Summary:
The field of lymphedema surgery has grown tremendously in recent years. In particular, the diversity of available donor sites for vascularized lymph node transfer has increased, and new donor sites are emerging. Researchers have explored a number of different donor sites, and their reports have demonstrated promising results with each site. Unfortunately, there are limited studies providing a comprehensive analysis of the available donor sites focusing on both the technical aspects of the harvest, including complications and donor site morbidity, and the efficacy and outcomes following transfer. The present review aims to present a comprehensive analysis of the available donor sites for vascularized lymph node transfer and a summary of the experience from a single center of excellence.
INTRODUCTION
Vascularized lymph node transfer (VLNT) has proven to be an effective technique in treating lymphedema.1–3 Similar to a free tissue transfer, lymph nodes can be harvested from one location and transferred to the affected extremity to improve the lymphatic drainage from the swollen limb.4 The lymph node transfer requires a microvascular anastomosis to perfuse and maintain the viability of the lymph nodes, similar to any other free flap performed for breast reconstruction, lower extremity salvage, and head and neck reconstruction. The field of lymphedema surgery has experienced tremendous advancements, with improved imaging modalities, superior optics, and finer instrumentation, which have translated into significant improvements in treatment and patient quality of life.5,6 Coupled with these technological advancements, reconstructive microsurgeons have gained a greater understanding of lymph node physiology and the underlying mechanism of function for the transferred nodes.7,8 As the knowledge in the field of lymphedema surgery has grown, so has the number of potential lymph node donor sites.
Unfortunately, there is a paucity of studies examining the diverse number of available donor sites. Microsurgeons performing lymphedema surgery including VLNT must have a broad understanding and knowledge of the potential options in their armamentarium to best counsel patients regarding the advantages and disadvantages of each donor site and the most appropriate option for each patient. Most studies are limited by small numbers and focused on a single lymph node donor site. Only a few studies exist that provide a more comprehensive analysis, with only one study examining the breadth of donor sites from a single institution.9,10 The present study aims to fill this knowledge gap by providing insights into the most popular available donor sites, focusing on the technical aspects of lymph node harvest, preoperative planning, postoperative complications, and outcomes following VLNT.
Supraclavicular Lymph Nodes
The supraclavicular lymph nodes are a suitable donor site for both upper and lower extremity lymphedema and have proven to be effective, with low morbidity and high efficacy.11 Harvesting the supraclavicular lymph nodes is relatively straightforward and based on 3 landmarks: the clavicle, sternocleidomastoid muscle, and internal jugular vein.12 Within this triangular boundary, the lymph nodes representing a level 5 neck dissection can be safely harvested for VLNT. The primary pedicle consists of the transverse cervical vessels, which most commonly arise from the thyrocervical trunk but can arise directly from the subclavian artery. On average, the pedicle is just under 5 cm long with a slightly larger mean arterial diameter on the right than the left (2.03 ± 0.83 versus 1.80 ± 0.77 cm).13 The authors prefer to include an overlying skin paddle with the lymph nodes for monitoring purposes, although the supraclavicular nodes can also be harvested alone without a skin paddle (Fig. 1). Aside from the benefits of monitoring, including a skin paddle also provides additional volume, which is often helpful to fill the dead space created following release of scar contracture, particularly in the axilla for upper extremity lymphedema. Although the literature contains only one report of donor site lymphedema following harvest of supraclavicular nodes,14 the authors still recommend preoperative lymphoscintigraphy to avoid donor site lymphedema or worsening a patient’s upper extremity lymphedema if the supraclavicular nodes are harvested from the ipsilateral side. If the supraclavicular nodes are harvested from the left side, caution should be taken to avoid injury to the thoracic duct, which can lead to a devastating chyle leak.
Fig. 1.
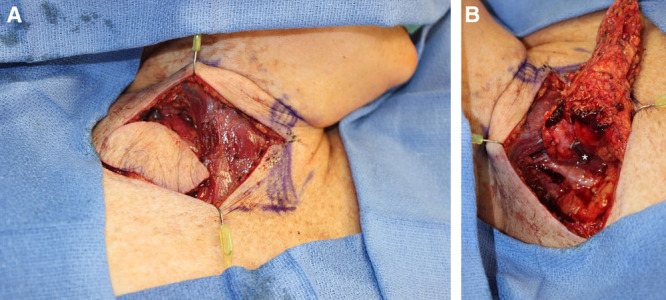
Intraoperative view of supraclavicular lymph node flap harvest. A, Supraclavicular node harvest including a skin paddle based off of a perforator which provides a means for flap monitoring and additional bulk. B, From the undersurface of the flap, the transverse cervical vessels can be visualized passing through the flap to perfuse the nodes and the skin paddle. *indicates the transverse cervical vessels.
The authors design the supraclavicular harvest based on the location of a perforator that is identified using a handheld Doppler. Once a signal locating a perforator is localized, the superior incision is made first in a natural skin crease, and the dissection is carried down to the level of the anterior scalene muscle, which represents the deep margin of the dissection. The omohyoid tendon is routinely divided during the harvest. The phrenic nerve should be identified and carefully protected. The lateral aspect of the dissection proceeds medially from the sternocleidomastoid muscle to the internal jugular vein. The transverse cervical vessels can be identified laterally or medially from their origin; however, the operating microsurgeon should be aware of the potential anatomic variation as far as the source vessels. The vessels should be traced to their take off to maximize their length and caliber. The inferior incision is then made to include a skin paddle with the flap and complete the flap harvest. The external jugular vein often passes through the flap and often is ligated during the harvest of the supraclavicular lymph nodes (See Video 1 [online], which displays a supraclavicular lymph node harvested from the right side which is recommended to avoid any potential injury to the thoracic duct. A skin paddle is harvested with the flap for monitoring purposes and for additional soft tissue volume.)
Video 1. Supraclavicular Lymph Node. Video 1 from “Comprehensive Overview of Available Donor Sites for Vascularized Lymph Node Transfer”.
Submental Lymph Nodes
The submental lymph nodes represent an ideal VLNT donor site in patients with excess tissue in this area where harvest of the nodes also improves the contour of the neck without the risk donor site lymphedema. Facial edema and donor site lymphedema following harvest of the submental nodes have never been reported. The submental lymph node flap is supplied by the submental branch of the facial artery and usually includes an average of three nodes, although there can be some variability in the number of nodes in this donor site basin.15–17
In general, a reliable skin paddle can be harvested with the submental lymph nodes, and again, this is the preferred technique for the authors to provide additional bulk in the recipient site, for monitoring, and for the potential theoretical benefit of the dermal lymphatics in improving drainage from the extremity (Fig. 2). The skin paddle is designed based on the amount of redundant tissue and laxity in the submental region, and as with the supraclavicular nodes, a handheld Doppler can be used to help identify a skin perforator arising from the submental artery. The skin incision is made in a natural crease, and the dissection proceeds through the platysma, which can be included in the flap or preserved. The submental nodes lie in close proximity to the submental artery adjacent to the mandible, and the dissection proceeds from an anteromedial to a posterolateral direction. The pedicle typically travels superior to the submandibular gland, but it can also travel through the gland, increasing the difficulty of the flap dissection. The marginal mandibular nerve almost always crosses superficial to the facial artery at the level of the mandible and should be identified and preserved during flap elevation.17,18
Fig. 2.
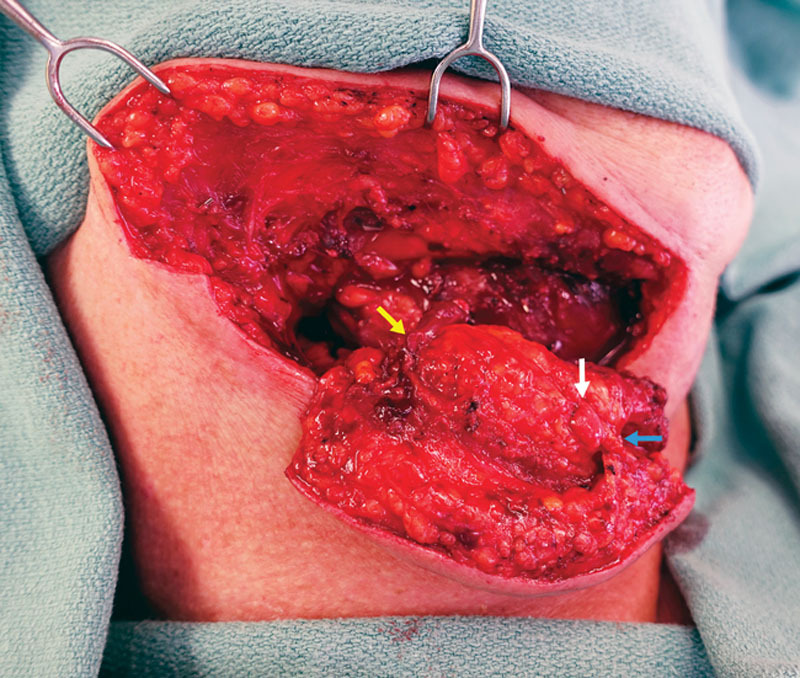
Submental lymph nodes harvested based off the submental artery. A skin paddle is also routinely harvested with the flap for monitoring and potentially for the benefit of dermal lymphatics that may aid the lymphatic drainage from the affected extremity. The yellow arrow indicates the main pedicle to the submental lymph nodes. The white arrow indicates a lymph node in the submental flap. The blue arrow represents a perforator supplying the skin paddle.
Superficial Inguinal Nodes
The superficial inguinal node transfer is a popular option, but is only usable for a patient with upper extremity lymphedema. Every patient undergoing an inguinal lymph node transfer should undergo preoperative lymphoscintigraphy to identify the sentinel nodes draining the leg to prevent donor site lymphedema. The superficial inguinal nodes are based off the superficial circumflex iliac vessels, but they can also be supplied by the superficial inferior epigastric vessels. Therefore, a skin paddle can also be harvested with the flap for monitoring and for additional bulk if needed. The superficial circumflex iliac perforator (SCIP) flap is essentially the skin paddle of the flap, so the flap is designed and harvested in a similar fashion.19,20 Again, a handheld Doppler is used to identify a perforator supplying the overlying skin (Fig. 3).
Fig. 3.
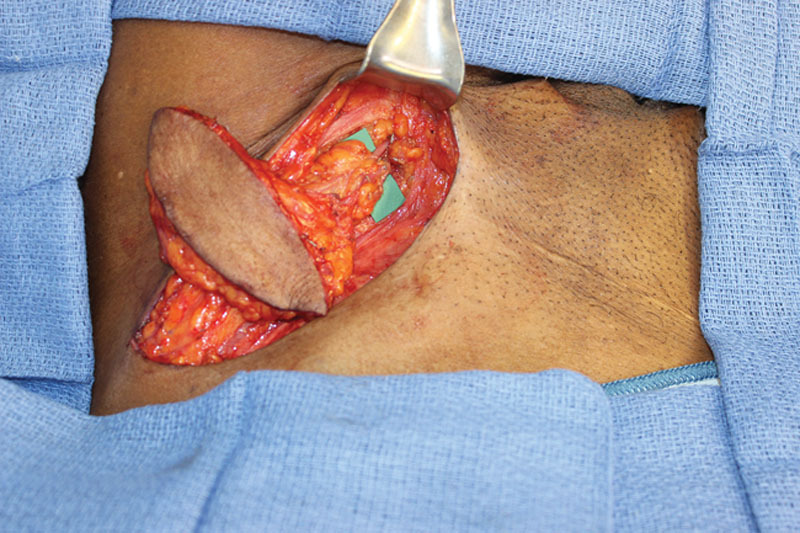
Inguinal lymph nodes based off the superficial circumflex iliac vessels with inclusion of the skin paddle which is the same as the superficial circumflex iliac perforator flap.
The axis of the superficial inguinal lymph nodes is oriented parallel to the inguinal ligament, and the skin incision is made in a natural skin crease with dimensions allowing for primary closure of the donor site. The dissection proceeds down to the level of the abdominal wall and fascia and progresses medially to maximize the length and caliber of the superficial circumflex iliac vessels. The inguinal nodes are often situated more medially and can be located near the junction between the superficial circumflex iliac and the superficial inferior epigastric vessels. Therefore, the flap can be based on the superficial inferior epigastric vessels as well. However, caution should be taken not to progress too medially as the sentinel nodes draining the lower extremities are typically located more medially. The lymph nodes included in the flap should be situated above the level of the inguinal ligament and lateral to the femoral vessels to avoid injuring or harvesting the sentinel nodes draining the leg (See Video 2 [online], which displays a cadaveric dissection of the inguinal lymph node donor site including a skin paddle which is the same skin paddle for a SCIP flap).
Video 2. Inguinal Lymph Node Transfer. Video 2 from “Comprehensive Overview of Available Donor Sites for Vascularized Lymph Node Transfer”.
In patients suffering from breast cancer-related lymphedema (BCRL) who are also interested in breast reconstruction, the inguinal nodes can be harvested safely in conjunction with a deep inferior epigastric perforator (DIEP) flap using a similar approach.21–23 The inguinal nodes remain attached to the DIEP flap based on a wide-based adipofascial pedicle, which is often sufficient to maintain perfusion and viability of the nodes. However, the superficial inguinal nodes should be harvested centered around either the superficial inferior epigastric or superficial circumflex iliac vessels. If the nodes are not adequately perfused through the DIEP pedicle, an additional anastomosis of the superficial vessels to recipient vessels in the axilla is warranted.
Lateral Thoracic Nodes
The lateral thoracic lymph node donor site historically has been associated with the highest complication rates among all donor sites with high rates of seromas, lymphoceles, and donor-site lymphedema. However, when performed with proper technique and training and taking appropriate precautions, the donor site morbidity of lateral thoracic node harvest is no greater than that of other donor sites.9 For patients with upper extremity lymphedema, harvest of the ipsilateral lateral thoracic nodes typically is not an option, as they have often been removed during the axillary dissection or can further compromise the drainage of the affected arm. As with the other donor sites, preoperative lymphoscintigraphy should be performed to minimize the risk of donor site lymphedema when harvesting the lateral thoracic nodes.
The lateral thoracic artery, which is the classic pedicle to the lateral thoracic lymph nodes, originates from the axillary artery, but anatomic variations can exist, with variable origin or complete absence of the lateral thoracic artery altogether. An alternative pedicle is the thoracodorsal artery, which can also supply the lateral thoracic nodes. However, if the thoracodorsal vessels are utilized for a lateral thoracic lymph node transfer, this sacrifices the latissimus dorsi and precludes the use of a workhorse flap in breast reconstruction. However, studies reporting on the combination of a latissimus dorsi flap with the lateral thoracic nodes have demonstrated promising results similar to those combining an abdominal based flap with the inguinal nodes for breast reconstruction and treatment of BCRL.24,25 As with the supraclavicular and inguinal nodes, the authors recommend including a skin paddle, which may be supplied by the lateral thoracic artery or the thoracodorsal artery. If the thoracodorsal pedicle is used, the perforator supplying the skin paddle is the perforator of the thoracodorsal artery perforator flap.25
The dissection of the lateral thoracic lymph nodes also begins with identification of the perforator to the overlying skin, which is classically approximately 8–10 cm caudal from the axillary fold in the midaxillary axis. The incision is made anteriorly, and the dissection proceeds posteriorly until a perforator is visualized, typically near the anterior border of the latissimus dorsi muscle. The lateral thoracic nodes are lateral to the breast and pectoralis major muscle and can extend deep between the muscle and the chest wall (Fig. 4). On average, three nodes in this location can be transferred safely based on either the lateral thoracic or thoracodorsal artery. The lateral thoracic pedicle is often shorter than the thoracodorsal pedicle, but has a usable average diameter of 2.2 mm and average length of 3.6 cm. The thoracodorsal pedicle can be considerably longer with a larger caliber artery and vein.26
Fig. 4.
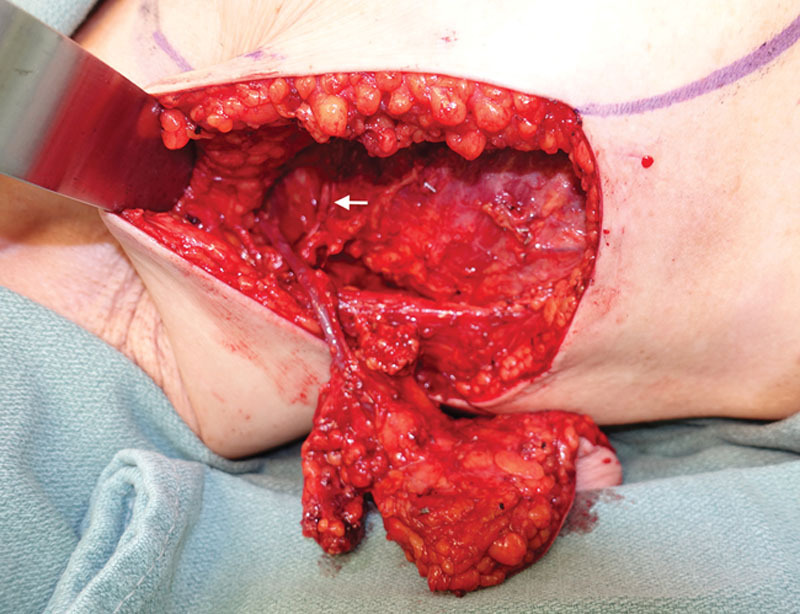
Lateral thoracic nodes harvested based off the lateral thoracic vessels including a skin paddle that can be used both for monitoring purposes as well as to provide additional bulk in the recipient site following release of scarring in the recipient bed. The white arrow identifies the thoracodorsal pedicle to the latissimus dorsi muscle.
Mesenteric Lymph Node
The mesenteric lymph nodes have gained in popularity as an alternate donor site for VLNT.27 The lymph nodes in the mesentery of the jejunum are best harvested using an open approach through a limited midline incision, where the jejunum can be externalized. The externalized segment allows for transillumination of the mesentery to best visualize the vascular arcade as well as the lymph nodes.28 Both the number of nodes taken and the pedicle should be carefully selected to avoid ligating a main branch of the mesentery, which can compromise the perfusion to that segment of the bowel. Consequently, the number of lymph nodes and bulk of soft tissue that can be harvested are very limited (Fig. 5).
Fig. 5.
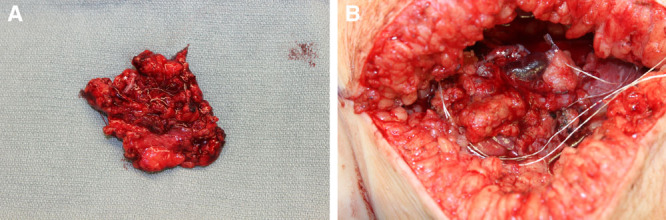
The lymph nodes residing in the mesentery of the jejunum can also be harvested for vascularized lymph node transfer. *indicates the pedicle to the mesenteric lymph nodes. A, Note the short pedicle length and small volume of the mesenteric lymph nodes. B, The lymph nodes are placed into the affected extremity and in this case, implantable Dopplers were used for monitoring, and a vein graft was needed for the arterial anastomosis. * identifies the implantable Doppler placed around the vein of for monitoring of the venous anastomosis. ** indicates the vein graft.
Using the mesenteric nodes has significant disadvantages, such as the need for intra-abdominal harvest via a laparotomy, which can be associated with a risk of injury to the bowel or other intra-abdominal structures, an incisional hernia, leakage of the bowel anastomosis, or ischemic bowel. However, use of the mesenteric nodes has never been associated with donor site lymphedema.
Gastroepiploic Nodes and Omentum
Another donor site that has been gaining in popularity is the free gastroepiploic lymph nodes and the greater omentum, which can be harvested using a minimally invasive laparoscopic approach or an upper epigastric incision.29 The omentum, including the gastroepiploic nodes, can be harvested based on the right and/or left gastroepiploic vessels and used for treatment of bilateral lymphedema or for dual-level lymph node transfer to the same extremity (Fig. 6). Anatomic studies have demonstrated an average of 3 nodes along the vascular pedicle. However, the omentum also contains lymphatic tissue or “lakes” that can aid in fluid absorption in the affected lymphedematous limb. The gastroepiploic arteries are typically of reasonable size, measuring nearly 2.5 mm in diameter, whereas the veins are often considerably larger but very thin walled.30
Fig. 6.
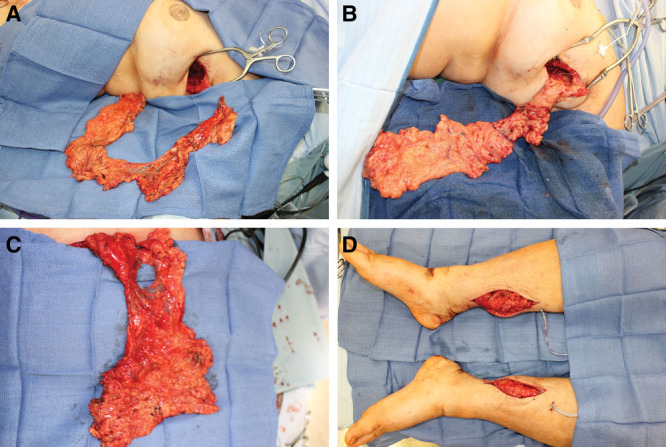
The omentum and gastroepiploic nodes provide sufficient volume as well as a large lymphatic organ that can provide volume to fill dead space following release of scar tissue. A and B, Omental and gastroepiploic nodes transferred into the axilla for treatment of breast cancer related lymphedema. C, Laparoscopic harvest of omentum through a minimally invasive approach. D, The omentum can be split into two separate flaps for treatment of bilateral lower extremity lymphedema.
Although use of the gastroepiploic nodes and omentum is not associated with a risk of donor site lymphedema, any intra-abdominal operation can be associated with risks of an incisional hernia, adhesions, or inadvertent injury to adjacent structures, similar to harvest of the mesenteric lymph nodes. Patients undergoing laparoscopic harvest should always be counseled regarding the potential need to convert to an open operation. Another rare but potential complication is pancreatitis. A complete history and thorough abdominal examination should be performed to ascertain whether a patient has had prior abdominal surgeries. Patients who have undergone a number of abdominal operations may not be candidates for a laparoscopic approach or for an omental flap at all. Although the authors often perform imaging to evaluate patient anatomy as well as the gastroepiploic vessels and superior mesenteric axis, preoperative imaging is of limited utility in assessment of adhesive disease burden and density. In patients for whom additional bulk and volume are needed for obliteration of the dead space following a thorough scar release, which commonly occurs in the axilla for patients with BCRL, the omentum is an excellent option for providing additional volume. We and others favor using indocyanine green angiography to confirm the viability and perfusion of the omentum that is transferred, and any areas that are poorly perfused should be removed.31
DISCUSSION
As the field of supermicrosurgery for lymphedema expands, new donor sites for VLNT are continuously emerging. Regarding the most common donor sites described, the decision to use one versus the other should be based on a thorough discussion with the patient about their advantages and disadvantages.32 Although the literature comparing the different donor sites is somewhat sparse, most studies have demonstrated excellent outcomes with each donor site. To our knowledge, no meta-analyses comparing the efficacy of the donor sites exists, but one comprehensive review demonstrated equivalent outcomes with all donor sites except for the lateral thoracic lymph nodes.9 However, this may reflect limited number of patients, early experience and early learning curves of the microsurgeons, and lack of consistency in follow-up and valid outcome measures. One study compared the submental donor site to the inguinal nodes and demonstrated superior outcomes with fewer complications in patients undergoing submental node transfer.33 Despite these findings, the study was limited due to small numbers and a clear bias favoring the submental donor site in terms of numbers, experience, and surgeon preference.
The authors have found all donor sites to have equivalent efficacy in terms of effectiveness in improving both lymphedema and limiting donor site morbidity. With appropriate training in lymphedema surgery, and safe, proper technique, all donor sites can produce excellent results in improving the lymphatic drainage from the affected limb. The primary factor in deciding which donor site to use is not the degree of improvement that can be achieved but considerations regarding each respective donor site. Historically, use of the lateral thoracic lymph nodes has been less efficacious than other VLNT donor sites and has been associated with the highest risk of donor site lymphedema, lymphoceles, and seromas. However, the inguinal nodal basin also has been associated with donor site complications. Only one case report of a supraclavicular node harvest precipitating lymphedema of the donor upper extremity has been reported, which is why we recommend performing preoperative lymphoscintigraphy for all patients undergoing a lateral thoracic, supraclavicular, or inguinal lymph node transfer to identify the sentinel nodes to be preserved during the node harvest. Reverse lymphatic mapping should also be performed to minimize the risk of donor site lymphedema.34
In addition to the risk of donor site lymphedema which has never been described following harvest of the submental or intra-abdominal nodes, donor site scarring should be discussed with patients. Although the inguinal and lateral thoracic donor site scars are easily concealed in clothing, the other donor sites can leave visible scars that may be concerning for patients. Overall, proponents of submental and supraclavicular lymph node transfer have demonstrated the scars at those sites are well tolerated but should be discussed with patients nonetheless. The gastroepiploic nodes and omentum as well as the jejunal mesenteric nodes are most commonly harvested via an open approach, which can leave relatively larger scars as well which may be an issue for some patients. Aside from the abdominal scars, patients should be cautioned and educated regarding the risks of an incisional hernia, adhesions, potential for a bowel obstruction, and other complications and injuries to the viscera and bowel. If the omentum is harvested laparoscopically, which is the authors’ preference, the patient should always be warned of the risk of needing to convert to an open operation. Although the authors favor a minimally invasive approach, this mandates advanced laparoscopic skills or perhaps collaboration with general surgery colleagues.
Another area of considerable debate with VLNT is the recipient location, specifically, whether the nodes should be placed distally or proximally in the affected extremity. In general, the authors favor placing the lymph nodes in an anatomic location proximally in the limb, although in certain circumstances, we have placed the nodes in the area of greatest severity or performed double lymph node transfers, both proximally and distally. However, for proximally placed nodes, the authors recommend a wide scar release, particularly in the axilla, which can have a dramatic impact on patients’ lymphedema and range of motion. A scar release alone is insufficient in treating lymphedema, and studies have demonstrated that vascularized tissue alone is also ineffective. An adequate scar release typically creates a sizable cavity and dead space that should be filled. Therefore, when considering the different lymph node donor sites, the volume deficit should also be considered. The omentum, inguinal, and lateral thoracic lymph nodes typically provide more volume than the supraclavicular, submental, and mesenteric nodes, which may be better suited for more distal placement. Overall, the ideal donor and recipient sites remain areas of considerable debate among those performing high-volume lymphedema surgery.35
Treatment of BCRL presents a unique circumstance as patients may be amenable to either lymphovenous bypass or VLNT. Although both approaches are remarkably effective in improving lymphedema, for patients also seeking breast reconstruction, the authors favor a combined approach that addresses both lymphedema and breast reconstruction. Recent studies demonstrated that autologous free flap reconstruction alone has no benefit in treating BCRL.36 As with any inguinal lymph node transfer, preoperative evaluation and imaging are always performed. To provide an aesthetic breast reconstruction and improve the lymphedema, the inguinal nodes are harvested in conjunction with the DIEP flap. The entire construct is then transferred to the chest, where the DIEP flap is used to reconstruct the breast, while the inguinal nodes are placed in the axilla following release of the scar. Indocyanine green angiography is used to determine whether an additional arterial or venous anastomosis is needed to perfuse the nodes. Historically, the DIEP flap with VLNT has been performed alone; however, the authors now recommend reconstructing the breast with the DIEP flap combined with lymphaticovenular anastomosis and VLNT. This combined approach, so-called Breast Reconstruction Including Lymphaticovenular anastomosis and Inguinal to Axillary Node Transfer, has demonstrated excellent results in addressing lymphedema and breast reconstruction in a single operation.
CONCLUSIONS
A number of different donor sites are available for VLNT. Although each donor site has distinct advantages and disadvantages, all of them seem to have equivalent benefit in treating lymphedema. The decision about which donor site to use should be based on donor site availability and morbidity, patient preference, surgeon comfort and experience, and recipient location.
Footnotes
Published online 25 March 2020.
Disclosure: The authors have no financial interest to declare in relation to the content of this article.
Related Digital Media are available in the full-text version of the article on www.PRSGlobalOpen.com.
REFERENCES
- 1.Raju A, Chang DW. Vascularized lymph node transfer for treatment of lymphedema: a comprehensive literature review. Ann Surg. 2015;261:1013–1023. [DOI] [PubMed] [Google Scholar]
- 2.Gould DJ, Mehrara BJ, Neligan P, et al. Lymph node transplantation for the treatment of lymphedema. J Surg Oncol. 2018;118:736–742. [DOI] [PubMed] [Google Scholar]
- 3.Schaverien MV, Badash I, Patel KM, et al. Vascularized lymph node transfer for lymphedema. Semin Plast Surg. 2018;32:28–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Silva AK, Chang DW. Vascularized lymph node transfer and lymphovenous bypass: novel treatment strategies for symptomatic lymphedema. J Surg Oncol. 2016;113:932–939. [DOI] [PubMed] [Google Scholar]
- 5.Patel KM, Lin CY, Cheng MH. A prospective evaluation of lymphedema-specific quality-of-life outcomes following vascularized lymph node transfer. Ann Surg Oncol. 2015;22:2424–2430. [DOI] [PubMed] [Google Scholar]
- 6.Schaverien MV, Coroneos CJ. Surgical treatment of lymphedema. Plast Reconstr Surg. 2019;144:738–758. [DOI] [PubMed] [Google Scholar]
- 7.Patel KM, Lin CY, Cheng MH. From theory to evidence: long-term evaluation of the mechanism of action and flap integration of distal vascularized lymph node transfers. J Reconstr Microsurg. 2015;31:26–30. [DOI] [PubMed] [Google Scholar]
- 8.Miranda Garcés M, Pons G, Mirapeix R, et al. Intratissue lymphovenous communications in the mechanism of action of vascularized lymph node transfer. J Surg Oncol. 2017;115:27–31. [DOI] [PubMed] [Google Scholar]
- 9.Scaglioni MF, Arvanitakis M, Chen YC, et al. Comprehensive review of vascularized lymph node transfers for lymphedema: outcomes and complications. Microsurgery. 2018;38:222–229. [DOI] [PubMed] [Google Scholar]
- 10.Ciudad P, Agko M, Perez Coca JJ, et al. Comparison of long-term clinical outcomes among different vascularized lymph node transfers: 6-year experience of a single center’s approach to the treatment of lymphedema. J Surg Oncol. 2017;116:671–682. [DOI] [PubMed] [Google Scholar]
- 11.Maldonado AA, Chen R, Chang DW. The use of supraclavicular free flap with vascularized lymph node transfer for treatment of lymphedema: a prospective study of 100 consecutive cases. J Surg Oncol. 2017;115:68–71. [DOI] [PubMed] [Google Scholar]
- 12.Ooi AS, Chang DW. 5-step harvest of supraclavicular lymph nodes as vascularized free tissue transfer for treatment of lymphedema. J Surg Oncol. 2017;115:63–67. [DOI] [PubMed] [Google Scholar]
- 13.Steinbacher J, Tinhofer IE, Meng S, et al. The surgical anatomy of the supraclavicular lymph node flap: a basis for the free vascularized lymph node transfer. J Surg Oncol. 2017;115:60–62. [DOI] [PubMed] [Google Scholar]
- 14.Lee M, McClure E, Reinertsen E, et al. Lymphedema of the upper extremity following supraclavicular lymph node harvest. Plast Reconstr Surg. 2015;135:1079e–1082e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Asuncion MO, Chu SY, Huang YL, et al. Accurate prediction of submental lymph nodes using magnetic resonance imaging for lymphedema surgery. Plast Reconstr Surg Glob Open. 2018;6:e1691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gustafsson J, Chu SY, Chan WH, et al. Correlation between quantity of transferred lymph nodes and outcome in vascularized submental lymph node flap transfer for lower limb lymphedema. Plast Reconstr Surg. 2018;142:1056–1063. [DOI] [PubMed] [Google Scholar]
- 17.Tzou CH, Meng S, Ines T, et al. Surgical anatomy of the vascularized submental lymph node flap: anatomic study of correlation of submental artery perforators and quantity of submental lymph node. J Surg Oncol. 2017;115:54–59. [DOI] [PubMed] [Google Scholar]
- 18.Cheng MH, Lin CY, Patel KM. A prospective clinical assessment of anatomic variability of the submental vascularized lymph node flap. J Surg Oncol. 2017;115:43–47. [DOI] [PubMed] [Google Scholar]
- 19.Suh HS, Jeong HH, Choi DH, et al. Study of the medial superficial perforator of the superficial circumflex iliac artery perforator flap using computed tomographic angiography and surgical anatomy in 142 patients. Plast Reconstr Surg. 2017;139:738–748. [DOI] [PubMed] [Google Scholar]
- 20.Goh TL, Park SW, Cho JY, et al. The search for the ideal thin skin flap: superficial circumflex iliac artery perforator flap–a review of 210 cases. Plast Reconstr Surg. 2015;135:592–601. [DOI] [PubMed] [Google Scholar]
- 21.Chang EI, Masià J, Smith ML. Combining autologous breast reconstruction and vascularized lymph node transfer. Semin Plast Surg. 2018;32:36–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nguyen AT, Chang EI, Suami H, et al. An algorithmic approach to simultaneous vascularized lymph node transfer with microvascular breast reconstruction. Ann Surg Oncol. 2015;22:2919–2924. [DOI] [PubMed] [Google Scholar]
- 23.Saaristo AM, Niemi TS, Viitanen TP, et al. Microvascular breast reconstruction and lymph node transfer for postmastectomy lymphedema patients. Ann Surg. 2012;255:468–473. [DOI] [PubMed] [Google Scholar]
- 24.Vibhakar D, Reddy S, Morgan-Hazelwood W, et al. Chimeric pedicled latissimus dorsi flap with lateral thoracic lymph nodes for breast reconstruction and lymphedema treatment in a hypercoagulable patient. Plast Reconstr Surg. 2014;134:494e–495e. [DOI] [PubMed] [Google Scholar]
- 25.Inbal A, Teven CM, Chang DW. Latissimus dorsi flap with vascularized lymph node transfer for lymphedema treatment: technique, outcomes, indications and review of literature. J Surg Oncol. 2017;115:72–77. [DOI] [PubMed] [Google Scholar]
- 26.Tinhofer IE, Meng S, Steinbacher J, et al. The surgical anatomy of the vascularized lateral thoracic artery lymph node flap-A cadaver study. J Surg Oncol. 2017;116:1062–1068. [DOI] [PubMed] [Google Scholar]
- 27.Coriddi M, Wee C, Meyerson J, et al. Vascularized jejunal mesenteric lymph node transfer: a novel surgical treatment for extremity lymphedema. J Am Coll Surg. 2017;225:650–657. [DOI] [PubMed] [Google Scholar]
- 28.Schaverien MV, Hofstetter WL, Selber JC. Vascularized jejunal mesenteric lymph node transfer for lymphedema: a novel approach. Plast Reconstr Surg. 2018;141:468e–469e. [DOI] [PubMed] [Google Scholar]
- 29.Nguyen AT, Suami H, Hanasono MM, et al. Long-term outcomes of the minimally invasive free vascularized omental lymphatic flap for the treatment of lymphedema. J Surg Oncol. 2017;115:84–89. [DOI] [PubMed] [Google Scholar]
- 30.Howell AC, Gould DJ, Mayfield C, et al. Anatomical basis of the gastroepiploic vascularized lymph node transfer: a radiographic evaluation using computed tomographic angiography. Plast Reconstr Surg. 2018;142:1046–1052. [DOI] [PubMed] [Google Scholar]
- 31.Coriddi M, Kenworthy E, Weinstein A, et al. The importance of indocyanine green near-infrared fluorescence angiography in perfusion assessment in vascularized omentum lymphatic transplant. J Surg Oncol. 2018;118:109–112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ciudad P, Manrique OJ, Date S, et al. A head-to-head comparison among donor site morbidity after vascularized lymph node transfer: pearls and pitfalls of a 6-year single center experience. J Surg Oncol. 2017;115:37–42. [DOI] [PubMed] [Google Scholar]
- 33.Ho OA, Lin CY, Pappalardo M, et al. Comparisons of submental and groin vascularized lymph node flaps transfer for breast cancer-related lymphedema. Plast Reconstr Surg Glob Open. 2018;6:e1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Dayan JH, Dayan E, Smith ML. Reverse lymphatic mapping: a new technique for maximizing safety in vascularized lymph node transfer. Plast Reconstr Surg. 2015;135:277–285. [DOI] [PubMed] [Google Scholar]
- 35.Pappalardo M, Patel K, Cheng MH. Vascularized lymph node transfer for treatment of extremity lymphedema: an overview of current controversies regarding donor sites, recipient sites and outcomes. J Surg Oncol. 2018;117:1420–1431. [DOI] [PubMed] [Google Scholar]
- 36.Engel H, Lin CY, Huang JJ, et al. Outcomes of lymphedema microsurgery for breast cancer-related lymphedema with or without microvascular breast reconstruction. Ann Surg. 2018;268:1076–1083. [DOI] [PubMed] [Google Scholar]


