdo M., Papadimitriou J., McMinn P. Morphological features of Murray Valley encephalitis virus infection in the central nervous system of Swiss mice. Int. J. Exp. Pathol. 2000;81:31–40. doi: 10.1046/j.1365-2613.2000.00135.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKinney E.C., Streilein J.W. On the extraordinary capacity of allogeneic epidermal Langerhans cells to prime cytotoxic T cells in vivo. J. Immunol. 1989;143:1560–1564. [PubMed] [Google Scholar]
- McMinn P.C., Dalgarno L., Weir R.C. A comparison of the spread of Murray Valley encephalitis viruses of high or low neuroinvasiveness in the tissues of Swiss mice after peripheral inoculation. Virology. 1996;220:414–423. doi: 10.1006/viro.1996.0329. [DOI] [PubMed] [Google Scholar]
- Megret F., Hugnot J.P., Falconar A., Gentry M.K., Morens D.M., Murray J.M., Schlesinger J.J., Wright P.J., Young P., Van Regenmortel M.H.V., Deubel V. Use of recombinant fusion proteins and monoclonal antibodies to define linear and discontinuous antigenic sites on the dengue virus envelope glycoprotein. Virology. 1992;187:480–491. doi: 10.1016/0042-6822(92)90450-4. [DOI] [PubMed] [Google Scholar]
- Mentor N.A., Kurane I. Dengue virus infection of human T lymphocytes. Acta Virol. 1997;41:175–176. [PubMed] [Google Scholar]
- Michel C.C. Capillary permeability and how it may change. J. Physiol. 1988;404:1–29. doi: 10.1113/jphysiol.1988.sp017275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitrakul C., Poshyachinda M., Futrakul P., Sangkawibha N., Ahandrik S. Hemostatic and platelet kinetic studies in dengue hemorrhagic fever. Am. J. Trop. Med. Hyg. 1977;26:975–984. doi: 10.4269/ajtmh.1977.26.975. [DOI] [PubMed] [Google Scholar]
- Moller A., Grabbe J., Czarnetzki B.M. Mast cells and their mediators in immediate and delayed immune reactions. Skin Pharmacol. 1991;4(Suppl 1):56–63. doi: 10.1159/000210983. [DOI] [PubMed] [Google Scholar]
- Moller A., Henz B.M., Grutzkau A., Lippert U., Aragane Y., Schwarz T., Kruger K.S. Comparative cytokine gene expression: Regulation and release by human mast cells. Immunology. 1998;93:289–295. doi: 10.1046/j.1365-2567.1998.00425.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moller A., Lippert U., Lessmann D., Kolde G., Hamann K., Welker P., Schadendorf D., Rosenbach T., Luger T., Czarnetzki B.M. Human mast cells produce IL-8. J. Immunol. 1993;151:3261–3266. [PubMed] [Google Scholar]
- Momburg F., Mullbacher A., Lobigs M. Modulation of transporter associated with antigen processing (TAP)-mediated peptide import into the endoplasmic reticulum by flavivirus infection. J. Virol. 2001;75:5663–5671. doi: 10.1128/JVI.75.12.5663-5671.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monath T.P. Dengue: The risk to developed and developing countries. Proc. Natl. Acad. Sci. USA. 1994;91:2395–2400. doi: 10.1073/pnas.91.7.2395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monath T.P., Arroyo J., Levenbook I., Zhang Z.X., Catalan J., Draper K., Guirakhoo F. Single mutation in the flavivirus envelope protein hinge region increases neurovirulence for mice and monkeys but decreases viscerotropism for monkeys: Relevance to development and safety testing of live, attenuated vaccines. J. Virol. 2002;76:1932–1943. doi: 10.1128/JVI.76.4.1932-1943.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monath T.P., Ballinger M.E., Miller B.R., Salaun J.J. Detection of yellow fever viral RNA by nucleic acid hybridization and viral antigen by immunocytochemistry in fixed human liver. Am. J. Trop. Med. Hyg. 1989;40:663–668. doi: 10.4269/ajtmh.1989.40.663. [DOI] [PubMed] [Google Scholar]
- Monath T.P., Cropp C.B., Harrison A.K. Mode of entry of a neurotropic arbovirus into the central nervous system: Reinvestigation of an old controversy. Lab. Invest. 1983;48:399–410. [PubMed] [Google Scholar]
- Montrucchio G., Alloatti G., Camussi G. Role of platelet-activating factor in cardiovascular pathophysiology. Physiol. Rev. 2000;80:1669–1699. doi: 10.1152/physrev.2000.80.4.1669. [DOI] [PubMed] [Google Scholar]
- Morzycki W., Sadowska J., Issekutz A.C. Interleukin-1 and tumour necrosis factor alpha induced polymorphonuclear leukocyte-endothelial cell adhesion and transendothelial migration in vitro: The effect of apical versus basal monolayer stimulation. Immunol. Lett. 1990;25:331–340. doi: 10.1016/0165-2478(90)90204-4. [DOI] [PubMed] [Google Scholar]
- Moser R., Schleiffenbaum B., Groscurth P., Fehr J. Interleukin 1 and tumor necrosis factor stimulate human vascular endothelial cells to promote transendothelial neutrophil passage. J. Clin. Invest. 1989;83:444–455. doi: 10.1172/JCI113903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munoz M.L., Cisneros A., Cruz J., Das P., Tovar R., Ortega A. Putative dengue virus receptors from mosquito cells. FEMS Microbiol. Lett. 1998;168:251–258. doi: 10.1111/j.1574-6968.1998.tb13281.x. [DOI] [PubMed] [Google Scholar]
- Murgue B., Cassar O., Deparis X. Plasma concentrations of sVCAM-1 and severity of dengue infections. J. Med. Virol. 2001;65:97–104. [PubMed] [Google Scholar]
- Navarro-Sanchez E., Altmeyer R., Amara A., Schwartz O., Fieschi F., Virelizier J.L., Arenzana-Seisdedos F., Desprès P. Dendritic-cell-specific ICAM3-grabbing non-integrin is essential for the productive infecton of human dendritic cells by mosquito-cell-derived dengue viruses. EMBO Rep. 2003;4(7 Suppl):1–6. doi: 10.1038/sj.embor.embor866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neff S., Sa-Carvalho D., Rieder E., Mason P.W., Blystone S.D., Brown E.J., Baxt B. Foot-and-mouth disease virus virulent for cattle utilizes the integrin alpha(v)beta3 as its receptor. J. Virol. 1998;72:3587–3594. doi: 10.1128/jvi.72.5.3587-3594.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ni H., Barrett A.D. Attenuation of Japanese encephalitis virus by selection of its mouse brain membrane receptor preparation escape variants. Virology. 1998;241:30–36. doi: 10.1006/viro.1997.8956. [DOI] [PubMed] [Google Scholar]
- Ni H., Ryman K.D., Wang H., Saeed M.F., Hull R., Wood D., Minor P.D., Watowich S.J., Barrett A.D. Interaction of yellow fever virus French neurotropic vaccine strain with monkey brain: Characterization of monkey brain membrane receptor escape variants. J. Virol. 2000;74:2903–2906. doi: 10.1128/jvi.74.6.2903-2906.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilsson G., Johnell M., Hammer C.H., Tiffany H.L., Nilsson K., Metcalfe D.D., Siegbahn A., Murphy P.M. C3a and C5a are chemotaxins for human mast cells and act through distinct receptors via a pertussis toxin-sensitive signal transduction pathway. J. Immunol. 1996;157:1693–1698. [PubMed] [Google Scholar]
- Nilsson G., Svensson V., Nilsson K. Constitutive and inducible cytokine mRNA expression in the human mast cell line HMC-1. Scand. J. Immunol. 1995;42:76–81. doi: 10.1111/j.1365-3083.1995.tb03628.x. [DOI] [PubMed] [Google Scholar]
- Nishioka K. Serum complement level in dengue hemorrhagic fever. Allerg. Immunol. (Leipz.) 1974;20–21:385–392. [PubMed] [Google Scholar]
- Okayama Y., Hagaman D.D., Metcalfe D.D. A comparison of mediators released or generated by IFN-gamma–treated human mast cells following aggregation of Fc gamma RI or Fc epsilon RI. J. Immunol. 2001;166:4705–4712. doi: 10.4049/jimmunol.166.7.4705. [DOI] [PubMed] [Google Scholar]
- Okayama Y., Hagaman D.D., Woolhiser M., Metcalfe D.D. Further characterization of FcgammaRII and FcgammaRII expression by cultured human mast cells. Int. Arch. Allergy Immunol. 2001;124:155–157. doi: 10.1159/000053696. [DOI] [PubMed] [Google Scholar]
- Okayama Y., Kirshenbaum A.S., Metcalfe D.D. Expression of a functional high-affinity IgG receptor, Fc gamma RI, on human mast cells: Up-regulation by IFN-gamma. J. Immunol. 2000;164:4332–4339. doi: 10.4049/jimmunol.164.8.4332. [DOI] [PubMed] [Google Scholar]
- Palucka A.K. Dengue virus and dendritic cells. Nature Med. 2000;6:748–749. doi: 10.1038/77470. [DOI] [PubMed] [Google Scholar]
- Passlick B., Flieger D., Ziegler-Heitbrock H.W. Identification and characterization of a novel monocyte subpopulation in human peripheral blood. Blood. 1989;74:2527–2534. [PubMed] [Google Scholar]
- Patel M., Yanagishita M., Roderiquez G., Bou-Habib D.C., Oravecz T., Hascall V.C., Norcross M.A. Cell-surface heparan sulfate proteoglycan mediates HIV-1 infection of T-cell lines. AIDS Res. Hum. Retrovir. 1993;9:167–174. doi: 10.1089/aid.1993.9.167. [DOI] [PubMed] [Google Scholar]
- Pavri K., Swe T., Ramamoorthy C.L., Chodankar V.P. Immunoglobulin E in dengue haemorrhagic fever (DHF) cases. Trans. R. Soc. Trop. Med. Hyg. 1979;73:451–452. doi: 10.1016/0035-9203(79)90174-3. [DOI] [PubMed] [Google Scholar]
- Pavri K.M., Prasad S.R. T suppressor cells: Role in dengue hemorrhagic fever and dengue shock syndrome. Rev. Infect. Dis. 1980;2:142–146. doi: 10.1093/clinids/2.1.142. [DOI] [PubMed] [Google Scholar]
- Peerschke E.I., Ghebrehiwet B. Human blood platelets possess specific binding sites for C1q. J. Immunol. 1987;138:1537–1541. [PubMed] [Google Scholar]
- Peerschke E.I., Ghebrehiwet B. Platelet receptors for the complement component C1q: Implications for hemostasis and thrombosis. Immunobiology. 1998;199:239–249. doi: 10.1016/S0171-2985(98)80030-2. [DOI] [PubMed] [Google Scholar]
- Peiris J.S., Porterfield J.S. Antibody-mediated enhancement of Flavivirus replication in macrophage-like cell lines. Nature. 1979;282:509–511. doi: 10.1038/282509a0. [DOI] [PubMed] [Google Scholar]
- Perussia B., Dayton E.T., Lazarus R., Fanning V., Trinchieri G. Immune interferon induces the receptor for monomeric IgG1 on human monocytic and myeloid cells. J. Exp. Med. 1983;158:1092–1113. doi: 10.1084/jem.158.4.1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pesci A., Majori M., Piccoli M.L., Casalini A., Curti A., Franchini D., Gabrielli M. Mast cells in bronchiolitis obliterans organizing pneumonia: Mast cell hyperplasia and evidence for extracellular release of tryptase. Chest. 1996;110:383–391. doi: 10.1378/chest.110.2.383. [DOI] [PubMed] [Google Scholar]
- Phanichyakarn P., Israngkura P.B., Krisarin C., Pongpanich B., Dhanamitta S., Valyasevi A. Studies on dengue hemorrhagic fever: IV. Fluorescence staining of the immune complexes on platelets. J. Med. Assoc. Thai. 1977;60:307–311. [PubMed] [Google Scholar]
- Phanichyakarn P., Pongpanich B., Israngkura P.B., Dhanamitta S., Valyasevi A. Studies on Dengue hemorrhagic fever: III. Serum complement (C3) and platelet studies. J. Med. Assoc. Thai. 1977;60:301–306. [PubMed] [Google Scholar]
- Phillpotts R.J., Stephenson J.R., Porterfield J.S. Antibody-dependent enhancement of tick-borne encephalitis virus infectivity. J. Gen. Virol. 1985;66:1831–1837. doi: 10.1099/0022-1317-66-8-1831. [DOI] [PubMed] [Google Scholar]
- Pincus S., Mason P.W., Konishi E., Fonseca B.A.L., Shope R.E., Rice C.M., Paoletti E. Recombinant vaccinia virus producing the prM and E proteins of yellow fever virus protects mice from lethal yellow fever encephalitis. Virology. 1992;187:290–297. doi: 10.1016/0042-6822(92)90317-i. [DOI] [PubMed] [Google Scholar]
- Pletnev S.V., Zhang W., Mukhopadhyay S., Fisher B.R., Hernandez R., Brown D.T., Baker T.S., Rossmann M.G., Kuhn R.J. Locations of carbohydrate sites on alphavirus glycoproteins show that E1 forms an icosahedral scaffold. Cell. 2001;105:127–136. doi: 10.1016/s0092-8674(01)00302-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ploegh H.L. Viral strategies of immune evasion. Science. 1998;280:248–253. doi: 10.1126/science.280.5361.248. [DOI] [PubMed] [Google Scholar]
- Pulimood A.B., Mathan M.M., Mathan V.I. Quantitative and ultrastructural analysis of rectal mucosal mast cells in acute infectious diarrhea. Dig. Dis. Sci. 1998;43:2111–2116. doi: 10.1023/a:1018875718392. [DOI] [PubMed] [Google Scholar]
- Ramos C., Sanchez G., Pando R.H., Baquera J., Hernandez D., Mota J., Ramos J., Flores A., Llausas E. Dengue virus in the brain of a fatal case of hemorrhagic dengue fever. J. Neurovirol. 1998;4:465–468. doi: 10.3109/13550289809114548. [DOI] [PubMed] [Google Scholar]
- Ramos-Castaneda J., Imbert J.L., Barron B.L., Ramos C. A 65-kDa trypsin-sensible membrane cell protein as a possible receptor for dengue virus in cultured neuroblastoma cells. J. Neurovirol. 1997;3:435–440. doi: 10.3109/13550289709031189. [DOI] [PubMed] [Google Scholar]
- Randolph V.B., Winkler G., Stollar V. Acidotropic amines inhibit proteolytic processing of flavivirus prM protein. Virology. 1990;174:450–458. doi: 10.1016/0042-6822(90)90099-d. [DOI] [PubMed] [Google Scholar]
- Ravetch J.V. Fc receptors: Rubor redux. Cell. 1994;78:553–560. doi: 10.1016/0092-8674(94)90521-5. [DOI] [PubMed] [Google Scholar]
- Rey F.A., Heinz F.X., Mandl C., Kunz C., Harrison S.C. The envelope glycoprotein from tick-borne encephalitis virus at 2A resolution. Nature. 1995;375:291–298. doi: 10.1038/375291a0. [DOI] [PubMed] [Google Scholar]
- Rice C.M. In: Fields Virology. 3rd Ed. Fields B.N., Knipe D.M., Howley P.M., editors. Lippincott-Raven; Philadelphia: 1996. pp. 931–959. [Google Scholar]
- Rico-Hesse R., Harrison L.M., Salas R.A., Tovar D., Nisalak A., Ramos C., Boshell J., de Mesa M.T., Nogueira R.M., da Rosa A.T. Origins of dengue type 2 viruses associated with increased pathogenicity in the Americas. Virology. 1997;230:244–251. doi: 10.1006/viro.1997.8504. [DOI] [PubMed] [Google Scholar]
- Rigau-Perez J.G., Clark G.G., Gubler D.J., Reiter P., Sanders E.J., Vorndam A.V. Dengue and dengue haemorrhagic fever. Lancet. 1998;352:971–977. doi: 10.1016/s0140-6736(97)12483-7. [DOI] [PubMed] [Google Scholar]
- Rink L., Kirchner H. Recent progress in the tumor necrosis factor–alpha field. Int. Arch. Allergy Immunol. 1996;111:199–209. doi: 10.1159/000237369. [DOI] [PubMed] [Google Scholar]
- Roebuck K.A., Finnegan A. Regulation of intercellular adhesion molecule-1 (CD54) gene expression. J. Leukocyte Biol. 1999;66:876–888. doi: 10.1002/jlb.66.6.876. [DOI] [PubMed] [Google Scholar]
- Rosen L., Khin M.M. Recovery of virus from the liver of children with fatal dengue: Reflections on the pathogenesis of the disease and its possible analogy with that of yellow fever. Res. Virol. 1989;140:351–360. doi: 10.1016/s0923-2516(89)80115-3. [DOI] [PubMed] [Google Scholar]
- Rothman A.L., Ennis F.A. Immunopathogenesis of dengue hemorrhagic fever. Virology. 1999;257:1–6. doi: 10.1006/viro.1999.9656. [DOI] [PubMed] [Google Scholar]
- Rothwell S.W., Putnak R., La Russa V.F. Dengue-2 virus infection of human bone marrow: Characterization of dengue-2 antigen-positive stromal cells. Am. J. Trop. Med. Hyg. 1996;54:503–510. doi: 10.4269/ajtmh.1996.54.503. [DOI] [PubMed] [Google Scholar]
- Ruangjirachuporn W., Boonpucknavig S., Nimmanitya S. Circulating immune complexes in serum from patients with dengue haemorrhagic fever. Clin. Exp. Immunol. 1979;36:46–53. [PMC free article] [PubMed] [Google Scholar]
- Rue C.A., Ryan P. Characterization of pseudorabies virus glycoprotein C attachment to heparan sulfate proteoglycans. J. Gen. Virol. 2002;83:301–309. doi: 10.1099/0022-1317-83-2-301. [DOI] [PubMed] [Google Scholar]
- Russell P.K., Intavivat A., Kanchanapilant S. Anti-dengue immunoglobulins and serum beta 1 c⧸a globulin levels in dengue shock syndrome. J. Immunol. 1969;102:412–420. [PubMed] [Google Scholar]
- Saadi S., Holzknecht R.A., Patte C.P., Platt J.L. Endothelial cell activation by pore-forming structures: Pivotal role for interleukin-1alpha. Circulation. 2000;101:1867–1873. doi: 10.1161/01.cir.101.15.1867. [DOI] [PubMed] [Google Scholar]
- Saadi S., Holzknecht R.A., Patte C.P., Stern D.M., Platt J.L. Complement-mediated regulation of tissue factor activity in endothelium. J. Exp. Med. 1995;182:1807–1814. doi: 10.1084/jem.182.6.1807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saadi S., Platt J.L. Transient perturbation of endothelial integrity induced by natural antibodies and complement. J. Exp. Med. 1995;181:21–31. doi: 10.1084/jem.181.1.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sa-Carvalho D., Rieder E., Baxt B., Rodarte R., Tanuri A., Mason P.W. Tissue culture adaptation of foot-and-mouth disease virus selects viruses that bind to heparin and are attenuated in cattle. J. Virol. 1997;71:5115–5123. doi: 10.1128/jvi.71.7.5115-5123.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahaphong S., Riengrojpitak S., Bhamarapravati N., Chirachariyavej T. Electron microscopic study of the vascular endothelial cell in dengue haemorrhagic fever. SE. Asian J. Trop. Med. Publ. Hlth. 1980;11:194–204. [PubMed] [Google Scholar]
- Saito H., Miura K., Takahashi G., Ebisawa M., Matsumoto K., Shichijo M., Onda T., Iikura Y., Yanagihara Y., Ra C. Development of tryptase-positive KU812 cells cultured in the presence of Steel factor. Int. Arch. Allergy Immunol. 1995;107:330–332. doi: 10.1159/000237018. [DOI] [PubMed] [Google Scholar]
- Salas-Benito J., del Angel R. Identification of two surface proteins from C6⧸36 cells that bind dengue type 4 virus. J. Virol. 1997;71:7246–7252. doi: 10.1128/jvi.71.10.7246-7252.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheppler J.A., Nicholson J.K., Swan D.C., Ahmed-Ansari A., McDougal J.S. Down-modulation of MHC-I in a CD4+ T cell line, CEM-E5, after HIV-1 infection. J. Immunol. 1989;143:2858–2866. [PubMed] [Google Scholar]
- Schlesinger J.J., Brandriss M.W. Antibody-mediated infection of macrophages and macrophage-like cell lines with 17D-yellow fever virus. J. Med. Virol. 1981;8:103–117. doi: 10.1002/jmv.1890080204. [DOI] [PubMed] [Google Scholar]
- Schlesinger J.J., Brandriss M.W. Growth of 17D yellow fever virus in a macrophage-like cell line, U937: Role of Fc and viral receptors in antibody-mediated infection. J. Immunol. 1981;127:659–665. [PubMed] [Google Scholar]
- Schlesinger J.J., Chapman S.E. Influence of the human high-affinity IgG receptor FcgammaRI (CD64) on residual infectivity of neutralized dengue virus. Virology. 1999;260:84–88. doi: 10.1006/viro.1999.9816. [DOI] [PubMed] [Google Scholar]
- Schnittler H.-J., Wilke A., Gress T., Suttorp N., Drenckhahn D. Role of actin and myosin in the control of paracellular permeability in pig, cat and human vascular endothelium. J. Physiol. 1990;431:379–401. doi: 10.1113/jphysiol.1990.sp018335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz L.B., Austen K.F. Structure and function of the chemical mediators of mast cells. Prog. Allergy. 1984;34:271–321. [PubMed] [Google Scholar]
- Scott R.M., Nisalak A., Cheamudom U., Seridhoranakul S., Nimmannitya S. A preliminary report on the isolation of viruses from the platelets and leukocytes of dengue patients. Asian J. Infect. Dis. 1978;2:95–97. [Google Scholar]
- Scott R.M., Nisalak A., Cheamudon U., Seridhoranakul S., Nimmannitya S. Isolation of dengue viruses from peripheral blood leukocytes of patients with hemorrhagic fever. J. Infect. Dis. 1980;141:1–6. doi: 10.1093/infdis/141.1.1. [DOI] [PubMed] [Google Scholar]
- Selvan R.S., Kapadia H.B., Platt J.L. Complement-induced expression of chemokine genes in endothelium: Regulation by IL-1-dependent and -independent mechanisms. J. Immunol. 1998;161:4388–4395. [PubMed] [Google Scholar]
- Selvaraj P., Rosse W.F., Silber R., Springer T. The major Fc receptor in blood has a phosphatidylinositol anchor and is deficient in paroxysmal nocturnal haemoglobinuria. Nature. 1988;333:565–567. doi: 10.1038/333565a0. [DOI] [PubMed] [Google Scholar]
- Selye H. Mast cells and necrosis. Science. 1966;152:1371–1372. doi: 10.1126/science.152.3727.1371. [DOI] [PubMed] [Google Scholar]
- Selye H., Somogyi A., Mecs I. Influence of mast cells and vasoconstrictors upon various acute connective-tissue reactions. Angiologica. 1968;5:172–185. [PubMed] [Google Scholar]
- Seya T., Turner J.R., Atkinson J.P. Purification and characterization of a membrane protein (gp45–70) that is a cofactor for cleavage of C3b and C4b. J. Exp. Med. 1986;163:837–855. doi: 10.1084/jem.163.4.837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaio M.-F., Chang F.-Y., Hou S.-C. Complement pathway activity in serum from patients with classical dengue fever. Trans. Roy. Soc. Trop. Med. Hyg. 1992;86:672–675. doi: 10.1016/0035-9203(92)90186-g. [DOI] [PubMed] [Google Scholar]
- Shanley T.P., Warner R.L., Ward P.A. The role of cytokines and adhesion molecules in the development of inflammatory injury. Mol. Med. Today. 1995;1:40–45. doi: 10.1016/1357-4310(95)80019-0. [DOI] [PubMed] [Google Scholar]
- Shapiro D., Brandt W.E., Russell P.K. Change involving a viral membrane glycoprotein during morphogenesis of group B arboviruses. Virology. 1972;50:906–911. doi: 10.1016/0042-6822(72)90445-x. [DOI] [PubMed] [Google Scholar]
- Shen J., Devery J.M., King N.J. Adherence status regulates the primary cellular activation responses to the flavivirus West Nile. Immunology. 1995;84:254–264. [PMC free article] [PubMed] [Google Scholar]
- Shen J., Devery J.M., King N.J. Early induction of interferon-independent virus-specific ICAM-1 (CD54) expression by flavivirus in quiescent but not proliferating fibroblasts: Implications for virus-host interactions. Virology. 1995;208:437–449. doi: 10.1006/viro.1995.1174. [DOI] [PubMed] [Google Scholar]
- Shen J., To S.S., Schrieber L., King N.J. Early E-selectin, VCAM-1, ICAM-1, and late major histocompatibility complex antigen induction on human endothelial cells by flavivirus and comodulation of adhesion molecule expression by immune cytokines. J. Virol. 1997;71:9323–9332. doi: 10.1128/jvi.71.12.9323-9332.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shukla D., Liu J., Blaiklock P., Shworak N.W., Bai X., Esko J.D., Cohen G.H., Eisenberg R.J., Rosenberg R.D., Spear P.G. A novel role for 3-O-sulfated heparan sulfate in herpes simplex virus 1 entry. Cell. 1999;99:13–22. doi: 10.1016/s0092-8674(00)80058-6. [DOI] [PubMed] [Google Scholar]
- Shukla D., Spear P.G. Herpesviruses and heparan sulfate: An intimate relationship in aid of viral entry. J. Clin. Invest. 2001;108:503–510. doi: 10.1172/JCI13799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simmons D., Seed B. The Fcγ receptor of natural killer cells is a phospholipid-linked membrane protein. Nature. 1988;333:568–570. doi: 10.1038/333568a0. [DOI] [PubMed] [Google Scholar]
- Sittisombut N., Maneekarn N., Kanjanahaluethai A., Kasinrerk W., Viputtikul K., Supawadee J. Lack of augmenting effect of interferon-gamma on dengue virus multiplication in human peripheral blood monocytes. J. Med. Virol. 1995;45:43–49. doi: 10.1002/jmv.1890450109. [DOI] [PubMed] [Google Scholar]
- Smith C.W., Marlin S.D., Rothlein R., Toman C., Anderson D.C. Cooperative interactions of LFA-1 and Mac-1 with intercellular adhesion molecule-1 in facilitating adherence and transendothelial migration of human neutrophils in vitro. J. Clin. Invest. 1989;83:2008–2017. doi: 10.1172/JCI114111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sobel A.T., Bokisch V.A., Muller-Eberhard H.J. C1q deviation test for the detection of immune complexes, aggregates of IgG, and bacterial products in human serum. J. Exp. Med. 1975;142:139–150. doi: 10.1084/jem.142.1.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sparer T.E., Gooding L.R. Suppression of MHC class I antigen presentation by human adenoviruses. Curr. Top. Microbiol. Immunol. 1998;232:135–147. doi: 10.1007/978-3-642-72045-1_7. [DOI] [PubMed] [Google Scholar]
- Sperr W.R., Bankl H.C., Mundigler G., Klappacher G., Grosschmidt K., Agis H., Simon P., Laufer P., Imhof M., Radaszkiewicz T. The human cardiac mast cell: Localization, isolation, phenotype, and functional characterization. Blood. 1994;84:3876–3884. [PubMed] [Google Scholar]
- Springer T.A. Traffic signals on endothelium for lymphocyte recirculation and leukocyte emigration. Annu. Rev. Physiol. 1995;57:827–872. doi: 10.1146/annurev.ph.57.030195.004143. [DOI] [PubMed] [Google Scholar]
- Srichaikul T., Nimmannitya S., Sripaisarn T., Kamolsilpa M., Pulgate C. Platelet function during the acute phase of dengue hemorrhagic fever. SE. Asian J. Trop. Med. Publ. Hlth. 1989;20:19–25. [PubMed] [Google Scholar]
- Steele K.E., Linn M.J., Schoepp R.J., Komar N., Geisbert T.W., Manduca R.M., Calle P.P., Raphael B.L., Clippinger T.L., Larsen T., Smith J., Lanciotti R.S., Panella N.A., McNamara T.S. Pathology of fatal West Nile virus infections in native and exotic birds during the 1999 outbreak in New York City, New York. Vet. Pathol. 2000;37:208–224. doi: 10.1354/vp.37-3-208. [DOI] [PubMed] [Google Scholar]
- Stiasny K., Allison S.L., Marchler-Bauer A., Kunz C., Heinz F.X. Structural requirements for low-pH–induced rearrangements in the envelope glycoprotein of tick-borne encephalitis virus. J. Virol. 1996;70:8142–8147. doi: 10.1128/jvi.70.11.8142-8147.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauss J.H., Strauss E.G. Virus evolution: How does an enveloped virus make a regular structure? Cell. 2001;105:5–8. doi: 10.1016/S0092-8674(01)00291-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Studahl M., Rosengren L., Gunther G., Hagberg L. Difference in pathogenesis between herpes simplex virus type 1 encephalitis and tick-borne encephalitis demonstrated by means of cerebrospinal fluid markers of glial and neuronal destruction. J. Neurol. 2000;247:636–642. doi: 10.1007/s004150070134. [DOI] [PubMed] [Google Scholar]
- Sung J.S., Diwan A.R., Falkler W.A.J., Yang H.Y., Halstead S.B. Dengue carrier culture and antigen production in human lymphoblastoid lines. Intervirol. 1975;5:137–149. doi: 10.1159/000149891. [DOI] [PubMed] [Google Scholar]
- Suzuki T., Ogata A., Tashiro K., Nagashima K., Tamura M., Yasui K., Nishihira J. Japanese encephalitis virus up-regulates expression of macrophage migration inhibitory factor (MIF) mRNA in the mouse brain. Biochim. Biophys. Acta. 2000;1517:100–106. doi: 10.1016/s0167-4781(00)00262-1. [DOI] [PubMed] [Google Scholar]
- Suzumura A., Lavi E., Weiss S.R., Silberberg D.H. Coronavirus infection induces H-2 antigen expression on oligodendrocytes and astrocytes. Science. 1986;232:991–993. doi: 10.1126/science.3010460. [DOI] [PubMed] [Google Scholar]
- Szabo C., Billiar T.R. Novel roles of nitric oxide in hemorrhagic shock. Shock. 1999;12:1–9. doi: 10.1097/00024382-199907000-00001. [DOI] [PubMed] [Google Scholar]
- Takasaki T., Takada K., Kurane I. Electron microscopic study of persistent dengue virus infection: analysis using a cell line persistently infected with dengue-2 virus. Intervirol. 2001;44:48–54. doi: 10.1159/000050030. [DOI] [PubMed] [Google Scholar]
- Takeda A., Robinson J.E., Ho D.D., Debouck C., Haigwood N.L., Ennis F.A. Distinction of human immunodeficiency virus type 1 neutralization and infection enhancement by human monoclonal antibodies to glycoprotein 120. J. Clin. Invest. 1992;89:1952–1957. doi: 10.1172/JCI115802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeda A., Sweet R.W., Ennis F.A. Two receptors are required for antibody-dependent enhancement of human immunodeficiency virus type 1 infection: CD4 and Fc gamma R. J. Virol. 1990;64:5605–5610. doi: 10.1128/jvi.64.11.5605-5610.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamura M., Webster R.G., Ennis F.A. Neutralization and infection-enhancement epitopes of influenza A virus hemagglutinin. J. Immunol. 1993;151:1731–1738. [PubMed] [Google Scholar]
- Tartaglia L.A., Goeddel D.V. Two TNF receptors. Immunol. Today. 1992;13:151–153. doi: 10.1016/0167-5699(92)90116-O. [DOI] [PubMed] [Google Scholar]
- Tassaneetrithep B., Burgess T.H., Granelli-Piperno A., Trumpfheller C., Finke J., Sun W., Eller M.A., Pattanapanyasat K., Sarasombath S., Birx D.L., Steinman R.M., Schlesinger S., Marovich M.A. DC-SIGN (CD209) mediates dengue virus infection of human dendritic cells. J. Exp. Med. 2003;197:823–829. doi: 10.1084/jem.20021840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taweechaisupapong S., Sriurairatana S., Angsubhakorn S., Yoksan S., Bhamarapravati N. In vivo and in vitro studies on the morphological change in the monkey epidermal Langerhans cells following exposure to dengue 2 (16681) virus. SE. Asian J. Trop. Med. Publ. Hlth. 1996;27:664–672. [PubMed] [Google Scholar]
- Taweechaisupapong S., Sriurairatana S., Angsubhakorn S., Yoksan S., Khin M.M., Sahaphong S., Bhamarapravati N. Langerhans cell density and serological changes following intradermal immunisation of mice with dengue 2 virus. J. Med. Microbiol. 1996;45:138–145. doi: 10.1099/00222615-45-2-138. [DOI] [PubMed] [Google Scholar]
- Thein S., Aung M.M., Shwe T.N., Aye M., Zaw A., Aye K., Aye K.M., Aaskov J. Risk factors in dengue shock syndrome. Am. J. Trop. Med. Hyg. 1997;56:566–572. doi: 10.4269/ajtmh.1997.56.566. [DOI] [PubMed] [Google Scholar]
- Theofilopoulos A.N., Brandt W.E., Russell P.K., Dixon F.T. Replication of dengue-2 virus in cultured human lymphoblastoid cells and subpopulations of human peripheral leukocytes. J. Immunol. 1976;117:953–961. [PubMed] [Google Scholar]
- Timares L., Takashima A., Johnston S.A. Quantitative analysis of the immunopotency of genetically transfected dendritic cells. Proc. Natl. Acad. Sci. USA. 1998;95:13147–13152. doi: 10.1073/pnas.95.22.13147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomita M., Yamamoto K., Kobashi H., Ohmoto M., Tsuji T. Immunohistochemical phenotyping of liver macrophages in normal and diseased human liver. Hepatol. 1994;20:317–325. [PubMed] [Google Scholar]
- Tracey K.J., Cerami A. Tumor necrosis factor, other cytokines and disease. Annu. Rev. Cell Biol. 1993;9:317–343. doi: 10.1146/annurev.cb.09.110193.001533. [DOI] [PubMed] [Google Scholar]
- Trirawatanapong T., Chandran B., Putnak R., Padmanabhan R. Mapping of a region of dengue virus type-2 glycoprotein required for binding by a neutralizing monoclonal antibody. Gene. 1992;116:139–150. doi: 10.1016/0378-1119(92)90509-N. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tuchinda M., Dhorranintra B., Tuchinda P. Histamine content in 24-hour urine in patients with dengue haemorrhagic fever. SE. Asian J. Trop. Med. Publ. Hlth. 1977;8:80–83. [PubMed] [Google Scholar]
- Tuijnman W.B., Van Wichen D.F., Schuurman H.J. Tissue distribution of human IgG Fc receptors CD16, CD32 and CD64: an immunohistochemical study. APMIS. 1993;101:319–329. doi: 10.1111/j.1699-0463.1993.tb00117.x. [DOI] [PubMed] [Google Scholar]
- van Den Engel N.K., Heidenthal E., Vinke A., Kolb H., Martin S. Circulating forms of intercellular adhesion molecule (ICAM)-1 in mice lacking membranous ICAM-1. Blood. 2000;95:1350–1355. [PubMed] [Google Scholar]
- van der Most R.G., Corver J., Strauss J.H. Mutagenesis of the RGD motif in the yellow fever virus 17D envelope protein. Virol. 1999;265:83–95. doi: 10.1006/viro.1999.0026. [DOI] [PubMed] [Google Scholar]
- van de Winkel J.G.J., Anderson C.L. Biology of human immunoglobulin G Fc receptors. J. Leuk. Biol. 1991;49:511–524. doi: 10.1002/jlb.49.5.511. [DOI] [PubMed] [Google Scholar]
- van Epps D.E., Chenoweth D.E. Analysis of the binding of fluorescent C5a and C3a to human peripheral blood leukocytes. J. Immunol. 1984;132:2862–2867. [PubMed] [Google Scholar]
- Vassalli P. The pathophysiology of tumor necrosis factors. Annu. Rev. Immunol. 1992;10:411–452. doi: 10.1146/annurev.iy.10.040192.002211. [DOI] [PubMed] [Google Scholar]
- Vitarana T., de Silva H., Withana N., Gunasekera C. Elevated tumour necrosis factor in dengue fever and dengue haemorrhagic fever. Ceylon Med. J. 1991;36:63–65. [PubMed] [Google Scholar]
- Wang S., He R., Anderson R. PrM- and cell-binding domains of the dengue virus E protein. J. Virol. 1999;73:2547–2551. doi: 10.1128/jvi.73.3.2547-2551.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S., He R., Patarapotikul J., Innis B.L., Anderson R. Antibody-enhanced binding of dengue-2 virus to human platelets. Virol. 1995;213:254–257. doi: 10.1006/viro.1995.1567. [DOI] [PubMed] [Google Scholar]
- Wang S., Zang L., Feng M., Liang Z., Zheng S., Zhang L., Jiang Z., Chen D. Transmission electron microscopic study of the hemorrhagic spots in patients with epidemic hemorrhagic fever in the early stage. Ultrastruct. Pathol. 1997;21:281–287. doi: 10.3109/01913129709021924. [DOI] [PubMed] [Google Scholar]
- Wedi B., Lewrick H., Butterfield J.H., Kapp A. Human HMC-1 mast cells exclusively express the Fc gamma RII subtype of IgG receptor. Arch. Dermatol. Res. 1996;289:21–27. doi: 10.1007/s004030050147. [DOI] [PubMed] [Google Scholar]
- Weiner L.P., Cole G.A., Nathanson N. Experimental encephalitis following peripheral inoculation of West Nile virus in mice of different ages. J. Hyg. (Lond.) 1970;68:435–446. doi: 10.1017/s0022172400042339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss H.J., Halstead S.B. Studies of hemostasis in Thai hemorrhagic fever. J. Pediat. 1965;66:918–926. doi: 10.1016/s0022-3476(65)80068-3. [DOI] [PubMed] [Google Scholar]
- Wengler G. Cell-associated West Nile flavivirus is covered with E+pre-M protein heterodimers which are destroyed and reorganized by proteolytic cleavage during virus release. J. Virol. 1989;63:2521–2526. doi: 10.1128/jvi.63.6.2521-2526.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkelstein A., Kiss J.E. Immunohematologic disorders. JAMA. 1997;278:1982–1992. [PubMed] [Google Scholar]
- Wu S.J., Grouard-Vogel G., Sun W., Mascola J.R., Brachtel E., Putvatana R., Louder M.K., Filgueira L., Marovich M.A., Wong H.K., Blauvelt A., Murphy G.S., Robb M.L., Innes B.L., Birx D.L., Hayes C.G., Frankel S.S. Human skin Langerhans cells are targets of dengue virus infection. Nature Med. 2000;6:816–820. doi: 10.1038/77553. [DOI] [PubMed] [Google Scholar]
- Yadav M., Kamath K.R., Iyngkaran N., Sinniah M. Dengue haemorrhagic fever and dengue shock syndrome: Are they tumour necrosis factor-mediated disorders? FEMS Microbiol. Immunol. 1991;89:45–50. doi: 10.1111/j.1574-6968.1991.tb04969.x. [DOI] [PubMed] [Google Scholar]
- Yang K.D., Yeh W.T., Yang M.Y., Chen R.F., Shaio M.F. Antibody-dependent enhancement of heterotypic dengue infections involved in suppression of IFN-gamma production. J. Med. Virol. 2001;63:150–157. [PubMed] [Google Scholar]
- Zaki S.R., Shieh W.J., Greer P.W., Goldsmith C.S., Ferebee T., Katshitshi J., Tshioko F.K., Bwaka M.A., Swanepoel R., Calain P., Khan A.S., Lloyd E., Rollin P.E., Ksiazek T.G., Peters C.J. A novel immunohistochemical assay for the detection of Ebola virus in skin: Implications for diagnosis, spread, and surveillance of Ebola hemorrhagic fever. J. Infect. Dis. 1999;179(Suppl. 1):S36–S47. doi: 10.1086/514319. [DOI] [PubMed] [Google Scholar]
- Zhu X., Meng G., Dickinson B.L., Li X., Mizoguchi E., Miao L., Wang Y., Robert C., Wu B., Smith P.D., Lencer W.I., Blumberg R.S. MHC class I–related neonatal Fc receptor for IgG is functionally expressed in monocytes, intestinal macrophages, and dendritic cells. J. Immunol. 2001;166:3266–3276. doi: 10.4049/jimmunol.166.5.3266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zucker-Franklin D., Seremetis S., Zheng Z.Y. Internalization of human immunodeficiency virus type 1 and other retroviruses by megakaryocytes and platelets. Blood. 1990;75:1920–1923. [PubMed] [Google Scholar]
- Zwirner J., Fayyazi A., Gotze O. Expression of the anaphylatoxin C5a receptor in non-myeloid cells. Mol. Immunol. 1999;36:877–884. doi: 10.1016/s0161-5890(99)00109-1. [DOI] [PubMed] [Google Scholar]


